Wound Care
Every wound type, dressing, procedure, classification, and documentation pattern a wound care scribe needs on day one and beyond.
All diagrams on this page are sourced from published educational or institutional materials rather than AI generation. Each figure caption links to the original source, and the full diagram and guideline citations are collected in the references section at the bottom.
01 Skin Anatomy & Wound Healing Physiology
Wound care as a specialty sits at the intersection of dermatology, vascular surgery, plastic surgery, endocrinology, infectious disease, and rehabilitation medicine. Before a scribe can document a wound encounter intelligently, they need a working knowledge of skin architecture and the biology of healing — because every dressing, debridement, and advanced therapy is chosen to correct a specific failure in that biology.
Skin Layers
The skin has three functional layers. The epidermis is the outer avascular layer composed primarily of keratinocytes; it is responsible for the water-barrier function and is replenished from the basal layer. The dermis lies beneath and contains the vascular supply, collagen, elastin, sensory nerves, hair follicles, sebaceous glands, and sweat glands. Beneath the dermis is the subcutaneous tissue (hypodermis), a fat-rich cushioning layer that houses larger blood vessels and nerves. Wounds are described by which layers they involve: partial-thickness wounds stop within the dermis, while full-thickness wounds extend through the entire dermis into subcutaneous tissue or deeper (muscle, tendon, bone).
The Four Phases of Wound Healing
Normal wound healing moves through four overlapping phases. Hemostasis begins within seconds of injury: vasoconstriction, platelet aggregation, and fibrin clot formation stop bleeding. Inflammation dominates days 1–4, as neutrophils and then macrophages clear debris and bacteria and release cytokines and growth factors. Proliferation takes over from day 4 through roughly week 3: fibroblasts lay down collagen, new capillaries form (angiogenesis), granulation tissue fills the wound bed, and keratinocytes migrate across the surface (epithelialization). Remodeling continues for months to years, as type III collagen is replaced by stronger type I collagen and the scar matures. A healed wound only regains about 80% of the tensile strength of uninjured skin.
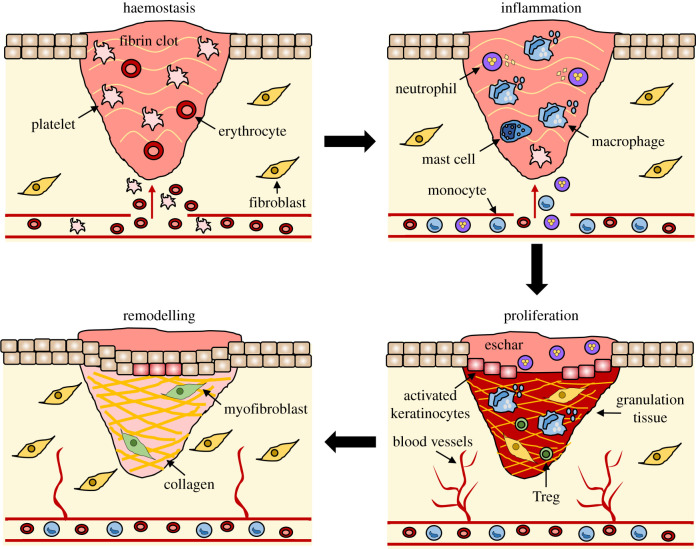
Acute vs Chronic Wounds
An acute wound is one expected to progress through these phases on a predictable timeline. A chronic wound is any wound that fails to heal in an orderly manner within an expected time frame — conventionally defined as a wound that has not substantially improved in 4 weeks or fully healed in 8–12 weeks. Chronic wounds are typically stuck in a prolonged inflammatory phase, with elevated matrix metalloproteinases (MMPs), senescent fibroblasts, heavy bacterial bioburden, and biofilm formation that actively block progression into the proliferative phase. Your job as a scribe in a wound clinic is essentially documenting the physician's effort to diagnose why a wound is stuck and to move it forward.
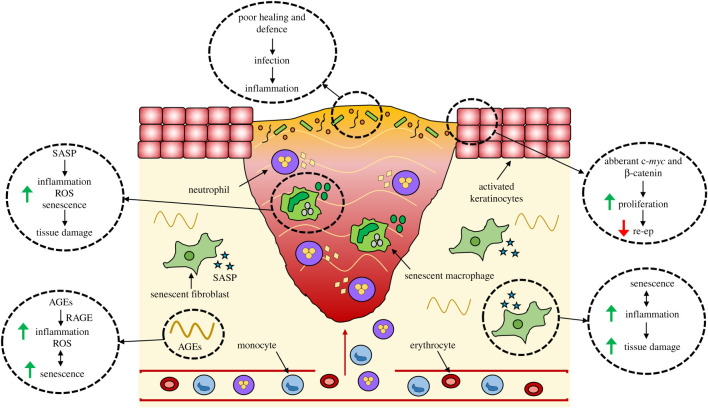
You will hear the term "stalled wound" constantly. When a wound has not decreased in size by 40–50% within 4 weeks of optimal care, guideline sources (Wound Healing Society guidelines) consider it a failure-to-heal trigger and recommend escalation to advanced therapy. Document the percent area reduction every visit if the clinician is tracking it — this is the single most important outcome measure in chronic wound care.
02 Scribe Documentation Framework
Wound clinic notes are usually SOAP-formatted, but the Objective section is unusually dense compared with most specialties because every wound requires precise measurement, description, and staging at every visit. The note must answer three questions: (1) What does this wound look like today, in measurable detail? (2) Is it better or worse than last visit? (3) What did we do about it today, and what did we instruct the patient to do at home?
CC: "non-healing foot ulcer," "wound follow-up," "new pressure injury," "dehisced abdominal incision."
HPI: Duration of the wound, mechanism (pressure, trauma, spontaneous, post-op, friction), prior treatments tried, interim dressing changes, pain score, drainage, odor, signs of infection, systemic symptoms (fever, chills), recent antibiotics, adherence to offloading or compression, nutritional status.
PMH: Diabetes (type, A1C, duration), peripheral arterial disease, venous insufficiency, immobility, incontinence, malnutrition, smoking, immunosuppression, prior amputations, renal disease, heart failure, collagen vascular disease.
Social: Home situation, caregiver support, ability to perform dressing changes, transportation to clinic, insurance coverage for supplies — all affect the realistic plan.
Location: Anatomically precise (e.g., "right lateral malleolus," "left plantar first metatarsal head," "sacrococcygeal midline"). Use laterality always.
Measurements: Length (head-to-toe) × width (side-to-side) × depth (deepest point) in centimeters. Document undermining and tunneling with clock-face orientation ("undermining 2 cm at 3 o'clock, tunneling 4 cm at 9 o'clock").
Wound bed: Percent granulation, percent slough, percent eschar. Use "100% beefy red granulation" or "50% yellow slough, 50% pink granulation" style descriptions.
Exudate: Amount (none, scant, minimal, moderate, heavy, copious) and character (serous, serosanguinous, sanguineous, purulent).
Wound edges: Attached, rolled (epibole), undermined, macerated, calloused/hyperkeratotic.
Periwound skin: Intact, erythematous, macerated, indurated, hemosiderin-stained, callused, denuded.
Odor & infection signs: Odor, local warmth, erythema > 2 cm beyond wound edge, increased pain, fluctuance, tracking.
Each wound is listed individually with its stage/classification, trajectory ("improving," "stable," "worsening," "stalled"), and the day's intervention: debridement (type, anesthesia used, tissue removed), dressing applied, offloading or compression prescribed, antibiotic decisions, imaging or lab orders, referrals, and follow-up interval. Photo documentation consent is noted for many wound practices.
Every wound clinic has a standardized wound flow sheet — either in Epic's wound LDA (Lines, Drains, Airways) module or a custom template. Learn yours on day one. Measurements, tissue composition percentages, and exudate characterization populate discrete fields, not narrative paragraphs. You must know where each element lives.
03 Wound Assessment & the TIME Framework Foundation
The TIME framework, endorsed by the Wound Healing Society and the World Union of Wound Healing Societies, is the universal mental model wound care physicians use when assessing any chronic wound. Every assessment you document should implicitly address all four domains, and most wound clinic templates are built around them.
| Letter | Domain | What to Document |
|---|---|---|
| T | Tissue (non-viable or deficient) | Percent granulation, slough, eschar, necrotic tissue. Presence of exposed muscle, tendon, bone. |
| I | Infection / Inflammation (bioburden) | Local signs of infection, biofilm suspicion, odor, purulence, surrounding cellulitis. |
| M | Moisture imbalance | Exudate volume and character; too dry (needs hydration) vs too wet (needs absorption). |
| E | Edge (non-advancing or undermined) | Rolled edges (epibole), undermining, tunneling, hyperkeratosis, maceration. |
Measuring the Wound
Standardized measurement is the only way to track healing trajectory. Length is measured head-to-toe, width is measured perpendicular (side-to-side), and depth is measured with a sterile cotton-tip applicator gently inserted into the deepest part of the wound. Undermining is tissue destruction under intact skin edges — documented as the depth and clock position (e.g., "undermining 1.5 cm from 2 to 5 o'clock"). Tunneling (also called sinus tract) is a narrow channel extending from the wound in a specific direction — documented as length and clock position.
A validated 13-item scoring system (size, depth, edges, undermining, necrotic tissue type and amount, exudate type and amount, surrounding skin color, peripheral edema, peripheral induration, granulation tissue, epithelialization). Each item is scored 1–5. Total score ranges 13 (best) to 65 (worst) and is used longitudinally to track healing. Some clinics use it at every visit, others at intake only.
A simpler NPIAP-endorsed tool that scores a wound on three items: surface area (0–10), exudate amount (0–3), and tissue type (0–4). Total score 0–17. Lower is better. Designed for pressure injuries but often used for any chronic wound.
04 Pressure Injuries Core Wound
A pressure injury (the term "pressure ulcer" was officially retired by the National Pressure Injury Advisory Panel in 2016) is localized damage to the skin and/or underlying soft tissue, usually over a bony prominence or related to a medical device, resulting from intense and/or prolonged pressure, often in combination with shear. The most common sites are the sacrum, coccyx, heels, ischial tuberosities, trochanters, and occiput. Device-related pressure injuries occur under oxygen tubing, nasogastric tubes, cervical collars, and endotracheal tube ties.
Pathophysiology
Sustained external pressure — particularly over a bony prominence — exceeds capillary closing pressure (~32 mmHg) and produces local ischemia. If pressure is not relieved, ischemia progresses to irreversible tissue necrosis. Shear forces (e.g., the sacral skin dragging against bed sheets as the head of bed is elevated) compound this by mechanically disrupting perfusion. Moisture, friction, malnutrition, immobility, incontinence, and reduced sensation all multiply the risk.
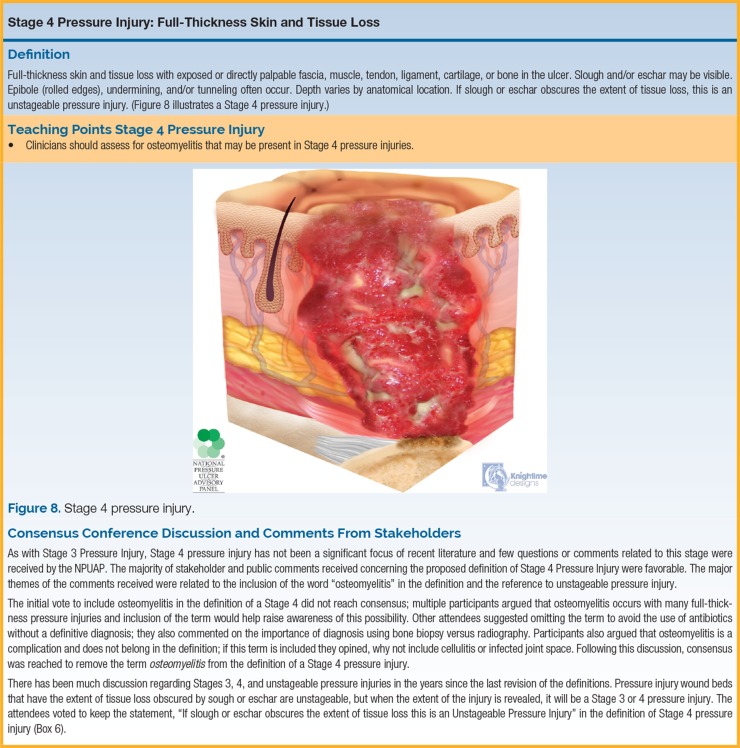
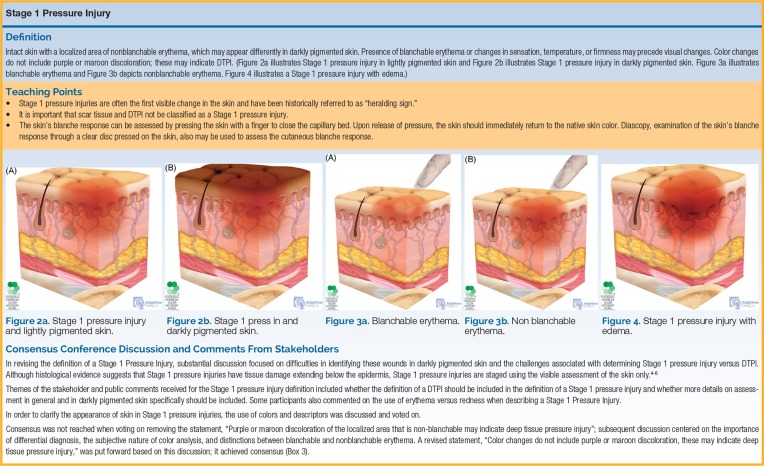
NPIAP / NPUAP Pressure Injury Staging
The 2016 NPIAP (formerly NPUAP) staging system is the universally used classification. Memorize every stage — you will chart this at every encounter.
| Stage | Description | Key Findings |
|---|---|---|
| Stage 1 | Non-blanchable erythema of intact skin | Skin intact. Localized red area that does not blanch with pressure. May be painful, firm, soft, warmer or cooler than adjacent tissue. |
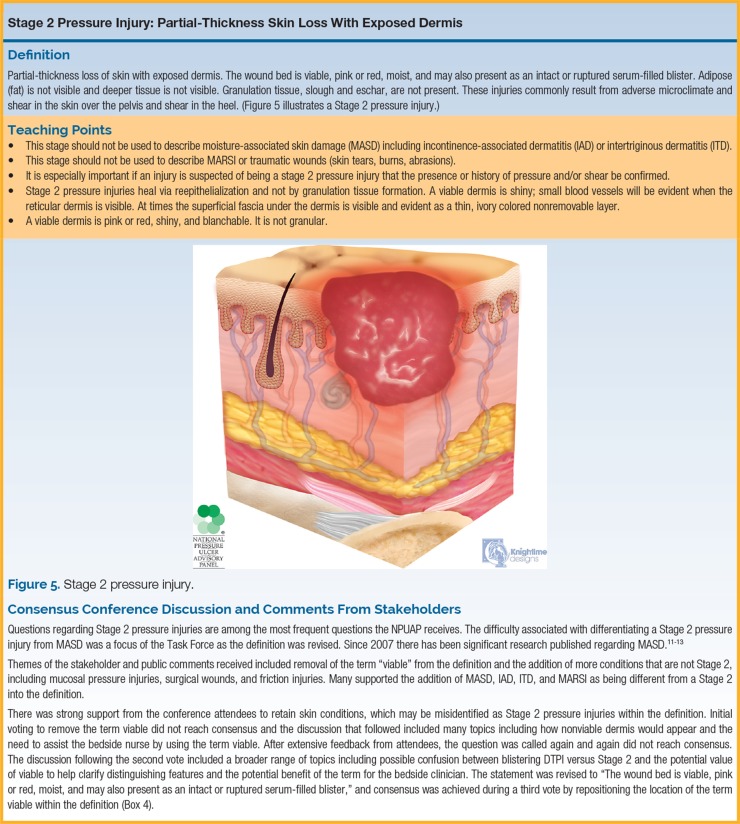
| Stage 2 | Partial-thickness skin loss with exposed dermis | Shallow open wound with a pink/red moist wound bed. May present as an intact or ruptured serum-filled blister. No slough or eschar. |
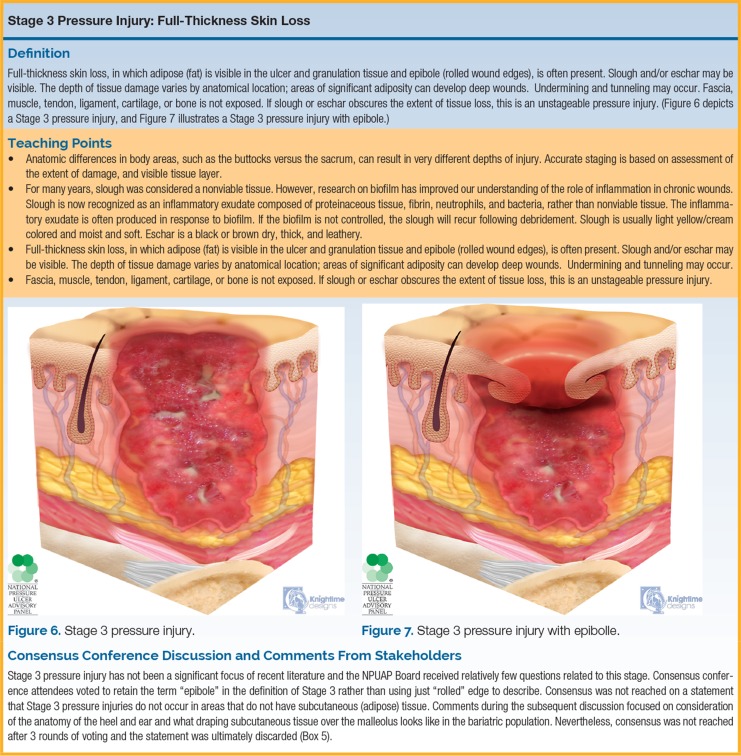
| Stage 3 | Full-thickness skin loss | Subcutaneous fat may be visible. No exposed muscle, tendon, ligament, cartilage, or bone. Slough and/or eschar may be present. May include undermining and tunneling. |
| Stage 4 | Full-thickness skin and tissue loss | Exposed muscle, tendon, ligament, cartilage, or bone. Slough, eschar, undermining, and tunneling often present. Osteomyelitis risk. |
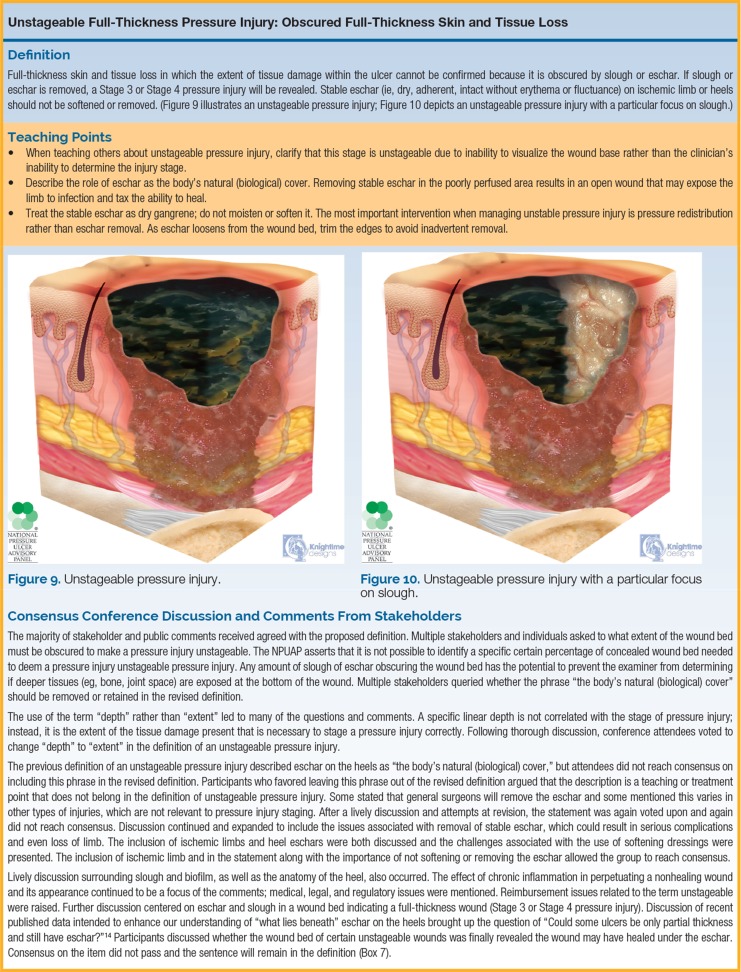
| Unstageable | Obscured full-thickness loss | Base covered by slough or eschar; depth cannot be determined until removed. By definition, it is at least Stage 3 or 4. |
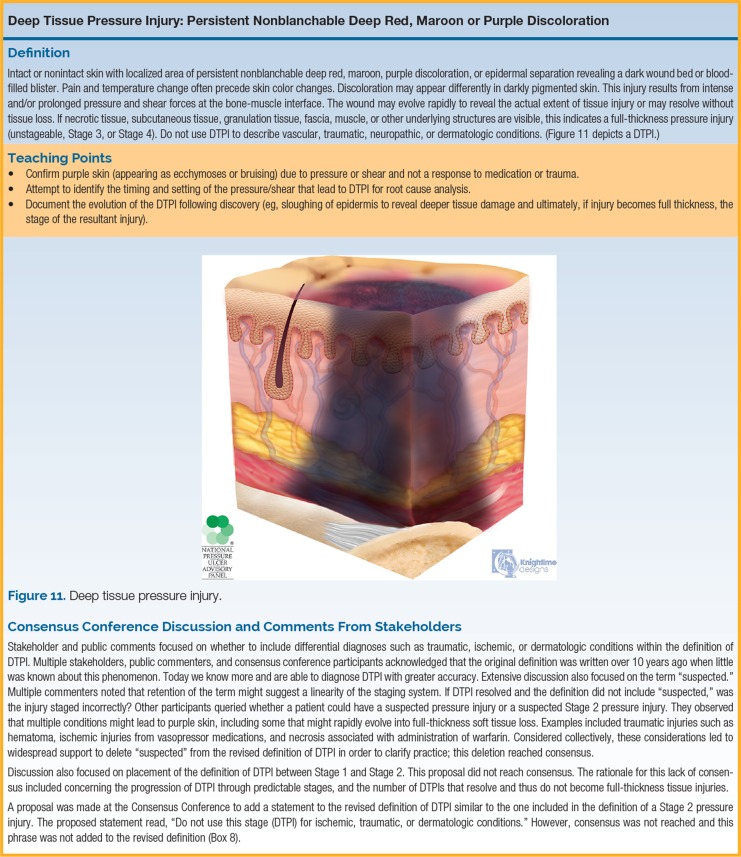
| Deep Tissue Injury (DTI) | Persistent non-blanchable deep red, maroon, or purple discoloration | Intact or non-intact skin with localized area of discoloration or blood-filled blister from damage of underlying soft tissue from pressure/shear. |
| Medical Device Related | Injury from a device used for therapy/diagnosis | Generally conforms to the shape of the device. Stage by the standard system. |
| Mucosal Membrane | Injury on mucous membrane | From a device in use at the location of the injury. Cannot be staged. |
Never "back-stage" a pressure injury. A Stage 4 that heals does not become Stage 3, 2, or 1 as it closes — it is documented as "healing Stage 4" or "closed Stage 4." The staging describes the maximum tissue damage ever observed. This is an NPIAP rule and auditors look for it.
Prevention & Management
Management follows the international NPIAP/EPUAP/PPPIA clinical practice guideline: pressure redistribution (turning every 2 hours, specialty mattresses, heel suspension), moisture management, nutrition optimization (protein 1.25–1.5 g/kg/day, vitamin C, zinc), and local wound care based on stage. Stage 3 and 4 wounds often need debridement, NPWT, and sometimes surgical flap closure. Unstageable wounds on heels with stable dry eschar are often not debrided — the eschar is left as a "biological cover" unless infection develops.
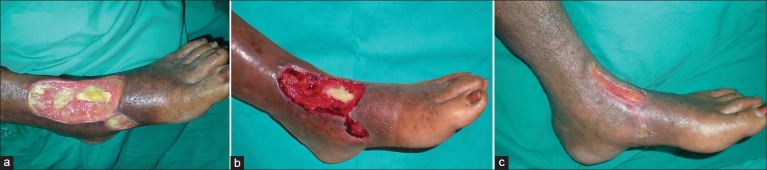
05 Diabetic Foot Ulcers (DFU) Core Wound
Diabetic foot ulcers are the single highest-volume diagnosis in most multidisciplinary wound clinics. Approximately 15–25% of patients with diabetes develop a foot ulcer in their lifetime, and DFU precedes roughly 85% of all diabetes-related lower-extremity amputations. The pathogenic triad is neuropathy, ischemia, and infection.
Pathophysiology
Peripheral neuropathy (sensory, motor, and autonomic) eliminates the protective sensation that normally prompts a person to shift weight off a painful area. The patient walks on a pressure point until the skin breaks down, then continues walking because they do not feel the wound. Motor neuropathy causes intrinsic foot muscle atrophy and toe clawing, shifting weight to the metatarsal heads. Autonomic neuropathy reduces sweating, producing dry, fissured skin. Concomitant PAD (especially tibial disease) compromises perfusion and healing. Hyperglycemia impairs neutrophil function, so infection spreads rapidly.
Clinical Presentation & Classification
DFUs are typically located at plantar pressure points (first and fifth metatarsal heads, great toe, heel) or at areas of footwear friction. Neuropathic ulcers have a surrounding callus ring; ischemic ulcers have pale, painful bases; neuro-ischemic ulcers combine both features. The two major classification systems are the Wagner system and the University of Texas (UT) system.
| Wagner Grade | Description |
|---|---|
| 0 | Pre- or post-ulcerative foot at risk. No open lesion but bony deformity, callus, or healed ulcer. |
| 1 | Superficial ulcer involving full skin thickness but no deeper tissues. |
| 2 | Deep ulcer penetrating to tendon, joint capsule, or bone without abscess or osteomyelitis. |
| 3 | Deep ulcer with abscess, osteomyelitis, or joint sepsis. |
| 4 | Localized gangrene (forefoot or heel). |
| 5 | Extensive gangrene involving the whole foot. |
The UT system is a grid with four grades (depth) and four stages (infection/ischemia modifiers). It has better prognostic validity than Wagner.
Grades (depth): 0 — pre/post-ulcerative, intact skin; I — superficial wound not involving tendon, capsule, or bone; II — wound penetrating to tendon or capsule; III — wound penetrating to bone or joint.
Stages (modifiers): A — clean (no infection, no ischemia); B — infection; C — ischemia; D — infection and ischemia. So a "UT IIIB" ulcer penetrates to bone and is infected; a "UT ID" ulcer is superficial but infected and ischemic.
Diabetic Foot Infection (DFI)
The IWGDF/IDSA 2023 guidelines define infection severity by extent of inflammation and systemic involvement. This classification drives antibiotic selection and hospitalization decisions.
| IWGDF/IDSA | Severity | Criteria |
|---|---|---|
| 1 | Uninfected | No symptoms or signs of infection. |
| 2 | Mild | Local infection (skin/subcutaneous) only, erythema ≤ 2 cm around ulcer, no systemic signs. |
| 3 | Moderate | Local infection with erythema > 2 cm OR involving deeper structures (abscess, osteomyelitis, septic arthritis, fasciitis), no SIRS. |
| 4 | Severe | Any local infection with SIRS (≥ 2 of: T > 38 or < 36, HR > 90, RR > 20, WBC > 12 or < 4). |
Management
The pillars of DFU care are offloading, debridement, infection control, vascular assessment, and glycemic control. Offloading with a total contact cast (TCC) is the gold standard for plantar neuropathic DFUs — it redistributes weight across the entire plantar surface and achieves healing rates above 80% in compliant patients. Alternatives include removable cast walkers, surgical shoes, and felted foam. Sharp debridement of callus and devitalized tissue is performed at nearly every visit. Every new DFU patient needs a vascular assessment (pulses, ABI/TBI) — if ischemia is present, revascularization precedes aggressive local wound care. Probe-to-bone (PTB) testing with a sterile blunt probe is a classic bedside test for osteomyelitis.
06 Venous Leg Ulcers (VLU) Core Wound
Venous leg ulcers account for the majority of lower extremity ulcers (~70%) and are the end-stage manifestation of chronic venous insufficiency (CVI). They are characteristically located in the gaiter area — the region between the mid-calf and the ankle, most commonly over the medial malleolus along the great saphenous vein territory.
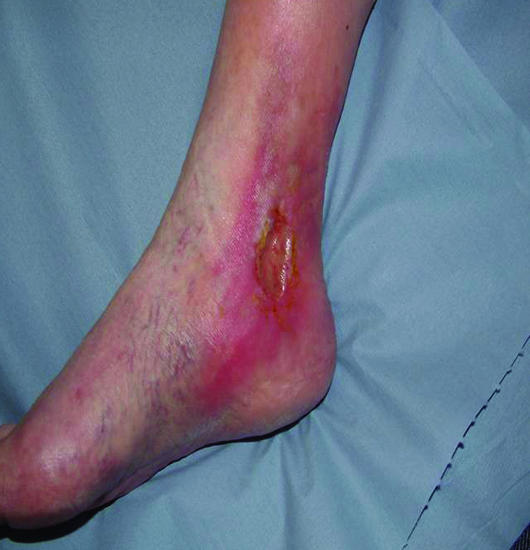
Pathophysiology
Incompetent venous valves (primary or secondary to prior DVT) allow reverse flow and pooling of blood in the lower extremity veins. Ambulatory venous hypertension transmits pressure to the dermal capillaries, causing fibrinogen leakage, perivascular fibrin cuff formation, red blood cell extravasation (producing hemosiderin deposition), leukocyte trapping, and chronic inflammation. The skin becomes hyperpigmented, thickened, and fragile (lipodermatosclerosis), and minor trauma opens a wound that cannot heal in the hypertensive venous environment.
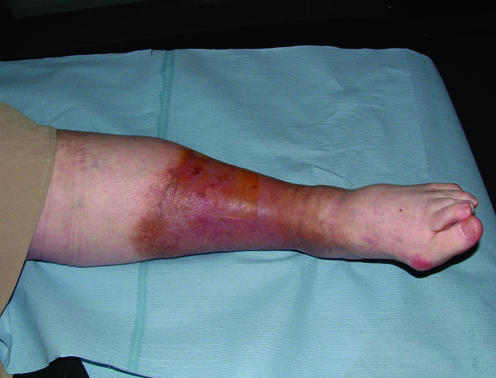
Clinical Features
VLUs are typically shallow with irregular borders, a granulating or sloughy wound bed, and moderate-to-heavy exudate. Surrounding skin shows hemosiderin staining (brown discoloration), lipodermatosclerosis ("inverted champagne bottle" leg), atrophie blanche (smooth white scarring), varicose veins, and pitting edema. Pain is typically dull, worse at the end of the day, and improved with leg elevation — the opposite of arterial pain.
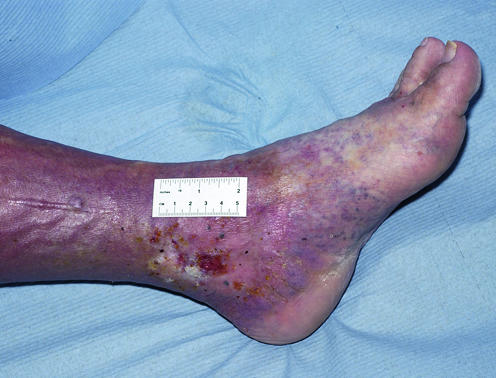
CEAP Classification
The CEAP classification (2020 revision) stages chronic venous disease clinically, etiologically, anatomically, and pathophysiologically. The clinical "C" component is what scribes document most often.
| Class | Clinical Finding |
|---|---|
| C0 | No visible or palpable signs of venous disease |
| C1 | Telangiectasias or reticular veins |
| C2 | Varicose veins (C2r = recurrent) |
| C3 | Edema |
| C4a | Pigmentation or eczema |
| C4b | Lipodermatosclerosis or atrophie blanche |
| C4c | Corona phlebectatica |
| C5 | Healed venous ulcer |
| C6 | Active venous ulcer (C6r = recurrent) |
Management
Compression therapy is the foundation. SVS/AVF CEAP-based guidelines and the ACP guideline on VLU recommend multilayer high-compression systems (30–40 mmHg at the ankle) in patients with an ABI ≥ 0.80. Options include multilayer bandaging systems (Profore, Coban 2), short-stretch bandages, Unna boots (zinc oxide paste), and compression stockings once healed. Pentoxifylline 400 mg TID is an evidence-supported oral adjunct. Treatment of underlying superficial reflux (endovenous ablation of the GSV or SSV) accelerates healing and reduces recurrence — the EVRA trial is the landmark evidence.
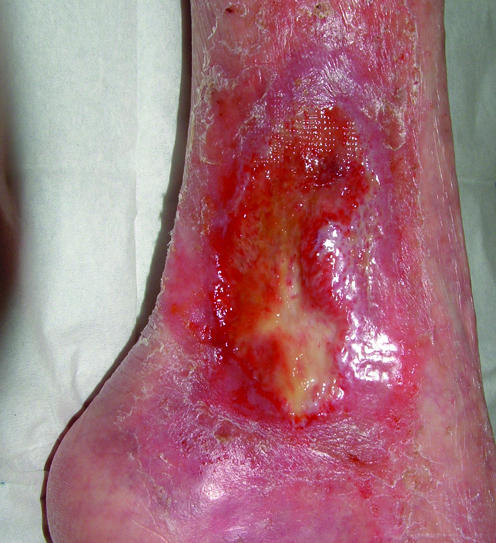
Before applying any compression, the physician will always confirm an ABI first. Compression on an ischemic leg (ABI < 0.50) can cause tissue necrosis. If the ABI is borderline (0.50–0.80), reduced compression (23–30 mmHg) may be used. Chart the ABI value in every VLU note.
07 Arterial & Mixed Arterial-Venous Ulcers Core Wound
Arterial ulcers result from inadequate arterial perfusion — almost always from atherosclerotic PAD, less commonly from thromboembolism, vasculitis, or Buerger disease. They are typically located at the most distal points of perfusion (toes, heels, lateral malleolus), are painful, dry, pale-based, and have "punched-out" well-demarcated borders. The surrounding skin is cool, shiny, hairless, with dependent rubor and elevational pallor.
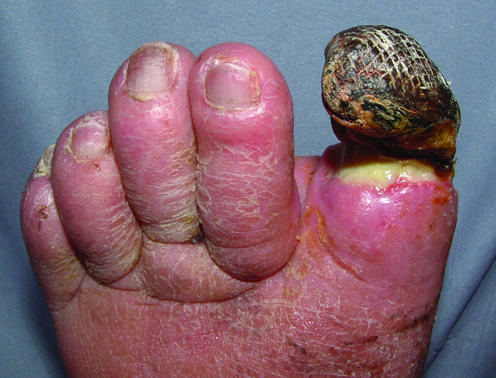
Mixed Arterial-Venous Ulcers
Approximately 15–20% of lower extremity ulcers have both arterial and venous components. These wounds present a therapeutic dilemma: compression (needed for the venous component) can worsen ischemia. An ABI of 0.50–0.80 defines the mixed category; modified compression (light, short-stretch) is used if tolerated. An ABI < 0.50 requires revascularization before compression can be safely applied.
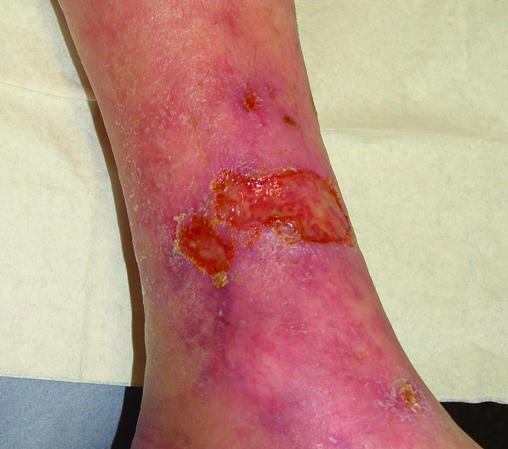
Martorell Hypertensive Ischemic Leg Ulcer
A specific arteriolosclerotic ulcer seen in patients with long-standing poorly controlled hypertension (and often diabetes). Located on the lateral or posterior calf, it is extremely painful, has a necrotic black base with surrounding livedo racemosa, and is often misdiagnosed as pyoderma gangrenosum. Management includes aggressive BP control, debridement, and often skin grafting.
Sickle Cell Leg Ulcers
Patients with sickle cell disease develop chronic, painful ulcers of the medial or lateral malleolus in late adolescence and young adulthood. Pathogenesis involves microvascular occlusion, chronic inflammation, and venous stasis. Management combines hydroxyurea, compression (if perfusion adequate), topical wound care, and sometimes transfusion therapy for non-healing wounds.
08 Surgical Wound Dehiscence Acute Wound
Dehiscence is partial or complete separation of a previously approximated surgical incision. It usually occurs between postoperative days 5 and 14. Risk factors include obesity, diabetes, malnutrition, chronic cough, steroid use, smoking, infection, hematoma, and increased intra-abdominal pressure. Superficial dehiscence involves the skin and subcutaneous tissue only; deep dehiscence extends through the fascia and is a surgical emergency because of the risk of evisceration (abdominal contents protruding through the open wound). The "pink fluid sign" — serosanguinous drainage from an otherwise well-appearing abdominal wound 5–7 days postop — is the classic warning of impending fascial dehiscence.
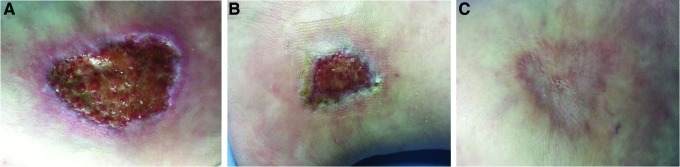
Management depends on depth. Superficial dehiscence is typically opened, irrigated, packed, and allowed to heal by secondary intention, often with NPWT. Fascial dehiscence requires urgent operative closure. Chronic non-healing surgical wounds are one of the most common indications for outpatient wound clinic referral.
09 Traumatic Wounds & Skin Tears Acute Wound
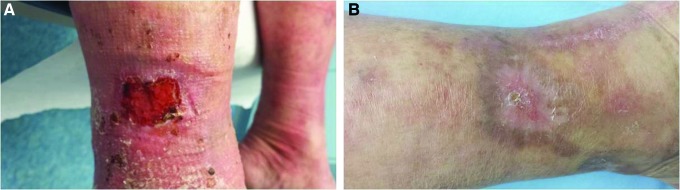
Traumatic wounds include lacerations, abrasions, avulsions, crush injuries, and bites. In the wound clinic, the most common traumatic presentation is the skin tear — a separation of skin layers usually in elderly or corticosteroid-using patients whose fragile skin shears from minor trauma (bumping into furniture, removing adhesive tape). The ISTAP classification grades skin tears:
| Type | Description |
|---|---|
| 1 | No skin loss; linear or flap tear with the flap fully approximable. |
| 2 | Partial flap loss; flap cannot be fully approximated. |
| 3 | Total flap loss exposing the entire wound bed. |
Management involves gentle cleansing, flap reapproximation if viable, non-adherent dressings (silicone contact layers, petrolatum gauze) to avoid re-tearing on removal, and arrow-labeling the dressing to show the direction of safe removal. Medical adhesive-related skin injury (MARSI) is a closely related diagnosis — skin damage from aggressive adhesive removal — prevented with silicone-based adhesives and adhesive remover wipes.
10 Burns (Acute and Chronic) Core Wound
Although acute major burns are managed at burn centers, outpatient wound clinics see small superficial burns, healed burns with contractures, and chronic burn-related wounds. Burns are classified by depth:
| Depth | Prior Name | Features |
|---|---|---|
| Superficial | First-degree | Epidermis only. Red, dry, painful. No blisters. Heals in 3–6 days without scarring. |
| Superficial partial-thickness | Second-degree (superficial) | Epidermis + superficial dermis. Red, moist, blistered, blanching, very painful. Heals in 7–21 days. |
| Deep partial-thickness | Second-degree (deep) | Epidermis + deep dermis. Pale/white or red, less blanching, decreased sensation. Heals > 21 days, usually with scarring. |
| Full-thickness | Third-degree | Entire dermis and into subcutaneous tissue. Leathery, dry, insensate. Will not heal without grafting. |
| Fourth-degree | — | Extends into muscle, tendon, or bone. |
Burn surface area is estimated by the Rule of Nines (adult): head 9%, each arm 9%, each leg 18%, anterior trunk 18%, posterior trunk 18%, perineum 1%. Topical antimicrobials historically included silver sulfadiazine (Silvadene) — though now often replaced by silver-impregnated dressings or bacitracin on the face. Chronic burn wounds may need debridement, skin grafting, or scar contracture release.
11 Atypical & Inflammatory Wounds Atypical
Any wound that fails to respond to standard care, has unusual morphology, or arises in an atypical location should raise suspicion for an atypical etiology. Biopsy is indicated when a wound does not respond to 4–6 weeks of appropriate therapy.
A neutrophilic dermatosis producing rapidly progressive, painful ulcers with violaceous undermined edges. Often associated with IBD, rheumatoid arthritis, or hematologic malignancy. Classic finding: pathergy — wounds worsen with debridement or trauma. Treatment is immunosuppression (systemic steroids, cyclosporine, biologics like infliximab), not aggressive debridement. Misdiagnosis as infection leads to repeated debridement and worsening disease.
Small-vessel vasculitis (leukocytoclastic, ANCA-associated, cryoglobulinemic) causes painful punched-out ulcers, often with surrounding palpable purpura. Workup includes ANA, ANCA, cryoglobulins, complement, hepatitis serologies, and biopsy. Treated with immunosuppression.
A life-threatening disorder of ESRD patients characterized by calcification of small dermal arterioles, producing exquisitely painful necrotic ulcers with violaceous retiform (net-like) borders. Mortality approaches 50%. Treatment includes sodium thiosulfate, strict phosphate control, wound care, pain management, and discontinuation of warfarin (which accelerates vascular calcification).
Squamous cell carcinoma arising in a chronic wound, scar, or burn (Marjolin ulcer). Any chronic wound of > 6 months that develops raised, friable, or hyperkeratotic edges needs biopsy. Basal cell carcinoma, melanoma, and cutaneous lymphoma can all masquerade as non-healing wounds.
Delayed radiation injury produces skin fibrosis, telangiectasia, atrophy, and chronic ulceration months to years after therapy. Hyperbaric oxygen therapy is the evidence-supported adjunct. Wounds heal poorly because of microvascular obliteration and fibrosis.
Autoimmune bullous disorders (pemphigus, bullous pemphigoid), factitial wounds (self-induced), infectious (atypical mycobacteria, deep fungal, cutaneous leishmaniasis), hidradenitis suppurativa, and Buerger disease (thromboangiitis obliterans) all can produce chronic wounds requiring specialist evaluation.
12 Infected Wounds, Biofilm & Osteomyelitis Infection
Every chronic wound is colonized with bacteria; not every colonized wound is infected. The distinction matters clinically and drives antibiotic decisions.
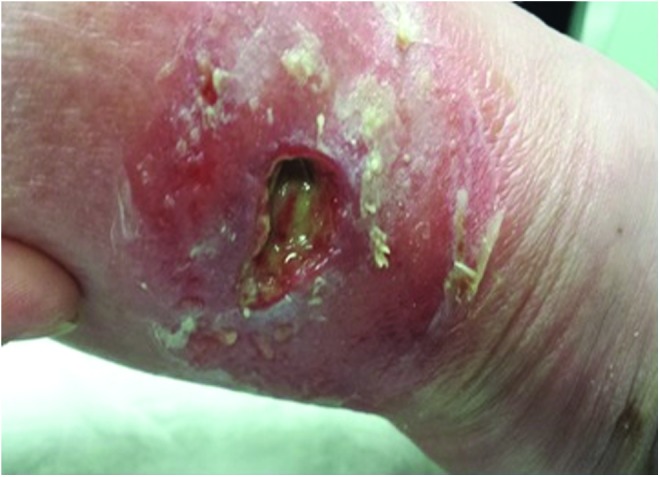
| Term | Definition |
|---|---|
| Contamination | Bacteria present, not replicating, no host response. |
| Colonization | Bacteria replicating, no host response, healing not impaired. |
| Local infection / critical colonization | Bacteria impairing healing with subtle signs (increased exudate, friable granulation, odor, delayed healing) without overt cellulitis. |
| Spreading infection | Erythema > 2 cm beyond wound edges, warmth, pain, cellulitis, lymphangitis. |
| Systemic infection | SIRS, sepsis, bacteremia, septic shock. |
Biofilm
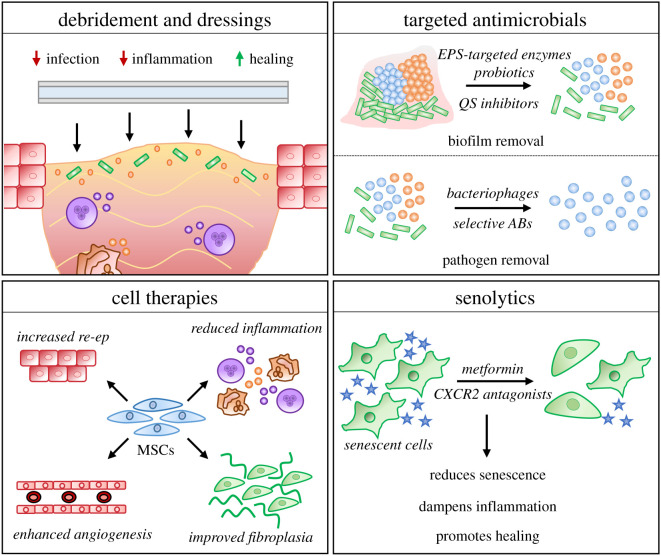
Biofilm is a structured community of bacteria embedded in a self-produced extracellular polymeric matrix that is resistant to antibiotics, antiseptics, and host immunity. Biofilm is present in the majority of chronic wounds and is a major cause of stalling. Mechanical disruption (debridement) is the single most effective way to disrupt biofilm; antimicrobial dressings (silver, iodine, PHMB, methylene blue/gentian violet) provide adjunctive control.
Osteomyelitis
When a wound extends to bone, osteomyelitis becomes the concern. Bedside probe-to-bone (PTB) testing with a sterile blunt probe has high positive predictive value in high-prevalence populations (DFU). Workup includes inflammatory markers (ESR, CRP), plain radiographs (may lag 2–3 weeks behind disease), MRI (gold standard for soft tissue and early bone involvement), and bone biopsy for definitive diagnosis and culture. Treatment combines prolonged antibiotics (typically 6 weeks IV or bioequivalent oral) and surgical debridement of infected bone.
Pain out of proportion to findings, rapidly advancing erythema, bullae, crepitus, and systemic toxicity define necrotizing fasciitis — a surgical emergency. Fournier gangrene is the perineal/scrotal variant. LRINEC score, CT findings of fascial gas, and rapid surgical consult are the workflow. Wound clinics do not manage these acutely — they transfer to the ED.
13 Moisture-Associated Skin Damage (MASD) Skin Injury
MASD is inflammation and erosion of the skin from prolonged exposure to urine, stool, sweat, wound drainage, or ostomy effluent. It is frequently mistaken for pressure injury but differs in mechanism and treatment. The four MASD subtypes:
| Type | Cause | Key Features |
|---|---|---|
| IAD | Incontinence-associated dermatitis (urine ± stool) | Diffuse erythema in perineum, buttocks, perigenital area, conforming to contact pattern. Not over a bony prominence. |
| ITD | Intertriginous dermatitis | Skin fold maceration (inframammary, pannus, groin, gluteal cleft) with mirror-image erythema. Often with candidal overgrowth. |
| Periwound MASD | Wound exudate | Macerated, white, boggy skin around a wound with heavy drainage. Interferes with epithelial migration. |
| Peristomal MASD | Ostomy effluent leak | Erythema, denudation around stoma. Requires ostomy appliance refitting. |
Management is protective: barrier creams (dimethicone, zinc oxide, petrolatum), polymer-based skin protectants (3M Cavilon), superabsorbent briefs and wicking products, and treatment of underlying candidal overgrowth with topical antifungals (nystatin, clotrimazole, miconazole powder).
The most common question on NPIAP audits is distinguishing IAD from Stage 1/2 pressure injury. Key differentiators: IAD is diffuse and not over a bony prominence; pressure injury is localized and over one. IAD has irregular, diffuse edges; pressure injury is well-demarcated. Document the distinction explicitly.
14 Lymphedema & Other Chronic Wound Settings Supporting
Lymphedema — protein-rich interstitial edema from impaired lymphatic drainage — is a major contributor to non-healing lower extremity wounds. Primary lymphedema is congenital; secondary lymphedema follows lymph node dissection, radiation, filariasis, recurrent cellulitis, or chronic venous insufficiency (phlebolymphedema). Skin changes include Stemmer sign (inability to pinch the skin at the base of the second toe), peau d'orange, papillomatosis, and lichenification. Management is complete decongestive therapy (CDT): manual lymphatic drainage, multilayer short-stretch compression bandaging, exercise, and meticulous skin care, followed by long-term compression garments or pneumatic compression devices.
Fistulas and Sinus Tracts
A fistula is an abnormal communication between two epithelialized surfaces (enterocutaneous, rectovaginal, urocutaneous). A sinus tract is a blind-ended tract leading from the skin to a deep cavity. Both produce complex wound management challenges — output containment (fistula bags, NPWT with fistula isolation), skin protection, and definitive surgical closure when possible.
15 Debridement Techniques Procedure
Debridement — the removal of non-viable tissue, debris, biofilm, and senescent cells — is the single most important wound clinic procedure. It converts a chronic wound into an acute-appearing wound and restores the proliferative phase. Debridement is billed by technique, depth, and surface area.
| Technique | Description | Indications / Notes |
|---|---|---|
| Sharp (bedside) | Selective removal of devitalized tissue with scalpel, curette, or scissors at the bedside | Most common. Performed by trained clinicians. Topical lidocaine often adequate. |
| Surgical (OR) | Excisional debridement under anesthesia, often extending into viable tissue | Extensive wounds, necrotizing infection, osteomyelitis, bone involvement. |
| Enzymatic | Topical application of collagenase (Santyl) to digest denatured collagen | Selective; suitable for patients who cannot tolerate sharp debridement. Slower (days to weeks). |
| Autolytic | Use of occlusive or semi-occlusive dressings (hydrogels, hydrocolloids) that harness endogenous enzymes in wound fluid | Slowest but painless and selective. Not for infected wounds. |
| Mechanical | Wet-to-dry gauze, pulsed lavage, monofilament pads (Debrisoft), ultrasonic debridement | Non-selective; wet-to-dry is outdated and discouraged because it removes granulation tissue with eschar. |
| Biological (maggot) | Sterile medical-grade Lucilia sericata larvae selectively digest necrotic tissue | FDA-cleared. Option for patients who cannot have sharp debridement. |
| Ultrasonic | Low-frequency contact or non-contact ultrasound (MIST, SonicOne) | Disrupts biofilm, stimulates healing, minimally painful. |
| Hydrosurgical | High-pressure saline jet cutting (Versajet) | Used in OR. Precise debridement of burns and complex wounds. |
Debridement billing requires precise depth documentation: CPT 11042 (skin/subcutaneous), 11043 (muscle/fascia), 11044 (bone). You must document which tissue layer was debrided and the surface area in square centimeters. Missing either element leads to denied claims.
16 Negative Pressure Wound Therapy (NPWT) Procedure
Negative pressure wound therapy (NPWT; also called vacuum-assisted closure, "wound vac," KCI V.A.C., PICO) applies sub-atmospheric pressure to a sealed wound via an open-pore foam or gauze filler connected to a canister through tubing. Typical settings are −125 mmHg continuous or intermittent, though lower pressures (−75 mmHg) are used on fragile tissue.
Mechanism
NPWT works by (1) removing interstitial fluid and reducing edema, (2) increasing local perfusion, (3) applying mechanical forces (macro- and microstrain) that stimulate granulation tissue, (4) containing exudate, and (5) approximating wound edges. Dressings are changed every 2–3 days (3×/week is standard).
Indications and Contraindications
Indications: Chronic non-healing wounds, surgical dehiscence, traumatic wounds, flaps and grafts (to bolster take), pressure injuries Stage 3–4, diabetic foot ulcers, open abdomen, sternotomy dehiscence.
Contraindications: Untreated osteomyelitis, malignancy in the wound, exposed unprotected blood vessels or organs, necrotic tissue with eschar, enteric fistulas that are not isolated.
NPWT with Instillation (NPWTi-d)
NPWT with instillation and dwell time (V.A.C. VeraFlo) periodically instills a topical solution (typically normal saline or a wound cleanser like Prontosan) into the wound, allows it to dwell 10–20 minutes, then resumes negative pressure. Indicated for heavily colonized or infected wounds not amenable to immediate closure.
17 Advanced Therapies & Skin Substitutes Procedure
When a wound has failed 4–6 weeks of standard care (debridement, offloading/compression, moisture balance, infection control), advanced therapies are considered. These are generally billed under Medicare as cellular and/or tissue-based products (CTPs) or bioengineered cellular and tissue products (BCTPs).
| Product | Type | Composition & Use |
|---|---|---|
| Apligraf | Bilayered living skin substitute | Neonatal keratinocytes and fibroblasts on bovine collagen. VLU, DFU. |
| Dermagraft | Living dermal substitute | Neonatal fibroblasts on bioabsorbable mesh. DFU. |
| Integra | Bilayer acellular dermal matrix | Bovine collagen/chondroitin with silicone outer layer. Burns, complex reconstruction. |
| Oasis | Acellular xenograft | Porcine small intestine submucosa. Venous ulcers, DFU, partial-thickness wounds. |
| EpiFix / Grafix | Amniotic membrane allograft | Human placental tissue with growth factors. DFU, VLU, complex wounds. |
| AlloDerm | Acellular dermal allograft | Cadaveric human dermis. Reconstructive use. |
| Biobrane | Biosynthetic dressing | Porcine collagen on nylon mesh. Superficial burns, donor sites. |
Hyperbaric Oxygen Therapy (HBOT)
HBOT delivers 100% oxygen at > 1 atmosphere (typically 2.0–2.4 ATA) in a chamber. Indications accepted by the Undersea & Hyperbaric Medical Society include Wagner grade 3 or higher DFU that has failed 30 days of standard care, delayed radiation injury, compromised flaps/grafts, refractory osteomyelitis, and necrotizing soft tissue infection. Typical protocol: 2 hours daily, 5 days/week, 20–40 sessions.
Other Advanced Modalities
Growth factors: Becaplermin (Regranex) — topical recombinant PDGF-BB for neuropathic DFUs. Carries a boxed warning for malignancy with heavy use.
Platelet-rich plasma (PRP): Autologous concentrated platelets applied topically or injected into wound margins.
Skin grafting: Split-thickness skin grafts (STSG) are the workhorse for large granulating wounds; punch grafts are used for small VLUs; cultured epithelial autografts (Epicel) for extensive burns.
Flap reconstruction: Local, regional, or free tissue transfer for wounds with exposed critical structures or that fail all simpler closures (managed by plastic surgery).
18 Offloading & Compression Therapy Procedure
Offloading (DFU & Pressure Injury)
| Device | Use | Notes |
|---|---|---|
| Total contact cast (TCC) | Gold standard for plantar neuropathic DFU | Non-removable. Changed weekly. Contraindicated with active infection or significant ischemia. |
| Removable cast walker (RCW) | Alternative to TCC when non-removable is not feasible | Can be made "instant TCC" by wrapping with fiberglass or cohesive bandage. |
| CROW boot | Charcot Restraint Orthotic Walker for Charcot foot | Custom molded ankle-foot orthosis. |
| Surgical/post-op shoe | Minor offloading, postoperative | Limited offloading efficacy alone. |
| Felted foam, half-shoes | Forefoot offloading | Simple, inexpensive. |
| Heel suspension boots | Pressure relief for heel ulcers in bedbound patients | Prevasore, Heelift, Waffle boot. |
| Specialty mattress | Whole-body pressure redistribution | Low-air-loss, alternating pressure, air-fluidized. |
Compression (VLU & Lymphedema)
| Product | Class | Notes |
|---|---|---|
| Profore, Coban 2, Dyna-Flex | Multilayer high compression | 30–40 mmHg. Stays on 3–7 days. VLU mainstay. |
| Unna boot | Zinc oxide paste bandage | Inelastic. Changed weekly. Inexpensive. |
| Short-stretch bandages | Low-resting, high-working pressure | Preferred in lymphedema CDT. |
| Compression stockings | Maintenance therapy after healing | Class I (15–20), II (20–30), III (30–40), IV (> 40 mmHg). |
| Pneumatic compression pumps | Intermittent sequential compression | Lymphedema, severe CVI, refractory wounds. |
| Velcro wrap devices | CircAid, FarrowWrap, ReadyWrap | Adjustable compression for self-management. |
19 Dressings — A to Z Reference
The dressing decision is driven by the wound bed (dry vs wet), depth, infection status, and patient factors. The universal principle, articulated by Winter in 1962 and endorsed by every modern guideline, is that a moist wound bed heals faster than a dry one. Match the dressing to the wound, not the other way around.
| Category | Examples | Use Case |
|---|---|---|
| Gauze (woven/non-woven) | Kerlix, 4×4s, Xeroform | Packing cavities, secondary dressing, wet-to-moist (not wet-to-dry). |
| Transparent film | Tegaderm, OpSite | Superficial wounds, IV site covers, secondary dressing. Retains moisture. |
| Hydrocolloid | DuoDERM, Comfeel, Tegasorb | Minimally exudative wounds. Autolytic debridement. Stage 2 pressure injuries. Not for infection. |
| Hydrogel | Solosite, IntraSite, Curasol | Dry wounds needing hydration. Rehydrates eschar for autolytic debridement. |
| Foam | Mepilex, Allevyn, Optifoam, Biatain | Moderate to heavy exudate. Stage 2–4 pressure injuries. Comfortable under compression. |
| Bordered foam (silicone) | Mepilex Border, Allevyn Life | Atraumatic, self-adherent. Sacral shape common for pressure-injury prevention. |
| Alginate | Kaltostat, Algisite, SeaSorb | Heavy exudate, tunneling/undermining. Gels on contact. Hemostatic. |
| Hydrofiber | Aquacel, Aquacel Ag | Heavy exudate. Gels on contact. Often used under compression. |
| Superabsorbent | Zetuvit Plus, Mextra | Very heavy exudate. Locks fluid away from periwound skin. |
| Silver antimicrobial | Acticoat, Aquacel Ag, Mepilex Ag, Silvercel | Critical colonization or local infection. Broad-spectrum including MRSA. |
| Iodine antimicrobial | Iodosorb, Inadine | Broad spectrum. Also debrides slough (cadexomer iodine). |
| Honey (medical-grade) | Medihoney | Antimicrobial, autolytic debridement. Manuka honey most studied. |
| PHMB / biguanide | Kerlix AMD, XCell | Antimicrobial without cytotoxicity concerns. |
| Methylene blue / gentian violet | Hydrofera Blue | Antimicrobial foam, bacteriostatic. |
| Collagen | Promogran, Stimulen, Puracol | Stalled wounds. Binds MMPs, provides scaffold. |
| Contact layer | Mepitel, Adaptic, Conformant | Non-adherent protective layer over fragile wounds. |
| Composite dressings | Combiderm, Covaderm | All-in-one absorbent + adherent border. |
20 Imaging, Labs & Diagnostics Diagnostics
Vascular Assessment
Every lower extremity wound needs a vascular assessment. Pulses (DP, PT), ankle-brachial index (ABI), toe-brachial index (TBI) (for diabetics/CKD with non-compressible vessels), transcutaneous oxygen measurement (TcPO2) (a TcPO2 < 40 mmHg predicts poor healing; > 40 mmHg is favorable), skin perfusion pressure (SPP), and duplex ultrasound for venous reflux or arterial stenosis mapping.

Imaging
| Modality | When Used |
|---|---|
| Plain radiograph | First-line for suspected osteomyelitis, foreign body, gas in soft tissue, Charcot changes. Findings lag 2–3 weeks. |
| MRI | Gold standard for osteomyelitis and soft tissue abscess. Distinguishes cellulitis from deeper infection. |
| CT | Deep abscess localization, gas in soft tissue (necrotizing infection), surgical planning. |
| Bone scan (triple phase) | Alternative to MRI when MRI contraindicated; less specific. |
| Tagged WBC scan | Differentiates infection from Charcot in equivocal cases. |
| Venous duplex | Confirms superficial and deep reflux, maps incompetent perforators. |
| Arterial duplex / CTA / MRA | Evaluates PAD prior to revascularization. |
Labs
CBC with differential, ESR, CRP, procalcitonin (sepsis), comprehensive metabolic panel (glucose, renal, liver), A1C, albumin and prealbumin (nutritional markers), vitamin D, zinc, vitamin C (healing cofactors), wound or deep tissue cultures (swab cultures of the wound surface are of limited value; tissue biopsy is preferred), and bone biopsy with culture for osteomyelitis.
21 Medications You Must Know Reference
Systemic Antibiotics
| Agent | Class | Use Case |
|---|---|---|
| Cephalexin (Keflex) | First-gen cephalosporin | Mild cellulitis (non-MRSA coverage). |
| Dicloxacillin | Antistaph penicillin | Mild cellulitis (MSSA). |
| TMP-SMX (Bactrim) | Sulfonamide | MRSA-suspected mild cellulitis; oral MRSA coverage. |
| Doxycycline | Tetracycline | MRSA coverage, bite wounds (with amox/clav). |
| Clindamycin | Lincosamide | MRSA (if susceptible), penicillin allergy, toxin suppression in NSTI. |
| Amoxicillin-clavulanate (Augmentin) | Aminopenicillin + BL inhibitor | Mild/moderate DFI; bite wounds. |
| Ampicillin-sulbactam (Unasyn) | Aminopenicillin + BL inhibitor | IV moderate DFI. |
| Piperacillin-tazobactam (Zosyn) | Extended-spectrum PCN | Moderate/severe DFI, hospital-acquired wound infection. |
| Ertapenem | Carbapenem | Moderate DFI, ESBL coverage. |
| Meropenem / Imipenem | Carbapenem | Severe DFI, necrotizing infection. |
| Vancomycin | Glycopeptide | IV MRSA coverage for moderate/severe infection, osteomyelitis. |
| Linezolid | Oxazolidinone | MRSA, VRE; oral bioequivalent. |
| Daptomycin | Lipopeptide | MRSA, osteomyelitis (not for pneumonia). |
| Ceftaroline | 5th-gen cephalosporin | MRSA with gram-negative coverage. |
| Ciprofloxacin / Levofloxacin | Fluoroquinolones | Gram-negative and Pseudomonas coverage; bone penetration. |
| Metronidazole | Nitroimidazole | Anaerobic coverage; added to quinolone or cephalosporin. |
Topical Antimicrobials
| Agent | Notes |
|---|---|
| Silver sulfadiazine (Silvadene) | Historical burn standard. Do not use on the face or in sulfa allergy. |
| Mupirocin (Bactroban) | MSSA/MRSA decolonization, small superficial wounds. |
| Bacitracin / Polymyxin B | Broad OTC. Avoid on large wounds (contact dermatitis, nephrotoxicity risk for polymyxin). |
| Mafenide acetate (Sulfamylon) | Burn eschar penetration; causes metabolic acidosis. |
| Nystatin / clotrimazole / miconazole | Candidal intertrigo, peristomal candidiasis. |
| Collagenase (Santyl) | Enzymatic debridement. |
| Becaplermin (Regranex) | Topical PDGF for DFU — boxed warning for malignancy. |
Supportive and Adjunctive Medications
| Agent | Use |
|---|---|
| Pentoxifylline | 400 mg TID for venous leg ulcer; rheologic agent, reduces blood viscosity. |
| Aspirin / clopidogrel | Antiplatelet therapy in PAD/DFU. |
| Statin | Atherosclerotic risk reduction for PAD/DFU. |
| Gabapentin / pregabalin | Neuropathic pain in diabetic neuropathy, painful ulcers. |
| Acetaminophen, NSAIDs, opioids | Analgesia. Avoid NSAIDs in CKD and in pyoderma gangrenosum active phase. |
| Vitamin C 500–1000 mg/day | Collagen synthesis cofactor. |
| Zinc sulfate 220 mg/day | Supplementation if deficient; not routine. |
| Protein supplementation (1.25–1.5 g/kg/day) | Wound healing nutrition. |
| Sodium thiosulfate | Calciphylaxis. |
| Systemic steroids / cyclosporine / infliximab | Pyoderma gangrenosum, vasculitis. |
| Prostanoids (iloprost) | Critical limb ischemia not amenable to revascularization (limited US availability). |
22 Classification Systems Reference
This section consolidates every grading and classification system referenced throughout the guide. Fully enumerated scales appear in their condition sections; this is a quick-reference map.

| System | What It Classifies | Scale |
|---|---|---|
| NPIAP / NPUAP | Pressure injury staging | Stage 1, 2, 3, 4, Unstageable, DTI, Medical Device, Mucosal |
| Wagner | DFU depth + gangrene | Grades 0–5 |
| University of Texas | DFU depth + infection/ischemia | Grades 0–III, Stages A–D (grid) |
| IWGDF/IDSA DFI | Diabetic foot infection severity | 1 (uninfected) to 4 (severe with SIRS) |
| CEAP | Chronic venous disease | C0–C6 clinical; E/A/P modifiers |
| WIfI | Limb threat in CLTI | Wound 0–3, Ischemia 0–3, Foot infection 0–3 |
| ISTAP | Skin tear | Types 1–3 |
| Burn depth | Burn severity | Superficial → Superficial partial → Deep partial → Full-thickness → Fourth-degree |
| Rule of Nines | Burn surface area | 9% head, 9% per arm, 18% per leg, 18% ant trunk, 18% post trunk, 1% perineum |
| PUSH tool | Pressure injury healing trajectory | Size + exudate + tissue; 0–17 |
| BWAT (Bates-Jensen) | Wound healing trajectory | 13 items; 13 (best) – 65 (worst) |
| Armstrong & Harkless | DFU depth/ischemia/infection (synonym for UT grid) | Grid 0–III / A–D |
| LRINEC | Necrotizing fasciitis risk score | CRP, WBC, Hb, Na, Cr, glucose; ≥ 6 suspicious, ≥ 8 high risk |
23 Physical Exam — The Wound Exam Reference
The wound-focused physical exam is performed at every visit and its findings populate discrete fields in the wound flow sheet. It is the most structured exam in clinical practice — essentially a checklist.
Location: [precise anatomic site, laterality]
Etiology: [pressure / venous / arterial / neuropathic / traumatic / surgical / atypical]
Duration: [weeks/months/years since onset]
Dimensions: [L × W × D cm]
Undermining: [depth at clock position]
Tunneling: [length at clock position]
Wound bed: [% granulation / % slough / % eschar]
Exudate: [amount & character]
Odor: [none / mild / strong]
Edges: [attached / rolled / macerated / calloused]
Periwound: [intact / erythematous / macerated / hemosiderin / indurated]
Pain: [0–10 scale]
Stage/Grade: [per applicable system]
Signs of infection: [absent / local / spreading / systemic]
Pulses: [DP/PT grading]
Sensation: [monofilament / vibration]
Beyond the wound itself, the lower extremity exam includes pulse grading, monofilament testing (10-g Semmes-Weinstein at 10 plantar sites for diabetic neuropathy screening), vibratory testing (128 Hz tuning fork), capillary refill, skin temperature, and Buerger test (elevational pallor and dependent rubor). For venous disease, document varicosities, hemosiderin staining, lipodermatosclerosis, atrophie blanche, and edema grading (1+ to 4+).
24 Abbreviations Master List Reference
Wound Types & Assessment
Procedures & Therapies
Vascular & Diagnostics
Guideline Bodies
25 Sample HPI Templates Reference
These templates demonstrate the rhythm and content of a strong wound clinic HPI. Pattern-match off them during your first weeks.
"Mr. [Name] is a 62-year-old male with type 2 diabetes mellitus (A1C 9.4), diabetic peripheral neuropathy, hypertension, and former 30-pack-year smoking history who presents for evaluation of a new right foot ulcer, present for approximately 3 weeks. He first noticed redness under the right first metatarsal head after wearing new work boots. The area progressed to an open ulcer which he has been covering with gauze and tape. He denies fever, chills, or systemic symptoms. He reports no pain (consistent with neuropathy). On exam there is a 1.8 × 1.5 × 0.4 cm plantar ulcer at the right first metatarsal head with surrounding hyperkeratotic callus, no undermining, 70% pink granulation and 30% yellow slough, scant serous drainage, no erythema or tracking, no foul odor. Probe-to-bone negative. DP and PT pulses palpable bilaterally. Monofilament absent at 8 of 10 plantar sites. Classified today as Wagner 1 / UT IA. Plan: sharp debridement of callus and slough, collagenase topical therapy, offloading with total contact cast, A1C optimization, and return in 1 week."
"Ms. [Name] is a 78-year-old bedbound female with advanced dementia, stroke with right hemiparesis, urinary incontinence, and cachexia (BMI 17.2, albumin 2.6) who is referred from skilled nursing facility for evaluation of a sacral pressure injury first noted 5 weeks ago. The wound has progressively enlarged despite twice-daily dressing changes with hydrocolloid. On exam there is a 6.2 × 5.0 × 2.8 cm sacrococcygeal wound with exposed gluteal fascia and a small area of visible bone at the wound base. Undermining 2 cm from 11 to 2 o'clock. Wound bed 60% yellow slough, 30% pink granulation, 10% black eschar at the inferior edge. Moderate serosanguinous exudate, mild foul odor. Periwound skin erythematous and macerated. Staged today as NPIAP Stage 4. Plan: sharp bedside debridement of slough and eschar, collagenase plus foam dressing, initiate NPWT at −125 mmHg continuous, nutrition consult for protein and vitamin C/zinc supplementation, low-air-loss mattress, q2-hour turning schedule at SNF. Will obtain MRI to evaluate for osteomyelitis."
"Mrs. [Name] is a 65-year-old female with obesity (BMI 34), chronic venous insufficiency, varicose veins, and remote history of left DVT following pregnancy who presents for follow-up of a left medial malleolar ulcer present for 4 months. The ulcer began spontaneously after minor trauma and has gradually enlarged. She reports heaviness and aching worse at the end of the day, improved with elevation. On exam there is a 3.4 × 2.6 × 0.3 cm shallow irregular ulcer over the left medial malleolus with 80% pink granulation and 20% yellow slough, moderate serous exudate, mild maceration of the periwound skin. Surrounding gaiter-area skin shows hemosiderin staining, lipodermatosclerosis, and 2+ pitting edema to the knee. DP and PT pulses 2+. ABI today 1.08 on the left. CEAP class C6. Plan: sharp debridement of slough, foam dressing under multilayer compression bandaging (Profore) with weekly changes, initiate pentoxifylline 400 mg TID, venous duplex to evaluate superficial reflux, referral to vascular for consideration of endovenous ablation."
"Mr. [Name] is a 58-year-old male 18 days status post open right hemicolectomy for colon cancer, complicated by superficial midline incision dehiscence noted by his surgeon at postoperative day 10. He presents for wound clinic care. He denies fever, chills, or abdominal pain. On exam the midline abdominal incision has a 5.5 × 1.8 × 2.0 cm open area at the inferior third with 50% pink granulation and 50% yellow slough, moderate serosanguinous drainage, no purulence, no fluctuance, fascia intact on digital exam. Periwound erythema minimal. No systemic signs of infection. Plan: sharp debridement of slough, initiate NPWT with black foam at −125 mmHg continuous, dressing changes 3×/week in clinic, continue oral antibiotic prophylaxis, nutrition optimization, return weekly."
"Ms. [Name] is a 46-year-old female with ulcerative colitis on mesalamine who presents with a rapidly progressive painful left lower extremity ulcer first appearing 3 weeks ago after a minor bump. The wound has enlarged despite oral cephalexin and local wound care at urgent care. She rates the pain 9/10. On exam there is a 4.2 × 3.8 × 0.6 cm ulcer over the left anterior tibia with violaceous undermined borders, surrounding erythema, and a boggy wound bed. DP and PT pulses 2+, ABI 1.10. No signs of systemic infection. Given the rapid progression, classic violaceous undermined borders, IBD association, and failure of antibiotic therapy, pyoderma gangrenosum is strongly suspected. Plan: defer aggressive debridement (pathergy risk), punch biopsy of wound edge to exclude vasculitis and infection, start systemic prednisone 40 mg daily, gentle silicone contact-layer dressing, pain control with gabapentin, gastroenterology and rheumatology co-management."
"Mr. [Name] is a 71-year-old male with type 2 diabetes, CKD stage 3, and PAD status post right SFA stenting who has a chronic right lateral foot wound present for 14 weeks, status post transmetatarsal amputation 8 weeks ago with incomplete healing of the lateral incision. Despite weekly sharp debridement, collagenase, and foam dressings, the wound has stalled with < 20% area reduction over 4 weeks. On exam there is a 3.0 × 2.2 × 1.2 cm wound at the lateral TMA site with 70% pink granulation and 30% slough, moderate serous drainage, no tunneling, no exposed bone. Recent ABI 0.82, TcPO2 48 mmHg. Plan: initiate NPWT with black foam at −125 mmHg continuous, dressing changes 3×/week, continue offloading with post-op shoe, re-evaluate for advanced therapy (skin substitute) in 2 weeks if no response, optimize glycemic control, continue aspirin and statin."
26 References & Sources
Clinical Practice Guidelines
Qaseem A et al. Treatment of pressure ulcers: ACP clinical practice guideline. Ann Intern Med. 2015.
Landmark Trials & Evidence
Diagram & Figure Sources
Figure 1: Structure of the skin. OpenStax College. CC BY 3.0.
Figure 2: Phases of wound healing. Wikimedia Commons. Public domain.
Figure 3: Pressure ulcer staging. Blausen Medical. CC BY 3.0.
Figure 4: Sacral pressure injury. Wikimedia Commons. Public domain.
Figure 5: Neuropathic diabetic foot ulcer. Wikimedia Commons. Public domain.
Figure 6: Venous leg ulcer. Wikimedia Commons. Public domain.
Figure 7: Negative pressure wound therapy. Wikimedia Commons. Public domain.
The best wound care scribes think like a wound care physician. When the doctor says "this is a stalled Stage 3 sacral pressure injury with biofilm and heavy exudate," a great scribe already knows the plan will involve sharp debridement, an antimicrobial dressing with high absorbency, and likely NPWT — and has started pre-populating the note. When the surgeon says "UT grade IIB with probe-to-bone positive," you know an MRI is coming and that antibiotics and possibly surgical debridement will follow.
Master the measurements. Memorize the staging systems cold. Know every dressing by its category, not just its brand name. Understand why offloading a DFU matters and why compression is the foundation of VLU care. Learn to distinguish pressure injury from IAD on sight. Above all, be precise: wound care documentation drives billing, legal defensibility, and payer audits more than in any other specialty. A sloppy note can cost the clinic thousands of dollars or invite a survey citation.
Welcome to wound care. It is detail-obsessed, visually oriented, deeply multidisciplinary work — and a well-trained scribe makes the clinic run.