Pediatric Surgery
Every diagnosis, congenital anomaly, procedure, medication, abbreviation, and documentation framework you need to succeed on day one in a pediatric surgical practice.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 Pediatric Surgery — Scope & Age-Based Essentials
Pediatric surgery is the surgical care of patients from the moment of birth (and sometimes before, in fetal surgery centers) through late adolescence. Unlike adult general surgery, where the patient population is relatively homogeneous physiologically, a pediatric surgeon in a single clinic day may care for a 900-gram premature neonate with necrotizing enterocolitis, a 6-week-old with projectile vomiting from pyloric stenosis, a 3-year-old with an inguinal hernia, a 10-year-old with appendicitis, and a 16-year-old with a pilonidal cyst. Each age group has different physiology, different normal values, different dosing, and different disease susceptibility.
Developmental Stages You Must Know
The language of pediatric surgery is age-based. You will hear and must chart these terms precisely:
Neonate: 0–28 days of life. Further divided into preterm (< 37 weeks gestational age), term (37–42 weeks), and post-term (> 42 weeks). Extremely low birth weight (ELBW) is < 1000 g, very low birth weight (VLBW) is < 1500 g, low birth weight (LBW) is < 2500 g. Corrected gestational age (CGA) is chronological age minus weeks of prematurity and is used to guide surgical timing in ex-preemies.
Infant: 1–12 months.
Toddler: 1–3 years.
Child: 3–12 years.
Adolescent: 12–18 (or 21) years. Older adolescents are managed on adult-style protocols with pediatric weight-based dosing.
Physiologic Pearls
Neonates and infants have larger body surface area relative to mass, immature thermoregulation, higher insensible fluid losses, lower glycogen stores (prone to hypoglycemia), and immature kidneys that concentrate urine poorly. Their tidal volumes are small and airways narrow, making them sensitive to even small amounts of airway edema. Blood volume is approximately 80–90 mL/kg in term neonates and up to 100 mL/kg in preterms — which is why what looks like a small amount of blood on a surgical field can be a meaningful percentage of circulating volume. Neonatal hemoglobin starts near 17 g/dL and physiologically nadirs around 9–11 g/dL by 8–12 weeks of life.
Fluids, Dosing & The 4-2-1 Rule
Pediatric maintenance fluids are calculated by the Holliday-Segar (4-2-1) rule: 4 mL/kg/hr for the first 10 kg, plus 2 mL/kg/hr for the next 10 kg, plus 1 mL/kg/hr for each additional kg. A 25 kg child therefore receives (10×4) + (10×2) + (5×1) = 65 mL/hr. Every medication is weight-based; nothing is ordered in adult-fixed doses.
Normal Pediatric Vital Signs
| Age | HR (bpm) | RR (/min) | SBP (mmHg) |
|---|---|---|---|
| Neonate (< 1 mo) | 100–180 | 30–60 | 60–90 |
| Infant (1–12 mo) | 100–160 | 25–50 | 70–100 |
| Toddler (1–3 yr) | 90–150 | 20–35 | 80–110 |
| Preschool (3–6 yr) | 80–130 | 20–30 | 85–110 |
| School (6–12 yr) | 70–120 | 16–25 | 90–120 |
| Adolescent (> 12) | 60–100 | 12–20 | 100–130 |
Always capture weight in kilograms, gestational age (for any neonate or ex-preemie), chronologic age in days (neonates) or months (infants), and corrected gestational age when the patient is still within the first year of life. These four numbers drive almost every downstream dosing and disposition decision. Never chart pounds — always kg.
02 Scribe Documentation Framework
Pediatric surgical notes are SOAP-structured but with several specialty-specific twists: the historian is almost never the patient, the birth history is part of every neonatal note, growth percentiles matter, and the physical exam language is developmentally staged.
Chief Complaint (CC): A one-line reason for the encounter framed in the caregiver's language when possible ("projectile vomiting for 1 week," "groin bulge noted at bath time," "referred for undescended left testis").
Historian: Always document who is giving the history (mother, father, foster parent, older sibling, EMS) and their reliability. In NAT screening this line becomes forensically important.
HPI: OLDCARTS adapted for peds — onset relative to birth or feeds, number of wet diapers, number and character of stools, emesis description (bilious vs non-bilious, projectile vs effortless, volume), oral intake trend, activity level, fever curve. Bilious emesis in any neonate is surgical until proven otherwise.
Birth History: Gestational age, birth weight, mode of delivery, APGAR scores, NICU course, prenatal ultrasound findings (polyhydramnios? known anomalies?), maternal infections, GBS status.
Feeding History: Breast vs formula, formula type, volume per feed, frequency, weight gain trajectory.
Development: Gross motor, fine motor, language, social milestones relative to age.
Immunizations: Up to date per AAP schedule? Last TdaP?
PMHx / PSHx / Meds / Allergies / FHx / SHx: Family history of Hirschsprung, CF, MEN, neurofibromatosis, familial polyposis, Beckwith-Wiedemann, cancer predisposition syndromes. Social history includes caregivers in the home, daycare, secondhand smoke exposure, and any DCFS/CPS history if NAT is on the differential.
Vitals: Pediatric vitals are age-specific. Tachycardia and tachypnea are the earliest signs of shock in children — hypotension is a late, ominous finding. Always document weight (kg), height/length, head circumference (under 2), and growth percentiles.
Exam: Tailored to age and cooperation — a screaming toddler's abdominal exam is limited; a sleeping infant is a gift. Document general appearance (well vs toxic, alert vs lethargic), hydration status (mucous membranes, capillary refill, fontanelle), respiratory effort, abdominal exam (distension, tenderness, bowel sounds, masses, hernias, scars), genitourinary exam (descended testes, hernia, hydrocele), back (sacral dimples, hair tufts), and skin (bruises — document pattern and location precisely when NAT is on the differential).
Results: CBC with differential, CMP/BMP, coagulation, blood gas (especially in neonates — capillary, arterial, or venous), lactate, CRP, blood culture, imaging findings. Always document specific values.
Each problem is listed with the plan immediately below. For surgical consults, end with disposition (admit, observe, operate, discharge), NPO status, IV fluid order, antibiotic choice, and who was contacted (NICU, PICU, anesthesia, social work). Consent conversations with parents/guardians must be documented including risks, benefits, alternatives, and that the parent verbalized understanding.
When a surgeon says "bilious emesis in a neonate," flag it in your head as a surgical emergency — malrotation with midgut volvulus until proven otherwise. The note should reflect that urgency: you are ordering STAT upper GI, calling NICU, starting IV fluids, making the patient NPO, and preparing an OR. Documentation speed matters here.
03 Congenital Diaphragmatic Hernia (CDH) Thoracic
CDH is a developmental failure of the diaphragm to close, most commonly at the posterolateral foramen of Bochdalek on the left (80–85%). Abdominal viscera herniate into the chest during fetal life, impairing lung development on the affected side and producing pulmonary hypoplasia and persistent pulmonary hypertension of the newborn (PPHN). A far smaller subset (< 5%) occurs anteromedially through the retrosternal foramen of Morgagni.
Clinical Presentation
Most CDH is diagnosed prenatally on second-trimester ultrasound. Postnatally, the newborn presents within minutes to hours of birth with respiratory distress, cyanosis, a scaphoid abdomen (because the gut is in the chest, not the belly), and shifted heart sounds. Breath sounds on the affected side are absent or diminished.
Management
Immediate management is not surgical — it is physiologic stabilization. Intubate (avoid bag-mask ventilation, which inflates the intrathoracic stomach and worsens lung compression), place a large NG/OG tube to decompress the stomach, establish vascular access (including umbilical lines in neonates), and optimize pulmonary hypertension with gentle ventilation, inhaled nitric oxide, and sometimes ECMO (extracorporeal membrane oxygenation). Surgical repair is performed days later once the infant is stable — via subcostal incision or thoracoscopy, with primary repair or patch closure (Gore-Tex, biologic mesh) for larger defects. The CDH EURO Consortium guidelines are the standard reference for stabilization and repair timing.
Never bag-mask ventilate a suspected CDH. Positive-pressure air fills the intrathoracic stomach, crushes the hypoplastic lung, and can precipitate cardiovascular collapse. Intubate immediately and place a sump NG tube to continuous low suction.
04 Tracheoesophageal Fistula & Esophageal Atresia (TEF/EA) Thoracic
TEF/EA is a failure of septation between the primitive foregut and trachea. The most common anatomy (~85%) is a proximal esophageal atresia with a distal tracheoesophageal fistula (Type C / Gross Type C). Isolated EA without a fistula (Type A) accounts for ~8%, and H-type fistula (intact esophagus with a communication to the trachea) is a rare presentation that can escape detection for months. The Spitz classification predicts survival based on birth weight and cardiac disease.
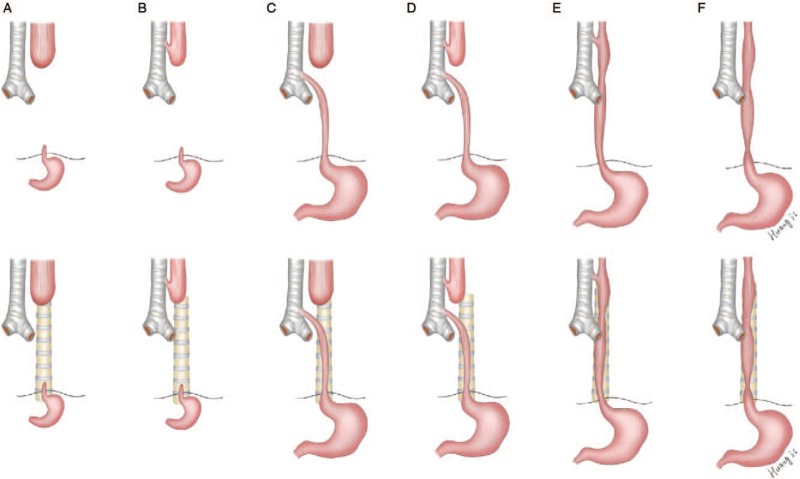
Clinical Presentation
Polyhydramnios on prenatal ultrasound is a clue. Postnatally, the newborn drools excessively, chokes with first feeding, and fails NG tube passage — a 10-Fr OG coils at ~10–12 cm on CXR, the classic diagnostic sign. A gasless abdomen on CXR indicates pure atresia (Type A); a gas-filled abdomen indicates a distal fistula is present.
Search for VACTERL associations (Vertebral, Anal, Cardiac, Tracheoesophageal, Esophageal, Renal, Limb) — cardiac lesions especially must be identified before OR because they affect anesthetic management.
Management
Place a Replogle sump in the proximal pouch to continuous low suction, elevate the head of bed, keep the infant NPO, start broad antibiotics if aspiration is suspected, obtain an echo and renal ultrasound for VACTERL workup. Surgical repair is typically a right posterolateral thoracotomy (or thoracoscopy) with fistula ligation and primary end-to-end esophageal anastomosis. Long-gap EA may require delayed repair, Foker traction elongation, or esophageal replacement.
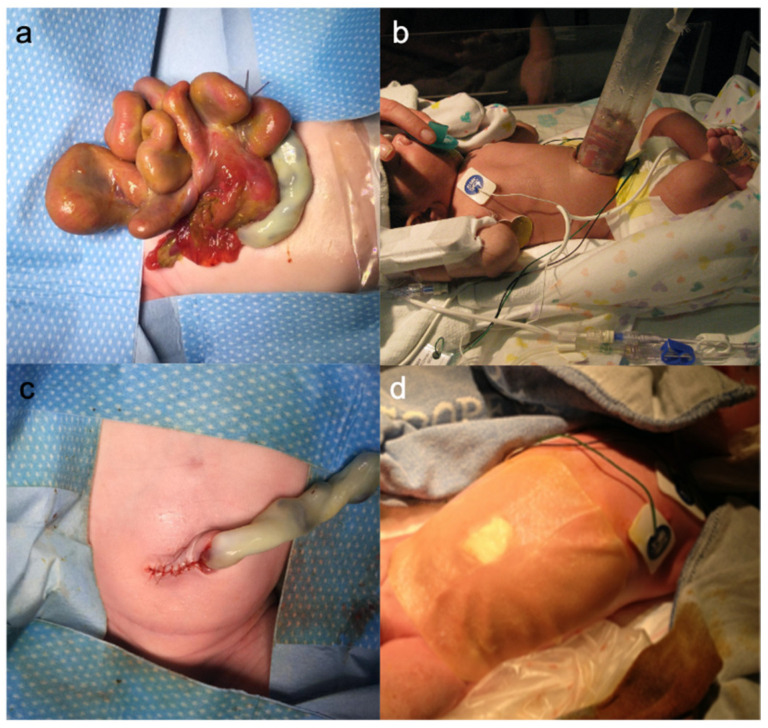
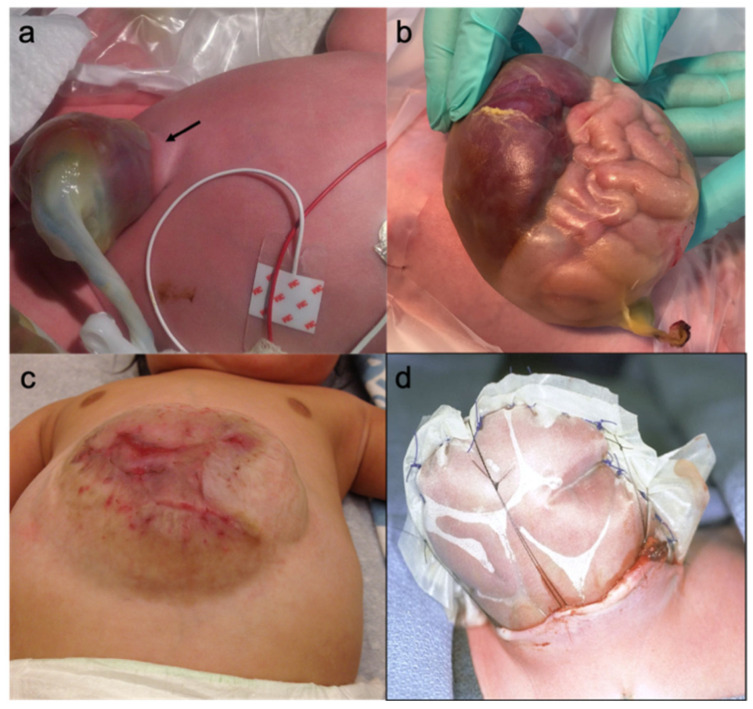
05 Abdominal Wall Defects: Gastroschisis & Omphalocele Abdominal Wall
The two major congenital abdominal wall defects differ in location, covering, associated anomalies, and prognosis — and confusing them in a chart is a serious documentation error.
| Feature | Gastroschisis | Omphalocele |
|---|---|---|
| Location | Right of umbilicus | Midline, through umbilical ring |
| Covering sac | None — bowel exposed | Present (amnion/peritoneum) |
| Contents | Bowel only (usually) | Bowel ± liver ± other viscera |
| Associated anomalies | Few (bowel atresia most common) | Many (cardiac, Beckwith-Wiedemann, trisomies, pentalogy of Cantrell) |
| Prognosis | Good (survival > 90%) | Depends on associated anomalies |
Management
Delivery at a center with pediatric surgery. Immediately after birth, wrap the exposed bowel (gastroschisis) or the intact sac (omphalocele) in warm saline gauze and a clear plastic bowel bag to prevent heat and fluid loss. Place the infant in the right lateral decubitus position to avoid mesenteric kinking. Start IV fluids (gastroschisis babies have tremendous insensible losses — often need 1.5–2× maintenance), NG decompression, and broad-spectrum antibiotics. Closure is either primary (if abdomen accommodates the viscera without excessive intra-abdominal pressure) or staged silo reduction (a Silastic spring-loaded silo is placed and gradually reduced over 3–10 days before fascial closure). Giant omphaloceles may be managed with topical escharotics (silver sulfadiazine or povidone) and delayed closure over months.
06 Hypertrophic Pyloric Stenosis (HPS) GI
HPS is hypertrophy of the pyloric muscle producing gastric outlet obstruction. It classically presents in a first-born male infant aged 3–6 weeks with progressive, projectile, non-bilious, post-prandial vomiting and an insatiable appetite afterward (the "hungry vomiter"). Incidence is ~2–4 per 1000 live births. Family history and erythromycin exposure are risk factors.
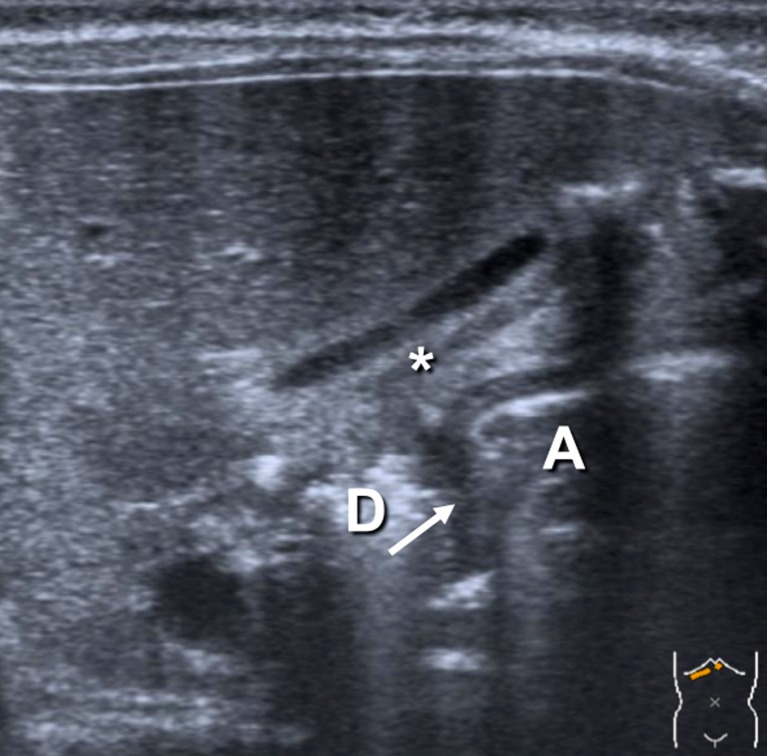
Diagnosis
Classic exam findings include a palpable "olive" in the right upper quadrant / epigastrium after gastric decompression and visible gastric peristaltic waves across the abdomen. The diagnostic imaging test is pyloric ultrasound: pyloric muscle thickness ≥ 3–4 mm and pyloric channel length ≥ 14–17 mm. Labs classically show a hypochloremic, hypokalemic metabolic alkalosis from loss of HCl in vomitus with paradoxical aciduria.
Management
HPS is a medical emergency, not a surgical emergency. The infant must be volume- and electrolyte-resuscitated before OR: normal saline boluses, then D5 ½ NS + 20 mEq/L KCl at 1.5–2× maintenance until the bicarbonate is < 30 and chloride is > 100. Operating on an alkalotic infant risks post-op apnea from CO2 retention. The procedure is laparoscopic (or open Ramstedt) pyloromyotomy — a longitudinal splitting of the pyloric muscle down to but not through the mucosa. Post-op feeds advance rapidly and discharge is usually within 24–48 hours. The APSA consensus on pyloromyotomy supports laparoscopic approach as standard.
07 Malrotation & Midgut Volvulus GI
During fetal development, the midgut rotates 270° counterclockwise around the superior mesenteric artery (SMA) and is fixed by the ligament of Treitz in the left upper quadrant and the cecum in the right lower quadrant, producing a broad mesenteric base. In malrotation, fixation fails. The narrow mesenteric pedicle predisposes to midgut volvulus, where the entire small bowel twists around the SMA, infarcts, and — without emergent surgery — kills the infant within hours.
Presentation & Diagnosis
Any neonate or infant with bilious emesis is assumed to have malrotation with volvulus until proven otherwise. Older children may present with intermittent abdominal pain, failure to thrive, or acutely with shock and peritonitis. The diagnostic study is an upper GI series (UGI): the ligament of Treitz should lie to the left of the spine at the level of the duodenal bulb. An abnormal position or a "corkscrew" or "bird's beak" duodenum is diagnostic. Ultrasound may show reversal of the normal SMA/SMV relationship.
Management
Operative: the Ladd procedure. Steps: (1) evisceration and derotation counterclockwise, (2) division of Ladd's bands (peritoneal bands running from the cecum across the duodenum), (3) widening of the mesenteric base, (4) placement of bowel in non-rotation (small bowel right, colon left), (5) incidental appendectomy (because the cecum is no longer in the RLQ and future appendicitis would be confusing). Necrotic bowel is resected.
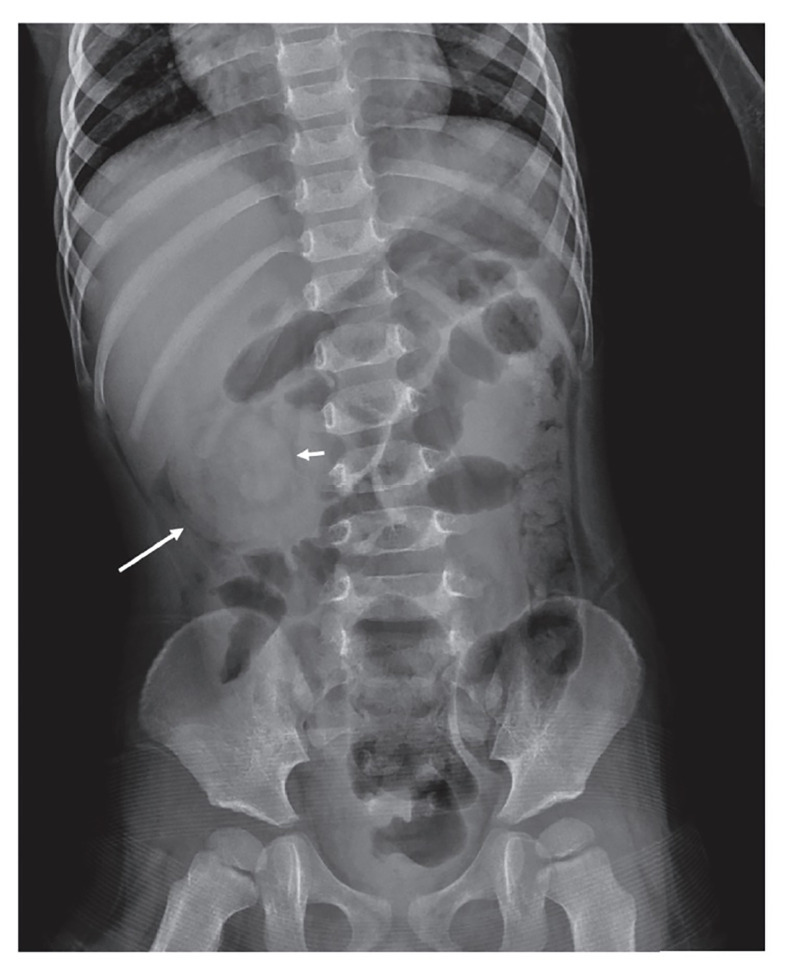
08 Intestinal Atresias GI
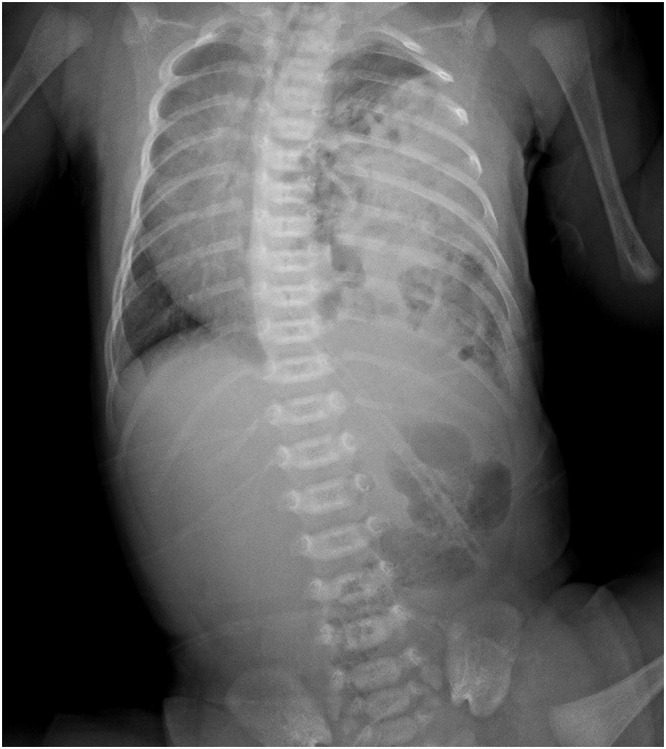
Intestinal atresias are developmental interruptions in bowel continuity. Duodenal atresia is most often a failure of recanalization; jejunoileal and colonic atresias are thought to arise from in utero vascular accidents.
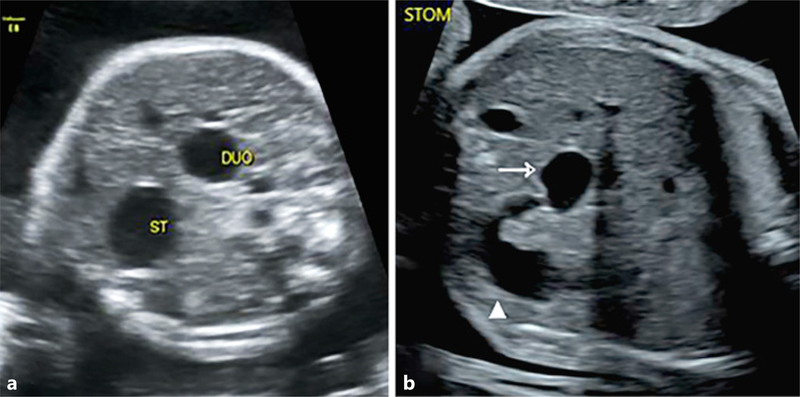
Classic "double bubble" sign on abdominal X-ray (dilated stomach + dilated proximal duodenum, no distal gas). Associated with Down syndrome in ~30%. Presents with bilious (or non-bilious if proximal to ampulla) emesis on day of life 1. Repair: duodenoduodenostomy (diamond-shaped anastomosis).
Grossman/Louw-Barnard classification Types I–IV: I = mucosal web, II = fibrous cord, IIIa = mesenteric defect with separated ends, IIIb = "apple-peel" (proximal jejunal atresia with distal ileum wrapped around a marginal artery), IV = multiple atresias. Presents with bilious emesis, distension, delayed meconium passage. Treatment is resection and primary anastomosis, tapering enteroplasty of a dilated proximal segment.
Rare (~5% of atresias). Presents with distension and failure to pass meconium. Contrast enema shows a microcolon distal to the obstruction. Repair is resection with primary anastomosis or staged with ostomy depending on bowel viability.
09 Hirschsprung Disease GI
Hirschsprung disease (HD) is congenital absence of ganglion cells in the distal bowel due to failed caudal migration of neural crest cells. The aganglionic segment cannot relax, producing a functional obstruction. Short-segment disease (rectosigmoid) accounts for ~80%; long-segment and total colonic aganglionosis are rarer and more difficult. Incidence is ~1 in 5000. Male predominance. Associated with Trisomy 21 (5–15% of HD patients).
Presentation
Failure to pass meconium within the first 48 hours of life (95% of healthy term neonates pass meconium by 24 hours). Progressive abdominal distension, bilious emesis, explosive stool on digital rectal exam (the "squirt sign"). Older undiagnosed children present with chronic severe constipation and failure to thrive. The most feared complication is Hirschsprung-associated enterocolitis (HAEC) — fever, foul explosive diarrhea, distension, and sepsis.
Diagnosis & Treatment
Contrast enema shows a distal narrow aganglionic segment with a transition zone and proximal dilation. Anorectal manometry shows absent recto-anal inhibitory reflex. The definitive diagnosis is rectal suction biopsy demonstrating absent ganglion cells with hypertrophic nerve trunks. Treatment is surgical pull-through, historically staged with colostomy but now often done in a single primary pull-through. Operative approaches include Soave (endorectal), Swenson (full-thickness), and Duhamel (retrorectal pouch). The APSA Hirschsprung guidelines discuss technique selection.
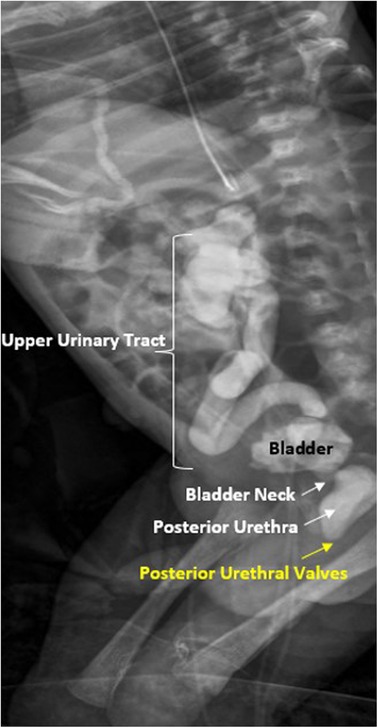
10 Anorectal Malformations & Cloaca GI
Anorectal malformations (ARM), formerly called "imperforate anus," are a spectrum from low defects (perineal fistula, easy repair) to high complex defects (rectourethral, rectovesical, or cloacal). The Krickenbeck classification (2005) is the current international system; the older Peña classification is still frequently quoted.
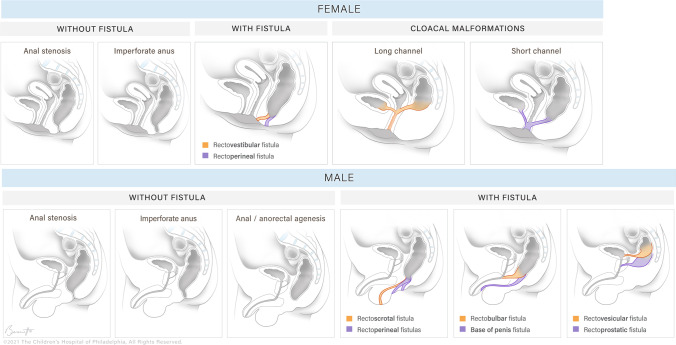
| Peña Subtype | Description | Prognosis |
|---|---|---|
| Perineal fistula (M/F) | Rectum opens to perineum anterior to normal position | Excellent — often primary PSARP |
| Rectourethral bulbar (M) | Fistula to bulbar urethra | Good |
| Rectourethral prostatic (M) | Fistula to prostatic urethra | Fair |
| Rectovesical (M) | Fistula to bladder neck | Poor — high incontinence risk |
| Vestibular fistula (F) | Rectum opens to vestibule posterior to vagina | Excellent |
| Cloaca (F) | Single perineal opening (rectum, vagina, urethra) | Complex, variable |
| No fistula | Blind-ending rectum, no communication | Variable (25% Down syndrome) |
Workup at birth: look for a fistula, check for VACTERL anomalies, spinal ultrasound for tethered cord, echo, renal ultrasound. High defects get a diverting colostomy at birth followed by posterior sagittal anorectoplasty (PSARP) at ~3 months, then colostomy takedown. Low defects can sometimes be repaired primarily without diversion.
11 Meconium Ileus & CF-Associated GI Disease GI
Meconium ileus is obstruction of the distal ileum by thick, inspissated meconium and is strongly associated with cystic fibrosis (nearly 100% of patients with meconium ileus have CF). Simple meconium ileus presents with distension, bilious emesis, and failure to pass meconium, with a "soap-bubble" appearance in the RLQ on X-ray and a microcolon on contrast enema. Complicated meconium ileus includes perforation, volvulus, atresia, or meconium pseudocyst formation.
Simple cases may respond to Gastrografin enema (hyperosmolar, draws water into the lumen and liquefies meconium) — both diagnostic and therapeutic. Failed medical management or complicated cases require operative intervention (enterotomy and irrigation, resection of necrotic or atretic bowel, ± ostomy). Every patient with meconium ileus requires CF genetic/sweat-chloride workup.
12 Necrotizing Enterocolitis (NEC) GI
NEC is a devastating inflammatory and ischemic disease of the neonatal intestine, predominantly affecting premature infants (the more premature, the higher the risk). It is the most common surgical GI emergency in the NICU. Pathophysiology involves an immature mucosal barrier, abnormal bacterial colonization, and inflammatory cascade triggering ischemia, pneumatosis, and full-thickness necrosis.
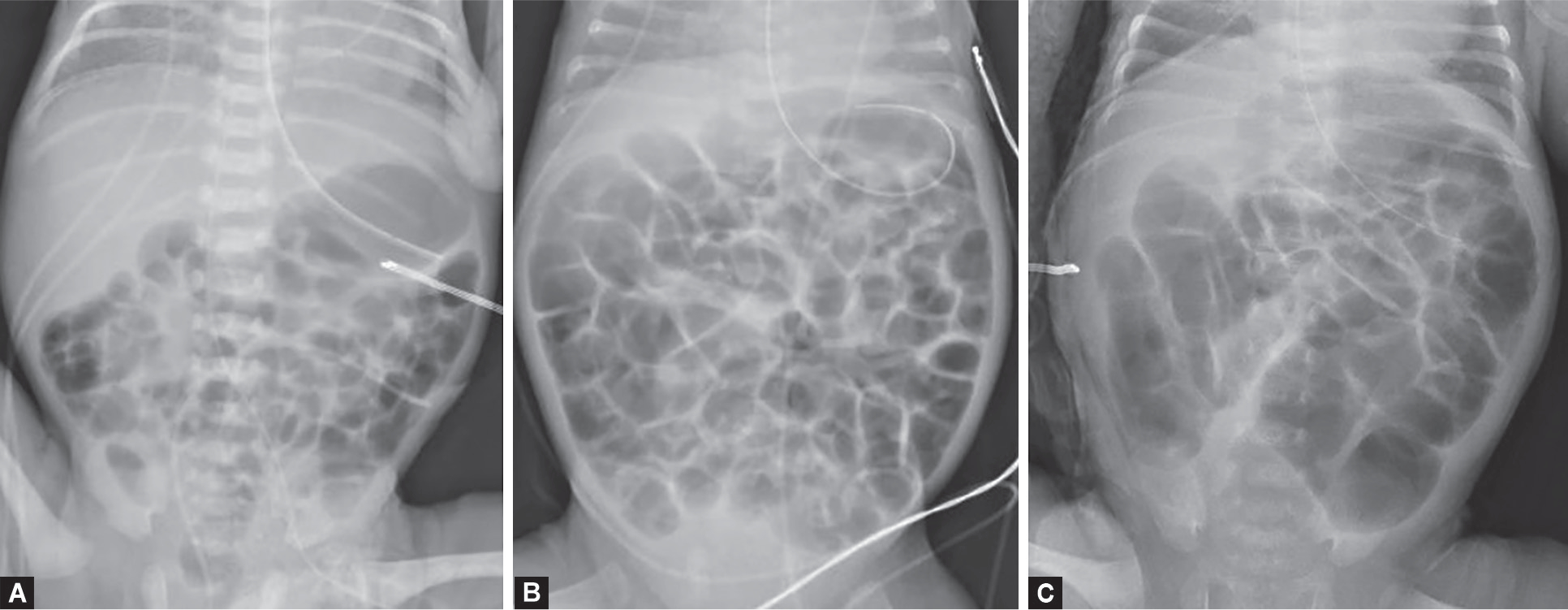
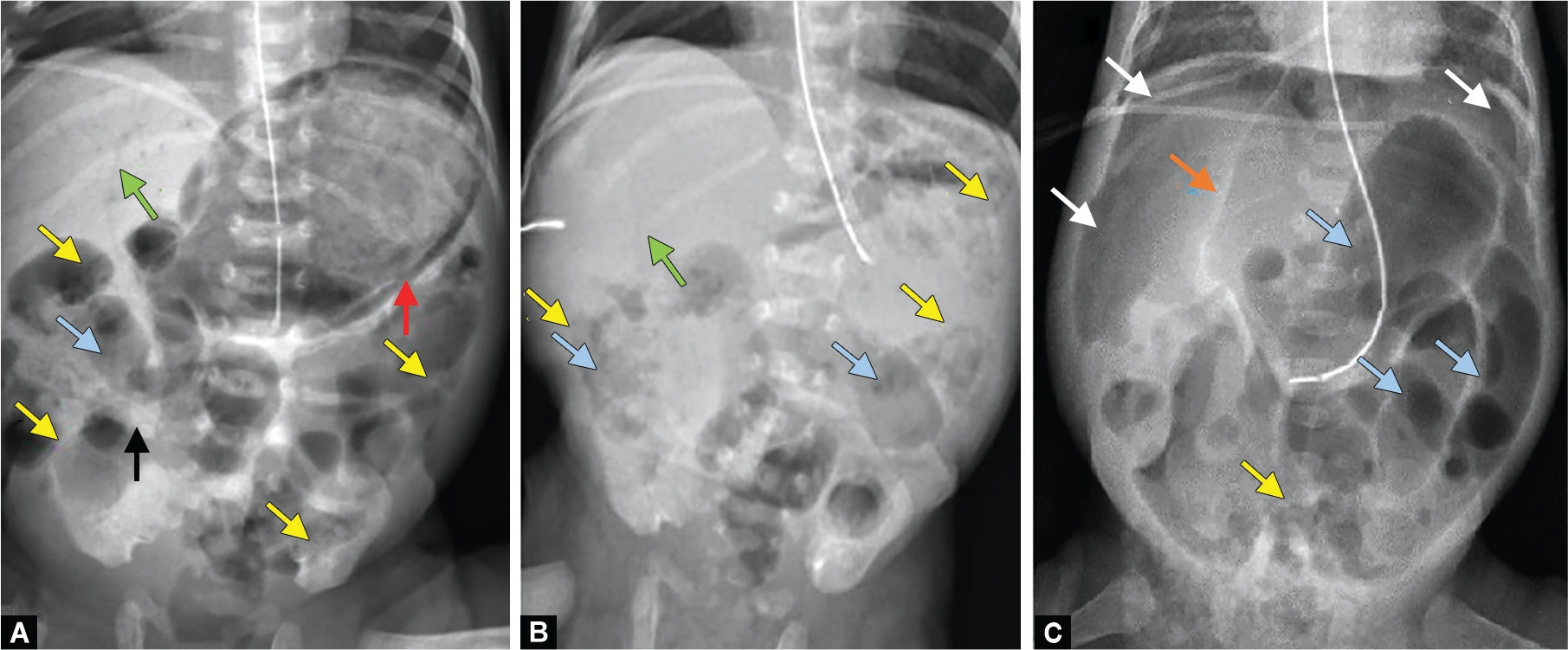
Presentation
The classic triad is abdominal distension, bloody stools, and feeding intolerance in a premature infant, often within the first 2–4 weeks of life. Progression brings temperature instability, apnea, lethargy, metabolic acidosis, thrombocytopenia, DIC, and shock.
Bell Staging
Stage IA (Suspected): Mild systemic signs (apnea, temp instability), mild GI signs (feeding intolerance, mild distension, guaiac-positive stool), non-specific X-ray.
Stage IB (Suspected): Same as IA plus grossly bloody stool.
Stage IIA (Definite, mildly ill): Same clinical signs plus absent bowel sounds, possibly RLQ tenderness, pneumatosis intestinalis on X-ray.
Stage IIB (Definite, moderately ill): Mild metabolic acidosis, mild thrombocytopenia, definite abdominal tenderness, ± cellulitis or RLQ mass, X-ray shows pneumatosis and/or portal venous gas, ± ascites.
Stage IIIA (Advanced, critically ill, bowel intact): Hypotension, bradycardia, severe apnea, mixed acidosis, DIC, neutropenia, generalized peritonitis, marked distension, X-ray with definite ascites.
Stage IIIB (Advanced, perforated): Same clinical picture plus pneumoperitoneum on X-ray. Surgical.
Management
Medical management for Stage I–II: NPO, NG decompression, broad-spectrum antibiotics (ampicillin + gentamicin + metronidazole; add vancomycin if central-line associated), parenteral nutrition, serial abdominal exams and X-rays every 6 hours, correction of thrombocytopenia and coagulopathy. Surgical indications include pneumoperitoneum (Stage IIIB), failure of medical management, fixed dilated loop, abdominal mass, erythema of abdominal wall, or progressive clinical deterioration. Operative options are peritoneal drain placement (for very small unstable neonates) or exploratory laparotomy with resection of necrotic bowel, primary anastomosis or diverting ostomy. The NECSTEPS and NICHD NEC trials continue to guide drain vs laparotomy decision-making.
13 Intussusception GI
Intussusception is telescoping of one segment of bowel (intussusceptum) into an adjacent distal segment (intussuscipiens). The most common location is ileocolic. Peak age is 6 months to 3 years. In children under 2, most cases are idiopathic — likely triggered by enlarged Peyer's patches after a viral URI or gastroenteritis. In older children or recurrent cases, look for a pathologic lead point (Meckel diverticulum, polyp, lymphoma, duplication cyst, Henoch-Schonlein purpura).
Presentation
Classic triad (present in < 50%): colicky abdominal pain (intermittent, drawing legs to abdomen, then returning to baseline), vomiting, and "currant jelly" stool (mix of blood and mucus — a late finding). A sausage-shaped RUQ mass may be palpable. Lethargy can be a presenting feature and is easily mistaken for sepsis or encephalopathy.
Management
Ultrasound is the diagnostic test of choice: the "target" sign on transverse, "pseudokidney" on longitudinal. First-line therapy for the stable child without peritonitis or perforation is air contrast enema reduction (success 80–90%). Hydrostatic (saline) enema is an alternative. The surgical team must be available before reduction because of perforation risk. Surgical reduction is indicated for failed enema, perforation, peritonitis, or a pathologic lead point. Manual reduction is attempted first; resection is performed if reduction fails or bowel is non-viable.
14 Meckel Diverticulum GI
Meckel diverticulum is a true diverticulum of the ileum — a persistent remnant of the omphalomesenteric (vitelline) duct. The mnemonic "rule of 2s": 2% of population, within 2 feet of the ileocecal valve, 2 inches long, 2 types of heterotopic mucosa (gastric, pancreatic), symptomatic usually before age 2, 2:1 male predominance. Presentations include painless lower GI bleeding (most common in children, from ectopic gastric mucosa ulcerating adjacent ileum), small bowel obstruction (intussusception with Meckel as lead point, or volvulus around an omphalomesenteric band), or diverticulitis mimicking appendicitis. Diagnosis is by Meckel scan (technetium-99m pertechnetate scintigraphy, which tags gastric mucosa). Treatment is diverticulectomy or segmental ileal resection.
15 Pediatric Appendicitis GI
Appendicitis is the most common surgical emergency in children. Pathophysiology is luminal obstruction (fecalith, lymphoid hyperplasia after viral illness) followed by distension, ischemia, bacterial overgrowth, and perforation.
Presentation
Classic progression: periumbilical pain migrating to RLQ, anorexia, nausea/vomiting, low-grade fever. Anorexia is a strongly suggestive pediatric finding. McBurney's point tenderness, Rovsing's sign (RLQ pain on LLQ palpation), Psoas sign, Obturator sign. Younger children (< 5 years) present atypically and have much higher perforation rates at presentation (> 80% in toddlers).
Workup & Scoring
Labs: WBC with left shift, CRP, urinalysis. The Pediatric Appendicitis Score (PAS) and Alvarado score risk-stratify for imaging and disposition decisions. Imaging: ultrasound first line (visualized non-compressible appendix > 6 mm with peri-appendiceal inflammation), CT only when ultrasound is equivocal and concern remains high, to minimize radiation.
Management
NPO, IV fluids, broad-spectrum antibiotics (ceftriaxone + metronidazole, or piperacillin-tazobactam). Definitive treatment: laparoscopic appendectomy. Perforated appendicitis with phlegmon or abscess may be managed with initial IV antibiotics and percutaneous drainage followed by interval appendectomy 6–8 weeks later. Non-operative management of uncomplicated appendicitis with antibiotics alone is an evolving option; see the APSA non-operative appendicitis data.
16 Inguinal, Umbilical & Other Hernias Abdominal Wall
Pediatric inguinal hernias are almost always indirect, resulting from a patent processus vaginalis. Incidence is ~1–5% of term infants, much higher (up to 30%) in preterms. Right-sided predominance (60%). Bilateral in ~10–15%. Repair is recommended at diagnosis because of the risk of incarceration, particularly in young infants.
Surgical options are open high ligation of the sac through a groin incision or laparoscopic repair (percutaneous internal ring suturing or intracorporeal ligation). Laparoscopy has the advantage of inspecting the contralateral side through the same access. Incarcerated hernias are reduced manually first (Trendelenburg position, sedation, gentle pressure); if reducible, semi-urgent repair within 24–48 hours after edema resolves; if irreducible or with signs of compromise, emergent OR. Hydroceles in infants under 2 are usually watched and resolve spontaneously; persistent or communicating hydroceles after age 2 are repaired like an inguinal hernia.
Umbilical hernias are extremely common (particularly in African-American infants and preterm infants) and the vast majority close spontaneously by age 4–5. Repair is indicated for persistent defects beyond age 4–5, fascial defects > 1.5 cm, incarceration (rare), or symptomatic pain.
Epigastric hernias (small midline linea alba defects with pre-peritoneal fat) do not close spontaneously and are repaired when diagnosed. Spigelian, femoral, and incisional hernias are rare in children but follow similar principles as in adults.
17 Undescended Testis, Hydrocele, Varicocele & Torsion Genitourinary
Cryptorchidism (undescended testis) affects ~3% of term males and ~30% of preterms. Spontaneous descent is uncommon after 6 months of age. Evaluation distinguishes palpable (inguinal canal, ectopic) from non-palpable (intra-abdominal, absent, atrophic) testes. Bilateral non-palpable testes with ambiguous genitalia raise concern for disorders of sexual development (DSD) and require urgent karyotype and endocrine workup before any surgery.
Surgical treatment is orchiopexy performed between 6–18 months of age per AUA guidelines to preserve fertility and reduce malignancy risk. Palpable testes: inguinal approach with hernia sac dissection and dartos pouch placement. Non-palpable: diagnostic laparoscopy with either a primary laparoscopic orchiopexy, Fowler-Stephens (2-stage) orchiopexy for high intra-abdominal testes, or orchiectomy for nubbin/atrophic remnants.
Communicating hydroceles reflect a patent processus — repaired like inguinal hernia. Varicoceles (dilated pampiniform plexus, almost always left-sided due to left spermatic vein drainage into the left renal vein) are repaired in adolescents with testicular growth arrest, pain, or infertility concerns.
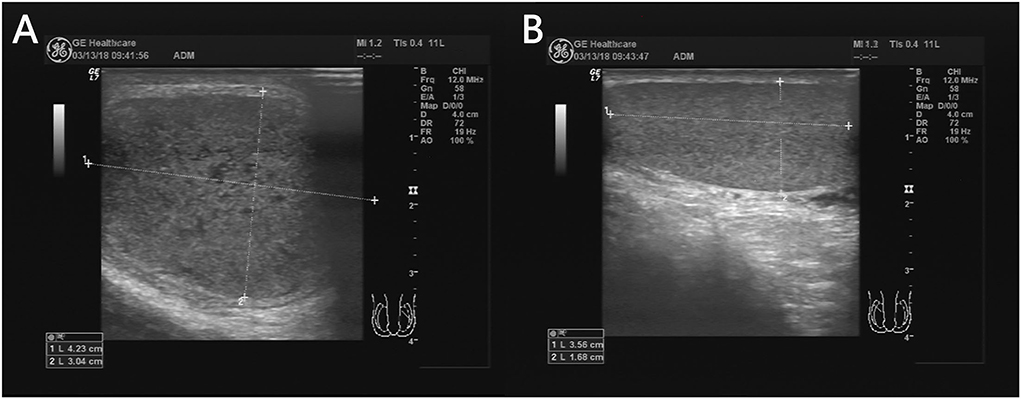
Testicular torsion is a surgical emergency. Peak ages are neonatal (extravaginal) and adolescent (intravaginal, "bell-clapper deformity"). Presents with sudden severe unilateral scrotal pain, nausea, high-riding transverse testis, absent cremasteric reflex. Doppler US shows absent flow. The 6-hour window for salvage is critical — do not wait for imaging if clinical suspicion is high. Treatment: urgent scrotal exploration, detorsion, bilateral orchiopexy (the contralateral side has the same anatomic predisposition).
Ovarian torsion presents with sudden severe unilateral lower abdominal pain ± mass, vomiting. Doppler ultrasound is the first-line imaging. Treatment is laparoscopic detorsion with ovary preservation whenever possible, even for black, bruised-appearing ovaries — recovery of function is surprising.
18 Pediatric Solid Tumors Oncology
Pediatric surgical oncology is a multidisciplinary enterprise in partnership with pediatric oncology, radiation oncology, and pathology, coordinated through Children's Oncology Group (COG) protocols.
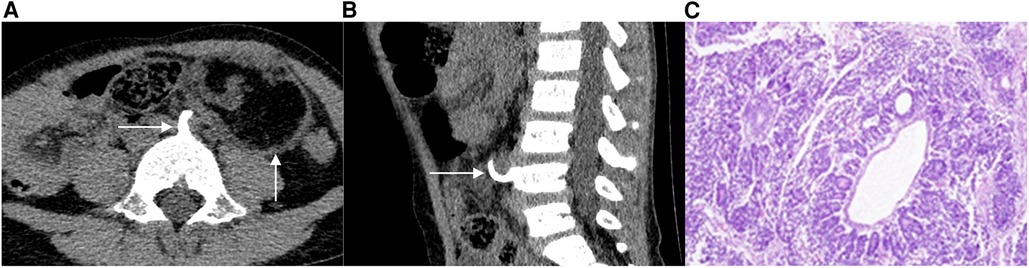
Wilms Tumor (Nephroblastoma)
Most common renal tumor of childhood. Peak age 2–5. Presents as an asymptomatic abdominal mass noted by caregivers at bath time, ± hematuria, hypertension. Associations: WAGR (Wilms, Aniridia, GU anomalies, Retardation), Beckwith-Wiedemann, Denys-Drash syndromes. Workup includes abdominal ultrasound, CT/MRI of abdomen, CXR (lung mets). Staging by COG:
Stage I: Tumor limited to kidney, completely resected.
Stage II: Tumor extends beyond kidney but completely resected (capsule breached, perirenal fat or vessels involved).
Stage III: Residual non-hematogenous tumor confined to abdomen (positive margin, nodes, rupture).
Stage IV: Hematogenous metastases (lung, liver, bone, brain) or nodes outside the abdomen.
Stage V: Bilateral renal involvement at diagnosis.
Treatment per COG: primary resection in North America for most stages, chemotherapy (vincristine, actinomycin-D, ± doxorubicin), radiation for advanced disease. See COG Renal Tumors Committee protocols.
Neuroblastoma
Most common extracranial solid tumor of childhood, arising from neural crest cells in the adrenal medulla or sympathetic chain. Median age < 2 years. Presents with abdominal mass, "raccoon eyes" periorbital bruising from metastases, opsoclonus-myoclonus-ataxia ("dancing eyes"), Horner syndrome (cervical primary), or paraspinal dumbbell tumors. Elevated urinary VMA and HVA. INSS staging:
Stage 1: Localized tumor, complete gross excision.
Stage 2A: Localized, incomplete resection, negative ipsilateral nodes.
Stage 2B: Localized, ± incomplete resection, positive ipsilateral nodes.
Stage 3: Unresectable unilateral tumor crossing midline, ± nodes; or midline tumor with bilateral extension.
Stage 4: Distant metastases (distant nodes, bone, marrow, liver, skin, other).
Stage 4S: Localized primary (stage 1/2A/2B) in infant < 12 months with dissemination limited to liver, skin, or <10% marrow (often regresses spontaneously).
Hepatoblastoma
Most common primary liver malignancy of childhood, typically in children < 3. Elevated alpha-fetoprotein (AFP) is characteristic. Treatment per COG protocols: neoadjuvant chemotherapy (cisplatin-based) followed by surgical resection (segmentectomy, lobectomy, extended hepatectomy, or transplant for unresectable tumors).
Rhabdomyosarcoma
Most common soft-tissue sarcoma of childhood, from embryonal mesenchyme. Common sites: head and neck (parameningeal, orbital), genitourinary (bladder, prostate, vagina, paratesticular), extremities. Two histologic types: embryonal (better prognosis) and alveolar. Treated with chemotherapy, radiation, and surgery per COG Soft Tissue Sarcoma Committee protocols.
19 Congenital Lung Lesions & Thoracic Disease Thoracic
Congenital lung lesions are increasingly diagnosed prenatally. Key entities:
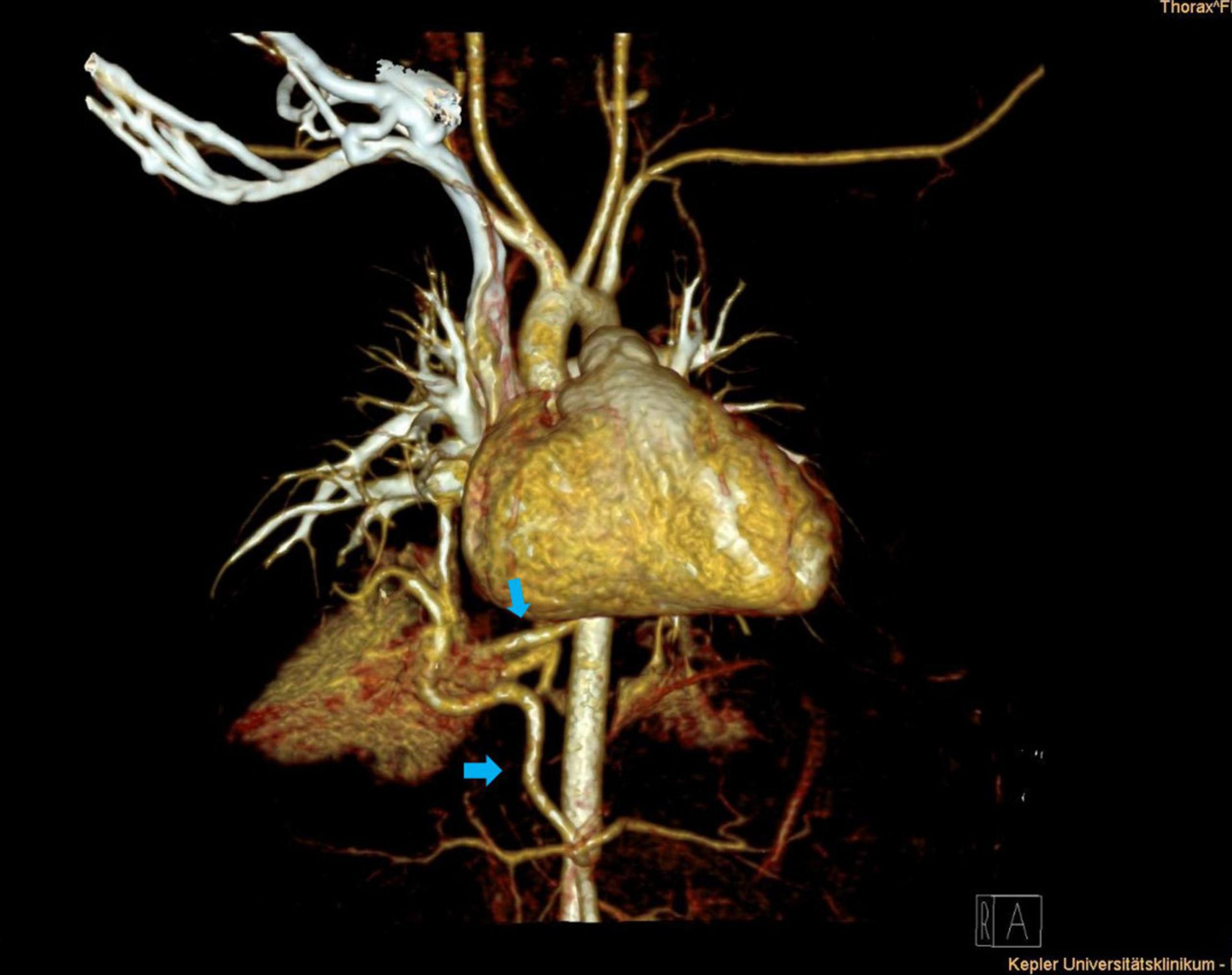
- Congenital pulmonary airway malformation (CPAM, formerly CCAM): Hamartomatous lung tissue with cystic and solid components. Stocker types I–IV. Can regress, remain asymptomatic, or present with recurrent infection and (rarely) malignant transformation. Elective lobectomy at 6–12 months is common.
- Bronchopulmonary sequestration: Non-functioning lung tissue with systemic arterial supply (often from the thoracic or abdominal aorta). Intralobar or extralobar. Treatment is resection, with meticulous attention to the anomalous systemic artery.
- Congenital lobar emphysema: Progressive hyperinflation of a lobe (most commonly LUL), causing mediastinal shift and respiratory distress. Treatment is lobectomy.
- Bronchogenic cyst: Foregut duplication cyst; resect if symptomatic or for diagnostic certainty.
20 Hepatobiliary: Biliary Atresia & Choledochal Cyst Hepatobiliary
Biliary atresia is a progressive obliterative cholangiopathy of unknown cause and the leading indication for pediatric liver transplant. Presents with persistent jaundice beyond 2 weeks of life, acholic (pale) stools, dark urine, and direct hyperbilirubinemia. Diagnosis is by HIDA scan (no excretion into the bowel), ultrasound (absent or triangular cord sign), and intraoperative cholangiogram. Treatment is the Kasai portoenterostomy — excision of the obliterated extrahepatic biliary tree and anastomosis of a Roux-en-Y limb of jejunum to the porta hepatis, ideally before 60 days of life for best outcomes. Many patients still progress to cirrhosis and require liver transplant.
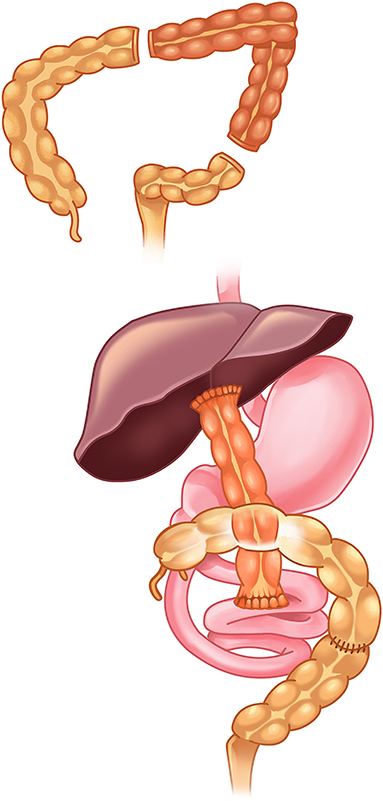
Choledochal cysts are congenital dilations of the biliary tree. Todani classification I–V. Present with jaundice, abdominal pain, and a palpable mass (the classic triad). Treated with complete cyst excision and hepaticojejunostomy due to malignancy risk.
21 Head & Neck Masses Head & Neck
Thyroglossal duct cyst: Midline neck mass that elevates with tongue protrusion, sitting just below the hyoid. Treatment is the Sistrunk procedure — cyst excision with the central portion of the hyoid bone and a core of tongue-base tissue to the foramen cecum.
Branchial cleft cysts/sinuses: Lateral neck, anterior to the sternocleidomastoid. Second branchial cleft is the most common, tracking between the ICA and ECA to the tonsillar fossa. Complete excision including the tract.
Lymphatic malformations (lymphangioma / cystic hygroma): Macrocystic, microcystic, or mixed. Often neck or axilla. Management includes sclerotherapy (doxycycline, bleomycin, OK-432) and/or surgical excision.
Infantile hemangiomas and vascular anomalies: Distinguish hemangiomas (proliferate then involute, GLUT-1 positive) from vascular malformations (do not involute). Problematic hemangiomas may be treated medically with propranolol per the AAP clinical practice guideline on infantile hemangioma.
22 Pediatric Trauma, Burns & Foreign Bodies Trauma
Pediatric trauma is the leading cause of childhood death in the US. Children compensate with tachycardia and peripheral vasoconstriction; hypotension is a terminal sign. Large heads, small airways, and thin compliant chest walls mean significant internal injury can occur without rib fractures.
Blunt abdominal trauma: Liver and spleen are the most injured solid organs. Non-operative management is the rule for hemodynamically stable patients with contrast-enhanced CT documentation. Follow the APSA solid-organ injury guidelines. Bed rest, serial hemoglobin monitoring, and hemodynamic watch. Surgery for hemodynamic instability, ongoing transfusion requirement, or hollow viscus injury.
Head injury: Use pediatric GCS (verbal subscore modified for infants — coos/babbles = 5, irritable cry = 4, cries to pain = 3, moans = 2, none = 1). PECARN decision rules reduce unnecessary head CT in low-risk children.
Non-accidental trauma (NAT): Red flags include history inconsistent with injury, developmentally impossible mechanism, patterned bruising, bruises in non-ambulatory infants ("those who don't cruise rarely bruise"), multiple fractures of different ages, spiral long bone fractures, metaphyseal corner fractures, posterior rib fractures, retinal hemorrhages, and delayed presentation. Document physical findings objectively and completely; notify the child protection team and law enforcement as required.
Burns: Estimate TBSA by the Lund-Browder chart (not the adult rule of nines — child head is proportionally larger). Fluid resuscitation: Parkland 4 mL/kg/%TBSA LR plus maintenance per 4-2-1. Transfer to burn center for > 10% TBSA partial thickness, full-thickness, hands/face/genitalia, electrical or chemical, or concern for NAT.
Foreign body ingestion: Most pass spontaneously. Button batteries in the esophagus are a true emergency — liquefactive necrosis can perforate within 2 hours. Endoscopic removal immediately. Give honey (age > 12 months) as a temporizing measure per the NASPGHAN button battery guideline. Multiple magnets can attract across bowel walls and cause perforations, fistulas, and obstruction — endoscopic or surgical removal is often required.
23 Pectus Deformities & Miscellaneous Entities Chest Wall
Pectus excavatum: Most common chest wall deformity, a posterior depression of the sternum. The Haller index (transverse chest diameter / anteroposterior diameter on CT) > 3.25 supports operative repair. The Nuss procedure is a minimally invasive pectus bar repair performed during adolescence; the Ravitch is an older open approach. Pectus carinatum (protuberant sternum) is typically managed with orthotic bracing; surgery is reserved for severe refractory cases.
Short bowel syndrome: Intestinal failure from extensive small bowel resection (often NEC survivors, gastroschisis with atresia, volvulus). Managed with TPN, gradual enteral advancement, specialized formulas, and, in refractory cases, bowel-lengthening procedures (STEP, Bianchi) or intestinal transplantation.
Congenital hyperinsulinism: Hypoglycemia from dysregulated insulin secretion. Medical therapy with diazoxide first line; refractory disease may require near-total pancreatectomy (for diffuse disease) or focal resection (for focal disease confirmed by 18F-DOPA PET).
GERD requiring fundoplication: Severe reflux in neurologically impaired children or those with failed medical management may require laparoscopic Nissen fundoplication, often combined with gastrostomy placement.
24 Pediatric Surgical Procedures — A to Z Procedures
| Procedure | Description / Indication |
|---|---|
| Laparoscopic pyloromyotomy | Longitudinal splitting of pyloric muscle for HPS |
| Ladd procedure | Derotation, Ladd's band division, broadening of mesentery, appendectomy for malrotation |
| Laparoscopic Nissen fundoplication | 360° gastric wrap for refractory GERD |
| Gastrostomy tube (PEG, lap, open Stamm) | Enteral access for feeding |
| Open / laparoscopic inguinal hernia repair | High ligation of hernia sac |
| Umbilical hernia repair | Fascial closure of umbilical defect |
| Orchiopexy | Mobilization and scrotal fixation of undescended testis |
| Fowler-Stephens orchiopexy | Two-stage for high intra-abdominal testis |
| Hydrocelectomy | Excision of hydrocele sac, processus ligation |
| Varicocelectomy | Ligation of dilated pampiniform veins |
| Laparoscopic / open appendectomy | Treatment of appendicitis |
| Radical nephroureterectomy | Wilms tumor resection |
| Neuroblastoma resection | Primary or after neoadjuvant chemotherapy |
| NEC peritoneal drain / laparotomy | Drainage vs resection ± ostomy for perforated NEC |
| Pull-through (Soave, Swenson, Duhamel) | Resection of aganglionic segment for Hirschsprung |
| PSARP | Posterior sagittal anorectoplasty for ARM |
| Kasai portoenterostomy | Roux-en-Y to porta hepatis for biliary atresia |
| Thoracoscopic lobectomy | Resection of CPAM, sequestration, lobar emphysema |
| CDH repair | Primary or patch diaphragm reconstruction |
| TEF/EA repair | Fistula ligation, end-to-end esophageal anastomosis |
| Gastroschisis/omphalocele closure | Primary vs staged silo reduction |
| Nuss procedure | Minimally invasive pectus excavatum repair with substernal bar |
| Bowel resection & ostomy | NEC, atresia, Hirschsprung, Crohn |
| Central line / port placement | Broviac, Hickman, Port-a-Cath for long-term access |
| ECMO cannulation | Neck (VA/VV) or central access for CDH, sepsis, cardiac failure |
| Endoscopic foreign body removal | Button battery, magnets, coins, sharp objects |
| Sistrunk procedure | Thyroglossal duct cyst excision with hyoid segment |
| STEP / Bianchi | Bowel-lengthening for short bowel syndrome |
25 Imaging, Medications & Fluids Reference
Imaging
Pediatric imaging is driven by the ALARA principle (As Low As Reasonably Achievable). Ultrasound and MRI are favored over CT whenever possible to minimize cumulative radiation. Key studies:
- Abdominal X-ray: NEC (pneumatosis, portal venous gas, pneumoperitoneum), obstruction (dilated loops, air-fluid levels, paucity of distal gas), foreign body.
- Upper GI series: Malrotation (position of ligament of Treitz), duodenal obstruction.
- Contrast enema: Hirschsprung (transition zone), meconium ileus (microcolon), intussusception reduction.
- Ultrasound: Pyloric stenosis, intussusception, appendicitis, testicular/ovarian torsion, hip effusion, intracranial (open fontanelle).
- HIDA scan: Biliary atresia (no bowel excretion at 24 h).
- Meckel scan: Tc-99m pertechnetate for ectopic gastric mucosa.
- MRI: Tethered cord, pelvic masses, sarcoma staging, CNS tumors.
- CT: Trauma, complex oncologic staging, abscess drainage planning.
Medications & Weight-Based Dosing
| Drug | Class / Use | Pediatric Dosing |
|---|---|---|
| Ampicillin | Penicillin — NEC, neonatal sepsis | 50–100 mg/kg/dose IV q6–12h |
| Gentamicin | Aminoglycoside — NEC, sepsis | 4–5 mg/kg/dose IV q24h (neonatal renal-adjusted) |
| Metronidazole | Anaerobic coverage — NEC, intra-abdominal | 7.5 mg/kg/dose IV q6–8h |
| Vancomycin | Gram-positive/MRSA — line infections | 10–15 mg/kg/dose IV q6–12h (trough-guided) |
| Ceftriaxone | Third-gen cephalosporin — appendicitis (not < 28 d) | 50–75 mg/kg/day IV q24h |
| Piperacillin-tazobactam | Broad coverage — perforated appy, complicated NEC | 100 mg/kg/dose piperacillin q6–8h |
| Acetaminophen | Analgesic / antipyretic | 10–15 mg/kg PO/PR q4–6h or 15 mg/kg IV q6h (max 75 mg/kg/day) |
| Ibuprofen | NSAID (> 6 months) | 5–10 mg/kg/dose PO q6–8h |
| Morphine | Opioid analgesia | 0.05–0.1 mg/kg/dose IV q2–4h |
| Fentanyl | Opioid — neonatal / procedural | 0.5–2 mcg/kg/dose IV |
| Ondansetron | Antiemetic | 0.15 mg/kg/dose IV/PO q8h (max 4 mg) |
| Ranitidine / famotidine | H2 blocker — stress ulcer, GERD | 1 mg/kg/dose q8–12h (famotidine) |
| Omeprazole / lansoprazole | PPI — GERD | 1 mg/kg/day PO daily |
| Diazoxide | Congenital hyperinsulinism | 5–15 mg/kg/day PO divided q8–12h |
| Propranolol | Infantile hemangioma | 1–3 mg/kg/day PO divided |
| Heparin | Line / ECMO anticoagulation | 10–28 U/kg/hr IV (PTT or anti-Xa titrated) |
Maintenance Fluids
First 10 kg: 4 mL/kg/hr. Next 10 kg (10–20): add 2 mL/kg/hr. Each kg above 20: add 1 mL/kg/hr. Standard maintenance fluid: D5 ½ NS + 20 mEq/L KCl (or isotonic balanced saline for surgical/resuscitation patients; isotonic solutions are now recommended to avoid iatrogenic hyponatremia per the AAP clinical practice guideline on maintenance IV fluids).
26 Classification Systems & Staging Reference
APGAR Score
Appearance (color): 0 blue/pale, 1 acrocyanotic, 2 completely pink.
Pulse (HR): 0 absent, 1 < 100, 2 ≥ 100.
Grimace (reflex): 0 none, 1 grimace, 2 cough/sneeze/cry.
Activity (tone): 0 limp, 1 some flexion, 2 active motion.
Respirations: 0 absent, 1 weak/irregular, 2 strong cry.
Total 0–10. < 4 severely depressed, 4–6 moderately depressed, 7–10 reassuring.
Ballard Score
Combines neuromuscular and physical maturity findings to estimate gestational age in a newborn, especially useful when dates are uncertain.
Pediatric GCS (Infant Modification)
5 = coos, babbles; 4 = irritable cry; 3 = cries to pain; 2 = moans to pain; 1 = no response. Motor and eye components are the same as adult GCS.
Salter-Harris Fracture Classification
I: Slipped — through the physis only.
II: Above — through the physis and metaphysis (most common).
III: Lower — through the physis and epiphysis into the joint.
IV: Transverse — through metaphysis, physis, and epiphysis.
V: cRush — compression injury to the physis (worst prognosis).
Spitz Classification (EA/TEF)
Risk stratification by birth weight and cardiac disease: I = BW > 1500 g, no major cardiac disease (best prognosis); II = BW < 1500 g OR major cardiac disease; III = BW < 1500 g AND major cardiac disease (highest mortality).
Bell Staging — NEC
See Section 12 for the full modified Bell classification I–IIIB.
Wilms & Neuroblastoma Staging
See Section 18 for COG Wilms staging I–V and INSS neuroblastoma staging 1–4S.
Ponseti Method (Clubfoot)
Non-operative treatment for congenital talipes equinovarus: serial weekly casting in a specific sequence (Cavus correction, Adductus, Varus, Equinus), percutaneous Achilles tenotomy in > 80%, followed by prolonged foot abduction bracing until age 4–5.
27 Physical Exam, Abbreviations & Sample HPIs
Pediatric Surgical Physical Exam Template
Gen: Well-appearing / toxic-appearing; alert / lethargic / irritable / consolable
Vitals: T ___ HR ___ RR ___ BP ___ SpO2 ___ Wt ___ kg (__%ile)
HEENT: Fontanelle ___; MMM; no scleral icterus
CV: RRR, no murmur, normal perfusion, cap refill ___
Pulm: CTAB, no retractions, no grunting
Abd: Soft / distended, +/- tender, +/- guarding, BS present, no masses, no hernias, umbilicus clean
GU: Male — testes descended bilaterally, no hernia, no hydrocele; Female — normal external genitalia
Back: No sacral dimple, tuft, or mass
Ext: Warm, well-perfused, pulses 2+, no edema
Skin: No rash, no bruising, no jaundice
Abbreviations Master List
Sample HPI Templates
"Baby [Name] is a 5-week-old full-term male (38+6 weeks, BW 3.4 kg, SVD, uncomplicated) first-born brought in by mother for 10 days of progressively worsening non-bilious, projectile post-prandial vomiting. Mother reports the infant vomits within 20 minutes of every breastfeed, immediately wants to feed again, and has had decreased wet diapers (3 in the last 24 hours) and no stool in 2 days. Weight today is 3.8 kg, down from 4.3 kg at the 1-month visit. On exam, the infant is lethargic but arousable, MMM slightly dry, sunken fontanelle, and there is a palpable olive in the epigastrium after NG decompression. Labs: Na 134, K 3.1, Cl 90, HCO3 34, pH 7.51 — consistent with hypochloremic, hypokalemic metabolic alkalosis. Pyloric US: muscle thickness 4.5 mm, channel length 18 mm. Plan: admit, NPO, NG decompression, resuscitation with NS bolus 20 mL/kg then D5 ½ NS + 20 KCl at 1.5× maintenance, serial electrolytes, surgery when HCO3 < 30 and Cl > 100."
"[Name] is a 14-month-old previously healthy male brought in by parents for 12 hours of intermittent severe abdominal pain. Parents describe episodes every 15–20 minutes where the child draws his knees to his chest, cries inconsolably for 2–3 minutes, then appears fine between episodes. He has vomited twice (non-bilious) and passed one stool with "red jelly-like" material this morning. No fever. Recent viral URI 1 week ago. On exam he is sleepy between episodes, abdomen soft, mildly tender RUQ with a possible sausage-shaped mass. Ultrasound shows a target sign in the right mid-abdomen consistent with ileocolic intussusception. Plan: NPO, IV access, labs, surgery consult available at bedside, air contrast enema reduction."
"[Name] is an 11-year-old previously healthy female with 24 hours of progressively worsening abdominal pain that began periumbilically and has now migrated to the right lower quadrant. She has had two episodes of non-bilious emesis, complete anorexia, and a low-grade fever to 38.2°C. No diarrhea, no dysuria, no vaginal discharge (pre-menarchal). On exam she is uncomfortable, walks hunched over, has focal tenderness to palpation at McBurney's point with voluntary guarding, positive Rovsing's sign, and rebound tenderness. WBC 14.8 with left shift, CRP 4.2, UA negative, pregnancy test negative. Ultrasound: non-compressible appendix measuring 9 mm with periappendiceal fluid. PAS 8. Plan: NPO, IV fluids, ceftriaxone/metronidazole, laparoscopic appendectomy."
"[Name] is a 6-month-old ex-32-week preterm male (current corrected GA 54 weeks, current weight 6.2 kg) referred by his pediatrician for a right inguinal bulge first noted by mother 2 weeks ago during diaper changes, particularly with crying and straining. The bulge reduces spontaneously when the infant is calm and has not caused feeding intolerance, vomiting, or irritability. No prior episodes of incarceration. On exam, a reducible right inguinal hernia is easily demonstrated with gentle abdominal pressure, testes are descended bilaterally, no left-sided hernia appreciated. Plan: schedule right open inguinal hernia repair with contralateral exploration discussed; parents counseled on signs of incarceration and when to return to the ED."
"[Name] is a 12-month-old full-term male referred for a non-palpable left testis noted at his well-child visits since birth. The right testis is fully descended. No other GU anomalies. Pregnancy and delivery were unremarkable. Family history is negative for testicular malignancy or infertility. On exam, the right testis is in the scrotum, normal size. The left hemiscrotum is empty, flat, and no testis is palpable in the inguinal canal or ectopic positions. Plan: diagnostic laparoscopy with either primary laparoscopic orchiopexy or staged Fowler-Stephens, depending on intraoperative findings. Risks, benefits, alternatives, and the possibility of an absent or atrophic nubbin were discussed with parents."
"[Name] is a 3-week-old ex-27-week female (CGA 30 weeks, current weight 1.1 kg) admitted to the NICU since birth for prematurity, now on full enteral feeds of 20 cal/oz human milk via OG tube. Overnight, the infant developed increased abdominal distension, three large-volume gastric residuals, one grossly bloody stool, and two apneic episodes. Exam: firm, distended, shiny abdomen with erythema of the periumbilical wall and decreased bowel sounds. Labs: WBC 4.2 (from 11), platelets 78 (from 220), lactate 4.1, metabolic acidosis. AXR: pneumatosis intestinalis in the RLQ and portal venous gas. Modified Bell Stage IIB. Plan: NPO, OG to LIS, broad-spectrum antibiotics (ampicillin, gentamicin, metronidazole), fluid resuscitation, PRBC and platelet transfusion, serial AXR every 6 hours, surgical team at bedside for ongoing reassessment."
Pediatric surgery lives at an unusual intersection of the extremes of medicine. You will see a 500-gram neonate in the NICU whose life depends on a correctly placed peritoneal drain, and later the same day see a healthy 15-year-old with an umbilical hernia. You will translate a frightened parent's words into a precise clinical note. You will document consent conversations that matter legally and emotionally. You will hear bilious emesis in an infant and know, before the attending says anything, that the next page will go to the OR.
The best scribes in this specialty know their weights, their doses, their developmental language, and their congenital anomalies cold. They pre-populate notes with the birth history and growth percentiles. They recognize VACTERL and trisomy 21 associations before they are mentioned. They never chart pounds. They write "bilious" or "non-bilious" and never just "vomiting." They document NAT findings with forensic precision. They know which studies to pre-load: ultrasound for pyloric stenosis, UGI for malrotation, contrast enema for Hirschsprung, and HIDA for biliary atresia.
Welcome to pediatric surgery. You will see medicine that adult services never encounter, and the patients you help today will carry that benefit for the next eighty years.
28 References & Sources
Clinical Practice Guidelines
Kolon TF, et al. Evaluation and Treatment of Cryptorchidism: AUA Guideline. J Urol. 2014.
APSA Consensus on Management of Infantile Hypertrophic Pyloric Stenosis. J Pediatr Surg. 2012.
Landmark Resources
Coran AG, Adzick NS, et al. Pediatric Surgery (7th ed). Elsevier. 2012.
Holcomb GW, Murphy JP, St. Peter SD. Holcomb and Ashcraft's Pediatric Surgery (7th ed). Elsevier. 2019.
Children's Oncology Group (COG) Soft Tissue Sarcoma, Renal Tumor, and Neuroblastoma Committee protocols (current active protocol series).
Diagram & Figure Sources
Figure 1: Gastroschisis. CDC/Public Health Image Library, Wikimedia Commons. Public domain.
Figure 2: Omphalocele. CDC/Public Health Image Library, Wikimedia Commons. Public domain.
Figure 3: Pyloric stenosis. Wikimedia Commons. Public domain.
Figure 4: Hirschsprung disease. Wikimedia Commons. Public domain.
Figure 5: Intussusception. Wikimedia Commons. CC BY-SA.