General Surgery
Every diagnosis, classification, procedure, technique, medication, complication, and management algorithm across the full scope of general surgery in one place.
01 Surgical Anatomy — Abdominal Wall & Peritoneum
The abdominal wall is the barrier a surgeon must traverse for every open and laparoscopic procedure. Understanding its layers, neurovascular supply, and weak points (where hernias form) is foundational to general surgery practice.
Layers of the Anterior Abdominal Wall
From superficial to deep: skin → Camper's fascia (superficial fatty layer of subcutaneous tissue) → Scarpa's fascia (deep membranous layer — continuous with Colles' fascia of the perineum; urine from urethral rupture tracks deep to Scarpa's) → external oblique (fibers run inferomedially, "hands in pockets" direction; aponeurosis forms the inguinal ligament inferiorly and the anterior rectus sheath) → internal oblique (fibers run superomedially; contributes to the conjoint tendon with the transversus) → transversus abdominis (fibers run transversely; its aponeurosis is the transversalis fascia anteriorly) → transversalis fascia (critical surgical plane — defects here define direct inguinal hernias) → preperitoneal fat → peritoneum.
The Rectus Sheath & Arcuate Line
The rectus abdominis muscles run vertically from the xiphoid/costal cartilages to the pubic symphysis, separated by the linea alba in the midline. Above the arcuate line (approximately midway between the umbilicus and pubic symphysis), the rectus sheath has an anterior and posterior layer — the posterior layer is formed by the internal oblique aponeurosis (posterior leaf) and transversus abdominis aponeurosis. Below the arcuate line, all three aponeurotic layers pass anterior to the rectus, leaving only transversalis fascia and peritoneum posteriorly. This is why laparoscopic totally extraperitoneal (TEP) hernia repair enters the preperitoneal space more easily below the arcuate line.
Inguinal Region
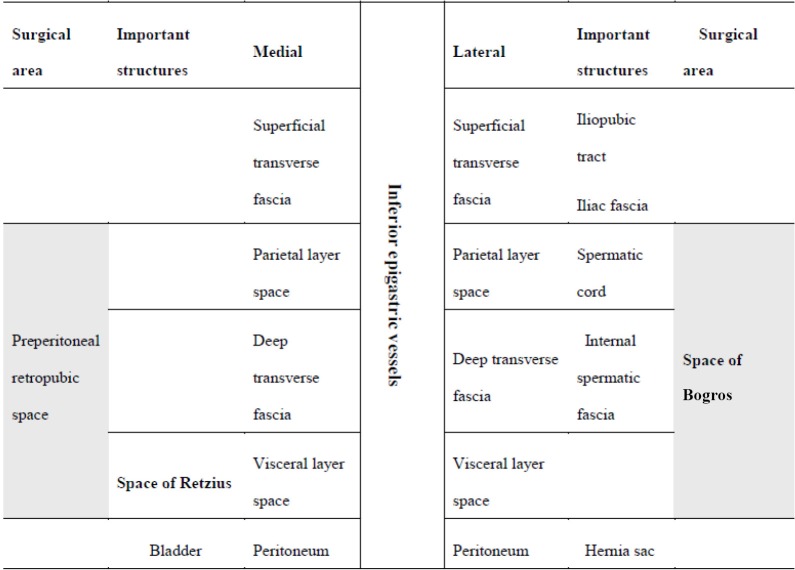
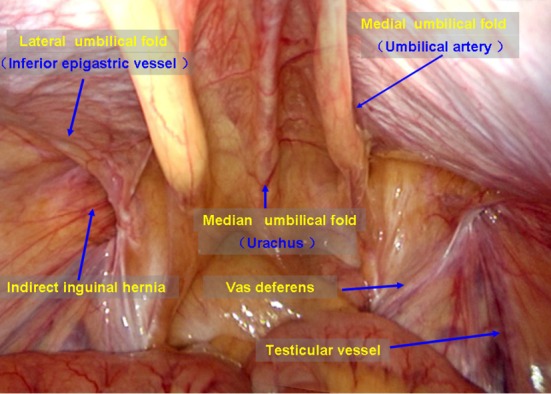
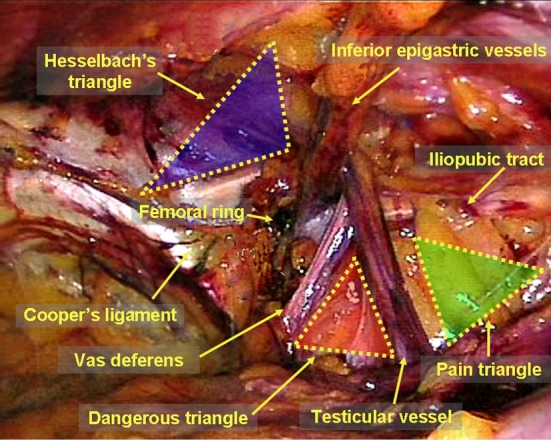
The inguinal canal runs from the deep (internal) inguinal ring to the superficial (external) inguinal ring. The deep ring is an opening in the transversalis fascia, lateral to the inferior epigastric vessels. The superficial ring is an opening in the external oblique aponeurosis. The canal contains the spermatic cord (or round ligament in women), which includes the vas deferens, testicular artery, pampiniform venous plexus, cremasteric artery, genital branch of genitofemoral nerve, and ilioinguinal nerve. The Hesselbach's triangle — bounded by the inguinal ligament inferiorly, the inferior epigastric vessels laterally, and the lateral border of the rectus sheath medially — defines the site of direct inguinal hernias. Indirect hernias exit through the deep ring, lateral to the inferior epigastric vessels.
Peritoneal Anatomy
The peritoneum is a serous membrane lining the abdominopelvic cavity. The parietal peritoneum lines the abdominal wall (somatic innervation — well-localized pain). The visceral peritoneum covers the organs (autonomic innervation — poorly localized, referred pain). The greater omentum hangs from the greater curvature of the stomach like an apron — it migrates to sites of inflammation ("the policeman of the abdomen"), often walling off perforations and abscesses. The lesser omentum connects the lesser curvature of the stomach to the liver; its free right edge contains the portal triad (hepatic artery, portal vein, common bile duct) — this is the hepatoduodenal ligament, which can be clamped during hemorrhage (the Pringle maneuver).
Key peritoneal spaces relevant to abscess formation: right subphrenic (between liver and diaphragm), right subhepatic / Morrison's pouch (the most dependent space in the supine position — fluid collects here first on FAST exam), left subphrenic, paracolic gutters (right gutter communicates with the subhepatic space; left gutter is partially blocked by the phrenicocolic ligament), and the pouch of Douglas / rectovesical pouch (most dependent space in the upright position).
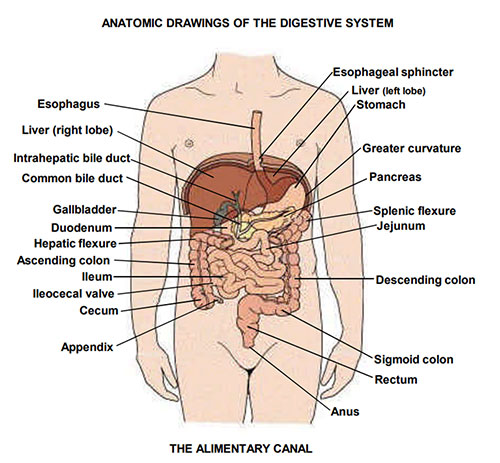
02 Surgical Anatomy — GI Tract, Biliary, & Hepatic
GI Tract Blood Supply
Three major arteries from the aorta supply the GI tract. The celiac trunk (T12) supplies the foregut (distal esophagus to the second portion of the duodenum, plus liver, spleen, and pancreas) via three branches: left gastric artery (lesser curvature of stomach, lower esophagus), splenic artery (spleen, pancreas body/tail, short gastrics to the fundus, left gastroepiploic), and common hepatic artery (→ proper hepatic artery → right and left hepatic arteries to the liver; gastroduodenal artery → right gastroepiploic + superior pancreaticoduodenal).
The superior mesenteric artery (SMA) (L1) supplies the midgut (duodenum distal to ampulla through the proximal two-thirds of the transverse colon) via the inferior pancreaticoduodenal artery, jejunal and ileal branches, ileocolic artery (terminal ileum, cecum, appendix), right colic artery, and middle colic artery. The inferior mesenteric artery (IMA) (L3) supplies the hindgut (distal one-third of the transverse colon through the upper rectum) via the left colic artery, sigmoid branches, and superior rectal artery.
Critical collateral pathways: the marginal artery of Drummond runs along the mesenteric border of the colon connecting SMA and IMA territories. The arc of Riolan (meandering mesenteric artery) is an inconstant but clinically important collateral between the middle colic and left colic arteries. The watershed areas — Griffiths' point (splenic flexure, SMA/IMA junction) and Sudeck's point (rectosigmoid, superior rectal/middle rectal junction) — are most vulnerable to ischemia during hypotension or after IMA ligation in aortic/colonic surgery.
Biliary Anatomy
Bile is produced by hepatocytes and drains through intrahepatic bile ducts → right and left hepatic ducts → join to form the common hepatic duct (CHD) → the cystic duct joins from the gallbladder to form the common bile duct (CBD) → the CBD travels in the hepatoduodenal ligament (anterior to the portal vein, to the right of the hepatic artery) → enters the head of the pancreas → joins the pancreatic duct of Wirsung → opens at the ampulla of Vater on the medial wall of the second portion of the duodenum, controlled by the sphincter of Oddi. Normal CBD diameter: up to 6 mm (add 1 mm per decade over 60; up to 10 mm post-cholecystectomy).
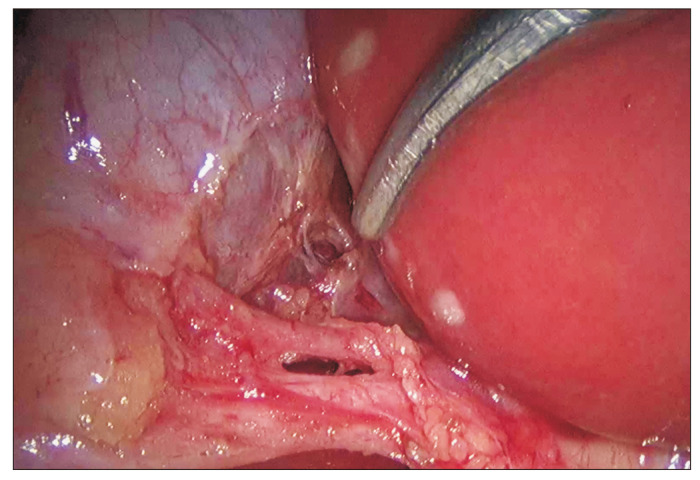
The Calot's triangle (cystohepatic triangle) is the critical landmark during cholecystectomy — bounded by the cystic duct inferiorly, the common hepatic duct medially, and the inferior edge of the liver superiorly. The cystic artery (usually a branch of the right hepatic artery) runs through Calot's triangle. Achieving the critical view of safety (CVS) — clearing the triangle of fat and fibrous tissue until only two structures (cystic duct and cystic artery) are seen entering the gallbladder — is the standard technique for preventing bile duct injury during laparoscopic cholecystectomy (Strasberg SM, 1995; PMID: 7559276).
Variant Biliary Anatomy
"Normal" biliary anatomy occurs in only about 55% of patients. Dangerous variants include: a short cystic duct running parallel to the CHD (risk of CBD injury during clip placement), an accessory hepatic duct draining directly into the cystic duct or gallbladder (risk of unrecognized bile leak), a replaced right hepatic artery arising from the SMA (runs behind the portal vein and through or near Calot's triangle — risk of arterial injury), and the classic Moynihan's hump (tortuous right hepatic artery looping through Calot's triangle, easily mistaken for the cystic artery).
Hepatic Segmentation (Couinaud Classification)
The liver is divided into 8 functionally independent segments, each with its own portal pedicle (portal vein, hepatic artery, bile duct) and hepatic venous drainage. The Cantlie line (from the gallbladder fossa to the IVC) divides the liver into right and left lobes along the plane of the middle hepatic vein. The falciform ligament divides the left lobe into a medial sector (segment IV) and lateral sector (segments II and III).
| Segment | Location | Surgical Relevance |
|---|---|---|
| I (caudate) | Posterior, between IVC and portal vein | Has independent portal and hepatic venous drainage; often hypertrophies in Budd-Chiari syndrome |
| II | Left lateral superior | Left lateral sectionectomy = segments II + III (for living-donor pediatric transplant) |
| III | Left lateral inferior | Easily accessed for biopsy; forms left lateral section with segment II |
| IV (IVa/IVb) | Left medial (between falciform and Cantlie line) | IVa = superior, IVb = inferior (quadrate lobe); left hepatectomy = II + III + IV |
| V | Right anterior inferior | Right hepatectomy = V + VI + VII + VIII |
| VI | Right posterior inferior | Common site of percutaneous biopsy access |
| VII | Right posterior superior | Difficult access; tumors here may require complex resection |
| VIII | Right anterior superior | Adjacent to middle and right hepatic veins superiorly |
03 The Surgical Physical Exam
Abdominal Examination
The surgical abdominal exam follows inspection → auscultation → percussion → palpation (auscultation before palpation/percussion to avoid stimulating bowel sounds). Inspection: distension, scars (prior surgery determines adhesion risk), visible peristalsis (obstruction), ecchymoses (Cullen's sign = periumbilical ecchymosis suggesting retroperitoneal hemorrhage or severe pancreatitis; Grey Turner's sign = flank ecchymosis, same significance). Auscultation: normal bowel sounds are intermittent every 5–15 seconds; high-pitched, tinkling, or rushes suggest mechanical obstruction; absent bowel sounds (>2 min in each quadrant) suggest ileus or peritonitis. Percussion: tympany over gas-filled bowel, dullness over solid organs or fluid; shifting dullness indicates ascites. Palpation: start away from the area of maximum pain; assess for voluntary vs involuntary guarding (involuntary = true peritoneal irritation), rigidity ("board-like" abdomen = diffuse peritonitis, surgical emergency), rebound tenderness, and masses.
Key provocative signs: Rovsing's sign (palpation of the LLQ causes RLQ pain — suggests appendicitis), psoas sign (RLQ pain with passive extension of right hip — retrocecal appendicitis), obturator sign (RLQ pain with internal rotation of flexed right hip — pelvic appendicitis), Murphy's sign (arrest of inspiration during RUQ palpation under the costal margin — acute cholecystitis), Carnett's sign (abdominal wall pain increases with tensing of the rectus — indicates abdominal wall pathology rather than intra-abdominal disease).
Hernia Examination
Examine the patient standing. Inspect the groin for bulges at rest and with Valsalva. The inguinal exam involves invaginating scrotal skin with the index finger into the external inguinal ring: a bulge felt at the fingertip (deep ring) suggests indirect hernia; a bulge against the finger pad (floor of the canal, Hesselbach's triangle) suggests direct hernia. This distinction is unreliable clinically — the definitive determination is made intraoperatively by the relationship to the inferior epigastric vessels. Femoral hernias present as a bulge below and lateral to the pubic tubercle (below the inguinal ligament), in the femoral canal — often small, often missed, high incarceration risk. Ventral hernias are examined supine and with abdominal wall tensing (Valsalva, sit-up) to assess size, reducibility, and fascial defect edges.
Digital Rectal Exam (DRE)
Essential in: lower GI bleeding, suspected appendicitis, anorectal complaints, trauma. Assess: sphincter tone (neurologic integrity), masses (rectal carcinoma within 7 cm of anal verge is palpable), tenderness (anterior tenderness suggests pelvic abscess or appendicitis), prostate (men), stool character and color (melena, hematochezia, occult blood), and presence of fecal impaction.
Wound Assessment
| CDC Wound Class | Definition | SSI Rate | Examples |
|---|---|---|---|
| I — Clean | Elective, no inflammation, no entry into GI/GU/respiratory tract | 1–3% | Thyroidectomy, hernia repair (without mesh infection risk factors) |
| II — Clean-contaminated | Controlled entry into GI/GU/respiratory tract without spillage | 5–8% | Cholecystectomy, elective colon resection with prep |
| III — Contaminated | Open traumatic wound, major spillage from GI tract, entry into infected biliary/urinary tract | 10–15% | Penetrating trauma, unprepared bowel surgery, bile spillage |
| IV — Dirty/Infected | Established infection, devitalized tissue, fecal contamination | 25–40% | Perforated diverticulitis, abscess drainage, fecal peritonitis |
04 Surgical Principles — Wound Healing, Sutures, Hemostasis
Phases of Wound Healing
| Phase | Timing | Key Events | Clinical Significance |
|---|---|---|---|
| Hemostasis | Minutes | Vasoconstriction, platelet plug, fibrin clot | Immediate hemostasis; foundation for subsequent phases |
| Inflammatory | Days 1–5 | Neutrophils (peak day 1–2), macrophages (peak day 2–4), cytokine release | Wound erythema/swelling is normal; absence of inflammation (immunosuppression, steroids) impairs healing |
| Proliferative | Days 4–21 | Fibroblasts produce collagen (type III initially), angiogenesis (granulation tissue), epithelialization, wound contraction (myofibroblasts) | Tensile strength increases rapidly; sutures provide most strength during this phase |
| Remodeling | Day 21 – 1 year+ | Type III collagen replaced by type I collagen, cross-linking increases | Maximum tensile strength reached ~80% of unwounded tissue (never returns to 100%); keloid/hypertrophic scar = excessive remodeling |
Wound Closure Types
Primary intention: edges approximated (sutures, staples, adhesive) — fastest healing, smallest scar, lowest infection risk in clean wounds. Secondary intention: wound left open to heal by granulation, contraction, and epithelialization — used for contaminated/infected wounds, some pilonidal cysts, perianal abscesses. Tertiary intention (delayed primary closure): wound initially left open (packed), then closed 3–5 days later once contamination/edema resolves — used for contaminated traumatic wounds, high-risk abdominal wounds.
Suture Materials
| Suture | Type | Absorption | Tensile Strength | Common Use |
|---|---|---|---|---|
| Chromic gut | Absorbable, natural (bovine/ovine intestine) | ~90 days | 50% at 7–10 days | GI anastomosis (inner layer), subcutaneous, oral mucosa |
| Polyglactin (Vicryl) | Absorbable, braided synthetic | 56–70 days | 75% at 14 days, 50% at 21 days | Most common general closure; fascia, subcuticular, GI anastomosis |
| Poliglecaprone (Monocryl) | Absorbable, monofilament synthetic | 91–119 days | 50–60% at 7 days, 0% at 21 days | Subcuticular skin closure (cosmetic); loses strength fast |
| Polydioxanone (PDS) | Absorbable, monofilament synthetic | 180–210 days | 70% at 14 days, 50% at 28 days | Fascia closure (midline laparotomy), tendon repair |
| Polypropylene (Prolene) | Non-absorbable, monofilament synthetic | None | Permanent | Skin (interrupted), vascular anastomosis, hernia repair |
| Nylon (Ethilon) | Non-absorbable, monofilament synthetic | Very slow degradation | ~80% at 1 year | Skin closure (interrupted or running) |
| Silk | Non-absorbable, braided natural | Loses strength over 1 year; effectively absorbed by 2 years | Variable | Ties (ligatures), drain sutures; highest tissue reaction |
Electrosurgery
Monopolar electrosurgery (Bovie): current flows from the active electrode through the patient to a grounding pad. Cut mode uses continuous low-voltage current — vaporizes tissue with minimal thermal spread. Coagulation mode uses intermittent high-voltage current — desiccates tissue, producing hemostasis with wider thermal spread. Blend mode combines cut and coag waveforms. Bipolar electrosurgery: current flows only between the two tips of a forceps — no grounding pad needed, minimal current spread — used near delicate structures (nerves, bowel, vessels). Advanced energy devices: ultrasonic (Harmonic scalpel) — vibrates at 55,500 Hz, denatures protein, seals vessels up to 5 mm; bipolar vessel sealing (LigaSure) — fuses vessel walls using pressure + bipolar energy, seals vessels up to 7 mm.
Principles of Hemostasis
Mechanical: direct pressure, suture ligation, clips (hemoclips, Weck clips for larger vessels), topical agents (thrombin, gelatin foam/Gelfoam, oxidized cellulose/Surgicel, microfibrillar collagen, fibrin sealant/Tisseel). For diffuse capillary oozing: electrocautery, argon beam coagulator (ionized argon gas conducts current — creates a thin, uniform eschar over large raw surfaces such as the liver bed after cholecystectomy). Packing: used in damage control surgery when the patient is too unstable for definitive hemostasis — perihepatic packing is the classic application in hepatic trauma.
05 Acute Appendicitis Emergency
The most common surgical emergency worldwide, with a lifetime risk of ~7–8%. Appendicitis results from luminal obstruction — fecalith (most common in adults), lymphoid hyperplasia (most common in children/young adults), neoplasm (consider in patients >40, especially if recurrent), or parasites — leading to distension, ischemia, bacterial overgrowth, and eventually perforation if untreated.
Clinical Presentation
Classic sequence: periumbilical visceral pain (due to midgut embryologic origin — T10 dermatome) → anorexia, nausea → pain migrates to McBurney's point (one-third of the distance from the ASIS to the umbilicus) as parietal peritoneal inflammation develops → low-grade fever. This classic progression occurs in only ~50% of patients. Atypical presentations: retrocecal appendix (flank/back pain, psoas sign positive), pelvic appendix (suprapubic pain, urinary symptoms, obturator sign positive), pregnant patients (pain displaced superiorly by the gravid uterus).
Alvarado (MANTRELS) Score
| Feature | Points |
|---|---|
| Migration of pain to RLQ | 1 |
| Anorexia | 1 |
| Nausea / Vomiting | 1 |
| Tenderness in RLQ | 2 |
| Rebound tenderness | 1 |
| Elevated temperature (>37.3 C) | 1 |
| Leukocytosis (>10,000) | 2 |
| Shift to left (neutrophilia >75%) | 1 |
| Total: 10. Score 1–4: unlikely; 5–6: equivocal (imaging recommended); 7–8: probable; 9–10: very probable. | |
Imaging
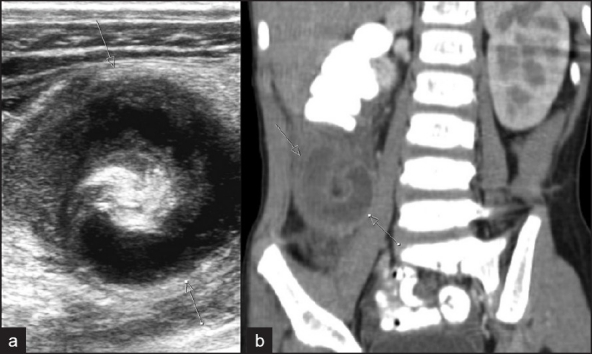
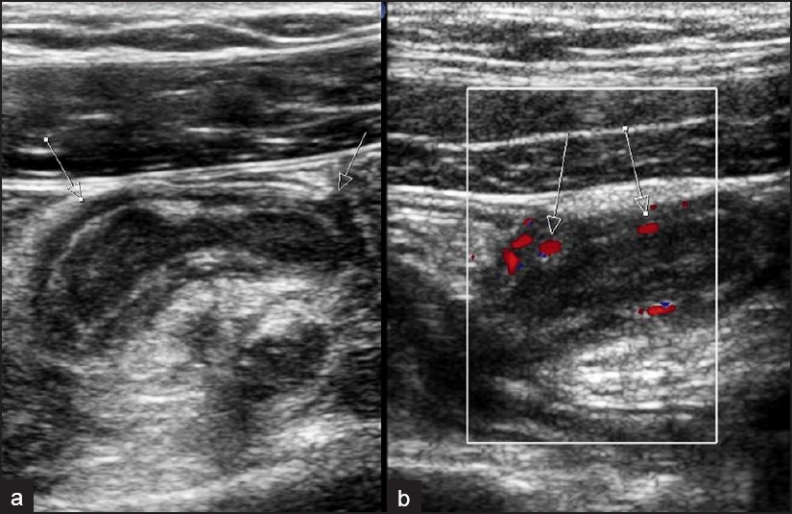
CT abdomen/pelvis with IV contrast is the gold standard in adults (sensitivity 94%, specificity 95%). Findings: dilated appendix >6 mm diameter, appendiceal wall thickening/enhancement, periappendiceal fat stranding, appendicolith, abscess. Ultrasound is first-line in children and pregnant patients (non-compressible tubular structure >6 mm in diameter, target sign on cross-section). MRI is an alternative in pregnancy when ultrasound is equivocal.
Management
Laparoscopic appendectomy is the standard of care — 3-port technique (umbilical 10/12 mm camera port, suprapubic 5 mm, LLQ 5 mm). The mesoappendix is divided with an energy device or between clips. The appendiceal base is secured with an endoloop (0 PDS or 0 Vicryl) or linear endostapler (particularly if the base is inflamed/necrotic). Stump is not routinely inverted. Irrigate the pelvis and right paracolic gutter. Open appendectomy via a McBurney (oblique) or Rocky-Davis (transverse) incision at McBurney's point is used when laparoscopy is unavailable or contraindicated.
Complicated appendicitis: if a well-formed periappendiceal abscess or phlegmon is found on imaging (typically >5 days of symptoms), initial management is IV antibiotics ± percutaneous drainage (if abscess >3 cm). Interval appendectomy 6–8 weeks later is recommended, particularly in patients >40 (to exclude underlying neoplasm). The CODA trial (PMID: 26193091) showed that antibiotics alone are a reasonable alternative to surgery for uncomplicated appendicitis, though ~30% ultimately undergo appendectomy within 90 days.
06 Acute Cholecystitis Emergency
Inflammation of the gallbladder, most commonly (90–95%) from gallstone impaction in the cystic duct. The obstructed gallbladder distends, wall becomes edematous and ischemic, and secondary bacterial infection often follows. Left untreated, progresses to gangrenous cholecystitis, empyema, or perforation. Acalculous cholecystitis (5–10%) occurs in critically ill ICU patients — due to gallbladder stasis and ischemia.
Diagnosis
Murphy's sign is 65% sensitive but highly specific when positive. Labs: leukocytosis, elevated CRP; mild transaminase elevations are common, but significantly elevated bilirubin (>4 mg/dL) or alkaline phosphatase should raise concern for choledocholithiasis or Mirizzi syndrome. Ultrasound is first-line imaging: gallstones, gallbladder wall thickening (>3 mm), pericholecystic fluid, sonographic Murphy's sign (focal tenderness over the gallbladder under the transducer). HIDA scan (hepatobiliary iminodiacetic acid) is used when ultrasound is equivocal — non-visualization of the gallbladder at 4 hours (or 60 minutes after morphine augmentation) confirms cystic duct obstruction (sensitivity 97%, specificity 90%).
Tokyo Guidelines (TG18) Severity Grading
| Grade | Criteria | Management |
|---|---|---|
| Grade I — Mild | Acute cholecystitis in a healthy patient without organ dysfunction; mild inflammatory changes confined to the gallbladder | Early laparoscopic cholecystectomy (within 72 hours of symptom onset) |
| Grade II — Moderate | Any of: WBC >18,000, palpable RUQ mass, duration >72 hours, marked local inflammation (gangrenous cholecystitis, pericholecystic abscess, hepatic abscess, biliary peritonitis) | Early cholecystectomy if experienced surgeon; otherwise antibiotics + delayed cholecystectomy or percutaneous cholecystostomy tube |
| Grade III — Severe | Organ dysfunction: cardiovascular (hypotension requiring vasopressors), neurological (decreased consciousness), respiratory (PaO2/FiO2 <300), renal (Cr >2.0), hepatic (INR >1.5), hematologic (platelets <100,000) | Urgent gallbladder drainage (percutaneous cholecystostomy) + ICU stabilization; cholecystectomy deferred until patient optimized |
Surgical Technique — Laparoscopic Cholecystectomy
Standard 4-port technique: 10/12 mm umbilical (camera), 10/12 mm epigastric (right of falciform), two 5 mm subcostal ports (midclavicular and anterior axillary lines). The gallbladder fundus is retracted cephalad, the infundibulum is retracted laterally and inferiorly to open Calot's triangle. The critical view of safety is achieved when: (1) the hepatocystic triangle is cleared of fat and fibrous tissue, (2) the lower third of the gallbladder is separated from the liver bed, and (3) only two structures (cystic duct and cystic artery) are seen entering the gallbladder. Only then are clips applied and structures divided. The gallbladder is dissected from the liver bed using electrocautery and extracted in a specimen bag. An intraoperative cholangiogram (IOC) may be performed to evaluate for CBD stones if indicated.
07 Small Bowel Obstruction Emergency
SBO accounts for ~15% of emergency surgical admissions. The bowel proximal to the obstruction dilates with gas and fluid, while the distal bowel decompresses. If the obstruction is not relieved, intraluminal pressure rises → venous congestion → arterial ischemia → necrosis → perforation.
Etiology
| Cause | Frequency | Key Features |
|---|---|---|
| Adhesions (post-surgical) | ~65–75% | Most common cause in developed countries; history of prior abdominal surgery (especially lower abdominal, pelvic) |
| Hernias (inguinal, ventral, internal) | ~10–15% | Most common cause worldwide; always examine the groins in SBO |
| Malignancy | ~5–10% | Extrinsic compression, intraluminal mass, or carcinomatosis |
| Crohn's disease | ~5% | Strictures from chronic inflammation or prior surgery |
| Other (gallstone ileus, intussusception, volvulus, foreign body, radiation enteritis) | ~5% | Gallstone ileus: air in the biliary tree (pneumobilia) + SBO + ectopic gallstone (Rigler's triad) |
Clinical Presentation & Diagnosis
Cardinal symptoms: colicky abdominal pain, nausea/vomiting (early and bilious in proximal SBO, late and feculent in distal SBO), abdominal distension (more prominent in distal SBO), obstipation (no flatus or stool — indicates complete obstruction). CT abdomen/pelvis with IV contrast is the imaging study of choice (sensitivity >90%): identifies the transition point (dilated proximal bowel → decompressed distal bowel), the cause (adhesive band, hernia, mass), and complications (closed-loop obstruction, strangulation, perforation). The "small bowel feces sign" (particulate matter in the small bowel proximal to the obstruction) indicates prolonged, high-grade obstruction.
Management Algorithm
Initial: NPO, IV fluid resuscitation (patients are profoundly volume-depleted from third-spacing and vomiting), NG tube decompression, foley catheter, serial abdominal exams, electrolyte correction. Indications for urgent surgery: peritonitis, closed-loop obstruction, pneumatosis intestinalis (bowel wall gas = ischemia/necrosis), portal venous gas, free air, strangulation signs (persistent tachycardia, fever, leukocytosis, lactic acidosis, CT signs of mesenteric edema or reduced bowel wall enhancement). Non-operative management (for adhesive SBO without strangulation signs): NG decompression, IV fluids, and observation for 24–72 hours. A water-soluble contrast study (Gastrografin) at 24–48 hours is both diagnostic (contrast in the colon on 24-hour film predicts resolution) and potentially therapeutic (hyperosmolar agent draws fluid into the lumen, stimulating peristalsis) (PMID: 17297533). Surgery is indicated if there is no improvement after 48–72 hours of conservative management.
08 Large Bowel Obstruction & Volvulus Emergency
LBO is less common than SBO but is more often caused by malignancy (~60%), followed by volvulus (~15%), diverticular stricture (~10%), and other causes (fecal impaction, Ogilvie's syndrome). The ileocecal valve determines whether the obstruction is "closed-loop" — a competent valve prevents decompression into the small bowel, causing progressive cecal distension. Cecal diameter >12 cm on imaging is associated with high perforation risk.
Sigmoid Volvulus
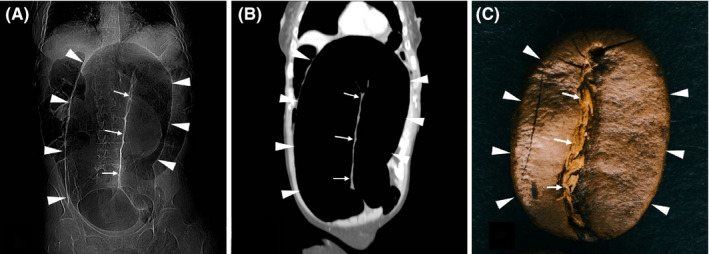
The most common type of volvulus in adults. A redundant sigmoid colon twists on its mesentery, causing closed-loop obstruction and mesenteric vascular compromise. Classic patient: elderly, institutionalized, chronic constipation, neuropsychiatric disease. X-ray: "coffee bean" sign — large dilated loop arising from the pelvis pointing toward the RUQ, with loss of haustral markings. CT: "whirl sign" at the point of torsion. Management: if no signs of ischemia/peritonitis → endoscopic decompression (flexible sigmoidoscopy with placement of a rectal tube) — successful in ~80% of cases. This is temporizing — the recurrence rate is 40–60%, so sigmoid colectomy (during the same admission after bowel prep, or as an interval procedure) is recommended. If gangrenous bowel is found → emergent resection (Hartmann's procedure — sigmoid colectomy with end colostomy and rectal stump).
Cecal Volvulus
The cecum and ascending colon twist (requires a mobile cecum — seen in ~10% of people with incomplete retroperitoneal fixation). Younger patients than sigmoid volvulus. X-ray: dilated cecum in the LUQ or epigastrium ("kidney bean" shape). Unlike sigmoid volvulus, endoscopic decompression is not effective for cecal volvulus. Treatment is surgical: right hemicolectomy (definitive) or cecopexy (higher recurrence rate).
Ogilvie's Syndrome (Acute Colonic Pseudo-Obstruction)
Massive colonic dilation without mechanical obstruction — a functional/motility disorder. Typically occurs in hospitalized, critically ill patients with multiple medical comorbidities, recent surgery, or electrolyte derangements. Treatment: correct electrolytes (potassium, magnesium), discontinue offending medications (opioids, anticholinergics), ambulate, neostigmine 2 mg IV over 3–5 minutes (under cardiac monitoring — may cause bradycardia; success rate ~90%); if refractory → colonoscopic decompression. Surgery only for perforation or failure of all conservative measures.
09 Perforated Viscus Emergency
Perforation of a hollow viscus is a surgical emergency. Free air on imaging (upright CXR shows subdiaphragmatic air; CT is more sensitive) confirms the diagnosis. The peritoneal contamination triggers chemical peritonitis (gastric acid in peptic ulcer perforation) and then bacterial peritonitis (fecal contamination in colonic perforation).
| Site | Common Causes | Key Features | Surgical Management |
|---|---|---|---|
| Stomach/Duodenum | Peptic ulcer (NSAIDs, H. pylori), malignancy | Sudden epigastric pain → diffuse peritonitis; anterior duodenal ulcers perforate freely, posterior ulcers erode into GDA → hemorrhage | Graham patch (omental patch over the perforation) ± definitive ulcer surgery (rarely needed in the H. pylori era) |
| Small bowel | Ischemia, Crohn's, typhoid, trauma, foreign body | Peritonitis with bilious/enteric content spillage | Resection with primary anastomosis (if clean, hemodynamically stable) or damage control with delayed anastomosis |
| Appendix | Appendicitis | RLQ peritonitis, walled-off abscess, or free perforation | Appendectomy; if diffuse peritonitis → washout; if abscess → drainage ± interval appendectomy |
| Colon | Diverticulitis, malignancy, ischemia, colonoscopy perforation, toxic megacolon | Fecal peritonitis = highest morbidity/mortality of all GI perforations | Hartmann's procedure (most common for emergent perforation), resection with primary anastomosis ± diverting ileostomy (selected stable patients), or damage control with temporary abdominal closure |
10 Mesenteric Ischemia Emergency
Mesenteric ischemia is a vascular emergency with mortality rates of 60–80% if bowel infarction occurs. The hallmark is "pain out of proportion to exam" — severe periumbilical pain with a relatively soft, non-tender abdomen early in the course. By the time peritoneal signs develop, bowel infarction has likely already occurred.
Types
| Type | % of Cases | Mechanism | Key Features |
|---|---|---|---|
| SMA embolism | ~50% | Cardiac embolism (AFib, mural thrombus, valve disease) lodges in the SMA (usually past the first jejunal branch) | Sudden onset; atrial fibrillation history; proximal jejunum often spared (embolus lodges distal to first jejunal branch) |
| SMA thrombosis | ~25% | Acute thrombosis on pre-existing atherosclerotic stenosis at the SMA origin | May have history of chronic mesenteric ischemia (postprandial pain, food fear, weight loss); more extensive bowel involvement |
| Non-occlusive mesenteric ischemia (NOMI) | ~20% | Splanchnic vasoconstriction from low cardiac output, vasopressors, dialysis, cocaine | ICU patients; no mesenteric vessel occlusion on angiography; treat the underlying shock, stop vasopressors if possible; intra-arterial papaverine |
| Mesenteric venous thrombosis | ~5% | SMV thrombosis from hypercoagulable states, portal hypertension, intra-abdominal inflammation | More insidious onset; CT shows SMV thrombus with bowel wall edema; most respond to anticoagulation alone; surgery only for peritonitis |
Management
CT angiography (CTA) is the diagnostic study of choice (sensitivity >95% for SMA occlusion). Labs: markedly elevated lactate (>2 mmol/L) suggests ischemia but is a late finding. Aggressive IV fluid resuscitation, broad-spectrum antibiotics, and heparin anticoagulation (prevent thrombus propagation). Definitive treatment for SMA embolism: surgical embolectomy (SMA exposed at the root of the mesentery, transverse arteriotomy, Fogarty balloon catheter embolectomy) or endovascular thrombectomy/thrombolysis. Second-look laparotomy at 24–48 hours is essential to reassess bowel viability and resect any additional non-viable segments — bowel viability is assessed by color (pink vs dusky), peristalsis, mesenteric arterial pulsation, and intravenous fluorescein (Wood's lamp examination) or ICG angiography.
11 Acute Pancreatitis Emergency
Acute inflammation of the pancreas, most commonly from gallstones (40%) and alcohol (30%). Other causes: hypertriglyceridemia (>1000 mg/dL), post-ERCP, medications (azathioprine, valproic acid, didanosine), autoimmune, trauma, scorpion stings. Diagnosis requires 2 of 3 criteria: (1) characteristic epigastric pain radiating to the back, (2) lipase ≥3× upper limit of normal, (3) characteristic imaging findings on CT.
Ranson's Criteria
| At Admission | At 48 Hours |
|---|---|
| Age >55 | Hematocrit drop >10% |
| WBC >16,000 | BUN increase >5 mg/dL |
| Glucose >200 mg/dL | Calcium <8 mg/dL |
| AST >250 IU/L | PaO2 <60 mmHg |
| LDH >350 IU/L | Base deficit >4 mEq/L |
| Fluid sequestration >6 L | |
| Score interpretation: 0–2: <1% mortality; 3–4: ~15% mortality; 5–6: ~40% mortality; ≥7: ~100% mortality | |
BISAP Score
| Criterion | Points |
|---|---|
| BUN >25 mg/dL | 1 |
| Impaired mental status (GCS <15) | 1 |
| SIRS (≥2 of: temp >38 or <36, HR >90, RR >20/PaCO2 <32, WBC >12K or <4K) | 1 |
| Age >60 | 1 |
| Pleural effusion on imaging | 1 |
| Score ≥3: increased risk of severe pancreatitis and mortality (>5% in-hospital mortality) | |
Revised Atlanta Classification (2012)
| Severity | Definition | Mortality |
|---|---|---|
| Mild | No organ failure, no local or systemic complications | <1% |
| Moderately severe | Transient organ failure (<48 hours) and/or local complications (peripancreatic fluid collection, pancreatic pseudocyst, acute necrotic collection, walled-off necrosis) | ~5% |
| Severe | Persistent organ failure (>48 hours) — respiratory (PaO2/FiO2 ≤300), cardiovascular (SBP <90 despite fluids), renal (Cr >1.9) | 20–40% |
Management
Supportive: aggressive IV fluid resuscitation (Lactated Ringer's preferred — PMID: 21748069), pain control (IV opioids, PCA), NPO initially then early oral feeding when tolerated (within 24–48 hours in mild disease; do not wait for lipase to normalize). Gallstone pancreatitis: cholecystectomy during the same admission (for mild disease) or after resolution of inflammation (for severe disease) — delays beyond 6 weeks have high recurrent pancreatitis rates. ERCP within 24 hours if concurrent cholangitis or persistent biliary obstruction. Infected pancreatic necrosis (suspected when a patient deteriorates after 7–10 days, gas within necrotic collection on CT, positive FNA culture): first-line is antibiotics (carbapenems penetrate necrotic tissue); if no improvement → step-up approach: percutaneous/endoscopic transgastric drainage first, then minimally invasive necrosectomy (VARD — video-assisted retroperitoneal debridement) only if drainage fails (PANTER trial; PMID: 20410514).
12 Gastrointestinal Hemorrhage Emergency
Upper GI Hemorrhage
Defined as bleeding proximal to the ligament of Treitz. Presents with hematemesis (bright red or "coffee ground"), melena, or both. Most common causes: peptic ulcer disease (35–50%), esophageal/gastric varices (10–20%), Mallory-Weiss tear (5–10%), erosive gastritis/esophagitis, Dieulafoy lesion (aberrant submucosal arteriole, usually in the gastric fundus — intermittent massive hemorrhage), angiodysplasia, malignancy, aortoenteric fistula (must consider in any patient with prior aortic graft + GI bleed — "herald bleed" may precede catastrophic hemorrhage by hours to days).
EGD within 24 hours is the primary diagnostic and therapeutic modality. Endoscopic therapies: injection (epinephrine 1:10,000 dilution — for temporary hemostasis only, always combined with another modality), thermal (bipolar electrocautery, heater probe), mechanical (hemoclips), and over-the-scope clips (OTSCs) for larger defects. The Forrest classification for peptic ulcers grades the endoscopic appearance:
| Forrest Class | Description | Rebleed Risk | Endoscopic Therapy |
|---|---|---|---|
| Ia | Spurting arterial hemorrhage | 90% | Yes — emergent |
| Ib | Oozing hemorrhage | 50% | Yes |
| IIa | Non-bleeding visible vessel | 40–50% | Yes |
| IIb | Adherent clot | 20–30% | Consider (remove clot, treat underlying lesion) |
| IIc | Flat pigmented spot | 5–10% | No |
| III | Clean base ulcer | <5% | No |
Surgical indications for upper GI bleeding: hemodynamic instability despite resuscitation, transfusion requirement >6 units in 24 hours, failed endoscopic hemostasis (two attempts), or recurrent bleeding after endoscopic therapy. Surgical options for bleeding peptic ulcer: oversewing the ulcer (U-stitch ligation of the bleeding vessel — GDA for posterior duodenal ulcers, left gastric artery for lesser curvature ulcers), with or without vagotomy/pyloroplasty.
Lower GI Hemorrhage
Defined as bleeding distal to the ligament of Treitz. Presents with hematochezia (bright red blood per rectum). Most common causes: diverticular bleeding (30–40% — painless, often massive, self-limited in 80%), angiodysplasia (arteriovenous malformations, typically in the right colon of elderly patients), colorectal neoplasm, hemorrhoids, IBD, ischemic colitis, radiation proctitis. Massive lower GI bleeding requiring >2 units transfusion is a diverticular bleed until proven otherwise.
Diagnostic workup: colonoscopy (after rapid bowel prep) is the preferred initial study when stable — both diagnostic and therapeutic. CT angiography (arterial phase) can detect active bleeding at rates as low as 0.3–0.5 mL/min and localizes the site for targeted intervention. Tagged RBC scan (technetium-99m) detects bleeding at 0.1–0.5 mL/min — more sensitive than CTA but less precise for localization. Mesenteric angiography detects bleeding at ≥0.5–1.0 mL/min and allows therapeutic embolization. Surgical indications: hemodynamic instability despite resuscitation, ongoing transfusion requirement (>4–6 units), or failure of endoscopic/angiographic interventions. If the bleeding source is localized → segmental colectomy. If the source cannot be identified despite all modalities → subtotal colectomy with ileorectal anastomosis is the operation of last resort (rebleed rate <5%, but significant morbidity).
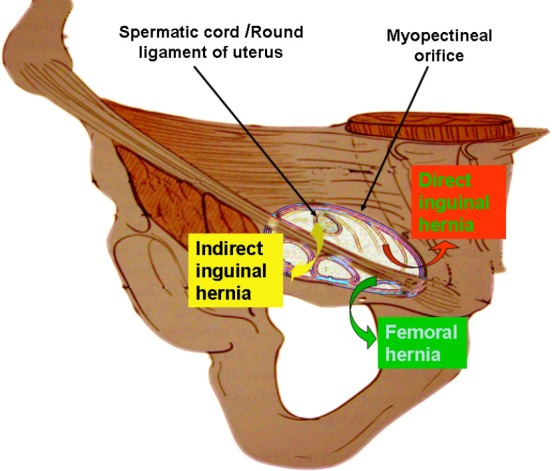
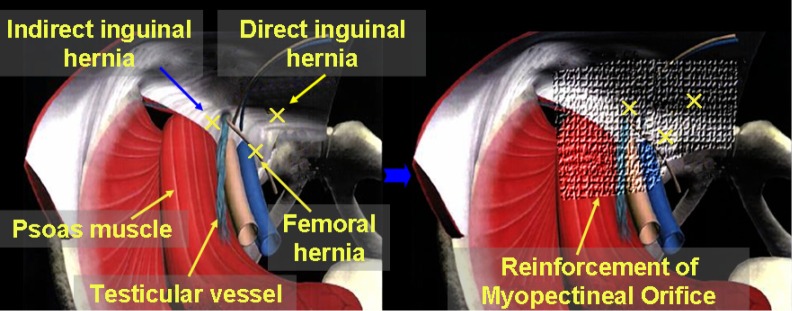
13 Inguinal Hernia
Inguinal hernias account for ~75% of all abdominal wall hernias. Lifetime risk: 27% for men, 3% for women. They are classified as indirect (through the deep inguinal ring, lateral to the inferior epigastric vessels — congenital, from a patent processus vaginalis; most common type overall and in all age groups) or direct (through the floor of the inguinal canal in Hesselbach's triangle, medial to the inferior epigastric vessels — acquired, from transversalis fascia weakness; associated with age, smoking, connective tissue disorders).
Nyhus Classification
| Type | Description |
|---|---|
| I | Indirect hernia with normal internal ring (pediatric type) |
| II | Indirect hernia with dilated internal ring; posterior wall intact |
| IIIa | Direct inguinal hernia (posterior wall defect) |
| IIIb | Indirect hernia with large internal ring ± direct component (posterior wall destroyed); includes sliding hernias, pantaloon hernias |
| IIIc | Femoral hernia |
| IV | Recurrent hernia (IVa = direct, IVb = indirect, IVc = femoral, IVd = combined) |
Open Repair — Lichtenstein Tension-Free Mesh Repair
The gold standard open technique. Inguinal incision from the pubic tubercle to the internal ring. External oblique aponeurosis opened. Spermatic cord mobilized. Cremaster fibers divided to identify the hernia sac — indirect sacs are dissected from the cord, reduced/ligated at the internal ring; direct sacs are simply inverted. A polypropylene mesh (lightweight, large-pore preferred — Ultrapro, Prolene Soft) is placed over the floor of the inguinal canal, sutured to the pubic tubercle medially, the inguinal ligament inferiorly, and the conjoint tendon/internal oblique superiorly. A slit is made laterally for the cord. The mesh is overlapped and tacked at the lateral apex. The ilioinguinal nerve should be identified and preserved (or if unavoidable, divided cleanly — a divided nerve causes less chronic pain than an entrapped one). Recurrence rate with mesh: 1–2%.
Laparoscopic Repair — TEP & TAPP
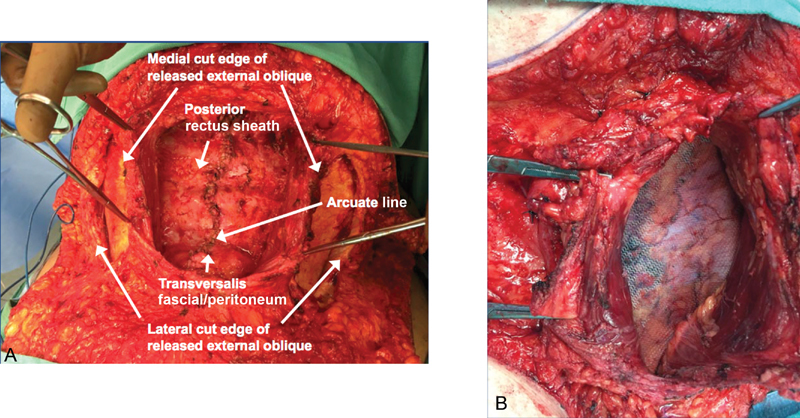
Totally Extraperitoneal (TEP): camera enters the preperitoneal space through an infraumbilical incision below the arcuate line. A dissecting balloon creates the working space. Two additional 5 mm ports placed in the midline. The hernia sac is reduced, and mesh (10 × 15 cm, covering the myopectineal orifice) is placed in the preperitoneal space without fixation (in most cases) or with absorbable tack fixation. Advantages: no peritoneal entry, less adhesion risk. Transabdominal Preperitoneal (TAPP): standard laparoscopic peritoneal access, peritoneum incised above the myopectineal orifice, preperitoneal space developed, mesh placed, peritoneum closed over the mesh. TAPP is technically easier and provides better visualization but requires peritoneal closure. Both have recurrence rates of 1–3% and offer the advantage of simultaneous bilateral repair through the same incisions.
Mesh Types
| Mesh Type | Material | Characteristics | Use |
|---|---|---|---|
| Heavyweight polypropylene | Polypropylene (>80 g/m2) | Strong, but higher foreign body reaction, stiffness, chronic pain | Largely replaced by lightweight meshes |
| Lightweight polypropylene | Polypropylene (25–50 g/m2), large pore (>1 mm) | Better tissue integration, less chronic pain, more flexibility | Standard for open and laparoscopic inguinal repair |
| Composite / ePTFE | Polypropylene + ePTFE/anti-adhesion barrier | One surface integrates with tissue, other surface prevents bowel adhesion | Intraperitoneal placement (IPOM for ventral hernia) |
| Biologic | Acellular dermal matrix (human, porcine, bovine) | Absorbable scaffold, remodels into host tissue; very expensive | Contaminated fields, infected hernia repairs |
14 Femoral Hernia
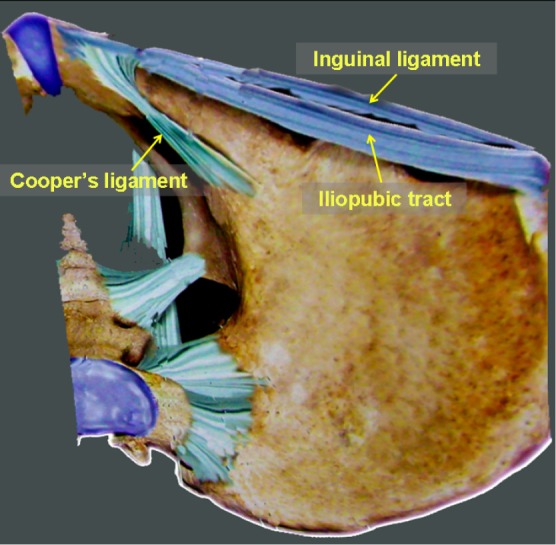
Protrusion through the femoral canal, located below the inguinal ligament, medial to the femoral vein, lateral to the lacunar ligament. Accounts for only ~5% of groin hernias but carries the highest strangulation risk of any hernia (~30–40% present with incarceration/strangulation). More common in women (due to wider pelvis), though inguinal hernias are still more common than femoral hernias even in women. The femoral canal is bounded: anteriorly by the inguinal ligament, posteriorly by the pectineal (Cooper's) ligament, medially by the lacunar (Gimbernat's) ligament, and laterally by the femoral vein — these rigid boundaries make this a tight ring prone to strangulation.
Repair: preperitoneal mesh repair (laparoscopic TEP/TAPP or open preperitoneal approach) is preferred — covers both the inguinal and femoral spaces. The McVay (Cooper's ligament) repair is the classic tissue repair that specifically addresses the femoral canal by suturing the conjoint tendon to Cooper's ligament. All incarcerated femoral hernias require emergent surgery with evaluation of bowel viability.
15 Ventral & Incisional Hernia
Ventral hernias include umbilical, epigastric, and incisional hernias. Incisional hernias occur in 10–20% of midline laparotomy closures (higher with emergency surgery, wound infection, obesity, diabetes, malnutrition, immunosuppression, tobacco use). They tend to enlarge over time due to progressive fascial attenuation (the "Swiss cheese" phenomenon of multiple fascial defects within a larger area of weakness).
Repair Techniques
| Technique | Description | Indications |
|---|---|---|
| Primary suture repair | Direct fascial edge approximation | Small defects <2 cm, low risk of recurrence |
| Open mesh repair — Onlay | Mesh placed anterior to the fascia (over the closed defect) | Simple, but higher SSI risk, higher recurrence than sublay |
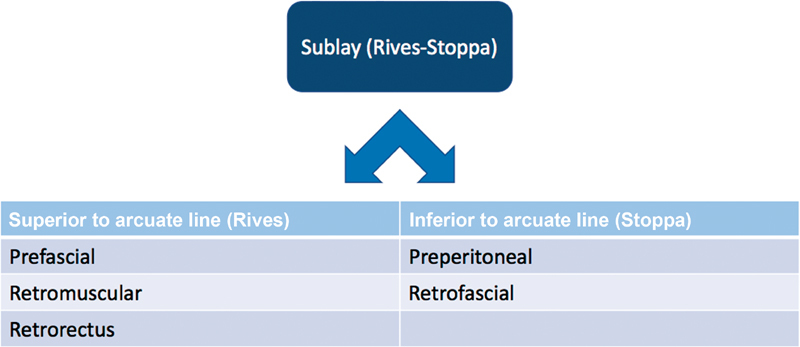
| Open mesh repair — Sublay (Rives-Stoppa) | Mesh placed in the retromuscular (retrorectus) space, behind the rectus muscles and anterior to the posterior rectus sheath | Gold standard for midline ventral/incisional hernias; mesh in a well-vascularized space separated from bowel by the posterior sheath |
| Open mesh repair — Underlay (IPOM) | Mesh placed intraperitoneally against the abdominal wall (requires anti-adhesion barrier mesh) | Used when preperitoneal/retromuscular space cannot be developed |
| Laparoscopic IPOM | Intraperitoneal mesh fixed with tacks and/or transfascial sutures | Smaller defects; less wound complications but higher recurrence than Rives-Stoppa for larger defects |
| Robotic retromuscular (rTAR) | Robotic-assisted transversus abdominis release with retromuscular mesh placement | Large/complex hernias; combines minimally invasive approach with ideal retromuscular mesh position |
| Component separation — Anterior (Ramirez) | Release of external oblique aponeurosis 1–2 cm lateral to the semilunar line, allowing medial advancement of the rectus complex up to 10 cm per side | Large defects with loss of domain; often combined with retromuscular mesh |
| Component separation — Posterior (TAR) | Release of the transversus abdominis muscle from the posterior rectus sheath, creating a large retromuscular space for mesh | Preferred over anterior release in many centers; better preserves abdominal wall vasculature |
16 Hiatal Hernia & Fundoplication
Hiatal Hernia Classification
| Type | Description | Frequency | Management |
|---|---|---|---|
| I — Sliding | GE junction migrates upward through the hiatus; the stomach remains in its normal axial alignment | ~95% | Medical GERD management; surgery (fundoplication) if refractory |
| II — Paraesophageal (rolling) | GE junction remains in normal position; the gastric fundus herniates alongside the esophagus through the hiatus | ~2% | Surgical repair (risk of volvulus, incarceration, strangulation) |
| III — Combined | Both the GE junction and the fundus herniate (elements of both Type I and II) | ~2% | Surgical repair (most common type seen in operative practice) |
| IV — Complex | Other organs (colon, spleen, omentum, small bowel) herniate through the hiatus alongside the stomach | <1% | Surgical repair (always) |
Nissen Fundoplication (360-degree Wrap)
The standard anti-reflux procedure. Performed laparoscopically: 5 ports. The esophagus is mobilized from the hiatus to achieve at least 2–3 cm of intra-abdominal esophageal length. The short gastric vessels are divided (using an ultrasonic device) to mobilize the fundus. The crura are closed posteriorly with permanent suture (0 or 2-0 Ethibond/Prolene). The fundus is wrapped 360 degrees around the distal esophagus ("shoe-shine" test confirms the wrap is floppy) and sutured (2–3 stitches, incorporating the anterior esophageal wall). A 56–60 Fr bougie is placed through the wrap to ensure it is not too tight. The wrap should be 2–3 cm long. Partial wraps (Toupet = 270 degrees posterior, Dor = 180 degrees anterior) are used in patients with esophageal dysmotility to reduce dysphagia risk.
17 Rare Hernias
| Hernia | Location / Definition | Key Features |
|---|---|---|
| Spigelian hernia | Through the spigelian fascia (lateral border of the rectus sheath) at the arcuate line level | Interparietal (between muscle layers) — often not palpable; high incarceration risk; diagnosed by CT or US; laparoscopic repair preferred |
| Obturator hernia | Through the obturator canal (obturator foramen of the pelvis) | Classic patient: thin, elderly woman; Howship-Romberg sign (medial thigh pain with internal rotation of the hip); often presents as SBO; diagnosed by CT; requires surgical reduction and repair |
| Littre's hernia | Any hernia sac containing a Meckel's diverticulum | Meckel's diverticulum as the sole content of the hernia sac; treat with hernia repair + diverticulectomy |
| Richter's hernia | Partial circumference of the bowel wall (antimesenteric border) is incarcerated | May strangulate and perforate without causing complete obstruction (lumen not fully occluded); classically occurs in femoral hernias |
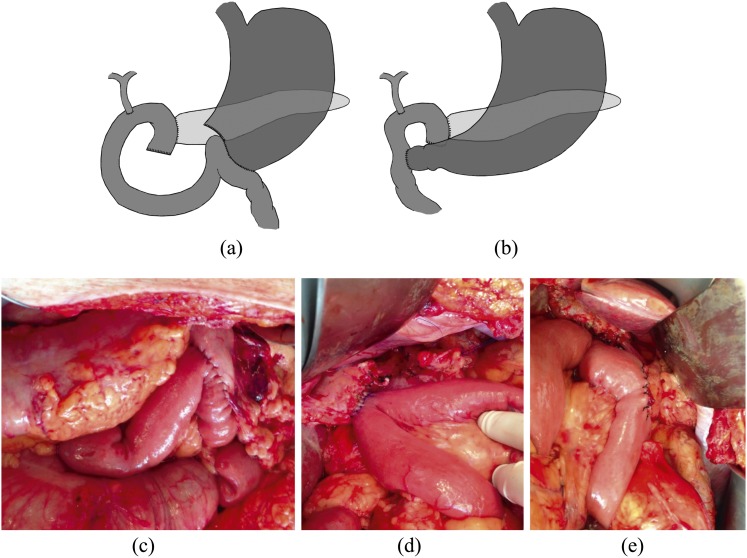
| Amyand's hernia | Inguinal hernia sac containing the appendix | If appendix is inflamed → appendectomy + hernia repair (without mesh due to contamination); if normal → hernia repair alone |
| Petersen's hernia | Internal hernia through the Petersen defect (behind a Roux limb, between the mesentery and transverse mesocolon) | Occurs after Roux-en-Y gastric bypass; presents as SBO; CT shows mesenteric swirl sign; requires emergent surgery; defects should be closed at the index operation |
| Lumbar hernia | Through the superior (Grynfeltt-Lesshaft) or inferior (Petit's) lumbar triangle | Rare; may be acquired (post-flank surgery, trauma) or congenital; palpable bulge in the flank that increases with straining |
18 Cholelithiasis & Biliary Disease
Gallstones are present in 10–15% of the US population; most (80%) are asymptomatic. Cholesterol stones (80%) form from supersaturated bile (excess cholesterol, decreased bile salts, gallbladder hypomotility). Pigment stones (20%) — black pigment stones (chronic hemolysis, cirrhosis) form in the gallbladder; brown pigment stones (biliary stasis, infection with beta-glucuronidase-producing bacteria) form in the bile ducts.
Clinical Presentations of Biliary Disease
| Condition | Pathophysiology | Presentation | Management |
|---|---|---|---|
| Biliary colic | Stone transiently impacted in the cystic duct | Episodic RUQ/epigastric pain, 30 min to several hours, postprandial, resolves spontaneously; no fever, normal labs | Elective cholecystectomy (prevents progression to cholecystitis) |
| Acute cholecystitis | Persistent cystic duct obstruction → gallbladder inflammation/infection | Persistent RUQ pain >6 hours, fever, Murphy's sign, leukocytosis | Early laparoscopic cholecystectomy (see section 06) |
| Choledocholithiasis | Stone in the CBD | Jaundice, elevated bilirubin/ALP/GGT; may cause acute pancreatitis if stone impacts at the ampulla | ERCP with sphincterotomy + stone extraction (pre- or post-cholecystectomy); laparoscopic CBD exploration is an alternative |
| Cholangitis | Bacterial infection of the biliary tree from obstruction (CBD stone, stricture, malignancy) | Charcot's triad (RUQ pain, fever, jaundice) — 50–70% sensitivity; Reynolds' pentad (adds hypotension + altered mental status) — indicates suppurative cholangitis, a surgical emergency | IV antibiotics + urgent biliary decompression (ERCP with sphincterotomy is first-line; percutaneous transhepatic cholangiography/drainage if ERCP fails) |
| Mirizzi syndrome | Stone impacted in the cystic duct or Hartmann's pouch compresses the CHD externally | Obstructive jaundice mimicking CBD stone or malignancy; MRCP/ERCP shows extrinsic compression of the CHD | Cholecystectomy (technically challenging, high conversion rate); Type I (external compression) — subtotal cholecystectomy may be safest; Types II–IV (cholecystobiliary fistula with progressive erosion into the CHD) — may require choledochoplasty or hepaticojejunostomy |
| Gallstone ileus | Large gallstone erodes through gallbladder wall into the duodenum (cholecystoduodenal fistula) → impacts in the terminal ileum | SBO in an elderly patient; Rigler's triad on CT: pneumobilia, SBO, ectopic gallstone | Enterolithotomy (remove the stone via enterotomy at the site of impaction); cholecystectomy/fistula repair at same operation or delayed (controversial) |
19 Hepatocellular Carcinoma
HCC is the most common primary liver malignancy worldwide. Major risk factors: chronic hepatitis B (even without cirrhosis — the virus is directly oncogenic), chronic hepatitis C, cirrhosis of any etiology (alcohol, NASH/MAFLD, autoimmune), aflatoxin B1 exposure (Aspergillus in contaminated grains — common in sub-Saharan Africa and Southeast Asia). Screening: ultrasound ± AFP every 6 months in all cirrhotic patients and chronic HBV carriers.
BCLC Staging System
| Stage | Tumor Burden | Liver Function | Performance Status | Treatment | Median Survival |
|---|---|---|---|---|---|
| 0 (Very early) | Single ≤2 cm, no vascular invasion | Child-Pugh A | ECOG 0 | Resection, ablation, or transplant | >5 years |
| A (Early) | Single or up to 3 nodules ≤3 cm each | Child-Pugh A–B | ECOG 0 | Resection (if adequate FLR), ablation (RFA/MWA), or transplant (Milan criteria) | >5 years |
| B (Intermediate) | Multinodular, beyond Milan | Child-Pugh A–B | ECOG 0 | Transarterial chemoembolization (TACE) | ~2.5 years |
| C (Advanced) | Portal invasion, extrahepatic spread | Child-Pugh A–B | ECOG 1–2 | Systemic therapy (atezolizumab + bevacizumab first-line per IMbrave150 trial; PMID: 32402160) | ~1 year |
| D (Terminal) | Any | Child-Pugh C | ECOG 3–4 | Best supportive care | <3 months |
Resection Criteria
Candidates for hepatic resection: no cirrhosis or well-compensated cirrhosis (Child-Pugh A), adequate future liver remnant (FLR) — generally ≥20% for normal liver, ≥30% for post-chemotherapy liver, ≥40% for cirrhotic liver — no portal hypertension (hepatic venous pressure gradient ≤10 mmHg), normal bilirubin, ECOG 0–1. If FLR is insufficient, portal vein embolization (PVE) of the side to be resected induces contralateral hypertrophy over 4–6 weeks (typically a 30–40% volume increase).
Milan Criteria for Transplant
A patient with HCC can receive a liver transplant if: single tumor ≤5 cm OR up to 3 tumors each ≤3 cm, no macrovascular invasion, no extrahepatic disease. 5-year post-transplant survival: ~70%. Expanded criteria (UCSF criteria, up-to-seven) are used at some centers but remain controversial. Bridging therapy (TACE, ablation) is used to prevent tumor progression while awaiting transplant.
20 Liver Metastases
The liver is the most common site of distant metastases from GI malignancies due to portal venous drainage. Colorectal liver metastases (CRLM) are the most important surgical indication — up to 25% of CRC patients have synchronous liver metastases at diagnosis, and another 25% develop metachronous metastases. Without treatment, median survival with CRLM is 6–12 months. With complete surgical resection, 5-year survival is 40–60% — making liver resection for CRLM one of the greatest achievements in surgical oncology.
Resectability Criteria
The current paradigm focuses not on what must be removed but on what must be left behind: (1) the ability to achieve complete resection (R0) of all disease, (2) preservation of an adequate FLR (≥20% with normal parenchyma) with intact vascular inflow, outflow, and biliary drainage, (3) at least two contiguous segments of liver remain. Number of metastases, size, and bilateral distribution are no longer absolute contraindications — what matters is whether R0 resection is achievable with adequate FLR. Neoadjuvant chemotherapy (FOLFOX, FOLFIRI ± biologics) can convert initially unresectable disease to resectable in 15–30% of patients.
Ablation (RFA or microwave ablation) is used for small (<3 cm) tumors that are surgically difficult to access or as an adjunct to resection. Hepatic artery infusion (HAI) chemotherapy delivers high-dose floxuridine directly to liver metastases via an implanted pump — used at specialized centers for unresectable CRLM.
21 Pancreatic Cancer
Pancreatic ductal adenocarcinoma (PDAC) is the third-leading cause of cancer death in the US with a 5-year survival of ~12%. Approximately 80% present with locally advanced or metastatic disease. Risk factors: smoking (2× risk), chronic pancreatitis, family history (BRCA2, PALB2, Lynch syndrome, FAMMM/CDKN2A, Peutz-Jeghers), new-onset diabetes in patients >50 (may be a paraneoplastic manifestation), obesity.
Resectability Criteria (NCCN)
| Category | Arterial Involvement | Venous Involvement | Approach |
|---|---|---|---|
| Resectable | No arterial contact (celiac, SMA, CHA) | No venous contact or ≤180-degree contact with SMV/PV without contour irregularity | Upfront surgery ± adjuvant chemo |
| Borderline resectable | ≤180-degree contact with SMA/celiac; contact with CHA without extension to celiac/proper hepatic artery bifurcation | >180-degree contact with SMV/PV, or ≤180 degrees with contour irregularity or thrombosis allowing reconstruction | Neoadjuvant chemotherapy (FOLFIRINOX or gem/nab-paclitaxel) → restaging → surgery if responding |
| Locally advanced (unresectable) | >180-degree contact with SMA or celiac; aortic involvement | Unreconstructible SMV/PV occlusion | Chemotherapy ± radiation; surgery only if dramatic downstaging |
| Metastatic | Any | Any | Palliative chemotherapy; biliary/gastric bypass for obstruction |
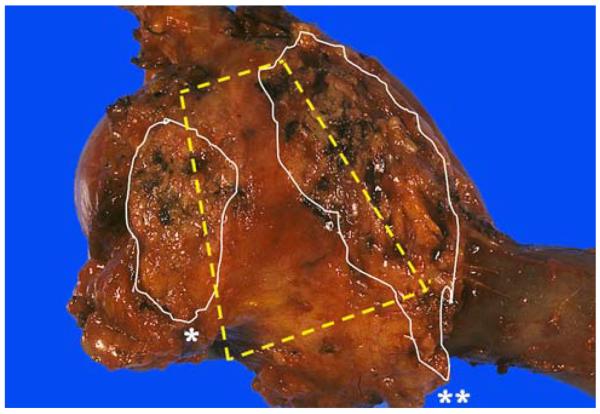
The Whipple Procedure (Pancreaticoduodenectomy)
The standard operation for pancreatic head/uncinate process tumors. Resected structures: pancreatic head, duodenum, distal CBD, gallbladder, and distal stomach (classic Whipple) or with pylorus preservation (pylorus-preserving pancreaticoduodenectomy, PPPD). Three anastomoses are then created: pancreaticojejunostomy (or pancreaticogastrostomy — most critical, as pancreatic fistula is the most feared complication), hepaticojejunostomy (biliary reconstruction), and gastrojejunostomy (or duodenojejunostomy in PPPD). Operative mortality at high-volume centers: 2–4%. The most common complication is delayed gastric emptying (DGE, 20–30%), managed conservatively with NG decompression and prokinetics. Pancreatic fistula (ISGPF grading: biochemical leak = grade A, requires treatment = grade B, organ failure/reoperation = grade C) occurs in 10–20% and may cause hemorrhage from pseudoaneurysm of the GDA stump or hepatic artery branches.
22 Pancreatic Cysts & IPMN
Pancreatic cysts are found incidentally in 2–15% of abdominal imaging studies. The critical question is always: does this cyst harbor or risk developing malignancy?
| Cyst Type | Demographics | Location | Fluid Analysis | Malignant Potential | Management |
|---|---|---|---|---|---|
| Serous cystadenoma (SCA) | Women, 60–70s | Anywhere; classic "honeycomb" microcystic pattern with central stellate scar | Low CEA, low amylase | Negligible (<1%) | Observation unless symptomatic |
| Mucinous cystic neoplasm (MCN) | Women, 40–50s | Body/tail (almost exclusively) | Elevated CEA (>192 ng/mL), low amylase; does NOT communicate with pancreatic duct | 15–30% | Surgical resection (distal pancreatectomy) |
| IPMN — Main duct | Men = women, 60–70s | Main pancreatic duct dilation ≥5 mm | Elevated CEA, elevated amylase (communicates with duct) | High (40–70%) | Surgical resection recommended |
| IPMN — Branch duct | Men = women, 60–70s | Side branch off the main duct; grape-like cluster | Elevated CEA, elevated amylase | Lower (15–25%) | Surveillance vs surgery based on high-risk stigmata/worrisome features |
| Solid pseudopapillary neoplasm (SPN) | Young women, 20–30s | Anywhere; solid and cystic components | Variable | Low-grade malignant (10–15% metastasize) | Surgical resection |
IPMN surveillance (Fukuoka/Revised 2017 guidelines): High-risk stigmata (main duct ≥10 mm, enhancing solid component, obstructive jaundice) → surgery. Worrisome features (cyst ≥3 cm, thickened cyst walls, main duct 5–9 mm, non-enhancing mural nodule, lymphadenopathy, abrupt change in duct caliber, cyst growth rate ≥5 mm/2 years) → EUS with FNA. No worrisome features → surveillance with MRI/MRCP (frequency varies by cyst size: <1 cm every 2 years, 1–2 cm yearly, 2–3 cm every 6–12 months).
23 Colorectal Cancer
Third most common cancer and second leading cause of cancer death in the US. The adenoma-carcinoma sequence (APC → KRAS → p53) accounts for ~70% of CRC; the microsatellite instability (MSI-H / Lynch syndrome) pathway accounts for ~15%. Screening: colonoscopy every 10 years beginning at age 45 (USPSTF 2021). Average time from adenoma to carcinoma: 10–15 years.
TNM Staging (AJCC 8th Edition, Simplified)
| Stage | T | N | M | 5-Year Survival |
|---|---|---|---|---|
| I | T1 (submucosa) or T2 (muscularis propria) | N0 | M0 | ~90% |
| IIA | T3 (subserosa/pericolorectal tissue) | N0 | M0 | ~80% |
| IIB | T4a (visceral peritoneum) or T4b (adjacent organs) | N0 | M0 | ~70% |
| IIIA | T1–T2 | N1 (1–3 nodes) | M0 | ~70% |
| IIIB | T3–T4a | N1 | M0 | ~50–60% |
| IIIC | Any T | N2 (≥4 nodes) | M0 | ~30–40% |
| IVA | Any T | Any N | M1a (1 site) | ~15% |
| IVB | Any T | Any N | M1b (≥2 sites) or M1c (peritoneal) | ~5% |
Surgical Principles
Curative resection requires: en bloc removal of the tumor with adequate margins (at least 5 cm proximal and distal for colon cancer; for rectal cancer, a 1–2 cm distal margin is acceptable with total mesorectal excision), ligation of the named feeding vessel at its origin (to include the regional lymph nodes), and harvest of at least 12 lymph nodes for adequate staging. Specific operations by tumor location: cecum/ascending colon → right hemicolectomy (ileocolic, right colic, and right branch of middle colic vessels); transverse colon → extended right hemicolectomy; splenic flexure/descending colon → left hemicolectomy; sigmoid → sigmoid colectomy (IMA ligation); rectum → low anterior resection (LAR) with total mesorectal excision (TME) for mid/upper rectal tumors, or abdominoperineal resection (APR) with permanent colostomy for very low tumors unable to achieve a safe distal margin.
Neoadjuvant Therapy for Rectal Cancer
Locally advanced rectal cancer (T3/T4 or node-positive on MRI) receives neoadjuvant therapy before surgery. The current standard is total neoadjuvant therapy (TNT): systemic chemotherapy (FOLFOX or CAPOX) followed by chemoradiation (5-FU or capecitabine with long-course radiation 50.4 Gy), or short-course radiation (25 Gy in 5 fractions) followed by systemic chemotherapy. Pathologic complete response (pCR = ypT0N0) occurs in 15–30% and is associated with excellent prognosis. A watch-and-wait (organ preservation) approach is being studied for clinical complete responders after TNT (OPRA trial; PMID: 35247940).
24 Diverticular Disease
Colonic diverticula are acquired pseudodiverticula (mucosa and submucosa herniating through the muscular wall at points of penetration of the vasa recta). Most common in the sigmoid colon (highest intraluminal pressure due to smallest diameter — Laplace's law). Prevalence increases with age: ~50% by age 60, ~70% by age 80. Risk factors: low-fiber diet, obesity, physical inactivity, smoking, NSAIDs.
Hinchey Classification — Complicated Diverticulitis
| Stage | Description | Management |
|---|---|---|
| I | Pericolic/mesenteric abscess | IV antibiotics; percutaneous drainage if abscess >3–4 cm |
| II | Pelvic, distant, or retroperitoneal abscess | IV antibiotics + percutaneous drainage; delayed elective sigmoid colectomy |
| III | Generalized purulent peritonitis (perforated diverticulum with purulent contamination) | Emergent surgery: Hartmann's procedure (sigmoid colectomy, end colostomy, rectal stump) or resection with primary anastomosis ± diverting loop ileostomy; laparoscopic lavage has been trialed but is controversial |
| IV | Generalized fecal peritonitis (free perforation with fecal spillage) | Emergent surgery: Hartmann's procedure (highest morbidity/mortality category) |
Elective sigmoid colectomy is no longer recommended based solely on the number of episodes. Current indications: complicated diverticulitis (fistula, stricture, abscess requiring drainage), recurrent attacks with significant quality-of-life impairment, immunosuppressed patients (higher perforation risk), and inability to exclude malignancy. The sigmoid colon is resected, and a primary colorectal anastomosis is created (the proximal resection margin should extend to where the colon is soft and nondiseased; the distal margin must be at the rectum — leaving behind diseased sigmoid is associated with recurrence).
25 Inflammatory Bowel Disease — Surgical Management
Crohn's Disease
Transmural, skip-lesion inflammation that can affect any part of the GI tract (most commonly the terminal ileum and colon). Surgical indications: failure of medical therapy, stricture causing obstruction, abscess, fistula (enteroenteric, enterocutaneous, enterovesical, enterovaginal), perforation, hemorrhage, and cancer (increased CRC risk with colonic Crohn's). Surgical principle: conserve bowel length — limited resection of only grossly diseased segments with microscopically positive margins accepted (unlike cancer surgery). Strictureplasty (Heineke-Mikulicz for short strictures <10 cm, Finney for 10–25 cm) opens a stricture without removing bowel. Recurrence occurs in ~50% at 10 years, most commonly at the anastomosis — endoscopic surveillance is recommended.
Ulcerative Colitis
Continuous mucosal inflammation limited to the colon and rectum. Surgical indications: failure of medical therapy (refractory to biologics, immunomodulators), toxic megacolon, massive hemorrhage, dysplasia/cancer (15–20% lifetime CRC risk in pancolitis), growth retardation in children. Definitive operation: total proctocolectomy with ileal pouch-anal anastomosis (IPAA/J-pouch) — the entire colon and rectum are removed, and a J-shaped reservoir is constructed from the terminal ileum and anastomosed to the anal canal (at the dentate line, preserving the anal sphincter mechanism). Usually performed in 2–3 stages: (1) total abdominal colectomy with end ileostomy (in the emergent/acute setting), (2) completion proctectomy with J-pouch creation and diverting loop ileostomy, (3) ileostomy reversal. Main long-term complication: pouchitis (~30–50% of patients) — treated with antibiotics (metronidazole, ciprofloxacin).
26 Anorectal Disease
Hemorrhoids
Internal hemorrhoids arise above the dentate line (visceral innervation — painless unless thrombosed/strangulated); graded I (bleeding only), II (prolapse with spontaneous reduction), III (prolapse requiring manual reduction), IV (irreducible). Treatment: Grade I–II → dietary fiber, topical agents, rubber band ligation (most common office procedure — necrosis and slough of the hemorrhoid over 5–7 days). Grade III → rubber band ligation or hemorrhoidectomy. Grade IV → excisional hemorrhoidectomy (Ferguson = closed, Milligan-Morgan = open). Stapled hemorrhoidopexy (PPH) has higher recurrence. External hemorrhoids are below the dentate line (somatic innervation — painful). Acute thrombosed external hemorrhoid within 48–72 hours → elliptical excision (not just incision and drainage — incomplete drainage leads to recurrence).
Anal Fissure
A tear in the anoderm, most commonly in the posterior midline (due to relatively poor blood supply from the inferior rectal artery at the posterior commissure). Atypical location (lateral, multiple, or anterior in men) should raise suspicion for Crohn's disease, HIV, syphilis, or malignancy. Acute fissures (<6 weeks): dietary fiber, warm sitz baths, topical nitroglycerin 0.4% ointment or topical diltiazem 2% cream (relax the internal anal sphincter, improving blood flow). Chronic fissures (>6 weeks, with sentinel pile, hypertrophied anal papilla, and visible internal sphincter fibers): botulinum toxin injection (20–30 units into the internal sphincter) or lateral internal sphincterotomy (LIS — partial division of the internal anal sphincter from the intersphincteric groove — definitive treatment with >95% healing rate; small risk of incontinence to flatus).
Anorectal Abscess & Fistula
Abscesses originate from infection of the anal glands (cryptoglandular origin). Types by location: perianal (most common, superficial), ischiorectal (lateral to the external sphincter), intersphincteric (between internal and external sphincter), and supralevator (above the levator ani — rare, complex). Treatment: incision and drainage — all anorectal abscesses require surgical drainage; antibiotics alone are insufficient (except in immunocompromised patients or overlying cellulitis without fluctuance). Approximately 30–50% of drained abscesses develop a fistula-in-ano.
Goodsall's Rule
Predicts the course of a fistula tract based on the external opening: if the external opening is anterior to an imaginary transverse line through the anus → the fistula tract runs radially (straight) to the nearest crypt. If the external opening is posterior → the tract curves to the posterior midline. Exception: anterior openings >3 cm from the anal verge may also curve posteriorly.
Fistula management depends on the relationship to the sphincter complex: intersphincteric or low transsphincteric (involving <30% of the external sphincter) → fistulotomy (lay open). High transsphincteric, suprasphincteric, or extrasphincteric → sphincter-sparing techniques: seton placement (draining seton to allow maturation, then cutting seton or staged fistulotomy), LIFT (ligation of the intersphincteric fistula tract), endorectal advancement flap, fibrin glue, or anal fistula plug. MRI pelvis is the gold standard for mapping complex fistulas (Parks classification).
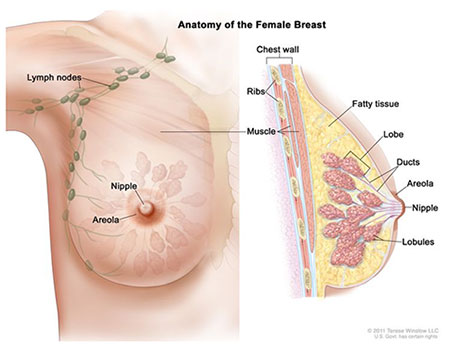
27 Breast Cancer
Breast cancer is the most common cancer in women (excluding skin cancer) and the second leading cause of cancer death in women. Lifetime risk: ~13% (1 in 8 women). Screening mammography (annual or biennial beginning at age 40–50, depending on guidelines) reduces breast cancer mortality by 20–30%. High-risk patients (BRCA1/2 carriers, >20% lifetime risk) should receive annual mammography plus breast MRI beginning at age 25–30.
Molecular Subtypes
| Subtype | ER/PR | HER2 | Ki-67 | Prognosis | Treatment |
|---|---|---|---|---|---|
| Luminal A | +/+ | − | Low (<20%) | Best | Endocrine therapy (tamoxifen or aromatase inhibitor) |
| Luminal B | +/+ (often lower) | − or + | High (>20%) | Intermediate | Endocrine therapy + chemotherapy ± anti-HER2 |
| HER2-enriched | −/− | + | High | Aggressive (improved with targeted therapy) | Chemotherapy + anti-HER2 (trastuzumab ± pertuzumab) |
| Triple-negative (basal-like) | −/− | − | High | Worst (highest recurrence, no targeted therapy) | Chemotherapy (anthracycline + taxane); immunotherapy (pembrolizumab) if PD-L1+ |
Surgical Options
Breast-conserving surgery (BCS/lumpectomy) + whole breast radiation is oncologically equivalent to mastectomy for early-stage breast cancer (stages I–II) — established by the NSABP B-06 trial (PMID: 12065600) with 20-year follow-up confirming equivalent survival. BCS requires: (1) negative margins (no ink on tumor for invasive disease, 2 mm margin for DCIS), (2) ability to achieve acceptable cosmetic result, (3) patient can receive radiation. Contraindications to BCS: multicentric disease, diffuse microcalcifications, inability to achieve negative margins, prior chest wall radiation, pregnancy (relative — radiation delayed until postpartum).
Mastectomy: total (simple) mastectomy removes all breast tissue including the nipple-areolar complex. Modified radical mastectomy adds level I/II axillary lymph node dissection. Skin-sparing and nipple-sparing mastectomy are oncologically equivalent for appropriate candidates and allow immediate reconstruction.
Sentinel Lymph Node Biopsy (SLNB)
The sentinel node is the first node(s) draining the breast tumor, identified using technetium-99m sulfur colloid (injected pre-op, detected with a gamma probe) and/or isosulfan blue/methylene blue dye (injected intraoperatively). If the sentinel node is negative → no further axillary surgery (false-negative rate ~5%). If 1–2 sentinel nodes are positive and the patient is undergoing BCS + radiation → no completion axillary lymph node dissection (ALND) is needed (ACOSOG Z0011 trial; PMID: 21304082). If ≥3 positive sentinel nodes, or positive nodes with mastectomy without radiation planned → completion ALND.
28 Benign Breast Disease
| Condition | Age/Demographics | Features | Cancer Risk | Management |
|---|---|---|---|---|
| Fibroadenoma | 15–35 years | Firm, rubbery, well-circumscribed, mobile ("breast mouse"); hormonally responsive (may enlarge during pregnancy) | No increased risk (unless complex/proliferative) | Observation if <2 cm and classic features on imaging/biopsy; excision if enlarging, >3 cm, or atypical histology |
| Fibrocystic changes | 30–50 years | Cyclic breast pain and lumpiness, worse premenstrually; may include cysts, fibrosis, adenosis, ductal hyperplasia | No increased risk (unless atypical hyperplasia found on biopsy) | Reassurance, supportive bra, NSAIDs; aspiration of symptomatic cysts |
| Phyllodes tumor | 40–50 years | Large, rapidly growing, smooth, well-circumscribed; leaf-like (phyllodes) pattern on histology; benign, borderline, or malignant variants | Malignant variant metastasizes hematogenously (lungs); does NOT metastasize to lymph nodes | Wide local excision with ≥1 cm margins (negative margins critical — high local recurrence); no role for SLNB; no benefit from adjuvant chemotherapy/radiation for most |
| Intraductal papilloma | 30–50 years | Bloody/serous nipple discharge (most common cause of pathologic nipple discharge); small mass near areola | Slight increase (1.5–2× for solitary; higher for multiple papillomas) | Duct excision (central duct excision for central papilloma, segmental excision for peripheral papillomas) |
| Gynecomastia | Males; puberty, elderly, drugs | Benign proliferation of male breast glandular tissue; distinguish from pseudogynecomastia (fat) and male breast cancer (hard, eccentric, fixed) | Male breast cancer rare but possible | Observation in pubertal boys (resolves in 90%); discontinue offending drugs (spironolactone, marijuana, estrogens, cimetidine); surgical excision if persistent and bothersome |
29 Thyroid — Nodules, Cancer, & Thyroidectomy
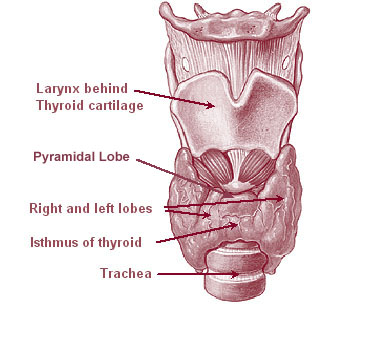
Thyroid Nodule Workup
Thyroid nodules are extremely common (50–65% of the population on ultrasound), but only ~5% are malignant. Workup: TSH first — if low (hyperfunctioning nodule), obtain a thyroid uptake scan (hot nodules are almost never malignant → no biopsy needed). If TSH is normal or high → ultrasound to assess features and guide biopsy. Suspicious ultrasound features (ACR TI-RADS): solid composition, hypoechogenicity, taller-than-wide shape, irregular margins, echogenic foci (microcalcifications). FNA biopsy is indicated based on nodule size and TI-RADS score (TR3 ≥2.5 cm, TR4 ≥1.5 cm, TR5 ≥1 cm).
Bethesda System for Reporting Thyroid Cytopathology
| Category | Diagnosis | Malignancy Risk | Usual Management |
|---|---|---|---|
| I | Nondiagnostic/unsatisfactory | 5–10% | Repeat FNA in 4–6 weeks |
| II | Benign | 0–3% | Clinical/ultrasound follow-up |
| III | Atypia of undetermined significance (AUS) / Follicular lesion of undetermined significance (FLUS) | 10–30% | Repeat FNA, molecular testing (Afirma, ThyroSeq), or diagnostic lobectomy |
| IV | Follicular neoplasm / Suspicious for follicular neoplasm | 25–40% | Diagnostic lobectomy (cannot distinguish follicular adenoma from carcinoma on cytology — need surgical pathology to assess capsular/vascular invasion) |
| V | Suspicious for malignancy | 50–75% | Lobectomy or total thyroidectomy |
| VI | Malignant | 97–99% | Total thyroidectomy (for tumors >1 cm, bilateral, or aggressive histology) or lobectomy (for <1 cm low-risk papillary carcinoma) |
Thyroid Cancer Types
| Type | % of Thyroid Cancers | Features | Prognosis |
|---|---|---|---|
| Papillary | 80–85% | Psammoma bodies on histology, "Orphan Annie" nuclei; lymph node metastases common; multifocal in 30% | Excellent (10-year survival >95%) |
| Follicular | 10–15% | Hematogenous spread (bone, lung) rather than lymphatic; Hurthle cell variant is more aggressive | Good (10-year survival ~90%) |
| Medullary | 3–5% | Arises from parafollicular C cells; produces calcitonin (tumor marker); 25% hereditary (MEN2A, MEN2B, FMTC — RET proto-oncogene mutations); amyloid deposits on histology | Moderate (10-year survival ~75%); does NOT respond to RAI |
| Anaplastic | 1–2% | Rapidly enlarging neck mass in elderly, locally invasive (trachea, esophagus, vessels); often on background of prior differentiated thyroid cancer | Dismal (median survival 3–6 months); often unresectable; may respond to BRAF + MEK inhibitor (dabrafenib + trametinib) if BRAF V600E mutated |
Thyroidectomy Complications
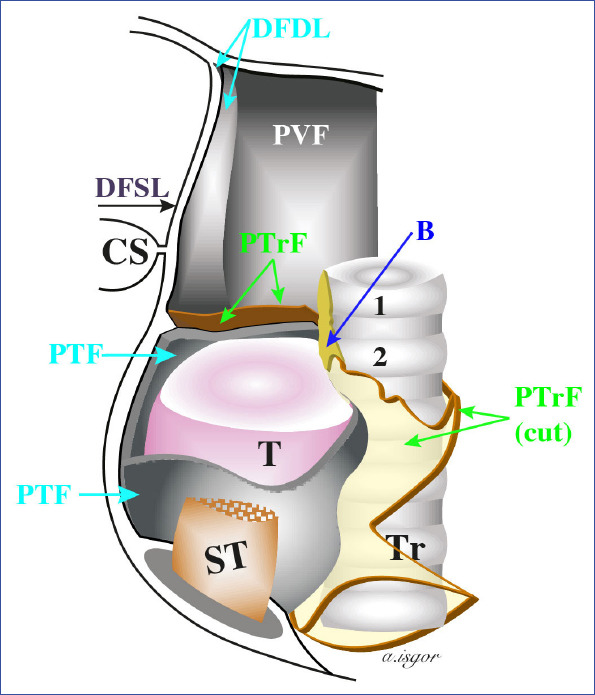
Recurrent laryngeal nerve (RLN) injury: unilateral → hoarseness (vocal cord paralysis); bilateral → airway obstruction requiring emergent reintubation or tracheostomy. Risk: 1–2% for experienced surgeons; intraoperative nerve monitoring (NIM) is used to identify the nerve but has not been definitively proven to reduce injury rates. The RLN runs in the tracheoesophageal groove; the nerve is identified and preserved by dissecting along its course — the nerve is "found, not sought" (systematic identification rather than blind avoidance). The non-recurrent laryngeal nerve (direct branch from the vagus, occurs in ~0.5% on the right, associated with aberrant right subclavian artery) is at high risk of injury if not recognized.
Hypoparathyroidism: transient (10–40%) or permanent (<2%) hypocalcemia from parathyroid injury/devascularization. Symptoms: perioral/digital tingling, Chvostek's sign (facial nerve tapping → facial muscle twitch), Trousseau's sign (BP cuff → carpopedal spasm). Treatment: IV calcium gluconate for symptomatic hypocalcemia; oral calcium carbonate + calcitriol for ongoing supplementation. Devascularized parathyroid glands should be autotransplanted (minced and placed into the sternocleidomastoid or forearm muscle).
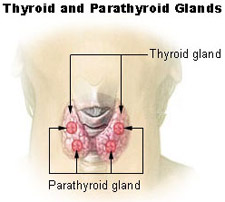
30 Parathyroid & Hyperparathyroidism
Primary hyperparathyroidism is the most common cause of hypercalcemia in the outpatient setting. Caused by a single parathyroid adenoma in 85%, four-gland hyperplasia in 10–15% (especially in MEN1/2A and familial hyperparathyroidism), double adenoma in 3–5%, and parathyroid carcinoma in <1%. Presentation: "bones, stones, groans, and psychiatric overtones" — osteoporosis/osteitis fibrosa cystica, nephrolithiasis/nephrocalcinosis, abdominal pain/constipation/pancreatitis, depression/confusion/fatigue. Many patients are asymptomatic (detected on routine bloodwork: elevated calcium + elevated or inappropriately normal PTH).
Preoperative Localization
Sestamibi scan (technetium-99m sestamibi with SPECT): the tracer is taken up by both thyroid and parathyroid tissue, but washes out faster from the thyroid — persistent uptake at 2–3 hours indicates a parathyroid adenoma (sensitivity ~80% for single adenoma). 4D-CT of the neck: multiplanar imaging with contrast showing the characteristic rapid enhancement and washout pattern of parathyroid adenomas (sensitivity ~90%). Ultrasound: identifies 60–80% of adenomas; operator-dependent but no radiation. Concordant localization on two studies allows a focused (minimally invasive) parathyroidectomy — targeted exploration of the identified gland through a small incision, confirmed by intraoperative PTH monitoring (Miami criterion: PTH drops >50% from the highest pre-excision level at 10 minutes post-excision → cure). If localization is discordant or negative → bilateral neck exploration (all four glands identified).
Surgical Indications (Asymptomatic Primary Hyperparathyroidism — 2014 Guidelines)
Surgery recommended if any of: calcium >1 mg/dL above upper limit of normal, eGFR <60, T-score ≤ −2.5 at any site (or vertebral fracture), age <50, 24-hour urine calcium >400 mg/day, or nephrolithiasis/nephrocalcinosis on imaging. Parathyroidectomy is the only definitive cure — medical management (cinacalcet for calcium reduction, bisphosphonates for bone protection) is reserved for patients who are not surgical candidates.
31 Adrenal Surgery
Adrenal Incidentaloma Workup
Adrenal masses found incidentally on abdominal imaging occur in ~5% of CT scans. Every incidentaloma requires two assessments: (1) Is it hormonally active? and (2) Is it malignant? Hormonal workup: 1 mg overnight dexamethasone suppression test (screening for subclinical Cushing's — cortisol >1.8 mcg/dL is abnormal), 24-hour urine catecholamines/metanephrines or plasma free metanephrines (screening for pheochromocytoma), and aldosterone-to-renin ratio (if hypertensive — screening for Conn's syndrome/primary aldosteronism). Malignancy assessment: size is the strongest predictor (<4 cm → ~2% malignancy; 4–6 cm → ~6%; >6 cm → ~25%). CT characteristics: benign adenomas are homogeneous, lipid-rich (≤10 HU on unenhanced CT, >60% absolute contrast washout at 15 minutes); malignant features include size >4 cm, >10 HU, irregular borders, heterogeneity, and growth on follow-up imaging.
Pheochromocytoma
"Rule of 10s" (approximate, based on older data): 10% bilateral, 10% extra-adrenal (paraganglioma), 10% malignant, 10% pediatric, 10% familial (MEN2A/2B, VHL, NF1, SDH mutations). Classic presentation: episodic hypertension, headache, sweating, palpitations, pallor. Preoperative preparation is mandatory: alpha-blockade first (phenoxybenzamine 10–20 mg BID, uptitrated over 10–14 days until orthostatic hypotension develops; OR doxazosin for a shorter-acting alternative), then beta-blockade (only AFTER alpha-blockade is established — unopposed beta-blockade causes hypertensive crisis from unchecked alpha-mediated vasoconstriction), high-sodium diet and IV fluids to expand intravascular volume. Laparoscopic adrenalectomy is the standard approach; the anesthesiologist must be prepared for intraoperative hypertensive surges (nitroprusside, nicardipine, phentolamine) and post-resection hypotension (volume, vasopressors).
Conn's Syndrome (Primary Aldosteronism)
The most common cause of secondary hypertension (5–10% of all hypertensive patients). Presents with hypertension + hypokalemia (though many are normokalemic). Screening: aldosterone-to-renin ratio (ARR) >30 with aldosterone >15 ng/dL. Confirmatory testing: oral sodium loading or IV saline infusion (failure to suppress aldosterone confirms the diagnosis). Subtype differentiation: adrenal vein sampling (AVS) is the gold standard to lateralize aldosterone production (unilateral adenoma vs bilateral hyperplasia) — critical because unilateral disease → laparoscopic adrenalectomy (curative in 30–60% of patients for hypertension), while bilateral hyperplasia → medical management with spironolactone (25–100 mg daily) or eplerenone (25–50 mg BID).
32 Melanoma
The most lethal skin cancer, accounting for only ~5% of skin cancers but the majority of skin cancer deaths. Incidence is rising. Risk factors: UV exposure (intermittent intense exposure → sunburns), fair skin (Fitzpatrick types I–II), numerous moles (>50), atypical/dysplastic nevi, family history (CDKN2A mutations), prior melanoma, and immunosuppression. The ABCDEs of melanoma: Asymmetry, Border irregularity, Color variation, Diameter >6 mm, Evolving.
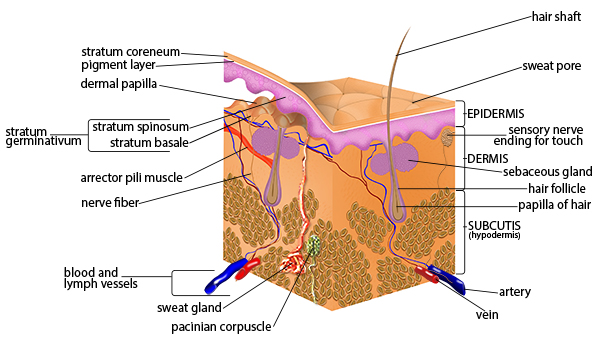
Breslow Depth & Wide Excision Margins
| Breslow Depth (Thickness) | Recommended Excision Margin | SLNB Recommended? |
|---|---|---|
| In situ (melanoma in situ) | 0.5–1 cm | No |
| ≤1.0 mm | 1 cm | Consider if 0.8–1.0 mm, ulcerated, or high mitotic rate |
| 1.01–2.0 mm | 1–2 cm | Yes |
| 2.01–4.0 mm | 2 cm | Yes |
| >4.0 mm | 2 cm | Yes |
Clark Levels (Historical, Largely Replaced by Breslow)
| Level | Depth of Invasion |
|---|---|
| I | Confined to epidermis (in situ) |
| II | Invades papillary dermis |
| III | Fills papillary dermis |
| IV | Invades reticular dermis |
| V | Invades subcutaneous fat |
Sentinel Lymph Node Biopsy in Melanoma
SLNB is the most important prognostic procedure in clinically node-negative melanoma. Indicated for Breslow >0.8 mm or any depth with ulceration, high mitotic rate, or lymphovascular invasion. Technique: intradermal injection of technetium-99m sulfur colloid and/or blue dye around the primary site (or biopsy scar); intraoperative gamma probe localization and visual identification of blue-stained nodes. If SLNB is positive, completion lymph node dissection (CLND) was traditionally performed but the MSLT-II trial (PMID: 28614720) showed no melanoma-specific survival benefit for CLND vs nodal observation with ultrasound surveillance — CLND is now optional, primarily reserved for bulky nodal disease or when surveillance is unreliable.
33 Soft Tissue Sarcoma
Soft tissue sarcomas (STS) are rare (<1% of adult cancers) mesenchymal tumors arising from muscle, fat, fibrous tissue, nerves, or blood vessels. Over 50 histologic subtypes exist; the most common in adults are undifferentiated pleomorphic sarcoma (formerly MFH), liposarcoma, leiomyosarcoma, and synovial sarcoma. Most present as a painless, enlarging mass — any soft tissue mass >5 cm, deep to the fascia, or rapidly growing should be presumed sarcoma until proven otherwise.
Workup: MRI (extremity/trunk) or CT (retroperitoneal) for local staging; chest CT for pulmonary metastases (sarcomas spread hematogenously to the lungs, NOT lymph nodes — SLNB has no role). Core needle biopsy (NOT excisional biopsy for a suspected sarcoma — excisional biopsy contaminates tissue planes and complicates subsequent definitive surgery). Treatment: wide local excision with negative margins (R0, ≥1 cm or intact fascial barrier) is the cornerstone. Adjuvant radiation (50–60 Gy) reduces local recurrence for high-grade, deep, >5 cm tumors. The role of chemotherapy is limited (doxorubicin-based regimens for high-grade, large tumors, especially in extremity sarcoma). Retroperitoneal sarcomas (most commonly liposarcoma or leiomyosarcoma) are resected en bloc with adjacent organs as needed; no proven benefit of radiation or chemotherapy.
34 ATLS & Initial Assessment Emergency
Primary Survey (ABCDE)
| Step | Assessment | Interventions |
|---|---|---|
| A — Airway with C-spine protection | Is the airway patent? Can the patient speak? Stridor? Facial/neck trauma? | Jaw thrust, chin lift, oropharyngeal/nasopharyngeal airway, endotracheal intubation, surgical airway (cricothyroidotomy) if intubation fails |
| B — Breathing | Respiratory rate, chest wall movement, auscultation, tracheal position, JVD, subcutaneous emphysema | Needle decompression (2nd ICS midclavicular line or 5th ICS anterior axillary line) for tension pneumothorax; tube thoracostomy (chest tube, 28–36 Fr, 5th ICS anterior to mid-axillary line) for pneumothorax/hemothorax; occlusive dressing for open pneumothorax |
| C — Circulation | Pulse rate/quality, BP, skin color/temperature, obvious hemorrhage, FAST exam | Two large-bore IVs (16-gauge or larger), blood type and crossmatch, massive transfusion protocol (1:1:1 ratio of pRBC:FFP:platelets), direct pressure on external bleeding, tourniquet for extremity hemorrhage, pelvic binder for unstable pelvic fracture, emergent surgery for non-compressible torso hemorrhage |
| D — Disability | GCS (best eye + verbal + motor response, scale 3–15), pupil size/reactivity, lateralizing signs | GCS ≤8 → intubate for airway protection; identify signs of herniation (unilateral dilated pupil, decerebrate posturing); mannitol 1 g/kg or hypertonic saline for acute herniation |
| E — Exposure/Environment | Fully undress patient, logroll to examine back, prevent hypothermia | Warm blankets, warm IV fluids, warm environment (trauma triad of death: hypothermia, acidosis, coagulopathy) |
FAST Exam (Focused Assessment with Sonography in Trauma)
Four views: RUQ (Morrison's pouch — between liver and right kidney, most dependent space in the supine position), LUQ (splenorenal recess), suprapubic/pelvic (pouch of Douglas or rectovesical space), and subxiphoid (pericardial effusion/tamponade). The E-FAST adds bilateral anterior chest views to detect pneumothorax (absent lung sliding). FAST is for detecting free fluid — it does not grade organ injury. A positive FAST in a hemodynamically unstable blunt trauma patient → immediate exploratory laparotomy.
35 Solid Organ Injury — Spleen & Liver Emergency
Splenic Injury (AAST Grading)
| Grade | Description | Management |
|---|---|---|
| I | Subcapsular hematoma <10% surface area; capsular laceration <1 cm deep | Non-operative management (NOM) — observation, serial exams, serial Hgb |
| II | Subcapsular hematoma 10–50% surface area; intraparenchymal hematoma <5 cm; laceration 1–3 cm deep, no trabecular vessel involvement | NOM in hemodynamically stable patients |
| III | Subcapsular hematoma >50% or expanding; ruptured subcapsular/intraparenchymal hematoma ≥5 cm; laceration >3 cm deep or involving trabecular vessels | NOM if stable ± angioembolization; surgery if unstable |
| IV | Laceration involving segmental or hilar vessels with major devascularization (>25% of spleen) | NOM with angioembolization if stable; splenectomy if unstable or failed NOM |
| V | Completely shattered spleen; hilar vascular injury devascularizing the spleen | Splenectomy (almost always operative) |
Post-splenectomy considerations: vaccinations against encapsulated organisms — Streptococcus pneumoniae (PCV13 then PPSV23), Neisseria meningitidis (MenACWY + MenB), and Haemophilus influenzae type b — ideally 14 days post-splenectomy (or pre-operatively for elective splenectomy). Overwhelming post-splenectomy infection (OPSI) is rare (0.5–1% lifetime risk) but carries >50% mortality. Patients should carry a medical alert card and receive early antibiotics for any febrile illness.
Hepatic Injury (AAST Grading)
| Grade | Description | Management |
|---|---|---|
| I | Subcapsular hematoma <10%; laceration <1 cm deep | NOM |
| II | Subcapsular hematoma 10–50%; intraparenchymal hematoma <10 cm; laceration 1–3 cm deep, <10 cm long | NOM |
| III | Subcapsular hematoma >50% or expanding; intraparenchymal hematoma >10 cm; laceration >3 cm deep | NOM if stable ± angioembolization; surgery if unstable |
| IV | Parenchymal disruption of 25–75% of a lobe; active intraparenchymal bleeding | Angioembolization or surgery (perihepatic packing) |
| V | Parenchymal disruption >75% of a lobe; juxtahepatic venous injury (hepatic veins/retrohepatic IVC) | Emergent surgery — damage control with perihepatic packing, Pringle maneuver, vascular repair; juxtahepatic venous injuries carry very high mortality |
Non-operative management is successful in >85% of blunt liver injuries in hemodynamically stable patients. Indications for angioembolization: CT evidence of active contrast extravasation (blush), large hemoperitoneum, or high-grade injury in a stable patient. Delayed complications of hepatic injury: biloma (contained bile collection → percutaneous drainage), hepatic abscess, hemobilia (blood in the biliary tree → GI bleed with jaundice after liver trauma → angioembolization), and bilhemia (bile entering the venous system through hepatic vein injury).
36 Damage Control Surgery Emergency
Damage control surgery (DCS) is a staged approach used in severely injured patients who are developing or at risk for the "lethal triad" of hypothermia (core temp <35°C), acidosis (pH <7.2, lactate >5, base deficit > −6), and coagulopathy (INR >1.5, fibrinogen <100). The goal is to abbreviate the initial operation to restore physiology rather than complete anatomy.
Three Phases
| Phase | Location | Goals | Details |
|---|---|---|---|
| Phase 1 — Abbreviated laparotomy | OR | Control hemorrhage, control contamination, temporary abdominal closure | Perihepatic packing, splenectomy, ligation of bleeding vessels, stapled bowel resection WITHOUT anastomosis (leave ends in discontinuity), temporary abdominal closure (negative-pressure wound therapy / Bogota bag / Wittmann patch) |
| Phase 2 — Resuscitation | ICU | Correct the lethal triad | Warm the patient (Bair Hugger, warm fluids, warm room), correct acidosis (resuscitate with blood products, fluids, vasopressors), correct coagulopathy (massive transfusion protocol 1:1:1, TXA within 3 hours, fibrinogen replacement, calcium), optimize hemodynamics |
| Phase 3 — Definitive repair | OR (24–72 hours later) | Complete reconstruction | Remove packing (carefully — risk of re-bleeding), create bowel anastomoses, definitive vascular repair, fascia closure (if possible — if not, temporary closure continued with planned serial washouts until closure achieved or abdominal wall reconstruction needed) |
37 Laparoscopic Surgery Fundamentals
Insufflation & Pneumoperitoneum
CO2 is the standard insufflation gas (rapidly absorbed, does not support combustion, inexpensive). Target intra-abdominal pressure: 12–15 mmHg. Pressures >15 mmHg impair venous return (decreased preload), decrease cardiac output, and reduce renal/splanchnic blood flow. Access methods: Veress needle (closed technique — inserted at the umbilicus or Palmer's point (LUQ, left subcostal) in patients with prior midline surgery), Hasson technique (open cut-down — safest in patients with prior surgery or adhesions), and optical trocar (direct visualization through the trocar as it traverses the layers).
Port Placement Principles
The camera port is typically at the umbilicus (natural weak point in the abdominal wall, cosmetically hidden). Working ports are placed so that: (1) instruments approach the target at a 60-degree angle to each other (optimal ergonomics), (2) the camera is between the two working ports (triangulation), (3) ports are at least 5 cm apart to avoid instrument clashing ("sword fighting"), (4) the camera views the operative field in the same orientation as the surgeon's hands. Standard port sizes: 5 mm (instruments), 10/12 mm (camera and stapler ports). Fascial closure is required for all ports ≥10 mm (to prevent port-site hernia — incidence ~1% for 10–12 mm ports).
Complications Specific to Laparoscopy
| Complication | Mechanism | Prevention/Management |
|---|---|---|
| Trocar/Veress needle injury (bowel, vascular) | Blind entry through the abdominal wall; most common with Veress needle | Open (Hasson) technique in high-risk patients; inspect all entry sites at the end of the case; vascular injury to aorta/iliac vessels at Veress entry requires emergent laparotomy |
| CO2 embolism | CO2 enters an open vein (rare but can be fatal) | Mill-wheel murmur on auscultation; Trendelenburg, left lateral decubitus, aspirate gas from central line |
| Subcutaneous emphysema | CO2 tracking into subcutaneous tissue from port sites or preperitoneal insufflation | Self-limited; ensure ports are in the peritoneal cavity; crepitus on exam; resolves with time after desufflation |
| Shoulder pain (referred) | Residual CO2 irritating the diaphragm → referred pain via phrenic nerve (C3–5) | Evacuate as much CO2 as possible at end of case; NSAIDs; self-limited |
38 Bowel Anastomosis
Principles
A successful bowel anastomosis requires: adequate blood supply to both ends (healthy pink bowel, mesenteric pulsation), absence of tension, no distal obstruction, and a well-nourished/non-septic patient. The anastomosis can be performed hand-sewn (interrupted or running sutures — typically 3-0 or 4-0 absorbable, e.g., PDS or Vicryl — single-layer full-thickness or two-layer with an inner absorbable running and outer interrupted seromuscular silk) or stapled (linear stapler for side-to-side/functional end-to-end anastomosis, circular stapler / EEA for end-to-end colorectal anastomosis). Configurations: end-to-end, end-to-side, side-to-side. For small bowel, a side-to-side (functional end-to-end) stapled anastomosis is the most common technique — the antimesenteric borders of both limbs are aligned, a linear stapler (GIA) fires through both walls creating the common channel, and the open end is closed with a linear stapler (TA) or hand-sewn.
Anastomotic Leak
The most feared complication of bowel surgery. Overall leak rates: small bowel 1–3%, ileocolic 3–4%, colorectal 5–15% (higher for low rectal anastomoses — within 5 cm of the anal verge, leak rates up to 15–20%, which is why a diverting loop ileostomy is often created to protect these anastomoses). Presentation: typically postoperative day 5–7, with fever, tachycardia, abdominal pain, ileus, leukocytosis, elevated CRP — or may present insidiously. Diagnosis: CT with oral and rectal contrast (extravasation of contrast, free air, abscess). Management depends on severity: contained leak with small abscess → percutaneous drainage + antibiotics + bowel rest; free peritonitis → re-exploration, washout, and usually takedown of the anastomosis with proximal diverting ostomy (or Hartmann's procedure for left-sided leaks).
39 Ostomy Creation & Management
Types
| Ostomy Type | Location | Output | Indications |
|---|---|---|---|
| End ileostomy | RLQ | Liquid, high-volume (800–1200 mL/day), enzyme-rich (corrosive to skin) | Total proctocolectomy (UC), protection of distal anastomosis if loop not feasible, Crohn's disease |
| Loop ileostomy | RLQ | Same character as end ileostomy | Temporary fecal diversion to protect a distal colorectal/coloanal anastomosis; easily reversed |
| End colostomy | LLQ (sigmoid/descending) | Formed stool, lower volume | Hartmann's procedure (emergent sigmoid resection), APR (permanent), distal obstruction |
| Loop colostomy | Usually transverse colon (RUQ) | Semi-formed stool | Temporary diversion for distal obstruction, perineal wound healing, anorectal sepsis |
Technical Points
The stoma site should be marked preoperatively (by a stoma nurse or surgeon) — ideally within the rectus muscle, away from bony prominences (costal margin, ASIS, iliac crest), skin folds, scars, and the umbilicus. A circular skin disc is excised, the rectus fascia is cruciate-incised, and the rectus muscle is spread (not cut). The bowel is delivered through the abdominal wall without tension. For an ileostomy, 3–5 cm of bowel should protrude above the skin to create a "Brooke" (everted, nippled) stoma — the eversion prevents enzyme-rich effluent from contacting the peristomal skin. For a colostomy, the stoma is matured flush with the skin.
Complications
| Complication | Timing | Features | Management |
|---|---|---|---|
| Ischemia/necrosis | Early (0–48 hours) | Dark/dusky stoma (should be pink/red); assess depth of necrosis with a glass test tube and light | Superficial necrosis → observe; necrosis below fascial level → urgent revision |
| Retraction | Early or late | Stoma sinks below skin level → poor pouching, skin excoriation | Convex pouching system; surgical revision if severe |
| Parastomal hernia | Late | Most common long-term complication (~30–50%); bulge around stoma | Supportive belt; surgical repair with mesh (Sugarbaker or keyhole technique) if symptomatic |
| Prolapse | Late | Excessive bowel protrusion; more common with loop colostomies | Manual reduction; elective revision or conversion to end stoma |
| High-output ileostomy | Any | Output >1500 mL/day → dehydration, electrolyte derangement (hyponatremia, hypokalemia, hypomagnesemia, metabolic acidosis) | Oral rehydration solution (WHO-ORS), loperamide (up to 16 mg/day), codeine phosphate, dietary modification, IV fluids if severe |
40 Central Lines & Surgical Access
Central Venous Catheter (CVC) Insertion
Standard sites: internal jugular vein (IJV) — preferred for most indications, easy ultrasound visualization, compressible; subclavian vein — lowest infection rate, higher pneumothorax risk, not easily compressible (avoid in coagulopathy); femoral vein — easiest access in emergencies, highest infection rate, contraindicated for long-term use. All CVCs should be placed under ultrasound guidance (reduces complications, recommended by multiple societies) and using maximal barrier precautions (cap, mask, sterile gown, sterile gloves, large sterile drape) to reduce central line-associated bloodstream infections (CLABSI).
Seldinger technique: Access the vein with an 18-gauge finder needle under ultrasound guidance → confirm venous blood (dark, non-pulsatile) → advance the J-tip guidewire → remove the needle → make a skin nick → advance the dilator over the wire → remove the dilator → advance the catheter over the wire → remove the wire (ALWAYS maintain control of the wire to prevent wire embolism) → confirm position with CXR (tip at the cavoatrial junction). Immediate complications: pneumothorax (subclavian > IJ), arterial puncture, air embolism (prevented by Trendelenburg positioning), arrhythmia from wire in the heart, hemothorax.
Surgically Implanted Ports & Tunneled Catheters
Port-a-Cath: subcutaneous reservoir with a catheter tunneled into the IJV or subclavian vein; accessed with a Huber needle through the skin. Indicated for long-term intermittent IV access (chemotherapy, TPN). Tunneled catheter (Hickman, Broviac): catheter exits the skin through a subcutaneous tunnel (the tunnel with a Dacron cuff promotes tissue ingrowth, reducing infection and dislodgement). Indicated for long-term continuous access (hemodialysis, prolonged antibiotics). PICC (peripherally inserted central catheter): inserted via the basilic or brachial vein, tip positioned at the cavoatrial junction; for intermediate-term access (weeks to months); risk of upper extremity DVT.
41 Imaging & Diagnostics
| Modality | Primary Use in General Surgery | Key Notes |
|---|---|---|
| Ultrasound (US) | RUQ (gallstones, cholecystitis), FAST (trauma), hernia, breast masses, thyroid nodules, guided procedures | First-line for RUQ pain; operator-dependent; no radiation; real-time guided biopsy/drainage |
| CT abdomen/pelvis | Acute abdomen (appendicitis, diverticulitis, SBO, LBO), staging, abscess localization, trauma | With IV contrast unless contraindicated; oral contrast adds value for bowel obstruction evaluation; look for transition point, free air, free fluid, contrast blush |
| CT angiography (CTA) | Mesenteric ischemia, GI bleeding localization, vascular anatomy for surgical planning | Arterial phase detects active bleeding at ≥0.3 mL/min |
| MRI abdomen | Liver lesion characterization (hepatocyte-specific contrast — gadoxetate/Eovist), rectal cancer staging (T and N staging with high-resolution rectal protocol), soft tissue sarcoma | Superior soft tissue contrast; no radiation; slower, more expensive |
| MRCP | Biliary/pancreatic duct anatomy, CBD stones, pancreatic cysts, strictures | Non-invasive alternative to ERCP for diagnosis (not therapy); heavily T2-weighted images highlight fluid-filled structures |
| HIDA scan | Acute cholecystitis (when US equivocal), biliary dyskinesia (with CCK stimulation — EF <35% is abnormal) | Non-visualization of gallbladder at 4 hours = cystic duct obstruction; morphine augmentation accelerates the study |
| PET/CT (FDG) | Staging and restaging of malignancy (CRC, melanoma, sarcoma), detecting recurrence, assessing treatment response | Identifies metabolically active tissue; false positives with infection/inflammation; false negatives with mucinous tumors and after recent chemotherapy |
| Mammography | Breast cancer screening and diagnostic evaluation | BI-RADS 0–6 classification; digital breast tomosynthesis (3D mammography) improves sensitivity in dense breasts |
| EGD / Colonoscopy | GI hemorrhage (diagnosis + treatment), colorectal cancer screening, biopsy | Both diagnostic and therapeutic; requires bowel prep for colonoscopy; procedural sedation |
| ERCP | CBD stone extraction, biliary stenting, sphincterotomy | Therapeutic endoscopy; 5–10% risk of post-ERCP pancreatitis; rectal indomethacin reduces this risk |
| EUS (endoscopic ultrasound) | Pancreatic mass biopsy, T-staging of GI cancers, IPMN evaluation, submucosal lesion assessment | High-resolution imaging of the GI wall layers and adjacent structures; FNA capability |
42 Classification Systems (All)
| System | Used For | Key Features |
|---|---|---|
| Alvarado (MANTRELS) score | Acute appendicitis | 10-point scoring system; ≥7 = highly probable (see Section 05) |
| Tokyo Guidelines (TG18) | Acute cholecystitis severity | Grade I (mild), II (moderate), III (severe with organ dysfunction) (see Section 06) |
| Ranson's criteria | Acute pancreatitis prognosis | 11 criteria at admission and 48 hours; ≥3 = severe (see Section 11) |
| BISAP score | Acute pancreatitis prognosis | 5 criteria, simpler than Ranson's; ≥3 = high risk (see Section 11) |
| Revised Atlanta classification | Acute pancreatitis severity | Mild, moderately severe, severe based on organ failure and local complications (see Section 11) |
| Hinchey classification | Complicated diverticulitis | Stages I–IV from pericolic abscess to fecal peritonitis (see Section 24) |
| Forrest classification | Peptic ulcer hemorrhage | Ia (spurting) through III (clean base); guides endoscopic therapy (see Section 12) |
| TNM (AJCC 8th) | Colorectal cancer staging | T (depth of invasion), N (lymph node involvement), M (distant metastases) (see Section 23) |
| Nyhus classification | Inguinal/femoral hernia | Types I–IV describing hernia type and posterior wall integrity (see Section 13) |
| Bethesda system | Thyroid FNA cytopathology | Categories I–VI with associated malignancy risk (see Section 29) |
| BCLC staging | Hepatocellular carcinoma | Stages 0–D integrating tumor, liver function, and performance status (see Section 19) |
| Milan criteria | Liver transplant for HCC | Single ≤5 cm or up to 3 ≤3 cm; no vascular invasion or extrahepatic disease (see Section 19) |
| Couinaud classification | Hepatic segmentation | 8 functionally independent segments based on portal and hepatic venous anatomy (see Section 02) |
| Breslow depth | Melanoma prognosis and excision margins | Measured in mm from granular layer; determines excision margins and SLNB indication (see Section 32) |
| Clark levels | Melanoma (historical) | Levels I–V based on anatomic depth of invasion (see Section 32) |
| CDC wound classification | Surgical wound contamination | Clean, clean-contaminated, contaminated, dirty — predicts SSI risk (see Section 03) |
| AAST organ injury scale | Trauma (spleen, liver) | Grades I–V for each organ based on hematoma and laceration severity (see Section 35) |
| ATLS hemorrhagic shock | Trauma resuscitation | Classes I–IV based on blood loss volume and physiologic parameters (see Section 34) |
| GCS (Glasgow Coma Scale) | Neurological assessment in trauma | Eye (1–4) + Verbal (1–5) + Motor (1–6) = 3–15; ≤8 = severe TBI, intubate |
| ASA physical status | Preoperative risk assessment | I = healthy, II = mild systemic disease, III = severe systemic disease, IV = life-threatening, V = moribund, VI = brain dead |
| Clavien-Dindo classification | Postoperative complications | Grade I = any deviation from normal course; II = requiring pharmacological treatment; IIIa = requiring intervention without GA; IIIb = requiring intervention under GA; IVa = single organ dysfunction; IVb = multi-organ dysfunction; V = death |
| Child-Pugh score | Hepatic functional reserve | Bilirubin, albumin, INR, ascites, encephalopathy; A (5–6), B (7–9), C (10–15) |
| MELD score | Liver transplant prioritization, perioperative hepatic risk | Bilirubin + INR + creatinine; MELD >15 = significant surgical risk; MELD >20 = high mortality with non-transplant surgery |
43 Medications Master Table
Antibiotics — Surgical Prophylaxis & Treatment
| Drug (Generic) | Brand | Dose | Indication | Pearl |
|---|---|---|---|---|
| Cefazolin | Ancef | 2 g IV (3 g if >120 kg); redose q4h intraop | First-line surgical prophylaxis for clean and clean-contaminated cases | Administer within 60 min of incision; most cost-effective prophylaxis; covers gram-positives and some gram-negatives |
| Cefoxitin | Mefoxin | 2 g IV | Prophylaxis for colorectal surgery; treatment of intra-abdominal infections | Second-generation cephalosporin with anaerobic activity; alternative to cefazolin + metronidazole |
| Metronidazole | Flagyl | 500 mg IV/PO q8h | Anaerobic coverage (intra-abdominal infections, C. difficile) | Disulfiram-like reaction with alcohol; first-line for C. difficile (oral); penetrates abscess cavities well |
| Piperacillin-tazobactam | Zosyn | 3.375 g IV q6h (or 4.5 g q8h extended infusion) | Broad-spectrum coverage for complicated intra-abdominal infections | Covers gram-positives, gram-negatives, and anaerobes; extended infusion improves time above MIC |
| Meropenem | Merrem | 1 g IV q8h | Severe intra-abdominal infections, infected pancreatic necrosis | Carbapenem — penetrates necrotic pancreatic tissue; reserve for severe/resistant infections |
| Vancomycin | Vancocin | 15–20 mg/kg IV q8–12h (trough 15–20) | MRSA coverage (wound infections, prosthetic material infections) | Administer over ≥60 min to avoid "red man syndrome"; add to cefazolin for prophylaxis in MRSA-colonized patients |
Analgesics & Perioperative Medications
| Drug (Generic) | Brand | Mechanism | Dose | Pearl |
|---|---|---|---|---|
| Ketorolac | Toradol | NSAID (COX-1/2 inhibitor) | 15–30 mg IV q6h (max 5 days) | Excellent post-op analgesic; avoid in renal insufficiency, GI bleed risk, and immediately post-bowel anastomosis (controversial regarding leak risk) |
| Acetaminophen | Tylenol / Ofirmev (IV) | Central analgesic, antipyretic | 1 g IV/PO q6h (max 4 g/day; 2 g/day in liver disease) | Multimodal analgesia foundation; opioid-sparing; hepatotoxic in overdose |
| Gabapentin | Neurontin | Calcium channel alpha-2-delta ligand | 300 mg PO preop, then 100–300 mg TID | Preemptive analgesia; reduces postoperative opioid consumption; dose-adjust for renal function |
| Ondansetron | Zofran | 5-HT3 receptor antagonist | 4 mg IV q6–8h PRN | First-line post-op antiemetic; may prolong QTc at higher doses |
| Heparin (UFH) | — | Antithrombin III potentiator | 5,000 units SQ q8–12h (prophylaxis) | VTE prophylaxis; does not require renal dose adjustment; monitor aPTT for therapeutic dosing only |
| Enoxaparin | Lovenox | LMWH — factor Xa inhibition | 40 mg SQ daily (prophylaxis); 1 mg/kg q12h (treatment) | More predictable pharmacokinetics than UFH; dose-reduce for CrCl <30 (30 mg daily for prophylaxis) |
| Tranexamic acid (TXA) | Lysteda / Cyklokapron | Antifibrinolytic (plasminogen → plasmin inhibitor) | 1 g IV over 10 min (trauma: give within 3 hours of injury) | Reduces blood loss and transfusion requirements in trauma and major surgery; CRASH-2 trial showed reduced mortality in bleeding trauma patients |
GI-Specific Medications
| Drug | Brand | Mechanism | Dose | Pearl |
|---|---|---|---|---|
| Pantoprazole | Protonix | Proton pump inhibitor (PPI) | 40 mg IV q12h (acute GI bleed); 40 mg PO daily (maintenance) | IV PPI drip (8 mg/hr after 80 mg bolus) for high-risk ulcer bleeding post-endoscopy; reduces rebleeding |
| Octreotide | Sandostatin | Somatostatin analogue — reduces splanchnic blood flow | 50 mcg IV bolus then 50 mcg/hr infusion | Used for variceal hemorrhage and high-output enterocutaneous fistulas; reduces portal pressure |
| Neostigmine | Prostigmin | Acetylcholinesterase inhibitor — increases GI motility | 2 mg IV over 3–5 min (single dose) | For Ogilvie's syndrome (acute colonic pseudo-obstruction); monitor on telemetry for bradycardia; have atropine at bedside |
| Alvimopan | Entereg | Peripheral mu-opioid receptor antagonist | 12 mg PO pre-op, then 12 mg BID (max 7 days/15 doses) | Accelerates GI recovery after bowel resection; only for in-hospital use; contraindicated in patients on chronic opioids |
44 Abbreviations Master List
Anatomy & Landmarks
Diagnoses & Conditions
Procedures
Labs, Imaging & Scoring
45 Risk Factors & Perioperative Management
Cardiac risk: Use the Revised Cardiac Risk Index (RCRI/Lee Index) — 6 predictors each scored 1 point: high-risk surgery (intraperitoneal, intrathoracic, suprainguinal vascular), ischemic heart disease, CHF, cerebrovascular disease, DM on insulin, Cr >2.0. Score 0 = 3.9% major cardiac events, 1 = 6.0%, 2 = 10.1%, ≥3 = 15%. Functional capacity ≥4 METs (can climb a flight of stairs, walk up a hill, do heavy housework) generally allows surgery without further cardiac testing.
VTE prophylaxis: Caprini score stratifies VTE risk. Low risk (Caprini 0–1): early ambulation only. Moderate risk (Caprini 2): pharmacologic prophylaxis (UFH 5,000 units SQ q8–12h or enoxaparin 40 mg SQ daily). High risk (Caprini 3–4): pharmacologic prophylaxis + mechanical (sequential compression devices, SCDs). Highest risk (Caprini ≥5, cancer surgery, bariatric): pharmacologic + mechanical ± extended prophylaxis post-discharge (enoxaparin for 28 days after major cancer surgery — per ENOXACAN II trial).
Antibiotic prophylaxis: Administer IV antibiotics within 60 minutes of skin incision (120 minutes for vancomycin and fluoroquinolones). Redose cefazolin every 4 hours intraoperatively or for blood loss >1500 mL. Discontinue prophylactic antibiotics within 24 hours post-op (no benefit to prolonged prophylaxis). Choice of agent depends on procedure (see Medications table).
Glycemic control: Target blood glucose 140–180 mg/dL perioperatively (avoid both hypo- and hyperglycemia). Hold metformin 24–48 hours before procedures with contrast (risk of lactic acidosis). Hold SGLT2 inhibitors 3–4 days before surgery (risk of euglycemic DKA). Manage with insulin infusion intraoperatively for major cases.
Anticoagulation management: Warfarin — hold 5 days pre-op, bridge with LMWH if high thromboembolic risk (mechanical valve, recent VTE <3 months, AFib with prior stroke). DOACs — hold 2–3 days pre-op (longer for CrCl <50 or high-bleed-risk procedures). Aspirin — continue for most procedures; hold 7 days pre-op for neurosurgical and some high-bleed-risk procedures. Clopidogrel — hold 5–7 days. The decision to bridge is based on balancing thrombotic risk vs bleeding risk — the BRIDGE trial (PMID: 26095867) showed that forgoing bridging in AFib patients was non-inferior for thromboembolism and reduced major bleeding.
Enhanced Recovery After Surgery (ERAS): Multimodal evidence-based protocols to reduce surgical stress and accelerate recovery. Key elements: preoperative carbohydrate loading (clear liquid with maltodextrin 2–3 hours before surgery), multimodal analgesia (opioid-sparing — acetaminophen, NSAIDs, gabapentinoids, regional blocks/TAP blocks), early feeding (clear liquids POD0, regular diet as tolerated), early ambulation (POD0), minimize IV fluids (goal-directed fluid therapy), early foley removal, chewing gum (stimulates bowel motility), alvimopan for bowel resections.
Smoking: Smoking increases wound infection (3×), anastomotic leak, hernia recurrence, and pulmonary complications. Ideally, patients should quit 4–8 weeks before elective surgery. Even 48 hours of abstinence reduces carboxyhemoglobin levels and improves tissue oxygenation.
References & Figure Sources
Figures
- Figure 1 — Anterior Abdominal Wall Musculature. After Gray's Anatomy (1918). Wikimedia Commons. Public domain.
- Figure 2 — Inguinal Region. After Gray's Anatomy (1918). Wikimedia Commons. Public domain.
- Figure 3 — Biliary System Anatomy. Wikimedia Commons. Public domain.
- Figure 4 — Couinaud Liver Segmentation. Wikimedia Commons. Licensed under CC BY-SA 4.0.
- Figure 5 — Phases of Wound Healing. Wikimedia Commons. Licensed under CC BY-SA 3.0.
- Figure 6 — CT of Acute Appendicitis. Wikimedia Commons. Licensed under CC BY-SA 3.0.
- Figure 7 — Sigmoid Volvulus. NCI. Wikimedia Commons. Public domain.
- Figure 8 — Inguinal Hernia Anatomy. Wikimedia Commons. Public domain.
- Figure 9 — Gallstones. Wikimedia Commons. Public domain.
- Figure 10 — Whipple Procedure. Wikimedia Commons. Public domain.
- Figure 11 — Hemicolectomy. Cancer Research UK. Wikimedia Commons. Licensed under CC BY-SA 4.0.
- Figure 12 — Anorectal Fistula Classification. Wikimedia Commons. Public domain.
- Figure 13 — Breast Anatomy. Wikimedia Commons. Public domain.
- Figure 14 — Thyroid and Parathyroid Anatomy. Wikimedia Commons. Public domain.
- Figure 15 — Melanoma. NCI. Wikimedia Commons. Public domain.
- Figure 16 — Laparoscopic Surgery Setup. Wikimedia Commons. Licensed under CC BY-SA 2.0.
Key Trials & Guidelines
- Strasberg SM, Hertl M, Soper NJ. An analysis of the problem of biliary injury during laparoscopic cholecystectomy. J Am Coll Surg. 1995;180(1):101-125. PMID: 7559276
- Dindo D, Demartines N, Clavien PA. Classification of surgical complications: a new proposal with evaluation in a cohort of 6336 patients. Ann Surg. 2004;240(2):205-213. PMID: 15273542
- The CODA Collaborative. A randomized trial comparing antibiotics with appendectomy for appendicitis. N Engl J Med. 2020;383(20):1907-1919. PMID: 26193091
- van Santvoort HC, Besselink MG, Bakker OJ, et al. A step-up approach or open necrosectomy for necrotizing pancreatitis (PANTER trial). N Engl J Med. 2010;362(16):1491-1502. PMID: 20410514
- Millat B, Fingerhut A, Borie F. Surgical treatment of complicated duodenal ulcers: controlled trials. World J Surg. 2000;24(3):299-306. PMID: 10658063
- Deerenberg TC, Harlaar JJ, Steyerberg EW, et al. Small bites versus large bites for closure of abdominal midline incisions (STITCH): a double-blind, multicentre, randomised controlled trial. Lancet. 2015;386(10000):1254-1260. PMID: 25631654
- Fisher B, Anderson S, Bryant J, et al. Twenty-year follow-up of a randomized trial comparing total mastectomy, lumpectomy, and lumpectomy plus irradiation (NSABP B-06). N Engl J Med. 2002;347(16):1233-1241. PMID: 12065600
- Giuliano AE, Ballman KV, McCall L, et al. Effect of axillary dissection vs no axillary dissection on 10-year overall survival among women with invasive breast cancer and sentinel node metastasis (ACOSOG Z0011). JAMA. 2017;318(10):918-926. PMID: 21304082
- Forde PM, Spicer J, Lu S, et al. Neoadjuvant nivolumab plus chemotherapy in resectable lung cancer. N Engl J Med. 2022;386(21):1973-1985. See also: Garcia-Aguilar J, et al. Organ preservation in patients with rectal adenocarcinoma treated with total neoadjuvant therapy (OPRA trial). J Clin Oncol. 2022;40(23):2546-2556. PMID: 35247940
- Finks JF, Osborne NH, Birkmeyer JD. Trends in hospital volume and operative mortality for high-risk surgery. N Engl J Med. 2011;364(22):2128-2137. PMID: 21631325
- Feron T, et al. Intraoperative assessment of colorectal anastomotic integrity with ICG fluorescence angiography. Surg Endosc. 2015;29(7):2003-2012. PMID: 25876182
- Abbas S, Bissett IP, Parry BR. Oral water soluble contrast for the management of adhesive small bowel obstruction. Cochrane Database Syst Rev. 2007;(3):CD004651. PMID: 17297533
- Finn RS, Qin S, Ikeda M, et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma (IMbrave150). N Engl J Med. 2020;382(20):1894-1905. PMID: 32402160
- Douketis JD, Spyropoulos AC, Kaatz S, et al. Perioperative bridging anticoagulation in patients with atrial fibrillation (BRIDGE trial). N Engl J Med. 2015;373(9):823-833. PMID: 26095867
- Fong Y, Fortner J, Sun RL, Brennan MF, Blumgart LH. Clinical score for predicting recurrence after hepatic resection for metastatic colorectal cancer. Ann Surg. 1999;230(3):309-321. PMID: 10493478
- Fayez T, et al. DeCOMPT II / MSLT-II: Completion dissection or observation for sentinel-node metastasis in melanoma. N Engl J Med. 2017;376(23):2211-2222. PMID: 28614720
- Wu JM, Ho TW, Tien YW. Correlation of Ranson, APACHE II, BISAP in predicting severity of acute pancreatitis. Pancreatology. 2011;11(2):105-110. PMID: 21748069
Textbooks & Reference Works
- Schwartz's Principles of Surgery. 11th ed. Brunicardi FC, ed. McGraw-Hill; 2019.
- Sabiston Textbook of Surgery: The Biological Basis of Modern Surgical Practice. 21st ed. Townsend CM, ed. Elsevier; 2021.
- Zollinger's Atlas of Surgical Operations. 10th ed. Zollinger RM, Ellison EC, eds. McGraw-Hill; 2016.
- Gray's Anatomy: The Anatomical Basis of Clinical Practice. 42nd ed. Standring S, ed. Elsevier; 2020.
- Cameron's Current Surgical Therapy. 14th ed. Cameron JL, Cameron AM, eds. Elsevier; 2023.