Colorectal Surgery
Every diagnosis, classification, procedure, technique, medication, complication, and management algorithm across the full scope of colorectal surgery in one place.
01 Colorectal Anatomy
The large intestine extends from the ileocecal valve to the anus, measuring approximately 150 cm in length. Understanding segmental anatomy, blood supply, and lymphatic drainage is essential for oncologic resection, vascular ligation, and stoma formation.
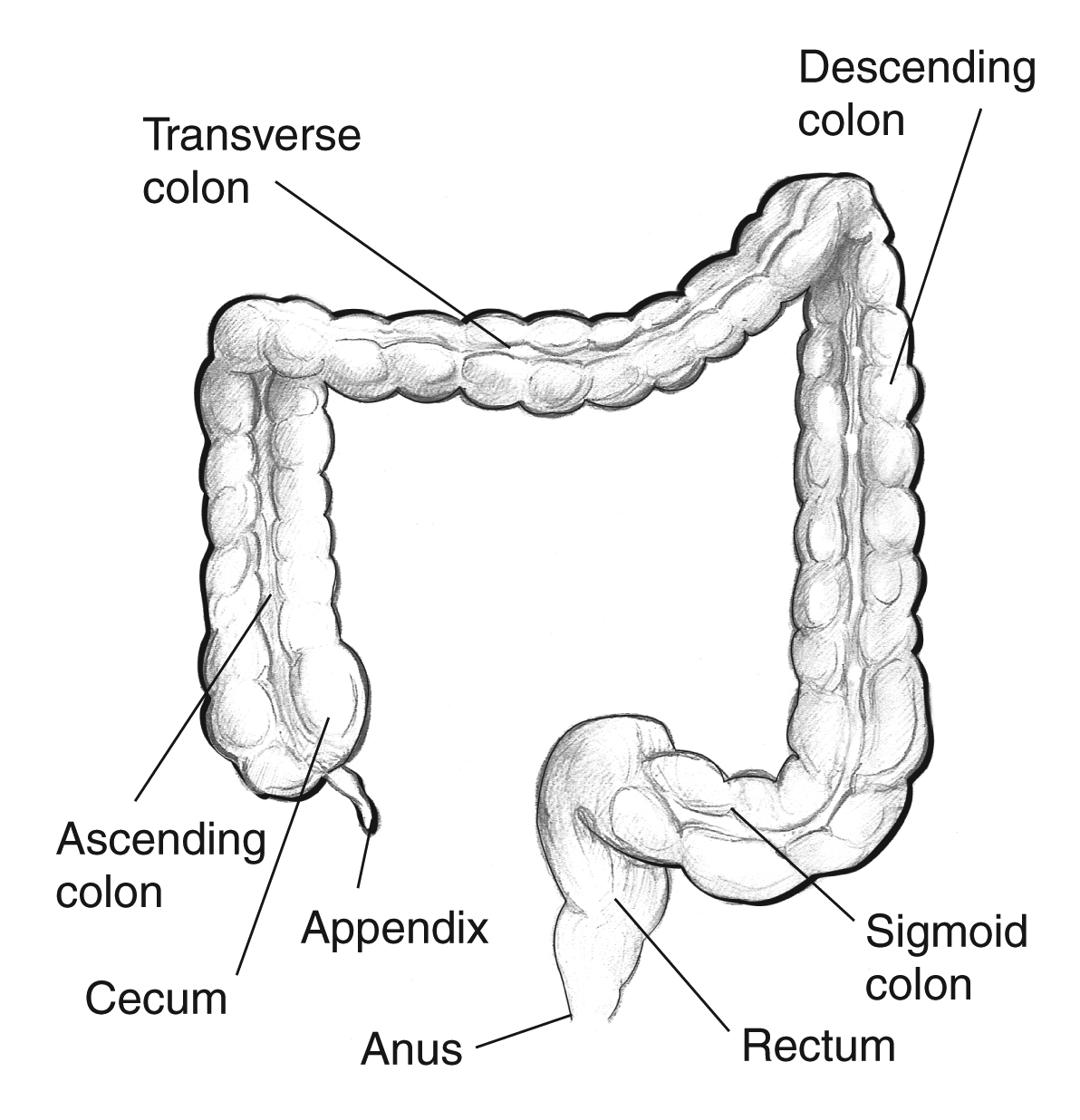
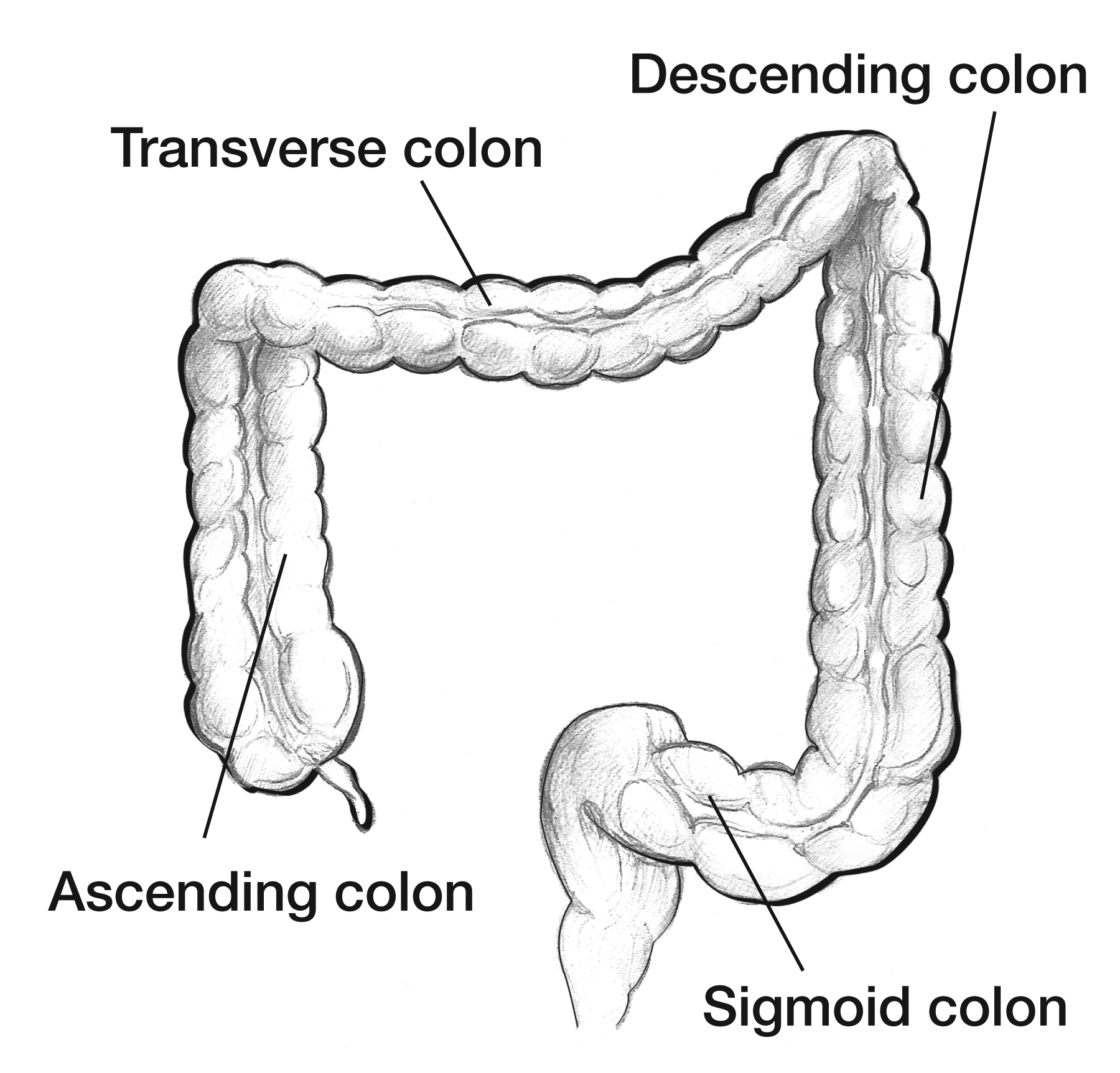
Colonic Segments
The cecum is the most proximal segment, a blind pouch located in the right iliac fossa. The appendix arises from the posteromedial wall of the cecum at the convergence of the three taeniae coli. The ascending colon is retroperitoneal (fixed posteriorly by Toldt's fascia) and extends to the hepatic flexure. The transverse colon is intraperitoneal, suspended by the transverse mesocolon, and extends from the hepatic flexure to the splenic flexure. The descending colon is retroperitoneal and runs from the splenic flexure to the sigmoid colon. The sigmoid colon is intraperitoneal with a variable-length mesentery and transitions to the rectum at the sacral promontory (S3).
Distinguishing Features of the Colon
Three features distinguish the colon from the small bowel: taeniae coli (three longitudinal muscle bands — anterior/free, posteromedial, and posterolateral — that converge at the appendix and fan out to form a complete longitudinal muscle layer at the rectum), haustra (sacculations between the taeniae), and appendices epiploicae (small fat-filled peritoneal outpouchings along the colon surface, most prominent on the sigmoid and transverse colon).
The Rectum
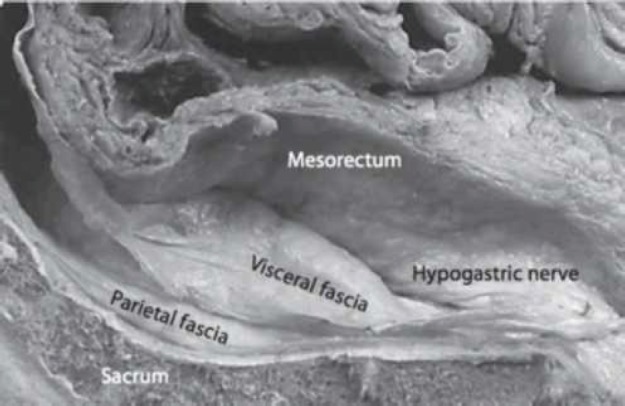
The rectum begins at the sacral promontory (S3) and extends approximately 12–15 cm to the anorectal junction (dentate line). It lacks taeniae coli and haustra. Surgically, the rectum is divided into thirds: upper third (12–15 cm from the anal verge, covered by peritoneum anteriorly), middle third (8–12 cm, peritoneum reflects off the anterior surface forming the rectovesical/rectouterine pouch), and lower third (0–8 cm, entirely extraperitoneal). The mesorectum is the fatty tissue envelope surrounding the rectum, enclosed by the mesorectal fascia (fascia propria of the rectum). It contains the superior rectal vessels, lymph nodes, and autonomic nerves. The circumferential resection margin (CRM) is the non-peritonealized mesorectal surface — a CRM of less than 1 mm is associated with significantly higher local recurrence rates.
Blood Supply
The arterial supply of the colon is derived from two major vessels:
Superior mesenteric artery (SMA) — supplies the cecum through the proximal two-thirds of the transverse colon via the ileocolic artery, right colic artery (variable, present in only 10–40% of individuals), and middle colic artery.
Inferior mesenteric artery (IMA) — supplies the distal transverse colon, descending colon, sigmoid colon, and upper rectum via the left colic artery, sigmoid arteries (2–5 branches), and superior rectal artery (terminal branch of the IMA).
The marginal artery of Drummond is a continuous arterial arcade running along the mesenteric border of the colon, connecting the SMA and IMA territories. It is formed by the anastomotic connections between the terminal branches of the ileocolic, right colic, middle colic, left colic, and sigmoid arteries. The arc of Riolan (meandering mesenteric artery) is a more central collateral vessel connecting the proximal SMA (via the middle colic) and IMA (via the left colic) trunks, present in some individuals, especially those with chronic mesenteric stenosis. A well-developed arc of Riolan is protective when the IMA is ligated during left-sided resections, as it maintains blood supply to the left colon via the SMA.
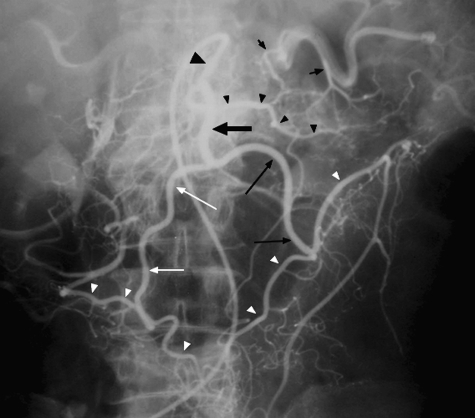
Watershed Areas
| Watershed | Location | Collateral | Clinical Significance |
|---|---|---|---|
| Griffiths' point | Splenic flexure | Between SMA (middle colic) and IMA (left colic) | Most common site of ischemic colitis |
| Sudeck's point | Rectosigmoid junction | Between last sigmoid artery and superior rectal artery | Risk of rectal stump ischemia after IMA ligation |
Rectal Blood Supply
The rectum has a triple blood supply: superior rectal artery (terminal branch of the IMA — main supply), middle rectal artery (branch of the internal iliac artery — supplies the lower third, variable), and inferior rectal artery (branch of the internal pudendal artery — supplies the anal canal below the dentate line). Venous drainage parallels the arterial supply: the superior rectal vein drains into the portal system (via the IMV), while the middle and inferior rectal veins drain into the systemic (caval) circulation via the internal iliac veins. This dual drainage explains why rectal cancers can metastasize to both the liver (portal) and lungs (systemic) directly.
Lymphatic Drainage
Lymphatic drainage follows the arterial supply. Colonic lymph nodes are classified into four tiers: epicolic (on the bowel wall), paracolic (along the marginal artery), intermediate (along the named arteries — ileocolic, right colic, middle colic, left colic, sigmoid), and principal/central (at the origin of the SMA/IMA). Adequate oncologic resection requires a minimum of 12 lymph nodes for proper staging. Central vascular ligation ensures complete lymphadenectomy along the draining arterial pedicle.
Autonomic Nerves
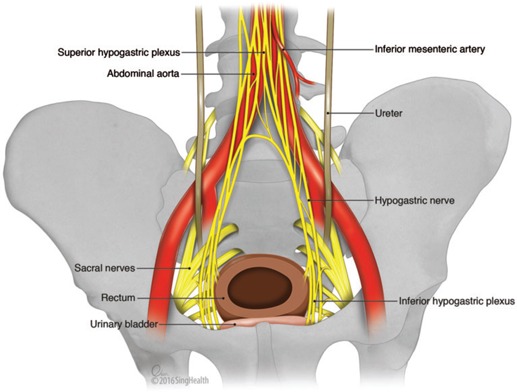
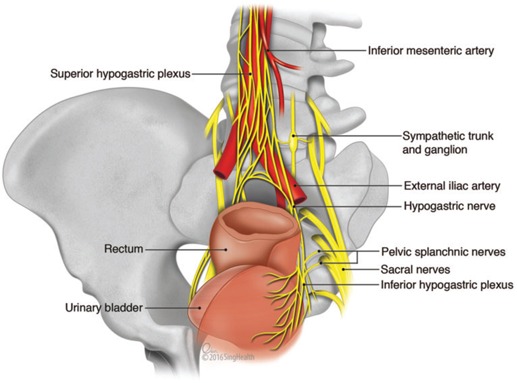
The hypogastric nerves (sympathetic, L2–L4) and pelvic splanchnic nerves (parasympathetic, S2–S4) course through the pelvis and are at risk during rectal surgery. Injury causes urinary dysfunction (atonic bladder, incomplete emptying) and sexual dysfunction (erectile dysfunction, retrograde ejaculation). The inferior hypogastric plexus (pelvic plexus) lies on the pelvic sidewall lateral to the mesorectum — sharp dissection in the areolar plane between the mesorectal fascia and the presacral fascia (Waldeyer's fascia) preserves these nerves. This nerve-sparing approach is a cornerstone of total mesorectal excision (TME).
02 Anorectal Anatomy
The anal canal is approximately 4 cm in length and extends from the anorectal ring (puborectalis sling) to the anal verge. Precise knowledge of anorectal anatomy is essential for managing hemorrhoids, fissures, fistulae, and anal cancer.
The Dentate (Pectinate) Line
The dentate line is the single most important landmark in anorectal surgery. Located approximately 2 cm from the anal verge, it represents the junction between the embryologic hindgut (endoderm) and proctodeum (ectoderm). Above the dentate line: columnar epithelium, visceral innervation (insensitive to pain), arterial supply from the superior rectal artery, venous drainage to the portal system (internal hemorrhoids), and lymphatic drainage to the inferior mesenteric nodes. Below the dentate line: squamous epithelium, somatic innervation (exquisitely sensitive to pain), arterial supply from the inferior rectal artery, venous drainage to the systemic system (external hemorrhoids), and lymphatic drainage to the inguinal nodes.
Anal Canal Lining
The anal transition zone (ATZ) is a 6–12 mm segment above the dentate line containing transitional epithelium (mixture of columnar, cuboidal, and squamous). The columns of Morgagni are 8–14 vertical folds of mucosa in the upper anal canal; at their base are the anal crypts, into which the anal glands (6–10) drain. Obstruction of these crypts and glands is the pathogenesis of cryptoglandular anorectal abscess and fistula-in-ano.
Sphincter Complex
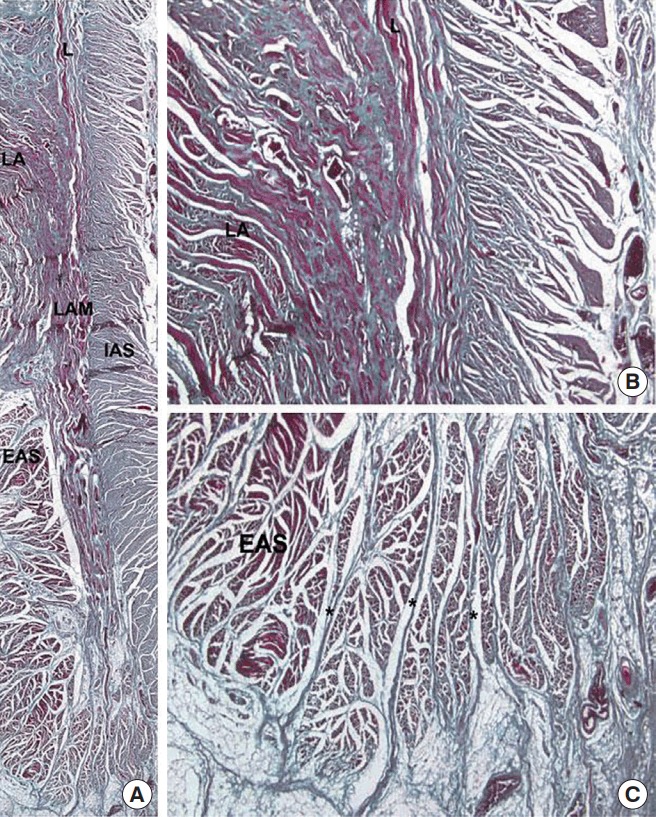
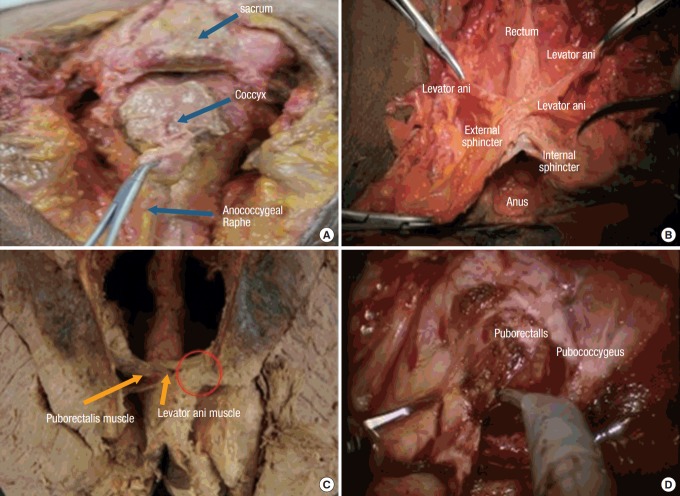
The internal anal sphincter (IAS) is a thickened continuation of the circular smooth muscle of the rectum. It is involuntary, tonically contracted (responsible for 70–85% of resting anal tone), and innervated by the autonomic nervous system. The external anal sphincter (EAS) is a voluntary striated muscle that encircles the IAS. It is innervated by the inferior rectal branch of the pudendal nerve (S2–S4) and provides squeeze pressure for continence. The puborectalis muscle forms a U-shaped sling around the anorectal junction, pulling the rectum anteriorly to create the anorectal angle (approximately 80–90 degrees at rest). This angle is the most important component of the continence mechanism.
Intersphincteric Space
The intersphincteric space lies between the IAS and EAS. It contains the intersphincteric portion of the anal glands and is the plane of dissection for intersphincteric resection in very low rectal cancers and for LIFT (ligation of the intersphincteric fistula tract) procedures. The intersphincteric groove can be palpated on digital rectal exam as a palpable sulcus between the two sphincters.
Spaces of the Anorectum
| Space | Location | Clinical Relevance |
|---|---|---|
| Perianal | Subcutaneous tissue surrounding the anal verge | Most common abscess location (60%); easily drained at bedside |
| Ischiorectal (ischioanal) | Lateral to the EAS, medial to the ischial tuberosity | Second most common abscess; can become horseshoe abscess via deep postanal space |
| Intersphincteric | Between IAS and EAS | Origin of cryptoglandular infection; 2nd most common abscess site |
| Supralevator | Above the levator ani, below the peritoneum | Rare abscess; drainage route depends on origin (intersphincteric vs ischiorectal) |
| Deep postanal (Courtney's space) | Behind the anal canal, between the levator ani and anococcygeal ligament | Connects the bilateral ischiorectal fossae — pathway for horseshoe abscess |
03 Colorectal Physiology
Water & Electrolyte Absorption
The colon receives approximately 1,000–1,500 mL of ileal effluent per day and absorbs 90% of the water and electrolytes, producing 100–200 mL of stool. The right colon is the primary site of water and sodium absorption (active sodium transport via epithelial sodium channels, ENaC). The left colon primarily stores and propels stool. Potassium is secreted into the colonic lumen. Loss of the colon (total colectomy) increases stool output to 500–1,500 mL/day; the small bowel adapts over 6–12 months, but patients remain at risk for dehydration and electrolyte depletion.
Colonic Motility
Three patterns of colonic motility: retrograde peristalsis (predominantly in the right colon, mixing content to maximize absorption), segmental contractions (haustral shuttling — non-propulsive, throughout the colon), and high-amplitude propagating contractions (HAPCs) (mass movements, 5–6 per day, propel stool from proximal to distal colon; stimulated by meals [gastrocolic reflex] and upon awakening). Abnormality of HAPCs underlies slow-transit constipation (colonic inertia).
Short-Chain Fatty Acids (SCFAs)
Bacterial fermentation of undigested carbohydrates produces short-chain fatty acids — butyrate, propionate, and acetate. Butyrate is the primary energy source for colonocytes (provides 60–70% of their energy needs). SCFAs stimulate sodium and water absorption, promote mucosal healing, and have anti-inflammatory properties. Diversion colitis (inflammation of a defunctionalized colon) results from butyrate deprivation and may be treated with SCFA enemas.
Defecation Physiology
Stool entry into the rectum causes distension, which is sensed by stretch receptors in the rectal wall and levator ani (the sampling reflex allows the upper anal canal to distinguish solid, liquid, and gas). This triggers the rectoanal inhibitory reflex (RAIR) — transient IAS relaxation mediated by the myenteric plexus. If defecation is appropriate, voluntary relaxation of the EAS and puborectalis straightens the anorectal angle from approximately 90 degrees to 130-140 degrees, and increased intra-abdominal pressure (Valsalva) expels the stool. If defecation is not appropriate, voluntary contraction of the EAS and puborectalis maintains continence until the rectum accommodates the stool bolus (rectal compliance).
The Microbiome
The colon harbors approximately 1011–1012 bacteria per gram of content — the highest density of microorganisms in the human body. The dominant phyla are Bacteroidetes and Firmicutes. The microbiome plays a role in vitamin K and B12 synthesis, bile acid metabolism, immune modulation, colonization resistance against pathogens (e.g., Clostridioides difficile), and potentially in colorectal carcinogenesis (Fusobacterium nucleatum has been associated with CRC). Antibiotic use disrupts the microbiome and predisposes to C. difficile infection — increasingly relevant in the perioperative setting.
04 The Colorectal Exam
Digital Rectal Exam (DRE)
The DRE is the cornerstone of anorectal assessment. With the patient in the left lateral decubitus (Sims') position or prone jackknife, inspect the perianal skin (tags, fissures, fistula openings, condylomata, dermatitis, excoriation). Then perform a lubricated digital exam assessing: resting tone (IAS function), squeeze pressure (EAS function), puborectalis contraction (posterior sling tightens during squeeze), rectal masses (distance from the anal verge, size, fixation, circumference involved), prostatic abnormalities (men), cervical/uterine abnormalities (women), and the quality of the stool (guaiac testing). For low rectal cancers, DRE can assess tumor fixation and clinical T stage.
Anoscopy
A beveled anoscope (e.g., Ives or Hill-Ferguson) provides direct visualization of the anal canal and distal rectum. It is the best tool for evaluating internal hemorrhoids (which are not visible on external exam unless prolapsing), anal fissures, and condylomata. It also permits office procedures: rubber band ligation, injection sclerotherapy, and biopsy.
Rigid Proctoscopy
A 25 cm rigid proctoscope allows assessment of the rectum up to 20–25 cm from the anal verge. It provides accurate measurement of tumor distance from the anal verge (more reliable than flexible endoscopy), biopsy capability, and assessment of rectal lumen and mucosal detail. This is essential for rectal cancer staging workup — the measured distance from the anal verge determines whether the tumor is "low" (0–5 cm), "mid" (5–10 cm), or "high" (10–15 cm), which directly affects surgical approach.
Anorectal Manometry
Anorectal manometry measures resting pressure (IAS), squeeze pressure (EAS), RAIR, rectal sensation thresholds, and rectal compliance. Indications: fecal incontinence evaluation, preoperative assessment before anorectal surgery (especially fistula surgery in patients with pre-existing continence issues), constipation workup (to identify dyssynergic defecation), and suspected Hirschsprung's disease.
Additional Diagnostic Studies
| Study | Purpose | Key Findings |
|---|---|---|
| Endoanal ultrasound (EAUS) | Evaluate sphincter integrity | Identifies sphincter defects (IAS/EAS), fistula tracts, rectal wall layers for T staging of early rectal cancers |
| MRI pelvis | Rectal cancer staging, fistula mapping | Best for assessing CRM, T stage, N stage, extramural vascular invasion (EMVI), and complex fistula anatomy |
| Defecography (fluoroscopic or MR) | Evaluate pelvic floor dynamics | Identifies rectocele, enterocele, intussusception, anismus (paradoxical puborectalis contraction) |
| Colonic transit study (Sitz marker) | Evaluate colonic motility | Patient ingests radiopaque markers; X-ray at day 5 — retention of >5/24 markers indicates slow transit |
| Balloon expulsion test | Screen for dyssynergic defecation | Inability to expel a 50 mL water-filled balloon within 1–3 minutes suggests pelvic floor dyssynergia |
05 Epidemiology, Risk Factors & Screening
Colorectal cancer (CRC) is the third most common cancer and the second leading cause of cancer death worldwide. In the United States, there are approximately 150,000 new cases and 53,000 deaths annually. The median age at diagnosis is 66, but incidence in adults under 50 is rising (early-onset CRC — a 2% annual increase since the mid-1990s).
Risk Factors
Non-modifiable: age >50, personal history of adenomatous polyps or CRC, family history (first-degree relative with CRC increases risk 2–3x), hereditary syndromes (Lynch, FAP), inflammatory bowel disease (UC > Crohn's, risk increases with duration and extent of disease).
Modifiable: obesity (BMI >30, RR 1.3), red/processed meat consumption, smoking (RR 1.2), heavy alcohol use (>3 drinks/day), physical inactivity, type 2 diabetes.
Protective: aspirin/NSAIDs (chemoprevention), high fiber diet, physical activity, calcium and vitamin D supplementation.
The Adenoma-Carcinoma Sequence
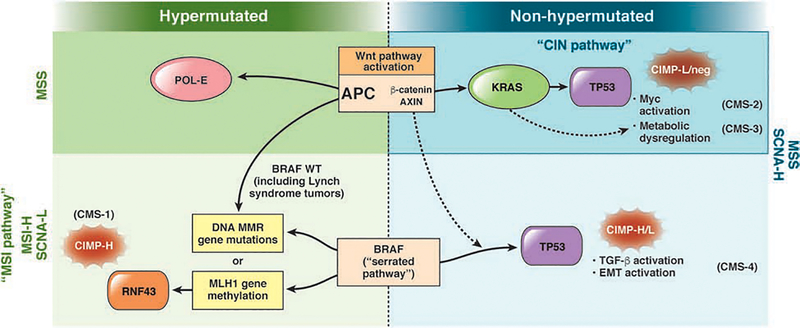
Most CRCs (70–80%) arise through the adenoma-carcinoma sequence — normal epithelium transforms to aberrant crypt foci, then to adenomatous polyp, then to carcinoma over approximately 10–15 years. Key molecular events: APC gene mutation (initiation, 5q), KRAS mutation (early adenoma to intermediate adenoma), DCC/SMAD4 loss (18q), and TP53 mutation (late adenoma to carcinoma, 17p). This chromosomal instability (CIN) pathway accounts for 85% of sporadic CRC. The alternative serrated neoplasia pathway (15%) involves sessile serrated lesions, BRAF V600E mutation, CpG island methylator phenotype (CIMP), and microsatellite instability (MSI-H) — more common in the right colon and in women.
Screening Guidelines
| Population | Start Age | Recommended Tests | Interval |
|---|---|---|---|
| Average risk | 45 (ACS, USPSTF 2021) | Colonoscopy (gold standard) | Every 10 years if normal |
| Average risk — alternatives | 45 | FIT (fecal immunochemical test) or MT-sDNA (Cologuard) | FIT annually; MT-sDNA every 3 years |
| 1st-degree relative with CRC <60 or 2+ relatives any age | 40, or 10 years before youngest case | Colonoscopy | Every 5 years |
| Lynch syndrome (confirmed) | 20–25, or 2–5 years before youngest cancer in family | Colonoscopy | Every 1–2 years |
| FAP (confirmed or at-risk) | 10–12 | Flexible sigmoidoscopy or colonoscopy | Annually until colectomy |
| UC/Crohn's colitis (extensive) | 8 years after symptom onset | Surveillance colonoscopy with chromoendoscopy | Every 1–3 years |
06 Hereditary Colorectal Cancer Syndromes
Lynch Syndrome (Hereditary Nonpolyposis Colorectal Cancer — HNPCC)
Lynch syndrome is the most common hereditary CRC syndrome, accounting for 2–4% of all CRC. It is caused by germline mutations in DNA mismatch repair (MMR) genes: MLH1, MSH2, MSH6, PMS2, or EPCAM (deletion causes MSH2 silencing). Inheritance is autosomal dominant. Tumors demonstrate microsatellite instability (MSI-H) and loss of MMR protein expression on immunohistochemistry.
All criteria must be met ("3-2-1" rule): 3 or more relatives with Lynch-associated cancer (CRC, endometrial, ovarian, ureter/renal pelvis, small bowel, hepatobiliary, stomach, brain); 2 successive generations affected; 1 case diagnosed before age 50; one should be a first-degree relative of the other two; FAP excluded.
CRC diagnosed <50 years; synchronous/metachronous Lynch-associated tumors; CRC with MSI-H histology (tumor-infiltrating lymphocytes, Crohn's-like reaction, mucinous/signet ring, medullary) in patient <60; CRC with one or more first-degree relatives with Lynch-associated tumor <50; CRC with two or more relatives with Lynch-associated tumors at any age.
Universal tumor testing: Current guidelines (NCCN) recommend that all newly diagnosed CRC undergo MSI/MMR testing regardless of age or family history. This identifies Lynch syndrome and also predicts response to immunotherapy (MSI-H tumors respond to pembrolizumab/nivolumab).
Surgical management: When a Lynch patient develops CRC, subtotal colectomy with ileorectal anastomosis (IRA) is preferred over segmental resection because the lifetime risk of metachronous CRC is 16% at 10 years and 40% at 20 years. The remaining rectum requires annual surveillance. For women, prophylactic hysterectomy and bilateral salpingo-oophorectomy should be discussed after childbearing is complete (endometrial cancer risk 40–60%, ovarian cancer risk 9–12%).
Familial Adenomatous Polyposis (FAP)
FAP is caused by a germline mutation in the APC gene (5q21), inherited in an autosomal dominant pattern. Patients develop hundreds to thousands of adenomatous polyps throughout the colon, typically by age 15–20. Without prophylactic surgery, 100% develop CRC by age 40. Approximately 25% of cases are de novo mutations.
Extracolonic manifestations: duodenal/periampullary adenomas (most common extraintestinal manifestation; leading cause of death after colectomy — lifetime risk of duodenal cancer 4–12%), desmoid tumors (10–20%, often mesenteric — can be life-threatening), fundic gland polyps (gastric), osteomas (mandible — Gardner syndrome), congenital hypertrophy of the retinal pigment epithelium (CHRPE), epidermoid cysts, supernumerary teeth, and hepatoblastoma in children.
Surgical management: Prophylactic colectomy is required, typically in late teens to early 20s. Options: total proctocolectomy with IPAA (J-pouch) — gold standard, eliminates all at-risk mucosa; or total colectomy with IRA — simpler, better functional outcomes, but retains rectal mucosa requiring lifelong surveillance (6-month proctoscopy). IRA is preferred in patients with few rectal polyps (<20) and those who prioritize fertility/function. IPAA is preferred when there are many rectal polyps or a known high-risk APC mutation (codon 1250–1464).
Attenuated FAP (AFAP)
10–99 polyps, later onset (mean age 55 for CRC), right colon predominance. May be managed with annual colonoscopy and polypectomy in early stages. Colectomy when polyps are unmanageable endoscopically.
Other Polyposis Syndromes
| Syndrome | Gene | Inheritance | Key Features | CRC Risk |
|---|---|---|---|---|
| MUTYH-associated polyposis (MAP) | MUTYH | Autosomal recessive | 10–100+ adenomas, phenocopies attenuated FAP | 43–100% |
| Peutz-Jeghers syndrome | STK11/LKB1 | AD | Hamartomatous GI polyps, mucocutaneous melanin spots (lips, buccal, fingers) | 39% |
| Juvenile polyposis syndrome | SMAD4, BMPR1A | AD | Multiple juvenile (hamartomatous) polyps in GI tract | 39–68% |
| Serrated polyposis syndrome | Unknown/polygenic | Variable | Multiple serrated polyps, especially proximal colon | Elevated (up to 50% lifetime) |
07 TNM Staging & Pathology
AJCC 8th Edition TNM Staging for Colorectal Cancer
| T Stage | Definition |
|---|---|
| Tis | Carcinoma in situ — intramucosal carcinoma (involvement of lamina propria with no extension through muscularis mucosae) |
| T1 | Tumor invades the submucosa |
| T2 | Tumor invades the muscularis propria |
| T3 | Tumor invades through the muscularis propria into pericolorectal tissues (subserosa or non-peritonealized pericolic/perirectal tissue) |
| T4a | Tumor penetrates the visceral peritoneum (serosa) |
| T4b | Tumor directly invades or is adherent to adjacent organs or structures |
| N Stage | Definition |
|---|---|
| N0 | No regional lymph node metastasis |
| N1a | 1 regional lymph node positive |
| N1b | 2–3 regional lymph nodes positive |
| N1c | Tumor deposit(s) in subserosa/mesentery without regional node metastasis |
| N2a | 4–6 regional lymph nodes positive |
| N2b | 7 or more regional lymph nodes positive |
| M Stage | Definition |
|---|---|
| M0 | No distant metastasis |
| M1a | Metastasis to one site/organ without peritoneal metastasis |
| M1b | Metastasis to two or more sites/organs without peritoneal metastasis |
| M1c | Peritoneal metastasis with or without other organ involvement |
Stage Grouping & Approximate 5-Year Survival
| Stage | TNM | Dukes | 5-Year OS |
|---|---|---|---|
| 0 | Tis N0 M0 | — | >95% |
| I | T1–T2 N0 M0 | A | 90–92% |
| IIA | T3 N0 M0 | B1 | 82–87% |
| IIB | T4a N0 M0 | B2 | 63–80% |
| IIC | T4b N0 M0 | B2 | 51–65% |
| IIIA | T1–T2 N1, T1 N2a | C1 | 69–89% |
| IIIB | T3–T4a N1, T2–T3 N2a, T1–T2 N2b | C2 | 50–69% |
| IIIC | T4a N2a, T3–T4a N2b, T4b N1–N2 | C2 | 28–47% |
| IVA | Any T, any N, M1a | D | 10–15% |
| IVB | Any T, any N, M1b | D | 5–8% |
| IVC | Any T, any N, M1c | D | <5% |
Key Pathologic Features
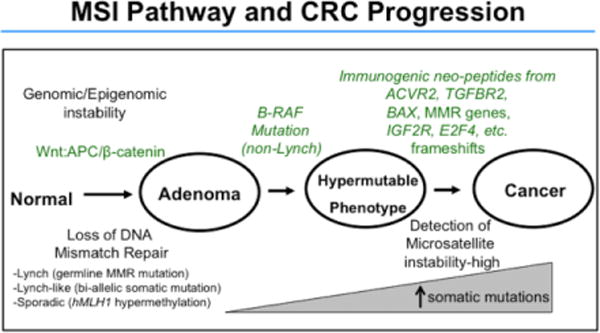
Microsatellite instability (MSI) — MSI-H tumors (15% of CRC) have deficient mismatch repair (dMMR). They are more common in the right colon, have a better prognosis stage-for-stage, do NOT benefit from 5-FU monotherapy in stage II disease, and respond to immune checkpoint inhibitors (pembrolizumab). KRAS/NRAS status — RAS wild-type tumors respond to anti-EGFR therapy (cetuximab, panitumumab); RAS-mutant tumors do not. BRAF V600E mutation — associated with poor prognosis, right-sided tumors, serrated pathway; targeted with encorafenib + cetuximab. HER2 amplification — emerging target for anti-HER2 therapy in RAS wild-type CRC.
Malignant Polyp (T1 CRC)
A polyp with invasive adenocarcinoma invading the submucosa. After polypectomy, oncologic resection is required if any of the following adverse features are present: positive margin (<1 mm), poorly differentiated histology, lymphovascular invasion (LVI), or Haggitt level 4 (sessile polyp with invasion into the stalk base/submucosa of the bowel wall). The Kikuchi classification applies to sessile polyps: sm1 (superficial submucosa), sm2 (mid submucosa), sm3 (deep submucosa) — sm3 has a 23% risk of lymph node metastasis and mandates surgery.
08 Surgical Management of Colon Cancer
Principles of Oncologic Resection
The goal of curative resection is complete removal of the tumor with adequate margins (at least 5 cm longitudinal margin, though 2 cm is accepted distally for rectal cancer) and complete lymphadenectomy along the primary feeding vessel. The operation is determined by tumor location and vascular anatomy.
Operations by Tumor Location
| Tumor Location | Operation | Vessel Ligated | Key Steps |
|---|---|---|---|
| Cecum, ascending colon | Right hemicolectomy | Ileocolic artery (at origin from SMA) | Mobilize right colon from Toldt's fascia; identify duodenum, right ureter, right gonadal vessels; ileocolic anastomosis |
| Hepatic flexure | Extended right hemicolectomy | Ileocolic + right branch of middle colic (or middle colic trunk) | Includes transverse colon; ileocolic or ileotransverse anastomosis |
| Transverse colon | Transverse colectomy or extended right/left hemicolectomy | Middle colic artery | Rarely done as isolated transverse colectomy; usually extended to right or left depending on location |
| Splenic flexure | Extended left hemicolectomy or extended right hemicolectomy | Left colic artery; IMA or middle colic | Technically challenging; requires mobilization of splenic flexure |
| Descending colon | Left hemicolectomy | IMA (high ligation at aortic origin) | Mobilize descending colon; identify left ureter, left gonadal vessels; colorectal or colocolic anastomosis |
| Sigmoid colon | Sigmoid colectomy | IMA or sigmoid arteries | High ligation of IMA ensures adequate lymphadenectomy; anastomosis to upper rectum; confirm adequate blood supply to proximal descending colon (test marginal artery flow) |
Emergency Presentation of Colon Cancer
Approximately 15–30% of colon cancers present as emergencies: obstruction (most common, especially left-sided/sigmoid cancers — presents with progressive constipation, distension, nausea/vomiting, absent flatus), perforation (either at the tumor site or proximal to an obstructing tumor — cecal blowout from closed-loop obstruction when the ileocecal valve is competent), and hemorrhage (significant bleeding requiring transfusion). Management of obstructing colon cancer: for right-sided tumors, right hemicolectomy with primary ileocolic anastomosis is safe even in the emergency setting. For left-sided tumors, options include: (1) resection with primary anastomosis (with or without on-table lavage/diverting ileostomy), (2) Hartmann procedure (sigmoid resection, end colostomy, rectal stump closure — safest in unstable patients), or (3) endoscopic self-expanding metallic stent (SEMS) as a bridge to elective surgery (controversial — the CReST trial showed no overall survival benefit, and concern exists about tumor seeding and perforation; currently ESGE recommends stent only in patients with incurable disease or unfit for emergency surgery).
Complete Mesocolic Excision (CME)
CME is the colon cancer equivalent of TME. It involves dissection in the embryologic plane between the mesocolic and retroperitoneal fascia, preserving an intact mesocolic envelope, with central vascular ligation. Hohenberger demonstrated that CME with central vascular ligation improves oncologic outcomes compared to standard resection (local recurrence 3.6% vs 6.5%, 5-year OS 89% vs 82%). While not universally adopted, CME principles are increasingly incorporated into colon cancer surgery. PMID: 19997731
Anastomotic Technique
Anastomoses can be created with hand-sewn (single or double layer) or stapled technique. For ileocolic anastomoses, a functional end-to-end (side-to-side) stapled technique is most common. For colorectal anastomoses after sigmoid or rectal resection, an end-to-end circular stapler (EEA) is used, creating a double-stapled anastomosis. Key principles: well-perfused tissue (check for bleeding from cut edges), tension-free, no twisting of the mesentery. An air leak test (insufflate rectum with rigid proctoscope under saline) confirms integrity after low anastomoses.
Laparoscopic vs Open
Multiple landmark RCTs have established the oncologic equivalence of laparoscopic and open colectomy: COST trial (2004) — no difference in 3-year OS, DFS, or recurrence; COLOR trial (2005) — similar long-term oncologic outcomes; CLASICC trial (2007) — equivalent outcomes for colon cancer (rectal cancer results were less clear). Benefits of laparoscopy: shorter hospital stay (5 vs 7 days), less pain, faster return of bowel function, fewer wound complications. Conversion rate is approximately 10–20%. PMID: 15483043
Adjuvant Chemotherapy for Colon Cancer
Stage I: Surgery alone — no adjuvant therapy. Stage II: Controversial. Standard approach is observation unless high-risk features are present (T4, poorly differentiated, LVI, perineural invasion, <12 nodes examined, positive margins, bowel obstruction/perforation at presentation). High-risk stage II: consider capecitabine or 5-FU/leucovorin monotherapy (MSI-H tumors may not benefit from 5-FU). Stage III: Adjuvant chemotherapy is standard — FOLFOX (5-FU, leucovorin, oxaliplatin) or CAPOX/XELOX (capecitabine + oxaliplatin) for 3–6 months. The IDEA collaboration showed that for low-risk stage III (T1–T3 N1), 3 months of CAPOX is non-inferior to 6 months, sparing oxaliplatin-related peripheral neuropathy. High-risk stage III (T4 or N2): 6 months remains standard. PMID: 29355075
09 Rectal Cancer — TME, Neoadjuvant Therapy & Organ Preservation
Total Mesorectal Excision (TME)
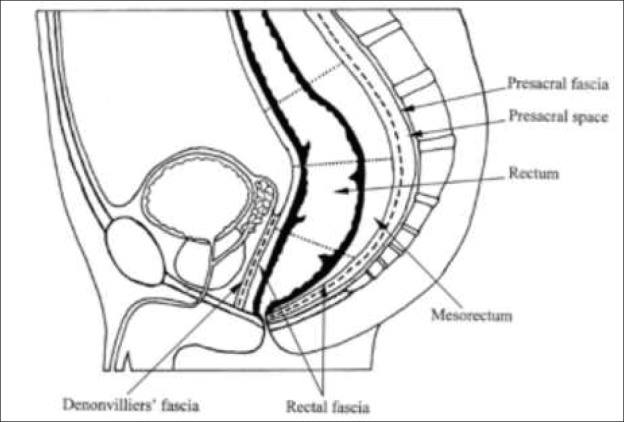
TME, described by R.J. Heald in 1982, is the oncologic standard for mid and low rectal cancer resection. The principle is sharp dissection under direct vision in the areolar tissue plane between the mesorectal fascia (visceral) and the presacral/pelvic sidewall fascia (parietal), excising the entire mesorectum as an intact, undamaged envelope. TME reduced local recurrence rates from 30–40% (conventional blunt dissection era) to <5%. Quality of TME is graded by the pathologist: complete (intact mesorectal surface, no defects deeper than 5 mm), near-complete (minor irregularities), or incomplete (defects to muscularis propria, visible tumor). PMID: 3319687
Surgical Options for Rectal Cancer
| Procedure | Indication | Key Details |
|---|---|---|
| Low anterior resection (LAR) | Mid/upper rectal cancer with adequate distal margin (≥1–2 cm) | TME + colorectal or coloanal anastomosis; often with diverting loop ileostomy (reversed at 8–12 weeks after leak check) |
| Abdominoperineal resection (APR) | Very low rectal/anal canal cancer where sphincter preservation is not possible | Combined abdominal and perineal approach; permanent end colostomy; includes excision of the entire anal canal and sphincter complex |
| Intersphincteric resection (ISR) | Very low rectal cancer (within 1–2 cm of dentate line) in selected patients | Dissection in the intersphincteric plane; preserves EAS; coloanal anastomosis (hand-sewn); avoids permanent stoma |
| Hartmann procedure | Emergency (obstruction, perforation) or frail patients | Sigmoid/rectal resection with end colostomy and rectal stump closure; no anastomosis |
| Local excision (TEM/TAMIS) | Selected T1 tumors (sm1, well-differentiated, no LVI, <3 cm, <30% circumference) | Full-thickness excision with clear margins; close surveillance required; if adverse features on pathology, completion radical resection recommended |
Neoadjuvant Therapy for Rectal Cancer
Neoadjuvant (preoperative) therapy is standard for locally advanced rectal cancer (T3/T4 or node-positive on MRI). Benefits: tumor downsizing/downstaging, increased R0 resection rate, improved sphincter preservation, and assessment of tumor response.
German Rectal Cancer Trial (2004): Established that preoperative chemoradiation (CRT) is superior to postoperative CRT — lower local recurrence (6% vs 13%), better compliance, fewer toxicities, higher sphincter preservation rates. Standard regimen: long-course CRT (50.4 Gy in 28 fractions + concurrent capecitabine or 5-FU), followed by surgery 6–8 weeks later, then adjuvant chemotherapy. PMID: 15496622
Total Neoadjuvant Therapy (TNT): The paradigm has shifted to delivering ALL systemic chemotherapy before surgery, rather than splitting it pre- and postoperative. This improves compliance (adjuvant chemo completion rates are only 50–60% after pelvic surgery) and increases pathologic complete response (pCR) rates.
RAPIDO trial (2021): Short-course radiation (5 x 5 Gy) → 6 cycles CAPOX or 9 cycles FOLFOX4 → surgery. Significantly improved disease-related treatment failure (23.7% vs 30.4%) and pCR rate (28% vs 14%). PMID: 33169015
PRODIGE 23 (2022): mFOLFIRINOX → CRT → surgery → adjuvant chemo vs standard CRT → surgery → adjuvant FOLFOX. TNT arm showed improved 3-year DFS (76% vs 69%) and pCR rate (28% vs 12%). PMID: 35660797
Organ Preservation — Watch and Wait
The OPRA trial (2022) randomized patients with locally advanced rectal cancer achieving a clinical complete response (cCR) after TNT to either watch-and-wait or TME surgery. At 3 years, the organ preservation rate was 53% in the watch-and-wait group (induction CRT → consolidation chemo arm). Patients who achieved and maintained a cCR had excellent oncologic outcomes without surgery. The watch-and-wait approach requires rigorous surveillance: DRE, proctoscopy, and rectal MRI every 3–4 months for 2 years, then every 6 months. Any regrowth is treated with salvage surgery (TME), which is curative in the vast majority. PMID: 36302838
10 Metastatic Colorectal Cancer — Liver & Lung
Colorectal Liver Metastases (CRLM)
The liver is the most common site of CRC metastasis (portal venous drainage). Approximately 15–25% of patients have synchronous liver metastases at diagnosis, and 50% will develop liver metastases during their disease course. Without treatment, median survival is 6–12 months. With modern multimodal therapy (chemotherapy + surgery), 5-year survival after complete hepatic resection is 40–58%.
Criteria for resectability: The traditional criteria (fewer than 4 metastases, no bilobar disease, size <5 cm) have been abandoned. The modern approach defines resectability based on: (1) ability to achieve R0 resection of all disease, (2) preservation of adequate future liver remnant (FLR) — typically >20% for normal liver, >30% with chemotherapy-related injury, >40% for cirrhosis, and (3) adequate hepatic inflow, outflow, and biliary drainage of the FLR. Strategies to increase FLR include portal vein embolization (PVE), associating liver partition and portal vein ligation for staged hepatectomy (ALPPS), and two-stage hepatectomy.
Timing of liver resection: For synchronous CRLM, three strategies exist: (1) simultaneous resection (colon + liver in one operation — selected patients), (2) classical approach (colon first → chemotherapy → liver resection), or (3) liver-first approach (chemotherapy → liver resection → colon resection — preferred when liver disease is the dominant threat and primary tumor is asymptomatic).
Lung Metastases
The lung is the second most common site of CRC metastasis and the most common site of extrahepatic metastasis. Pulmonary metastasectomy is offered to selected patients with: limited number of metastases, technically resectable disease, controlled primary tumor, and no unresectable extrapulmonary disease. Five-year survival after complete pulmonary metastasectomy is 30–50%. The PulMiCC trial (2020) questioned the benefit of pulmonary metastasectomy in a randomized setting, though methodological limitations tempered the conclusion. PMID: 31860952
Peritoneal Metastases
Peritoneal metastases (M1c) are present in 5–15% of CRC at diagnosis. Cytoreductive surgery (CRS) combined with hyperthermic intraperitoneal chemotherapy (HIPEC) is an option for selected patients with limited peritoneal disease (Peritoneal Cancer Index, PCI ≤20) and no extraperitoneal metastases. The PRODIGE 7 trial (2021) showed that adding HIPEC (oxaliplatin) to CRS did not improve OS compared to CRS alone, though the role of mitomycin-C-based HIPEC remains debated. PMID: 33524312
11 Crohn's Disease — Surgical Management
Approximately 70–80% of Crohn's disease (CD) patients will require at least one surgical procedure during their lifetime, though surgery is not curative — postoperative recurrence occurs in 50–60% within 10 years. The guiding principle is bowel conservation: resect only what is necessary, preserve as much bowel length as possible to prevent short bowel syndrome.
Indications for Surgery
Emergency: Free perforation, massive hemorrhage (rare), toxic megacolon/fulminant colitis unresponsive to medical therapy (72 hours), complete bowel obstruction.
Elective: Symptomatic stricture refractory to medical/endoscopic therapy, fistula (enterocutaneous, enterovesical, enterovaginal), abscess (percutaneous drainage first, then elective resection of diseased segment), failure of medical therapy (steroid dependence/refractory), dysplasia/cancer in Crohn's colitis, growth retardation in children.
Strictureplasty
Strictureplasty is a bowel-sparing technique that widens a strictured segment without resection. Ideal for multiple short strictures in the small bowel (especially in patients with prior resections or limited remaining bowel). Contraindications: active perforation at stricture site, phlegmon/abscess, internal fistula at stricture, dysplasia/malignancy, colonic strictures (higher cancer risk), and hypoalbuminemia (<2 g/dL).
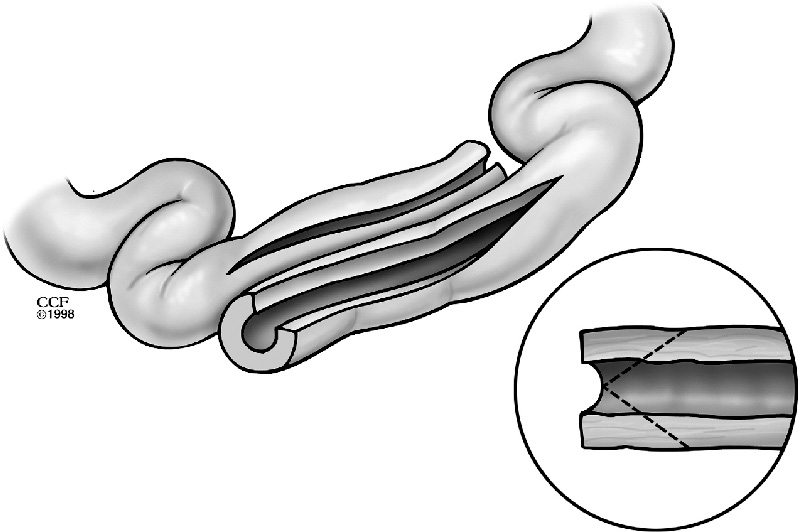
| Type | Stricture Length | Technique |
|---|---|---|
| Heineke-Mikulicz | <10 cm (short) | Longitudinal enterotomy across stricture, transverse closure (analogous to pyloroplasty) |
| Finney | 10–20 cm (intermediate) | U-shaped fold; side-to-side anastomosis |
| Michelassi (side-to-side isoperistaltic) | >20 cm (long) | The strictured segment is folded on itself and a long side-to-side anastomosis is created |
Ileocolic Resection
The most common operation for Crohn's disease is ileocolic resection (resection of the terminal ileum and cecum with ileocolic anastomosis), because the terminal ileum is the most commonly affected site. A wide (functional end-to-end, stapled side-to-side) anastomosis is preferred. Margins do NOT need to be microscopically free — resecting grossly normal bowel is sufficient. The Kono-S anastomosis (antimesenteric functional end-to-end) has shown lower endoscopic and surgical recurrence rates in some studies.
Crohn's Colitis
Options: segmental colectomy (for limited colonic disease with rectal sparing), subtotal colectomy with ileorectal anastomosis (diffuse colitis with rectal sparing — requires surveillance of retained rectum), or total proctocolectomy with end ileostomy (severe perianal disease, rectal involvement, or failed prior surgery). IPAA (J-pouch) is generally not recommended in Crohn's disease due to high pouch failure rates (up to 30–50%), though selected patients with isolated Crohn's colitis (no small bowel disease, no perianal disease) may be considered.
Perianal Crohn's Disease
Perianal fistulae affect 20–30% of CD patients. Management follows a stepwise algorithm: (1) EUA (examination under anesthesia) with abscess drainage and non-cutting seton placement — the seton maintains drainage and prevents abscess recurrence; (2) MRI pelvis to map all fistula tracts and identify occult collections; (3) combination medical therapy — anti-TNF (infliximab is the best studied agent for perianal Crohn's fistulae; the ACCENT II trial demonstrated that infliximab maintenance produces fistula closure in 36% at 54 weeks), antibiotics (ciprofloxacin 500 mg BID + metronidazole 500 mg TID for 8–12 weeks), and immunomodulators (azathioprine or 6-MP for maintenance); (4) definitive fistula repair (advancement flap, LIFT, or mesenchymal stem cell injection — the ADMIRE-CD trial showed darvadstrocel achieved combined remission in 50% of complex perianal Crohn's fistulae) only after optimizing medical therapy and ensuring abscess resolution. (5) Proctectomy with permanent end ileostomy is reserved for severe, refractory perianal disease with a destroyed anal canal, rectal stricture, or rectal cancer. PMID: 15175439
Postoperative Recurrence Prevention
Endoscopic recurrence after ileocolic resection is detected by colonoscopy at 6–12 months using the Rutgeerts score: i0 (no lesions), i1 (≤5 aphthous ulcers in neo-terminal ileum), i2 (>5 aphthous ulcers or larger lesions confined to ileocolonic anastomosis), i3 (diffuse aphthous ileitis), i4 (diffuse ileitis with large ulcers, nodules, or stenosis). Scores i2–i4 predict clinical recurrence and warrant medical prophylaxis. Current first-line prophylaxis after high-risk surgery: anti-TNF therapy (infliximab or adalimumab) started within 4 weeks of surgery. Lower-risk patients may be managed with thiopurines or nitroimidazole antibiotics (metronidazole 250 mg TID for 3 months). Smoking cessation is the single most important modifiable risk factor for postoperative recurrence.
12 Ulcerative Colitis — IPAA & J-Pouch
Unlike Crohn's disease, UC is surgically curable. Approximately 15–30% of UC patients ultimately require colectomy.
Indications for Surgery
Emergency: Fulminant colitis/toxic megacolon unresponsive to IV steroids + rescue therapy (infliximab or cyclosporine) within 3–5 days; perforation; massive hemorrhage. Elective: Refractory to maximal medical therapy (including biologics); steroid dependence; dysplasia (high-grade dysplasia, low-grade dysplasia if flat/multifocal, or associated with a mass); colorectal cancer; intolerable medication side effects; growth retardation in children.
Operative Strategy — Staged Approach
Emergency (3-stage): Stage 1 — subtotal colectomy with end ileostomy and Hartmann's closure of the rectal stump (sick patient, on steroids/biologics — minimize operative time and avoid pelvic dissection); Stage 2 — completion proctectomy with IPAA construction and diverting loop ileostomy (after patient recovers and steroids are weaned, typically 3–6 months); Stage 3 — loop ileostomy reversal (8–12 weeks after Stage 2, after confirming pouch integrity with contrast study).
Elective (2-stage — most common): Stage 1 — total proctocolectomy with IPAA and diverting loop ileostomy; Stage 2 — ileostomy reversal.
Elective (1-stage): Total proctocolectomy with IPAA, no diverting ileostomy. Selected low-risk patients (good nutrition, no steroids/biologics, tension-free pouch). Leak rate is slightly higher (5–8% vs 2–4% with diversion).
IPAA (J-Pouch) Construction
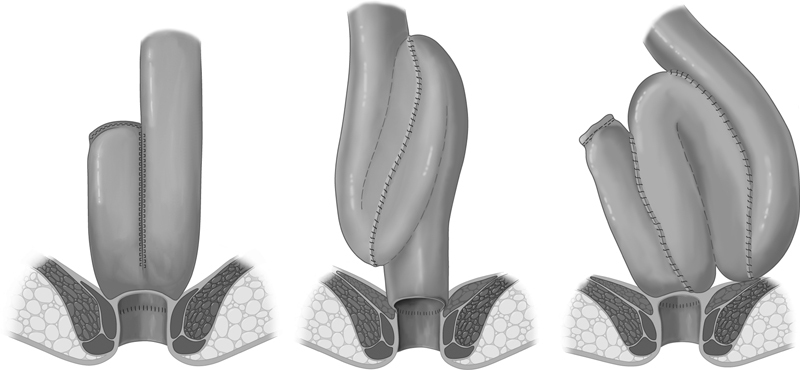
The ileal pouch-anal anastomosis (IPAA), also known as the J-pouch, is the gold standard restorative procedure for UC. The terminal ileum (15–20 cm limb length) is folded into a J-shape and stapled to create a reservoir. The pouch is anastomosed to the anal canal — either a stapled anastomosis (at the top of the anal canal, 1–2 cm above the dentate line, preserving the anal transition zone — better continence) or a mucosectomy with hand-sewn anastomosis (stripping the anal canal mucosa to the dentate line and suturing the pouch directly — used for dysplasia/cancer at the rectal cuff or FAP). The J-pouch configuration is preferred over S-pouch or W-pouch due to simplicity; all configurations have equivalent long-term functional outcomes.
Technical considerations: The pouch must reach the pelvic floor without tension — this usually requires full mobilization of the small bowel mesentery by dividing the peritoneum along the base of the mesentery up to the level of the duodenum (lengthening maneuvers). The ileocolic artery is preserved; the superior mesenteric artery is the vascular pedicle. If the pouch does not reach, additional length can be gained by selective division of mesenteric branches (preserving the arcade), scoring the peritoneum overlying the SMA, or (as a last resort) dividing the ileocolic artery.
Functional outcomes: After maturation (6–12 months), patients have an average of 5–8 bowel movements per day, approximately 1 at night. Continence is good in 85–95% of patients. Quality of life is generally excellent and comparable to the general population. Factors associated with worse functional outcomes: older age (>50), low hand-sewn anastomosis (mucosectomy), preoperative poor sphincter function, and chronic pouchitis.
Long-Term Outcomes & Complications
| Complication | Incidence | Management |
|---|---|---|
| Pouchitis | 30–50% at some point | Metronidazole or ciprofloxacin; chronic pouchitis → VSL#3 probiotics, budesonide, immunomodulators |
| Anastomotic stricture | 5–15% | Digital or endoscopic balloon dilation |
| Pouch fistula | 5–10% | Seton, advancement flap; consider Crohn's reclassification |
| Small bowel obstruction | 15–25% | Conservative initially; operative if complete or recurrent |
| Pelvic sepsis / leak | 5–10% | Drainage (percutaneous or transanal); may require diversion |
| Female infertility | 3x increased vs medical therapy | Laparoscopic approach may reduce adhesions; discuss fertility preservation preoperatively |
| Pouch failure (requiring excision/permanent ileostomy) | 5–10% at 10 years | Usually from chronic pouchitis, fistula, or reclassification as Crohn's |
13 Pouchitis & Pouch Complications
Pouchitis
Pouchitis is the most common long-term complication of IPAA, affecting 30–50% of patients within 5 years. It is non-specific inflammation of the ileal pouch reservoir. Symptoms: increased stool frequency, urgency, watery stool, cramping, pelvic discomfort, and occasionally hematochezia and fever. Diagnosis is based on the Pouchitis Disease Activity Index (PDAI), which combines symptoms, endoscopic findings (edema, granularity, friability, ulceration), and histologic features (polymorphonuclear infiltration, ulceration). A PDAI score of 7 or more defines pouchitis.
Treatment: First-line — metronidazole 500 mg TID or ciprofloxacin 500 mg BID for 14 days. Most patients respond. Chronic/refractory pouchitis (occurring in 5–15%): combination antibiotics (cipro + metronidazole or cipro + rifaximin), oral budesonide 9 mg daily, probiotics (VSL#3 — 3.6 x 1011 CFU/day shown to maintain remission), topical mesalamine suppositories, immunomodulators (azathioprine), and biologics (vedolizumab, infliximab) for the most refractory cases.
Crohn's Disease of the Pouch
Approximately 5–10% of patients who undergo IPAA for presumed UC are later reclassified as Crohn's disease when they develop pouch fistulae, strictures, or perianal disease. Management is medical (anti-TNF agents, immunomodulators) in most cases. Pouch excision is reserved for refractory disease.
14 Diverticulitis — Classification & Management
Diverticular disease is the most common condition of the colon in Western populations, affecting up to 60% of adults over 60. Diverticulosis is the presence of diverticula (false diverticula — outpouchings of mucosa and submucosa through the colonic wall at points of vasa recta penetration). Diverticulitis is inflammation/infection of a diverticulum, occurring in 10–25% of those with diverticulosis.
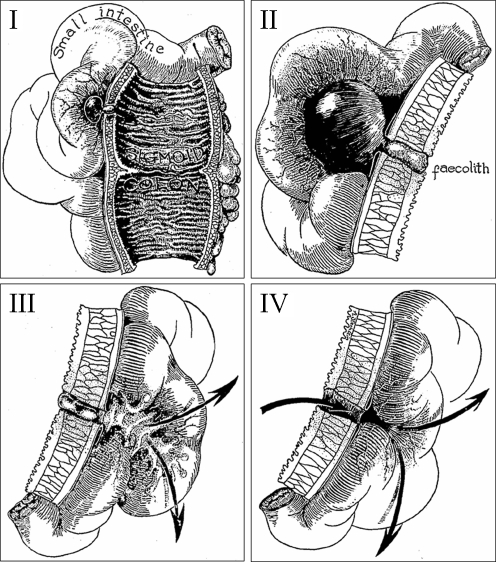
Hinchey Classification (Modified)
| Stage | Description | Management |
|---|---|---|
| 0 | Mild clinical diverticulitis (CT: colonic wall thickening, pericolic fat stranding) | Outpatient antibiotics (or observation alone — DIABOLO trial) |
| Ia | Confined pericolic phlegmon/inflammation | IV antibiotics |
| Ib | Pericolic abscess (<4 cm) | IV antibiotics; percutaneous drainage if no response |
| II | Pelvic, distant, or retroperitoneal abscess (>3–4 cm) | Percutaneous drainage + IV antibiotics; interval sigmoid colectomy 6–8 weeks later |
| III | Generalized purulent peritonitis (perforated diverticulum with purulent contamination) | Emergency surgery (see section 15) |
| IV | Generalized fecal peritonitis (free fecal contamination) | Emergency surgery — highest mortality (up to 35%) |
Uncomplicated Diverticulitis
Uncomplicated diverticulitis (Hinchey 0–Ia) is the most common presentation. Traditional management included NPO, IV antibiotics, and hospitalization. More recent evidence has shifted management: the DIABOLO trial (2017) demonstrated that observation without antibiotics is non-inferior to antibiotics for uncomplicated diverticulitis — no difference in time to recovery, complications, or recurrence at 24 months. Outpatient management with a clear liquid diet and oral antibiotics (if used: ciprofloxacin + metronidazole or amoxicillin-clavulanate for 7–10 days) is appropriate for immunocompetent patients without significant comorbidities. PMID: 28586827
When to Operate Electively
The traditional "2-strike rule" (operate after the second episode of uncomplicated diverticulitis) has been abandoned. Current guidelines (AGA 2015, ASCRS 2020) recommend individualized decision-making based on the severity of the episodes, persistent symptoms, quality of life impact, and patient comorbidities — not simply the number of episodes. Elective sigmoid colectomy is recommended for: complicated diverticulitis (Hinchey II or higher, even after successful initial non-operative management), fistula (colovesical, colovaginal), stricture causing obstruction, and inability to exclude malignancy.
15 Emergency Surgery for Complicated Diverticulitis
Hartmann Procedure vs Primary Anastomosis
For decades, the Hartmann procedure (sigmoid resection, end colostomy, rectal stump closure) was the default emergency operation for Hinchey III/IV diverticulitis. The alternative is sigmoid resection with primary anastomosis (PA) and diverting loop ileostomy. Multiple RCTs have compared the two approaches:
LADIES trial — DIVA arm (2019): For Hinchey III/IV perforated diverticulitis, PA with diverting ileostomy resulted in a significantly higher stoma reversal rate (94% vs 57%) and lower overall morbidity at 12 months. No significant difference in mortality or serious complications. PMID: 31402817
SCANDIV trial (2021): Hartmann vs PA with ileostomy for Hinchey III/IV. No significant difference in 90-day mortality or morbidity. Stoma reversal rate was higher with PA (73% vs 46%). PMID: 34042961
Current consensus: PA with diverting loop ileostomy is preferred over Hartmann in hemodynamically stable patients with Hinchey III disease operated by experienced surgeons. Hartmann procedure remains appropriate for hemodynamically unstable patients, Hinchey IV (fecal peritonitis), severe comorbidities, or surgeons with limited colorectal experience.
Laparoscopic Lavage
Laparoscopic peritoneal lavage (without resection) was investigated for Hinchey III diverticulitis in the LOLA arm of the LADIES trial, the SCANDIV trial, and the DILALA trial. Results were mixed — lavage had a lower stoma rate but a higher reoperation rate and failure rate compared to resection. It is not recommended as standard practice and has largely fallen out of favor.
Damage Control Surgery
In the severely septic, hemodynamically unstable patient (Hinchey IV), damage control principles apply: limited resection of the perforated segment, abdominal washout, temporary abdominal closure (negative pressure wound therapy), and ICU resuscitation. Return to the OR in 24–48 hours for definitive management (stoma creation or anastomosis) once the patient is stabilized.
16 Diverticular Bleeding
Diverticular bleeding is the most common cause of lower GI hemorrhage (30–40% of cases). It occurs when a vasa rectum erodes into the dome of a diverticulum. Bleeding is typically painless, acute, and self-limited (stops spontaneously in 70–80%). The right colon is the most common source despite diverticulosis being more prevalent on the left, because right-sided diverticula have wider necks exposing the vasa recta.
Management Algorithm
Resuscitation: Large-bore IV access, type and crossmatch, transfuse for hemoglobin <7 g/dL (or <9 g/dL if hemodynamically unstable or CAD). Correct coagulopathy. Hold anticoagulants/antiplatelets.
Localization: (1) Colonoscopy — after rapid bowel prep, colonoscopy can identify and treat the bleeding source (epinephrine injection, hemoclips, thermal coagulation). (2) CT angiography — for ongoing brisk bleeding (detects bleeding rates >0.3–0.5 mL/min); identifies the bleeding vessel. (3) Mesenteric angiography — for ongoing active bleeding (>0.5–1 mL/min), allows embolization (microcoils, gelfoam) of the bleeding vessel (90% success rate; risk of bowel ischemia 0–20%). (4) Tagged red blood cell scan — most sensitive for slow bleeding (>0.1 mL/min); localizes to a region but not a specific vessel.
Surgery: Reserved for massive hemorrhage refractory to endoscopic and angiographic intervention, or recurrent bleeding. If the bleeding source is localized, segmental colectomy is performed. If the source is unlocalized despite all modalities, subtotal colectomy with ileorectal anastomosis is necessary — rebleeding rate after blind segmental colectomy is unacceptably high (up to 30%).
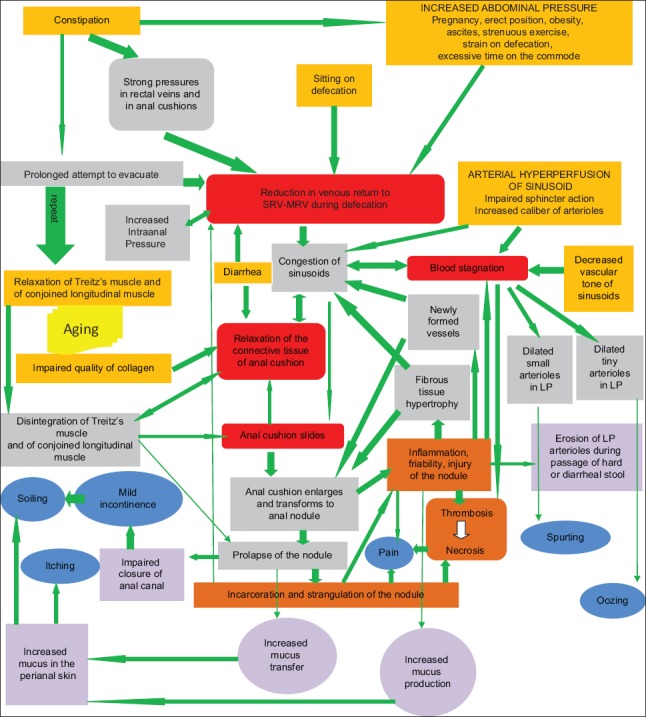
17 Hemorrhoids
Hemorrhoids are normal vascular cushions in the anal canal (left lateral, right anterolateral, right posterolateral positions) that contribute to fine continence. Hemorrhoidal disease occurs when these cushions become symptomatic — engorged, prolapsed, thrombosed, or bleeding.
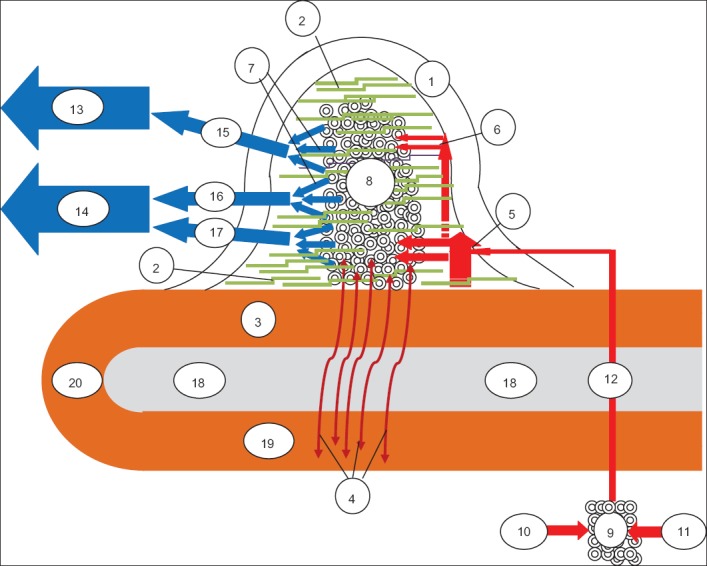
Classification
| Type | Origin | Covering | Innervation | Symptoms |
|---|---|---|---|---|
| Internal | Above the dentate line | Columnar mucosa | Visceral (painless) | Painless bleeding (bright red, on toilet paper/in bowl), prolapse, mucus discharge, pruritus |
| External | Below the dentate line | Anoderm (squamous) | Somatic (painful) | Pain (especially if thrombosed), swelling, skin tags, difficult hygiene |
Grading of Internal Hemorrhoids
| Grade | Description | Treatment |
|---|---|---|
| I | Bleed but do not prolapse | Conservative (fiber, sitz baths); rubber band ligation (RBL) if persistent bleeding |
| II | Prolapse with straining but reduce spontaneously | RBL (most effective office treatment); sclerotherapy; infrared coagulation |
| III | Prolapse requiring manual reduction | RBL (may need multiple sessions); excisional hemorrhoidectomy if RBL fails |
| IV | Irreducible prolapse (incarcerated) | Excisional hemorrhoidectomy; stapled hemorrhoidopexy |
Rubber Band Ligation (RBL)
The most effective and widely used office procedure for grade I–III internal hemorrhoids. A rubber band is applied to the base of the hemorrhoid 1–2 cm above the dentate line using a suction ligator. The band strangulates the hemorrhoidal tissue, which sloughs in 5–7 days. One hemorrhoid is banded per session; repeat at 4–6 week intervals. Success rate: 70–80%. Complications: pain (if band placed too close to the dentate line), bleeding (delayed, at 7–10 days when tissue sloughs), and rarely pelvic sepsis (life-threatening — fever, urinary retention, and severe pain after RBL requires urgent evaluation).
Excisional Hemorrhoidectomy
Milligan-Morgan (open) — excision of the hemorrhoidal tissue with the wound left open to heal by secondary intention. Most widely used worldwide. Ferguson (closed) — excision with primary closure of the wound. Faster healing, slightly more postoperative pain. Both approaches have equivalent long-term outcomes. The mucosal bridges between excision sites must be preserved to prevent anal stenosis.
Transanal hemorrhoidal dearterialization (THD/HAL-RAR) — Doppler-guided ligation of the hemorrhoidal arteries above the dentate line combined with mucopexy (recto-anal repair). Less postoperative pain than excisional hemorrhoidectomy; suitable for grade II–III hemorrhoids. Recurrence rates are similar to stapled hemorrhoidopexy.
Stapled hemorrhoidopexy (PPH — Procedure for Prolapse and Hemorrhoids) — a circumferential stapled mucosectomy above the dentate line using a circular stapler. This excises a ring of rectal mucosa and hemorrhoidal tissue and lifts the prolapsed hemorrhoids back into position. Less postoperative pain than conventional hemorrhoidectomy but higher recurrence rate. Risk of serious complications: staple line bleeding, rectovaginal fistula, rectal perforation, and chronic pelvic pain (if staple line incorporates rectal wall muscle).
Acute Thrombosed External Hemorrhoid
Sudden onset of severe perianal pain with a visible, tense, blue/purple subcutaneous nodule at the anal verge. If presenting within 48–72 hours of onset, excision (not just incision and drainage — simple I&D leads to a 50% recurrence rate because the clot re-forms) of the thrombosed hemorrhoid under local anesthesia provides immediate relief and prevents recurrence. An elliptical incision removes the entire thrombosed vein and overlying skin. After 72 hours, symptoms are typically improving and conservative management (sitz baths, analgesics, stool softeners, topical nitroglycerin) is appropriate. The thrombosis typically resolves over 2–4 weeks; the residual fibrotic skin tag can be excised electively if bothersome.
Hemorrhoidal Disease in Special Populations
Pregnancy: Hemorrhoids are common in the third trimester and postpartum due to increased pelvic venous pressure and constipation. Management is conservative (fiber, sitz baths, topical agents). Avoid surgical intervention during pregnancy unless there is acute incarceration with necrosis. Cirrhosis/portal hypertension: Anorectal varices (not true hemorrhoids) may occur. These require management of the underlying portal hypertension (TIPS, beta-blockers) rather than standard hemorrhoid treatments. Distinguish from hemorrhoids by their location (rectal varices are above the dentate line, not in the typical hemorrhoidal cushion positions) and their bluish, compressible appearance on anoscopy. Banding or surgery on anorectal varices risks massive hemorrhage. Immunosuppression: Exercise caution with any anorectal procedure in immunosuppressed patients (transplant recipients, HIV/AIDS with low CD4, active chemotherapy) — risk of severe sepsis and impaired healing.
18 Anal Fissure
An anal fissure is a longitudinal tear in the anoderm of the anal canal, typically in the posterior midline (90% of cases; 10% anterior — more common in women). The classic triad is pain during and after defecation (can last hours), bright red blood on toilet paper, and a visible tear at the anal verge.
Pathophysiology
Internal anal sphincter (IAS) hypertonia causes reduced blood flow to the posterior commissure of the anal canal (the posterior midline has the least blood supply — watershed zone). Ischemia impairs wound healing, creating a vicious cycle: fissure → pain → IAS spasm → ischemia → non-healing fissure.
Acute vs Chronic
Acute fissure (<6–8 weeks): fresh tear with clean edges. Chronic fissure (>8 weeks): visible white internal sphincter fibers at the base, sentinel pile (external skin tag at the distal end), hypertrophied anal papilla (at the proximal end), and thickened edges. Fissures NOT in the posterior or anterior midline ("lateral fissures") should raise suspicion for Crohn's disease, HIV, syphilis, tuberculosis, or anal cancer — biopsy is indicated.
Medical Management
First-line (effective in 50–70% of acute fissures): fiber supplementation (25–35 g/day), adequate hydration, stool softeners (docusate), sitz baths (warm water, 10–15 minutes TID), and avoidance of straining. Topical nitroglycerin 0.2–0.4% (applied to the anal margin BID–TID for 6–8 weeks) — acts as a nitric oxide donor, relaxing the IAS and increasing local blood flow. Main side effect: headache (20–30%). Alternative: topical diltiazem 2% (calcium channel blocker) — equivalent efficacy, fewer headaches. Botulinum toxin A injection (20–100 units into the IAS) — temporary IAS relaxation lasting 2–3 months; healing rate 60–80%; may cause transient incontinence.
Lateral Internal Sphincterotomy (LIS)
LIS is the gold standard surgical treatment for chronic anal fissure refractory to medical therapy (8–12 weeks). A partial (tailored) division of the IAS is performed through a small lateral incision. The sphincter is divided from its distal edge up to the level of the fissure apex (NOT the full length of the IAS). Healing rate: >95%. Risk of minor incontinence to flatus: 5–8%; frank fecal incontinence: <1%. LIS should be avoided or used with extreme caution in patients with pre-existing incontinence, prior anorectal surgery, obstetric injury, or Crohn's disease.
19 Anorectal Abscess & Fistula-in-Ano
Anorectal Abscess
Most anorectal abscesses originate from infection of an anal crypt gland (cryptoglandular theory). The infected gland drains into the intersphincteric space and can then extend to other spaces. Treatment is incision and drainage — antibiotics alone are insufficient. "The sun should never set on an undrained anorectal abscess."
| Abscess Type | Frequency | Drainage Approach |
|---|---|---|
| Perianal | 60% | Cruciate incision close to the anal verge; bedside/office under local anesthesia |
| Ischiorectal | 20% | Incision over the point of maximal fluctuance; may require OR for adequate drainage |
| Intersphincteric | 15% | Internal drainage via anoscopy into the anal canal |
| Supralevator | 5% | Route depends on origin — if intersphincteric origin, drain internally; if ischiorectal origin, drain through the ischiorectal fossa. NEVER drain through the levator (creates suprasphincteric fistula) |
| Horseshoe | Rare | Drain deep postanal space (Hanley procedure) + counter incisions in bilateral ischiorectal fossae |
Fistula-in-Ano
Approximately 30–50% of anorectal abscesses develop a fistula. A fistula is an abnormal tract connecting the anal canal (internal opening) to the perianal skin (external opening). The internal opening is almost always at the dentate line (at the infected crypt).
Goodsall's Rule
Goodsall's rule predicts the location of the internal opening based on the external opening: if the external opening is anterior to the transverse anal line, the fistula tract runs radially to the nearest crypt (straight tract). If the external opening is posterior to the transverse anal line, the tract curves posteriorly to the posterior midline (internal opening in the posterior commissure). Exception: external openings >3 cm from the anal verge anteriorly may still curve posteriorly.
Parks Classification of Fistulae
| Type | Frequency | Course | Treatment |
|---|---|---|---|
| Intersphincteric | 45% | Through IAS, along intersphincteric space, exits at anal verge | Fistulotomy (laying open) |
| Trans-sphincteric | 30% | Through both IAS and EAS into the ischiorectal fossa | Low: fistulotomy; High (involving >30% EAS): seton, LIFT, advancement flap |
| Suprasphincteric | 20% | Up through intersphincteric space, over puborectalis, down through ischiorectal fossa | Seton; advancement flap; NEVER fistulotomy (would divide the entire sphincter) |
| Extrasphincteric | 5% | From rectum above the sphincters, through the levator to the perineum | Treat the underlying cause (Crohn's, diverticulitis, pelvic abscess); usually requires fecal diversion |
Surgical Techniques for Fistula
Fistulotomy — laying open the entire fistula tract. Gold standard for simple intersphincteric and low trans-sphincteric fistulae involving <30% of the EAS. Healing rate: 92–97%. Risk of incontinence increases with the amount of sphincter divided.
Seton placement — a vessel loop or silk suture threaded through the fistula tract. Draining/loose seton: maintains drainage, prevents abscess recurrence, allows inflammation to subside — used as a bridge to definitive repair. Cutting seton: slowly tightened to cut through the sphincter while fibrosis occurs above — rarely used today due to unpredictable incontinence.
LIFT (Ligation of the Intersphincteric Fistula Tract) — the fistula tract is identified in the intersphincteric space, ligated, and divided. Success rate: 57–94%. Preserves sphincter integrity. Best for trans-sphincteric fistulae.
Endorectal advancement flap — a flap of rectal mucosa, submucosa, and a portion of the internal sphincter is advanced to cover the internal opening. Success rate: 50–80%. Used for high trans-sphincteric and suprasphincteric fistulae.
Other options: fibrin glue (limited success, 30–50%), bioprosthetic plug (variable results, 20–55%), VAAFT (video-assisted anal fistula treatment), FiLaC (fistula laser closure).
(1) Always identify the internal opening — this is the source; failure to address the internal opening guarantees recurrence. Hydrogen peroxide injection through the external opening during EUA can help identify the internal opening. (2) Determine how much sphincter the tract traverses before choosing a technique — sphincter-dividing procedures (fistulotomy) are only safe for low fistulae involving <30% of the EAS. (3) In patients with Crohn's disease, anterior fistulae in women, and patients with pre-existing incontinence, use sphincter-sparing techniques (LIFT, advancement flap, seton). (4) MRI pelvis is the gold standard for mapping complex fistulae — sensitivity >90% for identifying tracts and secondary extensions that may be missed on EUA alone. Combine MRI with EUA for the best outcomes in complex disease.
20 Pilonidal Disease
Pilonidal disease affects the natal cleft (gluteal crease) and is caused by penetration of shed hairs into the subcutaneous tissue, creating a foreign body reaction, chronic sinus tracts, and recurrent abscesses. Most common in young males (15–30 years), with risk factors including hirsutism, obesity, prolonged sitting, and deep natal cleft.
Acute Pilonidal Abscess
Treatment: incision and drainage lateral to the midline (off-midline incision heals faster and recurs less than midline incisions). Antibiotics are generally not needed unless there is surrounding cellulitis.
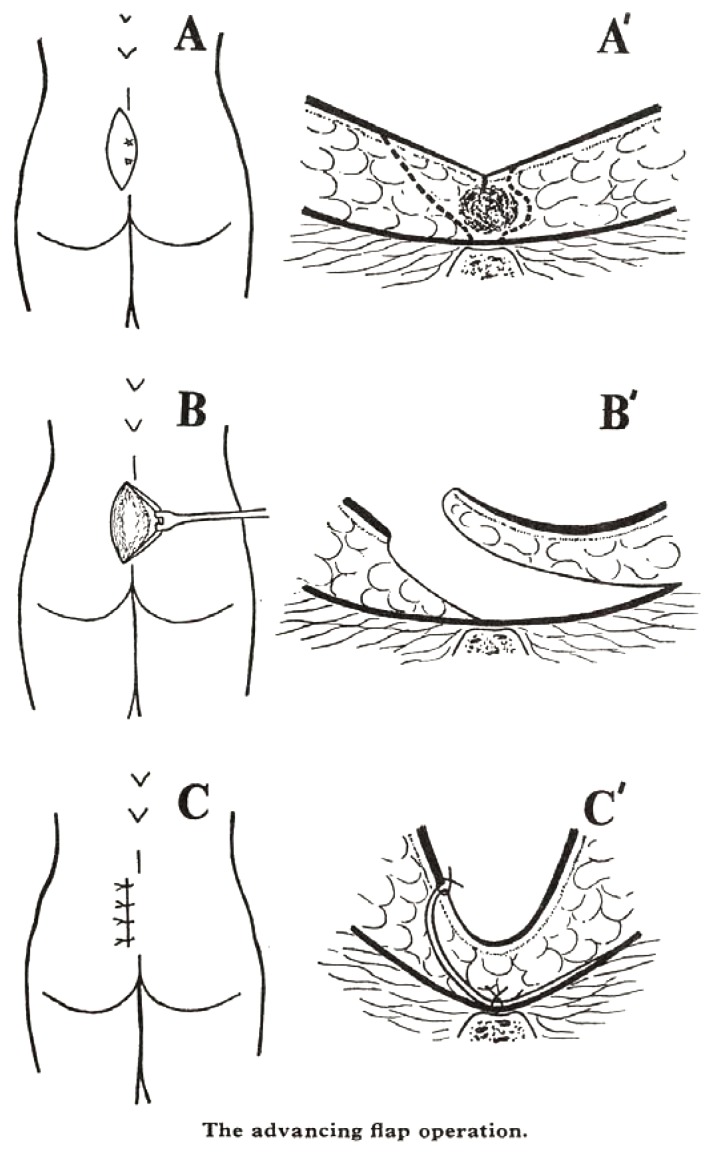
Chronic/Recurrent Pilonidal Disease
| Procedure | Technique | Recurrence | Key Points |
|---|---|---|---|
| Excision with open healing | Wide excision; heal by secondary intention | 5–15% | Simple; prolonged healing (6–12 weeks); good for small defects |
| Excision with primary midline closure | Excision and suture in midline | 10–30% | Fast healing but highest recurrence; NOT recommended |
| Off-midline closure (Karydakis/Bascom) | Excision with asymmetric closure, moving the wound away from the midline | 1–5% | Preferred technique; flattens the natal cleft |
| Limberg (rhomboid) flap | Rhomboid excision with rotation flap | 1–5% | For large/recurrent disease; excellent outcomes |
| Cleft lift (Bascom II) | Excision with Z-plasty to flatten the natal cleft | 1–3% | Best for complex/recurrent disease |
21 Anal Cancer
Anal canal cancer is uncommon (2–3% of GI malignancies) but increasing in incidence. The majority (85%) are squamous cell carcinomas (SCC), strongly associated with HPV infection (types 16 and 18). Risk factors: HPV, receptive anal intercourse, multiple sexual partners, HIV/immunosuppression, smoking, and history of other HPV-related cancers.
Anatomy-Based Definitions
Anal canal cancer extends from the anorectal ring to the anal verge. Anal margin cancer arises from the skin surrounding the anus (perianal skin, within 5 cm of the anal verge). Anal margin SCC behaves like skin cancer and is treated with wide local excision if T1–T2.
Staging (AJCC 8th Edition — Anal Canal)
| T Stage | Definition |
|---|---|
| T1 | Tumor ≤2 cm |
| T2 | Tumor >2 cm but ≤5 cm |
| T3 | Tumor >5 cm |
| T4 | Tumor invades adjacent organs (vagina, urethra, bladder; NOT sphincter invasion alone) |
The Nigro Protocol — Chemoradiation
The landmark contribution of Norman Nigro (1974) transformed anal cancer treatment from abdominoperineal resection (permanent colostomy) to organ-preserving chemoradiation (CRT). The Nigro protocol consists of concurrent radiation therapy (45–59 Gy) with 5-fluorouracil (1000 mg/m2/day continuous infusion, days 1–4 and 29–32) and mitomycin C (10–12 mg/m2 bolus, day 1). Complete response rate: 80–90%. Five-year overall survival: 70–80% for localized disease. PMID: 10735900
The ACT II trial (2013) confirmed that mitomycin C-based CRT is superior to cisplatin-based CRT and that maintenance chemotherapy after CRT provides no additional benefit. PMID: 23562698
APR for anal cancer is reserved for: persistent/recurrent disease after CRT (salvage APR — 5-year survival 40–60%), or rarely for T4 tumors with fecal incontinence at presentation. Immune checkpoint inhibitors (nivolumab, pembrolizumab) are used for metastatic anal SCC.
HPV Vaccination & Anal Dysplasia Screening
HPV vaccination (9-valent, Gardasil 9) prevents HPV 16/18 infection and reduces anal cancer risk. Screening for anal dysplasia (anal Pap smear → high-resolution anoscopy if abnormal) is recommended for high-risk populations (HIV-positive individuals, MSM, transplant recipients, women with cervical/vulvar dysplasia). Treatment of high-grade squamous intraepithelial lesion (HSIL) with ablation reduces progression to anal cancer — the ANCHOR trial (2022) demonstrated a 57% reduction in anal cancer with HSIL treatment in HIV-positive patients. PMID: 35704336
22 Rectal Prolapse
Rectal prolapse (procidentia) is a full-thickness intussusception of the rectal wall through the anus. Most common in elderly women (6:1 female-to-male ratio). Associated with chronic constipation/straining, pelvic floor weakness, pudendal neuropathy, and multiparous women.
Types
Full-thickness (complete) prolapse: all layers of the rectal wall protrude through the anus — visible concentric mucosal folds (distinguish from mucosal prolapse, which has radial folds). Mucosal prolapse: only the mucosa prolapses (radial folds) — associated with hemorrhoidal disease. Internal intussusception (occult prolapse): the rectum intussuscepts but does not exit the anal canal — seen on defecography; may cause obstructed defecation.
Surgical Approaches
| Approach | Procedure | Recurrence | Notes |
|---|---|---|---|
| Abdominal | Ventral mesh rectopexy (laparoscopic/robotic) | 3–5% | Preferred procedure in many centers; mesh placed on the anterior rectum and fixed to the sacral promontory; good functional outcomes; avoids posterior dissection (nerve-sparing) |
| Posterior suture rectopexy (Ripstein, Wells) | 3–8% | Posterior mobilization and fixation to the sacrum; mesh (Ripstein) or suture (Wells) rectopexy; higher constipation rates with posterior approaches | |
| Perineal | Altemeier (perineal rectosigmoidectomy) | 10–20% | Full-thickness excision of the prolapsed rectum via the perineum with coloanal anastomosis; often combined with levatoroplasty; best for frail/elderly patients unfit for abdominal surgery |
| Delorme (mucosal sleeve resection) | 15–30% | Mucosal stripping with plication of the denuded muscular wall; higher recurrence but minimal morbidity; for short-segment prolapse |
Preoperative Workup for Rectal Prolapse
A complete evaluation includes: colonoscopy (to exclude a lead point mass, especially in new-onset prolapse in older patients), anorectal manometry (to document baseline sphincter function — many prolapse patients have incontinence from chronic pudendal nerve stretch), and defecography (to identify concurrent enterocele, cystocele, or vaginal vault prolapse that may need simultaneous repair). In women with multicompartment pelvic floor prolapse, a combined approach with urogynecology should be considered.
Incarcerated Rectal Prolapse
An irreducible, edematous, or gangrenous rectal prolapse is a surgical emergency. Initial management includes application of granulated sugar or hypertonic saline to the prolapsed rectum to reduce edema (osmotic effect), followed by gentle manual reduction. If manual reduction fails or there is evidence of mucosal necrosis/gangrene, emergency perineal rectosigmoidectomy (Altemeier procedure) is performed. A gangrenous prolapse should NEVER be pushed back into the pelvis — this risks perforation and peritonitis.
23 Condylomata Acuminata & Fecal Incontinence
Condylomata Acuminata (Anal Warts)
Caused by HPV (types 6 and 11 are the most common — these are low-risk types causing warts, not cancer). Present as flesh-colored, papillomatous lesions in the perianal and intra-anal area. Treatment depends on extent: limited disease — topical therapy (imiquimod 5% cream, podofilox 0.5%, trichloroacetic acid 80–90%); extensive disease — surgical excision/fulguration under anesthesia. Always biopsy suspicious, atypical, or refractory lesions to exclude dysplasia or Buschke-Lowenstein tumor (verrucous carcinoma — giant condyloma). Recurrence rate is high (30–70%) because HPV persists in surrounding tissue.
Fecal Incontinence
Fecal incontinence affects 2–7% of the adult population and is underreported due to stigma. Causes include obstetric sphincter injury (most common in women), prior anorectal surgery (fistulotomy, hemorrhoidectomy, sphincterotomy), neurologic disorders, pelvic floor dysfunction, rectal prolapse, radiation proctitis, IBD, and aging.
Evaluation
History (onset, severity, stool consistency, obstetric history, prior anorectal surgery). Wexner (Cleveland Clinic) Incontinence Score (0–20, combining frequency of incontinence to gas, liquid, solid, need for pad, and lifestyle alteration). Anorectal manometry (resting and squeeze pressures), endoanal ultrasound (sphincter defects), pudendal nerve terminal motor latency (PNTML), and defecography.
Management
Conservative (first-line): Dietary modification (fiber supplementation to bulk stools), loperamide (increases IAS tone and reduces stool frequency), biofeedback/pelvic floor physical therapy (50–75% improvement), and anal plugs.
Surgical options:
| Procedure | Indication | Success |
|---|---|---|
| Sphincteroplasty (overlapping sphincter repair) | Focal EAS defect (typically anterior, post-obstetric) | 50–70% initially; long-term results decline (50% at 5 years) |
| Sacral nerve stimulation (SNS / InterStim) | Incontinence refractory to conservative therapy; intact or repaired sphincters | 70–80% achieve ≥50% improvement; now first-line surgical option |
| Injectable bulking agents (Solesta) | Passive incontinence with intact sphincters | 50% improvement; effect wanes over time |
| Colostomy | Severe, refractory incontinence impacting quality of life | Definitive; significant QoL improvement in selected patients |
24 Ileostomy & Colostomy — Types & Technique
Types of Stomas
| Type | Description | Common Indications | Output |
|---|---|---|---|
| End ileostomy (Brooke) | Terminal ileum divided; proximal end matured as stoma; distal end resected or as Hartmann's stump | Total proctocolectomy (UC, FAP); Crohn's after proctectomy | 500–1500 mL/day; liquid/paste |
| Loop ileostomy | Loop of ileum exteriorized; opened on antimesenteric border; both limbs visible (proximal functional, distal defunctionalized) | Fecal diversion to protect distal anastomosis (LAR, IPAA); emergency diversion | Similar to end ileostomy |
| End colostomy | Divided colon; proximal end matured as stoma | APR (permanent); Hartmann procedure (potentially reversible) | 100–300 mL/day; formed/semiformed |
| Loop colostomy | Loop of colon (usually transverse or sigmoid) exteriorized | Emergency decompression (obstruction); diversion for perineal wounds or distal pathology | Variable |
Stoma Site Marking
Preoperative stoma site marking by an enterostomal therapy (ET) nurse is essential. Principles: the stoma should be placed through the rectus abdominis muscle (reduces parastomal hernia risk), on a flat surface visible to the patient, away from skin creases, scars, bony prominences (iliac crest, costal margin), and the umbilicus. The patient should be assessed in sitting, standing, and supine positions. Mark the site with indelible ink. For ileostomies, the typical site is the right lower quadrant. For colostomies, the typical site is the left lower quadrant.
Brooke Ileostomy Technique
A circular skin excision is made at the marked site, carried down through subcutaneous tissue, anterior rectus sheath (cruciate incision), rectus muscle (split, not cut), and posterior rectus sheath/peritoneum. The aperture should admit two fingers. The ileum is delivered through the abdominal wall without tension, ensuring adequate blood supply (pink, bleeding mesentery). The stoma is matured by everting the ileal edge and suturing full-thickness bowel to the dermal-epidermal junction with absorbable sutures — creating a Brooke eversion (2–3 cm spout). The spout directs effluent into the appliance and prevents peristomal skin erosion from the alkaline ileal content.
25 Stoma Complications & Reversal
Stoma Complications
| Complication | Incidence | Presentation | Management |
|---|---|---|---|
| Parastomal hernia | 30–50% (most common late complication) | Bulge around stoma; may cause obstruction, incarceration | Sugarbaker or Keyhole mesh repair (open or laparoscopic); stoma relocation |
| Stoma prolapse | 2–10% | Telescoping of bowel through the stoma; more common with loop stomas and transverse colostomies | Local revision if symptomatic; relocation if recurrent |
| Stoma retraction | 3–15% | Stoma sinks below skin level; difficulty with appliance seal; peristomal skin breakdown | Convex appliance; local revision with re-eversion; complete revision if severe |
| Stenosis | 2–10% | Narrowing of the stoma at the skin or fascial level; obstructive symptoms | Digital dilation; local revision |
| High-output ileostomy | 10–20% | Output >1500–2000 mL/day; dehydration, electrolyte depletion (hyponatremia, hypokalemia, hypomagnesemia, metabolic acidosis), AKI | Oral rehydration solution (Na >90 mmol/L — WHO ORS or homemade), loperamide (up to 16 mg/day), codeine phosphate, omeprazole (reduces gastric secretion), dietary modification; IV fluids if severe; monitor renal function |
| Peristomal skin complications | Up to 70% | Irritant dermatitis (most common), allergic dermatitis, candidiasis, pyoderma gangrenosum | Proper appliance fit; skin barrier products (stoma powder, barrier rings); antifungals for candida; treat underlying cause |
| Necrosis/ischemia | 1–5% (early postop) | Dusky/black stoma; assess depth with pen light/test tube — if ischemia is superficial and above fascial level, observe; if deep (below fascial level), urgent revision | Observation if superficial; operative revision if below fascia (risk of perforation and peritonitis) |
Stoma Reversal
Loop ileostomy reversal is typically performed 8–12 weeks after the index operation. Prerequisites: confirmed anastomotic integrity (contrast enema or flexible endoscopy), adequate nutrition, resolution of sepsis, and completion of adjuvant chemotherapy (if applicable — many oncologists prefer completion of chemo before reversal). Technique: circumferential incision around the stoma, mobilize the bowel from the abdominal wall, resect the stoma site, and perform a stapled or hand-sewn side-to-side/end-to-end anastomosis. Complications of reversal: SSI (5–15%), anastomotic leak (1–3%), SBO (5–10%), and incisional hernia at the stoma site (up to 30%).
Hartmann reversal is a significantly more complex operation requiring re-laparotomy or laparoscopic approach, adhesiolysis, identification of the rectal stump, and colorectal anastomosis. It carries a morbidity rate of 20–40% and anastomotic leak rate of 5–10%. Up to 40% of Hartmann patients never undergo reversal (due to comorbidities, patient preference, or technical difficulty).
26 Obstructed Defecation & Pelvic Organ Prolapse
Obstructed Defecation Syndrome (ODS)
ODS is the inability to effectively evacuate rectal contents despite the urge to defecate. Causes include dyssynergic defecation (paradoxical contraction of the puborectalis/EAS during attempted defecation — the most common cause), rectocele, enterocele, rectal intussusception, and rectal prolapse.
Dyssynergic Defecation (Anismus)
Diagnosed by anorectal manometry (paradoxical increase in anal canal pressure during attempted defecation) and balloon expulsion test (inability to expel within 1–3 minutes). Treatment: biofeedback therapy is the mainstay (70% success rate) — patients are trained to relax the pelvic floor during defecation using visual/auditory feedback from manometric or EMG sensors. Surgery is NOT indicated for dyssynergic defecation.
Rectocele
An anterior bulge of the rectum into the vaginal septum, caused by weakness of the rectovaginal septum. Common in multiparous women. Symptomatic when requiring digital vaginal pressure ("splinting") to evacuate. Small rectoceles (<2 cm) are typically incidental. Surgical repair is indicated for symptomatic rectoceles >3 cm that fail conservative management (fiber, biofeedback). Approaches: transanal (endorectal flap repair — reinforces the rectovaginal septum from the rectal side), transvaginal (posterior colporrhaphy), or transperineal.
Rectal Intussusception
Infolding of the rectal wall during defecation — can be intrarectal (does not reach the anal canal) or intra-anal (extends into but not beyond the anal canal). Intrarectal intussusception is very common on defecography and is usually an incidental finding. Symptomatic intussusception causing ODS may be treated with ventral mesh rectopexy (laparoscopic). The relationship between internal intussusception and progression to full-thickness prolapse is debated.
Solitary Rectal Ulcer Syndrome (SRUS)
An uncommon condition associated with rectal prolapse/intussusception and straining. Presents with rectal bleeding, mucus discharge, tenesmus, and a characteristic shallow ulcer on the anterior rectal wall (5–10 cm from the anal verge). Biopsy shows fibromuscular obliteration of the lamina propria (pathognomonic). Treatment: behavioral modification (avoid straining), biofeedback, and treat underlying prolapse if present. Refractory cases may require rectopexy or diversion.
27 Chronic Constipation & Colonic Inertia
Evaluation of Chronic Constipation
Chronic constipation is defined by Rome IV criteria: two or more of the following for at least 3 months with onset at least 6 months prior: straining >25% of defecations, lumpy/hard stools >25%, sensation of incomplete evacuation >25%, sensation of anorectal obstruction >25%, manual maneuvers >25%, and fewer than 3 spontaneous bowel movements per week. The workup should include: colonoscopy (to exclude organic pathology, especially in patients >45 or with alarm features), anorectal manometry (to exclude dyssynergic defecation), balloon expulsion test, and colonic transit study (Sitz marker study).
Colonic Transit Study Interpretation
The patient ingests 24 radiopaque markers (day 0) and an abdominal X-ray is obtained on day 5. Normal: ≤5 markers remaining (80% passage). Slow transit constipation (colonic inertia): diffuse marker retention throughout the colon (>5 markers scattered). Outlet obstruction (functional outlet delay): markers clustered in the rectosigmoid. Mixed pattern: features of both.
Slow Transit Constipation (Colonic Inertia)
Surgery is reserved for severe, medically refractory slow-transit constipation after excluding outlet obstruction (dyssynergic defecation) and secondary causes (hypothyroidism, medications, neurologic disease). The procedure of choice is subtotal colectomy with ileorectal anastomosis. Patient selection is critical — satisfaction rate is 80–90% in well-selected patients (confirmed slow transit on repeat studies, normal anorectal function, no significant psychiatric comorbidity, realistic expectations). Potential complications: diarrhea (10–20%), recurrent constipation (5–10%), SBO (10–15%), and IRA anastomotic complications.
Megacolon & Megarectum
Idiopathic megacolon is a dilated, atonic colon (cecum >12 cm, sigmoid >6.5 cm on imaging) in the absence of an identifiable organic cause. Megarectum is a dilated rectum (>6.5 cm at the pelvic brim on lateral X-ray). These conditions cause severe, refractory constipation and may result from chronic fecal retention (behavioral/psychogenic) or neuronal abnormalities. Workup includes excluding Hirschsprung's disease (rectal biopsy for ganglion cells) and Chagas disease (in endemic areas). Management is stepwise: aggressive disimpaction (manual and/or GoLYTELY whole-bowel irrigation), then bowel retraining (scheduled toileting, fiber, osmotic laxatives). Surgery (subtotal colectomy for megacolon, or Duhamel/Soave procedure for megarectum) is reserved for failure of prolonged conservative management.
Acute Colonic Pseudo-Obstruction (Ogilvie Syndrome)
Massive colonic dilation without mechanical obstruction, typically occurring in hospitalized, critically ill, or postoperative patients. Cecal diameter >12 cm on imaging raises concern for impending perforation. Management: correct electrolyte abnormalities (hypokalemia, hypomagnesemia), discontinue offending medications (opioids, anticholinergics), decompression via nasogastric tube, rectal tube, ambulation, and knee-to-chest positioning. If the cecum is >12 cm and not responding to conservative measures within 24–48 hours, neostigmine 2 mg IV (acetylcholinesterase inhibitor) is given as a slow bolus over 3–5 minutes with cardiac monitoring (risk of bradycardia — atropine at bedside). Success rate: 80–90%. Colonoscopic decompression is an alternative. Surgery (cecostomy or subtotal colectomy) is reserved for perforation or failure of all other measures.
28 Colonoscopy & Polypectomy
Colonoscopy
Diagnostic and therapeutic colonoscopy is the most fundamental procedure in colorectal surgery. The colonoscope (typically 130–170 cm) is advanced from the anus to the cecum (identified by the appendiceal orifice, ileocecal valve, and triradiate fold/crow's foot). Cecal intubation rate should be ≥95% (quality indicator). Withdrawal time should be ≥6 minutes (associated with higher adenoma detection rate, ADR). ADR benchmark: ≥25% overall (30% in men, 20% in women).
Polyp Management
| Polyp Size | Technique | Details |
|---|---|---|
| Diminutive (≤5 mm) | Cold forceps polypectomy or cold snare | Cold snare preferred; "resect and discard" strategy acceptable for rectosigmoid diminutive polyps |
| Small (6–9 mm) | Cold snare polypectomy (CSP) | CSP is now preferred over hot snare for polyps <10 mm — lower risk of delayed bleeding and perforation |
| Large (10–19 mm) | Hot snare polypectomy or EMR | Submucosal injection (saline/methylene blue) lifts the polyp for safer resection; piecemeal EMR acceptable |
| Giant (≥20 mm) | Endoscopic mucosal resection (EMR) or endoscopic submucosal dissection (ESD) | ESD enables en bloc resection — better for histologic assessment of submucosal invasion; requires advanced training; higher perforation rate (3–5%) |
Complications of Colonoscopy
| Complication | Incidence | Management |
|---|---|---|
| Perforation | 0.01–0.1% (diagnostic); 0.1–0.5% (therapeutic/polypectomy) | Small, recognized immediately → endoscopic clip closure; large or delayed → CT, NPO, IV antibiotics; free air with peritonitis → emergency surgery (primary repair or resection) |
| Post-polypectomy bleeding | 0.3–1% (immediate or delayed up to 14 days) | Immediate: endoscopic hemostasis (clips, epinephrine injection, thermal coagulation); Delayed: repeat colonoscopy for hemostasis; rarely requires angiographic embolization or surgery |
| Post-polypectomy syndrome (transmural burn) | 0.5–1% | Localized peritoneal inflammation from cautery without perforation; presents with fever, localized pain, leukocytosis; CT shows focal wall thickening but no free air. Treat conservatively: NPO, IV antibiotics, serial exams |
| Splenic injury | Rare (0.004%) | Excessive traction on splenocolic ligament; left upper quadrant pain after colonoscopy; CT diagnosis; may require splenectomy |
Surveillance After Polypectomy
| Finding | Repeat Colonoscopy |
|---|---|
| Normal (no polyps) | 10 years |
| 1–2 tubular adenomas <10 mm | 7–10 years |
| 3–4 tubular adenomas <10 mm | 3–5 years |
| ≥5 adenomas, or any adenoma ≥10 mm, or tubulovillous/villous histology, or HGD | 3 years |
| Piecemeal resection of adenoma ≥20 mm | 6 months (to check for residual), then 1 year, then 3 years |
| Sessile serrated lesion <10 mm, no dysplasia | 5–10 years |
| Sessile serrated lesion ≥10 mm, or with dysplasia | 3 years |
29 Open, Laparoscopic & Robotic Colorectal Surgery
Minimally Invasive Surgery (MIS) for Colorectal Disease
Laparoscopic and robotic-assisted approaches are now the standard for most elective colorectal operations. Benefits over open surgery include shorter hospital stay (by 1–3 days), less postoperative pain, faster return of bowel function, fewer wound complications (SSI, incisional hernia), and comparable oncologic outcomes.
Landmark Trials
COST (2004): Laparoscopic vs open colectomy for colon cancer — equivalent 3-year OS, DFS, recurrence. Shorter hospital stay with laparoscopy. PMID: 15483043
COLOR (2005): Laparoscopic vs open for colon cancer — non-inferior 3-year DFS. PMID: 15998781
COLOR II (2015): Laparoscopic vs open for rectal cancer — non-inferior locoregional recurrence at 3 years. PMID: 25735922
ACOSOG Z6051 (2015) / ALaCaRT (2015): Laparoscopic TME for rectal cancer — did NOT demonstrate non-inferiority of laparoscopic vs open for pathologic outcomes (composite of CRM, distal margin, TME completeness). Interpretation is debated; many experts continue to support laparoscopic rectal cancer surgery in experienced hands. PMID: 26452660
ROLARR (2017): Robotic vs laparoscopic TME — no significant difference in conversion rate (primary endpoint) or short-term outcomes. Robotic approach may benefit in the narrow male pelvis and for obese patients. PMID: 28728955
Robotic Surgery
Robotic-assisted colorectal surgery offers wristed instruments, 3D magnified visualization, surgeon ergonomics, and a stable camera platform. Particularly advantageous in the deep pelvis (rectal surgery, TME) where the articulating instruments facilitate precise dissection. Disadvantages: cost (capital investment and disposables), longer operative time (especially during the learning curve), lack of tactile feedback, and larger port site incisions for the robotic arms. The adoption of robotic colorectal surgery continues to increase rapidly.
Natural Orifice & Single-Incision Approaches
Single-incision laparoscopic surgery (SILS) uses a single multi-channel port (typically through the umbilicus) for colectomy. Cosmetic benefits but technically challenging with instrument crowding. Natural orifice specimen extraction (NOSE) — after laparoscopic colectomy, the specimen is extracted transanally or transvaginally, avoiding an extraction site incision. This reduces wound complications and may improve cosmetic outcomes. Both approaches are gaining popularity in experienced centers but have not shown significant clinical advantage over conventional multi-port laparoscopy in RCTs.
30 Transanal Approaches — TAMIS, TEM & taTME
Transanal Endoscopic Microsurgery (TEM)
Developed by Gerhard Buess in the 1980s, TEM uses a specialized rigid operating rectoscope (4 cm diameter) with magnified stereoscopic visualization and CO2 insufflation to perform full-thickness excision of rectal lesions. Indications: benign rectal tumors (large villous adenomas), T1 rectal cancers with favorable features (sm1, well-differentiated, no LVI, <3 cm, <30% circumference, <8 cm from the anal verge). TEM provides superior excision quality and lower recurrence rates compared to conventional transanal excision.
Transanal Minimally Invasive Surgery (TAMIS)
TAMIS uses a single-incision laparoscopic port (GelPOINT Path or SILS port) inserted through the anus, combined with standard laparoscopic instruments and camera. It provides similar access and outcomes to TEM at lower cost and with a shorter learning curve. Same indications as TEM. A full-thickness excision is performed with 1 cm margins, and the defect is typically closed with absorbable sutures (though closure is not always necessary for small defects).
Transanal Total Mesorectal Excision (taTME)
taTME is a hybrid approach to rectal cancer resection: the abdominal portion (colon mobilization, IMA ligation) is performed laparoscopically, while the pelvic dissection and TME are performed from below (transanally). The theoretical advantage is improved visualization and access in the deep pelvis (where laparoscopic instruments have limited reach), potentially yielding better TME quality and distal margin for low rectal cancers. However, the Norwegian national study (2019) reported an unacceptable local recurrence rate (9.5% at 2.4 years), prompting a national moratorium on taTME in Norway. PMID: 31476773 Subsequent international registry data (COLOR III) and experienced centers report more favorable outcomes, suggesting that taTME is technique-sensitive and requires structured training and proctoring. taTME should only be performed in high-volume centers with appropriate training programs.
31 Enhanced Recovery After Surgery (ERAS)
The ERAS protocol (developed by Henrik Kehlet) is a multimodal, evidence-based perioperative care pathway that has become standard in colorectal surgery. ERAS reduces hospital stay by 2–3 days, decreases complication rates by 30–50%, and improves patient satisfaction.
Preoperative: Patient education and counseling; carbohydrate loading (clear liquid with maltodextrin 2–3 hours before induction — reduces insulin resistance); no prolonged fasting (clear liquids up to 2 hours, solids up to 6 hours before anesthesia); avoid/minimize mechanical bowel prep for colon surgery (consider MBP + oral antibiotics for left-sided/rectal cases); no routine nasogastric tube; DVT prophylaxis (LMWH); prehabilitation when feasible.
Intraoperative: Minimally invasive approach when possible; goal-directed fluid therapy (avoid overhydration); maintain normothermia (forced-air warming); epidural or TAP block for analgesia (opioid-sparing); avoid drains when possible; avoid routine NG tube.
Postoperative: Early oral intake (liquid diet POD 0, regular diet POD 1); early mobilization (out of bed POD 0, ambulate POD 1); multimodal analgesia (acetaminophen, NSAIDs/COX-2 inhibitors, gabapentinoids, local anesthetics — minimize opioids); gum chewing (promotes bowel motility); early removal of urinary catheter (POD 1 or intraoperatively for colon cases); VTE prophylaxis (extended — 28 days for cancer patients); audit and protocol compliance tracking.
ERAS Outcome Metrics
| Metric | Pre-ERAS (Traditional) | Post-ERAS |
|---|---|---|
| Median length of stay (colon) | 7–10 days | 3–5 days |
| Median length of stay (rectal) | 8–14 days | 4–7 days |
| Overall complication rate | 30–40% | 15–25% |
| Readmission rate (30-day) | 10–15% | 8–12% (may initially increase with shorter stays) |
| Return of bowel function | POD 4–5 | POD 2–3 |
| Protocol compliance (≥70% of elements) | N/A | Target ≥80% — higher compliance correlates with better outcomes |
32 Bowel Preparation & Antibiotic Prophylaxis
Mechanical Bowel Preparation (MBP)
MBP clears fecal content from the colon prior to surgery. Options: PEG-based lavage (GoLYTELY — 4 liters; or low-volume split-dose MiraLAX + Gatorade — 238 g PEG 3350 in 64 oz), sodium phosphate (OsmoPrep — oral tablets; higher risk of renal injury, use with caution), and sodium sulfate (SuPrep — low-volume, well-tolerated). The role of MBP alone (without oral antibiotics) in reducing SSI has been questioned — MBP alone may not reduce SSI and increases patient discomfort.
Oral Antibiotics
The addition of oral antibiotics to MBP has been shown to significantly reduce SSI and anastomotic leak rates. The standard regimen is neomycin 1g + metronidazole (Flagyl) 1g orally at 1 PM, 2 PM, and 10 PM the day before surgery (or similar timing to achieve three doses the afternoon/evening before surgery). The Michigan Surgical Quality Collaborative data (2014) demonstrated that combined MBP + oral antibiotics reduced SSI from 5.3% to 3.2% compared to MBP alone. PMID: 25072009
The MBP + Oral Abx Debate
For years, MBP was used universally. Several European RCTs (2004–2007) showed no benefit of MBP alone over no prep, leading many surgeons to abandon MBP. However, the US experience showed that MBP combined with oral antibiotics (not MBP alone) reduces SSI. The current evidence supports the combination (MBP + oral abx) but NOT MBP alone. For right colectomy, some evidence suggests MBP may not be necessary even with oral abx (less fecal loading); however, for left-sided and rectal operations, combined MBP + oral abx is the standard of care.
IV Antibiotic Prophylaxis
Standard IV prophylaxis for colorectal surgery (per SCIP/ACS guidelines): cefazolin 2g IV + metronidazole 500 mg IV within 60 minutes of incision. Alternative for beta-lactam allergy: clindamycin 900 mg IV + gentamicin 5 mg/kg IV, or metronidazole 500 mg IV + ciprofloxacin 400 mg IV. Re-dose cefazolin every 3–4 hours intraoperatively (or if blood loss >1500 mL). Postoperative antibiotics should be discontinued within 24 hours of surgery.
33 IBD Medications
| Drug Class | Examples | Mechanism | Surgical Implications |
|---|---|---|---|
| 5-Aminosalicylates (5-ASA) | Mesalamine (Asacol, Pentasa, Lialda), sulfasalazine | Topical anti-inflammatory (inhibits prostaglandin/leukotriene synthesis) | Safe perioperatively; no need to discontinue |
| Corticosteroids | Prednisone, budesonide, methylprednisolone | Broad immunosuppression, anti-inflammatory | Increased SSI, anastomotic leak, poor wound healing; taper/wean before elective surgery when possible; stress-dose steroids perioperatively if on chronic steroids (>3 weeks) |
| Thiopurines | Azathioprine, 6-mercaptopurine (6-MP) | Purine analog; inhibits lymphocyte proliferation | Modest increase in postoperative infectious complications; generally continued perioperatively |
| Methotrexate | Methotrexate (SQ or oral) | Dihydrofolate reductase inhibitor; anti-inflammatory at low doses | Hold 1–2 weeks before and after surgery; teratogenic |
| Anti-TNF agents | Infliximab (Remicade), adalimumab (Humira), certolizumab (Cimzia) | Monoclonal antibodies binding TNF-alpha | Controversial — some studies suggest increased postoperative infections; ECCO guidelines suggest continuing through surgery; time elective surgery to trough (4–8 weeks after last dose) |
| Anti-integrin | Vedolizumab (Entyvio) | Anti-alpha4beta7 integrin; gut-selective (blocks lymphocyte trafficking to GI mucosa) | Gut-selective → may be safer perioperatively than systemic immunosuppressants; emerging data suggests no increased postop complications |
| Anti-IL-12/23 | Ustekinumab (Stelara) | Monoclonal antibody targeting IL-12 and IL-23 p40 subunit | Limited surgical data; generally continued perioperatively |
| JAK inhibitors | Tofacitinib (Xeljanz), upadacitinib (Rinvoq) | Inhibit Janus kinases; modulate cytokine signaling | Short half-life (advantage for perioperative management); hold 5–7 days before surgery; increased VTE risk |
34 Chemotherapy & Targeted Therapy for CRC
Chemotherapy Backbone
| Regimen | Components | Indication | Key Toxicity |
|---|---|---|---|
| FOLFOX | 5-FU + leucovorin + oxaliplatin | Adjuvant stage III colon; neoadjuvant rectal (TNT); metastatic 1st line | Peripheral neuropathy (cumulative, dose-limiting — cold-triggered in early cycles, persistent in late); myelosuppression; hepatic sinusoidal injury (blue liver) |
| FOLFIRI | 5-FU + leucovorin + irinotecan | Metastatic 1st or 2nd line | Diarrhea (early cholinergic — atropine; late secretory — loperamide); myelosuppression; alopecia |
| CAPOX (XELOX) | Capecitabine + oxaliplatin | Adjuvant stage III colon (especially 3-month for low-risk); metastatic 1st line | Hand-foot syndrome (palmar-plantar erythrodysesthesia); neuropathy; diarrhea |
| Capecitabine alone | Oral fluoropyrimidine (prodrug of 5-FU) | Adjuvant high-risk stage II; concurrent with radiation for rectal cancer | Hand-foot syndrome; diarrhea; myelosuppression |
Targeted Agents
| Agent | Target | Indication | Requirement |
|---|---|---|---|
| Bevacizumab (Avastin) | VEGF (anti-angiogenesis) | Metastatic CRC, 1st or 2nd line | Hold 4–6 weeks before and after surgery (impairs wound healing, risk of bleeding, GI perforation) |
| Cetuximab (Erbitux) | EGFR | Metastatic CRC, left-sided, RAS wild-type | KRAS/NRAS wild-type required; acneiform rash (correlates with response) |
| Panitumumab (Vectibix) | EGFR | Metastatic CRC, left-sided, RAS wild-type | KRAS/NRAS wild-type required |
| Encorafenib + cetuximab (BEACON) | BRAF + EGFR | Metastatic CRC with BRAF V600E mutation | BEACON trial — improved OS vs chemotherapy (9.3 vs 5.9 months) |
| Pembrolizumab (Keytruda) | PD-1 (immune checkpoint) | MSI-H/dMMR CRC — 1st line metastatic (KEYNOTE-177); emerging neoadjuvant role | MSI-H/dMMR testing required |
| Nivolumab +/- ipilimumab | PD-1 +/- CTLA-4 | MSI-H/dMMR metastatic CRC (CheckMate 142) | MSI-H/dMMR testing required |
Tumor sidedness is a strong prognostic and predictive factor. Left-sided CRC (splenic flexure to rectum): better prognosis; responds to anti-EGFR therapy (cetuximab/panitumumab) in RAS wild-type disease. Right-sided CRC (cecum to transverse colon): worse prognosis; does NOT benefit from anti-EGFR therapy even if RAS wild-type; more likely to be MSI-H; consider immunotherapy if MSI-H. First-line metastatic: left-sided + RAS wild-type → FOLFOX/FOLFIRI + cetuximab; right-sided or RAS mutant → FOLFOX/FOLFIRI + bevacizumab; MSI-H → pembrolizumab.
35 Classification Systems (All)
TNM Staging — AJCC 8th Edition
See Section 07 for complete TNM staging tables.
Dukes Classification (Historical)
| Stage | Description | TNM Equivalent |
|---|---|---|
| A | Tumor confined to bowel wall (not through muscularis propria) | T1–T2 N0 |
| B | Tumor extends through bowel wall | T3–T4 N0 |
| C | Lymph node metastasis present | Any T, N1–N2 |
| D (Turnbull modification) | Distant metastasis | Any T, any N, M1 |
Hinchey Classification
See Section 14 for modified Hinchey classification of diverticulitis.
Parks Classification of Fistula-in-Ano
See Section 19 for Parks classification details.
Hemorrhoid Grading (Goligher)
See Section 17 for internal hemorrhoid classification.
Wexner (Cleveland Clinic) Fecal Incontinence Score
| Category | Never (0) | Rarely (1) | Sometimes (2) | Usually (3) | Always (4) |
|---|---|---|---|---|---|
| Incontinence to solid | 0 | 1 | 2 | 3 | 4 |
| Incontinence to liquid | 0 | 1 | 2 | 3 | 4 |
| Incontinence to gas | 0 | 1 | 2 | 3 | 4 |
| Wears pad | 0 | 1 | 2 | 3 | 4 |
| Lifestyle alteration | 0 | 1 | 2 | 3 | 4 |
Total score: 0 (perfect continence) to 20 (complete incontinence). Frequency definitions: Never = 0, Rarely = <1/month, Sometimes = <1/week but ≥1/month, Usually = <1/day but ≥1/week, Always = ≥1/day.
Clavien-Dindo Classification of Surgical Complications
| Grade | Definition | Examples |
|---|---|---|
| I | Any deviation from normal course; no pharmacologic, surgical, endoscopic, or radiologic intervention | Wound infection opened at bedside; antiemetics; antipyretics; electrolyte replacement |
| II | Requiring pharmacologic treatment not allowed for Grade I; blood transfusions; TPN | Blood transfusion; antibiotics for UTI; TPN for prolonged ileus |
| IIIa | Intervention not under general anesthesia | Percutaneous drainage of abscess; endoscopic stenting of stricture |
| IIIb | Intervention under general anesthesia | Reoperation for anastomotic leak; washout for abscess |
| IVa | Single organ dysfunction (including dialysis) | Renal failure requiring dialysis; respiratory failure requiring intubation |
| IVb | Multi-organ dysfunction | Septic shock with MOF |
| V | Death | — |
Peritoneal Cancer Index (PCI)
The PCI divides the abdomen into 13 regions (0–8 for the abdomen, 9–12 for the small bowel). Each region is scored 0–3 based on tumor size (0 = no tumor, 1 = ≤0.5 cm, 2 = 0.5–5 cm, 3 = >5 cm or confluence). Total PCI: 0–39. A PCI ≤20 is generally considered amenable to CRS + HIPEC for colorectal peritoneal metastases.
36 Complications Master Table
| Complication | Incidence | Risk Factors | Presentation | Management |
|---|---|---|---|---|
| Anastomotic leak | 3–15% (higher for low rectal) | Low anastomosis, male, obesity, steroids, radiation, tension, poor blood supply, smoking | Fever, tachycardia, peritonitis, feculent/purulent drain output; may be insidious (day 5–10); CT with rectal contrast | Contained (small): antibiotics, percutaneous drainage, NPO; Uncontained (large, peritonitis): OR — washout + diversion (proximal ileostomy or Hartmann) +/- drain |
| Surgical site infection (SSI) | 5–15% | Obesity, diabetes, smoking, steroids, emergency surgery, prolonged operative time | Incisional erythema, warmth, tenderness, purulent drainage (POD 3–7) | Open wound, drain purulence, antibiotics if cellulitis; wound vac for deep SSI |
| Postoperative ileus | 10–25% | Open surgery, opioid use, extensive dissection, electrolyte imbalance, peritonitis | Nausea, vomiting, abdominal distension, absent flatus/bowel function beyond POD 3–5 | Conservative: NPO, NGT decompression if vomiting, ambulation, minimize opioids, correct electrolytes; alvimopan (Entereg) for opioid-induced ileus |
| Small bowel obstruction (SBO) | 5–15% (higher after open/pelvic surgery) | Prior surgery, adhesions, hernia, radiation | Colicky abdominal pain, vomiting, distension, obstipation; CT shows transition point | Initial conservative management (NGT, IV fluids); surgery if complete obstruction, closed loop, strangulation, or failure to resolve in 48–72 hours |
| Urinary retention | 5–15% | Pelvic dissection (rectal surgery), epidural analgesia, opioids, male, BPH | Inability to void, suprapubic discomfort, bladder volume >400 mL on scan | In-out catheterization; Foley catheter if recurrent; alpha-blocker (tamsulosin); timed voiding trial |
| Urinary/sexual dysfunction (post-TME) | Urinary: 5–30%; Sexual: 10–35% | Low TME, APR, male sex, nerve injury (hypogastric, pelvic splanchnic) | Urinary: retention, incontinence; Sexual: erectile dysfunction, retrograde ejaculation, dyspareunia (female) | Careful nerve-sparing TME technique; urodynamic evaluation; PDE5 inhibitors for ED; may be permanent |
| Stoma complications | See Section 25 | — | — | See Section 25 |
| Venous thromboembolism (VTE) | 2–5% (without prophylaxis: 15–30%) | Cancer, pelvic surgery, prolonged immobility, obesity | DVT: leg swelling, pain; PE: dyspnea, tachycardia, chest pain | Extended VTE prophylaxis (LMWH 28 days for cancer surgery); treatment: therapeutic anticoagulation |
37 Medications Master Table
| Drug | Class | Dose | Key Notes |
|---|---|---|---|
| Cefazolin | Surgical prophylaxis | 2g IV (>120 kg: 3g); re-dose q3–4h | Standard prophylaxis for colorectal surgery (+ metronidazole) |
| Metronidazole (IV) | Surgical prophylaxis | 500 mg IV x1 | Anaerobic coverage; combine with cefazolin |
| Neomycin + Metronidazole (PO) | Oral bowel prep antibiotics | 1g each PO x 3 doses (1 PM, 2 PM, 10 PM day before surgery) | Combined with MBP; reduces SSI |
| Ciprofloxacin | Fluoroquinolone | 500 mg PO BID or 400 mg IV BID | Diverticulitis; pouchitis |
| Metronidazole (PO) | Nitroimidazole | 500 mg PO TID | Diverticulitis; pouchitis; perianal Crohn's; C. difficile (mild) |
| Loperamide | Antidiarrheal (opioid agonist) | 2–4 mg PRN (max 16 mg/day for high-output stoma) | Slows GI transit; increases IAS tone; high-output ileostomy |
| Alvimopan (Entereg) | Peripheral mu-opioid antagonist | 12 mg PO pre-op, then 12 mg BID (max 7 days, max 15 doses) | Accelerates return of bowel function after open colorectal surgery; hospital-restricted |
| Mesalamine | 5-ASA | 2.4–4.8 g/day PO or rectal (suppository/enema) | UC maintenance; cuffitis after IPAA |
| Infliximab | Anti-TNF | 5 mg/kg IV at 0, 2, 6 weeks then q8 weeks | UC and CD; perianal Crohn's fistula |
| Vedolizumab | Anti-integrin | 300 mg IV at 0, 2, 6 weeks then q8 weeks | Gut-selective; UC and CD; may be safer perioperatively |
| 5-FU + leucovorin | Antimetabolite | Various protocols (de Gramont, Roswell Park) | Backbone of CRC chemotherapy |
| Oxaliplatin | Platinum agent | 85–130 mg/m2 IV q2–3 weeks | FOLFOX/CAPOX; dose-limiting neuropathy; hepatic sinusoidal injury |
| Capecitabine | Oral fluoropyrimidine | 1000–1250 mg/m2 BID x 14 days, q3 weeks | Oral alternative to IV 5-FU; hand-foot syndrome |
| Bevacizumab | Anti-VEGF | 5–10 mg/kg IV q2–3 weeks | Hold 4–6 weeks before/after surgery; GI perforation risk |
| Cetuximab | Anti-EGFR | 400 mg/m2 loading, then 250 mg/m2 weekly (or 500 mg/m2 q2 weeks) | RAS wild-type only; left-sided CRC; acneiform rash |
| Pembrolizumab | Anti-PD-1 | 200 mg IV q3 weeks | MSI-H/dMMR CRC; 1st-line metastatic (KEYNOTE-177) |
| GTN 0.2% ointment | Nitric oxide donor | Apply to anal margin BID–TID x 6–8 weeks | Anal fissure; headache in 20–30% |
| Diltiazem 2% ointment | Calcium channel blocker | Apply to anal margin BID x 6–8 weeks | Anal fissure; alternative to GTN; fewer headaches |
| Botulinum toxin A | Neurotoxin (IAS relaxation) | 20–100 units injected into IAS | Chronic anal fissure; effect lasts 2–3 months |
38 Abbreviations Master List
| Abbreviation | Meaning |
|---|---|
| APR | Abdominoperineal resection |
| ATZ | Anal transition zone |
| CME | Complete mesocolic excision |
| CRC | Colorectal cancer |
| CRLM | Colorectal liver metastases |
| CRM | Circumferential resection margin |
| CRT | Chemoradiation therapy |
| DRE | Digital rectal exam |
| EAS | External anal sphincter |
| EAUS | Endoanal ultrasound |
| EMR | Endoscopic mucosal resection |
| ERAS | Enhanced Recovery After Surgery |
| ESD | Endoscopic submucosal dissection |
| FAP | Familial adenomatous polyposis |
| FOLFIRI | 5-FU + leucovorin + irinotecan |
| FOLFOX | 5-FU + leucovorin + oxaliplatin |
| HGD | High-grade dysplasia |
| HIPEC | Hyperthermic intraperitoneal chemotherapy |
| HNPCC | Hereditary nonpolyposis colorectal cancer (Lynch syndrome) |
| IAS | Internal anal sphincter |
| IBD | Inflammatory bowel disease |
| IMA | Inferior mesenteric artery |
| IPAA | Ileal pouch-anal anastomosis (J-pouch) |
| IRA | Ileorectal anastomosis |
| ISR | Intersphincteric resection |
| LAR | Low anterior resection |
| LIFT | Ligation of the intersphincteric fistula tract |
| LIS | Lateral internal sphincterotomy |
| LVI | Lymphovascular invasion |
| MAP | MUTYH-associated polyposis |
| MBP | Mechanical bowel preparation |
| MMR | Mismatch repair |
| MSI-H | Microsatellite instability-high |
| ODS | Obstructed defecation syndrome |
| pCR | Pathologic complete response |
| PCI | Peritoneal Cancer Index |
| PPH | Procedure for Prolapse and Hemorrhoids (stapled hemorrhoidopexy) |
| RAIR | Rectoanal inhibitory reflex |
| RBL | Rubber band ligation |
| SBO | Small bowel obstruction |
| SCC | Squamous cell carcinoma |
| SMA | Superior mesenteric artery |
| SNS | Sacral nerve stimulation |
| SSI | Surgical site infection |
| TAMIS | Transanal minimally invasive surgery |
| taTME | Transanal total mesorectal excision |
| TEM | Transanal endoscopic microsurgery |
| TME | Total mesorectal excision |
| TNT | Total neoadjuvant therapy |
| UC | Ulcerative colitis |
| VTE | Venous thromboembolism |
39 Key Trials, Guidelines & References
Figures
| Figure | Description | Source |
|---|---|---|
| Figure 1 | Large Intestine Anatomy | Wikimedia Commons, BruceBlaus, CC BY 3.0 |
| Figure 2 | Anal Canal Anatomy | Wikimedia Commons, Public domain |
Key Trials & Guidelines
| Trial / Guideline | Year | Key Finding | PMID |
|---|---|---|---|
| COST (laparoscopic vs open colectomy) | 2004 | Laparoscopic colectomy oncologically equivalent to open | 15483043 |
| German Rectal Cancer Trial | 2004 | Preoperative CRT superior to postoperative CRT for rectal cancer | 15496622 |
| Heald — TME principles | 1982 | TME reduced rectal cancer local recurrence from 30–40% to <5% | 3319687 |
| Hohenberger — CME | 2009 | CME with central vascular ligation improves oncologic outcomes for colon cancer | 19997731 |
| IDEA collaboration | 2018 | 3 months CAPOX non-inferior to 6 months for low-risk stage III colon cancer | 29355075 |
| RAPIDO trial | 2021 | TNT (short-course RT → chemo → surgery) improved pCR and disease control for rectal cancer | 33169015 |
| OPRA trial | 2022 | Watch-and-wait for clinical complete response after TNT; 53% organ preservation at 3 years | 36302838 |
| DIABOLO trial | 2017 | Observation without antibiotics non-inferior to antibiotics for uncomplicated diverticulitis | 28586827 |
| LADIES trial — DIVA arm | 2019 | Primary anastomosis + ileostomy superior to Hartmann for perforated diverticulitis (stoma reversal) | 31402817 |
| SCANDIV trial | 2021 | No mortality difference between Hartmann and PA for Hinchey III/IV; higher stoma reversal with PA | 34042961 |
| ACT II (anal cancer) | 2013 | Mitomycin C-based CRT superior to cisplatin-based CRT for anal SCC | 23562698 |
| ANCHOR trial | 2022 | HSIL treatment reduces anal cancer in HIV-positive patients by 57% | 35704336 |
| COLOR II (laparoscopic rectal surgery) | 2015 | Laparoscopic vs open rectal cancer — non-inferior locoregional recurrence | 25735922 |
| ACOSOG Z6051 | 2015 | Laparoscopic TME did not meet non-inferiority for pathologic outcomes vs open | 26452660 |
| ROLARR (robotic vs laparoscopic TME) | 2017 | No difference in conversion rate between robotic and laparoscopic rectal surgery | 28728955 |
| Michigan SQC — Oral antibiotics + MBP | 2014 | Combined MBP + oral antibiotics reduce SSI in colorectal surgery | 25072009 |
Emerging Concepts
| Topic | Description | Current Status |
|---|---|---|
| Circulating tumor DNA (ctDNA) | Postoperative ctDNA detection identifies minimal residual disease after curative resection; predicts recurrence months before radiologic detection | DYNAMIC trial showed ctDNA-guided adjuvant therapy reduces unnecessary chemo in stage II colon cancer without compromising recurrence-free survival; rapidly entering clinical practice |
| Immunotherapy for dMMR CRC | Dostarlimab (anti-PD-1) achieved 100% clinical complete response in 12 patients with dMMR locally advanced rectal cancer (NEJM 2022) | Landmark proof-of-concept; larger trials ongoing; potential to eliminate surgery/radiation in select dMMR rectal cancers |
| Fluorescence-guided surgery (ICG) | Indocyanine green (ICG) angiography assesses anastomotic perfusion intraoperatively; near-infrared fluorescence confirms blood supply to transected bowel ends | Retrospective data suggest reduced anastomotic leak rates; prospective RCTs ongoing (PILLAR III); increasingly standard in robotic colorectal surgery |
| Artificial intelligence in colonoscopy | AI-assisted polyp detection (CADe) and characterization (CADx) during colonoscopy | Multiple RCTs demonstrate improved adenoma detection rate by 10–15% absolute increase; FDA-cleared systems available (GI Genius, CAD EYE) |
Textbooks
The ASCRS Textbook of Colon and Rectal Surgery, 4th edition (Steele et al.) — comprehensive colorectal surgery reference.
Corman's Colon and Rectal Surgery, 6th edition (Corman, Bergamaschi) — classic surgical text.
Current Surgical Therapy, 14th edition (Cameron, Cameron) — board-review format for surgical management.
Sabiston Textbook of Surgery, 21st edition (Townsend et al.) — general surgery reference with colorectal chapters.
Schwartz's Principles of Surgery, 11th edition (Brunicardi et al.) — foundational surgical textbook.
Keighley & Williams' Surgery of the Anus, Rectum and Colon, 4th edition — definitive reference for anorectal and colorectal surgery.
NCCN Clinical Practice Guidelines in Oncology: Colon Cancer / Rectal Cancer / Anal Carcinoma — updated annually; freely accessible at nccn.org.
ASCRS Clinical Practice Guidelines — evidence-based guidelines for diverticulitis, CRC, hemorrhoids, fistula, IBD, and other colorectal conditions; published in Diseases of the Colon & Rectum.
ECCO (European Crohn's and Colitis Organisation) Guidelines — comprehensive guidelines for surgical management of Crohn's disease and ulcerative colitis.