Surgical Oncology
Every cancer, operation, staging system, medication, abbreviation, and documentation framework you need to survive day one in surgical oncology clinic and the OR.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 Surgical Oncology — Principles & Anatomy Essentials
Surgical oncology is the surgical subspecialty dedicated to the diagnosis, staging, and operative management of solid tumors. Unlike general surgery, the surgical oncologist works within a multidisciplinary team — medical oncology, radiation oncology, pathology, radiology, genetics, and reconstructive surgery — because curing a cancer rarely depends on the operation alone. A surgical oncology clinic may see breast cancer in the morning, pancreatic cancer at midday, and a retroperitoneal sarcoma in the afternoon. Scribes must master documentation across many organ systems simultaneously.
The core operative principles are the same regardless of organ: achieve a negative margin (R0) resection, perform an adequate regional lymphadenectomy for staging, minimize tumor spillage, preserve function wherever possible, and provide accurate pathologic staging to guide adjuvant therapy. R0 means no microscopic tumor at the margin; R1 means microscopic residual; R2 means gross residual disease.
Anatomy by Organ System — Quick Orientation
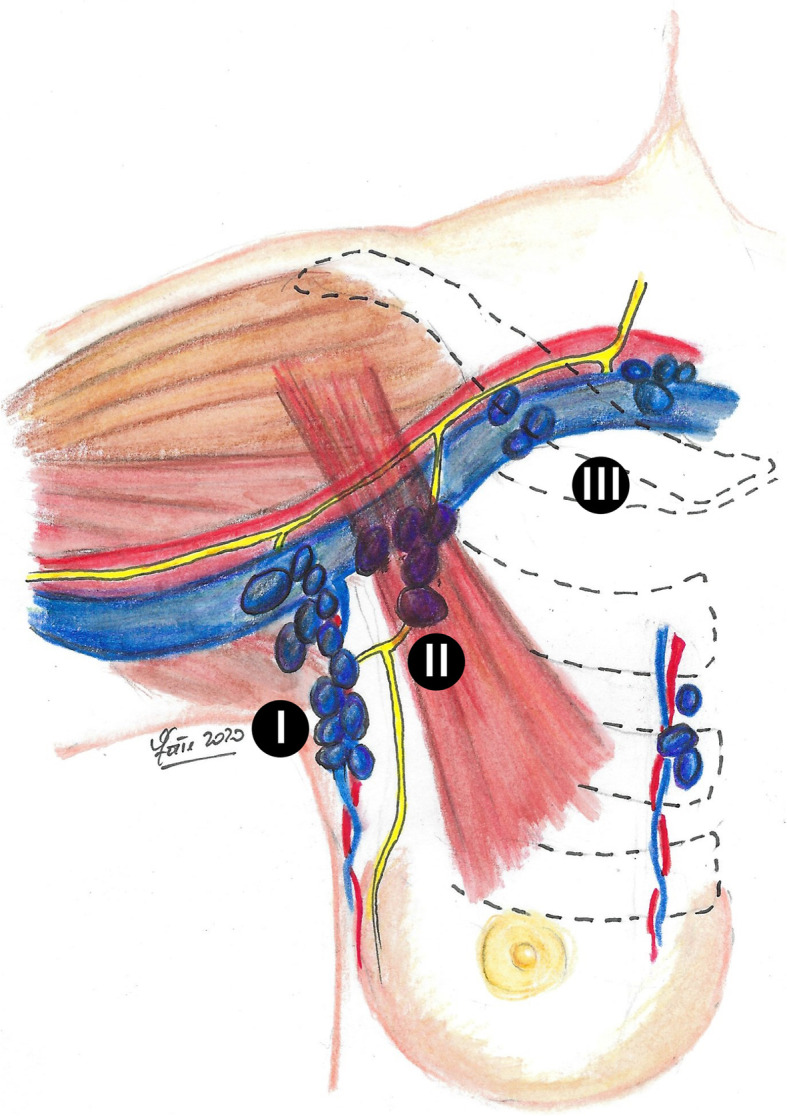
The breast sits on the pectoralis major, drains to the axillary lymph nodes (levels I, II, III relative to pectoralis minor) and internal mammary chain. The stomach has perigastric nodal stations (1–6) and extraperigastric stations (7–16) that define D1, D2, and D3 lymphadenectomy. The pancreas lies retroperitoneally: head and uncinate wrap around the SMA/SMV (the "pancreatic head" operation is the Whipple); body and tail extend toward the splenic hilum. The liver is divided into 8 Couinaud segments based on portal and hepatic venous drainage; a right hepatectomy removes segments V–VIII, left hepatectomy II–IV. The retroperitoneum contains the kidneys, adrenals, aorta, IVC, ureters, and loose areolar tissue where sarcomas arise silently until they reach enormous size.
When the attending says "R0," "sentinel node negative," or "clear margins > 2 mm," they are describing adequacy of resection — chart these phrases verbatim. These are the words that drive whether adjuvant therapy is indicated.
02 Multidisciplinary Cancer Care & Tumor Board
Nearly every cancer patient in a modern surgical oncology practice is reviewed at a multidisciplinary tumor board before, during, or after treatment. This is where the surgeon, medical oncologist, radiation oncologist, radiologist, pathologist, genetic counselor, and sometimes psycho-oncology, palliative care, and social work meet to agree on a treatment plan.
Screening → Diagnosis → Staging → Neoadjuvant therapy → Surgery → Pathologic staging → Adjuvant therapy → Surveillance → Survivorship (or recurrence / palliation). Every surgical oncology note must localize the patient on this continuum — is this a new diagnosis, a preoperative visit, a post-op visit, a surveillance visit, or a recurrence evaluation?
NCCN produces the most widely cited clinical practice guidelines in the United States, with disease-specific panels for breast, colon, pancreas, melanoma, sarcoma, thyroid, gastric, esophageal, hepatobiliary, and more (NCCN Clinical Practice Guidelines in Oncology). European practice follows ESMO Clinical Practice Guidelines, and systemic therapy recommendations are heavily shaped by ASCO guidelines. Screening recommendations come from the USPSTF cancer screening recommendations.
Every note on a cancer patient should identify the current line of therapy and whether the patient is treatment-naïve, on neoadjuvant therapy, post-op within 90 days, on adjuvant therapy, in surveillance, or has recurrence/metastasis. These phrases matter for billing, trial eligibility, and medicolegal clarity.
03 Scribe Documentation Framework
The Oncology SOAP Note
Surgical oncology notes are SOAP-structured but heavier on staging, prior-treatment history, and multidisciplinary planning than general surgery notes. Every new-patient HPI must capture: the tissue diagnosis (when, where, by whom), the histology and grade, receptor status (breast, gastric HER2, colon MSI, sarcoma molecular), the clinical TNM stage, any neoadjuvant therapy already given, prior oncologic surgery, and the genetic testing status.
Presenting problem: Palpable mass, screening abnormality, incidental imaging finding, obstructive symptoms, bleeding, weight loss, pain.
Tissue diagnosis: Date of biopsy, method (core needle, excisional, EUS-FNA, punch, endoscopic), institution, histology, grade, biomarkers.
Staging workup: Imaging modalities and dates (CT chest/abdomen/pelvis, MRI, PET-CT, mammogram, endoscopic ultrasound, bone scan), tumor markers (CA 15-3, CA 19-9, CEA, CA 125, AFP, LDH, chromogranin A).
Treatment history: Prior surgery, chemotherapy regimens (by name and number of cycles), radiation (dose and fractions), immunotherapy, targeted therapy, clinical trial enrollment.
Performance status: ECOG 0–4 and/or Karnofsky 100–0. Document on every visit.
Weight trend: Unintentional weight loss > 5–10% over 3–6 months is prognostically important (cancer cachexia, obstruction, malabsorption).
Vitals: Include weight and BMI every visit; trend over time. Document height at baseline for BSA calculation.
Exam: Focused exam of the primary site and regional nodes, plus a general oncologic review (supraclavicular nodes, liver edge, ascites, skin, surgical scars).
Results: Pathology report verbatim for the relevant specimens, receptor panels, imaging impressions with measurements (RECIST 1.1 target lesions when on therapy), tumor markers with trend, germline and somatic genetic test results.
"[Age/sex] with [histology] of [primary site], [clinical or pathologic TNM stage], [receptor/biomarker profile], [current point on continuum]. Plan: [surgery / neoadjuvant / adjuvant / surveillance / palliation]. Discussed at [tumor board date]. Risks/benefits/alternatives reviewed including [procedure]. Shared decision-making documented. Consent obtained. Next steps: [imaging, labs, appointments]."
04 Breast Cancer Breast
Breast cancer is the most common visceral cancer in women and the bread-and-butter of most surgical oncology practices. The diagnosis arrives via screening mammogram, a palpable mass, nipple discharge, or skin change. Your job as a scribe is to capture histology, receptor status, stage, genetic risk, and the multidisciplinary plan.
Histologic Subtypes
- Ductal carcinoma in situ (DCIS): Non-invasive; confined to ducts. Treated with lumpectomy + radiation or mastectomy; no axillary staging required for pure DCIS but SLNB often performed if mastectomy is planned.
- Invasive ductal carcinoma (IDC): ~75% of invasive breast cancers. Forms a discrete mass.
- Invasive lobular carcinoma (ILC): ~10–15%. Diffuse, "Indian-file" pattern, often mammographically occult, frequently multifocal or bilateral.
- Triple-negative breast cancer (TNBC): ER–/PR–/HER2–. Aggressive, often in younger or BRCA1 patients; responds to chemotherapy and PD-L1 immunotherapy (pembrolizumab).
- HER2-positive: HER2/neu amplified or 3+ on IHC. Highly responsive to trastuzumab, pertuzumab, and T-DM1.
- Hormone receptor–positive (HR+): ER+ and/or PR+; responsive to endocrine therapy (tamoxifen, aromatase inhibitors).
- Inflammatory breast cancer (IBC): Clinical diagnosis of peau d'orange, erythema, warmth, and breast enlargement over < 6 months; always T4d; requires neoadjuvant chemotherapy before mastectomy + radiation.
- Paget disease of the nipple: Eczematous, scaling nipple lesion; often harbors underlying DCIS or invasive carcinoma.
Workup & Staging
Diagnostic mammogram and targeted ultrasound establish the BI-RADS category. BI-RADS 4 or 5 prompts image-guided core needle biopsy. Pathology reports include histology, grade (Nottingham / SBR), ER, PR, HER2 (IHC 0/1+/2+/3+, with FISH for 2+), Ki-67, and sometimes Oncotype DX for HR+/HER2– node-negative disease to guide adjuvant chemotherapy decisions. Staging imaging for clinical stage II+ or symptoms: CT C/A/P, bone scan, or PET-CT. Breast MRI is used for lobular histology, dense breasts, occult primary, or high-risk screening.
Surgical Management
Lumpectomy (breast-conserving surgery, BCS) + whole-breast radiation is oncologically equivalent to mastectomy for stage I–II disease (NSABP B-06). Mastectomy variants: simple/total (removes breast tissue + nipple-areolar complex), skin-sparing (preserves skin envelope for reconstruction), nipple-sparing (preserves NAC when oncologically appropriate), and modified radical (mastectomy + axillary dissection). Axillary staging: sentinel lymph node biopsy (SLNB) for clinically node-negative; axillary lymph node dissection (ALND, levels I–II) for clinically positive nodes, SLN positive not meeting Z11 criteria, or after neoadjuvant therapy with residual disease.
The ACOSOG Z0011 trial (Giuliano et al., JAMA 2011) established that women with T1–T2 cN0 disease undergoing lumpectomy + whole-breast RT with 1–2 positive sentinel nodes do not require completion ALND. These "Z11 criteria" are recited at every breast tumor board.
cT1–T2, cN0, 1–2 positive SLNs, breast-conserving surgery with whole-breast radiation, and planned adjuvant systemic therapy. All five must be true.
NCCN Breast Cancer guideline is the standard reference for sequencing of surgery, radiation, and systemic therapy. For BRCA1/2 carriers, bilateral prophylactic mastectomy reduces breast cancer risk by > 90% and is offered alongside risk-reducing salpingo-oophorectomy.
For every breast cancer HPI, document: laterality, quadrant, size on imaging, histology, grade, ER/PR/HER2, Ki-67, clinical TNM, and genetic testing status. If any are missing, the note is incomplete.
05 Melanoma & Cutaneous Malignancy Skin
Melanoma arises from cutaneous melanocytes and is the deadliest common skin cancer. Scribes will see new diagnoses referred from dermatology after punch or excisional biopsy, patients for wide local excision (WLE) with or without sentinel node biopsy, and surveillance visits for resected or metastatic disease.
Subtypes & Presentation
Subtypes include superficial spreading (most common), nodular, lentigo maligna (chronically sun-damaged skin of elderly), acral lentiginous (palms, soles, nail beds, more common in darker skin), and desmoplastic. The ABCDE mnemonic — Asymmetry, Border irregularity, Color variegation, Diameter > 6 mm, Evolution — guides lay and physician recognition.
Staging & Surgical Margins
Breslow thickness (in millimeters) is the single most important prognostic factor and determines surgical margins and whether to perform SLNB. Ulceration and mitotic rate refine stage.
| Breslow Thickness | WLE Margin | SLNB |
|---|---|---|
| In situ | 0.5–1.0 cm | No |
| ≤ 1.0 mm | 1.0 cm | Discuss if > 0.8 mm or ulcerated |
| 1.01–2.0 mm | 1–2 cm | Yes |
| 2.01–4.0 mm | 2.0 cm | Yes |
| > 4.0 mm | 2.0 cm | Yes |
The MSLT-II trial (Faries et al., NEJM 2017) established that completion lymph node dissection after positive SLNB does not improve melanoma-specific survival; nodal observation with ultrasound is now standard. For unresectable or stage IV disease, systemic therapy is driven by BRAF V600 mutation status (dabrafenib + trametinib) and PD-1 inhibition (nivolumab, pembrolizumab, and the ipilimumab + nivolumab combination). NCCN Melanoma guideline is the reference.
Other cutaneous malignancies a surgical oncologist may manage include Merkel cell carcinoma (aggressive neuroendocrine skin cancer, often requires WLE + SLNB + adjuvant RT), dermatofibrosarcoma protuberans (DFSP), and high-risk squamous cell carcinoma requiring nodal dissection or Mohs referral.
06 Soft Tissue Sarcoma & GIST Sarcoma
Soft tissue sarcomas are a heterogeneous group of mesenchymal malignancies comprising < 1% of adult cancers but requiring specialized multidisciplinary care. Over 70 histologic subtypes exist — the most common are undifferentiated pleomorphic sarcoma (UPS), liposarcoma (well-differentiated, dedifferentiated, myxoid, pleomorphic), leiomyosarcoma, synovial sarcoma, myxofibrosarcoma, and angiosarcoma.
Extremity Sarcoma
Presents as a painless, enlarging mass. Any soft-tissue mass > 5 cm, deep to fascia, enlarging, or painful deserves MRI with contrast and referral before biopsy. Always biopsy along the planned incision line so that the biopsy tract can be excised en bloc at definitive surgery. Treatment is wide local excision with 1–2 cm margin (or anatomical barrier such as fascia), often with neoadjuvant or adjuvant radiation to reduce local recurrence. Amputation is now rare; limb-salvage is the default.
Retroperitoneal Sarcoma
Presents late, often enormous, with vague abdominal discomfort or early satiety. The majority are liposarcoma or leiomyosarcoma. Surgery involves compartmental resection — removing the tumor en bloc with adjacent organs (kidney, colon, psoas, diaphragm) to achieve R0 resection. The TARPSWG (Trans-Atlantic Retroperitoneal Sarcoma Working Group) leads modern standardization.
GIST
Gastrointestinal stromal tumor (GIST) arises from the interstitial cells of Cajal, most often in the stomach (60%) or small bowel. Driven by activating KIT (CD117) or PDGFRA mutations — hence the transformative effect of imatinib. Surgical principles: R0 resection with intact pseudocapsule; no routine lymphadenectomy (GIST rarely spreads to nodes); wedge resection or segmental bowel resection is preferred over radical procedures. High-risk tumors (size > 5 cm, mitotic rate > 5/50 HPF, non-gastric site, rupture) receive adjuvant imatinib for 3 years.
NCCN Soft Tissue Sarcoma guideline and the Memorial Sloan Kettering sarcoma nomogram (MSKCC nomogram, inputs: age, size, histology, grade, depth, site) are the usual decision aids cited at sarcoma tumor board.
07 Gastric & Esophageal Cancer GI
Gastric Cancer
Most gastric adenocarcinomas present at locally advanced stage with epigastric pain, early satiety, weight loss, or iron-deficiency anemia. Endoscopy with biopsy establishes diagnosis; EUS stages depth (T) and regional nodes (N); CT and often staging laparoscopy assess metastasis. HER2 and PD-L1 are tested on biopsy. Signet-ring cell and diffuse-type (Lauren classification) histologies are aggressive.
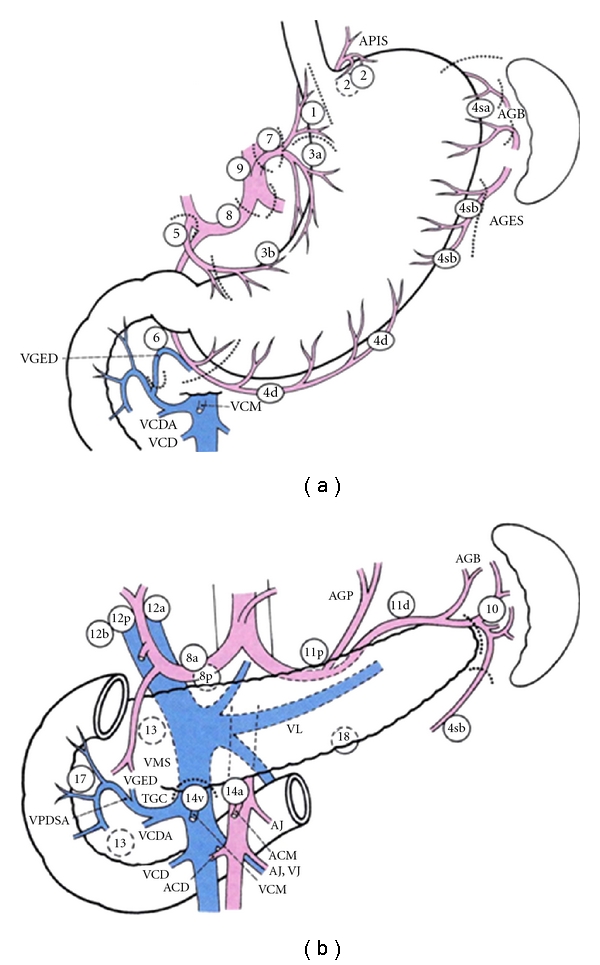
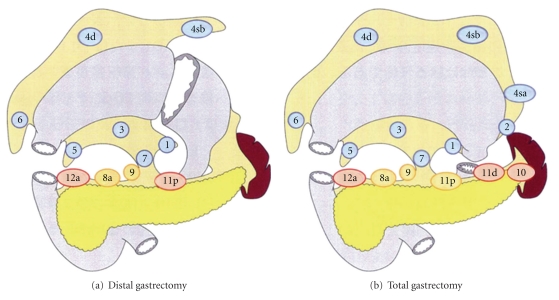
Treatment for resectable disease is perioperative chemotherapy (FLOT: 5-FU, leucovorin, oxaliplatin, docetaxel) plus D2 gastrectomy — partial (distal), subtotal, or total based on tumor location. D2 lymphadenectomy removes the perigastric (stations 1–6) plus extraperigastric nodes along the left gastric, common hepatic, celiac, splenic, and splenic hilum (stations 7, 8a, 9, 10, 11). The NCCN Gastric Cancer guideline reflects the FLOT4 trial (Al-Batran et al., Lancet 2019).
Esophageal Cancer
Two main histologies: squamous cell carcinoma (upper/mid esophagus, smoking/alcohol) and adenocarcinoma (lower esophagus / GE junction, GERD and Barrett esophagus). Presents with dysphagia progressing from solids to liquids, weight loss, and odynophagia. Workup: EGD with biopsy, EUS, CT C/A/P, PET-CT.
Standard-of-care for locally advanced disease is neoadjuvant chemoradiation (CROSS regimen: carboplatin/paclitaxel + 41.4 Gy) followed by esophagectomy — Ivor Lewis (laparotomy + right thoracotomy with intrathoracic anastomosis), McKeown (three-field: abdomen, right chest, left neck), transhiatal (abdomen + neck, no thoracotomy), or minimally invasive/robotic versions. The CROSS trial (van Hagen et al., NEJM 2012) established neoadjuvant chemoradiation as standard.
08 Pancreatic Cancer & Pancreatic NETs Pancreas
Pancreatic ductal adenocarcinoma (PDAC) is lethal: 5-year survival ~12% overall, with only ~20% of patients presenting with resectable disease. Scribes will document painless jaundice (head lesions), weight loss, new-onset diabetes, steatorrhea, and epigastric/back pain.
Resectability
Resectability is defined by vascular contact with the SMA, celiac axis, common hepatic, SMV, and portal vein. The four categories used in every pancreas tumor board are: resectable (no arterial contact, < 180° venous contact without irregularity), borderline resectable (limited arterial contact or reconstructible venous involvement), locally advanced (unreconstructible), and metastatic.
Surgical Management
Head and uncinate tumors: pancreaticoduodenectomy (Whipple) removes the pancreatic head, duodenum, gallbladder, distal bile duct, and proximal jejunum with reconstruction via pancreaticojejunostomy, hepaticojejunostomy, and gastrojejunostomy. Body/tail: distal pancreatectomy with splenectomy (spleen-preserving in benign disease). Multifocal disease or chronic involvement: total pancreatectomy (producing brittle diabetes).
Neoadjuvant FOLFIRINOX or gemcitabine/nab-paclitaxel is increasingly used for borderline resectable and resectable disease. CA 19-9 is the key tumor marker — document pre- and post-op values. NCCN Pancreatic Adenocarcinoma guideline is the reference.
Pancreatic Neuroendocrine Tumors (PNET)
PNETs are a separate disease from PDAC — indolent, arising from islet cells, may be functional (insulinoma, gastrinoma, glucagonoma, VIPoma, somatostatinoma) or non-functional. Surgery ranges from enucleation (small, benign-appearing) to formal pancreatectomy with lymphadenectomy. Liver metastases are common and often resected or ablated. MEN1 syndrome must be considered in multifocal PNETs.
09 Hepatobiliary Malignancies Liver / Biliary
Hepatocellular Carcinoma (HCC)
HCC almost always arises in the setting of chronic liver disease — hepatitis B, hepatitis C, alcoholic cirrhosis, NAFLD/NASH, hemochromatosis. AFP is elevated in ~60%. Diagnosis is often made radiographically in a cirrhotic liver using LI-RADS criteria on multiphase CT or MRI without needing biopsy. Treatment is driven by the Barcelona Clinic Liver Cancer (BCLC) staging system incorporating tumor burden, liver function (Child-Pugh), and performance status. Options: resection (solitary tumor, preserved function), ablation (RFA, MWA), transarterial chemoembolization (TACE), radioembolization (Y90), systemic therapy (atezolizumab/bevacizumab, lenvatinib), and liver transplant for those meeting Milan criteria (1 tumor ≤ 5 cm or up to 3 tumors each ≤ 3 cm).
Cholangiocarcinoma
Classified by anatomic location: intrahepatic (mass in hepatic parenchyma, treated with hepatectomy), perihilar / Klatskin tumor (at the biliary confluence, often requires extended hepatectomy + caudate resection + bile duct excision + biliary-enteric reconstruction), and distal (treated with Whipple). Bismuth-Corlette classification (I–IV) describes hilar extent.
Gallbladder Cancer
Often discovered incidentally on pathology after cholecystectomy for presumed benign disease. T1b or deeper requires re-operation for extended cholecystectomy: resection of segments IVb/V of the liver plus portal lymphadenectomy (stations 8, 12, 13).
NCCN Hepatobiliary Cancers guideline covers all three.
Liver-Directed Therapies
For colorectal and neuroendocrine liver metastases, hepatic resection + ablation is potentially curative. Parenchymal-sparing resection and two-stage hepatectomy with portal vein embolization are advanced strategies. The hepatic artery infusion (HAI) pump (floxuridine delivered directly to liver metastases) is offered at select centers for colorectal liver metastases. TACE and Y90 radioembolization are delivered by interventional radiology for unresectable disease.
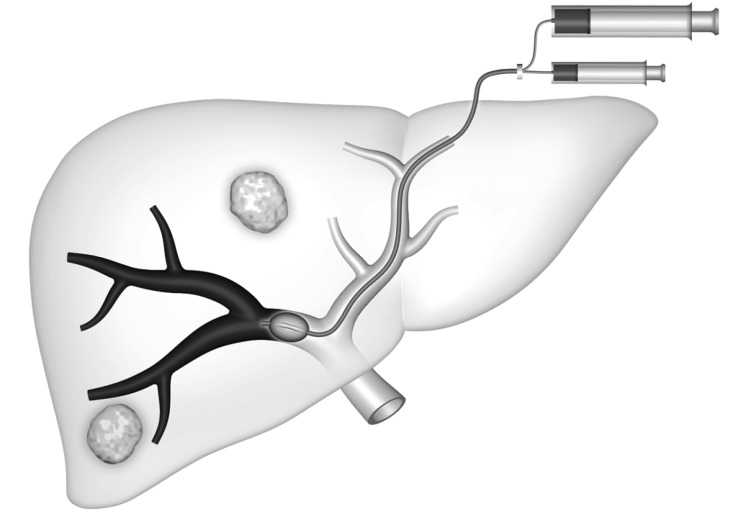
10 Colorectal & Anal Cancer (Surgical Onc Overlap) GI
Although most colorectal cancer is managed by colorectal surgery, surgical oncologists frequently participate in liver metastasectomy, recurrent/locally advanced cases, peritoneal disease, and hereditary syndromes. Key documentation elements: MSI/MMR status, KRAS/NRAS/BRAF mutational profile, CEA level, prior resection and chemotherapy history, synchronous versus metachronous metastases.
Surgical oncology involvement peaks in three scenarios: (1) colorectal liver metastases (resection ± ablation, neoadjuvant FOLFOX/FOLFIRI, sometimes biologics), (2) peritoneal carcinomatosis (see Section 11), and (3) hereditary syndromes such as FAP requiring total proctocolectomy or Lynch syndrome requiring extended resection and counseling. NCCN Colon Cancer guideline.
Anal squamous cell carcinoma is primarily treated with Nigro protocol chemoradiation (5-FU + mitomycin C + RT) with surgery (APR) reserved for salvage. Pelvic exenteration may be required for recurrent disease invading adjacent organs.
11 Peritoneal Carcinomatosis, CRS & HIPEC Peritoneum
Cytoreductive surgery (CRS) with hyperthermic intraperitoneal chemotherapy (HIPEC) is a defining surgical oncology intervention for peritoneal surface malignancies: appendiceal mucinous neoplasms / pseudomyxoma peritonei, peritoneal mesothelioma, selected colorectal peritoneal metastases, and ovarian cancer.
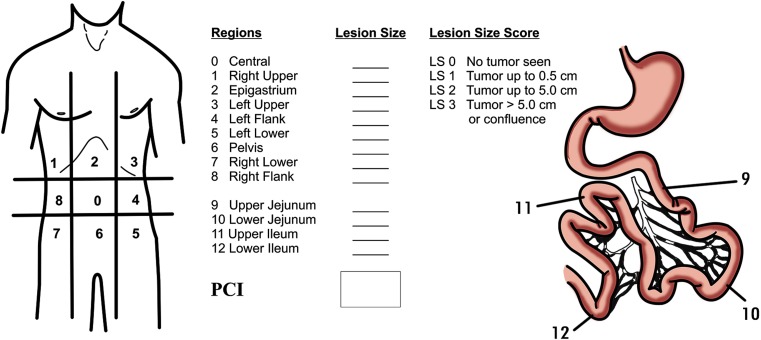
The operation involves complete peritonectomy (stripping the peritoneum and resecting involved organs) to achieve a completeness of cytoreduction (CC) score of 0 or 1 (no residual nodule > 2.5 mm), followed by instillation of heated chemotherapy (mitomycin C, oxaliplatin, or cisplatin at ~42°C) circulated in the abdomen for 30–90 minutes. The peritoneal cancer index (PCI, 0–39) scores disease burden across 13 abdominopelvic regions and is the most commonly cited prognostic metric.
High morbidity: prolonged ileus, anastomotic leak, hematologic toxicity (neutropenia, thrombocytopenia), bile leak, fluid shifts, AKI, delayed gastric emptying. Clavien-Dindo grade III–IV complications occur in 25–40%.
12 Endocrine Malignancy — Thyroid, Parathyroid, Adrenal Endocrine
Thyroid Cancer
- Papillary (PTC, ~80%): Excellent prognosis; lymphatic spread. Treated by lobectomy or total thyroidectomy ± central neck dissection, RAI ablation for intermediate/high risk.
- Follicular (FTC, ~10%): Hematogenous spread (lung, bone). Diagnosis often requires surgical pathology (FNA cannot distinguish follicular adenoma from carcinoma).
- Medullary (MTC, ~4%): Arises from parafollicular C cells; secretes calcitonin and CEA; associated with MEN2A / MEN2B (RET mutation); requires total thyroidectomy + central neck dissection.
- Anaplastic (ATC, ~1%): Highly lethal; rapidly enlarging neck mass; median survival months; treatment is palliative ± BRAF-targeted therapy when applicable.
NCCN Thyroid Carcinoma guideline and ATA guidelines inform the extent of surgery and RAI decisions.
Parathyroid Carcinoma
Rare; presents with markedly elevated calcium (> 14 mg/dL) and PTH (often 5–10× normal), a palpable neck mass, and renal/bone disease. Treatment is en bloc resection of the parathyroid with ipsilateral thyroid lobe; avoid capsular violation.
Adrenal Cancer (Adrenocortical Carcinoma, ACC)
Presents as a large (> 4–6 cm) adrenal mass ± hormonal hypersecretion (Cushing syndrome, virilization, feminization, hyperaldosteronism). Workup includes dexamethasone suppression, plasma metanephrines (always exclude pheochromocytoma before surgery), aldosterone/renin, DHEAS, 17-OH progesterone. Treatment is open adrenalectomy with en bloc resection and mitotane for adjuvant therapy. Laparoscopic adrenalectomy is reserved for tumors of clearly benign imaging features.
13 Neuroendocrine Tumors (Carcinoid & NETs) NET
Neuroendocrine tumors arise from enterochromaffin cells throughout the GI tract, pancreas, and lung. Most are low-grade (Ki-67 < 3%, G1) or intermediate (3–20%, G2), with high-grade neuroendocrine carcinoma (G3) being an aggressive exception. The classic carcinoid syndrome (flushing, diarrhea, bronchospasm, right-sided valvular heart disease) occurs when liver metastases bypass hepatic metabolism of serotonin.
Workup: chromogranin A, 24-hour urine 5-HIAA, Ga-68 DOTATATE PET (most sensitive imaging), CT/MRI. Management depends on site and grade: small bowel NETs require segmental resection with mesenteric lymphadenectomy and incidental hepatic metastasectomy; pancreatic NETs per Section 8; somatostatin analogues (octreotide, lanreotide) for hormone control and tumor stabilization; PRRT (Lu-177 DOTATATE) for progressive disease; everolimus and sunitinib for pancreatic NETs.
14 Oligometastatic Disease & Hereditary / Prophylactic Surgery Genetics
Oligometastatic disease — a small, potentially resectable number of metastases (classically ≤ 5 lesions in ≤ 2–3 organs) — is increasingly treated with curative intent. Colorectal and neuroendocrine liver metastases and sarcoma pulmonary metastases are the archetypes. Lung metastasectomy (wedge, segmentectomy) is performed via VATS or thoracotomy.
BRCA1/2: Bilateral prophylactic mastectomy and bilateral salpingo-oophorectomy by age 35–40.
FAP (APC): Prophylactic total proctocolectomy with IPAA in adolescence/early adulthood given near-certain colon cancer risk.
Lynch syndrome (MLH1/MSH2/MSH6/PMS2): Extended colectomy (subtotal) at index cancer; consideration of hysterectomy/BSO.
MEN2 (RET): Prophylactic total thyroidectomy — by age 5 for MEN2A, by age 1 for MEN2B, based on codon-specific risk.
CDH1 (hereditary diffuse gastric cancer): Prophylactic total gastrectomy recommended in mutation carriers.
15 Surgical Oncology Procedures — A to Z
This table is the scribe's quick-reference for procedure names as they are spoken in clinic and the OR. Use it to correctly chart what was performed, proposed, or declined.
| Procedure | What It Involves | Primary Indication |
|---|---|---|
| Lumpectomy / partial mastectomy | Excision of tumor with rim of normal breast tissue, with wire/seed localization if non-palpable | Early-stage breast cancer, DCIS |
| Simple (total) mastectomy | Removal of entire breast including NAC, preserving pectoralis | DCIS, prophylactic, multifocal disease |
| Modified radical mastectomy | Simple mastectomy + axillary level I–II dissection | Locally advanced breast cancer with clinically positive nodes |
| Skin-sparing / nipple-sparing mastectomy | Preserves skin envelope ± NAC for immediate reconstruction | Appropriate oncologic anatomy, reconstruction planned |
| Sentinel lymph node biopsy (SLNB) | Injection of radiotracer ± blue dye; excision of first draining node(s) | Breast, melanoma, Merkel, vulvar, endometrial staging |
| Axillary lymph node dissection (ALND) | En bloc removal of level I and II axillary nodes | Node-positive breast cancer not meeting Z11 |
| Wide local excision (WLE) | Resection with defined margin around primary lesion | Melanoma, sarcoma, cutaneous malignancy |
| Inguinal lymphadenectomy | Superficial ± deep groin node dissection | Melanoma, vulvar, anal, penile cancer |
| Neck dissection | Level I–V; central compartment for thyroid; modified radical for H&N | Thyroid, head and neck, parotid, melanoma |
| Total / hemi / completion thyroidectomy | Removal of both, one, or remaining thyroid lobe with RLN and parathyroid preservation | Thyroid carcinoma, MEN2 prophylaxis |
| Parathyroidectomy | Excision of hyperfunctioning or malignant gland(s) | Primary hyperparathyroidism, parathyroid cancer |
| Adrenalectomy (open or laparoscopic) | Unilateral adrenal removal | ACC, pheochromocytoma, functional adenoma |
| Splenectomy | Total or partial splenic removal | Distal pancreatectomy, lymphoma staging (rare) |
| Distal pancreatectomy | Resection of pancreatic body/tail, usually with splenectomy | Body/tail PDAC, PNET, mucinous cysts |
| Whipple (pancreaticoduodenectomy) | Pancreas head, duodenum, gallbladder, distal CBD, proximal jejunum; triple reconstruction | Head/uncinate PDAC, distal cholangiocarcinoma, duodenal/ampullary cancer |
| Total pancreatectomy | Whole gland + duodenum + spleen; results in brittle diabetes | Multifocal IPMN, diffuse PDAC |
| Subtotal / total gastrectomy with D2 | Partial or complete stomach resection with extended nodal dissection | Gastric adenocarcinoma |
| Esophagectomy (Ivor Lewis, McKeown, transhiatal, MIE) | Resection of esophagus with gastric conduit reconstruction | Esophageal cancer |
| Hepatic resection (segmentectomy, hemihepatectomy, extended, trisegmentectomy) | Anatomic or nonanatomic parenchymal resection | HCC, CRC mets, cholangiocarcinoma |
| Klatskin resection | Extended hepatectomy + caudate + bile duct excision + biliary reconstruction | Perihilar cholangiocarcinoma |
| Extended cholecystectomy | Cholecystectomy + liver segments IVb/V + portal lymphadenectomy | Gallbladder cancer T1b or deeper |
| Hepatic artery infusion (HAI) pump | Subcutaneous pump with catheter to hepatic artery delivering floxuridine | CRC liver metastases at select centers |
| Thermal / cryo ablation | RFA, MWA, cryoablation under imaging guidance | Small HCC, liver mets, lung mets, soft tissue lesions |
| TACE / Y90 radioembolization | Interventional radiology delivery of chemo or radioactive microspheres via hepatic artery | Unresectable HCC, hepatic metastases |
| CRS + HIPEC | Complete peritonectomy + heated intraperitoneal chemotherapy | Peritoneal carcinomatosis from appendiceal, CRC, ovarian, mesothelioma |
| Retroperitoneal sarcoma resection | Compartmental en bloc resection, often multivisceral | Retroperitoneal liposarcoma, leiomyosarcoma |
| Sternotomy / thoracotomy / VATS metastasectomy | Wedge or anatomic lung resection | Sarcoma, CRC, melanoma, RCC lung metastases |
| Robotic oncologic surgery | Da Vinci platform for abdominal, thoracic, and pelvic oncologic operations | Most modern GI and hepatobiliary oncologic surgery |
| Oncoplastic breast surgery | Combined lumpectomy + local tissue rearrangement for cosmesis | Breast cancer with unfavorable tumor/breast ratio |
16 Perioperative Oncology & Post-Op Complications
Cancer patients present unique perioperative challenges: malnutrition, immunosuppression from chemotherapy, bone marrow suppression, deconditioning, and elevated thromboembolic risk. Enhanced Recovery After Surgery (ERAS) protocols are now standard for most oncologic operations — carbohydrate loading, minimal fasting, multimodal opioid-sparing analgesia (acetaminophen, gabapentinoids, NSAIDs where safe, regional blocks, TAP/epidural/paravertebral), early mobilization, and early feeding.
Pre-op antibiotics: Cefazolin ± metronidazole within 60 minutes of incision; vancomycin for MRSA risk; ertapenem or piperacillin-tazobactam for GI cases.
VTE prophylaxis: Cancer patients are high risk. SC heparin or enoxaparin pre-op and post-op; extended 28-day prophylaxis after major abdominal/pelvic oncologic surgery is guideline-recommended per ASCO VTE guideline (Key et al., JCO 2020).
Multimodal analgesia: Scheduled acetaminophen, gabapentin, celecoxib if appropriate, regional block, transition to oral opioids on POD 1–2.
Nutrition: Albumin < 3.0 and/or > 10% weight loss warrants preoperative optimization; consider immunonutrition drinks or enteral supplementation.
Procedure-Specific Complications
- Whipple: Postoperative pancreatic fistula (POPF, graded A/B/C by ISGPF), delayed gastric emptying, post-pancreatectomy hemorrhage, bile leak, intra-abdominal abscess, anastomotic leak.
- Esophagectomy: Anastomotic leak (cervical > intrathoracic), chyle leak, RLN injury, pneumonia, conduit ischemia, atrial fibrillation.
- Mastectomy / ALND: Seroma, lymphedema, axillary web syndrome, intercostobrachial nerve numbness, capsular contracture in reconstruction.
- Hepatectomy: Bile leak, post-hepatectomy liver failure (ISGLS 50:50 criteria), ascites, pleural effusion, hemorrhage.
- Gastrectomy: Anastomotic leak, dumping syndrome, vitamin B12 deficiency, iron deficiency, nutritional decline.
- Thyroidectomy: RLN injury (hoarseness), hypocalcemia (transient or permanent hypoparathyroidism), hematoma requiring emergent evacuation.
- CRS/HIPEC: High overall morbidity as noted in Section 11.
Complications are graded with the Clavien-Dindo classification:
| Grade | Description |
|---|---|
| I | Deviation from normal post-op course; no drug, surgical, radiologic, or endoscopic intervention required (antiemetics, antipyretics, wound opening at bedside OK) |
| II | Requires pharmacologic treatment outside the Grade I list, including transfusion and TPN |
| IIIa | Requires surgical, endoscopic, or radiologic intervention without general anesthesia |
| IIIb | Requires intervention under general anesthesia |
| IVa | Life-threatening complication with single-organ dysfunction requiring ICU |
| IVb | Multi-organ dysfunction requiring ICU |
| V | Death of the patient |
17 Imaging, Staging & Pathology Reports
Surgical oncology documentation is imaging- and pathology-heavy. Every note references report findings and measurements; scribes must transcribe verbatim numbers and impressions.
CT chest/abdomen/pelvis with contrast: Standard staging study for most solid tumors. Look for primary size, T descriptor, regional nodes, distant metastases (liver, lung, peritoneum, bone).
MRI: Liver (HCC and CRC mets with eovist/primovist), rectum (pelvic MRI for T/N staging), soft tissue mass (extremity sarcoma), breast (occult primary, lobular, dense breasts).
PET-CT (FDG): Lymphoma, esophageal, lung, melanoma, head/neck; used to detect occult metastasis and monitor response.
Endoscopic ultrasound (EUS): Esophageal, gastric, pancreatic — defines depth of invasion and enables FNA.
DOTATATE PET: Most sensitive modality for well-differentiated neuroendocrine tumors.
Ultrasound: Breast, thyroid, superficial masses, nodal basins after melanoma SLNB.
RECIST 1.1 (Response Criteria)
RECIST 1.1 categorizes treatment response on imaging: CR complete response (all target lesions gone); PR partial response (≥ 30% decrease in sum of target diameters); SD stable disease; PD progressive disease (≥ 20% increase or new lesion). Target lesions are measurable, typically up to 5 (2 per organ).
Pathology Report Elements
Every final pathology report on a resection includes: specimen description, gross findings, histologic type and grade, size, margins (distance in mm from each margin), lymphovascular and perineural invasion, lymph nodes examined/positive, extranodal extension, receptor/biomarker panel, and pathologic TNM stage. Synoptic reports following the CAP (College of American Pathologists) protocol are the standard format and should be cited verbatim in the assessment.
18 Medications You Must Know
Surgical oncologists don't prescribe most chemotherapy themselves, but they must document what the patient has received and manage perioperative medications. Know these drugs by both generic and brand names.
Perioperative
| Drug | Class / Use | Notes |
|---|---|---|
| Cefazolin (Ancef) | First-gen cephalosporin | Standard pre-op antibiotic; redose if case > 4 h or EBL > 1500 mL |
| Ertapenem (Invanz) | Carbapenem | Whipple, contaminated cases, biliary surgery |
| Metronidazole (Flagyl) | Antianaerobic | Added to cefazolin for colorectal, hepatobiliary |
| Enoxaparin (Lovenox) | LMWH | VTE prophylaxis — 40 mg SC qD; extended 28 days post-major abdominal oncologic surgery |
| Heparin SC | Unfractionated heparin | 5000 U SC TID for VTE prophylaxis; preferred in renal failure |
| Acetaminophen (Tylenol / IV Ofirmev) | Non-opioid analgesic | Scheduled 1 g Q6H; cornerstone of ERAS |
| Ketorolac (Toradol) | NSAID | Avoid in renal failure, bleeding risk, or immediately after GI anastomosis |
| Gabapentin / pregabalin | Gabapentinoid | Opioid-sparing adjunct |
| Ondansetron (Zofran) | 5-HT3 antagonist | PONV prophylaxis |
| Dexamethasone | Corticosteroid | PONV, analgesic adjunct |
Oncologic Medications (Overview — Chart What the Patient is On)
| Drug / Regimen | Class | Commonly Used For |
|---|---|---|
| FOLFIRINOX | Cytotoxic combo (5-FU, leucovorin, irinotecan, oxaliplatin) | Pancreatic adenocarcinoma (neo/adj, metastatic) |
| Gemcitabine + nab-paclitaxel | Cytotoxic combo | Pancreatic adenocarcinoma |
| FLOT | 5-FU, leucovorin, oxaliplatin, docetaxel | Perioperative gastric/GEJ cancer |
| FOLFOX / FOLFIRI | Cytotoxic combo | Colorectal, gastric, pancreas |
| CROSS (carboplatin + paclitaxel + RT) | Chemoradiation | Neoadjuvant esophageal |
| Trastuzumab (Herceptin) | Anti-HER2 monoclonal | HER2+ breast, gastric |
| Pertuzumab (Perjeta) | Anti-HER2 | HER2+ breast with trastuzumab |
| T-DM1 (Kadcyla), T-DXd (Enhertu) | HER2 ADC | HER2+/low breast |
| Pembrolizumab (Keytruda) / Nivolumab (Opdivo) | PD-1 inhibitor | Melanoma, MSI-H colon, esophageal, gastric, TNBC, HCC |
| Ipilimumab (Yervoy) | CTLA-4 inhibitor | Melanoma (often combined with nivolumab) |
| Dabrafenib + trametinib | BRAF + MEK inhibitor | BRAF V600E melanoma, ATC |
| Imatinib (Gleevec) | KIT tyrosine kinase inhibitor | GIST (neoadjuvant, adjuvant, metastatic) |
| Tamoxifen | SERM | HR+ breast cancer (pre/postmenopausal) |
| Anastrozole, letrozole, exemestane | Aromatase inhibitors | HR+ breast, postmenopausal |
| Octreotide / lanreotide | Somatostatin analogue | Carcinoid syndrome, NET tumor control |
| Lu-177 DOTATATE (Lutathera) | Peptide receptor radionuclide therapy | Progressive well-differentiated NET |
| Atezolizumab + bevacizumab | PD-L1 + anti-VEGF | Unresectable HCC (first-line per IMbrave150) |
| Mitomycin C | Alkylating | HIPEC, anal SCC (with 5-FU) |
19 Classification & Staging Systems (AJCC 8)
All staging in the United States follows AJCC Cancer Staging Manual 8th Edition. The following tables enumerate the most important systems you will hear cited in surgical oncology clinic. Every cancer has its own T, N, and M definitions — below is the conceptual overview and condensed stage-group framework.
| Grade | Clinical Description |
|---|---|
| 0 | Fully active, no restrictions — pre-illness baseline |
| 1 | Restricted in strenuous activity but ambulatory and able to do light work |
| 2 | Ambulatory, self-care, unable to work, up and about > 50% of waking hours |
| 3 | Limited self-care, confined to bed or chair > 50% of waking hours |
| 4 | Completely disabled, no self-care, totally confined to bed or chair |
| 5 | Dead |
100 = normal, no complaints; 90 = minor symptoms; 80 = normal activity with effort; 70 = self-care, unable to work; 60 = occasional assistance; 50 = considerable assistance and frequent medical care; 40 = disabled, special care needed; 30 = severely disabled, hospitalization indicated; 20 = very sick, active support needed; 10 = moribund; 0 = dead.
Breast Cancer AJCC 8 (Anatomic)
| Stage | T | N | M |
|---|---|---|---|
| 0 | Tis (DCIS) | N0 | M0 |
| IA | T1 | N0 | M0 |
| IB | T0–1 | N1mi | M0 |
| IIA | T0–1 N1 / T2 N0 | — | M0 |
| IIB | T2 N1 / T3 N0 | — | M0 |
| IIIA | T0–3 N2 / T3 N1 | — | M0 |
| IIIB | T4 (inflammatory, skin/chest wall) | N0–2 | M0 |
| IIIC | Any T | N3 | M0 |
| IV | Any T | Any N | M1 |
AJCC 8 also incorporates prognostic staging with ER, PR, HER2, and grade — a small T1 TNBC can upstage, and a large HR+ HER2+ low-grade can downstage.
Colon Cancer AJCC 8
| Stage | T | N | M |
|---|---|---|---|
| 0 | Tis | N0 | M0 |
| I | T1–T2 | N0 | M0 |
| IIA / IIB / IIC | T3 / T4a / T4b | N0 | M0 |
| IIIA–IIIC | T1–T4 | N1–N2 | M0 |
| IVA / IVB / IVC | Any | Any | M1a / M1b / M1c (peritoneum) |
Gastric Cancer AJCC 8 (Condensed)
| Stage | Clinical Correlate |
|---|---|
| IA | T1 N0 — mucosal/submucosal, no nodes |
| IB | T1 N1 / T2 N0 |
| II | Progressively deeper invasion and/or 1–2 positive nodes |
| III | T4 or ≥ 3 positive nodes |
| IV | M1 (distant, peritoneal, non-regional nodes) |
Pancreatic AJCC 8
| Stage | T | N | M |
|---|---|---|---|
| IA | T1 (≤ 2 cm) | N0 | M0 |
| IB | T2 (2–4 cm) | N0 | M0 |
| IIA | T3 (> 4 cm) | N0 | M0 |
| IIB | T1–T3 | N1 (1–3 nodes) | M0 |
| III | Any T4 or N2 (≥ 4 nodes) | — | M0 |
| IV | Any | Any | M1 |
Melanoma AJCC 8 (T by Breslow)
| T Stage | Thickness | Ulceration |
|---|---|---|
| T1a | < 0.8 mm | No |
| T1b | < 0.8 mm with ulceration OR 0.8–1.0 mm | ± |
| T2a/b | 1.01–2.0 mm | No / Yes |
| T3a/b | 2.01–4.0 mm | No / Yes |
| T4a/b | > 4.0 mm | No / Yes |
Lung Cancer AJCC 8 (Condensed)
| Stage | Rough Descriptor |
|---|---|
| IA1–IA3 | T1 ≤ 3 cm, N0, M0 |
| IB | T2a (> 3–4 cm), N0, M0 |
| II | T2b–T3 or N1 |
| IIIA–IIIC | T4 or N2–N3 |
| IVA / IVB | Single vs multiple distant metastases |
Sarcoma Grading & MSKCC Nomogram
Soft tissue sarcoma is graded 1–3 (FNCLCC: differentiation + mitoses + necrosis). The MSKCC sarcoma nomogram predicts sarcoma-specific survival from age, tumor size, histology, grade, depth, and anatomic site — it's the number the surgeon quotes in a consent discussion when estimating prognosis.
20 Physical Exam — The Surgical Oncology Exam
Surgical oncology exam is organ-directed but always includes a general oncologic survey: performance status observation, cachexia, jaundice, adenopathy (cervical, supraclavicular/Virchow, axillary, inguinal), liver edge, ascites, and palpable masses.
"Symmetric breasts without overlying skin change, dimpling, peau d'orange, or nipple retraction. [Side/quadrant]: palpable firm, [size in cm] mass at [clock position], [distance from nipple], [mobile/fixed], [tender/non-tender]. No axillary, supraclavicular, or infraclavicular adenopathy bilaterally. Contralateral breast without mass."
"Abdomen soft, non-distended, non-tender. No palpable mass, hepatomegaly, splenomegaly, or ascites. No surgical scars [or: well-healed midline laparotomy scar]. No palpable umbilical (Sister Mary Joseph) nodule. No inguinal adenopathy."
"Total body skin exam performed. [Site] with [size] pigmented lesion, well-healed WLE scar, no satellite or in-transit lesions. No palpable adenopathy in [draining nodal basin]. Remainder of skin without suspicious pigmented lesions."
21 Risk Factors, Genetics & Comorbidities
Cancer risk factor documentation is central to the surgical oncology HPI. Every patient's note should capture the following domains, because they drive screening, genetic referral, and treatment decisions.
Tobacco: Pack-years, current vs former, quit date. Drives lung, esophageal, gastric, pancreatic, bladder, H&N cancer risk.
Alcohol: Drinks per day/week. Liver, esophageal, H&N, breast cancer.
Family history: First- and second-degree relatives with cancer, ages at diagnosis, sites. Two or more primaries, early onset (< 50), or rare combinations trigger genetics referral.
Germline testing: BRCA1/2, PALB2, TP53 (Li-Fraumeni), CDH1, Lynch panel, APC, RET, MEN1, VHL, PTEN. Document results explicitly — "Panel negative" and "not yet tested" are both meaningful.
Environmental/occupational: Asbestos (mesothelioma), radiation, HBV/HCV/HIV, HPV, H. pylori, Barrett esophagus, cirrhosis, IBD.
Performance & nutrition: ECOG, weight change, albumin/prealbumin, sarcopenia on imaging.
Comorbidities affecting operative risk: CAD, CHF (EF), COPD (FEV1), CKD, cirrhosis (Child-Pugh, MELD), diabetes (A1C), anticoagulation.
22 Abbreviations Master List
Diseases & Histology
Procedures
Staging, Imaging & Pathology
Medications & Perioperative
23 Sample HPI Templates
These templates read like real surgical oncology clinic notes. Use them as pattern templates during your first weeks.
"Ms. [Name] is a 54-year-old postmenopausal female with no prior oncologic history referred for newly diagnosed right breast cancer. She was found to have an abnormal screening mammogram on [date] showing a 1.6 cm spiculated mass at the right breast 10 o'clock, 4 cm from the nipple (BI-RADS 5). Ultrasound-guided core needle biopsy on [date] demonstrated invasive ductal carcinoma, Nottingham grade 2, ER 95% / PR 80% / HER2 negative (IHC 1+), Ki-67 15%. Staging MRI shows no additional ipsilateral or contralateral lesions. Physical exam without palpable adenopathy. Denies breast pain, nipple discharge, skin change, weight loss, or bone pain. ECOG 0. Genetic testing pending; family history notable for maternal aunt with breast cancer at age 62. Clinical stage cT1c N0 M0. Discussed breast-conserving surgery with sentinel lymph node biopsy versus mastectomy. Patient elects lumpectomy with SLNB and whole-breast radiation, with likely adjuvant endocrine therapy pending Oncotype DX."
"Mr. [Name] is a 62-year-old fair-skinned male with a history of multiple sunburns and prior non-melanoma skin cancers, referred after punch biopsy of a changing pigmented lesion on the left upper back. Pathology (dermatopathology, [date]) reports malignant melanoma, superficial spreading type, Breslow depth 1.4 mm, non-ulcerated, mitotic rate 2/mm², deep and peripheral margins involved on the shave biopsy. He denies constitutional symptoms and there are no palpable lymph nodes in bilateral axillae or cervical chains. CT chest/abdomen/pelvis obtained for baseline imaging without evidence of metastasis. Clinical stage cT2a. Plan: wide local excision with 1–2 cm margin and sentinel lymph node biopsy of the left axilla with lymphoscintigraphy. Risks and benefits reviewed including lymphedema, seroma, and false-negative SLN."
"Mr. [Name] is a 68-year-old male who presented to his PCP 3 weeks ago with painless jaundice, dark urine, and 12-pound unintentional weight loss over 2 months. CT pancreas protocol revealed a 2.8 cm hypoenhancing mass in the pancreatic head with upstream biliary and pancreatic ductal dilation. ERCP with metal stent placement relieved the jaundice; total bilirubin has decreased from 14 to 3.2. EUS with FNA confirmed pancreatic ductal adenocarcinoma, moderately differentiated. CA 19-9 = 420. Staging shows no evidence of metastatic disease; tumor abuts the SMV over < 180° without contour deformity and no arterial contact — resectable. ECOG 1. Comorbidities include T2DM (A1C 7.4) and HTN. No personal or family history of pancreatic cancer. Presented at multidisciplinary pancreas tumor board; consensus recommendation is neoadjuvant FOLFIRINOX followed by pancreaticoduodenectomy. Risks, benefits, and alternatives of Whipple discussed in detail; patient agrees to proceed with neoadjuvant therapy."
"Mrs. [Name] is a 57-year-old female with 4 months of left-sided abdominal fullness and early satiety. CT abdomen/pelvis demonstrates a 22 cm heterogeneous retroperitoneal mass encasing the left kidney, displacing the left colon, and abutting the psoas. MRI characterizes the mass as predominantly fatty with nodular enhancing components consistent with dedifferentiated liposarcoma. Core biopsy along the planned surgical incision confirms dedifferentiated liposarcoma, FNCLCC grade 2. No pulmonary metastases on CT chest. ECOG 1. She is otherwise healthy. Case discussed at sarcoma tumor board; plan is compartmental en bloc resection including left nephrectomy and left hemicolectomy, with potential adjuvant radiation. Detailed consent obtained covering multivisceral resection, blood transfusion, bowel anastomosis or ostomy, nephrectomy, and recurrence risk per MSKCC nomogram."
"Mr. [Name] is a 49-year-old male referred for evaluation of peritoneal carcinomatosis from a low-grade appendiceal mucinous neoplasm. He initially underwent appendectomy at an outside hospital 6 months ago for presumed appendicitis; pathology revealed low-grade appendiceal mucinous neoplasm with acellular mucin on the peritoneum. Follow-up CT demonstrates scalloping of the liver, omental caking, and right lower quadrant mucinous deposits. CEA 18, CA 19-9 45, CA 125 normal. ECOG 0. Diagnostic laparoscopy estimated PCI approximately 16 with disease predominantly in the right upper quadrant and pelvis; small bowel appears spared. Plan: cytoreductive surgery with peritonectomy, likely right hemicolectomy, omentectomy, and HIPEC with mitomycin C. Extensive preoperative counseling performed regarding expected ICU stay, prolonged recovery, and Clavien-Dindo III–IV complication rates."
"Mr. [Name] is a 70-year-old male post-op day 14 from classic pancreaticoduodenectomy for a 2.5 cm pancreatic head adenocarcinoma. Final pathology: moderately differentiated PDAC, 2.4 cm, pT2 pN1 (2/22 nodes positive), R0 resection (closest margin 3 mm at the SMA), LVI positive, PNI positive. Post-op course complicated by ISGPF grade B pancreatic fistula managed with maintenance of operative drain and percutaneous drainage of a 4 cm peri-anastomotic collection on POD 9. He is tolerating a regular diet, ambulating independently, with stable drain output of 30 mL/day. ECOG 1. Wound healing well without erythema or drainage. Plan: continue drain, transition off pancreatic enzyme replacement as tolerated, initiate adjuvant mFOLFIRINOX at 6–8 weeks post-op pending full recovery, and enrollment in surveillance CT imaging every 3 months with CA 19-9."
Surgical oncology is the most multidisciplinary surgical specialty. The best scribes understand that a surgical oncology note is not just a surgical note — it must capture histology, receptors, stage, genetics, prior treatments, and multidisciplinary plans with equal precision. When the attending says "pT2 pN1 M0 HR+ HER2-negative grade 2 IDC status post BCS with Z11-sparing of ALND, Oncotype 18, adjuvant AI planned," a great scribe writes exactly that.
Learn to read pathology reports and imaging reports before clinic starts. Know the staging rules for the diseases your attending operates on. Keep a running list of the patient's tumor board discussions and trial eligibility. Memorize tumor marker units and normal ranges. Anticipate which drugs come up for which cancer — FOLFIRINOX for pancreas, FLOT for gastric, CROSS for esophagus, nivo/ipi for melanoma — because you will hear them daily.
Welcome to surgical oncology. The learning curve is steep, the documentation is dense, and the work is among the most meaningful in medicine.
24 References & Sources
Clinical Practice Guidelines
NCCN Cutaneous Melanoma Guideline.
NCCN Soft Tissue Sarcoma Guideline.
NCCN Gastric Cancer Guideline.
NCCN Pancreatic Adenocarcinoma Guideline.
NCCN Hepatobiliary Cancers Guideline.
NCCN Thyroid Carcinoma Guideline.
ESMO Clinical Practice Guidelines. European Society for Medical Oncology.
ASCO Clinical Practice Guidelines. American Society of Clinical Oncology.
USPSTF Cancer Screening Recommendations. U.S. Preventive Services Task Force.
Landmark Clinical Trials
Diagram & Figure Sources
Figure 1: Breast Cancer Anatomy. U.S. National Cancer Institute via Wikimedia Commons. Public domain.
Figure 2: Lymph Node Architecture. Wikimedia Commons. Public domain.
Figure 3: Pancreatic Anatomy. Cancer Research UK via Wikimedia Commons. CC BY-SA 4.0.
Figure 4: Cutaneous Melanoma. U.S. National Cancer Institute via Wikimedia Commons. Public domain.
Figure 5: Pancreatic Cancer Staging Diagram. Cancer Research UK via Wikimedia Commons. CC BY-SA 4.0.
Figure 6: Whipple Procedure. U.S. National Cancer Institute via Wikimedia Commons. Public domain.