Pulmonology / Critical Care
Every diagnosis, procedure, medication, classification, and documentation framework a scribe needs to succeed in pulmonary and critical care medicine.
All diagrams on this page are sourced from published educational or institutional materials (OpenStax, Wikimedia Commons, Blausen Medical, Gray's Anatomy) rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 The Respiratory System — Anatomy & Physiology
Pulmonology covers the airways, lung parenchyma, pleura, pulmonary vasculature, mediastinum, chest wall, respiratory muscles, and the neural control of breathing. Critical care adds multi-organ failure management layered on top of respiratory failure. A scribe who understands the basic architecture of the airway tree, the gas exchange unit, and the mechanics of ventilation can follow almost any pulmonary clinic or ICU discussion.
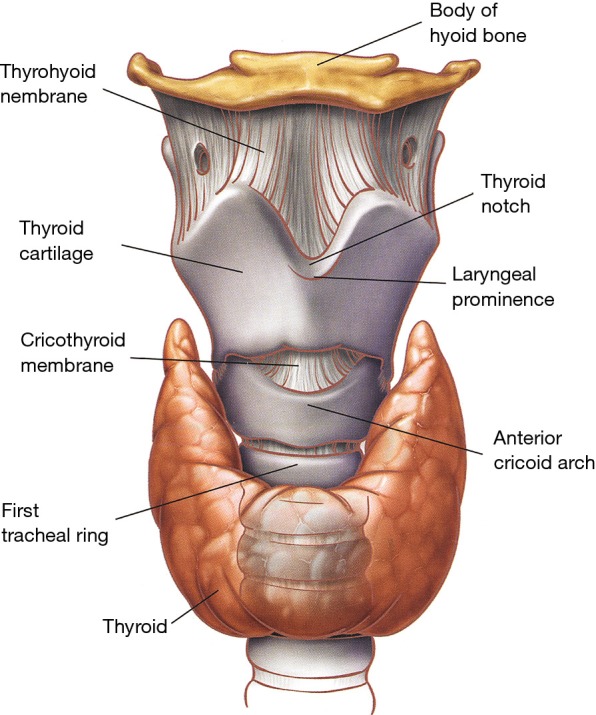
Conducting Airways & Gas Exchange Unit
Air enters through the nose and mouth, passes the pharynx and larynx, and enters the trachea. The trachea bifurcates at the carina (roughly T4-T5) into the right and left main bronchi. The right main bronchus is wider, shorter, and more vertical — which is why aspirated material and misplaced endotracheal tubes preferentially go right. Main bronchi divide into lobar bronchi (three on the right: RUL, RML, RLL; two on the left: LUL which includes the lingula, and LLL), then into segmental and subsegmental bronchi, down through roughly 23 generations of branching to the terminal bronchioles and respiratory bronchioles, which lead to the alveolar ducts and alveoli. Gas exchange happens across the thin alveolar-capillary membrane, where oxygen diffuses into pulmonary capillary blood and carbon dioxide diffuses out.
Pulmonary Circulation & Gas Exchange
The right ventricle pumps deoxygenated blood through the pulmonary artery, which branches with the bronchial tree out to the pulmonary capillaries surrounding the alveoli. Oxygenated blood returns through pulmonary veins to the left atrium. Pulmonary circulation is a low-pressure, high-compliance system; the normal mean pulmonary artery pressure is 14 mmHg, and values above 20 mmHg at rest define pulmonary hypertension. Hypoxic pulmonary vasoconstriction (HPV) is a unique reflex where poorly ventilated alveoli trigger local vasoconstriction to shunt blood away and preserve V/Q matching — but chronic hypoxia causes diffuse vasoconstriction and eventual pulmonary hypertension.
Mechanics of Breathing
The diaphragm is the primary muscle of inspiration. Accessory muscles (sternocleidomastoid, scalenes, intercostals) activate when respiratory work increases. Expiration is normally passive (elastic recoil) but becomes active in obstructive disease. Key physiologic relationships to know: compliance (change in volume per change in pressure — low in ILD/ARDS), resistance (opposition to flow — high in asthma/COPD), and the V/Q ratio (matching of ventilation to perfusion; mismatching is the dominant cause of hypoxemia in most lung disease).
Memorize the five causes of hypoxemia: V/Q mismatch (most common — PNA, PE, COPD), shunt (ARDS, pulmonary edema — does not correct with 100% O2), hypoventilation (opioids, neuromuscular, OHS), diffusion limitation (ILD, emphysema), and low inspired FiO2 (altitude). When the attending says "this is shunt physiology," you know they mean blood is passing alveoli that are not ventilated at all.
02 Scribe Documentation Framework
The SOAP Note in Pulmonology
Pulmonary clinic notes and ICU progress notes both follow a SOAP-style skeleton, but the content differs. Clinic encounters emphasize symptom chronology, functional capacity, and PFT/imaging trends. ICU notes are system-based and often written as "by-system" progress notes with numeric data dominating.
CC: Classic chief complaints include "shortness of breath," "chronic cough," "wheezing," "abnormal chest imaging," "follow-up lung nodule," or "sleep study results review."
HPI: Always capture dyspnea characterization (at rest vs exertional, level of exertion that provokes it, orthopnea, paroxysmal nocturnal dyspnea), cough (productive vs dry, sputum color and quantity, blood-tinged or frank hemoptysis), wheeze, chest tightness, and functional capacity (flights of stairs, blocks walked, activities limited). Quantify with mMRC dyspnea scale or CAT scores when possible.
ROS: Constitutional (weight loss, fevers, night sweats — red flags for TB and malignancy), ENT (sinus symptoms, post-nasal drip), cardiac (orthopnea, PND, edema), GI (reflux — a common asthma trigger), neurologic (daytime sleepiness for OSA workup).
PMHx/SHx: Smoking history in pack-years (packs/day × years) is mandatory on every pulmonary patient. Also document vaping, marijuana, occupational exposures (asbestos, silica, coal, birds, molds, metals), biomass fuel exposure, TB exposure, travel, pets, and family history of lung disease.
Vitals: Respiratory rate, SpO2 on room air (and on supplemental O2 with flow rate), temperature, heart rate, blood pressure. Document whether the patient is on home O2 and at what liter flow.
Exam: General appearance (in distress, tripoding, pursed-lip breathing, accessory muscle use), chest inspection (barrel chest, AP diameter, scars), palpation (tactile fremitus, chest expansion), percussion (resonant, dull, hyperresonant), auscultation (breath sounds in all fields, wheezes, rhonchi, crackles, rub).
Data: PFT values (FEV1, FVC, FEV1/FVC ratio, TLC, DLCO, % predicted), ABG (pH, PaO2, PaCO2, HCO3, A-a gradient), chest imaging findings, 6-minute walk distance with pre/post SpO2, polysomnography indices (AHI, ODI, nadir SpO2).
Pulmonary/CCM notes almost always use a problem-based plan. Number each problem (e.g., 1. Acute hypoxemic respiratory failure; 2. Community-acquired pneumonia; 3. Atrial fibrillation; 4. Acute kidney injury). Under each, document current assessment, diagnostics ordered, treatment, and decision points. In the ICU, the note is often written by system: Neuro, CV, Pulm, GI, Renal, ID, Heme, Endo, Prophylaxis, Dispo.
When a patient is on supplemental oxygen, you must chart the exact delivery device and flow rate: 2 L NC, 6 L simple mask, 40% venti mask, 40 L/min 60% HFNC, BiPAP 14/6 with 40% FiO2, ventilator settings (mode, TV, RR, PEEP, FiO2). "On oxygen" is not acceptable documentation in pulmonary medicine.
03 Asthma Obstructive
Asthma is a chronic inflammatory airway disease characterized by reversible bronchoconstriction, airway hyperresponsiveness, and airway remodeling. It affects about 8% of US adults and is one of the top three diagnoses in any pulmonary clinic.
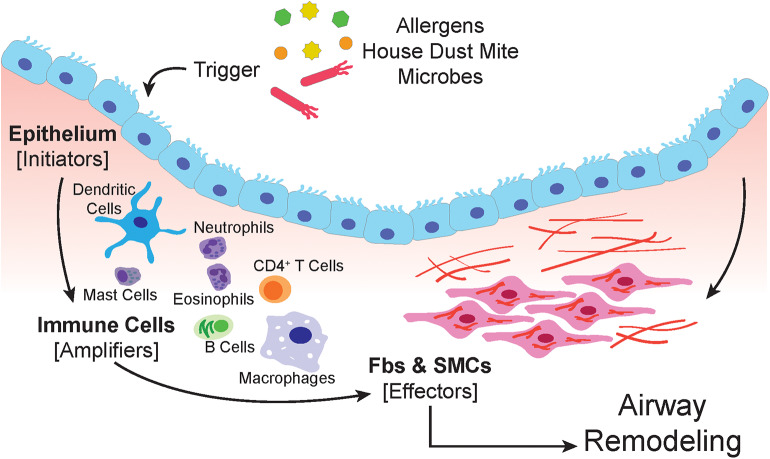
Pathophysiology & Presentation
Type 2 (Th2) inflammation dominates the typical asthma phenotype. Eosinophils, IgE, mast cells, IL-4, IL-5, and IL-13 drive airway edema, mucus plugging, and bronchoconstriction. Patients present with episodic wheezing, cough (especially nocturnal or post-exercise), chest tightness, and dyspnea. Triggers include upper respiratory infections, allergens (dust mites, pets, pollen, mold), exercise, cold air, tobacco smoke, occupational exposures, aspirin/NSAIDs (AERD), and GERD.
Diagnosis & Control Assessment
Diagnosis requires demonstrating variable expiratory airflow limitation. Spirometry showing an FEV1/FVC ratio below the lower limit of normal with a bronchodilator reversibility of ≥12% and ≥200 mL rise in FEV1 is classic. When PFTs are normal, a methacholine challenge with a PC20 ≤8 mg/mL or exercise challenge can be diagnostic. FeNO >25 ppb suggests eosinophilic airway inflammation.
| Step | Preferred Controller | Reliever |
|---|---|---|
| 1 | As-needed low-dose ICS-formoterol | ICS-formoterol PRN |
| 2 | Daily low-dose ICS or PRN ICS-formoterol | ICS-formoterol or SABA |
| 3 | Low-dose ICS-LABA | ICS-formoterol (MART) |
| 4 | Medium-dose ICS-LABA | ICS-formoterol (MART) |
| 5 | High-dose ICS-LABA + add-on (tiotropium, anti-IgE, anti-IL5/5R, anti-IL4Rα, anti-TSLP) | ICS-formoterol |
| Severity | Symptoms/Week | Night Awakenings | SABA Use | FEV1 |
|---|---|---|---|---|
| Intermittent | ≤2/wk | ≤2/mo | ≤2/wk | ≥80% |
| Mild Persistent | >2/wk, not daily | 3-4/mo | >2/wk | ≥80% |
| Moderate Persistent | Daily | >1/wk, not nightly | Daily | 60-80% |
| Severe Persistent | Throughout day | Often nightly | Several/day | <60% |
Management
Contemporary management follows the GINA 2024 Global Strategy for Asthma Management and Prevention, which recommends ICS-containing therapy for everyone with asthma (including step 1) and abandons SABA-only therapy because of mortality signals. Severe asthma with a Type 2 high phenotype (eosinophils, elevated FeNO, IgE-mediated) is treated with biologics: omalizumab (anti-IgE), mepolizumab/reslizumab/benralizumab (anti-IL5/IL5R), dupilumab (anti-IL4Rα), and tezepelumab (anti-TSLP — works regardless of phenotype).
Asthma Exacerbation
Acute exacerbations present with worsening dyspnea, wheeze, cough, and chest tightness, often with a URI trigger. ED/hospital management: oxygen to SpO2 93-95%, nebulized albuterol (SABA) plus ipratropium, systemic corticosteroids (prednisone 40-50 mg PO daily × 5 days or methylprednisolone IV), and magnesium sulfate IV for severe cases. Intubation is reserved for impending respiratory failure.
A "quiet chest" in a severe asthma exacerbation is ominous — it means airflow is so reduced that no wheeze is audible. Document the transition from wheezing to silent chest, worsening mental status, rising CO2, and accessory muscle use. This is the patient who is about to require intubation.
04 COPD & Asthma-COPD Overlap Obstructive
COPD is the third leading cause of death worldwide. It is a chronic, progressive, largely irreversible airflow limitation caused by chronic inflammation of the airways and destruction of lung parenchyma, overwhelmingly due to cigarette smoking (though biomass fuel exposure, occupational exposures, and alpha-1 antitrypsin deficiency also cause it).
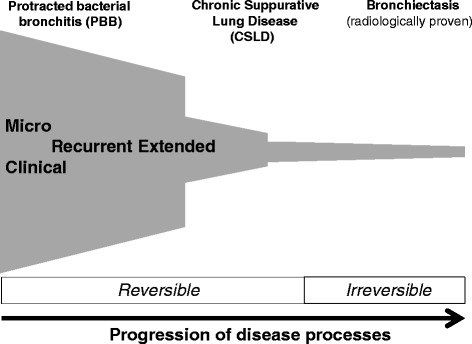
Pathophysiology & Phenotypes
Two overlapping processes occur. Chronic bronchitis is clinically defined as productive cough for at least 3 months in each of 2 consecutive years; pathologically it is goblet cell hyperplasia, mucus hypersecretion, and small airway inflammation. Emphysema is the permanent destruction of alveolar walls distal to the terminal bronchioles, producing enlarged airspaces, loss of elastic recoil, and loss of surface area for gas exchange. Centrilobular emphysema (upper lobe predominant) is the smoking-related pattern; panacinar emphysema (lower lobe predominant) is classic for alpha-1 antitrypsin deficiency.
Diagnosis
Diagnosis requires spirometry demonstrating a post-bronchodilator FEV1/FVC ratio <0.70. Severity is then graded by FEV1 % predicted (GOLD 1-4), and the overall assessment incorporates symptoms (mMRC or CAT) and exacerbation history (the ABE groups, which replaced the older ABCD grouping in GOLD 2023).
| GOLD Stage | FEV1 % Predicted | Severity |
|---|---|---|
| GOLD 1 | ≥80% | Mild |
| GOLD 2 | 50-79% | Moderate |
| GOLD 3 | 30-49% | Severe |
| GOLD 4 | <30% | Very severe |
| Group | Exacerbations (past year) | Symptoms (mMRC/CAT) |
|---|---|---|
| A | 0 or 1 (not leading to hospitalization) | mMRC 0-1 or CAT <10 |
| B | 0 or 1 (not leading to hospitalization) | mMRC ≥2 or CAT ≥10 |
| E | ≥2 moderate OR ≥1 hospitalization | Any |
Management
Management follows the 2024 GOLD Report. Core interventions: smoking cessation (the only intervention that alters natural history), vaccinations (flu, COVID, pneumococcal, RSV, Tdap), pulmonary rehabilitation (improves dyspnea and quality of life), and inhaler therapy escalated by group. Group A gets a bronchodilator; Group B gets LABA+LAMA; Group E gets LABA+LAMA with ICS added if blood eosinophils ≥300 or frequent exacerbations. Long-term supplemental oxygen is indicated for resting hypoxemia (PaO2 ≤55 or SpO2 ≤88%) and reduces mortality.
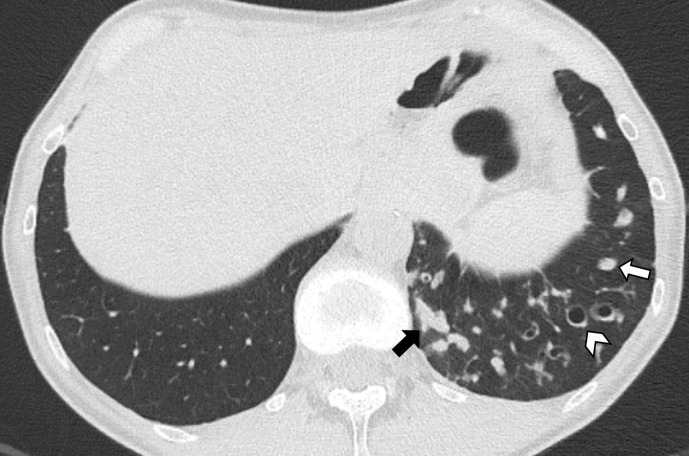
Acute Exacerbation of COPD (AECOPD)
Exacerbations are defined by worsening dyspnea, increased sputum volume, and increased sputum purulence (Anthonisen criteria). Treatment: short-acting bronchodilators (albuterol + ipratropium nebs), systemic steroids (prednisone 40 mg PO × 5 days), antibiotics when 2 of 3 Anthonisen criteria or requiring mechanical ventilation (azithromycin, doxycycline, or amoxicillin-clavulanate), and NIV (BiPAP) for hypercapnic respiratory failure, which reduces mortality and intubation rates.
Asthma-COPD Overlap (ACO)
Some patients have features of both — typically older smokers with an atopic history and partially reversible obstruction. GINA/GOLD no longer treat ACO as a single entity; instead, identify whether Type 2 inflammation (eosinophils, atopy) dominates and include ICS early. Documentation should describe both components rather than using the ACO label alone.
When a COPD patient is admitted with an exacerbation, always chart the home oxygen baseline, the number of exacerbations in the last year, prior ICU admissions or intubations, and the current inhaler regimen. These drive both risk stratification and discharge planning.
05 Bronchiectasis & Cystic Fibrosis Obstructive
Bronchiectasis is the permanent, pathological dilation of bronchi caused by chronic inflammation and airway wall destruction, usually from a vicious cycle of infection and inflammation. CT imaging shows dilated airways with bronchial wall thickening, "tram tracks," and "signet rings" (bronchus larger than its accompanying artery).
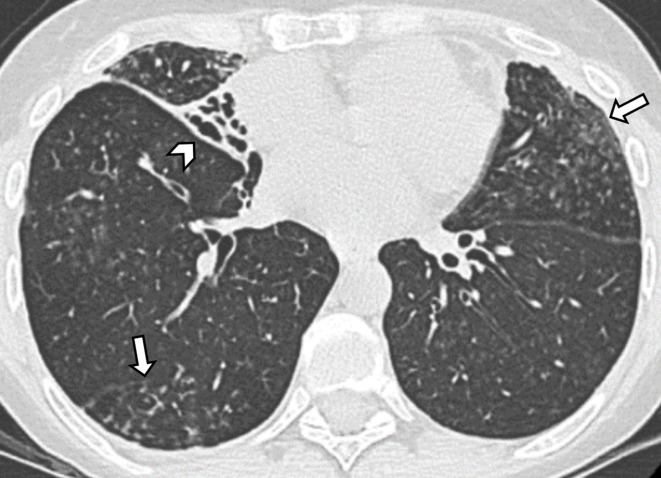
Etiologies
Bronchiectasis has dozens of causes: post-infectious (TB, NTM, severe pneumonia in childhood), cystic fibrosis, primary ciliary dyskinesia, immunodeficiency (CVID, hypogammaglobulinemia), ABPA, alpha-1 antitrypsin deficiency, rheumatologic disease (RA, Sjogren), aspiration, and idiopathic. The workup includes CF genetic testing, immunoglobulins, HIV, ABPA testing (total IgE, Aspergillus IgE), alpha-1 levels, rheum serologies, and sputum cultures (bacterial, mycobacterial, fungal).
Presentation & Management
Patients present with chronic productive cough (often copious purulent sputum), recurrent respiratory infections, hemoptysis, and progressive dyspnea. Management centers on airway clearance (oscillating PEP devices, high-frequency chest wall oscillation vest, nebulized hypertonic saline), treatment of exacerbations with targeted antibiotics (based on sputum cultures — Pseudomonas is common), chronic suppression (inhaled tobramycin or aztreonam for Pseudomonas, macrolide three-times-weekly for exacerbation prevention), and treatment of the underlying cause.
Cystic Fibrosis
CF is an autosomal recessive disease caused by CFTR mutations (most commonly ΔF508), leading to defective chloride and bicarbonate transport. Thick, dehydrated airway secretions drive chronic infection (Pseudomonas, Staph aureus, Burkholderia, NTM), bronchiectasis, pancreatic insufficiency, CF-related diabetes, and infertility. Modern management includes CFTR modulators — ivacaftor, lumacaftor/ivacaftor, tezacaftor/ivacaftor, and elexacaftor/tezacaftor/ivacaftor (Trikafta, a triple combination that has dramatically improved outcomes in patients with at least one F508del mutation). Airway clearance, nebulized dornase alfa, inhaled hypertonic saline, pancreatic enzyme replacement, and aggressive treatment of pulmonary exacerbations remain core therapies.
06 Interstitial Lung Disease Restrictive
ILD is an umbrella term for >200 heterogeneous disorders that share inflammation and fibrosis of the lung interstitium — the space between alveolar walls and capillaries. The result is restrictive physiology (reduced TLC and FVC), decreased DLCO, and hypoxemia especially with exertion.
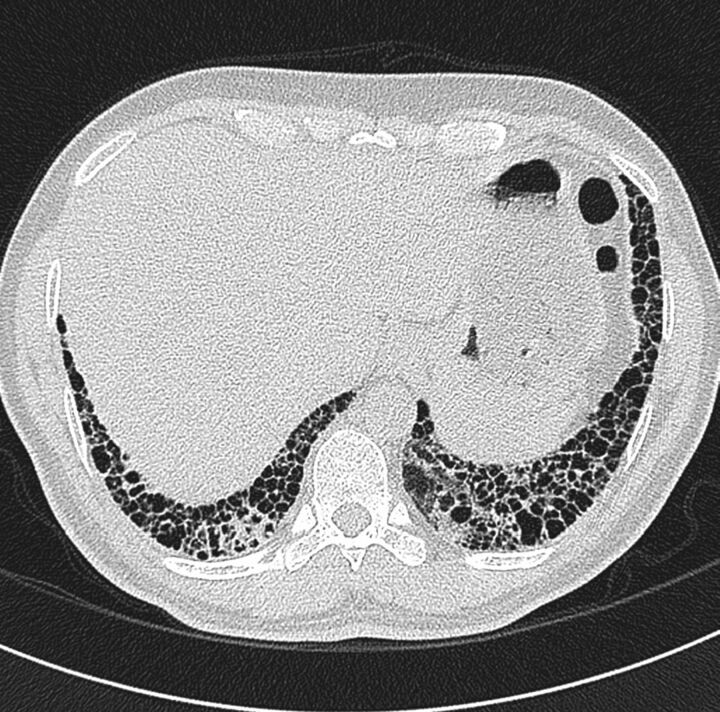
Major ILD Categories
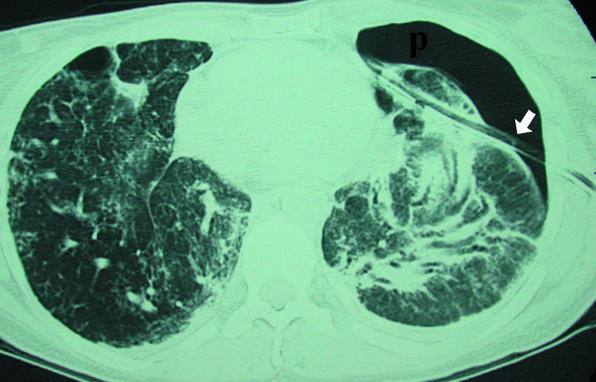
The prototypical progressive fibrotic ILD. Usually affects men over 60 with a smoking history; presents with progressive exertional dyspnea, dry cough, and bibasilar "velcro" crackles with clubbing. HRCT shows a usual interstitial pneumonia (UIP) pattern: peripheral, basal, reticular opacities with honeycombing and traction bronchiectasis. Diagnosis follows the 2022 ATS/ERS/JRS/ALAT IPF Clinical Practice Guideline. Treatment: antifibrotics (pirfenidone or nintedanib) slow progression; no therapy reverses fibrosis. Lung transplant is the only curative option. Median survival untreated ~3-5 years.
More inflammation than fibrosis on biopsy. Cellular NSIP responds well to immunosuppression; fibrotic NSIP behaves more like IPF. Strongly associated with connective tissue disease (scleroderma, polymyositis/dermatomyositis, Sjogren). CT shows bilateral, symmetric, basal-predominant ground glass with minimal honeycombing.
Immune-mediated reaction to inhaled antigens (bird droppings, mold, hot tub mycobacteria, farming antigens). Can be acute, subacute, or chronic fibrotic. HRCT shows upper/mid lung centrilobular ground glass nodules, mosaic attenuation, and air trapping on expiration. Treatment: antigen avoidance is essential, plus corticosteroids; chronic fibrotic HP may get antifibrotics.
Non-caseating granulomatous disease of unknown etiology with a predilection for young Black adults and middle-aged Scandinavians. Bilateral hilar lymphadenopathy is the classic finding. Scadding stages 0-IV stage the CXR appearance. Extrapulmonary involvement is common (skin, eye, cardiac, neuro). Workup: EBUS-TBNA of mediastinal nodes for tissue diagnosis, serum ACE, calcium, EKG. Treatment: observation for asymptomatic disease, corticosteroids for symptomatic or organ-threatening involvement, steroid-sparing agents (methotrexate, azathioprine) for refractory cases, per ATS sarcoidosis guidelines.
RA, scleroderma, polymyositis/dermatomyositis, MCTD, Sjogren, and SLE can all cause ILD. Scleroderma-ILD is the most common (NSIP pattern typically). Workup includes ANA, RF, anti-CCP, ENA panel, anti-Scl70, anti-Jo1, and MDA5. Treatment is immunosuppression tailored to the underlying disease, and nintedanib is approved for progressive pulmonary fibrosis regardless of underlying etiology.
| Stage | Chest X-Ray Findings |
|---|---|
| 0 | Normal CXR |
| I | Bilateral hilar lymphadenopathy (BHL) only |
| II | BHL + parenchymal infiltrates |
| III | Parenchymal infiltrates without BHL |
| IV | Pulmonary fibrosis |
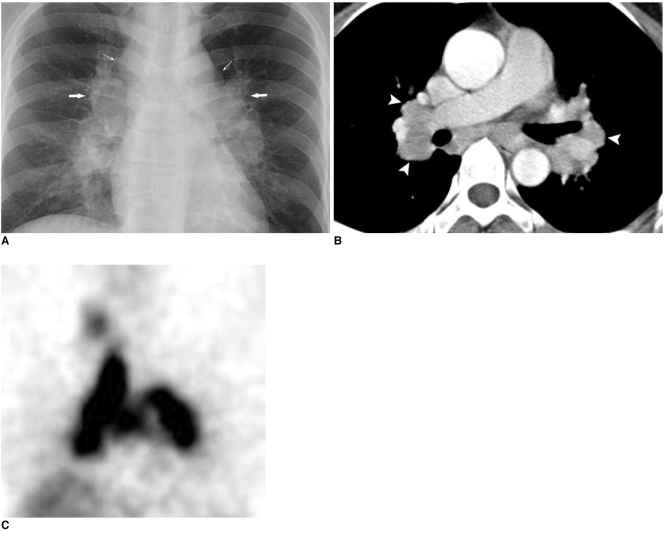
Workup
Every ILD patient needs: full PFTs with DLCO, HRCT with prone and expiratory views, 6-minute walk test, autoimmune serologies, HP precipitins and detailed environmental history, and sometimes surgical lung biopsy (VATS) or transbronchial cryobiopsy for definitive diagnosis. All cases should be discussed at a multidisciplinary ILD conference (MDD).
07 Pulmonary Hypertension Vascular
Pulmonary hypertension (PH) is defined as a mean pulmonary artery pressure (mPAP) >20 mmHg at rest by right heart catheterization. The WHO clinical classification organizes PH into five groups based on mechanism, because treatment differs radically between them.
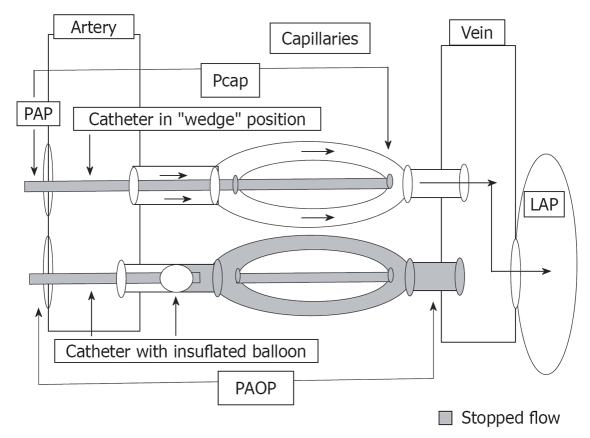
| Group | Category | Examples |
|---|---|---|
| 1 | Pulmonary arterial hypertension (PAH) | Idiopathic, heritable, drug-induced, CTD (scleroderma), HIV, portopulmonary, congenital heart disease |
| 2 | PH due to left heart disease | HFrEF, HFpEF, valvular disease (most common cause overall) |
| 3 | PH due to lung disease/hypoxia | COPD, ILD, OSA, OHS, high altitude |
| 4 | Chronic thromboembolic PH (CTEPH) | Unresolved organized thromboembolic obstruction |
| 5 | PH with unclear/multifactorial mechanisms | Sarcoidosis, hematologic disease, metabolic disease |
Workup & Management
Evaluation starts with echocardiography (elevated RVSP, RV dilation/dysfunction, septal flattening, tricuspid regurgitation), followed by V/Q scan (mandatory to exclude CTEPH), PFTs, overnight oximetry, HIV/hepatitis/autoimmune labs, and definitive right heart catheterization. Only Group 1 PAH is treated with PAH-targeted therapy: PDE5 inhibitors (sildenafil, tadalafil), endothelin receptor antagonists (bosentan, ambrisentan, macitentan), prostanoids (epoprostenol, treprostinil, iloprost, selexipag), and the soluble guanylate cyclase stimulator (riociguat, also approved for CTEPH). Group 2 is treated by optimizing left heart disease; Group 3 by treating underlying lung disease and hypoxia; Group 4 with pulmonary thromboendarterectomy or balloon pulmonary angioplasty.
08 Pulmonary Embolism & DVT Vascular
Pulmonary embolism is the obstruction of pulmonary arterial flow by a thrombus, almost always arising from a lower-extremity DVT. PE is the third most common cause of cardiovascular death.
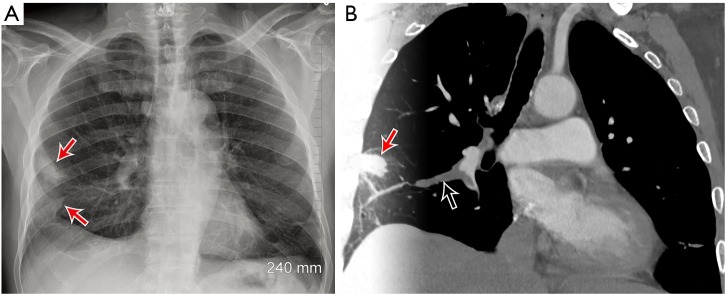
Presentation & Diagnosis
Classic presentation includes acute dyspnea, pleuritic chest pain, tachycardia, tachypnea, and hypoxemia; massive PE presents with hypotension or cardiac arrest. Hemoptysis, syncope, or unexplained RV failure on echo should also trigger PE workup. Initial risk stratification uses the Wells criteria and PERC rule; low probability gets D-dimer (age-adjusted cutoff: age × 10 ng/mL for patients >50), intermediate/high probability proceeds directly to CT pulmonary angiography (CTPA).
| Criterion | Points |
|---|---|
| Clinical signs of DVT | 3.0 |
| PE is #1 diagnosis (or equally likely) | 3.0 |
| Heart rate >100 | 1.5 |
| Immobilization ≥3 days or surgery in prior 4 weeks | 1.5 |
| Prior DVT/PE | 1.5 |
| Hemoptysis | 1.0 |
| Malignancy (treatment within 6 months) | 1.0 |
Interpretation: ≤4 = "PE unlikely" (D-dimer); >4 = "PE likely" (CTPA).
Classification & Management
PE is classified as massive (hemodynamically unstable — SBP <90), submassive/intermediate (stable but with RV strain on imaging and/or positive troponin/BNP), or low-risk. Per the CHEST 2021 VTE guidelines, anticoagulation is the cornerstone — DOACs (apixaban, rivaroxaban) are first-line for most patients; LMWH is preferred in cancer (though DOACs increasingly used); warfarin is reserved for specific settings (antiphospholipid syndrome, mechanical valves). Systemic thrombolysis (alteplase) is indicated for massive PE; catheter-directed thrombolysis or thrombectomy (EKOS, FlowTriever) is used for select intermediate-high-risk patients. IVC filters are reserved for patients with contraindications to anticoagulation.
When charting a PE, always document the PESI or simplified PESI score, RV strain on CT or echo, troponin and BNP, and whether the patient received thrombolysis, catheter-directed therapy, or anticoagulation alone. These drive disposition (floor vs ICU) and follow-up.
09 Pneumonia & Lower Respiratory Infection Infectious
Pneumonia is infection of the lung parenchyma. Classification is epidemiological (CAP, HAP, VAP, HCAP is largely abandoned) and influences likely pathogens and antibiotic choice.
Community-Acquired Pneumonia (CAP)
Most common pathogens: Streptococcus pneumoniae, Haemophilus influenzae, Mycoplasma pneumoniae, Chlamydia pneumoniae, Legionella, respiratory viruses (influenza, RSV, SARS-CoV-2). Presentation: fever, productive cough, pleuritic chest pain, dyspnea, and focal crackles or consolidation on exam. Diagnosis requires a chest X-ray or CT showing infiltrate. Severity is stratified with CURB-65 or Pneumonia Severity Index (PSI/PORT).
Confusion, Urea >7 mmol/L (BUN >19), Respiratory rate ≥30, Blood pressure (SBP <90 or DBP ≤60), age ≥65. Each criterion = 1 point.
0-1: outpatient. 2: short hospital stay or closely supervised outpatient. 3-5: hospitalize; 4-5 consider ICU.
Treatment follows the 2019 ATS/IDSA CAP guidelines. Outpatient healthy adults: amoxicillin, doxycycline, or a macrolide. Outpatients with comorbidities: beta-lactam plus macrolide or doxycycline, or respiratory fluoroquinolone (levofloxacin, moxifloxacin) monotherapy. Inpatient non-severe: beta-lactam + macrolide, or respiratory FQ. Inpatient severe/ICU: beta-lactam (ceftriaxone, ampicillin-sulbactam) + macrolide or FQ; add MRSA and Pseudomonas coverage only when specific risk factors are present.
Hospital-Acquired & Ventilator-Associated Pneumonia
HAP occurs ≥48 hours after admission; VAP ≥48 hours after intubation. Empirical coverage targets MDR gram-negatives (Pseudomonas, ESBL Enterobacterales) and MRSA: piperacillin-tazobactam or cefepime or meropenem, plus vancomycin or linezolid, with a second anti-pseudomonal agent in high-risk patients. De-escalate based on cultures.
Aspiration & Lung Abscess
Aspiration pneumonitis is chemical injury from gastric acid; aspiration pneumonia adds bacterial infection (mixed oropharyngeal flora, including anaerobes). Patients at risk: altered mental status, dysphagia, stroke, alcohol/drug use, seizures. Classic location: superior segment of the RLL (supine aspiration). Treat with ampicillin-sulbactam or ceftriaxone + metronidazole; lung abscess often needs prolonged antibiotics.
10 Tuberculosis & NTM Infectious
TB is caused by Mycobacterium tuberculosis. Latent TB infection (LTBI) is asymptomatic infection detected by IGRA (QuantiFERON, T-SPOT) or tuberculin skin test; active TB is symptomatic disease with cough >2-3 weeks, hemoptysis, night sweats, weight loss, fevers, and often cavitary upper-lobe disease on imaging. Diagnosis requires sputum AFB smear, mycobacterial culture, and NAAT (Xpert MTB/RIF). Patients are placed in airborne isolation.
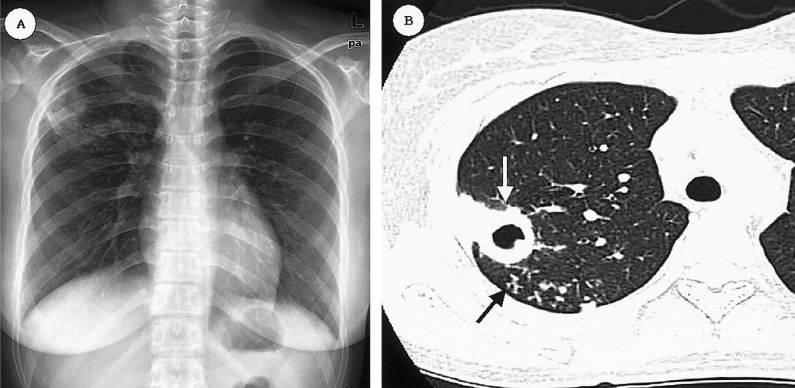
Treatment of active pulmonary TB: the RIPE regimen — rifampin, isoniazid, pyrazinamide, ethambutol — for 2 months, then rifampin + isoniazid for 4 additional months. Drug-resistant TB (MDR, XDR) requires longer, more complex regimens. LTBI is treated with isoniazid × 9 months, rifampin × 4 months, or 3HP (isoniazid + rifapentine weekly × 12 weeks).
Non-tuberculous mycobacteria (NTM) — Mycobacterium avium complex (MAC), M. abscessus, M. kansasii — cause indolent pulmonary infection often in patients with underlying bronchiectasis (the "Lady Windermere syndrome" is MAC in older thin women with RML/lingular bronchiectasis). Treatment is a multi-drug macrolide-based regimen for 12+ months after culture conversion.
11 Pleural Disease Pleural
Pleural Effusion
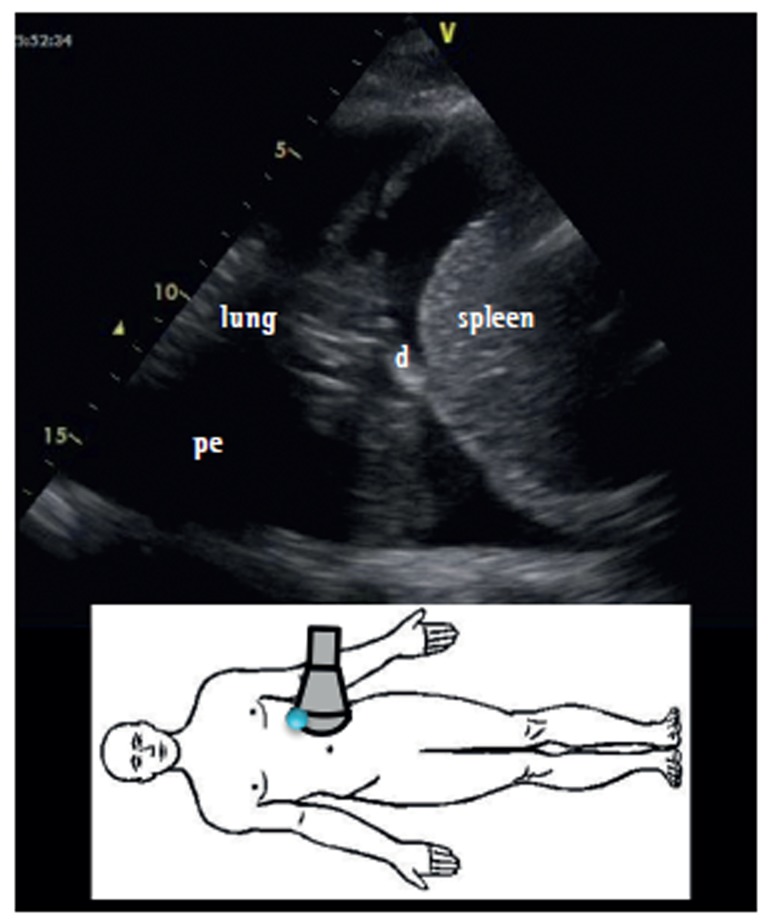
Fluid accumulation in the pleural space. Use Light's criteria to distinguish transudate from exudate — an effusion is exudative if any of: pleural protein/serum protein >0.5, pleural LDH/serum LDH >0.6, or pleural LDH >2/3 upper limit of normal serum LDH. Transudates (CHF, cirrhosis, nephrotic syndrome, atelectasis) arise from imbalance of hydrostatic/oncotic pressure. Exudates (pneumonia/parapneumonic, malignancy, PE, TB, rheumatologic) arise from inflammation or impaired lymphatic drainage. Thoracentesis samples are sent for cell count with differential, pH, glucose, LDH, protein, Gram stain and culture, cytology, and adenosine deaminase for TB.
Empyema & Complicated Parapneumonic Effusion
A parapneumonic effusion becomes complicated (pH <7.20, glucose <40, positive Gram stain) or frankly empyematous (pus) when bacterial infection invades the pleural space. These require chest tube drainage (≥14 Fr) and often intrapleural fibrinolytics (tPA + DNase) or surgical decortication (VATS) if loculated.
Pneumothorax
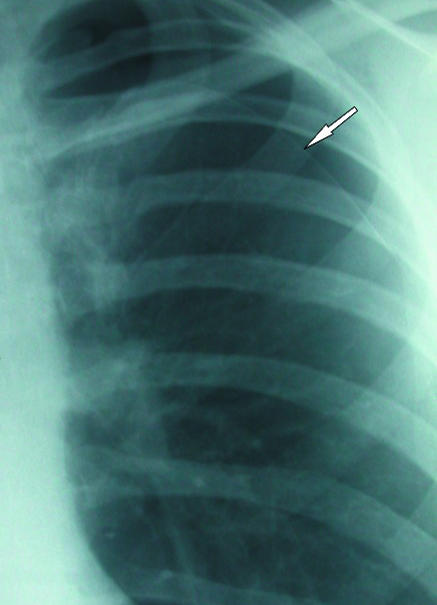
Air in the pleural space. Primary spontaneous pneumothorax (young tall thin smokers) and secondary spontaneous pneumothorax (COPD, CF, ILD) present with sudden chest pain and dyspnea. Traumatic and iatrogenic (after central line, thoracentesis, biopsy) are common in hospital settings. Tension pneumothorax is a clinical diagnosis (hypotension, tracheal deviation away from the side, absent breath sounds, distended neck veins) requiring immediate needle decompression followed by chest tube placement.
12 Lung Cancer & Pulmonary Nodules Oncologic
Lung cancer remains the leading cause of cancer death. The dominant histologic distinction is non-small cell lung cancer (NSCLC, ~85% — adenocarcinoma, squamous, large cell) versus small cell lung cancer (SCLC, ~15%). Workup: tissue diagnosis (bronchoscopy, EBUS-TBNA, CT-guided core biopsy), staging CT chest/abdomen/pelvis, PET-CT, brain MRI, and molecular testing on NSCLC adenocarcinoma for actionable alterations (EGFR, ALK, ROS1, BRAF, KRAS G12C, MET, RET, NTRK, HER2) plus PD-L1 expression.
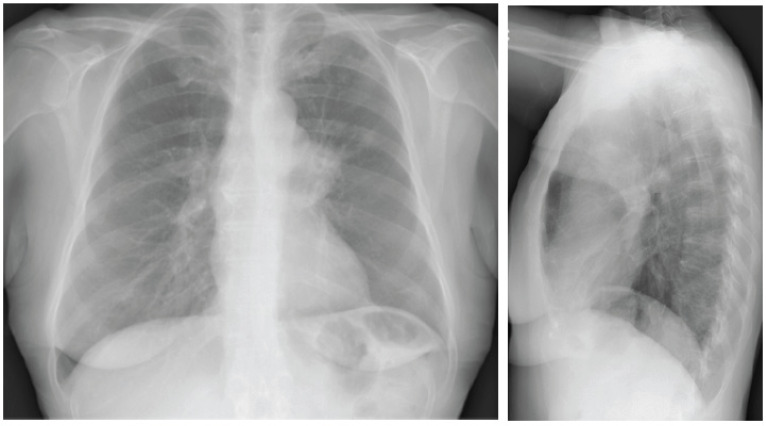
Screening
The USPSTF 2021 lung cancer screening recommendation is annual low-dose CT (LDCT) for adults age 50-80 with a 20+ pack-year smoking history who currently smoke or quit within the last 15 years.
Management
NSCLC early stage (I-II): surgical resection (lobectomy with mediastinal lymph node sampling) is the standard; SBRT for non-operable candidates. Stage III: multimodal therapy (concurrent chemoradiation followed by durvalumab consolidation). Stage IV: systemic therapy guided by biomarkers — targeted TKIs for driver mutations, immunotherapy (pembrolizumab, atezolizumab, nivolumab) often combined with chemotherapy, per NCCN NSCLC Guidelines. SCLC: limited stage gets concurrent chemoradiation and PCI; extensive stage gets platinum/etoposide + atezolizumab.
Solitary Pulmonary Nodule — Fleischner Society Guidelines
Incidental pulmonary nodules are extraordinarily common. Management depends on size, solid vs subsolid character, and patient risk.
| Nodule Size | Low Risk | High Risk |
|---|---|---|
| <6 mm | No routine follow-up | Optional CT at 12 months |
| 6-8 mm | CT at 6-12 months, then 18-24 months | CT at 6-12 months, then 18-24 months |
| >8 mm | CT at 3 months, PET-CT, or tissue | CT at 3 months, PET-CT, or tissue |
| Type | Size | Follow-Up |
|---|---|---|
| Ground glass | <6 mm | No routine follow-up |
| Ground glass | ≥6 mm | CT at 6-12 months, then every 2 years to 5 years |
| Part-solid | <6 mm | No routine follow-up |
| Part-solid | ≥6 mm | CT at 3-6 months; if stable and solid component <6 mm, annual CT × 5 years |
See the Fleischner Society 2017 recommendations for the complete algorithm.
13 Sleep-Disordered Breathing Sleep
Obstructive Sleep Apnea (OSA)
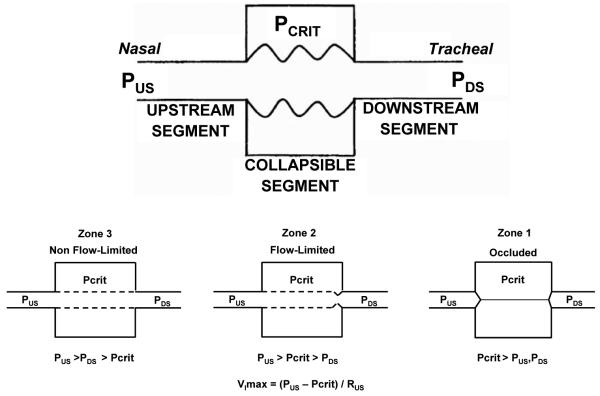
Repeated partial or complete upper airway collapse during sleep producing apneas, hypopneas, oxygen desaturation, and cortical arousals. Risk factors: obesity, male sex, neck circumference >17 inches (men) / >16 (women), retrognathia, large tonsils, family history. Symptoms: loud habitual snoring, witnessed apneas, gasping/choking arousals, nonrestorative sleep, morning headaches, excessive daytime sleepiness (quantify with Epworth Sleepiness Scale). Screening tools: STOP-BANG, Berlin questionnaire.
Diagnosis is by polysomnography (PSG) in-lab or home sleep apnea test (HSAT) for uncomplicated moderate-to-high probability adults without major comorbidities, per AASM OSA diagnosis guideline. Severity is graded by apnea-hypopnea index (AHI).
| Severity | AHI (events/hour) |
|---|---|
| Normal | <5 |
| Mild | 5-14 |
| Moderate | 15-29 |
| Severe | ≥30 |
Treatment: CPAP is first-line for moderate-to-severe OSA; alternatives include oral appliances (mandibular advancement devices), positional therapy, weight loss, upper airway surgery (uvulopalatopharyngoplasty, maxillomandibular advancement), and hypoglossal nerve stimulation (Inspire) in selected patients.
Central Sleep Apnea (CSA)
Apneas without respiratory effort, due to loss of central respiratory drive. Causes: heart failure (Cheyne-Stokes respiration), opioids, high altitude, stroke, brainstem lesions, idiopathic. Treatment: address the underlying cause; adaptive servo-ventilation (ASV) for CSA with normal EF, but contraindicated in HFrEF with EF ≤45%.
Obesity Hypoventilation Syndrome (OHS)
Defined by obesity (BMI ≥30), awake daytime hypercapnia (PaCO2 ≥45 mmHg), and sleep-disordered breathing — after excluding other causes of hypoventilation. Most have severe OSA. Treatment: PAP therapy (CPAP or BPAP), weight loss (including bariatric surgery), and careful O2 titration to avoid worsening hypercapnia.
14 ARDS & Mechanical Ventilation Critical Care
Acute Respiratory Distress Syndrome
ARDS is acute-onset hypoxemic respiratory failure due to increased pulmonary capillary permeability and non-cardiogenic pulmonary edema. Common triggers: pneumonia, sepsis, aspiration, trauma, pancreatitis, transfusion, drug reactions.
| Criterion | Requirement |
|---|---|
| Timing | Within 1 week of known insult or new/worsening respiratory symptoms |
| Imaging | Bilateral opacities on CXR/CT not fully explained by effusion, collapse, or nodules |
| Origin of edema | Not fully explained by cardiac failure or fluid overload (objective assessment, e.g., echo, if no risk factor) |
| Mild | PaO2/FiO2 200-300 on PEEP ≥5 |
| Moderate | PaO2/FiO2 100-200 on PEEP ≥5 |
| Severe | PaO2/FiO2 ≤100 on PEEP ≥5 |
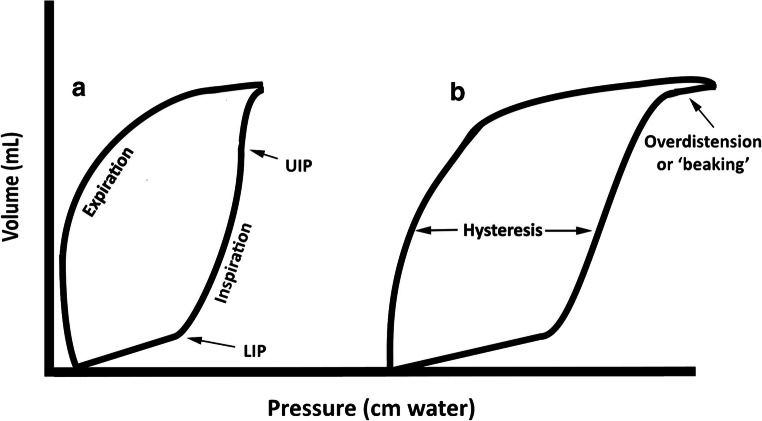
Management is supportive: lung-protective ventilation (tidal volume 4-6 mL/kg predicted body weight, plateau pressure <30 cmH2O, driving pressure <15 cmH2O), higher PEEP strategies, conservative fluid management, prone positioning for moderate-severe ARDS, neuromuscular blockade in selected severe cases, and ECMO as rescue therapy. Treat the underlying cause.
Mechanical Ventilation Basics
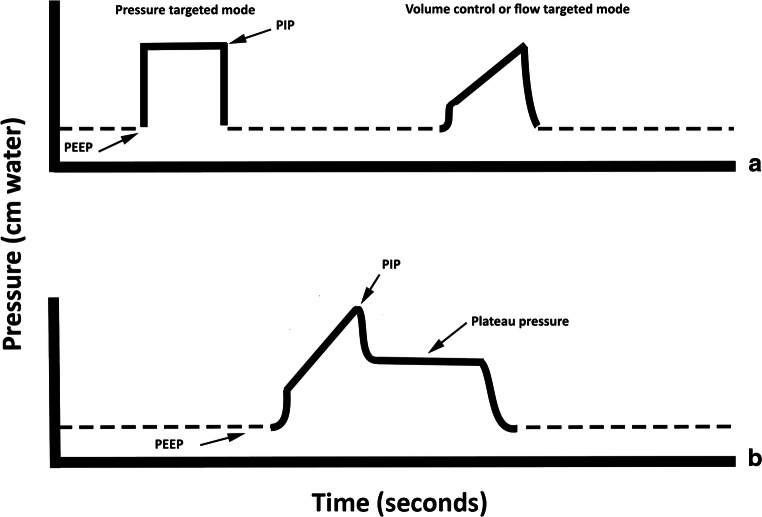
Scribes in the ICU must chart ventilator settings accurately. Know the modes and what each parameter means.
Mode: AC/VC (assist control volume control), PC (pressure control), PRVC (pressure-regulated volume control), SIMV, PSV (pressure support), CPAP, APRV. TV: tidal volume (mL). RR: respiratory rate (set and total). PEEP: positive end-expiratory pressure (cmH2O). FiO2: fraction of inspired oxygen (0.21-1.00). PIP: peak inspiratory pressure. Pplat: plateau pressure (static airway pressure). I:E ratio: inspiratory to expiratory ratio. PS: pressure support.
Document every change: "Vent settings adjusted from AC/VC TV 450, RR 18, PEEP 8, FiO2 0.50 to AC/VC TV 450, RR 20, PEEP 10, FiO2 0.60 for worsening hypoxemia; pH 7.30, PaCO2 50, PaO2 68."
Weaning follows a daily spontaneous breathing trial (SBT) when the patient meets readiness criteria (improving underlying process, adequate oxygenation on FiO2 ≤0.40 and PEEP ≤5-8, hemodynamically stable, able to initiate breaths). The RSBI (Rapid Shallow Breathing Index = RR/TV in liters) <105 predicts successful extubation.
15 Hemoptysis & Airway Emergencies Critical Care
Hemoptysis is expectoration of blood from the lower respiratory tract. Massive hemoptysis is variably defined (>100-600 mL in 24 hours or any volume causing hemodynamic or respiratory compromise) and is a true airway emergency — the problem is airway flooding and asphyxiation, not exsanguination.
Causes: bronchiectasis, bronchitis, TB, NTM, fungal infection (aspergilloma), lung cancer, pulmonary embolism with infarction, vasculitis (GPA, MPA, anti-GBM), mitral stenosis, AVMs, trauma. Initial management: protect airway (place the bleeding lung down, selective intubation of the non-bleeding side with a double-lumen or mainstem advancement), reverse coagulopathy, urgent CT angiography, bronchoscopy to localize, and bronchial artery embolization (BAE) by interventional radiology as definitive therapy in most cases.
16 Pulmonary Procedures Procedures
Scribes will document procedure notes for a range of bedside, bronchoscopic, and pleural interventions. Learn the core elements: indication, consent, time-out, technique, findings, specimens sent, estimated blood loss, complications, and post-procedure plan.
| Procedure | Description | Common Indications |
|---|---|---|
| Flexible bronchoscopy | Fiberoptic scope through nose/mouth/ETT into airway; inspection, BAL, brushings, biopsies | Hemoptysis, persistent infiltrate, foreign body, mucus plug, diagnosis of suspected infection or malignancy |
| Rigid bronchoscopy | Metal scope under general anesthesia; large working channel | Massive hemoptysis, central airway obstruction, foreign body, stent placement |
| EBUS-TBNA | Endobronchial ultrasound with transbronchial needle aspiration of mediastinal/hilar lymph nodes | Lung cancer staging, sarcoidosis diagnosis, mediastinal lymphadenopathy workup |
| Transbronchial biopsy | Peripheral lung tissue sampling via bronchoscope under fluoroscopy or navigation | ILD, peripheral nodules, transplant rejection surveillance |
| Transbronchial cryobiopsy | Larger lung biopsy using cryoprobe | ILD diagnosis when surgical biopsy is high-risk |
| Bronchoalveolar lavage (BAL) | Saline instilled and aspirated from subsegment | Infection workup (especially immunocompromised), ILD workup, alveolar hemorrhage |
| Navigational bronchoscopy / Robotic (Monarch, Ion) | Electromagnetic or robotic guidance to peripheral nodules | Diagnosis of peripheral pulmonary nodules |
| Thoracentesis | Needle or catheter drainage of pleural effusion | Diagnostic and therapeutic effusion drainage |
| Pleural biopsy | Closed needle (Abrams, Cope) or image-guided core biopsy of pleura | Suspected TB or malignant pleural disease |
| Medical thoracoscopy / pleuroscopy | Direct visualization of pleura through small incision under moderate sedation | Undiagnosed exudative effusion, talc pleurodesis |
| Chest tube (tube thoracostomy) | Pigtail (8-14 Fr) or large-bore (20-36 Fr) tube placed in pleural space | Pneumothorax, hemothorax, empyema, malignant effusion |
| Indwelling pleural catheter (PleurX) | Tunneled pleural catheter for recurrent malignant effusion | Recurrent malignant pleural effusion |
| Arterial blood gas (ABG) | Radial (most common), brachial, or femoral arterial puncture | Assessment of oxygenation, ventilation, acid-base |
| PFTs | Spirometry, lung volumes (plethysmography or helium dilution), DLCO, 6MWT | Diagnosis and monitoring of all chronic lung disease |
| Methacholine challenge | Inhalation of escalating methacholine doses with spirometry | Diagnosis of asthma when PFTs are normal |
| Polysomnography (PSG) | Overnight in-lab study with EEG, EOG, EMG, ECG, airflow, effort, SpO2 | Diagnosis of OSA, CSA, parasomnia, narcolepsy, PLMD |
| Home sleep apnea test (HSAT) | Portable multi-channel recording (airflow, effort, SpO2) | Uncomplicated suspected moderate-severe OSA |
| Lung biopsy (surgical / VATS) | Video-assisted thoracoscopic wedge biopsy | Definitive ILD diagnosis, peripheral nodules |
| LVRS / bullectomy | Lung volume reduction surgery removing emphysematous tissue; endobronchial valves (Zephyr, Spiration) as less invasive alternative | Severe upper-lobe emphysema with hyperinflation |
| Lung transplant | Single or bilateral lung transplantation | End-stage ILD, COPD, CF, PAH |
Bronchoscopy notes must document oxygen delivery and sedation (versed, fentanyl, propofol), topical anesthesia (lidocaine amount and concentration), each airway inspected (right/left main, each lobar, each segment), findings (mucosal appearance, secretions, lesions), samples taken and sent (BAL from which subsegment, brushings, biopsies, needle aspirations), and any complications.
17 PFTs, Imaging & Diagnostics Diagnostics
Pulmonary Function Tests
PFTs measure three things: spirometry (airflow), lung volumes (capacity), and diffusing capacity (gas transfer).
| Pattern | FEV1/FVC | FVC | TLC | DLCO |
|---|---|---|---|---|
| Obstructive (asthma, COPD) | <0.70 (or <LLN) | Normal or ↓ | Normal or ↑ | Normal (asthma), ↓ (emphysema) |
| Restrictive | Normal or ↑ | ↓ | ↓ | ↓ (parenchymal) / Normal (extraparenchymal) |
| Mixed | <0.70 | ↓ | ↓ | Variable |
| Isolated low DLCO | Normal | Normal | Normal | ↓ |
A bronchodilator response is positive if FEV1 or FVC improves by ≥12% AND ≥200 mL after albuterol. DLCO reflects alveolar-capillary gas transfer and is reduced in emphysema, ILD, PAH, and anemia (correct for hemoglobin). 6-minute walk test (6MWT) measures functional capacity; a drop in SpO2 ≥4% or below 88% is clinically significant.
ABG Interpretation
Normal values: pH 7.35-7.45, PaCO2 35-45, PaO2 80-100, HCO3 22-26, SaO2 >95%. The A-a gradient = PAO2 - PaO2; normal is approximately (age/4) + 4. A widened A-a gradient indicates V/Q mismatch, shunt, or diffusion limitation. Chart ABGs as: "pH/PaCO2/PaO2/HCO3/SaO2 on (room air or O2 device)."
Chest Imaging
CXR: cheap and fast; limited sensitivity. Look at lines (mediastinum, silhouettes), bones, breathing (lung fields), and soft tissues. CT chest without contrast: parenchymal disease, nodules, ILD. HRCT: thin-slice, used for ILD pattern recognition and bronchiectasis. CTPA: PE protocol with bolus contrast. PET-CT: cancer staging and nodule characterization (SUV >2.5 is suspicious). V/Q scan: PE when CTPA is contraindicated; CTEPH evaluation.
18 Medications You Must Know Meds
Inhaled Therapies
| Class | Generic (Brand) | Use |
|---|---|---|
| SABA | Albuterol (ProAir, Ventolin, Proventil); levalbuterol (Xopenex) | Rescue bronchodilator |
| SAMA | Ipratropium (Atrovent) | Acute COPD/asthma adjunct |
| LABA | Salmeterol (Serevent); formoterol; olodaterol | Maintenance bronchodilator (always with ICS in asthma) |
| LAMA | Tiotropium (Spiriva); umeclidinium; glycopyrrolate; aclidinium | Maintenance bronchodilator in COPD; add-on in severe asthma |
| ICS | Fluticasone, budesonide, beclomethasone, mometasone, ciclesonide | Controller for asthma; COPD with frequent exacerbations or high eosinophils |
| ICS-LABA | Fluticasone-salmeterol (Advair), budesonide-formoterol (Symbicort), fluticasone-vilanterol (Breo), mometasone-formoterol (Dulera) | Asthma and COPD maintenance |
| LABA-LAMA | Olodaterol-tiotropium (Stiolto), vilanterol-umeclidinium (Anoro), formoterol-glycopyrrolate (Bevespi), indacaterol-glycopyrrolate (Utibron) | COPD maintenance |
| ICS-LABA-LAMA (triple) | Fluticasone-umeclidinium-vilanterol (Trelegy), budesonide-glycopyrrolate-formoterol (Breztri) | COPD with high exacerbation risk; severe asthma |
Oral & Systemic Therapies
| Class | Examples | Use |
|---|---|---|
| LTRA | Montelukast (Singulair), zafirlukast | Asthma add-on; allergic rhinitis |
| Theophylline | Theophylline ER | Second-line asthma/COPD; narrow therapeutic index |
| PDE4 inhibitor | Roflumilast (Daliresp) | Severe COPD with chronic bronchitis and exacerbations |
| Systemic steroids | Prednisone, methylprednisolone, dexamethasone | Exacerbations (asthma, COPD, ILD, sarcoidosis) |
| Macrolides (chronic) | Azithromycin 3×/wk | Exacerbation prevention in COPD, non-CF bronchiectasis, DPB |
| Mucolytics | N-acetylcysteine, dornase alfa (CF), hypertonic saline | Airway clearance in CF, bronchiectasis |
Biologics for Severe Asthma
| Agent | Target | Indication |
|---|---|---|
| Omalizumab (Xolair) | Anti-IgE | Severe allergic asthma with elevated IgE |
| Mepolizumab (Nucala) | Anti-IL5 | Severe eosinophilic asthma; EGPA; HES; CRSwNP |
| Reslizumab (Cinqair) | Anti-IL5 | Severe eosinophilic asthma (IV) |
| Benralizumab (Fasenra) | Anti-IL5Rα | Severe eosinophilic asthma |
| Dupilumab (Dupixent) | Anti-IL4Rα (blocks IL-4/IL-13) | Severe Type 2 asthma, CRSwNP, atopic dermatitis, EoE |
| Tezepelumab (Tezspire) | Anti-TSLP | Severe asthma regardless of phenotype |
Antifibrotics
| Agent | Mechanism | Indication |
|---|---|---|
| Pirfenidone (Esbriet) | Pleiotropic antifibrotic/anti-inflammatory | IPF |
| Nintedanib (Ofev) | Multi-kinase inhibitor (VEGFR, FGFR, PDGFR) | IPF, SSc-ILD, progressive fibrosing ILD |
Pulmonary Hypertension
| Class | Examples | Route |
|---|---|---|
| PDE5 inhibitors | Sildenafil (Revatio), tadalafil (Adcirca) | PO |
| ERAs | Bosentan (Tracleer), ambrisentan (Letairis), macitentan (Opsumit) | PO |
| Prostanoids | Epoprostenol (Flolan, Veletri), treprostinil (Remodulin, Tyvaso, Orenitram), iloprost (Ventavis), selexipag (Uptravi) | IV / SC / inhaled / PO |
| sGC stimulator | Riociguat (Adempas) | PO — PAH and CTEPH |
CFTR Modulators
Ivacaftor (Kalydeco), lumacaftor/ivacaftor (Orkambi), tezacaftor/ivacaftor (Symdeko), elexacaftor/tezacaftor/ivacaftor (Trikafta). Genotype-dependent; Trikafta is now standard for most patients ≥2 years old with at least one F508del mutation.
19 Classification Systems Reference
| Grade | Description |
|---|---|
| 0 | Only breathless with strenuous exercise |
| 1 | SOB when hurrying on level ground or walking up slight hill |
| 2 | Walks slower than peers on level ground, or must stop for breath at own pace |
| 3 | Stops for breath after ~100 meters or after a few minutes on level ground |
| 4 | Too breathless to leave the house or breathless on dressing/undressing |
BMI, Obstruction (FEV1 % predicted), Dyspnea (mMRC), Exercise (6-minute walk distance). Each scored 0-3 (except BMI 0-1); total 0-10. Higher score = higher 4-year mortality.
Eight items (cough, phlegm, chest tightness, breathlessness, activities, confidence, sleep, energy) scored 0-5 each, total 0-40. CAT ≥10 = high symptom burden (drives ABE grouping).
0-10 scale of perceived breathlessness used during 6MWT and pulmonary rehab. 0 = nothing at all; 10 = maximal.
20-variable score stratifying CAP patients into classes I-V. Class I-II = outpatient; III = brief observation; IV-V = inpatient, V often ICU. Uses age, comorbidities, vitals, and labs (BUN, Na, glucose, hematocrit, pO2, pH).
See also: Wells criteria (s08), CURB-65 (s09), GOLD GOLD 1-4 and ABE (s04), GINA steps and NAEPP severity (s03), Fleischner nodule rules (s12), Scadding sarcoid stages (s06), Berlin ARDS (s14), WHO PH groups (s07), OSA AHI severity (s13).
20 Physical Exam — The Pulmonary Exam
The pulmonary physical exam follows Inspection, Palpation, Percussion, Auscultation (IPPA). Document specific findings in each zone rather than summary labels.
General: Alert, in no acute distress / using accessory muscles / tripoding / pursed-lip breathing. Speaks in full sentences / short phrases / single words.
HEENT: No nasal flaring; oropharynx clear; no cyanosis of lips.
Neck: Trachea midline; no JVD; no cervical lymphadenopathy; no accessory muscle use.
Chest inspection: Symmetric chest wall expansion; no barrel chest / increased AP diameter; no scars or deformity.
Palpation: Tactile fremitus symmetric; no tenderness; chest wall expansion equal.
Percussion: Resonant throughout / dullness at left base / hyperresonance in right upper lobe.
Auscultation: Breath sounds equal bilaterally; clear to auscultation in all lung fields / expiratory wheezes throughout / bibasilar crackles / rhonchi in right mid lung / decreased breath sounds left base with dullness / pleural rub right mid lung. No stridor.
Wheezes: continuous musical high-pitched — airway narrowing (asthma, COPD, foreign body, tumor).
Rhonchi: low-pitched snoring — large airway secretions; clears with cough.
Crackles (rales): discontinuous popping — fine (ILD, pulmonary edema, early PNA) vs coarse (CHF, pneumonia, bronchiectasis).
Stridor: inspiratory high-pitched from upper airway obstruction — emergency.
Pleural rub: coarse grating with respiration; suggests pleuritis, PE, pleural infection.
Always document whether crackles clear with cough (suggests secretions) or not (suggests parenchymal disease). And when charting "decreased breath sounds at left base," also describe percussion and tactile fremitus — dullness + decreased fremitus suggests effusion; dullness + increased fremitus suggests consolidation.
21 Abbreviations Master List
Anatomy & Physiology
Diagnoses
Procedures & Diagnostics
Ventilation & Critical Care
22 Sample HPI Templates
Use these as frameworks for the most common pulmonary and critical care encounters.
"Ms. [Name] is a 32-year-old female with a history of moderate persistent asthma (GINA step 3, on fluticasone-salmeterol 250/50 BID and albuterol PRN) who presents to clinic with a 4-day history of worsening dyspnea, cough, and wheezing in the setting of a viral URI. She reports needing her rescue albuterol every 2-3 hours with only partial relief, two nocturnal awakenings per night from cough, and inability to complete her normal half-mile walk without stopping. She denies fever, chest pain, hemoptysis, or known sick contacts. She has had three prior ED visits for asthma in the last 12 months but no prior intubations. She smokes 3-4 cigarettes per day. On exam: RR 22, SpO2 94% RA, diffuse expiratory wheezes bilaterally, prolonged expiratory phase. Peak flow 58% of personal best."
"Mr. [Name] is a 71-year-old male with severe COPD (GOLD 3, Group E, baseline FEV1 38% predicted), 60-pack-year smoking history (current smoker), home O2 2 L continuous, on tiotropium and budesonide-formoterol, admitted for a 5-day history of increased dyspnea, change in sputum from white to yellow-green, and increased cough productivity. He used 6 albuterol nebulizers at home on the day of presentation without improvement. He has had 3 exacerbations in the past year, including one requiring BiPAP. He denies chest pain, fever, or leg swelling. On arrival: RR 28, SpO2 86% on 2 L, increased to 91% on 4 L NC; diffuse expiratory wheezes, prolonged expiratory phase, accessory muscle use; ABG pH 7.32, PaCO2 58, PaO2 62 on 4 L."
"Mr. [Name] is a 63-year-old male, 35-pack-year former smoker (quit 4 years ago), with hypertension and hyperlipidemia, referred for evaluation of an incidentally discovered right upper lobe pulmonary nodule. CT chest obtained for chronic cough 2 weeks ago demonstrated an 11 mm solid spiculated nodule in the right upper lobe posterior segment with no mediastinal or hilar lymphadenopathy. He denies hemoptysis, weight loss, fevers, or night sweats. He has no personal history of malignancy. Family history is notable for lung cancer in his father (smoker, diagnosed at 68). On exam lungs are clear bilaterally. Plan is PET-CT to characterize metabolic activity and navigational bronchoscopy versus CT-guided biopsy if FDG-avid."
"Ms. [Name] is a 58-year-old female with non-CF bronchiectasis (post-tuberculous, treated in childhood) on chronic azithromycin and oscillating PEP airway clearance, who presents with a 2-day history of blood-tinged sputum progressing today to 3 episodes of frank hemoptysis estimated at 50-75 mL total. She denies chest pain, pleuritic pain, extremity swelling, or recent travel. No history of anticoagulant use. On exam: HR 98, RR 20, SpO2 95% RA, coarse crackles and rhonchi in the right mid and lower lung fields, no stridor. CTA chest demonstrates stable bronchiectasis with a hypertrophied right bronchial artery. Hemoglobin 12.2, coagulation studies normal. Interventional radiology consulted for bronchial artery embolization."
"Mr. [Name] is a 48-year-old male with obesity (BMI 38), hypertension, and type 2 diabetes, referred by his primary care physician for evaluation of suspected obstructive sleep apnea. He reports loud habitual snoring reported by his wife, multiple witnessed apneic episodes, gasping arousals, nonrestorative sleep, morning headaches, and excessive daytime sleepiness (Epworth Sleepiness Scale 16/24). STOP-BANG score 6. He has nearly fallen asleep while driving twice in the past month. Neck circumference 18 inches. Examination shows Mallampati class IV airway and enlarged tonsils. Plan for home sleep apnea test given high pretest probability and absence of significant cardiopulmonary comorbidity."
"Mrs. [Name] is a 66-year-old female with a history of rheumatoid arthritis on methotrexate who presents with a 6-month history of progressive exertional dyspnea, now mMRC grade 3, and a persistent dry cough. She denies fevers, hemoptysis, orthopnea, PND, or leg swelling. She has never smoked. She is not aware of any environmental or occupational exposures. On exam: SpO2 93% on room air dropping to 87% with ambulation, fine bibasilar velcro crackles, no clubbing. HRCT demonstrates bibasilar, peripheral reticulation and traction bronchiectasis without honeycombing, consistent with a fibrotic NSIP pattern. PFTs show FVC 62% predicted, TLC 64%, DLCO 48%. Plan to complete autoimmune serologies, discuss at multidisciplinary ILD conference, and consider initiation of immunosuppression versus antifibrotic therapy depending on progression."
23 References & Sources
Clinical Practice Guidelines
Landmark Clinical Trials
King TE, et al. ASCEND: A Phase 3 Trial of Pirfenidone in Patients with IPF. N Engl J Med. 2014.
Richeldi L, et al. INPULSIS: Efficacy and Safety of Nintedanib in IPF. N Engl J Med. 2014.
Diagram & Figure Sources
Figure 1: Major Respiratory Organs. OpenStax Anatomy & Physiology. CC BY 3.0.
Figure 2: Bronchial Tree. OpenStax Anatomy & Physiology. CC BY 3.0.
Figure 3: Lung Volumes and Capacities. OpenStax Anatomy & Physiology. CC BY 3.0.
Figure 4: Asthma Pathophysiology. Blausen Medical via Wikimedia Commons. CC BY 3.0.
Figure 5: Metered-Dose Inhaler. Blausen Medical via Wikimedia Commons. CC BY 3.0.
Figure 6: Pulmonary Embolism. Blausen Medical via Wikimedia Commons. CC BY 3.0.
Pulmonology and critical care live at the intersection of physiology, pharmacology, and rapid decision-making. A great scribe in this field understands that the numbers matter — FEV1 percent predicted, P/F ratio, AHI, ABI of the ABG, peak and plateau pressures — and captures them precisely. When the attending says "the patient has a P/F of 120 on PEEP 12," you already know this is moderate ARDS by Berlin criteria and can anticipate the discussion of proning, paralysis, and driving pressure.
Know the inhalers by brand and generic. Know which biologic targets which cytokine. Know the Fleischner rules cold. Know when a nodule needs PET versus biopsy versus watch-and-wait. Anticipate the plan from the HPI and the imaging before the physician speaks. Pre-read the radiology reports, the PFT numbers, and the most recent discharge summary. Respiratory medicine rewards the scribe who comes prepared.
Welcome to pulmonary and critical care medicine. Every patient you chart here is breathing a little easier because of how carefully you captured their story.