Reproductive Endocrinology
Every diagnosis, ART procedure, medication, classification system, abbreviation, and documentation framework a new REI scribe needs to succeed.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 Reproductive Physiology — HPG Axis & the Menstrual Cycle
Reproductive endocrinology and infertility (REI) is the subspecialty of obstetrics and gynecology that manages disorders of the reproductive hormonal axis, infertility, and assisted reproductive technology (ART). An REI scribe must understand the hypothalamic-pituitary-gonadal (HPG) axis because every diagnosis, medication, and stimulation protocol manipulates some node of this pathway.
The HPG Axis
The hypothalamus secretes gonadotropin-releasing hormone (GnRH) in a pulsatile fashion into the hypophyseal portal circulation. GnRH stimulates the anterior pituitary to release two gonadotropins: follicle-stimulating hormone (FSH) and luteinizing hormone (LH). In females, FSH drives recruitment and growth of ovarian follicles and stimulates granulosa cells to produce estradiol via aromatization of androgens. LH stimulates theca cells to produce androgens (the substrate for aromatization) and, at mid-cycle, triggers ovulation. In males, FSH supports Sertoli cells and spermatogenesis, while LH stimulates Leydig cells to produce testosterone.
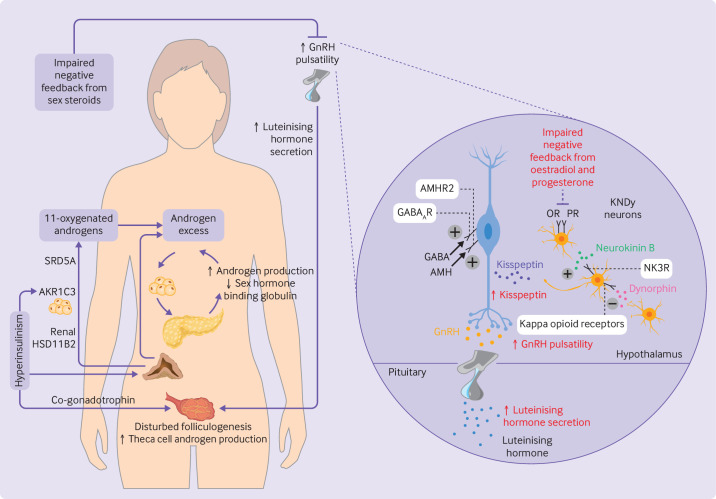
Feedback is tightly regulated. Estradiol and progesterone exert negative feedback on GnRH and FSH/LH throughout most of the cycle, but late-follicular-phase estradiol (rising above ~200 pg/mL sustained for ~48 hours) flips to positive feedback, triggering the LH surge. Inhibin B (granulosa cell product in the follicular phase) and inhibin A (corpus luteum product in the luteal phase) selectively suppress FSH.
The Menstrual Cycle
An idealized cycle is 28 days long, but anything between 21 and 35 days with cycle-to-cycle variation under 7 days is considered ovulatory. The cycle has two hormonally defined phases separated by ovulation:
Follicular phase (days 1–14): Menstruation begins on day 1. Low estradiol and progesterone allow FSH to rise, recruiting a cohort of antral follicles. A single dominant follicle (typically the most FSH-sensitive) is selected around day 5–7 and produces rising estradiol. The endometrium is in the proliferative phase — estradiol-driven glandular and stromal growth. When estradiol peaks (~200–300 pg/mL from a mature follicle 18–22 mm), the LH surge fires and ovulation occurs ~36 hours later.
Luteal phase (days 14–28): The ruptured follicle becomes the corpus luteum, producing progesterone (and continued estradiol). The endometrium enters the secretory phase — tortuous glands, stromal edema, and a receptive implantation window (~days 20–24). If no pregnancy occurs, the corpus luteum involutes, progesterone drops, and menstruation begins. If implantation occurs, the embryo secretes hCG, rescuing the corpus luteum until placental steroidogenesis takes over at ~7–9 weeks (luteal-placental shift).
Female Reproductive Anatomy
The female reproductive tract consists of the ovaries, fallopian tubes (with fimbriated ends, ampullary segment where fertilization occurs, and interstitial segment passing through the uterine wall), uterus (fundus, body, lower uterine segment, cervix), and vagina. Each ovary is anchored by the ovarian ligament, suspensory ligament of the ovary (containing the ovarian vessels), and mesovarium. Knowing these relationships matters when interpreting imaging and dictation from egg retrievals, laparoscopies, and HSG reports.
Cycle day 1 is always the first day of full menstrual flow — not spotting. REI clinicians order "day 3 labs" (FSH, LH, estradiol) on cycle days 2, 3, or 4, and you will routinely chart phrases like "CD3 labs" and "monitoring on CD7." Know the difference between CD (cycle day) and SD (stim day) — stim day 1 is the first day of gonadotropin injections, not the first day of the cycle.
02 Scribe Documentation Framework in REI
REI notes blend an OB/GYN-style gynecologic history with a fertility-specific narrative. A new consult can easily take 45 minutes and generates a dense multi-page note. Follow-ups during active ART cycles are short, number-heavy, and follow a repeating template.
Duration of infertility: "Attempting conception for [X] months, unprotected intercourse [frequency]." ASRM defines infertility as failure to conceive after 12 months of unprotected intercourse (6 months if female partner is ≥ 35).
Obstetric history: G_P_ notation — gravida, para (term/preterm/abortion/living). Include any prior pregnancies with both partners, outcomes, delivery mode, complications, and prior losses with gestational age.
Menstrual history: LMP, cycle length and regularity, flow, dysmenorrhea, intermenstrual bleeding, menarche.
Gynecologic history: STIs, PID, prior pelvic surgery, fibroids, endometriosis, abnormal Pap smears, contraceptive history.
Prior infertility workup/treatment: Previously completed HSG, SIS, semen analysis, day-3 labs, AMH, prior IUI or IVF cycles with protocols, number of retrievals, eggs/embryos, transfers, outcomes.
Partner history: Age, medical issues, prior children, prior fertility, medications, exposures, surgeries (especially varicocele repair, vasectomy, or testicular procedures).
Genetic/family history: Consanguinity, known genetic conditions, ethnicity-based carrier risks (e.g., Ashkenazi, Mediterranean, African), family history of early menopause or RPL.
During a stim cycle, notes are brief and numeric. A sample monitoring note:
CD8 / Stim Day 6 — Antagonist protocol
Meds: Gonal-F 225, Menopur 75, Cetrotide 0.25 started today
E2: 1840 LH: 2.1 P4: 0.6
TVUS: R ovary — 14, 13, 12, 11, 10, 9 mm
L ovary — 15, 14, 13, 12, 11, 9, 9 mm
Endometrium: 8.2 mm trilaminar
Plan: continue current doses, return in 2 days
You will write "G_P_" constantly. Remember the TPAL format inside parentheses: G3P1021 = gravida 3, 1 term, 0 preterm, 2 abortions (miscarriages or terminations), 1 living child. An infertility patient with two miscarriages and no births is G2P0020.
03 The Infertility Workup — Female & Male Evaluation
The standard infertility workup evaluates four functional domains: ovulation, ovarian reserve, tubal patency, and sperm. Uterine cavity evaluation is added when indicated. ASRM Practice Committee: Fertility evaluation of infertile women (2021) defines the baseline evaluation.
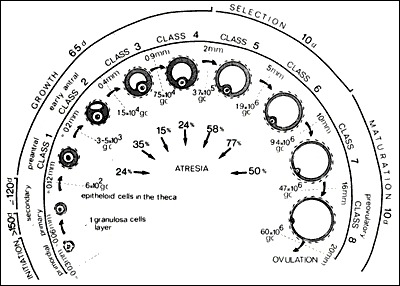
Ovulation: Cycle history, mid-luteal progesterone (day ~21, > 3 ng/mL confirms ovulation), LH ovulation predictor kits, basal body temperature (rarely used now).
Ovarian reserve: Day-3 FSH and estradiol, anti-Müllerian hormone (AMH), and antral follicle count (AFC) by transvaginal ultrasound.
Tubal patency: Hysterosalpingogram (HSG) or HyCoSy (sonohysterography with contrast). Laparoscopy with chromopertubation if high suspicion for pelvic pathology.
Uterine cavity: Transvaginal ultrasound; saline infusion sonohysterogram (SIS) or diagnostic hysteroscopy if suspected polyps, fibroids, adhesions, or septum.
Endocrine screening: TSH, prolactin, and when indicated 17-OH progesterone, androgens (total and free testosterone, DHEA-S), fasting glucose/insulin, lipids.
Semen analysis: The cornerstone. Collected after 2–5 days of abstinence. Evaluates volume, concentration, total sperm count, motility, progressive motility, and morphology (Kruger strict criteria). Repeat in 4–6 weeks if abnormal. AUA/ASRM Male Infertility Guideline (2021).
History/exam: Pubertal development, prior paternity, testicular injury/torsion, cryptorchidism, mumps orchitis, exposures (heat, radiation, chemo), anabolic steroid or testosterone use, varicocele, genital exam.
Endocrine/genetic: FSH, LH, total testosterone, prolactin if abnormal exam or very low count. Karyotype and Y-chromosome microdeletion if severe oligospermia or azoospermia. Cystic fibrosis gene testing if congenital bilateral absence of the vas deferens.
Do not confuse AMH with FSH. AMH is produced by small growing follicles and reflects the resting follicle pool (ovarian reserve). It is cycle-day independent. FSH is a pituitary marker of ovarian responsiveness that must be drawn on cycle day 2–4. A high day-3 FSH (> 10–12) and a low AMH (< 1.0–1.1 ng/mL) both suggest diminished ovarian reserve.
04 Ovulatory & Anovulatory Disorders Endocrine
Anovulation and oligo-ovulation account for roughly 25% of female infertility. The WHO classification, adopted by REI practice, divides anovulation into three groups:
| Group | Description | Typical Example |
|---|---|---|
| WHO I | Hypogonadotropic hypogonadal anovulation — low FSH, low estradiol | Hypothalamic amenorrhea, Kallmann syndrome |
| WHO II | Normogonadotropic normoestrogenic anovulation — normal FSH/estradiol, disordered signaling | PCOS (the overwhelming majority) |
| WHO III | Hypergonadotropic hypoestrogenic anovulation — high FSH, low estradiol | Premature ovarian insufficiency, perimenopause |
A separate WHO category (sometimes called Group IV) is hyperprolactinemic anovulation, covered in its own section below. Management of anovulation depends on the category: WHO II responds to ovulation induction with letrozole or clomiphene; WHO I requires exogenous gonadotropins or pulsatile GnRH; WHO III generally requires donor egg or aggressive IVF with poor prognosis.
05 Polycystic Ovary Syndrome (PCOS) Endocrine
PCOS is the most common endocrine disorder in reproductive-age women (8–13% prevalence) and the single most common cause of anovulatory infertility. It is a syndrome — meaning it is diagnosed by a constellation of features rather than a single test.
Pathophysiology
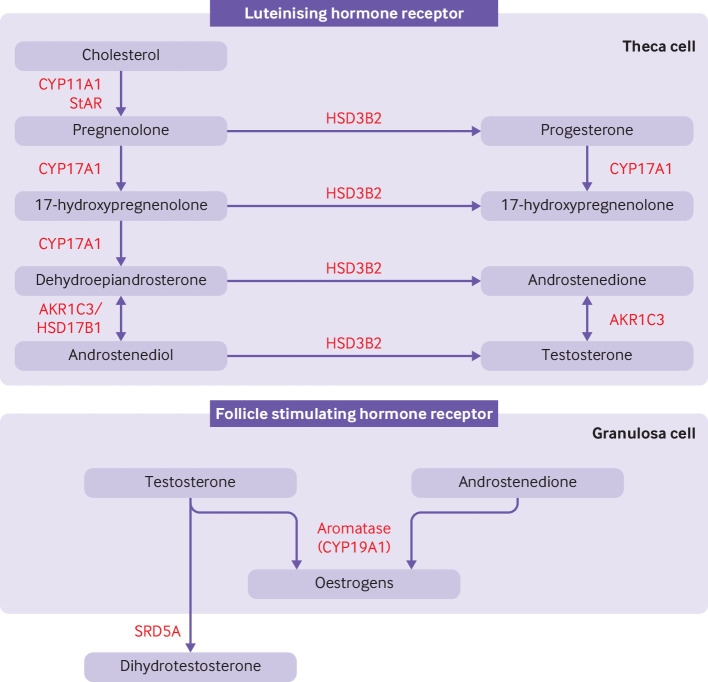
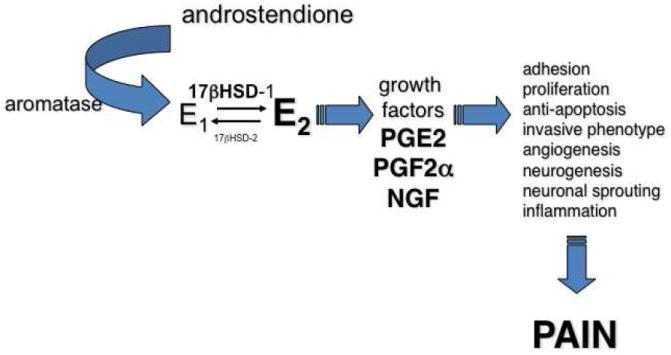
PCOS is driven by a combination of increased GnRH pulse frequency (favoring LH over FSH), ovarian and adrenal hyperandrogenism, and peripheral insulin resistance. Elevated LH stimulates theca cell androgen production; relative FSH insufficiency impairs aromatization and follicular maturation, leaving many small antral follicles arrested at ~5–8 mm — the classic polycystic ovarian morphology. Insulin resistance amplifies ovarian androgen production and reduces SHBG, raising free testosterone.
Diagnosis: Rotterdam Criteria
The Rotterdam criteria (2003), reaffirmed by the 2023 International PCOS Guideline, require 2 of 3 of the following after exclusion of other causes (thyroid disease, hyperprolactinemia, non-classical CAH, Cushing, androgen-secreting tumor):
| Criterion | Definition |
|---|---|
| 1. Oligo- or anovulation | Cycles > 35 days or < 8 cycles/year |
| 2. Clinical or biochemical hyperandrogenism | Hirsutism, acne, androgenic alopecia, or elevated total/free testosterone |
| 3. Polycystic ovarian morphology on ultrasound | ≥ 20 follicles 2–9 mm per ovary OR ovarian volume > 10 mL (on high-frequency TVUS) — AMH > ~4.7 ng/mL can substitute in adults |
Clinical Presentation & Workup
Patients present with oligomenorrhea/amenorrhea, infertility, hirsutism, acne, obesity, and acanthosis nigricans. Long-term risks include endometrial hyperplasia and carcinoma (from unopposed estrogen), type 2 diabetes, metabolic syndrome, NAFLD, obstructive sleep apnea, and cardiovascular disease. Workup includes total testosterone, DHEA-S, 17-OH progesterone (to rule out non-classical CAH), TSH, prolactin, 2-hour OGTT or HbA1c, lipid panel, and pelvic ultrasound.
Management
Non-fertility: Lifestyle modification (first-line; even 5–10% weight loss restores ovulation in many), combined oral contraceptives for cycle regulation and androgen symptoms, metformin for insulin resistance and metabolic risk, spironolactone for hirsutism, endometrial protection with progestin.
Fertility: Letrozole is first-line ovulation induction (PPCOS II trial) — superior live-birth rate compared to clomiphene in PCOS. Second-line: gonadotropins, laparoscopic ovarian drilling. Third-line: IVF (with attention to OHSS risk — PCOS patients are high responders).
PCOS patients are the classic high responders to ovarian stimulation. Expect the REI doctor to use low-dose gonadotropin protocols, antagonist cycles, and agonist (Lupron) triggers to reduce OHSS risk. When you hear "freeze-all" or "dual trigger," high responder physiology is usually the reason.
06 Diminished Ovarian Reserve & Premature Ovarian Insufficiency Endocrine
Diminished ovarian reserve (DOR) describes a quantitative and/or qualitative reduction in the remaining oocyte pool for age. It is not a disease per se but a prognostic finding associated with reduced response to stimulation and lower cycle success rates. ASRM: Testing and interpreting measures of ovarian reserve (2020) is the reference.
| Test | Concerning Value | Notes |
|---|---|---|
| Day-3 FSH | > 10–15 IU/L | Must be paired with estradiol < 60–80 pg/mL (elevated E2 can falsely normalize FSH) |
| AMH | < 1.0–1.1 ng/mL | Best single test; cycle-day independent |
| Antral follicle count | < 5–7 total (both ovaries) | TVUS on cycle day 2–5 |
| Clomiphene challenge test | Day-10 FSH > 10 | Historical, rarely used today |
Premature Ovarian Insufficiency (POI)
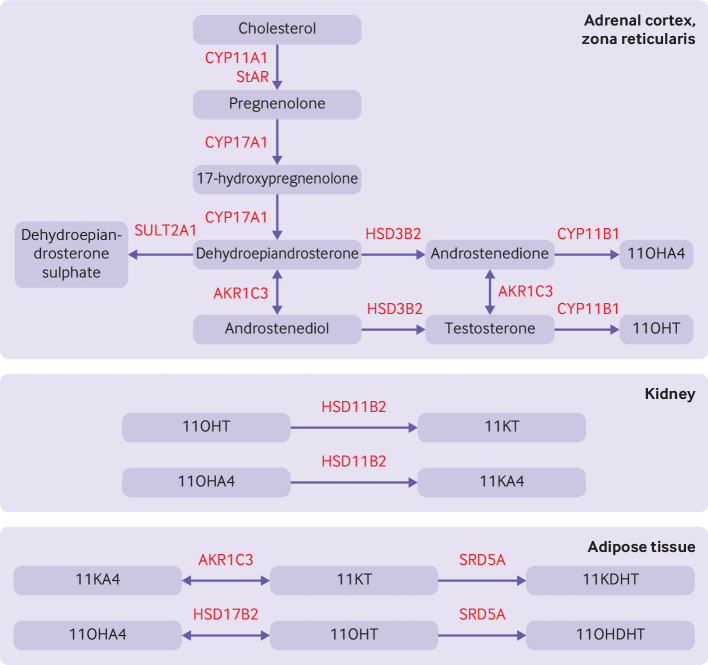
POI, formerly "premature ovarian failure," is defined as hypergonadotropic hypoestrogenic ovarian dysfunction in women younger than 40, with amenorrhea (or markedly oligomenorrhea) for at least 4 months and two FSH levels > 25–40 IU/L at least one month apart. Etiologies include idiopathic, genetic (Turner 45,X mosaicism, fragile X premutation FMR1), autoimmune (oophoritis, polyglandular syndromes), iatrogenic (chemotherapy, pelvic radiation, bilateral oophorectomy), and infectious. Workup includes karyotype, FMR1, adrenal autoantibodies, TSH, and thyroid autoantibodies. Management emphasizes hormone replacement therapy until natural menopause age (~51), bone health, cardiovascular risk management, and — for fertility — donor egg IVF. Spontaneous pregnancies occur in ~5–10% and cannot be reliably induced.
07 Hypogonadotropic Hypogonadism & Hypothalamic Amenorrhea Endocrine
Hypogonadotropic hypogonadism is the hypothalamic or pituitary failure to stimulate the ovaries — low FSH, low LH, low estradiol. The most common REI presentation is functional hypothalamic amenorrhea (FHA) from low body weight, excessive exercise, or chronic stress. Other etiologies include Kallmann syndrome (congenital GnRH deficiency with anosmia), pituitary adenomas, Sheehan syndrome, infiltrative disease (sarcoid, hemochromatosis), and prior cranial irradiation.
Workup includes FSH/LH/estradiol, prolactin, TSH, pituitary MRI if central cause suspected, and assessment of energy availability. Endocrine Society guideline on FHA (2017) emphasizes restoration of energy balance (nutrition, reduced exercise, psychological support) as first-line management. For fertility, pulsatile GnRH (where available) or exogenous gonadotropins (FSH plus LH activity) induce ovulation. Clomiphene does not work in FHA because there is no estrogen-driven feedback to block.
08 Hyperprolactinemia & Pituitary Disorders Endocrine
Prolactin suppresses GnRH pulsatility, producing oligomenorrhea/amenorrhea, anovulation, galactorrhea, and infertility. Causes include pregnancy (always exclude first), medications (antipsychotics, metoclopramide, SSRIs, methyldopa, verapamil), hypothyroidism (TRH stimulates prolactin), prolactinoma (microadenoma < 10 mm, macroadenoma ≥ 10 mm), stalk effect from a non-prolactin-secreting sellar mass, chronic kidney disease, and stress/recent breast exam.
Evaluation: repeat prolactin fasting and without recent breast/nipple stimulation, TSH, pregnancy test, pituitary MRI if persistent elevation or very high levels (> 100 ng/mL usually indicates adenoma). Management is dopamine agonist therapy — cabergoline (first line, 0.25–0.5 mg twice weekly) is preferred over bromocriptine for efficacy and tolerability. Most micro- and macroadenomas shrink substantially on cabergoline, and ovulation/fertility often return within weeks to months. Endocrine Society hyperprolactinemia guideline (2011).
09 Endometriosis & Adenomyosis Pelvic
Endometriosis is the presence of endometrial-like glands and stroma outside the uterine cavity, most commonly on the peritoneum, ovaries (endometriomas / "chocolate cysts"), pelvic ligaments, and bowel/bladder serosa. It affects 6–10% of reproductive-age women and up to 30–50% of women with infertility. It contributes to infertility via distorted pelvic anatomy, adhesions, impaired folliculogenesis, altered peritoneal environment, and reduced implantation.
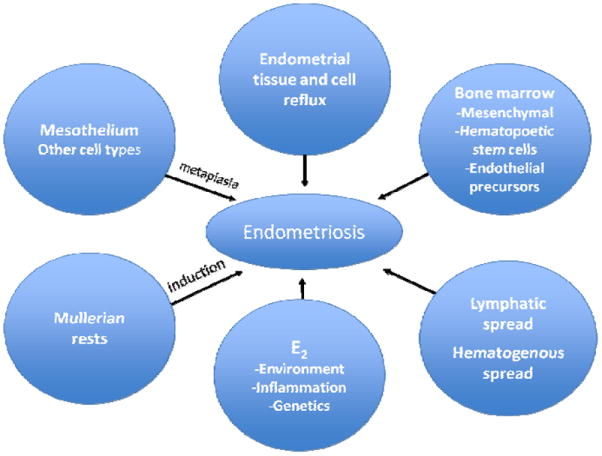
Clinical Presentation & Diagnosis
Cyclic pelvic pain, dysmenorrhea, dyspareunia (especially deep), dyschezia, infertility, abnormal bleeding. Physical exam may reveal uterosacral nodularity, fixed retroverted uterus, adnexal tenderness. TVUS identifies endometriomas; MRI characterizes deep infiltrating endometriosis. Definitive diagnosis is histologic from laparoscopic biopsy, though empiric diagnosis and treatment are now common. ESHRE Endometriosis Guideline (2022) supports clinical and imaging diagnosis without mandatory laparoscopy.
ASRM (rASRM) Staging
| Stage | Score | Description |
|---|---|---|
| I — Minimal | 1–5 | Few isolated superficial implants, minimal or no adhesions |
| II — Mild | 6–15 | More superficial implants, minimal adhesions; possible small endometriomas |
| III — Moderate | 16–40 | Multiple implants, deep peritoneal or ovarian endometriomas, mild to moderate adhesions |
| IV — Severe | > 40 | Large endometriomas, dense adhesions, cul-de-sac obliteration, deep infiltrating disease |
Management
For pain: NSAIDs, combined oral contraceptives, progestins (norethindrone acetate, dienogest), GnRH agonists (leuprolide) with add-back therapy, GnRH antagonists (elagolix, relugolix), and laparoscopic excision or ablation. For fertility: expectant management in mild disease, surgical excision may improve spontaneous conception in early stages, IVF for moderate-severe disease or failed other therapies. Endometrioma resection is weighed carefully because cystectomy can further reduce ovarian reserve.
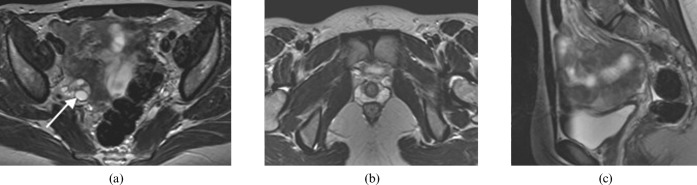
Adenomyosis
Ectopic endometrial glands within the myometrium, causing a bulky globular uterus, heavy menstrual bleeding, dysmenorrhea, and reduced implantation/pregnancy rates. Imaging features on TVUS and MRI include a thickened junctional zone (> 12 mm on MRI), myometrial cysts, and asymmetric myometrial thickening. Management mirrors endometriosis; definitive cure is hysterectomy (not fertility-compatible).
10 Uterine Fibroids, Polyps & Intrauterine Adhesions Uterine
Uterine Fibroids (Leiomyomas)
Benign smooth-muscle tumors of the uterus, extremely common (up to 70% lifetime prevalence). Classified by location — submucosal (protruding into the cavity, highest fertility impact), intramural, and subserosal. The FIGO leiomyoma classification (0–8) subdivides these further, with Types 0–2 being submucosal:
| Type | Location |
|---|---|
| 0 | Pedunculated intracavitary (submucosal) |
| 1 | Submucosal, < 50% intramural |
| 2 | Submucosal, ≥ 50% intramural |
| 3 | Intramural, contacts endometrium |
| 4 | Intramural |
| 5 | Subserosal, ≥ 50% intramural |
| 6 | Subserosal, < 50% intramural |
| 7 | Pedunculated subserosal |
| 8 | Other (cervical, parasitic) |
Submucosal and cavity-distorting intramural fibroids reduce implantation and increase miscarriage. Management for fertility includes hysteroscopic myomectomy (Types 0–2), abdominal or laparoscopic/robotic myomectomy (Types 3–4 large), and expectant management for small non-cavity-distorting fibroids. ASRM: Removal of myomas in asymptomatic patients (2017).
Endometrial Polyps
Benign focal overgrowths of endometrial glands and stroma projecting into the cavity. Associated with abnormal uterine bleeding and reduced implantation. Diagnosed by SIS or hysteroscopy; removal by hysteroscopic polypectomy before IVF is routine.
Asherman Syndrome / Intrauterine Adhesions
Scarring within the uterine cavity, most often following aggressive D&C (especially after retained products of conception or postpartum). Presents with hypomenorrhea, amenorrhea, cyclic pain, recurrent loss, or infertility. Diagnosed by hysteroscopy (gold standard), SIS, or HSG. Treatment is hysteroscopic adhesiolysis followed by estrogen therapy and adhesion prevention strategies (balloon stent, gel, IUD). Prognosis depends on severity per the ESGE classification.
11 Tubal Factor, Hydrosalpinx & Congenital Uterine Anomalies Anatomic
Tubal Factor Infertility
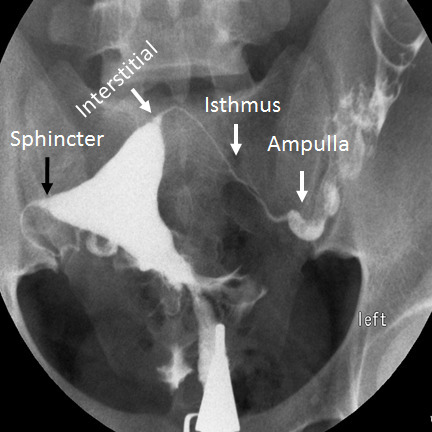
Accounts for ~25–35% of female infertility. Causes include prior pelvic inflammatory disease (chlamydia, gonorrhea), tubal surgery, prior ectopic pregnancy, endometriosis, tuberculosis, and prior tubal ligation. Diagnosis is by hysterosalpingogram (HSG) — bilateral free spill indicates patent tubes; unilateral or bilateral blockage, hydrosalpinx, or cornual occlusion is documented.
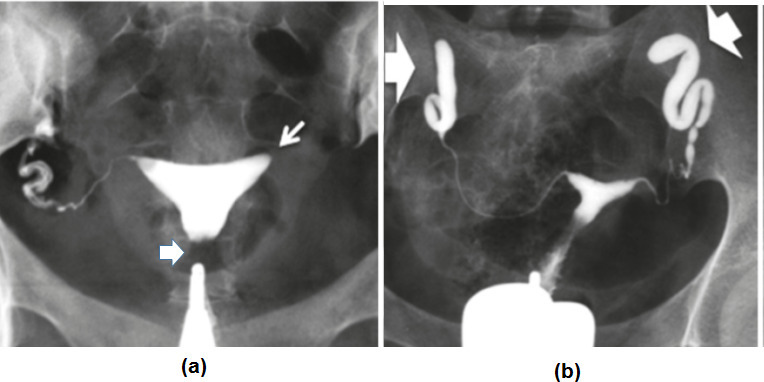
Hydrosalpinx
A fluid-filled obstructed fallopian tube, visible as a tubular cystic adnexal structure on ultrasound. Hydrosalpinx fluid is embryotoxic and halves IVF implantation/live birth. ASRM: Role of tubal surgery in the era of ART (2021) recommends salpingectomy or proximal tubal occlusion before IVF for any ultrasound-visible hydrosalpinx.
Congenital Uterine (Müllerian) Anomalies
Abnormal fusion of the paired paramesonephric (Müllerian) ducts during embryogenesis. The ASRM/ESHRE classification includes:
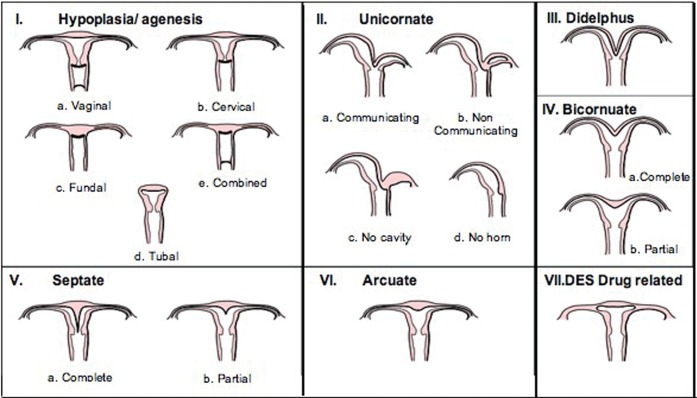
| Class | Anomaly | Fertility Impact |
|---|---|---|
| I | Segmental agenesis / hypoplasia (includes MRKH) | Absolute — requires surrogacy |
| II | Unicornuate uterus | Increased preterm birth, malpresentation |
| III | Uterus didelphys (complete duplication) | Generally preserved fertility, increased preterm birth |
| IV | Bicornuate uterus | Increased preterm birth, malpresentation |
| V | Septate uterus | Most strongly associated with RPL and infertility; amenable to hysteroscopic resection |
| VI | Arcuate uterus | Normal variant, minimal impact |
| VII | DES-related anomaly (T-shaped) | Historical, decreasing |
3D ultrasound and MRI distinguish septate from bicornuate uteri — the distinction is critical because septate is corrected hysteroscopically, while bicornuate requires abdominal metroplasty (rarely done).
12 Male Factor Infertility Male
Male factor contributes to ~40–50% of infertility (as sole or contributing factor). The semen analysis is the cornerstone of evaluation. WHO 2021 (6th edition) reference lower limits define the common abnormalities:
| Parameter | Lower Reference Limit |
|---|---|
| Volume | ≥ 1.4 mL |
| Sperm concentration | ≥ 16 million/mL |
| Total sperm count | ≥ 39 million/ejaculate |
| Total motility | ≥ 42% |
| Progressive motility | ≥ 30% |
| Normal morphology (Kruger strict) | ≥ 4% |
| Vitality | ≥ 54% |
Terminology
Oligospermia — low sperm count; asthenospermia — poor motility; teratospermia — abnormal morphology; oligoasthenoteratospermia (OAT) — all three; azoospermia — no sperm in ejaculate; aspermia — no ejaculate (retrograde ejaculation, obstruction).
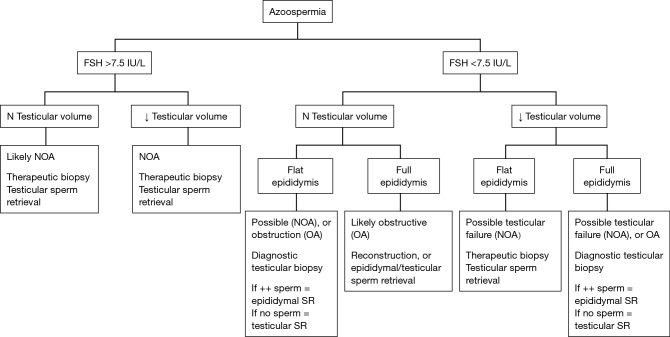
Azoospermia — Obstructive vs Non-Obstructive
Obstructive (OA): Normal testicular volume, normal FSH, normal testosterone. Causes: CBAVD (cystic fibrosis), post-infectious, iatrogenic (vasectomy, hernia repair). Sperm can usually be retrieved surgically (PESA, MESA, TESA). Non-obstructive (NOA): Small testes, elevated FSH, variable testosterone. Causes: Klinefelter 47,XXY, Y-chromosome microdeletion (AZFa/b/c), cryptorchidism, chemotherapy/radiation, Sertoli-cell-only syndrome. Sperm retrieval requires microdissection TESE with variable success (30–60%).
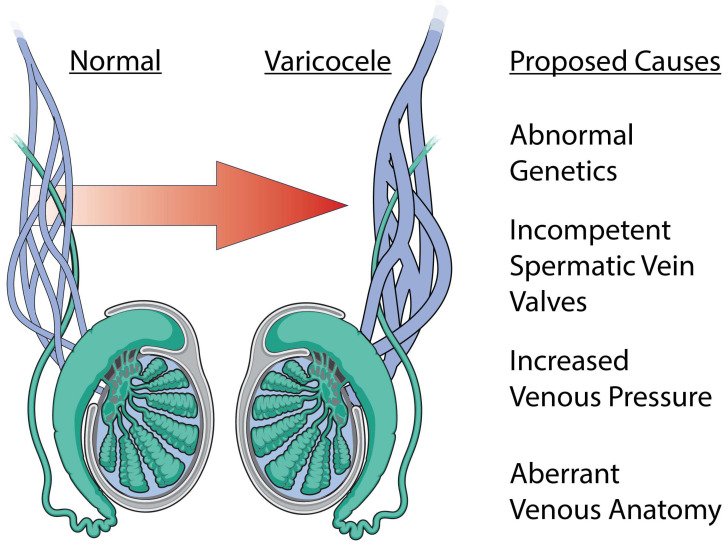
Varicocele
Dilated pampiniform plexus veins, usually left-sided (90%). Associated with abnormal semen parameters via testicular hyperthermia and oxidative stress. Graded by palpation: Grade 1 palpable only with Valsalva, Grade 2 palpable at rest, Grade 3 visible through scrotal skin. Varicocelectomy is offered to men with palpable varicocele, abnormal semen, and otherwise unexplained infertility per AUA/ASRM Male Infertility Guideline (2021).
Male Hypogonadism & Exogenous Testosterone
Exogenous testosterone suppresses pituitary LH/FSH and shuts down spermatogenesis — a common, underrecognized cause of secondary male infertility. Management: stop testosterone, treat with clomiphene, hCG, and/or hMG to restore intratesticular testosterone and spermatogenesis. This often takes 6–18 months. Sperm DNA fragmentation testing may be ordered for unexplained infertility, recurrent IVF failure, or RPL.
13 Recurrent Pregnancy Loss (RPL) Loss
ASRM defines recurrent pregnancy loss as two or more failed clinical pregnancies documented by ultrasound or histology (ESHRE definition allows biochemical losses). Only ~50% of RPL workups identify a definitive cause. ASRM Practice Committee: Evaluation of RPL (2012) and ESHRE RPL Guideline (2018).
1. Genetic: Parental karyotypes (balanced translocation in 2–5%), products of conception karyotype/microarray (aneuploidy is the leading cause, rises sharply with maternal age).
2. Anatomic: SIS, hysteroscopy, or 3D ultrasound for septate uterus, submucosal fibroids, polyps, intrauterine adhesions.
3. Endocrine: TSH, prolactin, HbA1c, consider progesterone, prolactin, and diabetes screening. Uncontrolled thyroid disease and diabetes impair implantation and early pregnancy.
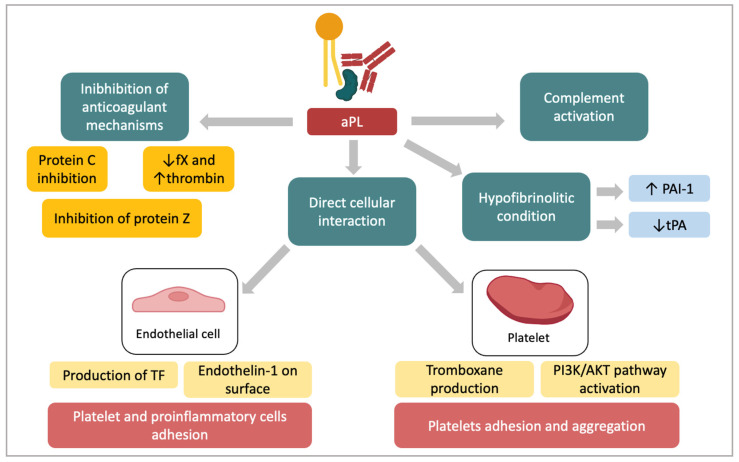
4. Immunologic/Thrombophilic: Antiphospholipid syndrome panel (lupus anticoagulant, anti-cardiolipin IgG/IgM, anti-beta-2 glycoprotein IgG/IgM — requires two positive results 12 weeks apart). Heritable thrombophilia testing (Factor V Leiden, prothrombin G20210A, protein C/S, antithrombin) is NOT routinely recommended for RPL unless personal/family VTE history.
5. Lifestyle/Environmental: Smoking, alcohol, obesity, recreational drugs, caffeine, BMI extremes.
Management
Targeted based on identified cause: hysteroscopic septoplasty, progesterone supplementation (controversial, most benefit in women with 3+ prior losses), levothyroxine for hypothyroidism, anticoagulation (low-dose aspirin + LMWH) for confirmed antiphospholipid syndrome, IVF with PGT-SR for balanced translocation carriers, IVF with PGT-A considered in recurrent aneuploid losses. MTHFR genotyping is NOT recommended — it is not an established RPL cause despite historical testing. Empiric treatments (IVIG, intralipid, lymphocyte immunotherapy) are not evidence-based and generally discouraged.
14 Unexplained & Secondary Infertility Evaluation
Unexplained infertility is a diagnosis of exclusion — ovulatory cycles, normal semen analysis, patent tubes, and a normal uterine cavity after a complete workup. It accounts for ~15–30% of couples. Management ladder: expectant management (younger women), ovulation induction with timed intercourse, OI plus IUI, and IVF. ASRM: Evidence-based treatments for couples with unexplained infertility (2020).
Secondary infertility is inability to conceive after a prior successful pregnancy. The workup is identical to primary infertility; age-related decline in ovarian reserve and interval-acquired issues (new male factor, tubal disease, retained products or adhesions from a prior delivery) are common.
15 Fertility Preservation & Oncofertility Preservation
Fertility preservation covers patients facing gonadotoxic therapy (chemotherapy, pelvic radiation, bilateral oophorectomy or orchiectomy), those with a gender identity transition plan, and those pursuing elective ("social") preservation. ASCO Fertility Preservation Guideline update (2018) is the standard oncology-facing reference.
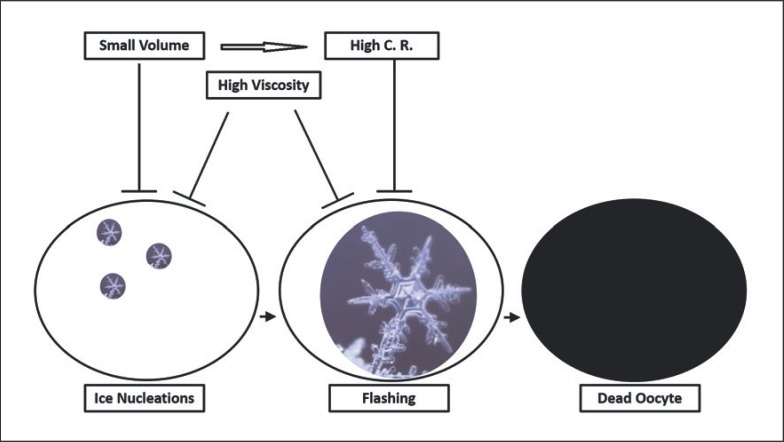
Oocyte cryopreservation (egg freezing): Standard of care. Controlled ovarian stimulation followed by retrieval and vitrification of mature (MII) oocytes. Success depends heavily on age at freeze and number of eggs banked.
Embryo cryopreservation: Highest established success rates. Requires sperm source at time of freezing.
Sperm cryopreservation: Before chemo/radiation, gender-affirming care, or vasectomy. Simple and low-cost.
Ovarian tissue cryopreservation: Surgical removal and freezing of ovarian cortex strips. Previously experimental, now standard of care per ASRM. Only option for prepubertal girls and women needing immediate chemotherapy.
Testicular tissue cryopreservation: Experimental, used in prepubertal boys.
Random-start protocols (stimulation begun in the luteal phase rather than waiting for the next menses) are commonly used in oncofertility to minimize delay to chemotherapy. Letrozole co-administration is used for hormone-sensitive cancers (e.g., breast cancer) to keep peak estradiol lower during stim.
16 Third-Party Reproduction & Donor Gametes Third-Party
Third-party reproduction involves donor eggs, donor sperm, donor embryos, or a gestational carrier. REI clinics perform extensive screening, counseling, and psychological evaluation before these cycles.
Donor egg IVF: Indicated for POI, severe DOR, prior poor IVF response, genetic disease, or advanced maternal age. Donors are screened for medical history, genetic carrier status, infectious disease (per FDA 21 CFR 1271), and undergo psychological evaluation. Cycles can be fresh (synchronized with recipient endometrial prep) or frozen donor egg bank cycles.
Donor sperm: Used for azoospermia without viable retrieval, severe genetic disease, single women by choice, and same-sex female couples. All donor sperm is quarantined for 6 months with repeat infectious disease testing per FDA.
Gestational carrier (GC): A woman who carries a pregnancy conceived from intended parents' (or donors') gametes via embryo transfer. Distinct from traditional surrogacy (which uses the carrier's own egg and is rarely performed today). Indicated for absolute uterine factor (MRKH, hysterectomy, severe Asherman), medical contraindication to pregnancy, or same-sex male couples. Legal contracts are mandatory and state laws vary widely. ASRM guidance on gestational carriers (2022).
17 Assisted Reproductive Technology (ART) Procedures Procedure
Ovulation Induction (OI)
Letrozole (Femara) 2.5–7.5 mg days 3–7 — an aromatase inhibitor; first-line in PCOS and unexplained infertility per PPCOS II. Clomiphene (Clomid) 50–150 mg days 3–7 or 5–9 — a SERM blocking hypothalamic estrogen feedback, raising endogenous FSH. Both typically produce 1–2 mature follicles. Cycles are monitored with follicle tracking ultrasound and timed intercourse or IUI. Expect the REI doctor to order CD3 baseline, mid-cycle monitoring, and trigger hCG 36 hours before ovulation/insemination.
Intrauterine Insemination (IUI)
Washed, concentrated sperm is placed directly into the uterine cavity using a soft catheter at the time of ovulation. Indicated for mild male factor, cervical factor, unexplained infertility, same-sex couples, and donor sperm cycles. Typically combined with OI. Success rates of 8–15% per cycle depending on age and diagnosis.
Controlled Ovarian Stimulation (COS) for IVF
Exogenous gonadotropins recruit and grow multiple follicles to maturity, overriding normal cohort selection. Two dominant protocols:
GnRH antagonist protocol: Most common in modern REI. Baseline ultrasound and labs CD2–3, start gonadotropins (FSH ± LH) CD2–3, add GnRH antagonist (cetrorelix/Cetrotide or ganirelix/Ganirelix) on ~stim day 5–6 or when lead follicle ≥ 14 mm to suppress the endogenous LH surge. Trigger with hCG or leuprolide when ≥ 2–3 lead follicles reach 17–18 mm. Retrieval 35–36 hours after trigger.
Long GnRH agonist (luteal Lupron) protocol: Begin leuprolide in the mid-luteal phase of the preceding cycle for pituitary downregulation, then start gonadotropins after menses. Once dominant in the past, still used for select patients (e.g., endometriosis, very tight synchronization needs).
Microdose flare, mini-IVF, natural cycle IVF: Specialized protocols for poor responders or patients wanting minimal medication.
Agonist (Lupron) trigger: Used in high responders (PCOS, high AMH) to reduce OHSS risk; must be paired with freeze-all or luteal support because it causes short-lived LH surge insufficient to maintain corpus luteum.
Egg Retrieval (Oocyte Pickup, OPU)
Transvaginal ultrasound-guided needle aspiration of follicles under IV sedation, typically 10–20 minutes. Each follicle is aspirated and flushed; follicular fluid is handed to embryology to identify cumulus-oocyte complexes. Number and maturity (MII, MI, GV) are documented. Risks include bleeding, infection, anesthesia, damage to adjacent structures, and OHSS.

Fertilization — Conventional IVF vs ICSI
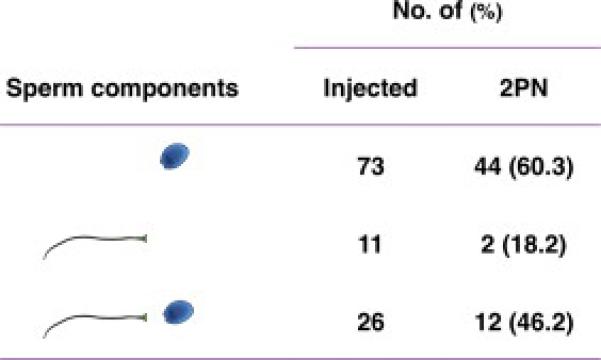
Conventional insemination: ~100,000 motile sperm are placed in the dish with each oocyte. Intracytoplasmic sperm injection (ICSI): A single sperm is microinjected into each mature (MII) oocyte. Indications: severe male factor, prior fertilization failure, surgically retrieved sperm, PGT cycles (to avoid paternal DNA contamination). Fertilization is assessed at ~16–18 hours for pronuclei.
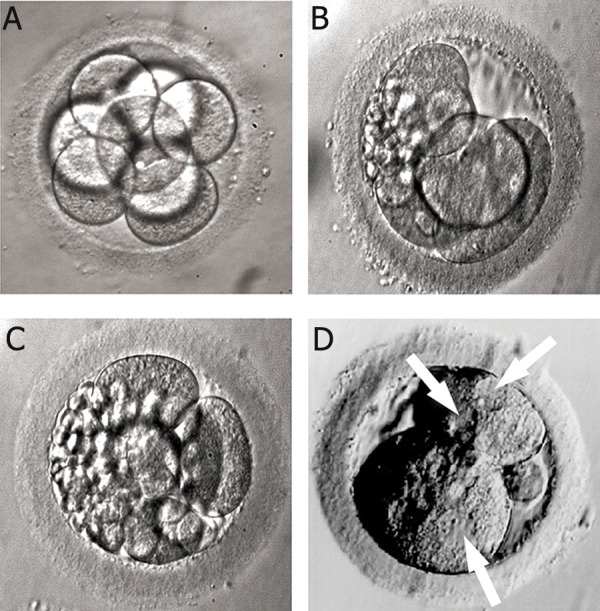
Embryo Culture & Transfer
Embryos are cultured to cleavage stage (day 3, ~8 cells) or blastocyst stage (day 5–6). Blastocyst culture allows better selection and is the norm in most US programs. Fresh embryo transfer — transfer during the same cycle as retrieval. Frozen embryo transfer (FET) — embryos are vitrified and transferred in a subsequent programmed or natural cycle. FET has become more common because it allows PGT, avoids OHSS, and may improve outcomes in certain populations. Endometrial preparation for programmed FET: estradiol (oral, transdermal, or vaginal) for ~2 weeks to build lining, then progesterone (IM, vaginal, or combined) to create the implantation window, with transfer on progesterone day 5 (blastocyst) or day 3 (cleavage).
Preimplantation Genetic Testing (PGT)
Performed on a trophectoderm biopsy of blastocyst-stage embryos, with embryos frozen awaiting results.
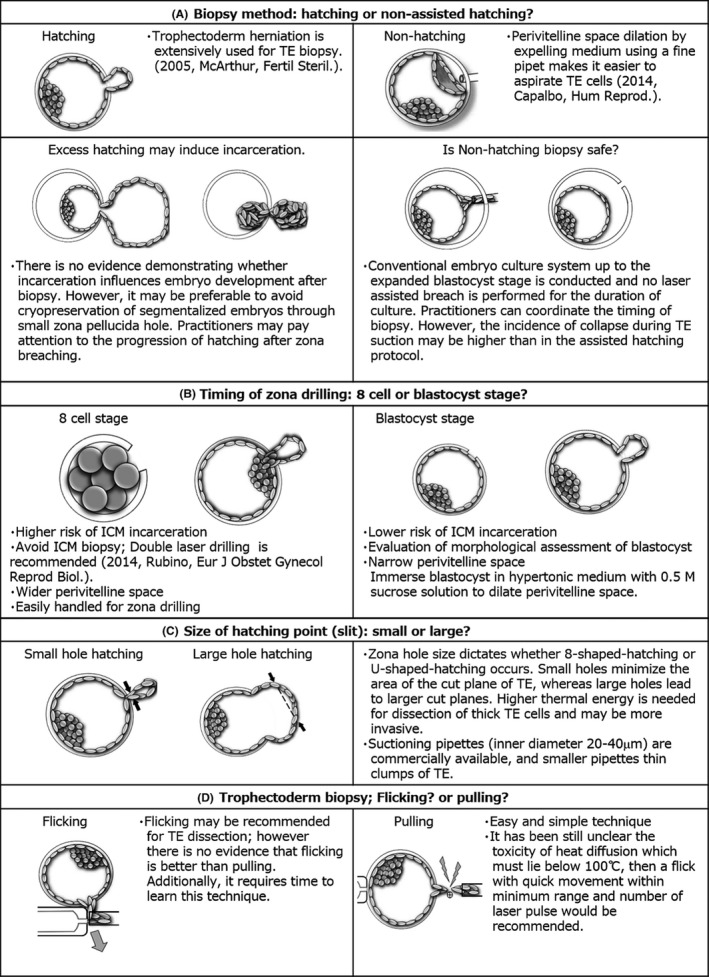
- PGT-A (aneuploidy): Screens for chromosomal abnormalities. Improves per-transfer success and may reduce miscarriage; does not improve cumulative live birth in the general population and is controversial in younger patients.
- PGT-M (monogenic): Tests for specific single-gene disorders (cystic fibrosis, sickle cell, Huntington, BRCA, fragile X, etc.) — requires custom probe development.
- PGT-SR (structural rearrangement): For carriers of balanced translocations and inversions.
Mosaic embryos (mixed euploid/aneuploid cell lines on biopsy) are a distinct result category; low-level mosaic embryos may be transferred with appropriate counseling and have produced healthy live births.
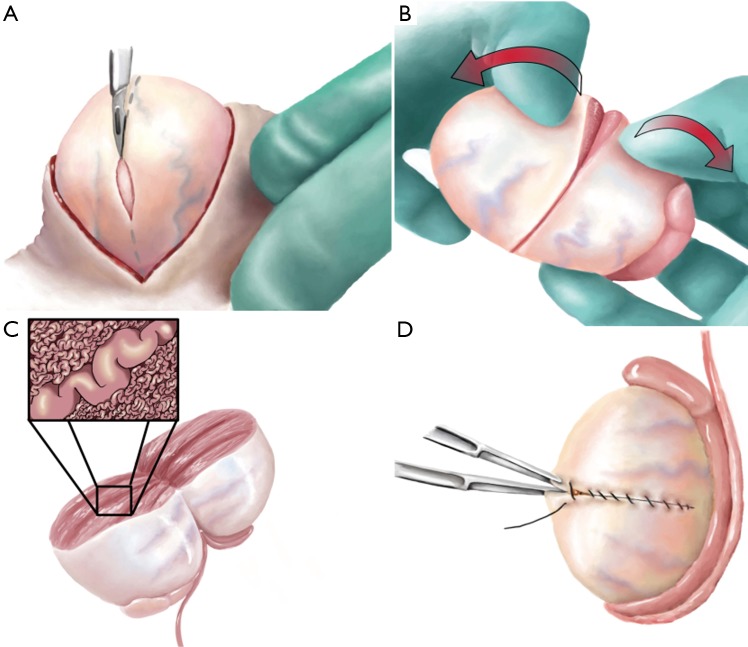
Surgical Sperm Retrieval
PESA — Percutaneous epididymal sperm aspiration. MESA — Microsurgical epididymal sperm aspiration (open, microscope-assisted). TESA — Testicular sperm aspiration (needle). TESE / micro-TESE — Testicular sperm extraction (open biopsy; microdissection variant for non-obstructive azoospermia). Sperm retrieved is typically used with ICSI.
Diagnostic & Office Procedures
Hysterosalpingogram (HSG): Fluoroscopic study where iodinated contrast is injected through the cervix to outline the cavity and tubes. Performed in follicular phase after menses, before ovulation. Reports comment on cavity contour, tubal fill, free spill, loculation, and any hydrosalpinx.
Saline infusion sonogram (SIS) / sonohysterogram: Transvaginal ultrasound with saline infusion to distend the cavity and detect polyps, submucosal fibroids, and adhesions.
Hysteroscopy: Direct visualization of the cavity with a thin scope; used diagnostically and for operative procedures (polypectomy, myomectomy, septoplasty, adhesiolysis).
Laparoscopy: Minimally invasive surgical exploration, used for endometriosis diagnosis/treatment, salpingectomy, tubal reconstruction, and ovarian cystectomy.
Endometrial biopsy: Office sampling for histologic dating, hyperplasia screening, or chronic endometritis (CD138 staining).
18 OHSS & Other ART Complications Complication
Ovarian hyperstimulation syndrome (OHSS) is an iatrogenic complication of ovarian stimulation driven by VEGF-mediated capillary leak in hCG-exposed, hyperstimulated ovaries. Presents 3–10 days after trigger (early) or after pregnancy establishes (late). High-risk features: PCOS, high AMH (> 3.5), high AFC, prior OHSS, peak estradiol > 3500–5000, > 20 follicles on stim ultrasound.

| Grade | Features |
|---|---|
| Mild | Abdominal distension, mild nausea, enlarged ovaries (< 8 cm), no ascites |
| Moderate | Ultrasound-evident ascites, ovaries 8–12 cm, weight gain, moderate nausea/vomiting |
| Severe | Clinical ascites ± pleural effusion, hemoconcentration (Hct > 45%), oliguria, hypoproteinemia, electrolyte disturbance, ovaries > 12 cm |
| Critical | Tense ascites or large pleural effusion, Hct > 55%, WBC > 25k, renal failure, thromboembolism, ARDS |
ASRM: Prevention and treatment of OHSS (2016) emphasizes prevention: antagonist protocols, agonist triggers in high responders, freeze-all cycles, cabergoline prophylaxis, and individualized gonadotropin dosing. Management: supportive care with fluids, analgesia, VTE prophylaxis, paracentesis for tense ascites, and admission for severe disease.
Other Complications
Multiple gestation: Historic main risk of IVF; now mitigated by elective single embryo transfer (eSET). Ectopic pregnancy: ~2–4% after IVF (higher in tubal factor). Bleeding, infection, injury at retrieval. Anesthesia complications. Implantation failure and repeat IVF failure trigger further workup: repeat cavity evaluation, endometrial biopsy for chronic endometritis, thyroid and prolactin, PGT-A consideration, sperm DNA fragmentation, immunologic evaluation in select cases.
When a patient calls with severe bloating, shortness of breath, and rapid weight gain after an IVF retrieval, chart "concern for OHSS" as the top-of-mind diagnosis. Document weight, abdominal girth, urine output, symptoms, and any vitals the MA obtained. OHSS can progress rapidly from moderate to severe.
19 Imaging, Labs & Diagnostics Diagnostics
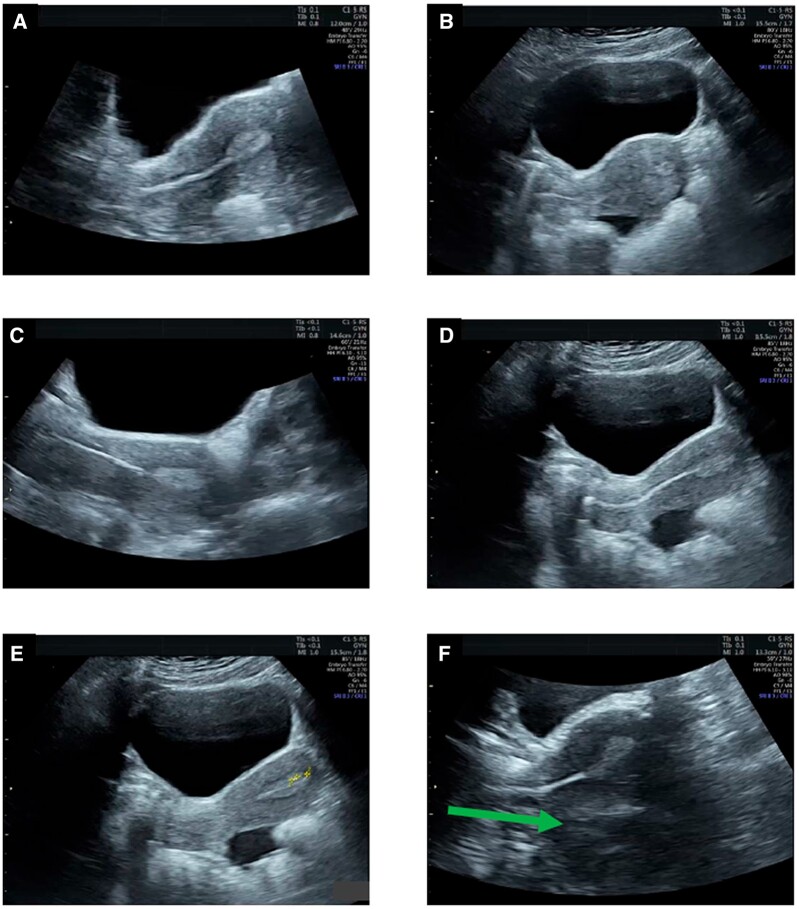
Transvaginal Ultrasound (TVUS)
The workhorse of REI. Used for baseline ovarian and uterine assessment, AFC, follicle tracking during stim, endometrial measurement, cyst characterization, and embryo transfer guidance. You will document: ovarian volumes, follicle number by size, endometrial thickness (mm) and pattern (trilaminar/triple-stripe, homogeneous, hyperechoic), and any abnormalities (cysts, fibroids, polyps, hydrosalpinx, free fluid).
Key Lab Values
| Lab | Normal / Target | Clinical Use |
|---|---|---|
| Day-3 FSH | < 10 IU/L | Ovarian reserve |
| Day-3 estradiol | < 60–80 pg/mL | Must be low for FSH to be interpretable |
| AMH | 1.0–4.0 ng/mL (varies by age) | Ovarian reserve, OHSS risk |
| TSH | < 2.5 mIU/L preconception | Thyroid optimization for fertility |
| Prolactin | < 25 ng/mL | Anovulation workup |
| Mid-luteal progesterone | > 3 ng/mL | Confirms ovulation |
| HbA1c | < 6.5% preconception | Glycemic control |
| Stim-cycle estradiol | ~200–300 per mature follicle | Response tracking |
| Beta-hCG (pregnancy test) | Doubling every 48–72 h early | Pregnancy confirmation post-ET |
20 Medications You Must Know Meds
| Drug (Generic) | Brand | Mechanism / Use |
|---|---|---|
| Letrozole | Femara | Aromatase inhibitor; first-line OI, especially PCOS |
| Clomiphene citrate | Clomid, Serophene | SERM; blocks hypothalamic estrogen feedback, raises FSH |
| Follitropin alfa/beta | Gonal-F, Follistim | Recombinant FSH; injectable stimulation |
| Menotropins (hMG) | Menopur, Repronex | FSH + LH activity from urinary source |
| Urofollitropin | Bravelle (discontinued) | Purified urinary FSH |
| hCG (choriogonadotropin) | Novarel, Pregnyl, Ovidrel | LH-like; trigger shot for ovulation/final maturation |
| Leuprolide acetate | Lupron | GnRH agonist; downregulation or agonist trigger |
| Cetrorelix | Cetrotide | GnRH antagonist; prevents LH surge during stim |
| Ganirelix | Ganirelix, Orgalutran | GnRH antagonist; alternative to cetrorelix |
| Drug | Form | Use |
|---|---|---|
| Progesterone in oil (PIO) | IM injection | Luteal phase and FET support |
| Vaginal progesterone | Endometrin, Crinone, compounded suppositories | Luteal/FET support |
| Estradiol | Oral (Estrace), transdermal (Vivelle, Climara), vaginal | Endometrial prep for FET, HRT, donor cycles |
| Medroxyprogesterone | Provera | Withdrawal bleed induction, cycle regulation |
| Combined OCPs | Various | Cycle regulation, pretreatment, PCOS |
| Norethindrone acetate | Aygestin | Endometriosis, cycle suppression |
| Drug | Use |
|---|---|
| Metformin | PCOS insulin resistance, reduces OHSS risk |
| Cabergoline | Hyperprolactinemia (1st line); OHSS prophylaxis (0.5 mg daily for 8 days) |
| Bromocriptine | Hyperprolactinemia (alternative) |
| Low-dose aspirin (81 mg) | APS; empiric in RPL and ART (variable evidence) |
| Heparin / LMWH (enoxaparin) | APS, high-risk thrombophilia during pregnancy |
| Levothyroxine | Thyroid optimization to TSH < 2.5 preconception |
| Prenatal vitamin + folic acid (400–800 mcg) | Preconception standard |
| Testosterone, DHEA (oral 75 mg) | Poor responder / DOR adjunct (variable evidence) |
| Growth hormone | Poor responder adjunct in select protocols |
| Doxycycline | Periprocedural prophylaxis (HSG, retrieval with hydrosalpinx) |
| Methylprednisolone | Periprocedural immunomodulation (protocol-dependent) |
| GnRH antagonists (elagolix, relugolix) | Endometriosis, fibroids |
21 Classification Systems & Criteria Reference
Most classifications referenced in REI notes are already enumerated above in their condition sections — Rotterdam (PCOS), rASRM (endometriosis), FIGO (fibroids), WHO (anovulation), ASRM (Müllerian), WHO 2021 (semen), OHSS severity. Two more are worth memorizing:
At least 2 of 3:
- Advanced maternal age (≥ 40) or other risk factor for poor response
- Previous poor ovarian response (≤ 3 oocytes with conventional stim)
- Abnormal ovarian reserve test (AFC < 5–7 or AMH < 0.5–1.1 ng/mL)
Two episodes of poor response after maximal stimulation are sufficient even without age or abnormal reserve test.
| Group | Age | Ovarian Reserve | Prior Response |
|---|---|---|---|
| 1 | < 35 | Normal (AFC ≥ 5, AMH ≥ 1.2) | Unexpected poor response |
| 2 | ≥ 35 | Normal | Unexpected poor response |
| 3 | < 35 | Low | Expected poor response |
| 4 | ≥ 35 | Low | Expected poor response |
22 Physical Exam & Pelvic Exam in REI
The REI physical exam is focused and targeted. Full exams happen at the new consult; subsequent monitoring visits are usually a TVUS only.
General: BMI, body habitus, signs of androgen excess (hirsutism, acne, alopecia, acanthosis nigricans).
Thyroid: Goiter, nodules, tenderness.
Breast: Tanner stage if relevant, galactorrhea on expression.
Abdomen: Surgical scars, masses, tenderness.
External genitalia: Tanner stage, clitoromegaly (virilization), vulvar findings.
Speculum: Cervix appearance, discharge, lesions.
Bimanual: Uterine size, position, mobility, tenderness; adnexal masses or tenderness; cul-de-sac nodularity (endometriosis). Document normal findings explicitly — "uterus anteverted, normal size, mobile, non-tender; adnexa without masses; no nodularity."
On pelvic ultrasound you will write: "Uterus anteverted, normal contour, [X.X] cm. Endometrial stripe [X] mm, [pattern]. Right ovary [volume], [N] antral follicles, [dominant follicle sizes]. Left ovary [volume], [N] antral follicles, [dominant follicle sizes]. No hydrosalpinx. No free fluid." This template repeats across every monitoring visit.
23 Abbreviations Master List
Cycle & Physiology
Diagnoses
Procedures & ART
Medications
24 Sample HPI Templates
These templates reflect the typical rhythm, content, and terminology of an REI clinic note. Use them as scaffolds.
"Ms. [Name] is a 34-year-old G0 female presenting with her 36-year-old male partner for initial evaluation of primary infertility of 14 months. They have had regular unprotected intercourse 2–3 times weekly without conception. Menarche age 12, cycles every 28–30 days, 4–5 days of flow, mild dysmenorrhea, no intermenstrual bleeding. LMP 10 days ago. No prior pregnancies. GynHx: normal Paps, no STIs, no pelvic surgery. No galactorrhea, hirsutism, or weight changes. Partner is healthy, G0, no prior paternity, no genital surgery or exposures. No family history of early menopause, infertility, or genetic disease. She takes a prenatal vitamin; no other medications. No tobacco or drug use; social alcohol. Today we obtained baseline CD10 TVUS, reviewed the infertility workup plan, ordered CD3 labs (FSH, E2, AMH, TSH, prolactin), semen analysis, and scheduled HSG in the follicular phase next cycle."
"Mrs. [Name] is a 38-year-old G2P0020 with unexplained infertility and two prior first-trimester losses, currently on stim day 7 of her second IVF cycle using an antagonist protocol. Medications: Gonal-F 300 units and Menopur 75 units nightly since CD2, Cetrotide 0.25 mg added on SD5. She reports mild bloating, no significant pelvic pain, no nausea, no shortness of breath, and no weight gain. Today's E2 is 2340 pg/mL, LH 1.8, P4 0.6. TVUS: right ovary 6 follicles measuring 16, 15, 14, 13, 12, 11 mm; left ovary 7 follicles measuring 17, 16, 15, 14, 13, 12, 10 mm. Endometrium 9 mm trilaminar. Plan: continue current doses, return in 2 days for repeat labs and ultrasound; anticipate hCG trigger in 1–2 days with retrieval 36 hours later."
"Ms. [Name] is a 29-year-old G0 female with PCOS diagnosed in adolescence presenting with 18 months of primary infertility. Menses every 45–90 days since menarche, last period 7 weeks ago. BMI 31. She reports moderate hirsutism of the chin and abdomen and intermittent acne. Prior workup confirmed Rotterdam criteria with elevated total testosterone, polycystic ovarian morphology on TVUS, and oligo-ovulation. Normal TSH, prolactin, and 17-OH progesterone; HbA1c 5.8. Partner semen analysis within normal limits. Prior HSG with bilateral tubal patency and normal cavity. She has attempted lifestyle modification with 10-lb weight loss over 6 months. Today we discussed ovulation induction with letrozole starting at 2.5 mg days 3–7 of her next cycle with mid-cycle TVUS follicle tracking and timed intercourse, and reviewed risk of multiples, OHSS, and the step-up plan if no response."
"Mrs. [Name] is a 32-year-old G3P0030 presenting for recurrent pregnancy loss evaluation after three consecutive first-trimester losses at 6, 8, and 9 weeks' gestation, all confirmed by ultrasound, over the past 18 months. POC karyotype from the most recent loss showed 46,XX (euploid). No living children. Cycles regular at 29 days, ovulatory by LH kits. Medical history unremarkable; no thromboses, no autoimmune disease. Partner is healthy, no prior paternity elsewhere. No consanguinity. Today we reviewed the ASRM RPL workup and ordered parental karyotypes, SIS for cavity evaluation, TSH, prolactin, HbA1c, and an antiphospholipid antibody panel. Next visit to review results and formulate a pretreatment plan."
"Mr. [Name] is a 35-year-old male presenting with his 33-year-old female partner for evaluation of severe male factor infertility. Two semen analyses 6 weeks apart demonstrate oligoasthenoteratospermia — concentration 3 million/mL, progressive motility 12%, normal morphology 1% (Kruger). No prior paternity. History notable for cryptorchidism with bilateral orchidopexy at age 2 and a left varicocelectomy at age 22. No exogenous testosterone. Exam demonstrates bilateral small but descended testes and no palpable varicocele. Endocrine labs: FSH 14 IU/L, LH 6.2, total testosterone 310 ng/dL, prolactin normal. Karyotype and Y-chromosome microdeletion pending. Female partner has normal cycles, normal AMH, patent tubes, and normal cavity. Discussed ICSI-based IVF as primary path forward pending genetic results; will refer to urology for repeat evaluation and potential micro-TESE counseling."
"Ms. [Name] is a 31-year-old G0 with PCOS and high AMH (6.8) s/p egg retrieval 5 days ago (28 oocytes retrieved, 22 mature) presenting with progressive abdominal distension, bloating, nausea, and a 6-lb weight gain over 3 days. She denies chest pain, shortness of breath, fever, or calf pain. She triggered with dual hCG/Lupron and is doing a freeze-all. On exam, BP 112/70, HR 96, abdomen distended with shifting dullness, mild diffuse tenderness, no peritoneal signs. TVUS shows enlarged ovaries bilaterally (right 10 cm, left 11 cm) and moderate pelvic free fluid. Labs: Hct 44, WBC 13, Cr 0.9, electrolytes normal. Assessment: moderate early OHSS. Plan: oral hydration with electrolyte-rich fluids, high-protein intake, daily weights and abdominal girth, strict return precautions for worsening symptoms, VTE precautions, and follow-up in 2 days."
REI runs on numbers, protocols, and repeating rhythms. The best scribes know the stim protocols cold, anticipate which labs and measurements the doctor needs to review before they ask, pre-populate monitoring notes with yesterday's values so trends are visible at a glance, and can translate "we'll do an antagonist with 300 of Gonal-F and 75 of Menopur, Cetrotide on day 5, triggering when the lead is 18" into a clean note in real time.
Learn the medications by both generic and brand names — patients and pharmacists use the brands, physicians switch between both. Know the difference between CD and SD. Always document how many follicles were seen, how many were retrieved, how many fertilized, how many made blastocyst, and how many were biopsied and frozen — this is the cycle ledger every patient carries forward. And remember that this is an emotionally charged specialty: your notes will be read by patients who are living through years of hope and loss. Clear, accurate, and compassionate documentation matters.
Welcome to REI. It is a detail-dense specialty with one of the most grateful patient populations in medicine.
25 References & Sources
Clinical Practice Guidelines
ESHRE Endometriosis Guideline Group. ESHRE guideline: endometriosis. Hum Reprod Open. 2022.
ASRM Practice Committee. Evaluation and treatment of recurrent pregnancy loss. Fertil Steril. 2012.
ESHRE Guideline Group on RPL. Recurrent pregnancy loss guideline. Hum Reprod Open. 2018.
Landmark Clinical Trials
Diagram & Figure Sources
Figure 1: HPG axis diagram. Wikimedia Commons, Artoria2e5. Public domain.
Figure 2: Menstrual cycle integrated diagram. Wikimedia Commons, Chris 73. CC BY-SA 3.0.
Figure 3: Female reproductive system anatomy. OpenStax College / Wikimedia Commons. CC BY 3.0.
Figure 4: Ovarian follicle development. OpenStax College / Wikimedia Commons. CC BY 3.0.
Figure 5: IVF procedure overview. Blausen Medical / Wikimedia Commons. CC BY 3.0.