Transplant Surgery
Every organ, immunosuppressant, rejection pattern, opportunistic infection, procedure, classification, and documentation framework you need to succeed on day one on the transplant service.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 Transplant Immunology — Foundations
Transplant surgery exists at the intersection of technical surgery and applied immunology. Every operation is only half the story — the other half is preventing the recipient's immune system from destroying the graft. As a scribe, you cannot document rejection, drug changes, or infection without understanding the basic immunology that drives every clinical decision on the service.
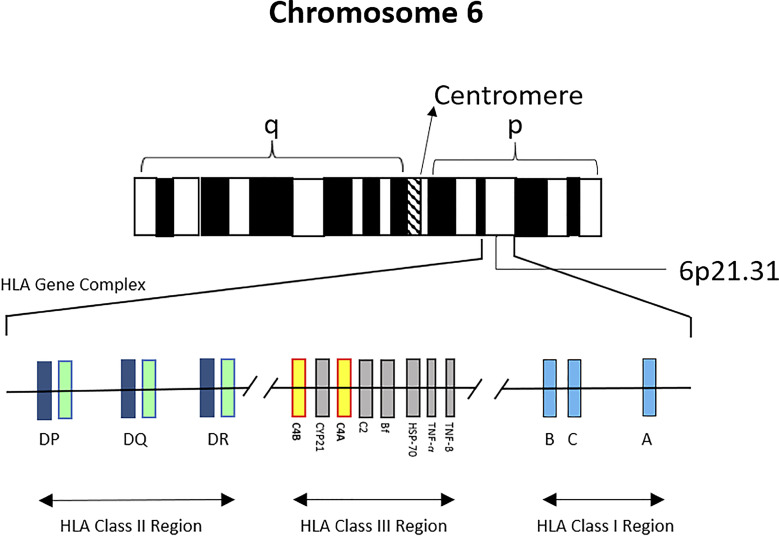
The HLA System
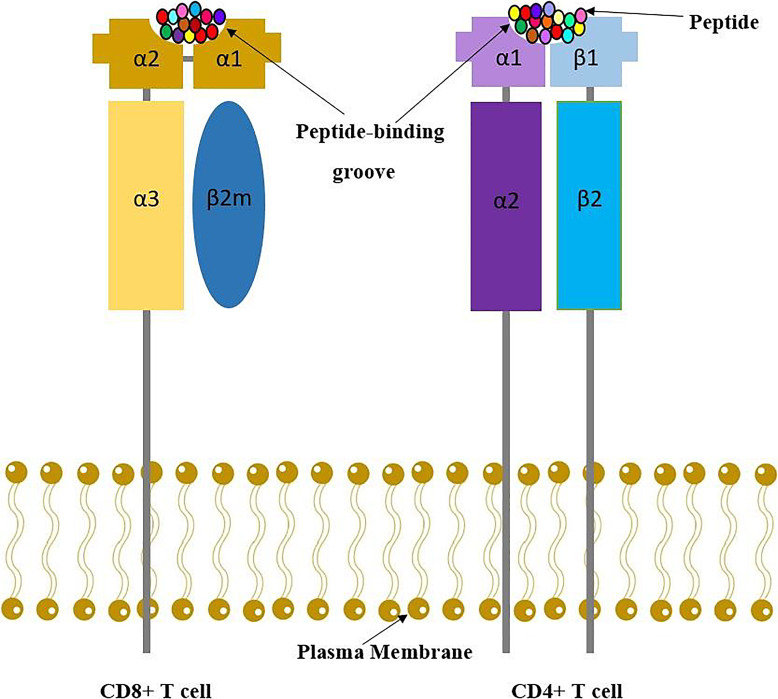
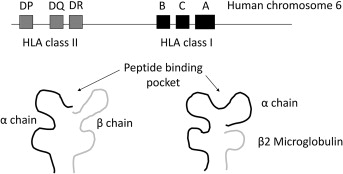
The Human Leukocyte Antigen (HLA) system is the human version of the Major Histocompatibility Complex (MHC) and is encoded on chromosome 6. HLA molecules present peptides to T cells and are the primary target of graft rejection. Class I molecules (HLA-A, HLA-B, HLA-C) are expressed on almost all nucleated cells and present to CD8+ cytotoxic T cells. Class II molecules (HLA-DR, HLA-DQ, HLA-DP) are expressed on antigen-presenting cells (dendritic cells, B cells, macrophages) and present to CD4+ helper T cells.
In deceased donor kidney allocation, compatibility is scored across six loci: two A, two B, and two DR alleles — a "zero-antigen mismatch" (0 ABDR MM) is the best possible match and earns priority points. For non-renal organs, HLA matching is generally not used for allocation (time and organ ischemia matter more), but a pre-existing donor-specific antibody still matters for every organ.
ABO Blood Group
ABO compatibility is the first and most rigid biological barrier. Natural anti-A and anti-B antibodies cause hyperacute rejection within minutes if violated. Standard rules mirror transfusion: O donors are universal, AB recipients are universal. ABO-incompatible (ABOi) transplantation is possible in kidney and liver transplantation using desensitization protocols (plasmapheresis, IVIG, rituximab) to reduce isoagglutinin titers below a critical threshold (typically 1:8 or 1:16).
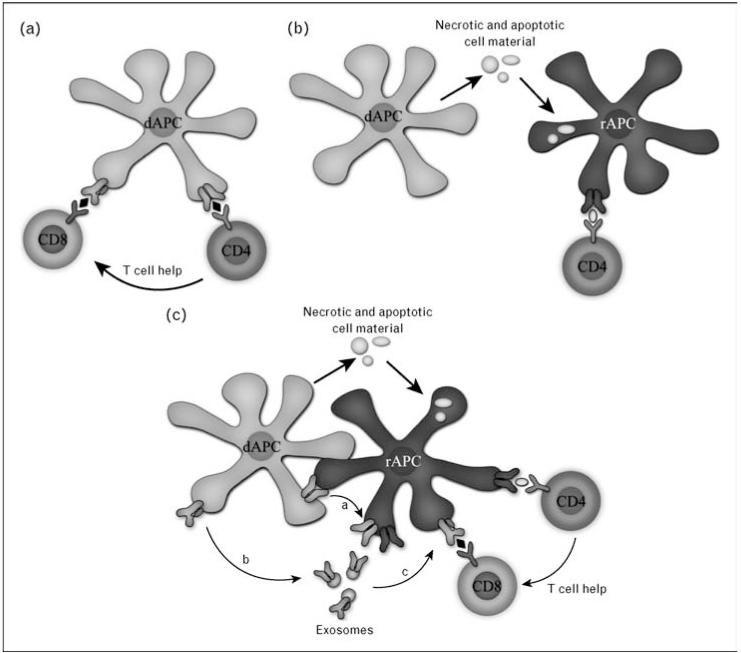
Alloantigen Recognition Pathways
T cells recognize donor antigen through three pathways. The direct pathway uses donor APCs presenting donor peptides directly to recipient T cells and dominates early acute cellular rejection. The indirect pathway uses recipient APCs processing and presenting donor peptides — this pathway dominates chronic rejection and drives donor-specific antibody (DSA) formation. The semi-direct pathway involves membrane transfer of intact donor MHC onto recipient APCs.
Key Immunologic Tests
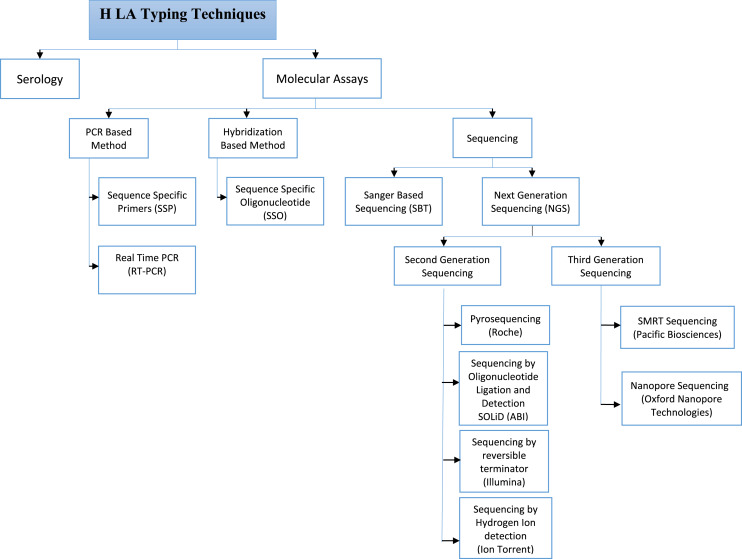
HLA typing: High-resolution molecular typing of recipient (and donor) at all major HLA loci.
Panel Reactive Antibody (PRA) / Calculated PRA (cPRA): Percentage of the donor pool to which the recipient has preformed antibodies. A cPRA of 99% means the recipient will react to 99 of every 100 donors.
Solid phase antibody testing (Luminex single antigen bead): Identifies specific HLA antibodies and assigns a Mean Fluorescence Intensity (MFI) value.
Crossmatch: Mixes recipient serum with donor lymphocytes. Complement-dependent cytotoxicity (CDC) crossmatch and flow cytometric crossmatch (FXM) must be negative before transplant. A positive T-cell CDC crossmatch is an absolute contraindication.
Donor-specific antibodies (DSA): HLA antibodies directed against the specific donor's HLA; the single most important biomarker for antibody-mediated rejection.
You will constantly hear MFI values, cPRA percentages, and "DSA positive/negative" during clinic. Write them down verbatim — fellows will ask you to read them back. "Class I DSA to A2 MFI 4200" is exactly how it should appear in the note.
02 Scribe Documentation Framework
Transplant notes are among the most complex in medicine because they must simultaneously track the graft, the immunosuppression regimen, infection prophylaxis, opportunistic surveillance, and the patient's underlying comorbidities. Most transplant centers use structured note templates, but you still need to understand what belongs where.
The Transplant HPI
The HPI for a transplant follow-up is never just "patient is doing well." It must document, every visit:
- Organ transplanted & date ("Status post deceased donor kidney transplant 05/12/2023, now POD #318")
- Donor type (DDKT vs LDKT; DCD vs DBD; living related vs living unrelated)
- Induction agent used (ATG, basiliximab, alemtuzumab)
- Current maintenance regimen (tacrolimus/MMF/prednisone is the classic triple therapy)
- Most recent trough level (tacrolimus, cyclosporine, sirolimus — the "window" is narrow)
- Graft function markers (creatinine for kidney; LFTs for liver; troponin/echo for heart; spirometry for lung)
- Rejection history (episodes, dates, Banff/ISHLT grades, treatment)
- Infection history (CMV, BK, EBV status of donor and recipient, any viremia episodes)
- Prophylaxis status (valganciclovir, TMP-SMX, fluconazole start/stop dates)
1. Graft function: Trend creatinine/LFT/troponin. Compare to baseline. Stable? Declining? Reason?
2. Immunosuppression: Current doses, most recent trough, target range, any adjustments.
3. Rejection surveillance: DSA screen result, protocol biopsy due?
4. Infection surveillance: CMV PCR, BK PCR, EBV PCR as indicated. Prophylaxis on or off?
5. Comorbidities: Hypertension, diabetes (new-onset diabetes after transplant — NODAT), dyslipidemia, bone health, skin cancer surveillance.
6. Vaccination & health maintenance: No live vaccines ever post-transplant.
Never write "labs reviewed" without the numbers. A transplant note lives or dies on the creatinine trend, the tacrolimus trough, and the BK/CMV viral load. Pull them into the A/P every visit.
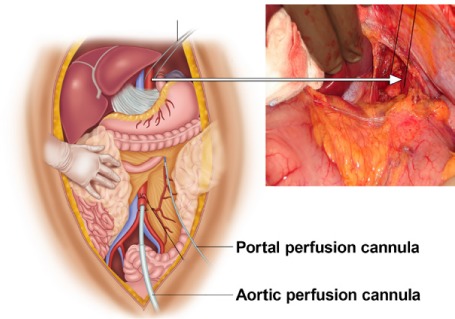
03 Donor Evaluation & Organ Procurement
Deceased Donor Categories
Donation after Brain Death (DBD) donors have been declared neurologically dead but have an intact cardiac circulation supported by the ventilator until organ retrieval. This is the historically standard donor and still yields the best outcomes for thoracic organs. Donation after Circulatory Death (DCD) donors have a non-survivable injury but do not meet brain death criteria; after withdrawal of life support in the OR, death is declared after a mandatory "no-touch" observation period (typically 2 or 5 minutes), and procurement begins. DCD donors yield kidneys, livers, lungs, and increasingly hearts (using either normothermic regional perfusion or direct procurement with ex-situ machine perfusion). OPTN DCD policy governs the protocol.
Living Donor Evaluation
Living donation is the standard of care for kidneys and a growing option for liver (right or left lobe hepatectomy). Evaluation includes a comprehensive medical workup (cardiac, pulmonary, renal function with iothalamate GFR or DTPA, cancer screening), psychosocial assessment by an independent donor advocate, ABO and crossmatch testing, and detailed cross-sectional imaging (CT angiogram for kidney, CT/MRI volumetry for liver).
Kidney Paired Donation (KPD)
When a recipient has a willing living donor who is ABO or HLA incompatible, KPD exchanges donors between multiple pairs so each recipient receives a compatible kidney. Chains (using a non-directed altruistic donor) can extend across dozens of pairs nationally through the UNOS KPD program or the National Kidney Registry.
Donor Management in the ICU
Once a donor is declared brain-dead and consent is obtained, the donor is managed aggressively to preserve organ function until procurement. Goals: mean arterial pressure >65, CVP 4-10, urine output 1-3 mL/kg/hr, PaO2/FiO2 ratio >300, normoglycemia, and correction of diabetes insipidus with DDAVP or vasopressin.
04 Brain Death & DCD Determination
Brain death determination is a medical and legal prerequisite for DBD organ donation. The 2023 update to the AAN consensus practice guideline on brain death/death by neurologic criteria provides the current standard.
Prerequisites: Known catastrophic irreversible brain injury; exclusion of confounders (hypothermia <36°C, severe metabolic/endocrine disturbance, drug intoxication, neuromuscular blockade).
Clinical exam: Coma with no response to noxious stimulation; absent brainstem reflexes (pupillary, corneal, oculocephalic, oculovestibular/cold calorics, gag, cough); no spontaneous respirations.
Apnea test: Pre-oxygenation to 100%, disconnect from ventilator, observe for respiratory effort for 8-10 minutes while monitoring ABG. Positive test = no respiratory effort and PaCO2 rise to ≥60 or 20 mmHg above baseline.
Ancillary testing (if apnea test cannot be completed or exam confounded): EEG, cerebral angiography, nuclear perfusion scan (HMPAO SPECT), or transcranial Doppler.
DCD "No-Touch" Interval
For DCD, after withdrawal of life-sustaining treatment and cardiac arrest, a mandatory observation period (typically 2 to 5 minutes depending on institutional policy and state law) must elapse with no spontaneous return of circulation before death can be declared and procurement begun. Warm ischemia time begins at the onset of hypotension (functional warm ischemia) and directly impacts organ viability.
05 Allocation Systems (OPTN/UNOS)
In the United States, the Organ Procurement and Transplantation Network (OPTN), operated under contract by the United Network for Organ Sharing (UNOS), governs all allocation. Each organ has its own scoring system.
Kidney: KDPI and EPTS
The Kidney Donor Profile Index (KDPI) assigns each deceased donor kidney a percentile from 0% (best) to 100% (worst) based on 10 donor characteristics (age, height, weight, ethnicity, history of hypertension, history of diabetes, cause of death, creatinine, HCV status, DCD status). The Estimated Post-Transplant Survival (EPTS) score is calculated for candidates based on age, time on dialysis, prior transplant, and diabetes status. Top 20% EPTS recipients are preferentially allocated KDPI <20% kidneys (longevity matching).
Liver: MELD-3.0
MELD (Model for End-Stage Liver Disease) predicts 3-month mortality on the waitlist. The current MELD-3.0 formula includes bilirubin, INR, creatinine, sodium, albumin, and sex (a correction for the historical disadvantage to female candidates). Score range 6-40. Higher = sicker = higher priority. Status 1A is reserved for acute fulminant liver failure with expected survival <7 days. HCC exception points are awarded to candidates meeting Milan criteria.
Heart: 6-Tier Status
Revised in 2018, heart allocation uses 6 status tiers. Status 1 is the highest (VA ECMO, non-dischargeable BiVAD); Status 6 is the lowest (stable outpatient listed for transplant). See the full enumeration in Section 19.
Lung: Lung Allocation Score (LAS) / CAS
The Lung Allocation Score (LAS) was introduced in 2005 and replaced in 2023 with the Composite Allocation Score (CAS), which balances medical urgency, post-transplant survival, biological disadvantages (height, blood type, sensitization), patient access, and placement efficiency.
Pancreas
Pancreas allocation follows ABO, waiting time, CPRA, and distance, with separate lists for pancreas alone (PTA), pancreas after kidney (PAK), and simultaneous pancreas-kidney (SPK).
06 Kidney Transplantation
Indications & Recipient Evaluation
Kidney transplantation is the treatment of choice for end-stage renal disease (ESRD). Common etiologies include diabetic nephropathy (the single most common cause in the US), hypertensive nephrosclerosis, glomerulonephritis (FSGS, IgA nephropathy, membranous, lupus), polycystic kidney disease (ADPKD), and reflux/obstructive uropathy. Preemptive transplantation (before dialysis initiation) yields the best outcomes. KDIGO Clinical Practice Guideline on the Evaluation and Management of Candidates for Kidney Transplantation is the standard reference.
Donor Options
- Living Donor Kidney Transplant (LDKT): Best outcomes. Laparoscopic or robotic donor nephrectomy. Graft half-life >15 years.
- Deceased Donor Kidney Transplant (DDKT) — DBD: Standard donor source.
- DDKT — DCD: Higher rate of delayed graft function but equivalent long-term outcomes.
- ABO-incompatible (ABOi): Desensitization protocol with plasmapheresis, IVIG, and rituximab.
- HLA-incompatible (positive crossmatch): Similar desensitization; worse outcomes than compatible transplant.
- Kidney Paired Donation (KPD): Donor exchange across incompatible pairs.
Surgical Technique
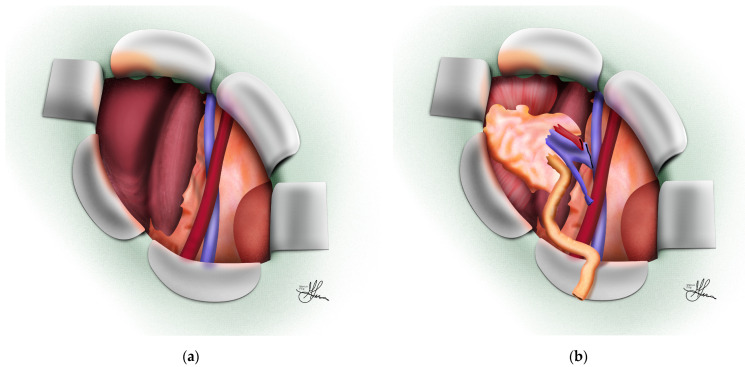
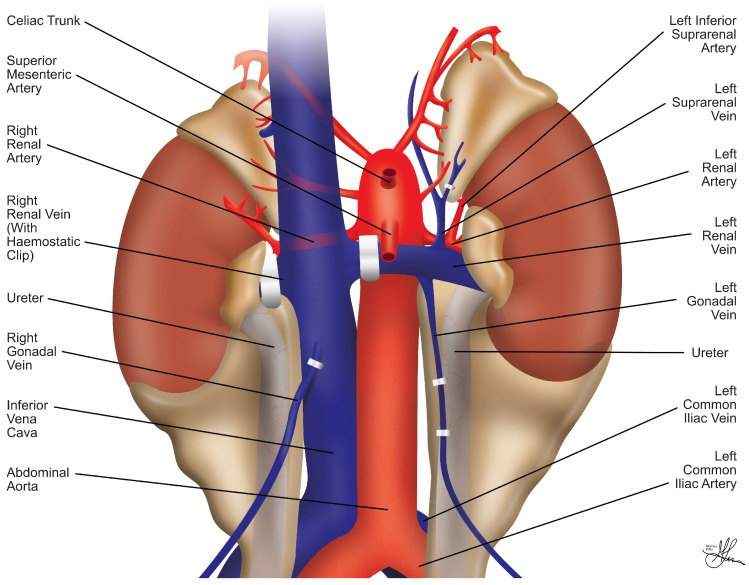
The allograft is placed extraperitoneally in the iliac fossa (right iliac fossa is most common). The renal artery is anastomosed end-to-side to the external iliac artery, the renal vein end-to-side to the external iliac vein, and the ureter is implanted into the bladder using either the Lich-Gregoir extravesical technique (most common) or the Politano-Leadbetter intravesical technique. A ureteral stent is typically left in place and removed at 4-6 weeks.
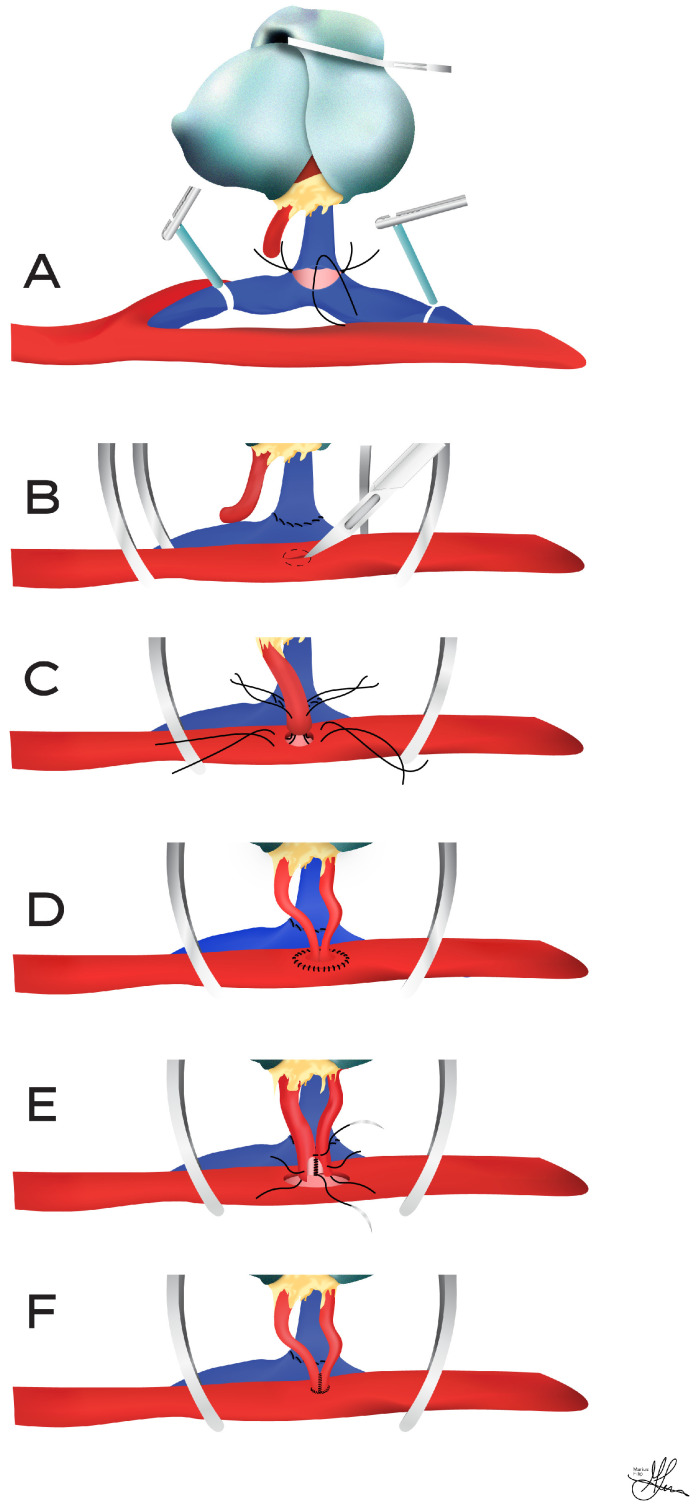
Post-Operative Course
Expected immediate graft function: brisk urine output and a rapid fall in creatinine. Delayed graft function (DGF), defined as the need for dialysis in the first week post-transplant, occurs in 20-40% of DCD and marginal DBD kidneys. Primary nonfunction (PNF) is the failure of the graft to ever function and is rare.
Anuria/oliguria: Work up with Doppler ultrasound immediately — rule out vascular thrombosis (renal artery or vein), urinary obstruction, and urine leak.
Sudden rise in creatinine: Consider acute rejection, CNI toxicity, BK nephropathy, or obstruction.
Lymphocele: Presents as fluid collection around graft, can cause ureteral obstruction or leg edema.
07 Liver Transplantation
Indications
The most common indications for liver transplant in adults are non-alcoholic steatohepatitis (NASH, now MASH), alcohol-associated liver disease (ALD), chronic hepatitis C (declining dramatically in the DAA era), hepatocellular carcinoma (HCC) within Milan criteria, and acute liver failure. Other indications include primary biliary cholangitis (PBC), primary sclerosing cholangitis (PSC), autoimmune hepatitis, Wilson's disease, hereditary hemochromatosis, alpha-1 antitrypsin deficiency, and pediatric biliary atresia.
HCC exception points require meeting Milan criteria: single lesion ≤5 cm, or up to 3 lesions each ≤3 cm, no vascular invasion, no extrahepatic spread. The AASLD Guidelines for Liver Transplantation summarize contemporary management.
Graft Types
- Whole liver (deceased donor): Standard.
- Split liver: One donor liver divided — typically left lateral segment (segments 2-3) for a pediatric recipient and extended right (segments 1, 4-8) for an adult.
- Living donor liver transplant (LDLT): Right lobe (segments 5-8) for adult recipient; left lateral segment for pediatric recipient.
- DCD liver: Sensitive to warm ischemia; higher rate of ischemic cholangiopathy.
- Normothermic machine perfusion (NMP) & hypothermic oxygenated perfusion (HOPE): Ex-situ machine perfusion techniques that allow organ viability assessment and reduce ischemia-reperfusion injury. OrganOx NMP randomized trial.
Surgical Technique
Two techniques dominate: the classical (bicaval) technique, in which the recipient IVC is resected en bloc with the native liver and the donor suprahepatic and infrahepatic cava are anastomosed end-to-end, and the piggyback technique, in which the recipient IVC is preserved and the donor suprahepatic cava is anastomosed to a common orifice formed by the recipient hepatic veins. The portal vein is reconstructed next, followed by the hepatic artery, and finally the biliary anastomosis (duct-to-duct in most adults, Roux-en-Y hepaticojejunostomy if the recipient duct is diseased — as in PSC).
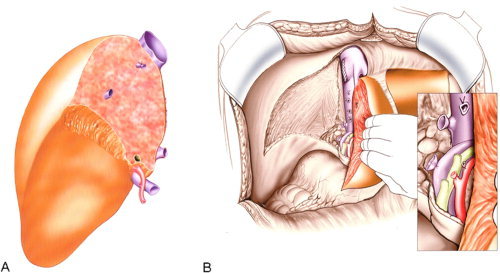
Complications Unique to Liver Transplant
- Hepatic artery thrombosis (HAT): Catastrophic early; mandates urgent re-exploration or re-transplant. Late HAT causes ischemic cholangiopathy.
- Portal vein thrombosis (PVT): Rare but serious.
- Biliary leak: At anastomosis or T-tube site.
- Biliary stricture: Anastomotic or non-anastomotic (ischemic type, DCD-associated).
- Primary nonfunction: Failure of the graft to ever work — Status 1A re-listing.
- Small-for-size syndrome: In LDLT when graft-to-recipient weight ratio is too low.
- Recurrent disease: HCV (with DAA treatment now), autoimmune, PBC, PSC, NASH, HCC.
08 Pancreas & Islet Transplantation
Pancreas transplantation cures type 1 diabetes (and highly selected insulin-dependent type 2) by restoring endogenous insulin secretion. Three configurations:
- Simultaneous Pancreas-Kidney (SPK): The most common configuration, for type 1 diabetics with ESRD.
- Pancreas After Kidney (PAK): A pancreas transplant in a patient who already has a functioning kidney allograft.
- Pancreas Transplant Alone (PTA): For type 1 diabetics with preserved renal function but severe hypoglycemia unawareness or brittle diabetes.
Surgical Technique
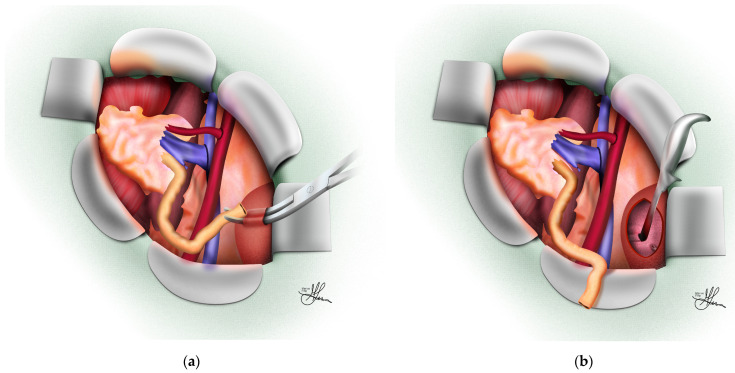
The pancreas is transplanted with a segment of donor duodenum. Arterial inflow is typically via a donor iliac artery Y-graft connecting the superior mesenteric artery and splenic artery to the recipient common iliac. Venous outflow can be systemic (to the iliac vein) or portal (to the superior mesenteric vein). Exocrine drainage is either enteric (to recipient jejunum, standard) or bladder (historical; allows monitoring of urinary amylase but causes metabolic acidosis and cystitis).
Islet Cell Transplantation
Islets are isolated from a donor pancreas and infused into the recipient's portal vein, where they engraft in the liver sinusoids. Indicated for severe hypoglycemia unawareness. The CIT-07 trial demonstrated durable freedom from severe hypoglycemic events. FDA approval of donislecel (Lantidra) came in 2023.
09 Heart Transplantation
Indications
End-stage heart failure refractory to maximal medical and device therapy, with expected one-year survival without transplant <50%. Etiologies: ischemic cardiomyopathy, non-ischemic dilated cardiomyopathy, hypertrophic cardiomyopathy, restrictive cardiomyopathy, congenital heart disease (adult congenital), and amyloid cardiomyopathy. The ISHLT listing criteria for heart transplantation govern candidacy.
Bridging Strategies
- Durable LVAD (HeartMate 3): Centrifugal continuous-flow pump; most common bridge.
- Temporary MCS: Impella 5.5, TandemHeart, Centrimag.
- VA ECMO: For cardiogenic shock; Status 1 listing.
- IABP: Counterpulsation bridge.
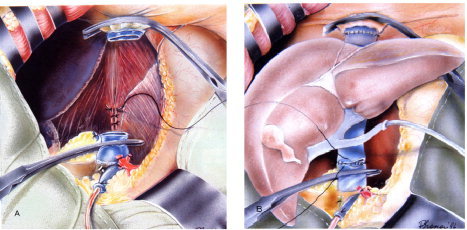
Surgical Technique
Two techniques: the bicaval technique (standard; separate SVC and IVC anastomoses preserve right atrial geometry and sinus node function) and the historical biatrial (Lower-Shumway) technique. DCD heart transplantation has been enabled by normothermic regional perfusion (NRP) and ex-situ normothermic machine perfusion (TransMedics OCS Heart).
Post-Transplant Surveillance
Endomyocardial biopsy (EMB) via internal jugular approach is the gold standard for rejection surveillance. Schedule: weekly for first month, tapering through the first year. Non-invasive alternatives: gene expression profiling (AlloMap) and donor-derived cell-free DNA (dd-cfDNA). Cardiac allograft vasculopathy (CAV) is the leading cause of late graft loss and is surveilled with annual coronary angiography and IVUS.
10 Lung Transplantation
Indications
End-stage lung disease with expected survival <2 years without transplant. Common indications: idiopathic pulmonary fibrosis (IPF) — now the single most common indication, COPD/emphysema, cystic fibrosis (CF), pulmonary arterial hypertension (PAH), alpha-1 antitrypsin deficiency, sarcoidosis, and connective tissue disease-associated ILD. ISHLT consensus document for lung transplant candidate selection.
Single vs Bilateral
Bilateral sequential lung transplant (BLT) is the dominant procedure. Single lung transplant (SLT) is reserved for selected older patients with non-septic lung disease (IPF, emphysema) and offers shorter wait times. CF and septic lung disease (bronchiectasis) require bilateral transplant. PAH historically required heart-lung but BLT is now standard with right ventricular recovery.
Surgical Technique
Clamshell or bilateral anterior thoracotomy ("clamshell incision") for BLT; posterolateral thoracotomy for SLT. Three anastomoses per lung: bronchus (telescoped or end-to-end), pulmonary artery, and left atrial cuff (containing the pulmonary veins). Intraoperative support ranges from off-pump to VA ECMO depending on hemodynamics and oxygenation.
Primary Graft Dysfunction (PGD)
PGD is the lung equivalent of reperfusion injury and occurs in up to 30% of lung transplants. Graded 0-3 by PaO2/FiO2 ratio and chest X-ray findings within 72 hours of reperfusion. PGD grade 3 is a leading cause of early mortality.
Chronic Lung Allograft Dysfunction (CLAD)
CLAD is the late graft failure pattern unique to lung transplant, manifesting as bronchiolitis obliterans syndrome (BOS) or restrictive allograft syndrome (RAS). Surveillance: spirometry (FEV1 is the key number), chest CT, and transbronchial biopsy for rejection surveillance.
11 Intestinal, Multi-Visceral & VCA
Intestinal Transplantation
Indicated for irreversible intestinal failure with life-threatening TPN complications (liver failure, loss of central venous access, recurrent line sepsis, severe dehydration). Three configurations: isolated intestine, combined liver-intestine, and multi-visceral (stomach, duodenum, pancreas, intestine ± liver). The most immunogenic of all solid organ transplants, requiring the highest immunosuppression.
Vascularized Composite Allotransplantation (VCA)
VCA encompasses transplantation of tissues containing skin, muscle, bone, nerve, and vessels as a single functional unit. Examples include hand/upper extremity transplant, face transplant, and (rarely) uterine transplant. Skin is the most immunogenic tissue in the body, making VCA prone to rejection despite aggressive immunosuppression.
12 HSCT / Bone Marrow Transplant Overview
Hematopoietic stem cell transplant (HSCT) is not solid organ transplant but scribes on inpatient consult services will encounter these patients. HSCT replaces the recipient's hematopoietic system with donor stem cells, typically for hematologic malignancies (AML, ALL, MDS, lymphoma, multiple myeloma), bone marrow failure syndromes, and inherited immunodeficiencies.
Autologous: Patient's own stem cells (for myeloma, lymphoma). No rejection, no GVHD.
Allogeneic — matched sibling donor (MSD): Best outcomes.
Allogeneic — matched unrelated donor (MUD): Via NMDP registry.
Haploidentical: Half-matched family donor with post-transplant cyclophosphamide.
Umbilical cord blood: Banked cord units, tolerant of HLA mismatch.
Graft-versus-Host Disease (GVHD) is the signature complication: donor T cells attack recipient tissues. Acute GVHD targets skin (rash), gut (diarrhea), and liver (cholestatic LFTs). Chronic GVHD mimics autoimmune disease across multiple organs. Prophylaxis: tacrolimus or cyclosporine plus methotrexate, or post-transplant cyclophosphamide for haplo.
13 Transplant Procedures — A to Z
| Procedure | Description | Key Documentation |
|---|---|---|
| Deceased donor multi-organ procurement | Simultaneous retrieval of thoracic and abdominal organs from DBD/DCD donor. | WIT, CIT, procurement anatomy, anomalies. |
| Laparoscopic donor nephrectomy | Hand-assisted or pure lap removal of a living donor kidney (typically left for longer vein). | Number of renal arteries, vein length, warm ischemia time. |
| Robotic donor nephrectomy | Robotic-assisted variant. | Console time, conversion rate. |
| Living donor hepatectomy | Right lobe (adult recipient) or left lateral (pediatric recipient) donation. | Graft volume, GRWR, remnant volume, biliary anatomy. |
| Kidney transplant | Extraperitoneal iliac fossa placement with vascular and ureteral anastomosis. | CIT, WIT, reperfusion time, urine output in OR. |
| Orthotopic liver transplant | Bicaval or piggyback technique with portal, arterial, and biliary reconstruction. | CIT, reperfusion syndrome, transfusion volume, biliary reconstruction type. |
| Pancreas transplant | Intra-abdominal placement with Y-graft inflow, enteric or bladder drainage. | Drainage type, venous drainage (systemic vs portal). |
| Orthotopic heart transplant | Bicaval technique; CPB with cardioplegic arrest. | Ischemia time, inotrope requirement, post-bypass echo. |
| Bilateral sequential lung transplant | Clamshell incision; sequential implantation of each lung. | Ischemia time per side, ECMO use, PGD grade. |
| Back-table preparation | Bench work on the graft — vessel reconstruction, trimming, arterial Y-graft. | Number of vessels reconstructed, technique. |
| Normothermic machine perfusion (NMP) | Ex-situ perfusion with oxygenated blood at 37°C for viability assessment. | Perfusion time, lactate clearance, bile production (liver). |
| Hypothermic machine perfusion (HMP) | Pulsatile cold perfusion for kidney preservation. | Flow, resistance, pump time. |
| Kidney allograft biopsy | Percutaneous ultrasound-guided core biopsy. | Indication (protocol vs for cause), number of cores, complications. |
| Endomyocardial biopsy | Right IJ approach via bioptome to RV septum. | Number of samples, grade, complications. |
| Transbronchial lung biopsy | Via bronchoscope for CLAD and rejection surveillance. | Lobe sampled, pathology grade. |
| Plasmapheresis | Apheresis circuit via central line; removes circulating antibodies. | Number of plasma volumes exchanged, replacement fluid. |
| Ureteral stent removal | Cystoscopic removal at 4-6 weeks post kidney transplant. | Residual blood, symptoms. |
Cold ischemia time (CIT) and warm ischemia time (WIT) are sacred numbers. CIT = cross-clamp in donor to reperfusion in recipient. WIT = from cross-clamp or rewarming to reperfusion. Every transplant op note must have them.
14 Rejection — Diagnosis & Treatment
Rejection Categories by Timing & Mechanism
Hyperacute rejection: Minutes to hours. Preformed antibodies (ABO or DSA) cause complement activation and graft thrombosis. Now virtually eliminated by modern crossmatching.
Accelerated acute rejection: Days. Memory response in previously sensitized recipient.
Acute cellular rejection (ACR, T-cell mediated, TCMR): Days to months. T-cell infiltration of the graft. Responds to high-dose steroids and/or ATG.
Acute antibody-mediated rejection (AMR): Days to months. De novo DSA formation with peritubular capillaritis, C4d staining, and complement activation. Treated with plasmapheresis, IVIG, rituximab, ± bortezomib, ± eculizumab.
Chronic rejection: Months to years. Chronic active TCMR or chronic active AMR; drives transplant glomerulopathy (kidney), CAV (heart), CLAD (lung), and vanishing bile duct syndrome (liver).
Rejection Workup
Suspected rejection is confirmed by biopsy. Adjunct: Luminex DSA screen, C4d immunostaining (marker of complement activation in AMR), and donor-derived cell-free DNA (dd-cfDNA, noninvasive rejection screen). The biopsy is graded according to organ-specific classification systems: Banff for kidney, pancreas, liver; ISHLT for heart and lung. See Section 19 for full enumeration.
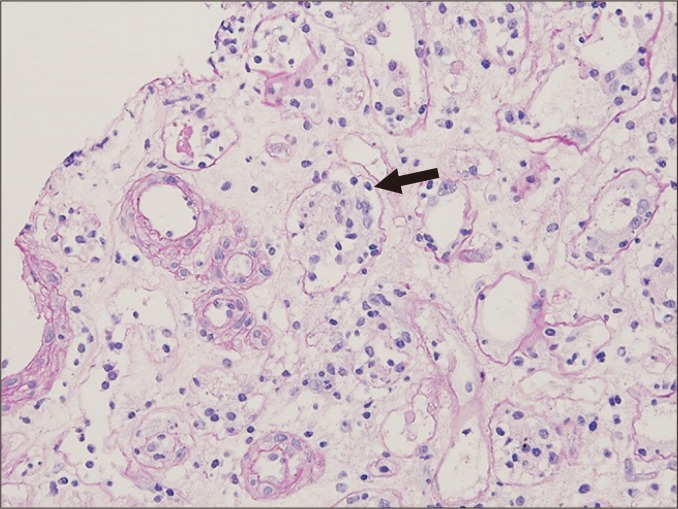
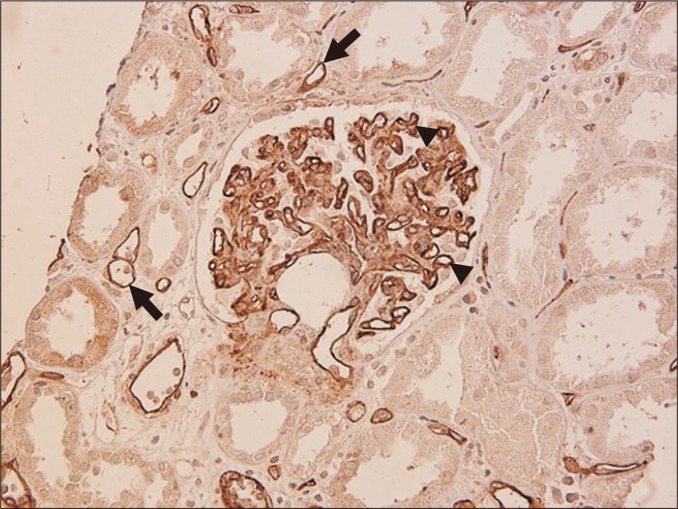
Treatment
| Rejection Type | First-Line Treatment | Second-Line / Adjunct |
|---|---|---|
| Mild ACR (Banff IA) | Methylprednisolone pulse 250-500 mg x 3 days | Optimize maintenance; switch CsA to tacrolimus. |
| Moderate-severe ACR (Banff IB-III) | ATG (thymoglobulin) 1.5 mg/kg x 5-10 days | Alemtuzumab; CMV and PJP prophylaxis reinforced. |
| AMR | Plasmapheresis + IVIG | Rituximab, bortezomib, eculizumab (for complement-active AMR). |
| Chronic active AMR | Optimize immunosuppression, ACEi/ARB | No proven therapy; clinical trials. |
| Cardiac rejection pAMR 2-3 | IV methylprednisolone + plasmapheresis + IVIG | Rituximab, bortezomib. |
15 Induction & Maintenance Immunosuppression
Induction Agents
Induction is a short course of intense immunosuppression at the time of transplant to blunt the early alloimmune response.
| Agent | Class | Dose | Key Notes |
|---|---|---|---|
| Basiliximab (Simulect) | Anti-CD25 mAb | 20 mg IV x 2 doses | Non-depleting; lowest-risk induction. |
| Antithymocyte globulin (ATG, Thymoglobulin) | Rabbit polyclonal anti-T-cell | 1.5 mg/kg/day x 3-7 days | T-cell depleting; cytokine release, requires premeds. |
| Alemtuzumab (Campath) | Anti-CD52 mAb | 30 mg IV x 1 | Profound lymphocyte depletion; used off-label. |
Maintenance Immunosuppression
Classic regimen: triple therapy — a calcineurin inhibitor (CNI), an antimetabolite, and prednisone.
| Drug | Class | Mechanism | Target/Monitoring | Key Side Effects |
|---|---|---|---|---|
| Tacrolimus (Prograf, Envarsus) | CNI | Blocks calcineurin → inhibits IL-2 | Trough 5-12 ng/mL (varies by organ and time) | Nephrotoxicity, tremor, hyperglycemia, HTN, hyperkalemia, alopecia. |
| Cyclosporine (Neoral, Sandimmune) | CNI | Blocks calcineurin | C0 or C2 level | Nephrotoxicity, HTN, hirsutism, gum hyperplasia. |
| Mycophenolate mofetil (MMF, CellCept) | Antimetabolite | Inhibits IMPDH → purine synthesis | Not routinely drug-level monitored | Diarrhea, leukopenia, teratogenic (REMS). |
| Mycophenolic acid (Myfortic) | Antimetabolite | Enteric-coated MPA | — | Better GI tolerance than MMF. |
| Azathioprine (Imuran) | Antimetabolite | Purine analog | TPMT testing | Myelosuppression, hepatitis. |
| Sirolimus (Rapamune) | mTOR inhibitor | Blocks mTOR signaling | Trough 5-15 ng/mL | Proteinuria, wound healing, pneumonitis, dyslipidemia. |
| Everolimus (Zortress) | mTOR inhibitor | mTOR blockade | Trough 3-8 ng/mL | Same class; shorter half-life. |
| Prednisone | Corticosteroid | Broad anti-inflammatory | Clinical | NODAT, osteoporosis, Cushingoid, cataract. |
| Belatacept (Nulojix) | Costim blocker | CTLA4-Ig blocks CD80/86 | Monthly infusion | EBV-seronegative patients excluded (PTLD risk). |
Increase tacrolimus/cyclosporine levels (CYP3A4 inhibitors): azoles (fluconazole, voriconazole, posaconazole), macrolides (erythromycin, clarithromycin), protease inhibitors, diltiazem, verapamil, grapefruit juice.
Decrease CNI levels (CYP3A4 inducers): rifampin, phenytoin, carbamazepine, phenobarbital, St. John's wort.
16 Opportunistic Infections & Prophylaxis
Infection timelines in the post-transplant patient follow a predictable pattern. Months 0-1: nosocomial and surgical site infections. Months 1-6: opportunistic infections (CMV, PJP, BK, fungi). Beyond 6 months: community-acquired and late opportunistic infections.
Key Opportunistic Pathogens
The single most important viral pathogen. Highest risk: donor positive / recipient negative (D+/R-). Presentations: viremia, syndrome (fever, cytopenias), tissue-invasive disease (colitis, pneumonitis, hepatitis, retinitis). Diagnosis: quantitative PCR. Treatment: IV ganciclovir or oral valganciclovir. Resistance (UL97, UL54 mutations) treated with foscarnet, cidofovir, letermovir, or maribavir. AST CMV guidelines.
Primary concern in kidney transplant. Causes BK viremia and BK nephropathy (BKVAN). Diagnosis: serum BK PCR, biopsy with SV40 staining. Treatment: reduce immunosuppression (first-line), IVIG, cidofovir (limited evidence), leflunomide.
EBV-seronegative recipient of EBV+ organ is highest risk. Presentations range from mononucleosis-like illness to aggressive B-cell lymphoma (post-transplant lymphoproliferative disorder, PTLD). Treatment: reduce immunosuppression, rituximab, chemotherapy.
Diffuse interstitial pneumonia with hypoxemia. Diagnosis: BAL with silver stain or PCR. Treatment: TMP-SMX plus steroids if PaO2 <70. Prophylaxis with TMP-SMX for 6-12 months prevents most cases.
Other Opportunists
- Candida: Mucosal and systemic; fluconazole prophylaxis.
- Aspergillus: Especially lung transplant. Voriconazole or isavuconazole.
- Cryptococcus: Meningitis, pneumonia.
- Toxoplasma gondii: Especially heart transplant (cyst reactivation). TMP-SMX covers this.
- Nocardia: Pulmonary and brain abscess. TMP-SMX.
- JC virus: PML (progressive multifocal leukoencephalopathy) — rare but devastating.
- Mycobacterium tuberculosis: Reactivation; isoniazid prophylaxis for LTBI pre-transplant.
Prophylaxis Regimens
| Prophylaxis | Target | Standard Regimen | Duration |
|---|---|---|---|
| Valganciclovir | CMV | 900 mg PO daily (dose-adjusted for CrCl) | 3-6 months (high-risk up to 12) |
| TMP-SMX | PJP, Toxo, Nocardia, UTI | SS daily or DS 3x/week | 6-12 months (lifelong in lung) |
| Fluconazole | Candida | 200-400 mg PO daily | 1-3 months |
| Voriconazole / Posaconazole / Itraconazole | Aspergillus | Varies by drug | Lung: 3-6 months |
| Isoniazid (+ B6) | LTBI | 300 mg PO daily | 9 months |
| Nystatin / Clotrimazole troches | Oral thrush | Swish and swallow | 1-3 months |
Always document the donor/recipient CMV and EBV serology pairings (D+/R-, D+/R+, D-/R+, D-/R-). These pairings drive prophylaxis duration and risk counseling.
17 Post-Transplant Malignancy & PTLD
Chronic immunosuppression raises the risk of de novo malignancy. The major categories:
- Non-melanoma skin cancer (SCC, BCC): The most common malignancy post-transplant, with a 65-fold increased risk of SCC. Annual dermatology exam and photoprotection counseling are standard.
- PTLD: A spectrum from polyclonal B-cell hyperplasia to aggressive DLBCL. EBV-driven in most early cases. Treatment: reduce IS, rituximab, R-CHOP.
- Kaposi sarcoma: HHV-8 driven.
- Anogenital cancers: HPV-associated.
- Solid-organ cancers: Elevated risk for lung, kidney, liver, thyroid.
Switching from CNI to an mTOR inhibitor (sirolimus or everolimus) may reduce skin cancer and Kaposi sarcoma risk given their anti-proliferative properties.
18 Surgical & Medical Complications
Vascular Complications
- Renal artery/vein thrombosis: Early graft loss; Doppler US for diagnosis.
- Hepatic artery thrombosis: Re-exploration or re-transplant.
- Transplant renal artery stenosis (TRAS): Uncontrolled HTN, elevated Cr; duplex US and angioplasty/stent.
Urologic Complications (Kidney)
- Ureteral leak: Fluid collection with high creatinine content; percutaneous drainage and stent/reoperation.
- Ureteral stricture: Usually at bladder anastomosis; stent or surgical revision.
- Lymphocele: Drainage via percutaneous or peritoneal window.
Biliary Complications (Liver)
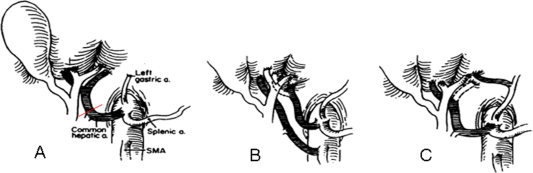
- Biliary leak: Typically at anastomosis or T-tube; ERCP stent placement.
- Anastomotic stricture: ERCP with balloon dilation and stent.
- Non-anastomotic (ischemic-type) stricture: Associated with DCD donors, long CIT.
Medical Complications
- New-onset diabetes after transplant (NODAT): Tacrolimus and steroids both contribute.
- Post-transplant hypertension: CNI-induced; ACEi/ARB or CCB.
- Dyslipidemia: Statins; interactions with CNI (avoid simvastatin with cyclosporine).
- CNI nephrotoxicity: Acute (vasoconstrictive) and chronic (interstitial fibrosis).
- Bone disease: Steroid-induced osteoporosis; DEXA surveillance.
- Recurrent disease: FSGS, IgA, HCV (DAA eradication), NASH, PBC, PSC, autoimmune hepatitis.
19 Classification Systems — Full Enumeration
Banff Kidney Allograft Rejection (abbreviated)
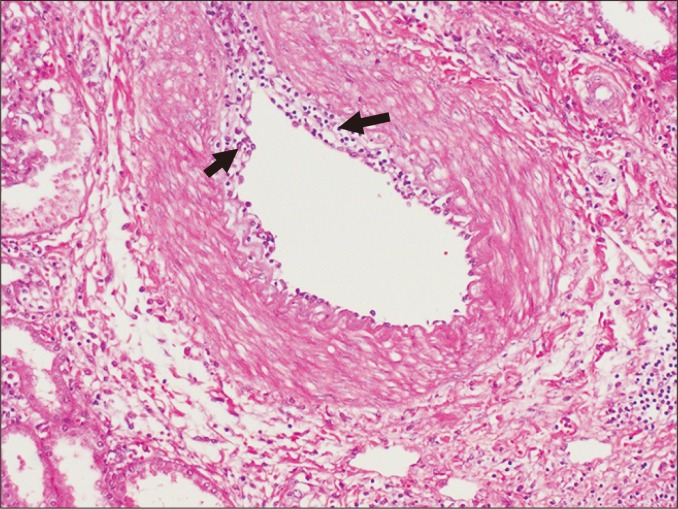
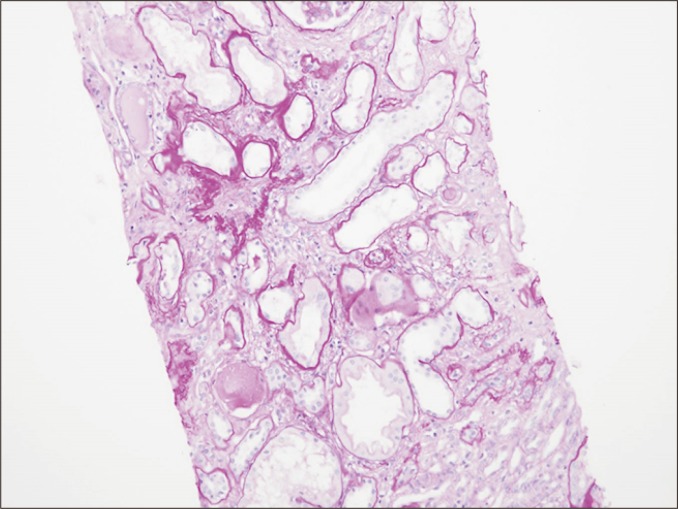
| Banff Category | Finding |
|---|---|
| Category 1 | Normal biopsy or nonspecific changes. |
| Category 2 | Antibody-mediated rejection (active or chronic active). |
| Category 3 | Borderline changes: suspicious for acute TCMR. |
| Category 4 — IA | TCMR: tubulitis t2 with interstitial inflammation i2-3. |
| Category 4 — IB | TCMR: tubulitis t3 with i2-3. |
| Category 4 — IIA | Mild-moderate intimal arteritis (v1). |
| Category 4 — IIB | Severe intimal arteritis (v2). |
| Category 4 — III | Transmural arteritis / fibrinoid necrosis (v3). |
| Category 5 | Interstitial fibrosis & tubular atrophy (IF/TA). |
| Category 6 | Other (BK, drug toxicity, recurrent disease). |
Banff 2019 Kidney Classification.
ISHLT Acute Cellular Rejection — Heart
| Grade | Finding |
|---|---|
| Grade 0R | No rejection. |
| Grade 1R (mild) | Interstitial and/or perivascular infiltrate with up to 1 focus of myocyte damage. |
| Grade 2R (moderate) | Two or more foci of infiltrate with associated myocyte damage. |
| Grade 3R (severe) | Diffuse infiltrate with multifocal myocyte damage ± edema, hemorrhage, vasculitis. |
ISHLT pAMR (Antibody-Mediated Rejection) — Heart
| Grade | Finding |
|---|---|
| pAMR 0 | Negative histology and immunopathology. |
| pAMR 1 (H+) | Histologic AMR alone (positive H, negative I). |
| pAMR 1 (I+) | Immunopathologic AMR alone (negative H, positive I). |
| pAMR 2 | Both histologic and immunopathologic AMR present. |
| pAMR 3 | Severe AMR with interstitial hemorrhage, capillary fragmentation, fibrinoid necrosis. |
MELD-3.0 Components
Score uses: bilirubin, INR, creatinine, sodium, albumin, sex, and interactions. Range 6-40; higher = sicker. Used for liver allocation.
Child-Pugh Score
| Parameter | 1 Point | 2 Points | 3 Points |
|---|---|---|---|
| Bilirubin (mg/dL) | <2 | 2-3 | >3 |
| Albumin (g/dL) | >3.5 | 2.8-3.5 | <2.8 |
| INR | <1.7 | 1.7-2.3 | >2.3 |
| Ascites | None | Mild | Moderate/severe |
| Encephalopathy | None | Grade 1-2 | Grade 3-4 |
Class A 5-6, Class B 7-9, Class C 10-15.
OPTN Heart Allocation Status (2018)
| Status | Criteria |
|---|---|
| Status 1 | VA ECMO, non-dischargeable BiVAD or TAH, MCS with life-threatening arrhythmia. |
| Status 2 | Non-dischargeable LVAD, IABP, percutaneous endovascular MCS, durable LVAD with device complication. |
| Status 3 | Dischargeable LVAD for 30 days of discretionary use, multiple inotropes, VA ECMO >7d, durable MCS complications. |
| Status 4 | Stable on durable LVAD, single inotrope, congenital heart disease, HCM, amyloid, ischemic HD with angina refractory to medical therapy, retransplant. |
| Status 5 | On multiple waitlists (combined organ recipients). |
| Status 6 | All other eligible candidates. |
KDPI / EPTS
KDPI: 0-100% from 10 donor variables; lower is better. EPTS: candidate score based on age, dialysis time, diabetes, prior transplant; lower = longer expected survival. Longevity matching pairs top 20% EPTS with KDPI <20%.
PGD Grading (Lung)
| Grade | PaO2/FiO2 | CXR Edema |
|---|---|---|
| 0 | >300 | No edema |
| 1 | >300 | Diffuse edema present |
| 2 | 200-300 | Diffuse edema present |
| 3 | <200 | Diffuse edema present |
20 Transplant Physical Exam
General
Document Cushingoid features (moon facies, buffalo hump, striae, central adiposity — common with chronic steroids), tremor (a CNI signature), gum hypertrophy and hirsutism (cyclosporine), acne and weight gain (steroids), and overall frailty.
Organ-Specific Exam
Right or left lower quadrant Gibson/Rutherford-Morrison incision (check for drainage, erythema, dehiscence). Palpate for graft size, tenderness, bruit over the graft (possible TRAS). Check for lymphocele (fluctuance). Auscultate native kidney bruits. Assess for peripheral edema.
Mercedes or "hockey stick" subcostal incision. Palpate liver edge (normally at the costal margin). Check for ascites, caput medusae (residual portal hypertension), scleral icterus, asterixis, RUQ tenderness. Auscultate bruits.
Median sternotomy. JVD, lung crackles, S3, peripheral edema. Listen for murmurs (tricuspid regurgitation from biopsy injury). Absent carotid sinus response (denervated heart). HR is typically 90-100 at rest due to denervation.
Clamshell (anterior thoracotomy) or bilateral incisions. Auscultate for equal breath sounds, crackles, wheezing, and bronchial breath sounds (anastomotic narrowing). Digital clubbing (if pre-transplant lung disease).
21 Abbreviations Master List
Organs & Procedures
Allocation & Immunology
Rejection & Pathology
Medications
Infections
22 Sample HPI Templates
58-year-old woman with ESRD secondary to type 2 diabetic nephropathy and hypertension, on in-center hemodialysis MWF via a left upper arm brachiocephalic AVF since 2022, presenting for initial kidney transplant evaluation. PMH notable for HTN, HLD, T2DM on basal-bolus insulin (last A1c 7.2%), prior NSTEMI 2019 (s/p PCI LAD), and tobacco use disorder (quit 2020). She reports stable exercise tolerance (climbs two flights without angina) and no recent hospitalizations. Dialysis access is functional without recirculation. She has a potential living donor (her daughter, blood type O, in evaluation). Cardiac clearance, cancer screening (mammography, colonoscopy, cervical), and PPD testing are up to date.
62-year-old man s/p DDKT (KDPI 42%, DCD donor, induction with ATG, maintenance tacrolimus/MMF/prednisone) on POD 45, presenting for routine outpatient follow-up. He reports stable energy, no fevers, no dysuria, no graft pain, and good PO intake. Tacrolimus trough today 8.4 ng/mL (target 8-10). Creatinine 1.3 (baseline 1.2 at discharge, nadir 1.1). MMF 1000 mg BID without GI intolerance; prednisone tapered to 10 mg daily. CMV PCR negative (D+/R+, on valganciclovir prophylaxis). BK PCR undetectable. TMP-SMX DS MWF. Urine output remains brisk with no edema. Ureteral stent removal scheduled for next week.
66-year-old man with Child-Pugh B cirrhosis secondary to chronic hepatitis C (SVR achieved 2019 with DAA) and HCC presenting for transplant listing. Initial diagnosis of a 3.2 cm segment VI LI-RADS 5 lesion on MRI 8 months ago, treated with TACE and subsequent Y-90 radioembolization; most recent MRI demonstrates one residual 2.5 cm lesion without vascular invasion or extrahepatic spread, meeting Milan criteria. Current MELD-Na 16, with HCC exception points pending to MELD 28. AFP stable at 24. He endorses mild ascites controlled on furosemide/spironolactone and no episodes of hepatic encephalopathy. EGD 6 months ago showed small varices without red signs.
64-year-old man with idiopathic pulmonary fibrosis (diagnosed 2022, on pirfenidone) presenting for lung transplant listing evaluation. Over the past 6 months he has experienced worsening dyspnea (now mMRC 3), 15-pound weight loss, and a drop in FVC from 62% to 48% predicted and DLCO from 41% to 28%. He requires 3 L/min nasal cannula O2 at rest and 5 L/min with ambulation. Recent 6-minute walk distance 285 meters with SpO2 nadir 84%. Echocardiogram shows estimated PASP 48 mmHg with normal LV function. No history of aspiration, reflux well-controlled on omeprazole. Cardiac catheterization, bone density, cancer screening, and dental evaluation completed; nutrition and psych clearances in progress.
44-year-old woman s/p LDKT (sister donor, 1 ABDR MM, basiliximab induction) POD 98 presenting with a 0.4 mg/dL rise in creatinine (from 1.1 to 1.5) over 2 weeks and new-onset mild graft tenderness. She denies fevers, dysuria, or medication changes. Tacrolimus trough 6.8 (target 6-8). BK PCR negative, CMV PCR negative. UA with trace protein and no cells. Doppler ultrasound shows normal arterial waveforms with RI 0.72 (no rise from baseline). DSA screen sent today (pending). For-cause kidney biopsy scheduled tomorrow given concern for acute rejection vs CNI toxicity vs evolving BK.
55-year-old man s/p OLT (DBD whole liver, MELD 32 at transplant for NASH cirrhosis) now 4 months post-transplant, presenting with 5 days of fatigue, low-grade fevers to 100.8°F, leukopenia (WBC 2.4), and elevated LFTs (AST 96, ALT 110, ALP 180, T.bili 1.4 — up from baseline AST/ALT 25/22). D+/R- CMV serology at transplant. He completed 90 days of valganciclovir prophylaxis 4 weeks ago. CMV PCR today 45,000 IU/mL. Abdominal ultrasound shows patent vessels and no biliary dilation. Plan: admit for IV ganciclovir, reduce MMF dose, hold azathioprine if added, monitor viral load twice weekly, and evaluate for tissue-invasive disease if LFTs do not improve.
The transplant service is a team sport: nephrology/hepatology/cardiology/pulmonology co-manage with transplant surgery, and infectious disease is on speed dial. When the fellow dictates "discussed with ID, will start foscarnet," document the consultation verbatim — the medical record is how the team communicates.
23 References & Sources
Clinical Practice Guidelines
ISHLT. Listing Criteria for Heart Transplantation. J Heart Lung Transplant. 2016.
Loupy A et al. The Banff 2019 Kidney Meeting Report. Am J Transplant. 2020.
Landmark Clinical Trials & Key Studies
Diagram & Figure Sources
Figure 1: MHC Class I Antigen Presentation. Wikimedia Commons. Public domain.
Figure 2: Organ Donation Symbol. Wikimedia Commons. Public domain.
Figure 3: Kidney Transplant Diagram. Wikimedia Commons. Public domain.
Figure 4: Liver Transplant. Wikimedia Commons / NIH. Public domain.
Figure 5: Heart Transplant Diagram. Wikimedia Commons. Public domain.
Figure 6: Calcineurin-NFAT Signaling Pathway. Wikimedia Commons. Public domain.
Closing Note
Transplant surgery rewards scribes who respect the complexity: a single patient has a graft, an immunosuppression regimen, a rejection history, an infection surveillance schedule, and a new set of chronic comorbidities layered on top of the reason they needed a transplant in the first place. Learn the rhythms of the service — the protocol biopsies, the trough checks, the viral load trends — and your notes will become the fellows' favorite reference point during the next clinic. A great transplant scribe makes complicated patients feel navigable, and that is the highest compliment this service offers.