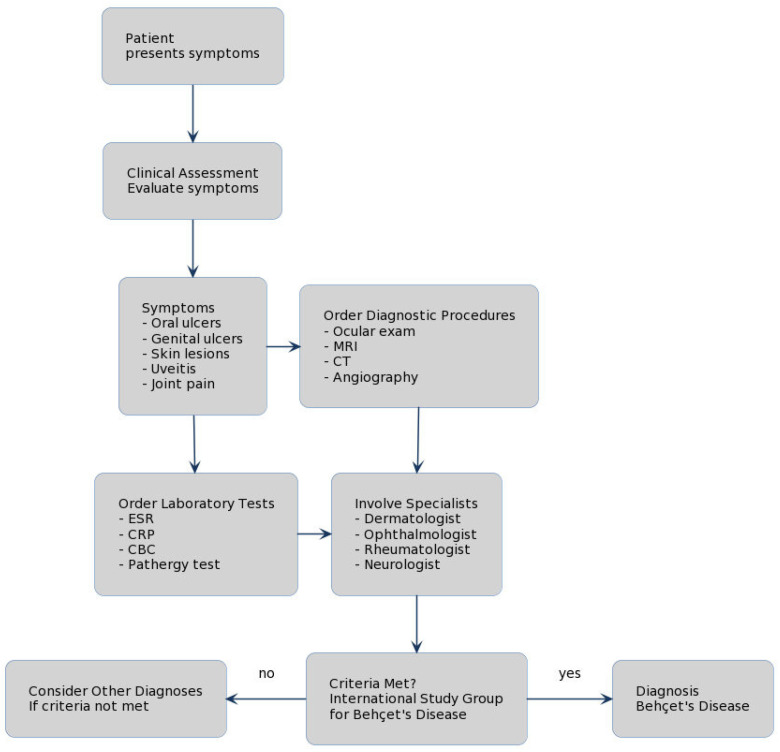
Rheumatology
Every diagnosis, autoantibody, procedure, DMARD, classification criterion, and documentation framework you need to succeed on day one and beyond.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 Joint Anatomy & Immunology Essentials
Rheumatology sits at the intersection of musculoskeletal medicine and clinical immunology. Its patients almost always have one of two problems: joint pain or systemic autoimmune disease, and usually both. To document fluently you need a working knowledge of how a synovial joint is built, how the immune system drives autoimmune inflammation, and how the two intersect in each major rheumatologic disease.
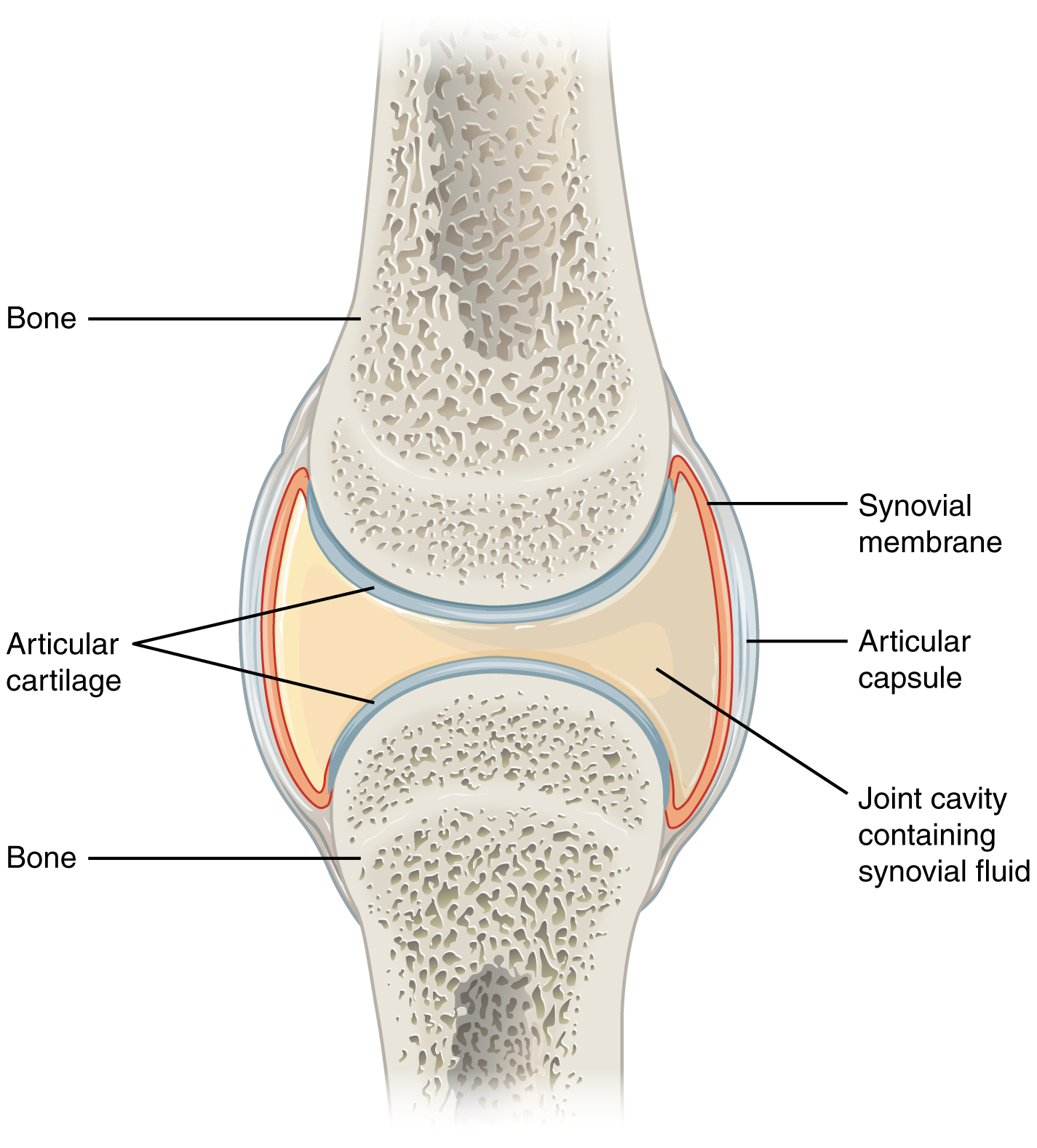
The Synovial Joint
A synovial joint is the body's standard freely movable joint. The articulating ends of the bones are capped by hyaline articular cartilage, a smooth, avascular, low-friction surface. Surrounding the joint is a tough fibrous capsule lined on its inner surface by the synovial membrane (synovium), which secretes synovial fluid into the joint space. Synovial fluid is an ultrafiltrate of plasma enriched with hyaluronic acid; it lubricates the joint and nourishes the avascular cartilage. Ligaments stabilize the joint externally, tendons attach muscles across it, and bursae cushion pressure points.
Almost every rheumatologic disease attacks one or more of these structures. Rheumatoid arthritis targets the synovium, driving a proliferative pannus that erodes cartilage and subchondral bone. Osteoarthritis is cartilage-first disease with secondary bony changes. Gout deposits monosodium urate crystals in synovial fluid. Septic arthritis infects it. Spondyloarthropathies attack the enthesis — the site where tendon or ligament inserts into bone — producing enthesitis rather than primary synovitis.
Joints You Must Know By Name
Rheumatologists describe joint involvement with precise abbreviations. In the hand, from distal to proximal: the distal interphalangeal (DIP) joints, proximal interphalangeal (PIP) joints, and metacarpophalangeal (MCP) joints. The thumb's equivalents are the IP and MCP; the base of the thumb is the first carpometacarpal (CMC) joint, a classic OA site. The wrist is functionally the radiocarpal and intercarpal joints. In the foot, the metatarsophalangeal (MTP) joints, especially the first MTP, are the classic site for gout; the DIPs and PIPs of the toes parallel the hand.
Larger joints include the shoulder (glenohumeral), elbow, hip, knee, and ankle (tibiotalar). The sacroiliac (SI) joints and the zygapophyseal (facet) joints of the spine are the central battleground of axial spondyloarthritis. The temporomandibular joints (TMJs) are involved in RA and juvenile idiopathic arthritis. Always chart joint involvement bilaterally when possible — symmetry is a key feature of RA.
Immunology Essentials
Autoimmune disease arises when the adaptive immune system loses self-tolerance. B cells produce autoantibodies; T cells (particularly CD4+ helper subsets such as Th1, Th2, Th17, and T follicular helper cells) orchestrate and amplify tissue damage through cytokine release. The cytokines that matter most in rheumatology are tumor necrosis factor alpha (TNF-α), interleukin-6 (IL-6), interleukin-1 (IL-1), interleukin-17 (IL-17), interleukin-23 (IL-23), and the type I interferons (especially IFN-α in SLE). Every biologic DMARD you will document targets one of these molecules or the cells that make them.
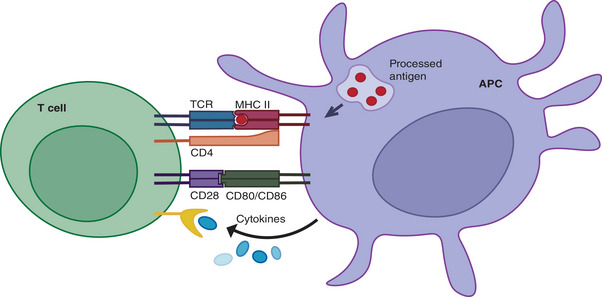
Complement is a plasma protein cascade (C1–C9) that amplifies inflammation and lyses targets; in SLE, immune complexes consume C3 and C4, so low complement levels signal active disease. HLA (human leukocyte antigen) alleles confer genetic susceptibility: HLA-B27 for ankylosing spondylitis, HLA-DR4 (shared epitope) for RA, HLA-DR3 for SLE and Sjögren.
Rheumatology notes are dense with joint abbreviations. When the physician says "tender and swollen MCPs and PIPs bilaterally, DIPs spared," you should immediately know this points to RA rather than OA (which hits DIPs and spares MCPs). Memorize the pattern: RA = MCP/PIP/wrist; OA = DIP/PIP/1st CMC; gout = 1st MTP; PsA = DIPs and "ray" involvement.
02 Scribe Documentation Framework
The Rheumatology SOAP Note
Rheumatology notes are SOAP-format but heavy on two things other specialties don't emphasize: the joint count and the medication timeline. Every visit the physician will document a tender joint count (TJC) and swollen joint count (SJC), often out of 28 or 66/68, and every visit they will review which DMARDs the patient is on, at what dose, for how long, and with what response.
Chief Complaint: "Joint pain follow-up," "new polyarthralgia evaluation," "lupus flare," "gout flare," "rash and arthritis."
HPI: Use OLDCARTS but add rheumatology-specific questions. Duration of morning stiffness (> 60 minutes suggests inflammatory arthritis; < 30 minutes suggests mechanical/OA). Pattern — which joints, symmetric or asymmetric, small or large, axial or peripheral. Constitutional symptoms (fever, weight loss, fatigue, night sweats). Extra-articular features (rash, oral ulcers, photosensitivity, Raynaud phenomenon, sicca symptoms, dyspnea, hemoptysis, neuropathy, eye redness/uveitis). Prior DMARD trials with reason for discontinuation (inefficacy vs intolerance vs insurance).
ROS: Rheumatologic ROS is broad because autoimmune diseases are multisystem. Always ask about eyes (dry eyes, red eyes, vision change), mouth (dry mouth, ulcers), skin (rash, photosensitivity, alopecia, Raynaud), cardiopulmonary (chest pain, dyspnea, cough), GI (dysphagia, reflux, diarrhea), GU (dysuria, hematuria), neuro (headache, weakness, numbness), and constitutional.
PMHx, Meds, Allergies, Social, Family: Document prior infections (TB, hepatitis B/C, HIV — all screened before biologics), vaccination status (especially zoster, pneumococcal, influenza), pregnancy status for women of childbearing age (methotrexate is teratogenic), alcohol use (methotrexate liver toxicity), and family history of autoimmune disease.
Vitals: BP, HR, temperature (fever in flare or infection), weight.
Joint Exam: Inspection (swelling, deformity, erythema), palpation (tenderness, warmth, effusion, synovial thickening), range of motion (active and passive), and stability. Document a tender and swollen joint count. The DAS28 uses 28 joints (shoulders, elbows, wrists, MCPs 1–5, PIPs 1–5, knees). The full 66/68 count adds feet and additional joints.
Skin, Mucosa, Lungs, Heart, Abdomen, Neuro: Target the ROS positives. Rash description (malar, discoid, photosensitive, palpable purpura), oral ulcers, lung auscultation for crackles (ILD), cardiac rubs (pericarditis), abdominal exam, and a focused neuro exam when indicated.
Results: ESR, CRP, CBC, CMP, UA, autoantibodies (ANA, RF, anti-CCP, dsDNA, ENA panel, ANCA), complement (C3, C4), imaging (X-ray, ultrasound with power Doppler, MRI), and medication labs (A1C, lipids, LFTs for MTX, lipid panel for JAKi).
Each rheumatologic diagnosis gets a line item with disease activity ("RA, moderate disease activity by DAS28-CRP 4.1"), a medication action ("continue methotrexate 20 mg weekly, increase adalimumab to every week"), monitoring labs, vaccinations, and return-to-clinic interval. Always document shared decision-making for biologic starts and pregnancy planning.
Rheumatology is a longitudinal specialty. The physician will constantly compare today's exam and labs to prior visits. Before clinic, pre-populate the note with prior DMARD history, last DAS28 or CDAI score, last labs, and last imaging. A scribe who has the trajectory ready saves the physician enormous time.
03 Rheumatoid Arthritis (RA) Inflammatory
Rheumatoid arthritis is the prototypical chronic inflammatory arthritis and the single most common diagnosis in a general rheumatology clinic. It affects roughly 0.5–1% of the adult population, is three times more common in women than men, and peaks in incidence between ages 30 and 60. Untreated RA causes progressive joint destruction, deformity, disability, and a measurable reduction in life expectancy driven primarily by cardiovascular disease.
Pathophysiology
RA begins with loss of tolerance to citrullinated self-proteins. B cells produce rheumatoid factor (RF) and anti-cyclic citrullinated peptide antibodies (anti-CCP / ACPA); these can appear years before symptoms. T cells and macrophages infiltrate the synovium and release TNF-α, IL-6, and IL-1, driving synovial proliferation (pannus), neovascularization, and recruitment of osteoclasts that erode bone at the joint margin. Cartilage is destroyed by matrix metalloproteinases.
Clinical Presentation
Classic RA is a symmetric polyarthritis of the small joints of the hands and feet — MCPs, PIPs, wrists, and MTPs — with morning stiffness lasting more than an hour. The DIPs are characteristically spared (unlike OA and PsA). As disease progresses patients develop the classic deformities: ulnar deviation at the MCPs, swan-neck (PIP hyperextension with DIP flexion) and boutonnière (PIP flexion with DIP hyperextension) deformities, and Z-thumb. Constitutional symptoms include fatigue, low-grade fever, and weight loss.
Extra-articular disease includes rheumatoid nodules (especially over extensor surfaces), rheumatoid lung (interstitial lung disease, pleural effusions), secondary Sjögren syndrome, scleritis/episcleritis, pericarditis, vasculitis, and Felty syndrome (RA + splenomegaly + neutropenia). Cervical spine involvement at C1–C2 (atlantoaxial subluxation) can be life-threatening and must be documented before any general anesthesia.
Diagnostic Workup & Classification
RA is classified by the 2010 ACR/EULAR RA classification criteria, which assign points across four domains — joint involvement, serology, acute-phase reactants, and symptom duration — with a score ≥ 6/10 classifying definite RA.
| Domain | Finding | Points |
|---|---|---|
| Joint involvement | 1 large joint | 0 |
| 2–10 large joints | 1 | |
| 1–3 small joints | 2 | |
| 4–10 small joints | 3 | |
| > 10 joints (at least 1 small) | 5 | |
| Serology | Negative RF and anti-CCP | 0 |
| Low-positive RF or anti-CCP | 2 | |
| High-positive RF or anti-CCP | 3 | |
| Acute phase | Normal CRP and ESR | 0 |
| Abnormal CRP or ESR | 1 | |
| Duration | < 6 weeks | 0 |
| ≥ 6 weeks | 1 |
Score ≥ 6/10 = definite RA.
Imaging: plain radiographs of hands and feet look for periarticular osteopenia, joint space narrowing, and marginal erosions. Ultrasound with power Doppler detects subclinical synovitis and is increasingly used at the bedside. MRI is the most sensitive modality for early erosions and bone marrow edema.
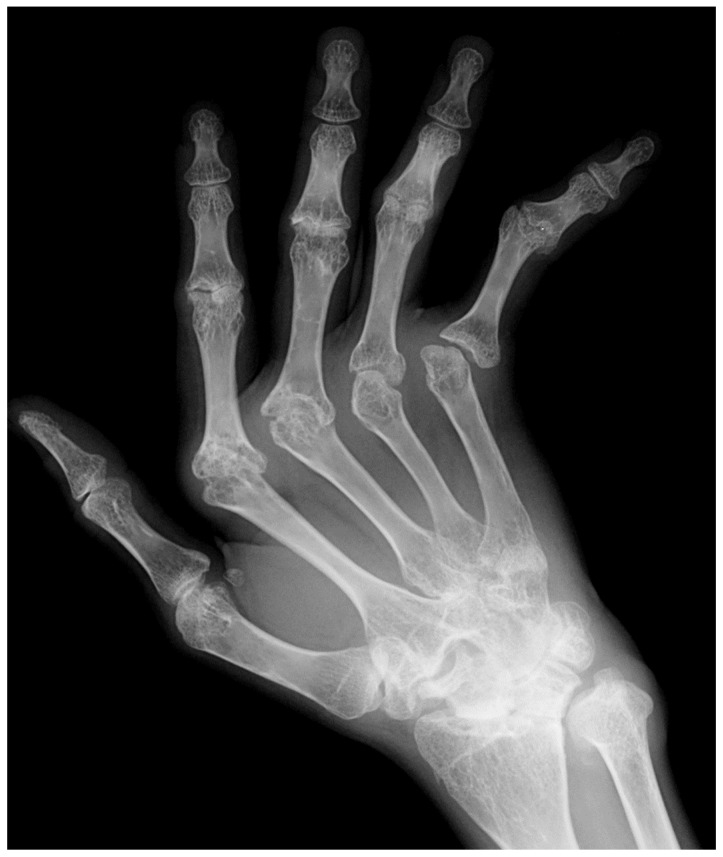
Disease Activity Measures
Document one of the validated disease activity scores at every RA visit: DAS28-CRP or DAS28-ESR (composite of tender joint count, swollen joint count, inflammatory marker, and patient global), CDAI (clinical disease activity index; no lab), or SDAI (adds CRP to CDAI). Categories for DAS28: remission < 2.6, low 2.6–3.2, moderate > 3.2–5.1, high > 5.1.
Management
Treatment follows a treat-to-target strategy aimed at remission or low disease activity. Per the 2021 ACR RA treatment guideline, methotrexate (MTX) is the anchor csDMARD, started at 15 mg weekly and titrated to 20–25 mg weekly with folic acid 1 mg daily. If response is inadequate after 3 months, add or switch to a biologic or targeted synthetic DMARD — typically a TNF inhibitor (etanercept, adalimumab, infliximab, certolizumab, golimumab), IL-6 receptor inhibitor (tocilizumab, sarilumab), abatacept (T-cell co-stimulation blocker), rituximab (anti-CD20 B-cell depletion), or a JAK inhibitor (tofacitinib, baricitinib, upadacitinib). Glucocorticoids are used as short-term bridge therapy only. The ORAL Surveillance trial showed increased cardiovascular and malignancy risk with tofacitinib versus TNFi in high-risk patients, which now shapes JAKi prescribing.
04 Osteoarthritis (OA) Degenerative
Osteoarthritis is the most common joint disease in the world and the most common reason an adult sees a physician for joint pain. While primarily managed by primary care and orthopedics, rheumatologists frequently see OA in the context of atypical presentations, inflammatory flares of "erosive OA," and distinguishing OA from inflammatory arthritis.
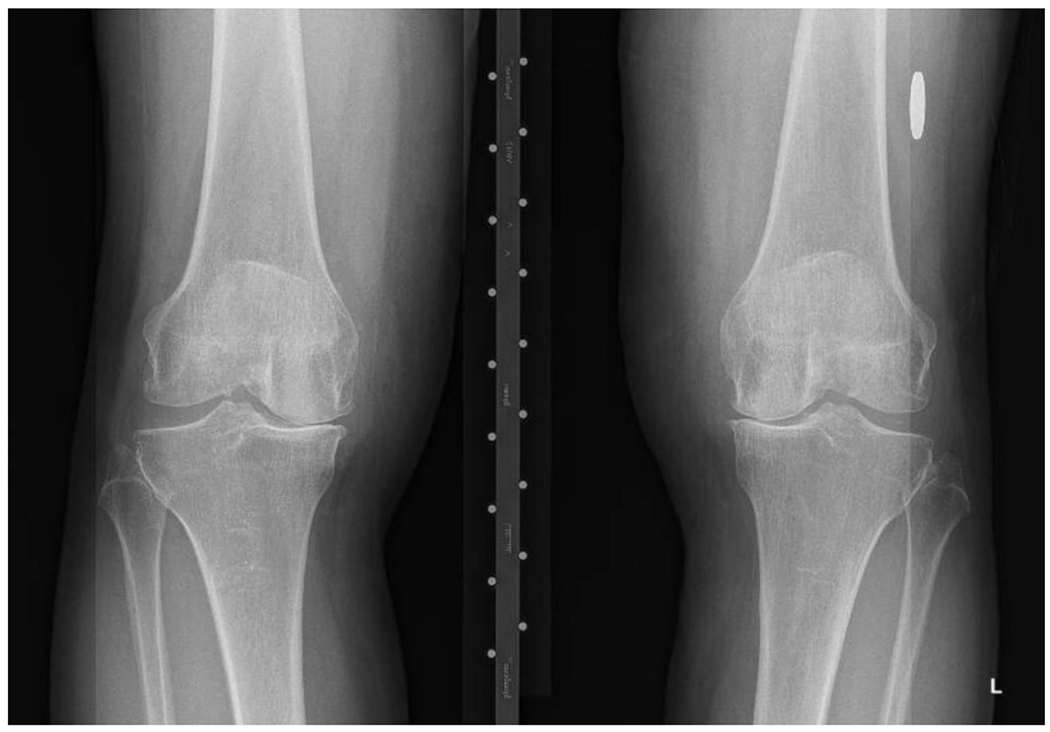
Pathophysiology
OA is a disease of the whole joint — not just "wear and tear." Cartilage loses its normal collagen II matrix and proteoglycans, the subchondral bone remodels and produces osteophytes, the synovium becomes mildly inflamed, and the joint capsule thickens. Mechanical stress, obesity, prior trauma, malalignment, and genetic factors all contribute.
Clinical Presentation
OA causes activity-related joint pain that worsens through the day and improves with rest. Morning stiffness is typically brief (< 30 minutes), distinguishing it from inflammatory arthritis. Affected joints are the DIPs (producing Heberden nodes), PIPs (Bouchard nodes), first CMC, hips, knees, and spine (cervical and lumbar facet joints). Unlike RA, OA rarely affects the MCPs, wrists, elbows, ankles, or shoulders unless there is a secondary cause.
Diagnostic Workup
OA is a clinical and radiographic diagnosis. X-rays show the classic tetrad: joint space narrowing, osteophytes, subchondral sclerosis, and subchondral cysts. Labs are by definition normal — no elevated inflammatory markers, no autoantibodies. The Kellgren-Lawrence grading system (Grade 0–4) is used to quantify radiographic severity.
Management
Per the 2019 ACR/AF OA guideline, first-line management is non-pharmacologic: weight loss, exercise (especially aquatic and strength training for knee/hip OA), and physical therapy. Pharmacologic options include topical NSAIDs (first-line for knee and hand OA), oral NSAIDs (with GI and cardiovascular risk consideration), intra-articular corticosteroid injections, and duloxetine for knee OA. Acetaminophen has limited efficacy. Intra-articular hyaluronic acid and stem cell injections are not strongly recommended. Joint replacement is reserved for end-stage disease refractory to conservative care.
05 Systemic Lupus Erythematosus (SLE) Connective Tissue
SLE is the prototype multisystem autoimmune disease. It predominantly affects women of childbearing age (female:male ratio 9:1) and is more prevalent and more severe in Black, Hispanic, and Asian patients. It can attack virtually every organ — skin, joints, kidneys, blood, brain, heart, lungs — and is characterized by flares and remissions.
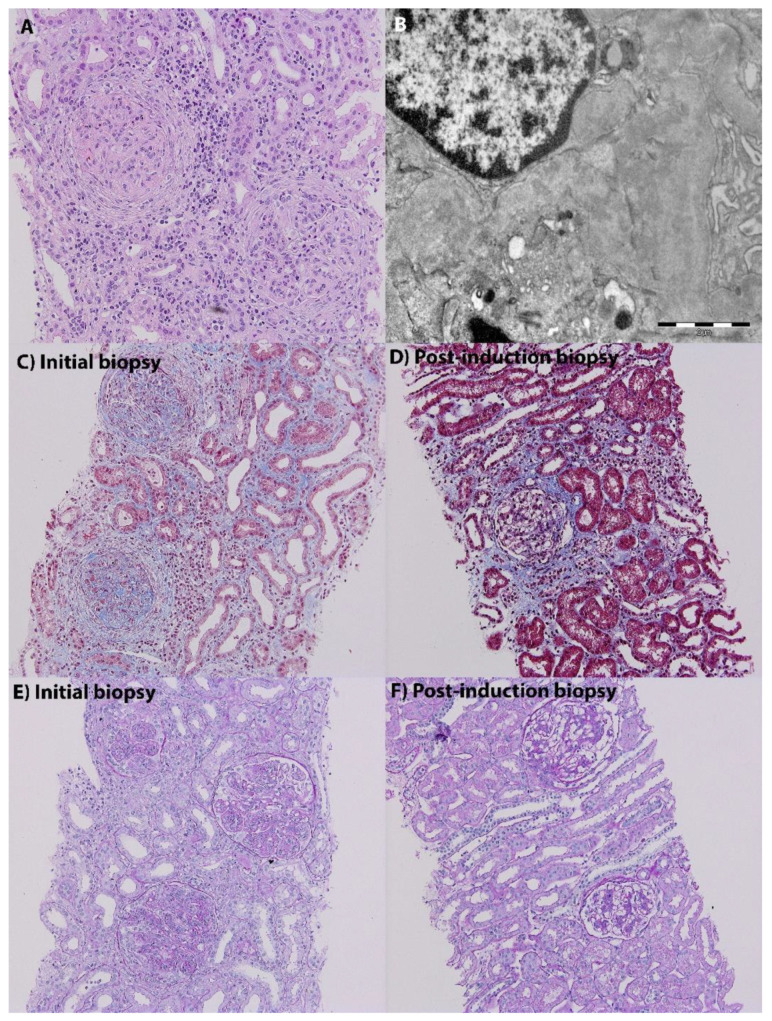
Pathophysiology
SLE is driven by loss of tolerance to nuclear self-antigens. Defective clearance of apoptotic cells exposes nuclear debris; plasmacytoid dendritic cells sense this debris and produce type I interferon, which drives an autoimmune cascade. B cells produce antinuclear, anti-dsDNA, anti-Sm, and other autoantibodies that form immune complexes, deposit in tissues (especially kidney and skin), activate complement, and cause end-organ damage.
Clinical Presentation
The mnemonic SOAP BRAIN MD captures classic features: Serositis, Oral ulcers, Arthritis (non-erosive), Photosensitivity, Blood (cytopenias), Renal, ANA, Immunologic, Neuro, Malar rash, Discoid rash. Key organ involvement includes lupus nephritis (classified I–VI by the ISN/RPS system, with Class III/IV being proliferative and most severe), neuropsychiatric lupus, hematologic disease (AIHA, ITP, lymphopenia), and serositis.
Diagnostic Workup & Classification
SLE is classified by the 2019 EULAR/ACR SLE classification criteria, which require a positive ANA ≥ 1:80 as an entry criterion, then weighted clinical and immunologic criteria across seven clinical and three immunologic domains; a total score ≥ 10 classifies SLE. Key labs: ANA (> 95% sensitive), anti-dsDNA (specific, correlates with nephritis activity), anti-Sm (specific), anti-Ro/La (Sjögren overlap, neonatal lupus risk), anti-RNP (MCTD overlap), antiphospholipid antibodies (lupus anticoagulant, anticardiolipin, anti-β2GPI), and low C3/C4 (active disease).
Disease Activity Measures
Two scoring systems dominate: SLEDAI-2K (Systemic Lupus Erythematosus Disease Activity Index, 24 weighted items, score 0–105) and BILAG-2004 (British Isles Lupus Assessment Group, which grades each of 9 organ systems A–E where A = severe active disease, E = never involved).
Management
Per the 2023 EULAR SLE recommendations, hydroxychloroquine (HCQ) is the cornerstone — nearly every lupus patient should be on it unless contraindicated. It reduces flares, improves survival, and is safe in pregnancy. Glucocorticoids are used for flares with a goal of tapering to ≤ 5 mg/day. Moderate disease is managed with MTX, azathioprine, or mycophenolate mofetil. Severe organ disease (nephritis, neuropsych, severe hematologic) requires high-dose glucocorticoids plus cyclophosphamide or mycophenolate induction. Biologics include belimumab (anti-BLyS/BAFF) and anifrolumab (type I interferon receptor blocker). Annual ophthalmology screening is required for HCQ retinal toxicity.
Document hydroxychloroquine daily dose in mg/kg of actual body weight — the recommended maximum is 5 mg/kg/day to minimize retinal toxicity. Also document the date of the last ophthalmology exam. Physicians will review this at every visit.
06 Sjögren Disease Connective Tissue
Sjögren disease (formerly Sjögren syndrome) is a chronic autoimmune exocrinopathy with lymphocytic infiltration of the salivary and lacrimal glands, producing the classic sicca symptoms of dry eyes and dry mouth. It can be primary or secondary to another connective tissue disease (most commonly RA or SLE).
Clinical Presentation
Patients complain of gritty dry eyes (keratoconjunctivitis sicca), difficulty swallowing dry food without water, dental caries, parotid swelling, fatigue, arthralgia, and Raynaud phenomenon. Extra-glandular features include interstitial lung disease, interstitial nephritis with renal tubular acidosis, peripheral neuropathy (sometimes small-fiber), cutaneous vasculitis, and a markedly increased lifetime risk of MALT lymphoma arising in the parotid.
Diagnostic Workup
Labs: positive ANA, anti-Ro/SSA and anti-La/SSB antibodies, polyclonal hypergammaglobulinemia, and sometimes positive RF. Objective dryness is documented by Schirmer test (≤ 5 mm wetting in 5 minutes), ocular staining score, and unstimulated whole salivary flow (≤ 0.1 mL/min). Minor salivary gland (lip) biopsy showing focal lymphocytic sialadenitis with a focus score ≥ 1 is the gold standard histologic finding and often the decisive test.
Management
Symptomatic care with artificial tears, punctal plugs, pilocarpine or cevimeline (muscarinic agonists for xerostomia), and aggressive dental care. Hydroxychloroquine is used for arthralgia and fatigue. Systemic disease (vasculitis, neuropathy, nephritis, lung disease) may require glucocorticoids, methotrexate, azathioprine, mycophenolate, or rituximab.
07 Systemic Sclerosis (Scleroderma) Connective Tissue
Systemic sclerosis is a connective tissue disease characterized by small-vessel vasculopathy, autoimmunity, and progressive fibrosis of the skin and internal organs. It is uncommon but devastating when organ involvement is severe.
Subsets
Two main subsets are defined by the extent of skin involvement:
- Limited cutaneous (lcSSc): skin thickening distal to elbows and knees (plus face); slower progression; associated with pulmonary arterial hypertension; anti-centromere antibody positive. Includes the CREST phenotype (Calcinosis, Raynaud, Esophageal dysmotility, Sclerodactyly, Telangiectasia).
- Diffuse cutaneous (dcSSc): skin thickening proximal to elbows and knees and on the trunk; rapid progression; early interstitial lung disease and scleroderma renal crisis; anti-Scl-70 (topoisomerase I) and anti-RNA polymerase III positive.
Clinical Features
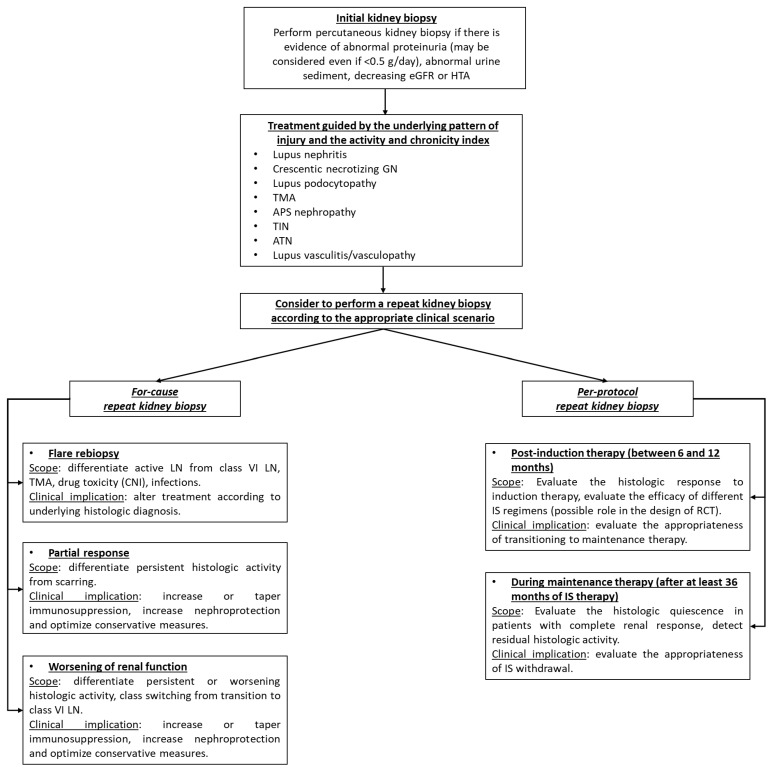
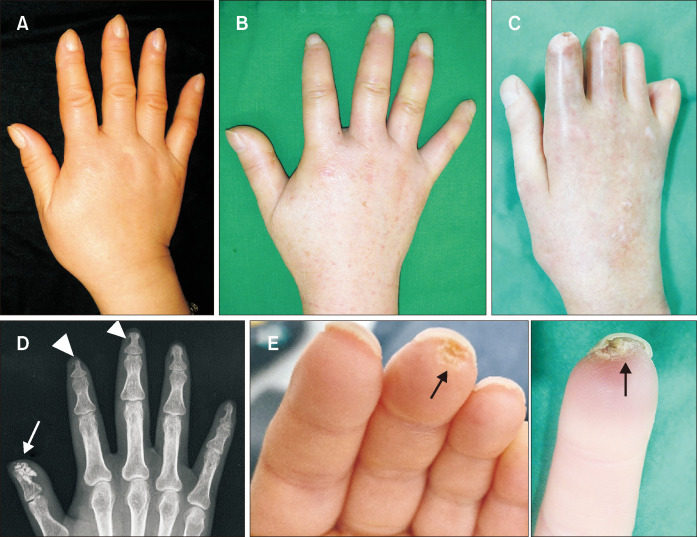
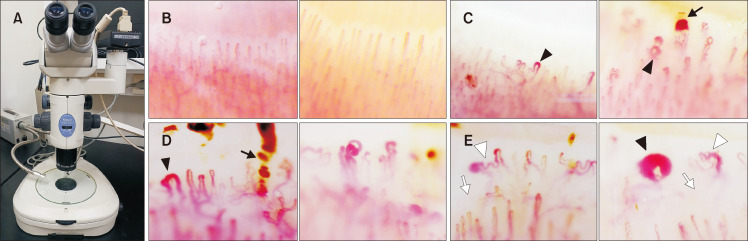
Raynaud phenomenon is nearly universal and often the first symptom, sometimes preceding other features by years. Nailfold capillaroscopy shows dilated capillaries, dropout, and hemorrhages in SSc, distinguishing it from primary Raynaud. Skin changes progress from edematous → indurated → atrophic. GI involvement causes reflux, dysmotility, small intestinal bacterial overgrowth, and "watermelon stomach" (GAVE). Lung involvement includes ILD (usually NSIP pattern) and PAH. Scleroderma renal crisis is a life-threatening malignant hypertension with AKI, classically in dcSSc on high-dose steroids; managed urgently with ACE inhibitors.
Management
Per the 2017 EULAR scleroderma recommendations, treatment is organ-directed: calcium channel blockers and PDE5 inhibitors for Raynaud and digital ulcers; mycophenolate, cyclophosphamide, nintedanib, or tocilizumab for ILD; ACE inhibitors for renal crisis; PAH-specific therapy (endothelin receptor antagonists, PDE5 inhibitors, prostacyclins) for pulmonary hypertension; PPI for reflux; prokinetics for dysmotility. There is no single disease-modifying therapy.
08 Inflammatory Myopathies (PM, DM, IBM, IMNM) Connective Tissue
The idiopathic inflammatory myopathies are a group of autoimmune muscle diseases causing proximal weakness and elevated muscle enzymes. The major subtypes are polymyositis (PM), dermatomyositis (DM), inclusion body myositis (IBM), and immune-mediated necrotizing myopathy (IMNM).
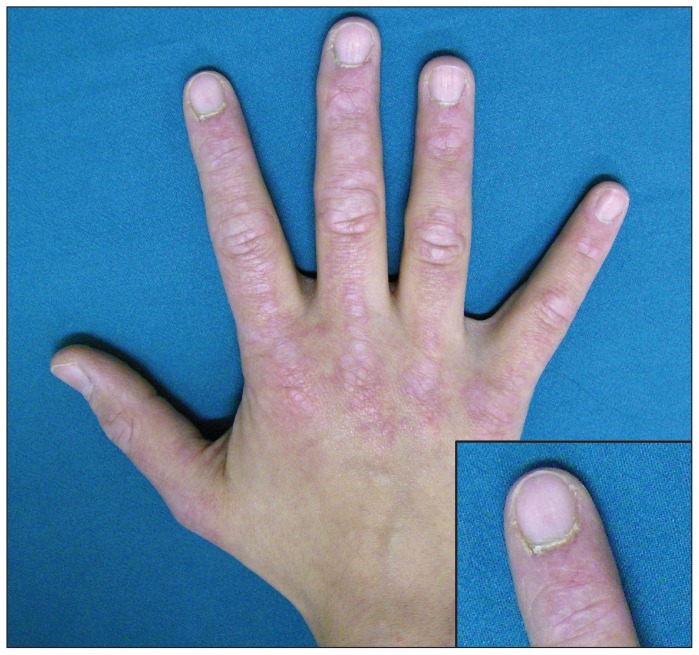
Clinical Features
All four share symmetric proximal muscle weakness (difficulty rising from a chair, climbing stairs, lifting objects overhead). DM adds characteristic skin findings: heliotrope rash (violaceous eyelid discoloration), Gottron papules (scaly papules over MCPs and PIPs), shawl sign, V-sign, mechanic's hands, and calcinosis cutis. IBM is distinguished by asymmetric involvement, early finger flexor and quadriceps weakness, older age at onset, and poor response to immunosuppression. IMNM is associated with statin exposure (anti-HMGCR antibodies) and anti-SRP antibodies.
Diagnostic Workup
CK is elevated (often > 1,000, sometimes > 10,000 in IMNM). Aldolase, AST, ALT, and LDH may also rise. Autoantibodies include the myositis-specific antibodies (MSAs): Jo-1 and other anti-synthetase antibodies (antisynthetase syndrome — myositis + ILD + arthritis + mechanic's hands + Raynaud), anti-Mi-2 (classic DM), anti-MDA5 (amyopathic DM with rapidly progressive ILD), anti-TIF1γ and anti-NXP2 (DM with malignancy risk), anti-SRP, anti-HMGCR. Electromyography shows myopathic changes. MRI of thigh muscles shows edema. Muscle biopsy is definitive. Adults with DM require cancer screening given the paraneoplastic association.
Management
High-dose glucocorticoids are first-line, with steroid-sparing agents including methotrexate, azathioprine, mycophenolate, IVIG, and rituximab. IBM is refractory to immunosuppression; management is primarily supportive and physical therapy.
09 MCTD, UCTD & Antiphospholipid Syndrome Connective Tissue
Mixed Connective Tissue Disease (MCTD)
MCTD is an overlap syndrome with features of SLE, scleroderma, and polymyositis, defined by high-titer anti-U1-RNP antibodies. Patients typically have Raynaud, puffy hands, arthritis, myositis, and esophageal dysmotility; pulmonary hypertension is a leading cause of mortality.
Undifferentiated Connective Tissue Disease (UCTD)
UCTD describes patients with symptoms and autoantibodies suggesting autoimmune connective tissue disease but who do not meet full classification criteria for a specific disease. A subset will eventually evolve into SLE, scleroderma, or another defined entity.
Antiphospholipid Syndrome (APS)
APS is an acquired thrombophilia defined by vascular thrombosis (arterial, venous, or microvascular) or obstetric morbidity (recurrent early miscarriage, fetal loss, severe preeclampsia) plus persistent positivity (≥ 12 weeks apart) for one or more antiphospholipid antibodies: lupus anticoagulant (LA), anticardiolipin (aCL) IgG/IgM, or anti-β2 glycoprotein I (anti-β2GPI) IgG/IgM. Triple-positive patients (all three) carry the highest thrombotic risk. Catastrophic APS (CAPS) is a rare, rapidly progressive multi-organ thrombotic syndrome with high mortality.
Management: lifelong warfarin (INR 2–3 for venous events, higher for arterial) for thrombotic APS. DOACs are not recommended in triple-positive APS after rivaroxaban failed the TRAPS trial. Obstetric APS is managed with aspirin plus prophylactic LMWH during pregnancy. The 2019 EULAR APS guidelines are the standard reference.
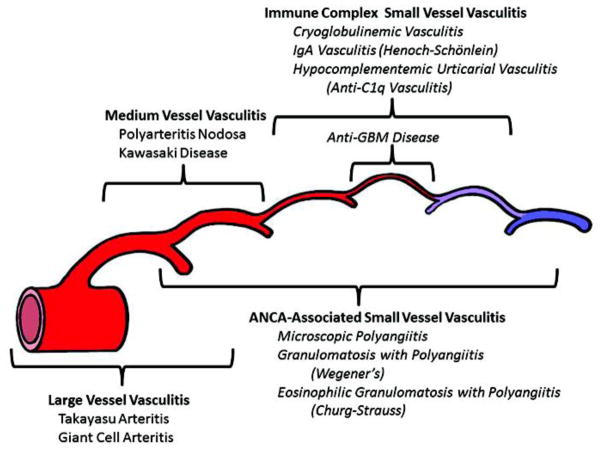
10 ANCA-Associated Vasculitis (GPA, MPA, EGPA) Vasculitis
The ANCA-associated vasculitides (AAVs) are small-vessel necrotizing vasculitides with few or no immune complex deposits, associated with antineutrophil cytoplasmic antibodies (ANCA). The three entities are granulomatosis with polyangiitis (GPA, formerly Wegener), microscopic polyangiitis (MPA), and eosinophilic granulomatosis with polyangiitis (EGPA, formerly Churg-Strauss).
GPA (Wegener)
Classic triad: upper airway (chronic sinusitis, saddle-nose deformity, subglottic stenosis, nasal crusting/bleeding), lower airway (pulmonary nodules, cavities, alveolar hemorrhage), and glomerulonephritis (rapidly progressive, pauci-immune crescentic). Strongly associated with c-ANCA / anti-PR3.
MPA
Small-vessel vasculitis without granulomas; dominated by rapidly progressive glomerulonephritis and pulmonary capillaritis/alveolar hemorrhage. Strongly associated with p-ANCA / anti-MPO.
EGPA (Churg-Strauss)
Three phases: allergic (asthma, rhinitis), eosinophilic (eosinophilia, eosinophilic infiltrates), and vasculitic (mononeuritis multiplex, cutaneous vasculitis, cardiac involvement). Peripheral eosinophilia is characteristic. Only 30–40% are ANCA-positive (usually p-ANCA/MPO).
Management
Induction with glucocorticoids plus either rituximab or cyclophosphamide per the 2021 ACR/VF ANCA vasculitis guideline; rituximab is non-inferior and preferred in relapsing disease and in women of childbearing age. Plasma exchange may be considered in severe pulmonary-renal disease. Maintenance with rituximab, azathioprine, or methotrexate. Mepolizumab (anti-IL-5) is approved for EGPA. Avacopan (C5a receptor antagonist) has a steroid-sparing role.
11 Large-Vessel Vasculitis (GCA, Takayasu) & PMR Vasculitis
Giant Cell Arteritis (GCA)
GCA is a granulomatous vasculitis of large and medium-sized arteries, particularly the cranial branches of the external carotid. It affects adults > 50 (almost always > 70) and is the most common primary vasculitis of the elderly.
Clinical features: new-onset temporal headache, scalp tenderness, jaw claudication, visual symptoms (amaurosis fugax or sudden permanent vision loss from anterior ischemic optic neuropathy), and constitutional symptoms. Up to half of GCA patients also have polymyalgia rheumatica. ESR and CRP are markedly elevated. Temporal artery biopsy remains the diagnostic gold standard; vascular ultrasound looking for the halo sign is increasingly used. The 2022 ACR/EULAR GCA classification criteria provide the updated framework.
Management: start high-dose glucocorticoids immediately on clinical suspicion — do not wait for biopsy if vision is threatened. 40–60 mg prednisone daily for uncomplicated GCA; IV methylprednisolone 1 g daily × 3 for visual involvement. Tocilizumab (IL-6R inhibitor) is an approved steroid-sparing agent that achieves sustained remission at lower cumulative steroid doses.
Takayasu Arteritis
Takayasu is a granulomatous vasculitis of the aorta and its major branches, affecting younger women (< 40, often much younger). Patients present with constitutional symptoms, limb claudication, absent or asymmetric pulses ("pulseless disease"), blood pressure discrepancies between arms, bruits, and sometimes stroke or renovascular hypertension. MRA or CTA of the aorta and great vessels is the imaging of choice. Management is high-dose glucocorticoids plus a steroid-sparing agent (methotrexate, azathioprine, TNFi, tocilizumab).
Polymyalgia Rheumatica (PMR)
PMR is a clinical syndrome of age > 50 with bilateral shoulder and/or hip girdle pain and stiffness (worse in the morning, lasting > 45 minutes), elevated inflammatory markers, and a rapid response to low-dose prednisone (15–20 mg/day). It frequently overlaps with GCA, and every PMR patient must be screened clinically for GCA features at every visit. The 2012 ACR/EULAR PMR provisional classification criteria are the standard reference.
12 Small- & Medium-Vessel Vasculitis Vasculitis
Beyond ANCA-associated vasculitis, the small- and medium-vessel vasculitides include several clinically important entities:
- IgA vasculitis (Henoch-Schönlein purpura): palpable purpura (lower extremities), arthritis, abdominal pain, and IgA nephropathy. Most common in children. Self-limited in most cases.
- Cryoglobulinemic vasculitis: immune complex deposition of cryoglobulins; classic triad of palpable purpura, arthralgia, and weakness; often from hepatitis C. Types I (monoclonal), II (mixed), III (polyclonal).
- Hypersensitivity vasculitis / cutaneous small-vessel vasculitis: drug- or infection-triggered small-vessel vasculitis limited to the skin.
- Polyarteritis nodosa (PAN): necrotizing vasculitis of medium-sized arteries without glomerulonephritis (key distinction from MPA); associated with hepatitis B. Presents with constitutional symptoms, mononeuritis multiplex, skin ulcers, livedo, mesenteric ischemia, and renal infarction (not glomerulonephritis). Angiography classically shows microaneurysms.
- Kawasaki disease: medium-vessel vasculitis of childhood; prolonged fever, conjunctivitis, rash, extremity changes, cervical lymphadenopathy; coronary aneurysm risk; treated with IVIG and aspirin.
13 Gout & Pseudogout (CPPD) Crystal
Crystal arthropathies are among the most common reasons for acute rheumatology consultation. Definitive diagnosis requires joint aspiration with crystal analysis under polarized microscopy.
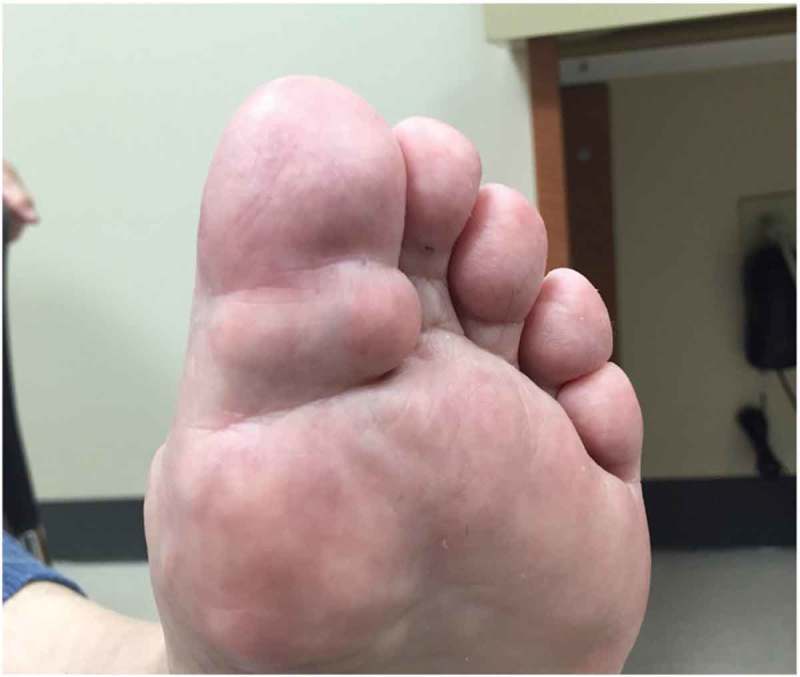
Gout
Gout results from deposition of monosodium urate (MSU) crystals in joints and soft tissues due to chronic hyperuricemia (serum urate > 6.8 mg/dL, the solubility limit). Risk factors include male sex, obesity, diet (purines, alcohol, fructose), diuretics, CKD, and genetics. Presentations include acute gout flare (rapid-onset monoarticular arthritis, classically first MTP — podagra), intercritical gout, chronic tophaceous gout, and urate nephrolithiasis. Polarized microscopy shows needle-shaped, negatively birefringent crystals.
Management (2020 ACR gout guideline):
- Acute flare: NSAIDs, colchicine, or glucocorticoids (oral, intra-articular, or IM). Start within 24 hours.
- Urate-lowering therapy (ULT): indicated for ≥ 2 flares/year, tophi, radiographic damage, or urate nephrolithiasis. Allopurinol is first-line (start low 100 mg/day and titrate to target serum urate < 6 mg/dL); HLA-B*58:01 screening recommended in high-risk populations before starting. Febuxostat is an alternative. Pegloticase is for refractory tophaceous gout. Always use anti-inflammatory prophylaxis (colchicine 0.6 mg daily or low-dose NSAID) during the first 3–6 months of ULT to prevent paradoxical flares.
Entry: at least one episode of swelling, pain, or tenderness in a peripheral joint or bursa. Sufficient criterion: MSU crystals present in a symptomatic joint, bursa, or tophus. Otherwise a weighted score across clinical (pattern, time course, tophi), lab (serum urate, synovial analysis), and imaging (ultrasound double contour sign, DECT urate deposition, X-ray erosions) domains; score ≥ 8 classifies gout.
Calcium Pyrophosphate Deposition (CPPD / Pseudogout)
CPPD is caused by deposition of calcium pyrophosphate dihydrate crystals. Presentations include asymptomatic chondrocalcinosis (seen on X-ray as linear calcification in cartilage, especially meniscus and triangular fibrocartilage of the wrist), acute CPP crystal arthritis ("pseudogout," often knee or wrist), chronic CPP inflammatory arthritis (can mimic RA), and OA with CPPD. Associations: hyperparathyroidism, hemochromatosis, hypomagnesemia, hypophosphatasia. Crystals are rhomboid, weakly positively birefringent. Treatment of acute attacks mirrors gout (NSAIDs, colchicine, steroids); no urate-lowering equivalent exists.
14 Spondyloarthropathies (AS, PsA, ReA, IBD-SpA) Spondyloarthritis
The spondyloarthropathies (SpA) are a family of seronegative (RF-negative) inflammatory arthritides sharing HLA-B27 association, axial skeleton involvement, enthesitis, dactylitis, uveitis, and extra-articular features. Subtypes: ankylosing spondylitis (AS), psoriatic arthritis (PsA), reactive arthritis (ReA), IBD-associated arthritis, and undifferentiated SpA.
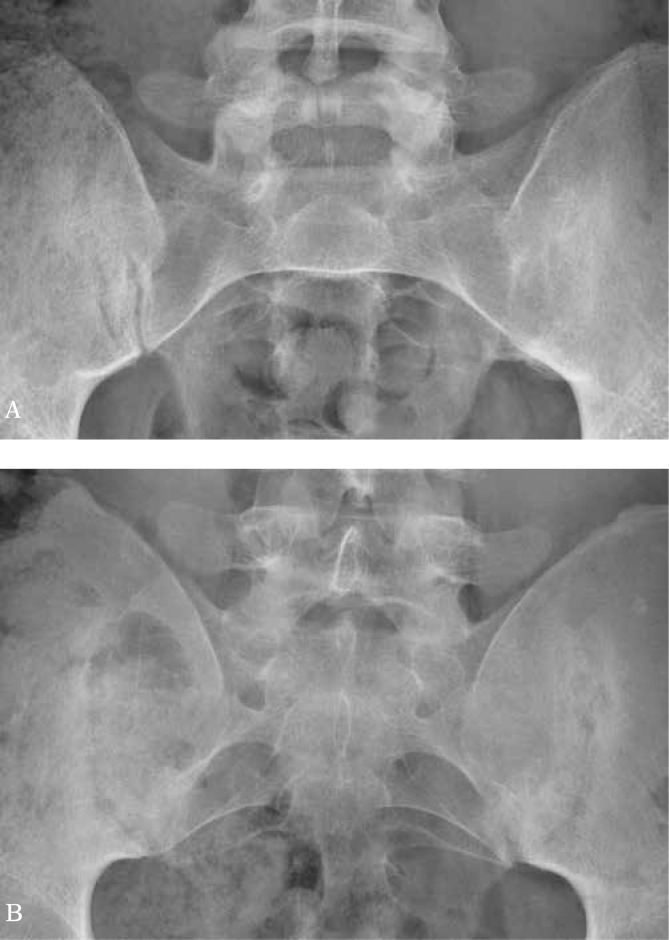
Ankylosing Spondylitis / Axial Spondyloarthritis
AS is chronic inflammation of the axial skeleton, particularly the sacroiliac joints and spine. Classic presentation: young adult man with inflammatory back pain (insidious, age < 45, > 3 months, morning stiffness > 30 minutes, improves with exercise not rest, nocturnal awakening). Exam shows reduced chest expansion, positive Schober test, and tenderness at the SI joints. Late findings include the fused "bamboo spine." Extra-articular features include anterior uveitis (most common), aortitis, IBD, and psoriasis. The ASAS axial SpA classification criteria branch into an imaging arm (MRI active sacroiliitis or radiographic sacroiliitis) and a clinical arm (HLA-B27 + additional SpA features).
Management: NSAIDs (first-line, often continuous for symptomatic disease), physical therapy, TNF inhibitors (all five are effective), IL-17 inhibitors (secukinumab, ixekizumab), and JAK inhibitors (upadacitinib, tofacitinib). csDMARDs do not work for axial disease.
Psoriatic Arthritis (PsA)
PsA affects roughly 30% of psoriasis patients and has several patterns: asymmetric oligoarthritis, symmetric polyarthritis mimicking RA, DIP-predominant, axial (AS-like), and the destructive arthritis mutilans ("pencil-in-cup" deformity on X-ray). Features include nail pitting and onycholysis, dactylitis ("sausage digit"), enthesitis (especially Achilles, plantar fascia), and uveitis. The CASPAR criteria are the standard for classification.
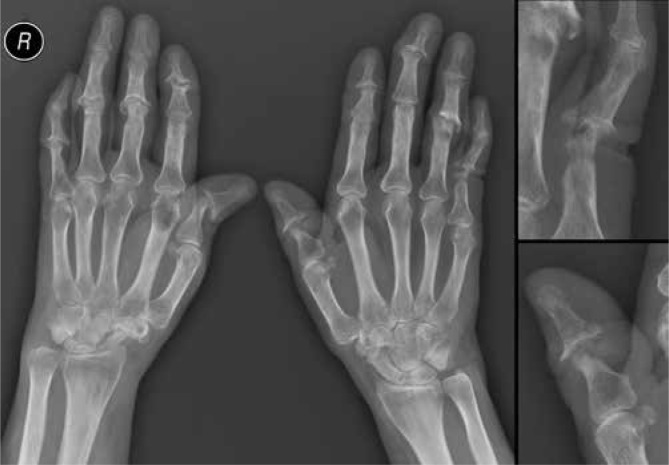
Management includes NSAIDs, csDMARDs (methotrexate, sulfasalazine, leflunomide — mostly for peripheral disease), TNFi, IL-17 inhibitors, IL-12/23 (ustekinumab), IL-23 (guselkumab, risankizumab), abatacept, apremilast (PDE4 inhibitor), and JAK inhibitors.
Reactive Arthritis (ReA)
ReA is a sterile post-infectious oligoarthritis occurring days to weeks after GI (Salmonella, Shigella, Campylobacter, Yersinia) or GU (Chlamydia trachomatis) infection. Classic triad ("can't see, can't pee, can't climb a tree"): conjunctivitis, urethritis, arthritis. Keratoderma blennorrhagicum and circinate balanitis are characteristic skin findings. Most cases resolve within 6 months. NSAIDs first; csDMARDs or TNFi for chronic disease.
IBD-Associated Arthritis (Enteropathic)
Inflammatory arthritis occurs in up to 20% of IBD patients. Type 1 is pauciarticular and parallels IBD flares; type 2 is polyarticular and runs independently. Axial involvement (sacroiliitis, spondylitis) is also common and does not parallel bowel disease. TNF inhibitors that treat both gut and joints (infliximab, adalimumab) are preferred; avoid IL-17i in IBD because they can worsen colitis.
15 Juvenile Idiopathic Arthritis & Adult-Onset Still Disease Autoinflammatory
Juvenile Idiopathic Arthritis (JIA)
JIA is the umbrella term for chronic arthritis of unknown cause beginning before age 16 and lasting > 6 weeks. ILAR subtypes: oligoarticular (≤ 4 joints), polyarticular RF-negative, polyarticular RF-positive, systemic (Still disease — quotidian fevers, salmon-colored rash, hepatosplenomegaly, lymphadenopathy, serositis), enthesitis-related (ERA, an early axSpA), psoriatic JIA, and undifferentiated. Uveitis screening (especially ANA-positive oligoarticular JIA) is mandatory. Macrophage activation syndrome (MAS) is a life-threatening cytokine storm complication of systemic JIA.
Adult-Onset Still Disease (AOSD)
AOSD is the adult equivalent of systemic JIA: quotidian (daily) fever spikes, evanescent salmon-pink rash that comes with the fever, arthritis, sore throat, lymphadenopathy, hepatosplenomegaly, extreme hyperferritinemia (often > 10,000 ng/mL), neutrophilic leukocytosis, and transaminitis. Management includes NSAIDs, glucocorticoids, methotrexate, and IL-1 (anakinra, canakinumab) or IL-6 (tocilizumab) inhibitors.
16 Fibromyalgia Functional
Fibromyalgia is a chronic central pain amplification syndrome characterized by widespread musculoskeletal pain, fatigue, non-restorative sleep, cognitive dysfunction ("fibro fog"), and high rates of comorbid mood and anxiety disorders. It is not inflammatory, not autoimmune, and not joint destructive — but it is extremely common in rheumatology clinic, both as a primary diagnosis and as an overlay on other rheumatologic diseases.
Diagnosis
The 2016 revised ACR criteria use the Widespread Pain Index (WPI) and the Symptom Severity Scale (SSS). Labs are normal by definition; extensive autoantibody testing is discouraged because false positives lead to misdiagnosis. The tender-point exam is no longer required.
Management
First-line is non-pharmacologic: patient education, aerobic and strength exercise, cognitive behavioral therapy, and sleep optimization. Pharmacologic options include duloxetine, milnacipran, pregabalin, gabapentin, and low-dose amitriptyline. Opioids and long-term NSAIDs are avoided.
17 IgG4-Related Disease, Sarcoidosis, Behçet & Relapsing Polychondritis Multisystem
IgG4-Related Disease
IgG4-RD is a fibroinflammatory disease characterized by tumefactive lesions, lymphoplasmacytic infiltrates rich in IgG4+ plasma cells, storiform fibrosis, and obliterative phlebitis on biopsy. Can affect virtually any organ: pancreas (autoimmune pancreatitis type 1), salivary glands (Mikulicz disease, Küttner tumor), lacrimal glands, retroperitoneum (retroperitoneal fibrosis), aorta (periaortitis), kidneys, thyroid (Riedel), and lung. Serum IgG4 is often but not always elevated. Treatment is glucocorticoids and rituximab.
Sarcoidosis (Rheumatologic Overlap)
Sarcoidosis is a multisystem granulomatous disease that rheumatology sees because of arthritis, uveitis, and parenchymal involvement. Löfgren syndrome (acute sarcoid) presents with the triad of bilateral hilar lymphadenopathy, erythema nodosum, and migratory ankle arthritis — often self-limited. Chronic sarcoid arthritis is more insidious. Treatment ranges from NSAIDs for mild disease to glucocorticoids, methotrexate, and TNFi for severe systemic involvement.
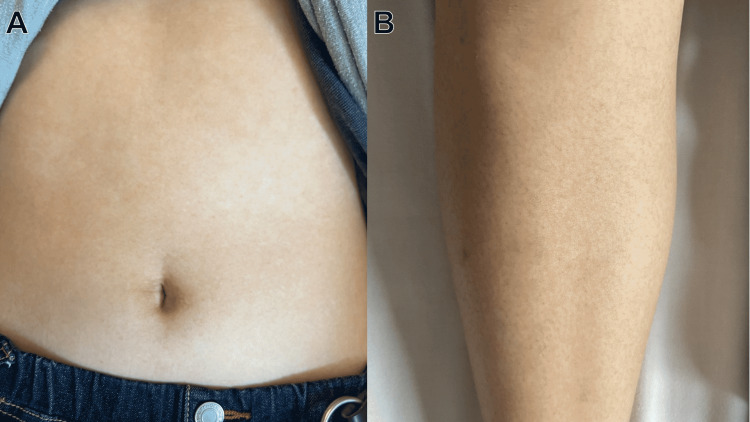
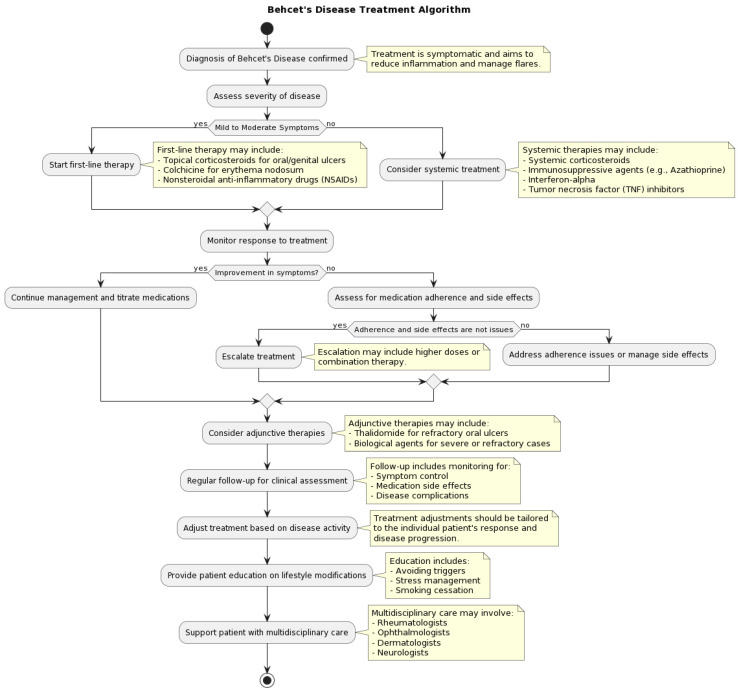
Behçet Disease
Behçet is a variable-vessel vasculitis characterized by recurrent oral aphthae, genital ulcers, uveitis (often severe posterior or panuveitis), skin lesions (erythema nodosum, pseudofolliculitis), and pathergy reaction. Venous and arterial thrombosis, CNS involvement, and GI involvement can occur. Most common along the "Silk Road." Management ranges from colchicine for mucocutaneous disease to glucocorticoids, azathioprine, TNFi, and apremilast for systemic disease.
Relapsing Polychondritis
Autoimmune attack on cartilage: auricular chondritis (painful, red, swollen ear sparing the lobule), nasal chondritis (saddle-nose deformity), laryngotracheal chondritis (airway collapse — life-threatening), and inflammatory eye and ear disease. Treatment is glucocorticoids and steroid-sparing agents.
18 Septic Arthritis & Osteoporosis Overlap Overlap
Septic Arthritis
Septic arthritis is a rheumatologic emergency. Any acutely hot, swollen, painful joint — especially monoarticular — must be aspirated before steroid injection to exclude infection. Classic organisms: Staphylococcus aureus (most common overall), streptococci, gram-negatives in the elderly and immunocompromised, Neisseria gonorrhoeae in sexually active young adults (often disseminated with tenosynovitis, dermatitis, and migratory polyarthralgia). Synovial fluid in septic arthritis shows WBC typically > 50,000 with > 75% neutrophils, positive gram stain (only 50%), and positive culture. Management is urgent drainage (surgical or arthroscopic washout) plus IV antibiotics. Patients on biologics are at elevated risk and must be screened at every visit for infection.
Osteoporosis in Rheumatology
Rheumatology patients have high osteoporosis risk because of inflammatory disease, glucocorticoid exposure, and immobility. Glucocorticoid-induced osteoporosis is covered by the 2017 ACR GIOP guideline. Document bone density (DEXA T-score), FRAX score, calcium/vitamin D supplementation, and current therapy (bisphosphonates, denosumab, teriparatide/abaloparatide, romosozumab).
19 Procedures — Injections, Aspirations & Biopsies Procedural
Procedures in rheumatology are primarily diagnostic joint aspiration (arthrocentesis) and therapeutic joint or soft-tissue injection. You will document consent, site, approach, medication, volume, fluid appearance, complications, and patient tolerance for every procedure.
| Procedure | Indication | Key Scribe Details |
|---|---|---|
| Knee arthrocentesis | Effusion for diagnosis (crystals, infection) or therapeutic drainage | Superolateral or medial mid-patellar approach; document fluid volume, appearance, labs sent |
| Knee injection | OA, inflammatory arthritis flare, bursitis | Triamcinolone 40 mg or methylprednisolone 40–80 mg with lidocaine |
| Shoulder injection (glenohumeral, subacromial, AC) | OA, rotator cuff impingement, adhesive capsulitis, AC arthropathy | Posterior approach most common; ultrasound guidance improves accuracy |
| Hip injection | Trochanteric bursitis (common), intra-articular OA (image-guided) | Intra-articular hip injections require fluoroscopy or ultrasound |
| Wrist, MCP, PIP injection | RA, PsA, OA flare | Small volumes; dorsal approach; triamcinolone 10–20 mg |
| Ankle, subtalar, MTP injection | OA, gout, inflammatory arthritis | Small-joint technique; fluoroscopy often used |
| Elbow injection | Lateral epicondylitis, olecranon bursitis, RA | Document site specifically (joint vs bursa vs epicondyle) |
| Trigger point injection | Myofascial pain | Local anesthetic ± low-dose steroid into taut band |
| Tendon sheath injection | De Quervain tenosynovitis, trigger finger | Avoid intratendinous injection; small volume |
| Nailfold capillaroscopy | Raynaud evaluation, SSc vs primary Raynaud | Document capillary density, dilation, dropout, hemorrhages |
| Minor salivary gland (lip) biopsy | Sjögren evaluation | Referred to oral surgery or ENT; focus score ≥ 1 is the histologic threshold |
| Temporal artery biopsy | GCA evaluation | Referred to vascular or general surgery; do not delay steroids |
Consent obtained after discussion of risks, benefits, and alternatives. Joint/site identified and marked. Skin prepped with chlorhexidine in sterile fashion. Under sterile technique, a [gauge] needle was introduced via [approach]. [Fluid aspirated — volume, color, clarity; sent for cell count, crystals, gram stain, culture]. [Medication: triamcinolone __ mg in __ mL of 1% lidocaine] was injected without difficulty. Needle withdrawn, hemostasis achieved, bandage applied. Patient tolerated the procedure well with no immediate complications. Post-procedure precautions reviewed.
When documenting arthrocentesis, always note if the fluid was sent for crystals and culture. A missing crystal result on a flare note is the most common omission that will get your note sent back. Also note the post-procedure plan for steroid injections in diabetics — expect a transient glucose rise.
20 Labs, Serologies & Imaging Diagnostics
Core Labs
Every rheumatology visit typically includes CBC (anemia of chronic disease, leukopenia in SLE, thrombocytopenia), CMP (renal function, LFTs for medication monitoring), UA (proteinuria and hematuria for lupus nephritis, vasculitis), ESR, and CRP. ESR is slower and influenced by age, sex, and anemia; CRP is more acute and hepatically driven. Both can be normal in active disease and should never be interpreted in isolation.
Autoantibodies
| Antibody | Disease Association | Interpretation Notes |
|---|---|---|
| ANA | SLE (>95%), SSc, Sjögren, MCTD, DM, AIH, many others | Screening test; pattern (homogeneous, speckled, nucleolar, centromere) helps narrow; titer matters (> 1:160 more meaningful) |
| Anti-dsDNA | SLE (specific) | Correlates with lupus nephritis activity |
| Anti-Sm | SLE (specific) | Low sensitivity, very high specificity |
| Anti-Ro/SSA, Anti-La/SSB | Sjögren, SLE, neonatal lupus (congenital heart block risk) | Must document if pregnant |
| Anti-U1-RNP | MCTD (high titer) | Can be present in SLE at lower titers |
| Anti-Scl-70 (topoisomerase I) | Diffuse cutaneous SSc, ILD | Poor prognosis marker |
| Anti-centromere | Limited cutaneous SSc (CREST) | PAH association |
| Anti-RNA polymerase III | Diffuse SSc, renal crisis, malignancy | Aggressive skin disease |
| Anti-Jo-1 (and other anti-synthetases) | Antisynthetase syndrome | Myositis + ILD + mechanic's hands + arthritis + Raynaud |
| Anti-Mi-2, anti-MDA5, anti-TIF1γ, anti-NXP2, anti-SRP, anti-HMGCR | DM / PM / IMNM subsets | Myositis-specific antibodies; each carries prognostic meaning |
| RF | RA, Sjögren, cryoglobulinemia, chronic infection | Sensitivity ~70% in RA; not specific |
| Anti-CCP (ACPA) | RA (specific) | More specific than RF; erosive disease predictor |
| c-ANCA / anti-PR3 | GPA | Strongly associated |
| p-ANCA / anti-MPO | MPA, EGPA, drug-induced vasculitis | Strongly associated |
| Antiphospholipid panel (LA, aCL, anti-β2GPI) | APS, SLE | Must be confirmed ≥ 12 weeks apart |
| Complement (C3, C4, CH50) | SLE, cryoglobulinemia | Low in active disease |
| HLA-B27 | AS, axSpA, reactive arthritis, anterior uveitis | Supports diagnosis in clinical context |
Imaging
Plain radiographs remain the first-line imaging for inflammatory arthritis; look for periarticular osteopenia, joint space narrowing, and erosions (RA), chondrocalcinosis (CPPD), sacroiliitis and syndesmophytes (AS), pencil-in-cup (PsA), and tophi/overhanging edges (gout). Musculoskeletal ultrasound with power Doppler is used extensively to detect subclinical synovitis, tenosynovitis, enthesitis, tophi (double contour sign), and to guide injections. MRI is the most sensitive modality for early erosions, bone marrow edema, sacroiliitis, and myositis. CT angiography or MRA is used for large-vessel vasculitis. DECT (dual-energy CT) can non-invasively identify urate deposition in chronic gout. DEXA assesses bone density.
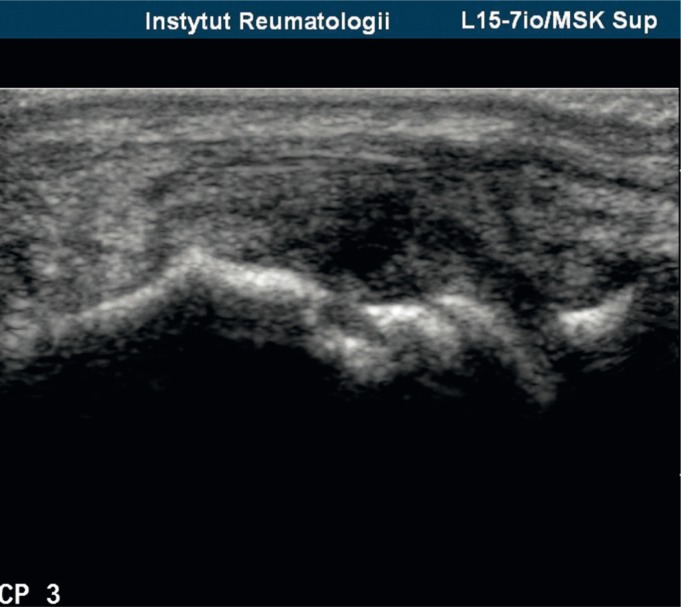
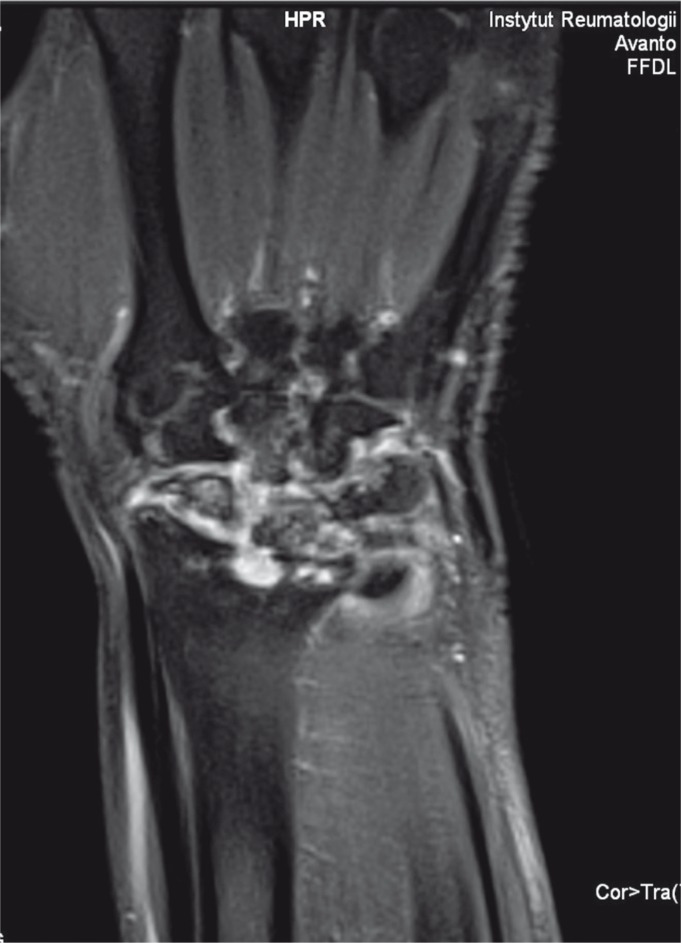
21 Medications — DMARDs, Biologics & Beyond Medications
Rheumatology is a medication-heavy specialty. You must know every drug class, mechanism, brand name, and monitoring requirement. All biologics and JAK inhibitors require baseline screening for TB (QuantiFERON or PPD), hepatitis B and C, and HIV, and a review of vaccination status. Live vaccines are contraindicated while on biologics and JAK inhibitors.
NSAIDs
Non-selective (ibuprofen, naproxen, indomethacin, diclofenac, meloxicam) and COX-2 selective (celecoxib). Used for symptomatic relief in OA, crystal arthritis, and spondyloarthritis. GI, renal, and cardiovascular toxicity limit long-term use.
Glucocorticoids
Prednisone, prednisolone, methylprednisolone, dexamethasone. Potent anti-inflammatory and immunosuppressive. Used as bridge therapy (RA), first-line induction (vasculitis, myositis, SLE flare, GCA, PMR), and intra-articular injections. Chronic toxicities: osteoporosis, diabetes, hypertension, weight gain, infection, cataracts, avascular necrosis, adrenal suppression.
Conventional Synthetic DMARDs (csDMARDs)
| Drug | Typical Dose | Key Indications | Monitoring / Notes |
|---|---|---|---|
| Methotrexate (Trexall, Rheumatrex, Otrexup) | 15–25 mg weekly PO/SQ | RA, PsA, JIA, myositis, vasculitis maintenance | CBC, LFTs, Cr every 1–3 mo; folic acid 1 mg daily; teratogenic; avoid alcohol; hepatotoxicity, pneumonitis |
| Hydroxychloroquine (Plaquenil) | ≤ 5 mg/kg/day (actual body weight) | SLE, Sjögren, RA, mild PsA | Baseline and annual ophthalmology screening; safe in pregnancy |
| Sulfasalazine (Azulfidine) | 2–3 g/day | RA, peripheral PsA, IBD-SpA | G6PD check; CBC, LFTs; sulfa allergy |
| Leflunomide (Arava) | 10–20 mg daily | RA, PsA | LFTs; long half-life (cholestyramine washout); teratogenic |
| Azathioprine (Imuran) | 1–3 mg/kg/day | SLE, vasculitis, IBD, myositis maintenance | TPMT activity before starting; CBC, LFTs |
| Mycophenolate mofetil (CellCept) | 1–3 g/day divided | Lupus nephritis, SSc-ILD, myositis, vasculitis | CBC; teratogenic; GI side effects |
| Cyclophosphamide (Cytoxan) | IV or PO pulse | Severe SLE, ANCA vasculitis induction | CBC, UA (hemorrhagic cystitis), mesna, infertility risk, malignancy risk |
| Cyclosporine / Tacrolimus | Variable | Lupus nephritis, refractory disease | BP, Cr, drug levels |
| Colchicine | 0.6 mg BID or daily | Gout, CPPD, FMF, Behçet, pericarditis | Dose reduce in CKD; avoid with strong CYP3A4 / P-gp inhibitors |
Biologic DMARDs (bDMARDs)
| Target | Drugs | Primary Uses |
|---|---|---|
| TNF-α | Etanercept (Enbrel), Adalimumab (Humira), Infliximab (Remicade), Certolizumab (Cimzia), Golimumab (Simponi) | RA, PsA, AS, JIA, IBD (not etanercept), uveitis |
| IL-6 receptor | Tocilizumab (Actemra), Sarilumab (Kevzara) | RA, GCA, JIA, CRS; tocilizumab for systemic sclerosis |
| IL-1 | Anakinra (Kineret), Canakinumab (Ilaris), Rilonacept (Arcalyst) | AOSD, systemic JIA, gout (refractory), CAPS, FMF |
| IL-17A | Secukinumab (Cosentyx), Ixekizumab (Taltz), Bimekizumab (Bimzelx) | PsA, AS, psoriasis; avoid in IBD |
| IL-12/23 (p40) | Ustekinumab (Stelara) | PsA, psoriasis, IBD |
| IL-23 (p19) | Guselkumab (Tremfya), Risankizumab (Skyrizi) | PsA, psoriasis |
| CTLA-4-Ig (T-cell co-stim) | Abatacept (Orencia) | RA, PsA, JIA |
| Anti-CD20 (B cell) | Rituximab (Rituxan) | RA, ANCA vasculitis, SLE (off-label), Sjögren, myositis, IgG4-RD |
| BLyS/BAFF | Belimumab (Benlysta) | SLE, lupus nephritis |
| Type I IFN receptor | Anifrolumab (Saphnelo) | SLE |
| IL-5 | Mepolizumab (Nucala) | EGPA |
| C5a receptor | Avacopan (Tavneos) | ANCA vasculitis (steroid-sparing) |
Targeted Synthetic DMARDs (tsDMARDs)
JAK inhibitors (JAKi): tofacitinib (Xeljanz), baricitinib (Olumiant), upadacitinib (Rinvoq). Used in RA, PsA, AS, JIA, AD, UC, alopecia areata. The FDA JAKi boxed warning covers MACE, malignancy, thrombosis, and mortality signals in higher-risk patients. Apremilast (Otezla): PDE4 inhibitor for PsA, psoriasis, Behçet.
Urate-Lowering & Gout-Specific
Allopurinol (Zyloprim): xanthine oxidase inhibitor, first-line ULT. Febuxostat (Uloric): alternative XOI; CV warning. Probenecid: uricosuric; avoid in CKD and nephrolithiasis. Pegloticase (Krystexxa): recombinant uricase for refractory tophaceous gout; infusion-only; methotrexate co-therapy reduces anti-drug antibodies.
Vaccinations & Pregnancy
The 2022 ACR vaccination guideline and the 2020 ACR reproductive health guideline are the standard references for immunization and pregnancy planning in patients on immunosuppressives. Key points: inactivated flu, pneumococcal, COVID-19, and recombinant zoster vaccines are recommended; live vaccines (MMR, live zoster, yellow fever) are contraindicated on biologics/JAKi; HCQ, sulfasalazine, azathioprine, and certain TNFi are considered pregnancy-compatible; methotrexate, mycophenolate, cyclophosphamide, and leflunomide are teratogenic and must be held before conception.
22 Classification Systems — Full Enumeration Reference
| Category | DAS28-ESR / CRP | Clinical Meaning |
|---|---|---|
| Remission | < 2.6 | Treatment target |
| Low | 2.6 – 3.2 | Acceptable if sustained |
| Moderate | > 3.2 – 5.1 | Escalate therapy |
| High | > 5.1 | Escalate therapy |
| Grade | Meaning |
|---|---|
| A | Severe active disease requiring major therapy change |
| B | Moderate activity requiring treatment adjustment |
| C | Mild stable activity |
| D | Previously involved, now inactive |
| E | Never involved |
| Class | Histology | Clinical Correlation |
|---|---|---|
| I | Minimal mesangial | Normal UA, no treatment |
| II | Mesangial proliferative | Mild proteinuria/hematuria |
| III | Focal proliferative (< 50% glomeruli) | Induction immunosuppression |
| IV | Diffuse proliferative (≥ 50% glomeruli) | Most severe; aggressive induction |
| V | Membranous | Nephrotic-range proteinuria |
| VI | Advanced sclerosing | ≥ 90% globally sclerosed; ESRD course |
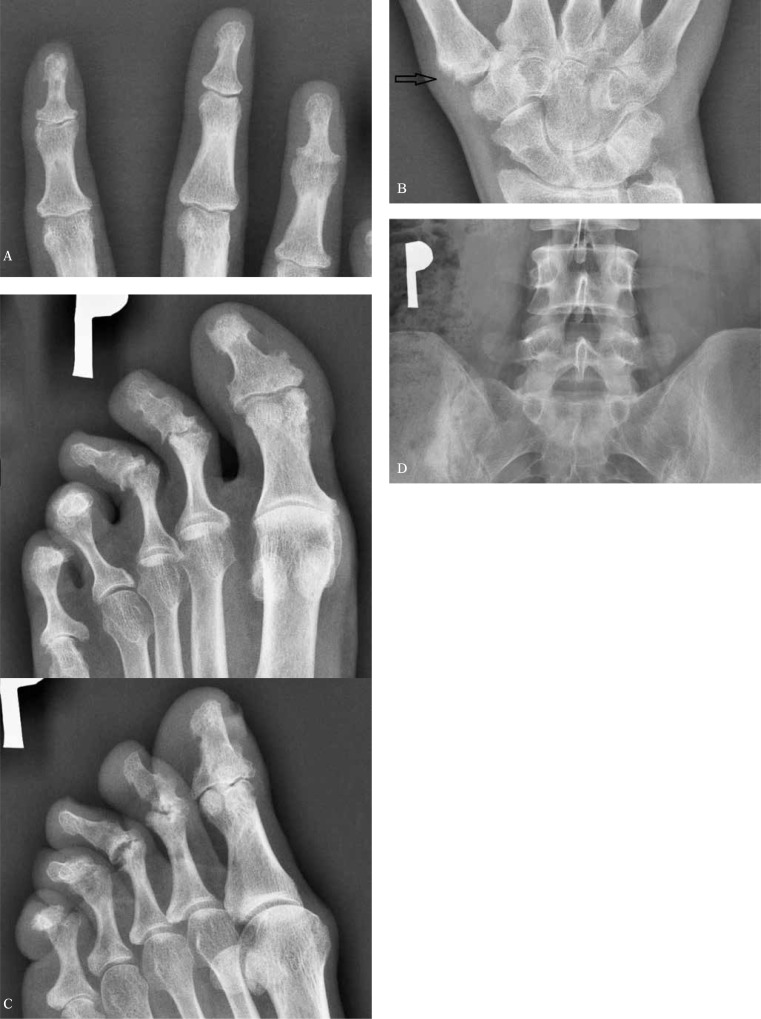
Entry: established inflammatory articular disease (joint, spine, or entheseal). Score ≥ 3 points from:
- Current psoriasis (2 points); personal history of psoriasis (1); family history of psoriasis (1)
- Typical psoriatic nail dystrophy (pitting, onycholysis, hyperkeratosis) (1)
- Negative RF (1)
- Current dactylitis or history of dactylitis documented by a rheumatologist (1)
- Radiographic evidence of juxta-articular new bone formation (1)
For patients with back pain ≥ 3 months and age of onset < 45, classify as axSpA if:
- Imaging arm: sacroiliitis on imaging (MRI active inflammation or definite X-ray sacroiliitis per modified New York) + ≥ 1 SpA feature
- Clinical arm: HLA-B27 positive + ≥ 2 SpA features
SpA features include inflammatory back pain, arthritis, enthesitis (heel), uveitis, dactylitis, psoriasis, IBD, good NSAID response, family history, HLA-B27, elevated CRP.
Entry: age ≥ 50 with a diagnosis of medium- or large-vessel vasculitis. Score ≥ 6 points classifies GCA. Criteria include positive temporal artery biopsy (+5), halo sign on temporal artery ultrasound (+5), morning stiffness in shoulders/neck, jaw or tongue claudication, new temporal headache, scalp tenderness, abnormal temporal artery on exam, sudden visual loss, and large-vessel involvement on imaging.
Required: age ≥ 50, bilateral shoulder aching, abnormal ESR and/or CRP. Score ≥ 4 (without ultrasound) or ≥ 5 (with ultrasound) from: morning stiffness > 45 min (2), hip pain or limited ROM (1), absence of RF and ACPA (2), absence of peripheral joint pain (1), plus ultrasound criteria.
23 Physical Exam — The Rheumatologic Exam Exam
The rheumatologic exam is the most specialized part of documentation. The physician performs a focused joint and skin exam every visit, and you need to capture it accurately and efficiently.
General: Well-appearing, no acute distress.
Skin: No rash. No nodules. No Raynaud changes. Nailfolds normal.
HEENT: No oral ulcers. Salivary pools normal. No scalp tenderness.
Cardiopulm: RRR, no rubs. Lungs clear to auscultation; no crackles.
Musculoskeletal:
Cervical spine: Full ROM, non-tender.
Shoulders: Full ROM bilaterally, no impingement, no effusion.
Elbows: Full ROM, no effusion, no nodules.
Wrists: Non-tender, no synovitis, no deformity.
MCPs 1–5: Non-tender, no synovitis bilaterally.
PIPs 1–5: Non-tender, no synovitis bilaterally.
DIPs 1–5: Non-tender, no Heberden nodes.
Hips: Full ROM, no groin pain with FABER.
Knees: No effusion, no warmth, stable ligaments.
Ankles: No effusion, non-tender.
MTPs: Non-tender, no synovitis. No podagra.
Entheses: Achilles and plantar fascia non-tender.
TJC 0/28 SJC 0/28
Neuro: Strength 5/5 throughout. Sensation intact.
Beyond joint counts, the rheumatologic exam includes skin (rashes, nodules, digital ulcers, nailfold capillaries, livedo, purpura, calcinosis, sclerodactyly, alopecia), mucosa (oral and nasal ulcers, dry mucous membranes, parotid swelling), eyes (conjunctival injection, uveitis), lungs (crackles of ILD), heart (pericardial rub, murmurs), abdomen (hepatosplenomegaly), and a focused neuro exam for proximal weakness, mononeuritis, or cranial nerve deficits. For spondyloarthritis, document the modified Schober test and chest expansion. For scleroderma, document the modified Rodnan skin score. For myositis, document manual muscle testing (MMT-8).
Learn the homunculus joint map many rheumatologists use to record tender and swollen joints. Each clinic has a preferred form — ask which one to use. Your job is to transcribe the findings into the narrative exam section AND tabulate the joint counts accurately. Always count tender and swollen separately; they are not the same thing.
24 Abbreviations, Sample HPIs & Final Note Reference
Diseases
Labs & Serologies
Medications & Scores
Sample HPIs
"Ms. [Name] is a 43-year-old woman referred by her PCP for evaluation of symmetric polyarthritis. She describes 4 months of progressive bilateral hand and wrist pain with 2 hours of morning stiffness that improves with activity. She reports swelling of the MCPs and PIPs bilaterally and difficulty opening jars and buttoning clothes. She denies rash, oral ulcers, photosensitivity, Raynaud, sicca symptoms, dyspnea, or back pain. ROS positive for fatigue; negative for fever, weight loss, and chest pain. Outside labs show ESR 48, CRP 2.1, RF 96 IU/mL, and anti-CCP > 250 U/mL. Hand X-rays demonstrate periarticular osteopenia without frank erosions. No prior DMARD exposure. She is TB- and hepatitis-screened, UTD on vaccinations, and not pregnant or planning pregnancy."
"Mr. [Name] is a 58-year-old man with a history of HTN on HCTZ, CKD stage 3, obesity, and 3 prior gout flares presenting with 36 hours of severe pain, swelling, and erythema of the right first MTP. Pain began in the early morning and reached a 9/10 by noon. He denies fever, chills, trauma, or recent alcohol binge. On exam the right first MTP is exquisitely tender, swollen, warm, and erythematous — classic podagra. Joint aspiration yielded cloudy yellow fluid; polarized microscopy demonstrated intracellular negatively birefringent needle-shaped crystals, consistent with MSU. Most recent serum urate 8.9 mg/dL. He is not currently on ULT. Plan: oral prednisone taper for this flare given CKD; discuss starting allopurinol with anti-inflammatory prophylaxis once the acute flare settles."
"Ms. [Name] is a 29-year-old woman with SLE (dx 2022, ANA 1:1280 speckled, anti-dsDNA positive, anti-Sm positive, low C3/C4, prior Class IV lupus nephritis in remission on mycophenolate 1500 mg BID and hydroxychloroquine 300 mg daily [4.5 mg/kg]) presenting for routine follow-up. She reports stable energy, no new rashes, no oral ulcers, no arthralgias, no chest pain, no shortness of breath. No recent infections. She is compliant with medications and sunscreen. Last ophthalmology exam 3 months ago — no HCQ retinopathy. Last labs: Cr 0.8, UPCR 0.3, C3 92, C4 18, anti-dsDNA 22 IU/mL (down from 45), CBC normal. SLEDAI-2K = 2. She is using reliable contraception and is not currently planning pregnancy."
"Mr. [Name] is a 52-year-old man referred for evaluation of 6 weeks of polyarticular joint pain involving the knees, ankles, wrists, and fingers. He reports asymmetric involvement, morning stiffness of 20 minutes, and dactylitis of the right third toe. He has a 10-year history of plaque psoriasis on the elbows and scalp with nail pitting. He denies back pain, uveitis, IBD symptoms, or family history of rheumatic disease. On exam, synovitis of the right 3rd DIP, left 2nd PIP, and right knee effusion; dactylitis of the right third toe; nail pitting and onycholysis. RF and anti-CCP negative. Plain films show marginal erosions of the right 3rd DIP. Assessment: psoriatic arthritis meeting CASPAR criteria. Plan: start methotrexate, screen for latent TB and hepatitis, and consider TNFi if inadequate response."
"Mrs. [Name] is a 76-year-old woman presenting with 3 weeks of new bilateral temporal headache, scalp tenderness when combing her hair, jaw pain after a few minutes of chewing, and bilateral shoulder and hip girdle aching with > 1 hour of morning stiffness. She denies vision change, diplopia, fevers, or weight loss. Exam reveals tender, nodular right temporal artery without palpable pulse, bilateral shoulder and hip girdle tenderness with painful abduction, no synovitis. ESR 88, CRP 9.2, Hb 10.8, platelets 540. Temporal artery biopsy scheduled. Prednisone 60 mg daily started today for clinical suspicion of GCA with PMR overlap; she was counseled on red flags for visual loss and instructed to go to the ED immediately if any vision change occurs. PPI and calcium/vitamin D initiated; DEXA ordered; tocilizumab discussed as a steroid-sparing option."
"Ms. [Name] is a 34-year-old woman self-referred for 8 months of diffuse arthralgia, fatigue, and intermittent Raynaud of the fingertips in cold weather. She denies frank joint swelling, morning stiffness, rash, oral ulcers, photosensitivity, sicca symptoms, dyspnea, or weight change. Family history notable for maternal SLE. Outside labs: ANA 1:320 speckled, negative dsDNA, negative Sm, negative RNP, negative Ro/La, normal C3/C4, normal CBC, ESR 14. On exam no synovitis, no rash, normal nailfold capillaries by bedside. Assessment: positive ANA with nonspecific symptoms, not meeting any classification criteria for defined CTD at this time; likely early UCTD vs fibromyalgia overlay. Plan: reassure regarding low current risk, counsel on photoprotection and smoking avoidance, baseline urinalysis, return in 6 months or sooner for any new rash, synovitis, pleuritic chest pain, or cytopenias."
References & Sources
Key guidelines cited above include the 2010 ACR/EULAR RA classification, 2021 ACR RA treatment guideline, 2019 EULAR/ACR SLE classification, 2023 EULAR SLE recommendations, 2019 ACR/AF OA guideline, 2020 ACR gout guideline, 2021 ACR/VF ANCA vasculitis guideline, 2022 ACR/EULAR GCA classification, 2012 ACR/EULAR PMR classification, CASPAR PsA criteria, ASAS axSpA criteria, 2017 EULAR scleroderma recommendations, 2019 EULAR APS guidelines, 2017 ACR GIOP guideline, 2022 ACR vaccination guideline, and the 2020 ACR reproductive health guideline. Figure sources are listed in each figure caption and include Wikimedia Commons, OpenStax College (CC BY 3.0), and Blausen Medical (CC BY 3.0).
The best rheumatology scribes are not just fast typists — they understand the immunologic logic behind the documentation. When a physician says "young woman with malar rash, oral ulcers, pleuritic chest pain, positive ANA and low complement," you should already know this is SLE and start thinking about SLEDAI components, hydroxychloroquine dosing, and the need to rule out nephritis. When they say "70-year-old with new headache, jaw claudication, ESR 90," you should have "GCA" at the top of your problem list and "high-dose prednisone today, biopsy this week" in your plan.
Rheumatology is a longitudinal specialty — pre-populate every visit with prior DMARD history, last disease activity score, last labs, and last imaging. Know medications by both generic and brand names. Know which drugs are teratogenic, which require TB screening, and which are safe in pregnancy. Anticipate the plan based on the diagnosis and the trajectory, not just the current snapshot.
Welcome to rheumatology. The learning curve is steep, the vocabulary is enormous, and the patients are often among the most complex in medicine — but the continuity and the diagnostic puzzles make it one of the most rewarding specialties to scribe.