Bariatric / Metabolic Surgery
Every diagnosis, procedure, classification, complication, medication, metabolic outcome, nutritional guideline, and management algorithm across the full scope of bariatric and metabolic surgery in one place.
01 GI Anatomy & Hormonal Physiology
Gastric Anatomy
The stomach is a J-shaped organ divided into four regions: the cardia (immediately distal to the GEJ), the fundus (dome-shaped area superior to the cardia, primary reservoir for ghrelin-secreting cells), the body/corpus (largest portion, contains parietal cells producing HCl and intrinsic factor, and chief cells producing pepsinogen), and the antrum/pylorus (thicker muscularis, G cells produce gastrin). The stomach wall consists of four layers: mucosa, submucosa, muscularis propria (three layers — inner oblique, middle circular, outer longitudinal), and serosa.
The blood supply derives from the celiac trunk: the left gastric artery (largest contributor to the lesser curvature, must be preserved or carefully managed in all bariatric procedures), right gastric artery (from the proper hepatic artery), left gastroepiploic artery (from splenic artery), right gastroepiploic artery (from gastroduodenal artery — the primary blood supply to the gastric pouch in RYGB derives from the left gastric and short gastric arteries), and short gastric arteries (from the splenic artery to the fundus). Venous drainage parallels the arterial supply, ultimately draining into the portal vein.
Lymphatic Drainage & Surgical Relevance
The stomach's lymphatic drainage follows the arterial supply and is divided into four zones by the Japanese classification: Zone I (right cardia — along the left gastric artery to celiac nodes), Zone II (greater curvature — along the gastroepiploic arteries to splenic and pancreatic nodes), Zone III (pyloric — along the right gastric artery to hepatic nodes), and Zone IV (lesser curvature — along the left gastric artery). In bariatric surgery, lymphatic disruption is generally not clinically significant, but awareness of lymphatic drainage is important when preoperative EGD identifies suspicious lesions (incidental early gastric cancer has been found in 0.1-0.3% of bariatric surgery specimens — the resected fundus/body after LSG should always be sent for pathological examination).
The vagus nerve provides parasympathetic innervation. The anterior (left) vagus gives off the hepatic branch (to the liver) and the criminal nerve of Grassi (branch to the fundus — if not divided, can cause persistent acid secretion). The posterior (right) vagus gives off the celiac branch. In sleeve gastrectomy, transection of the short gastric vessels and fundus removes much of the vagal fundic innervation. In RYGB, the small gastric pouch is effectively vagotomized.
Small Bowel Anatomy
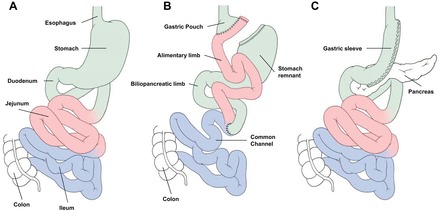
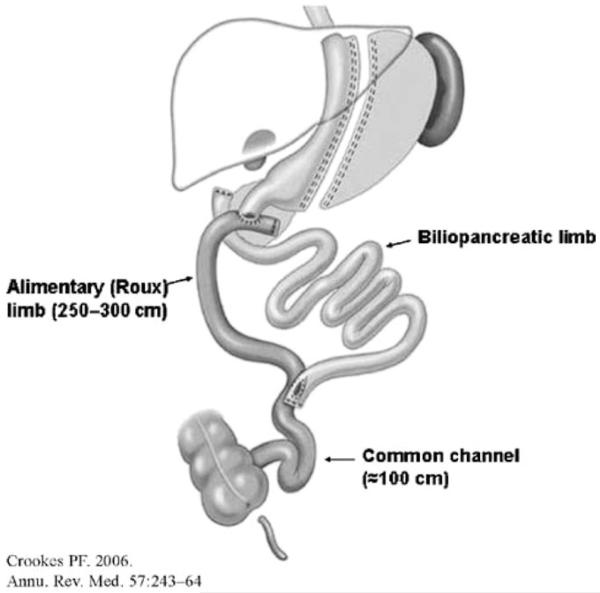
The small intestine measures approximately 300-800 cm (average ~600 cm) from the ligament of Treitz to the ileocecal valve. It is divided into the duodenum (~25 cm, C-shaped, retroperitoneal except for the first portion; site of iron and calcium absorption, bile/pancreatic duct entry at the ampulla of Vater), the jejunum (proximal ~40% of the remaining small bowel, larger villi, thicker wall, more prominent plicae circulares, primary site for folate, water-soluble vitamin absorption), and the ileum (distal ~60%, thinner wall, Peyer's patches, primary site for B12 and bile salt absorption). The ligament of Treitz marks the duodenojejunal junction and is the key landmark in all bypass procedures for identifying the starting point of the jejunal Roux limb.
In Roux-en-Y anatomy, three limb lengths must be defined: the biliopancreatic (BP) limb (from ligament of Treitz to the jejunojejunostomy — typically 30-75 cm in standard RYGB), the Roux (alimentary) limb (from the gastrojejunostomy to the jejunojejunostomy — typically 75-150 cm), and the common channel (from the jejunojejunostomy to the ileocecal valve — where digestion and absorption of nutrients occurs with bile and pancreatic enzymes). In malabsorptive procedures (BPD/DS), the common channel is shortened to 75-100 cm.
Gut Hormones & the Enteroendocrine Axis
Bariatric surgery produces weight loss and metabolic improvement through hormonal mechanisms that go far beyond simple restriction or malabsorption:
| Hormone | Source | Action | Change After RYGB | Change After LSG |
|---|---|---|---|---|
| Ghrelin | Gastric fundus (X/A-like cells) | Orexigenic (appetite stimulant); growth hormone release; increases gastric motility | Minimal change or slight decrease (fundus remains in excluded stomach) | Markedly decreased (fundus removed) |
| GLP-1 (glucagon-like peptide-1) | L-cells of distal ileum and colon | Incretin effect (glucose-dependent insulin secretion); suppresses glucagon; delays gastric emptying; promotes satiety via CNS | Markedly increased (expedited nutrient delivery to L-cells) | Increased (rapid gastric emptying) |
| PYY (peptide YY) | L-cells of distal ileum and colon | Anorexigenic (suppresses appetite via hypothalamus); slows gastric emptying ("ileal brake") | Markedly increased | Increased |
| GIP (glucose-dependent insulinotropic polypeptide) | K-cells of duodenum and proximal jejunum | Incretin effect; promotes fat storage in adipocytes | Decreased (duodenum bypassed) | Variable |
| CCK (cholecystokinin) | I-cells of duodenum and jejunum | Gallbladder contraction; pancreatic enzyme secretion; satiety signaling | Variable | Increased |
| Bile acids | Hepatocytes; enterohepatic circulation | FXR/TGR5 receptor agonism; GLP-1 release; energy expenditure via brown fat activation | Markedly increased (altered enterohepatic circulation) | Increased |
| Oxyntomodulin | L-cells | GLP-1 and glucagon receptor agonist; energy expenditure; appetite suppression | Increased | Increased |
Gut Microbiome
The gut microbiome undergoes profound changes after bariatric surgery. After RYGB, there is a shift from Firmicutes-dominant flora (associated with obesity and increased caloric harvest) toward Bacteroidetes, Proteobacteria (particularly Gammaproteobacteria), and Verrucomicrobia (Akkermansia muciniphila — associated with improved metabolic health). These microbial shifts occur within weeks of surgery and persist long-term. Fecal transplant from post-RYGB patients to germ-free mice transfers a lean phenotype, confirming that the altered microbiome independently contributes to weight loss and metabolic improvement (Liou et al., PMID: 23535730).
After LSG, microbiome changes are less dramatic but still significant — increased Bacteroidetes-to-Firmicutes ratio and increased microbial diversity. Short-chain fatty acid (SCFA) production by gut bacteria (butyrate, propionate, acetate) influences gut hormone secretion, intestinal barrier integrity, and hepatic glucose metabolism. The altered bile acid pool after RYGB (increased primary bile acids, altered FXR/TGR5 signaling) is a major driver of microbiome remodeling.
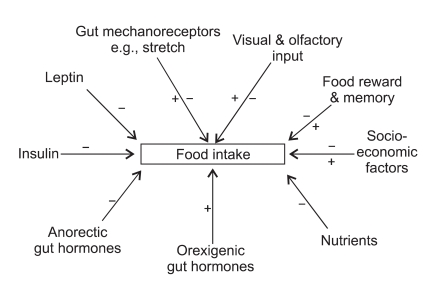
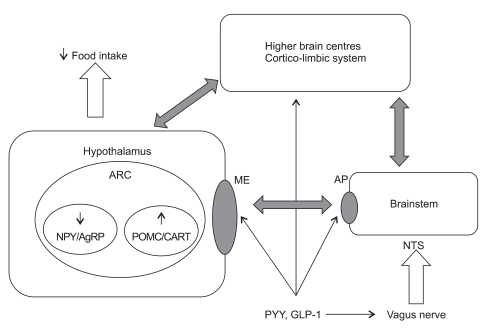
Neurohormonal Appetite Regulation
The hypothalamic arcuate nucleus integrates peripheral signals to regulate energy balance. Two key neuronal populations: NPY/AgRP neurons (orexigenic — stimulate hunger, decrease energy expenditure; activated by ghrelin, inhibited by leptin/insulin) and POMC/CART neurons (anorexigenic — suppress appetite, increase energy expenditure; activated by leptin/insulin, inhibited by ghrelin). These neurons project to second-order neurons in the paraventricular nucleus (PVN), lateral hypothalamus, and ventromedial hypothalamus. The nucleus tractus solitarius (NTS) in the brainstem receives vagal afferent signals from the gut (CCK, GLP-1, PYY, gastric distension) and relays satiety information to the hypothalamus.
After RYGB, the hormonal milieu shifts powerfully toward satiety: increased GLP-1 and PYY (anorexigenic), decreased ghrelin effect (though levels may not fall significantly), increased bile acid signaling, and altered vagal tone. The net effect is reduced hunger, earlier satiation, and changes in food preferences (reduced desire for high-fat, high-sugar foods — the "food reward" system in the mesolimbic dopaminergic pathway is modulated).
02 Obesity Physiology & Metabolic Syndrome
BMI Classification
| Category | BMI (kg/m²) | WHO Class |
|---|---|---|
| Underweight | < 18.5 | — |
| Normal weight | 18.5 – 24.9 | — |
| Overweight | 25.0 – 29.9 | Pre-obese |
| Obesity Class I | 30.0 – 34.9 | Class I |
| Obesity Class II | 35.0 – 39.9 | Class II |
| Obesity Class III (severe/morbid) | 40.0 – 49.9 | Class III |
| Super obesity | 50.0 – 59.9 | — |
| Super-super obesity | ≥ 60.0 | — |
Adipose Tissue Biology
White adipose tissue (WAT) is the predominant storage form; excess visceral WAT drives metabolic dysfunction through secretion of pro-inflammatory adipokines (TNF-α, IL-6, resistin) and reduced secretion of the insulin-sensitizing adipokine adiponectin. Adipocyte hypertrophy leads to hypoxia, macrophage infiltration (crown-like structures), and a chronic low-grade inflammatory state. Brown adipose tissue (BAT) dissipates energy as heat via uncoupling protein-1 (UCP1) and is metabolically favorable; bariatric surgery increases BAT activity. Beige/brite adipocytes are inducible thermogenic cells within WAT depots, activated by cold exposure, exercise, and bile acid signaling (FXR/TGR5 pathways).
Metabolic Syndrome
Defined by the harmonized criteria (IDF/AHA/NHLBI 2009) — requires ≥ 3 of 5 criteria:
| Criterion | Threshold |
|---|---|
| Waist circumference | ≥ 102 cm (M), ≥ 88 cm (F) — population-specific |
| Triglycerides | ≥ 150 mg/dL (or on Rx) |
| HDL cholesterol | < 40 mg/dL (M), < 50 mg/dL (F) (or on Rx) |
| Blood pressure | ≥ 130/85 mmHg (or on Rx) |
| Fasting glucose | ≥ 100 mg/dL (or on Rx for T2DM) |
Insulin Resistance & Pathogenesis of T2DM in Obesity
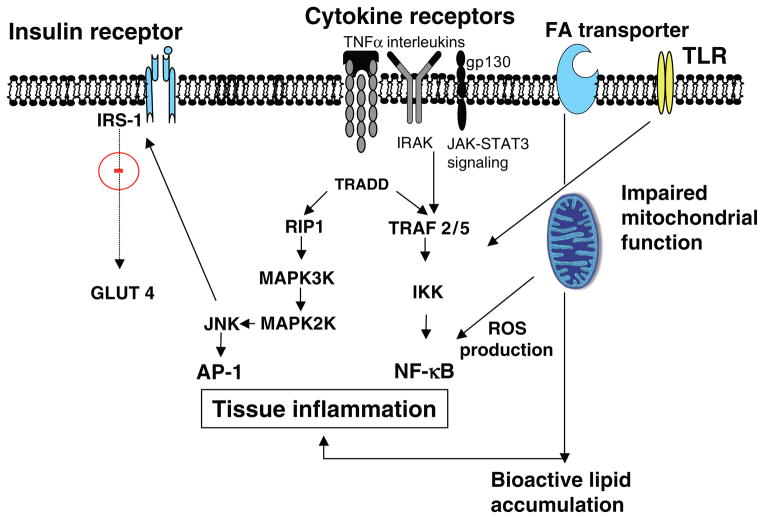
Obesity drives insulin resistance through multiple mechanisms: lipotoxicity (ectopic fat deposition in liver, muscle, and pancreas impairs insulin signaling via diacylglycerol and ceramide accumulation), chronic inflammation (adipose tissue macrophage infiltration produces TNF-α and IL-6, which activate JNK and IKK-β pathways, phosphorylating IRS-1 at serine residues and blocking insulin signal transduction), endoplasmic reticulum (ER) stress (unfolded protein response in hepatocytes contributes to hepatic insulin resistance), and altered adipokine profile (reduced adiponectin, increased resistin and retinol-binding protein-4).
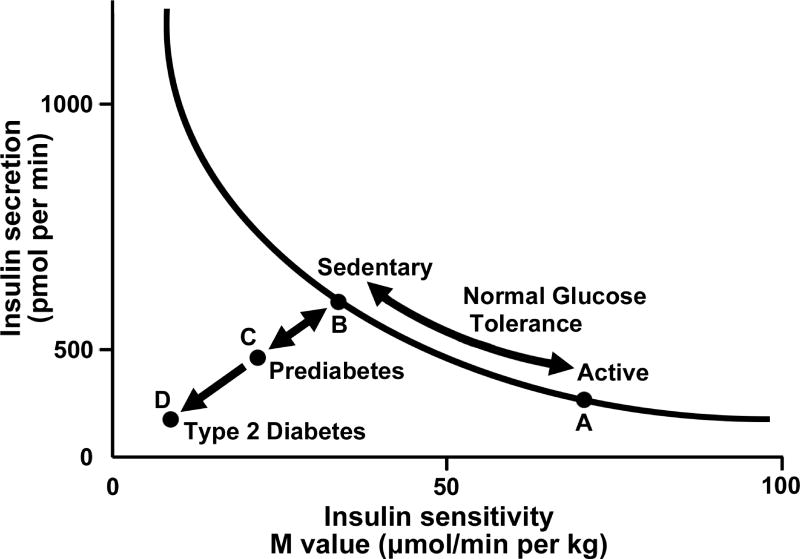
The progression from insulin resistance to T2DM requires beta-cell failure: initially, the pancreas compensates with hyperinsulinemia; over time, glucotoxicity and lipotoxicity cause beta-cell apoptosis (amyloid deposition, oxidative stress). At diagnosis of T2DM, approximately 50-80% of beta-cell function has been lost. This is why C-peptide level (≥ 2.0 ng/mL = preserved beta-cell reserve) is the strongest predictor of T2DM remission after bariatric surgery — patients with earlier-stage disease and preserved beta-cell function respond best.
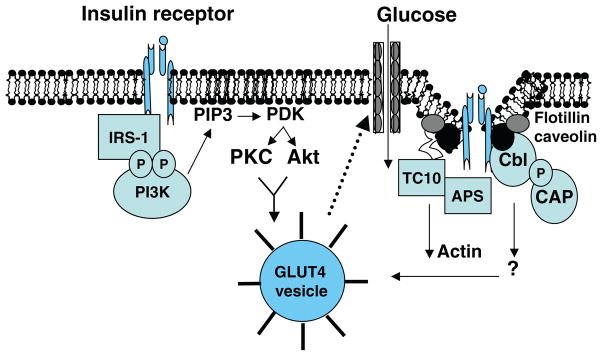
Set Point Theory & Energy Homeostasis
The body defends a genetically influenced "set point" weight through the leptin-melanocortin pathway: adipocytes secrete leptin proportional to fat mass; leptin acts on the arcuate nucleus of the hypothalamus, activating anorexigenic POMC/CART neurons and inhibiting orexigenic NPY/AgRP neurons. After diet-induced weight loss, leptin levels fall disproportionately, triggering adaptive thermogenesis (reduced resting energy expenditure beyond what is predicted by lost mass), increased ghrelin, and heightened hunger — driving weight regain. This metabolic adaptation persists for years (Rosenbaum et al., PMID: 18842775). Bariatric surgery appears to "reset" the defended body weight to a lower point, producing sustained hormonal changes that favor weight maintenance.
Genetics of Obesity
Obesity is a polygenic disorder with heritability estimated at 40-70%. Key genetic pathways include:
| Gene/Pathway | Function | Clinical Significance |
|---|---|---|
| MC4R (melanocortin-4 receptor) | Hypothalamic satiety signaling; most common monogenic obesity gene | Mutations present in 2-6% of severely obese patients; autosomal dominant with variable penetrance; patients have hyperphagia and hyperinsulinemia from childhood |
| LEP / LEPR (leptin / leptin receptor) | Adipocyte-derived hormone signaling satiety to hypothalamus | Rare autosomal recessive; congenital leptin deficiency causes extreme early-onset obesity; treatable with recombinant leptin (metreleptin) |
| POMC (pro-opiomelanocortin) | Precursor to α-MSH (anorexigenic peptide) | Deficiency causes obesity, adrenal insufficiency, and red hair; setmelanotide (MC4R agonist) is FDA-approved treatment |
| FTO (fat mass and obesity-associated) | Most replicated GWAS signal for BMI; affects appetite regulation | Common variants (40-65% of population carry risk allele); each allele increases BMI by 0.35 kg/m² |
| Prader-Willi Syndrome | Imprinting disorder (chromosome 15q11-13); hypothalamic dysfunction | Hyperphagia, intellectual disability, hypogonadism; bariatric surgery outcomes are poor (high complication rates, limited weight loss due to persistent hyperphagia) |
03 The Bariatric Exam & Preoperative Assessment
History & Physical Examination
The bariatric-specific history should assess: weight history (onset of obesity, maximum lifetime weight, prior weight loss attempts and methods, rate of weight gain), dietary history (eating patterns — structured meals vs grazing, nighttime eating, binge eating episodes, types of calories consumed, liquid calorie intake), exercise history (current activity level, barriers to exercise — joint pain, dyspnea, space constraints), comorbidity inventory (screen for all obesity-related conditions), medication review (identify weight-promoting medications — insulin, sulfonylureas, thiazolidinediones, steroids, antipsychotics, certain antidepressants, gabapentin/pregabalin, beta-blockers), substance use (alcohol, tobacco, marijuana, opioids), surgical history (prior abdominal operations — assess feasibility of laparoscopic approach), and reproductive plans (contraception counseling — pregnancy should be delayed 12-18 months).
The physical examination should include: vitals (with appropriately sized BP cuff — using a standard cuff on a large arm falsely elevates readings), BMI and waist circumference, neck circumference (> 43 cm in men and > 41 cm in women predicts OSA), abdominal exam (prior surgical scars — assess for ventral hernia; hepatomegaly; assessment of pannus for planning port placement), skin exam (acanthosis nigricans — marker of insulin resistance; intertriginous rashes; stasis dermatitis), extremities (venous stasis changes, lymphedema, peripheral edema), and Mallampati score (airway assessment for anesthetic planning).
Cardiac Risk Evaluation
All bariatric surgery candidates require assessment of cardiovascular risk. The ACC/AHA Perioperative Guidelines classify bariatric surgery as an intermediate-risk procedure (estimated cardiac risk 1-5%). Assessment includes resting ECG, echocardiography (if clinical suspicion for heart failure, valvular disease, or pulmonary hypertension), and functional capacity assessment. Patients with ≥ 4 METs of functional capacity without symptoms generally do not require further cardiac testing. Stress testing is indicated for patients with active cardiac conditions or poor functional capacity with ≥ 3 clinical risk factors (Lee's Revised Cardiac Risk Index).
STOP-BANG Questionnaire for OSA Screening
| Letter | Question | Scoring |
|---|---|---|
| S | Snoring — Do you snore loudly? | Yes = 1 |
| T | Tired — Do you often feel tired, fatigued, or sleepy during daytime? | Yes = 1 |
| O | Observed — Has anyone observed you stop breathing during sleep? | Yes = 1 |
| P | Pressure — Are you being treated for high blood pressure? | Yes = 1 |
| B | BMI > 35 kg/m²? | Yes = 1 |
| A | Age > 50 years? | Yes = 1 |
| N | Neck circumference > 40 cm (16 inches)? | Yes = 1 |
| G | Gender — Male? | Yes = 1 |
Interpretation: Score 0-2 = low risk; 3-4 = intermediate risk; 5-8 = high risk of moderate-severe OSA. In the bariatric population, most patients score ≥ 3 by default (BMI > 35, often hypertensive, large neck circumference).
Obstructive Sleep Apnea Screening
Prevalence of OSA in bariatric surgery candidates is 60-80%. Screening tools include the STOP-BANG questionnaire (score ≥ 5 = high probability of moderate-severe OSA) and the Epworth Sleepiness Scale. All patients with positive screens require polysomnography (PSG). Patients with confirmed moderate-severe OSA (≥ 15 events/hour) should be initiated on CPAP preoperatively (ideally 4-6 weeks before surgery) and instructed to bring their device for postoperative use. Untreated severe OSA increases risk of postoperative respiratory failure, arrhythmia, and sudden death.
Psychological Evaluation
A comprehensive psychosocial evaluation by a licensed mental health professional is required by most insurance payers and recommended by ASMBS guidelines. Key areas assessed: eating disorder history (binge eating disorder present in ~25-50% of candidates — not an absolute contraindication but requires treatment), active substance abuse (must be in remission), uncontrolled psychiatric illness (severe depression, psychosis), cognitive capacity for informed consent and adherence to postoperative regimen, and social support. Risk of new-onset alcohol use disorder is increased after RYGB (~7-10%, likely due to altered alcohol pharmacokinetics — faster absorption, higher peak BAC, and cross-addiction transfer).
Preoperative Nutritional Labs
| Lab | Rationale | Frequency |
|---|---|---|
| CBC with differential | Baseline anemia assessment (iron, B12, folate deficiency common in obese) | Baseline |
| CMP (BMP + LFTs) | Hepatic function, renal function, glucose, electrolytes | Baseline |
| HbA1c | Glycemic control in T2DM patients; screen for undiagnosed diabetes | Baseline |
| Lipid panel | Cardiovascular risk stratification | Baseline |
| Iron studies (Fe, TIBC, ferritin) | Iron deficiency common (30-40% of obese patients preoperatively) | Baseline |
| Vitamin B12 | Baseline; deficiency in 2-18% preoperatively | Baseline |
| Thiamine (B1) | Critical: deficiency risk with prolonged vomiting postop | Baseline |
| 25-OH Vitamin D | Deficiency in 60-90% of bariatric candidates | Baseline |
| PTH, calcium | Secondary hyperparathyroidism screening | Baseline |
| Folate (RBC) | Baseline for women of childbearing age | Baseline |
| Zinc, copper | Baseline before malabsorptive procedures | Baseline (BPD/DS, RYGB) |
| Vitamin A | Deficiency risk with malabsorptive procedures | Baseline (BPD/DS) |
Additional Preoperative Requirements
Upper endoscopy (EGD) — recommended by ASMBS for all RYGB candidates (to identify H. pylori, Barrett's, ulcers, hiatal hernia) as the excluded stomach cannot be endoscoped postoperatively. For sleeve gastrectomy, EGD is selectively recommended. H. pylori testing and eradication are required before all bypass procedures. Abdominal ultrasound — screens for cholelithiasis; concomitant cholecystectomy is no longer routinely recommended but ursodiol prophylaxis (300 mg BID for 6 months postop) reduces gallstone formation from 30% to < 2%. Pulmonary function testing — indicated in patients with known lung disease or BMI > 50. Preoperative weight loss — 2-4 week liquid diet (or 5-10% body weight loss) is commonly required to reduce liver volume and facilitate laparoscopic visualization.
04 Patient Selection Criteria
NIH 1991 Consensus Criteria (Traditional)
The landmark 1991 NIH Consensus Conference established criteria that remained the standard for three decades:
- BMI ≥ 40 kg/m² without comorbidities, OR
- BMI ≥ 35 kg/m² with at least one obesity-related comorbidity (T2DM, hypertension, OSA, NAFLD, GERD, pseudotumor cerebri, Pickwickian syndrome, weight-bearing joint disease)
- Failure of non-surgical weight loss attempts
- Acceptable operative risk
- Well-informed and motivated patient
Updated ASMBS/IFSO 2022 Consensus Guidelines
The 2022 joint ASMBS/IFSO statement significantly expanded indications (Eisenberg et al., PMID: 36336720):
- BMI ≥ 35 kg/m² — surgery recommended regardless of comorbidities
- BMI 30-34.9 kg/m² — surgery recommended for patients with metabolic disease (T2DM, NAFLD/NASH, dyslipidemia, hypertension, OSA) who have not achieved durable weight loss or comorbidity improvement with non-surgical methods
- BMI thresholds should be adjusted for Asian populations (reduce by 2.5 units: ≥ 32.5 without comorbidities, ≥ 27.5 with metabolic disease)
- Surgery should not be denied based on age alone (adolescents ≥ Tanner stage IV and patients aged 65+ can benefit)
Contraindications
| Absolute | Relative |
|---|---|
| Active substance abuse disorder | BMI < 30 (except in clinical trials) |
| Uncontrolled severe psychiatric illness | Crohn's disease (relative for bypass) |
| Inability to comply with nutritional supplementation | Previous extensive abdominal surgery |
| Active malignancy requiring treatment | Cirrhosis (Child-Pugh B/C) |
| Inability to provide informed consent | Active H. pylori (treat before surgery) |
| Medically unfit for general anesthesia (ASA V) | Barrett's esophagus (relative for LSG; prefer RYGB) |
Special Populations
Adolescents: Bariatric surgery is increasingly performed in adolescents (age 13-17) with severe obesity (BMI ≥ 35 with comorbidities or ≥ 40 with or without comorbidities). The Teen-LABS study (PMID: 26444015) demonstrated excellent weight loss outcomes (26-28% TWL at 3 years after RYGB) with comorbidity resolution rates similar to adults. Requirements: Tanner stage IV-V, skeletal maturity (or near-maturity), and a multidisciplinary evaluation including pediatric psychologist and nutritionist. LSG is the most commonly performed procedure in adolescents.
Elderly (≥ 65 years): Advanced age is not an absolute contraindication. However, complication rates are modestly higher (15-20% vs 10-15% in younger patients), and weight loss may be less robust. The primary goal in elderly patients is comorbidity improvement (T2DM remission, OSA resolution, improved mobility) rather than cosmetic weight loss. LSG is often preferred for its shorter operative time and lower complication profile.
Super-obese (BMI ≥ 50): These patients carry significantly higher operative risk (3-5 fold increase in complications vs BMI 35-45). Strategies include: preoperative weight loss (liquid diet, IGB placement, or GLP-1 RA therapy), staged approach (LSG first, followed by conversion to DS or RYGB at lower BMI), and centers of excellence with high-volume surgeons. Liver retraction is particularly challenging due to massive hepatomegaly.
Cirrhosis: Compensated cirrhosis (Child-Pugh A) is manageable — LSG is the preferred procedure (avoids bypass physiology and preserves endoscopic access). Decompensated cirrhosis (Child-Pugh B/C) generally precludes standard bariatric surgery — LSG may be performed as a bridge to liver transplantation in select centers. MELD score ≥ 15 correlates with significantly increased perioperative mortality.
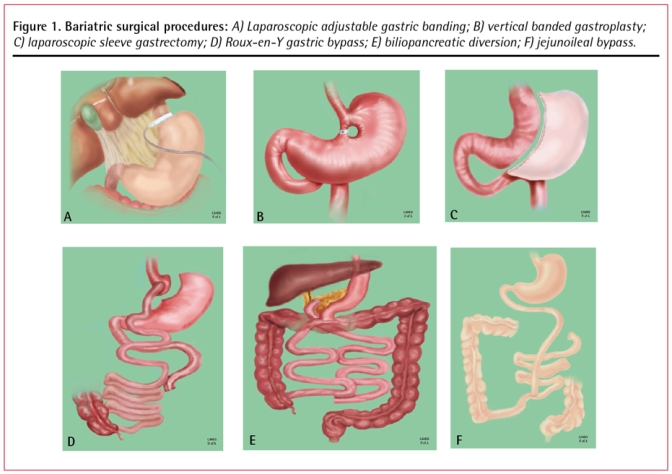
Procedure Selection Algorithm
Choosing the optimal bariatric procedure is individualized based on patient characteristics, comorbidities, and goals:
| Clinical Scenario | Preferred Procedure | Rationale |
|---|---|---|
| BMI 30-35 with T2DM | RYGB or OAGB | Highest T2DM remission rates among standard procedures; strongest metabolic effect for moderate obesity |
| BMI 35-45, no GERD, no Barrett's | LSG or RYGB | Patient preference; both effective; LSG has slightly lower morbidity; RYGB has slightly better metabolic outcomes |
| BMI 35-45 with severe GERD or Barrett's | RYGB (preferred) | RYGB resolves GERD in 85-95%; LSG may worsen GERD and is relatively contraindicated with Barrett's |
| BMI ≥ 50 (super-obese) | RYGB, or staged LSG → DS/SADI-S | Super-obese patients may benefit from the more powerful BPD/DS if staged safely |
| Crohn's disease | LSG (preferred) | Bypass procedures risk anastomotic complications at diseased bowel; sleeve avoids small bowel manipulation |
| Organ transplant candidate (renal, liver) | LSG | Avoids altered drug absorption of bypass; preserves endoscopic access; shorter procedure; lower nutritional risk |
| Chronic NSAID requirement | LSG (preferred) | NSAIDs are contraindicated after RYGB due to marginal ulcer risk; LSG has intact pylorus |
| Iron deficiency anemia (pre-existing severe) | LSG or LAGB | RYGB and BPD/DS bypass the duodenum (primary iron absorption site), worsening anemia |
| Failed LSG (weight regain) | Conversion to RYGB or SADI-S | RYGB is most common conversion; adds malabsorptive component |
| Failed LSG (severe GERD) | Conversion to RYGB | RYGB reliably resolves post-sleeve GERD in > 90% |
05 Metabolic Outcomes & T2DM Remission
T2DM Remission by Procedure
Type 2 diabetes remission is the most dramatic metabolic outcome of bariatric surgery. Remission is defined as HbA1c < 6.0% (complete) or < 6.5% (partial) without diabetes medications for ≥ 1 year.
| Procedure | T2DM Remission at 1 yr | T2DM Remission at 5 yr | %EWL at 5 yr |
|---|---|---|---|
| RYGB | 75-85% | 50-65% | 60-75% |
| LSG | 55-70% | 40-55% | 50-65% |
| BPD/DS | 90-98% | 85-95% | 70-80% |
| SADI-S | 85-95% | 75-90% | 65-80% |
| LAGB | 40-55% | 25-35% | 40-50% |
| OAGB | 75-85% | 55-70% | 60-75% |
DiaRem & ABCD Scores
The DiaRem Score predicts T2DM remission probability after RYGB based on 4 variables: age, HbA1c, use of insulin, and type of other diabetes medications (score 0-22; score 0-2 = 87-88% remission; score 18-22 = 0-5% remission; Still et al., PMID: 24170248).
The ABCD Score (Lee et al.) incorporates: Age (< 40 = 1 point), BMI (≥ 35 = 1 point), C-peptide (≥ 2.0 ng/mL = 1 point, ≥ 5.0 = 2 points), and Duration of T2DM (< 4 years = 1 point). A higher score predicts greater likelihood of remission. C-peptide ≥ 2.0 ng/mL (reflecting preserved beta-cell function) is the strongest predictor of T2DM remission.
Mechanisms of T2DM Remission
The mechanisms by which bariatric surgery achieves T2DM remission are multifactorial and procedure-specific:
| Mechanism | Timeline | Procedure Relevance |
|---|---|---|
| Caloric restriction | Immediate (days) | All procedures — reduced caloric intake improves hepatic insulin sensitivity within 48-72 hours |
| Incretin enhancement (GLP-1, PYY) | Days to weeks | RYGB > OAGB > LSG — expedited nutrient delivery to L-cells in distal ileum; "hindgut hypothesis" |
| Foregut exclusion | Days to weeks | RYGB, OAGB, BPD/DS — bypassing the duodenum removes putative "anti-incretin" signals; "foregut hypothesis" |
| Bile acid changes | Weeks to months | RYGB, BPD/DS — increased circulating bile acids activate FXR and TGR5 receptors, improving insulin sensitivity and GLP-1 secretion |
| Gut microbiome remodeling | Weeks to months | All procedures — shift from Firmicutes to Bacteroidetes; increased Akkermansia muciniphila |
| Reduced adipose tissue inflammation | Months | All procedures — weight loss reduces TNF-α, IL-6, and macrophage infiltration; increases adiponectin |
| Reduced ectopic fat | Months | All procedures — reduced hepatic and pancreatic fat improves insulin sensitivity and beta-cell function |
| Beta-cell recovery | Months to years | All procedures — reduced glucolipotoxicity allows partial beta-cell functional recovery (limited by disease duration) |
Resolution of Other Comorbidities
| Comorbidity | Resolution Rate (RYGB) | Resolution Rate (LSG) |
|---|---|---|
| Hypertension | 60-75% | 50-65% |
| Dyslipidemia | 70-85% | 60-75% |
| Obstructive sleep apnea | 75-90% | 65-80% |
| GERD | 85-95% (gold standard for GERD) | Variable; may worsen (de novo GERD 15-35%) |
| NAFLD/NASH | 85-95% (steatosis); 60-70% (fibrosis) | 80-90% (steatosis); 50-65% (fibrosis) |
| Depression | 55-65% | 50-60% |
06 Obesity-Related Comorbidities
NAFLD / NASH
Non-alcoholic fatty liver disease affects > 90% of bariatric surgery candidates. NAFLD encompasses a spectrum: simple steatosis (≥ 5% hepatocyte fat content) → NASH (steatosis with inflammation and hepatocyte ballooning) → fibrosis → cirrhosis. Bariatric surgery is the most effective intervention: NASH resolution occurs in 60-85% of patients, fibrosis improves in 30-65%, and progression to cirrhosis is markedly reduced (Lassailly et al., PMID: 31401286). Compensated cirrhosis (Child-Pugh A) is not a contraindication; decompensated cirrhosis (Child-Pugh B/C) typically precludes standard bariatric surgery (consider sleeve gastrectomy as bridge to transplant).
Polycystic Ovary Syndrome (PCOS)
PCOS is the most common endocrine disorder in reproductive-age women, affecting 6-20%. Obesity exacerbates hyperandrogenism, insulin resistance, and anovulation. After bariatric surgery, 50-75% of patients achieve resolution of menstrual irregularity, 40-60% have reduced hirsutism, and spontaneous fertility is restored in many previously anovulatory patients. Contraception is mandatory for 12-18 months postoperatively due to risk of nutritional deficiency during rapid weight loss phase.
Obesity Hypoventilation Syndrome (OHS / Pickwickian)
OHS is defined as obesity (BMI ≥ 30) plus awake alveolar hypoventilation (PaCO2 ≥ 45 mmHg) not attributable to other causes. Prevalence in bariatric candidates with BMI > 50 is 10-20%. Associated with pulmonary hypertension (cor pulmonale in severe cases), biventricular failure, and high perioperative mortality (up to 4-8% without preoperative optimization). Preoperative noninvasive positive pressure ventilation (BiPAP) is essential. Resolution after bariatric surgery exceeds 75%.
Pseudotumor Cerebri (Idiopathic Intracranial Hypertension)
Strongly associated with obesity in young women. Presents with headache, visual disturbances, papilledema, and CSF opening pressure > 25 cmH2O. Bariatric surgery produces resolution or improvement in 90-95% of cases (often within weeks of surgery, before major weight loss occurs). Weight loss of 5-10% can significantly reduce intracranial pressure.
Obesity & Cancer Risk
Obesity is associated with significantly increased risk of at least 13 cancers (IARC 2016): endometrial (RR 2.5-7.0 — strongest association), esophageal adenocarcinoma (RR 4.0-5.0), gastric cardia, colorectal (RR 1.3-1.5), hepatocellular (RR 1.5-2.0), gallbladder, pancreatic (RR 1.5-2.0), renal cell (RR 1.5-2.5), meningioma, thyroid, multiple myeloma, breast (postmenopausal) (RR 1.2-1.5), and ovarian. Mechanisms include chronic hyperinsulinemia (IGF-1 signaling promotes cell proliferation), chronic inflammation (IL-6, TNF-α), elevated estrogen (aromatase activity in adipose tissue), and altered adipokine signaling. The SOS study demonstrated that bariatric surgery reduces cancer incidence by 42% in women (primarily through reduction of endometrial, breast, and colorectal cancers).
Obesity-Related Cardiomyopathy
Obesity cardiomyopathy is characterized by LV dilation and eccentric hypertrophy (due to chronic volume overload from increased blood volume), diastolic dysfunction (lipotoxic infiltration of the myocardium), and eventual systolic heart failure. BMI > 40 is associated with a 2-fold increased risk of heart failure. After bariatric surgery, LV mass decreases by 15-25%, diastolic function improves, and heart failure symptoms resolve or improve in 60-80% of patients within 1-2 years.
Weight-Bearing Joint Disease
Every 1 kg of body weight exerts approximately 4 kg of force across the knee during walking. Bariatric surgery reduces the need for total knee arthroplasty (TKA) and total hip arthroplasty (THA); when joint replacement is necessary, prior bariatric surgery reduces perioperative complication rates (infection, DVT, dislocation). Optimizing BMI to < 40 (ideally < 35) before arthroplasty is recommended by most orthopedic guidelines. Beyond the mechanical benefits, weight loss reduces systemic inflammation (lower CRP, IL-6) which contributes to joint inflammation and osteoarthritis progression.
Venous Stasis Disease & Lymphedema
Chronic venous insufficiency and lymphedema are common in patients with severe obesity (BMI > 50). Elevated intra-abdominal pressure from visceral adiposity impedes venous return from the lower extremities, leading to chronic venous hypertension, stasis dermatitis, lipodermatosclerosis, and venous ulceration. Lymphedema occurs when the lymphatic system is overwhelmed by the combination of venous congestion and excess adipose tissue compressing lymphatic channels. After bariatric surgery, 60-70% of patients with chronic venous disease experience significant improvement, and venous ulcer healing rates improve dramatically with weight loss.
07 Nutritional Deficiencies & Supplementation
Deficiency Risk by Procedure
| Nutrient | LAGB | LSG | RYGB | BPD/DS | Mechanism |
|---|---|---|---|---|---|
| Iron | Low | Moderate | High (20-55%) | Very high (25-70%) | Bypassed duodenum (primary absorption site); reduced gastric acid impairs Fe3+ → Fe2+ conversion |
| Vitamin B12 | Low | Low-Mod | High (15-35%) | Very high (20-50%) | Reduced IF production (small pouch); bypassed duodenum/proximal jejunum |
| Folate | Low | Low | Moderate (5-15%) | Moderate (10-20%) | Bypassed proximal jejunum; usually prevented by standard MVI |
| Calcium | Low | Low-Mod | High | Very high (25-70%) | Bypassed duodenum; reduced acid for calcium solubility; vitamin D deficiency |
| Vitamin D | Moderate | Moderate | High (25-50%) | Very high (50-80%) | Fat-soluble vitamin; fat malabsorption; sequestration in adipose tissue |
| Thiamine (B1) | Low | Moderate | Moderate (5-10%) | Moderate | Poor intake; persistent vomiting; carbohydrate-heavy refeeding |
| Vitamin A | Low | Low | Low-Mod | Very high (50-70%) | Fat-soluble vitamin; fat malabsorption in BPD/DS |
| Copper | Low | Low | Moderate (5-15%) | High (10-25%) | Bypassed duodenum; zinc supplementation competes with absorption |
| Zinc | Low | Low-Mod | Moderate (10-20%) | High (30-50%) | Bypassed duodenum/proximal jejunum |
ASMBS Supplementation Guidelines
| Supplement | LAGB / LSG | RYGB | BPD/DS |
|---|---|---|---|
| Multivitamin with minerals | 1 daily | 2 daily | 2 daily (high potency) |
| Calcium citrate + Vit D | 1200-1500 mg/day + D3 3000 IU | 1200-1500 mg/day + D3 3000 IU | 1800-2400 mg/day + D3 3000-5000 IU |
| Iron (with vitamin C) | 18 mg/day (MVI) | 45-60 mg/day elemental | 45-60 mg/day elemental |
| Vitamin B12 | Per MVI | 1000 µg/day SL or 1000 µg/month IM | 1000 µg/day SL or 1000 µg/month IM |
| Vitamin A (BPD/DS) | — | — | 5000-10000 IU/day |
| Vitamin E (BPD/DS) | — | — | 15 mg/day |
| Vitamin K (BPD/DS) | — | — | 300 µg/day |
08 Roux-en-Y Gastric Bypass (RYGB)
Surgical Technique
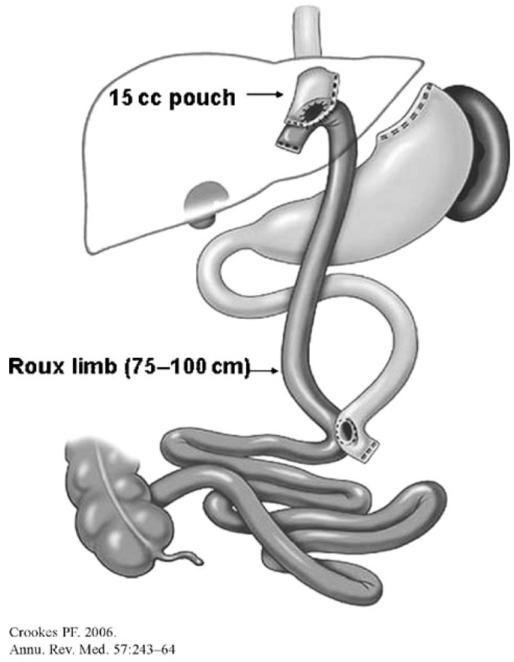
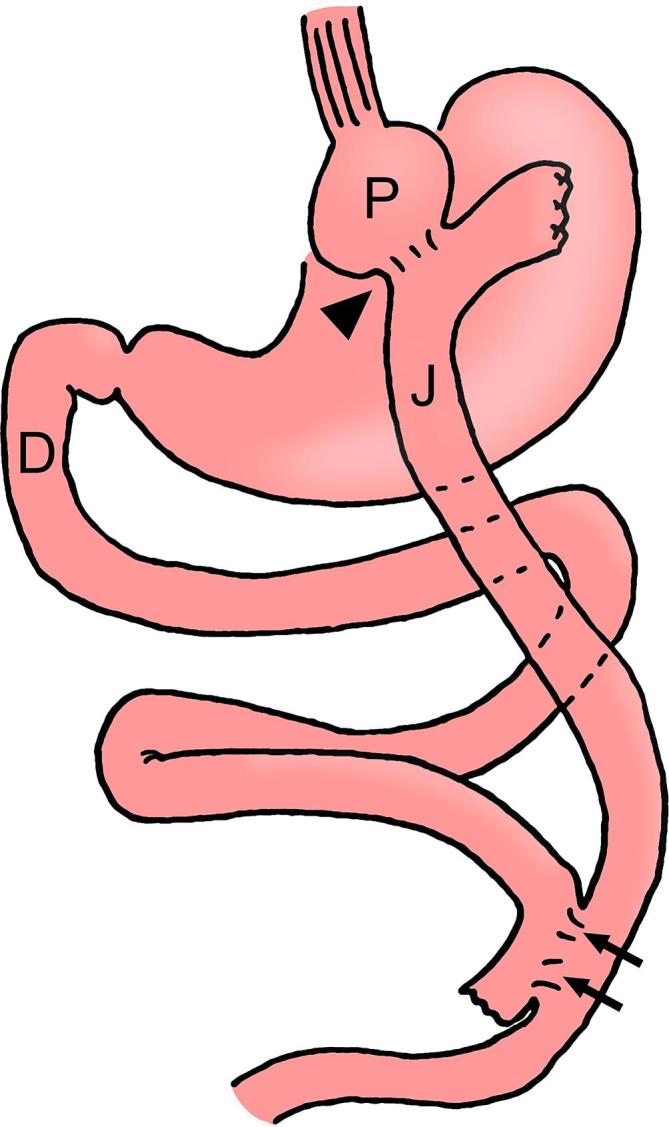
The Roux-en-Y gastric bypass is a combined restrictive and malabsorptive procedure; it is considered the "gold standard" bariatric operation. The key steps of the laparoscopic technique:
- Gastric pouch creation: A small (15-30 mL) gastric pouch is created along the lesser curvature using sequential firings of a linear endoscopic stapler. The pouch is divided completely from the gastric remnant. The first stapler firing begins at the lesser curvature approximately 3-4 cm distal to the GEJ. A calibrating tube (32-36 Fr bougie) may guide pouch sizing.
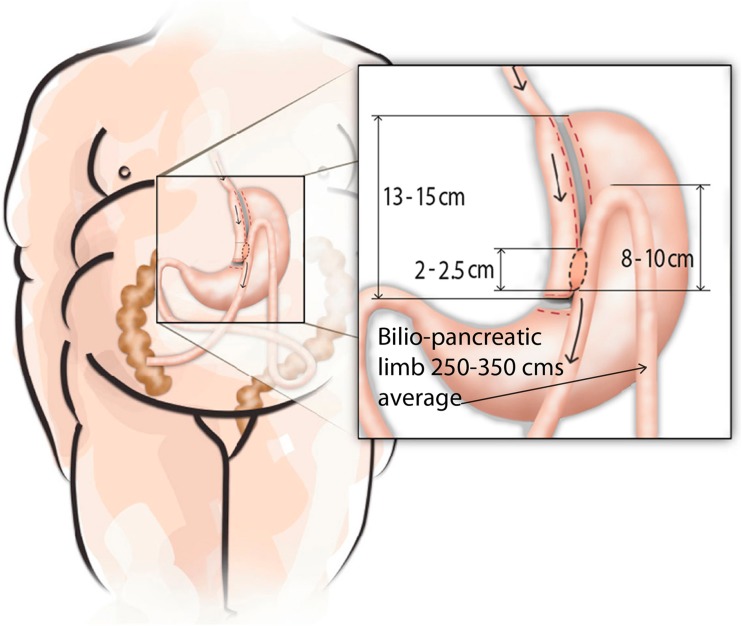
- Roux limb creation: The jejunum is identified at the ligament of Treitz. The jejunum is divided 30-75 cm distal to the ligament of Treitz (creating the biliopancreatic limb). The distal end of the divided jejunum is then measured distally 75-150 cm to create the Roux (alimentary) limb.
- Gastrojejunostomy (GJ): The Roux limb is brought up to the gastric pouch (antecolic, antegastric approach is most common) and a GJ anastomosis is created — linear stapled (25-30 mm opening) or hand-sewn or circular stapled (21-25 mm EEA). The GJ is the most critical anastomosis and the most common site of leak (1-5%).
- Jejunojejunostomy (JJ): A side-to-side jejunojejunostomy is created where the biliopancreatic limb meets the Roux limb, restoring intestinal continuity. This is performed with a linear stapler.
- Mesenteric defect closure: Petersen's space (between the Roux limb mesentery and the transverse mesocolon) and the jejunojejunostomy mesenteric defect are closed with non-absorbable sutures or clips to prevent internal hernia (incidence drops from 5-10% to < 1% with closure).
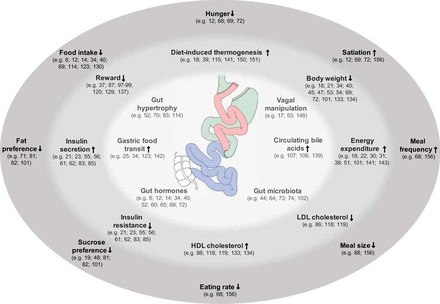
Mechanism of Weight Loss
RYGB achieves weight loss through multiple mechanisms: restriction (small 15-30 mL pouch limits meal volume), mild malabsorption (bypassed duodenum and proximal jejunum), hormonal changes (markedly increased GLP-1, PYY; altered bile acid signaling), altered gut microbiome (increased Gammaproteobacteria, Fusobacteria), vagal disruption, and food aversions (dumping physiology discourages simple sugar intake).
Outcomes
Expected %EWL at 1 year: 65-80%; at 5 years: 55-70%. Mean %TWL: 25-35%. T2DM remission: 75-85% at 1 year, 50-65% at 5 years. 30-day mortality: 0.1-0.3%. Overall complication rate: 10-15%. Long-term nutritional deficiency rate: 30-50% without adherent supplementation (Arterburn et al., PMID: 32167525).
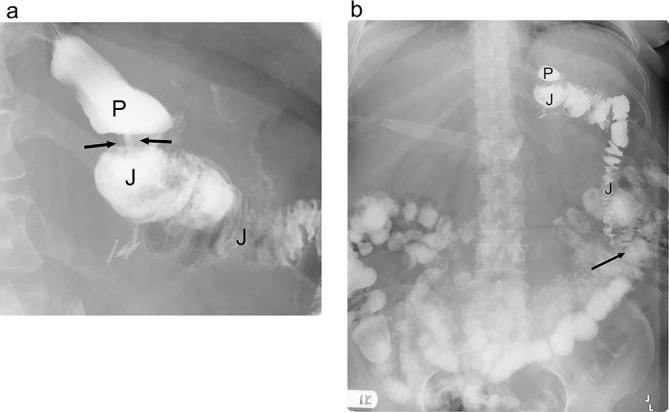
Antecolic vs Retrocolic Roux Limb
The Roux limb can be routed antecolic (in front of the transverse colon and its mesentery) or retrocolic (through the transverse mesocolon). The antecolic approach is now preferred by most surgeons because it avoids creating a mesocolic defect (reducing internal hernia risk) and simplifies the procedure. However, the antecolic route requires a longer Roux limb to reach the pouch without tension. The retrocolic approach provides a shorter, more direct route to the pouch but creates an additional mesenteric defect (mesocolic window) that must be closed to prevent internal hernia. Studies show internal hernia rates of 1-3% with antecolic and 3-5% with retrocolic approaches when defects are routinely closed.
Gastric Remnant Considerations
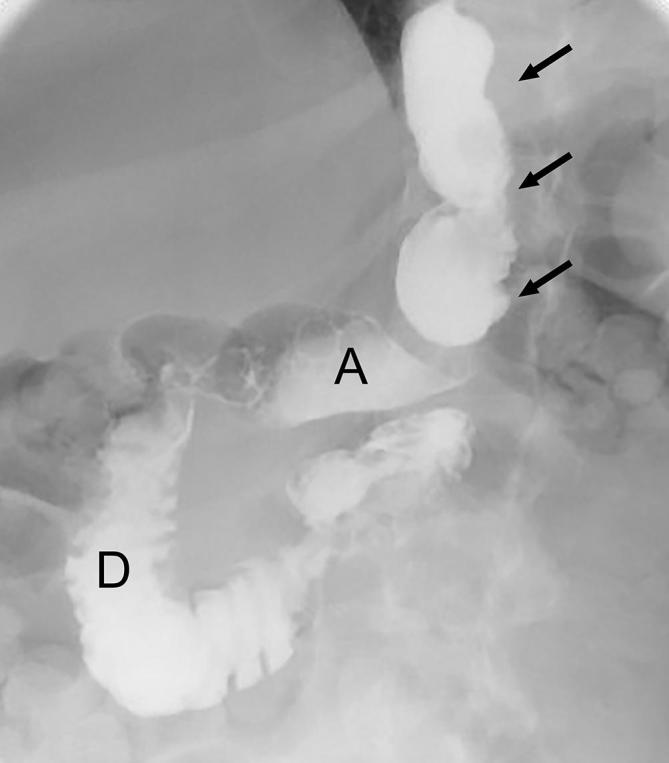
The excluded gastric remnant after RYGB cannot be accessed by standard upper endoscopy — this is a critical consideration for long-term surveillance. Options for remnant evaluation include: double-balloon enteroscopy (can reach the remnant in 60-80% of cases), laparoscopy-assisted transgastric endoscopy (inserting an endoscope through the remnant wall intraoperatively), and CT enterography (for mass lesions). Concern about occult pathology in the excluded stomach is one reason some surgeons advocate for preoperative EGD in all RYGB candidates and a relative argument for LSG in patients with family history of gastric cancer. A gastrogastric fistula (abnormal communication between the pouch and remnant) occurs in 1-6% of patients and presents with weight regain, marginal ulcer, or GERD — diagnosis by UGI series or EGD, treatment is surgical revision.
09 Sleeve Gastrectomy (LSG)
Surgical Technique
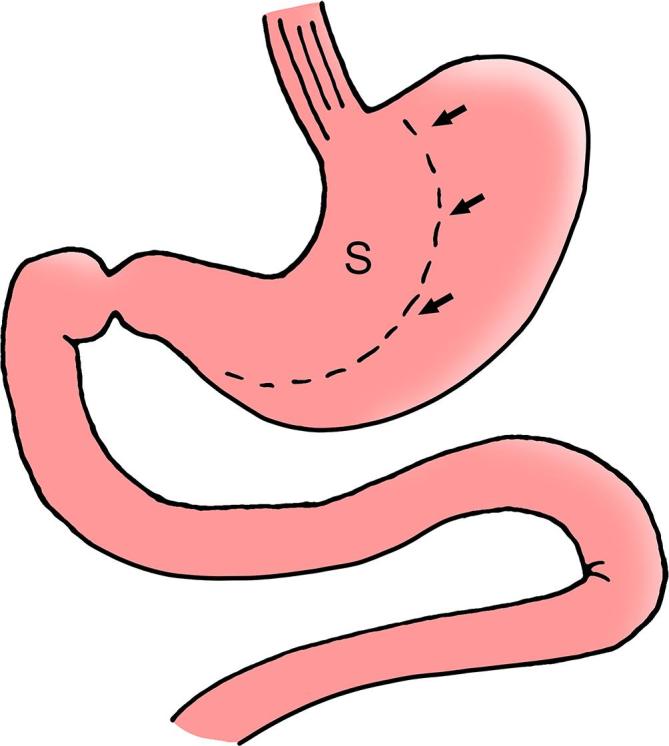
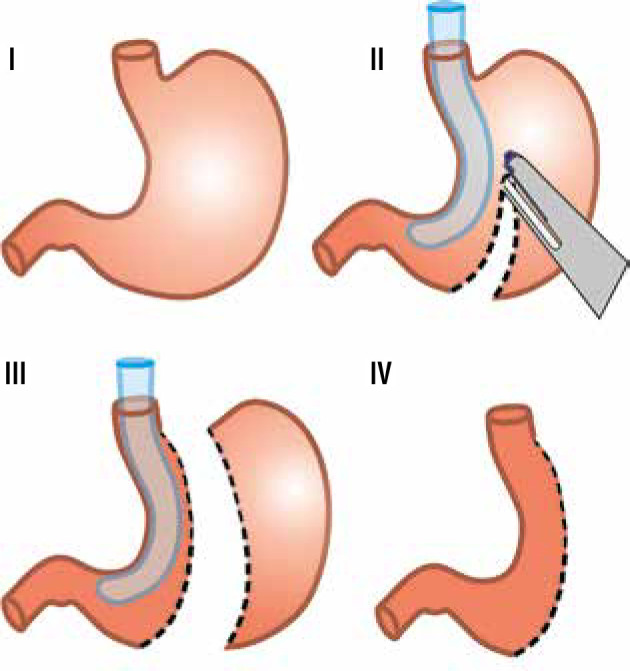
Laparoscopic sleeve gastrectomy (LSG) is a purely restrictive procedure (though hormonal changes contribute significantly). It has become the most commonly performed bariatric procedure worldwide (~60% of all bariatric procedures globally as of 2023).
- Greater curvature devascularization: The greater omentum is separated from the greater curvature of the stomach beginning 4-6 cm proximal to the pylorus and extending to the angle of His, dividing the short gastric vessels and the gastroepiploic arcade. An energy device (ultrasonic shears or bipolar vessel sealer) is used.
- Bougie calibration: A bougie (typically 36-40 Fr; 32 Fr bougie associated with more strictures, 40 Fr associated with less weight loss) is passed transorally along the lesser curvature to calibrate the sleeve.
- Staple-line transection: Sequential firings of a linear endoscopic stapler (60 mm loads; green or gold cartridges for thicker antrum, blue or purple for thinner fundus) create the sleeve, starting 4-6 cm from the pylorus and extending to the angle of His. Typically 5-7 stapler firings are required. The resected fundus and body (60-80% of stomach volume) are removed.
- Staple-line reinforcement: Options include buttressing material (bovine pericardium strips — Peristrips, absorbable polymer — Seamguard), oversewing (running suture), or no reinforcement. The evidence is mixed; reinforcement may reduce staple-line bleeding but has not definitively proven to reduce leak rates.
Key Technical Points
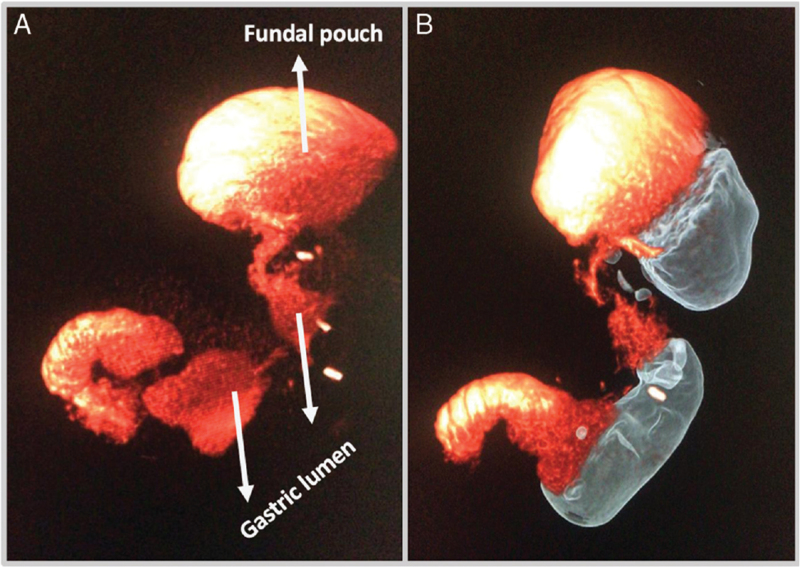
Start the staple line 4-6 cm from the pylorus — too close (< 2 cm) risks pyloric narrowing and delayed gastric emptying; too far (> 6 cm) leaves excessive antrum and may compromise weight loss. The incisura angularis (angularis incisure) is the most common site of stricture — careful lateral traction and precise stapler alignment prevent kinking. At the angle of His, the last stapler firing must be close to the left crus of the diaphragm but should not incorporate esophageal tissue. Complete mobilization of the fundus from the left crus is essential to avoid a retained fundus ("neo-fundus" or "dog ear"), which may cause inadequate weight loss and persistent ghrelin secretion.
Outcomes
Expected %EWL at 1 year: 55-70%; at 5 years: 45-60%. Mean %TWL: 20-30%. T2DM remission: 55-70% at 1 year. 30-day mortality: 0.05-0.2% (lowest of all bariatric procedures). Overall complication rate: 5-10%. Staple-line leak rate: 1-3% (most commonly at the GEJ/upper sleeve). De novo GERD: 15-35%.
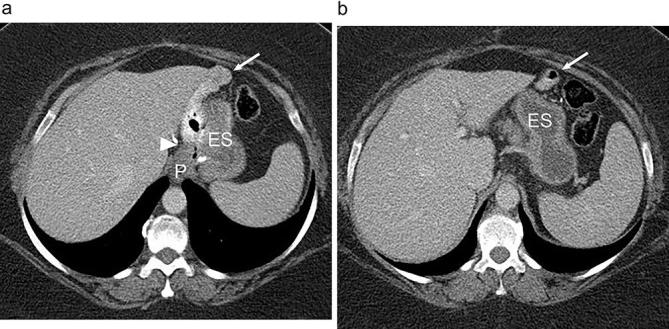
Hiatal Hernia & LSG
Hiatal hernia is present in 20-40% of bariatric surgery candidates (often asymptomatic and only discovered intraoperatively). During LSG, concomitant hiatal hernia repair (posterior cruroplasty with non-absorbable sutures) is strongly recommended, as unrepaired hiatal hernia significantly increases postoperative GERD risk. The dissection of the left crus during fundus mobilization provides excellent exposure for hiatal hernia repair. Some centers routinely perform posterior cruroplasty even without an obvious hernia defect as a prophylactic measure against de novo GERD.
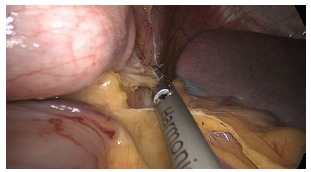
Leak After Sleeve Gastrectomy
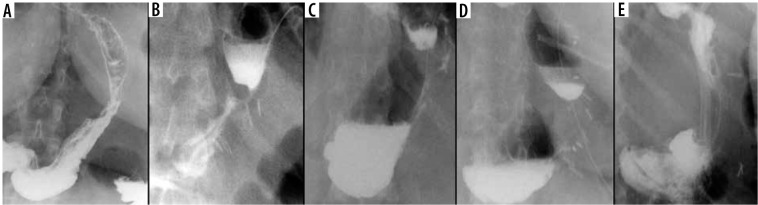
The staple-line leak after LSG occurs most commonly at the proximal sleeve near the GEJ (85-90% of leaks). The mechanism is typically ischemia (the proximal staple line is the most distal point from the remaining blood supply) combined with increased intraluminal pressure (the narrow tubular sleeve, especially at the incisura, creates a high-pressure zone that directs force proximally toward the GEJ). Leaks after LSG are classified by timing: early (POD 1-3, usually technical — staple misfires), intermediate (POD 4-7, usually ischemic), and late (POD > 7, usually related to ischemia and tissue necrosis). Late sleeve leaks at the GEJ are notoriously resistant to healing and may require months of drainage, stenting, and ultimately conversion to RYGB.
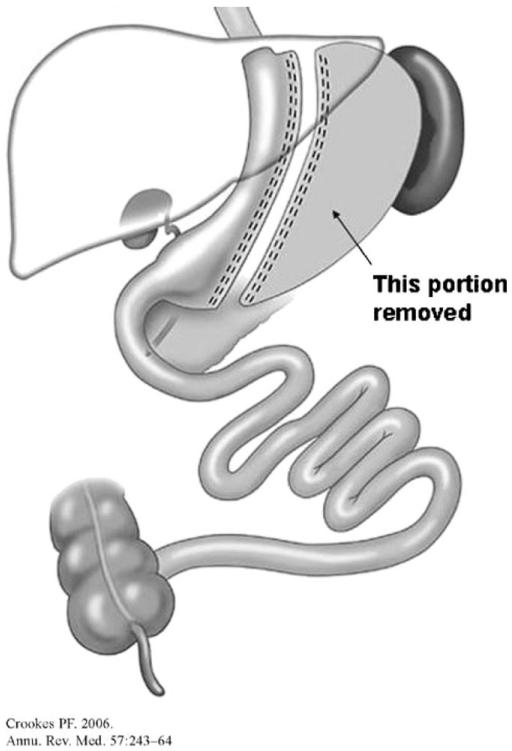
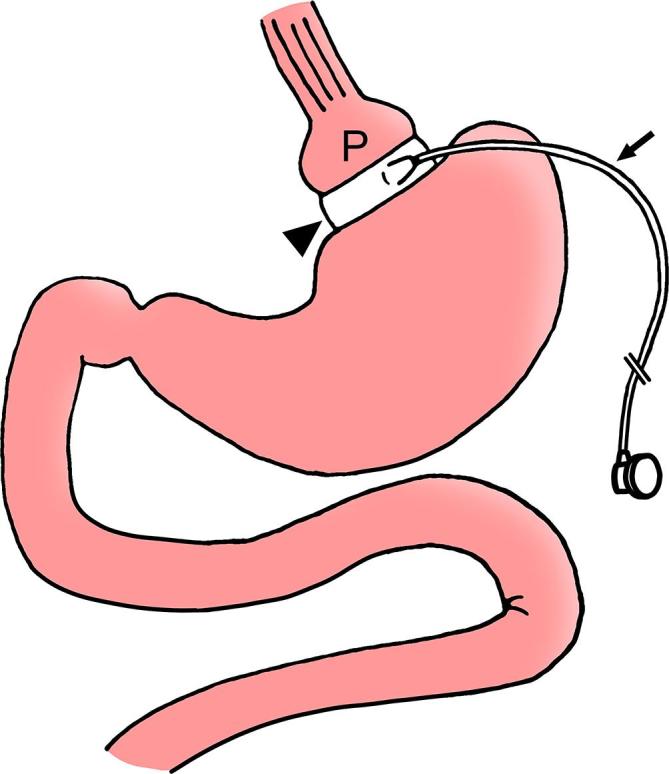
10 One Anastomosis Gastric Bypass (OAGB/MGB)
Surgical Technique
The One Anastomosis Gastric Bypass (also known as Mini Gastric Bypass, MGB) is a simplified bypass with a single anastomosis. Key steps:
- Long gastric pouch: A narrow, tubular pouch is created along the lesser curvature, approximately 15-18 cm in length (longer than the RYGB pouch), from the crow's foot (junction of the nerve of Latarjet branches) to the angle of His.
- Loop gastrojejunostomy: A loop of jejunum (measured 150-250 cm from the ligament of Treitz — the afferent/biliopancreatic limb) is anastomosed to the gastric pouch in an antecolic fashion. This single anastomosis avoids the need for a Roux limb and jejunojejunostomy.
Bile Reflux Concern
The primary criticism of OAGB is the theoretical risk of bile reflux into the gastric pouch and esophagus (since there is no Roux limb to divert bile). However, the long tubular pouch design minimizes this risk, and multiple large series have shown clinically significant bile reflux in < 1-2% of patients. The afferent limb of 150-250 cm creates sufficient distance to reduce bile exposure. Some centers perform routine EGD surveillance to monitor for bile gastritis or Barrett's-like changes.
Outcomes
%EWL at 5 years: 60-75% (comparable to RYGB). T2DM remission: 75-85%. Mortality: 0.1-0.2%. Advantages: shorter operative time, single anastomosis (fewer leak sites), easier revisional potential. OAGB is endorsed by IFSO as a standard procedure (IFSO position statement, 2018; PMID: 29327301).
OAGB vs RYGB — Comparative Summary
| Parameter | OAGB | RYGB |
|---|---|---|
| Anastomoses | 1 (gastrojejunostomy only) | 2 (GJ + JJ) |
| Operative time | 60-90 minutes | 90-150 minutes |
| Leak rate | 0.5-1.5% | 1-5% |
| Internal hernia risk | Lower (fewer mesenteric defects) | 2-9% without defect closure |
| Bile reflux risk | Theoretical concern (1-2% clinical significance) | Minimal (Roux limb diverts bile) |
| Nutritional deficiency | Similar to RYGB or slightly higher (longer BP limb) | Moderate (standard limb lengths) |
| Revisional potential | Easier (single anastomosis to revise or convert) | More complex (two anastomoses) |
| Long-term data | Less mature (< 15 years of large-series data) | Extensive (> 30 years of follow-up data) |
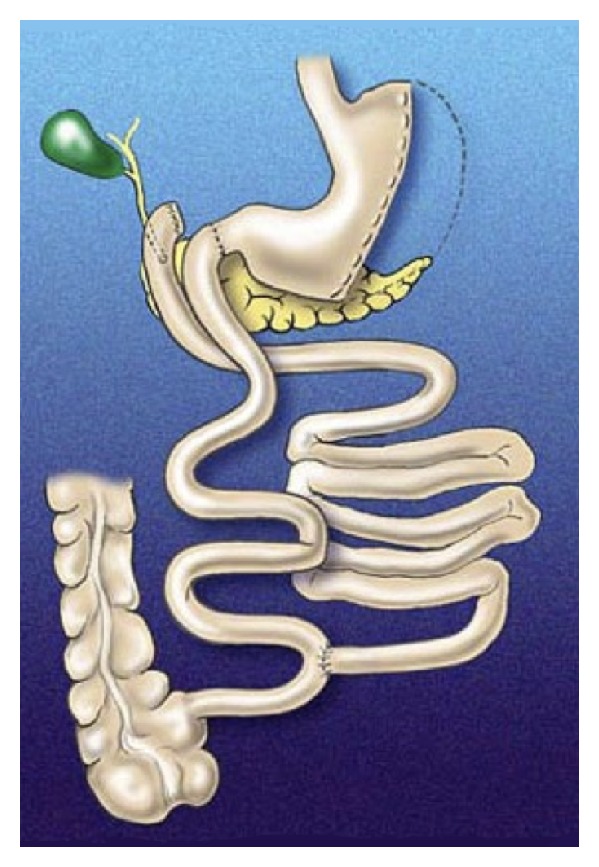
11 BPD/DS & SADI-S
Classic Biliopancreatic Diversion with Duodenal Switch (BPD/DS)
The BPD/DS is the most powerful bariatric procedure for weight loss and metabolic improvement but carries the highest nutritional risk. Originally described by Scopinaro (BPD) and modified by Hess and Marceau (DS).
- Sleeve gastrectomy: Performed as the first component (as described in section 09).
- Duodenal transection: The duodenum is divided 2-4 cm distal to the pylorus (preserving the pylorus — critical difference from Scopinaro BPD).
- Alimentary limb: The ileum is measured 250 cm proximally from the ileocecal valve; at this point the ileum is divided, and the distal end is anastomosed to the proximal duodenal stump (duodenoileostomy).
- Common channel: Only 75-100 cm of terminal ileum constitutes the common channel (where food mixes with bile/pancreatic secretions).
- Biliopancreatic limb: The proximal end of the divided ileum is anastomosed to the alimentary limb 75-100 cm proximal to the ileocecal valve (ileoileostomy).
SADI-S (Single Anastomosis Duodenoileal Bypass with Sleeve)
The SADI-S is a simplified modification using a loop duodenoileostomy (single anastomosis) instead of the Roux-en-Y configuration. The common channel is typically 250-300 cm (longer than classic BPD/DS, reducing malnutrition risk while maintaining excellent metabolic outcomes). ASMBS endorsed SADI-S as a standard bariatric procedure in 2020 (PMID: 32440366).
Outcomes
BPD/DS: %EWL at 5 years: 70-80%; T2DM remission: 90-98% (the highest of any procedure). However, protein malnutrition occurs in 5-15%, fat-soluble vitamin (A, D, E, K) deficiency in 50-70%, and calcium/vitamin D deficiency leading to metabolic bone disease in 25-50%. Steatorrhea and malodorous flatulence are common. BPD/DS is typically reserved for patients with BMI ≥ 50 or those with severe metabolic disease requiring maximal intervention.
12 Adjustable Gastric Banding (LAGB)
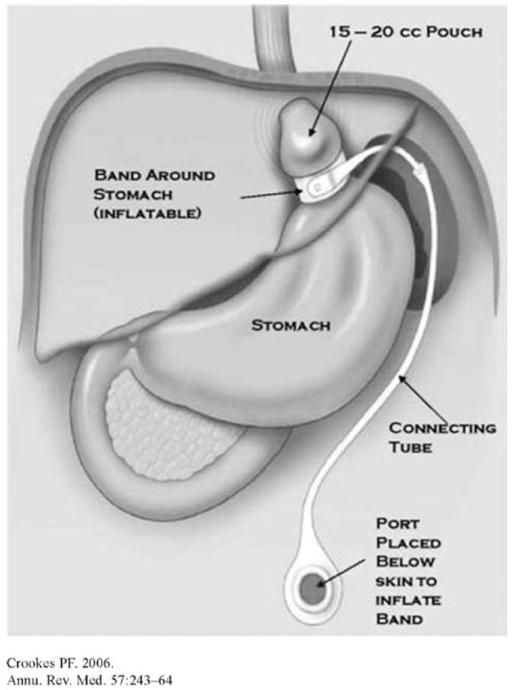
Overview
The laparoscopic adjustable gastric band (LAGB) — marketed as the Lap-Band or Realize band — was one of the most popular bariatric procedures from 2000-2010 but has declined dramatically (< 1% of bariatric procedures in 2023 in the US and Europe). The procedure involves placing an inflatable silicone band around the proximal stomach (at the angle of His), creating a small proximal pouch above the band. The band is connected via tubing to a subcutaneous access port, allowing percutaneous adjustments (saline injection to tighten, removal to loosen).
Complications & Reasons for Decline
| Complication | Incidence | Management |
|---|---|---|
| Band slippage / prolapse | 5-15% | Band deflation; operative repositioning or removal |
| Band erosion (into gastric lumen) | 1-3% | Endoscopic or surgical band removal |
| Pouch dilation | 10-20% | Band deflation; dietary counseling; conversion to sleeve or RYGB |
| Port complications (flipping, infection, tubing disconnect) | 5-10% | Port revision or replacement |
| Dysphagia / food intolerance | 20-40% | Band adjustment (deflation) |
| Esophageal dilation | 5-15% | Band deflation; monitor; may require removal |
| Weight regain / inadequate loss | 30-50% | Conversion to RYGB or LSG |
Long-term reoperation rate: 30-60% (the highest of any bariatric procedure). Band removal rate: 25-50% by 10 years. For these reasons, LAGB is no longer recommended as a primary bariatric procedure by most centers and guidelines. The most common conversion is LAGB-to-LSG (single stage) or LAGB-to-RYGB.
13 Revisional & Conversion Surgery
Indications for Revisional Surgery
Revisional bariatric surgery is performed for: inadequate weight loss (< 50% EWL or < 20% TWL), significant weight regain (regain of > 25% of lost weight), complications of the index procedure (chronic leak, stricture, refractory GERD, band complications), or metabolic disease recurrence (T2DM relapse). Revisional surgery accounts for approximately 10-15% of all bariatric procedures and carries higher complication rates (15-30%) than primary procedures.
Common Conversions
| Index Procedure | Conversion Options | Key Considerations |
|---|---|---|
| LAGB | LSG (most common), RYGB, OAGB | Band removal + conversion can be single or two-stage; assess for esophageal dilation |
| LSG (inadequate loss) | RYGB, OAGB, SADI-S, Re-sleeve | RYGB is most common conversion; re-sleeve only if dilated sleeve with no GERD |
| LSG (severe GERD) | RYGB (gold standard) | RYGB reliably resolves post-sleeve GERD in > 90%; Conversion to RYGB preferred over long-term PPI |
| RYGB (weight regain) | Pouch/stoma revision, distalization (lengthening BP limb), conversion to BPD/DS | TORe (transoral outlet reduction via endoscopy) for dilated GJ stoma (> 2 cm) |
| RYGB (marginal ulcer) | Revision of GJ, resection of ulcer bed, truncal vagotomy | Rule out H. pylori, NSAID use, smoking, ischemia, foreign body (suture/staple) |
| VBG (vertical banded gastroplasty) | RYGB, LSG | Historical procedure; complex adhesions expected |
Technical Challenges
Revisional surgery is technically demanding due to: altered anatomy, dense adhesions, fibrosis at prior staple lines, reduced tissue vascularity, and distorted landmarks. Preoperative upper GI series and/or EGD are mandatory. Complication rates for revisional surgery are approximately 2-3 times higher than for primary procedures. A 2-stage approach (e.g., band removal, then conversion 3-6 months later) is safer in complex cases, especially when significant inflammation or nutritional deficiency is present.
Weight Regain Workup Algorithm
When a post-bariatric patient presents with significant weight regain, a systematic evaluation is required:
- Behavioral assessment: Dietary recall (caloric intake, grazing patterns, liquid calories, return to sweets/carbohydrates); exercise habits; eating disorder screening (binge eating relapse); psychological assessment (depression, stress eating); support group attendance
- Anatomic evaluation: UGI series (pouch size, stoma diameter, staple line integrity, gastrogastric fistula); EGD (stoma measurement — dilated if > 2 cm; marginal ulcer; gastrogastric fistula)
- Hormonal/metabolic evaluation: Thyroid function (TSH, free T4); HbA1c; cortisol (if Cushing's suspected); screen for medications causing weight gain (antipsychotics, corticosteroids, insulin, sulfonylureas, beta-blockers)
- Intervention ladder: Dietary counseling and behavioral modification → pharmacotherapy (GLP-1 RA as adjunct — increasingly used) → endoscopic revision (TORe for dilated stoma) → surgical revision (pouch revision, distalization, or conversion to more malabsorptive procedure)
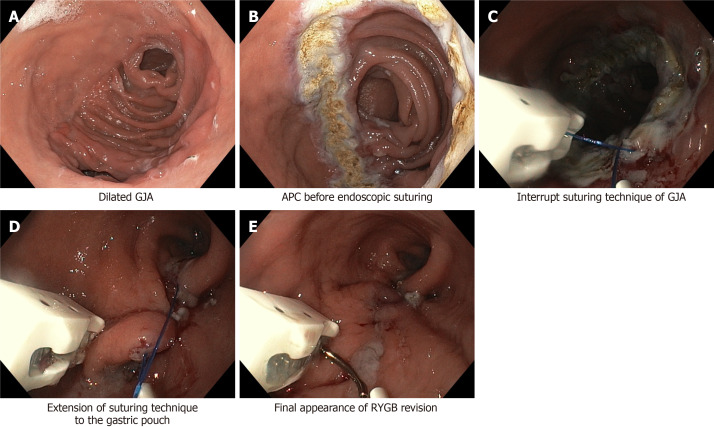
14 Endoscopic Bariatric Therapies
Intragastric Balloons (IGB)
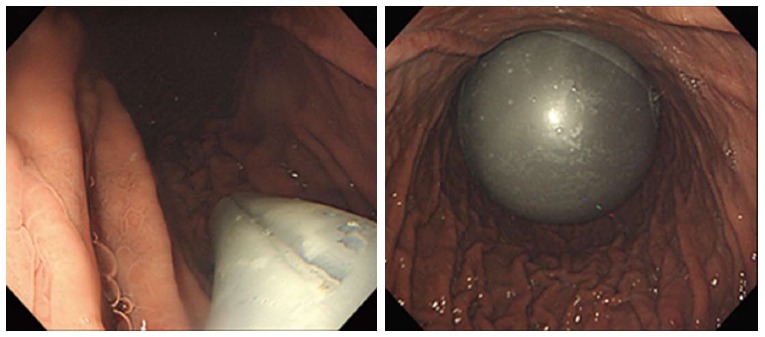
Orbera (single fluid-filled balloon, 400-700 mL saline; placed for 6 months), Obalon (swallowable gas-filled capsules, up to 3 balloons), and Spatz (adjustable balloon, 12-month duration). Average %TWL: 10-15% at 6 months. Complications: nausea/vomiting (30-50% in first week), balloon deflation/migration (1-3%, risk of bowel obstruction), gastric perforation (0.1-0.3%), pancreatitis (rare). IGBs are typically used as a bridge to surgery in high-BMI patients, for patients unwilling or unfit for surgery, or for temporary preoperative weight loss.
Endoscopic Sleeve Gastroplasty (ESG)
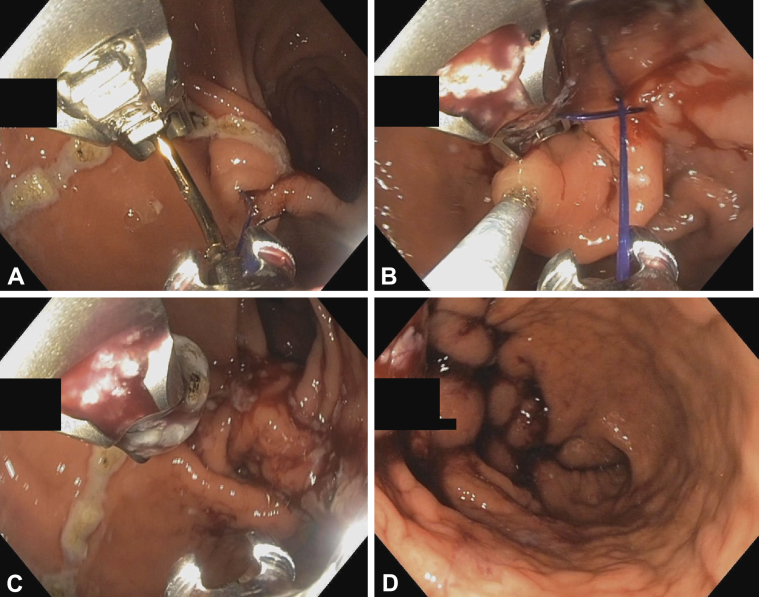
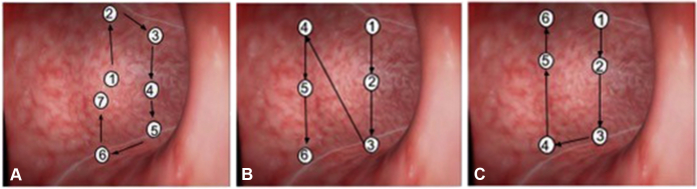
ESG uses a full-thickness endoscopic suturing device (OverStitch, Apollo Endosurgery) to create a series of interrupted or running sutures along the greater curvature of the stomach, reducing gastric volume by ~70%. Average %TWL: 15-20% at 1 year, 15-18% at 2 years. Advantages: outpatient procedure, no incisions, shorter recovery (2-3 days vs 1-2 weeks for LSG), lower complication rate (< 2% serious adverse events). Disadvantages: less durable weight loss than surgical options, limited long-term data (> 5 years), and not covered by most insurers. ESG is emerging as an option for patients with BMI 30-40 who prefer a less invasive approach (Abu Dayyeh et al., PMID: 27567955).
Intragastric Balloon Complications & Management
| Complication | Incidence | Presentation | Management |
|---|---|---|---|
| Accommodation syndrome | 30-50% | Severe nausea, vomiting, abdominal pain in first 3-7 days | Antiemetics (ondansetron, scopolamine), antispasmodics, PPI; usually self-limited |
| Balloon deflation | 1-3% | Sudden loss of restriction; green/blue urine (methylene blue dye leaks from balloon as safety indicator) | Urgent endoscopic retrieval — deflated balloon can migrate distally and cause SBO |
| Gastric ulcer/erosion | 2-5% | Epigastric pain, hematemesis | Balloon removal; PPI therapy |
| Gastric perforation | 0.1-0.3% | Acute abdomen, peritonitis | Emergency Surgical repair; balloon removal |
| Pancreatitis | < 0.5% | Epigastric pain radiating to back, elevated lipase | Balloon repositioning or removal; supportive care |
| Spontaneous hyperinflation | Rare (Obalon gas-filled) | Acute gastric outlet obstruction | Emergency endoscopic deflation and retrieval |
Gastric Electrical Stimulation
Gastric electrical stimulation (GES) involves implantation of electrodes on the gastric serosa connected to a pulse generator (similar to a cardiac pacemaker). The MAESTRO Rechargeable System (vBloc) uses vagal nerve blockade to reduce hunger signals — FDA approved in 2015 for BMI 40-45 or BMI 35-39.9 with one comorbidity. Results are modest: 8-10% EWL greater than sham at 12 months (ReCharge trial). The device has not gained widespread adoption due to modest efficacy and frequent need for recharging. GES for gastroparesis (Enterra device) is a related but distinct technology.
Other Endoscopic Interventions
AspireAssist — a percutaneous gastrostomy tube that allows drainage of a portion of gastric contents after meals; FDA approved in 2016 but rarely used due to poor patient acceptance. Duodenal mucosal resurfacing (DMR) — thermal ablation of the duodenal mucosa to alter incretin signaling; investigational. Transpyloric shuttle — a device that delays gastric emptying; limited data. Primary obesity surgery endoluminal (POSE) — creates folds in the gastric fundus and body; largely superseded by ESG.
15 Early Postoperative Complications
Postoperative Day 1 Management Protocol
Standard postoperative management on the first day after bariatric surgery:
| System | Assessment/Intervention | Red Flags |
|---|---|---|
| Cardiovascular | Heart rate monitoring q4h; target HR < 100 bpm; assess for orthostatic hypotension | Persistent HR > 120 (leak until proven otherwise); new arrhythmia; hypotension unresponsive to fluids |
| Respiratory | Incentive spirometry q1h while awake; head of bed ≥ 30°; CPAP for OSA patients; SpO2 monitoring | SpO2 < 92% on room air; increasing O2 requirements; labored breathing |
| GI | Clear liquid sips (30 mL/15 min); assess for nausea/vomiting; check drain output (if placed) | Bilious or feculent drain output; inability to tolerate any liquids; abdominal distension |
| Renal | Strict I/O; target urine output ≥ 0.5 mL/kg/h (use ideal body weight); check BMP | Oliguria (< 30 mL/h); dark urine (rhabdomyolysis); hyperkalemia; rising creatinine |
| VTE | SCDs on both legs; initiate LMWH 6-12h postop; early ambulation (within 2-4 hours) | Unilateral leg swelling; calf tenderness; acute dyspnea or pleuritic chest pain (PE) |
| Pain | Multimodal: acetaminophen 1g IV/PO q6h + gabapentin 300 mg PO BID; minimize opioids | Pain out of proportion to expected (leak, rhabdomyolysis); shoulder pain (diaphragmatic irritation from leak) |
| Glucose | POC glucose q6h for all patients; insulin protocol for T2DM patients (reduced doses) | Hypoglycemia in T2DM patients (medication doses may need immediate reduction) |
Anastomotic / Staple-Line Leak
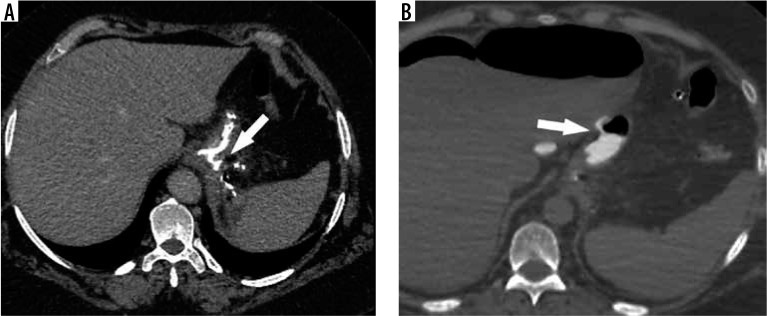
Emergency The most feared early complication. Incidence: RYGB 1-5% (most at the GJ anastomosis), LSG 1-3% (most at the GEJ/upper sleeve), BPD/DS 2-5%. Leak typically presents on POD 1-7 but can be delayed. Mortality from uncontrolled leak: 10-40%.
| Parameter | Detail |
|---|---|
| Presentation | Tachycardia (> 120 bpm — most sensitive early sign), fever, tachypnea, left shoulder pain, abdominal pain (may be absent in obese patients), oliguria, leukocytosis, elevated CRP/procalcitonin |
| Diagnosis | CT abdomen/pelvis with oral water-soluble contrast (sensitivity 75-90%); upper GI series with Gastrografin (sensitivity 50-75%); clinical suspicion often sufficient for surgical exploration |
| Management — acute (< 72h) | Operative re-exploration: washout, repair if feasible, wide drainage, NG decompression. Consider controlled fistula approach with drain placement near the leak |
| Management — subacute (3-14d) | Interventional radiology-guided percutaneous drainage; NPO with TPN; endoscopic stent placement (fully covered metal stent, 4-8 weeks); endoscopic vacuum therapy (Endo-SPONGE) |
| Management — chronic (> 14d) | Endoscopic stenting; septotomy; fistulojejunostomy; Roux-en-Y drainage. Chronic sleeve leaks at the GEJ are notoriously difficult — may require conversion to RYGB |
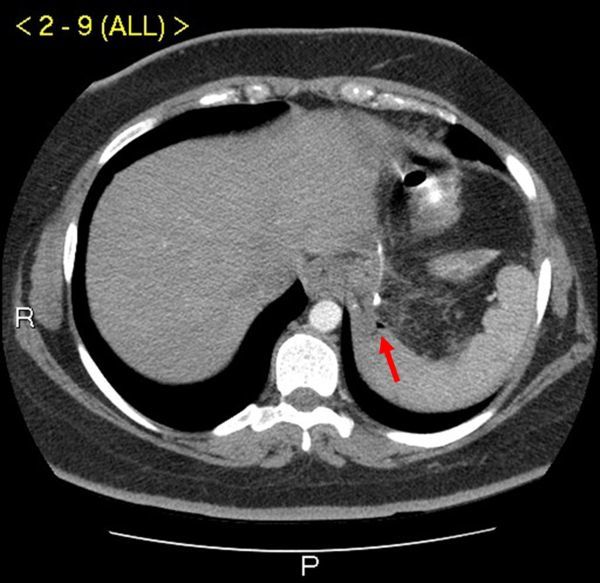
Hemorrhage
Postoperative bleeding occurs in 1-4% of cases and can be intraluminal (GI bleeding from staple lines — presents with hematemesis, melena, or tachycardia/hypotension) or extraluminal (intraabdominal bleeding from staple lines, omental vessels, trocar sites, or spleen — presents with tachycardia, falling Hb, drain output). Management: resuscitation, correction of coagulopathy (reverse any anticoagulation), EGD for intraluminal bleeding (clipping or epinephrine injection), and operative exploration for hemodynamically significant extraluminal bleeding or failure of endoscopic therapy.
Venous Thromboembolism (PE/DVT)
Emergency PE is a leading cause of death after bariatric surgery (accounting for 30-50% of perioperative mortality). Risk factors: BMI > 50, prior VTE history, immobility, OHS, hypercoagulable state. The 30-day PE rate is 0.3-1.0%. Prophylaxis is critical:
- Mechanical: SCDs (sequential compression devices) intraoperatively and until fully ambulatory
- Pharmacological: Enoxaparin 40 mg SQ BID (weight-based dosing for BMI > 40), or heparin 5000 units SQ TID; start 2-6 hours postop
- Extended prophylaxis: Consider 2-4 weeks of LMWH after discharge for high-risk patients (prior VTE, BMI > 50, immobility)
- IVC filter: Not routinely recommended; consider only for patients with recent (< 3 month) VTE who cannot be anticoagulated
Portomesenteric Vein Thrombosis
Portomesenteric vein thrombosis (PMVT) is a rare but serious complication occurring in 0.3-1.0% of patients after LSG (less common after RYGB). Risk factors include dehydration, systemic inflammatory response from surgery, and compression of portal tributaries during sleeve creation. Presentation is often delayed (POD 5-14): severe epigastric or diffuse abdominal pain, nausea, vomiting, and elevated liver enzymes. Diagnosis: CT angiography showing filling defects in the portal vein, superior mesenteric vein, or splenic vein. Treatment: systemic anticoagulation with heparin (bridging to warfarin or DOAC) for 3-6 months. Early diagnosis is critical — delayed treatment can lead to mesenteric ischemia, bowel infarction, and death. Thrombophilia workup should be considered in confirmed cases.
Marginal Ulcer Perforation
Emergency Perforation of a marginal ulcer occurs in 1-2% of post-RYGB patients with marginal ulcers. Presents with acute-onset severe epigastric pain, peritonitis (rigid abdomen, guarding), tachycardia, and free air on upright CXR or CT. Management: emergent operative exploration with omental patch repair (Graham patch), washout, and wide drainage. After stabilization, the underlying cause must be addressed: smoking cessation, NSAID avoidance, H. pylori eradication, and high-dose PPI therapy. Recurrent perforated marginal ulcers despite medical optimization may require surgical revision of the GJ with truncal vagotomy.
Bowel Obstruction
Small bowel obstruction (SBO) after bariatric surgery occurs in 2-5% of RYGB patients and < 1% of LSG patients. Causes include: internal hernia (most common cause after RYGB — 50-70% of SBO cases), adhesive band, port-site hernia (especially at the 12-15 mm port site — routine fascial closure of all port sites ≥ 10 mm is recommended), and jejunojejunostomy stricture. Unique to bariatric patients: SBO can present atypically — abdominal distension is less apparent due to body habitus, and pain may be poorly localized. CT sensitivity for SBO is lower in obese patients due to image degradation. A high index of suspicion and low threshold for diagnostic laparoscopy are essential.
Respiratory Complications
Atelectasis is the most common postoperative respiratory complication (incidence 20-40%), particularly in patients with BMI > 50 and OHS. Risk factors: supine positioning (reduces FRC by 50% in obese patients), general anesthesia, pain-limited deep breathing. Prevention: early mobilization (upright positioning within 2-4 hours), incentive spirometry, CPAP/BiPAP in patients with OSA. Aspiration pneumonia is a concern due to increased intra-abdominal pressure, delayed gastric emptying, and GERD. Postoperative respiratory failure requiring reintubation occurs in 1-3% and is associated with unrecognized OHS, over-sedation (increased opioid sensitivity), and OSA.
Rhabdomyolysis
Rhabdomyolysis occurs in 1-7% of bariatric surgery patients (especially those with BMI > 50 and prolonged operative times > 4 hours). Risk factors: supine positioning with pressure on gluteal muscles, prolonged operative time, and male sex. Presentation: gluteal/back pain, dark urine, elevated CK (> 5000 U/L). Prevention: adequate padding, limiting operative time, and early ambulation. Treatment: aggressive IV fluid resuscitation (target urine output > 200 mL/h), alkalization of urine (sodium bicarbonate), and monitoring for acute kidney injury.
16 Late Complications
Marginal Ulcer
Marginal ulcers occur at or near the gastrojejunal (GJ) anastomosis after RYGB in 1-16% of patients. Risk factors: smoking (most significant modifiable risk factor — 3-5x increased risk), NSAID use, large pouch with parietal cells, H. pylori infection, ischemia at the anastomosis, and foreign body (exposed staples or sutures). Presentation: epigastric pain, nausea, GI bleeding (hematemesis, melena), or perforation. Diagnosis: EGD (gold standard). Management: PPI therapy (high-dose, 40 mg BID for 8-12 weeks), smoking cessation, discontinue NSAIDs, H. pylori eradication if positive. Refractory ulcers may require surgical revision (resection of the ulcer bed, recreation of the GJ, truncal vagotomy).
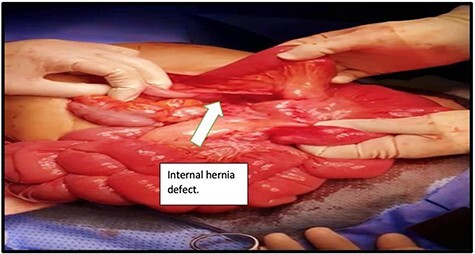
Internal Hernia
Internal hernias are the most common cause of small bowel obstruction after RYGB (incidence 2-9% with laparoscopic approach, lower with routine mesenteric defect closure). Two primary defects:
- Petersen's space: The space between the Roux limb mesentery and the transverse mesocolon (in retrocolic RYGB) or between the Roux limb mesentery and the retroperitoneum (in antecolic RYGB)
- Jejunojejunostomy (JJ) mesenteric defect: The defect in the mesentery at the JJ anastomosis
Presentation: intermittent or acute abdominal pain (often postprandial), nausea/vomiting, SBO. CT findings: the "swirl sign" (mesenteric whirl), clustered small bowel loops, and mesenteric edema. Internal hernia can progress to bowel strangulation and necrosis if not recognized. Treatment: emergent/urgent laparoscopic exploration and reduction with defect closure. Even with negative CT but high clinical suspicion, diagnostic laparoscopy is indicated.
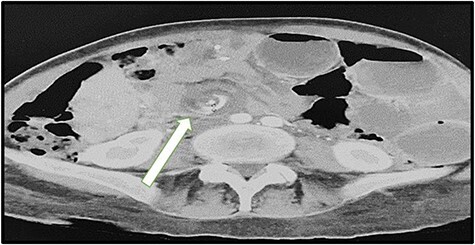
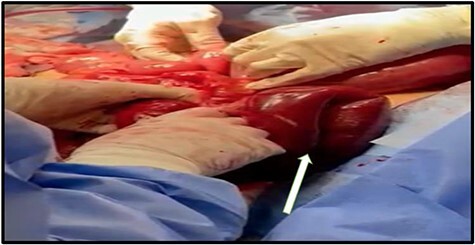
Stricture
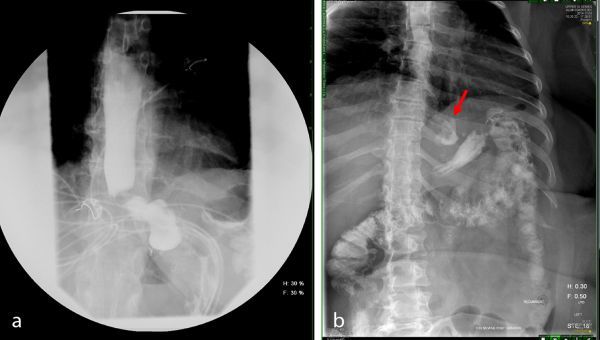
Anastomotic stricture occurs in 3-8% of RYGB patients (at the GJ) and 0.5-3% of LSG patients (at the incisura angularis). Presents at 3-12 weeks as progressive dysphagia, nausea, vomiting. Diagnosis: UGI series and/or EGD. Treatment: endoscopic balloon dilation (typically 12-15 mm TTS balloon, serial dilations may be needed). Strictures refractory to 3 endoscopic dilations may require surgical revision.
GERD After Sleeve Gastrectomy
De novo GERD after LSG develops in 15-35% of patients. Mechanisms: destruction of the angle of His (antireflux mechanism), increased intragastric pressure (in a narrow sleeve), LES dysfunction, and hiatal hernia (often unrecognized at index operation). Long-term GERD after sleeve may progress to erosive esophagitis (LA grade C/D) and Barrett's esophagus. Management: PPI therapy; hiatal hernia repair (if present); if refractory, conversion to RYGB (definitive solution, resolves GERD in > 90%).
Gastrogastric Fistula
A gastrogastric fistula (GGF) is an abnormal communication between the gastric pouch and the excluded gastric remnant after RYGB. Incidence: 1-6% (higher with undivided gastric partition, which is no longer performed). GGF allows acid and food from the pouch to enter the remnant, causing weight regain (the remnant re-enters the digestive circuit, expanding effective gastric volume), marginal ulceration (acid exposure at the GJ), and GERD. Diagnosis: UGI series shows contrast flowing from pouch to remnant; EGD may visualize the fistula tract. Treatment: surgical excision of the fistula tract with re-separation and reinforcement of the pouch-remnant partition. Endoscopic closure (clips, suturing) has limited success for large fistulae.
Chronic Nausea & Vomiting
Persistent nausea and vomiting after bariatric surgery (beyond the initial postoperative period) affects 5-15% of patients. The differential diagnosis includes: stricture (most common — at GJ after RYGB or incisura after LSG), marginal ulcer, internal hernia (intermittent SBO), GERD, food intolerance (often to specific proteins like red meat), gastric dysmotility (gastroparesis of the remnant stomach or sleeve), small bowel bacterial overgrowth (SIBO) (from blind loop/bypassed limbs — presents with bloating, diarrhea, B12 deficiency), gallstone disease, and adhesive disease. Workup: EGD, UGI series, CT abdomen, gastric emptying study, and trial of dietary modification. Treatment depends on etiology.
Cholelithiasis
Rapid weight loss is the strongest risk factor for gallstone formation — incidence is approximately 30-40% within the first year after bariatric surgery without prophylaxis. Gallstone formation results from supersaturation of bile with cholesterol during mobilization of adipose stores. Ursodiol prophylaxis (300 mg PO BID for 6 months postoperatively) reduces the incidence to < 2% and is recommended by ASMBS guidelines. Routine concomitant cholecystectomy is no longer recommended (only 10-15% of gallstones become symptomatic). Cholecystectomy should be performed for symptomatic cholelithiasis or choledocholithiasis. In post-RYGB patients, ERCP is technically challenging due to altered anatomy — options include laparoscopic-assisted transgastric ERCP, double-balloon enteroscopy-assisted ERCP, or laparoscopic common bile duct exploration.
Metabolic Bone Disease
Long-term bone health is a major concern after malabsorptive procedures. Mechanisms include: calcium malabsorption (bypassed duodenum — primary calcium absorption site), vitamin D deficiency (fat-soluble vitamin malabsorption), secondary hyperparathyroidism (PTH elevation in response to hypocalcemia drives bone resorption), and altered bone metabolism markers (elevated CTX, decreased osteocalcin). DXA bone density scanning shows 5-10% decrease in bone mineral density at the hip and femoral neck within 2 years of RYGB, and greater losses after BPD/DS. Fracture risk is increased 1.3-2.0 fold after RYGB compared to age-matched obese controls. Prevention: adequate calcium citrate supplementation, vitamin D repletion (target 25-OH D > 30 ng/mL), weight-bearing exercise, and DXA monitoring.
Hair Loss (Telogen Effluvium)
Telogen effluvium — diffuse, non-scarring hair loss — occurs in 30-50% of bariatric surgery patients, typically beginning 3-6 months postoperatively and peaking at 6-9 months. The rapid weight loss and nutritional changes push hair follicles from the growth phase (anagen) into the resting phase (telogen) prematurely. Contributing factors include protein deficiency (the most important), iron deficiency, zinc deficiency, and biotin deficiency. Management: ensure adequate protein intake (≥ 60-80 g/day), check and correct iron, zinc, and biotin levels, and reassure the patient (hair loss is typically self-limited and resolves by 12-18 months postop as weight stabilizes and nutrition normalizes). Minoxidil is rarely needed.
Kidney Stones (Nephrolithiasis)
Hyperoxaluria after RYGB increases the risk of calcium oxalate nephrolithiasis by 2-3 fold. Mechanism: unabsorbed fatty acids in the colon bind to calcium (which would normally bind oxalate), leaving free oxalate to be absorbed — enteric hyperoxaluria. Urine oxalate levels increase by 40-100% after RYGB. Prevention: adequate calcium supplementation (calcium binds dietary oxalate in the gut), hydration (> 2L/day), and low-oxalate diet (avoid spinach, rhubarb, chocolate, nuts, tea). 24-hour urine oxalate monitoring is recommended for symptomatic patients.
17 Dumping Syndrome
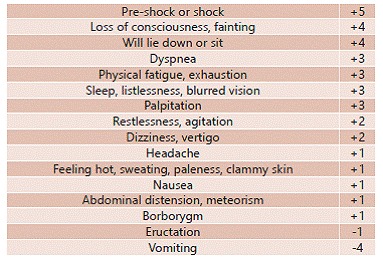
Early Dumping
Early dumping syndrome occurs 10-30 minutes after eating, primarily after RYGB (10-40% of patients). Pathophysiology: rapid emptying of hyperosmolar food from the gastric pouch into the jejunum causes fluid shifts into the intestinal lumen (osmotic effect) and release of vasoactive mediators (serotonin, bradykinin, VIP). Symptoms: nausea, crampy abdominal pain, diarrhea, bloating, diaphoresis, flushing, tachycardia, dizziness, and vasomotor collapse (in severe cases).
Late Dumping (Reactive Hypoglycemia)
Late dumping occurs 1-3 hours after eating, particularly after carbohydrate-rich meals. Pathophysiology: rapid glucose absorption causes hyperglycemia → exaggerated GLP-1 and insulin secretion → reactive hypoglycemia (blood glucose < 50 mg/dL). Symptoms: diaphoresis, tremor, confusion, weakness, syncope. Severe cases can cause loss of consciousness and seizures. Post-bariatric hyperinsulinemic hypoglycemia (PBHH), also known as nesidioblastosis, is a rare (< 1%) but severe form of late dumping with recurrent neuroglycopenic episodes.
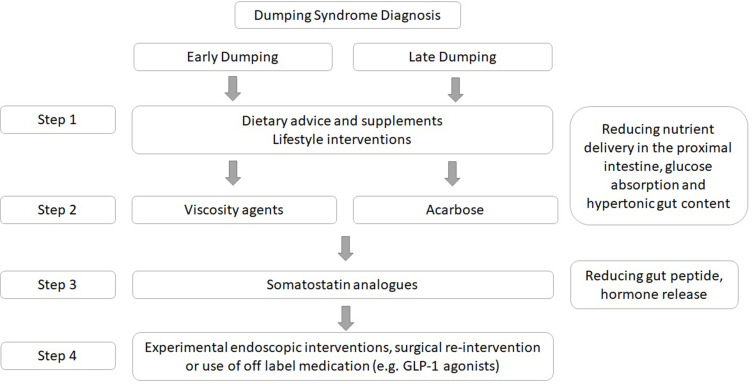
Management
| Strategy | Detail |
|---|---|
| Dietary modification (first-line) | Small frequent meals (5-6/day); avoid simple sugars; increase protein and complex carbohydrates; eat solids before liquids; avoid drinking with meals |
| Acarbose | Alpha-glucosidase inhibitor (50-100 mg with meals); delays carbohydrate absorption; most effective for late dumping |
| Octreotide | Somatostatin analogue (50-100 µg SQ before meals or long-acting 20-30 mg IM monthly); inhibits gut hormone release; for severe refractory cases |
| Diazoxide | Potassium channel opener; inhibits insulin secretion; for severe PBHH |
| Continuous glucose monitoring | Useful for documenting hypoglycemic episodes in PBHH |
| Surgical intervention | Roux limb reversal, gastric pouch restriction, or subtotal pancreatectomy — reserved for refractory neuroglycopenic PBHH |
18 Nutritional Emergencies
Wernicke Encephalopathy (Thiamine/B1 Deficiency)
Emergency Wernicke encephalopathy is the most dangerous nutritional emergency after bariatric surgery. Incidence: 0.5-1% but likely underdiagnosed. Typically occurs weeks to months postoperatively, especially in patients with prolonged vomiting, poor oral intake, or IV dextrose administration without thiamine supplementation.
The classic triad (present in only 16% of cases): encephalopathy (confusion, apathy, altered consciousness), oculomotor dysfunction (nystagmus, lateral rectus palsy, conjugate gaze palsy), and ataxia (cerebellar dysfunction, wide-based gait). Untreated Wernicke encephalopathy can progress to Korsakoff syndrome (irreversible anterograde and retrograde amnesia with confabulation).
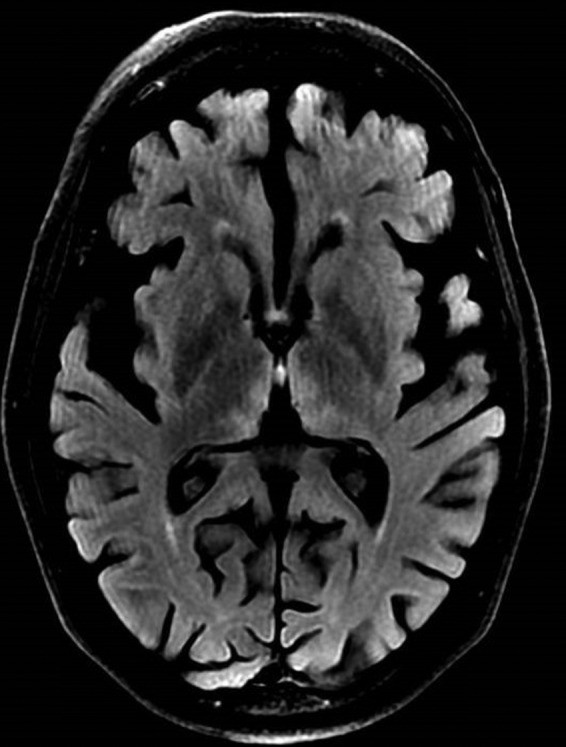
Treatment — URGENT: IV thiamine 200-500 mg TID for 3-5 days, followed by oral thiamine 100 mg TID. Administer thiamine BEFORE glucose/dextrose (glucose metabolism consumes thiamine and can precipitate acute Wernicke in a depleted patient). MRI may show hyperintensity in the mammillary bodies and periaqueductal gray matter on T2/FLAIR sequences.
Copper Deficiency Myelopathy
Copper deficiency can develop months to years after bypass procedures (especially BPD/DS and RYGB), presenting as a clinical syndrome mimicking subacute combined degeneration (similar to B12 deficiency): myelopathy with posterior column dysfunction (loss of proprioception, sensory ataxia), peripheral neuropathy, and cytopenias (anemia, neutropenia). Diagnosis: serum copper < 70 µg/dL and ceruloplasmin < 20 mg/dL. Treatment: copper supplementation (2-4 mg/day oral or IV). Key pitfall: excessive zinc supplementation (commonly prescribed after bariatric surgery) competes with copper for intestinal absorption via metallothionein and is a major cause of iatrogenic copper deficiency.
Severe Protein Malnutrition
Occurs primarily after BPD/DS (5-15%) and long-limb RYGB. Presents with hypoalbuminemia (< 3.0 g/dL), edema, hair loss, poor wound healing, and immune dysfunction. Severe cases (albumin < 2.0) may require hospitalization for parenteral nutrition. Refractory protein malnutrition after BPD/DS may necessitate surgical revision (lengthening the common channel).
Vitamin A Deficiency
Vitamin A deficiency is most common after BPD/DS (incidence 50-70% without supplementation). Fat-soluble vitamin A requires bile salts for absorption — the short common channel in BPD/DS limits bile-nutrient mixing time. Night blindness (nyctalopia) is the earliest clinical manifestation, followed by xerophthalmia (dry eyes, Bitot's spots — foamy patches on the conjunctiva), keratomalacia (corneal ulceration), and ultimately irreversible blindness. Additionally, vitamin A deficiency impairs immune function (increased susceptibility to infections) and skin integrity (follicular hyperkeratosis). Treatment: oral vitamin A 50,000-100,000 IU/day until levels normalize; in severe cases, IM vitamin A. Prevention: 5,000-10,000 IU/day for BPD/DS patients.
Iron Deficiency Anemia
Iron deficiency is the most common long-term nutritional deficiency after bariatric surgery, affecting 20-55% of post-RYGB and 25-70% of post-BPD/DS patients. Mechanisms: bypassed duodenum (primary iron absorption site), reduced gastric acid (impairs conversion of non-heme Fe3+ to absorbable Fe2+), and reduced dietary intake of red meat. Premenopausal women are at highest risk. Oral iron must be taken with vitamin C (enhances absorption) and separated from calcium, PPIs, and antacids (which impair absorption). Refractory iron deficiency despite oral supplementation requires IV iron infusions (ferric carboxymaltose 750 mg IV × 2 doses, or iron sucrose 200-300 mg IV). Severe anemia (Hb < 7 g/dL) may require blood transfusion.
Folate Deficiency & Neural Tube Defects
Folate deficiency occurs in 5-15% of post-RYGB patients (the proximal jejunum, where folate is primarily absorbed, is bypassed). Folate deficiency is of particular concern in women of childbearing age due to the risk of neural tube defects (NTDs) — spina bifida and anencephaly — in offspring. All women of reproductive age after bariatric surgery should take folic acid supplementation (≥ 400 µg/day in standard MVI; 800-1000 µg/day is recommended by some guidelines; 4 mg/day if planning pregnancy or history of NTDs). RBC folate is a more accurate indicator of tissue folate stores than serum folate. Folate supplementation should begin preoperatively and continue lifelong.
Vitamin B12 Deficiency
B12 deficiency develops in 15-35% of post-RYGB patients by 3-5 years. Mechanism: the small gastric pouch produces minimal intrinsic factor (IF), and the bypassed duodenum is the primary site of IF-B12 complex absorption. Presentation includes megaloblastic anemia, glossitis, peripheral neuropathy (paresthesias in hands and feet), and cognitive impairment. Severe untreated B12 deficiency can cause subacute combined degeneration of the spinal cord (posterior and lateral column demyelination). Sublingual B12 (1000 µg/day) or IM B12 (1000 µg/month) is the standard prevention. Serum methylmalonic acid (MMA) is a more sensitive marker of tissue B12 deficiency than serum B12 levels alone.
19 Enhanced Recovery & Perioperative Protocols
ERAS Protocol for Bariatric Surgery
Enhanced Recovery After Surgery (ERAS) protocols have been widely adopted in bariatric surgery, reducing length of stay from 3-5 days to 1-2 days (many centers now perform LSG as same-day surgery). Key elements:
| Phase | Intervention |
|---|---|
| Preoperative | Patient education and expectation setting; clear liquids until 2h before induction; carbohydrate-rich drink 2-3h preop (reduces insulin resistance); preoperative analgesia (acetaminophen 1g PO, gabapentin 300-600 mg); anxiety reduction (avoid long-acting sedatives) |
| Intraoperative | Goal-directed fluid therapy (avoid overhydration — crystalloid < 2L); multimodal analgesia (minimize opioids; ketorolac 15-30 mg IV if no RYGB anastomosis concern; local anesthetic infiltration at port sites; TAP block); PONV prophylaxis (ondansetron 4 mg + dexamethasone 4-8 mg + scopolamine patch); normothermia (forced-air warming) |
| Postoperative | Early ambulation (within 2-4 hours); clear liquid diet POD0 (some centers); VTE prophylaxis (SCDs + LMWH); multimodal analgesia (scheduled acetaminophen, NSAIDs if appropriate, gabapentinoids; minimize opioids); avoid routine UGI swallow study (perform only for clinical concern); discharge criteria-based (not time-based) |
VTE Prophylaxis
Bariatric surgery patients are at elevated VTE risk due to obesity, immobility, venous stasis, and chronic inflammatory state. Standard prophylaxis combines mechanical (SCDs) and pharmacological (weight-based LMWH) measures. For high-risk patients (BMI > 50, prior VTE, known thrombophilia, OHS), extended chemoprophylaxis (2-4 weeks post-discharge) is recommended (Finks et al., PMID: 21247680).
Anesthetic Considerations for the Obese Patient
The morbidly obese patient presents unique anesthetic challenges:
| Challenge | Clinical Implication | Strategy |
|---|---|---|
| Difficult airway | Increased Mallampati score, limited neck extension, redundant pharyngeal tissue, OSA | Awake fiberoptic intubation kit available; ramped position (head of bed elevated 25-30 degrees, external auditory meatus level with sternal notch); video laryngoscopy as first-line |
| Reduced FRC | FRC decreases 50-70% in supine obese patients; rapid desaturation during apnea (safe apnea time may be < 1-2 minutes vs 8-10 minutes in normal BMI) | Preoxygenation with 100% O2 for 3-5 minutes; CPAP during preoxygenation; head-up positioning; apneic oxygenation (high-flow nasal cannula during intubation) |
| Altered pharmacokinetics | Increased volume of distribution for lipophilic drugs; unpredictable drug distribution | Dose lipophilic drugs (benzodiazepines, propofol) based on ideal or adjusted body weight; hydrophilic drugs (neuromuscular blockers) based on ideal body weight; sugammadex based on actual body weight |
| Positioning | Nerve injury, rhabdomyolysis, pressure injuries | Adequate padding of all pressure points; arms on padded armboards (avoid tucking); reverse Trendelenburg (improves respiratory mechanics and reduces ICP) |
| Postoperative monitoring | OSA patients at high risk for respiratory depression; opioid sensitivity | Continuous pulse oximetry for 24-48h; CPAP postoperatively; minimize opioids (multimodal analgesia); avoid sedatives |
Glycemic Management
Patients with T2DM require careful perioperative glucose management. Metformin is held 24-48 hours before surgery. Sulfonylureas are held on the day of surgery. Insulin is typically reduced by 50-80% in the immediate postoperative period (insulin sensitivity improves rapidly after RYGB). Target blood glucose: 140-180 mg/dL perioperatively. After discharge, close follow-up is essential — many patients require rapid reduction or discontinuation of diabetes medications to avoid hypoglycemia. T2DM remission can occur within days of RYGB (before significant weight loss), mediated by incretin changes.
Postoperative Dietary Progression
| Phase | Duration | Allowed Foods | Key Instructions |
|---|---|---|---|
| Phase 1: Clear liquids | POD 0-1 | Water, sugar-free gelatin, broth, sugar-free popsicles, decaf tea | Sip slowly; 30 mL (1 oz) every 15 minutes; no straws (swallowed air causes distension) |
| Phase 2: Full liquids | Weeks 1-2 | Protein shakes (whey isolate — goal 60-80 g protein/day), skim milk, strained cream soups, sugar-free yogurt | Protein intake is priority; avoid carbonated beverages; 64 oz fluids/day minimum |
| Phase 3: Pureed foods | Weeks 2-4 | Pureed lean meats, scrambled eggs, cottage cheese, hummus, pureed beans, applesauce | Consistency of baby food; protein first at each meal; 3 meals/day, 1/4 cup portions |
| Phase 4: Soft foods | Weeks 4-8 | Soft cooked fish, ground meats, soft-cooked vegetables, canned fruits (in water) | Chew thoroughly (20-30 chews per bite); stop eating at first sense of fullness; no drinking with meals |
| Phase 5: Regular diet | Week 8+ | All foods tolerated; emphasis on protein, vegetables, fruits; avoid simple sugars and fried foods | Protein first (60-80 g/day for RYGB/LSG; 80-120 g/day for BPD/DS); lifelong small portions; avoid grazing |
Exercise Recommendations
Exercise is a critical component of long-term weight maintenance after bariatric surgery. ASMBS guidelines recommend: weeks 1-4: walking (start with 5-10 minutes, gradually increase to 30 minutes/day); weeks 4-8: low-impact aerobic exercise (swimming, cycling, elliptical); after 8 weeks: progressive resistance training (2-3 sessions/week — essential for preserving lean muscle mass during rapid weight loss) plus aerobic exercise (150-300 minutes/week of moderate-intensity activity). Patients who maintain ≥ 200 minutes/week of moderate exercise have significantly less weight regain at 5 years. Resistance training is particularly important: rapid weight loss causes 20-30% of weight lost to be lean body mass (muscle and bone); resistance training can reduce lean mass loss to 10-15%.
20 Long-Term Follow-Up & Surveillance
Follow-Up Schedule
| Timepoint | Assessments |
|---|---|
| 2 weeks postop | Wound check; diet advancement (full liquids to pureed); weight; review medications (adjust diabetes meds) |
| 6 weeks postop | Weight; diet advancement (soft to regular foods); labs (CMP, CBC); exercise counseling |
| 3 months | Weight; nutritional labs (see below); comorbidity reassessment; medication adjustment |
| 6 months | Weight; nutritional labs; body composition assessment; psychological well-being; support group engagement |
| 12 months | Comprehensive: weight, nutritional labs, DXA (bone density for BPD/DS), comorbidity status, pregnancy counseling |
| Annually thereafter | Weight; nutritional labs (CBC, CMP, iron studies, B12, folate, 25-OH Vit D, PTH, thiamine, zinc, copper); comorbidity review; behavioral assessment; cancer screening (per age-appropriate guidelines) |
Postoperative Diet & Behavior Milestones
| Timepoint | Expected Behavior | Warning Signs |
|---|---|---|
| 1 month | Tolerating full liquids and pureed foods; protein intake ≥ 60 g/day; 64 oz fluids/day; taking all supplements | Persistent vomiting, dehydration, inability to tolerate liquids, nausea with supplements |
| 3 months | Eating soft/regular foods; 3 meals/day; protein-first eating; walking 30 min/day; support group participation | Grazing/snacking behavior, skipping meals, returning to pre-surgery eating patterns, not exercising |
| 6 months | Well-established eating routine; consistent exercise; 10-20% TWL; improved comorbidities; emotional adjustment | Binge eating relapse, alcohol use, emotional eating, relationship difficulties, unrealistic body expectations |
| 12 months | Maximum weight loss approaching; dietary diversity; 150+ min exercise/week; all supplements taken; regular follow-up | Weight plateau frustration, supplement non-adherence, loss to follow-up, disordered eating, substance use |
| 2+ years | Weight maintenance phase; lifelong supplement adherence; annual labs; ongoing behavioral awareness | Weight regain (> 5% from nadir), grazing, liquid calories, loss of support network, depression |
Lab Monitoring
Minimum annual labs for all post-bariatric patients: CBC, CMP, iron studies (Fe, TIBC, ferritin), vitamin B12, folate, 25-OH vitamin D, PTH, calcium. Additional for RYGB: thiamine, zinc, copper. Additional for BPD/DS: vitamins A, E, K, and INR; 24-hour urine calcium; DXA scan. Premenopausal women should have ferritin checked every 3-6 months due to high iron deficiency risk.
Weight Regain
Transfer Addiction & Behavioral Changes
After bariatric surgery, patients can no longer use food as a primary coping mechanism or reward, leading some to develop transfer addiction (also called "addiction transfer" or "cross-addiction"). New compulsive behaviors may include: alcohol use disorder (7-10% after RYGB — see section 20), compulsive spending/shopping, gambling, exercise addiction, sexual compulsivity, and substance abuse. The neurobiological basis involves the dopaminergic mesolimbic reward pathway — food activates the same reward circuitry as other addictive substances, and when food reward is reduced after surgery, the brain may seek alternative dopamine sources. Preoperative screening for addictive tendencies, postoperative behavioral monitoring, and support group participation are protective factors.
Weight regain (> 25% of maximum weight lost) occurs in 20-35% of patients by 5-10 years. Causes include: anatomic factors (pouch/stoma dilation, staple line disruption), behavioral factors (grazing, high-calorie liquid intake, loss of dietary restriction), metabolic adaptation, and psychosocial factors (depression, life stressors, disordered eating relapse). Management is multimodal: behavioral/dietary counseling, pharmacotherapy (GLP-1 receptor agonists are increasingly used as adjunct), endoscopic revision (TORe for GJ dilation), or surgical revision.
Alcohol Use After Bariatric Surgery
The risk of new-onset alcohol use disorder (AUD) is significantly increased after RYGB (7-10% incidence, approximately 2-fold higher than pre-surgery rates). Mechanisms include: altered alcohol pharmacokinetics (after RYGB, alcohol is absorbed rapidly in the Roux limb without first-pass gastric metabolism by alcohol dehydrogenase — peak blood alcohol levels are 50-100% higher and reached faster than pre-surgery), cross-addiction transfer (patients who previously used food for emotional regulation may substitute alcohol), and neurobiological changes (altered dopaminergic reward signaling). The King et al. study (2012, PMID: 22710042) demonstrated that AUD prevalence increased from 7.6% pre-surgery to 9.6% in the second postoperative year specifically after RYGB. LAGB and LSG patients did not show the same increase. Preoperative counseling about alcohol risk and ongoing screening (AUDIT-C questionnaire) are essential components of long-term follow-up.
Mental Health & Suicide Risk
While bariatric surgery improves depression, anxiety, and quality of life for the majority of patients, a concerning signal of increased suicide risk exists in the post-bariatric population. A meta-analysis by Peterhänsel et al. found that the rate of suicide after bariatric surgery is approximately 4.1 per 10,000 person-years — higher than the general population and the obese non-surgical population. Risk factors include: pre-existing psychiatric disorders, substance abuse history, social isolation (particularly if the patient's primary social activity was food-related), and unmet expectations about body image or relationship outcomes. Ongoing mental health screening and support group participation are protective.
Body Contouring After Weight Loss
After massive weight loss (typically > 100 lbs or > 50% EWL), excess skin folds can cause hygiene issues (intertriginous dermatitis, fungal infections), functional limitations, psychological distress, and body image dissatisfaction. Body contouring procedures include panniculectomy/abdominoplasty (most common), brachioplasty, thigh lift, breast reduction/mastopexy, and lower body lift. These are typically performed ≥ 18 months after bariatric surgery when weight has stabilized. Nutritional status (albumin, prealbumin) should be optimized before contouring surgery to reduce wound healing complications.
Pregnancy After Bariatric Surgery
Pregnancy is recommended to be delayed 12-18 months after bariatric surgery to avoid nutritional deficiency during rapid weight loss. Bariatric surgery reduces gestational diabetes (60-80% reduction), preeclampsia, and macrosomia compared to obese controls. However, increased risks include: SGA (small for gestational age) infants, preterm birth, and nutritional deficiency (especially iron, B12, folate, calcium). Monthly nutritional monitoring is recommended throughout pregnancy. Adjustable bands should be deflated during pregnancy.
Bariatric Surgery Center of Excellence Criteria
The MBSAQIP (Metabolic and Bariatric Surgery Accreditation and Quality Improvement Program) — a joint program of the ACS and ASMBS — accredits bariatric surgery centers based on quality metrics. Requirements include: minimum annual case volume (25 stapled bariatric procedures for accredited centers, 50 for comprehensive centers), dedicated bariatric program coordinator, multidisciplinary team (surgeon, dietitian, psychologist, internist), 24/7 OR availability with bariatric-capable equipment (appropriate-weight-capacity OR tables, specialized retractors, long instruments), ICU availability, critical care support for complications, prospective data collection in the national MBSAQIP registry, and annual quality review. Centers with MBSAQIP accreditation have demonstrated 30-40% lower complication rates and 50% lower mortality compared to non-accredited facilities.
21 Classification Systems & Staging
Edmonton Obesity Staging System (EOSS)
| Stage | Description | Clinical Features |
|---|---|---|
| 0 | No apparent risk factors | No obesity-related risk factors, no physical symptoms, no functional limitations |
| 1 | Subclinical risk factors | Borderline hypertension, impaired fasting glucose, elevated liver enzymes, mild physical symptoms (dyspnea on exertion, aches), mild functional limitations |
| 2 | Established chronic disease | Hypertension requiring treatment, T2DM, OSA, PCOS, NAFLD, osteoarthritis, GERD, moderate functional limitations, mild-moderate psychological distress |
| 3 | Significant end-organ damage | MI, heart failure, diabetic complications (retinopathy, nephropathy), OSA with cor pulmonale, significant psychological disorders, significant functional limitations |
| 4 | Severe disability/end-stage | Severe disabling conditions, severe functional limitations, severe psychological distress, end-stage organ damage |
Clavien-Dindo Classification of Surgical Complications
| Grade | Definition | Bariatric Example |
|---|---|---|
| I | Deviation from normal course; no pharmacological, endoscopic, or radiological intervention needed | Wound infection managed with bedside opening |
| II | Pharmacological treatment (beyond antiemetics, antipyretics, analgesics, diuretics); blood transfusion; TPN | Postoperative hemorrhage requiring transfusion; UTI requiring antibiotics |
| IIIa | Intervention without general anesthesia | IR-guided percutaneous drainage of abscess; endoscopic balloon dilation of stricture |
| IIIb | Intervention under general anesthesia | Operative re-exploration for leak or internal hernia |
| IVa | ICU management; single-organ dysfunction | Postoperative respiratory failure requiring ventilation |
| IVb | ICU management; multiorgan dysfunction | Sepsis from uncontrolled leak with ARDS and AKI |
| V | Death | — |
ASMBS Outcome Reporting Standards
The ASMBS recommends standardized reporting of weight-loss outcomes using the following metrics:
- %EWL (percent excess weight loss) = [(initial weight − current weight) / (initial weight − ideal body weight)] × 100. Ideal body weight is typically calculated at BMI 25.
- %TWL (percent total weight loss) = [(initial weight − current weight) / initial weight] × 100. Preferred metric as it is independent of arbitrary ideal body weight.
- %EBMIL (percent excess BMI loss) = [(initial BMI − current BMI) / (initial BMI − 25)] × 100.
- Adequate weight loss: ≥ 50% EWL or ≥ 20% TWL at 12-18 months.
Comprehensive Procedure Comparison
| Parameter | LSG | RYGB | OAGB | BPD/DS | LAGB |
|---|---|---|---|---|---|
| Mechanism | Restrictive + hormonal | Restrictive + malabsorptive + hormonal | Restrictive + malabsorptive + hormonal | Primarily malabsorptive + hormonal | Purely restrictive |
| OR time (min) | 45-90 | 90-150 | 60-90 | 150-240 | 30-60 |
| 30-day mortality | 0.05-0.2% | 0.1-0.3% | 0.1-0.2% | 0.5-1.0% | < 0.1% |
| %EWL at 5 yr | 45-60% | 55-70% | 60-75% | 70-80% | 40-50% |
| T2DM remission 5 yr | 40-55% | 50-65% | 55-70% | 85-95% | 25-35% |
| GERD effect | May worsen (15-35% de novo) | Resolves (85-95%) | Bile reflux risk (1-2%) | Variable | May worsen |
| Nutritional risk | Low-moderate | Moderate-high | Moderate-high | Very high | Low |
| Reversibility | Irreversible | Technically reversible (rarely done) | Reversible | Technically reversible (rarely done) | Fully reversible |
| Endoscopic access to remnant | N/A (no excluded stomach) | Limited (requires special techniques) | Limited | N/A (no excluded stomach) | N/A |
| Long-term reoperation rate | 5-15% | 10-20% | 5-10% | 15-25% | 30-60% |
Obesity Staging — King's Obesity Staging Criteria
| Stage | Criteria | Recommended Intervention |
|---|---|---|
| A (At risk) | BMI ≥ 30 with no comorbidities, no functional limitations | Lifestyle modification; consider pharmacotherapy |
| B (Borderline) | BMI ≥ 30 with mild comorbidities (pre-diabetes, borderline HTN) | Intensive lifestyle + pharmacotherapy; consider surgery if BMI ≥ 35 |
| C (Complicated) | BMI ≥ 30 with established comorbidities requiring treatment (T2DM, HTN, OSA) | Bariatric surgery recommended per ASMBS/IFSO 2022 if BMI ≥ 30 with metabolic disease |
| D (Disabled) | Severe obesity with significant functional impairment, end-organ damage | Bariatric surgery with multidisciplinary optimization; may need staged approach |
ASA Physical Status Classification (Perioperative Context)
| Class | Definition | Bariatric Relevance |
|---|---|---|
| ASA I | Normal healthy patient | Rare in bariatric candidates |
| ASA II | Mild systemic disease | BMI 35-40 without major comorbidities |
| ASA III | Severe systemic disease | Most bariatric patients (BMI ≥ 40, or BMI 35-40 with T2DM/OSA/HTN) |
| ASA IV | Severe systemic disease, constant threat to life | Super-obese with OHS, cor pulmonale, or severe heart failure |
| ASA V | Moribund; not expected to survive without surgery | Absolute contraindication to elective bariatric surgery |
22 Medications Master Table
Anti-Obesity Medications (Pharmacotherapy)
| Medication | Mechanism | Dose | Expected %TWL | Key Side Effects / Notes |
|---|---|---|---|---|
| Semaglutide (Wegovy) | GLP-1 receptor agonist | 2.4 mg SQ weekly (titrate over 16 weeks) | 12-17% | Nausea, vomiting, diarrhea, pancreatitis risk, contraindicated in MTC/MEN2 family history; STEP trials (PMID: 33567185) |
| Tirzepatide (Zepbound) | Dual GIP/GLP-1 receptor agonist | 5-15 mg SQ weekly | 15-22% | Nausea, diarrhea; SURMOUNT trials; most effective single anti-obesity medication |
| Liraglutide (Saxenda) | GLP-1 receptor agonist | 3.0 mg SQ daily | 5-8% | Nausea, pancreatitis risk; SCALE trials |
| Phentermine/topiramate ER (Qsymia) | Sympathomimetic + anticonvulsant | 7.5/46 to 15/92 mg daily | 7-10% | Dry mouth, paresthesias, constipation, cognitive impairment; teratogenic (pregnancy cat X); EQUIP, CONQUER trials |
| Naltrexone/bupropion ER (Contrave) | Opioid antagonist + aminoketone | 32/360 mg daily | 4-6% | Nausea, headache, insomnia; contraindicated in seizure disorder, opioid use; COR trials |
| Orlistat (Xenical/Alli) | Pancreatic lipase inhibitor | 120 mg TID with meals | 3-4% | Steatorrhea, fecal urgency, fat-soluble vitamin malabsorption; poor tolerability |
| Phentermine | Sympathomimetic (norepinephrine release) | 15-37.5 mg daily | 3-5% | Insomnia, dry mouth, tachycardia; DEA Schedule IV; approved for short-term (< 12 weeks) only |
Vitamin & Mineral Supplements
| Supplement | Form | Post-RYGB Dose | Post-BPD/DS Dose |
|---|---|---|---|
| Multivitamin with minerals | Chewable or liquid (first 3 months), then capsule | 2 daily | 2 daily (high potency) |
| Calcium citrate | Chewable or capsule (divided doses, 500-600 mg each) | 1200-1500 mg/day | 1800-2400 mg/day |
| Vitamin D3 | Capsule or liquid | 3000 IU/day (titrate to 25-OH D > 30) | 3000-5000 IU/day |
| Iron (ferrous sulfate / fumarate) | With vitamin C (enhances absorption) | 45-60 mg elemental/day | 45-60 mg elemental/day |
| Vitamin B12 | Sublingual, oral, or IM | 1000 µg/day SL or 1000 µg/month IM | 1000 µg/day SL or 1000 µg/month IM |
| Vitamin A | Capsule | Per MVI | 5000-10000 IU/day |
| Vitamin K | Capsule | Per MVI | 300 µg/day |
Commonly Used Perioperative Medications
| Category | Medication | Dose/Route | Notes |
|---|---|---|---|
| VTE prophylaxis | Enoxaparin | 40 mg SQ BID (BMI > 40); start 2-6h postop | Weight-based dosing; anti-Xa level monitoring for extremes |
| PPI (ulcer prophylaxis) | Omeprazole / Pantoprazole | 40 mg PO/IV daily | Recommended for 90 days after RYGB; longer if marginal ulcer risk |
| Ursodiol (gallstone prevention) | Ursodeoxycholic acid | 300 mg PO BID for 6 months | Reduces gallstone formation from ~30% to < 2% |
| Antiemetic | Ondansetron | 4 mg IV/PO q8h PRN | Standard PONV prophylaxis |
| Analgesic | Acetaminophen | 1000 mg PO/IV q6h | First-line; avoid NSAIDs after RYGB (marginal ulcer risk) |
| Analgesic (NSAID) | Ketorolac / Ibuprofen | 15-30 mg IV / 400 mg PO | AVOID after RYGB; acceptable short-term after LSG |
Medication Absorption Changes After Bariatric Surgery
Bariatric surgery alters drug pharmacokinetics through multiple mechanisms — this has critical implications for medication management:
| Factor | Change After RYGB/BPD-DS | Clinical Implications |
|---|---|---|
| Gastric pH | Increased (less acidic — reduced parietal cell mass) | Reduced absorption of weak acids (aspirin, ketoconazole); improved absorption of weak bases (metformin) |
| Gastric emptying | Markedly accelerated (no pyloric regulation in RYGB) | Faster drug absorption; higher peak levels; shorter duration of action for sustained-release formulations |
| Absorptive surface area | Reduced (bypassed duodenum and proximal jejunum) | Reduced bioavailability of drugs absorbed primarily in duodenum/proximal jejunum |
| Intestinal transit | Altered (shortened absorptive segment) | Extended-release and enteric-coated formulations may not fully dissolve — avoid or monitor closely |
| First-pass metabolism | Altered (intestinal CYP3A4 bypassed in RYGB) | Increased bioavailability of CYP3A4 substrates (cyclosporine, tacrolimus, statins) |
Key medication considerations: Avoid extended-release, enteric-coated, and large tablet formulations (switch to immediate-release, liquid, or chewable forms). Levothyroxine absorption is reduced — monitor TSH closely and adjust dose. Oral contraceptives — absorption may be unpredictable after RYGB/BPD-DS; consider non-oral alternatives (IUD, implant, patch, ring). Immunosuppressants (tacrolimus, cyclosporine) — levels may be highly variable; frequent monitoring essential. Psychiatric medications (SSRIs, lithium) — absorption changes require dose adjustment and level monitoring.
23 Imaging & Diagnostics
Leak Detection Algorithm
A systematic approach to suspected anastomotic/staple-line leak:
- Clinical suspicion: Persistent tachycardia (> 120 bpm), fever (> 38.5°C), tachypnea, left shoulder pain (diaphragmatic irritation), oliguria, peritonitis, or elevated inflammatory markers (WBC > 15,000, CRP > 150 mg/L, procalcitonin > 1.0 ng/mL)
- Initial imaging: CT abdomen/pelvis with oral water-soluble contrast (sensitivity 75-90%) — look for contrast extravasation, extraluminal air beyond expected postop pneumoperitoneum, fluid collections
- If CT negative but high clinical suspicion: UGI series with Gastrografin followed by thin barium (higher sensitivity for small contained leaks); or direct operative exploration (definitive diagnosis)
- Hemodynamically unstable patient: Skip imaging — proceed directly to operative exploration (do not delay definitive treatment for imaging in a septic, unstable patient)
- Classification of leak: Type I (subclinical — microperforations detected on routine imaging without symptoms; manage conservatively); Type II (clinical — symptomatic leak with localized contamination; drainage + stent or VAC); Type III (clinical with generalized peritonitis or sepsis; requires operative washout)
Upper GI Series (UGI / Barium Swallow)
Indications: Evaluation of suspected leak (use water-soluble contrast — Gastrografin — initially; if negative, follow with thin barium for higher sensitivity), stricture (filling defect, delayed contrast passage), marginal ulcer (mucosal irregularity), and pouch/stoma sizing. Sensitivity for leak: 50-75% (lower than CT; false negatives are common in contained leaks). Routine postoperative UGI: no longer recommended as standard practice (ERAS protocols) — perform only when clinical concern exists.
CT Abdomen/Pelvis
CT with oral water-soluble contrast is the study of choice for suspected leak (sensitivity 75-90%), abscess, or internal hernia. Key findings:
- Leak: Extravasation of oral contrast, pneumoperitoneum (beyond expected postoperative air), fluid collection near the anastomosis, wall defect
- Internal hernia: Swirl sign (mesenteric rotation), clustered bowel loops, mesenteric edema, displaced JJ anastomosis
- Pulmonary embolism: CTA chest with IV contrast — filling defect in pulmonary arteries
- Portal vein thrombosis: Rare but reported (0.3-1%) after LSG; CT with IV contrast shows filling defect in portal/mesenteric veins
Esophagogastroduodenoscopy (EGD)
EGD is the gold standard for evaluating: marginal ulcer, stricture (and therapeutic dilation), band erosion, Barrett's surveillance (post-LSG), gastrogastric fistula (RYGB), and stomal dilation (TORe). In post-RYGB patients, the excluded stomach cannot be reached by standard EGD — deep enteroscopy, device-assisted enteroscopy, or interventional radiology-guided percutaneous access to the remnant stomach may be needed for evaluation.
Endoscopic Evaluation & Therapeutic Interventions
Endoscopic interventions specific to bariatric surgery patients include:
| Intervention | Indication | Technique |
|---|---|---|
| Balloon dilation | GJ stricture (RYGB), incisura stricture (LSG) | TTS balloon (12-15 mm), serial dilations; avoid exceeding 15 mm at GJ (risk of perforation) |
| Stent placement | Staple-line leak (LSG), GJ leak (RYGB) | Fully covered SEMS (self-expanding metal stent); 4-8 week duration; migration rate 15-30% |
| Endoscopic vacuum therapy | Chronic leak/fistula (Endo-SPONGE/Endo-VAC) | Polyurethane sponge placed in abscess cavity via endoscope; changed every 3-5 days; promotes granulation |
| TORe (transoral outlet reduction) | GJ stoma dilation (> 2 cm) with weight regain | Argon plasma coagulation of stoma margin + endoscopic suturing (OverStitch) to reduce stoma to 8-12 mm |
| Septotomy | Chronic gastrogastric fistula | Endoscopic division of septum between pouch and remnant |
| Band removal (endoscopic) | Band erosion with > 50% intraluminal migration | Band cut endoscopically and removed through the mouth |
HIDA Scan (Hepatobiliary Iminodiacetic Acid Scan)
Used to evaluate biliary dysfunction (biliary dyskinesia with EF < 35%) in post-bariatric patients with right upper quadrant pain and negative ultrasound. Gallstone disease is common after rapid weight loss (incidence ~30% in the first year without ursodiol prophylaxis).
Radiological Signs Summary
| Finding | Modality | Diagnosis | Significance |
|---|---|---|---|
| Swirl sign | CT | Internal hernia | Mesenteric vessels rotating around a central axis; sensitivity 78-100% |
| Small bowel feces sign | CT | SBO (late/high-grade) | Feculent material in dilated small bowel proximal to obstruction |
| Free air (pneumoperitoneum) | CT / upright CXR | Perforation or leak | Expected for 24-48h postop; concerning if increasing or persistent beyond 48h |
| Contrast extravasation | CT with oral contrast / UGI | Anastomotic or staple-line leak | Sensitivity 75-90% (CT) or 50-75% (UGI); false negatives possible with contained leaks |
| Dilated pouch / stoma | UGI / EGD | Weight regain after RYGB | Pouch > 6 cm or stoma > 2 cm associated with weight regain |
| Filling defect in portal/mesenteric veins | CTA | PMVT | Rare but serious; more common after LSG |
| Mammillary body hyperintensity | MRI (T2/FLAIR) | Wernicke encephalopathy | Thiamine deficiency; sensitivity ~50% (clinical diagnosis often precedes MRI findings) |
24 Abbreviations Master List
| Abbreviation | Full Term |
|---|---|
| ASMBS | American Society for Metabolic and Bariatric Surgery |
| BAT | Brown adipose tissue |
| BMI | Body mass index (kg/m²) |
| BP limb | Biliopancreatic limb |
| BPD/DS | Biliopancreatic diversion with duodenal switch |
| CCK | Cholecystokinin |
| CPAP | Continuous positive airway pressure |
| DXA | Dual-energy X-ray absorptiometry |
| EGD | Esophagogastroduodenoscopy |
| ERAS | Enhanced Recovery After Surgery |
| ESG | Endoscopic sleeve gastroplasty |
| %EWL | Percent excess weight loss |
| GEJ | Gastroesophageal junction |
| GIP | Glucose-dependent insulinotropic polypeptide |
| GJ | Gastrojejunostomy |
| GLP-1 | Glucagon-like peptide-1 |
| HIDA | Hepatobiliary iminodiacetic acid (scan) |
| IF | Intrinsic factor |
| IFSO | International Federation for the Surgery of Obesity and Metabolic Disorders |
| IGB | Intragastric balloon |
| JJ | Jejunojejunostomy |
| LAGB | Laparoscopic adjustable gastric banding |
| LMWH | Low-molecular-weight heparin |
| LSG | Laparoscopic sleeve gastrectomy |
| MGB | Mini gastric bypass (= OAGB) |
| NAFLD | Non-alcoholic fatty liver disease |
| NASH | Non-alcoholic steatohepatitis |
| OAGB | One anastomosis gastric bypass |
| OHS | Obesity hypoventilation syndrome |
| OSA | Obstructive sleep apnea |
| PBHH | Post-bariatric hyperinsulinemic hypoglycemia |
| PCOS | Polycystic ovary syndrome |
| PPI | Proton pump inhibitor |
| PSG | Polysomnography |
| PYY | Peptide YY |
| RYGB | Roux-en-Y gastric bypass |
| SADI-S | Single anastomosis duodenoileal bypass with sleeve gastrectomy |
| SCD | Sequential compression device |
| T2DM | Type 2 diabetes mellitus |
| TORe | Transoral outlet reduction (endoscopic) |
| %TWL | Percent total weight loss |
| UGI | Upper gastrointestinal (series) |
| VTE | Venous thromboembolism |
| WAT | White adipose tissue |
25 Landmark Trials & Evidence
Swedish Obese Subjects (SOS) Study
The SOS Study is the largest and longest prospective controlled study of bariatric surgery (Sjöström et al., PMID: 17715408). Design: prospective, matched, non-randomized; 2,010 surgical patients vs 2,037 matched obese controls; median follow-up 10+ years (up to 20 years). Key findings: surgery reduced overall mortality by 29% (HR 0.71), cardiovascular events by 33%, cancer incidence by 42% (in women), and T2DM incidence by 78%. Weight loss at 10 years: RYGB 25% TWL, VBG 16%, LAGB 14% (vs 1-2% in controls). This study provided the first Level I evidence that bariatric surgery reduces mortality.
STAMPEDE Trial
The STAMPEDE trial (Schauer et al., PMID: 28641132) was a landmark RCT comparing RYGB vs LSG vs intensive medical therapy in 150 patients with T2DM and BMI 27-43. Primary endpoint: HbA1c ≤ 6.0% at 5 years. Results: 29% of RYGB and 23% of LSG patients achieved the primary endpoint vs only 5% of medical therapy patients (p < 0.01 for both). Both surgical groups had significant improvements in triglycerides, HDL, and quality of life. This trial established bariatric surgery as superior to medical therapy for T2DM management.
SM-BOSS Trial
The SM-BOSS trial (Peterli et al., PMID: 29234903) was a multicenter RCT comparing LSG vs RYGB in 217 patients. At 5 years, %EBMIL was 61.1% for LSG vs 68.3% for RYGB (non-significant difference). T2DM remission was similar. However, RYGB had superior GERD outcomes, while LSG had fewer nutritional deficiencies. Concluded that both procedures provide similar weight loss and metabolic outcomes at 5 years.
SLEEVE vs BYPASS (SLEEVEPASS) Trial
The SLEEVEPASS trial (Salminen et al., PMID: 29340679) randomized 240 patients to LSG vs RYGB. At 5 years, %EWL was 49% for LSG vs 57% for RYGB (non-significant). RYGB had higher remission of T2DM (50% vs 33%, p = 0.04) and dyslipidemia. LSG had more GERD requiring medication. Conclusion: both procedures are effective; RYGB may have an advantage for T2DM and dyslipidemia.
GATEWAY Trial
The GATEWAY trial (Schiavon et al., PMID: 28641240) was an RCT evaluating RYGB + medical therapy vs medical therapy alone for hypertension in patients with BMI 30-39.9. At 12 months, 83.7% of the surgical group achieved ≥ 30% reduction in antihypertensive medications (primary endpoint) vs 12.8% in the medical group (p < 0.001). Complete remission of hypertension (off all medications): 51% surgical vs 0% medical. This trial was pivotal in supporting bariatric surgery for metabolic disease at lower BMI thresholds.
Emerging Procedures & Future Directions
The field of bariatric and metabolic surgery continues to evolve:
| Innovation | Description | Status |
|---|---|---|
| Robotic bariatric surgery | Robot-assisted (da Vinci) RYGB and LSG; potentially improved ergonomics, visualization, and suturing precision | Widely adopted; comparable outcomes to laparoscopic; higher cost; shorter learning curve for complex revisional cases |
| Endoscopic sleeve gastroplasty (ESG) revisional | ESG performed as a revision for dilated sleeve or post-RYGB pouch dilation | Emerging; early data show 10-15% additional TWL |
| Duodenal mucosal resurfacing (DMR / Revita) | Endoscopic thermal ablation of duodenal mucosa to alter incretin signaling and improve glycemic control | Investigational; targeting T2DM in patients with BMI 28-40; Phase III trials underway |
| Magnetic sphincter augmentation (LINX) after LSG | Magnetic bead device placed at LES to treat post-sleeve GERD as alternative to RYGB conversion | Early experience; limited data; potential role in mild-moderate post-sleeve GERD |
| GLP-1 RA + surgery combination | Preoperative GLP-1 RA to enhance liver shrinkage and preoperative weight loss; postoperative use for weight recidivism | Rapidly expanding practice; optimal timing and duration under investigation |
| Stomach intestinal pylorus-sparing surgery (SIPS) | Variation of SADI-S with a slightly different intestinal configuration; single anastomosis duodenoileal bypass | Increasingly performed; some centers use interchangeably with SADI-S |
Bariatric Surgery Global Trends
According to IFSO global registry data, the worldwide distribution of bariatric procedures (2023) is: LSG ~60%, RYGB ~20%, OAGB ~10%, BPD/DS/SADI-S ~3%, LAGB < 1%, and other/revisional ~6%. The number of bariatric procedures performed worldwide has grown from approximately 350,000/year in 2011 to over 800,000/year in 2023, yet this represents treatment of less than 1% of eligible patients. The massive treatment gap between surgical eligibility and surgical utilization remains the greatest challenge in the field.
Additional Key Evidence
| Study | Year | Key Finding | Reference |
|---|---|---|---|
| Buchwald meta-analysis | 2004 | T2DM resolved in 76.8% of bariatric patients; 86% for BPD/DS | PMID: 15494575 |
| Adams et al. (Utah mortality) | 2007 | RYGB reduced long-term mortality by 40% vs matched controls | PMID: 17715409 |
| Mingrone et al. | 2015 | BPD and RYGB superior to medical therapy for T2DM remission at 5 years | PMID: 25654259 |
| ASMBS/IFSO 2022 Consensus | 2022 | Expanded indications: BMI ≥ 30 with metabolic disease qualifies for surgery | PMID: 36336720 |
| STEP 1 (semaglutide) | 2021 | Semaglutide 2.4 mg/wk: 14.9% TWL vs 2.4% placebo at 68 weeks | PMID: 33567185 |
| SURMOUNT-1 (tirzepatide) | 2022 | Tirzepatide 15 mg: 22.5% TWL at 72 weeks (most effective anti-obesity drug) | PMID: 35658024 |