Pain Management
Every pain syndrome, interventional procedure, medication, classification, and documentation framework you need to succeed on day one and beyond.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 Pain Neurobiology — Pathways & Mechanisms
Pain medicine is a multidisciplinary specialty that manages acute, chronic, cancer, and interventional pain. Unlike most fields where "the diagnosis" drives care, pain medicine is organized around mechanisms of pain. A single patient with low back pain may simultaneously have nociceptive (facet arthropathy), neuropathic (radiculopathy), and nociplastic (central sensitization) components, and the documentation must capture each driver distinctly because treatment differs for each.
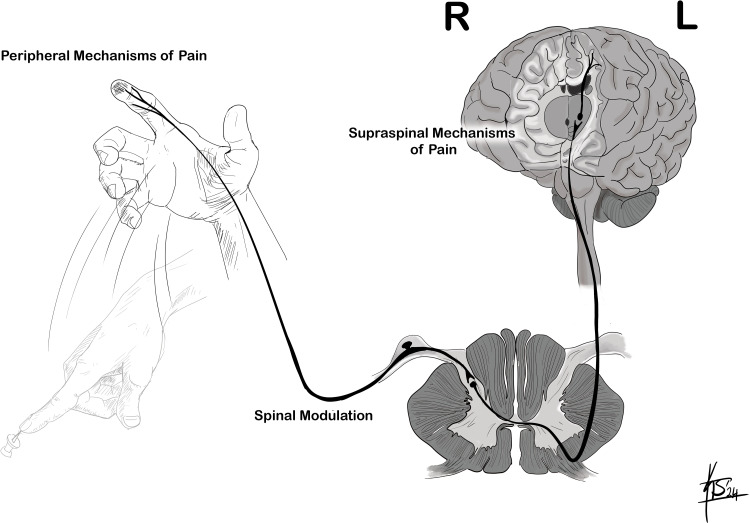
The Four Pain Mechanisms
Every pain complaint a scribe documents can be classified into one or more of four mechanism categories. Clinicians will say things like "predominantly neuropathic with a nociplastic overlay," and you are expected to chart this language accurately.
Activation of peripheral nociceptors (A-delta and C fibers) by actual or threatened tissue damage. Further divided into somatic (skin, muscle, bone, joint — well-localized, sharp, aching) and visceral (hollow organs, capsules — poorly localized, cramping, referred). Examples: osteoarthritis, fracture pain, post-operative incisional pain, sickle cell vaso-occlusive pain. Typically responds to NSAIDs, acetaminophen, and opioids.
Pain caused by a lesion or disease of the somatosensory nervous system. Descriptors include burning, electric, shooting, lancinating, pins-and-needles, and it follows a dermatomal or peripheral nerve distribution. Examples: diabetic peripheral neuropathy, post-herpetic neuralgia, trigeminal neuralgia, radiculopathy, phantom limb. Responds preferentially to gabapentinoids, SNRIs, and tricyclic antidepressants — not NSAIDs.
Pain arising from altered central nociceptive processing without clear tissue damage or nerve lesion. The IASP formalized this third mechanism category in 2017. Examples: fibromyalgia, non-specific chronic low back pain with central sensitization, irritable bowel syndrome, chronic pelvic pain, tension-type headache. Widespread, amplified, often with allodynia and hyperalgesia. Responds to duloxetine, milnacipran, low-dose naltrexone, and non-pharmacologic modalities (CBT, graded exercise).
The most common category clinically. Examples: cancer pain (tumor invasion + nerve compression + bone destruction), chronic low back pain (disc degeneration + radiculopathy + muscle spasm + central sensitization), CRPS (peripheral nerve injury + inflammatory + autonomic + central). Always document each component that is contributing.
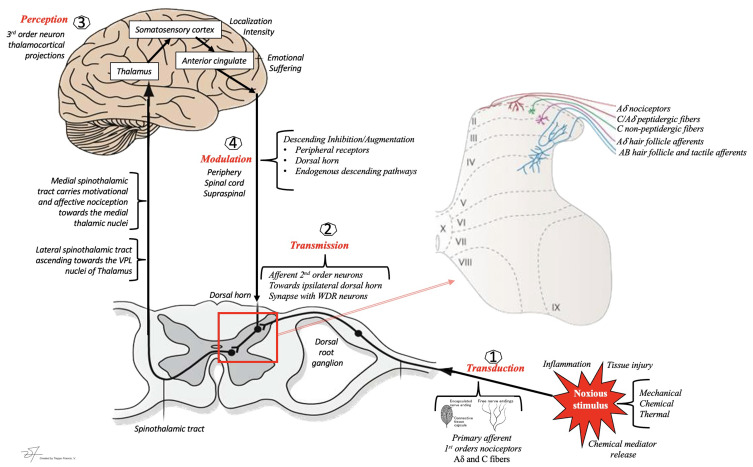
Ascending & Descending Pathways
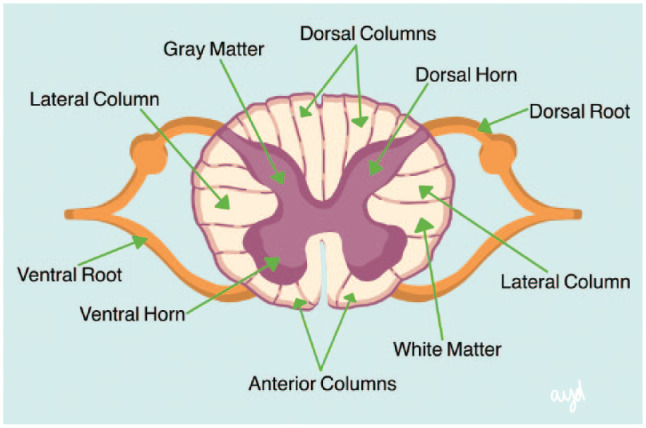
A noxious stimulus activates primary afferent nociceptors whose cell bodies sit in the dorsal root ganglion (DRG). These first-order neurons synapse in the dorsal horn of the spinal cord (Rexed laminae I, II, V), release glutamate and substance P, and excite second-order neurons that decussate and ascend in the spinothalamic tract to the thalamus. Third-order neurons project from thalamus to the primary somatosensory cortex, insula, and anterior cingulate cortex — the "pain matrix." A parallel descending modulatory system from the periaqueductal gray (PAG) and rostroventral medulla releases serotonin, norepinephrine, and endogenous opioids at the dorsal horn to dampen incoming signals. This anatomy explains why SNRIs (duloxetine, venlafaxine), TCAs, and opioids all work as analgesics — they augment descending inhibition or mimic endogenous opioids.
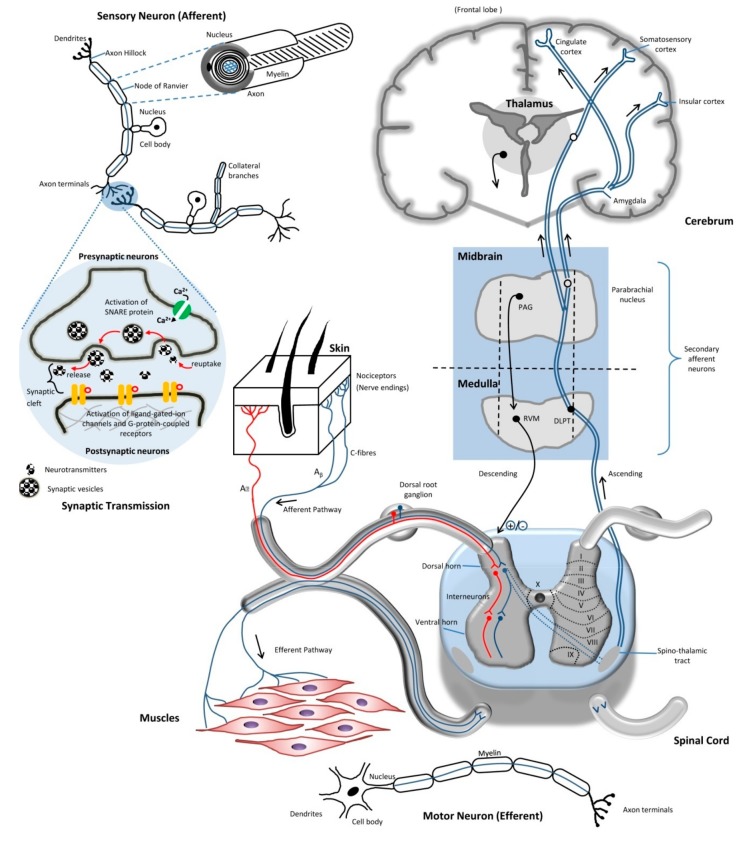
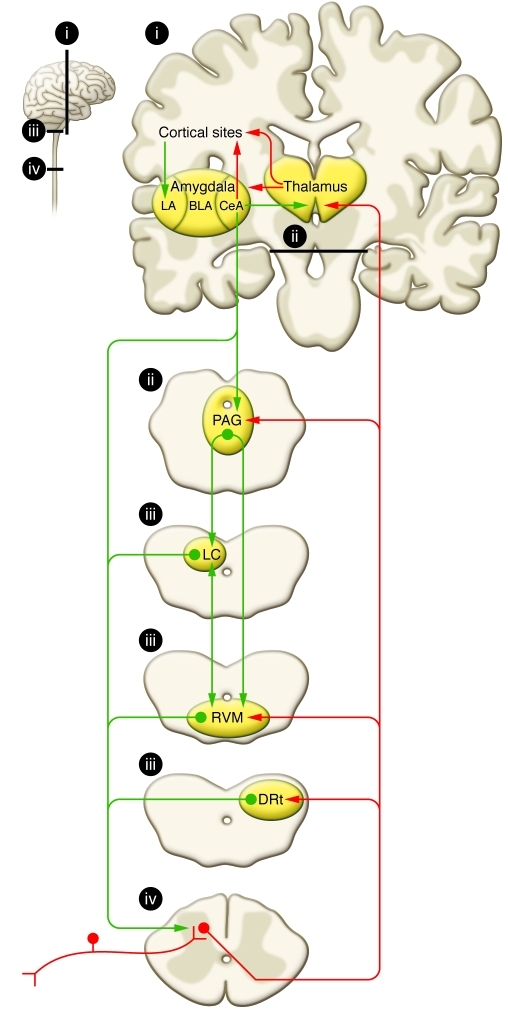
Peripheral & Central Sensitization
When tissue injury or nerve damage persists, both peripheral nociceptors and central neurons become hyperexcitable. Peripheral sensitization lowers nociceptor firing thresholds (producing primary hyperalgesia at the site of injury). Central sensitization amplifies signals at the dorsal horn through NMDA-receptor activation, producing allodynia (pain from a normally non-painful stimulus such as light touch), secondary hyperalgesia spreading beyond the injury site, and temporal summation ("wind-up"). Understanding this mechanism is the reason chronic pain patients describe pain that "does not match" the imaging — the nervous system, not just the tissue, is the generator.
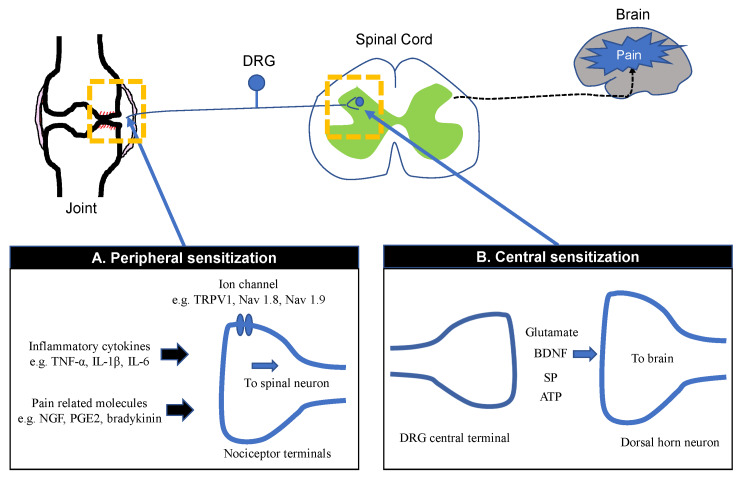
When a clinician says "the patient has allodynia and hyperalgesia in a glove distribution," they are describing central/peripheral sensitization — not making a casual observation. Always chart these exact words. They drive both diagnosis (CRPS, neuropathy) and treatment selection (gabapentinoids, SNRIs, topical lidocaine).
02 Scribe Documentation Framework
Pain encounters fall into four templates: new consultation, medication follow-up, procedure visit, and post-procedure follow-up. Each has its own required elements, and you will learn to toggle between them fluidly.
Provokes/Palliates, Quality, Region/Radiation, Severity (NRS 0–10 current / average / worst / best in past week), Timing (constant, intermittent, diurnal pattern), Understanding (patient's belief about cause). Always capture functional impact: sleep, ambulation, ADLs, work, mood. Pain documentation that is missing a functional narrative is incomplete.
(1) Physical function — walking distance, stairs, ability to sit/stand, yard work, lifting. (2) Sleep — hours, awakenings due to pain, use of sleep aids. (3) Mood — depression, anxiety, catastrophizing (PHQ-9, GAD-7 if documented). (4) Work/ADLs — employment status, disability claim, able to dress/bathe/cook independently. These domains drive medical necessity for interventional procedures and controlled substance continuation.
The Controlled Substance Visit
Any encounter where opioids, benzodiazepines, or other schedule II–IV medications are prescribed requires additional documentation beyond the standard SOAP note. This is not optional — it is the scaffolding of defensible pain practice and is mandated by state medical boards, DEA, and the 2022 CDC Clinical Practice Guideline for Prescribing Opioids.
1. Pain intensity (NRS) and functional improvement since last visit — "the 4 A's" (Analgesia, Activities, Adverse effects, Aberrant behavior).
2. PDMP (Prescription Drug Monitoring Program) review date and findings.
3. Urine drug screen (UDS) date and results consistency.
4. Controlled substance agreement (pain contract) on file.
5. Morphine milligram equivalent (MME) daily dose calculation.
6. Naloxone co-prescription documented.
7. Opioid Risk Tool (ORT) or SOAPP-R score if new patient.
8. Discussion of risks, alternatives, and goals of therapy.
When the physician says "PDMP reviewed, no red flags" — chart the exact phrase plus the date of query. Never write "PDMP normal" if the physician did not use that word. These notes are subpoenaed more often than you think, and documentation precision protects both the patient and the practice.
03 Chronic Low Back Pain Axial Spine
Chronic low back pain (CLBP) is the single highest-volume diagnosis in any pain clinic. Low back pain lasting > 12 weeks is considered chronic. The scribe's job is to classify it by the suspected pain generator because management diverges dramatically.
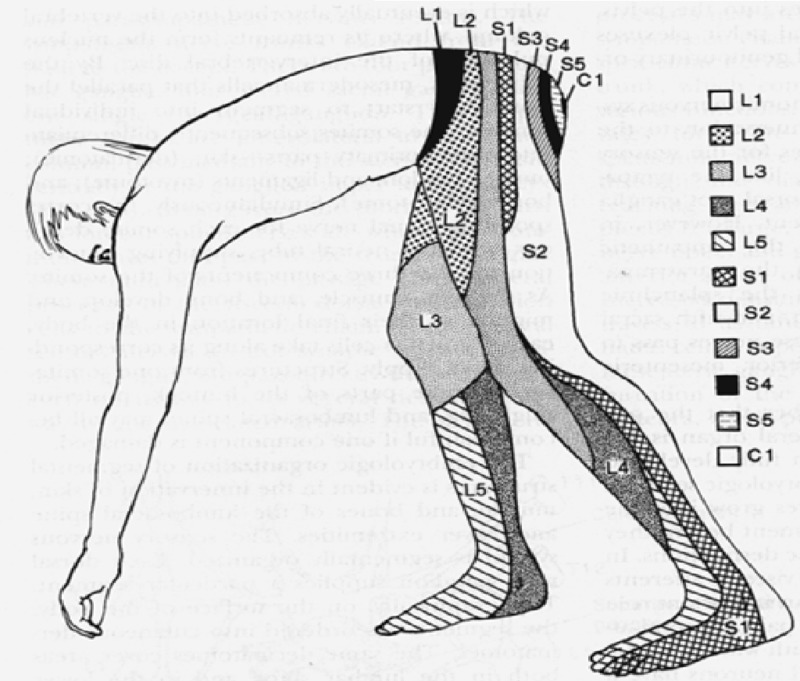
Three Clinical Subtypes
Pain localized to the lumbar region without radiation below the knee. Usually nociceptive from facet joints, discs, sacroiliac joints, or paraspinal musculature. Worsened by extension (facet) or flexion/sitting (discogenic). Does not follow a dermatomal pattern.
Sharp, lancinating, electric pain radiating in a dermatomal distribution below the knee, most commonly L5 (dorsum of foot) or S1 (lateral foot/heel). Caused by nerve root irritation/compression from disc herniation, foraminal stenosis, or lateral recess stenosis. Positive straight leg raise. Neuropathic in quality and responds to gabapentinoids + epidural steroid injection.
Bilateral leg heaviness, cramping, and weakness brought on by standing or walking and relieved by sitting or lumbar flexion ("shopping cart sign"). Classic presentation of central lumbar spinal stenosis. Must be distinguished from vascular claudication (relieved by standing still, no flexion benefit) and typically requires MRI confirmation.
Red Flags That Change Management Immediately
Any of these findings demand urgent imaging and escalation: saddle anesthesia, bowel/bladder dysfunction, progressive motor weakness (cauda equina), fever + IV drug use (epidural abscess), history of cancer with new back pain (metastasis), unexplained weight loss, major trauma, or osteoporosis with acute pain (vertebral compression fracture).
Management
First-line is a combination of NSAIDs, acetaminophen, activity modification, physical therapy, and cognitive-behavioral therapy. The American College of Physicians 2017 low back pain guideline recommends non-pharmacologic therapy (exercise, CBT, acupuncture, spinal manipulation) as first-line. When conservative therapy fails, interventional options are selected based on the suspected generator: transforaminal epidural steroid injection for radicular pain, medial branch block → radiofrequency ablation for facet-mediated pain, SI joint injection for sacroiliac dysfunction, or spinal cord stimulator trial for failed back surgery syndrome.
04 Failed Back Surgery / Post-Laminectomy Syndrome Axial Spine
Also called post-laminectomy syndrome or persistent spinal pain syndrome type 2, FBSS refers to persistent or recurrent low back and/or leg pain after one or more lumbar spine surgeries. Estimated to affect 20–40% of post-lumbar-surgery patients. The pain generator may be recurrent disc herniation, epidural fibrosis (scar tissue), adjacent segment disease, arachnoiditis, pseudoarthrosis at a fusion site, or central sensitization. FBSS is one of the primary FDA-approved indications for spinal cord stimulation and dorsal root ganglion stimulation.
Treatment is layered: neuropathic medications (gabapentin, pregabalin, duloxetine), targeted interventions (transforaminal ESI if radicular, adhesiolysis if epidural fibrosis), and neuromodulation (SCS trial) for refractory cases. SENZA-RCT established high-frequency (10 kHz) SCS superiority over traditional tonic stimulation for back and leg pain.
05 Facet Arthropathy & Sacroiliac Joint Dysfunction Axial Spine
Facet (Zygapophyseal) Joint Pain
Facet joints are paired synovial joints at each spinal level. Degenerative arthropathy produces axial pain that worsens with extension and rotation, is typically unilateral or bilateral paraspinal, and may refer into the buttock or posterior thigh but rarely below the knee. Diagnosis is clinical but confirmed by dual diagnostic medial branch blocks (MBB) — two separate injections at the nerves innervating the suspected facet joints. A positive response (>50–80% pain relief for the expected anesthetic duration, on both blocks) qualifies the patient for radiofrequency ablation (RFA), also called medial branch neurotomy, which produces 6–18 months of relief by thermally lesioning the medial branch nerves.
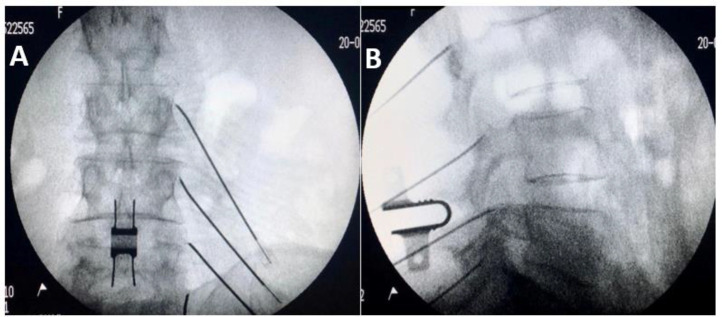
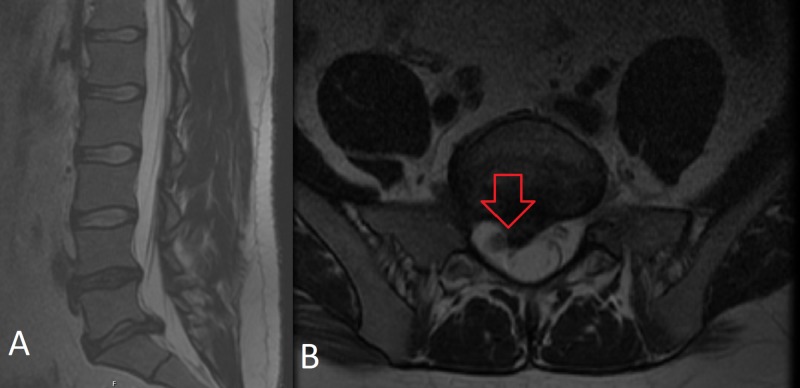
Sacroiliac Joint Dysfunction
The SI joint transmits load between the spine and pelvis and is a common source of low back and buttock pain, particularly in post-fusion patients, pregnant/postpartum women, and after trauma. Pain is usually unilateral, centered over the posterior superior iliac spine (PSIS), and may refer into the posterior thigh. Provocation tests (FABER/Patrick, Gaenslen, thigh thrust, compression, distraction) are positive. Three or more positive maneuvers increase diagnostic confidence. Treatment progresses from PT → intra-articular SI injection → lateral branch RFA → SI joint fusion (iFuse, SImmetry) for refractory cases.
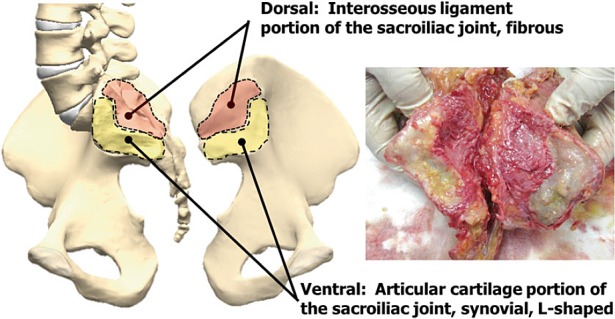
Document the exact percentage of pain relief after a diagnostic block (e.g., "80% relief lasting 5 hours"). Insurers require dual positive MBBs before authorizing RFA, and the percent-relief numbers go directly into the prior authorization. A fuzzy "patient reported good relief" is not enough.
06 Disc Herniation & Radiculopathy Axial Spine
Intervertebral disc herniation — protrusion, extrusion, or sequestration of the nucleus pulposus through the annulus fibrosus — compresses or inflames an exiting nerve root producing radicular pain in a dermatomal distribution. In the lumbar spine, the most common levels are L4–L5 (affecting the L5 root, with dorsum-of-foot pain, great toe weakness, and diminished sensation) and L5–S1 (affecting S1, with posterior calf/lateral foot pain, diminished Achilles reflex, and gastrocnemius weakness). In the cervical spine, C5–C6 and C6–C7 dominate, producing arm pain with specific motor and reflex patterns.
Cervical Radiculopathy Patterns
| Root | Pain / Sensory | Motor Weakness | Reflex |
|---|---|---|---|
| C5 | Lateral deltoid | Deltoid, biceps | Biceps |
| C6 | Lateral forearm, thumb | Biceps, wrist extensors | Brachioradialis |
| C7 | Middle finger, posterior arm | Triceps, wrist flexors | Triceps |
| C8 | Medial forearm, small finger | Finger flexors, intrinsics | — |
| T1 | Medial arm | Hand intrinsics | — |
Management
Most disc herniations improve with 6–12 weeks of conservative care (NSAIDs, gabapentin, PT, activity modification). Transforaminal epidural steroid injections are first-line interventional for persistent radicular pain. Surgical referral is triggered by progressive motor weakness, cauda equina, intractable pain despite injection, or failure of 6–12 weeks of conservative management.
07 Spinal Stenosis & Neurogenic Claudication Axial Spine
Central, lateral recess, or foraminal narrowing of the spinal canal from degenerative changes (hypertrophied ligamentum flavum, facet hypertrophy, disc bulge, spondylolisthesis). Lumbar spinal stenosis presents with neurogenic claudication; cervical spinal stenosis with myelopathy (wide-based gait, hand clumsiness, hyperreflexia, positive Hoffmann, urinary urgency).
Epidural steroid injection (interlaminar or transforaminal) provides short- to medium-term relief. MILD (Minimally Invasive Lumbar Decompression) percutaneously debulks hypertrophied ligamentum flavum. Vertiflex Superion is an interspinous spacer implanted percutaneously to indirectly decompress the canal. Open laminectomy remains the definitive surgical option for severe stenosis with functional limitation.
08 Headache & Craniofacial Pain Syndromes Craniofacial
Headache disorders are frequently co-managed with neurology, but pain medicine handles interventional treatment and many refractory cases.
Headache on ≥ 15 days/month for > 3 months with migrainous features on ≥ 8 days/month. OnabotulinumtoxinA (Botox) per the PREEMPT protocol (155 units across 31 sites, every 12 weeks) is FDA-approved. CGRP monoclonal antibodies (erenumab, fremanezumab, galcanezumab, eptinezumab) are the newest preventive class.
Severe unilateral orbital/supraorbital/temporal pain with ipsilateral autonomic features (ptosis, miosis, lacrimation, rhinorrhea, conjunctival injection) in episodic cluster periods. High-flow oxygen and subcutaneous sumatriptan abort attacks. Occipital nerve blocks and sphenopalatine ganglion blocks are used for bridging and refractory cases.
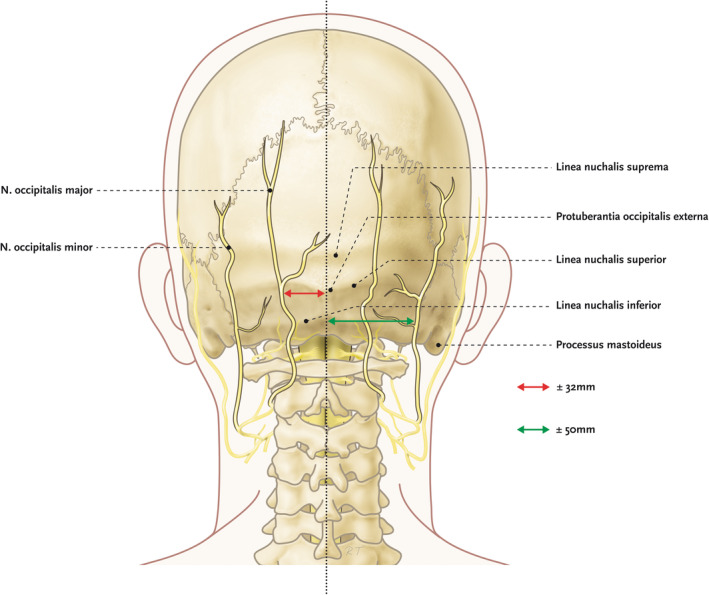
Sharp, shooting pain in the distribution of the greater/lesser occipital nerves, often from C2–C3 facet arthropathy or occipital nerve entrapment. Occipital nerve block with lidocaine + steroid is diagnostic and therapeutic. Cervicogenic headache refers from upper cervical structures and responds to third occipital nerve / C2–C3 medial branch blocks and RFA.
Paroxysmal, lancinating, electric-shock-like pain in one or more trigeminal divisions (V1/V2/V3), often triggered by chewing, talking, or light touch to a cutaneous trigger zone. First-line: carbamazepine or oxcarbazepine. Refractory: microvascular decompression, percutaneous glycerol rhizotomy, balloon compression, or gamma knife radiosurgery.
Headache ≥ 15 days/month in a patient with pre-existing primary headache who has overused acute headache medication (triptans, combination analgesics, opioids, butalbital ≥ 10 days/month; simple analgesics ≥ 15 days/month) for > 3 months. Treatment is withdrawal of the overused agent plus bridging therapy and initiation of prevention.
09 Neuropathic Pain Syndromes Neuropathic
Neuropathic pain arises from lesion of the somatosensory nervous system. Evaluation tools include the DN4 questionnaire and painDETECT. The IASP NeuPSIG treatment recommendations form the backbone of pharmacologic management.
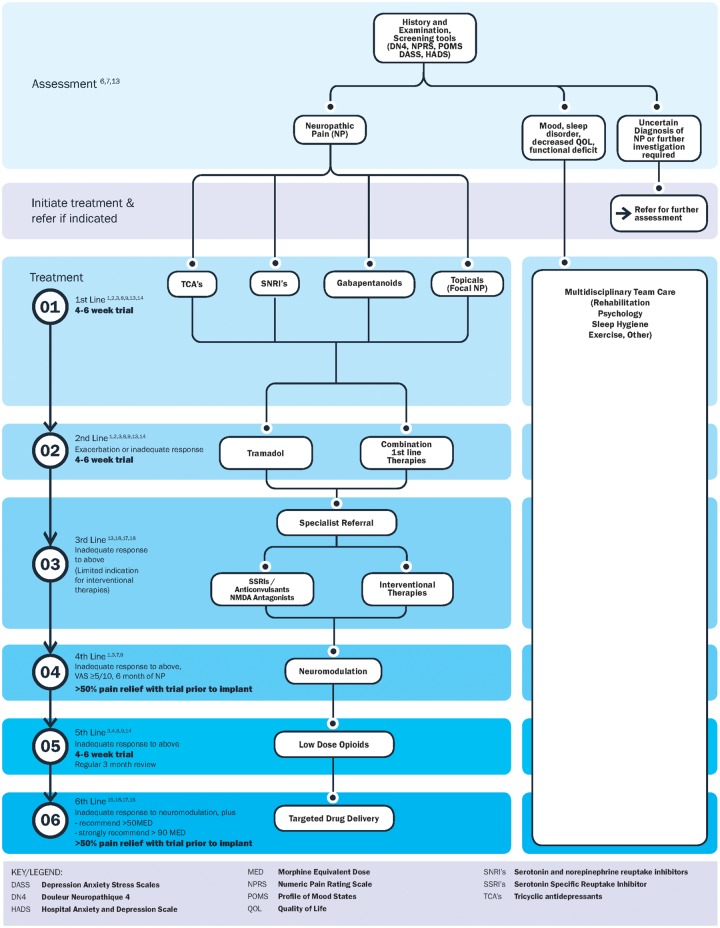
Symmetric stocking-glove distribution of burning, tingling, numbness, and allodynia. Diabetic peripheral neuropathy (DPN) is the most common cause in pain clinics. Chemotherapy-induced peripheral neuropathy (CIPN) is prominent with taxanes, platinums, vincristine, and bortezomib. First-line: duloxetine (FDA-approved for DPN), gabapentin, pregabalin, TCAs. Topical lidocaine 5% patch and capsaicin 8% patch are adjuncts.
Persistent dermatomal burning/allodynia > 90 days after a shingles outbreak, most commonly thoracic or trigeminal (V1). Prevention is the recombinant zoster vaccine (Shingrix). Treatment: gabapentinoids, TCAs, topical lidocaine 5% patch (FDA-approved for PHN), topical capsaicin 8%.
Phantom limb pain (perceived in the absent limb) and residual limb (stump) pain affect a large majority of amputees. Mechanisms include cortical reorganization, ectopic firing in neuromas, and central sensitization. Treatment: gabapentin, mirror therapy, targeted muscle reinnervation, peripheral nerve stimulation, and intrathecal therapy for refractory cases.
Carpal tunnel (median at wrist), cubital tunnel (ulnar at elbow), meralgia paresthetica (lateral femoral cutaneous), tarsal tunnel (posterior tibial), common peroneal at fibular head, piriformis syndrome (sciatic). Managed with splinting, steroid injection under ultrasound, hydrodissection, and neurolysis.
10 Complex Regional Pain Syndrome Neuropathic
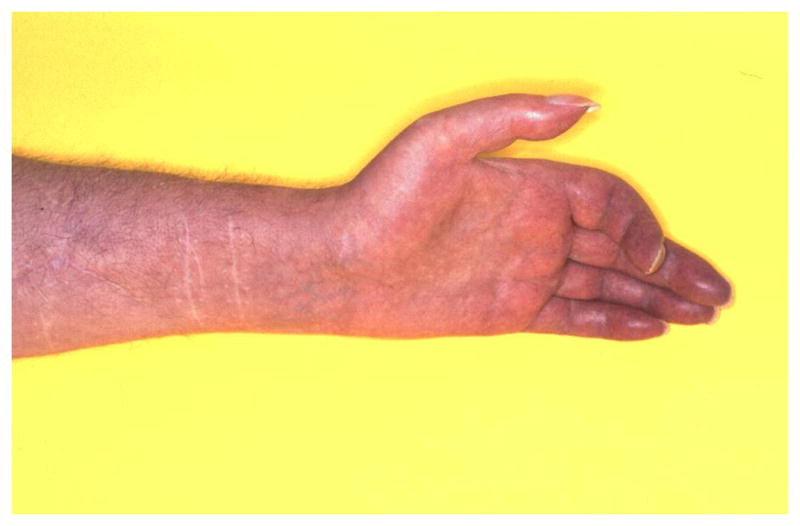
CRPS is a disproportionate pain syndrome following injury or surgery, characterized by regional sensory, motor, autonomic, and trophic disturbances. CRPS Type I (formerly reflex sympathetic dystrophy) has no identifiable nerve lesion; Type II (formerly causalgia) follows a defined nerve injury. Diagnosis is clinical and uses the Budapest criteria.
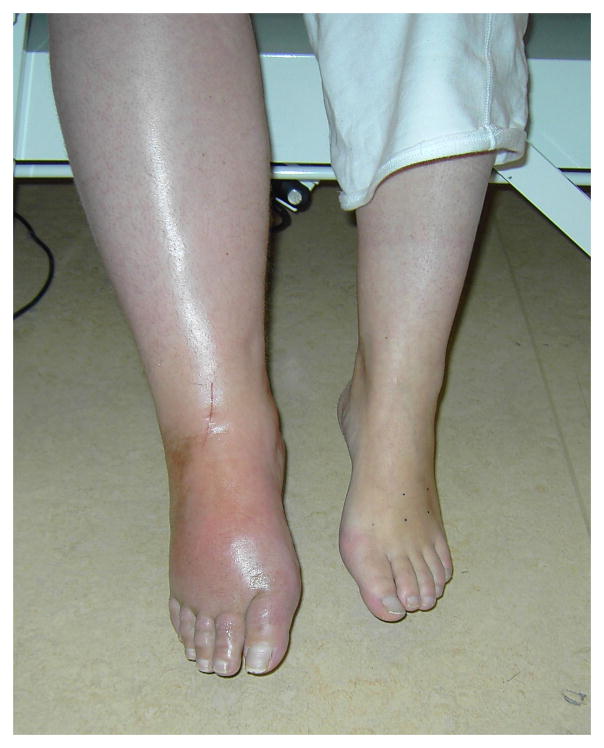
(1) Continuing pain disproportionate to any inciting event.
(2) Must report at least one symptom in three of four categories:
• Sensory: hyperalgesia, allodynia
• Vasomotor: temperature asymmetry, skin color changes
• Sudomotor/edema: edema, sweating changes
• Motor/trophic: decreased ROM, weakness, tremor, nail/hair/skin changes
(3) Must display at time of evaluation at least one sign in two or more of these categories.
(4) No other diagnosis better explains the signs and symptoms.
Management
Early aggressive multidisciplinary treatment is key. Physical/occupational therapy with desensitization and graded motor imagery is foundational. Pharmacologic: gabapentin, pregabalin, amitriptyline, bisphosphonates (pamidronate, neridronate), and short oral steroid bursts in early disease. Interventional: stellate ganglion block for upper extremity, lumbar sympathetic block for lower extremity. Ketamine infusion, dorsal root ganglion (DRG) stimulation (FDA-approved for CRPS of the lower extremity), and spinal cord stimulation for refractory cases.
11 Myofascial, Fibromyalgia & Nociplastic Pain Nociplastic
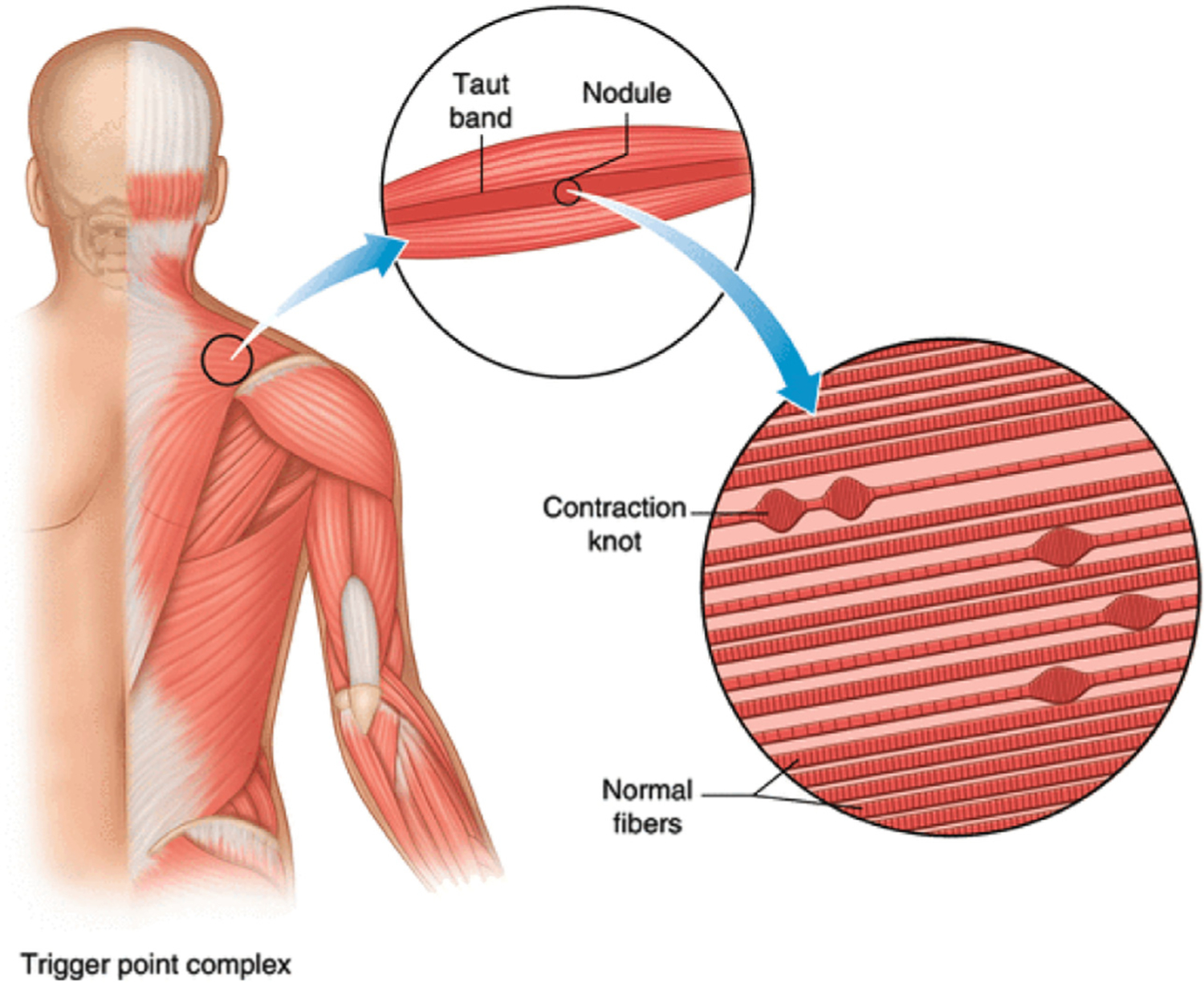
Regional pain originating from trigger points — discrete hyperirritable nodules within taut bands of skeletal muscle that refer pain in predictable patterns. Common in trapezius, levator scapulae, quadratus lumborum, gluteus medius, and piriformis. Treatment: trigger point injection with local anesthetic (with or without steroid), dry needling, stretching, and correction of postural/ergonomic contributors.
Widespread pain (above and below the waist, both sides of body, axial), chronic fatigue, non-restorative sleep, and cognitive dysfunction ("fibro fog"). Diagnosed using the 2016 ACR criteria (Widespread Pain Index + Symptom Severity Scale). FDA-approved medications: pregabalin, duloxetine, milnacipran. Non-pharmacologic: graded aerobic exercise, CBT, tai chi, sleep hygiene. Opioids are not recommended. Overlaps substantially with rheumatology.
12 Cancer, Visceral & Pelvic Pain Visceral/Cancer
Cancer pain is typically mixed — nociceptive (bone metastasis, soft tissue invasion), neuropathic (nerve compression, chemotherapy), and visceral (organ capsule stretch, obstruction). The WHO analgesic ladder remains the global framework.
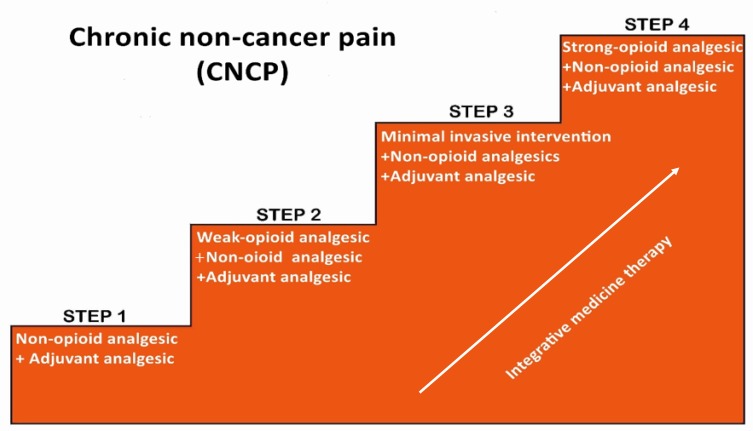
Step 1 — Mild pain: Non-opioid (acetaminophen, NSAID) ± adjuvant.
Step 2 — Moderate pain: Weak opioid (tramadol, codeine, low-dose hydrocodone) + non-opioid ± adjuvant.
Step 3 — Severe pain: Strong opioid (morphine, oxycodone, hydromorphone, fentanyl, methadone) + non-opioid ± adjuvant.
Step 4 (modern addition): Interventional — intrathecal pump, neurolytic blocks, vertebral augmentation.
Interventional Options for Cancer Pain
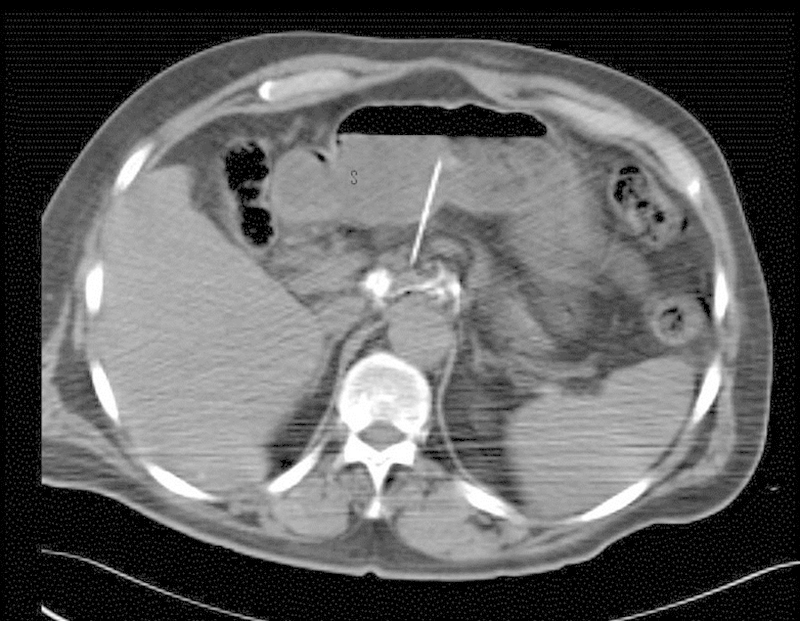
- Celiac plexus block/neurolysis — upper abdominal visceral pain (pancreatic cancer).
- Superior hypogastric plexus block — pelvic visceral pain (cervical, prostate, rectal cancer).
- Ganglion impar block — perineal/rectal/vaginal pain.
- Intercostal nerve block / cryoablation — chest wall pain, rib metastases.
- Kyphoplasty / vertebroplasty — painful vertebral compression fractures (osteoporotic or metastatic).
- Intrathecal pump (Medtronic SynchroMed) — refractory cancer pain; delivers morphine, ziconotide, bupivacaine, or clonidine directly to CSF.
Interstitial cystitis/bladder pain syndrome, chronic prostatitis/chronic pelvic pain syndrome, vulvodynia, pudendal neuralgia, and endometriosis-related pain are commonly co-managed. Pudendal nerve blocks, ganglion impar blocks, and pelvic floor physical therapy are mainstays.
13 Post-Surgical & Post-Thoracotomy Pain Neuropathic
Chronic post-surgical pain (CPSP) develops after 10–50% of major surgeries. Recognized syndromes include post-thoracotomy pain syndrome (intercostal nerve injury), post-mastectomy pain syndrome (intercostobrachial neuralgia), post-inguinal herniorrhaphy pain (ilioinguinal/iliohypogastric entrapment), and post-amputation pain. Management combines neuropathic medications, intercostal nerve blocks, cryoablation or pulsed radiofrequency of intercostal nerves, and peripheral nerve stimulation. Intercostal neuralgia and abdominal wall pain (anterior cutaneous nerve entrapment syndrome, ACNES) are diagnosed with targeted nerve blocks.
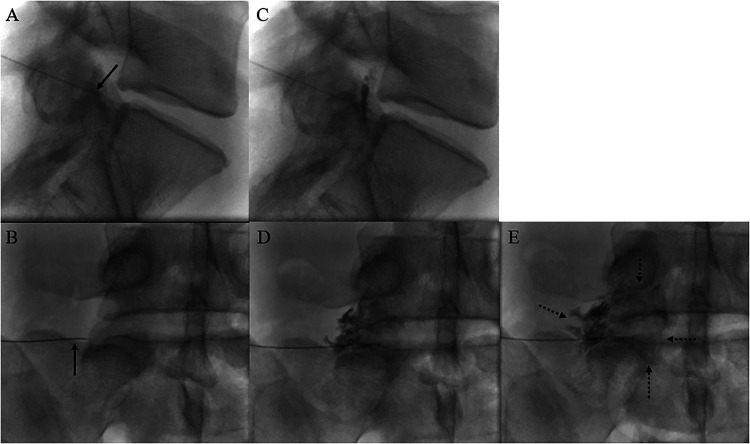
14 Interventional Procedures — A to Z Procedural
Interventional pain procedures form the backbone of any pain clinic's revenue and clinical value. You will document dozens of these per week. Each procedure has specific consent language, fluoroscopy or ultrasound guidance, injectate composition, and post-procedure instructions.
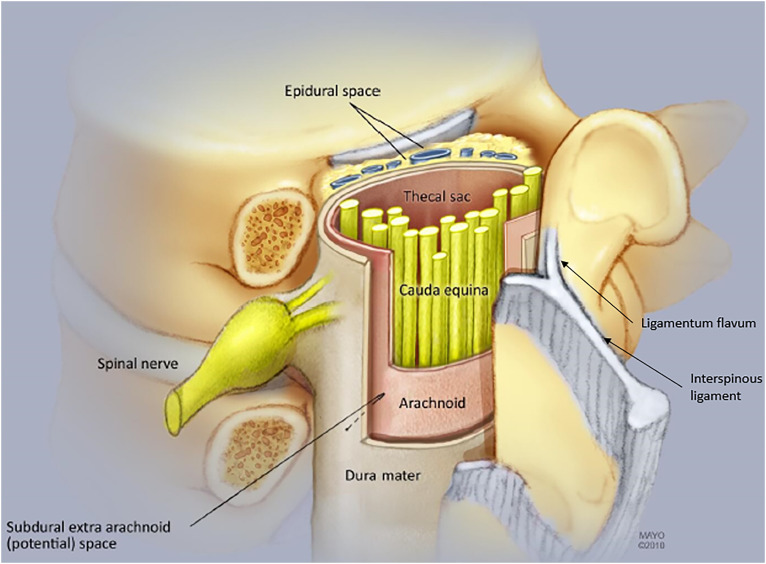
Spine Injections
| Procedure | Indication | Key Details |
|---|---|---|
| Interlaminar ESI (cervical, thoracic, lumbar) | Axial + multilevel radicular pain | Loss-of-resistance technique; fluoro with contrast |
| Transforaminal ESI | Unilateral radiculopathy at a specific level | "Safe triangle" approach; dexamethasone preferred cervically (embolic risk) |
| Caudal epidural | Post-surgical back/multilevel lumbar pain | Through sacral hiatus; highest-volume injection |
| Selective nerve root block (SNRB) | Diagnostic level localization | Very small volume at a single root |
| Facet joint injection | Suspected facet-mediated axial pain | Intra-articular LA + steroid |
| Medial branch block (MBB) | Diagnostic for RFA candidacy | Two positive blocks required by most payers |
| Radiofrequency ablation (RFA) | After positive MBBs | 80 °C x 90 seconds; 6–18 months relief |
| SI joint injection | Sacroiliac dysfunction | Intra-articular under fluoro or ultrasound |
| Lateral branch RFA (SI) | SI joint pain after positive diagnostic block | Targets S1–S3 lateral branches + L5 dorsal ramus |
| Discography | Diagnostic provocation for discogenic pain | Concordant pain reproduction at pathologic level |
| Basivertebral nerve ablation (Intracept) | Vertebrogenic low back pain with Modic changes | Transpedicular RFA of the basivertebral nerve |
| Kyphoplasty / vertebroplasty | Vertebral compression fracture | Cement augmentation through pedicle |
| MILD procedure | Central lumbar stenosis from ligamentum flavum hypertrophy | Percutaneous ligamentum debulking |
| Vertiflex Superion | Moderate lumbar stenosis with flexion relief | Interspinous spacer implant |
Sympathetic & Visceral Blocks
| Block | Target | Indications |
|---|---|---|
| Stellate ganglion block | C6/C7 sympathetic chain | Upper extremity CRPS, herpes zoster, hot flashes, PTSD (investigational) |
| Lumbar sympathetic block | L2–L4 sympathetic chain | Lower extremity CRPS, ischemic pain, phantom limb |
| Celiac plexus block | Anterocrural T12–L1 | Pancreatic cancer, upper abdominal visceral pain |
| Superior hypogastric plexus block | L5–S1 anterior | Pelvic visceral cancer pain |
| Ganglion impar block | Sacrococcygeal junction | Perineal/rectal/coccydynia pain |
| Sphenopalatine ganglion block | Pterygopalatine fossa | Cluster headache, chronic migraine, trigeminal neuralgia (V2) |
Peripheral & Soft Tissue Procedures
- Trigger point injection (TPI) — local anesthetic into myofascial trigger points.
- Occipital nerve block — greater and/or lesser occipital nerve for occipital neuralgia, cervicogenic headache, chronic migraine.
- Intercostal nerve block / cryoablation — rib fractures, post-thoracotomy pain, chest wall metastases.
- Peripheral nerve blocks — suprascapular, genicular (knee OA), pudendal, ilioinguinal, lateral femoral cutaneous (meralgia paresthetica), saphenous, tibial.
- Genicular nerve RFA — chronic knee osteoarthritis pain in patients who cannot or will not undergo TKA.
- Botox (onabotulinumtoxinA) — chronic migraine (PREEMPT), cervical dystonia, spasticity, sialorrhea, off-label myofascial pain.
- Intra-articular injections — hip, knee, shoulder, ankle with steroid or hyaluronic acid viscosupplementation (Synvisc, Euflexxa, Orthovisc).
- Regenerative — PRP, prolotherapy, stem cell injections (largely cash-pay, evidence mixed).
- Ketamine infusion — subanesthetic (0.1–0.5 mg/kg/hr) for CRPS, refractory neuropathic pain, treatment-resistant depression.
- Lidocaine infusion — 1–5 mg/kg over 30–60 minutes for neuropathic pain.
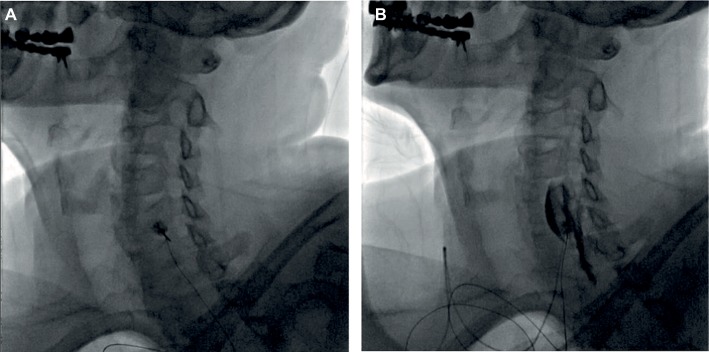
15 Neuromodulation & Implantable Devices Procedural
Neuromodulation is the fastest-growing segment of interventional pain medicine.
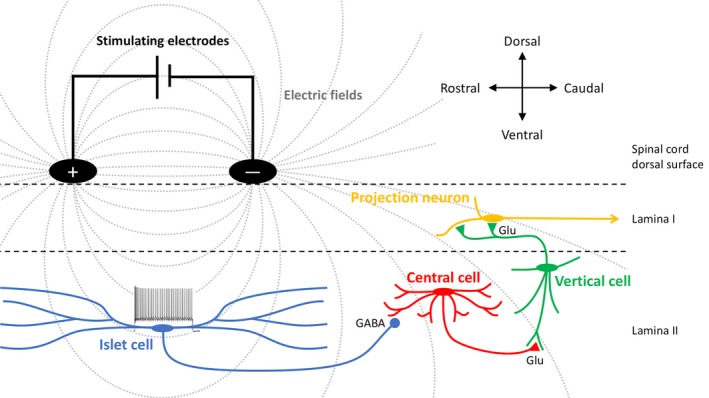
Epidural leads deliver electrical stimulation to the dorsal columns, modulating pain signaling. Indicated for FBSS, CRPS, painful diabetic neuropathy, refractory angina, and ischemic limb pain. Workflow: psychological clearance → 5–7 day percutaneous trial → permanent implant if ≥ 50% pain relief and functional improvement. Waveforms: traditional tonic (paresthesia-based), high-frequency 10 kHz (HF10, paresthesia-free, Nevro Senza), burst (Abbott), closed-loop ECAP-controlled (Saluda Evoke), differential target multiplexed (Medtronic).
Abbott Axium/Proclaim DRG. FDA-approved for lower extremity CRPS and causalgia. More focal coverage than SCS for groin, foot, and knee pain that is hard to capture with dorsal column stimulation.
Percutaneous leads placed near peripheral nerves (occipital, brachial plexus, suprascapular, tibial, genicular, intercostal, radial, axillary). Systems include SPRINT PNS (60-day temporary) and StimRouter (permanent). Indications include post-amputation pain, post-surgical neuralgia, occipital neuralgia, shoulder pain after stroke.
Implanted programmable pump (Medtronic SynchroMed II, Flowonix Prometra) delivers medication directly to the intrathecal space. FDA-approved agents: morphine, ziconotide (Prialt — non-opioid N-type calcium channel blocker from cone snail toxin), and baclofen (for spasticity). Off-label: bupivacaine, clonidine, hydromorphone, fentanyl. Indicated for refractory cancer pain and severe chronic non-cancer pain after failed conservative and interventional care.
16 Post-Procedure Care & Complications
Every procedure visit includes structured post-procedure documentation: vitals pre/post, sedation level (most pain procedures are done awake or with minimal sedation), immediate pain score change, neurologic exam unchanged, and return precautions.
Epidural abscess / meningitis: fever, severe spinal pain, new neurologic deficit — emergent MRI.
Epidural hematoma: progressive weakness, saddle anesthesia, bowel/bladder change — emergent decompression within 8–12 hours.
Intravascular injection (TFESI): particulate steroid embolism into radicular artery can cause spinal cord infarction — dexamethasone (non-particulate) preferred for cervical TFESI.
Pneumothorax: after thoracic, stellate ganglion, intercostal, or paravertebral block — dyspnea, chest pain, absent breath sounds.
Post-dural puncture headache: positional headache after inadvertent dural puncture — conservative care, caffeine, epidural blood patch.
Vasovagal syncope: very common intraprocedurally — always document BP, HR.
Local anesthetic systemic toxicity (LAST): perioral numbness, tinnitus, seizure, cardiac arrest — intralipid rescue.
Expected post-injection course: most patients have 1–3 days of injection-site soreness, a "steroid flare" (paradoxical pain increase for 24–72 hours) in up to 10%, a glucose spike in diabetics lasting 3–7 days, and facial flushing or insomnia from systemic steroid absorption. Peak benefit from epidural steroids is 1–3 weeks post-procedure. Follow-up is typically 2–4 weeks after injection to assess response.
17 Imaging & Diagnostics
Pain physicians rely on MRI, CT, X-ray, EMG/NCS, and diagnostic nerve blocks to identify the pain generator and justify intervention.
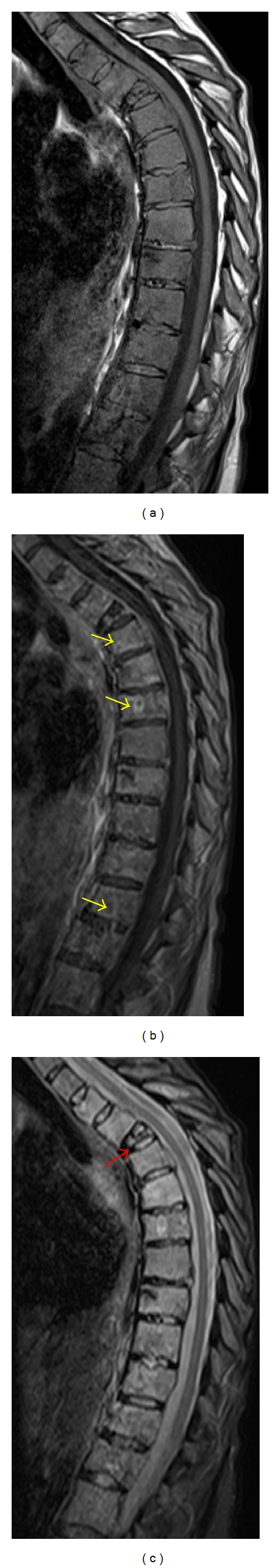
| Modality | Primary Use | What to Chart |
|---|---|---|
| MRI lumbar/cervical spine | Disc, nerve root, stenosis, tumor, infection | Level, side, Pfirrmann grade, foraminal/central narrowing |
| X-ray spine (flex/ext) | Instability, spondylolisthesis, alignment | Listhesis grade, motion on dynamic views |
| CT spine | Bony detail, fracture, post-fusion hardware | Fracture morphology, hardware integrity |
| DEXA | Osteoporosis screening before kyphoplasty | T-score |
| EMG / NCS | Confirm radiculopathy, neuropathy, entrapment | Active denervation, chronic changes |
| Diagnostic nerve block | Confirm pain generator | Percent relief, duration of relief |
| Fluoroscopy intraop | Procedure guidance | Contrast spread, needle position |
| Ultrasound | Soft tissue blocks, joint injection | Needle tip, spread pattern |
MRI Reading Vocabulary
You will chart terms like disc desiccation, annular fissure, posterior central protrusion, broad-based disc bulge, foraminal extrusion, ligamentum flavum hypertrophy, facet arthropathy, Modic type I/II endplate changes, Schmorl node, spondylolisthesis grade I–IV, pars defect (spondylolysis), central canal stenosis, lateral recess stenosis, neural foraminal narrowing (mild/moderate/severe), Tarlov cyst. Pfirrmann grading (I–V) scores disc degeneration on T2-weighted MRI.
18 Medications — Multimodal Analgesia
Multimodal analgesia layers agents with different mechanisms to maximize relief and minimize opioid exposure.
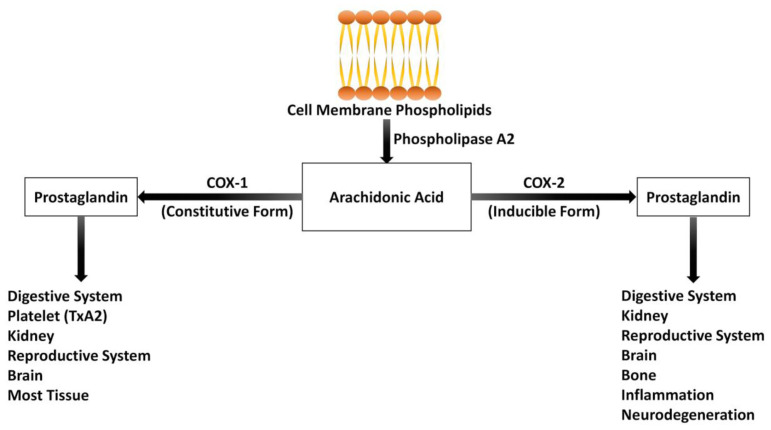
Non-Opioid Analgesics
| Drug | Brand | Class / Mechanism | Notes |
|---|---|---|---|
| Acetaminophen | Tylenol | Central COX | Max 3–4 g/day; hepatotoxic |
| Ibuprofen | Motrin, Advil | Non-selective NSAID | GI, renal, CV risk |
| Naproxen | Aleve | Non-selective NSAID | Longer half-life |
| Meloxicam | Mobic | Preferential COX-2 | Once daily dosing |
| Celecoxib | Celebrex | Selective COX-2 | Less GI risk; CV caution |
| Ketorolac | Toradol | IV/IM NSAID | Max 5 days |
| Diclofenac topical | Voltaren gel | Topical NSAID | Localized OA pain |
Neuropathic & Adjuvant Agents
| Drug | Brand | Use | Notes |
|---|---|---|---|
| Gabapentin | Neurontin | Neuropathic pain, PHN | Titrate; sedation, edema |
| Pregabalin | Lyrica | DPN, PHN, fibromyalgia, spinal cord injury pain | Schedule V; weight gain |
| Duloxetine | Cymbalta | DPN, fibromyalgia, chronic musculoskeletal pain | SNRI; BP check |
| Venlafaxine | Effexor XR | Neuropathic, migraine prevention | SNRI |
| Milnacipran | Savella | Fibromyalgia | SNRI |
| Amitriptyline | Elavil | Neuropathic, migraine, tension HA | TCA; anticholinergic, cardiac |
| Nortriptyline | Pamelor | Neuropathic | Better tolerated TCA |
| Carbamazepine | Tegretol | Trigeminal neuralgia (first-line) | HLA-B*1502 screening |
| Oxcarbazepine | Trileptal | Trigeminal neuralgia | Hyponatremia |
| Topiramate | Topamax | Migraine prevention | Cognitive dulling, weight loss |
| Lamotrigine | Lamictal | Central pain, trigeminal neuralgia | Slow titration (SJS) |
| Baclofen | Lioresal | Spasticity, trigeminal neuralgia | Do not stop abruptly |
| Tizanidine | Zanaflex | Muscle spasm | Sedation, hypotension |
| Cyclobenzaprine | Flexeril | Acute muscle spasm | Short-term use |
| Methocarbamol | Robaxin | Muscle spasm | Milder sedation |
| Ketamine | Ketalar | CRPS, neuropathic, refractory pain | NMDA antagonist; PO/IV/IN |
| Low-dose naltrexone | LDN | Fibromyalgia, CRPS | Off-label; 1.5–4.5 mg nightly |
Topical Agents
- Lidocaine 5% patch (Lidoderm) — FDA-approved PHN; used off-label for localized musculoskeletal and neuropathic pain. 12 hours on / 12 off.
- Capsaicin 8% patch (Qutenza) — in-office application for PHN, DPN of the feet, HIV neuropathy.
- Diclofenac gel 1% (Voltaren) — topical NSAID for hand/knee OA.
- Compounded creams — ketamine, gabapentin, amitriptyline, baclofen, clonidine, lidocaine combinations.
Opioids
| Drug | Brand | Form | Notes |
|---|---|---|---|
| Morphine IR / ER | MSIR, MS Contin, Kadian | PO, IV | Reference opioid for MME |
| Oxycodone IR / ER | Roxicodone, OxyContin | PO | Most commonly prescribed |
| Hydrocodone / APAP | Norco, Vicodin | PO | Schedule II since 2014 |
| Hydromorphone | Dilaudid, Exalgo ER | PO, IV | ~4x morphine potency |
| Fentanyl patch | Duragesic | Transdermal | 72 h; opioid-tolerant only |
| Methadone | Dolophine | PO | NMDA + mu; long, variable half-life; QTc |
| Buprenorphine | Belbuca, Butrans, Subutex, Suboxone | SL, buccal, TD, IM | Partial agonist; ceiling; increasingly first-line for chronic pain |
| Tapentadol | Nucynta | PO | Mu agonist + NRI |
| Tramadol | Ultram | PO | Weak mu + SNRI; seizure, serotonin syndrome |
| Codeine | Tylenol #3/#4 | PO | CYP2D6 variability |
| Naloxone | Narcan | IN, IM | Co-prescribe for MME ≥ 50 or risk factors |
Interventional Medications
Corticosteroids injected during procedures include methylprednisolone (Depo-Medrol), triamcinolone (Kenalog), betamethasone (Celestone), and dexamethasone (non-particulate, preferred for cervical transforaminal). Local anesthetics include lidocaine (fast onset, short duration), bupivacaine (slower, longer), and ropivacaine (less cardiotoxic). Hyaluronic acid viscosupplementation (sodium hyaluronate — Synvisc, Euflexxa, Orthovisc, Supartz) is injected into osteoarthritic joints.
19 Opioid Stewardship & Risk Mitigation
The 2022 CDC Clinical Practice Guideline for Prescribing Opioids for Pain replaced the 2016 version and emphasizes individualized, non-restrictive care while retaining the core principles of caution, multimodality, and risk mitigation.
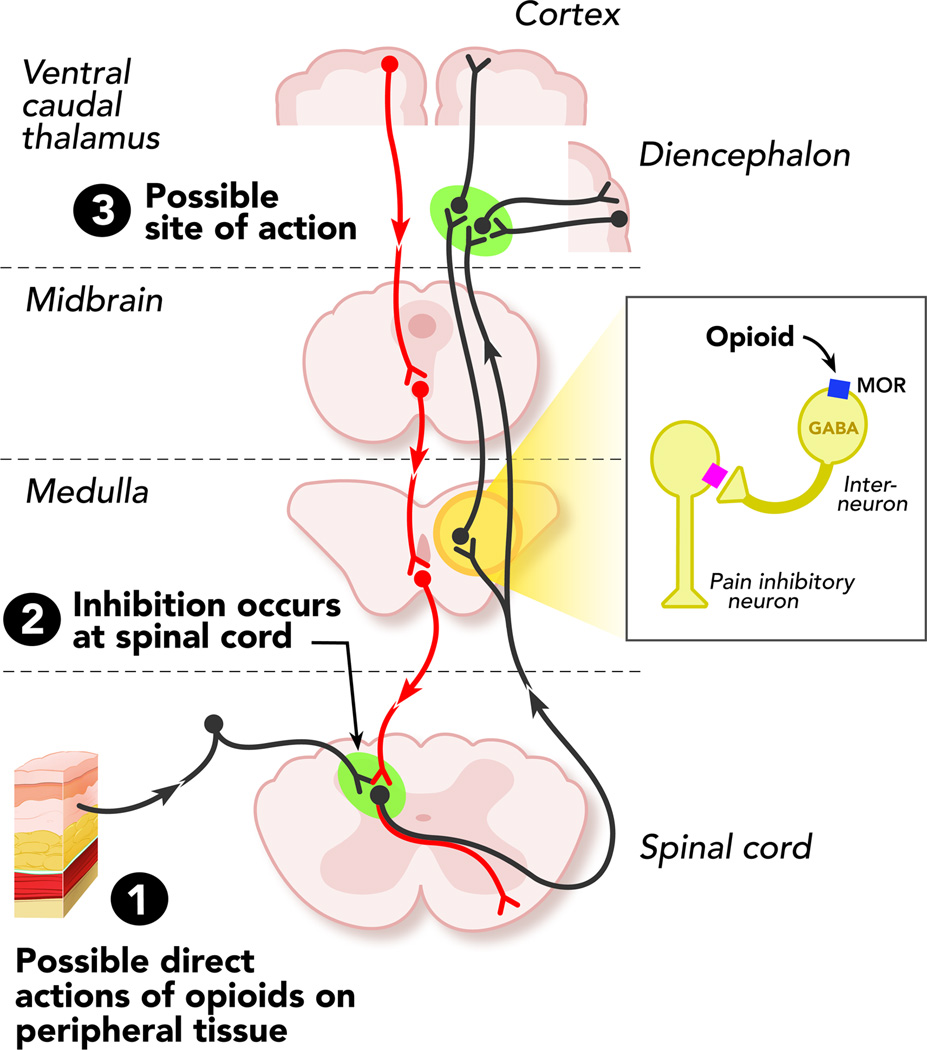
Morphine 1 mg PO = 1 MME
Hydrocodone 1 mg = 1 MME
Oxycodone 1 mg = 1.5 MME
Hydromorphone 1 mg = 4 MME
Oxymorphone 1 mg = 3 MME
Tramadol 1 mg = 0.1 MME
Fentanyl patch 25 mcg/hr ≈ 60 MME/day
Methadone: non-linear — 4 MME (at ≤ 20 mg/day), 10 MME (at 61–80 mg/day) — always verify with a calculator.
CDC thresholds: Caution ≥ 50 MME/day. Avoid ≥ 90 MME/day when possible or carefully justify. Co-prescribe naloxone for patients ≥ 50 MME/day, concurrent benzodiazepines, history of overdose, or SUD.
Opioid Risk Tool (ORT) — 10-item self-report; stratifies risk low/moderate/high.
SOAPP-R (Screener and Opioid Assessment for Patients with Pain — Revised) — 24-item; identifies aberrant behavior risk.
COMM (Current Opioid Misuse Measure) — 17-item; assesses current aberrant behavior in patients already on opioids.
PDMP — state prescription drug monitoring program; must be reviewed at every controlled substance visit per most state laws.
UDS — urine drug screen; presence/absence pattern assessed for consistency with prescribed regimen and undisclosed substances.
Paradoxical worsening of pain and development of diffuse allodynia with escalating opioid doses. Recognizing OIH is a skill because it resembles tolerance — but dose escalation worsens OIH, whereas tolerance may respond. Treatment: opioid rotation (often to buprenorphine or methadone), taper, ketamine, and non-opioid multimodal therapy.
Nearly universal side effect. Prophylaxis with osmotic laxative (polyethylene glycol) + stimulant (senna) at opioid initiation. Peripherally acting mu-opioid receptor antagonists (PAMORAs): methylnaltrexone (Relistor), naloxegol (Movantik), naldemedine (Symproic). Lubiprostone is also approved.
20 Classification & Assessment Systems
Pain Intensity Scales
Numeric Rating Scale (NRS 0–10): 0 = no pain, 10 = worst imaginable. Default in most U.S. pain clinics. Document current, average (past week), worst, and least.
Visual Analog Scale (VAS): 100 mm line marked by the patient; converted to 0–100.
Wong-Baker FACES: pediatric and limited-communication adults.
Verbal Rating Scale: none / mild / moderate / severe.
McGill Pain Questionnaire (MPQ): sensory, affective, evaluative descriptors for qualitative assessment.
Functional & Disability Indices
0–20%: Minimal disability. 21–40%: Moderate. 41–60%: Severe. 61–80%: Crippled. 81–100%: Bed-bound or exaggerated. Ten sections scoring pain, personal care, lifting, walking, sitting, standing, sleeping, sex life, social life, traveling.
Same 0–50 point structure applied to neck pain. Same severity bands as ODI.
Neuropathic Pain Screens
DN4: 10-item tool combining history and bedside exam; score ≥ 4/10 suggests neuropathic pain. painDETECT: validated questionnaire for identifying a neuropathic component. LANSS: Leeds Assessment of Neuropathic Symptoms and Signs.
CRPS
Budapest Clinical Criteria (see Section 10) is the current diagnostic standard.
FDA Opioid Risk Classification & Scheduling
| Schedule | Examples | Prescribing Notes |
|---|---|---|
| Schedule II | Morphine, oxycodone, hydrocodone, fentanyl, methadone, hydromorphone | No refills; written or e-prescribed (EPCS) |
| Schedule III | Buprenorphine, ketamine, codeine combos ≤ 90 mg/dose | 5 refills / 6 months |
| Schedule IV | Tramadol, benzodiazepines, carisoprodol | 5 refills / 6 months |
| Schedule V | Pregabalin (Lyrica), low-dose codeine cough preparations | Limited restrictions |
21 Physical Exam — The Pain Exam
The pain-specific exam blends musculoskeletal, neurologic, and observational components. Chart it in the same order every time.
Inspection: posture, gait (antalgic, Trendelenburg, steppage), surgical scars, skin changes, muscle atrophy.
Palpation: spinous processes, paraspinal muscles, SI joints, greater trochanters, ischial tuberosity, piriformis, trigger points.
Range of motion: cervical/lumbar flexion, extension, lateral bending, rotation — documented in degrees or as % of normal with pain provocation.
Provocative tests: straight leg raise (L5/S1), femoral stretch (L2–L4), Spurling (cervical radiculopathy), FABER/Patrick (hip/SI), Gaenslen, thigh thrust, compression, distraction (SI), Tinel (median/ulnar), Phalen, Hoffmann, Lhermitte, Waddell signs.
Motor strength: MRC 0–5 by myotome.
Sensory: light touch, pinprick, temperature by dermatome; allodynia/hyperalgesia mapping.
Reflexes: biceps, brachioradialis, triceps, patellar, Achilles (0 absent, 1+ diminished, 2+ normal, 3+ brisk, 4+ clonus); Babinski.
Coordination/gait: tandem, heel, toe walking; Romberg.
0 no contraction • 1 flicker • 2 active movement with gravity eliminated • 3 against gravity • 4 against resistance (4−, 4, 4+) • 5 normal.
Waddell signs (superficial tenderness, non-anatomic pain, distraction inconsistency, regional disturbance, overreaction) are pain-behavior observations, not proof of malingering. Chart them as the physician states them and never editorialize.
22 Abbreviations Master List
Diagnoses & Syndromes
Procedures
Medications & Assessment
23 Sample HPI Templates
The following HPIs represent the most common encounter types in a pain clinic. Study them until the rhythm is automatic.
"Mr. [Name] is a 54-year-old male with a history of type 2 diabetes, hypertension, obesity (BMI 34), and a remote L4–L5 microdiscectomy (2018) who presents as a new consult for chronic low back pain with right lower extremity radiation. Pain began insidiously 8 months ago without inciting trauma. He describes a constant 6/10 axial lumbar ache with intermittent 9/10 sharp electric pain radiating down the posterior right thigh into the lateral calf and dorsum of the foot. Pain is worsened by prolonged sitting, forward bending, and sneezing; partially relieved by walking and lying supine. He endorses subjective right foot dragging and numbness along the dorsum of the foot. Denies bowel/bladder changes, saddle anesthesia, or fevers. Has trialed ibuprofen 800 mg TID, cyclobenzaprine 10 mg QHS, 6 weeks of physical therapy, and a chiropractor without durable benefit. Sleep is disrupted 3–4 nights per week by pain; he walks less than 1 block before stopping. Functional goals: return to part-time landscaping work and play with grandchildren."
"Ms. [Name] is a 62-year-old female with persistent bilateral lower extremity burning neuropathic pain following an L4–S1 posterior lumbar fusion (2022), returning for evaluation for spinal cord stimulator trial. Despite successful radiographic fusion, she describes constant 7/10 burning and electric pain in both feet and calves with allodynia to sock contact. She has failed gabapentin 1200 mg TID (limited by edema), duloxetine 60 mg daily, two bilateral transforaminal ESIs at L5–S1 (< 25% relief), and a structured PT program. MME currently 35 (oxycodone 5 mg TID). PHQ-9 11 (moderate depression); psychology clearance completed. She is highly motivated for opioid reduction and improved ambulation. Plan discussed for SCS trial with HF10 waveform pending insurance authorization."
"Mr. [Name] is a 68-year-old male with long-standing type 2 diabetes (A1C 8.4), hypertension, and CKD stage 3 who presents for management of painful diabetic peripheral neuropathy. He describes symmetric burning and tingling in both feet that ascends to the mid-calf, worse at night and interfering with sleep (currently averaging 4 hours/night). Pain is 7/10 average, 10/10 worst, with allodynia to bedsheets. DN4 score 7/10 consistent with neuropathic pain. He failed gabapentin 900 mg TID due to dizziness and lower extremity edema. Currently tolerating duloxetine 30 mg daily with partial benefit. Exam today shows reduced vibratory and monofilament sensation to the mid-calf bilaterally, absent Achilles reflexes, and 5/5 strength. Today we will uptitrate duloxetine to 60 mg and add lidocaine 5% patches to the feet overnight."
"Ms. [Name] is a 41-year-old female who presents 10 weeks following an ORIF of a right distal radius fracture with progressive disproportionate pain of the right hand and forearm. She describes constant 8/10 burning pain with severe allodynia to light touch, intermittent color changes (blotchy red/blue), temperature asymmetry (right hand cooler than left), swelling, and progressive inability to move the fingers. Occupational therapy has been limited by pain. Exam demonstrates right hand edema, mottled skin, 2°C temperature asymmetry, hyperalgesia, and markedly reduced active and passive ROM of the fingers. Meets Budapest clinical criteria for CRPS Type I. Today we will proceed with right stellate ganglion block, initiate gabapentin 300 mg TID, continue intensive hand therapy with desensitization and mirror therapy, and discuss DRG stimulation candidacy if refractory."
"Mr. [Name] is a 71-year-old male 9 months post right upper lobectomy for stage IB NSCLC, presenting for persistent right chest wall neuropathic pain in the distribution of the T5–T7 intercostal nerves. He describes a constant 5/10 burning with intermittent 8/10 shooting pain along the thoracotomy scar, worsened by deep inspiration, coughing, and right shoulder motion. He has allodynia in a 10 cm band along the scar. Oncologically NED on recent surveillance imaging. Previously trialed gabapentin (discontinued due to sedation) and tramadol (minimal benefit). Today we will proceed with diagnostic right T5–T7 intercostal nerve blocks with plan for intercostal cryoablation if diagnostic response is positive."
"Mr. [Name] is a 58-year-old male on chronic opioid therapy for severe lumbar spondylosis and bilateral knee osteoarthritis, returning for routine 28-day follow-up and refill. Current regimen: oxycodone ER 20 mg BID + oxycodone IR 5 mg TID PRN (total 75 MME/day), gabapentin 600 mg TID, duloxetine 60 mg daily, meloxicam 15 mg daily, topical diclofenac. Pain averages 5/10 (down from 8/10 pre-treatment); walks 20 minutes continuously; maintains part-time work. Denies sedation, constipation (on Miralax), or respiratory symptoms. PDMP reviewed today — single prescriber, single pharmacy, no co-prescribed controlled substances. UDS from prior visit appropriate (positive for oxycodone, negative for all non-prescribed substances). Controlled substance agreement on file and reviewed. Naloxone nasal spray prescription current. Discussed ongoing goals, tapering considerations, and buprenorphine as a future option. Plan: continue current regimen, refill 28 days, UDS today, return in 4 weeks."
The best pain scribes think in mechanisms. When a patient describes burning foot pain worse at night in a diabetic, you should be mentally typing "neuropathic, DPN pattern, duloxetine candidate" before the physician has finished talking. When a patient reports buttock pain with extension relieved by flexion, you should be anticipating "facet arthropathy, MBB workup." This anticipation is what separates a good scribe from a great one.
Pain medicine is also the specialty where documentation precision has the highest legal and regulatory stakes. Chart the MME. Chart the PDMP date. Chart the exact percent of pain relief after a diagnostic block. Chart naloxone co-prescription. Chart functional status, not just pain scores. These details drive prior authorization, board defense, and patient safety simultaneously.
Welcome to pain management. You are joining a specialty that demands empathy for suffering, skepticism for narrative inconsistency, and the discipline to document both clearly.
24 References & Sources
Clinical Practice Guidelines
Landmark Clinical Trials & Classification Sources
Deer TR et al. ACCURATE Study: DRG Stimulation vs. SCS for CRPS and Causalgia. Pain. 2017.
Dworkin RH et al. PREEMPT: OnabotulinumtoxinA for chronic migraine. Headache. 2010.
Diagram & Figure Sources
Figure 1: Ascending Sensory Pathway — OpenStax Anatomy & Physiology. CC BY 3.0.
Figure 2: The Brain — OpenStax Anatomy & Physiology. CC BY 3.0.
Figure 3: Dermatomes — OpenStax Anatomy & Physiology. CC BY 3.0.
Figure 4: Herniated Disc — Blausen Medical. CC BY 3.0.
Figure 5: Epidural Steroid Injection — Blausen Medical. CC BY 3.0.
Figure 6: Spinal Cord Stimulator. Public domain, U.S. federal government.
Figure 7: Wong-Baker FACES Pain Rating Scale. Public domain.