Cardiothoracic Surgery
Every diagnosis, classification, procedure, technique, medication, complication, and management algorithm across the full scope of cardiothoracic surgery in one place.
01 Cardiac Anatomy
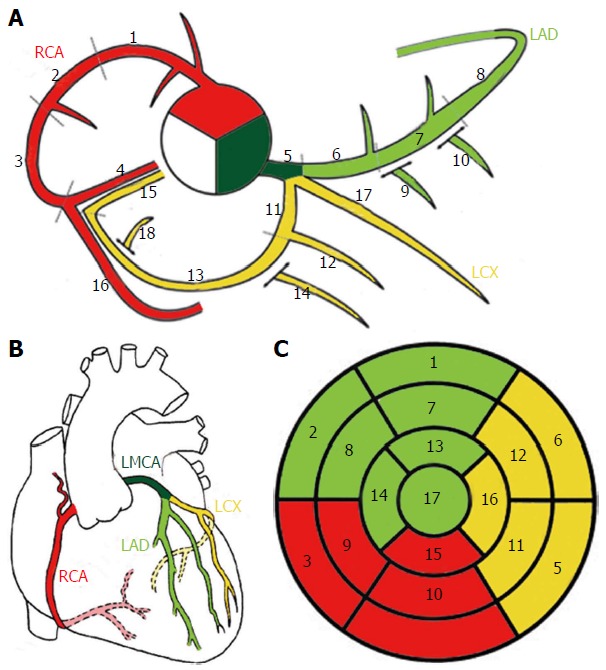
Coronary Circulation
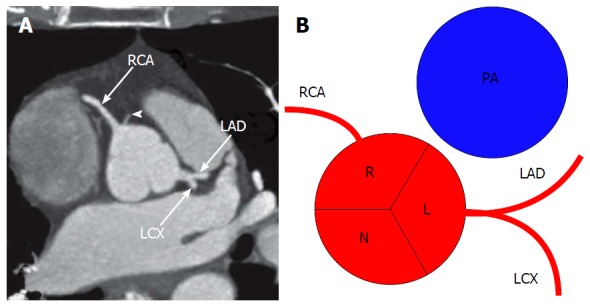
The heart receives its blood supply from two coronary arteries arising from the aortic root just above the aortic valve cusps (the sinuses of Valsalva). The left main coronary artery (LMCA) arises from the left coronary sinus and bifurcates (after 1-2 cm) into the left anterior descending artery (LAD) and the left circumflex artery (LCx). In some patients, a ramus intermedius arises as a trifurcation branch. The right coronary artery (RCA) arises from the right coronary sinus and courses in the right atrioventricular groove.
LAD: Supplies the anterior wall of the LV, anterior two-thirds of the interventricular septum, and the apex. Gives off septal perforators and diagonal branches. The LAD is the most commonly diseased coronary artery and the single most important vessel for surgical revascularization (LIMA-to-LAD graft).
LCx: Courses in the left atrioventricular groove, supplying the lateral and posterolateral LV wall. Gives off obtuse marginal (OM) branches. In ~15% of patients the LCx is dominant (supplies the PDA).
RCA: Supplies the right ventricle, SA node (in ~55% of patients), AV node (in ~85% of right-dominant patients), and the inferior wall of the LV. The RCA gives rise to the posterior descending artery (PDA) in a right-dominant circulation (~85% of the population). Right dominance means the PDA arises from the RCA; left dominance (~8%) means the PDA arises from the LCx; codominant (~7%) indicates both contribute.
Valvular Anatomy
The heart has four valves arranged in the cardiac skeleton (fibrous trigones): two atrioventricular (AV) valves — mitral (bicuspid, two leaflets: anterior and posterior) and tricuspid (three leaflets: anterior, posterior, septal) — and two semilunar valves — aortic (three cusps: left, right, noncoronary) and pulmonic (three cusps). The mitral valve has a complex apparatus: annulus, leaflets, chordae tendineae, and papillary muscles (anterolateral — dual blood supply from LAD and LCx, and posteromedial — single supply from PDA, making it more vulnerable to ischemic rupture).
The aortic valve cusps are named by the coronary ostia they face: the left coronary cusp (LCC), the right coronary cusp (RCC), and the noncoronary cusp (NCC). The NCC is adjacent to the membranous septum and the AV node — excessive decalcification or suturing in this area during AVR can cause heart block. The aortic annulus is in close proximity to the mitral valve anteriorly and the bundle of His posteriorly.
Conduction System
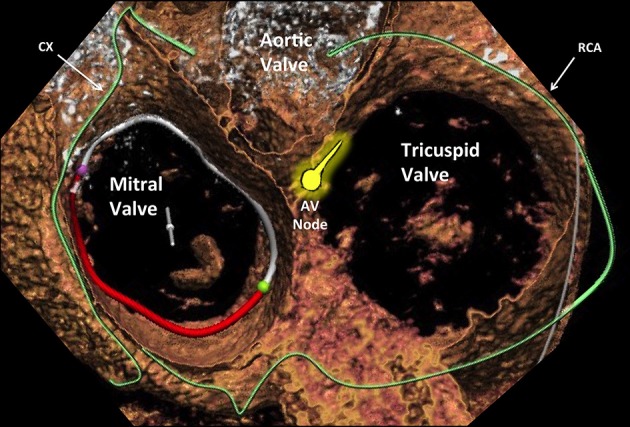
The sinoatrial (SA) node lies at the junction of the SVC and right atrium (supplied by the SA nodal artery — from RCA in ~55%, from LCx in ~45%). The atrioventricular (AV) node is located in the triangle of Koch (bounded by the coronary sinus ostium, tendon of Todaro, and tricuspid valve annulus) — supplied by the AV nodal artery (branch of the PDA, which comes from the RCA in ~85%). The bundle of His penetrates the central fibrous body and divides into the left and right bundle branches. The left bundle has two fascicles (anterosuperior and posteroinferior).
Pericardium
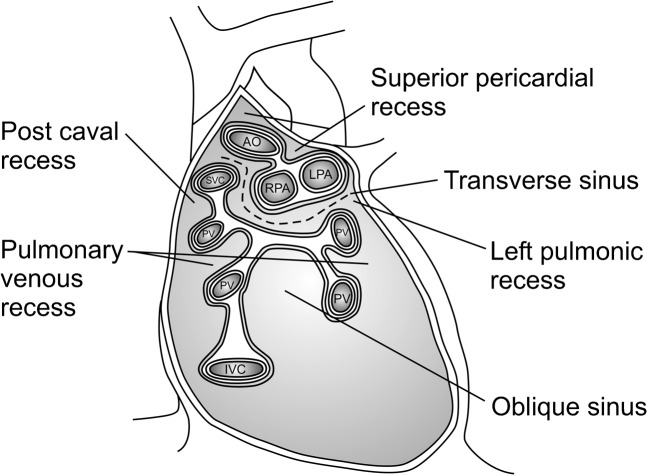
A double-layered fibroserous sac consisting of the visceral pericardium (epicardium) — adherent to the heart surface — and the parietal pericardium — a tough fibrous outer layer. Between them lies the pericardial cavity, which normally contains 15-50 mL of serous fluid. Two important pericardial sinuses: the transverse sinus (behind the great arteries, in front of the SVC — the surgeon passes a finger or clamp through this sinus to encircle the aorta during CPB cannulation) and the oblique sinus (behind the left atrium, bounded by the pulmonary veins). The pericardium receives its blood supply from the internal mammary (thoracic) arteries and pericardiophrenic branches. The phrenic nerves course along the pericardium bilaterally — injury during cardiac surgery causes diaphragmatic paralysis.
Great Vessels & Cardiac Chambers
The right atrium (RA) receives deoxygenated blood from the SVC, IVC, and coronary sinus. Important internal landmarks: crista terminalis (muscular ridge separating the smooth and trabeculated portions), fossa ovalis (remnant of the foramen ovale — site of ASD closure devices), Eustachian valve (directs IVC flow toward the fossa ovalis in fetal life), and the Thebesian valve (guards the coronary sinus ostium). The right ventricle (RV) is a thin-walled, crescent-shaped chamber that generates ~1/5 the stroke work of the LV. Key structures: moderator band (contains the right bundle branch), conus arteriosus (infundibulum — outflow tract to the pulmonary artery), and the tricuspid papillary muscles (anterior, posterior, septal).
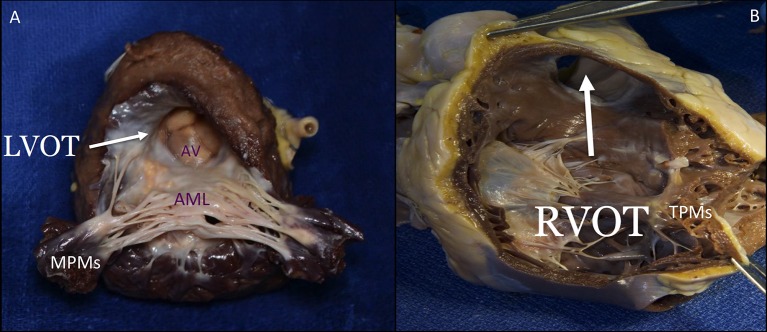
The left atrium (LA) is the most posterior cardiac chamber (directly anterior to the esophagus — hence TEE provides excellent views). Receives oxygenated blood from four pulmonary veins (right superior/inferior, left superior/inferior). The LA appendage is the most common site of thrombus formation in atrial fibrillation. The left ventricle (LV) is thick-walled (8-12 mm) and conical. The LV outflow tract (LVOT) is bounded by the interventricular septum anteriorly, the anterior mitral leaflet posteriorly, and the aortic valve superiorly — obstruction here occurs in hypertrophic obstructive cardiomyopathy (HOCM) and after excessive septal myectomy.
02 Thoracic & Pulmonary Anatomy
Bronchopulmonary Segments
The right lung has three lobes (upper, middle, lower) separated by the oblique and horizontal fissures, with 10 bronchopulmonary segments. The left lung has two lobes (upper — includes the lingula, and lower) separated by the oblique fissure, with 8-10 segments. The right main bronchus is wider, shorter, and more vertical than the left — aspirated foreign bodies preferentially enter the right side (especially the right lower lobe).
Right upper lobe segments: Apical (B1), posterior (B2), anterior (B3). Right middle lobe: Lateral (B4), medial (B5). Right lower lobe: Superior (B6), medial basal (B7), anterior basal (B8), lateral basal (B9), posterior basal (B10). Left upper lobe: Apicoposterior (B1+2), anterior (B3), superior lingular (B4), inferior lingular (B5). Left lower lobe: Superior (B6), anteromedial basal (B7+8), lateral basal (B9), posterior basal (B10).
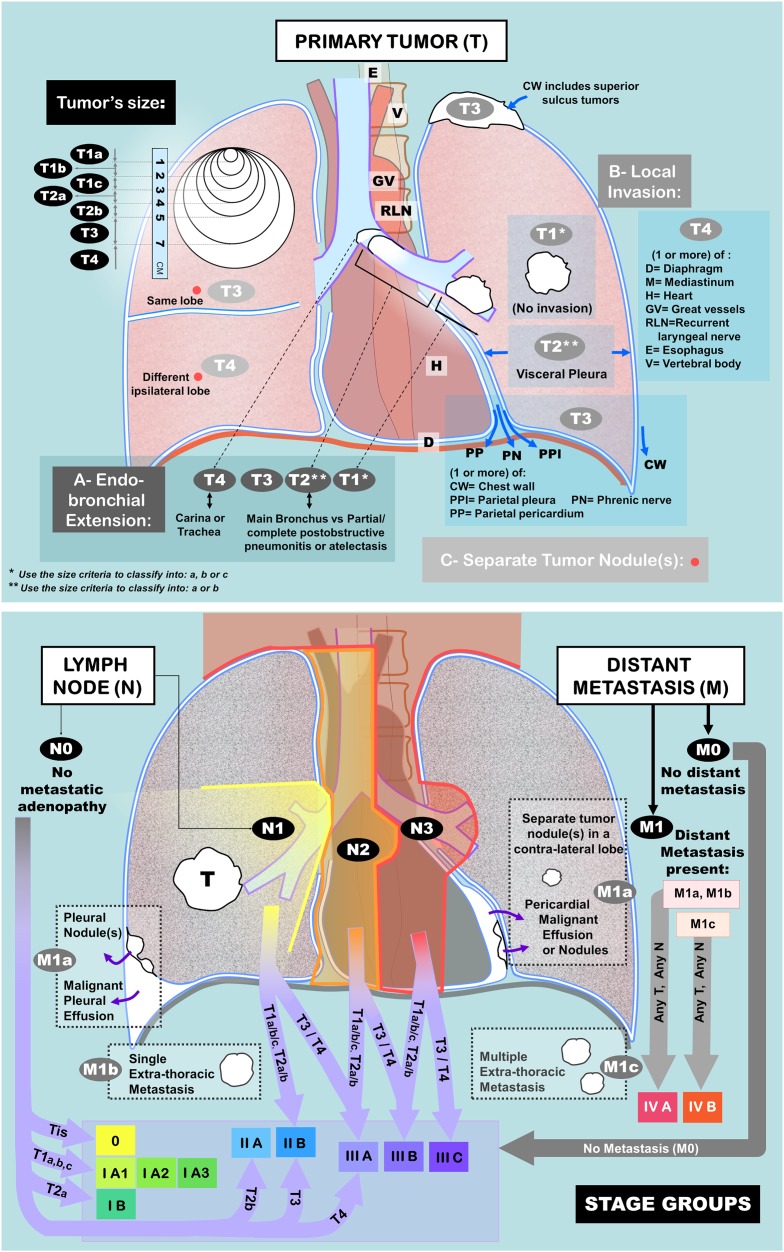
Mediastinal Compartments
The mediastinum is divided into compartments for differential diagnosis of masses:
Anterior (prevascular) mediastinum: Thymus, lymph nodes, fat, thyroid (substernal extension). Masses: thymoma, lymphoma, teratoma (germ cell tumor), thyroid goiter — the classic "4 T's".
Middle (visceral) mediastinum: Heart, pericardium, great vessels, trachea, main bronchi, esophagus, thoracic duct, vagus and phrenic nerves. Masses: lymphadenopathy, bronchogenic cyst, pericardial cyst, esophageal duplication cyst.
Posterior (paravertebral) mediastinum: Thoracic spine, sympathetic chain, descending aorta, azygos/hemiazygos veins, thoracic duct (lower portion). Masses: neurogenic tumors (schwannoma, neurofibroma, ganglioneuroma — most common posterior mediastinal mass), meningocele, esophageal lesions.
Pleural Spaces & Chest Wall
The pleural cavity is a potential space between the visceral pleura (adherent to lung surface) and the parietal pleura (lines the chest wall, diaphragm, and mediastinum). Normally contains ~5 mL of serous fluid. The costodiaphragmatic recess is the lowest extent of the pleural space — it dips to the level of the 12th rib posteriorly (important: posterior chest tube placement should target this recess for drainage). An upright CXR requires ~200 mL to detect a pleural effusion; a lateral decubitus requires ~50 mL.
The chest wall consists of 12 pairs of ribs, the sternum (manubrium, body, xiphoid process), intercostal muscles, and the diaphragm. The intercostal neurovascular bundle runs in the costal groove along the inferior edge of each rib in the order vein-artery-nerve (VAN) from superior to inferior. Chest tubes and thoracentesis are inserted over the top of the rib (superior border) to avoid the neurovascular bundle.
Trachea & Carina
The trachea begins at the level of C6 (cricoid cartilage) and bifurcates at the carina at the level of T4-T5 (angle of Louis). It is ~11-12 cm long and consists of 16-20 C-shaped cartilaginous rings (the posterior membranous wall lacks cartilage and is shared with the esophagus). The right main bronchus angles ~25 degrees from vertical; the left ~45 degrees. The subcarinal space (station 7 lymph nodes) is a critical staging location in lung cancer — accessed by EBUS, EUS, or mediastinoscopy. The azygos vein arches over the right main bronchus to drain into the SVC — its location is relevant during right upper lobectomy and esophagectomy.
Key Thoracic Nerves
Phrenic nerve (C3-C5): Descends along the anterior surface of the anterior scalene, enters the chest, and courses along the pericardium. Innervates the diaphragm; injury causes hemidiaphragm paralysis. Recurrent laryngeal nerve: The left RLN loops under the aortic arch at the level of the ligamentum arteriosum; the right RLN loops under the right subclavian artery. Both ascend in the tracheoesophageal groove to innervate the vocal cords. Left RLN injury is a risk during aortic arch surgery, PDA ligation, and left-sided thoracic procedures — causes hoarseness and aspiration risk. Vagus nerve (CN X): Courses through the mediastinum, giving off the recurrent laryngeal nerves; contributes to pulmonary and esophageal plexuses.
03 Cardiopulmonary Physiology
Cardiac Output Determinants
Cardiac output (CO) = Heart rate (HR) × Stroke volume (SV). Normal CO is ~5 L/min. Stroke volume is determined by three factors: preload (end-diastolic volume — Frank-Starling mechanism), afterload (wall stress, approximated by SVR), and contractility (intrinsic myocardial force generation, independent of loading conditions). The Frank-Starling law states that increased preload (myocardial fiber stretch) increases SV up to a plateau — beyond which further preload causes pulmonary congestion without improved output (the descending limb of the curve in heart failure).
Cardiac index (CI) = CO / body surface area (BSA). Normal CI = 2.5-4.0 L/min/m². CI < 2.2 L/min/m² = cardiogenic shock territory. Systemic vascular resistance (SVR) = (MAP - CVP) / CO × 80. Normal SVR = 800-1200 dynes·s/cm&sup5;. Mixed venous oxygen saturation (SvO2) measured from the PA catheter reflects the balance between oxygen delivery and consumption — normal 65-75%; < 60% suggests inadequate CO or increased extraction.
Coronary Physiology
The coronary arteries fill primarily during diastole (unlike all other systemic arteries, which fill during systole) because the intramyocardial pressure during systole compresses the coronary vessels, especially in the subendocardium. This makes the subendocardium the most vulnerable zone for ischemia — it is the "last meadow" of coronary flow. Conditions that reduce diastolic time (tachycardia) or diastolic pressure (aortic regurgitation, sepsis) impair coronary perfusion.
Pulmonary Physiology Essentials
Preoperative pulmonary function tests (PFTs) are critical before lung resection: FEV1 (forced expiratory volume in 1 second) and DLCO (diffusing capacity for carbon monoxide) are the two most important values. Predicted postoperative (ppo) FEV1 is calculated based on the number of functioning segments to be removed. ppoFEV1 > 40% predicted and ppoDLCO > 40% predicted are thresholds for lobectomy without further testing. If ppo values are < 40%, exercise testing (stair climb, shuttle walk, or formal CPET with VO2max) is needed. VO2max > 15 mL/kg/min supports operability; < 10 mL/kg/min is prohibitive for major resection.
| Parameter | Safe for Lobectomy | Safe for Pneumonectomy | High Risk / Further Testing Needed |
|---|---|---|---|
| ppoFEV1 | > 40% predicted | > 40% predicted | < 40% |
| ppoDLCO | > 40% predicted | > 40% predicted | < 40% |
| VO2max | > 15 mL/kg/min | > 20 mL/kg/min | < 10 mL/kg/min (prohibitive) |
Myocardial Oxygen Supply-Demand Balance
Understanding myocardial oxygen balance is fundamental to cardiothoracic surgery. Oxygen demand is determined by: heart rate (most important modifiable factor), wall tension (afterload), contractility, and LV mass. Oxygen supply depends on: coronary blood flow (determined by coronary perfusion pressure and diastolic time), hemoglobin concentration, and arterial oxygen saturation. The heart extracts ~70-80% of delivered oxygen at baseline (highest extraction ratio of any organ), leaving minimal reserve — increased demand can only be met by increased flow, not increased extraction. This is why coronary stenosis causes ischemia with exertion before it causes ischemia at rest.
Cardiopulmonary Interactions
Positive-pressure ventilation increases intrathoracic pressure, which reduces venous return (preload) to the right heart but reduces LV afterload (transmural pressure). In patients with normal RV function, the net effect of positive-pressure ventilation is usually minimal. However, in patients with RV failure, positive-pressure ventilation can severely compromise CO by reducing preload to an already failing RV. Conversely, in LV failure, positive-pressure ventilation can be beneficial by reducing LV afterload. Understanding these interactions is critical for ventilator management after cardiac surgery, especially when weaning from mechanical ventilation in patients with RV dysfunction.
04 The Cardiothoracic Exam & Preoperative Assessment
Cardiac Risk Stratification
Two primary scoring systems dominate cardiothoracic surgical risk assessment:
The European System for Cardiac Operative Risk Evaluation II is a logistic regression model that predicts operative mortality for cardiac surgery. Variables include: patient factors (age, sex, renal function — creatinine clearance, extracardiac arteriopathy, poor mobility, prior cardiac surgery, chronic lung disease, active endocarditis, critical preoperative state, diabetes on insulin), cardiac factors (NYHA class, CCS angina class, LV function — EF categories, recent MI, pulmonary hypertension), and operative factors (urgency, weight of intervention, surgery on thoracic aorta). Provides a predicted mortality percentage. Available as an online calculator at euroscore.org.
The Society of Thoracic Surgeons (STS) Risk Calculator is the most widely used risk model in North America. Predicts operative mortality and major morbidity for CABG, valve surgery, and combined procedures. Incorporates > 40 variables including demographics, comorbidities, hemodynamics, and procedural details. STS predicted risk of mortality (PROM) is reported as a percentage. An STS-PROM ≥ 8% is considered extreme risk and is the threshold that may qualify patients for transcatheter approaches (TAVR over SAVR). STS short-term risk scores also predict renal failure, prolonged ventilation, stroke, reoperation, and deep sternal wound infection.
Preoperative Cardiac Workup
Standard preoperative evaluation includes: transthoracic echocardiography (TTE) — EF, wall motion abnormalities, valvular function, RV function, PA systolic pressure; coronary angiography (cardiac catheterization) — mandatory before valve surgery and for patients with suspected CAD; CTA/MRA for aortic pathology; carotid duplex ultrasound — indicated before CABG in patients ≥ 65, known carotid disease, left main disease, peripheral vascular disease, history of stroke/TIA, or carotid bruit; PFTs — before any lung resection or if there is concern for significant pulmonary disease.
Frailty and Functional Status
Frailty is increasingly recognized as a predictor of poor outcomes after cardiac surgery. The 5-meter walk test (> 6 seconds = frail), grip strength, Katz Activities of Daily Living, and the Essential Frailty Toolset (EFT) are used. Frailty scores are now incorporated into decision-making for TAVR vs SAVR (Heart Team approach). A 6-minute walk test < 150 meters is a marker of poor functional status that increases perioperative risk.
Preoperative Optimization
Antiplatelet management: Aspirin can be continued through cardiac surgery. Clopidogrel and ticagrelor should be held 5 days before elective CABG; prasugrel 7 days. In urgent cases, platelet transfusion may be needed intraoperatively. Anticoagulation: Warfarin should be stopped 5 days preoperatively (bridge with heparin if high thrombotic risk — mechanical valve, recent VTE). DOACs: stop 2-4 days preoperatively depending on renal function and specific agent. Diabetes: Target glucose 140-180 mg/dL perioperatively — tight glycemic control reduces sternal wound infection. Hold metformin 48 hours before surgery (lactic acidosis risk with contrast/hypoperfusion). Continue insulin with dose adjustments. Anemia: Preoperative Hgb < 12 g/dL is an independent risk factor for transfusion and adverse outcomes. Preoperative iron supplementation and/or erythropoietin in elective cases with time allows optimization.
Carotid Disease in Cardiac Surgery Patients
Significant carotid stenosis is present in 5-10% of patients undergoing CABG. The management of combined carotid and coronary disease is debated: (1) Staged approach — CEA first, CABG later (or vice versa): Reduces the complexity of each operation but exposes the patient to the risk of the second procedure during the interval. (2) Combined/simultaneous CEA + CABG: Single anesthetic and recovery, but longer operative time and potentially higher combined stroke and cardiac event risk. (3) CAS (carotid stenting) before CABG: Less invasive than CEA but requires DAPT (which complicates CABG within days). Current approach is individualized: symptomatic carotid stenosis (≥ 50%) — address carotid first or combined; asymptomatic carotid stenosis (≥ 80%) — consider staged or combined; asymptomatic < 80% — proceed with CABG alone. The CREST trial showed equivalent outcomes for CEA vs CAS in general, but surgical patients tend to be younger and CAS patients older.
Imaging for Cardiothoracic Surgery
Transthoracic echocardiography (TTE): First-line for valve assessment, EF, wall motion. Transesophageal echocardiography (TEE): Superior resolution for mitral valve anatomy, aortic pathology, endocarditis vegetations, and intracardiac thrombus; mandatory intraoperatively for valve repair assessment. Coronary angiography: Gold standard for coronary anatomy; mandatory before valve surgery in patients ≥ 40 or with CAD risk factors. Cardiac CT: Coronary CT angiography is increasingly used as a non-invasive alternative; CT is essential for aortic pathology (aneurysm sizing, dissection anatomy, calcification mapping for cannulation strategy) and preoperative planning for redo sternotomy (relationship of cardiac structures to sternum). Cardiac MRI: Best for myocardial viability (late gadolinium enhancement — predicts recovery after revascularization), tissue characterization (myocarditis, sarcoid, iron overload), and complex congenital heart disease anatomy. PET/CT: Standard for lung cancer staging and prosthetic valve endocarditis diagnosis (FDG uptake around the prosthesis).
05 Coronary Artery Bypass Grafting (CABG)
Indications for CABG
Current ACC/AHA guideline class I indications for CABG include:
- Left main disease (≥ 50% stenosis) — CABG is superior to PCI (EXCEL trial controversy notwithstanding; 5-year data favor CABG)
- Three-vessel disease — especially with reduced LV function (EF < 50%) or diabetes
- Two-vessel disease with proximal LAD involvement and either diabetes, LV dysfunction, or significant ischemia on stress testing
- Failed PCI or PCI-unsuitable anatomy (chronic total occlusions, heavily calcified lesions, bifurcation lesions)
- CABG + valve surgery when concomitant significant CAD is present
The SYNTAX score quantifies the complexity of coronary artery disease based on angiographic features (number of lesions, location, bifurcation involvement, calcification, total occlusions). Low SYNTAX (≤ 22): PCI and CABG have similar outcomes — PCI reasonable. Intermediate SYNTAX (23-32): CABG generally preferred, especially with diabetes. High SYNTAX (≥ 33): CABG is clearly superior to PCI. The SYNTAX score is now a cornerstone of the Heart Team discussion.
Graft Selection
Graft choice is one of the most consequential decisions in CABG. Patency rates differ dramatically between conduits:
| Conduit | Target Territory | 1-Year Patency | 10-Year Patency | Key Notes |
|---|---|---|---|---|
| LIMA (left internal mammary/thoracic artery) | LAD (gold standard) | > 98% | 90-95% | Single most important graft; improves survival; resistant to atherosclerosis |
| RIMA (right internal mammary artery) | LCx or LAD (if BIMA) | > 95% | 85-90% | Bilateral IMA (BIMA) grafting controversial — ART trial showed no survival advantage at 10 years; increased sternal wound infection risk in diabetics and obese patients |
| Radial artery | LCx, RCA (≥ 70-90% target stenosis) | ~92% | 80-85% | Prone to spasm (give CCB or nitrate postop); superior to SVG in high-grade stenosis targets (RADIAL trial) |
| Saphenous vein graft (SVG) | Any territory | 85-90% | 50-60% | Most commonly used conduit; prone to neointimal hyperplasia (1-5 years) and atherosclerosis (> 5 years); endoscopic harvest reduces wound complications |
On-Pump vs Off-Pump CABG
On-pump CABG (ONCAB): Performed with cardiopulmonary bypass (CPB) and cardioplegic arrest. The heart is still and bloodless, providing optimal surgical conditions. This remains the standard approach in most centers. Off-pump CABG (OPCAB): Performed on the beating heart using mechanical stabilizers (e.g., Octopus device). Avoids the inflammatory response and potential neurological complications of CPB. The ROOBY trial (VA; 2009) showed worse graft patency and a higher rate of repeat revascularization with OPCAB compared to ONCAB at 1 year. The CORONARY trial (2016, 5-year data) showed no significant difference in death, MI, stroke, or repeat revascularization between ONCAB and OPCAB. Current consensus: OPCAB may benefit select high-risk patients (porcelain aorta, severe aortic atherosclerosis, CKD), but ONCAB remains the standard for most patients.
Surgical Technique Highlights
CABG is typically performed via median sternotomy. After harvesting conduits, CPB is established (aortic and right atrial cannulation), the aorta is cross-clamped, and cardioplegia is delivered (see Section 20). Distal anastomoses are constructed first (graft-to-coronary artery), followed by proximal anastomoses (graft-to-aorta) — either during cross-clamp or with a partial occlusion clamp. The LIMA is typically used as an in situ pedicled graft (left intact at its subclavian origin). SVGs are reversed (valves) or stripped of valves before use. The "no-touch" SVG harvest technique preserves the perivascular tissue surrounding the vein, improving long-term patency.
Redo CABG & Special Situations
Redo sternotomy carries significant risk — patent LIMA or SVG grafts may be adherent to the posterior sternum and can be injured during re-entry. Preoperative CT angiography is mandatory to map graft position relative to the sternum. Femoral cannulation for CPB before sternal re-entry ("peripheral cannulation first") is a common safety strategy. Porcelain aorta (severely calcified ascending aorta): standard aortic cross-clamping and proximal anastomoses are dangerous (stroke risk from calcium emboli). Strategies include: no-touch aorta with OPCAB, proximal anastomotic devices (Heartstring), or alternative arterial inflow (axillary artery, innominate artery). Epiaortic ultrasound is used intraoperatively to identify safe sites for cannulation and clamping.
Postoperative CABG Care
Aspirin 81-325 mg within 6 hours postoperatively (Class I) — improves SVG patency. DAPT (aspirin + clopidogrel 75 mg or ticagrelor 90 mg BID) for 12 months if the patient had ACS preoperatively. Beta-blockers should be continued or initiated postoperatively (reduces POAF). Statin therapy should be continued. Cardiac rehabilitation referral is standard. SVG failure is common: ~10-15% occlude within the first year (thrombosis), ~25% by 5 years (neointimal hyperplasia), and ~50% by 10 years (atherosclerosis). Antiplatelet therapy, statins, and smoking cessation are the cornerstones of graft preservation.
06 Landmark Trials & Revascularization Strategy
SYNTAX Trial (2009; PMID: 19228612): Left main or 3-vessel disease randomized to PCI (TAXUS stent) vs CABG. At 5 years, CABG had lower MACCE in 3-vessel disease and intermediate/high SYNTAX scores. PCI was non-inferior only in low SYNTAX scores. Established the SYNTAX score as the key decision-making tool.
FREEDOM Trial (2012; PMID: 23121323): Diabetic patients with multivessel CAD. CABG significantly reduced death and MI compared to PCI with DES at 5 years (all-cause mortality 10.9% vs 16.3%). CABG had more strokes. Conclusion: CABG is the preferred revascularization strategy in diabetics with multivessel disease.
ISCHEMIA Trial (2020; PMID: 32227756): Stable CAD patients with moderate-to-severe ischemia on stress testing. An initial invasive strategy (PCI or CABG) did not reduce death or MI compared to optimal medical therapy alone at 3.2 years. Changed paradigm: stable CAD — even with significant ischemia — should be managed with optimal medical therapy first. Note: excluded left main disease.
ART Trial (2019; PMID: 31582132): Bilateral IMA (BIMA) vs single IMA (SIMA) in CABG. At 10 years, no significant survival difference (BIMA 22.6% vs SIMA 22.2% mortality). High crossover rate from BIMA to SIMA (14%). BIMA may benefit younger patients with longer life expectancy.
RADIAL Trial (2018; PMID: 30508392): Radial artery vs saphenous vein as second conduit in CABG. Radial artery had significantly lower incidence of adverse cardiac events and better patency at 5 years. Supports use of radial artery over SVG as the second conduit of choice.
Current Revascularization Strategy Summary
| Clinical Scenario | Recommended Strategy | Evidence |
|---|---|---|
| Left main, low SYNTAX (≤ 22) | PCI or CABG (Heart Team decision) | EXCEL, NOBLE (contradictory) |
| Left main, intermediate/high SYNTAX | CABG preferred | SYNTAX, EXCEL 5-year |
| 3-vessel CAD | CABG preferred (especially with diabetes) | SYNTAX, FREEDOM |
| 3-vessel CAD, diabetic | CABG strongly preferred | FREEDOM |
| 2-vessel with proximal LAD | CABG preferred if diabetes or LV dysfunction | Guidelines consensus |
| Stable CAD with ischemia (no left main) | Optimal medical therapy first | ISCHEMIA |
| STEMI with multivessel CAD | PCI to culprit lesion; CABG or staged PCI for residual disease | COMPLETE trial |
07 Aortic Valve Disease
Aortic Stenosis (AS)
The most common valvular indication for surgery in adults. Etiologies: degenerative/calcific (most common in elderly, typically > 65), bicuspid aortic valve (most common congenital cardiac anomaly, prevalence ~1-2%, presents earlier at age 40-60), and rheumatic (commissural fusion, now uncommon in developed countries).
Classic symptom triad of severe AS: angina (5-year survival without surgery), syncope (3-year survival), heart failure (2-year survival). Once symptoms develop, the natural history is grim without intervention.
| Parameter | Mild | Moderate | Severe |
|---|---|---|---|
| Aortic valve area (AVA) | > 1.5 cm² | 1.0-1.5 cm² | < 1.0 cm² |
| Mean gradient | < 20 mmHg | 20-40 mmHg | > 40 mmHg |
| Peak velocity | < 3.0 m/s | 3.0-4.0 m/s | > 4.0 m/s |
| Indexed AVA | > 0.85 cm²/m² | 0.6-0.85 cm²/m² | < 0.6 cm²/m² |
Surgical Aortic Valve Replacement (SAVR)
Indications for intervention (Class I): Severe AS with symptoms; severe AS undergoing other cardiac surgery (CABG, aortic surgery); severe AS with EF < 50%. Approach: median sternotomy, CPB, aortotomy above the sinotubular junction, excision of native valve and decalcification of the annulus, sizing and implantation of prosthetic valve, closure of aortotomy.
| Prosthesis Type | Durability | Anticoagulation | Ideal Candidate |
|---|---|---|---|
| Mechanical (St. Jude, On-X) | Lifetime (> 30 years) | Lifelong warfarin (INR 2.0-3.0 for aortic; 2.5-3.5 for mitral) | Age < 50, willing/able to take warfarin, no contraindication to anticoagulation |
| Bioprosthetic (Carpentier-Edwards, Hancock, porcine or bovine pericardial) | 10-20 years (accelerated degeneration in younger patients) | Aspirin only (warfarin for first 3-6 months is optional/debated) | Age ≥ 50 (aortic), ≥ 65-70 (mitral), contraindication to anticoagulation, women of childbearing age (warfarin is teratogenic) |
| Sutureless / rapid-deployment (Perceval, INTUITY) | Similar to bioprosthetic | Same as bioprosthetic | Minimally invasive AVR, small annulus, redo surgery, short cross-clamp time desired |
TAVR vs SAVR
Transcatheter aortic valve replacement (TAVR) has transformed AS management. Based on the PARTNER and Evolut trials: Prohibitive/extreme risk: TAVR is standard (PARTNER 1B, Evolut Extreme Risk). High risk: TAVR is non-inferior or superior to SAVR (PARTNER 1A, CoreValve High Risk). Intermediate risk: TAVR is non-inferior to SAVR (PARTNER 2, SURTAVI). Low risk: TAVR is non-inferior to SAVR at 2 years (PARTNER 3, Evolut Low Risk). Key concern with TAVR in younger patients is valve durability, paravalvular leak, need for pacemaker (~10-20% with self-expanding valves), and long-term data beyond 5-10 years are still maturing.
Aortic Regurgitation (AR)
Etiologies: Acute AR — aortic dissection (emergency surgery), endocarditis (valve destruction), trauma. Chronic AR — bicuspid aortic valve, aortic root dilation (Marfan, annuloaortic ectasia), rheumatic disease, degenerative. Chronic AR is tolerated for years because the LV dilates (volume overload); surgery is indicated for severe AR with symptoms, EF ≤ 55%, or severe LV dilation (LV end-systolic dimension > 50 mm or indexed LVESD > 25 mm/m²).
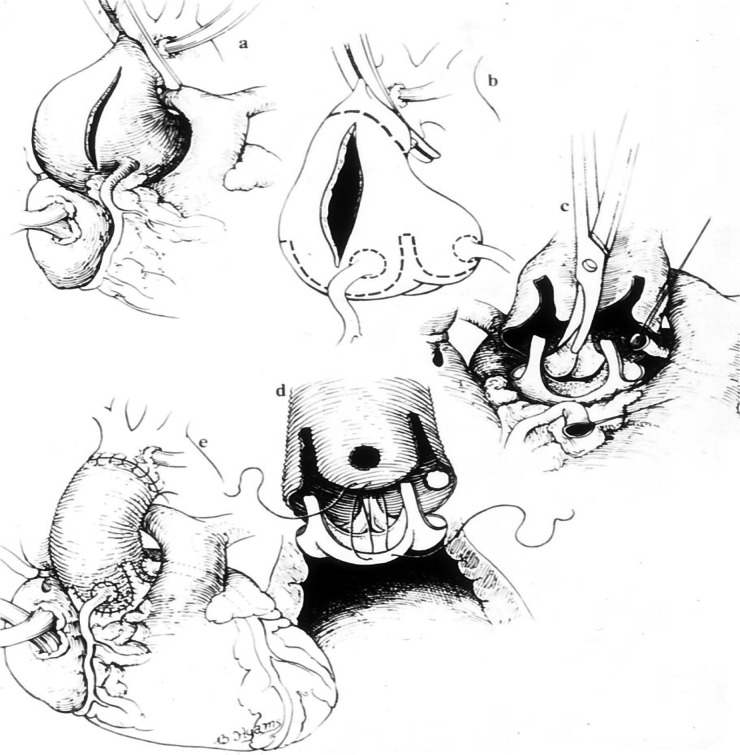
Valve-Sparing Aortic Root Replacement
When AR is caused by aortic root dilation rather than intrinsic valve disease, the native aortic valve can be preserved:
- David procedure (valve reimplantation): The native aortic valve is reimplanted inside a Dacron tube graft. The graft replaces the entire root and provides external support to the annulus, preventing future annular dilation. Preferred for Marfan patients.
- Yacoub procedure (valve remodeling): The aortic root sinuses are replaced with a scalloped Dacron graft, preserving the native valve and reconstructing the sinuses of Valsalva. Does NOT fix the annulus — higher late failure rate due to annular dilation. Less commonly performed now.
Bicuspid Aortic Valve (BAV)
The most common congenital cardiac anomaly, present in ~1-2% of the population (male:female ratio 3:1). The valve has two functional cusps instead of three, most commonly with fusion of the right and left coronary cusps (type 1 — ~70%). BAV is associated with: accelerated aortic stenosis (calcific degeneration occurs decades earlier than tricuspid valves), aortic regurgitation, aortic root and ascending aortic dilation (BAV aortopathy — a connective tissue abnormality independent of hemodynamic effects), coarctation of the aorta, and infective endocarditis. Guidelines recommend aortic root/ascending aorta replacement at ≥ 5.0-5.5 cm in BAV patients, or at ≥ 4.5 cm if undergoing AVR. First-degree relatives of BAV patients should be screened with echocardiography.
Low-Flow, Low-Gradient Aortic Stenosis
A diagnostic challenge: AVA < 1.0 cm² (severe) but mean gradient < 40 mmHg (not meeting gradient criteria for severe). Two forms: (1) Classic low-flow, low-gradient AS with reduced EF: EF < 50%, low stroke volume (SVI < 35 mL/m²). Must distinguish true severe AS from "pseudo-severe" AS (valve appears stenotic only because the weakened ventricle cannot open it fully). Dobutamine stress echo differentiates: true severe AS shows increased gradient (> 40 mmHg) without significant increase in AVA; pseudo-severe AS shows increase in AVA (> 1.0 cm²) with improved EF. Patients with true severe AS benefit from SAVR even with low EF. (2) Paradoxical low-flow, low-gradient AS with preserved EF: Normal EF but low stroke volume due to small LV cavity, concentric hypertrophy, or diastolic dysfunction. Diagnosis confirmed by CT calcium scoring (Agatston score > 2000 in men, > 1200 in women confirms severe AS) and clinical correlation.
Prosthetic Valve Complications
Structural valve degeneration (SVD): Bioprosthetic valves degenerate over time — leaflet calcification, fibrosis, and tearing. Younger patients (< 40) have faster degeneration. Paravalvular leak (PVL): Regurgitation around the sewing ring — occurs in ~5-10% of surgical prostheses; higher after TAVR. Significant PVL causes hemolytic anemia (fragmented RBCs on smear, elevated LDH, haptoglobin depressed) and heart failure. Treatment: percutaneous closure with Amplatzer devices or surgical re-repair. Prosthetic valve thrombosis: More common with mechanical valves in the mitral position. Presents with new or increased transvalvular gradient, muffled opening/closing clicks, and heart failure. Treatment: thrombolysis (tPA) for right-sided or left-sided thrombosis in high-surgical-risk patients; surgical thrombectomy or valve replacement for left-sided thrombosis in acceptable surgical candidates. Patient-prosthesis mismatch (PPM): Prosthetic valve EOA too small relative to BSA (indexed EOA < 0.85 cm²/m² for aortic; < 1.2 cm²/m² for mitral). Causes persistent transvalvular gradients and failure to relieve symptoms. Prevention: choose the largest valve size that fits; consider root enlargement (Nicks, Manouguian, or Konno procedures) if the annulus is too small for an adequate valve.
08 Mitral Valve Disease
Mitral Regurgitation (MR)
MR is classified by the Carpentier classification based on leaflet motion: Type I — normal leaflet motion (annular dilation, leaflet perforation from endocarditis); Type II — excessive leaflet motion (leaflet prolapse from myxomatous degeneration/Barlow's disease, chordal rupture, papillary muscle rupture); Type IIIa — restricted leaflet motion in systole and diastole (rheumatic disease — thickened, fibrotic leaflets); Type IIIb — restricted leaflet motion in systole only (functional/ischemic MR — papillary muscle displacement from LV dilation).
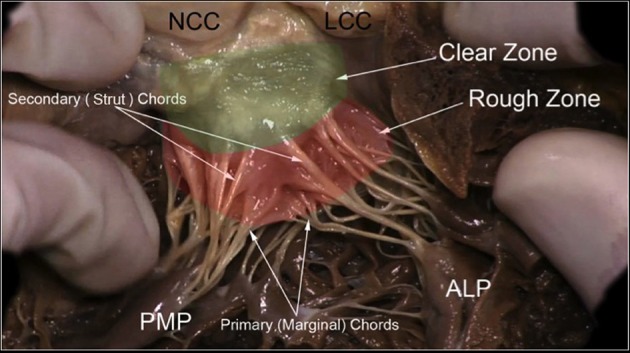
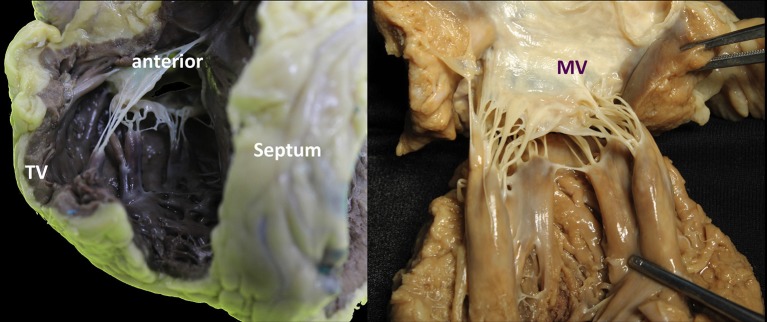
Primary (Degenerative) MR — Repair vs Replacement
Mitral valve repair is strongly preferred over replacement for primary (degenerative) MR. Repair preserves the subvalvular apparatus, maintains LV geometry, avoids anticoagulation, and has lower operative mortality (~1% vs 4-6% for replacement). The cornerstone repair technique involves: triangular or quadrangular resection of the prolapsing posterior leaflet segment (most commonly P2), artificial neochordae (ePTFE Gore-Tex sutures) for anterior leaflet prolapse, and a rigid or semi-rigid annuloplasty ring to restore annular geometry and prevent future dilation. Repair success rates at experienced centers exceed 95% for posterior leaflet pathology.
Mitral Stenosis (MS)
Almost always rheumatic in etiology. Characterized by commissural fusion, leaflet thickening, and chordal shortening. Severe MS is defined as mitral valve area < 1.0 cm² (normal 4-6 cm²). Symptoms typically appear when MVA < 1.5 cm² — dyspnea, orthopnea, hemoptysis, atrial fibrillation, and thromboembolism. Initial intervention for favorable anatomy: percutaneous mitral balloon commissurotomy (PMBC) — guided by the Wilkins score (echocardiographic score 0-16 assessing leaflet mobility, thickening, calcification, and subvalvular involvement; score ≤ 8 is favorable for PMBC). Surgical options: open commissurotomy or mitral valve replacement (MVR) for heavily calcified/fibrotic valves or failed PMBC.
Functional (Secondary) MR
Results from LV dilation (ischemic or dilated cardiomyopathy) displacing the papillary muscles and restricting leaflet coaptation — Carpentier type IIIb. The valve leaflets themselves are normal. Management is primarily medical (guideline-directed medical therapy for heart failure, CRT if indicated). Surgical repair with restrictive annuloplasty reduces MR but does NOT improve survival compared to replacement (CTSN Severe Ischemic MR Trial, PMID: 24245543). MitraClip (transcatheter edge-to-edge repair): The COAPT trial (2018; PMID: 30280640) showed significant reduction in heart failure hospitalizations and death in patients with functional MR despite maximal GDMT. The MITRA-FR trial, however, showed no benefit — the difference was in patient selection (COAPT selected patients with disproportionate MR relative to LV dilation).
09 Tricuspid Valve Disease & Endocarditis
Tricuspid Regurgitation (TR)
The "forgotten valve" — TR is commonly functional (secondary) due to right ventricular dilation from pulmonary hypertension, left-sided heart disease, or atrial fibrillation. Significant TR is present in ~4% of the population and is associated with reduced survival. Indications for surgical repair: severe TR in patients undergoing left-sided valve surgery (tricuspid annuloplasty is added — Class I if severe TR; Class IIa if moderate TR or annular dilation ≥ 40 mm even with less-than-severe TR). Isolated tricuspid surgery is considered for severe symptomatic TR refractory to medical therapy.
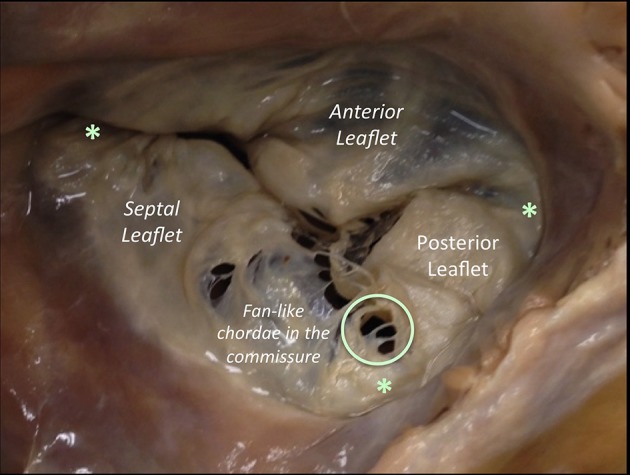
Surgical options: tricuspid annuloplasty ring (most common — restores annular geometry), DeVega annuloplasty (purse-string suture technique — simpler but higher recurrence), or tricuspid valve replacement (for severely destroyed valves — bioprosthetic strongly preferred over mechanical due to high thrombosis risk of mechanical valves in the tricuspid position). Transcatheter tricuspid valve interventions (TriClip edge-to-edge repair, EVOQUE valve replacement) are emerging for high-surgical-risk patients, with the TRILUMINATE trial demonstrating safety and efficacy of TriClip.
Infective Endocarditis — Surgical Indications
Endocarditis is managed medically with prolonged IV antibiotics (4-6 weeks), but surgery is indicated for specific complications:
- Heart failure due to valvular dysfunction (most common indication for surgery in endocarditis)
- Uncontrolled infection — persistent bacteremia > 5-7 days despite appropriate antibiotics, perivalvular abscess (especially aortic root abscess), fungal endocarditis (Candida, Aspergillus)
- Prevention of embolism — large vegetation (> 10 mm) with embolic events; very large vegetation (> 15 mm) even without embolic events (especially left-sided, mobile, on the mitral valve anterior leaflet)
- Prosthetic valve endocarditis (PVE) with dehiscence, abscess, or persistent bacteremia
Surgical approach: aggressive debridement of all infected tissue, abscess drainage, valve repair (if feasible) or replacement. Aortic root abscess may require root replacement with homograft (lower reinfection rate compared to prosthetic valve). In right-sided (tricuspid) endocarditis (typically in IV drug users), surgery is reserved for persistent sepsis, large vegetations (> 20 mm), or right heart failure. Valve excision without replacement ("valvectomy") was historically done but has fallen out of favor; tricuspid repair or bioprosthetic replacement is preferred.
10 Thoracic Aortic Aneurysm
Definition & Etiology
A thoracic aortic aneurysm (TAA) is defined as dilation of the thoracic aorta to ≥ 1.5 times the expected normal diameter (approximately ≥ 4.5 cm for the ascending aorta in adults). Etiologies: degenerative (medial degeneration — most common), connective tissue disorders (Marfan syndrome — FBN1 mutation, Loeys-Dietz syndrome — TGFBR mutations, vascular Ehlers-Danlos — COL3A1 mutation), bicuspid aortic valve-associated aortopathy, atherosclerotic, post-dissection, mycotic/infected, and post-traumatic.
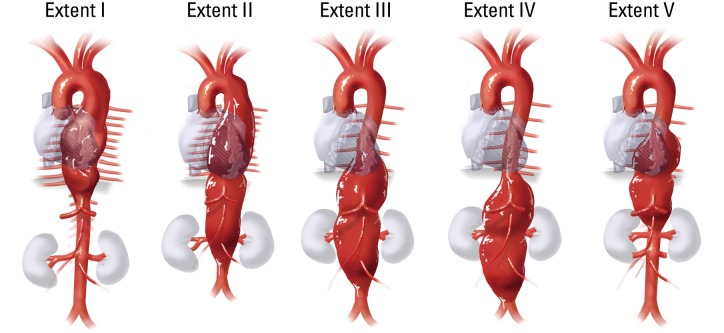
Crawford Classification of Thoracoabdominal Aortic Aneurysms (TAAA)
| Type | Extent | Spinal Cord Ischemia Risk |
|---|---|---|
| Extent I | Left subclavian to above renal arteries | Moderate |
| Extent II | Left subclavian to aortic bifurcation (entire DTA + abdominal aorta) | Highest (~20-30%) |
| Extent III | Mid-descending thoracic aorta (T6) to aortic bifurcation | Moderate-high |
| Extent IV | Diaphragm (T12) to aortic bifurcation (thoracoabdominal portion) | Lower |
| Extent V | Below T6 to above renal arteries | Moderate |
Surgical Thresholds for Repair
| Location / Condition | Threshold for Elective Repair |
|---|---|
| Ascending aorta (degenerative) | ≥ 5.5 cm |
| Ascending aorta with Marfan syndrome | ≥ 5.0 cm (or ≥ 4.5 cm with risk factors: family history of dissection, rapid growth > 0.5 cm/year, severe AR) |
| Ascending aorta with Loeys-Dietz | ≥ 4.2-4.4 cm (or by internal diameter on TEE ≥ 4.0 cm) |
| Ascending aorta with bicuspid aortic valve | ≥ 5.0-5.5 cm (if undergoing AVR, prophylactic root/ascending replacement at ≥ 4.5 cm) |
| Descending thoracic aorta | ≥ 5.5-6.0 cm |
| Thoracoabdominal aorta | ≥ 5.5-6.0 cm |
| Rate of growth threshold | > 0.5 cm/year regardless of diameter |
Connective Tissue Disorders & the Aorta
Several genetic conditions predispose to aortic aneurysm and dissection at younger ages, with lower diameter thresholds for intervention:
| Disorder | Gene / Defect | Key Features | Surgical Threshold |
|---|---|---|---|
| Marfan syndrome | FBN1 (fibrillin-1) | Tall stature, arachnodactyly, lens subluxation, aortic root dilation, mitral valve prolapse | Aortic root ≥ 5.0 cm (or ≥ 4.5 cm with risk factors) |
| Loeys-Dietz syndrome | TGFBR1/2 (TGF-beta receptors) | Hypertelorism, bifid uvula/cleft palate, arterial tortuosity, aggressive aortic disease at smaller diameters | Aortic root ≥ 4.0-4.2 cm (most aggressive threshold) |
| Vascular Ehlers-Danlos (type IV) | COL3A1 (type III collagen) | Translucent skin, easy bruising, arterial/visceral rupture; tissue is extremely friable — surgery is high-risk | Surgery only for acute complications (extremely fragile tissues make elective surgery dangerous) |
| Turner syndrome (45,X) | Monosomy X | Short stature, webbed neck, bicuspid aortic valve (30%), coarctation, aortic dissection risk | Aortic size index ≥ 2.5 cm/m² (adjusted for small body size) |
All patients with connective tissue disorders and aortic pathology should be managed at specialized aortic centers. First-degree relatives of patients with heritable thoracic aortic disease (HTAD) should be screened with echocardiography and genetic testing when available. Beta-blockers (and in Marfan syndrome, losartan per the Pediatric Heart Network trial) are used to reduce aortic wall stress and slow growth, though evidence for losartan was mixed.
Surveillance of Thoracic Aortic Disease
Patients with known thoracic aortic dilation below surgical threshold require serial imaging. Recommended intervals: stable < 4.0 cm — repeat in 12 months to establish growth rate, then every 12-24 months if stable. 4.0-4.5 cm — annual imaging (CT or MRI; MRI preferred for young patients to avoid cumulative radiation). > 4.5 cm or rapid growth — imaging every 6 months. Growth rate > 0.5 cm/year is an indication for surgery regardless of absolute diameter. Echocardiography is adequate for aortic root surveillance but cannot visualize the descending aorta — CT or MRI is required for complete thoracic aortic surveillance.
Chronic Type B Dissection
After the acute phase (> 14 days), many type B dissections become chronic. The false lumen may remain patent (higher risk of late aneurysmal dilation — 60-70% will develop aneurysmal degeneration over 5 years), become partially thrombosed (highest risk of adverse aortic events), or completely thrombose (most favorable). Indications for intervention in chronic type B dissection: aneurysmal dilation of the false lumen (≥ 5.5-6.0 cm), rapid growth (> 1 cm/year), persistent pain, or malperfusion. TEVAR can be performed for chronic dissection but is more complex than in the acute setting — the septum between true and false lumen becomes fibrotic and rigid, and fenestrations may be needed. Open thoracoabdominal repair may be required for extensive chronic dissection with aneurysmal degeneration.
Post-dissection surveillance is lifelong: CTA at 1, 3, 6, and 12 months after the acute event, then annually. Monitor for: aortic diameter increase, false lumen growth, new entry/re-entry tears, branch vessel compromise, and development of new dissection. Aggressive blood pressure control (target < 130/80 mmHg) with beta-blockers is maintained indefinitely. First-degree relatives should be screened with echocardiography, as 20% of thoracic aortic dissections have a familial component.
11 Acute Aortic Dissection
Classification Systems
Stanford Type A: Any dissection involving the ascending aorta (regardless of the entry tear site). This is a surgical emergency — mortality increases ~1-2% per hour without surgery. Stanford Type B: Dissection involving only the descending aorta (distal to the left subclavian artery). Usually managed medically unless complicated.
DeBakey Type I: Originates in the ascending aorta and extends to at least the aortic arch (often the entire aorta). DeBakey Type II: Originates in and is confined to the ascending aorta. DeBakey Type III: Originates in the descending aorta — IIIa extends distally but confined to thorax; IIIb extends below the diaphragm. Stanford A = DeBakey I + II. Stanford B = DeBakey III.
Acute Type A Dissection — Emergency Surgery
Presentation: sudden, severe "tearing" or "ripping" chest pain radiating to the back, blood pressure differential between arms, aortic regurgitation murmur, pulse deficits, and signs of malperfusion (stroke, limb ischemia, mesenteric ischemia, renal failure). Diagnosis: CT angiography (CTA) is the imaging modality of choice (sensitivity/specificity > 95%); TEE is valuable intraoperatively and at the bedside if the patient is too unstable for CT.
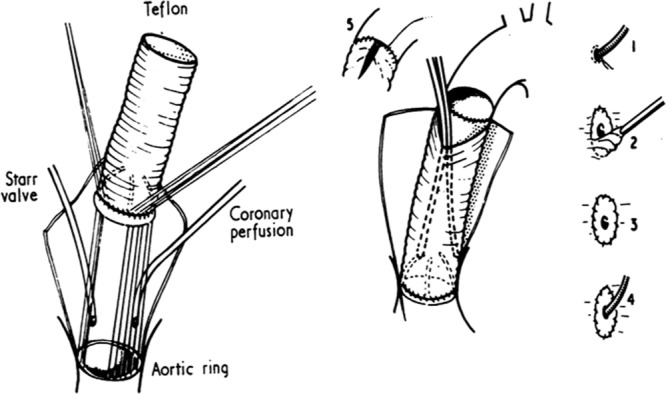
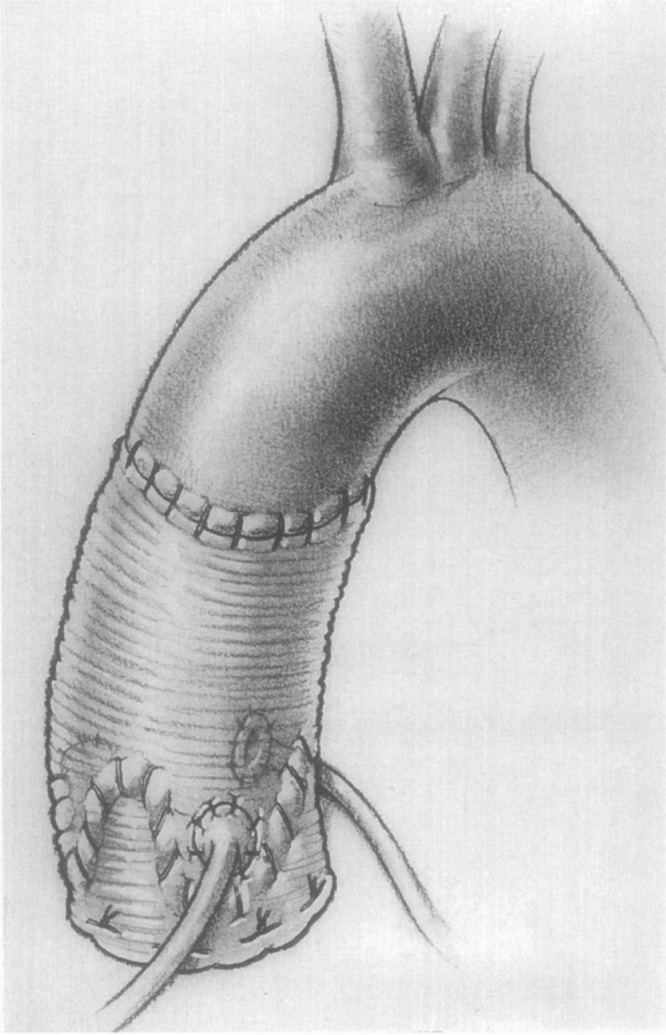
Surgical principles: The goal is to replace the ascending aorta with a Dacron tube graft, resect the primary intimal tear (entry site), and restore flow to the true lumen. The operation is performed under deep hypothermic circulatory arrest (DHCA) at 18-20°C or moderate hypothermic circulatory arrest (24-28°C) with selective antegrade cerebral perfusion (SACP). Arch involvement may require hemiarch or total arch replacement with the "elephant trunk" or "frozen elephant trunk" technique for staged descending aortic repair. Concomitant aortic root replacement (Bentall) is performed if the root is aneurysmal or the coronary ostia are dissected. Operative mortality for acute type A repair is 10-25% at experienced centers.
Acute Type B Dissection
Uncomplicated Type B: Managed medically with aggressive blood pressure control — target SBP 100-120 mmHg and heart rate < 60 bpm using IV esmolol or labetalol (beta-blockers are first-line to reduce aortic wall stress, dP/dt). Add nicardipine or nitroprusside if additional vasodilation is needed (never use vasodilators alone without beta-blockade — reflex tachycardia increases shear stress).
Complicated Type B (indications for intervention): malperfusion syndrome (visceral, renal, limb ischemia), rupture or impending rupture, uncontrolled pain despite medical therapy, rapid aortic expansion, and refractory hypertension. Treatment: TEVAR (thoracic endovascular aortic repair) — covers the primary entry tear to promote true lumen expansion and false lumen thrombosis. The INSTEAD-XL trial (PMID: 23918683) showed that preemptive TEVAR in stable Type B dissection improves aorta-specific survival at 5 years compared to medical therapy alone.
Acute Aortic Syndromes
In addition to classic aortic dissection, two related entities form the spectrum of acute aortic syndromes:
Intramural hematoma (IMH): Hemorrhage within the aortic media without an intimal tear or false lumen flow on imaging. Thought to result from rupture of vasa vasorum. CT appearance: crescent-shaped high-attenuation area in the aortic wall without flow. Management mirrors dissection: Type A IMH — surgical repair (high risk of progression to classic dissection or rupture); Type B IMH — medical management with serial imaging (many resolve spontaneously; ~10% progress to dissection).
Penetrating atherosclerotic ulcer (PAU): Ulceration of an atherosclerotic plaque that penetrates the internal elastic lamina into the media. Most common in the descending aorta of elderly hypertensive patients. Can lead to IMH, pseudoaneurysm, or rupture. Management: TEVAR for symptomatic or enlarging PAU in the descending aorta; surgical repair for ascending aorta PAU.
12 Aortic Root Surgery & TEVAR
Bentall Procedure (Composite Valve-Graft Replacement)
The Bentall procedure replaces the aortic root entirely: the aortic valve, sinuses of Valsalva, and ascending aorta are replaced with a composite graft (a valved conduit — mechanical or bioprosthetic valve sewn into a Dacron tube graft). The coronary arteries are reimplanted into the graft as "buttons" (Cabrol modification uses interposition grafts to the coronaries). Indications: aortic root aneurysm with unrepairable aortic valve disease, annuloaortic ectasia, acute type A dissection with root involvement, and Marfan syndrome (when valve-sparing root replacement is not feasible).
TEVAR (Thoracic Endovascular Aortic Repair)
Endovascular stent-graft deployment in the descending thoracic aorta via femoral artery access. Indications: descending thoracic aortic aneurysm (≥ 5.5 cm), complicated type B dissection, traumatic aortic injury (blunt aortic transection — typically at the aortic isthmus, just distal to the left subclavian), penetrating aortic ulcer, and intramural hematoma. Requirements: adequate proximal and distal "landing zones" (at least 2 cm of healthy aorta for seal), appropriate femoral/iliac access (if not, iliac conduit may be needed).
Key complications of TEVAR: endoleak (Type I — at the graft seal zones, requires reintervention; Type II — from branch vessels, usually benign; Type III — graft component separation; Type IV — graft porosity), spinal cord ischemia (coverage of long aortic segments or coverage of the left subclavian artery origin increases risk; subclavian revascularization prior to TEVAR is recommended if covering the left subclavian), stroke (from wire/catheter manipulation in the arch), and access complications.
Traumatic Aortic Injury
Blunt thoracic aortic injury (BTAI) typically results from high-speed deceleration (motor vehicle collisions, falls). The most common site of injury is the aortic isthmus — just distal to the left subclavian artery at the ligamentum arteriosum, where the relatively mobile aortic arch meets the fixed descending aorta. BTAI is graded by the Society for Vascular Surgery: Grade I (intimal tear) — observe with repeat imaging; Grade II (intramural hematoma) — TEVAR or close observation; Grade III (pseudoaneurysm) — TEVAR (now first-line for Grade III-IV); Grade IV (free rupture) — emergent repair (most die before reaching the hospital). CTA is the diagnostic modality of choice. TEVAR has largely replaced open repair for BTAI, with lower paraplegia and mortality rates. In polytrauma patients, permissive hypotension for other injuries conflicts with the need for aggressive BP control for the aortic injury — this is managed by staged intervention (address life-threatening hemorrhage first, then TEVAR within 24 hours).
Aortic Arch Surgery — Cerebral Protection
Aortic arch operations require a period of circulatory arrest because the arch vessels must be clamped or detached. Cerebral protection strategies include:
- Deep hypothermic circulatory arrest (DHCA): Cool to 18-20°C, stop the pump, and operate in a bloodless field. Safe circulatory arrest time: ~30-40 minutes at 18°C. Beyond this, neurological injury risk increases significantly.
- Selective antegrade cerebral perfusion (SACP): Cold blood is perfused directly into the cerebral vessels (via the brachiocephalic/innominate and left carotid arteries, or via the right axillary artery) during circulatory arrest. Allows moderate hypothermia (24-28°C) with extended safe arrest times (> 60 minutes). Now the most commonly used technique for complex arch surgery.
- Retrograde cerebral perfusion (RCP): Cold blood is infused retrograde via the SVC into the cerebral venous system. Provides minimal cerebral oxygen delivery but helps flush air and debris from the cerebral vessels. Less commonly used now compared to SACP.
The elephant trunk technique is used for extensive aortic disease involving the arch and descending aorta: the arch is replaced and a free-floating graft ("trunk") is left hanging in the proximal descending aorta. A second-stage operation (open or TEVAR) completes the descending aortic repair. The frozen elephant trunk combines the arch replacement with a stent-graft that is deployed into the descending aorta in a single stage, reducing the need for a second operation.
13 Lung Cancer — Staging & Surgical Management
Non-Small Cell Lung Cancer (NSCLC) Staging
Lung cancer is the leading cause of cancer death worldwide. NSCLC (~85% of lung cancers) includes adenocarcinoma (most common, ~40%), squamous cell carcinoma (~25%), and large cell carcinoma (~10%). Small cell lung cancer (~15%) is generally not treated surgically.
T (Tumor): T1a (≤ 1 cm) | T1b (> 1-2 cm) | T1c (> 2-3 cm) | T2a (> 3-4 cm) | T2b (> 4-5 cm) | T3 (> 5-7 cm or chest wall/pericardium/phrenic nerve invasion, separate nodule in same lobe) | T4 (> 7 cm or mediastinum/diaphragm/heart/great vessels/recurrent laryngeal nerve/esophagus/vertebral body/carina invasion, or separate nodule in different ipsilateral lobe)
N (Nodes): N0 (no nodal metastasis) | N1 (ipsilateral peribronchial/hilar nodes) | N2 (ipsilateral mediastinal/subcarinal nodes) | N3 (contralateral mediastinal/hilar nodes, scalene or supraclavicular nodes)
M (Metastasis): M0 (no distant metastasis) | M1a (separate nodule in contralateral lung, pleural/pericardial nodules or malignant effusion) | M1b (single extrathoracic metastasis) | M1c (multiple extrathoracic metastases)

Stage Groupings & Surgical Resectability
| Stage | TNM | 5-Year Survival | Treatment |
|---|---|---|---|
| IA1-IA3 | T1a-c N0 M0 | 77-92% | Surgical resection (lobectomy or segmentectomy for ≤ 2 cm) |
| IB | T2a N0 M0 | 68% | Surgical resection ± adjuvant chemo (if ≥ 4 cm) |
| IIA | T2b N0 M0 | 60% | Surgical resection + adjuvant chemotherapy |
| IIB | T1-2 N1 or T3 N0 | 53% | Surgical resection + adjuvant chemotherapy |
| IIIA | T1-2 N2, T3 N1, T4 N0-1 | 36% | Multimodal: neoadjuvant chemo/immunotherapy + surgery, or concurrent chemoRT |
| IIIB-IIIC | T3-4 N2-3 | 13-26% | Usually unresectable; definitive chemoradiation |
| IV | Any T, any N, M1 | < 10% | Systemic therapy (chemo, immunotherapy, targeted therapy); palliative surgery |
Surgical Resection Types
Lobectomy: Resection of an entire lobe — the gold standard for operable NSCLC. Can be performed via open thoracotomy, VATS (video-assisted thoracoscopic surgery), or robotic-assisted approaches. VATS lobectomy has become the preferred approach at many centers (shorter recovery, less pain, equivalent oncologic outcomes).
Segmentectomy (anatomical): Resection of a bronchopulmonary segment with its segmental bronchus, artery, and vein. The JCOG0802/WJOG4607L trial (2022; PMID: 35220659) and CALGB 140503 trial showed non-inferior or superior outcomes with segmentectomy vs lobectomy for peripheral NSCLC ≤ 2 cm. This has shifted practice toward sublobar resection for small peripheral tumors.
Pneumonectomy: Resection of the entire lung. Reserved for central tumors not amenable to lobectomy. Right pneumonectomy carries higher morbidity/mortality than left (larger volume of lung tissue removed, higher risk of postpneumonectomy pulmonary edema and bronchopleural fistula). Mortality: 5-8% (right) vs 2-4% (left).
Sleeve resection: Resection of a segment of the main bronchus or pulmonary artery with reanastomosis — a parenchyma-sparing alternative to pneumonectomy for centrally located tumors. Preferred over pneumonectomy when technically feasible due to lower morbidity and preserved lung function.
Mediastinal Staging
Accurate nodal staging is critical. PET/CT is standard for staging — PET-positive mediastinal nodes require pathologic confirmation before declaring a patient unresectable. Endobronchial ultrasound (EBUS) with transbronchial needle aspiration (TBNA) and endoscopic ultrasound (EUS) are first-line invasive mediastinal staging modalities. Mediastinoscopy (cervical approach, accessing stations 2R, 4R, 2L, 4L, and 7) remains the gold standard if EBUS/EUS is negative but clinical suspicion for N2 disease remains high. Chamberlain procedure (anterior mediastinotomy) specifically accesses the aortopulmonary window (station 5) and para-aortic (station 6) nodes that cannot be reached by standard cervical mediastinoscopy.
Adjuvant & Neoadjuvant Therapy for NSCLC
Adjuvant chemotherapy (cisplatin-based doublet × 4 cycles) is standard for resected stage IB (≥ 4 cm), II, and IIIA NSCLC — improves 5-year survival by ~5% absolute. Adjuvant targeted therapy: osimertinib (EGFR TKI) for resected stage IB-IIIA NSCLC with EGFR exon 19 deletion or L858R mutation — the ADAURA trial (PMID: 32955177) showed dramatic improvement in DFS (HR 0.17). Adjuvant immunotherapy: atezolizumab (anti-PD-L1) for resected stage II-IIIA NSCLC with PD-L1 ≥ 1% (IMpower010 trial). Neoadjuvant immunotherapy + chemotherapy: the CheckMate 816 trial established nivolumab + platinum-based chemo as a neoadjuvant standard, showing improved pathologic complete response rate (24% vs 2.2%) and event-free survival. Neoadjuvant chemoimmunotherapy is increasingly adopted as it allows assessment of treatment response on the surgical specimen.
Superior Sulcus (Pancoast) Tumors
Tumors arising at the lung apex that invade the chest wall, brachial plexus, subclavian vessels, and/or stellate ganglion. Classic presentation: shoulder/arm pain radiating down the ulnar distribution (C8-T1 involvement), Horner syndrome (miosis, ptosis, anhidrosis — from stellate ganglion involvement), and arm weakness/atrophy. Treatment: trimodality therapy — neoadjuvant concurrent chemoradiation (cisplatin + etoposide + 45-50 Gy) followed by en bloc surgical resection (extended posterolateral thoracotomy or anterior transcervical approach for anterior tumors involving subclavian vessels). Complete pathologic response occurs in ~30% and is associated with improved 5-year survival (~50% vs ~30% for incomplete response). Vertebral body invasion (T4) was previously considered unresectable but select cases now undergo hemivertebrectomy with spine stabilization.
Lung Metastasectomy
Surgical resection of pulmonary metastases from extrathoracic primary cancers (colorectal, renal cell, sarcoma, melanoma, head and neck) is performed when: the primary tumor is controlled, no extrathoracic disease (or limited and treatable), the patient can tolerate resection, and all metastases are technically resectable. Approach: wedge resection (most common — parenchyma-sparing) via VATS or thoracotomy. For colorectal metastases, 5-year survival after complete resection is 35-50% (vs < 5% without surgery). The PulMiCC trial (2020) challenged the benefit of pulmonary metastasectomy for colorectal cancer, though it was underpowered and did not change widespread practice.
14 Esophageal Cancer & Esophagectomy
Esophageal Cancer Overview
Two main histologic types: squamous cell carcinoma (SCC) — associated with smoking and alcohol, more common in upper/mid esophagus, higher incidence in Asia and Africa — and adenocarcinoma — associated with GERD, Barrett's esophagus, and obesity, predominantly in the lower esophagus/GEJ, now the most common type in Western countries. The progression from Barrett's esophagus to adenocarcinoma follows the metaplasia-dysplasia-carcinoma sequence: GERD causes chronic inflammation of the distal esophagus, leading to intestinal metaplasia (Barrett's), then low-grade dysplasia, high-grade dysplasia, and ultimately invasive carcinoma. Endoscopic surveillance of Barrett's esophagus is recommended: every 3-5 years for non-dysplastic Barrett's, every 6-12 months for low-grade dysplasia, and immediate intervention (endoscopic mucosal resection or radiofrequency ablation) for high-grade dysplasia or T1a tumors. Staging follows the AJCC 8th edition TNM system. Staging workup: EGD with biopsy, CT chest/abdomen/pelvis, PET/CT, and endoscopic ultrasound (EUS) for T and N staging.
Esophagectomy Approaches
| Approach | Description | Indications / Advantages |
|---|---|---|
| Ivor Lewis (transthoracic) | Laparotomy (gastric mobilization, creation of gastric conduit) + right thoracotomy (esophageal mobilization, intrathoracic anastomosis) | Standard for mid/lower esophageal and GEJ tumors; allows thoracic lymphadenectomy; most common approach |
| McKeown (three-incision) | Right thoracotomy + laparotomy + left cervical incision (cervical anastomosis) | Upper/mid esophageal tumors; cervical anastomosis allows higher resection margin; cervical leak is easier to manage than intrathoracic leak |
| Transhiatal (Orringer) | Laparotomy + cervical incision — esophagus bluntly dissected through the hiatus without thoracotomy | Avoids thoracotomy (lower pulmonary morbidity); cervical anastomosis; no thoracic lymphadenectomy (disadvantage) |
| Minimally invasive (MIE / RAMIE) | Thoracoscopic/robotic + laparoscopic approach | Reduced wound complications, shorter recovery; TIME trial showed reduced pulmonary complications vs open. ROBOT trial confirmed feasibility of robotic approach. |
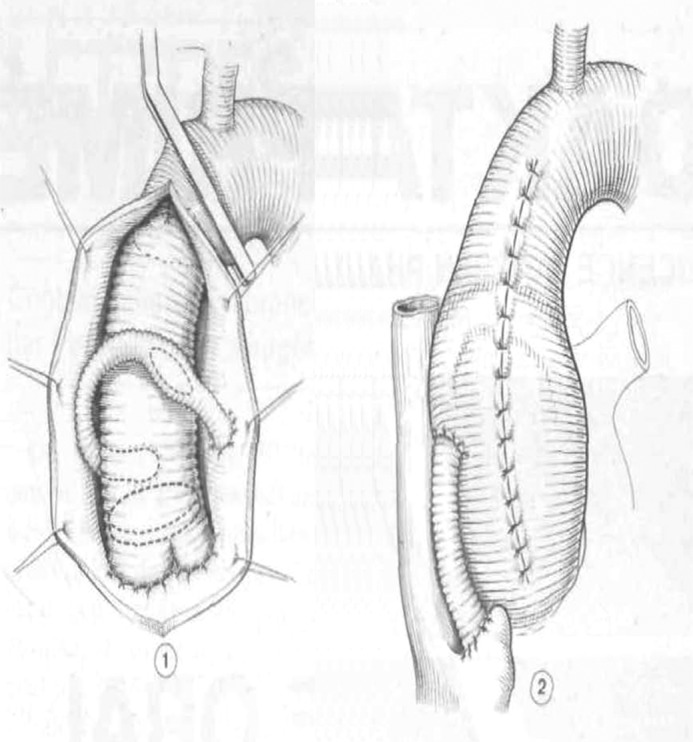
The conduit for esophageal replacement is most commonly the stomach (gastric pull-up), brought up through the posterior mediastinum or substernal route. Alternatives (if stomach unavailable): colon interposition (left or right colon) or jejunal interposition (free jejunal graft with microvascular anastomosis for cervical esophageal reconstruction).
Neoadjuvant Therapy
The CROSS trial (2012; PMID: 22646630) established neoadjuvant chemoradiation (carboplatin/paclitaxel + 41.4 Gy radiation) followed by surgery as the standard of care for locally advanced (T2-3, N0-1) esophageal cancer. Complete pathologic response (ypT0N0) was achieved in 29% of patients and was associated with significantly improved survival. The CheckMate 577 trial (2020; PMID: 33789008) showed that adjuvant nivolumab after neoadjuvant chemoRT + esophagectomy improves disease-free survival in patients who did not achieve a complete pathologic response.
Esophageal Perforation (Boerhaave Syndrome)
Boerhaave syndrome is a spontaneous transmural esophageal perforation, classically occurring in the left posterolateral distal esophagus after forceful vomiting/retching (often after heavy eating and drinking). It is a surgical emergency with a mortality of 20-40% even with treatment. Presentation: severe chest/epigastric pain after vomiting, subcutaneous emphysema, Mackler triad (vomiting, chest pain, subcutaneous emphysema), and signs of sepsis/mediastinitis. Diagnosis: CTA chest (pneumomediastinum, pleural effusion, esophageal thickening); water-soluble contrast esophagram (Gastrografin first, then thin barium if Gastrografin is negative — Gastrografin is less sensitive but avoids barium-induced mediastinitis).
Management depends on timing and contamination: Early (< 24 hours), contained perforation: Primary repair (two-layer closure) + buttressed with muscle flap (intercostal, diaphragm, pleural) + wide drainage + IV antibiotics. Late (> 24 hours), extensive contamination: Debridement + wide drainage + esophageal diversion (cervical esophagostomy + gastrostomy) with delayed reconstruction, or primary repair if tissue quality permits + drainage. Endoscopic management: Increasingly used for small perforations — esophageal stenting (covered self-expanding metal stent), endoscopic vacuum therapy (EVT/Endo-SPONGE), and endoscopic clip closure. Regardless of approach, nutrition must be established (jejunostomy tube or TPN) and broad-spectrum antibiotics covering gram-negatives and anaerobes are mandatory.
Esophageal Motility Disorders
Achalasia: Failure of LES relaxation + absent esophageal peristalsis. Surgical treatment: Heller myotomy — laparoscopic/robotic division of the LES muscle fibers (cardiomyotomy) + partial fundoplication (Dor anterior wrap most common — prevents reflux without increasing dysphagia). Per-oral endoscopic myotomy (POEM) — endoscopic submucosal tunnel myotomy; equivalent efficacy to Heller myotomy at 2 years but higher rates of GERD (no fundoplication). Zenker's diverticulum: A pharyngeal pouch (posterior mucosal herniation through Killian's triangle between the cricopharyngeus and inferior pharyngeal constrictor). Presents with dysphagia, regurgitation of undigested food, halitosis, and aspiration. Treatment: cricopharyngeal myotomy + diverticulectomy or diverticulopexy (open transcervical approach) or endoscopic stapler-assisted diverticulotomy for smaller diverticula.
Paraesophageal Hernia
Classified as: Type I (sliding) — GEJ migrates upward through the hiatus; most common (95%); usually managed medically. Type II (true paraesophageal) — GEJ stays in normal position but the gastric fundus herniates through the hiatus alongside the esophagus. Type III (combined) — both the GEJ and fundus herniate. Type IV — other organs herniate in addition to the stomach (colon, spleen, omentum). Types II-IV carry risk of gastric volvulus, incarceration, strangulation, and perforation. Surgical repair is indicated for symptomatic paraesophageal hernias and asymptomatic large hernias in fit patients. Approach: laparoscopic repair — reduction of the hernia sac, closure of the crural defect (posterior cruroplasty with sutures, with or without mesh reinforcement for large defects), and fundoplication (Nissen 360-degree wrap or Toupet 270-degree partial wrap). Recurrence rates are 15-30% even with mesh reinforcement.
Diaphragmatic Injuries & Eventration
Traumatic diaphragmatic rupture: Occurs in both blunt (high-speed MVC — usually large, left-sided tears due to the protective effect of the liver on the right) and penetrating trauma. Diagnosis is often missed acutely — CXR may show elevated hemidiaphragm, bowel/nasogastric tube in the chest, or loss of diaphragmatic contour. CT sensitivity is ~70% acutely. Repair: primary closure with non-absorbable suture (prolene or ethibond); mesh reinforcement for large defects. Acute injuries are repaired via laparotomy; chronic hernias via thoracotomy (adhesions are better approached from the chest).
Diaphragm eventration: Congenital or acquired (phrenic nerve injury after cardiac surgery, birth trauma) elevation of all or part of the hemidiaphragm without a true defect. If symptomatic (dyspnea, orthopnea, exercise intolerance), surgical treatment is diaphragm plication — folding and suturing the redundant diaphragm to flatten it and prevent paradoxical motion. Can be performed via VATS or thoracotomy with excellent results and improved pulmonary function.
15 Mediastinal Masses
The "4 T's" of the Anterior Mediastinum
The most common anterior mediastinal masses are: Thymoma, Teratoma (and other germ cell tumors), Terrible lymphoma (Hodgkin's and non-Hodgkin's), and Thyroid (substernal goiter). Approach: CT chest with contrast is the initial imaging study. If a tissue diagnosis is needed before surgery, CT-guided core biopsy or anterior mediastinotomy (Chamberlain procedure) is performed. Surgical resection via median sternotomy or VATS is indicated for thymoma and mature teratoma; lymphoma is treated with chemotherapy/radiation (tissue diagnosis first, do NOT resect).
Thymoma
The most common primary anterior mediastinal neoplasm in adults. ~30-50% of patients with thymoma have myasthenia gravis (MG), and ~10-15% of MG patients have a thymoma. Masaoka-Koga staging:
| Stage | Description | Treatment |
|---|---|---|
| I | Completely encapsulated, no microscopic capsular invasion | Surgical resection alone (thymectomy) |
| IIA | Microscopic capsular invasion | Surgical resection ± adjuvant RT |
| IIB | Macroscopic invasion into surrounding fat or grossly adherent to pleura/pericardium | Surgical resection + adjuvant RT |
| III | Invasion into neighboring organs (lung, great vessels, pericardium) | Neoadjuvant chemo → surgery + adjuvant RT (if resectable) |
| IVA | Pleural or pericardial dissemination | Chemotherapy ± radiation |
| IVB | Lymphogenous or hematogenous metastasis | Chemotherapy |
Other Mediastinal Masses
Posterior mediastinal masses: Neurogenic tumors are most common — schwannoma, neurofibroma (benign, in adults), ganglioneuroma, neuroblastoma (malignant, in children). Most are resectable via VATS. "Dumbbell" tumors extend through the neural foramen into the spinal canal — require combined neurosurgical and thoracic approach.
Middle mediastinal masses: Bronchogenic cysts (most common congenital mediastinal cyst — typically near the carina, may cause airway compression; resect even if asymptomatic due to risk of infection and growth), pericardial cysts (benign, usually right cardiophrenic angle, observation if asymptomatic), and lymphadenopathy (sarcoidosis, metastatic disease, Castleman disease).
Mediastinal germ cell tumors: Include mature teratoma (benign — surgical excision is curative; contains ectoderm, mesoderm, and endoderm derivatives such as teeth, hair, cartilage), seminoma (radiosensitive; treat with radiation + chemotherapy; excellent prognosis), and non-seminomatous germ cell tumors (NSGCT — embryonal carcinoma, yolk sac tumor, choriocarcinoma; check tumor markers: AFP and beta-hCG; treatment: cisplatin-based chemotherapy then surgical resection of residual mass). Serum AFP is NOT elevated in pure seminoma — if AFP is elevated, treat as NSGCT regardless of histology. Mediastinal NSGCT are associated with Klinefelter syndrome (47,XXY).
Substernal goiter: Extension of a thyroid goiter below the thoracic inlet into the anterior mediastinum. Most can be resected via a cervical collar incision with traction, but large goiters with intrathoracic extension below the aortic arch may require a partial sternotomy or thoracotomy for safe removal. Preoperative CT is essential to evaluate relationship to great vessels, tracheal compression/deviation, and extension pattern. Tracheomalacia (softening of tracheal cartilage from chronic compression) may require temporary postoperative intubation or rarely tracheal stenting.
16 Pleural Disease & Chest Wall Tumors
Pleural Effusion
Categorized as transudative (CHF, cirrhosis, nephrotic syndrome — Light's criteria: protein ratio < 0.5, LDH ratio < 0.6, LDH < 2/3 upper limit of normal) or exudative (infection, malignancy, PE, autoimmune — meets one or more Light's criteria). Malignant pleural effusion: most commonly from lung, breast, or lymphoma metastasis. Management: thoracentesis (diagnostic and therapeutic), pleurodesis (talc — via VATS or slurry through chest tube) for recurrent symptomatic effusions, or indwelling pleural catheter (PleurX) for symptomatic relief in patients with trapped lung or poor performance status.
Massive Hemoptysis
Defined as > 500-600 mL of blood expectorated in 24 hours (or any amount causing hemodynamic instability or respiratory compromise). Most common causes: bronchiectasis, lung cancer, tuberculosis, aspergilloma (mycetoma), and bronchial artery erosion. Immediate management: (1) protect the airway — intubate with a large (8.0+) ETT; if the bleeding source is known, position patient with bleeding lung dependent (to protect the contralateral lung from aspiration); selective intubation of the non-bleeding main bronchus if necessary. (2) Bronchoscopy — rigid bronchoscopy is preferred (better suctioning, airway control, and intervention capability); flexible can be used initially for localization. (3) Bronchial artery embolization (interventional radiology) — first-line definitive treatment; success rate ~85% for immediate cessation but recurrence in ~20-30%. (4) Surgical resection — for recurrent or massive hemoptysis refractory to embolization (lobectomy or pneumonectomy). For aspergilloma causing massive hemoptysis, surgical resection (lobectomy or wedge) is the definitive treatment when the patient can tolerate it.
Empyema
Infected pleural space, usually from parapneumonic effusion (most common cause), post-surgical, esophageal perforation, or trauma. Three stages: Stage I (exudative) — free-flowing fluid, thin, low WBC; treat with antibiotics + thoracentesis or tube thoracostomy. Stage II (fibrinopurulent) — fibrin strands and loculations develop; may respond to chest tube + intrapleural fibrinolytics (tPA/DNase), but VATS decortication is often needed. Stage III (organized) — thick fibrous peel traps the lung (trapped lung); requires surgical decortication (VATS or thoracotomy) to free the lung and allow re-expansion.
Mesothelioma
Malignant pleural mesothelioma is strongly associated with asbestos exposure (long latency of 20-40 years). Presents with pleural effusion, dyspnea, and chest pain. Diagnosis requires pleural biopsy (thoracoscopy preferred). Epithelioid subtype has the best prognosis; sarcomatoid has the worst. Treatment: multimodal — surgery (if resectable: extended pleurectomy/decortication [P/D] is preferred over extrapleural pneumonectomy [EPP] based on the MARS and MARS-2 trials showing no benefit of EPP and high morbidity) + chemotherapy (cisplatin/pemetrexed) + radiation. The addition of immunotherapy (nivolumab + ipilimumab, CheckMate 743 trial) has shown survival benefit in unresectable disease.
Chest Wall Tumors
Primary chest wall tumors can be benign (osteochondroma, fibrous dysplasia, desmoid tumor) or malignant (chondrosarcoma — most common primary malignant chest wall tumor, Ewing's sarcoma, osteosarcoma, soft tissue sarcomas). Treatment for malignant tumors: wide excision with 2-4 cm margins (including involved ribs above and below). Chest wall reconstruction is required for defects > 5 cm or > 2 ribs: use prosthetic mesh (Prolene, Gore-Tex, or methylmethacrylate "sandwich" for rigid reconstruction) + muscle flap coverage (latissimus dorsi, pectoralis major, serratus anterior, rectus abdominis, or omental flap).
Pneumothorax
Primary spontaneous pneumothorax (PSP): Typically occurs in tall, thin, young males aged 15-35 due to rupture of apical subpleural blebs. Treatment: small (< 2 cm on CXR), asymptomatic — observation with supplemental O2; moderate-large or symptomatic — aspiration or chest tube (small bore 14-22 Fr with Heimlich valve or underwater seal). Indications for surgery: recurrent ipsilateral PSP (second episode), persistent air leak > 5-7 days, bilateral PSP, hemopneumothorax, or high-risk occupation (pilot, diver). Surgical approach: VATS with bleb resection (wedge/stapler) + pleurodesis (mechanical abrasion or apical pleurectomy). Recurrence after VATS: ~5% (vs ~30% after chest tube alone).
Secondary spontaneous pneumothorax: Occurs in patients with underlying lung disease (COPD, cystic fibrosis, Pneumocystis pneumonia). More dangerous due to limited respiratory reserve. Lower threshold for chest tube insertion and surgical intervention. Tension pneumothorax: Life-threatening emergency — positive pressure builds up in the pleural space due to a one-way valve mechanism. Signs: hypotension, tracheal deviation away from the affected side, absent breath sounds, distended neck veins. Treatment: immediate needle decompression (14-gauge needle, 2nd intercostal space, midclavicular line) followed by tube thoracostomy.
Tracheal Surgery
Tracheal stenosis: Most commonly post-intubation (cuff site or stoma site after tracheostomy). Can also result from inflammatory conditions (granulomatosis with polyangiitis, relapsing polychondritis) or malignancy. Management depends on etiology and length of stenosis: endoscopic dilation/stenting for short-term relief or poor surgical candidates; tracheal resection and primary anastomosis for benign stenosis up to ~4-5 cm in length (approximately 50% of the trachea). The anastomosis is tension-free using laryngeal release maneuvers (suprahyoid or thyrohyoid release) if needed. Grillo developed many of the foundational techniques. Tracheal tumors: Rare — squamous cell carcinoma and adenoid cystic carcinoma are the two most common primary tracheal malignancies. Treatment: surgical resection with primary anastomosis when feasible; radiation for unresectable disease.
17 Heart Transplantation
Listing Criteria
Heart transplantation is indicated for end-stage heart failure (NYHA class III-IV) refractory to maximal medical therapy, with an expected 1-year survival < 50% without transplant. Specific indications include: VO2max < 12-14 mL/kg/min on cardiopulmonary exercise testing (or < 50% predicted), recurrent life-threatening arrhythmias refractory to all therapies, refractory cardiogenic shock requiring mechanical circulatory support, and severe ischemic heart disease with inoperable anatomy and refractory angina.
- Fixed pulmonary hypertension — PVR > 5 Wood units that does not decrease with vasodilator testing (a transplanted right ventricle cannot handle high PVR and will fail acutely)
- Active systemic infection or sepsis
- Active malignancy (exceptions: skin cancer, early-stage cancers with > 5-year disease-free survival)
- Severe irreversible renal or hepatic dysfunction (unless combined organ transplant planned)
- Severe peripheral or cerebrovascular disease not amenable to revascularization
- Active substance abuse, lack of social support, psychiatric illness that impairs compliance
- Age is relative — most programs accept patients up to ~70 years, with individualized assessment
Surgical Technique
Two approaches: Bicaval technique (now standard) — recipient heart is excised leaving a cuff of left atrium (with pulmonary veins), and the donor heart is anastomosed at the left atrial cuff, IVC, SVC, pulmonary artery, and aorta. Preserves atrial geometry and sinus node function. Biatrial technique (Lower-Shumway) — older approach with left and right atrial cuff anastomoses; higher rate of atrial arrhythmias and tricuspid regurgitation.
Immunosuppression
Standard triple therapy: tacrolimus (calcineurin inhibitor — cornerstone, target trough 10-15 ng/mL early, 5-10 ng/mL long-term), mycophenolate mofetil (MMF) (antimetabolite, 1-1.5 g BID), and prednisone (tapered over months — many centers attempt complete steroid withdrawal by 6-12 months). Induction therapy with basiliximab (IL-2 receptor antagonist) or anti-thymocyte globulin (ATG) is used in the perioperative period.
Rejection surveillance: endomyocardial biopsy (EMB) — performed per protocol (weekly initially, then monthly, then quarterly). ISHLT grading: Grade 0R (no rejection), 1R (mild — focal or diffuse infiltrate without myocyte damage), 2R (moderate — multifocal infiltrate with myocyte damage), 3R (severe — diffuse infiltrate with myocyte necrosis and edema). Treatment of rejection: pulse methylprednisolone (1 g IV daily × 3 days) for cellular rejection; plasmapheresis, IVIG, and rituximab for antibody-mediated rejection (AMR).
Donor Selection & Organ Procurement
Donor criteria: brain-dead donor, age typically < 55 (though extended criteria donors are increasingly used), no significant cardiac disease (echocardiographic EF > 40-45%), negative serologies for HIV and active hepatitis, limited ischemic time (ideally < 4 hours — 6 hours is the upper limit; longer ischemic times increase primary graft dysfunction risk). Size matching: donor-to-recipient weight ratio ideally 0.8-1.2; undersized donors for recipients with elevated PVR increases risk of RV failure. Donor management after brain death includes maintaining hemodynamic stability (may require vasopressors, hormonal resuscitation with T3/T4, corticosteroids, vasopressin, and insulin). Procurement technique: median sternotomy, inspection of the heart, aortic cross-clamp with cold cardioplegia, excision with adequate cuffs of great vessels.
Post-Transplant Complications
Primary graft dysfunction (PGD): Early graft failure within 24 hours of transplantation — the leading cause of 30-day mortality. Manifests as severe biventricular failure requiring mechanical support (ECMO, Impella, IABP). Risk factors: prolonged ischemic time, donor-recipient size mismatch, elevated recipient PVR. Infection: Leading cause of death in the first year — opportunistic infections (CMV, Pneumocystis, Aspergillus, Toxoplasma) due to immunosuppression. Prophylaxis: valganciclovir (CMV), trimethoprim-sulfamethoxazole (PCP), and nystatin/fluconazole (Candida). Malignancy: Long-term immunosuppression increases risk — skin cancer (most common) and post-transplant lymphoproliferative disorder (PTLD, usually EBV-driven B-cell lymphoma). Treatment of PTLD: reduce immunosuppression, rituximab, chemotherapy.
18 Mechanical Circulatory Support
Left Ventricular Assist Device (LVAD)
The HeartMate 3 (HM3) is the current-generation LVAD — a centrifugal-flow, fully magnetically levitated pump. The impeller is suspended without mechanical bearings, reducing shear stress on blood elements and significantly lowering pump thrombosis rates compared to the axial-flow HeartMate II. Implanted with an inflow cannula in the LV apex and outflow graft to the ascending aorta; the driveline exits the skin (percutaneous) and connects to an external controller and batteries.
Bridge to transplant (BTT): Support while awaiting donor organ. Bridge to candidacy (BTC): Stabilize the patient to reassess transplant eligibility (e.g., reversible pulmonary hypertension, deconditioning). Destination therapy (DT): Permanent support in transplant-ineligible patients. The MOMENTUM 3 trial (PMID: 30575491) demonstrated superiority of HM3 over HMII in survival free from disabling stroke and reoperation for pump malfunction at 2 years.
INTERMACS profiles: 1 = critical cardiogenic shock ("crash and burn"); 2 = progressive decline on inotropes; 3 = stable but inotrope-dependent; 4 = resting symptoms on oral therapy; 5 = exertion intolerant; 6 = exertion limited; 7 = NYHA III. Profiles 1-3 are the most common indications for LVAD implantation.
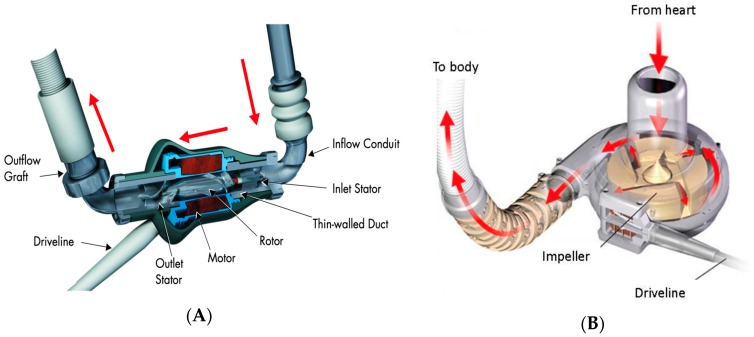
LVAD Complications
Bleeding — GI bleeding is the most common complication (~20-30%), caused by acquired von Willebrand syndrome (shear-mediated destruction of large vWF multimers) and arteriovenous malformations (AVMs — the continuous-flow physiology promotes AVM formation). Management: endoscopy, octreotide for recurrent GI AVMs, and danazol or thalidomide in refractory cases. Anticoagulation management is challenging (withholding increases pump thrombosis risk). Driveline infection — chronic risk due to the percutaneous driveline; meticulous exit-site care is essential; superficial infections treated with antibiotics and local wound care; deep infections may require driveline relocation or device exchange. Pump thrombosis — much reduced with HM3 but still occurs; presents with elevated LDH (> 2.5× upper limit of normal), elevated plasma-free hemoglobin, dark urine (hemoglobinuria), heart failure symptoms, and abnormal pump parameters (increased power consumption). Treatment: increase anticoagulation (heparin drip), tPA in select cases, or urgent device exchange for refractory thrombosis. Right ventricular failure — ~20-30% of LVAD patients develop RV failure due to increased venous return to an unsupported RV and interventricular septal shift; may require temporary RVAD (CentriMag) support. Stroke — both hemorrhagic and ischemic; hemorrhagic stroke is more common with LVADs due to anticoagulation and acquired coagulopathy; ischemic stroke from pump thrombosis or embolism.
LVAD Patient Management Pearls
LVAD patients have unique clinical considerations: (1) Blood pressure measurement: Continuous-flow LVADs generate reduced pulse pressure — BP is often measured as a single mean pressure using a Doppler over the brachial artery (target MAP 70-80 mmHg). Automated cuffs may not detect a BP. (2) Auscultation: A continuous low-pitched hum is heard over the precordium (pump sound). Changes in pump hum (higher pitch, intermittent) may indicate alarm conditions. (3) Emergency situations: If the LVAD stops functioning, the patient has near-zero cardiac output (the conduit creates a "sump" effect). Chest compressions are controversial and generally avoided (risk of dislodging the inflow cannula); focus on troubleshooting the device (check connections, batteries, controller). (4) Anticoagulation: Warfarin (target INR 2.0-3.0) + aspirin 81-325 mg daily. INR monitoring is essential. DOACs are NOT used with LVADs.
ECMO (Extracorporeal Membrane Oxygenation)
Veno-arterial (VA) ECMO: Provides both cardiac and respiratory support. Blood is drained from the venous system (typically right atrium via femoral vein or directly via right atrial cannula), passed through an oxygenator, and returned to the arterial system (typically femoral artery or ascending aorta). Used for cardiogenic shock, post-cardiotomy syndrome, bridge to decision in cardiac arrest. Veno-venous (VV) ECMO: Provides respiratory support only (no cardiac support). Blood is drained from and returned to the venous system (dual-lumen cannula in the right IJ, or femoral-IJ configuration). Used for severe ARDS (the EOLIA trial suggested a trend toward reduced 60-day mortality with VV ECMO for severe ARDS).
Impella
A percutaneous microaxial flow pump placed across the aortic valve (via femoral artery) that aspirates blood from the LV and expels it into the ascending aorta. Impella CP: ~3.7 L/min flow support; Impella 5.0/5.5: ~5-5.5 L/min (requires axillary artery cutdown). Used for high-risk PCI support, acute MI with cardiogenic shock (though the DANGER trial raised questions about routine use in AMI cardiogenic shock), and bridge to LVAD/transplant.
19 Congenital Heart Disease, Pericardial Disease & Cardiac Tumors
Adult Congenital Heart Disease (ACHD)
Atrial septal defect (ASD): Secundum (most common, ~70%) — amenable to device closure (Amplatzer) or surgical patch closure. Primum — associated with AV canal defects, requires surgical repair. Sinus venosus — associated with partial anomalous pulmonary venous return (PAPVR), requires surgical repair (Warden procedure). Indications for closure: right heart volume overload (RV dilation), Qp:Qs ≥ 1.5:1, paradoxical embolism, and symptomatic patients.
Ventricular septal defect (VSD): In adults, most hemodynamically significant VSDs have been repaired in childhood. Residual or missed VSDs causing LV volume overload (Qp:Qs ≥ 2:1), heart failure, or aortic regurgitation (due to prolapse of the right coronary cusp into the VSD — common with supracristal VSDs) warrant surgical closure with patch.
Tetralogy of Fallot (TOF) — adult management: Most patients have been repaired in infancy. The four features of TOF are: (1) ventricular septal defect, (2) overriding aorta, (3) RV outflow tract obstruction, (4) right ventricular hypertrophy. Late issues in repaired TOF patients include: pulmonary regurgitation (from transannular patch repair) causing RV dilation — indications for pulmonary valve replacement include progressive RV dilation (RV end-diastolic volume index > 150 mL/m² on MRI), RV dysfunction, exercise intolerance, and arrhythmias (especially sustained VT). Residual VSD, RV outflow tract obstruction, and aortic root dilation may also require intervention. Transcatheter pulmonary valve implantation (Melody valve, SAPIEN) is increasingly used in appropriate anatomy to avoid repeat sternotomy.
Coarctation of the aorta: Discrete narrowing of the aorta typically at the aortic isthmus (just distal to the left subclavian artery, near the ligamentum arteriosum). Associated with bicuspid aortic valve (50-80%), intracranial aneurysms (Circle of Willis — berry aneurysms), and Turner syndrome. In adults, presents with hypertension (upper extremities), weak femoral pulses, rib notching on CXR (from dilated intercostal collateral arteries), and a systolic murmur. Treatment: stent placement (endovascular — increasingly first-line in adults) or surgical resection with end-to-end anastomosis or interposition graft. Residual or recurrent coarctation after childhood repair is common — requires lifelong surveillance.
Lung Transplantation
Lung transplantation is indicated for end-stage lung disease refractory to medical therapy, with an expected 2-year survival < 50% without transplant. Common indications: COPD/emphysema (most common), idiopathic pulmonary fibrosis (IPF — worst prognosis without transplant), cystic fibrosis, pulmonary arterial hypertension, and alpha-1 antitrypsin deficiency. The Lung Allocation Score (LAS) prioritizes donor lungs based on medical urgency and expected post-transplant survival (range 0-100; higher score = higher priority). Contraindications: active malignancy, significant other organ dysfunction, non-adherence, active substance abuse, and BMI > 35.
Types: Bilateral lung transplant (BLT) — standard for CF, pulmonary hypertension, and most patients < 65; performed via bilateral anterolateral thoracotomies (clamshell incision) or bilateral thoracosternotomy; sequential anastomoses (bronchial, pulmonary artery, pulmonary veins/left atrial cuff). Single lung transplant (SLT) — acceptable for COPD and IPF in select patients; performed via thoracotomy. Median survival: ~6 years overall. Immunosuppression is similar to heart transplant (tacrolimus + MMF + prednisone). Major complications: primary graft dysfunction (ARDS-like picture within 72 hours), acute cellular rejection (transbronchial biopsy for diagnosis), chronic lung allograft dysfunction (CLAD) — formerly called bronchiolitis obliterans syndrome (BOS) — the leading cause of late morbidity and mortality; presents as progressive, irreversible decline in FEV1. Infection is the leading cause of death in the first year.
Pericardial Disease
Constrictive pericarditis: Chronic inflammation leads to fibrous thickening and/or calcification of the pericardium, restricting diastolic filling. Classic hemodynamic findings: equalization of diastolic pressures across all four chambers, "square root" sign (rapid early diastolic filling followed by abrupt plateau), and discordant ventricular pressure changes with respiration. Key distinguishing feature from restrictive cardiomyopathy: ventricular interdependence (one ventricle filling impairs the other — respiratory variation in filling pressures). Imaging: CT shows pericardial thickening > 4 mm and/or calcification; cardiac MRI can assess inflammation (late gadolinium enhancement suggests reversible inflammatory constriction that may respond to anti-inflammatory therapy). Treatment of established constriction: pericardiectomy — radical removal of the pericardium from phrenic nerve to phrenic nerve. This is a challenging operation with significant morbidity (mortality 6-12%); recovery of diastolic function may take weeks to months.
Cardiac Tumors
Hypertrophic Obstructive Cardiomyopathy (HOCM)
Asymmetric septal hypertrophy causing dynamic LVOT obstruction exacerbated by conditions that reduce preload (dehydration, Valsalva) or afterload (vasodilation), or increase contractility (inotropes, exercise). Surgical treatment: septal myectomy (Morrow procedure) — performed via aortotomy, a trough of septal muscle is resected from the subaortic area, widening the LVOT. This is the gold standard with > 95% success rate and < 1% mortality at experienced centers. The LVOT gradient should be reduced to < 30 mmHg. Alternative: alcohol septal ablation (interventional cardiology — alcohol injection into a septal perforator branch to create a controlled infarct in the septum; higher rate of heart block and late recurrence compared to myectomy; preferred for patients who are not surgical candidates). Myectomy is preferred in patients who also need concomitant cardiac surgery (mitral repair, CABG).
Cardiac papillary fibroelastoma: The second most common primary cardiac tumor. Small (< 1.5 cm), pedunculated papillary tumor typically found on the aortic or mitral valve leaflets. Usually discovered incidentally on echocardiography. Risk of systemic embolization (stroke, coronary embolism). Treatment: surgical excision (valve-sparing when possible) is recommended for left-sided tumors due to embolic risk; right-sided tumors can be observed if asymptomatic.
Atrial myxoma: The most common primary cardiac tumor (~50% of all primary cardiac tumors). Typically a pedunculated mass arising from the interatrial septum near the fossa ovalis, most commonly in the left atrium (~75%). Classic triad: obstructive symptoms (mimicking mitral stenosis — dyspnea, orthopnea), embolic phenomena (stroke, peripheral embolization), and constitutional symptoms (fever, weight loss, elevated ESR — "cardiac fever"). Diagnosis: echocardiography. Treatment: surgical excision with the base of the interatrial septum (to prevent recurrence) and patch closure of the septum. Recurrence rate: ~3% for sporadic myxoma, ~12-22% for Carney complex (familial myxoma syndrome — autosomal dominant, multiple myxomas, skin pigmentation, endocrine tumors).
20 Cardiopulmonary Bypass & Myocardial Protection
CPB Circuit
The cardiopulmonary bypass (CPB) circuit consists of: venous cannula(e) (right atrial — two-stage cannula for most cases; bicaval cannulation for open-heart procedures requiring right atriotomy), venous reservoir, membrane oxygenator (provides gas exchange), heat exchanger (for cooling/rewarming), arterial pump (roller or centrifugal), arterial filter, and arterial cannula (ascending aorta). Blood is drained by gravity into the venous reservoir, pumped through the oxygenator and heat exchanger, filtered, and returned to the aorta.
Cannulation & Initiation
Standard: aortic cannulation (distal ascending aorta) and right atrial venous cannulation. Before cannulation, heparin is given (300-400 units/kg IV) and confirmed by activated clotting time (ACT > 480 seconds) before initiating CPB. After cannulation and achieving target ACT, CPB is initiated ("on pump"), the aorta is cross-clamped (isolating the heart from the systemic circulation), and cardioplegia is delivered to arrest the heart.
Myocardial Protection — Cardioplegia
Cardioplegia is a hyperkalemic solution that depolarizes the myocardium and arrests the heart in diastole, dramatically reducing myocardial oxygen consumption. Delivered antegrade (via aortic root or directly into coronary ostia) and/or retrograde (via coronary sinus catheter). Two major formulations:
del Nido cardioplegia: Originally developed for pediatric surgery, now widely used in adults. Single-dose solution (can achieve 60-90 minutes of arrest with one dose) containing: Plasma-Lyte A base, mannitol, MgSO4, NaHCO3, KCl (achieving K+ ~24 mEq/L in the final solution), and lidocaine. Mixed 4:1 (crystalloid:blood). Advantages: single dose simplifies the operation, long safe arrest time, reduced potassium load to the patient.
Buckberg (blood) cardioplegia: Uses the patient's own blood as a base mixed with crystalloid cardioplegia concentrate. Delivered as a warm induction dose, then cold maintenance doses every 15-20 minutes, and a warm "hot shot" reperfusion dose before cross-clamp removal. Rationale: blood provides oxygen-carrying capacity and buffering. More complex to administer (multiple redosing) but well-studied in long cross-clamp time operations.
Weaning from CPB
Steps for separation from bypass: (1) Rewarm to 36-37°C. (2) De-air the heart (transesophageal echo guided). (3) Remove the aortic cross-clamp — the heart reperfuses and typically resumes electrical activity (may need defibrillation if VF). (4) Ensure adequate heart rate and rhythm (temporary epicardial pacing wires for bradycardia or heart block). (5) Gradually reduce CPB flow as the heart assumes the work. (6) Assess hemodynamics (TEE for ventricular function, valve function, air). (7) Once stable, fully wean off CPB and administer protamine to reverse heparin (1-1.3 mg protamine per 100 units of heparin given; given slowly — rapid administration causes hypotension and pulmonary vasoconstriction; rare anaphylaxis risk, especially in patients with fish allergy or prior protamine exposure).
Intraoperative TEE
Transesophageal echocardiography is now standard during cardiac surgery. Key roles: (1) Pre-bypass assessment — confirm preoperative diagnosis, evaluate valve pathology, assess LV/RV function, identify aortic atherosclerosis (epiaortic ultrasound is complementary), and guide cannulation strategy. (2) Post-bypass assessment — confirm adequacy of valve repair (no significant residual MR after mitral repair; acceptable gradients after AVR), rule out new wall motion abnormalities suggesting graft occlusion, evaluate de-airing, and assess ventricular function. (3) Hemodynamic monitoring — real-time assessment of volume status, contractility, and response to interventions. A new finding on intraoperative TEE (e.g., previously undiagnosed severe MR) may change the surgical plan. The cardiac anesthesiologist and surgeon jointly interpret TEE findings.
Hypothermia During CPB
Temperature management during CPB is tailored to the procedure: Normothermic (36-37°C): Standard for uncomplicated CABG — reduces CPB time (no cooling/rewarming); may reduce coagulopathy. Mild hypothermia (32-35°C): Common for valve surgery — provides some organ protection with minimal metabolic derangement. Moderate hypothermia (24-28°C): Used for aortic arch surgery with SACP — allows extended circulatory arrest times with cerebral perfusion. Deep hypothermia (18-20°C): Deep hypothermic circulatory arrest (DHCA) — provides maximal organ protection during total circulatory arrest; safe for ~30-40 minutes. Cooling is achieved via the heat exchanger on the CPB circuit. Rewarming must be gradual (no more than 10°C gradient between blood and water temperature; core temperature should not exceed 37°C to avoid cerebral hyperthermia). Complications of hypothermia: coagulopathy (platelet dysfunction, impaired clotting cascade), cardiac arrhythmias, and prolonged CPB time.
Blood Conservation
Cell salvage (Cell Saver): Collects shed blood from the operative field, washes and concentrates red blood cells, and returns them to the patient. Standard in cardiac surgery. Antifibrinolytics: Tranexamic acid (TXA) — most commonly used; typical dosing: 1 g loading + 1 g in CPB circuit + 1 g infusion over the case (or 30 mg/kg load + 16 mg/kg/hr infusion + 2 mg/kg in pump prime). High-dose TXA may increase seizure risk. Aminocaproic acid (Amicar) — alternative; typical dosing: 5 g load + 5 g in circuit + 1 g/hr infusion. Aprotinin was removed from the market (BART trial showed increased mortality) but has been reintroduced in limited settings in some countries.
21 Postoperative ICU Management
Hemodynamic Management
After cardiac surgery, patients are monitored with arterial line, PA catheter (in complex cases), central venous catheter, and TEE as needed. Target parameters: MAP 65-80 mmHg, CI > 2.2 L/min/m², CVP 8-12 mmHg, SvO2 > 65%, urine output > 0.5 mL/kg/hr. Common hemodynamic issues:
| Problem | Hemodynamic Pattern | Treatment |
|---|---|---|
| Low CO / cardiogenic shock | Low CI, elevated SVR, elevated filling pressures | Inotropes (milrinone, dobutamine, epinephrine); IABP or Impella if refractory |
| Vasoplegia (distributive) | Low SVR, normal or high CI, low filling pressures | Vasopressors (norepinephrine first-line, vasopressin as adjunct); methylene blue (1.5-2 mg/kg) for refractory vasoplegia |
| RV failure | Elevated CVP, low PA pressures, low CI | Avoid volume overload; milrinone (also reduces PVR); inhaled nitric oxide or epoprostenol for pulmonary vasodilation; consider RVAD |
| Tamponade | Equalized filling pressures, low CI, pulsus paradoxus | Emergent reoperation (re-sternotomy); maintain preload and heart rate until OR |
| Hypovolemia | Low filling pressures, low CI, elevated SVR | Volume resuscitation (crystalloid, colloid, blood products as indicated) |
Chest Tube Management
Mediastinal and/or pleural chest tubes are placed at the end of cardiac surgery. Monitor output hourly. Excessive bleeding: > 200 mL/hr for 2 consecutive hours, > 400 mL in the first hour, or > 1500 mL total in the first 12 hours warrants re-exploration. Before re-exploration, ensure correction of coagulopathy: check ACT, PT/INR, PTT, fibrinogen (> 200 mg/dL target), platelets (> 100K), and administer protamine, FFP, platelets, cryoprecipitate, or factor concentrates as needed.
Postoperative Atrial Fibrillation (POAF)
The most common complication after cardiac surgery, occurring in 20-40% of CABG patients and up to 50-60% of valve surgery patients. Peak incidence on postoperative days 2-3. Associated with increased ICU/hospital stay, stroke risk, and long-term AF. Prevention: amiodarone prophylaxis (most effective — started preoperatively or on POD 0: 400 mg BID for 7 days, or IV load followed by oral) or beta-blocker continuation postoperatively. Treatment of POAF: rate control with beta-blockers (metoprolol) or amiodarone; rhythm control with amiodarone or DC cardioversion if hemodynamically unstable; anticoagulation (heparin then warfarin) if AF persists > 48 hours.
Sternal Wound Care
The sternotomy is closed with stainless steel wires. Deep sternal wound infection (DSWI) / mediastinitis: Incidence 1-3%, mortality 10-25%. Risk factors: diabetes (especially poorly controlled), obesity, bilateral IMA harvest, prolonged CPB time, reoperation. Organisms: Staphylococcus aureus (most common), coagulase-negative staphylococci, gram-negative bacteria. Treatment: surgical debridement + rewiring (if early, stable sternum) or sternal resection + muscle flap closure (pectoralis major advancement flaps most common; rectus abdominis or omental flaps for extensive defects) + prolonged IV antibiotics (4-6 weeks). Vacuum-assisted closure (VAC) can be used as a bridge to definitive closure.
Ventilator Management Post-Cardiac Surgery
Most cardiac surgery patients arrive intubated to the ICU. Standard initial ventilator settings: volume-controlled ventilation, Vt 6-8 mL/kg ideal body weight, PEEP 5 cmH2O, FiO2 titrated to SpO2 > 94%, rate 12-16/min. Fast-track extubation (within 4-6 hours of ICU arrival) is standard for uncomplicated cases — criteria: hemodynamically stable, adequate gas exchange (PaO2 > 60 on FiO2 ≤ 0.4), minimal chest tube output, alert and cooperative, temperature > 36°C, no active bleeding. Delayed extubation (> 24 hours) is associated with increased pneumonia, prolonged ICU stay, and mortality. Risk factors for delayed extubation: preoperative COPD, EF < 30%, prolonged CPB time (> 120 min), reoperation for bleeding, and stroke.
Renal Protection & Fluid Management
AKI after cardiac surgery occurs in 5-30% of patients and 2-5% require renal replacement therapy. Risk factors: preoperative CKD, diabetes, prolonged CPB (> 120 min), low CO, nephrotoxin exposure (contrast, aminoglycosides), and prolonged aortic cross-clamp time. Preventive strategies: maintain adequate perfusion pressure on CPB (MAP > 65 mmHg, or > 75 in patients with preexisting hypertension), avoid nephrotoxins, goal-directed fluid therapy (avoid both hypovolemia and fluid overload), and early initiation of CRRT if oliguric AKI develops with fluid overload. Fenoldopam and "renal-dose dopamine" have NOT been shown to prevent AKI.
Neurological Complications
Stroke after cardiac surgery is classified as: Type I — focal neurological deficit (stroke, TIA) occurring in 1-5% of cardiac surgery patients; and Type II — diffuse encephalopathy (delirium, confusion, cognitive decline without focal deficit) occurring in 10-30%. Risk factors for type I: aortic atherosclerosis (most important — epiaortic ultrasound-guided cannulation reduces stroke risk), prior stroke, carotid stenosis, advanced age, prolonged CPB, atrial fibrillation, and aortic arch manipulation. Management of perioperative stroke: emergent CT head (rule out hemorrhagic stroke before anticoagulation), neurology consultation, consider thrombectomy for large-vessel occlusion if within the time window (up to 24 hours in select patients). Postoperative delirium is managed with reorientation, sleep hygiene, early mobilization, avoiding deliriogenic medications (benzodiazepines, anticholinergics), and low-dose antipsychotics (haloperidol, quetiapine) if necessary for agitation.
Postpericardiotomy Syndrome
An inflammatory syndrome occurring 1-6 weeks after cardiac surgery (or any procedure that opens the pericardium). Presents with: fever, pleuritic chest pain, pericardial and/or pleural effusion, elevated ESR and CRP. Mechanism: autoimmune response to pericardial/myocardial antigens. Incidence: 10-40% of cardiac surgery patients. Diagnosis: clinical — must exclude other causes (wound infection, pneumonia, tamponade from surgical bleeding). Treatment: NSAIDs (ibuprofen 600 mg TID) + colchicine (0.5 mg BID — the COPPS and COPPS-2 trials showed that perioperative colchicine reduces postpericardiotomy syndrome incidence). Corticosteroids reserved for refractory cases (promote dependence and recurrence). Most cases are self-limited. Tamponade requiring pericardiocentesis or surgical drainage occurs in < 5%.
Enhanced Recovery After Surgery (ERAS) in Cardiac/Thoracic Surgery
ERAS protocols are increasingly applied to cardiothoracic surgery to improve outcomes and reduce length of stay. Key components: preoperative education and optimization, minimization of preoperative fasting (clear fluids up to 2 hours before surgery), multimodal analgesia (regional anesthesia when possible — paravertebral block, erector spinae plane block, or epidural for thoracotomy; avoid excessive opioids), early extubation (within 4-6 hours), early mobilization (out of bed POD 0 or 1), early chest tube removal, early enteral nutrition, and standardized postoperative care pathways. In thoracic surgery, ERAS protocols have been shown to reduce hospital stay by 1-3 days and decrease pulmonary complications.
22 Surgical Approaches & Key Procedures
Median Sternotomy
The median sternotomy is the most common approach in cardiac surgery. An incision from the sternal notch to the xiphoid process; the sternum is divided with an oscillating saw. Provides excellent exposure to the heart, great vessels, and both pleural spaces. Closed with stainless steel wires (typically 6-8 wires). Complications: sternal dehiscence (0.5-5%), DSWI/mediastinitis (1-3%), and phrenic nerve injury.
Thoracotomy
Posterolateral thoracotomy: Standard for open thoracic surgery (lung resection, esophagectomy, descending aortic surgery). Incision along the 5th or 6th intercostal space from the midaxillary line posteriorly; divides the latissimus dorsi and serratus anterior muscles. Provides excellent exposure but is the most painful surgical approach in the body. Anterolateral thoracotomy: Used for emergency access (e.g., ED thoracotomy — "clam shell" if bilateral), minimally invasive cardiac surgery (mini-thoracotomy for MICS AVR or mitral valve surgery). Muscle-sparing thoracotomy: Retracts rather than divides the latissimus dorsi — less postoperative pain and shoulder dysfunction.
VATS (Video-Assisted Thoracoscopic Surgery)
Performed through 1-4 small incisions (ports) with camera and instruments. Now the preferred approach for: anatomic lobectomy, segmentectomy, wedge resection, mediastinal mass excision, pleural biopsy, decortication, sympathectomy, and pericardial window. Advantages: less pain, shorter hospital stay, faster recovery, reduced immunosuppression. Multiport VATS uses 3-4 ports; uniportal VATS (single-incision) is gaining popularity.
Robotic-Assisted Surgery
The da Vinci surgical system provides 3D visualization, wristed instruments, and tremor filtration. Used in: robotic lobectomy (increasing adoption, oncologic outcomes equivalent to VATS), robotic-assisted mitral valve repair, robotic CABG (for LIMA harvest or direct LIMA-to-LAD anastomosis), and robotic thymectomy. Advantages: improved ergonomics for the surgeon, potentially better visualization in confined spaces. Disadvantages: longer operative times, higher cost, lack of tactile feedback.
Pericardiocentesis & Pericardial Window
Pericardiocentesis: Needle drainage of pericardial effusion, typically performed under echocardiographic guidance using a subxiphoid approach. The needle is directed toward the left shoulder at a 15-30 degree angle. Used for acute tamponade or diagnostic sampling. Pericardial window: Surgical creation of a permanent opening in the pericardium for recurrent effusions (most commonly malignant). Can be done via subxiphoid approach (local/general anesthesia, drains into the peritoneal cavity) or VATS (drains into the pleural space). VATS pericardial window also allows tissue biopsy and inspection for metastatic disease.
Bronchoscopy
Flexible bronchoscopy: Standard diagnostic tool — visualization of the tracheobronchial tree, bronchoalveolar lavage (BAL), brushings, transbronchial biopsy, and EBUS-TBNA for mediastinal staging. Performed under conscious sedation or general anesthesia. Rigid bronchoscopy: Requires general anesthesia; provides a larger working channel for: massive hemoptysis management (tamponade, ice-cold saline, vasoconstrictors), foreign body retrieval, airway stent placement, tumor debulking (laser, cryotherapy, electrocautery), and tracheal dilation.
Chest Tube Insertion (Tube Thoracostomy)
Indications: pneumothorax (traumatic, spontaneous, iatrogenic), hemothorax, pleural effusion requiring drainage, empyema, and postoperative drainage after thoracic/cardiac surgery. Technique (standard intercostal approach): Position the patient with the arm abducted overhead. Site: typically the 4th-5th intercostal space in the anterior-to-midaxillary line ("safe triangle" — bounded by the anterior border of the latissimus dorsi, the lateral border of the pectoralis major, a line superior to the horizontal level of the nipple, and the apex of the axilla). Incise the skin, blunt-dissect through the intercostal muscles over the top of the rib (to avoid the neurovascular bundle), enter the pleural space with a finger sweep (to confirm entry and ensure no adhesions), and advance the tube posteriorly and superiorly. Secure with suture and connect to a water seal drainage system (-20 cmH2O suction initially).
Tube size selection: large bore (28-36 Fr) for hemothorax and empyema; medium (20-28 Fr) for pneumothorax; small bore pigtail catheters (8-14 Fr) for simple effusions and pneumothorax (increasingly used, with equivalent efficacy in many scenarios). Removal criteria: air leak resolved (for pneumothorax — confirm with water seal trial for 6-24 hours), output < 200-300 mL/day (for effusions), and stable CXR. Remove the chest tube during end-expiration or during a Valsalva maneuver to prevent air entry.
Intra-Aortic Balloon Pump (IABP)
A percutaneous mechanical circulatory support device placed via the femoral artery with the balloon tip positioned in the descending aorta, just distal to the left subclavian artery. Operates on the principle of counterpulsation: the balloon inflates during diastole (augmenting coronary perfusion pressure) and deflates during systole (reducing LV afterload and myocardial oxygen demand). Provides modest hemodynamic support (~0.5-1.0 L/min increase in CO). Indications: cardiogenic shock as a bridge, refractory angina, support during high-risk PCI, and weaning from CPB. Contraindications: moderate-severe aortic regurgitation (augmenting diastolic pressure worsens regurgitation), aortic dissection, severe peripheral vascular disease (access issues). The IABP-SHOCK II trial showed no mortality benefit of IABP in cardiogenic shock complicating MI, but it remains widely used as an adjunct.
Temporary Mechanical Circulatory Support Comparison
| Device | Support Level | Mechanism | Duration | Key Complications |
|---|---|---|---|---|
| IABP | ~0.5-1.0 L/min augmentation | Counterpulsation (diastolic augmentation + systolic unloading) | Days to weeks | Limb ischemia, balloon rupture (helium embolism), thrombocytopenia |
| Impella CP | ~3.5-4.0 L/min | Microaxial pump across aortic valve (LV to ascending aorta) | Days (up to 14) | Hemolysis, limb ischemia, aortic valve injury, device migration |
| Impella 5.0/5.5 | ~5.0-5.5 L/min | Same as CP; larger caliber via axillary cutdown | Days to weeks | Same + axillary artery injury, brachial plexus injury |
| TandemHeart | ~4.0-5.0 L/min | Centrifugal pump; LA drainage via transseptal cannula, return to femoral artery | Days | Tamponade (transseptal puncture), residual ASD, limb ischemia |
| VA ECMO (peripheral) | ~3.0-6.0 L/min | External centrifugal pump + oxygenator (femoral V to femoral A) | Days to weeks | Limb ischemia, Harlequin syndrome, LV distension, bleeding, hemolysis |
| CentriMag (RVAD) | Up to 10 L/min | Extracorporeal centrifugal pump for RV support (RA to PA) | Up to 30 days | Bleeding, infection, thrombosis |
Minimally Invasive Cardiac Surgery (MICS)
Alternatives to full median sternotomy for select cardiac operations: Right mini-thoracotomy (5-7 cm, 4th intercostal space): Used for mitral valve repair/replacement and tricuspid valve surgery; femoral or axillary cannulation for CPB with endoaortic balloon occlusion (EndoClamp) for cardioplegic arrest. Upper hemisternotomy or right parasternal approach: Used for minimally invasive AVR (MICS-AVR). Totally endoscopic (robotic) CABG: LIMA harvest and LIMA-to-LAD anastomosis performed through port incisions without sternotomy. Advantages of MICS approaches: reduced blood loss, shorter ICU and hospital stay, faster return to normal activity, improved cosmetic results, and less sternal wound complications. Disadvantages: longer operative times, learning curve, limited exposure for complex pathology, and need for specialized equipment.
23 ECMO Cannulation & Management
VA ECMO Cannulation
Peripheral VA ECMO: Femoral vein (drainage) + femoral artery (return). Most common configuration for emergent cannulation. Key complication: limb ischemia distal to the femoral arterial cannula — a distal perfusion cannula (DPC) (antegrade cannula in the superficial femoral artery) must be placed to prevent limb ischemia. Monitor: Harlequin syndrome (also called North-South syndrome or differential hypoxemia) — occurs when the native heart provides poorly oxygenated blood to the upper body while ECMO provides oxygenated blood to the lower body. Detected by comparing right radial SpO2 to lower extremity SpO2; if upper body SpO2 is lower, consider switching to central VA ECMO or adding a VV ECMO cannula (VA-V configuration).
Central VA ECMO: Right atrial (drainage) + ascending aorta (return). Requires sternotomy or re-sternotomy. Used for post-cardiotomy failure to wean from CPB. Provides fully oxygenated blood to the proximal aorta (avoiding Harlequin syndrome). Complication: bleeding from the chest, especially if the sternum is left open ("open chest").
VV ECMO Cannulation
Configurations: (1) Dual-lumen cannula (Avalon/Crescent) placed in the right internal jugular vein — drains from SVC and IVC, returns oxygenated blood directed toward the tricuspid valve. Single access site but requires fluoroscopic or TEE guidance for positioning. (2) Femoral-IJ configuration — drainage from femoral vein, return via IJ vein. Higher risk of recirculation.
ECMO Management Targets
| Parameter | VA ECMO Target | VV ECMO Target |
|---|---|---|
| Flow | 50-80 mL/kg/min (enough to maintain end-organ perfusion) | 60-80 mL/kg/min |
| Anticoagulation | Heparin infusion, aPTT 40-60 sec (or anti-Xa 0.2-0.4) | Same |
| Sweep gas (FiO2 on circuit) | Titrate to PaO2 > 60 mmHg | Titrate to SpO2 > 88% |
| Ventilator settings | Lung rest (low tidal volume, low FiO2, moderate PEEP) | Lung rest (ultra-protective: Vt 3-4 mL/kg, low rate) |
| Hgb target | > 7-8 g/dL (some advocate > 10 in cardiogenic shock) | > 7-8 g/dL |
| MAP target | > 65 mmHg (may need vasopressors) | > 65 mmHg |
24 Inotropes, Vasopressors & Hemodynamic Agents
| Agent | Mechanism | Dose Range | Hemodynamic Effect | Key Notes |
|---|---|---|---|---|
| Milrinone | PDE-3 inhibitor (inodilator) | 0.125-0.75 mcg/kg/min (load 50 mcg/kg over 10 min — often omitted due to hypotension) | ↑ inotropy, ↓ SVR, ↓ PVR | Ideal for low CO + elevated PVR (RV failure support); renally cleared — dose reduce in CKD; causes vasodilation (may need concurrent vasopressor) |
| Dobutamine | Beta-1 > Beta-2 agonist | 2-20 mcg/kg/min | ↑ inotropy, ↑ HR, mild ↓ SVR | First-line inotrope for acute decompensated HF; increases myocardial oxygen demand; tachyarrhythmia risk |
| Epinephrine | Alpha-1, Beta-1, Beta-2 agonist | 0.01-0.5 mcg/kg/min | ↑ inotropy, ↑ HR, ↑ SVR (at higher doses) | Potent inotrope and vasopressor; first-line for anaphylaxis and cardiac arrest; may cause lactic acidosis and hyperglycemia |
| Norepinephrine | Alpha-1 >> Beta-1 agonist | 0.01-3 mcg/kg/min | ↑ SVR, mild ↑ inotropy, reflex ↓ HR | First-line vasopressor for hypotension; preferred in vasoplegia; increases afterload (avoid in cardiogenic shock as sole agent) |
| Vasopressin | V1 receptor agonist | 0.01-0.04 units/min (fixed dose, not titrated in most protocols) | ↑ SVR (does not increase PVR as much as norepinephrine) | Second-line vasopressor; useful in vasoplegia and catecholamine-resistant hypotension; does not increase heart rate |
| Phenylephrine | Pure alpha-1 agonist | 0.1-5 mcg/kg/min | ↑ SVR, reflex ↓ HR | Pure vasoconstrictor; useful for hypotension with adequate CO; avoid in cardiogenic shock (increases afterload without inotropy) |
| Dopamine | Dose-dependent: D1 (low), Beta-1 (mid), Alpha-1 (high) | 2-20 mcg/kg/min | Variable: ↑ renal perfusion (low dose), ↑ inotropy (mid), ↑ SVR (high) | Less commonly used now; "renal-dose dopamine" is a myth; higher arrhythmia risk than norepinephrine per SOAP II trial |
25 Anticoagulation, Antiarrhythmics & Immunosuppression
Anticoagulation for Prosthetic Valves
| Valve Type / Position | Anticoagulation | Target INR |
|---|---|---|
| Mechanical aortic valve (low risk — On-X, St. Jude bileaflet) | Warfarin + aspirin 81 mg | 2.0-3.0 (On-X: 1.5-2.0 after 3 months per PROACT trial) |
| Mechanical mitral valve | Warfarin + aspirin 81 mg | 2.5-3.5 |
| Bioprosthetic aortic valve | Aspirin 81 mg (warfarin for first 3-6 months is center-dependent) | If warfarin used: 2.0-3.0 |
| Bioprosthetic mitral valve | Warfarin for 3-6 months then aspirin | 2.0-3.0 |
| TAVR | Aspirin + clopidogrel for 3-6 months, then aspirin alone (or aspirin alone per POPular TAVI trial) | N/A |
Antiarrhythmics for Postoperative AF
Amiodarone: The most effective agent for prevention and treatment of postoperative AF. Prophylactic regimen: 200-400 mg PO BID starting 7 days preoperatively or 1 g IV over 24 hours then 200 mg PO BID. Treatment of new-onset POAF: 150 mg IV bolus over 10 minutes, then 1 mg/min for 6 hours, then 0.5 mg/min for 18 hours, then transition to oral 200-400 mg daily. Side effects: bradycardia, hypotension (IV formulation), thyroid dysfunction (both hypo- and hyperthyroidism), pulmonary toxicity (rare with short-course use), hepatotoxicity, corneal microdeposits. Beta-blockers (metoprolol 5 mg IV q5min up to 15 mg; then 25-100 mg PO BID): first-line for rate control, should be continued postoperatively if patient was on preoperatively.
Immunosuppression for Heart Transplant
| Agent | Class | Dose / Monitoring | Key Toxicities |
|---|---|---|---|
| Tacrolimus | Calcineurin inhibitor | 0.05-0.1 mg/kg/day divided BID; trough 10-15 (early), 5-10 (long-term) | Nephrotoxicity, hyperglycemia, tremor, hypertension, neurotoxicity |
| Mycophenolate mofetil (MMF) | Inosine monophosphate dehydrogenase inhibitor | 1-1.5 g PO BID | GI toxicity (diarrhea, nausea), bone marrow suppression (leukopenia), teratogenic |
| Prednisone | Corticosteroid | High-dose initially (1 mg/kg), taper to 5 mg/day or off by 6-12 months | Diabetes, osteoporosis, infection, Cushing's, weight gain, cataracts, AVN |
| Basiliximab | Anti-IL-2R monoclonal Ab (induction) | 20 mg IV on POD 0 and POD 4 | Generally well-tolerated; GI symptoms |
| Anti-thymocyte globulin (ATG) | Polyclonal T-cell depleting Ab (induction) | 1.5 mg/kg/day IV for 3-5 days | Profound lymphopenia, thrombocytopenia, serum sickness, increased infection/malignancy risk |
| Everolimus / Sirolimus | mTOR inhibitor (alternative to MMF) | Trough 3-8 ng/mL (everolimus) | Impaired wound healing, hyperlipidemia, mouth ulcers, proteinuria; may slow CAV progression |
Chemotherapy for Thoracic Malignancies
Key regimens: NSCLC adjuvant: cisplatin + vinorelbine (or cisplatin + pemetrexed for non-squamous) × 4 cycles. NSCLC neoadjuvant immunotherapy: nivolumab + platinum-based chemo (CheckMate 816 — neoadjuvant nivolumab + chemo significantly improved pCR rate and event-free survival in resectable NSCLC). Esophageal cancer: CROSS regimen (carboplatin AUC 2 weekly + paclitaxel 50 mg/m² weekly + 41.4 Gy concurrent radiation). Mesothelioma: cisplatin + pemetrexed (with or without bevacizumab). Thymoma: cisplatin-based combinations (CAP: cyclophosphamide, doxorubicin, cisplatin) for advanced/unresectable disease.
Pain Management in Thoracic Surgery
Post-thoracotomy pain is among the most severe surgical pain and is a major risk factor for pulmonary complications (atelectasis, pneumonia) if inadequately treated. Multimodal analgesia is essential:
| Technique | Description | Advantages / Disadvantages |
|---|---|---|
| Thoracic epidural | Catheter at T5-T8; bupivacaine + fentanyl infusion | Gold standard for thoracotomy pain; excellent analgesia; risk: epidural hematoma (rare), hypotension, urinary retention |
| Paravertebral block (PVB) | Local anesthetic injected into the paravertebral space; single shot or catheter | Equivalent analgesia to epidural with fewer side effects; less hypotension; unilateral block |
| Erector spinae plane (ESP) block | Fascial plane block; catheter placed at the level of the erector spinae muscle | Easier to perform, lower complication risk; increasingly used for both thoracotomy and sternotomy pain |
| Intercostal nerve block | Local anesthetic injected near each intercostal nerve | Simple; limited duration (single shot); intraoperative application common |
| IV PCA (patient-controlled analgesia) | Opioid (hydromorphone or morphine) patient-controlled boluses | Adjunct to regional techniques; avoid as sole modality; risk: respiratory depression, ileus |
| Multimodal oral agents | Acetaminophen 1 g q6h + NSAIDs (ketorolac 15-30 mg q6h) + gabapentin 300 mg TID | Reduce opioid requirements by 30-50%; transition from IV to oral as soon as tolerating PO |
26 Classification Systems
Cardiac Surgery Risk Scores
| Score | Purpose | Key Variables |
|---|---|---|
| EuroSCORE II | Predicted operative mortality for cardiac surgery | Age, sex, CrCl, LVEF, NYHA, CCS, PA pressure, urgency, thoracic aorta surgery, prior cardiac surgery |
| STS Risk Score | Predicted mortality + morbidity for CABG, valve, combined | > 40 variables; provides PROM, PROMM, specific complication risks |
| SYNTAX Score | Coronary anatomy complexity (guides CABG vs PCI) | Number, location, complexity of coronary lesions |
| NYHA Functional Classification | Heart failure symptom severity | I = no limitation; II = mild limitation; III = marked limitation; IV = symptoms at rest |
| CCS Angina Classification | Angina severity | I = strenuous exertion; II = moderate exertion; III = mild exertion; IV = rest angina |
Aortic Classifications
| System | Classification |
|---|---|
| Stanford | A = ascending aorta involved (surgical emergency); B = descending only (usually medical) |
| DeBakey | I = ascending + arch + descending; II = ascending only; IIIa = descending (above diaphragm); IIIb = descending (below diaphragm) |
| Crawford (TAAA) | I-V extent classification based on proximal and distal extent of thoracoabdominal aneurysm |
Lung Cancer Staging (AJCC 8th Edition)
See Section 13 for complete TNM staging. Key stage groupings: Stage I (T1-2a N0) = surgical resection; Stage II (T2b N0, T1-2 N1, T3 N0) = surgery + adjuvant chemo; Stage IIIA (N2 involvement) = multimodal; Stage IIIB-IV = generally non-surgical.
Esophageal Cancer Staging (AJCC 8th Edition)
T staging: Tis (high-grade dysplasia/in situ) | T1a (lamina propria/muscularis mucosa) | T1b (submucosa) | T2 (muscularis propria) | T3 (adventitia) | T4a (resectable adjacent structures — pleura, pericardium, diaphragm) | T4b (unresectable structures — aorta, vertebral body, trachea). N staging: N0 | N1 (1-2 regional nodes) | N2 (3-6 nodes) | N3 (≥ 7 nodes). Stages I-III are potentially resectable; Stage IV is palliative.
ISHLT Heart Transplant Rejection Grading
| Grade | Histology | Treatment |
|---|---|---|
| 0R | No rejection | None |
| 1R (mild) | Focal/diffuse perivascular or interstitial infiltrate without myocyte damage | Observation or augment immunosuppression |
| 2R (moderate) | Multifocal infiltrate with myocyte damage | Pulse methylprednisolone 1 g/day × 3 |
| 3R (severe) | Diffuse infiltrate with myocyte necrosis ± edema, hemorrhage, vasculitis | Pulse steroids + ATG; consider hemodynamic support |
Masaoka-Koga Staging (Thymoma)
Stage I (encapsulated) | IIA (microscopic capsular invasion) | IIB (macroscopic invasion into fat/pleura) | III (invasion into adjacent organs) | IVA (pleural/pericardial dissemination) | IVB (distant metastasis). See Section 15 for full details.
Modified WHO Classification of Thymoma
| Type | Histology | Behavior |
|---|---|---|
| A | Spindle/oval cells; no atypia | Benign; excellent prognosis |
| AB | Mixed type A + lymphocyte-rich areas | Low-grade malignancy; good prognosis |
| B1 | Lymphocyte-rich; resembles normal thymus | Low-grade malignancy |
| B2 | Mixed epithelial and lymphocytic; scattered epithelial cells | Moderate malignancy |
| B3 | Predominantly epithelial; minimal lymphocytes | Moderate-high malignancy |
| C | Thymic carcinoma — clear cytological atypia | High-grade malignancy; poor prognosis; often unresectable |
Carpentier Classification (Mitral Regurgitation)
Type I = normal motion; Type II = excessive motion (prolapse); Type IIIa = restricted in systole and diastole (rheumatic); Type IIIb = restricted in systole only (functional/ischemic). See Section 8 for details.
INTERMACS Profiles (Mechanical Circulatory Support)
| Profile | Description | Time to MCS |
|---|---|---|
| 1 — Critical cardiogenic shock ("crash and burn") | Life-threatening hypotension despite escalating inotropes/vasopressors and IABP; lactic acidosis, end-organ failure | Hours |
| 2 — Progressive decline ("sliding on inotropes") | Inotrope-dependent with worsening renal function, nutritional status, or end-organ perfusion despite inotropes | Days |
| 3 — Stable but inotrope-dependent | Hemodynamically stable on continuous inotropes; unable to wean | Weeks to months |
| 4 — Resting symptoms | Symptoms at rest on oral therapy; frequent rehospitalizations; no inotrope dependence | Weeks to months |
| 5 — Exertion intolerant | Comfortable at rest, unable to do any activity; living at home | Variable |
| 6 — Exertion limited ("walking wounded") | Comfortable at rest, able to do mild activity; fatigue within minutes | Variable |
| 7 — Advanced NYHA III | Patient in NYHA III without recent decompensation | Not imminent |
Wilkins Score (Mitral Balloon Valvotomy Eligibility)
| Feature | 1 Point | 2 Points | 3 Points | 4 Points |
|---|---|---|---|---|
| Mobility | Highly mobile, tip only restricted | Midportion and base have reduced mobility | Valve moves forward in diastole mainly from the base | No or minimal forward movement |
| Thickening | Leaflets near normal (4-5 mm) | Midleaflet thickening, margins normal | Thickening through entire leaflet (5-8 mm) | Considerable thickening (> 8 mm) |
| Calcification | Single area of brightness | Scattered areas confined to margins | Extending into mid-leaflets | Extensive, throughout leaflets |
| Subvalvular | Minimal thickening below leaflets | Thickening of chordae, extending up to one-third of length | Thickening extending to distal third of chordae | Extensive thickening of all chordae, extending to papillary muscles |
Total score 0-16. Score ≤ 8 is favorable for percutaneous mitral balloon commissurotomy. Score > 8 predicts poor outcome with balloon valvotomy — surgical commissurotomy or MVR preferred.
Lung Cancer TNM Quick Reference
| T Stage | Size / Features |
|---|---|
| T1a / T1b / T1c | ≤ 1 cm / > 1-2 cm / > 2-3 cm; surrounded by lung parenchyma |
| T2a / T2b | > 3-4 cm / > 4-5 cm; or involves main bronchus (not carina), visceral pleura, or associated atelectasis |
| T3 | > 5-7 cm; chest wall, pericardium, phrenic nerve invasion; separate nodule(s) in same lobe |
| T4 | > 7 cm; mediastinum, diaphragm, heart, great vessels, trachea, recurrent laryngeal nerve, esophagus, vertebra, carina invasion; separate nodule(s) in different ipsilateral lobe |
STS-PROM Risk Categories
| Risk Category | STS-PROM | Implication |
|---|---|---|
| Low risk | < 4% | Favors SAVR (long-term durability data better than TAVR) |
| Intermediate risk | 4-8% | SAVR or TAVR both acceptable (Heart Team decision) |
| High risk | ≥ 8% | TAVR generally preferred (lower periprocedural risk) |
| Extreme / prohibitive risk | ≥ 15% or other prohibitive factors | TAVR strongly preferred; SAVR futile in some cases |
Postoperative Bleeding — Transfusion Triggers
| Product | Trigger / Indication | Target |
|---|---|---|
| Packed red blood cells (pRBCs) | Hgb < 7 g/dL (restrictive strategy per TRICS III trial); < 8 g/dL if active bleeding or hemodynamic instability | Hgb 7-9 g/dL |
| Platelets | Platelet count < 100K with active bleeding; < 50K prophylactically | > 100K if ongoing bleeding |
| Fresh frozen plasma (FFP) | INR > 1.5 with active bleeding | INR < 1.5 |
| Cryoprecipitate | Fibrinogen < 200 mg/dL with active bleeding | Fibrinogen > 200 mg/dL |
| Prothrombin complex concentrate (PCC / Kcentra) | Warfarin reversal or factor concentrate for refractory coagulopathic bleeding | Per clinical response |
27 Complications
| Complication | Incidence | Recognition | Management |
|---|---|---|---|
| Postoperative bleeding | 3-5% require re-exploration | Chest tube output > 200 mL/hr × 2 hrs; hemodynamic instability | Correct coagulopathy (protamine, FFP, platelets, cryo, TXA); re-exploration if medical management fails |
| Cardiac tamponade | 1-3% after cardiac surgery | Hypotension, elevated CVP, equalized filling pressures, low CO, muffled heart sounds; TEE: RV/RA compression | Emergent re-sternotomy; maintain preload and HR as bridge |
| Stroke | 1-3% after CABG; 1-5% after valve surgery | New focal neurological deficit on awakening or postoperatively | Emergent CT head; neurology consultation; consider thrombectomy if large-vessel occlusion within time window |
| Postoperative AF | 20-40% (CABG); 50-60% (valve) | Irregular rhythm on telemetry, POD 2-3 peak | Rate control (beta-blocker, amiodarone); anticoagulation if persistent > 48 hrs; cardioversion if unstable |
| Deep sternal wound infection / Mediastinitis | 1-3% | Sternal instability, wound drainage, fever, leukocytosis, positive cultures | Surgical debridement, rewiring or muscle flap closure, IV antibiotics × 4-6 weeks |
| Acute kidney injury (AKI) | 5-30% (stage 1-3); 2-5% require RRT | Rise in creatinine, oliguria | Optimize hemodynamics, avoid nephrotoxins, RRT if needed |
| Chylothorax | 0.5-2% (higher with esophagectomy) | Milky chest tube output; pleural fluid triglycerides > 110 mg/dL | NPO + TPN or low-fat diet (MCT-based) + octreotide; thoracic duct ligation (VATS or thoracotomy) if output > 1 L/day for > 5-7 days |
| Recurrent laryngeal nerve injury | 1-5% (aortic arch surgery, esophagectomy) | Hoarseness, aspiration, weak cough | Speech therapy; laryngoscopy to confirm vocal cord paralysis; medialization thyroplasty if persistent |
| Phrenic nerve injury | 1-5% (LIMA harvest, redo surgery, topical cooling) | Hemidiaphragm elevation on CXR, difficulty weaning from ventilator | Usually recovers over months; diaphragm plication if symptomatic and persistent |
| Spinal cord ischemia / Paraplegia | 2-10% (TAAA repair); < 3% (TEVAR) | Lower extremity weakness or paralysis on awakening | Immediate CSF drainage (target CSF pressure < 10 mmHg), raise MAP > 90 mmHg, optimize hemoglobin, avoid hypoxia |
| Bronchopleural fistula (BPF) | 1-4% (pneumonectomy); < 1% (lobectomy) | Air leak, subcutaneous emphysema, empyema, contralateral aspiration | Chest tube drainage, NPO, bronchoscopic assessment; surgical closure + muscle flap reinforcement |
| Postpericardiotomy syndrome | 10-40% | Fever, pleuritic chest pain, pericardial/pleural effusion 1-6 weeks postop; elevated ESR/CRP | NSAIDs + colchicine; corticosteroids for refractory cases; pericardiocentesis if tamponade |
| Vasoplegia | 5-25% after CPB | Hypotension with low SVR, normal or high CO, warm extremities; refractory to vasopressors | Norepinephrine + vasopressin; methylene blue (1.5-2 mg/kg) for refractory cases; hydroxocobalamin |
| Heart block | 2-5% (AVR), < 1% (CABG) | New conduction abnormality on ECG; hemodynamic compromise if complete block | Temporary epicardial pacing; permanent pacemaker if persistent > 5-7 days (or immediately for complete block post-TAVR/AVR) |
| Prosthetic valve endocarditis | 1-4% (early < 60 days; late > 60 days) | Fever, new murmur, embolic phenomena; positive blood cultures; vegetations on TEE | Prolonged IV antibiotics (6+ weeks); surgical debridement + valve re-replacement for abscess, dehiscence, or persistent sepsis |
| Pneumonia / Ventilator-associated pneumonia | 5-10% | Fever, purulent secretions, new infiltrate on CXR, leukocytosis | Broad-spectrum antibiotics (de-escalate based on culture); aggressive pulmonary toilet; early extubation reduces risk |
Emergent re-sternotomy (also called "re-exploration" or "take-back") is a life-saving procedure performed in the cardiac surgery ICU for: cardiac tamponade (the most urgent indication — can cause PEA arrest), massive hemorrhage (chest tube output meeting re-exploration criteria), or cardiac arrest within 10 days of cardiac surgery (open cardiac massage is indicated per Society of Thoracic Surgeons guidelines — external chest compressions on a fresh sternotomy risk sternal wire disruption and injury to the heart/grafts). Protocol: in extremis, re-sternotomy can be performed at the bedside in the ICU — the surgeon removes sternal wires, opens the chest, evacuates clot, and performs internal cardiac massage and/or identifies and controls the bleeding source. The patient is then transferred to the OR for definitive repair.
28 Medications Master Table
| Drug | Class | Typical Dose | Key Indication in CT Surgery |
|---|---|---|---|
| Heparin (UFH) | Anticoagulant | 300-400 U/kg for CPB; 80 U/kg bolus + 18 U/kg/hr for treatment | CPB anticoagulation; ECMO; VTE treatment |
| Protamine | Heparin reversal | 1-1.3 mg per 100 U heparin; slow IV push | Reversal of heparin after CPB |
| Warfarin | Vitamin K antagonist | Titrate to target INR | Mechanical prosthetic valves, AF, VTE |
| Aspirin | Antiplatelet | 81-325 mg daily | Post-CABG (SVG protection), bioprosthetic valves |
| Clopidogrel | P2Y12 inhibitor | 75 mg daily | Post-TAVR, DAPT post-stenting; hold 5-7 days before surgery |
| Tranexamic acid (TXA) | Antifibrinolytic | 1 g load + 1 g in pump + 1 g infusion | Blood conservation during CPB |
| Aminocaproic acid | Antifibrinolytic | 5 g load + 1 g/hr infusion | Alternative to TXA |
| Milrinone | PDE-3 inhibitor | 0.125-0.75 mcg/kg/min | Low CO with elevated PVR; post-CPB |
| Dobutamine | Beta-1 agonist | 2-20 mcg/kg/min | Low cardiac output syndrome |
| Norepinephrine | Alpha-1 > Beta-1 agonist | 0.01-3 mcg/kg/min | Vasoplegia post-CPB |
| Vasopressin | V1 agonist | 0.01-0.04 U/min | Vasoplegia refractory to norepinephrine |
| Epinephrine | Alpha/Beta agonist | 0.01-0.5 mcg/kg/min | Severe cardiogenic shock; cardiac arrest |
| Amiodarone | Class III antiarrhythmic | 150 mg IV bolus, then 1 mg/min × 6 hr, then 0.5 mg/min; oral 200-400 mg/day | POAF prevention and treatment |
| Metoprolol | Beta-blocker | 5 mg IV q5min (max 15 mg); 25-100 mg PO BID | Rate control; POAF prevention; anti-impulse therapy in dissection |
| Esmolol | Ultra-short-acting beta-blocker | 500 mcg/kg load, then 50-300 mcg/kg/min | Acute aortic dissection BP/HR control |
| Nicardipine | IV calcium channel blocker | 5-15 mg/hr IV | Adjunct BP control in aortic dissection |
| Inhaled nitric oxide (iNO) | Selective pulmonary vasodilator | 20-40 ppm | RV failure with elevated PVR; post-transplant |
| Tacrolimus | Calcineurin inhibitor | 0.05-0.1 mg/kg/day; target trough 5-15 ng/mL | Transplant immunosuppression (cornerstone) |
| Mycophenolate mofetil | Antimetabolite | 1-1.5 g PO BID | Transplant immunosuppression |
| Prednisone | Corticosteroid | Taper from 1 mg/kg to 5 mg/day | Transplant immunosuppression; rejection treatment |
| Cisplatin | Platinum-based chemotherapy | Per protocol (typically 75 mg/m²) | NSCLC adjuvant, esophageal ca (CROSS), mesothelioma |
| Pemetrexed | Antifolate | Per protocol (500 mg/m²) | Non-squamous NSCLC, mesothelioma |
| Nivolumab | Anti-PD-1 immunotherapy | Per protocol | Adjuvant esophageal ca (CheckMate 577), neoadjuvant NSCLC (CM 816), mesothelioma |
29 Abbreviations Master List
References & Figure Sources
Figures
- Figure 1 — Coronary Artery Anatomy. Wikimedia Commons. Public domain.
- Figure 2 — Bronchial Tree and Lung Lobes. NIH. Wikimedia Commons. Public domain.
- Figure 3 — Aortic Dissection Classification (Stanford). Wikimedia Commons. Public domain.
Key Trials & Guidelines
- Serruys PW, Morice MC, Kappetein AP, et al. Percutaneous coronary intervention versus coronary-artery bypass grafting for severe coronary artery disease (SYNTAX trial). N Engl J Med. 2009;360(10):961-972. PMID: 19228612
- Farkouh ME, Domanski M, Sleeper LA, et al. Strategies for multivessel revascularization in patients with diabetes (FREEDOM trial). N Engl J Med. 2012;367(25):2375-2384. PMID: 23121323
- Maron DJ, Hochman JS, Reynolds HR, et al. Initial invasive or conservative strategy for stable coronary disease (ISCHEMIA trial). N Engl J Med. 2020;382(15):1395-1407. PMID: 32227756
- Taggart DP, Benedetto U, Gerry S, et al. Bilateral versus single internal-thoracic-artery grafts at 10 years (ART trial). N Engl J Med. 2019;380(5):437-446. PMID: 31582132
- Gaudino M, Benedetto U, Bakaeen F, et al. Radial-artery or saphenous-vein grafts in coronary-artery bypass surgery (RADIAL trial). N Engl J Med. 2018;378(22):2069-2077. PMID: 30508392
- Stone GW, Kappetein AP, Sabik JF, et al. Five-year outcomes after PCI or CABG for left main coronary disease (EXCEL trial). N Engl J Med. 2019;381(19):1820-1830. PMID: 31562798
- Goldstone AB, Chiu P, Baiocchi M, et al. Mechanical or biologic prostheses for aortic-valve and mitral-valve replacement. N Engl J Med. 2017;377(19):1847-1857. PMID: 29117490
- Acker MA, Parides MK, Perrault LP, et al. Mitral-valve repair versus replacement for severe ischemic mitral regurgitation (CTSN trial). N Engl J Med. 2014;370(1):23-32. PMID: 24245543
- Stone GW, Lindenfeld J, Abraham WT, et al. Transcatheter mitral-valve repair in patients with heart failure (COAPT trial). N Engl J Med. 2018;379(24):2307-2318. PMID: 30280640
- Nienaber CA, Kische S, Rousseau H, et al. Endovascular repair of type B aortic dissection (INSTEAD-XL trial). Circ Cardiovasc Interv. 2013;6(4):407-416. PMID: 23918683
- van Hagen IM, Shapiro LM, Hien Dat Nguyen, et al. Preoperative chemoradiotherapy for esophageal or junctional cancer (CROSS trial). N Engl J Med. 2012;366(22):2074-2084. PMID: 22646630
- Kelly RJ, Ajani JA, Kuzdzal J, et al. Adjuvant nivolumab in resected esophageal or gastroesophageal junction cancer (CheckMate 577). N Engl J Med. 2021;384(13):1191-1203. PMID: 33789008
- Mehra MR, Uriel N, Naka Y, et al. A fully magnetically levitated left ventricular assist device (MOMENTUM 3 trial). N Engl J Med. 2019;380(17):1618-1627. PMID: 30575491
- Saji L, Aoki J, Suzuki T, et al. Segmentectomy versus lobectomy in small-sized peripheral non-small-cell lung cancer (JCOG0802/WJOG4607L). Lancet. 2022;399(10335):1607-1617. PMID: 35220659
Textbooks & Reference Works
- Sabiston and Spencer's Surgery of the Chest. 10th ed. Sellke FW, del Nido PJ, Swanson SJ, eds. Elsevier; 2022.
- Cardiac Surgery in the Adult. 6th ed. Cohn LH, Adams DH, eds. McGraw-Hill; 2023.
- Shields' General Thoracic Surgery. 8th ed. LoCicero J III, Feins RH, Colson YL, Rocco G, eds. Wolters Kluwer; 2019.
- Kirklin/Barratt-Boyes Cardiac Surgery. 4th ed. Kouchoukos NT, Blackstone EH, Hanley FL, Kirklin JK, eds. Elsevier; 2013.
- Gray's Anatomy: The Anatomical Basis of Clinical Practice. 42nd ed. Standring S, ed. Elsevier; 2020.
- The Washington Manual of Cardiothoracic Surgery. Krupnick AS, Kreisel D, eds. Lippincott Williams & Wilkins; 2017.
- Glenn's Thoracic and Cardiovascular Surgery. 7th ed. Baue AE, Geha AS, Hammond GL, et al., eds. Appleton & Lange; 2005.
Key Online Resources
- STS National Database & Risk Calculator: sts.org/resources/risk-calculator — Operative risk prediction for CABG, valve, and combined procedures.
- EuroSCORE II Calculator: euroscore.org — European cardiac surgery risk scoring system.
- AJCC Cancer Staging (8th Edition): TNM staging for lung, esophageal, and mediastinal malignancies.
- ACC/AHA Valvular Heart Disease Guidelines (2020/2021 update): Comprehensive guidelines for diagnosis and management of valvular heart disease.
- AATS/STS Guidelines on Thoracic Aortic Disease (2022 update): Recommendations for management of thoracic aortic aneurysm and dissection.