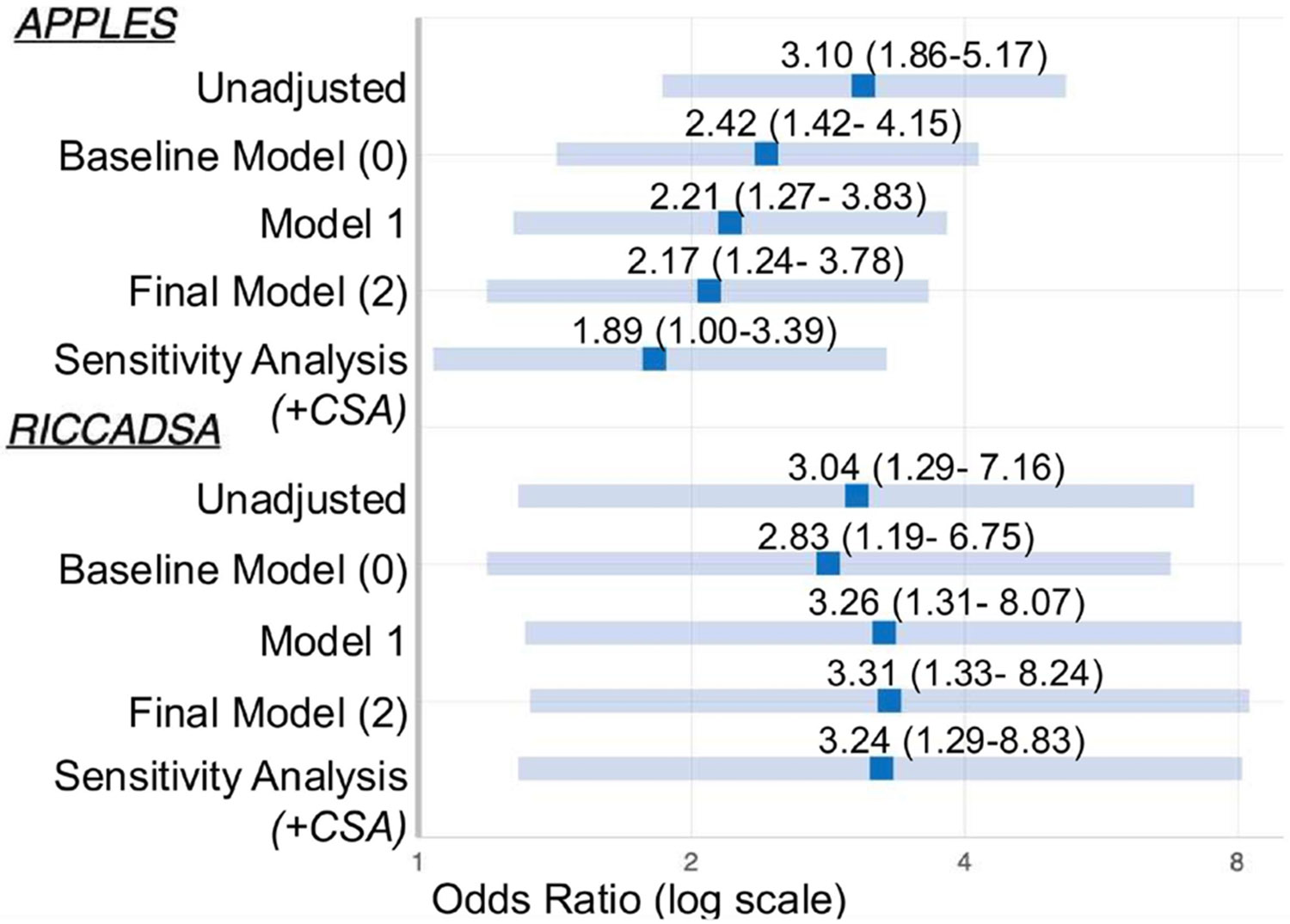
Sleep Medicine
Every diagnosis, sleep study, PAP device, medication, abbreviation, and documentation framework you need to scribe competently in a sleep clinic or sleep lab.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 Sleep Neurophysiology — Essentials
Sleep medicine sits at the intersection of pulmonology, neurology, otolaryngology, and psychiatry. To chart competently, a scribe must understand how normal sleep is organized, what physiologic signals are recorded during a sleep study, and what happens to the airway, brain, and autonomic nervous system when sleep goes wrong.
Sleep Architecture — Stages of Sleep
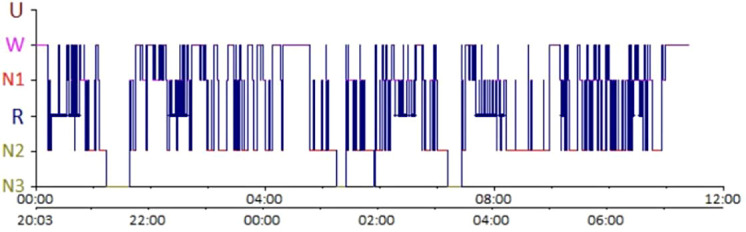
Normal adult sleep is divided into non-REM (NREM) and REM (rapid eye movement) sleep. NREM is further subdivided into three stages — N1 (light transitional sleep), N2 (the bulk of total sleep time, characterized by sleep spindles and K-complexes on EEG), and N3 (slow-wave or “deep” sleep, dominated by delta waves, most restorative). REM sleep is defined by low-amplitude mixed-frequency EEG, rapid conjugate eye movements, and skeletal muscle atonia driven by active inhibition from the brainstem. A healthy adult cycles through NREM and REM approximately every 90–110 minutes, completing 4–6 cycles per night. Slow-wave sleep predominates in the first half of the night; REM periods lengthen toward the morning.
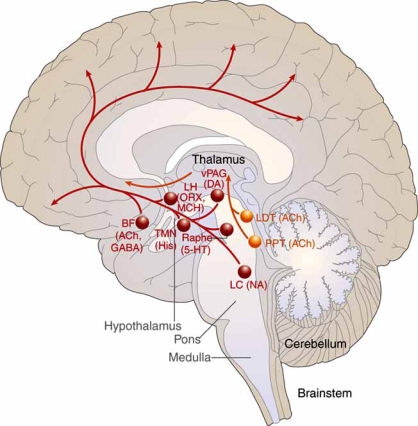
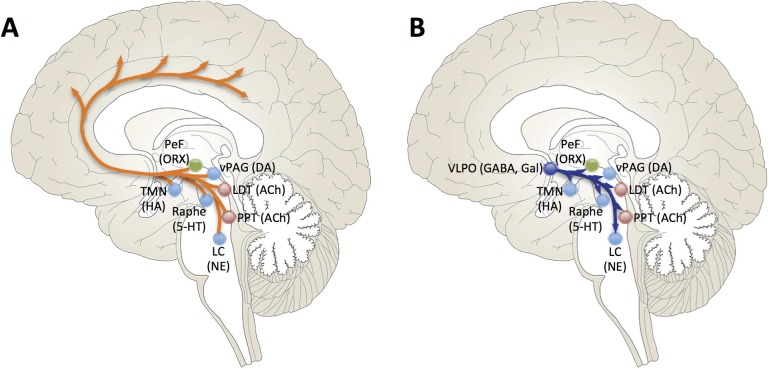
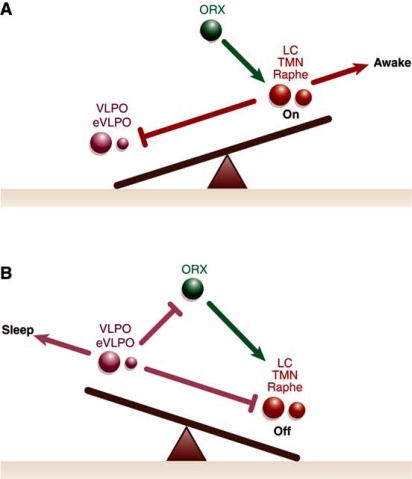
Neuroanatomy of Sleep-Wake Control
Wakefulness is maintained by the ascending reticular activating system (ARAS), a network of brainstem nuclei that release acetylcholine, norepinephrine, serotonin, dopamine, and histamine up into the thalamus and cortex. The lateral hypothalamus adds orexin (hypocretin), a stabilizing neuropeptide whose loss causes narcolepsy type 1. The ventrolateral preoptic nucleus (VLPO) of the anterior hypothalamus promotes sleep by GABAergic inhibition of the arousal system. REM sleep is generated in the pontine tegmentum; lesions in the subcoeruleus region produce REM sleep behavior disorder (RBD) by releasing the normal REM atonia.
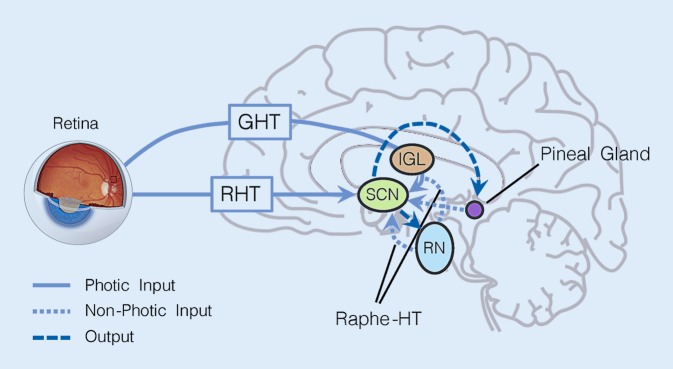
Circadian Rhythm
The suprachiasmatic nucleus (SCN) of the hypothalamus is the master circadian pacemaker, entrained to the light-dark cycle via retinohypothalamic input from melanopsin-containing retinal ganglion cells. The SCN drives melatonin secretion by the pineal gland (rising in the evening, peaking in the middle of the night, suppressed by light) and drives daily variation in core body temperature, cortisol, and sleep propensity. The two-process model of sleep regulation combines Process S (homeostatic sleep drive that builds during wakefulness via adenosine accumulation) and Process C (circadian alerting signal from the SCN). Scribes should understand these terms because clinicians will dictate them, especially for circadian rhythm disorders and shift-work patients.
Upper Airway Anatomy for OSA
The pharynx is a collapsible muscular tube extending from the nasal choanae to the larynx, divided into the nasopharynx, oropharynx (soft palate to hyoid), and hypopharynx. During wakefulness, pharyngeal dilator muscles (genioglossus, tensor palatini) keep the airway patent. During sleep, muscle tone drops and the airway narrows, particularly at the retropalatal and retroglossal levels. In obstructive sleep apnea, anatomic crowding (large tongue, long soft palate, enlarged tonsils, retrognathia, obesity with parapharyngeal fat) combined with loss of muscle tone leads to repetitive collapse.
You will hear clinicians say things like “the patient had REM-predominant OSA with an REM AHI of 42 and an NREM AHI of 8.” Know that this means obstruction is concentrated in REM sleep (where atonia worsens airway collapsibility). Chart both numbers — they drive treatment decisions.
02 Scribe Documentation Framework
Sleep medicine encounters fall into a handful of predictable buckets: new consultations for snoring/witnessed apneas/daytime sleepiness, insomnia evaluations, restless legs, narcolepsy or hypersomnia workups, parasomnia visits, and follow-ups centered on PAP compliance or medication response. Most clinics use a SOAP format, but the history is heavily templated around sleep-specific questions the generic HPI ignores.
Every new sleep consult should document: bedtime and wake time on workdays and free days, sleep latency (minutes to fall asleep), number and duration of awakenings, total sleep time (TST), nap habits, snoring (loudness, bed-partner complaints), witnessed apneas or gasping/choking arousals, nocturia, morning headaches, dry mouth, daytime sleepiness (quantified with the Epworth Sleepiness Scale), unrefreshing sleep, cataplexy-like episodes, hypnagogic/hypnopompic hallucinations, sleep paralysis, leg discomfort at rest, dream enactment, and caffeine/alcohol/nicotine/cannabis use.
Vitals & anthropometrics: BP, HR, height, weight, BMI, neck circumference (> 17 inches in men or > 16 inches in women is a major OSA risk factor). Upper airway exam: Modified Mallampati class, tonsil grade, nasal patency, retrognathia. Prior sleep studies: Date, type (in-lab PSG vs HSAT), AHI, lowest SpO2, time below 90%, sleep efficiency, REM percentage, PLM index. PAP data download: Usage days, average hours per night, residual AHI, leak, pressure settings.
The A&P in sleep medicine typically enumerates each sleep disorder with ICD-10, then attaches a plan: order a PSG vs HSAT, start or titrate PAP, change mask interface, trial a medication (and for controlled substances document the DEA considerations), refer to ENT for DISE or Inspire evaluation, behavioral sleep medicine referral for CBT-I, iron studies for RLS, or repeat study after weight loss/surgery.
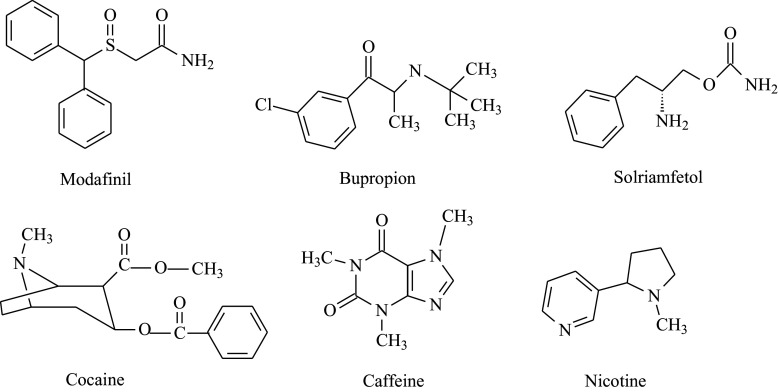
Always chart the Epworth Sleepiness Scale number and each individual item score. Sleep clinicians use the total (0–24) to track treatment response and to justify medications like modafinil. A score > 10 is considered abnormal daytime sleepiness.
03 Obstructive Sleep Apnea (OSA)
Obstructive sleep apnea is repetitive partial or complete collapse of the upper airway during sleep despite ongoing respiratory effort. It is the single most common diagnosis in a sleep clinic, affecting an estimated 10–30% of middle-aged adults, and the majority of your charting days will revolve around it. Untreated OSA is associated with hypertension, stroke, atrial fibrillation, insulin resistance, motor-vehicle accidents, and increased all-cause mortality.
Pathophysiology
Collapse is driven by an imbalance between the collapsing pressure (negative inspiratory suction generated by the diaphragm) and the dilating force (tone of the pharyngeal dilator muscles, especially the genioglossus). During sleep, neural drive to dilator muscles decreases; in REM sleep it collapses almost entirely. Each obstructive event produces hypoxemia, hypercapnia, and an arousal that fragments sleep. Surges of sympathetic activity during events drive blood-pressure spikes and contribute to the cardiovascular consequences of untreated disease.
Clinical Presentation
Classic symptoms include loud habitual snoring, witnessed apneas, gasping/choking arousals, nonrestorative sleep, morning headaches, dry mouth, nocturia, excessive daytime sleepiness, difficulty concentrating, and irritability. Bed-partner history is often more reliable than the patient’s. Women and older adults more often present with insomnia, fatigue, or mood symptoms rather than classic snoring.
Diagnostic Workup
Screen with the STOP-BANG questionnaire (Snoring, Tiredness, Observed apneas, high blood Pressure, BMI > 35, Age > 50, Neck > 40 cm, male Gender — 1 point each). A score of 0–2 is low risk, 3–4 intermediate, and 5–8 high risk. Confirm with either in-lab polysomnography (PSG, gold standard) or a home sleep apnea test (HSAT) in uncomplicated adults with high pre-test probability and no significant cardiopulmonary disease, per the AASM 2017 Clinical Practice Guideline on Diagnostic Testing for Adult OSA. The USPSTF 2022 OSA screening statement found insufficient evidence to screen asymptomatic adults but recommends testing any adult with symptoms or observed apneas.
| Severity | AHI (events/hour) | Clinical context |
|---|---|---|
| Normal | < 5 | No sleep-disordered breathing |
| Mild OSA | 5 – 14.9 | Treat if symptomatic or with comorbidities |
| Moderate OSA | 15 – 29.9 | PAP therapy indicated |
| Severe OSA | ≥ 30 | PAP strongly indicated; expedite treatment |
Respiratory event definitions: Apnea = ≥ 90% airflow drop for ≥ 10 seconds. Hypopnea = ≥ 30% airflow drop for ≥ 10 seconds with either ≥ 3% desaturation or an arousal (AASM recommended rule) or with ≥ 4% desaturation (CMS-required rule). RERA = respiratory effort–related arousal.
Management
First-line treatment for moderate-to-severe OSA is positive airway pressure (PAP) therapy, per the AASM 2019 Clinical Practice Guideline on PAP Therapy. Alternatives and adjuncts include weight loss (bariatric surgery for BMI ≥ 35), oral appliances (mandibular advancement devices) for mild-to-moderate OSA or PAP-intolerant patients, positional therapy for positional OSA, upper airway stimulation (Inspire hypoglossal nerve stimulator), and surgical options (UPPP, maxillomandibular advancement, tonsillectomy in pediatric OSA). Avoid alcohol and sedatives, which worsen airway collapsibility.
When a patient is on CPAP, every follow-up note should contain four numbers from the device download: percent days used, average hours of use on days used, residual AHI, and 95th-percentile leak. Medicare compliance requires ≥ 4 hours on ≥ 70% of nights over a 30-day window in the first 90 days.
04 Central Sleep Apnea (CSA)
Central sleep apnea is a cessation of airflow caused by loss of ventilatory drive — there is no respiratory effort during the event, unlike OSA. CSA is far less common than OSA and usually occurs in specific clinical contexts.
Subtypes
- Idiopathic CSA — no identifiable cause; often presents with insomnia or nocturnal awakenings.
- Cheyne-Stokes breathing — a crescendo-decrescendo pattern of tidal volume with apneic or hypopneic pauses, classically seen in heart failure with reduced ejection fraction and after stroke. The cycle length (30–90 seconds) reflects prolonged circulation time.
- Opioid-induced CSA — chronic opioid use suppresses the respiratory rhythm generator, producing ataxic breathing and central apneas.
- Treatment-emergent CSA (complex sleep apnea) — central apneas that appear after CPAP is initiated for apparent OSA; often resolves with time or requires ASV.
- High-altitude periodic breathing — hypoxia-driven instability above ~2,500 m.
- CSA due to medical conditions — brainstem strokes, Arnold-Chiari malformation, renal failure.
Management
Treat the underlying cause first (optimize heart failure, taper opioids when possible). PAP options include CPAP, BiPAP in spontaneous-timed mode (BiPAP-ST), and adaptive servo-ventilation (ASV). Important safety note: per the SERVE-HF trial, ASV is contraindicated in symptomatic heart failure with EF ≤ 45% because it was associated with increased cardiovascular mortality. Phrenic nerve stimulators (remedé) are an emerging option for moderate-to-severe CSA.
05 Obesity Hypoventilation Syndrome (OHS)
Obesity hypoventilation syndrome (Pickwickian syndrome) is defined by obesity (BMI ≥ 30), awake chronic hypercapnia (PaCO2 > 45 mmHg), and sleep-disordered breathing, with no alternative cause of hypoventilation. Roughly 90% of OHS patients also have OSA. Untreated OHS carries high mortality — higher than OSA alone — from right heart failure, pulmonary hypertension, and respiratory failure.
Workup & Management
Suspect OHS when a patient with severe obesity has a serum bicarbonate ≥ 27 mEq/L (a sensitive, inexpensive screen for chronic CO2 retention). Confirm with an awake ABG. Obtain PSG with CO2 monitoring (end-tidal or transcutaneous). First-line therapy per the ATS 2019 OHS guideline is PAP therapy — CPAP for stable OHS with concomitant severe OSA, otherwise BiPAP (or volume-assured pressure support, AVAPS). Weight loss is the only definitive cure; bariatric surgery is strongly recommended for eligible patients.
06 Complex / Mixed Apneas, UARS & Snoring
Mixed apnea: An event that starts as a central apnea (no effort) and transitions into an obstructive apnea as effort returns against a collapsed airway. Scored as obstructive by AASM convention. Complex sleep apnea = treatment-emergent CSA (see Section 4). Upper airway resistance syndrome (UARS): A phenotype in which patients have repeated arousals from increased respiratory effort without meeting AHI criteria for OSA. Often young, thin women with fatigue, insomnia, and chronic headaches. Diagnosed by a high RDI (RERAs included) with a normal AHI. Primary snoring: Snoring without airflow limitation, desaturation, or arousals — a social problem rather than a medical one, though it may be a precursor to OSA.
07 Insomnia Disorders
Insomnia disorder is defined by dissatisfaction with sleep quantity or quality associated with difficulty initiating sleep, maintaining sleep, or early-morning awakening, occurring despite adequate opportunity for sleep, with daytime impairment. Chronic insomnia requires symptoms ≥ 3 nights per week for ≥ 3 months (ICSD-3-TR). Short-term insomnia lasts < 3 months.
Subtypes & Phenotypes
- Primary (idiopathic) insomnia: No identifiable medical, psychiatric, or substance cause.
- Comorbid insomnia: Co-occurring with depression, anxiety, chronic pain, PTSD, OSA, RLS, menopause, or medication side effects. The most common presentation.
- Psychophysiological insomnia: Conditioned arousal and heightened bedtime anxiety (“I can never sleep in my own bed”).
- Paradoxical insomnia (sleep-state misperception): Patient reports severe insomnia but PSG shows normal sleep duration and architecture.
- Idiopathic insomnia: Lifelong, early-onset insomnia without clear precipitant.
Assessment Tools
Document the Insomnia Severity Index (ISI) (0–28; 0–7 no insomnia, 8–14 subthreshold, 15–21 moderate, 22–28 severe) and/or the Pittsburgh Sleep Quality Index (PSQI) (> 5 suggests poor sleep quality). A sleep diary for 1–2 weeks is the single most valuable behavioral assessment. Actigraphy can supplement the diary.
Management
Per the AASM 2021 Clinical Practice Guideline on Behavioral and Psychological Treatments for Chronic Insomnia, cognitive behavioral therapy for insomnia (CBT-I) is first-line for chronic insomnia disorder and is more durable than medications. CBT-I components include sleep restriction, stimulus control, cognitive restructuring, sleep hygiene education, and relaxation training. Pharmacotherapy (see Section 16) per the AASM 2017 Clinical Practice Guideline for the Pharmacologic Treatment of Chronic Insomnia is second-line and should be used at the lowest effective dose for the shortest duration, preferring DORAs (suvorexant, lemborexant, daridorexant), low-dose doxepin, or ramelteon.

08 Narcolepsy & Central Disorders of Hypersomnolence
Narcolepsy is a chronic neurologic disorder of sleep-wake state instability, classically presenting with excessive daytime sleepiness and intrusion of REM phenomena into wakefulness.
Narcolepsy Type 1 (with cataplexy)
Caused by autoimmune loss of hypothalamic orexin (hypocretin) neurons. The tetrad: (1) excessive daytime sleepiness, (2) cataplexy — sudden bilateral loss of muscle tone triggered by strong emotion (laughter is classic), (3) sleep paralysis, and (4) hypnagogic/hypnopompic hallucinations. Disrupted nocturnal sleep is a fifth feature. Diagnosis requires EDS for ≥ 3 months plus either cataplexy with MSLT findings, or CSF hypocretin-1 ≤ 110 pg/mL. MSLT must show mean sleep latency ≤ 8 minutes and ≥ 2 sleep-onset REM periods (SOREMPs); one SOREMP on the preceding PSG can count toward the total.
Narcolepsy Type 2 (without cataplexy)
Same EDS and MSLT criteria but no cataplexy and normal CSF hypocretin.
Idiopathic Hypersomnia
EDS with long sleep times, severe sleep inertia (“sleep drunkenness”), and unrefreshing naps. MSLT mean sleep latency ≤ 8 minutes with < 2 SOREMPs, or total 24-hour sleep time ≥ 11 hours.
Kleine-Levin Syndrome
Rare, episodic hypersomnia (sleeping 16–20 hours/day) lasting days to weeks with cognitive/behavioral changes (hyperphagia, hypersexuality, derealization), typically in adolescent males. Between episodes, patients are normal.
Management
Per the AASM 2021 Clinical Practice Guideline on Treatment of Central Disorders of Hypersomnolence, wake-promoting agents (modafinil, armodafinil, solriamfetol, pitolisant) are used for EDS, with sodium oxybate (Xyrem) or low-sodium oxybate (Xywav) reserved for patients with significant cataplexy or when stimulants are inadequate. Venlafaxine or fluoxetine are used off-label for isolated cataplexy. Scheduled naps, good sleep hygiene, and driving safety counseling are essential.
09 Restless Legs Syndrome & PLMD
Restless legs syndrome (RLS / Willis-Ekbom disease) is a sensorimotor disorder defined by the IRLSSG criteria: (1) an urge to move the legs, usually accompanied by uncomfortable sensations; (2) worse at rest; (3) relieved by movement; (4) worse in the evening or night; and (5) not solely explained by another condition (leg cramps, positional discomfort, arthralgia). Prevalence is 5–10% of adults.
Workup
Always order ferritin, iron, TIBC, transferrin saturation. Iron deficiency is the most common reversible cause of RLS. Target ferritin > 75–100 ng/mL and transferrin saturation > 20% in RLS patients. The International RLS Study Group severity scale (IRLS) is a 10-item patient-reported instrument: 0 none, 1–10 mild, 11–20 moderate, 21–30 severe, 31–40 very severe.
Periodic Limb Movement Disorder (PLMD)
Stereotyped, repetitive flexion movements of the legs during sleep (PLMS = periodic limb movements of sleep; PLMD = PLMS causing sleep disturbance or daytime impairment). PLMS are scored on PSG: > 15/hour in adults is abnormal. PLMS frequently coexists with RLS but is a distinct disorder.
Management
Per the AASM 2025 Clinical Practice Guideline on Treatment of RLS and PLMD and the updated IRLSSG/AASM framework, first-line therapy has shifted from dopamine agonists to alpha-2-delta ligands (gabapentin enacarbil, pregabalin, gabapentin) because of the high long-term risk of augmentation with dopamine agonists. Dopamine agonists (pramipexole, ropinirole, rotigotine patch) remain second-line. IV iron (ferric carboxymaltose) is recommended for symptomatic patients with ferritin ≤ 75 ng/mL. Opioids (low-dose oxycodone, methadone) are reserved for severe refractory cases. Avoid: serotonergic antidepressants, antihistamines, dopamine antagonists, and alcohol, all of which worsen RLS.
10 REM Sleep Behavior Disorder & NREM Parasomnias
REM Sleep Behavior Disorder (RBD)
RBD is loss of the normal REM atonia, allowing patients to physically enact their dreams — punching, kicking, jumping out of bed, and injuring themselves or bed partners. Dreams are typically action-packed and confrontational. RBD is strongly associated with alpha-synucleinopathies: > 80% of idiopathic RBD patients will eventually develop Parkinson disease, dementia with Lewy bodies, or multiple system atrophy, often years to decades later. Diagnosis requires PSG showing REM sleep without atonia (RSWA) plus clinical history of dream enactment.
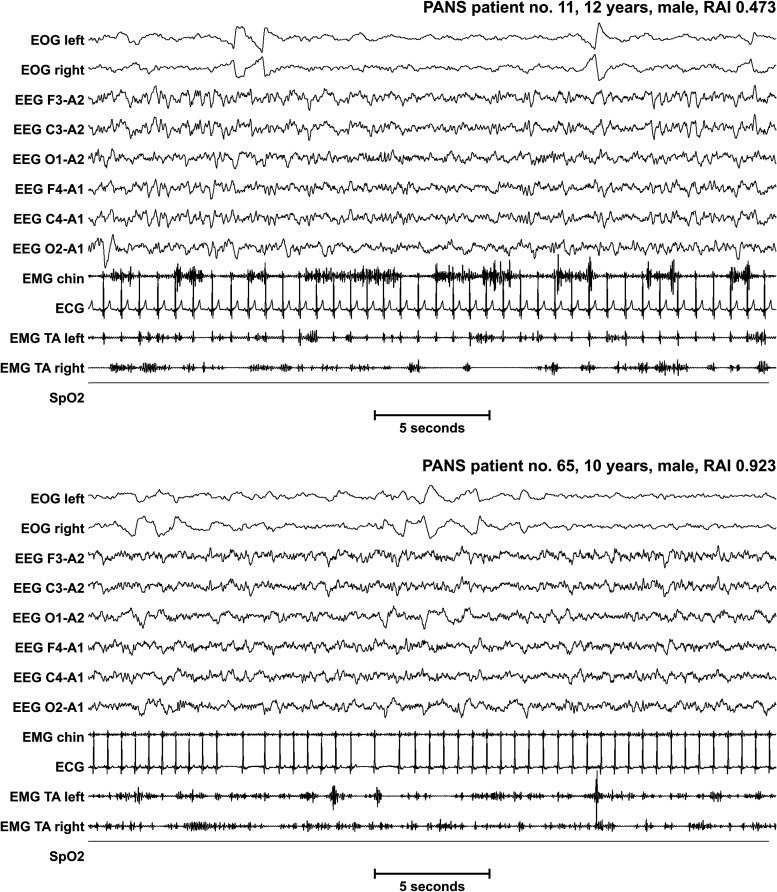
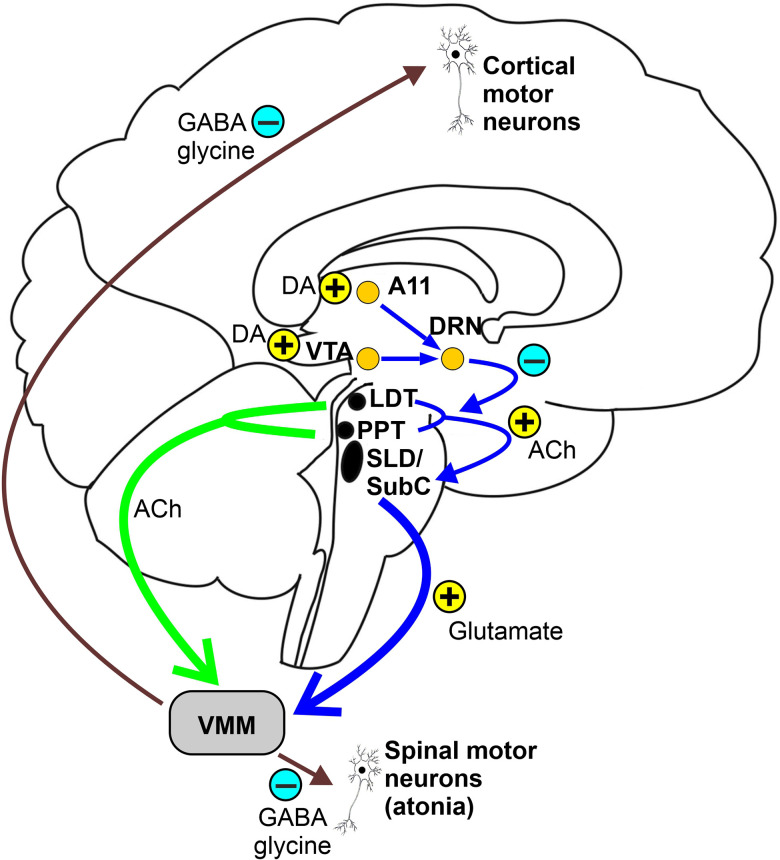
Management per the AASM Best Practice Guide on Treatment of RBD: bedroom safety (pad floor, remove sharp objects, sleep alone or with barrier), melatonin 3–12 mg at bedtime (first-line, favorable side-effect profile), or clonazepam 0.25–2 mg (caution in OSA, falls, dementia). Counsel patients on the neurodegenerative risk and schedule neurologic follow-up.
NREM Parasomnias (Disorders of Arousal)
NREM parasomnias arise from incomplete arousal from slow-wave sleep, typically in the first third of the night. Patients are not truly awake, do not remember the events, and may be difficult to redirect. Common in children; triggers include sleep deprivation, fever, stress, OSA (which fragments sleep and triggers arousals), and certain medications (zolpidem).
- Confusional arousals: Disoriented, sometimes agitated behavior in bed without ambulation.
- Sleepwalking (somnambulism): Walking and complex behaviors with open eyes and glassy stare. Can include leaving the house, driving, cooking.
- Sleep terrors (pavor nocturnus): Sudden arousal with screaming, autonomic surge (tachycardia, diaphoresis, mydriasis), and inconsolability. Distinct from nightmares, which occur in REM.
- Sleep-related eating disorder (SRED): Recurrent episodes of involuntary eating during a partial arousal, often of bizarre food combinations.
- Sexsomnia: Sexual behaviors during sleep without conscious awareness.
Evaluate for and treat any underlying OSA, ensure adequate sleep, remove triggering medications, create a safe environment. Pharmacotherapy (clonazepam) is reserved for injurious or recurrent cases.
11 Other Parasomnias & Sleep-Related Movement Disorders
- Nightmare disorder: Recurrent, well-remembered disturbing dreams in REM sleep; treat with image rehearsal therapy (IRT) and prazosin, especially for PTSD-associated nightmares.
- Sleep-related hallucinations: Hypnagogic (falling asleep) or hypnopompic (waking); vivid visual or auditory; can be normal or part of narcolepsy.
- Sleep paralysis: Transient inability to move on sleep-wake transition; isolated episodes are common in normal adults, recurrent paralysis suggests narcolepsy.
- Exploding head syndrome: Perception of a loud bang or flash of light at sleep onset; benign, reassure.
- Sleep-related bruxism: Tooth grinding during sleep; chart dental wear, jaw pain, bed-partner complaints. Occlusal splint is first-line.
- Sleep-related leg cramps: Sudden painful calf/foot cramps during sleep; stretching, hydration; quinine avoided due to adverse effects.
- Catathrenia (sleep-related groaning): Prolonged expiratory groaning during REM; benign.
- Sleep fragmentation / sleep deprivation: Not a formal ICSD diagnosis per se but a common documentation topic; chart TST, WASO (wake after sleep onset), and sleep efficiency.
- Fatal familial insomnia (FFI): Rare autosomal-dominant prion disease (PRNP mutation); progressive insomnia, dysautonomia, and death within ~18 months. Extraordinarily rare but should be in the scribe’s vocabulary.
12 Circadian Rhythm Sleep-Wake Disorders
Circadian rhythm disorders are misalignments between the endogenous circadian clock and the desired or required sleep-wake schedule. Total sleep quality is often normal when the patient sleeps on their own schedule; problems arise from external demands. Management is based on the AASM 2015 Clinical Practice Guideline on the Treatment of Intrinsic Circadian Rhythm Sleep-Wake Disorders.
| Disorder | Pattern | Treatment |
|---|---|---|
| Delayed sleep-wake phase (DSWPD) | “Night owls” — cannot fall asleep until 2–6 AM; teens and young adults | Morning bright light + evening low-dose melatonin (0.3–0.5 mg); chronotherapy |
| Advanced sleep-wake phase (ASWPD) | “Early birds” — asleep by 6–8 PM, wake at 2–4 AM; older adults | Evening bright light therapy |
| Irregular sleep-wake rhythm | No defined sleep period; 3+ bouts in 24h; dementia, brain injury | Structured activity schedule, daytime light, evening melatonin |
| Non-24-hour sleep-wake (Non-24) | Free-running rhythm; common in blind individuals without light perception | Tasimelteon; melatonin in sighted patients |
| Shift work disorder | Insomnia and sleepiness from atypical work hours | Strategic napping, caffeine, modafinil/armodafinil, light exposure timing |
| Jet lag disorder | Transient misalignment after rapid timezone change | Timed light exposure, melatonin, short-acting hypnotics |
13 Sleep Studies — PSG, HSAT, MSLT, MWT
Sleep studies are the procedural heart of sleep medicine. A scribe must know what each study measures, when it is ordered, and what the report says.
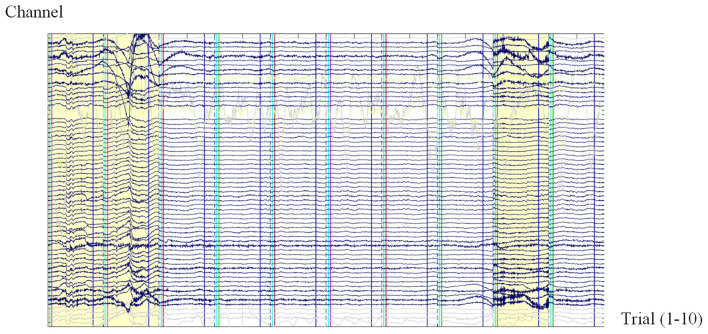
The gold-standard attended overnight study. Records EEG (frontal, central, occipital), EOG (eye movements), chin EMG (tone), ECG, nasal pressure and oronasal thermistor (airflow), thoracic and abdominal respiratory effort belts (RIP), SpO2, leg EMG (tibialis anterior, for PLMS), body position, and snore sensor. Indications: suspected OSA with comorbidities (CHF, COPD, stroke, neuromuscular disease), parasomnia evaluation, titration studies, suspected PLMD, and narcolepsy workups (preceding MSLT).
Unattended, limited-channel study for diagnosis of OSA only. Type III devices record airflow (cannula), respiratory effort, SpO2, and sometimes HR and position. HSAT reports the respiratory event index (REI) rather than AHI because denominator is recording time, not sleep time — typically underestimates true AHI. Indicated for uncomplicated adults with high pre-test probability of moderate-to-severe OSA and no significant cardiopulmonary comorbidity, per AASM 2017 diagnostic testing guidelines.
Objective measure of daytime sleep tendency, performed the day after an in-lab PSG. Five 20-minute nap opportunities at 2-hour intervals. Measures mean sleep latency and counts sleep-onset REM periods (SOREMPs). Narcolepsy criteria: mean sleep latency ≤ 8 minutes and ≥ 2 SOREMPs (a REM period within 15 min of the preceding PSG can count toward the 2). Patients must be off REM-suppressing medications (SSRIs, SNRIs, TCAs) for at least 2 weeks and have adequate sleep the preceding 2 weeks (documented by sleep diary or actigraphy).
Measures a patient’s ability to stay awake in a soporific environment. Four 40-minute trials, seated in a dimly lit room. Used to assess treatment response in narcolepsy/hypersomnia and for occupational fitness-for-duty (pilots, commercial drivers).
Other Diagnostic Tools
Actigraphy: Wrist-worn accelerometer that estimates sleep-wake patterns over 1–2 weeks. Used for insomnia, circadian rhythm disorders, and prior to MSLT. Sleep diary: Patient-completed log of bedtime, wake time, naps, caffeine, and alcohol. Drug-induced sleep endoscopy (DISE): ENT performs flexible endoscopy during propofol-induced sleep to identify sites of airway collapse before surgical or Inspire planning.
The PSG report has standard elements you will chart: TST, sleep efficiency (TST/TIB), sleep latency, REM latency, stage percentages, AHI (total and supine/REM), lowest SpO2, time with SpO2 < 90%, PLM index, arousal index, heart rhythm notes. Pull each one verbatim into the note; don’t paraphrase.
14 PAP Therapy — CPAP, BiPAP, ASV, AVAPS
Positive airway pressure pneumatically splints the upper airway open. The device, mode, pressures, interface, and humidification are all scribe-charted fields.
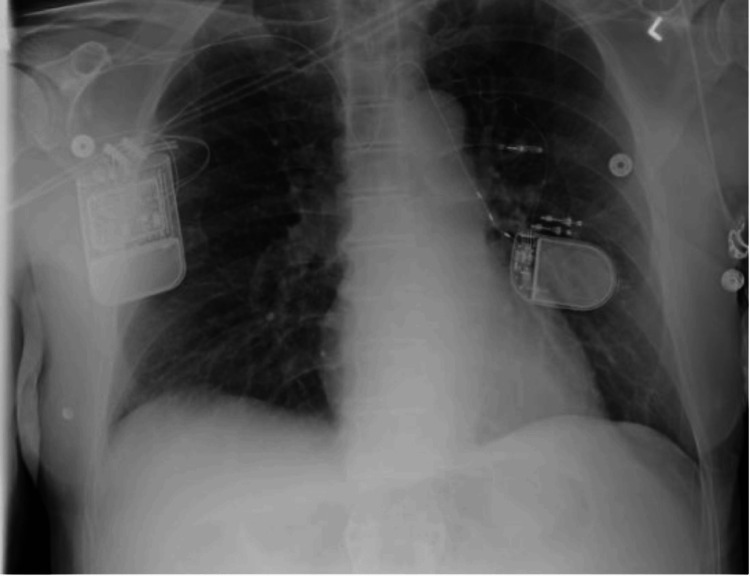
| Mode | What It Delivers | Typical Indication |
|---|---|---|
| CPAP | Single fixed pressure throughout inspiration and expiration | OSA without significant comorbidity |
| APAP (AutoPAP) | Auto-adjusting pressure within a set range | OSA, especially when pressure needs vary (REM, supine) |
| BiPAP-S | Separate IPAP and EPAP, spontaneously triggered | CPAP intolerance, high pressure needs, OHS |
| BiPAP-ST | BiPAP with backup rate | Central apneas, hypoventilation |
| ASV | Auto-adjusting pressure support targeting ventilation | Treatment-emergent CSA, idiopathic CSA; contraindicated in symptomatic HFrEF EF ≤ 45% |
| AVAPS / iVAPS | Volume-assured pressure support | OHS, neuromuscular disease, restrictive chest wall |
Interfaces & Accessories
Mask choices include nasal pillows (smallest, least claustrophobic), nasal mask, full face (oronasal) mask (mouth breathers, higher pressures), hybrid masks, and total face masks. Heated humidifiers, heated tubing, and chinstraps are commonly added. Ramp feature starts at a low pressure and increases to prescribed setting over ~20 minutes to ease falling asleep.
Titration & Follow-Up
In-lab titration gradually increases pressure to eliminate obstructive events, snoring, and flow limitation across body positions and sleep stages. Split-night studies perform diagnosis in the first half and titration in the second when severe OSA is confirmed early. Follow-up visits chart usage data, residual AHI, leak, symptom response (ESS), and troubleshoot problems (aerophagia, mask leak, skin breakdown, claustrophobia, nasal congestion, dry mouth).
15 Surgical & Device Procedures for OSA
| Procedure | Description | Indication |
|---|---|---|
| Drug-induced sleep endoscopy (DISE) | Flexible endoscopy during propofol sedation to map airway collapse (VOTE: velum, oropharynx, tongue base, epiglottis) | Planning Inspire or surgical candidacy |
| Upper airway stimulation (Inspire) | Implanted hypoglossal nerve stimulator; senses respiration and stimulates tongue protrusion during inspiration | Moderate-severe OSA, BMI < 32–35, CPAP failure/intolerance, complete concentric collapse excluded on DISE |
| Uvulopalatopharyngoplasty (UPPP) | Resection of uvula, soft palate, and tonsils to enlarge retropalatal airway | Selected patients with retropalatal obstruction |
| Expansion sphincter pharyngoplasty | Modified palatopharyngeal surgery preserving more tissue | Alternative to UPPP |
| Tonsillectomy & adenoidectomy | First-line surgical treatment for pediatric OSA | Pediatric OSA with adenotonsillar hypertrophy |
| Maxillomandibular advancement (MMA) | Le Fort I + bilateral sagittal split osteotomy, advancing facial skeleton 10–12 mm | Severe OSA refractory to PAP; most effective surgical option |
| Septoplasty / turbinate reduction | Improves nasal airflow | Adjunct to improve CPAP tolerance, not a standalone OSA cure |
| Tracheostomy | Bypasses the upper airway entirely | Salvage for severe OSA refractory to everything else |
| Bariatric surgery | Sleeve gastrectomy, RYGB | OSA with BMI ≥ 35; definitive therapy when weight is the driver |
| Oral appliance (MAD) | Mandibular advancement device fit by sleep dentist; advances mandible 5–10 mm | Mild-moderate OSA, CPAP-intolerant, primary snoring |
Light therapy boxes (10,000 lux, 30 minutes in the morning) are prescribed for DSWPD and seasonal affective disorder. Melatonin protocols: low-dose (0.3–0.5 mg) 5–7 hours before desired sleep onset as a chronobiotic for DSWPD; 3–5 mg at bedtime as a soporific in insomnia or RBD.
16 Medications You Must Know
| Drug Class | Generic (Brand) | Mechanism / Notes |
|---|---|---|
| Z-drugs | Zolpidem (Ambien), zaleplon (Sonata), eszopiclone (Lunesta) | Selective GABA-A (BZ1) agonists. Sleep-driving, complex sleep behaviors (FDA boxed warning). Shorter acting for sleep onset, eszopiclone for maintenance. |
| Benzodiazepines | Temazepam (Restoril), triazolam (Halcion), estazolam, flurazepam | Non-selective GABA-A. Tolerance, dependence, falls in elderly. Avoid in OSA. |
| DORAs (orexin antagonists) | Suvorexant (Belsomra), lemborexant (Dayvigo), daridorexant (Quviviq) | Block orexin signaling; low abuse potential. AASM-preferred for chronic insomnia. |
| Melatonin receptor agonist | Ramelteon (Rozerem) | MT1/MT2 agonist; no controlled-substance status; for sleep-onset insomnia. |
| Sedating antidepressants | Doxepin 3–6 mg (Silenor), trazodone, mirtazapine | Low-dose doxepin FDA-approved for sleep maintenance. Trazodone widely used off-label despite limited evidence. |
| Antihistamines | Diphenhydramine, doxylamine | Over-the-counter; avoid chronic use especially in elderly (anticholinergic burden). |
| Drug | Use | Notes |
|---|---|---|
| Modafinil (Provigil) | Narcolepsy, shift work disorder, residual EDS on CPAP | Dopamine reuptake modulator; reduces OCP efficacy; Schedule IV |
| Armodafinil (Nuvigil) | Same as modafinil | R-enantiomer; longer half-life |
| Solriamfetol (Sunosi) | EDS in narcolepsy or OSA | DA/NE reuptake inhibitor; Schedule IV |
| Pitolisant (Wakix) | Narcolepsy, EDS and cataplexy | H3 inverse agonist; non-scheduled |
| Sodium oxybate (Xyrem) | Narcolepsy (EDS + cataplexy) | GHB salt; two nightly doses; Schedule III; REMS program |
| Low-sodium oxybate (Xywav) | Narcolepsy, idiopathic hypersomnia | Lower sodium burden; same REMS |
| Methylphenidate, amphetamines | EDS when above fail | Schedule II; cardiovascular monitoring |
| Venlafaxine, fluoxetine, clomipramine | Cataplexy (off-label) | REM-suppressing effect |
| Drug | Class | Notes |
|---|---|---|
| Gabapentin enacarbil (Horizant) | Alpha-2-delta ligand | FDA-approved for moderate-severe RLS; first-line |
| Pregabalin (Lyrica), gabapentin | Alpha-2-delta ligand | First-line; less augmentation than DAs |
| Pramipexole (Mirapex) | Dopamine agonist | Effective but high long-term augmentation rate |
| Ropinirole (Requip) | Dopamine agonist | Same concerns |
| Rotigotine (Neupro) | Dopamine agonist transdermal patch | More stable 24h level, lower augmentation |
| Carbidopa/levodopa | Dopamine precursor | Limited use — highest augmentation rate |
| Oxycodone, methadone | Opioid | Severe refractory RLS only |
| Ferric carboxymaltose (Injectafer) | IV iron | Ferritin ≤ 75 ng/mL |
Melatonin: OTC; chronobiotic dosing 0.3–0.5 mg for circadian phase shifting; soporific dosing 1–5 mg. Ramelteon: MT agonist for DSWPD. Tasimelteon (Hetlioz): FDA-approved for non-24-hour sleep-wake disorder. Prazosin: Alpha-1 antagonist for PTSD-associated nightmares. Clonazepam: Benzodiazepine used in RBD and injurious NREM parasomnias. Melatonin 3–12 mg: First-line for RBD.
17 Classification Systems & Severity Scales
Eight situations, each scored 0 (would never doze) to 3 (high chance of dozing). Total 0–24.
| Total | Interpretation |
|---|---|
| 0–5 | Lower normal daytime sleepiness |
| 6–10 | Higher normal daytime sleepiness |
| 11–12 | Mild excessive daytime sleepiness |
| 13–15 | Moderate EDS |
| 16–24 | Severe EDS |
Snoring, Tiredness, Observed apneas, high blood Pressure, BMI > 35, Age > 50, Neck > 40 cm, male Gender (1 point each).
| Score | OSA Risk |
|---|---|
| 0–2 | Low |
| 3–4 | Intermediate |
| 5–8 | High (especially for moderate-severe OSA) |
7-item patient-reported questionnaire, total 0–28.
| Score | Interpretation |
|---|---|
| 0–7 | No clinically significant insomnia |
| 8–14 | Subthreshold insomnia |
| 15–21 | Moderate insomnia |
| 22–28 | Severe insomnia |
19 self-rated items across 7 component scores (sleep quality, latency, duration, efficiency, disturbances, medication use, daytime dysfunction). Global score > 5 suggests poor sleep quality.
| Total | Severity |
|---|---|
| 0 | None |
| 1–10 | Mild |
| 11–20 | Moderate |
| 21–30 | Severe |
| 31–40 | Very severe |
| Class | What You See (mouth open, tongue out) |
|---|---|
| I | Soft palate, uvula, tonsillar pillars all visible |
| II | Soft palate and uvula visible |
| III | Only soft palate and base of uvula visible |
| IV | Only hard palate visible |
Higher classes correlate with higher OSA risk and more difficult airway.
| Grade | Description |
|---|---|
| 0 | Tonsils within pillars (prior tonsillectomy) |
| 1+ | < 25% of oropharyngeal width |
| 2+ | 25–50% |
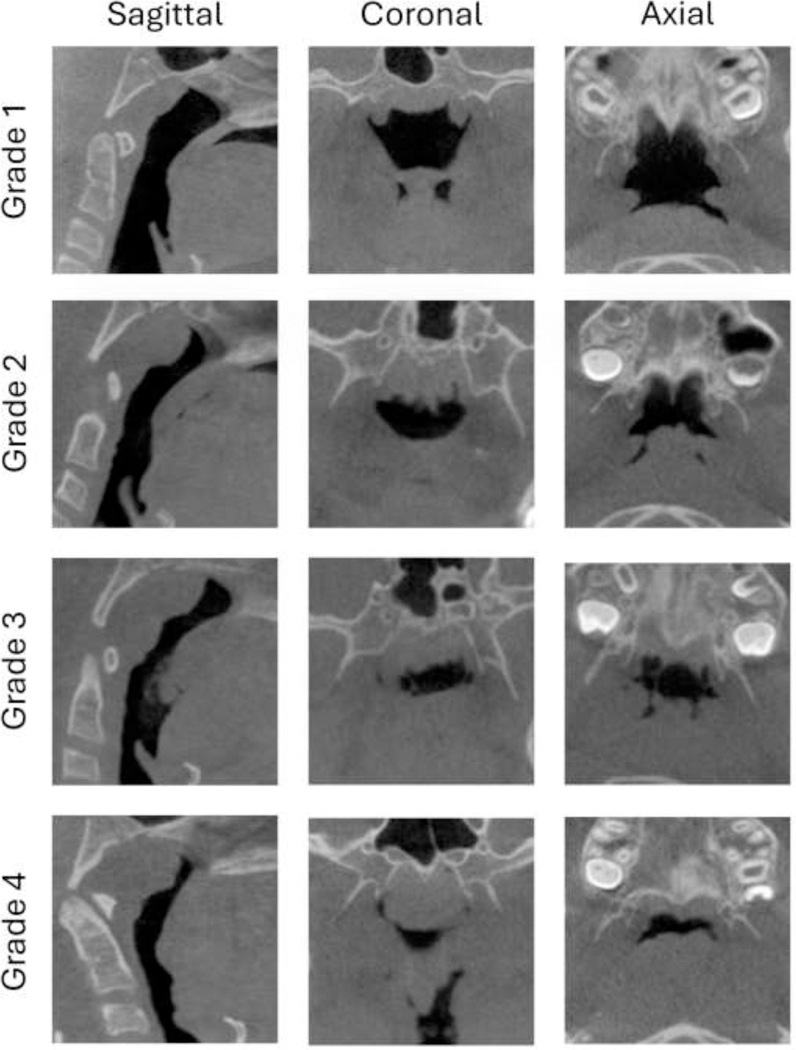
| 3+ | 50–75% |
| 4+ | > 75% (“kissing tonsils”) |
Mean sleep latency ≤ 8 minutes across 5 naps, plus ≥ 2 sleep-onset REM periods (SOREMPs). A SOREMP on the preceding overnight PSG can substitute for one of the MSLT SOREMPs.
18 Physical Exam — The Sleep Exam
The sleep exam is focused and repetitive — you will document essentially the same elements every visit. Capture them efficiently.
Vitals: BP ___/___, HR ___, SpO2 ___%, BMI ___, Neck circumference ___ in
General: Alert, NAD, [body habitus]
HEENT: Nasal passages ___ (patent/congested/deviated septum)
Oropharynx: Mallampati class ___, tonsils ___, elongated uvula (y/n)
Dentition: intact / worn / bruxism facets
Neck: supple, no thyromegaly, no JVD
Mandible: normal / retrognathia / micrognathia
Lungs: CTA bilaterally
CV: RRR, no murmurs, no peripheral edema
Neuro: CN II–XII intact, no focal deficits, normal gait
Skin: no dependent rubor / no trophic changes
Key maneuvers: neck circumference (tape at cricothyroid membrane), Mallampati (patient sits, opens mouth wide, sticks tongue out without phonation), tonsil grading, observation for retrognathia in profile, and nasal exam for deviated septum or turbinate hypertrophy. In narcolepsy workups, look for evidence of cataplexy on emotional prompting only if the clinician elects to; document objectively.
19 Risk Factors & Comorbidities
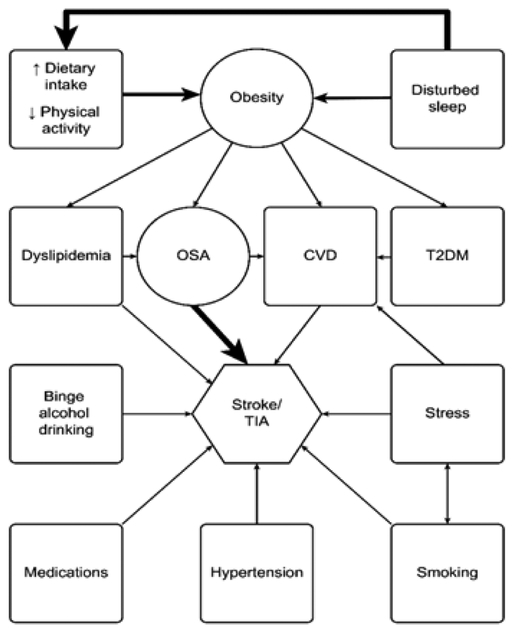
Obesity: BMI and neck circumference. Central adiposity narrows the upper airway and reduces FRC.
Hypertension: Resistant HTN is a major OSA association. Document current regimen and home BP readings.
Diabetes / metabolic syndrome: A1C; OSA worsens insulin resistance, and treatment improves glycemic control modestly.
Atrial fibrillation: Treating OSA improves rhythm control and reduces recurrence after ablation.
Heart failure: Both OSA and CSA common; drives PAP mode selection (beware ASV in HFrEF).
Stroke / TIA: OSA is independently associated with recurrent stroke; screen every stroke patient.
COPD / asthma: “Overlap syndrome” = OSA + COPD, high morbidity; chart nocturnal SpO2.
Psychiatric comorbidity: Depression, anxiety, PTSD, bipolar — enormously important for insomnia and nightmare disorder.
Medications: Opioids (CSA), benzodiazepines (OSA worsening), sedating antihistamines, beta-blockers (melatonin suppression), SSRIs/SNRIs (RBD unmasking, REM suppression confounds MSLT).
Substance use: Alcohol (worsens OSA), cannabis, nicotine, caffeine (quantify intake and timing).
Occupational / driving risk: Commercial drivers, pilots, heavy-machinery operators. Document any drowsy-driving episodes or motor-vehicle accidents.
20 Abbreviations Master List
Diagnoses & Phenomena
Studies & Parameters
PAP & Therapeutics
21 Sample HPI Templates
These templates show the rhythm and content of a good sleep-medicine HPI. Pattern-match off them during your first week.
“Mr. [Name] is a 54-year-old male with hypertension, hyperlipidemia, and BMI 34 referred by his PCP for evaluation of loud habitual snoring and witnessed apneas. His wife reports nightly snoring audible through closed doors and several witnessed pauses per night terminated by gasping. He describes unrefreshing sleep, waking with dry mouth and morning headaches 3–4 mornings per week, and nocturia 2–3 times nightly. Bedtime 10:30 PM, sleep latency ~15 minutes, wake 6:00 AM with an alarm. Denies insomnia, cataplexy, sleep paralysis, hypnagogic hallucinations, or dream enactment. ESS today 14/24. STOP-BANG 6/8 (all items positive except age and male—wait, both positive; neck 17.5 in). Neck circumference 17.5 in, Mallampati class III, tonsils 2+. Plan: in-lab PSG given BMI and HTN.”
“Ms. [Name] is a 42-year-old female with generalized anxiety disorder and perimenopausal symptoms presenting with 2 years of sleep-maintenance insomnia. She falls asleep within 15 minutes of bedtime (10:30 PM) but wakes 3–4 times nightly, often lying awake for 45–90 minutes each time, with anxious rumination. Final awakening 5:00 AM, unable to return to sleep. Estimated TST 4.5–5.5 hours. No snoring, witnessed apneas, leg discomfort, or dream enactment. Uses melatonin 5 mg and diphenhydramine intermittently without benefit; tried zolpidem 5 mg in the past with hangover effect. Caffeine 2 cups coffee before noon. Alcohol 1–2 glasses wine in evening. ISI 19/28, consistent with moderate insomnia. PHQ-9 8, GAD-7 12. Plan: referral to behavioral sleep medicine for CBT-I; discussed DORA (lemborexant) as pharmacologic adjunct if CBT-I insufficient; counseled on alcohol’s fragmenting effect.”
“Mrs. [Name] is a 58-year-old female with iron-deficiency anemia and chronic kidney disease stage 3 presenting with 3 years of progressive uncomfortable leg sensations occurring in the evening when sitting or lying down. She describes a ‘crawling, creeping’ sensation deep in the calves, relieved by walking or stretching, and worst between 9 PM and 1 AM. Symptoms delay sleep onset by 45–90 minutes nightly. Bed partner notes occasional kicking movements during sleep. IRLS severity 26/40 (severe). Ferritin 32 ng/mL, TSAT 14%. Currently on no RLS-specific therapy; takes citalopram 20 mg for depression. Denies cataplexy, RBD features, or EDS. Plan: trial gabapentin enacarbil 600 mg at 5 PM; IV ferric carboxymaltose infusion given ferritin < 75; discussed that SSRIs can worsen RLS but will defer change pending psychiatry input.”
“Mr. [Name] is a 23-year-old male college student with 3 years of progressive daytime sleepiness, now severe enough to cause sleep attacks during lectures and driving. He sleeps 8–9 hours nightly with normal sleep latency but wakes unrefreshed. Describes 2–3 episodes per month of bilateral knee buckling and head-drop triggered by laughter, lasting seconds, consciousness preserved — concerning for cataplexy. Endorses hypnagogic hallucinations (hearing his name called as he falls asleep) and sleep paralysis 1–2 times per week. No snoring or witnessed apneas. ESS 20/24. Not on SSRIs or REM-suppressing medications. Plan: in-lab PSG followed by MSLT; counseled to avoid driving until evaluation complete; two-week sleep diary prior to MSLT; actigraphy ordered.”
“Mr. [Name] is a 68-year-old male with hyperlipidemia presenting with 18 months of dream enactment behavior reported by his wife. Episodes occur in the second half of the night; she describes him punching, kicking, and shouting, with one episode 2 months ago in which he struck her in the face and another where he fell out of bed and sustained a scalp laceration. He recalls vivid, confrontational dreams matching the enacted behavior. No snoring or apneas. Not on SSRI/SNRI. Subtle hyposmia on review of systems; spouse has noted very mild slowing of movement over the past year. Plan: in-lab video PSG to confirm REM sleep without atonia; bedroom safety counseling (padded floor, remove nightstand, consider sleeping alone or with barrier); melatonin 6 mg QHS; neurology referral for evaluation of possible prodromal alpha-synucleinopathy.”
“Ms. [Name] is a 61-year-old female with severe OSA (AHI 48 on 2024 PSG) returning for 3-month CPAP follow-up. Prescribed APAP 8–15 cmH2O with full-face mask and heated humidifier. Device download: usage 26/30 days, average 6 hours 42 minutes on days used, residual AHI 2.1, 95th-percentile pressure 11.4, 95th-percentile leak 18 L/min. She reports markedly improved daytime alertness (ESS down from 16 to 7), resolution of morning headaches, and her spouse confirms cessation of witnessed apneas. Complains of mild nasal congestion and occasional aerophagia. Plan: continue APAP unchanged; add saline nasal spray and raise head of bed for aerophagia; meets Medicare adherence criteria; renew DME order for annual supplies.”
The best sleep scribes live inside a handful of numbers: AHI, lowest SpO2, time below 90%, ESS, ISI, PAP usage hours, residual AHI, and leak. When a clinician says “she is adherent but has breakthrough events,” you already know to pull the device download and chart residual AHI and leak. When they say “this looks like REM-predominant OSA,” you chart both REM and NREM AHI. When they say “the MSLT was positive,” you chart mean sleep latency and the number of SOREMPs.
Understand the difference between an HSAT REI and an in-lab PSG AHI, between CPAP and ASV, between cataplexy and a drop attack. Know the RLS criteria by memory. Know that dopamine agonists cause augmentation. Know that ASV is contraindicated in HFrEF. Know that an ISI of 22 is severe and that CBT-I is first-line. Know that RBD predicts Parkinson disease.
Welcome to sleep medicine. The patients are grateful, the data are objective, and the difference CPAP makes in someone’s life is among the most satisfying things you will witness in outpatient medicine.
22 References & Sources
Clinical Practice Guidelines
American Academy of Sleep Medicine Clinical Practice Guidelines (index).
AAFP. Diagnosis and Management of Obstructive Sleep Apnea in Adults. Am Fam Physician. 2016.
Landmark Clinical Trials & Key Studies
Diagram & Figure Sources
Figure 1: Sleep Hypnogram. Wikimedia Commons. Public domain.
Figure 2: The Brain. OpenStax College via Wikimedia Commons. CC BY 3.0.
Figure 3: Upper airway obstruction in sleep apnea. Wikimedia Commons, Habib M’henni. Public domain.
Figure 4: Obstructive sleep apnea. Wikimedia Commons. Public domain.
Figure 5: Polysomnography REM stage. Wikimedia Commons. Public domain.
Figure 6: CPAP device. Wikimedia Commons. Public domain.