Palliative Care / Hospice
Every symptom, syndrome, medication, opioid conversion, goals-of-care framework, abbreviation, and documentation pattern you need to chart palliative encounters with confidence.
All diagrams on this page are sourced from published educational and institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 Palliative Care Principles & Scope
Palliative care is specialized medical care for people living with serious illness. Its focus is the relief of physical symptoms, psychological distress, and existential suffering, as well as support for families and caregivers. Unlike hospice, palliative care can be delivered concurrently with disease-directed therapy at any stage of a serious illness — a newly diagnosed cancer patient receiving chemotherapy, a heart failure patient awaiting LVAD, and a dialysis patient with advanced CKD are all appropriate for a palliative consult.
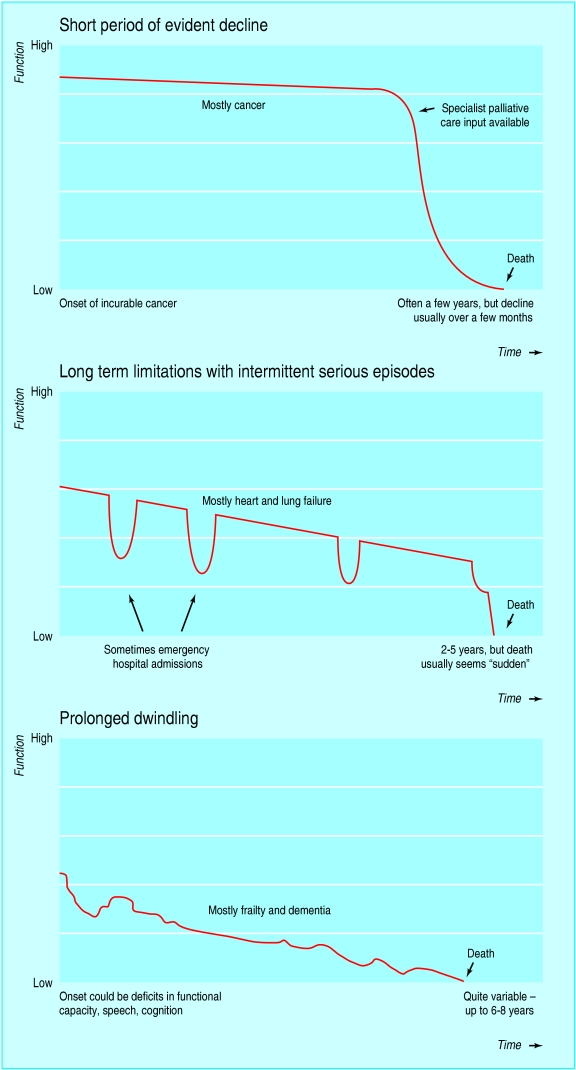
Hospice, by contrast, is a structured Medicare/Medicaid benefit for patients with a prognosis of six months or less if the illness runs its natural course, who elect to forgo curative treatment of their terminal diagnosis. Hospice care is most often delivered in the home, but can also occur in hospice inpatient units, skilled nursing facilities, or general inpatient (GIP) hospital beds. All hospice is palliative care, but not all palliative care is hospice — this distinction is one of the most common sources of confusion for new scribes.
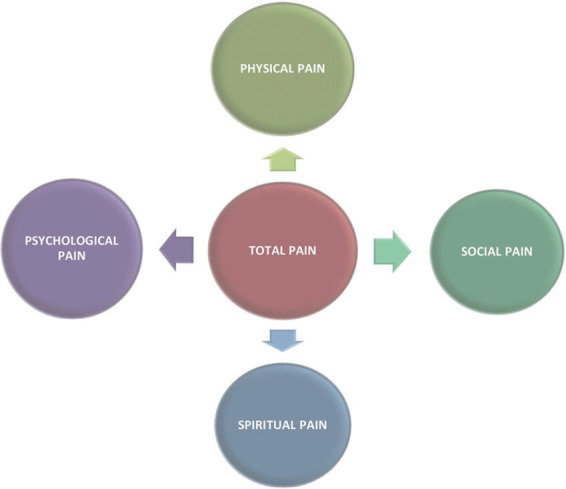
Every palliative encounter touches these eight domains, which mirror the NCP Clinical Practice Guidelines for Quality Palliative Care (4th ed.): (1) structure & process, (2) physical aspects, (3) psychological & psychiatric, (4) social, (5) spiritual/religious/existential, (6) cultural, (7) care of the patient nearing end of life, and (8) ethical and legal.
Palliative teams are interdisciplinary by design: physician or advanced practice provider, nurse, social worker, chaplain, and often pharmacist, music therapist, and bereavement counselor. Consults are generated by oncology, cardiology, pulmonology, critical care, nephrology, neurology, hospital medicine, and primary care, and the chief complaint is rarely "one thing" — it is almost always a combination of uncontrolled symptoms plus an unresolved decision-making need.
Never chart "palliative" as a synonym for "dying." A patient in active chemotherapy for metastatic breast cancer with uncontrolled neuropathic pain is a palliative patient. The attending will correct you fast if your HPI says "the patient is here for end-of-life care" when she is in fact here for an opioid titration.
02 Scribe Documentation Framework
The Palliative HPI
A palliative HPI is symptom-centric, goal-centric, and psychosocial-centric all at once. It typically opens with a one-line summary of the patient's life-limiting illness — "78-year-old man with metastatic castration-resistant prostate cancer to bone and liver, ECOG 3, admitted with uncontrolled pain and new confusion" — and then flows through the chief symptom, the trajectory of disease, prior treatments, current medications (with special attention to opioid regimen and breakthrough use), symptom burden across the body systems (pain, dyspnea, nausea, constipation, mood, sleep, appetite), and finally the patient's understanding of illness and goals.
- One-liner: age, sex, primary serious illness, stage/trajectory, functional status (ECOG/PPS/KPS), reason for consult
- Disease trajectory: date of diagnosis, treatments received, most recent imaging/labs, hospitalizations in past 6 months
- Symptom review (ESAS): pain, tiredness, drowsiness, nausea, appetite, shortness of breath, depression, anxiety, wellbeing, other — each 0–10
- Pain inventory: location(s), quality, intensity at rest/with movement, temporal pattern, radiation, exacerbating/relieving factors, current regimen, breakthrough use in last 24 hours, side effects
- Psychosocial: who lives at home, primary caregiver, surrogate, advance directive status, code status, spiritual/religious preferences
- Understanding & goals: what the patient knows, what they hope for, what they worry about, what they are willing or unwilling to accept in treatment
The Palliative A/P
The assessment/plan is organized by symptom, not by organ system. A typical plan will have separate paragraphs for pain, dyspnea, nausea, constipation, anxiety, and goals of care, each with a one-sentence assessment (is it controlled, worsening, or new?) followed by concrete interventions. For goals-of-care encounters, the plan paragraph documents the content of the conversation: who was present, what was discussed, what was decided, what remains unresolved, and what the next step is.
Always quote the patient directly when possible. "I just want to be comfortable" or "I don't want to be on a breathing machine again" carries enormous documentation weight in a goals-of-care note. Use quotation marks and attribute the speaker (patient, spouse, daughter).
03 Pain Assessment & Classification symptom
Pain is the most common reason for palliative consultation. The first task in any pain encounter is to characterize the pain mechanistically, because the right analgesic depends on what kind of pain you are treating.
Mechanistic Categories
- Somatic nociceptive pain — from skin, muscle, bone, joint. Well localized, aching, throbbing, sharp with movement. Classic example: bone metastases. Responds well to opioids, NSAIDs, acetaminophen, bisphosphonates, radiation.
- Visceral nociceptive pain — from hollow or solid organs. Poorly localized, deep, cramping, squeezing, often referred (pancreatic pain to mid-back, liver capsule pain to right shoulder). Responds to opioids and, when organ-specific, adjuvants such as glucocorticoids (liver capsule stretch) or antispasmodics.
- Neuropathic pain — from nerve injury or dysfunction. Burning, shooting, electric, tingling, numb, allodynic. Examples: chemotherapy-induced peripheral neuropathy, post-herpetic neuralgia, radiculopathy from epidural metastasis, plexopathy. Only partially responsive to opioids; requires adjuvants (gabapentinoids, TCAs, SNRIs).
- Mixed pain — the majority of cancer pain. A lung cancer patient with a Pancoast tumor has somatic chest wall pain, visceral mediastinal pain, and neuropathic brachial plexus pain all at once.
- Breakthrough pain — a transitory exacerbation of pain that "breaks through" otherwise adequately controlled background pain. Subtypes: incident (movement-provoked), spontaneous (unpredictable), and end-of-dose failure (wearing off before next scheduled dose). Requires a rapid-onset short-acting opioid, typically 10–20% of the total daily opioid dose q1–2h PRN.
Pain Assessment Tools
| Tool | Population | What it measures |
|---|---|---|
| NRS (0–10 Numeric Rating) | Cognitively intact adults | Self-reported intensity |
| Wong-Baker FACES | Pediatric, cognitively impaired, non-English speakers | Visual analog |
| PAINAD | Patients with advanced dementia | Breathing, vocalization, facial expression, body language, consolability (0–10) |
| CPOT | ICU / intubated | Facial expression, movement, muscle tension, ventilator compliance |
| Brief Pain Inventory (BPI) | Cancer pain | Intensity + interference with 7 life domains |
| ESAS-r | All palliative patients | 9-symptom 0–10 screen including pain |
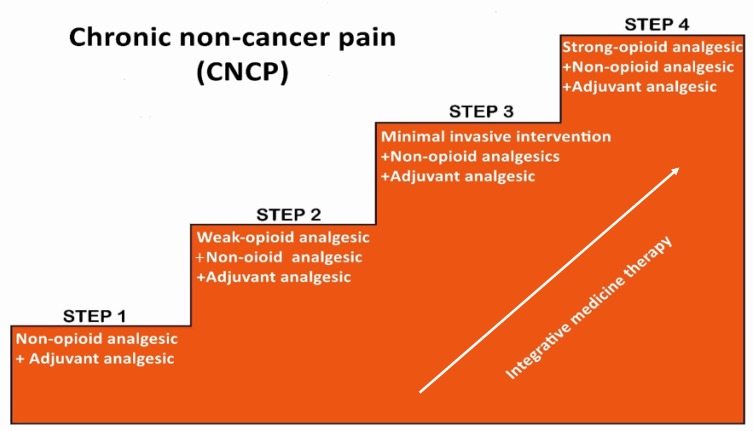
The WHO Analgesic Ladder
The original WHO Analgesic Ladder prescribed a three-step approach: Step 1 non-opioids (acetaminophen, NSAIDs) for mild pain, Step 2 weak opioids (codeine, tramadol) for moderate pain, and Step 3 strong opioids (morphine, oxycodone, hydromorphone, fentanyl, methadone) for severe pain. Modern practice generally skips Step 2 for cancer pain and moves directly from non-opioids to a low-dose strong opioid, with adjuvants added at every step.
Document pain like a forensic accountant. Location, quality, intensity now, intensity at worst, intensity at best, what makes it better, what makes it worse, how many breakthrough doses in the last 24 hours, how much relief each dose gave, how long the relief lasted, and any side effects. This level of detail is what drives the next titration decision.
04 Opioid Pharmacology & Equianalgesic Conversion medications
Opioids are the cornerstone of moderate-to-severe pain management in palliative care. A competent scribe must recognize every commonly used opioid, its routes, its relative potency, and the conversion math, because the physician will dictate conversions out loud and expect them to land in the plan correctly.
Opioids in Routine Use
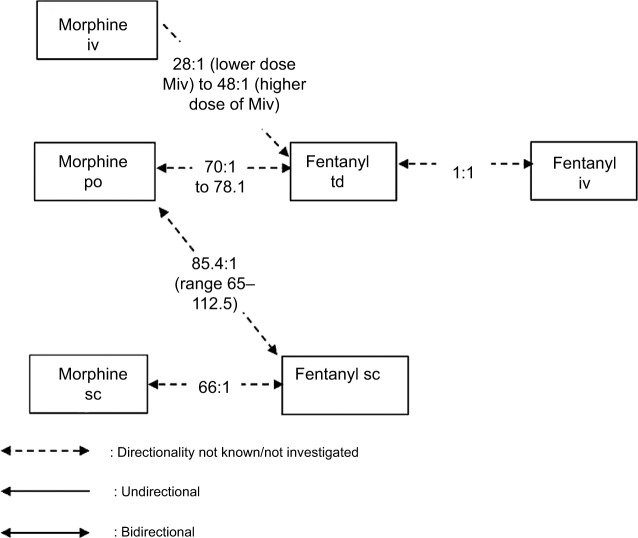
- Morphine (MS Contin, MSIR, Roxanol oral solution, Duramorph IV) — the reference opioid. PO, SL, PR, SC, IV, IT. Avoid in severe renal failure (active metabolite M6G accumulates).
- Oxycodone (Roxicodone IR, OxyContin ER) — PO only. ~1.5× as potent as PO morphine.
- Hydromorphone (Dilaudid) — PO, SC, IV. Safer than morphine in renal failure. ~4× PO morphine potency PO, ~20× PO morphine IV.
- Fentanyl (Duragesic patch, Sublimaze IV, Actiq, Fentora, Subsys) — highly lipophilic, transdermal patch for stable chronic pain, rapid-onset transmucosal formulations for breakthrough. Patch reaches steady state in 12–24 hours; do not use for acute titration.
- Methadone (Dolophine) — NMDA antagonism gives it a role in neuropathic and opioid-tolerant pain. Long and variable half-life, QTc prolongation, numerous drug interactions, non-linear equianalgesic conversion. Only experienced clinicians should initiate.
- Buprenorphine (Butrans patch, Belbuca buccal film, Subutex, Sublocade) — partial mu agonist with ceiling on respiratory depression. Increasingly used for chronic pain and for patients with OUD history. Does not require opioid tapering before initiation with microdosing protocols.
- Tramadol (Ultram) — weak opioid + SNRI activity. Seizure risk, serotonin syndrome risk with SSRIs/SNRIs.
- Codeine — prodrug metabolized to morphine via CYP2D6; unreliable due to poor and ultra-rapid metabolizers. Largely displaced in modern palliative practice.
Equianalgesic Conversion Table
| Opioid | PO / PR dose | SC / IV dose | Ratio to PO morphine |
|---|---|---|---|
| Morphine | 30 mg | 10 mg | 1 (reference) |
| Oxycodone | 20 mg | — | 1.5× |
| Hydromorphone | 7.5 mg | 1.5 mg | 4× PO, 20× IV |
| Hydrocodone | 30 mg | — | 1× |
| Fentanyl transdermal | 25 mcg/hr patch ≈ 50–60 mg PO morphine/24h | — | |
| Codeine | 200 mg | — | 0.15× |
| Tramadol | 120 mg | — | 0.25× |
| Methadone | Variable; ratio 4:1 at low MEDD up to 20:1 at >1000 mg MEDD | Specialist only | |
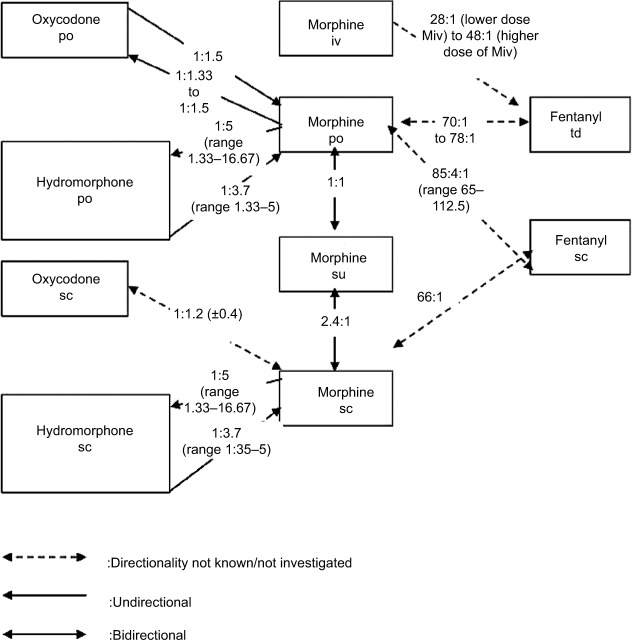
Opioid Rotation Principles
Opioid rotation is indicated for uncontrolled pain despite titration, intolerable side effects (neurotoxicity, myoclonus, hyperalgesia, severe constipation), or organ dysfunction. The standard approach: (1) calculate the total 24-hour morphine equivalent daily dose (MEDD) including scheduled and breakthrough doses, (2) convert to the new opioid using the equianalgesic table, (3) reduce by 25–50% for incomplete cross-tolerance, and (4) divide by the appropriate dosing interval. The NCCN Adult Cancer Pain Guidelines and ASCO chronic pain in cancer survivors guideline are the standard citations.
Opioid Side Effects
Every opioid order must be paired with a bowel regimen. Constipation is universal and does not develop tolerance. Other predictable side effects include nausea (usually improves within days), sedation (tolerance develops), pruritus (histamine release, worse with morphine), urinary retention, myoclonus (dose-related, suggests neurotoxicity), and respiratory depression (the only truly dangerous effect; naloxone-reversible). Suspect opioid-induced neurotoxicity (OIN) when a patient develops delirium, hyperalgesia, allodynia, or myoclonus on escalating opioid doses — the treatment is rotation, hydration, and dose reduction, not more opioid.
05 Adjuvant Analgesics & Neuropathic Pain medications
Adjuvants are non-opioid medications that enhance analgesia, particularly for neuropathic, bone, and visceral pain. They are the second pillar of cancer pain management.
| Class | Examples | Primary indication |
|---|---|---|
| Gabapentinoids | Gabapentin (Neurontin), pregabalin (Lyrica) | Neuropathic pain, CIPN, radiculopathy |
| TCAs | Nortriptyline, desipramine, amitriptyline | Neuropathic pain, insomnia |
| SNRIs | Duloxetine (Cymbalta), venlafaxine | CIPN, neuropathic pain, depression |
| Glucocorticoids | Dexamethasone | Bone pain, liver capsule pain, nerve compression, cord compression, ICP, anorexia |
| Bisphosphonates / RANK-L inhibitor | Zoledronic acid, pamidronate, denosumab | Bone metastasis pain, SRE prevention |
| NMDA antagonists | Ketamine (subanesthetic), methadone | Opioid-refractory, neuropathic, complex regional |
| Local anesthetics | Lidocaine 5% patch, IV lidocaine infusion, mexiletine | Post-herpetic neuralgia, neuropathic |
| Antispasmodics | Baclofen, tizanidine, cyclobenzaprine | Muscle spasm, MS-related pain |
| Topicals | Capsaicin, diclofenac gel, compounded creams | Localized neuropathic and musculoskeletal pain |
| Cannabinoids | Dronabinol, nabilone, state-legal THC/CBD products | Adjuvant for refractory pain, nausea, anorexia |
For established neuropathic pain, first-line agents are gabapentin, pregabalin, duloxetine, or a TCA — supported by the NeuPSIG pharmacotherapy recommendations. For bone metastases, dexamethasone plus radiation plus a bisphosphonate or denosumab is the standard combination. For malignant bowel obstruction pain, scheduled octreotide and scopolamine can reduce secretions and cramping.
06 Dyspnea & Respiratory Symptoms symptom
Dyspnea is the subjective sensation of breathlessness and is the second most common distressing symptom at the end of life. It is not synonymous with hypoxia — patients with normal oxygen saturation can be profoundly dyspneic, and patients with SpO2 of 85% can feel fine. The sensation of dyspnea originates from mismatch between respiratory drive and mechanical output, with major input from chemoreceptors, mechanoreceptors, and affective circuits.
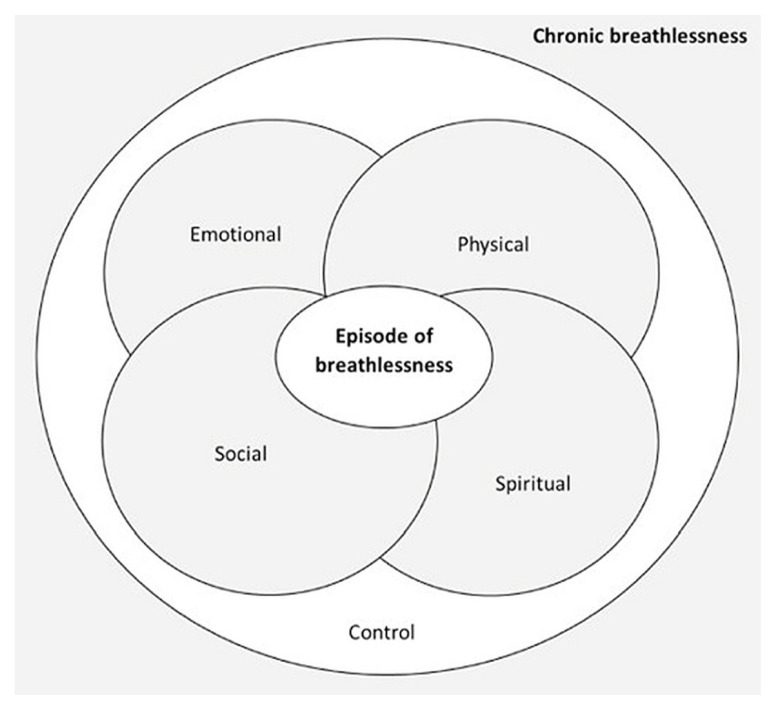
- Pleural effusion (consider thoracentesis or indwelling pleural catheter)
- Pulmonary embolism
- Pneumonia
- Heart failure exacerbation
- COPD exacerbation
- Airway obstruction (stent, radiation, debulking)
- Anemia (transfusion if goals-appropriate)
- Anxiety amplifying baseline dyspnea
Management
Nonpharmacologic measures are first-line and underused: a fan blowing air across the face reliably reduces the sensation of dyspnea via trigeminal V2 afferents, upright positioning, pursed-lip breathing, cool room temperature, energy conservation, and reassurance. Supplemental oxygen should be reserved for hypoxemic patients; in non-hypoxemic palliative patients, the Abernethy palliative oxygen trial showed that room air via nasal cannula was equivalent to oxygen.
Pharmacologically, low-dose opioids are the mainstay. Morphine 2.5–5 mg PO or 1–2 mg IV/SC q2–4h is typical for opioid-naive patients; opioid-tolerant patients need a dose titration of their existing regimen. Benzodiazepines (lorazepam 0.5–1 mg) are added for the anxiety component, not as primary dyspnea treatment. Nebulized bronchodilators, glucocorticoids (for airway edema or lymphangitic spread), and noninvasive ventilation may be considered depending on the goals-of-care trajectory.
07 Nausea, Vomiting & GI Symptoms symptom
Palliative nausea is treated by mechanism, not by trial-and-error. Identifying the likely dominant pathway directs you to the most effective antiemetic.
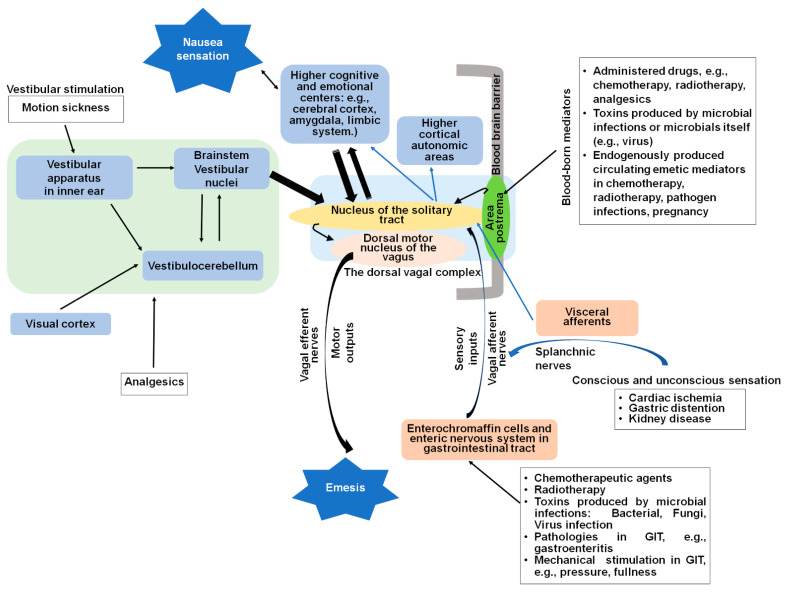
| Mechanism | Receptors | First-line agent(s) |
|---|---|---|
| Chemotherapy/opioid (CTZ) | D2, 5-HT3, NK1 | Haloperidol, prochlorperazine, ondansetron, aprepitant |
| Vestibular/motion | H1, ACh | Meclizine, scopolamine, dimenhydrinate |
| Gastric stasis | D2, 5-HT4 | Metoclopramide (avoid in complete obstruction) |
| Bowel obstruction | Multiple | Haloperidol + octreotide + dexamethasone + scopolamine |
| Raised ICP | Mass effect | Dexamethasone, mannitol |
| Anxiety | GABA | Lorazepam |
| Refractory / mixed | Multi-receptor | Olanzapine (broad receptor profile) |
Metoclopramide is a prokinetic — excellent for opioid-induced gastroparesis but contraindicated in complete mechanical bowel obstruction, where increased peristalsis causes pain and may precipitate perforation. Olanzapine 2.5–5 mg has become a favored agent for refractory nausea because it blocks D2, 5-HT2, 5-HT3, H1, and muscarinic receptors simultaneously and is supported by randomized data for chemotherapy-induced and advanced cancer nausea.
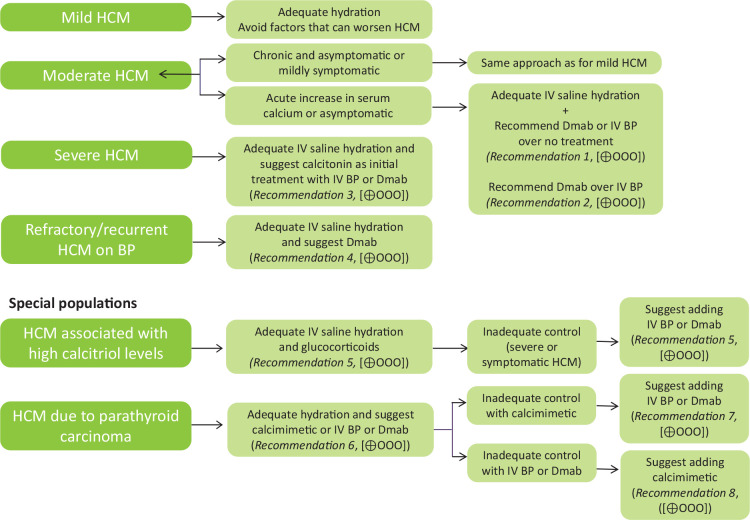
08 Constipation, Malignant Bowel Obstruction & Ascites symptom
Opioid-Induced Constipation
Every patient on opioids needs a scheduled bowel regimen from day one. The standard is a stimulant laxative (senna) plus an osmotic laxative (polyethylene glycol) titrated to a soft bowel movement every 1–2 days. Stool softeners alone (docusate) are ineffective for opioid-induced constipation and have been largely abandoned as monotherapy.
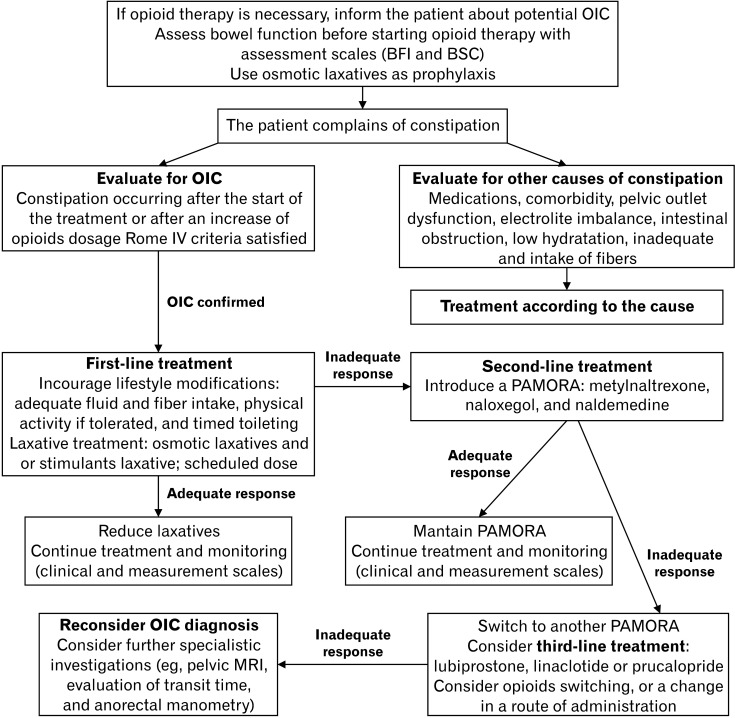
For refractory opioid-induced constipation despite a full bowel regimen, peripherally-acting mu-opioid receptor antagonists (PAMORAs) — methylnaltrexone (Relistor), naloxegol (Movantik), naldemedine (Symproic) — restore bowel function without reversing central analgesia.
Malignant Bowel Obstruction (MBO)
MBO is obstruction of the small or large bowel by intra-abdominal malignancy, most commonly ovarian, colorectal, gastric, or pancreatic cancer. Presentation is crampy abdominal pain, distension, nausea, vomiting (feculent if distal), and absent bowel movements. Management depends on surgical candidacy and goals of care. Nonsurgical management ("medical NG-free regimen") is the classic palliative approach: NPO or clear liquids, IV hydration as goals allow, and the triad of dexamethasone (reduces peritumoral edema), octreotide (reduces GI secretions), and haloperidol or scopolamine for symptoms. Venting PEG or nasogastric decompression is reserved for vomiting refractory to medical management.
Malignant Ascites
Malignant ascites causes abdominal distension, dyspnea, early satiety, and nausea. Therapeutic paracentesis provides rapid symptomatic relief. For recurrent ascites, a tunneled indwelling peritoneal catheter (PleurX) allows home drainage and avoids repeated hospital visits. Diuretics are largely ineffective for peritoneal carcinomatosis ascites (as opposed to cirrhotic ascites).
09 Anorexia, Cachexia & Fatigue symptom
Cancer cachexia is a multifactorial syndrome of ongoing loss of skeletal muscle mass (with or without fat loss) that cannot be fully reversed by conventional nutritional support and leads to progressive functional decline.
It is driven by proinflammatory cytokines (TNF, IL-1, IL-6), proteolysis-inducing factor, and altered hypothalamic appetite regulation — not simply by inadequate caloric intake. Artificial nutrition and hydration at end of life do not reverse cachexia and may increase symptom burden; this is a critical counseling point for families.
Pharmacologic appetite stimulation is modestly effective. Dexamethasone 2–4 mg daily improves appetite and sense of wellbeing for weeks at a time. Megestrol acetate (Megace) 160–800 mg/day promotes weight gain but carries VTE risk and adrenal suppression. Mirtazapine addresses appetite + sleep + mood simultaneously and is frequently chosen for multi-symptom patients. Olanzapine low-dose has emerging data for cancer cachexia.
Fatigue is the most common symptom in advanced cancer (80–90% prevalence). Reversible contributors should be addressed first: anemia, hypothyroidism, hypogonadism, depression, insomnia, uncontrolled pain, medication side effects, adrenal insufficiency. Nonpharmacologic management with energy conservation and graded exercise has the strongest evidence. Psychostimulants (methylphenidate 2.5–10 mg BID, modafinil) are used selectively for opioid-related sedation and fatigue, with mixed trial data.
10 Delirium, Anxiety & Depression symptom
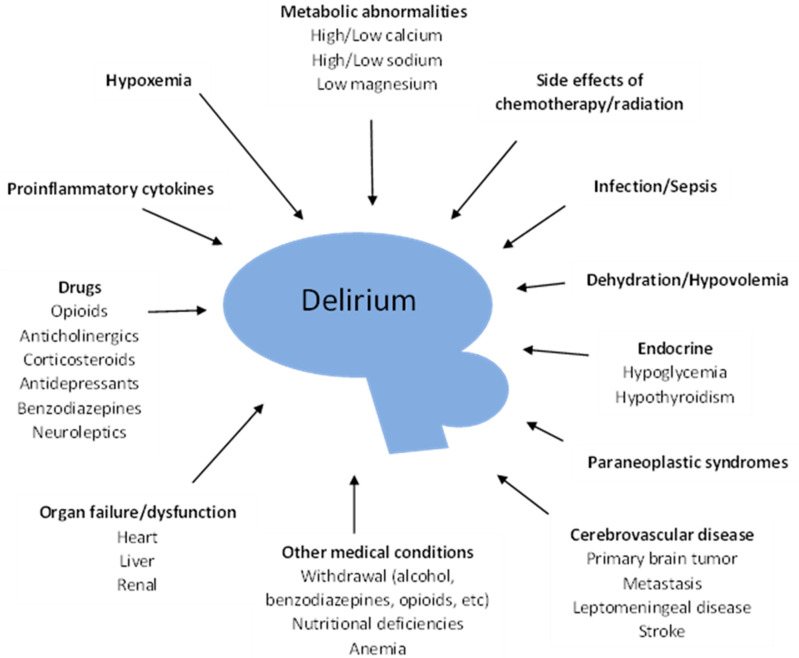
Delirium
Delirium occurs in up to 85% of patients in the final days of life (terminal delirium). It presents as acute onset of inattention, disorganized thinking, and altered level of consciousness, and can be hyperactive (agitation, restlessness, hallucinations), hypoactive (somnolence, withdrawal, often missed), or mixed. Reversible precipitants to screen for include medications (opioids, benzodiazepines, anticholinergics), infection, dehydration, hypercalcemia, hyponatremia, hypoxia, urinary retention, constipation, and pain.
Nonpharmacologic management is first-line: reorientation, familiar voices, minimizing room changes, promoting sleep-wake cycle, treating reversible causes. For pharmacologic control of distressing agitation, low-dose haloperidol (0.5–2 mg PO/SC/IV) is the classic agent, with olanzapine, quetiapine, or risperidone as alternatives. Benzodiazepines should generally be avoided because they worsen delirium, except in alcohol/benzodiazepine withdrawal and in refractory terminal agitation where sedation is the goal.
Anxiety & Depression
Anxiety and depression in advanced illness are treated similarly to primary psychiatric conditions but with attention to time horizon. SSRIs (citalopram, sertraline, escitalopram) and SNRIs (duloxetine, venlafaxine) take 4–6 weeks — appropriate for patients with months of prognosis, not days. Mirtazapine is preferred when sleep, appetite, and nausea coexist. Methylphenidate produces mood improvement within 24–48 hours and is an option for patients with short prognosis. For acute anxiety, lorazepam 0.25–1 mg PO/SL/IV provides rapid relief.
Always screen for suicidal ideation and for demoralization distinct from depression; referral to a palliative psychologist, psychiatrist, or chaplain is frequently appropriate. The NCCN Distress Thermometer is a validated one-question screen (0–10) with an accompanying problem checklist.
11 Terminal Phase: Secretions, Agitation, Palliative Sedation end of life
In the final hours to days of life, patients develop a predictable pattern of changes: decreased oral intake, decreased urine output, mottling of extremities, cool skin, Cheyne-Stokes respirations, altered consciousness, and pooled oral secretions producing the death rattle.
Respiratory Secretions ("Death Rattle")
The rattling sound is caused by air moving through secretions in the upper airway in a patient too weak to swallow or clear them. It is often more distressing to family than to the patient. Management: reposition the patient (lateral or semiprone), limit IV fluids, and use an antimuscarinic to dry secretions. The three common anticholinergics are:
- Glycopyrrolate 0.2 mg SC/IV q4h — does not cross BBB, least sedating.
- Scopolamine (hyoscine hydrobromide) 1.5 mg transdermal patch q72h or 0.4 mg SC — crosses BBB, can cause delirium.
- Hyoscyamine (Levsin) 0.125–0.25 mg SL q4h PRN.
- Atropine 1% ophthalmic drops given sublingually 1–2 drops q4h.
Terminal Agitation
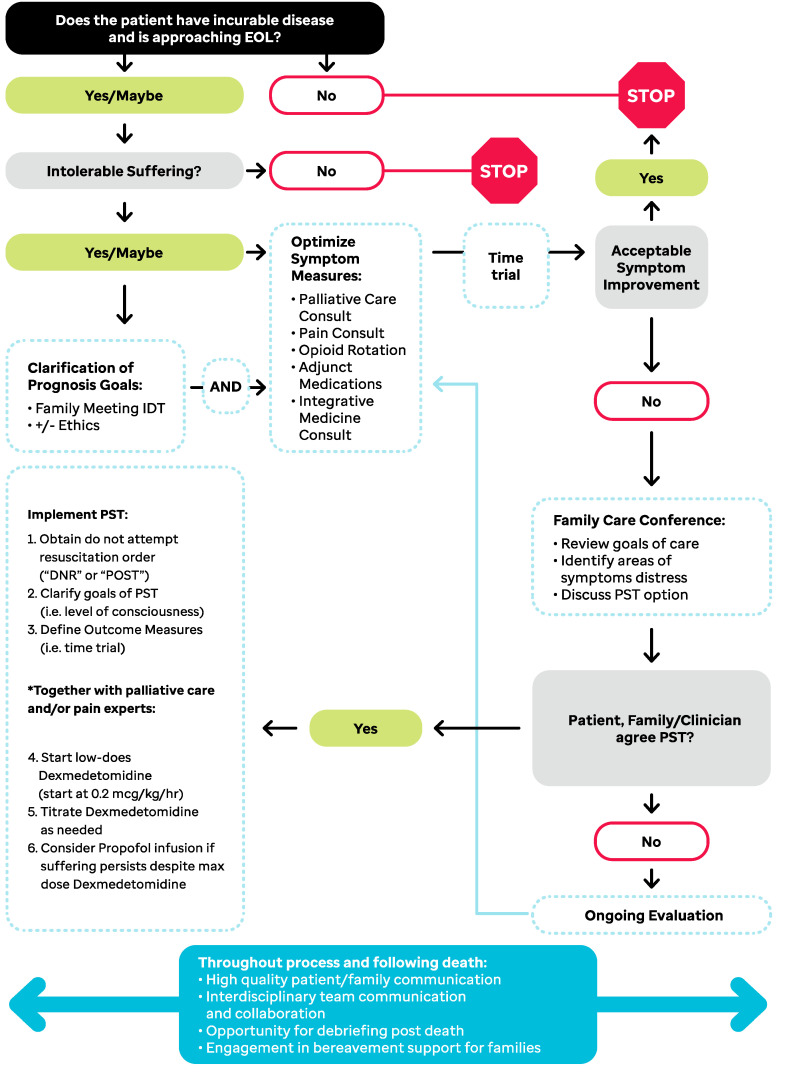
Restlessness and agitation at end of life that does not respond to treatment of reversible causes (pain, urinary retention, hypoxia, constipation, medications) and fails neuroleptic trials may require palliative sedation. This is the monitored use of sedating medication to reduce awareness of intolerable suffering that is refractory to standard palliative interventions. The intent is symptom relief, not hastening death — this distinction (proportionality and double effect) is core to the ethical framework and must be documented. Common agents are midazolam continuous infusion, phenobarbital, or propofol in select ICU settings. The EAPC framework for palliative sedation is the standard reference.
- Refractory symptom clearly defined (usually delirium, dyspnea, or pain)
- Standard interventions tried and failed
- Goals of care aligned with sedation (comfort only)
- Patient or surrogate informed consent, documented verbatim if possible
- Intent documented as symptom relief, not acceleration of death
- Level of sedation monitored (RASS scale or similar)
12 Other Symptoms: Pruritus, Hiccups, Mucositis, Wounds symptom
- Pruritus — opioid-induced (rotate, low-dose ondansetron, antihistamines), cholestatic (rifampin, ursodeoxycholic acid, naltrexone, sertraline), uremic (gabapentin, UVB), paraneoplastic.
- Hiccups (singultus) — first-line baclofen 5–10 mg TID or metoclopramide; second-line chlorpromazine (only FDA-approved agent), gabapentin, haloperidol.
- Mucositis — oral care, magic mouthwash (viscous lidocaine + diphenhydramine + antacid ± nystatin), systemic opioids for severe pain, nutritional support.
- Oral care — meticulous mouth care every 1–2 hours at end of life prevents thrush, mucosal breakdown, and discomfort. Ice chips, swabs, artificial saliva.
- Pressure injuries — at end of life, the goal is comfort and prevention of worsening rather than healing. Turning every 2–4 hours may be abandoned if repositioning causes distress (Kennedy terminal ulcer is expected at end of life).
- Malignant wounds — odor (metronidazole gel or PO), bleeding (tranexamic acid, Surgicel, epinephrine-soaked gauze), exudate (absorbent dressings, charcoal), pain (topical opioid gels).
- Lymphedema — complete decongestive therapy: manual lymphatic drainage, compression, skin care, exercise. Avoid BP cuffs and IVs in affected limb.
- Dehydration at end of life — parenteral hydration does not prolong life or reduce delirium in imminently dying patients and may worsen secretions, edema, and ascites. Discuss clearly with families.
- Urinary symptoms — retention (catheter), spasm (oxybutynin, B&O suppository), tenesmus (steroids, lidocaine).
- Fever — acetaminophen scheduled, cool cloths; antibiotics only if consistent with goals of care.
13 Prognostication Tools & Performance Scales assessment
Accurate prognostication is central to palliative decision-making. Clinicians are famously optimistic (by a factor of 3–5 in advanced cancer), so structured tools are used to anchor the conversation.
Karnofsky Performance Status (KPS)
| Score | Description |
|---|---|
| 100 | Normal; no complaints; no evidence of disease |
| 90 | Minor signs/symptoms; normal activity |
| 80 | Normal activity with effort |
| 70 | Cares for self; unable to carry on normal activity or work |
| 60 | Requires occasional assistance but cares for most needs |
| 50 | Requires considerable assistance and frequent medical care |
| 40 | Disabled; requires special care and assistance |
| 30 | Severely disabled; hospitalization indicated, death not imminent |
| 20 | Very sick; hospitalization and active supportive treatment necessary |
| 10 | Moribund; fatal processes progressing rapidly |
| 0 | Dead |
Palliative Performance Scale (PPS)
| PPS | Ambulation | Activity / disease | Self-care | Intake | Consciousness |
|---|---|---|---|---|---|
| 100% | Full | Normal, no disease | Full | Normal | Full |
| 90% | Full | Normal, some disease | Full | Normal | Full |
| 80% | Full | Normal with effort | Full | Normal/reduced | Full |
| 70% | Reduced | Unable to do normal work | Full | Normal/reduced | Full |
| 60% | Reduced | Unable to do hobbies/housework | Occasional assistance | Normal/reduced | Full or confusion |
| 50% | Mainly sit/lie | Unable to do any work | Considerable assistance | Normal/reduced | Full or confusion |
| 40% | Mainly in bed | Unable to do most activity | Mainly assistance | Normal/reduced | Full/drowsy/confusion |
| 30% | Totally bedbound | Unable to do any activity | Total care | Reduced | Full/drowsy/confusion |
| 20% | Totally bedbound | Unable, extensive disease | Total care | Minimal sips | Full/drowsy/confusion |
| 10% | Totally bedbound | Unable, extensive disease | Total care | Mouth care only | Drowsy/coma |
| 0% | Death | — | — | — | — |
Other Prognostic Tools
- ECOG (Zubrod) — 0 fully active; 1 restricted strenuous activity; 2 ambulatory >50% of day, cannot work; 3 in bed/chair >50% of day; 4 completely disabled, bedbound; 5 dead.
- Palliative Prognostic Score (PaP) — weighted score using clinician prediction, KPS, anorexia, dyspnea, WBC, and lymphocyte %. Stratifies 30-day survival into three groups.
- Palliative Prognostic Index (PPI) — uses PPS, oral intake, edema, dyspnea at rest, and delirium to predict survival of <3 weeks or <6 weeks.
- PRISM (Prospective Risk Index) — predicts hospital mortality in advanced cancer admissions.
- Surprise Question — "Would you be surprised if this patient died in the next year?" A no answer has strong validity for triggering palliative/hospice referral.
14 Goals of Care, SPIKES & Family Meetings communication
A goals-of-care (GOC) discussion aligns medical care with what matters most to the patient. It is a procedure as much as a chest tube is, with preparation, consent, technique, and documentation.

SPIKES Protocol for Breaking Bad News
- S — Setting: private, seated, tissues available, key family present, interpreter if needed
- P — Perception: "What have the other doctors told you?"
- I — Invitation: "How much do you want to know about what is going on?"
- K — Knowledge: warning shot ("I'm afraid I have difficult news"), then clear information in plain language, small chunks
- E — Emotions: respond to emotion with empathic statements (NURSE: name, understand, respect, support, explore)
- S — Strategy & Summary: next steps, who is doing what, when you will meet again
The Family Meeting
A well-run family meeting follows a predictable flow: introduction and naming of participants; establishing the patient's medical situation and prognosis in shared language; exploring the patient's values and prior expressed wishes; making recommendations aligned with those values; and closing with concrete plans. Recommendations should be framed, not menu-listed ("Based on everything you have told me, I would recommend we focus on comfort and transition to hospice"). The VitalTalk communication framework and Serious Illness Conversation Guide (Ariadne Labs) are the standard references.
- Participants: names/relationships of all attendees, including medical team members
- Purpose: why the meeting was called (e.g., new diagnosis of metastatic disease, transition of care, code status clarification)
- Patient/family understanding of illness (direct quotes preferred)
- Information shared by team: diagnosis, prognosis, treatment options presented
- Values and goals elicited: what the patient values, fears, and hopes for
- Decisions reached (code status, hospice election, specific treatments accepted/declined)
- Unresolved questions and follow-up plan
15 Code Status, Advance Directives, POLST/MOLST legal
Code Status Terminology
- Full code — all resuscitative efforts including CPR, intubation, defibrillation, vasopressors
- DNR / DNAR / AND — do not resuscitate / do not attempt resuscitation / allow natural death; no CPR, no defibrillation
- DNI — do not intubate; excludes endotracheal intubation but may still include NIV, CPR chest compressions, etc.
- DNR/DNI — the most common combined order
- Comfort care / comfort measures only (CMO) — focus exclusively on comfort; discontinue disease-directed interventions
- Limited trial of therapy — time-limited escalation with predefined goals (e.g., "72-hour trial of pressors, reassess")
Advance Directives vs. POLST/MOLST
An advance directive (living will + durable power of attorney for healthcare) is a legal document expressing the patient's wishes for future care if they become unable to speak for themselves. It is patient-signed, not a medical order, and does not translate directly into an actionable order set at the bedside.
A POLST (Physician Orders for Life-Sustaining Treatment, also called MOLST, POST, or MOST depending on state) is a portable medical order signed by a clinician that follows the patient across care settings. It translates the goals in an advance directive into actionable orders covering CPR, medical interventions, and artificial nutrition. POLST is intended for patients with serious illness or frailty — not the general population. See the National POLST program for state-specific forms.
16 Capacity, Surrogates & Ethics legal
Decisional Capacity
Capacity is a clinical determination (anyone can make it) distinct from competency (a legal determination by a court). Capacity is decision-specific and time-specific. The four-part Appelbaum-Grisso standard asks whether the patient can: (1) understand the relevant information, (2) appreciate how it applies to their situation, (3) reason through the options, and (4) communicate a consistent choice. A patient may lack capacity to refuse dialysis while retaining capacity to name a surrogate.
Surrogate Decision-Making Hierarchy
When a patient lacks capacity, the decision-maker is determined by the state surrogate statute. A typical hierarchy: (1) court-appointed guardian, (2) durable power of attorney for healthcare, (3) spouse, (4) adult child, (5) parent, (6) sibling, (7) other relatives, (8) close friend, (9) attending physician as last resort with ethics consultation. The surrogate uses substituted judgment ("what would the patient want?") first, and best interest only when prior wishes are unknown.
Withholding vs. Withdrawing Treatment
Ethically and legally, withholding a treatment and withdrawing a treatment are equivalent. The decision to discontinue mechanical ventilation, dialysis, artificial nutrition, or vasopressors is not euthanasia; it is a withdrawal of an unwanted or non-beneficial intervention allowing the natural course of disease to proceed. The principle of double effect supports using medications (opioids, benzodiazepines) to treat symptoms during withdrawal even when they may shorten life, provided the intent is symptom relief and the dose is proportionate. Ethics consultation is appropriate for persistent disagreement, concerns about surrogate motivation, or medically inappropriate (futile) treatment requests.
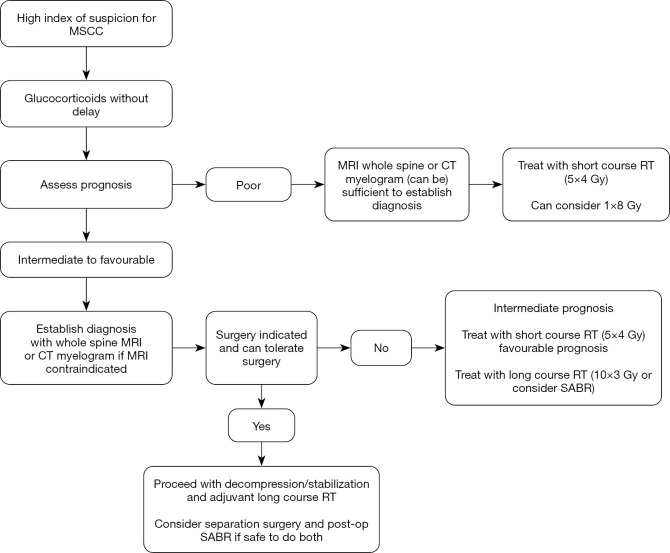
17 Hospice Eligibility & Medicare Criteria hospice
The Medicare Hospice Benefit requires two physician certifications (the hospice medical director and the attending) that the patient has a prognosis of six months or less if the disease runs its normal course. Patients elect hospice and waive Medicare coverage for curative treatment of the terminal diagnosis (not for unrelated problems). Benefit periods are two 90-day periods followed by unlimited 60-day periods, each requiring recertification and a face-to-face visit after the third benefit period per CMS hospice regulations.
Levels of Hospice Care
- Routine home care — the default; care delivered in home or SNF
- Continuous home care — 8–24 hours/day of nursing during a crisis (severe pain, dyspnea, agitation)
- General inpatient (GIP) — inpatient hospice for symptom management that cannot be provided at home
- Respite care — up to 5 consecutive days of inpatient care to give family caregivers a break
Disease-Specific Hospice Eligibility Highlights
- Cancer: metastatic or unresectable disease, declining PPS ≤70%, ECOG ≥3, refusing or not candidate for further disease-directed therapy
- Heart failure: NYHA IV despite optimal therapy, EF ≤20%, symptomatic arrhythmias, cardiac cachexia, recurrent admissions
- COPD: dyspnea at rest, FEV1 <30% predicted, resting hypoxemia, cor pulmonale, unintentional weight loss, tachycardia at rest
- Dementia: FAST stage 7C or greater (unable to ambulate, dress, bathe, incontinent, speech ≤6 intelligible words) plus a recent complication (aspiration pneumonia, stage 3–4 pressure ulcer, recurrent UTI, sepsis, weight loss >10%)
- Liver disease: INR >1.5, albumin <2.5, plus refractory ascites, SBP, HRS, HE, or variceal bleed
- Renal failure: not pursuing dialysis, CrCl <10 (<15 in diabetics), symptomatic uremia
- Stroke/coma: PPS ≤40%, poor nutritional intake, aspiration pneumonia, pressure ulcers, recurrent infection
- ALS: declining FVC, declining functional status, dysphagia with weight loss, refusal of PEG or ventilation
18 Palliative Procedures & PCA Management procedures
| Procedure | Indication | Key documentation |
|---|---|---|
| Therapeutic paracentesis | Symptomatic ascites | Volume removed, relief, albumin replacement if applicable |
| Indwelling peritoneal (PleurX) | Recurrent malignant ascites | Home drainage teaching, dose/schedule |
| Thoracentesis | Symptomatic pleural effusion | Volume, cytology, relief |
| Indwelling pleural catheter | Recurrent MPE | Drainage schedule, pleurodesis attempts |
| Venting gastrostomy | Refractory MBO with vomiting | Output, pain relief |
| PCA initiation | Severe pain | Basal, bolus, lockout, 1-hr limit, PCA tries vs given |
| SC opioid infusion | No IV access at end of life | Site, rate, breakthrough doses |
| Nerve block / celiac plexus | Pancreatic/upper abdominal pain | IR or anesthesia referral |
| Intrathecal pump | Refractory cancer pain | Medication, rate, dose titration |
| Palliative radiation | Bone mets, SVC, cord compression, bleeding | Fractionation (e.g., 8 Gy × 1, 20 Gy in 5 fx) |
PCA Management
A patient-controlled analgesia (PCA) pump delivers an opioid via IV or SC route with three programmable settings the scribe must chart at every encounter:
- Continuous (basal) rate — e.g., morphine 1 mg/hr; provides background analgesia
- Demand (bolus) dose — e.g., morphine 1 mg; delivered when the patient presses the button
- Lockout interval — e.g., 10 minutes; minimum time between successful boluses
- 1-hour or 4-hour dose limit — safety ceiling
- PCA tries vs attempts — ratio of button presses to delivered doses; high ratio of tries to given doses suggests inadequate analgesia
Continuous SC infusion is the preferred route at end of life when the patient has lost IV access and cannot swallow. Typical sites are anterior chest, abdomen, or thigh; needle is rotated every 3–7 days. Multiple medications (morphine + haloperidol + midazolam, or "bag of kindness") can be combined in a single syringe driver per compatibility tables.
19 Medications You Must Know medications
| Drug | Brand | Class | Primary use in palliative care |
|---|---|---|---|
| Morphine | MSIR, MS Contin, Roxanol | Strong opioid | Pain, dyspnea |
| Oxycodone | Roxicodone, OxyContin | Strong opioid | Pain |
| Hydromorphone | Dilaudid | Strong opioid | Pain in renal failure |
| Fentanyl patch | Duragesic | Strong opioid | Stable chronic pain, PO route unavailable |
| Methadone | Dolophine | Mu + NMDA antagonist | Neuropathic pain, rotation |
| Buprenorphine | Butrans, Belbuca | Partial mu agonist | Chronic pain, OUD history |
| Tramadol | Ultram | Weak opioid + SNRI | Mild-moderate pain |
| Naloxone | Narcan | Opioid antagonist | Respiratory depression reversal |
| Methylnaltrexone | Relistor | PAMORA | Opioid-induced constipation |
| Gabapentin | Neurontin | Gabapentinoid | Neuropathic pain, hiccups, pruritus |
| Pregabalin | Lyrica | Gabapentinoid | Neuropathic pain |
| Duloxetine | Cymbalta | SNRI | CIPN, neuropathic pain, depression |
| Nortriptyline | Pamelor | TCA | Neuropathic pain, insomnia |
| Ketamine | Ketalar | NMDA antagonist | Refractory pain |
| Lidocaine patch | Lidoderm | Local anesthetic | Focal neuropathic pain |
| Dexamethasone | Decadron | Glucocorticoid | Pain, nausea, appetite, cord compression, ICP |
| Megestrol | Megace | Progestin | Appetite stimulation |
| Mirtazapine | Remeron | NaSSA | Appetite, sleep, depression, nausea |
| Methylphenidate | Ritalin | Stimulant | Fatigue, opioid sedation, depression (rapid onset) |
| Haloperidol | Haldol | Butyrophenone | Delirium, nausea, agitation |
| Olanzapine | Zyprexa | Atypical antipsychotic | Refractory nausea, delirium, anxiety |
| Metoclopramide | Reglan | Prokinetic | Gastroparesis, CTZ nausea |
| Ondansetron | Zofran | 5-HT3 antagonist | Chemo nausea, opioid nausea |
| Prochlorperazine | Compazine | Phenothiazine | Nausea |
| Promethazine | Phenergan | Phenothiazine + H1 | Nausea |
| Lorazepam | Ativan | Benzodiazepine | Anxiety, dyspnea anxiety, seizures, sedation |
| Midazolam | Versed | Benzodiazepine | Palliative sedation, terminal agitation |
| Phenobarbital | Luminal | Barbiturate | Refractory terminal agitation/sedation |
| Scopolamine patch | Transderm Scop | Anticholinergic | Secretions, nausea |
| Glycopyrrolate | Robinul | Anticholinergic | Secretions (does not cross BBB) |
| Hyoscyamine | Levsin | Anticholinergic | Secretions, visceral cramps |
| Atropine drops | — | Anticholinergic | Sublingual secretion control |
| Octreotide | Sandostatin | Somatostatin analog | MBO secretions, diarrhea, variceal bleed |
| Senna / PEG | Senokot / MiraLAX | Laxatives | Opioid-induced constipation |
| Zoledronic acid | Zometa | Bisphosphonate | Bone pain, hypercalcemia, SRE prevention |
| Denosumab | Xgeva | RANK-L inhibitor | Bone mets, SRE prevention |
| Tranexamic acid | Lysteda | Antifibrinolytic | Malignant wound bleeding, hemoptysis |
20 Classification & Assessment Systems reference
Edmonton Symptom Assessment System (ESAS-r)
The ESAS-r is a nine-symptom self-report tool, each scored 0–10, collected at every palliative visit:
- Pain (no pain → worst possible pain)
- Tiredness (no tiredness → worst possible)
- Drowsiness
- Nausea
- Lack of appetite
- Shortness of breath
- Depression
- Anxiety
- Wellbeing (best → worst)
- "Other problem" (free text)
FAST Scale for Dementia (Functional Assessment Staging)
| Stage | Clinical findings |
|---|---|
| 1 | Normal adult |
| 2 | Subjective memory deficit (normal aging) |
| 3 | Decreased job functioning, early dementia |
| 4 | Difficulty with complex tasks (finances, travel) |
| 5 | Requires assistance with choosing clothes |
| 6a–e | Needs help dressing, bathing, toileting; urinary incontinence; fecal incontinence |
| 7a | Speech limited to ≤6 intelligible words/day |
| 7b | Speech limited to single intelligible word |
| 7c | Nonambulatory |
| 7d | Unable to sit up independently |
| 7e | Unable to smile |
| 7f | Unable to hold head up |
Hospice eligibility for dementia generally requires FAST 7C or greater plus a qualifying complication.
NYHA Heart Failure Class
- I — no limitation of ordinary activity
- II — slight limitation; comfortable at rest, ordinary activity causes symptoms
- III — marked limitation; less than ordinary activity causes symptoms
- IV — symptoms at rest; any activity worsens discomfort
RASS (Richmond Agitation-Sedation Scale)
| Score | Term | Description |
|---|---|---|
| +4 | Combative | Violent, immediate danger to staff |
| +3 | Very agitated | Pulls at tubes/catheters, aggressive |
| +2 | Agitated | Frequent nonpurposeful movement |
| +1 | Restless | Anxious but not aggressive |
| 0 | Alert and calm | — |
| -1 | Drowsy | Not fully alert, sustained awakening to voice |
| -2 | Light sedation | Briefly awakens to voice |
| -3 | Moderate sedation | Movement to voice but no eye contact |
| -4 | Deep sedation | No response to voice, movement to physical stimulation |
| -5 | Unarousable | No response to voice or physical stimulation |
21 Abbreviations Master List reference
22 Sample HPI Templates templates
HPI 1 — Inpatient Pain Crisis Consult
Mr. L is a 64-year-old man with metastatic pancreatic adenocarcinoma (diagnosed 11 months ago, s/p FOLFIRINOX x 8, now on second-line gemcitabine/nab-paclitaxel), ECOG 3, PPS 40%, admitted yesterday from home with a 3-day history of escalating mid-epigastric and mid-back pain described as "deep, boring, constant, 9/10," radiating to bilateral flanks, worsened by lying supine and by meals, partially relieved only briefly by his home oxycodone. His outpatient regimen is MS Contin 60 mg PO BID with oxycodone 15 mg PO q3h PRN for breakthrough; in the past 24 hours he has used 12 breakthrough doses with only transient relief. He endorses associated nausea without emesis, decreased appetite, constipation (last BM 4 days ago), fatigue, poor sleep from pain, and low mood. Denies fever, jaundice change, hematemesis, melena, or focal weakness. ESAS today: pain 9, tired 8, drowsy 4, nausea 5, appetite 2, SOB 3, depression 6, anxiety 5, wellbeing 8. He is accompanied by his wife (surrogate), is a full code at present, and has an advance directive naming his wife as DPOA-HC. Palliative consult was placed by oncology for pain crisis management and initial goals-of-care discussion.
HPI 2 — Goals of Care / Family Meeting Consult
Mrs. R is an 82-year-old woman with advanced Alzheimer's dementia (FAST 7C, nonambulatory, minimal verbal output, requires total care), hypertension, atrial fibrillation on apixaban, and CKD stage 4, admitted from her long-term care facility with her fourth aspiration pneumonia in the past 6 months. She is currently on IV ceftriaxone and azithromycin, requires 3 L NC for SpO2 >92%, and is intermittently agitated. She has lost 18 lb (11% body weight) over the past 3 months. Family meeting requested by the hospitalist team to discuss prognosis and goals of care. Present: patient (nonverbal), daughter (DPOA-HC), son, hospitalist, palliative physician, bedside RN, chaplain. Prior advance directive from 2019 expresses a wish to "not be on machines" and to "die peacefully at home." The team shared that the trajectory is of progressive decline with recurrent aspiration events, that each pneumonia carries increasing mortality, and that the underlying dementia is terminal and incurable. Daughter expressed her mother's long-standing value of independence and the family's prior discussions that she would not want a feeding tube. After discussion, the family agreed to transition to comfort-focused care with home hospice, to de-escalate IV antibiotics after current course, to make the patient DNR/DNI, and to discontinue apixaban given bleeding risk without meaningful benefit at this stage. Hospice intake initiated today; anticipated discharge tomorrow.
HPI 3 — Dyspnea in Advanced Heart Failure
Mr. T is a 71-year-old man with NYHA IV nonischemic cardiomyopathy (EF 15%), s/p CRT-D, chronic kidney disease stage 4, and anemia of chronic disease, with his sixth admission in 12 months for acute decompensation and progressive dyspnea at rest. On maximally tolerated GDMT (carvedilol, sacubitril-valsartan at reduced dose limited by SBP and Cr, spironolactone held for hyperkalemia, torsemide 80 mg BID) with ongoing congestion despite escalation. Cardiology has advised that he is not a transplant or LVAD candidate due to age, frailty, and renal dysfunction. Palliative consulted for dyspnea management and GOC. He reports breathlessness with minimal exertion (walking from bed to bathroom), orthopnea requiring 3 pillows, and a sensation of "suffocation" at night that wakes him from sleep. ESAS: pain 2, tired 9, drowsy 6, nausea 1, appetite 3, SOB 9, depression 7, anxiety 8, wellbeing 9. He lives with his wife, has adult children nearby, and is a full code. He states, "I am tired of coming to the hospital. I want to be home." Plan to initiate low-dose morphine 2 mg PO q4h scheduled and 2 mg q2h PRN for dyspnea, lorazepam 0.5 mg PO q6h PRN for anxiety, and fan therapy; convene family meeting tomorrow for hospice discussion.
HPI 4 — Terminal Agitation / Imminently Dying
Mrs. K is an 88-year-old woman on inpatient hospice (GIP level) for end-stage metastatic breast cancer to bone, liver, and brain, day 3 of admission, PPS 10%, RASS +2, with increasing restlessness over the past 12 hours despite scheduled morphine 4 mg SC q4h and haloperidol 1 mg SC q6h. She is picking at bedsheets, calling out intermittently, and grimacing with turning. She is no longer taking oral intake, is producing minimal urine, and has mottling to the mid-thighs. Family at bedside (two daughters, one son). Evaluated for reversible precipitants: no urinary retention (Foley in place with minimal output), no palpable fecal impaction, no suggestion of infection, no new medications. Discussed with daughters the transition to a SC midazolam infusion starting at 0.5 mg/hr with 1 mg q30min PRN for breakthrough agitation, and increased morphine to 6 mg q4h, framed as proportional sedation to relieve refractory terminal agitation with the goal of comfort, not hastening death. Daughters verbalized understanding and agreement. Chaplain at bedside. Prognosis: hours to short days. Family updated, code status remains DNR/DNI/CMO.
HPI 5 — Outpatient New Palliative Consult
Ms. J is a 57-year-old woman with metastatic non-small cell lung cancer (EGFR wild-type, KRAS G12C mutated, on sotorasib second line after progression on pembrolizumab-carboplatin-pemetrexed), ECOG 2, PPS 70%, referred by medical oncology to outpatient palliative for symptom management and advance care planning. She reports right-sided chest wall pain from a rib metastasis (6/10, sharp with deep breathing, constant dull ache at rest 4/10), chemotherapy-induced peripheral neuropathy in feet (burning, 5/10), fatigue, anxiety about scan results, and insomnia. Current regimen: acetaminophen 650 mg TID, tramadol 50 mg TID, gabapentin 300 mg TID. No breakthrough opioid. ESAS: pain 6, tired 7, drowsy 3, nausea 2, appetite 5, SOB 3, depression 5, anxiety 8, wellbeing 6. Lives with her husband and 16-year-old daughter, works part-time as a teacher. Has not completed an advance directive. Understands her cancer is incurable but hopes for "meaningful time." Plan: rotate tramadol to oxycodone 5 mg q4h PRN, start oxycodone ER 10 mg q12h after breakthrough use established; increase gabapentin to 600 mg TID; refer to palliative RT for the rib lesion; initiate advance care planning conversation over several visits; referral to social work for family support.
HPI 6 — Hospice Transition Consult
Mr. B is a 74-year-old man with ALS diagnosed 3 years ago, now with severe bulbar involvement and respiratory insufficiency (FVC 28% predicted), declining PEG and tracheostomy, admitted to the hospital after a choking episode at home. He is using BiPAP 16/8 with backup rate for comfort, requires full assistance for all ADLs, and has lost 22 lb over the past 4 months. ESAS today: pain 3 (cramping), tired 9, drowsy 5, nausea 2, appetite 2, SOB 7, depression 4, anxiety 6, wellbeing 8. He communicates with an eye-gaze device and clearly states he wants to "go home and stay home." Prior advance directive rejects intubation, tracheostomy, and artificial nutrition. Wife at bedside, supportive of his wishes, is his DPOA-HC. Palliative consult for hospice transition. He meets hospice eligibility for ALS with declining FVC, declining functional status, dysphagia with weight loss, and refusal of invasive interventions. Discussed home hospice with continued BiPAP use for comfort, morphine for dyspnea, glycopyrrolate for secretions, lorazepam for anxiety, and bowel regimen. Family verbalized understanding of prognosis (days to short weeks). Hospice referral placed; discharge planned tomorrow.
23 References & Sources
Clinical Practice Guidelines
NCCN Clinical Practice Guidelines in Oncology: Adult Cancer Pain.
NCCN Clinical Practice Guidelines in Oncology: Palliative Care.
NCCN Guidelines for Distress Management (Distress Thermometer).
NICE NG31. Care of dying adults in the last days of life. 2015.
NICE NG142. End of life care for adults: service delivery.
Landmark Trials & Evidence
Temel JS et al. Early Palliative Care for Patients with Metastatic NSCLC. NEJM. 2010.
Thomas J et al. Methylnaltrexone for opioid-induced constipation. NEJM. 2008.
Navari RM et al. Olanzapine for chronic nausea in advanced cancer. JAMA Oncol. 2019.
Back AL et al. VitalTalk communication framework.
Baile WF et al. SPIKES — A Six-Step Protocol for Delivering Bad News. Oncologist. 2000.
Diagram & Figure Sources
Figure 1: Hospice care bedside. Wikimedia Commons. Public domain.
Figure 2: Wong-Baker FACES Pain Rating Scale. Wikimedia Commons.
24 A Final Word
Palliative care scribing is different from any other specialty. Your notes are not just billing documents — they become part of the permanent record of how a patient's last months, weeks, or hours were managed, how decisions were made, and how families were supported. A well-documented goals-of-care conversation prevents unwanted ICU admissions. A clearly charted pain regimen prevents a crisis call at 3 a.m. An accurately quoted family wish protects the patient's autonomy long after they can no longer speak.
Be precise with numbers (opioid doses, ESAS scores, intervals), direct with quotations, and respectful in tone. When a patient says "I am ready," write those exact words. When a daughter says "we don't want her to suffer," write those exact words. The attending can make the medical decisions; your job is to make sure the record tells the truth of what happened in the room. Do that well and you will be invaluable to every team you work with.