Vascular Surgery
Every diagnosis, condition, procedure, medication, abbreviation, and documentation framework you need to succeed on day one and beyond.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 The Vascular System — Anatomy Essentials
Vascular surgery covers every blood vessel in the body except the coronary arteries (managed by cardiothoracic/interventional cardiologists) and the intracranial vessels (managed by neurosurgeons). This means a vascular surgeon's domain includes the aorta in its entirety, the carotid arteries, the visceral (mesenteric and renal) arteries, the iliac arteries, the entire lower and upper extremity arterial tree, and all corresponding veins and lymphatic structures.
Arterial Anatomy You Must Know
The aorta is the body's largest artery. It exits the heart as the ascending aorta, curves through the aortic arch (giving off the brachiocephalic trunk, left common carotid, and left subclavian arteries), descends through the thorax as the descending thoracic aorta, passes through the diaphragm, and becomes the abdominal aorta. Below the kidneys (infrarenal), it bifurcates into the common iliac arteries at approximately the level of the umbilicus (L4 vertebral body).
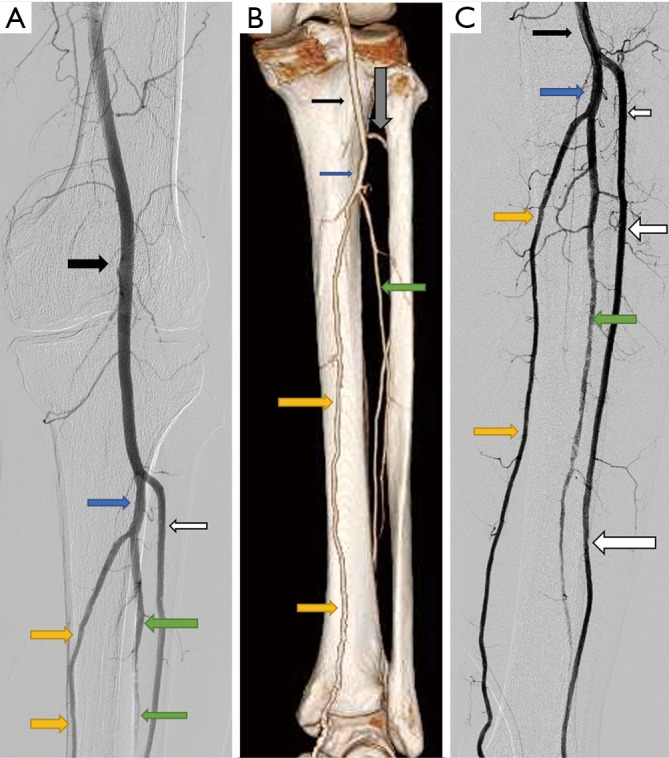
The lower extremity arterial tree follows a predictable sequence: common iliac → external iliac → common femoral artery (CFA) → superficial femoral artery (SFA) and profunda femoris (deep femoral) → popliteal artery → trifurcation into the anterior tibial (AT), posterior tibial (PT), and peroneal arteries. The AT continues as the dorsalis pedis (DP) on the dorsum of the foot, while the PT gives rise to the medial and lateral plantar arteries on the sole. The SFA is the single most common site for atherosclerotic occlusive disease in the lower extremity, particularly at the adductor canal (Hunter's canal).
The carotid system begins with the common carotid artery (CCA), which bifurcates at the level of C3–C4 into the internal carotid artery (ICA) — supplying the brain — and the external carotid artery (ECA) — supplying the face and scalp. The carotid bulb is the dilated area at the bifurcation and is the most common site of plaque buildup.
Venous Anatomy You Must Know
The deep venous system of the lower extremity runs parallel to the arterial system: posterior tibial veins, peroneal veins, anterior tibial veins drain into the popliteal vein, which becomes the femoral vein, then the common femoral vein, and then the external iliac vein and common iliac vein draining into the inferior vena cava (IVC). The superficial venous system consists of the great saphenous vein (GSV) — running medially from the ankle to the saphenofemoral junction (SFJ) in the groin — and the small saphenous vein (SSV) — running posteriorly from the ankle to the saphenopopliteal junction (SPJ). Perforator veins connect the superficial to the deep system through the fascia.
Know the anatomy cold. The surgeon will say things like "The patient has a high-grade SFA lesion at Hunter's canal" or "GSV reflux to below the knee" and you are expected to chart this immediately. The SFA is NOT actually superficial — it runs deep in the thigh, sandwiched between the sartorius and adductor muscles.
02 Scribe Documentation Framework
The SOAP Note in Vascular Surgery
Many vascular surgery encounters are documented in a SOAP-style format. As a scribe, you are writing the physician's note in real time, so understanding each section's purpose and what belongs where is essential.
Chief Complaint (CC): The single reason the patient is being seen today, in the patient's own words or as a brief medical statement. Examples: "right leg pain with walking," "follow-up AAA surveillance," "non-healing wound left foot."
History of Present Illness (HPI): This is the story. Use the OLDCARTS mnemonic — Onset, Location, Duration, Character, Aggravating factors, Relieving factors, Timing, Severity. In vascular surgery, always document walking distance before claudication onset, rest pain (especially nocturnal), wound history, and prior interventions.
Review of Systems (ROS): Document a medically relevant, problem-focused ROS rather than trying to force a 14-system checklist into every note. In vascular clinic, key positives and negatives often include cardiovascular (chest pain, palpitations, dyspnea on exertion), neurological (amaurosis fugax, TIA symptoms, focal weakness), extremities (swelling, discoloration, temperature changes, non-healing wounds), and pulmonary (shortness of breath, cough).
Past Medical History (PMHx), Surgical History (PSHx), Medications, Allergies, Social History (SHx), Family History (FHx): Always capture diabetes (type, A1C), hypertension, hyperlipidemia, CKD/ESRD, coronary artery disease, atrial fibrillation, smoking history (pack-years and quit date), and prior vascular procedures. Family history of aneurysmal disease or early cardiovascular death is highly relevant.
Vitals: Blood pressure in both arms (arm-to-arm difference matters for subclavian stenosis/TOS), heart rate, BMI.
Physical Exam: The vascular-specific exam includes pulse examination (carotid, brachial, radial, femoral, popliteal, dorsalis pedis, posterior tibial — each graded 0 = absent, 1+ = diminished, 2+ = normal, 3+ = bounding), presence of bruits (carotid, femoral, abdominal), skin assessment of the lower extremities (color, temperature, trophic changes, hair loss, dependent rubor, pallor on elevation), edema grading (1+ to 4+), wound assessment using the WIfI classification if applicable, and varicose vein mapping.
Results: Labs (CBC, BMP, coagulation studies, A1C, lipid panel), imaging (duplex ultrasound findings, CTA/MRA results, ABI values). Always document specific numbers, not just "normal" or "abnormal."
The physician's medical decision-making. Document each diagnosis with its ICD-10 code if available, followed by the specific plan: lifestyle modifications, medication changes, imaging orders, referrals, or procedural planning. In vascular surgery, the plan often includes antiplatelet/anticoagulation management, risk factor modification (smoking cessation is paramount), compression therapy, wound care plans, exercise prescriptions, and surgical or endovascular intervention scheduling.
Classic HPI Elements
Older documentation teaching refers to eight classic HPI elements. You do not need to count these elements to determine the office visit level under current outpatient E/M rules, but they remain a useful checklist for capturing a complete vascular history. The eight elements are: Location (which leg? bilateral? which artery territory?), Quality (cramping? burning? sharp? heaviness?), Severity (pain scale 0–10), Duration (how long has this been going on?), Timing (constant? intermittent? nocturnal?), Context (occurs with walking? at rest? after standing all day?), Modifying factors (relieved by rest? dangling legs off the bed? elevation? compression stockings?), and Associated signs/symptoms (numbness? tingling? skin color change? swelling?).
In vascular surgery HPI, always capture claudication distance (how many blocks or minutes of walking before pain forces the patient to stop), whether the patient experiences rest pain (pain at rest, often worse at night, relieved by dangling the leg), and whether there is any tissue loss (ulcers, gangrene). These three features map directly to Fontaine/Rutherford staging and determine whether the patient has lifestyle-limiting claudication versus limb-threatening ischemia.
03 Peripheral Arterial Disease (PAD) Arterial
PAD is the single most common diagnosis in vascular surgery. It refers to atherosclerotic narrowing or occlusion of the arteries supplying the extremities, overwhelmingly affecting the lower limbs. PAD is a systemic manifestation of atherosclerosis — patients with PAD have a significantly elevated risk of myocardial infarction, stroke, and cardiovascular death, not just limb problems.
Pathophysiology
Atherosclerotic plaque — composed of lipids, calcium, fibrous tissue, and inflammatory cells — gradually narrows the arterial lumen. The most commonly affected segments are the superficial femoral artery (especially at the adductor canal), the aortoiliac segment, and the tibial vessels. As the artery narrows, blood flow at rest may remain adequate because of compensatory vasodilation and collateral vessel development. However, during exercise, the increased metabolic demand of working muscles cannot be met, producing the classic symptom of intermittent claudication — muscular pain, cramping, or fatigue that occurs with walking and resolves within minutes of rest.
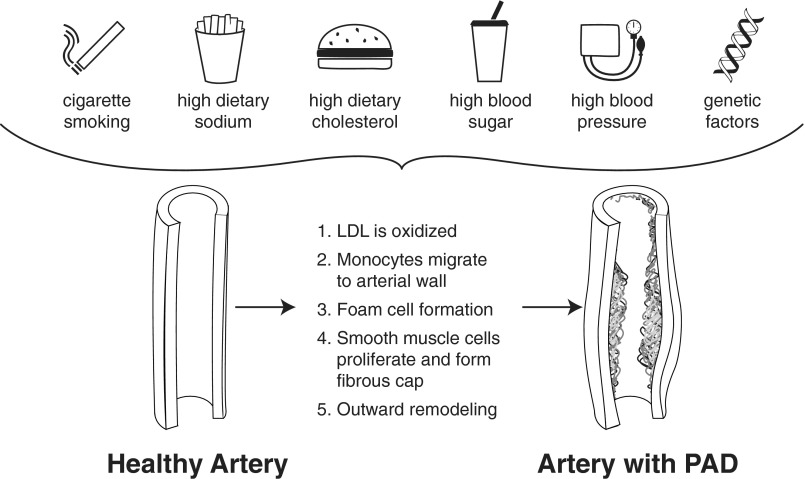
Clinical Presentations
The hallmark symptom: reproducible leg pain (usually calf, but can be buttock/thigh for aortoiliac disease, or foot for tibial disease) that occurs at a consistent walking distance and resolves within 2–5 minutes of standing rest. The location of claudication tells you the level of disease: buttock/hip/thigh claudication suggests aortoiliac disease; calf claudication suggests SFA/popliteal disease; foot claudication suggests tibial disease. Aortoiliac occlusive disease (also called Leriche syndrome when bilateral and complete) classically presents with the triad of buttock claudication, absent femoral pulses, and erectile dysfunction.
As PAD progresses, the blood supply becomes insufficient even at rest. Rest pain typically affects the forefoot and toes, is worse at night when the legs are elevated (loss of gravity-assisted perfusion), and is characteristically relieved by dangling the leg off the side of the bed. This is a sign of chronic limb-threatening ischemia (CLTI) and represents a much more serious stage of disease.
The most advanced presentation: non-healing ulcers, gangrene (dry or wet), or both. Arterial ulcers are typically located on the toes, heel, or lateral malleolus, are often painful, have well-demarcated "punched-out" borders, a pale or necrotic base, and lack granulation tissue. Gangrene that is dry (mummified, black, not infected) can sometimes be managed conservatively with auto-amputation, while wet gangrene (infected, foul-smelling, spreading) is a surgical emergency often requiring urgent amputation.
ABI — The Critical Number
The ankle-brachial index (ABI) is the cornerstone diagnostic test. It is the ratio of systolic blood pressure at the ankle (higher of dorsalis pedis or posterior tibial) to systolic blood pressure in the arm (higher of the two arms). You will document ABI values constantly in vascular clinic.
| ABI Value | Interpretation | Clinical Correlation |
|---|---|---|
| 1.00 – 1.40 | Normal | No significant PAD |
| 0.91 – 0.99 | Borderline | Equivocal; may need exercise ABI |
| 0.70 – 0.90 | Mild PAD | Often asymptomatic or mild claudication |
| 0.50 – 0.69 | Moderate PAD | Claudication with reduced walking distance |
| < 0.50 | Severe PAD / CLTI | Rest pain, non-healing wounds, gangrene |
| > 1.40 | Non-compressible (calcified) | Unreliable — order TBI or duplex instead |
ABIs > 1.40 are falsely elevated due to calcified, non-compressible arteries — common in diabetics and CKD/ESRD patients. The toe-brachial index (TBI) is used instead because digital arteries are less susceptible to calcification. A TBI < 0.70 is diagnostic of PAD. Always check if the patient is diabetic before interpreting ABIs.
Management Overview
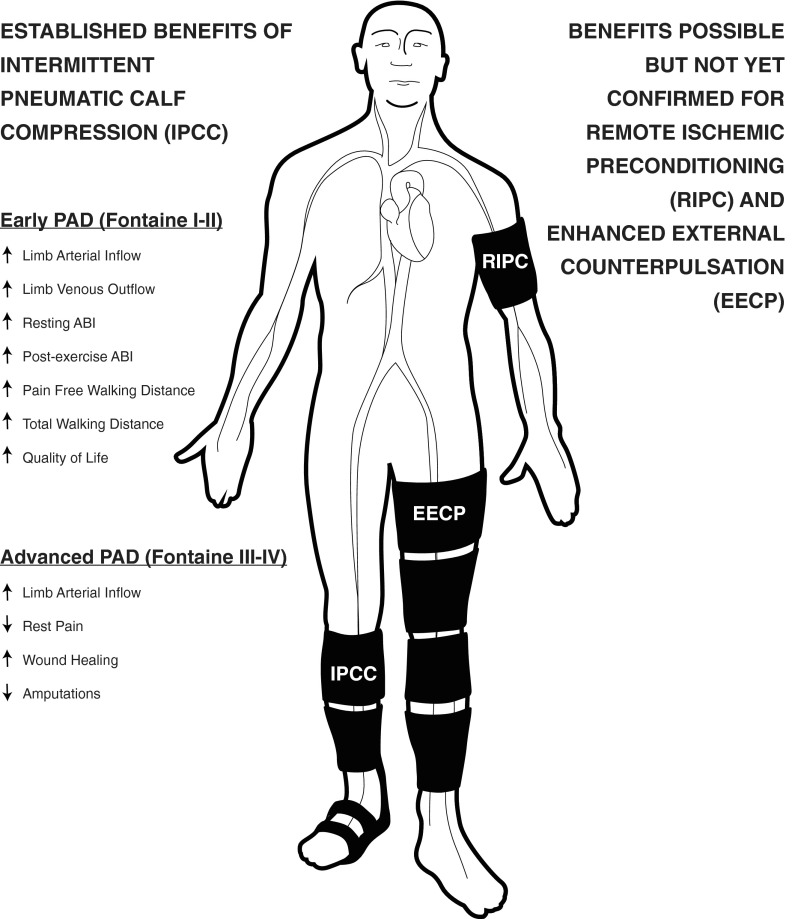
Recent PAD guidance continues to emphasize supervised exercise therapy, risk-factor control, and individualized revascularization. For limb-threatening disease, the SVS Global Vascular Guidelines provide the widely used PLAN and GLASS framework for choosing endovascular-first versus bypass-first strategies.
Vascular surgeons often manage PAD medically before considering intervention. The first-line treatment for intermittent claudication is supervised exercise therapy (SET) — a structured walking program, typically 3 sessions per week for 12+ weeks, where the patient walks until near-maximum claudication pain, rests, and repeats. SET has been shown to double walking distance. Medical therapy includes aggressive cardiovascular risk factor modification: antiplatelet therapy (aspirin 81 mg daily or clopidogrel 75 mg daily), high-intensity statin therapy (atorvastatin 40–80 mg), antihypertensive therapy (targeting BP < 130/80), diabetes management (A1C < 7%), and — critically — smoking cessation. Cilostazol (Pletal) 100 mg BID is the only FDA-approved medication that directly improves claudication symptoms; it is a phosphodiesterase III inhibitor with antiplatelet and vasodilatory properties. Cilostazol is contraindicated in heart failure.
Revascularization — either endovascular (angioplasty, stenting, atherectomy) or open surgical (bypass, endarterectomy) — is reserved for patients with lifestyle-limiting claudication refractory to exercise and medical therapy, or for patients with CLTI (rest pain, tissue loss). The choice between endovascular and open approaches depends on the anatomy (location and extent of disease), patient fitness for surgery, and durability requirements.
04 Carotid Artery Disease & Cerebrovascular Arterial
Carotid artery disease is a common vascular diagnosis. Atherosclerotic plaque builds up at the carotid bifurcation, narrowing the internal carotid artery (ICA). This is dangerous because plaque can rupture and send emboli to the brain, causing a stroke or transient ischemic attack (TIA).
Symptomatic vs. Asymptomatic
This distinction is the single most important factor in determining treatment. A patient is symptomatic if they have had a stroke, TIA, or amaurosis fugax (transient monocular blindness — described as a "curtain coming down over one eye") ipsilateral to the carotid stenosis within the past 6 months. A patient is asymptomatic if they have carotid stenosis discovered incidentally (often from a bruit heard on exam or duplex done for other reasons) without any neurological events.
Degree of Stenosis
Carotid stenosis is graded by the percentage of luminal narrowing, most commonly assessed by carotid duplex ultrasound. Common duplex criteria are: PSV 125–230 cm/sec with EDV 40–100 cm/sec and ICA/CCA ratio 2.0–4.0 suggest 50–69% stenosis, while PSV > 230 cm/sec with EDV > 100 cm/sec and ICA/CCA ratio > 4.0 suggest ≥ 70% stenosis. Near-occlusion can have variable or deceptively low velocities, so CTA or MRA is often used for clarification before intervention.
Symptomatic stenosis: CEA is generally recommended for 70–99% stenosis when perioperative risk is acceptable, and is considered on a case-by-case basis for 50–69% stenosis. When revascularization is chosen, treatment should ideally occur within 14 days of the neurological event. CAS or TCAR are alternatives in selected patients, especially when CEA risk is high or neck anatomy is unfavorable for open surgery.
Asymptomatic high-grade stenosis: Best medical therapy is mandatory. CEA, CAS, or TCAR may be considered only in carefully selected patients with high-grade stenosis, acceptable procedural risk, and reasonable life expectancy; this is not a blanket recommendation for every asymptomatic lesion above a single threshold.
ESVS carotid guidance is useful when you need a source for symptomatic timing, peri-procedural risk thresholds, or how carefully selected asymptomatic high-grade lesions are approached.
Procedures
Carotid endarterectomy (CEA) is the gold-standard open surgical procedure. The surgeon makes a neck incision along the anterior border of the sternocleidomastoid muscle, exposes the carotid bifurcation, clamps the artery, opens it (arteriotomy), physically removes the plaque, and closes the artery — often with a bovine pericardium or Dacron patch to widen the lumen. The patient is often under general anesthesia, though some centers use regional/local anesthesia with intraoperative neurological monitoring.
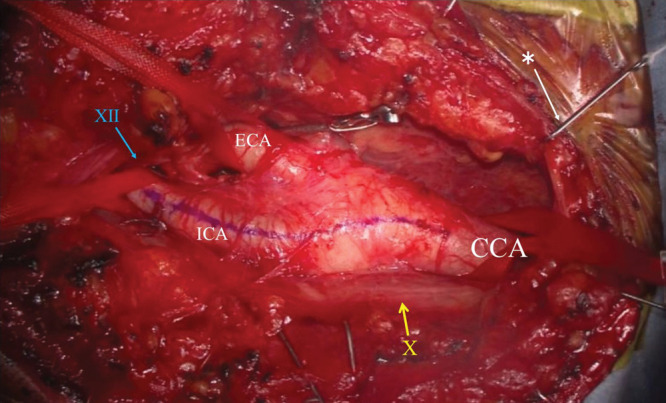
Carotid artery stenting (CAS) is a minimally invasive alternative performed via femoral or radial artery access. A catheter-delivered stent is placed across the stenosis. An embolic protection device (EPD) — either a distal filter or proximal balloon occlusion system — is used to catch embolic debris during the procedure. CAS is typically reserved for patients who are high surgical risk for CEA (prior neck radiation, contralateral occlusion, recurrent stenosis after prior CEA).
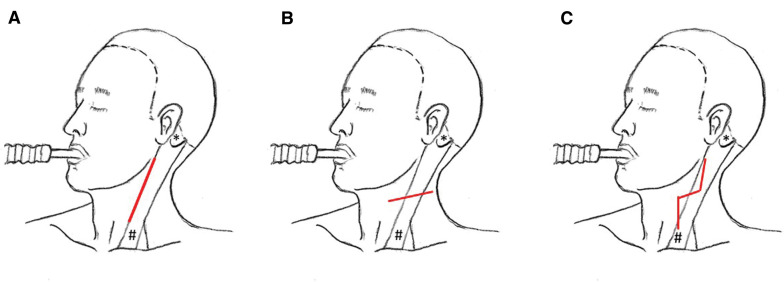
Transcarotid artery revascularization (TCAR) is a newer hybrid approach: direct surgical access to the common carotid artery through a small incision above the clavicle, with flow reversal (blood is diverted through an external filter circuit and returned via a femoral vein sheath) to provide embolic protection during stent deployment. TCAR combines the embolic safety of flow reversal with the minimally invasive nature of stenting.
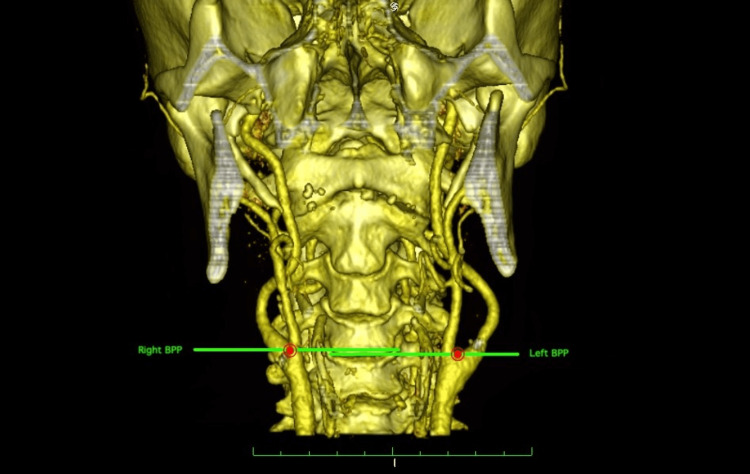
When charting carotid disease, always document: (1) symptomatic or asymptomatic, (2) percent stenosis and how it was measured (duplex PSV/EDV, CTA, MRA), (3) laterality (right, left, bilateral), (4) neurological symptoms if any (TIA symptoms, amaurosis fugax, completed stroke with residual deficits). If the patient had a TIA, document the date and nature of symptoms precisely — this is medically and legally important.
05 Aortic Aneurysms (AAA & TAA) Aortic
An aneurysm is an abnormal dilation of a blood vessel to greater than 1.5 times its normal diameter. For the infrarenal abdominal aorta, the normal diameter is approximately 2.0 cm, so an abdominal aortic aneurysm (AAA) is defined as a diameter ≥ 3.0 cm. Aneurysms are dangerous because they can rupture, causing massive internal hemorrhage with a mortality rate exceeding 80% for ruptured AAAs that do not reach the operating room, and approximately 50% overall including those who reach surgery.
Abdominal Aortic Aneurysm (AAA)
USPSTF screening guidance is the cleanest citation for one-time ultrasound screening in men aged 65–75 who have ever smoked, while the 2022 ACC/AHA aortic guideline is the broader source for surveillance and repair thresholds.
AAAs are overwhelmingly infrarenal (below the renal arteries) and affect men more than women (6:1 ratio). Risk factors include male sex, age > 65, smoking (the strongest modifiable risk factor — responsible for more than 75% of AAAs), family history of AAA (especially first-degree relatives), hypertension, and connective tissue disorders (Marfan syndrome, Ehlers-Danlos syndrome, Loeys-Dietz syndrome). Most AAAs are asymptomatic and discovered incidentally on imaging or screening ultrasound. The USPSTF recommends a one-time screening abdominal ultrasound for all men aged 65–75 who have ever smoked.
Size Thresholds and Surveillance
| AAA Diameter | Management | Surveillance Interval |
|---|---|---|
| < 3.0 cm | Normal — no follow-up needed | — |
| 3.0 – 3.9 cm | Small aneurysm — surveillance | Every 3 years |
| 4.0 – 4.9 cm | Moderate — surveillance | Every 12 months |
| 5.0 – 5.4 cm | Approaching threshold — close surveillance | Every 6 months |
| ≥ 5.5 cm (men) / ≥ 5.0 cm (women) | Elective repair indicated | — |
| Rapid growth > 0.5 cm/6 months | Repair indicated regardless of size | — |
| Symptomatic (any size) | Urgent/emergent repair | — |
Repair Options
Endovascular aneurysm repair (EVAR) is the minimally invasive approach: stent-grafts are deployed through femoral artery access to line the aneurysm from within, excluding it from blood flow. EVAR requires adequate "landing zones" — at least 10–15 mm of non-aneurysmal aorta below the renal arteries (proximal neck) and adequate iliac arteries (distal landing zones). EVAR has lower perioperative mortality than open repair (1–2% vs 3–5%) but requires lifelong surveillance imaging for endoleaks (blood leaking into the aneurysm sac around the graft).
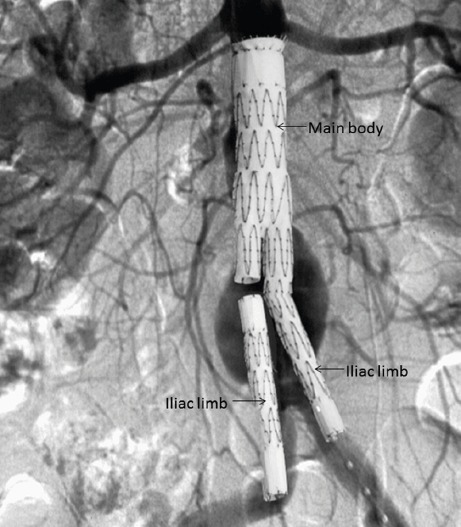

Open surgical repair (OSR) involves a midline laparotomy or retroperitoneal approach, clamping the aorta above and below the aneurysm, opening the aneurysm sac, and sewing in a prosthetic graft (typically Dacron) as a tube graft (if the iliac arteries are normal) or a bifurcated graft (if the iliacs are aneurysmal too). Open repair is more durable and does not require the same degree of surveillance, but has higher perioperative morbidity and a longer recovery.
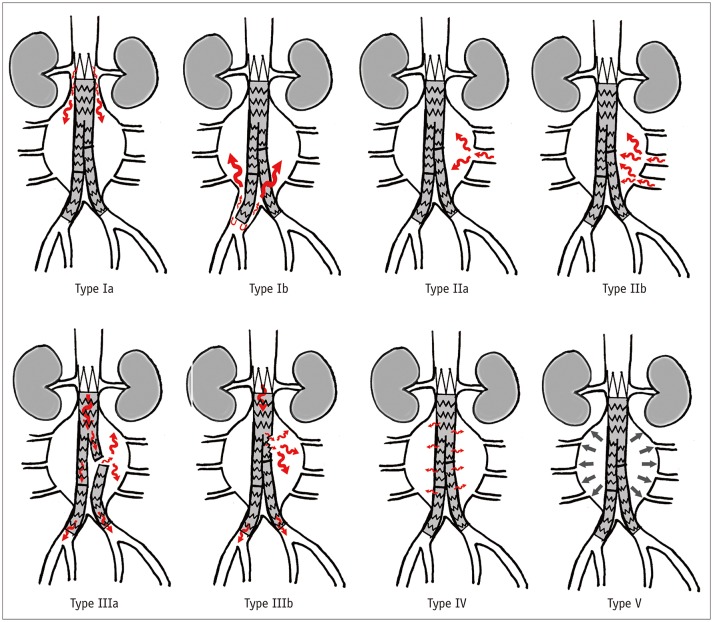
Type I: Leak at the attachment site (proximal or distal seal zone). Requires intervention — the graft is not sealing properly.
Type II: Retrograde flow from branch arteries (lumbar arteries, inferior mesenteric artery) filling the sac. Most common type. Usually observed; only treated if the aneurysm sac is expanding.
Type III: Graft defect — separation of components or fabric tear. Requires intervention.
Type IV: Porosity of the graft fabric. Rare with modern grafts; self-limited.
Type V (endotension): Sac expansion without a demonstrable leak. Controversial entity.
Thoracic Aortic Aneurysm (TAA)
The 2022 ACC/AHA aortic disease guideline is the key reference when you want a source for size thresholds, connective-tissue-disease exceptions, and surveillance language.
Aneurysms of the descending thoracic aorta are repaired with thoracic endovascular aortic repair (TEVAR) or open thoracotomy. The threshold for repair is typically ≥ 5.5–6.0 cm, or ≥ 4.5–5.0 cm in patients with connective tissue disorders. Thoracoabdominal aneurysms involve both the thoracic and abdominal aorta and are commonly described with the Crawford classification (classically Types I–IV, with a modified Type V in some references). They are among the most complex repairs in vascular surgery, requiring fenestrated or branched endografts or open surgical replacement with reimplantation of visceral arteries.
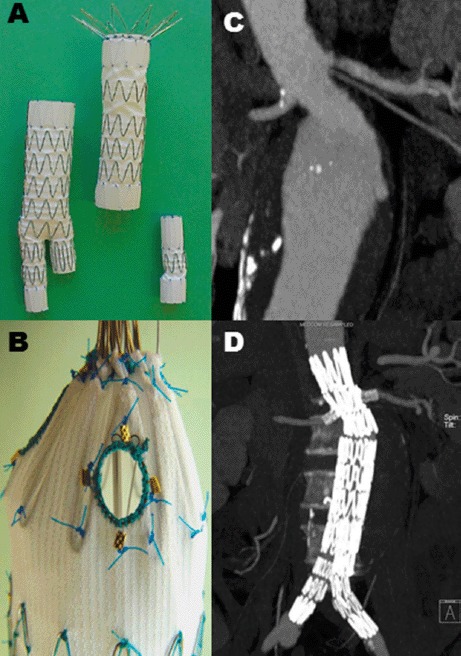
Always document the maximum diameter (in cm), the location (infrarenal, juxtarenal, suprarenal, thoracic, thoracoabdominal), morphology (fusiform vs saccular), and how it was measured (CT, ultrasound, MRA). For post-EVAR patients, document: sac size compared to prior, presence or absence of endoleak, and type of endoleak if present. These surveillance visits are a huge part of vascular clinic.
06 Aortic Dissection & Acute Aortic Syndromes Aortic
The 2022 ACC/AHA aortic guideline is the best single reference for acute aortic syndrome terminology, while INSTEAD-XL is the trial most often cited when TEVAR is discussed in subacute or chronic uncomplicated type B disease.
Aortic dissection is a tear in the inner layer (intima) of the aortic wall, allowing blood to dissect between the intima and the media, creating a "false lumen." This is a life-threatening emergency. The Stanford classification divides dissections into Type A (involving the ascending aorta — requires emergent open surgery, managed by cardiac surgery) and Type B (involving only the descending aorta distal to the left subclavian — managed by vascular surgery). The DeBakey classification is more granular: Type I involves the entire aorta, Type II involves only the ascending, and Type III involves only the descending.
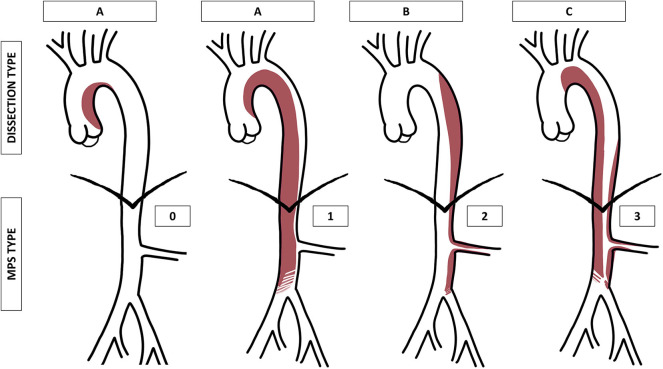
Uncomplicated Type B dissection is initially managed medically with aggressive blood pressure and heart rate control (target SBP < 120, HR < 60 using IV esmolol or labetalol acutely, then oral beta-blockers). Complicated Type B dissection — defined by malperfusion of organs or limbs, rupture, rapid expansion, or refractory pain/hypertension — requires urgent TEVAR to cover the primary entry tear and restore true lumen flow.
Other acute aortic syndromes include intramural hematoma (IMH) — hemorrhage within the aortic wall without an intimal flap, considered a precursor or variant of dissection — and penetrating atherosclerotic ulcer (PAU) — an ulcerated plaque that erodes through the intima into the media. Management depends on location: ascending (type A) IMH or PAU requires urgent surgical evaluation, while uncomplicated descending (type B) disease is often managed medically and complicated descending disease may require TEVAR.
07 Acute Limb Ischemia (ALI) Arterial
ALI is a surgical emergency defined as a sudden decrease in limb perfusion that threatens limb viability, typically occurring within 14 days. The classic teaching mnemonic is the 6 P's: Pain, Pallor, Pulselessness, Poikilothermia (cold), Paresthesia, and Paralysis. The last two (paresthesia and paralysis) are the most ominous and indicate motor/sensory nerve ischemia that may be irreversible if not treated rapidly.
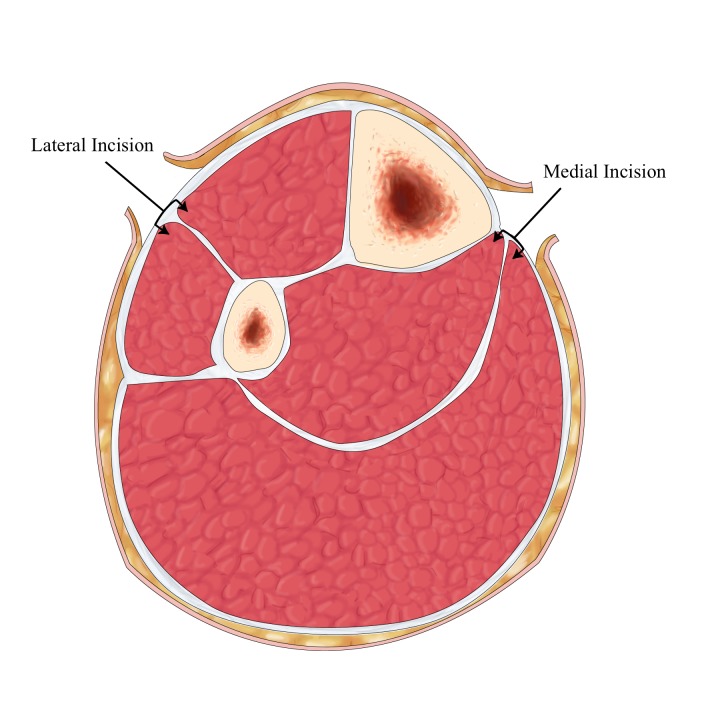
Etiology
The two main causes are embolism (typically from a cardiac source — atrial fibrillation is the most common; also mural thrombus after MI, valvular heart disease, or paradoxical embolism through a PFO) and thrombosis in situ (acute thrombosis of a pre-existing atherosclerotic lesion or bypass graft). Embolic ALI tends to present more dramatically because there are no pre-developed collateral vessels, whereas thrombotic ALI in a patient with chronic PAD may be less acute because collaterals have already formed.
Rutherford Classification for ALI
| Category | Description | Sensory Loss | Motor Deficit | Doppler Signals |
|---|---|---|---|---|
| I — Viable | Not immediately threatened | None | None | Audible arterial & venous |
| IIa — Marginally threatened | Salvageable if treated promptly | Minimal (toes) | None | Often inaudible arterial, audible venous |
| IIb — Immediately threatened | Salvageable with immediate revascularization | Beyond toes, rest pain | Mild to moderate | Inaudible arterial, audible venous |
| III — Irreversible | Major tissue loss or permanent nerve damage inevitable | Profound, anesthetic | Profound (paralysis, rigor) | Inaudible arterial & venous |
Treatment of ALI is time-critical. Options include surgical thromboembolectomy (often using a Fogarty balloon catheter passed through a cutdown incision), catheter-directed thrombolysis (CDT) (infusion of tPA directly into the clot via catheter), percutaneous mechanical thrombectomy, or surgical bypass. If the limb is Rutherford III (irreversible), primary amputation is typically indicated.
After revascularization of an acutely ischemic limb, watch for reperfusion injury and compartment syndrome. Reperfusion can cause rhabdomyolysis (monitor CK and urine myoglobin), hyperkalemia (can cause cardiac arrest), and metabolic acidosis. Compartment syndrome presents with increasing pain, tenseness of the compartments, and pain with passive stretch — treatment is emergent four-compartment fasciotomy.
08 Chronic Limb-Threatening Ischemia (CLTI) Arterial
The modern WIfI, GLASS, and PLAN language in this section comes from the SVS Global Vascular Guidelines on CLTI.
CLTI is the advanced stage of PAD, defined by the presence of ischemic rest pain, non-healing ischemic ulceration, or gangrene for > 2 weeks. This is the modern term replacing "critical limb ischemia (CLI)." CLTI carries a grim prognosis: without revascularization, approximately 25–40% of patients will require major amputation within 6 months, and 1-year mortality is approximately 20–25%.
The SVS WIfI classification (Wound, Ischemia, foot Infection) was developed to better stratify CLTI patients. Each component is graded 0–3:
| Grade | Wound (W) | Ischemia (I) | Foot Infection (fI) |
|---|---|---|---|
| 0 | No ulcer or gangrene | ABI ≥ 0.80, ankle pressure ≥ 100 mmHg | No infection |
| 1 | Small, shallow ulcer; no gangrene | ABI 0.60–0.79, ankle pressure 70–100 | Mild — local infection, skin/subQ only |
| 2 | Deeper ulcer, exposed bone/tendon, or gangrene limited to digits | ABI 0.40–0.59, ankle pressure 50–70 | Moderate — deeper abscess, cellulitis |
| 3 | Extensive ulcer or gangrene involving forefoot/midfoot | ABI ≤ 0.39, ankle pressure < 50 | Severe — sepsis, SIRS, limb-threatening |
Management always begins with risk factor modification and infection control if present. Revascularization planning uses CTA, MRA, or angiography to map the anatomy. The goal is to restore "straight-line flow" to the foot via at least one tibial or pedal artery. The best revascularization strategy (endovascular versus open bypass) is individualized using the SVS Global Vascular Guidelines PLAN framework: Patient risk, Limb staging (WIfI), ANatomic pattern (GLASS classification — Global Limb Anatomic Staging System).
09 Deep Vein Thrombosis (DVT) Venous
DVT is the formation of a blood clot within a deep vein, most commonly in the lower extremities (femoral, popliteal, iliac veins). The pathogenesis is described by Virchow's triad: venous stasis (immobilization, prolonged travel, heart failure), endothelial injury (surgery, trauma, central lines), and hypercoagulability (cancer, pregnancy, oral contraceptives, inherited thrombophilias like Factor V Leiden or Prothrombin 20210A mutation).
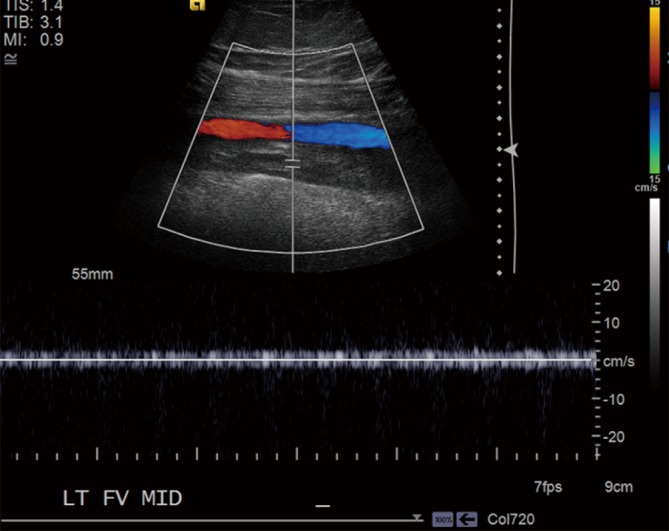
Classification
Proximal DVT (popliteal vein and above — femoral, iliac) carries a significant risk of pulmonary embolism (PE) and typically requires anticoagulation. Distal DVT (calf veins — gastrocnemius, soleal, peroneal, tibial veins) has a lower risk of PE; management is debated and may include serial surveillance imaging versus anticoagulation depending on risk factors. Iliofemoral DVT — involving the iliac and/or common femoral veins — is the most extensive form and may benefit from catheter-directed thrombolysis in young, otherwise healthy patients with low bleeding risk, especially if there is phlegmasia.
Phlegmasia cerulea dolens ("painful blue swelling") is a massive, limb-threatening DVT that obstructs nearly all venous outflow, causing a tense, cyanotic, painful limb. It can progress to venous gangrene and limb loss if not treated urgently with anticoagulation, elevation, and often catheter-directed thrombolysis or surgical thrombectomy. Phlegmasia alba dolens ("painful white swelling") is an earlier stage where the limb is swollen and pale.
Treatment
CHEST VTE guidance is the standard citation when you need a source for anticoagulation-first management, treatment duration, and when intervention is reserved for selected iliofemoral or limb-threatening presentations.
First-line treatment is systemic anticoagulation. For most DVTs, direct oral anticoagulants (DOACs) like rivaroxaban (Xarelto) or apixaban (Eliquis) are first-line. Heparin (unfractionated or LMWH like enoxaparin) is used as bridging therapy or in settings where rapid reversal may be needed (perioperative, renal failure). Warfarin (Coumadin) with a target INR of 2.0–3.0 is still used in some patients (mechanical heart valves, antiphospholipid syndrome). Duration of anticoagulation: 3 months for provoked DVT (identifiable trigger), often indefinite for unprovoked or recurrent DVT.
Vascular surgeons are involved in DVT management when intervention beyond anticoagulation is needed: catheter-directed thrombolysis (CDT), pharmacomechanical thrombectomy (PMT), IVC filter placement (for patients who cannot tolerate anticoagulation or have recurrent PE despite adequate anticoagulation), and venous stenting (for iliocaval venous compression/stenosis such as May-Thurner syndrome).
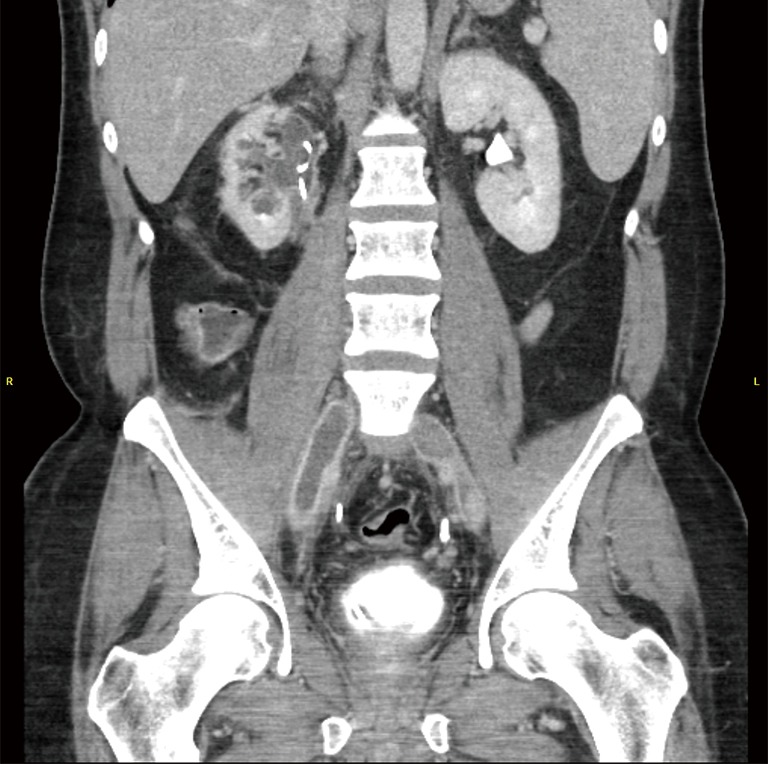
Compression of the left common iliac vein by the overlying right common iliac artery against the lumbar spine. This anatomic variant predisposes to left-sided iliofemoral DVT, particularly in young women. Treatment involves anticoagulation for the DVT, catheter-directed thrombolysis to clear the clot, followed by iliac vein stenting to relieve the compression.
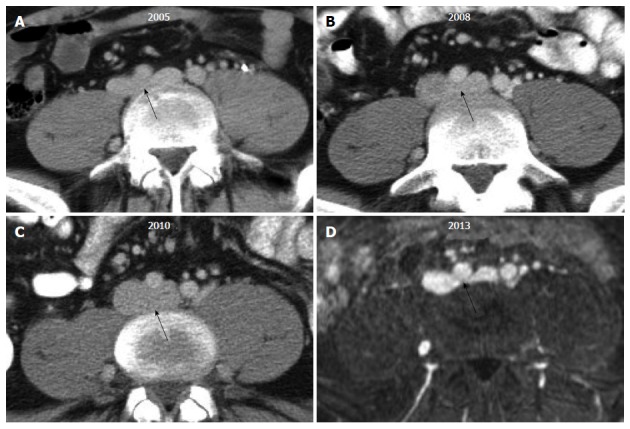
10 Chronic Venous Insufficiency (CVI) Venous
CVI results from incompetent (leaky) venous valves, leading to sustained venous hypertension in the lower extremities. The most common cause is prior DVT (post-thrombotic syndrome), followed by primary valvular incompetence. Venous blood pools in the legs, causing a cascade of symptoms: swelling, heaviness, aching, skin changes, and ultimately venous ulceration.
CEAP Classification
The universal classification system for chronic venous disease, covering Clinical, Etiological, Anatomic, and Pathophysiological categories. The clinical (C) grades are the ones you will chart most often:
| Class | Clinical Finding |
|---|---|
| C0 | No visible or palpable signs of venous disease |
| C1 | Telangiectasias (spider veins) or reticular veins |
| C2 | Varicose veins (> 3 mm diameter) |
| C3 | Edema without skin changes |
| C4a | Pigmentation (hemosiderin staining) or eczema |
| C4b | Lipodermatosclerosis or atrophie blanche |
| C5 | Healed venous ulcer |
| C6 | Active venous ulcer |
Venous ulcers are the most common type of lower-extremity ulcer and typically occur at the medial malleolus (the "gaiter area"). They tend to be shallow, irregularly shaped, with a pink/red granulating base, and surrounded by brown hemosiderin-stained skin. They are often weepy (exudative) and generally less painful than arterial ulcers, unless infected. Treatment is compression therapy — multi-layer bandaging or graduated compression stockings (typically 30–40 mmHg). Note: compression is contraindicated if the ABI is < 0.5 (severe arterial disease).
When documenting CVI, always record the CEAP class, laterality, presence of reflux on duplex (and which veins — GSV, SSV, perforators, deep system), and ulcer characteristics if present (size in cm, location, depth, wound bed description, surrounding skin, signs of infection). This is highly standardized documentation.
11 Varicose Veins & Superficial Venous Disease Venous
Varicose veins are dilated, tortuous superficial veins ≥ 3 mm in diameter, resulting from incompetent valves in the superficial venous system (usually the GSV or SSV). While often considered a cosmetic concern, varicose veins can cause significant symptoms: heaviness, aching, throbbing, burning, itching, night cramps, and restless legs. They may also lead to complications including superficial thrombophlebitis, bleeding, and progression to CVI with skin changes and ulceration.
Diagnosis is by clinical inspection and venous duplex ultrasound with reflux testing. The patient stands upright, and the Valsalva maneuver or calf compression/release is used to provoke reflux. Reflux is defined as retrograde flow lasting > 0.5 seconds in the superficial veins or > 1.0 second in the deep veins.
Treatment Options
Conservative management starts with compression stockings (20–30 or 30–40 mmHg), leg elevation, exercise, and weight management. When intervention is indicated, the mainstay is endovenous thermal ablation — either radiofrequency ablation (RFA, using the ClosureFast catheter) or endovenous laser ablation (EVLA/EVLT). These procedures deliver heat through a catheter inserted into the GSV or SSV under ultrasound guidance, causing the vein to seal shut. They are performed in the office under local anesthesia (tumescent anesthesia).
Alternatives include mechanochemical ablation (MOCA) using the ClariVein device (combines mechanical disruption with sclerosant injection), cyanoacrylate closure (VenaSeal — medical-grade glue injected into the vein), sclerotherapy (injection of a sclerosant like sodium tetradecyl sulfate or polidocanol to chemically destroy the vein — used for spider veins and smaller varicosities), and ambulatory phlebectomy (micro-incisions to remove varicose tributaries).
12 Dialysis Access (AVF, AVG, CVC) Special
KDOQI 2019 vascular access guidance is the key reference for access preference, maturation targets, and how fistulas, grafts, and catheters are prioritized.
Vascular surgeons create and maintain vascular access for hemodialysis in patients with end-stage renal disease (ESRD). The three types of hemodialysis access, in order of preference, are:
A surgically created direct connection between an artery and a vein, typically in the forearm (radiocephalic or Brescia-Cimino fistula) or upper arm (brachiocephalic or brachiobasilic fistula). The arterial pressure causes the vein to dilate and thicken ("mature" or "arterialize"), creating a durable access that can be repeatedly cannulated for dialysis. Maturation takes 4–8 weeks. The "rule of sixes" defines a mature fistula: flow rate ≥ 600 mL/min, diameter ≥ 6 mm, depth ≤ 6 mm from the skin surface, and at least 6 cm of usable length. AVFs have the best long-term patency and lowest infection rates.
A synthetic tube (typically ePTFE — expanded polytetrafluoroethylene, i.e., Gore-Tex) connecting an artery to a vein. Used when the patient's native veins are inadequate for a fistula. AVGs can be used sooner (often 2–3 weeks after placement) but have higher rates of thrombosis, infection, and stenosis (especially at the venous anastomosis) compared to AVFs.
Tunneled (Permacath) or non-tunneled catheters placed in the internal jugular, subclavian, or femoral veins. These are used when urgent dialysis is needed before an AVF/AVG has been created or matured, or when all other options have been exhausted. CVCs have the highest infection rates and are associated with central venous stenosis.
Common problems you will document in dialysis access patients include: stenosis (most commonly at the venous anastomosis of a graft or the juxta-anastomotic segment of a fistula — causing elevated venous pressures during dialysis, poor clearance, and prolonged bleeding after dialysis), thrombosis (access suddenly stops working — treated with surgical or endovascular thrombectomy), steal syndrome (diversion of arterial blood flow through the access causes ischemia to the hand — cold, painful, numb hand distal to the access), aneurysm/pseudoaneurysm (dilation at cannulation sites), and infection.
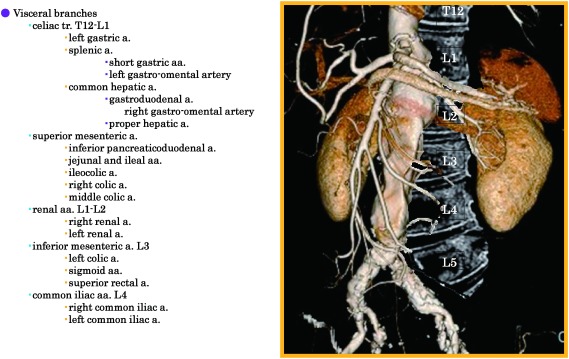
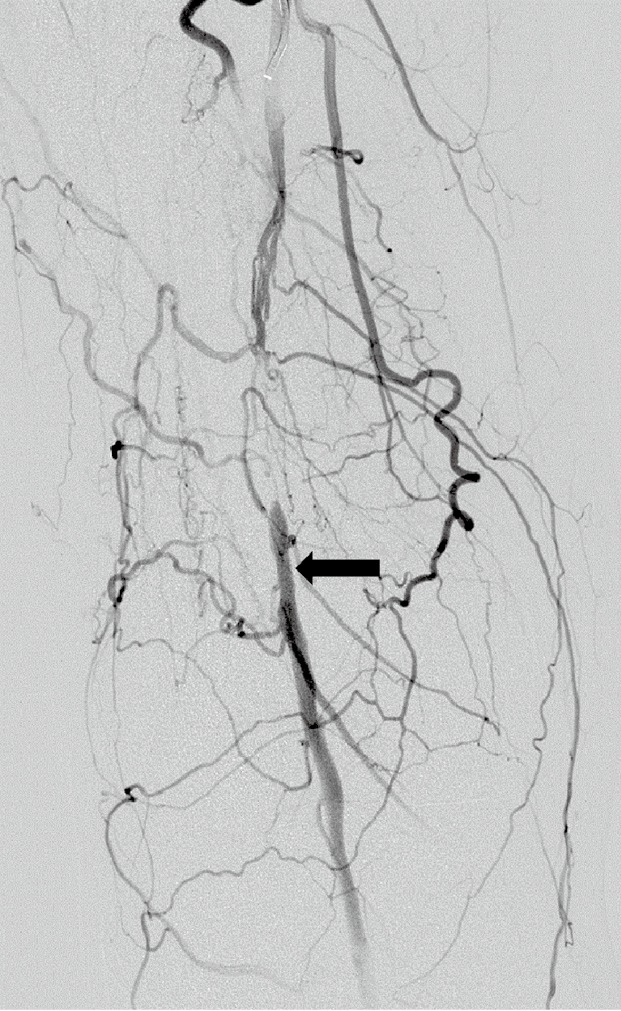
13 Mesenteric & Renal Artery Disease Arterial
Mesenteric Ischemia
Acute mesenteric ischemia (AMI) is a surgical emergency with mortality rates of 60–80%. Causes include superior mesenteric artery (SMA) embolism (most common, often from a cardiac source), SMA thrombosis (on pre-existing atherosclerotic disease), mesenteric venous thrombosis, and non-occlusive mesenteric ischemia (NOMI — vasospasm, usually in critically ill patients on vasopressors). The classic presentation is "pain out of proportion to exam" — severe abdominal pain with a relatively benign initial physical examination. CT angiography (CTA) is the diagnostic study of choice. Treatment is urgent revascularization (embolectomy, thrombolysis, or surgical bypass) combined with assessment of bowel viability (often requires exploratory laparotomy with possible bowel resection).
Chronic mesenteric ischemia (CMI) is caused by atherosclerotic occlusion of mesenteric arteries (usually ≥ 2 of the 3 mesenteric vessels — celiac, SMA, IMA — must be significantly stenosed or occluded to cause symptoms, due to extensive collateral networks). The classic triad is: postprandial abdominal pain ("intestinal angina" — pain within 15–30 minutes after eating, lasting 1–3 hours), food fear (sitophobia — patients avoid eating to avoid pain), and weight loss. Treatment is revascularization, either mesenteric stenting (endovascular, first-line) or surgical bypass.
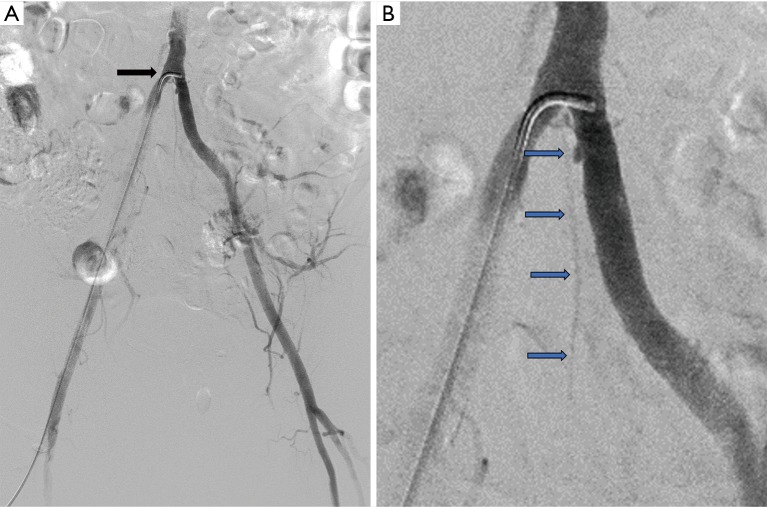
Renal Artery Stenosis (RAS)
Narrowing of the renal artery, most commonly due to atherosclerosis (affects the ostium/proximal renal artery, typically in older patients with other atherosclerotic risk factors) or fibromuscular dysplasia (FMD) (affects the mid-to-distal renal artery, classically in young women, producing a "string of beads" appearance on angiography). RAS can cause renovascular hypertension (resistant hypertension, especially new onset after age 55), and ischemic nephropathy (progressive renal failure). Screening is by renal artery duplex ultrasound. Treatment of atherosclerotic RAS is primarily medical (blood pressure control, statins); renal artery stenting is reserved for specific indications (failure of medical therapy, flash pulmonary edema, declining renal function). FMD is treated with balloon angioplasty (without stenting) and generally has excellent results.
14 Thoracic Outlet Syndrome (TOS) Special
TOS encompasses a group of disorders caused by compression of the neurovascular bundle (brachial plexus, subclavian artery, subclavian vein) as it passes through the thoracic outlet — the space between the clavicle, first rib, and scalene muscles. TOS is classified into three types:
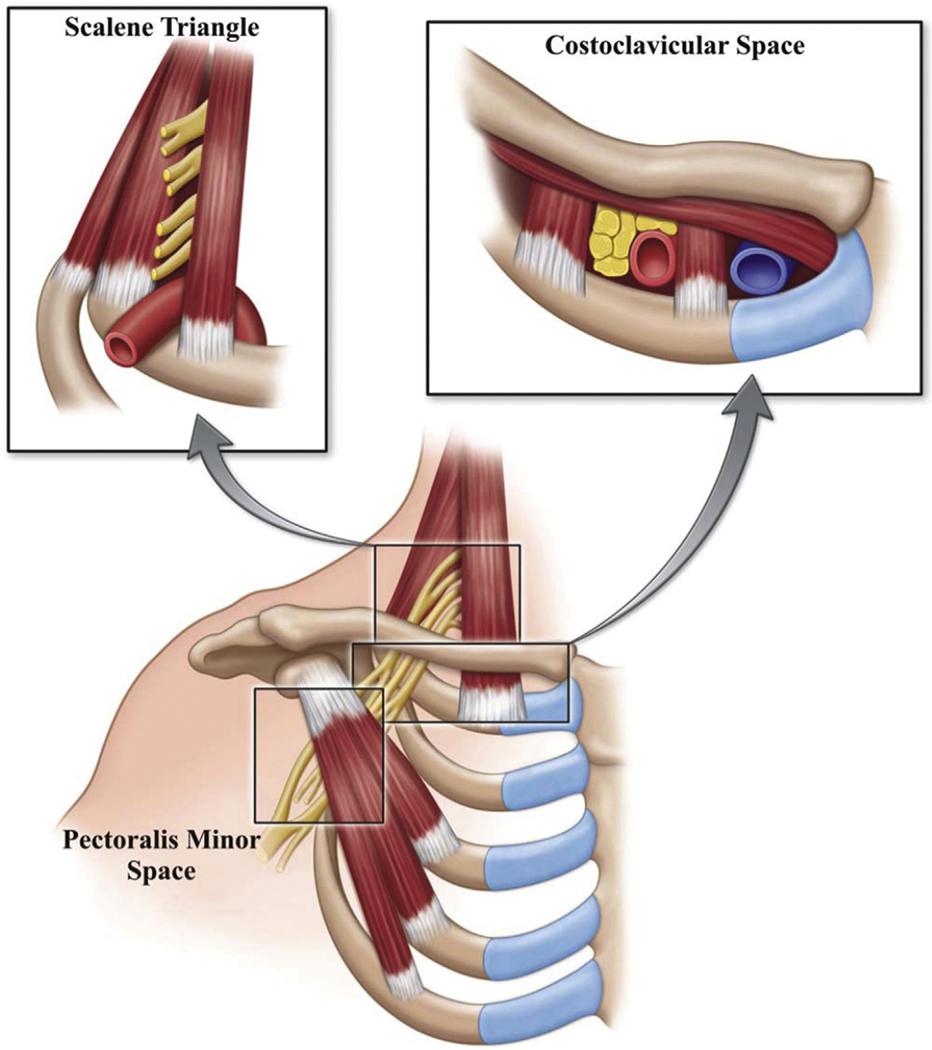
Neurogenic TOS (nTOS) — by far the most common (~95% of cases). Compression of the brachial plexus causes pain, numbness, tingling, and weakness in the arm and hand, typically exacerbated by overhead arm positioning. Diagnosis is largely clinical and supported by provocative maneuvers (Adson's test, Wright's test, Roos test/EAST — elevated arm stress test). Treatment starts with physical therapy; surgical first rib resection is reserved for refractory cases.
Venous TOS (vTOS) — also called Paget-Schroetter syndrome or "effort thrombosis." Compression of the subclavian vein leads to DVT of the upper extremity, often precipitated by repetitive overhead activity (swimmers, pitchers, weightlifters). Presents as sudden arm swelling, pain, and cyanosis. Treatment is anticoagulation, catheter-directed thrombolysis, and subsequent first rib resection to decompress the vein.
Arterial TOS (aTOS) — the rarest and most dangerous type. Compression of the subclavian artery (often by a cervical rib or anomalous first rib) can cause post-stenotic aneurysm formation and distal embolization (blue finger syndrome). Treatment is first rib resection and arterial reconstruction.
15 Diabetic Foot & Wound Care Special
IWGDF/IDSA diabetic foot infection guidance is the best citation for multidisciplinary wound assessment, infection severity, and when surgical source control is required.
Diabetic foot disease is an enormous part of vascular surgery practice. Diabetes causes both macrovascular disease (PAD — affecting the tibial arteries most severely in diabetics) and microvascular disease, combined with peripheral neuropathy (loss of protective sensation) and altered foot biomechanics (Charcot arthropathy). The result is that diabetic patients develop foot ulcers that do not heal, get infected, and can ultimately require amputation.
The key to diabetic foot management is a multidisciplinary approach: vascular assessment (ABIs/TBIs, duplex, CTA to assess perfusion), wound care (debridement, offloading with total contact casting or specialized shoes, advanced wound care products), infection management (antibiotics when indicated, surgical drainage of abscesses, I&D of osteomyelitis), and revascularization when ischemia is contributing. The WIfI classification (discussed under CLTI) is essential for risk-stratifying diabetic foot patients.
When documenting diabetic foot wounds, always record: exact location on the foot (plantar 1st metatarsal head, lateral 5th toe, heel, dorsum, etc.), dimensions (length × width × depth in cm), wound bed characteristics (granulation tissue, slough, eschar, exposed bone/tendon), presence of tunneling or undermining, surrounding skin (erythema, warmth, induration, maceration), signs of infection (purulence, fluctuance, crepitus, foul odor), and neurovascular status (sensation testing with Semmes-Weinstein monofilament, pulse palpation, capillary refill).
Amputation Levels
When tissue cannot be salvaged, amputation may be required. Levels in order of ascending severity: toe amputation, ray amputation (toe + its metatarsal), transmetatarsal amputation (TMA), Chopart amputation (midfoot), Syme amputation (ankle disarticulation), below-knee amputation (BKA), and above-knee amputation (AKA). The goal is always to preserve the most functional limb length possible.
16 Lymphedema Special
Lymphedema is the accumulation of protein-rich interstitial fluid due to impaired lymphatic drainage. Primary lymphedema results from congenital malformation of the lymphatic system (lymphedema praecox presents before age 35, lymphedema tarda presents after 35). Secondary lymphedema is far more common and results from damage to the lymphatic system by surgery (particularly axillary lymph node dissection for breast cancer), radiation therapy, infection (filariasis — the most common cause worldwide), malignancy, or trauma.
Clinically, lymphedema presents as progressive, non-pitting (or initially pitting) swelling that is often asymmetric, worsens through the day, and is improved by elevation overnight. A positive Stemmer's sign (inability to pinch the skin on the dorsum of the second toe into a fold) supports the diagnosis. Over time, the skin becomes thickened (fibrosis), develops a "peau d'orange" texture, and is prone to recurrent infections (cellulitis, lymphangitis). Treatment is lifelong and consists of complete decongestive therapy (CDT): manual lymphatic drainage massage, multi-layer compression bandaging, compression garments, exercise, and meticulous skin care. Surgery is rarely needed and is reserved for severe cases.
17 Vascular Procedures — A to Z Procedures
| Procedure | What It Is | When It's Used |
|---|---|---|
| Angioplasty (PTA) | Balloon dilation of a stenosed artery | PAD, renal artery stenosis, venous stenosis |
| Stenting | Metal mesh scaffold placed to hold artery/vein open | Iliac, SFA, carotid (CAS), venous (iliac, SVC) |
| Atherectomy | Removal of plaque by cutting, shaving, or laser | SFA/popliteal lesions, in-stent restenosis |
| CEA | Open removal of carotid plaque | Selected symptomatic carotid stenosis and carefully selected asymptomatic high-grade stenosis |
| CAS / TCAR | Carotid stenting (percutaneous or transcarotid) | High-risk CEA patients |
| EVAR | Endovascular stent-graft for AAA | AAA ≥ 5.5 cm with suitable anatomy |
| TEVAR | Thoracic endovascular aortic repair | TAA, Type B dissection, traumatic aortic injury |
| Open AAA repair | Laparotomy, aortic cross-clamp, graft interposition | AAA unsuitable for EVAR |
| Bypass grafting | New conduit (vein or prosthetic) around blocked artery | Femoral-popliteal (fem-pop), femoral-tibial, aortobifemoral |
| Embolectomy/Thrombectomy | Removal of clot (Fogarty balloon or mechanical) | ALI, graft thrombosis |
| Thrombolysis (CDT) | Catheter-delivered tPA to dissolve clot | ALI (IIa), iliofemoral DVT, graft thrombosis |
| IVC filter placement | Filter in the IVC to catch PE-causing emboli | Contraindication to anticoagulation with DVT/PE |
| AVF/AVG creation | Dialysis access creation | ESRD requiring hemodialysis |
| Fistulogram/Graftogram | Contrast study of dialysis access | Dysfunctional access — stenosis, thrombosis |
| Endovenous ablation (RFA/EVLT) | Thermal sealing of incompetent saphenous vein | Symptomatic varicose veins with GSV/SSV reflux |
| Sclerotherapy | Chemical injection to close small veins | Spider veins, small varicosities |
| Fasciotomy | Opening fascial compartments to relieve pressure | Compartment syndrome after revascularization |
| Amputation (toe/TMA/BKA/AKA) | Surgical removal of non-viable tissue | Non-salvageable ischemia, uncontrolled infection |
| Wound debridement | Removal of necrotic/infected tissue | Chronic wounds, diabetic foot ulcers |
18 Vascular Imaging & Diagnostics Special
| Modality | What It Shows | Key Notes for Scribes |
|---|---|---|
| Ankle-Brachial Index (ABI) | Ratio of ankle to arm blood pressure | Document exact numbers bilaterally; note if exercise ABIs were done |
| Toe-Brachial Index (TBI) | Same as ABI but at the toe; avoids calcification artifact | Used when ABI > 1.40 (diabetics, CKD) |
| Arterial Duplex Ultrasound | Visualizes arterial anatomy + measures flow velocities | Document PSV, stenosis locations, waveform patterns |
| Venous Duplex Ultrasound | Detects DVT (compressibility) and reflux | Document which veins affected, reflux duration in seconds |
| Carotid Duplex | Stenosis grading of carotid arteries by velocity criteria | PSV, EDV, ICA/CCA ratio — document all |
| CT Angiography (CTA) | Detailed 3D arterial anatomy with IV contrast | Gold standard for surgical planning; check creatinine/eGFR first |
| MR Angiography (MRA) | Arterial imaging without ionizing radiation | Check renal function and gadolinium agent class; group II agents are preferred in advanced CKD because NSF risk is very low |
| Digital Subtraction Angiography (DSA) | Real-time fluoroscopic imaging of arteries | Invasive; gold standard but used mainly during interventions |
| Segmental Pressures | BP at multiple levels (thigh, calf, ankle) to localize disease | A > 20 mmHg gradient between adjacent segments = significant stenosis |
| Pulse Volume Recordings (PVR) | Plethysmographic waveforms at multiple levels | Qualitative — normal has a sharp upstroke, flat waveforms indicate disease |
| Transcutaneous O₂ (TcPO₂) | Tissue oxygenation at skin level | > 40 mmHg = adequate for healing; < 30 mmHg = critical ischemia |
| Intravascular Ultrasound (IVUS) | Ultrasound probe inside the vessel during procedure | Used during venous stenting (iliac compression), aortic procedures |
19 Medications You Must Know Meds
Antiplatelet Agents
| Drug | Brand | Mechanism | Typical Use in Vasc Surg | Key Notes |
|---|---|---|---|---|
| Aspirin | — | Irreversible COX-1 inhibitor | All PAD, post-CEA, post-stenting | 81 mg or 325 mg daily; hold 7 days pre-op if needed |
| Clopidogrel | Plavix | Irreversible P2Y12 (ADP) receptor blocker | PAD, dual antiplatelet after stenting | 75 mg daily; hold 5 days pre-op |
| Ticagrelor | Brilinta | Reversible P2Y12 blocker | Post-ACS, sometimes PAD (EUCLID trial) | 90 mg BID; hold 5 days pre-op |
| Cilostazol | Pletal | PDE3 inhibitor → vasodilation + antiplatelet | Intermittent claudication | 100 mg BID; contraindicated in CHF |
| Dipyridamole + ASA | Aggrenox | Nucleoside transport inhibitor + COX inhibitor | Secondary stroke prevention | 200/25 mg BID |
Anticoagulants
| Drug | Brand | Mechanism | Key Notes |
|---|---|---|---|
| Heparin (UFH) | — | Activates antithrombin III → inhibits IIa and Xa | IV drip; monitor with aPTT; reversible with protamine |
| Enoxaparin | Lovenox | LMWH — primarily anti-Xa | SubQ injection; dose-adjust for renal impairment; monitor with anti-Xa levels if needed |
| Warfarin | Coumadin | Vitamin K antagonist (inhibits II, VII, IX, X) | Monitor with INR (target 2.0–3.0 usually); reverse with vitamin K, FFP, or PCC |
| Rivaroxaban | Xarelto | Direct factor Xa inhibitor | DVT/PE treatment; 2.5 mg BID + ASA for PAD (COMPASS/VOYAGER trials) |
| Apixaban | Eliquis | Direct factor Xa inhibitor | DVT/PE treatment; preferred in elderly/renal impairment |
| Dabigatran | Pradaxa | Direct thrombin (IIa) inhibitor | AF stroke prevention; reversible with idarucizumab (Praxbind) |
Other Key Medications
| Drug | Category | Relevance |
|---|---|---|
| Atorvastatin (Lipitor) 40–80 mg | High-intensity statin | All PAD patients — reduces cardiovascular events |
| Rosuvastatin (Crestor) 20–40 mg | High-intensity statin | Alternative to atorvastatin |
| Metoprolol, Atenolol | Beta-blocker | Rate/BP control; perioperative cardiac risk reduction |
| Amlodipine | Calcium channel blocker | HTN management, also used in Raynaud's phenomenon |
| Lisinopril, Losartan | ACE-I / ARB | HTN, renal protection in diabetics |
| Metformin | Biguanide | First-line diabetes med; routine IV iodinated contrast usually does not require a hold if eGFR is stable and there is no AKI |
| Pentoxifylline (Trental) | Methylxanthine | Older claudication drug — improves RBC deformability; modest benefit |
Metformin and contrast: For routine IV iodinated contrast, metformin is usually continued if the patient has no AKI and eGFR is at least 30. Hold it for AKI, eGFR < 30, or higher-risk arterial catheter studies that could affect renal perfusion, and resume according to local protocol once renal function is stable.
20 Classification Systems Special
Vascular surgery uses many classification systems. You do not need to memorize every detail, but you need to know what each system classifies and the basic stages. The most important ones:
| Classification | What It Classifies | Key Features |
|---|---|---|
| Fontaine | Chronic PAD severity | 4 stages based on clinical symptoms only (I–IV). European origin. |
| Rutherford | Chronic PAD severity | 7 categories (0–6) with objective hemodynamic data (ABI, PVR). US standard. |
| WIfI | CLTI / limb threat | Grades Wound, Ischemia, foot Infection (each 0–3). Predicts amputation risk. |
| CEAP | Chronic venous disease | Clinical (C0–C6), Etiologic, Anatomic, Pathophysiologic categories. |
| Stanford | Aortic dissection | Type A (ascending) vs Type B (descending only). |
| DeBakey | Aortic dissection | Type I (all), II (ascending only), III (descending only). |
| Crawford | Thoracoabdominal aneurysm | Classically Types I–IV; some references use a modified Type V. |
| TASC II | PAD lesion morphology | Types A–D based on lesion length/location; guides endovascular vs surgical approach. |
| GLASS | Infrainguinal anatomy in CLTI | Global Limb Anatomic Staging System — newer, replaces TASC for CLTI planning. |
| Rutherford (ALI) | Acute limb ischemia severity | Categories I, IIa, IIb, III — determines salvageability. |
21 Abbreviations Master List
You will hear and use these every single day. Know them cold.
Anatomy & Vessels
Diagnoses
Procedures & Imaging
Medications & Labs
22 Physical Exam — The Vascular Exam
The vascular physical exam is the most specialized part of documentation. The surgeon performs a focused exam every visit, and you need to capture it accurately and efficiently.
Pulses are documented bilaterally and graded: 0 = absent, 1+ = diminished, 2+ = normal, 3+ = bounding. "D" = Doppler signal (audible with handheld Doppler but not palpable). A common format in vascular clinic:
Carotid: 2+/2+ no bruits bilaterally
Femoral: 2+/2+ no bruits
Popliteal: 2+/1+
DP: 2+/D
PT: 2+/D
Beyond pulses, the vascular exam includes: inspection for trophic changes (hair loss on the distal leg/foot, shiny skin, thickened nails — signs of chronic ischemia), skin color assessment (pallor on elevation, dependent rubor — the foot turns white when raised and red when hanging down, a classic sign of severe PAD), capillary refill time (> 3 seconds is abnormal), skin temperature (cool limb = reduced perfusion), edema grading and distribution, wound inspection if applicable, and assessment of surgical scars from prior procedures. For venous disease: look for varicosities (standing position), hemosiderin staining, lipodermatosclerosis (hardened, darkened skin of the lower leg), venous ulcers, and correlate the exam with duplex reflux findings.
For carotid disease: palpate carotid pulses gently (never both simultaneously), auscultate for bruits (a bruit indicates turbulent flow from stenosis, but severe stenosis may have no bruit because flow is too reduced), and perform a focused neurological exam if the patient is symptomatic (cranial nerves, motor strength, sensation, speech).
23 Risk Factors & Comorbidities
Vascular disease does not exist in isolation. Almost every vascular patient has multiple comorbidities, and documenting them thoroughly is critical because they affect surgical risk, anesthetic choice, and long-term outcomes.
Smoking: The single most important modifiable risk factor for both arterial and aneurysmal disease. Document pack-years (packs per day × years smoked) and quit date if applicable. Active smoking increases graft failure, amputation risk, and cardiovascular events. Smoking cessation counseling should be documented at every visit.
Diabetes mellitus: Document type (1 or 2), most recent A1C, current medications (insulin, metformin, sulfonylureas, GLP-1 agonists), and presence of diabetic complications (neuropathy, retinopathy, nephropathy). Diabetics have accelerated atherosclerosis, especially in the tibial vessels, and impaired wound healing.
Hypertension: Target BP < 130/80 for most vascular patients. Document current medications and any evidence of end-organ damage.
Hyperlipidemia: Document LDL level and statin use. All PAD patients should be on a high-intensity statin regardless of LDL.
Chronic kidney disease (CKD) / ESRD: Document creatinine, eGFR, dialysis status (and type of access). CKD affects contrast use (contrast-induced nephropathy risk), medication dosing, and calcification patterns.
Coronary artery disease: Document prior MI, CABG, stents, ejection fraction if known. Cardiac clearance is often needed before major vascular surgery.
Atrial fibrillation: Important for embolic risk (carotid, mesenteric, ALI), anticoagulation management.
Obesity, sedentary lifestyle, family history of aneurysm, PAD, or premature cardiovascular disease are additional factors that should be documented.
24 Sample HPI Templates
These templates show the kind of documentation expected in a vascular surgery clinic note. Use them as frameworks to internalize the rhythm and content of a good vascular HPI.
"Mr. [Name] is a 68-year-old male with a history of hypertension, hyperlipidemia, type 2 diabetes mellitus (A1C 7.8), former 40-pack-year smoker (quit 2019), and known peripheral arterial disease who presents for evaluation of worsening bilateral lower extremity claudication. He reports cramping pain in both calves that begins after approximately 2 blocks of walking and resolves within 3–5 minutes of rest. Symptoms have been progressive over the past 6 months, now limiting his ability to grocery shop independently. He denies rest pain, non-healing wounds, or tissue loss. He is compliant with aspirin 81 mg daily, atorvastatin 80 mg daily, and cilostazol 100 mg BID. He has not participated in a supervised exercise program. Prior vascular history includes right SFA stenting in 2022 with good initial result."
"Mrs. [Name] is a 72-year-old female with a known 4.2 cm infrarenal abdominal aortic aneurysm, discovered incidentally on CT scan 18 months ago, presenting for routine surveillance. She is asymptomatic — denies abdominal or back pain, syncope, or pulsatile abdominal mass. Prior measurements: 3.8 cm (March 2024), 4.0 cm (September 2024), 4.2 cm (March 2025). Growth rate approximately 0.4 cm/year. Risk factors include 30-pack-year smoking history (quit 2020) and hypertension managed with amlodipine 5 mg daily. Family history positive for AAA (father required open repair at age 70). Today's ultrasound demonstrates the aneurysm at 4.3 cm maximal diameter."
"Ms. [Name] is a 34-year-old female presenting with 3 days of progressive left lower extremity swelling, pain, and redness. She is on combined oral contraceptive pills. She took a 5-hour flight 1 week ago. She denies chest pain, shortness of breath, or hemoptysis. No prior history of DVT/PE. No family history of thrombophilia. On exam, the left lower extremity is notably edematous from the mid-thigh distally, warm, and tender along the medial thigh. Venous duplex ultrasound obtained today demonstrates acute occlusive thrombus in the left common femoral and femoral veins."
"Mr. [Name] is a 75-year-old male referred for evaluation of high-grade right carotid stenosis. He was found to have a right carotid bruit on routine physical examination by his PCP. He is completely asymptomatic — denies any episodes of transient monocular blindness, contralateral weakness, numbness, speech difficulty, or prior TIA/stroke symptoms. Carotid duplex obtained 1 week ago demonstrates right ICA stenosis of 70–79% (PSV 280 cm/sec, EDV 110 cm/sec, ICA/CCA ratio 4.2) and left ICA stenosis of < 50%. Risk factors include hypertension, hyperlipidemia, and former smoking. He is on aspirin 81 mg, atorvastatin 40 mg, and lisinopril 20 mg daily."
The best vascular scribes are not just fast typists. They understand the clinical reasoning behind the documentation. When a surgeon says "the ABI is 0.4 with a non-healing heel ulcer," a great scribe immediately knows this is CLTI (Rutherford 5), will document it appropriately, and will already have the WIfI classification framework in mind. When the surgeon says "this is a 5.6 cm infrarenal AAA with a short infrarenal neck," the scribe understands this may not be suitable for standard EVAR and anticipates the surgeon discussing FEVAR or open repair.
Be an active listener. Learn the surgeon's preferences for phrasing, note structure, and level of detail. Ask questions during downtime. Read the imaging reports and labs before the patient is seen so you can pre-populate the note. Know the medications by both generic and brand names. Anticipate the plan based on the diagnosis. This guide gives you the knowledge foundation — the expertise comes from applying it in clinic every day.
Welcome to vascular surgery. The learning curve is steep, but the specialty is deeply rewarding.