Neurosurgery
Every diagnosis, classification, procedure, technique, medication, complication, and management algorithm across the full scope of neurosurgery in one place.
01 Neuroanatomy — Cranial, Ventricular, & Vascular
A thorough understanding of cranial anatomy is the foundation of neurosurgical practice. The skull base, cerebral vasculature, and ventricular system define the corridors and constraints of every neurosurgical approach.
Cranial Fossae
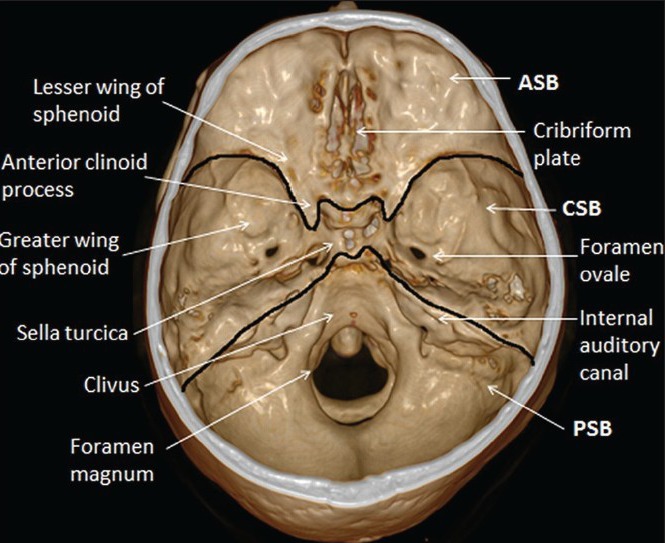
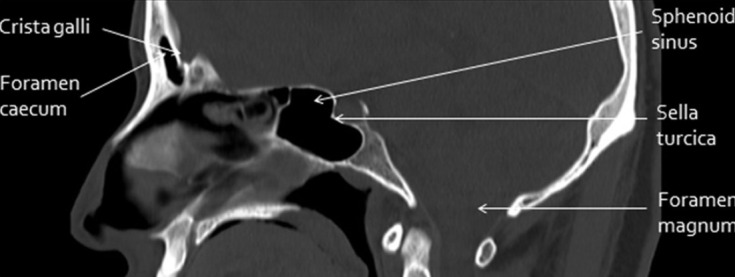
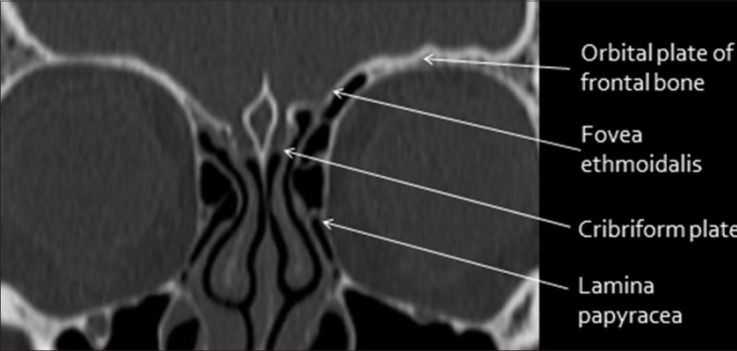
The interior of the skull base is divided into three fossae. The anterior cranial fossa is formed by the orbital plates of the frontal bone, cribriform plate of the ethmoid, and lesser wing of the sphenoid; it houses the frontal lobes and olfactory bulbs. The cribriform plate is the thinnest portion of the skull base — fractures here cause CSF rhinorrhea and anosmia. The middle cranial fossa is formed by the greater wing of the sphenoid and temporal bone; it contains the temporal lobes, pituitary fossa (sella turcica), and the cavernous sinus. Key foramina: foramen rotundum (V2), foramen ovale (V3, accessory meningeal artery), foramen spinosum (middle meningeal artery — epidural hematoma source), and the superior orbital fissure (CN III, IV, V1, VI, ophthalmic veins). The posterior cranial fossa is formed by the occipital bone and petrous temporal bones; it contains the cerebellum, brainstem, and CN IV–XII. The foramen magnum transmits the medulla oblongata, vertebral arteries, and spinal accessory nerves (CN XI ascending roots).
Cerebral Lobes & Functional Areas
The frontal lobe (anterior to the central sulcus) contains the primary motor cortex (precentral gyrus), Broca's area (dominant inferior frontal gyrus — expressive speech), supplementary motor area, and frontal eye fields. The parietal lobe contains the primary sensory cortex (postcentral gyrus) and association areas for spatial awareness; lesions of the non-dominant parietal lobe cause hemispatial neglect. The temporal lobe houses the primary auditory cortex (Heschl's gyrus), Wernicke's area (dominant posterior superior temporal gyrus — receptive speech), hippocampus (memory), and amygdala. The occipital lobe contains the primary visual cortex along the calcarine fissure. The insula, deep to the Sylvian fissure, is involved in pain processing, autonomic regulation, and taste.
Ventricular System
CSF is produced primarily by the choroid plexus (~500 mL/day; total CSF volume ~150 mL, turnover 3–4 times daily). Flow pathway: lateral ventricles → foramen of Monro (interventricular foramen) → third ventricle → cerebral aqueduct (of Sylvius) → fourth ventricle → exits via the foramina of Luschka (lateral, paired) and foramen of Magendie (midline, posterior) → subarachnoid space → absorbed at the arachnoid granulations into the superior sagittal sinus. Obstruction at any point in this pathway produces obstructive (non-communicating) hydrocephalus. The cerebral aqueduct is the narrowest point and the most common site of obstruction (aqueductal stenosis, tectal plate glioma, pineal region tumor).
Arterial Supply — Circle of Willis
The brain receives ~15% of cardiac output via two paired arterial systems. The internal carotid arteries (ICA) enter the skull through the carotid canals, traverse the cavernous sinus, and give off: ophthalmic artery (first intradural branch), posterior communicating artery (PComm), anterior choroidal artery, then bifurcate into the anterior cerebral artery (ACA) and middle cerebral artery (MCA). The vertebral arteries enter through the foramen magnum, give off the posterior inferior cerebellar arteries (PICA), then unite to form the basilar artery, which gives off the anterior inferior cerebellar arteries (AICA) and superior cerebellar arteries (SCA) before bifurcating into the posterior cerebral arteries (PCA).
The Circle of Willis connects these systems: ACA — anterior communicating artery (AComm) — contralateral ACA; ICA — PComm — PCA. A complete Circle of Willis is present in only ~25% of the population. The AComm is the most common site of intracranial aneurysms (~30%), followed by the PComm (~25%) and the MCA bifurcation (~20%).
Venous Sinuses
The dural venous sinuses are valveless channels between the periosteal and meningeal layers of dura. The superior sagittal sinus (SSS) runs in the superior margin of the falx cerebri from the crista galli to the confluence of sinuses (torcular Herophili). Parasagittal bridging veins drain cortical surface blood into the SSS — these are stretched and torn in acute subdural hematomas. The transverse sinuses run laterally from the torcular along the tentorium to become the sigmoid sinuses, which exit through the jugular foramina. The cavernous sinus surrounds the sella turcica and contains the ICA and CN VI traversing through its center, with CN III, IV, V1, and V2 in its lateral wall. Cavernous sinus thrombosis presents with proptosis, chemosis, ophthalmoplegia, and facial sensory loss.
Cerebral Arterial Territories & Stroke Syndromes
| Artery | Territory | Occlusion Syndrome |
|---|---|---|
| ACA | Medial frontal and parietal lobes, corpus callosum (anterior) | Contralateral leg weakness > arm; abulia (bilateral ACA); urinary incontinence; transcortical motor aphasia (dominant) |
| MCA — superior division | Lateral frontal lobe, motor strip (face/arm) | Contralateral face and arm weakness > leg; Broca's aphasia (dominant hemisphere) |
| MCA — inferior division | Lateral temporal and parietal lobes | Wernicke's aphasia (dominant hemisphere); hemispatial neglect (non-dominant); contralateral homonymous hemianopia or superior quadrantanopia |
| MCA — complete | Entire lateral hemisphere | Dense hemiplegia, hemisensory loss, hemianopia, global aphasia (dominant) or neglect (non-dominant) |
| PCA | Occipital lobe, medial temporal lobe, thalamus | Contralateral homonymous hemianopia with macular sparing; memory impairment (hippocampal); thalamic pain syndrome |
| Basilar artery | Pons, midbrain, cerebellum | "Locked-in syndrome" (ventral pontine infarction — quadriplegia, anarthria, preserved consciousness and vertical eye movements); basilar apex: "top of the basilar" syndrome with altered consciousness, visual field deficits, oculomotor abnormalities |
| PICA | Lateral medulla, inferior cerebellum | Wallenberg syndrome (lateral medullary syndrome): ipsilateral Horner's, facial pain/temp loss, ataxia, vocal cord paralysis; contralateral body pain/temp loss; dysphagia, vertigo, nystagmus |
Meninges
Three membranes envelop the brain and spinal cord. The dura mater is the outermost, toughest layer; in the cranium it has two layers (periosteal and meningeal) that separate to form the dural venous sinuses. Dural folds compartmentalize the intracranial space: the falx cerebri (separates cerebral hemispheres; contains the SSS superiorly and inferior sagittal sinus inferiorly) and the tentorium cerebelli (separates the cerebral hemispheres from the cerebellum; the tentorial notch transmits the midbrain and is the site of transtentorial/uncal herniation). The arachnoid mater is a delicate, avascular membrane beneath the dura; the subdural space is a potential space between the dura and arachnoid (where subdural hematomas collect). The pia mater is intimately adherent to the brain surface, following every gyrus and sulcus; the subarachnoid space (between arachnoid and pia) contains CSF and the major cerebral arteries.
02 Neuroanatomy — Spinal Cord & Peripheral Nerves
Spinal Cord Anatomy
The spinal cord extends from the foramen magnum to approximately the L1–L2 vertebral level in adults (the conus medullaris). Below this, the lumbar and sacral nerve roots form the cauda equina. The cord has two enlargements: the cervical enlargement (C4–T1, brachial plexus) and the lumbar enlargement (L1–S3, lumbosacral plexus). The filum terminale is a pial strand anchoring the conus to the coccyx.
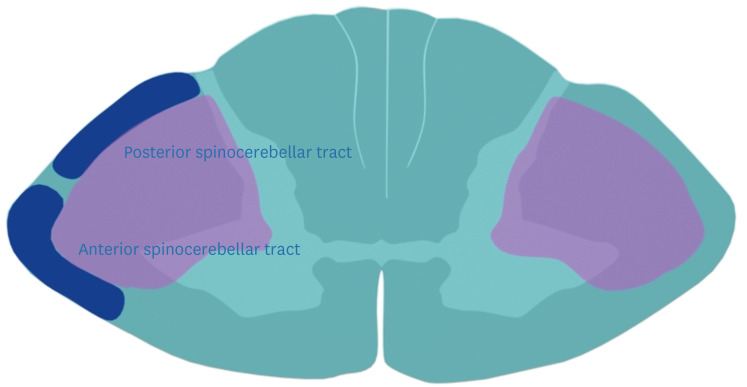
In cross-section, the cord has an H-shaped central gray matter surrounded by white matter tracts. Key tracts: the corticospinal (pyramidal) tract — descends in the lateral white matter (lateral corticospinal tract, crossed at medullary pyramids); lesions cause ipsilateral upper motor neuron weakness. The dorsal columns (fasciculus gracilis — lower extremities, medial; fasciculus cuneatus — upper extremities, lateral) carry proprioception, vibration, and fine touch; they are ipsilateral and cross in the medulla. The spinothalamic tract carries pain and temperature; fibers cross within 1–2 levels of entry and ascend contralaterally.
Spinal Cord Syndromes
| Syndrome | Mechanism | Clinical Features | Prognosis |
|---|---|---|---|
| Central cord | Hyperextension injury in elderly with cervical spondylosis | Upper extremity weakness > lower extremity ("cape-like" distribution); bladder dysfunction; variable sensory loss | Best prognosis of incomplete injuries; hands recover last |
| Brown-Sequard (hemisection) | Penetrating trauma, lateral mass lesion | Ipsilateral motor loss + dorsal column loss (proprioception/vibration); contralateral spinothalamic loss (pain/temp) 1–2 levels below | Best overall motor recovery |
| Anterior cord | Anterior spinal artery occlusion, burst fracture with retropulsed fragment | Bilateral motor paralysis + pain/temp loss; preserved proprioception/vibration (dorsal columns intact) | Worst prognosis of incomplete injuries |
| Posterior cord | Rare; posterior spinal artery stroke, B12 deficiency, tabes dorsalis | Loss of proprioception and vibration; preserved motor, pain, temp | Variable |
| Cauda equina | Large central disc herniation (usually L4–L5 or L5–S1), tumor | Bilateral leg pain, saddle anesthesia, urinary retention, decreased rectal tone — LMN pattern | Surgical emergency — best outcomes with decompression within 48 hours |
| Conus medullaris | Fractures at thoracolumbar junction, tumors | Early bladder/bowel dysfunction (S2–S4), saddle anesthesia, mixed UMN/LMN pattern; symmetric; may have preserved bulbocavernosus reflex | Variable |
Nerve Root Localization
| Root | Motor | Reflex | Sensory | Disc Level |
|---|---|---|---|---|
| C5 | Deltoid, biceps | Biceps (C5–C6) | Lateral arm (regimental badge area) | C4–C5 |
| C6 | Wrist extensors, biceps | Brachioradialis | Lateral forearm, thumb, index finger | C5–C6 |
| C7 | Triceps, wrist flexors, finger extensors | Triceps | Middle finger | C6–C7 |
| C8 | Finger flexors, hand intrinsics | None reliable | Medial forearm, ring and little finger | C7–T1 |
| L4 | Tibialis anterior (ankle dorsiflexion), quadriceps | Patellar (L3–L4) | Medial leg | L3–L4 |
| L5 | Extensor hallucis longus (great toe dorsiflexion), hip abductors | None reliable (medial hamstring) | Lateral leg, dorsum of foot | L4–L5 |
| S1 | Gastrocnemius/soleus (plantarflexion), peroneus longus/brevis | Achilles | Lateral foot, sole | L5–S1 |
03 The Neurosurgical Physical Exam
Glasgow Coma Scale (GCS)
The GCS is the most widely used scale for assessing level of consciousness in neurosurgical patients. Score ranges from 3 (worst) to 15 (best). It is composed of three components:
| Component | Response | Score |
|---|---|---|
| Eye Opening (E) | Spontaneous | 4 |
| To voice | 3 | |
| To pain | 2 | |
| None | 1 | |
| Verbal (V) | Oriented | 5 |
| Confused | 4 | |
| Inappropriate words | 3 | |
| Incomprehensible sounds | 2 | |
| None | 1 | |
| Motor (M) | Obeys commands | 6 |
| Localizes pain | 5 | |
| Withdrawal (flexion) | 4 | |
| Abnormal flexion (decorticate) | 3 | |
| Extension (decerebrate) | 2 | |
| None | 1 |
Mild TBI: GCS 13–15 | Moderate TBI: GCS 9–12 | Severe TBI: GCS 3–8 (intubation indicated)
The motor score is the single most predictive component. Always record the best response and note whether the patient is intubated (record as E_VTM_). A GCS of 8 or less warrants emergent intubation for airway protection.
Pupillary Examination
Assess size, symmetry, and reactivity. Unilateral fixed, dilated pupil (mydriasis): ipsilateral CN III compression — most commonly from uncal herniation pushing the temporal lobe through the tentorial notch, compressing CN III against the posterior clinoid. This is a neurosurgical emergency requiring immediate intervention to relieve the herniation. Bilateral fixed, dilated pupils: bilateral CN III compression from severe raised ICP or brainstem death — very poor prognosis. Bilateral pinpoint pupils: pontine lesion (hemorrhage or ischemia), opioid toxicity. Horner syndrome (miosis, ptosis, anhidrosis): disruption of the sympathetic chain — central (brainstem/spinal cord lesion), preganglionic (Pancoast tumor, carotid dissection), or postganglionic (cavernous sinus lesion).
Cranial Nerve Examination
CN I (olfactory): test each nostril separately with coffee, peppermint; anosmia occurs with cribriform plate fractures or olfactory groove meningiomas. CN II (optic): visual acuity, visual fields (confrontation), fundoscopy (papilledema = raised ICP), relative afferent pupillary defect (RAPD, Marcus Gunn pupil — swinging flashlight test). CN III, IV, VI: extraocular movements; CN III palsy = "down and out" eye, ptosis, mydriasis; CN IV palsy = hypertropia worsened with contralateral head tilt (Bielschowsky test); CN VI palsy = inability to abduct (often a false localizing sign of raised ICP). CN V: facial sensation in V1/V2/V3 distributions, masseter/temporalis strength, corneal reflex (afferent V1, efferent VII). CN VII: facial symmetry at rest and with movement; UMN lesion = contralateral lower face weakness (forehead spared); LMN lesion = ipsilateral entire hemiface weakness. CN VIII: hearing (finger rub, Rinne/Weber), vestibular function. CN IX/X: gag reflex, palate elevation (uvula deviates away from lesion), hoarseness. CN XI: sternocleidomastoid (turns head to opposite side), trapezius (shoulder shrug). CN XII: tongue protrusion (deviates toward the side of the lesion in LMN injury).
Motor Examination
Assess strength using the MRC (Medical Research Council) scale: 0 = no contraction; 1 = flicker; 2 = movement with gravity eliminated; 3 = movement against gravity; 4 = movement against resistance (4−, 4, 4+); 5 = normal. Assess tone (spasticity vs rigidity vs flaccidity), pronator drift (subtle corticospinal tract dysfunction), and deep tendon reflexes. Pathologic reflexes: Babinski sign (upgoing toe with plantar stimulation = UMN lesion); Hoffman sign (flicking the middle finger distal phalanx produces involuntary flexion of the thumb and index finger = cervical myelopathy).
Cerebellar Examination
The cerebellum coordinates movement, balance, and muscle tone. Test for: dysmetria (finger-nose-finger, heel-knee-shin — past-pointing), dysdiadochokinesia (rapid alternating movements), intention tremor (worsens approaching the target), ataxic gait (wide-based, unsteady), nystagmus (fast component toward the lesion side), and Romberg sign (proprioceptive, not cerebellar — patient falls with eyes closed if dorsal column deficit). Cerebellar lesions produce ipsilateral deficits.
Herniation Syndromes
| Type | Mechanism | Clinical Features |
|---|---|---|
| Uncal (transtentorial) | Medial temporal lobe (uncus) herniates through the tentorial notch | Ipsilateral CN III palsy (fixed dilated pupil) → contralateral hemiparesis (cerebral peduncle compression) → Kernohan's notch phenomenon (ipsilateral hemiparesis from contralateral peduncle compressed against the tentorium — false localizing) → coma → death if untreated |
| Central (descending transtentorial) | Bilateral, symmetric downward displacement of the diencephalon through the tentorial notch | Progressive decline through diencephalic → midbrain → pontine → medullary stages; bilateral small reactive pupils → bilateral fixed midposition pupils → bilateral fixed dilated pupils |
| Subfalcine (cingulate) | Cingulate gyrus herniates under the falx cerebri | Contralateral leg weakness (ACA compression); may precede uncal herniation; often seen as midline shift on imaging |
| Tonsillar | Cerebellar tonsils herniate through the foramen magnum | Brainstem compression → cardiorespiratory arrest; most lethal herniation syndrome; seen with posterior fossa masses |
| Upward (ascending transtentorial) | Posterior fossa mass or edema pushes cerebellum upward through the tentorial notch | Can occur after EVD placement for posterior fossa pathology (decompresses supratentorial space relative to infratentorial); pineal region compression → Parinaud syndrome (upgaze palsy) |
04 Neurosurgical Principles — ICP, CPP, & Brain Relaxation
Intracranial Pressure (ICP)
Normal ICP in adults is 5–15 mmHg (supine). The Monro-Kellie doctrine states that the cranial vault is a fixed volume; the sum of brain parenchyma (~80%), blood (~10%), and CSF (~10%) must remain constant. An increase in any one component must be offset by a decrease in another, or ICP will rise. Initially, CSF is displaced into the spinal subarachnoid space and venous blood is compressed out of the sinuses (compensatory mechanisms). Once compensation is exhausted, small additional volume increases cause dramatic ICP elevations (steep portion of the intracranial compliance curve).
Cushing's triad (late and ominous): hypertension (widened pulse pressure), bradycardia, irregular respirations. Other signs: headache (worse in morning, with Valsalva), nausea/vomiting (often projectile), papilledema, CN VI palsy (false localizing), altered consciousness. In infants: bulging fontanelle, splayed sutures, "sunset" eyes (upgaze palsy from compression of the tectal plate).
ICP Monitoring
Indications (BTF guidelines): severe TBI (GCS ≤8) with an abnormal CT, or with a normal CT if two or more of the following are present: age >40, unilateral or bilateral motor posturing, or systolic BP <90 mmHg. Types of monitors: External ventricular drain (EVD) — gold standard; catheter placed in the lateral ventricle (usually right, at Kocher's point: 11 cm posterior to the nasion, 3 cm lateral to midline, aimed at the medial canthus of the ipsilateral eye in the coronal plane and toward the tragus in the sagittal plane); allows both measurement and therapeutic CSF drainage. Intraparenchymal monitor (Camino, Codman) — placed in the brain parenchyma; cannot drain CSF but less infection risk and easier to place.
Cerebral Perfusion Pressure (CPP)
CPP = MAP − ICP. Target CPP: 60–70 mmHg (BTF guidelines recommend maintaining CPP ≥60 mmHg; aggressive attempts to keep CPP >70 mmHg are associated with ARDS and should be avoided). Cerebral autoregulation normally maintains constant cerebral blood flow (CBF) across MAP 50–150 mmHg; this autoregulation is often impaired after TBI, making CBF directly dependent on CPP.
ICP Management — Tiered Approach
Tier 0 (Prevention): Head of bed elevated 30°, head midline (avoid jugular venous compression), avoid hyperthermia, treat pain and agitation, prevent hypotension and hypoxia.
Tier 1: CSF drainage via EVD (10–20 mL increments, close after draining); osmotic therapy — mannitol 0.25–1 g/kg IV bolus (onset 15–30 min, duration 4–6 hr, keep serum osmolality <320 mOsm/L, monitor for renal toxicity) or hypertonic saline (23.4% NaCl, 30 mL via central line, or 3% NaCl infusion targeting Na 145–155 mEq/L).
Tier 2: Moderate hyperventilation (PaCO2 30–35 mmHg — vasoconstriction reduces CBV; avoid aggressive hyperventilation <25 mmHg as it may cause ischemia). Sedation with propofol (short-acting, allows serial neurologic exams; beware propofol infusion syndrome with doses >5 mg/kg/hr for >48 hr) or midazolam.
Tier 3: Barbiturate coma (pentobarbital 5–10 mg/kg loading dose, then 1–3 mg/kg/hr, titrate to ICP control or burst suppression on EEG; causes hypotension, requires vasopressors). Decompressive craniectomy — removal of a large bone flap (≥12 × 15 cm or bifrontal) to allow brain expansion; DECRA and RESCUEicp trials showed improved survival but increased severe disability (DECRA, PMID: 21434843; RESCUEicp, PMID: 27602507).
Brain Relaxation During Surgery
Techniques to achieve a "slack" brain during craniotomy: mannitol or hypertonic saline (given 30 min before dural opening); CSF drainage (lumbar drain placed preoperatively, or EVD, or opening the Sylvian cisterns to release CSF); mild hyperventilation (PaCO2 30–35 mmHg during the approach, return to normocarbia once the lesion is accessed); head elevation (promotes venous drainage); avoid jugular venous obstruction (head position, tape securing ETT); and appropriate anesthetic depth (avoid coughing, straining). If the brain remains "tight" despite these measures, consider the possibility of an evolving contralateral lesion (obtain intraoperative imaging or inspect the contralateral side).
Cerebral Blood Flow Physiology
Normal CBF is ~50 mL/100 g/min. Ischemic threshold: CBF <20 mL/100 g/min → neuronal dysfunction (reversible); CBF <10 mL/100 g/min → infarction (irreversible). Factors affecting CBF:
| Factor | Effect on CBF | Clinical Relevance |
|---|---|---|
| PaCO2 | CBF changes ~3% per 1 mmHg change in PaCO2 (range 20–60 mmHg) | Hyperventilation → vasoconstriction → reduced ICP (but risk of ischemia if PaCO2 <25 mmHg); hypoventilation → vasodilation → increased ICP |
| PaO2 | CBF increases when PaO2 <60 mmHg (significant vasodilation) | Maintain PaO2 >60 mmHg in neurosurgical patients; supplemental O2 above normal has minimal effect on CBF |
| Temperature | CBF decreases ~6–7% per 1°C decrease in temperature | Hypothermia reduces metabolic demand and CBF; hyperthermia increases metabolic demand and is deleterious in TBI and stroke |
| Hematocrit | Low Hct → decreased viscosity → increased CBF; high Hct → increased viscosity → decreased CBF | Optimal Hct ~30–33% for cerebral oxygen delivery; extreme hemodilution (Hct <25%) reduces oxygen-carrying capacity despite increased CBF |
| Anesthetic agents | Volatile agents (isoflurane, sevoflurane) → dose-dependent vasodilation → increased CBF; propofol, barbiturates → decreased CBF (coupled to decreased CMRO2) | TIVA (total intravenous anesthesia with propofol/remifentanil) preferred for neurosurgery — maintains autoregulation and reduces brain bulk |
05 Gliomas — WHO Classification & Management
Gliomas are the most common primary malignant brain tumors in adults, arising from glial cells (astrocytes, oligodendrocytes, ependymal cells). The 2021 WHO Classification of CNS tumors integrates molecular markers — particularly IDH mutation status and 1p/19q co-deletion — into diagnosis and grading.
WHO Classification (2021)
| Tumor Type | IDH Status | 1p/19q | WHO Grade | Key Features |
|---|---|---|---|---|
| Astrocytoma, IDH-mutant | Mutant | Intact | 2, 3, or 4 | ATRX loss, TP53 mutation; grade 4 if microvascular proliferation, necrosis, or CDKN2A/B homozygous deletion |
| Oligodendroglioma, IDH-mutant | Mutant | Co-deleted | 2 or 3 | "Fried egg" cells, chicken-wire vascularity; better prognosis; PCV chemo-responsive |
| Glioblastoma, IDH-wildtype | Wildtype | N/A | 4 | Grade 4 by definition; TERT promoter mutation, EGFR amplification, or +7/−10; median survival 14–16 months |
Glioblastoma (GBM)
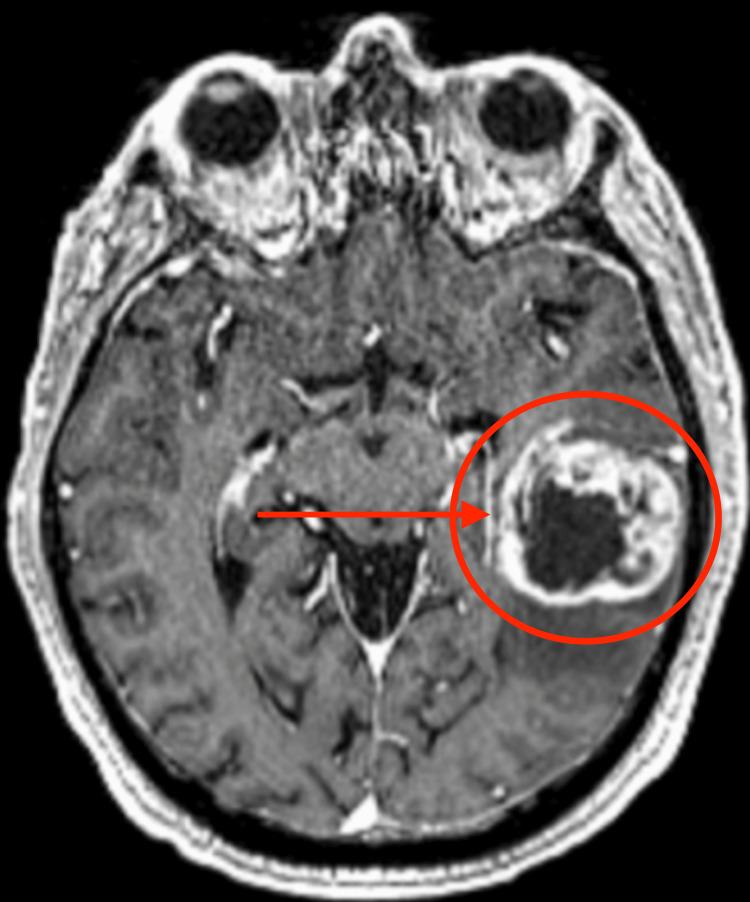
The most common and most aggressive primary brain tumor. Incidence peaks at age 55–85. Presents with headache, seizures, progressive focal neurologic deficit, or personality changes. MRI shows an irregularly enhancing mass with central necrosis and surrounding vasogenic edema (T2/FLAIR hyperintensity). Standard treatment (Stupp protocol): maximal safe resection → concurrent temozolomide (TMZ) + radiation (60 Gy in 30 fractions) → adjuvant TMZ for 6 cycles (Stupp et al., 2005; PMID: 15758009). MGMT promoter methylation predicts TMZ responsiveness (median survival ~21 months with methylation vs ~14 months without).
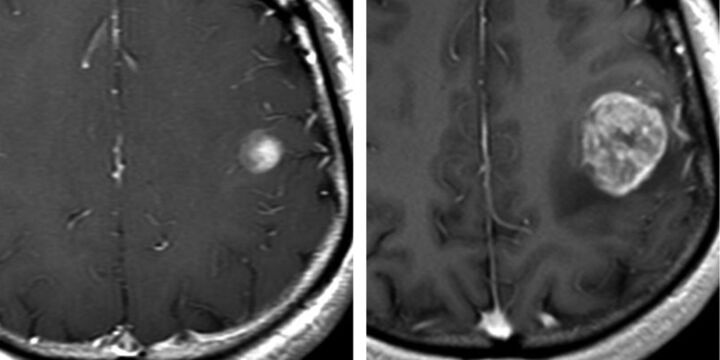
Low-Grade Gliomas (WHO Grade 2)
Present in younger patients (30–40s), often with seizures. MRI shows a non-enhancing, T2/FLAIR hyperintense lesion without necrosis. IDH-mutant astrocytomas and oligodendrogliomas have significantly better prognoses than IDH-wildtype tumors. Management: maximal safe resection (extent of resection correlates with OS); observation vs early adjuvant therapy based on risk factors. High-risk features (age >40, subtotal resection, tumor >6 cm, tumor crossing midline, non-oligodendroglioma histology, neurologic deficit) favor early radiation + chemotherapy (PCV for oligodendrogliomas — RTOG 9802, PMID: 27050206; TMZ also used).
Ependymoma
Arises from ependymal cells lining the ventricles. In children, most are posterior fossa (fourth ventricle); in adults, most are spinal (intradural intramedullary). WHO grade 2 or 3. The RELA fusion-positive variant (supratentorial) carries a worse prognosis. Treatment: gross total resection is the primary goal; adjuvant radiation for subtotal resection or anaplastic histology. Chemotherapy has limited efficacy in ependymomas.
Extent of Resection in Glioma Surgery
Multiple retrospective studies demonstrate that extent of resection (EOR) is one of the strongest modifiable prognostic factors in glioma surgery. For GBM, gross total resection (GTR, defined as no residual contrast enhancement on postoperative MRI within 48 hours) is associated with a median OS of ~16–18 months vs ~12–14 months for subtotal resection. For low-grade gliomas, EOR >90% is associated with significantly improved PFS and OS. Supramaximal resection (resecting beyond the contrast-enhancing margin into the FLAIR abnormality) is an active area of investigation and may further improve survival, particularly for IDH-mutant gliomas.
Intraoperative tools to maximize EOR: (1) Neuronavigation — integrates preoperative MRI with real-time surgical coordinates; accuracy limited by "brain shift" (brain displacement after CSF drainage and tumor resection). (2) Intraoperative MRI (iMRI) — allows real-time imaging during surgery to identify residual tumor before closure; increases GTR rates from ~60% to ~90% in some series; expensive, requires MRI-compatible instruments and specialized OR suite. (3) 5-ALA fluorescence — tumor fluoresces pink under blue light (see clinical pearl above). (4) Awake craniotomy with cortical/subcortical mapping — direct electrical stimulation identifies motor and language function in real time; allows resection up to the functional boundary; essential for tumors in or near eloquent cortex. (5) Intraoperative ultrasound — real-time, portable, and inexpensive; useful for identifying tumor margins, cystic components, and brain shift.
Molecular Markers in Glioma — Clinical Relevance
| Marker | Test Method | Clinical Significance |
|---|---|---|
| IDH1/2 mutation | Immunohistochemistry (R132H antibody covers ~90%), sequencing for non-canonical | Defines glioma subtype; IDH-mutant gliomas have dramatically better prognosis than IDH-wildtype; IDH-mutant astrocytoma grade 4 has better OS than IDH-wildtype GBM |
| 1p/19q co-deletion | FISH or LOH analysis | Defines oligodendroglioma diagnosis; predicts response to PCV chemotherapy; associated with better prognosis |
| MGMT promoter methylation | Methylation-specific PCR or pyrosequencing | Predicts TMZ response in GBM; methylated: median OS ~21 months (Stupp protocol); unmethylated: ~14 months; in elderly GBM, methylation status guides TMZ vs RT decision |
| ATRX loss | Immunohistochemistry | Lost in IDH-mutant astrocytomas (retained in oligodendrogliomas); associated with alternative lengthening of telomeres (ALT) |
| TERT promoter mutation | Sequencing | Present in GBM (IDH-wildtype) and oligodendrogliomas; in context of IDH-wildtype diffuse glioma, TERT mutation classifies as grade 4 GBM regardless of histology |
| CDKN2A/B homozygous deletion | FISH | In IDH-mutant astrocytoma, homozygous deletion upgrades to grade 4 regardless of histologic features |
| EGFR amplification | FISH or NGS | Common in GBM (~40%); in IDH-wildtype diffuse glioma, EGFR amplification classifies as grade 4 GBM; potential therapeutic target |
06 Meningiomas
Meningiomas are the most common primary intracranial tumors overall (~37% of all CNS tumors), arising from arachnoid cap cells. Most are benign (WHO grade 1, ~80%), slow-growing, and affect women more than men (2:1). They express progesterone receptors (may grow during pregnancy). Risk factors include prior cranial irradiation, NF2 (multiple meningiomas), and female sex.
Common Locations & Presentations
| Location | Frequency | Clinical Features | Surgical Considerations |
|---|---|---|---|
| Parasagittal / falcine | ~25% | Leg weakness (parasagittal compression of motor cortex), seizures | Involvement of SSS may preclude complete resection of posterior third (high risk of venous infarction) |
| Convexity | ~20% | Focal deficits related to location, seizures, headache | Usually amenable to complete resection |
| Sphenoid wing | ~20% | Medial: visual loss, cavernous sinus involvement; lateral: temporal swelling, seizures | Medial (clinoidal) meningiomas encase the ICA and involve CN II — high surgical risk |
| Olfactory groove | ~10% | Anosmia (often bilateral), personality changes (frontal lobe compression), Foster Kennedy syndrome (ipsilateral optic atrophy + contralateral papilledema) | Bifrontal craniotomy; preserve anterior cerebral arteries |
| Posterior fossa (CPA, petroclival) | ~10% | CPA: hearing loss, facial numbness, ataxia; Petroclival: multiple cranial neuropathies | Complex approaches (retrosigmoid, transpetrosal); high morbidity |
| Tuberculum sellae / planum sphenoidale | ~5% | Visual loss (compression of optic chiasm/nerves), often bilateral | Endoscopic endonasal or craniotomy approach; visual recovery depends on chronicity of compression |
Simpson Grading of Meningioma Resection
| Grade | Extent of Resection | 10-Year Recurrence |
|---|---|---|
| I | Complete resection including dural attachment and abnormal bone | ~9% |
| II | Complete resection with coagulation of dural attachment | ~19% |
| III | Complete resection without dural coagulation or extradural excision (e.g., invaded sinus) | ~29% |
| IV | Subtotal resection (tumor left in situ) | ~44% |
| V | Biopsy only (decompression) | N/A |
WHO Grading
Grade 1 (benign): ~80% of meningiomas; typical histologic subtypes (meningothelial, fibrous, transitional, psammomatous); slow growth; surgery alone is usually curative. Grade 2 (atypical): ~15–20%; defined by increased mitotic activity (4–19 mitoses per 10 HPF), brain invasion, or 3+ of 5 features (sheeting architecture, prominent nucleoli, high cellularity, small cell change, necrosis); higher recurrence rate; consider adjuvant radiation after subtotal resection. Grade 3 (anaplastic/malignant): ~1–3%; ≥20 mitoses per 10 HPF, or frank anaplasia; aggressive behavior with brain invasion and potential metastasis; adjuvant radiation recommended regardless of resection extent.
07 Pituitary Tumors
Pituitary adenomas account for ~15% of intracranial tumors. They are classified by size (microadenoma <10 mm vs macroadenoma ≥10 mm) and by hormonal activity (functioning vs non-functioning).
Functioning Adenomas
| Type | Hormone | Frequency | Clinical Features | First-Line Treatment |
|---|---|---|---|---|
| Prolactinoma | Prolactin | ~40% | Amenorrhea, galactorrhea, infertility (women); decreased libido, gynecomastia (men) | Medical — dopamine agonists (cabergoline preferred over bromocriptine); surgery only if medication intolerant or resistant |
| Somatotroph (GH) | Growth hormone | ~20% | Acromegaly (adults): coarsened facial features, enlarged hands/feet, OSA, diabetes, cardiomyopathy; Gigantism (children, before growth plate closure) | Surgical — transsphenoidal resection; medical if incomplete resection (somatostatin analogs: octreotide, lanreotide; GH receptor antagonist: pegvisomant) |
| Corticotroph (ACTH) | ACTH | ~5–10% | Cushing's disease: central obesity, moon facies, buffalo hump, striae, HTN, diabetes, osteoporosis, proximal myopathy | Surgical — transsphenoidal resection; bilateral adrenalectomy for refractory cases (risk of Nelson syndrome) |
| Thyrotroph (TSH) | TSH | <1% | Secondary hyperthyroidism | Surgical |
Non-Functioning Adenomas
Comprise ~30–40% of pituitary tumors. Typically present as macroadenomas with mass effect: bitemporal hemianopsia (compression of the optic chiasm from below — superior temporal fibers are compressed first), headache, and hypopituitarism (compression of the normal gland — typically GH lost first, then LH/FSH, TSH, ACTH, and prolactin last). Elevated prolactin may occur due to stalk effect (compression of the infundibulum preventing dopamine from reaching the anterior pituitary, causing mild prolactin elevation, typically <100 ng/mL — in contrast, prolactinomas cause prolactin levels proportional to tumor size: macroadenoma prolactinomas usually >200 ng/mL).
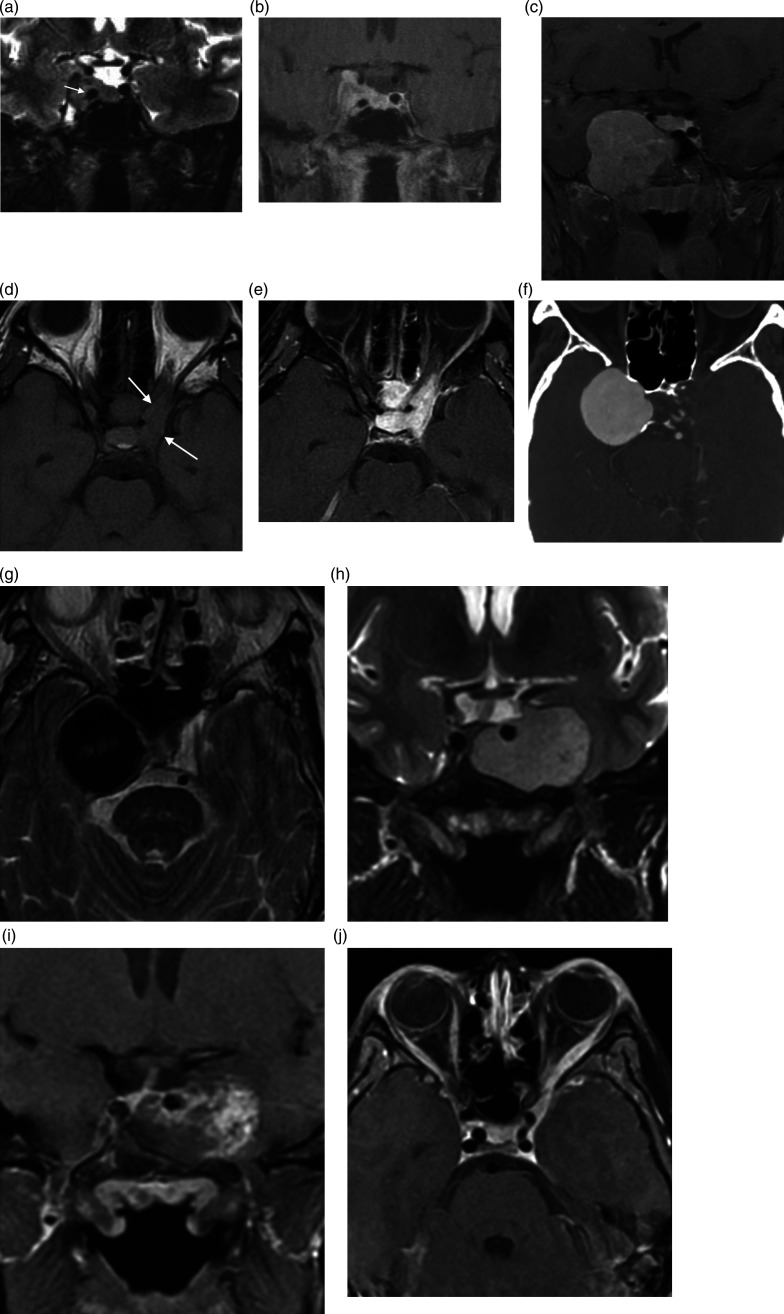
Transsphenoidal Surgery
The endoscopic endonasal transsphenoidal approach is the standard surgical technique for pituitary tumors. The approach traverses the nasal cavity → sphenoid ostium → sphenoid sinus → sellar floor (bone is thinned or removed) → dura is opened → tumor is removed. Key anatomic landmarks: the sphenoid septum (may be deflected), carotid prominences (lateral walls of the sphenoid sinus — must be identified to avoid ICA injury), opticocarotid recesses, and the sellar floor. Reconstruction of the sellar defect after tumor removal uses abdominal fat graft, fascia lata or synthetic dural substitute, and a nasoseptal flap (Hadad-Bassagasteguy flap) to prevent CSF leak.
Acute hemorrhage or infarction of a pituitary adenoma. Presents with sudden severe headache, visual loss (optic chiasm compression), ophthalmoplegia (cavernous sinus involvement), and potentially adrenal crisis (acute ACTH deficiency). Management: immediate IV stress-dose steroids (hydrocortisone 100 mg bolus, then 50 mg q8h), endocrine assessment, and urgent transsphenoidal decompression if visual deterioration or decreased consciousness is present. Visual outcomes are better with surgery within 7 days of onset (PMID: 21381868).
08 Metastatic Brain Tumors
Brain metastases are the most common intracranial tumors overall, outnumbering primary brain tumors ~10:1. The most common primary sources are lung (~40–50%), breast (~15–25%), melanoma (~5–20%), renal cell carcinoma, and colorectal cancer. Metastases tend to occur at the gray-white matter junction (hematogenous spread via end-arteries). Distribution follows blood flow: ~80% cerebral hemispheres, ~15% cerebellum, ~5% brainstem.
Management
Treatment decisions depend on number and size of metastases, performance status, controlled vs uncontrolled systemic disease, and histology. Options include:
Single metastasis, good KPS (≥70), controlled systemic disease: surgical resection + SRS to cavity (or SRS alone if ≤3 cm and surgically inaccessible). Limited metastases (2–4), all ≤3 cm: SRS alone (avoid WBRT if possible to preserve cognition). Multiple metastases (>4) or poor KPS: WBRT (hippocampal avoidance + memantine) or best supportive care. Radiosensitive histology (SCLC, lymphoma, germ cell): WBRT or chemo may be first-line. Melanoma or RCC (radioresistant): SRS preferred over WBRT; immunotherapy (checkpoint inhibitors) may have intracranial activity.
Surgical resection: indicated for single, accessible metastasis with good performance status and controlled systemic disease; also for tissue diagnosis when unknown primary, and for large symptomatic lesions causing mass effect. The landmark Patchell trial demonstrated that surgery + WBRT was superior to WBRT alone for single brain metastases (Patchell 1990, PMID: 2405271).
Stereotactic radiosurgery (SRS): single high-dose focused radiation (Gamma Knife, CyberKnife, LINAC-based); effective for 1–4 metastases, each ≤3 cm; increasingly used for more lesions. Local control rates ~80–90%. SRS to the resection cavity after surgery reduces local recurrence.
Whole brain radiation therapy (WBRT): 30 Gy in 10 fractions; used for multiple metastases (>4), leptomeningeal disease, or poor performance status. Associated with cognitive decline. Hippocampal avoidance WBRT + memantine can mitigate cognitive toxicity (NRG CC001, PMID: 31454575).
09 Posterior Fossa Tumors
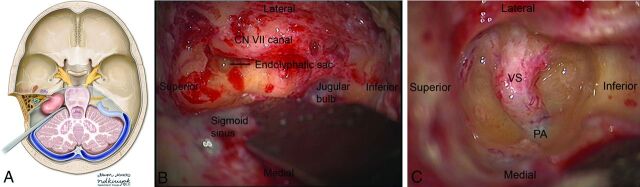
Vestibular Schwannoma (Acoustic Neuroma)
Benign tumor arising from Schwann cells of the vestibular portion of CN VIII, typically at the internal auditory canal (IAC). Presents with unilateral sensorineural hearing loss (most common symptom, insidious onset), tinnitus, and disequilibrium. Large tumors compress CN V (facial numbness) and CN VII, and may cause brainstem compression and hydrocephalus. Bilateral vestibular schwannomas are pathognomonic for NF2. MRI shows a enhancing CPA mass with extension into the IAC ("ice cream cone" appearance).
Management options: observation (small, asymptomatic, elderly patients — serial MRI); stereotactic radiosurgery (tumors ≤3 cm, tumor control ~95%); microsurgical resection via retrosigmoid, translabyrinthine (sacrifices hearing), or middle fossa approach (preserves hearing for small tumors). The goal is tumor control with preservation of facial nerve function (House-Brackmann grade I–II).
Medulloblastoma
The most common malignant brain tumor in children (~20% of pediatric brain tumors). Arises in the posterior fossa — from the cerebellar vermis in children (midline), from the cerebellar hemispheres in adults. WHO grade 4 embryonal tumor. Molecular subgroups (2021 WHO): WNT-activated (best prognosis, ~90% OS), SHH-activated (TP53-wildtype vs TP53-mutant), Group 3 (worst prognosis, often MYC-amplified), and Group 4 (most common). Presents with signs of obstructive hydrocephalus (headache, vomiting, ataxia, papilledema) and cerebellar signs. Can disseminate via CSF (drop metastases to the spine). Treatment: maximal safe resection → craniospinal irradiation (CSI) + boost → adjuvant chemotherapy (vincristine, cisplatin, cyclophosphamide).
Hemangioblastoma
Benign (WHO grade 1), highly vascular tumor, most commonly in the cerebellar hemispheres (posterior fossa). ~25% are associated with von Hippel-Lindau (VHL) syndrome (autosomal dominant; also associated with retinal hemangioblastomas, clear-cell renal cell carcinoma, pheochromocytoma, and pancreatic cysts/neuroendocrine tumors). MRI characteristically shows a cystic lesion with an enhancing mural nodule. The nodule is the tumor — the cyst wall is non-neoplastic (only the nodule needs to be resected). Key surgical principle: these are hypervascular; preoperative angiography and embolization may be considered for large tumors. Associated with polycythemia (tumor secretes erythropoietin).
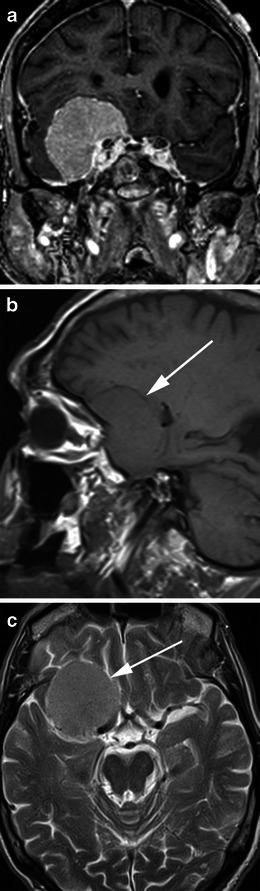
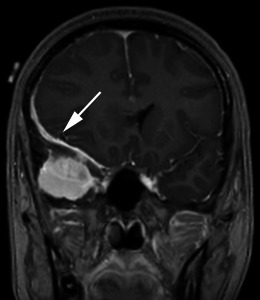
Craniopharyngioma
A benign (WHO grade 1) but locally aggressive epithelial tumor arising from Rathke's pouch remnants in the sellar/suprasellar region. Bimodal age distribution: children (5–15 years) and adults (45–65 years). Two histologic subtypes: adamantinomatous (more common in children; calcifications, cystic with "machinery oil" fluid; CTNNB1/beta-catenin mutations) and papillary (almost exclusively in adults; rarely calcified; BRAF V600E mutations — amenable to targeted therapy). Presents with visual loss (optic chiasm compression), endocrinopathy (panhypopituitarism, growth failure in children, diabetes insipidus), and headache. Surgery (transsphenoidal or transcranial) aims for gross total resection but must be balanced against hypothalamic injury risk (hypothalamic obesity, severe memory and behavioral deficits are devastating — subtotal resection + radiation may be preferred when hypothalamic involvement is significant). Recurrence rate after GTR: ~10–20%; after STR without radiation: ~50–70%.
Primary CNS Lymphoma (PCNSL)
Diffuse large B-cell lymphoma arising within the CNS, without systemic involvement at diagnosis. Increased incidence in immunocompromised patients (HIV/AIDS, transplant recipients) but also occurs in immunocompetent patients (median age 60). MRI: typically deep-seated (periventricular), homogeneously enhancing (in immunocompetent), may be multifocal. Key principle: suspected PCNSL should NOT be resected — surgical role is limited to biopsy for tissue diagnosis. Hold steroids before biopsy if possible (steroids cause rapid tumor lysis and may render biopsy non-diagnostic — the "vanishing tumor"). Treatment: high-dose methotrexate-based chemotherapy ± whole brain radiation; median survival ~3–5 years with treatment.
10 Intracranial Aneurysms & Subarachnoid Hemorrhage
Intracranial aneurysms are present in ~2–3% of the general population. Most are saccular ("berry") aneurysms at arterial bifurcations in the Circle of Willis. Risk factors for rupture: size (>7 mm significantly increases risk), location (posterior circulation, PComm), hypertension, smoking, family history, and conditions such as autosomal dominant polycystic kidney disease and Ehlers-Danlos type IV.
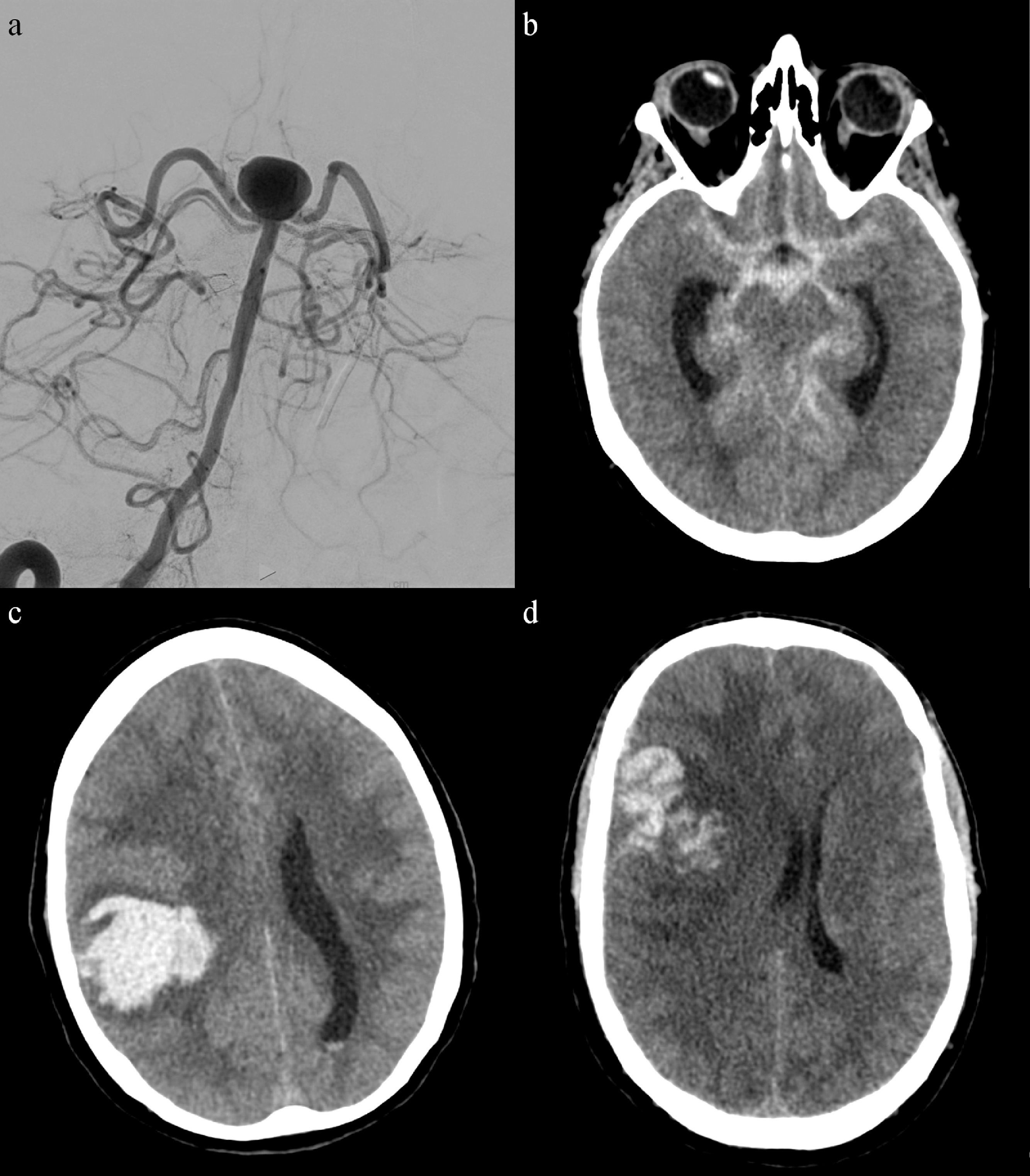
Subarachnoid Hemorrhage (SAH)
Rupture of an intracranial aneurysm causes SAH in ~85% of non-traumatic cases. Classic presentation: "worst headache of my life" (thunderclap headache), with or without loss of consciousness, nausea, meningismus, photophobia, and focal neurologic deficits. Mortality is ~30–40% overall; ~15% die before reaching the hospital.
Grading Scales
| Hunt-Hess Scale | ||
|---|---|---|
| Grade | Clinical Findings | Mortality |
| I | Asymptomatic or mild headache, slight nuchal rigidity | ~1% |
| II | Moderate to severe headache, nuchal rigidity, cranial nerve palsy (e.g., CN III from PComm aneurysm) | ~5% |
| III | Drowsiness, confusion, mild focal deficit | ~19% |
| IV | Stupor, moderate to severe hemiparesis, possible early decerebrate rigidity | ~42% |
| V | Deep coma, decerebrate rigidity, moribund appearance | ~77% |
| Modified Fisher Scale (CT-based, predicts vasospasm risk) | ||
|---|---|---|
| Grade | CT Findings | Vasospasm Risk |
| 0 | No SAH or IVH | Low |
| 1 | Thin SAH, no IVH | Low (~24%) |
| 2 | Thin SAH with IVH | Moderate (~33%) |
| 3 | Thick SAH, no IVH | High (~33%) |
| 4 | Thick SAH with IVH | Highest (~40%) |
| WFNS Scale (based on GCS and focal deficits) | ||
|---|---|---|
| Grade | GCS | Motor Deficit |
| I | 15 | Absent |
| II | 13–14 | Absent |
| III | 13–14 | Present |
| IV | 7–12 | Present or absent |
| V | 3–6 | Present or absent |
SAH Management
Secure the aneurysm early (within 24 hours of admission when feasible) to prevent rebleeding (rebleeding risk is highest in the first 24 hours, ~4% on day 1, then ~1.5%/day for the first 2 weeks).
Clipping vs coiling: The ISAT trial showed improved outcomes at 1 year with endovascular coiling vs surgical clipping for ruptured aneurysms suitable for either treatment (ISAT, PMID: 12414204). Clipping preferred for: MCA aneurysms, aneurysms with broad necks or branches arising from the dome, associated hematoma requiring evacuation. Coiling preferred for: posterior circulation aneurysms, elderly patients, poor-grade SAH.
Vasospasm (days 3–14, peak days 7–10): cerebral arterial vasospasm is the leading cause of morbidity in SAH survivors. Nimodipine 60 mg PO/NG q4h for 21 days reduces poor outcomes (does not prevent angiographic vasospasm but improves outcomes — PMID: 6498626). Monitor with transcranial Doppler (TCD) — MCA mean velocity >120 cm/s suggests vasospasm, >200 cm/s severe vasospasm. If symptomatic vasospasm (delayed cerebral ischemia, DCI): first-line = induced hypertension (euvolemia, target SBP 180–220 mmHg); if refractory, intraarterial verapamil or balloon angioplasty.
Hydrocephalus: acute (from blood obstructing CSF drainage) — treat with EVD. Chronic hydrocephalus (~20% of SAH patients) may require VP shunt.
Unruptured Intracranial Aneurysms — Management
The decision to treat an unruptured aneurysm balances the natural history risk of rupture against the procedural risk of treatment. Risk factors for rupture (PHASES score): Population (Japanese/Finnish highest risk), Hypertension, Age (>70 increases risk), Size (≥7 mm significantly higher risk; <7 mm AComm/PComm also higher risk than other locations), Earlier SAH (from another aneurysm), and Site (posterior circulation, PComm highest risk). The ISUIA (International Study of Unruptured Intracranial Aneurysms) provided key natural history data: for anterior circulation aneurysms <7 mm without prior SAH, the 5-year rupture risk is ~0%; however, this has been debated, and many small aneurysms do rupture. Treatment options: microsurgical clipping (definitive, durable, low retreatment rate but requires craniotomy), endovascular coiling (less invasive, higher retreatment rate ~17% at 5 years), flow diverter stents (Pipeline Embolization Device — for large, wide-necked ICA aneurysms; promotes endoluminal reconstruction; requires dual antiplatelet therapy for 6+ months), or observation with serial imaging (for small, asymptomatic aneurysms in low-risk locations in elderly patients).
SAH Complications Summary
| Complication | Timing | Incidence | Management |
|---|---|---|---|
| Rebleeding | Highest in first 24 hours | ~4% day 1; ~1.5%/day first 2 weeks | Early aneurysm securing (within 24 hours); short-term aminocaproic acid if delay |
| Vasospasm / DCI | Days 3–14 (peak days 7–10) | Angiographic: ~70%; symptomatic: ~30% | Nimodipine 60 mg q4h; induced hypertension; intraarterial vasodilators; balloon angioplasty |
| Acute hydrocephalus | Hours to days | ~20% | EVD placement; monitor for ventriculitis |
| Chronic hydrocephalus | Weeks to months | ~20% | VP shunt if persistent symptoms |
| Hyponatremia | Days 3–14 | ~30% | Distinguish SIADH (euvolemic — fluid restrict) from cerebral salt wasting (hypovolemic — replace with NS or hypertonic saline); CSW more common in SAH; avoid fluid restriction in SAH (risk of vasospasm) |
| Seizures | Acute: first 24 hours; late: weeks to months | ~5–10% | Short-term prophylaxis (levetiracetam preferred); treat acute seizures aggressively |
| Cardiac dysfunction | Hours to days | ~20–30% (ECG changes) | Neurogenic stunned myocardium; troponin elevation; regional wall motion abnormalities; usually self-limited; echo to guide fluid management |
11 Arteriovenous Malformations & Cavernous Malformations
Arteriovenous Malformations (AVMs)
AVMs are congenital tangles of abnormal arteries and veins without an intervening capillary bed, resulting in high-flow arteriovenous shunting. The nidus is the core of the AVM. Annual hemorrhage risk is ~2–4% per year (higher with prior hemorrhage, deep location, deep venous drainage, and associated aneurysms). Presentation: hemorrhage (~50%), seizures (~25%), headache, progressive neurologic deficit ("steal" phenomenon).
Spetzler-Martin Grading
| Feature | Points |
|---|---|
| Size: Small (<3 cm) | 1 |
| Medium (3–6 cm) | 2 |
| Large (>6 cm) | 3 |
| Eloquent cortex: No | 0 |
| Yes (motor, sensory, language, visual, thalamus, hypothalamus, internal capsule, brainstem, cerebellar peduncles, deep cerebellar nuclei) | 1 |
| Deep venous drainage: No (superficial only) | 0 |
| Yes (drainage through deep veins: internal cerebral veins, basal veins of Rosenthal, precentral cerebellar vein) | 1 |
Total score 1–5. Grade I–II: microsurgical resection is first-line (low morbidity, ~96–100% cure rate). Grade III: multimodality treatment — consider embolization + surgery, or radiosurgery for small nidi. Grade IV–V: high surgical risk; observation often preferred unless hemorrhage occurs; ARUBA trial showed better outcomes with medical management alone vs intervention in unruptured AVMs at 33-month follow-up (controversial, debated long-term applicability — ARUBA, PMID: 24444842).
Cavernous Malformations (Cavernomas)
Clusters of thin-walled, endothelium-lined sinusoidal channels without intervening brain parenchyma (no smooth muscle or elastic fibers). Low-flow lesions, angiographically occult ("occult vascular malformations"). Prevalence ~0.5%. MRI appearance: "popcorn" or "mulberry" on T2, with characteristic hemosiderin ring (dark rim on T2/GRE/SWI). Can be sporadic (single) or familial (multiple — autosomal dominant mutations in CCM1/KRIT1, CCM2, CCM3). Annual symptomatic hemorrhage rate ~0.5–1% per lesion per year (higher after initial hemorrhage, ~4.5%/year). Presentation: seizures (most common), hemorrhage, focal neurologic deficits. Treatment: surgical resection for accessible, symptomatic lesions (especially with recurrent hemorrhage or drug-resistant epilepsy); observation for deep-seated or brainstem lesions with minimal symptoms. Radiosurgery is controversial (may reduce rebleeding risk for inoperable brainstem cavernomas).
12 Stroke — Surgical Management
Decompressive Craniectomy for Malignant MCA Infarction
Large MCA territory infarctions may develop life-threatening cerebral edema peaking at 2–5 days (malignant MCA syndrome, mortality up to 80% with medical management alone). Decompressive hemicraniectomy (≥12 cm diameter bone flap removal with duraplasty) within 48 hours reduces mortality from ~78% to ~29% in patients ≤60 years (pooled analysis of DECIMAL, DESTINY, and HAMLET trials — PMID: 17290030). However, many survivors have significant disability (mRS 4). DESTINY II showed benefit even in patients >60 years, though severe disability rates are higher (DESTINY II, PMID: 24694544). Patient/family goals-of-care discussion is essential.
Replacement of the bone flap after decompressive craniectomy, typically performed 3–6 months later once brain swelling has resolved. Options: autologous bone flap (stored in a subcutaneous abdominal pocket or cryopreserved at −80°C) or synthetic implant (custom-made titanium mesh, PEEK, hydroxyapatite, or PMMA). Autologous bone has higher resorption rates (~20–30%, especially in pediatric patients and with prolonged storage), which may necessitate a synthetic cranioplasty. Syndrome of the trephined (sinking skin flap syndrome) — paradoxical neurological decline after large craniectomy due to atmospheric pressure on the brain and altered CSF dynamics; improves dramatically after cranioplasty.
Posterior Fossa Stroke
Cerebellar infarctions and hemorrhages carry a risk of rapid deterioration due to brainstem compression and obstructive hydrocephalus. Indications for suboccipital decompressive craniectomy: GCS decline, radiographic brainstem compression, acute hydrocephalus not responding to EVD alone. EVD placement alone may be sufficient for hydrocephalus without mass effect, but carries a risk of upward herniation if posterior fossa mass effect is not addressed.
Intracerebral Hemorrhage — Surgical Evacuation
The role of surgery for spontaneous ICH remains debated. The STICH trial showed no benefit of early surgical evacuation over medical management for deep ICH (STICH, PMID: 15639681). STICH II suggested possible benefit for superficial lobar hemorrhage (within 1 cm of cortical surface) in patients with GCS ≥9, though results were not statistically significant (STICH II, PMID: 23726393). MISTIE III evaluated minimally invasive catheter-based evacuation with alteplase — did not meet primary endpoint but subgroup analysis suggested benefit when ≥70% of clot is removed. Cerebellar hemorrhage >3 cm with brainstem compression is a class I indication for surgical evacuation.
Carotid Endarterectomy (CEA) & Stenting (CAS)
CEA is indicated for symptomatic carotid stenosis ≥50% (NASCET criteria — NASCET, PMID: 1852179) and asymptomatic carotid stenosis ≥60–70% in good surgical candidates with >5-year life expectancy (ACAS/ACST). CAS is an alternative for patients with hostile neck anatomy, prior neck radiation, or high surgical risk. The procedure should be performed within 2 weeks of the index event for symptomatic stenosis for maximum benefit.
CEA Surgical Technique
The patient is positioned supine with the head turned to the contralateral side. An incision is made along the anterior border of the sternocleidomastoid. The common carotid artery, internal carotid artery, and external carotid artery are exposed and controlled. Systemic heparin is given (5,000–8,000 units, ACT >250). Clamps are placed on the ICA, CCA, and ECA. An arteriotomy is made and the atherosclerotic plaque is dissected from the vessel wall in the endarterectomy plane (media-adventitia). The distal endpoint is carefully feathered to avoid a flap. Closure: primary closure or patch angioplasty (Dacron or bovine pericardium — patch closure reduces restenosis and perioperative stroke risk). A shunt (Javid, Pruitt-Inahara) may be placed during clamping to maintain cerebral perfusion — indications include: stump pressure <40–50 mmHg, contralateral carotid occlusion, EEG changes during clamping, or surgeon preference (routine shunting). Postoperative monitoring: frequent neurologic checks (stroke), blood pressure management (avoid hypertension → reperfusion hemorrhage; avoid hypotension → ischemia), and neck hematoma surveillance (expanding hematoma = emergent OR for wound exploration — can cause airway compromise).
Occurs in ~1–3% of patients after CEA or CAS, typically 2–7 days postoperatively. Results from impaired autoregulation in a chronically hypoperfused hemisphere that is suddenly exposed to normal perfusion pressures after revascularization. Presents with: ipsilateral headache (most common), seizures, focal neurologic deficits, and potentially intracerebral hemorrhage (most feared complication, ~0.5%). Risk factors: high-grade stenosis, contralateral occlusion, poor collateral circulation, and postoperative hypertension. Prevention and treatment: strict blood pressure control (SBP <120–140 mmHg), seizure prophylaxis if symptoms develop, and avoidance of antiplatelet/anticoagulation if hemorrhage occurs.
Endovascular Thrombectomy for Acute Ischemic Stroke
Mechanical thrombectomy for large-vessel occlusion (LVO) stroke is one of the most impactful advances in neurosurgery/neurointerventional radiology. Five landmark trials in 2015 (MR CLEAN, ESCAPE, EXTEND-IA, SWIFT PRIME, REVASCAT) demonstrated overwhelming benefit of endovascular thrombectomy + IV tPA vs IV tPA alone for anterior circulation LVO within 6 hours of symptom onset. The DAWN and DEFUSE 3 trials extended the treatment window to 24 hours in select patients with favorable perfusion imaging (small ischemic core with large penumbra — "mismatch" profile). Stent retriever (Solitaire, Trevo) and aspiration catheter (ADAPT technique, Penumbra) approaches are used. Target: TICI 2b/3 reperfusion (successful recanalization). NNT ~2.6 for reduced disability (one of the lowest in medicine).
13 Degenerative Disc Disease & Cervical Myelopathy
Cervical Radiculopathy
Most commonly caused by posterolateral disc herniation or foraminal stenosis from osteophytes. Presents with arm pain in a dermatomal distribution, sensory changes, and weakness corresponding to the affected nerve root (see Section 02 Nerve Root Localization table). Natural history is favorable — ~75% improve with conservative management (physical therapy, NSAIDs, epidural steroid injections). Surgical indications: failure of 6–12 weeks conservative management, progressive neurologic deficit, or intractable pain.
Anterior Cervical Discectomy and Fusion (ACDF)
The most common cervical spine procedure. Approach through a transverse skin incision on the left side of the neck (to avoid the recurrent laryngeal nerve, which loops lower and is more variable on the right). Platysma is divided, the plane between the carotid sheath laterally and the trachea/esophagus medially is developed. Longus colli muscles are retracted to expose the anterior spine. The disc is removed, osteophytes are drilled, and the neural foramen is decompressed. An interbody graft (structural allograft, PEEK cage, or cage filled with bone graft) is placed, often with an anterior cervical plate for stabilization. Fusion rates >95% for single-level ACDF.
Cervical Myelopathy
Compression of the spinal cord, most commonly from cervical spondylosis (disc-osteophyte complexes, ligamentum flavum hypertrophy, ossification of the posterior longitudinal ligament — OPLL). Presents insidiously with gait difficulty (wide-based, spastic), hand clumsiness (difficulty with buttons, writing), upper motor neuron signs (hyperreflexia, Hoffman sign, Babinski sign, clonus), and sensory changes. Natural history is progressive — surgery is recommended for moderate-to-severe myelopathy or progressive symptoms.
Surgical approaches: Anterior — ACDF (1–2 levels), anterior cervical corpectomy and fusion (ACCF, for retrovertebral compression), or multi-level ACDF. Posterior — laminectomy with lateral mass fusion (for ≥3 levels, maintained lordosis), or laminoplasty (open-door or French-door technique — expands the canal without fusion, preserves motion; best for 3–5 levels with maintained lordosis). Anterior approach is preferred for 1–2 levels or when the compressive pathology is anterior (disc, OPLL). Posterior approach is preferred for ≥3 levels with maintained lordosis. Combined anterior-posterior surgery may be needed for severe multilevel disease with kyphosis.
Cervical Disc Arthroplasty
Motion-preserving alternative to ACDF for single or two-level cervical radiculopathy or myelopathy. Multiple FDA-approved devices (Prestige LP, ProDisc-C, Mobi-C). Contraindications include significant facet arthropathy, instability, and osteoporosis. Long-term data shows non-inferiority to ACDF with potential reduction in adjacent segment disease rates.
Ossification of the Posterior Longitudinal Ligament (OPLL)
Progressive ossification of the PLL causes cervical myelopathy, predominantly in East Asian populations. Classification (by CT morphology): continuous (worst prognosis, extends across multiple segments), segmental, mixed, and localized. OPLL occupying >60% of the canal diameter is generally considered an indication for surgery. Anterior surgery (corpectomy) directly removes the compressive OPLL but carries higher risk of CSF leak (the dura may be adherent to or ossified with the OPLL — "dural ossification"). Posterior surgery (laminoplasty or laminectomy with fusion) indirectly decompresses by allowing the cord to drift posteriorly — generally preferred for multilevel OPLL in patients with maintained lordosis. For OPLL extending beyond the vertebral body margin ("hill-shaped"), anterior approaches may be needed regardless.
Increased mechanical stress on segments adjacent to a cervical or lumbar fusion. Symptomatic ASD occurs at a rate of ~2–3% per year after ACDF. Risk factors: pre-existing degenerative changes at adjacent levels, multilevel fusion (greater stress transfer), sagittal malalignment. ASD is a major argument for disc arthroplasty (motion preservation), though whether arthroplasty truly reduces ASD incidence remains debated. Treatment: extension of fusion or disc replacement at the affected adjacent level.
14 Lumbar Stenosis & Spondylolisthesis
Lumbar Spinal Stenosis
Narrowing of the spinal canal, lateral recesses, or neural foramina, most commonly from degenerative changes (facet hypertrophy, ligamentum flavum thickening, disc bulging). Classic presentation: neurogenic claudication — bilateral leg pain, heaviness, and numbness with walking, relieved by sitting or forward flexion (increased canal diameter in flexion). Must be distinguished from vascular claudication (pain with walking, relieved by standing still, absent pedal pulses, ABI <0.9).
Conservative management: physical therapy, epidural steroid injections. The SPORT trial showed that surgical decompression (laminectomy) provides significant benefit over non-operative treatment at 2 and 4 years, with some crossover benefit at 8 years (SPORT for stenosis, PMID: 18299459). Standard procedure: lumbar laminectomy (removal of the lamina and ligamentum flavum to decompress the thecal sac and nerve roots). Fusion is added when there is associated instability or spondylolisthesis.
Lumbar Disc Herniation
Most commonly at L4–L5 and L5–S1. The posterolateral direction of herniation compresses the traversing nerve root. Surgical indications: cauda equina syndrome (emergency), progressive neurologic deficit, or failure of 6 weeks of conservative management. Standard procedure: microdiscectomy (microscope or loupe-assisted, hemilaminotomy, removal of the herniated disc fragment). Success rates ~85–95% for leg pain relief. Recurrence rate ~5–15%.
Spondylolisthesis
Forward slippage of one vertebral body on another. Classification by Meyerding grade: Grade I = 0–25%, Grade II = 25–50%, Grade III = 50–75%, Grade IV = 75–100%, Grade V (spondyloptosis) = >100%.
| Type | Mechanism | Level | Key Features |
|---|---|---|---|
| Isthmic (Type II) | Pars interarticularis defect (spondylolysis), stress fracture | L5–S1 most common | Most common in young athletes (gymnasts, football linemen); bilateral pars defects allow forward slippage |
| Degenerative (Type III) | Facet joint arthropathy and disc degeneration | L4–L5 most common | Most common type in adults >50; associated with stenosis; pars is intact |
| Dysplastic (Type I) | Congenital deficiency of superior S1 or inferior L5 facets | L5–S1 | Higher risk of high-grade slippage in children |
| Traumatic (Type IV) | Acute fracture of the neural arch (not pars) | Variable | High-energy injury |
| Pathologic (Type V) | Bone disease (tumor, infection, metabolic) | Variable | Rare |
Surgical management: decompression alone may suffice for low-grade degenerative spondylolisthesis with stenosis, but fusion is typically added (posterior lumbar interbody fusion — PLIF, or transforaminal lumbar interbody fusion — TLIF). The SLIP II trial is evaluating the role of fusion for grade I degenerative spondylolisthesis. Isthmic spondylolisthesis failing conservative treatment is managed with posterolateral or interbody fusion. High-grade slips (III–V) require instrumented fusion with reduction consideration.
Lumbar Fusion Approaches
| Approach | Technique | Advantages | Risks/Limitations |
|---|---|---|---|
| PLIF (posterior lumbar interbody fusion) | Bilateral laminectomy, bilateral cage insertion through the disc space from posterior | Direct neural decompression; good interbody fusion rates | More neural retraction than TLIF; risk of dural tear, nerve root injury |
| TLIF (transforaminal lumbar interbody fusion) | Unilateral facetectomy, oblique cage insertion through Kambin's triangle | Less neural retraction (unilateral approach); can be done MIS | Risk of exiting nerve root injury; learning curve for MIS technique |
| ALIF (anterior lumbar interbody fusion) | Retroperitoneal approach (vascular surgeon assist at L5–S1), large anterior cage | Large cage restores lordosis and disc height; no posterior muscle dissection; excellent fusion rates | Vascular injury (iliac vessels), retrograde ejaculation (presacral plexus), visceral injury; requires second stage for posterior fixation if supplemental instrumentation needed |
| XLIF/LLIF (lateral lumbar interbody fusion) | Lateral retroperitoneal, transpsoas approach | Large cage for coronal and sagittal correction; indirect decompression | Lumbar plexus injury (~20% transient thigh weakness/numbness); cannot access L5–S1; requires neuromonitoring |
| OLIF (oblique lateral interbody fusion) | Oblique retroperitoneal approach, anterior to the psoas | Avoids lumbar plexus (no transpsoas dissection); can access L5–S1 | Vascular injury (segmental vessels); sympathetic chain injury; less familiar to many surgeons |
15 Spinal Cord Tumors
Classification by Compartment
| Compartment | Common Tumors | Clinical Features | Surgical Principles |
|---|---|---|---|
| Extradural (~55%) | Metastases (most common), lymphoma, primary vertebral tumors (chordoma, osteosarcoma, giant cell tumor) | Pain (often first symptom), progressive myelopathy, radiculopathy; metastatic cord compression is a surgical emergency | Decompression (anterior or posterior) + stabilization; radiation for radiosensitive tumors; NOMS framework guides management |
| Intradural extramedullary (~40%) | Meningioma, schwannoma/neurofibroma (nerve sheath tumors) | Meningiomas: thoracic predilection, middle-aged women; Schwannomas: "dumbbell" shape extending through neural foramen | Gross total resection usually achievable through posterior approach with microsurgical technique; good prognosis |
| Intramedullary (~5%) | Ependymoma (most common in adults), astrocytoma (most common in children), hemangioblastoma | Central cord symptoms (cape-like sensory loss), progressive myelopathy, pain | Ependymoma: well-circumscribed, GTR often achievable with good outcomes; Astrocytoma: infiltrative, GTR usually not possible without significant morbidity — biopsy + debulk + radiation |
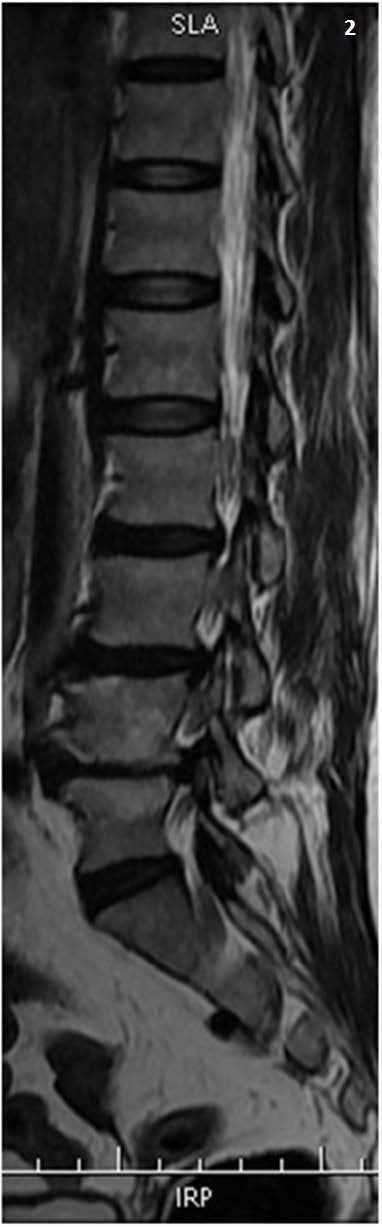
Metastatic Spinal Cord Compression (MSCC)
A neurosurgical/oncologic emergency. Most commonly from lung, breast, prostate, kidney, and myeloma. Presents with progressive back pain (worse supine, worse with Valsalva), followed by weakness, sensory level, and bowel/bladder dysfunction. The Tokuhashi score helps predict prognosis and guide operative vs palliative approach. The NOMS framework (Neurologic, Oncologic, Mechanical instability, Systemic disease) guides decision-making. The Patchell randomized trial demonstrated that direct decompressive surgery + RT was superior to RT alone for maintaining ambulatory status in MSCC (Patchell 2005, PMID: 15689584).
Evaluates mechanical instability in spinal metastases. Components: location (junctional = 3, mobile = 2, semi-rigid = 1, rigid = 0), mechanical pain (yes = 3, occasional = 1, pain-free = 0), bone lesion type (lytic = 2, mixed = 1, blastic = 0), alignment (subluxation = 4, de novo deformity = 2, normal = 0), vertebral body collapse (>50% = 3, <50% = 2, no collapse with >50% involved = 1, none = 0), posterolateral element involvement (bilateral = 3, unilateral = 1, none = 0). Score 0–6: stable; 7–12: indeterminate (surgical consultation); 13–18: unstable (surgical stabilization recommended).
16 Traumatic Brain Injury Emergency
Epidural Hematoma (EDH)
Arterial bleeding between the skull and dura, most commonly from rupture of the middle meningeal artery following a temporal bone fracture. Classic (but not always present) presentation: loss of consciousness → lucid interval → rapid deterioration with contralateral hemiparesis and ipsilateral pupil dilation (uncal herniation). CT shows a biconvex (lenticular) hyperdense collection that does not cross suture lines (dura is tightly adherent at sutures). Surgical indication: thickness >15 mm, midline shift >5 mm, or neurologic deterioration. Treatment: emergent craniotomy with hematoma evacuation and coagulation of the middle meningeal artery. Prognosis is excellent with prompt surgery (mortality <5% for uncomplicated EDH with early intervention).
Acute Subdural Hematoma (aSDH)
Venous bleeding from torn bridging veins between the cortex and dural sinuses, or from cortical arterial laceration. More common and more lethal than EDH. Associated with higher-energy mechanisms and often with underlying brain injury. CT shows a crescent-shaped hyperdense collection conforming to the brain surface, crossing suture lines. Surgical indications (BTF): thickness >10 mm, midline shift >5 mm, or GCS decline by ≥2 points. Treatment: emergent large trauma craniectomy/craniotomy with hematoma evacuation. Mortality remains high (~40–60%) due to associated parenchymal injury. Time from injury to surgery is critical — "the 4-hour rule" — mortality increases sharply when surgery is delayed beyond 4 hours (PMID: 16534088).
Chronic Subdural Hematoma (cSDH)
Occurs primarily in elderly, anticoagulated, or brain-atrophied patients. Develops over weeks following minor or unrecalled trauma. Bridging veins are stretched over the atrophied brain and tear easily. The hematoma develops a neomembrane with fragile capillaries that repeatedly bleed. CT shows a crescent-shaped hypodense or isodense collection (hyperdense if acute-on-chronic). Presentation: headache, cognitive decline, gait disturbance, hemiparesis. Treatment: burr hole drainage (1–2 burr holes with irrigation and subdural drain placement for 24–48 hours) — success rate ~80–90%. Craniotomy for recurrence, organized/loculated collections, or acute-on-chronic SDH. Middle meningeal artery embolization (MMAE) is an emerging treatment for preventing recurrence — the EMBOLISE trial showed significantly reduced recurrence and repeat surgery (EMBOLISE, PMID: 37856610).
Diffuse Axonal Injury (DAI)
Caused by rotational acceleration-deceleration forces shearing white matter axons. Grades: Grade 1 — gray-white matter junction (frontal, temporal); Grade 2 — corpus callosum (especially splenium); Grade 3 — dorsolateral brainstem (worst prognosis). CT may appear normal or show small punctate hemorrhages; MRI (especially SWI/DWI sequences) is more sensitive. No surgical treatment — management is supportive with ICP management as needed. Prognosis ranges from good recovery (mild DAI) to vegetative state or death (severe DAI with brainstem involvement).
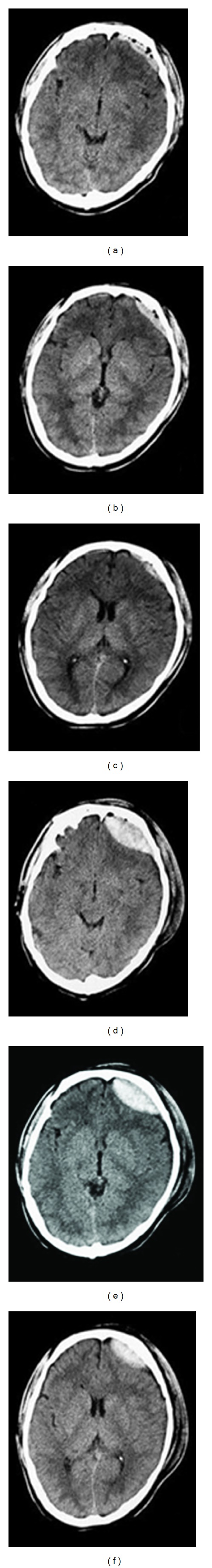
Traumatic Contusions
Bruising of the brain parenchyma, most commonly in the frontal and temporal poles (brain impacts the rough anterior and middle cranial fossa floor). Coup contusions occur at the impact site; contrecoup contusions occur opposite the impact. May expand ("blossom") over 24–48 hours — repeat imaging is essential. Surgical evacuation is considered if the contusion is >50 mL, causes >5 mm midline shift, or the patient deteriorates neurologically.
Skull Fractures
| Type | Features | Management |
|---|---|---|
| Linear | Most common; simple crack in the calvarium; often temporal bone (overlying middle meningeal artery) | Observation if non-displaced and no underlying pathology; if crosses a vascular groove or sinus, monitor for epidural hematoma |
| Depressed | Bone fragment depressed below the level of surrounding skull; open (scalp laceration) or closed | Surgical elevation if: depression > full thickness of skull, open fracture with contamination, underlying hematoma, cosmetic deformity, or neurologic deficit. Elevation within 24 hours for open depressed fractures (infection prevention) |
| Basilar | Fracture of the skull base. Anterior fossa: raccoon eyes (periorbital ecchymosis), CSF rhinorrhea, anosmia. Middle fossa: Battle sign (mastoid ecchymosis), hemotympanum, CSF otorrhea, CN VII/VIII palsy. Posterior fossa: rare, may injure vertebral arteries | Most manage conservatively. CSF leak: 70–80% resolve within 7 days with HOB elevation and stool softeners. Persistent CSF leak (>7 days): lumbar drain; surgical repair if lumbar drain fails. Prophylactic antibiotics for CSF leak are controversial (not routinely recommended) |
| Growing (pediatric) | Dural tear underlying a skull fracture in infants; brain pulsations prevent healing and cause progressive widening ("growing skull fracture" or "leptomeningeal cyst") | Surgical repair of dural defect + cranioplasty; diagnosed by progressive widening on serial imaging |
Penetrating TBI
Most commonly from gunshot wounds. Management: aggressive resuscitation, seizure prophylaxis (7 days), broad-spectrum antibiotics, CT to define projectile trajectory and associated injuries. Surgical indications: accessible hematoma with mass effect, debridement of necrotic tissue and accessible bone/projectile fragments (do NOT probe deep for projectiles), watertight dural closure. Objects in situ should not be removed outside the OR. Vascular imaging (CTA or angiography) is recommended to evaluate for traumatic aneurysm or vascular injury. Mortality is ~90% for bihemispheric, transventricular, or dominant-hemisphere gunshot wounds.
ICP monitoring for GCS ≤8 with abnormal CT. ICP treatment threshold: >22 mmHg. CPP target: 60–70 mmHg. Seizure prophylaxis: levetiracetam or phenytoin for 7 days (prevents early post-traumatic seizures but NOT late epilepsy). Nutrition: full caloric replacement by day 5 (at least by day 7). Avoid hypotension (SBP <100 in ages 50–69; SBP <110 in ages 15–49 or >70). Avoid hyperthermia. Steroids are contraindicated (CRASH trial — increased mortality with methylprednisolone — CRASH, PMID: 15474134).
17 Spinal Trauma & Spinal Cord Injury
ASIA (American Spinal Injury Association) Impairment Scale
| Grade | Definition |
|---|---|
| A — Complete | No motor or sensory function preserved in S4–S5 segments |
| B — Sensory incomplete | Sensory but not motor function preserved below the neurologic level, including S4–S5 |
| C — Motor incomplete | Motor function preserved below neurologic level; more than half of key muscles below the level have grade <3 |
| D — Motor incomplete | Motor function preserved below neurologic level; at least half of key muscles below the level have grade ≥3 |
| E — Normal | Motor and sensory function normal |
Cervical Spine Fractures
Atlas (C1) fractures: Jefferson fracture = burst fracture of the C1 ring from axial loading. The rule of Spence: if total lateral mass overhang >6.9 mm on open-mouth odontoid view, the transverse ligament is likely ruptured (unstable) — requires surgery. Stable Jefferson fractures are treated with a rigid cervical collar or halo vest.
Axis (C2) fractures: Odontoid (dens) fractures — Anderson-D'Alonzo classification: Type I = tip avulsion (stable, rare); Type II = base of the dens (most common, highest nonunion rate ~40% — treat with anterior odontoid screw or posterior C1–C2 fusion); Type III = extends into the C2 body (usually heals with external immobilization). Hangman's fracture = bilateral C2 pars interarticularis fracture (traumatic spondylolisthesis of C2). Levine-Edwards classification: Type I = <3 mm displacement, no angulation (rigid collar); Type II = >3 mm displacement and angulation (halo or surgery); Type IIA = minimal displacement, significant angulation (traction contraindicated — causes distraction; treat with halo or surgery); Type III = associated facet dislocation (requires surgery).
Subaxial Cervical Spine Injury Classification (SLICS)
| Component | Points |
|---|---|
| Morphology: No abnormality | 0 |
| Compression | 1 |
| Burst | +1 |
| Distraction | 3 |
| Rotation/translation | 4 |
| DLC (Disco-Ligamentous Complex): Intact | 0 |
| Indeterminate | 1 |
| Disrupted | 2 |
| Neurologic status: Intact | 0 |
| Root injury | 1 |
| Complete cord injury | 2 |
| Incomplete cord injury | 3 |
| Continuous cord compression with neuro deficit | +1 |
SLICS Score: ≤3 = non-operative; 4 = equivocal; ≥5 = surgical.
Thoracolumbar Injury Classification & Severity Score (TLICS)
| Component | Points |
|---|---|
| Morphology: Compression | 1 |
| Burst | +1 |
| Translational/rotational | 3 |
| Distraction | 4 |
| PLC (Posterior Ligamentous Complex): Intact | 0 |
| Suspected/indeterminate | 2 |
| Injured | 3 |
| Neurologic status: Intact | 0 |
| Nerve root | 2 |
| Cord/conus — complete | 2 |
| Cord/conus — incomplete | 3 |
| Cauda equina | 3 |
TLICS Score: ≤3 = non-operative; 4 = equivocal; ≥5 = surgical.
Cervical Spine Clearance
NEXUS (National Emergency X-Radiography Utilization Study): cervical spine imaging can be deferred if ALL five criteria are met: no midline cervical tenderness, no focal neurologic deficit, normal level of alertness, no intoxication, and no painful distracting injury. Sensitivity ~99% for significant injury.
Canadian C-Spine Rule: more specific than NEXUS. Three steps: (1) Any high-risk factor mandating imaging? (age ≥65, dangerous mechanism, paresthesias in extremities) → if yes, image. (2) Any low-risk factor allowing safe assessment of ROM? (simple rear-end MVC, sitting position in ED, ambulatory at any time, delayed onset of neck pain, absence of midline tenderness) → if no low-risk factors, image. (3) Can patient actively rotate neck 45° left and right? → if yes, no imaging needed.
Obtunded patient clearance: high-quality CT (with multiplanar reformats) is adequate for clearance in many centers. MRI within 48–72 hours is recommended by some guidelines if CT is negative but clinical concern persists (to detect ligamentous injury, disc herniation). Extended collar use in obtunded patients increases complications (skin breakdown, DVT, elevated ICP).
Spinal Shock vs Neurogenic Shock
| Feature | Spinal Shock | Neurogenic Shock |
|---|---|---|
| Definition | Temporary loss of all spinal cord function and reflexes below the level of injury | Hemodynamic instability from loss of sympathetic tone (vasodilation + bradycardia) |
| Onset/Duration | Immediate; lasts hours to weeks (bulbocavernosus reflex return marks end of spinal shock) | Immediate; may persist for 1–3 weeks |
| Clinical features | Flaccid paralysis, areflexia, loss of sensation below the level; inability to assess ASIA grade until spinal shock resolves | Hypotension, bradycardia (or inappropriate lack of tachycardia), warm/vasodilated extremities (unlike hypovolemic shock which is cool/vasoconstricted) |
| Treatment | Supportive; monitor for return of reflexes | IV fluids (first-line); vasopressors if refractory (norepinephrine or phenylephrine; avoid pure beta-agonists which may worsen bradycardia); atropine for symptomatic bradycardia |
18 Deep Brain Stimulation & Functional Procedures
Deep Brain Stimulation (DBS)
DBS involves the stereotactic implantation of electrodes into specific deep brain nuclei, connected to a subcutaneously placed pulse generator (similar to a cardiac pacemaker). The mechanism involves modulation (not ablation) of pathologic neural circuit activity.
| Indication | Target | Key Considerations |
|---|---|---|
| Parkinson's disease | Subthalamic nucleus (STN) or globus pallidus internus (GPi) | For medication-refractory motor fluctuations, dyskinesia, or tremor; STN-DBS allows greater reduction in medications; GPi-DBS may be better for dyskinesia; patient must be levodopa-responsive (except tremor-dominant PD) |
| Essential tremor | Ventral intermediate nucleus of thalamus (VIM) | Dramatic tremor reduction (~80%); unilateral usually sufficient for dominant hand; does not address other features |
| Dystonia | GPi | Best results in DYT1-positive generalized dystonia; improvement may take weeks to months (gradual, unlike PD); generally better in primary than secondary dystonia |
| OCD (humanitarian device exemption) | Ventral capsule/ventral striatum (VC/VS) or anterior limb of internal capsule | For severe, treatment-refractory OCD; response rate ~60% |
Ablative Procedures
With the advent of DBS and MRI-guided focused ultrasound (MRgFUS), radiofrequency ablation has become less common but remains relevant. MRgFUS thalamotomy (Exablate Neuro) is FDA-approved for essential tremor and tremor-dominant PD — incisionless ablation of the VIM nucleus using convergent ultrasound beams under real-time MRI thermometry. Pallidotomy (GPi ablation) is occasionally used for Parkinson's dyskinesia in patients who are not DBS candidates.
Other Functional Procedures
Trigeminal neuralgia: Microvascular decompression (MVD) is the gold-standard surgical treatment for medication-refractory trigeminal neuralgia — a vascular loop (usually the SCA) is separated from the trigeminal nerve root entry zone with a Teflon pad (pain-free rate ~80% at 5 years). Percutaneous procedures (radiofrequency rhizotomy, glycerol rhizolysis, balloon compression) and stereotactic radiosurgery are alternatives for patients who are poor surgical candidates. Hemifacial spasm: MVD (offending vessel, usually AICA or PICA, compressing CN VII at the root exit zone) is the definitive treatment.
Spasticity Management
Intrathecal baclofen pump (ITB): a programmable pump (placed subcutaneously in the abdomen) delivers baclofen directly into the CSF via a catheter in the lumbar intrathecal space. Indicated for severe spasticity from spinal cord injury, cerebral palsy, multiple sclerosis, or stroke that is refractory to oral medications. A screening trial (50–100 mcg intrathecal baclofen via lumbar puncture) must demonstrate ≥1-point reduction on the Ashworth scale before permanent implantation. Complications: catheter malfunction (kinking, migration, disconnection — most common), infection, overdose (flaccidity, respiratory depression, coma — treated with airway support), and withdrawal syndrome (rebound spasticity, hyperthermia, rhabdomyolysis, multiorgan failure, death — treat with oral/enteral baclofen, benzodiazepines, ICU monitoring; can mimic malignant hyperthermia or neuroleptic malignant syndrome).
Selective dorsal rhizotomy (SDR): selective sectioning of dorsal (sensory) rootlets L1–S1 to reduce afferent input driving spasticity. Primarily used in children with spastic diplegic cerebral palsy (ambulatory children ages 3–8 with predominantly lower-extremity spasticity and good underlying strength). Intraoperative EMG is used to identify and selectively cut the rootlets with the most abnormal responses (sustained, spreading activity). Significant reduction in spasticity with preserved motor strength; requires intensive post-operative physiotherapy.
Pain Procedures
Spinal cord stimulation (SCS): epidural electrode placement over the dorsal columns; generates paresthesias that mask pain signals. Indications: failed back surgery syndrome (FBSS), complex regional pain syndrome (CRPS), peripheral neuropathy, refractory angina. Trial phase (temporary electrode) precedes permanent implantation — ≥50% pain relief defines a successful trial. High-frequency (10 kHz) and burst stimulation paradigms provide paresthesia-free analgesia. Motor cortex stimulation (MCS): epidural electrode over the precentral gyrus for refractory central pain syndromes (thalamic pain, post-stroke pain, trigeminal neuropathic pain). Intrathecal drug delivery: morphine, ziconotide (non-opioid calcium channel blocker), or bupivacaine delivered via implanted pump for cancer-related pain or refractory non-malignant pain.
19 Epilepsy Surgery
Epilepsy surgery is indicated for drug-resistant epilepsy (failure of ≥2 appropriate antiseizure medications at adequate doses). Approximately one-third of epilepsy patients are drug-resistant. Presurgical evaluation includes: video-EEG monitoring (seizure semiology and EEG localization), MRI (structural lesion identification — hippocampal sclerosis, cortical dysplasia, tumor, vascular malformation), PET (interictal hypometabolism), neuropsychological testing, WADA test or fMRI (language and memory lateralization), and potentially invasive EEG monitoring (subdural grids, stereo-EEG — SEEG).
Surgical Procedures
| Procedure | Indication | Seizure Freedom Rate | Key Points |
|---|---|---|---|
| Anterior temporal lobectomy (ATL) | Mesial temporal lobe epilepsy (hippocampal sclerosis) | ~65–80% (Engel Class I) | Most effective epilepsy surgery; removes anterior temporal neocortex + amygdala + hippocampus; dominant-side surgery carries higher risk of verbal memory decline |
| Selective amygdalohippocampectomy (SAH) | Mesial temporal epilepsy | ~60–70% | Preserves lateral temporal neocortex; may have lower neuropsychological morbidity than ATL |
| Lesionectomy | Epileptogenic lesion (tumor, cavernoma, FCD) | Variable (~60–80%) | Resection of the lesion plus surrounding epileptogenic cortex |
| Corpus callosotomy | Generalized epilepsy with drop attacks (Lennox-Gastaut) | Palliative (~60–80% reduction in drop attacks) | Anterior two-thirds callosotomy; complete callosotomy for refractory cases (risk of disconnection syndrome) |
| Hemispherectomy / hemisphereotomy | Hemispheric epilepsy syndromes (Rasmussen encephalitis, hemimegalencephaly, Sturge-Weber) | ~70–85% | Functional hemispherectomy (disconnection) preferred over anatomic removal; best outcomes in children with early plasticity |
Neuromodulation for Epilepsy
Vagus nerve stimulation (VNS): left vagus nerve electrode connected to a chest-wall pulse generator; reduces seizure frequency by ~50% in ~50% of patients (palliative, not curative). Mechanism not fully understood — activates brainstem nuclei that modulate cortical excitability. Responsive neurostimulation (RNS, NeuroPace): closed-loop device implanted at the seizure focus that detects abnormal ECoG activity and delivers targeted electrical stimulation to abort seizures. Median seizure reduction ~50–60% at 2 years. Deep brain stimulation of the anterior nucleus of the thalamus (ANT-DBS): SANTE trial showed ~56% seizure reduction at 2 years (SANTE, PMID: 20307616).
Engel Outcome Classification
| Class | Description | Detail |
|---|---|---|
| I — Free of disabling seizures | Seizure-free or only non-disabling auras | Ia: completely seizure-free; Ib: non-disabling simple partial seizures only; Ic: some disabling seizures after surgery but free for ≥2 years; Id: generalized convulsions with AED discontinuation only |
| II — Rare disabling seizures | Almost seizure-free | IIa: initially free then rare seizures; IIb: rare disabling seizures from the start; IIc: seizures occurring upon AED reduction; IId: nocturnal seizures only |
| III — Worthwhile improvement | Meaningful seizure reduction | IIIa: meaningful seizure reduction; IIIb: prolonged seizure-free intervals ≥50% of follow-up |
| IV — No worthwhile improvement | No significant change | IVa: significant seizure reduction; IVb: no appreciable change; IVc: seizures worse |
Temporal Lobe Epilepsy — Mesial Temporal Sclerosis
Mesial temporal sclerosis (MTS) is the most common pathologic substrate of drug-resistant temporal lobe epilepsy. Characterized histologically by hippocampal neuronal loss (especially CA1 and CA3 subfields) and gliosis. MRI findings: hippocampal atrophy (decreased volume on coronal T1), increased T2/FLAIR signal in the hippocampus, and loss of internal hippocampal architecture. Clinical presentation: complex partial seizures (now termed "focal impaired awareness seizures") with an aura (epigastric rising sensation, fear, deja vu), followed by behavioral arrest, oroalimentary automatisms (lip-smacking, chewing), and contralateral hand automatisms with ipsilateral hand dystonia. Scalp EEG shows ipsilateral temporal epileptiform discharges. Surgical treatment (ATL or SAH) is the most effective treatment for drug-resistant TLE — the ERSET trial demonstrated superiority of surgery over continued medical therapy (Engel I outcome: 58% surgery vs 8% medical — ERSET, PMID: 22150006).
20 Hydrocephalus
Classification
Obstructive (non-communicating): blockage within the ventricular system (e.g., aqueductal stenosis, posterior fossa tumor obstructing the fourth ventricle, colloid cyst blocking the foramen of Monro). Communicating: impaired CSF absorption at the arachnoid granulations (post-SAH, post-meningitis, post-hemorrhage) or, rarely, CSF overproduction (choroid plexus papilloma). Ex vacuo: ventriculomegaly from brain parenchymal loss (atrophy) — NOT true hydrocephalus (no elevated pressure).
Treatment Options
Ventriculoperitoneal (VP) shunt: the most common definitive treatment. A ventricular catheter (typically right frontal via Kocher's point or right parieto-occipital via Keen's point) is connected to a one-way pressure-regulated valve, which drains via a distal catheter into the peritoneal cavity. Complications: obstruction (~30% at 1 year — most common cause of shunt failure; proximal catheter occlusion by choroid plexus is the most common site), infection (~5–10%, usually within 6 months; most common organisms: Staphylococcus epidermidis and S. aureus; treatment: shunt removal + external drainage + IV antibiotics for 10–14 days, then new shunt), over-drainage (slit ventricle syndrome, subdural hygromas/hematomas; prevented by programmable valves or antisiphon devices).
Endoscopic third ventriculostomy (ETV): endoscopic fenestration of the floor of the third ventricle, creating a direct communication between the third ventricle and the prepontine cistern, bypassing the aqueduct. Best for obstructive hydrocephalus (aqueductal stenosis). The ETV Success Score (ETVSS) predicts success based on age, etiology, and prior shunt: higher scores predict greater success. ETV may be combined with choroid plexus cauterization (ETV/CPC) in infants.
Normal Pressure Hydrocephalus (NPH)
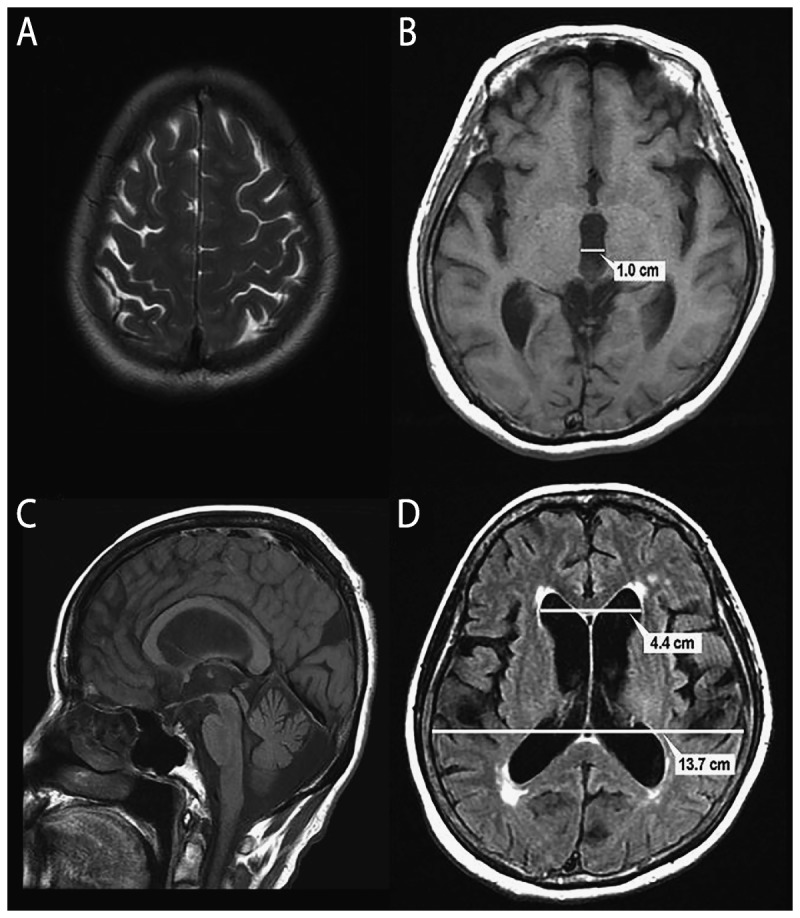
A form of communicating hydrocephalus in elderly patients. Classic triad (Hakim's triad): gait disturbance (wide-based, magnetic, shuffling — earliest and most responsive to treatment), urinary incontinence, and dementia (subcortical pattern — psychomotor slowing; latest and least responsive to treatment). Mnemonic: "wet, wacky, wobbly." Imaging: ventriculomegaly disproportionate to sulcal atrophy (Evans' index = maximal bicaudate diameter / maximal inner skull diameter >0.3). Diagnostic workup: large-volume lumbar puncture (30–50 mL CSF removal) — improvement in gait within 1–4 hours predicts shunt response (~80% positive predictive value). Extended lumbar drainage (3–5 days) is more sensitive. Treatment: VP shunt with a programmable valve (allows adjustment if over- or under-drainage occurs). Gait improves in ~80% of properly selected patients; cognitive improvement is less reliable.
Idiopathic Intracranial Hypertension (Pseudotumor Cerebri)
Elevated ICP without hydrocephalus, mass lesion, or abnormal CSF composition. Predominantly affects obese women of childbearing age. Modified Dandy criteria: symptoms/signs of elevated ICP (headache, papilledema, visual obscurations, CN VI palsy), elevated opening pressure on LP (>25 cmH2O), normal CSF composition, normal neuroimaging (may show empty sella, optic nerve sheath distension, transverse sinus stenosis on MRV). Medical treatment: weight loss, acetazolamide (reduces CSF production; 500 mg BID, titrate up to 2 g/day), topiramate (adjunct — also promotes weight loss). Surgical options for refractory cases or progressive visual loss: optic nerve sheath fenestration (ONSF — protects vision but may not relieve headache) or CSF diversion (VP shunt or LP shunt). Venous sinus stenting is an emerging treatment for patients with documented transverse sinus stenosis and pressure gradient >8 mmHg.
Slit Ventricle Syndrome
A complication of chronic VP shunt over-drainage, most commonly in children. Small slit-like ventricles with intermittent shunt obstruction cause episodic headaches, nausea, and lethargy. CT shows collapsed ventricles. The shunt cannot drain effectively because the choroid plexus and ependyma occlude the ventricular catheter tip. Management: programmable valve with higher opening pressure, antisiphon device, shunt revision with catheter repositioning, subtemporal decompression, or ETV (shunt independence).
21 Pediatric Neurosurgery
Craniosynostosis
Premature fusion of one or more cranial sutures, resulting in abnormal head shape. Incidence ~1 in 2,000 births. Classified by the suture involved:
| Suture | Head Shape | Frequency | Key Features |
|---|---|---|---|
| Sagittal | Scaphocephaly (dolichocephaly — long, narrow) | ~40–55% | Most common single-suture craniosynostosis; M:F 4:1; usually non-syndromic |
| Coronal (unilateral) | Anterior plagiocephaly (ipsilateral forehead flattening, contralateral frontal bossing, "harlequin" deformity on X-ray) | ~20–25% | Must distinguish from positional plagiocephaly (posterior flattening from supine positioning); coronal has ipsilateral brow retrusion |
| Metopic | Trigonocephaly (triangular forehead, hypotelorism) | ~15–20% | A midline metopic ridge is common and benign; true trigonocephaly with hypotelorism requires surgery |
| Lambdoid (unilateral) | Posterior plagiocephaly (true, with ipsilateral occipital flatness and contralateral frontal bossing) | ~2–4% | Rarest; must distinguish from positional (lambdoid: ipsilateral ear displaced inferoposteriorly; positional: ipsilateral ear displaced anteriorly) |
| Multiple sutures | Various: turricephaly, cloverleaf | Variable | Often syndromic (Crouzon, Apert, Pfeiffer, Saethre-Chotzen); higher risk of elevated ICP; may need staged operations |
Surgical treatment: open cranial vault remodeling (typically at 6–12 months) or endoscopic strip craniectomy with post-operative helmet therapy (best if performed before 3–4 months of age). Endoscopic approaches are less invasive with shorter hospital stays but require compliant helmet wear for months.
Chiari Malformations
Chiari I: tonsillar ectopia ≥5 mm below the foramen magnum. May be asymptomatic (incidental finding on MRI). Symptomatic patients present with suboccipital headache (worsened by Valsalva, cough, straining), neck pain, and may develop syringomyelia (cavitation of the spinal cord from altered CSF dynamics). Treatment: posterior fossa decompression (suboccipital craniectomy + C1 laminectomy ± duraplasty). Chiari II: associated with myelomeningocele; hindbrain herniation includes the cerebellar vermis, fourth ventricle, and lower brainstem through the foramen magnum. Chiari III: rare; herniation of posterior fossa contents into an occipitocervical encephalocele.
Myelomeningocele (Open Neural Tube Defect)
The most common open neural tube defect. Results from failure of posterior neural tube closure during the 4th week of gestation. The exposed neural placode is covered by a meningeal sac. ~90% are associated with Chiari II malformation and ~80% develop hydrocephalus requiring shunt placement. Prevention: maternal folic acid supplementation (0.4 mg/day preconception; 4 mg/day if prior affected pregnancy). Postnatal repair within 24–72 hours (closure of the neural placode and overlying layers). Prenatal repair (MOMS trial — PMID: 21306277) at 19–25 weeks gestation showed reduced need for shunting and improved motor outcomes at 30 months but increased risk of preterm delivery.
Tethered Cord Syndrome
The spinal cord is abnormally attached, restricting its normal ascent during growth. Causes: thick filum terminale (most common), lipomyelomeningocele, split cord malformation (diastematomyelia), dermoid/epidermoid, prior myelomeningocele repair. Presentation in children: progressive lower extremity weakness, foot deformity (pes cavus), scoliosis, urologic dysfunction (incontinence, recurrent UTIs). In adults: back/leg pain, progressive lower extremity symptoms, urologic dysfunction. MRI shows a low-lying conus (below L2 in adults) and the tethering lesion. Treatment: surgical untethering (section of the filum terminale or release of the tethering lesion).
Pediatric Brain Tumors — Overview
Brain tumors are the most common solid tumors in children and the leading cause of cancer-related death in pediatrics. Unlike adults, the majority of pediatric brain tumors arise in the posterior fossa (~60%). The four most common types:
| Tumor | Location | Age Peak | Key Features |
|---|---|---|---|
| Pilocytic astrocytoma (WHO 1) | Cerebellum (most common); also optic pathway/hypothalamus (NF1-associated), brainstem | 5–15 years | Most common pediatric brain tumor; cystic with enhancing mural nodule; BRAF fusion (tandem duplication); excellent prognosis after GTR (~95% 10-year OS); observation if GTR achieved |
| Medulloblastoma (WHO 4) | Posterior fossa (vermis in children, hemispheres in adults) | 3–8 years | Most common malignant pediatric brain tumor; CSF dissemination common (spine MRI required preoperatively); treatment: surgery + CSI + chemo (see Section 09) |
| Ependymoma (WHO 2–3) | Fourth ventricle (most common in children) | 1–5 years | Arises from the floor of the fourth ventricle; may extend through the foramen of Luschka (CPA) or foramen of Magendie; GTR is the most important prognostic factor; adjuvant RT for incomplete resection |
| Diffuse midline glioma, H3 K27-altered (WHO 4) | Pons (DIPG), thalamus, spinal cord | 5–10 years | Previously called diffuse intrinsic pontine glioma (DIPG); diagnosed clinically + MRI (biopsy increasingly performed for molecular profiling); no effective treatment; radiation provides transient palliation; median survival ~11 months; dismal prognosis |
Arachnoid Cysts
Congenital, developmental CSF-filled cysts within the arachnoid membrane. Most common location: middle cranial fossa (Sylvian fissure). Usually incidental findings. Most are asymptomatic and require no treatment (serial imaging only). Indications for surgery: symptomatic (headache attributable to the cyst, focal deficit, seizures directly related), growing on serial imaging, or associated hydrocephalus. Surgical options: endoscopic fenestration (preferred — opens the cyst into the basal cisterns), craniotomy with cyst wall excision/marsupialization, or cystoperitoneal shunt (least preferred due to shunt dependence).
22 Craniotomy Approaches
Major Craniotomy Approaches
| Approach | Indications | Key Steps / Landmarks | Structures at Risk |
|---|---|---|---|
| Pterional (frontotemporal) | Most versatile: anterior circulation aneurysms, Sylvian fissure pathology, sellar/parasellar tumors, anterior skull base lesions | Curvilinear incision behind the hairline; temporal muscle reflected; frontotemporal craniotomy with sphenoid wing drilling; open Sylvian fissure for access | Frontal branch of CN VII (runs in the temporoparietal fascia — protect by interfascial or subfascial dissection); MCA branches during Sylvian fissure splitting |
| Suboccipital / retrosigmoid | CPA tumors (vestibular schwannoma, meningioma), trigeminal neuralgia (MVD), posterior fossa pathology | Incision behind the ear; craniotomy posterior to the sigmoid sinus; lateral cerebellar retraction for CPA access | CN VII and VIII (CPA surgery); sigmoid and transverse sinuses; vertebral artery (if approach extended medially); venous air embolism (sitting position) |
| Midline suboccipital | Cerebellar tumors, fourth ventricle tumors (medulloblastoma, ependymoma), Chiari decompression | Midline incision; suboccipital craniotomy ± C1 laminectomy; telovelar approach for fourth ventricle access (avoids vermian splitting) | PICA, occipital sinus, cerebellar mutism (especially in children after vermian manipulation) |
| Bifrontal | Anterior skull base tumors (olfactory groove meningioma, craniopharyngioma), anterior interhemispheric approach | Coronal skin incision; bilateral frontal craniotomy; may require crista galli drilling; basal interhemispheric approach for midline pathology | SSS (must preserve or ligate anterior portion only — posterior third is critical); olfactory nerves; ACA |
| Endoscopic endonasal (transsphenoidal) | Pituitary adenomas, craniopharyngiomas, clival chordomas, CSF rhinorrhea repair, optic nerve decompression | Binostril approach through the nasal cavity, sphenoid sinus, to sella or extended to planum, clivus, or cribriform plate | ICA (lateral walls of sphenoid); optic nerves; CSF leak (nasoseptal flap reconstruction critical) |
| Interhemispheric (transcallosal) | Third ventricle tumors (colloid cyst, craniopharyngioma), lateral ventricle tumors, callosotomy | Parasagittal craniotomy; interhemispheric corridor between the falx and hemisphere; callosotomy for access to the ventricle | SSS, bridging veins (must be preserved or selectively sacrificed), pericallosal arteries, fornix (memory deficits) |
Patient Positioning
Proper head positioning is critical. The Mayfield head clamp (three-pin fixation) rigidly fixes the head for craniotomies. Positions: supine with head turned (pterional, frontal); lateral (park bench) (suboccipital, retrosigmoid); prone (midline suboccipital, posterior spine); sitting (posterior fossa — excellent gravity-assisted retraction, but risk of venous air embolism — requires precordial Doppler, end-tidal CO2 monitoring, and central venous access for air aspiration).
23 Stereotactic & Endoscopic Neurosurgery
Stereotactic Surgery
Stereotactic surgery uses a coordinate system (frame-based or frameless) to precisely target intracranial structures for biopsy, ablation, or electrode placement. Frame-based stereotaxy: a rigid frame (Leksell, CRW, or BRW) is fixed to the skull with pins, providing a Cartesian coordinate system. CT or MRI is obtained with the frame and localizer in place. Target coordinates are calculated. Accuracy ~1–2 mm. Frameless stereotaxy (neuronavigation): uses fiducial markers or anatomic landmarks registered to preoperative imaging (CT, MRI). Systems include optical (infrared cameras tracking reflective spheres on instruments — Medtronic StealthStation, Brainlab) and electromagnetic tracking. Accuracy ~2–3 mm. Used routinely for craniotomy planning, biopsy guidance, and intraoperative orientation.
Stereotactic Brain Biopsy
Indicated when tissue diagnosis is needed and resection is not feasible (deep-seated lesions, eloquent cortex, multiple lesions, lymphoma suspected — steroids should be withheld until biopsy for suspected lymphoma). Diagnostic yield >95% for frame-based biopsies. Complications: hemorrhage (~2–5%, symptomatic ~1%), infection, neurologic deficit. The target should avoid vessels (plan trajectory on contrast-enhanced MRI), ventricles (risk of hemorrhage into the CSF), and eloquent structures.
Endoscopic Neurosurgery
Rigid or flexible endoscopes are used for intraventricular procedures: ETV (see Section 20), tumor biopsy and removal (intraventricular tumors, colloid cysts), cyst fenestration, septum pellucidotomy (for trapped lateral ventricle), and aqueductoplasty. The endoscopic endonasal approach (see Section 22) is an expanded-corridor endoscopic technique for skull base pathology. Advantages of endoscopy include illumination, angled views, and minimally invasive access. Limitations: limited instrument maneuverability, difficulty with hemostasis, and a learning curve.
Stereotactic Radiosurgery (SRS)
Single-fraction, high-dose focused radiation delivered with submillimeter accuracy. Platforms: Gamma Knife (201 cobalt-60 sources converging on a focal point; requires frame fixation; most commonly used for intracranial targets), CyberKnife (robotic LINAC; frameless; can treat cranial and spinal targets), LINAC-based SRS (using specialized collimation or multileaf collimators). Indications: brain metastases (1–10 lesions, ≤3 cm), vestibular schwannoma, meningioma (residual/recurrent), AVM (<3 cm nidus), trigeminal neuralgia, and selected pituitary adenomas. For AVMs, the obliteration rate after SRS is ~70–80% at 3 years for small nidi. The latency period (1–3 years for AVM obliteration) is a key limitation — the patient remains at risk of hemorrhage during this interval.
24 Minimally Invasive Spine Surgery
MIS Principles
Minimally invasive spine surgery (MISS) uses tubular retractors, endoscopes, and specialized instruments to achieve the same surgical goals (decompression, fusion, stabilization) through smaller incisions with less muscle disruption. Benefits: reduced blood loss, shorter hospital stay, less postoperative pain, faster recovery. Drawbacks: steep learning curve, limited visualization, increased radiation exposure (fluoroscopy dependence).
Key MIS Procedures
MIS microdiscectomy: tubular retractor placed through a ~2 cm incision; microscope or endoscope used for disc removal. Outcomes equivalent to open microdiscectomy with less tissue damage. MIS TLIF (transforaminal lumbar interbody fusion): performed through a tubular retractor; percutaneous pedicle screws placed under fluoroscopic guidance; interbody cage placed through a unilateral transforaminal approach. Endoscopic spine surgery: full-endoscopic discectomy (transforaminal or interlaminar approach) using a working-channel endoscope; performed under local anesthesia or conscious sedation in some centers. Percutaneous pedicle screw fixation: screws placed through stab incisions under fluoroscopy guidance (K-wire, cannulated screw technique); used for trauma stabilization, tumor stabilization, and as part of MIS fusion constructs. Lateral interbody fusion (XLIF/LLIF): retroperitoneal, transpsoas approach to the lumbar disc space; allows placement of a large interbody cage for indirect decompression and coronal/sagittal correction; risk of lumbar plexus injury (neuromonitoring required); cannot be performed at L5–S1 (iliac crest blocks access).
Robotic-Assisted Spine Surgery
Robotic systems (Mazor X, ExcelsiusGPS, ROSA Spine) assist with pedicle screw placement through preoperative CT-based planning and intraoperative guidance. Studies show improved accuracy of screw placement (>98% accuracy vs ~92% freehand) with potential reduction in revision surgery, though operative time may be increased during the learning curve.
Spinal Fusion — Instrumentation Principles
Pedicle screws are the workhorse of spinal fixation — inserted through the pedicle into the vertebral body, providing three-column fixation. Screw diameter should be ~80% of the pedicle width. Placement techniques: freehand (anatomic landmarks — entry point at the junction of the transverse process and superior articular process for lumbar; use a gearshift to cannulate the pedicle), fluoroscopy-guided, navigation-guided, or robot-assisted. Lateral mass screws are used in the cervical spine (C3–C6); the Magerl technique is most common (entry point at center of lateral mass, aimed 25° lateral and 45° cephalad). C1 lateral mass screws and C2 pedicle or pars screws (Harms technique) are used for atlantoaxial fixation. Interbody cages (PEEK, titanium, or 3D-printed) provide anterior column support, restore disc height, and contain bone graft to promote fusion.
Autograft (iliac crest bone graft — ICBG): gold standard; provides osteogenic, osteoinductive, and osteoconductive properties. Donor site morbidity (pain, hematoma, infection) limits use. Local autograft from laminectomy bone is used when available. Allograft: cadaveric bone — osteoconductive and mildly osteoinductive; no donor site morbidity. Bone morphogenetic protein (BMP-2, Infuse): potent osteoinductive agent; FDA-approved for single-level anterior lumbar interbody fusion (ALIF). Off-label use is common but controversial — associated with ectopic bone formation, radiculitis (in posterior lumbar approaches), and possible cancer risk at high doses. Demineralized bone matrix (DBM) and ceramic bone substitutes (tricalcium phosphate, hydroxyapatite) serve as graft extenders.
25 Neuroimaging — CT, MRI, & Angiography
CT — Key Neurosurgical Applications
CT is the first-line imaging for acute neurosurgical emergencies. Non-contrast CT (NCCT): acute blood appears hyperdense (white) for approximately 1–2 weeks, then becomes isodense, then hypodense (chronic SDH is dark). Air (pneumocephalus) is hyperdense black. Bone is bright white. Key findings to identify: midline shift (measured at the septum pellucidum — >5 mm generally warrants surgical consideration), hydrocephalus (temporal horn dilation is an early sign), effacement of basal cisterns (impending herniation), skull fractures (linear, depressed, basilar). CT angiography (CTA): rapid assessment of cerebral vasculature — aneurysm detection, vessel occlusion (large-vessel stroke), vascular injury in trauma, and AVM characterization.
MRI — Key Sequences
| Sequence | Utility | Key Features |
|---|---|---|
| T1-weighted | Anatomy, fat, subacute hemorrhage | CSF is dark, fat is bright, gray matter is darker than white matter; subacute blood (methemoglobin) is bright |
| T2-weighted | Edema, pathology detection | CSF is bright, edema is bright; most pathology (tumors, infarcts, demyelination) is bright on T2 |
| FLAIR (T2 with CSF suppression) | Periventricular pathology, subtle cortical lesions, SAH | Suppresses CSF signal — bright CSF in FLAIR = abnormal (SAH, meningitis, leptomeningeal disease) |
| T1 post-contrast (gadolinium) | Tumor enhancement, infection, BBB breakdown | Enhancement = BBB disruption; ring-enhancing: GBM, metastasis, abscess, lymphoma; solid enhancement: meningioma, schwannoma |
| DWI / ADC | Acute ischemia, abscess, epidermoid | Acute infarct: bright on DWI, dark on ADC (restricted diffusion); abscess: restricted diffusion centrally (vs tumor necrosis which does not restrict) |
| SWI / GRE | Blood products, calcification, cavernomas | Exquisitely sensitive to hemosiderin (old blood) — "blooming artifact"; identifies microbleeds, cavernomas, DAI |
| MR spectroscopy | Tumor grading, tumor vs radiation necrosis | Elevated choline (cell membrane turnover) and decreased NAA (neuronal marker) suggest tumor; elevated lactate and lipid suggest necrosis or high-grade tumor |
| Perfusion MRI (DSC, ASL) | Tumor grade, tumor vs radiation necrosis, hemodynamics | Elevated rCBV suggests high-grade tumor or recurrence; low rCBV suggests radiation necrosis |
Cerebral Angiography (DSA)
Digital subtraction angiography (DSA) remains the gold standard for cerebral vascular imaging. Indications: definitive characterization of aneurysms (morphology, neck, relationship to branches), AVM evaluation (feeding arteries, nidus, draining veins, flow dynamics), fistula evaluation, vasospasm assessment and treatment, and therapeutic intervention (coiling, embolization, thrombectomy, angioplasty). Complications: stroke (~0.5–1%), groin hematoma, dissection, contrast reactions.
Key Imaging Patterns — Differential Diagnosis
The mnemonic "MAGIC DR" captures the major differential: Metastasis, Abscess, Glioblastoma, Infarct (subacute), Contusion (resolving), Demyelination (tumefactive MS), Radiation necrosis. Key distinguishing features: Abscess — restricted diffusion centrally (bright DWI, dark ADC), smooth thin wall, low rCBV on perfusion. GBM — thick, irregular, incomplete ring; elevated choline on spectroscopy; elevated rCBV. Metastasis — at the gray-white junction; multiple lesions; history of primary cancer; elevated rCBV. Radiation necrosis — occurs within the radiation field, typically 3–12 months post-RT; low rCBV; match on PET (low metabolism).
Brain Abscess — Neurosurgical Considerations
Brain abscesses are focal infections within the brain parenchyma, most commonly from contiguous spread (otitis media, sinusitis, dental infection), hematogenous dissemination (endocarditis, pulmonary AVM, IV drug use), or post-surgical/post-traumatic inoculation. Common organisms: Streptococcus spp. (most common), Staphylococcus aureus (post-surgical/traumatic), anaerobes, and polymicrobial. In immunocompromised: Toxoplasma (HIV, ring-enhancing, basal ganglia), Nocardia, Aspergillus. Treatment: antibiotics (empiric: third-gen cephalosporin + metronidazole ± vancomycin, for 6–8 weeks) + surgical drainage. Stereotactic aspiration is preferred for deep-seated or eloquent-area abscesses; craniotomy with excision for superficial, multiloculated, or fungal abscesses. Abscess ≥2.5 cm generally requires surgical intervention. Serial imaging to confirm response. Identify and treat the source (e.g., sinus, ear, dental).
26 Electrophysiological Monitoring
Intraoperative Neuromonitoring (IONM)
IONM is used during neurosurgical and spine procedures to detect impending neurologic injury in real time, allowing the surgeon to modify the approach before permanent damage occurs.
| Modality | What It Monitors | Key Applications | Alert Criteria |
|---|---|---|---|
| Somatosensory evoked potentials (SSEPs) | Dorsal column pathway (peripheral nerve → dorsal columns → thalamus → sensory cortex) | Spine surgery (posterior cord function), intracranial surgery near sensory pathways, vascular procedures | >50% amplitude decrease or >10% latency increase |
| Motor evoked potentials (MEPs) | Corticospinal tract (motor cortex → anterior horn cells → muscle) | Spine surgery (anterior cord function), intracranial surgery near motor cortex/pathways; more sensitive than SSEPs for motor deficit detection | >50% amplitude decrease (some centers use >80% or all-or-nothing criteria) |
| Electromyography (EMG) — free-running | Individual nerve root / peripheral nerve irritation | Spine surgery (nerve root manipulation), skull base surgery (facial nerve); detects mechanical irritation as spontaneous discharges | Sustained, high-frequency "train" discharges (bursts are typically benign) |
| EMG — triggered (stimulated) | Nerve root identification, pedicle screw accuracy | Pedicle screw placement: stimulating through the screw — low-threshold response (<6–11 mA) suggests medial wall breach and nerve root proximity | Threshold <6 mA (thoracic) or <11 mA (lumbar) — concern for breach |
| Brainstem auditory evoked potentials (BAEPs) | Auditory pathway (cochlea → CN VIII → brainstem) | CPA surgery (vestibular schwannoma), brainstem surgery; hearing preservation | Wave V latency prolongation >1 ms or >50% amplitude decrease |
| Electroencephalography (EEG) | Cortical electrical activity | Epilepsy surgery (electrocorticography — ECoG), carotid endarterectomy (detect ischemia), barbiturate coma monitoring (burst suppression) | Focal slowing, loss of fast activity, burst suppression |
27 Antiepileptics & Osmotic Agents
Antiepileptic Drugs (AEDs) in Neurosurgery
| Drug | Mechanism | Dosing | Key Points |
|---|---|---|---|
| Levetiracetam (Keppra) | SV2A modulation | 500–1500 mg IV/PO BID; load 20–60 mg/kg IV | First-line perioperative AED; no hepatic enzyme induction; no drug level monitoring needed; renal dose adjustment; side effects: irritability, behavioral changes |
| Phenytoin (Dilantin) | Na+ channel blockade | Load 15–20 mg/kg IV (rate ≤50 mg/min); maintenance 100 mg TID | Therapeutic level 10–20 mcg/mL (free level 1–2 mcg/mL); zero-order kinetics; many drug interactions (CYP inducer); IV causes hypotension, arrhythmia, purple glove syndrome; fosphenytoin preferred (faster infusion, less toxicity) |
| Lacosamide (Vimpat) | Na+ channel (slow inactivation) | 50–200 mg IV/PO BID; load 200–400 mg IV | Minimal drug interactions; well tolerated; increasing use as perioperative AED; may cause PR prolongation |
| Valproic acid (Depakote) | GABA enhancement, Na+ channel | 15–60 mg/kg/day divided BID-TID | Broad-spectrum; avoid in liver disease, pregnancy (teratogenic — neural tube defects); inhibits platelet function; may cause thrombocytopenia — problematic perioperatively |
Post-TBI: levetiracetam or phenytoin for 7 days reduces early post-traumatic seizures but does NOT prevent late epilepsy (Temkin 1990, PMID: 2299096). Post-craniotomy for tumor: AAN guidelines recommend against routine prophylaxis in patients without prior seizures; however, many neurosurgeons use perioperative prophylaxis (7–14 days) for supratentorial tumors. SAH: short-term prophylaxis (3–7 days) is reasonable; prolonged use is associated with worse outcomes. Levetiracetam is preferred over phenytoin in SAH (phenytoin associated with worse cognitive outcomes — PMID: 17898332).
Osmotic Agents
| Agent | Mechanism | Dosing | Onset / Duration | Key Points |
|---|---|---|---|---|
| Mannitol (20%) | Osmotic diuresis; reduces brain water; rheologic effect (decreases blood viscosity, improving microcirculation) | 0.25–1 g/kg IV bolus; may repeat q4–6h | Onset 15–30 min; duration 4–6 hr | Keep serum osmolality <320 mOsm/L; causes diuresis → hypotension, hypovolemia, AKI; check electrolytes and osmolality frequently; rebound ICP elevation with repeated dosing |
| Hypertonic saline (23.4%) | Osmotic gradient draws water from brain parenchyma; augments intravascular volume | 23.4%: 30 mL via central line over 10–20 min; 3%: 250 mL bolus or continuous infusion (target Na 145–155) | Onset 15–30 min; duration 4–6 hr | Does not cause diuresis — better for hypotensive patients; central pontine myelinolysis risk if sodium corrected too rapidly in chronic hyponatremia; requires central venous access for 23.4% |
28 Steroids, Anticoagulation Reversal, & Chemotherapy
Dexamethasone
The primary corticosteroid used in neurosurgery. Mechanism: reduces vasogenic edema by stabilizing the blood-brain barrier (decreases capillary permeability). Onset ~4–6 hours for edema reduction (maximal effect 24–72 hours). Dosing: brain tumors with edema — 10 mg IV loading dose, then 4 mg IV/PO q6h; taper as symptoms allow. Spinal cord compression — 10 mg IV bolus, then 4–6 mg q6h. Key side effects: hyperglycemia (tight glucose control recommended — hyperglycemia worsens neurologic outcomes), GI hemorrhage (PPI prophylaxis), immunosuppression, myopathy (proximal weakness with chronic use), insomnia, psychosis, avascular necrosis, adrenal suppression (taper after prolonged use, do not stop abruptly).
The CRASH trial (n = 10,008) demonstrated that high-dose methylprednisolone in TBI significantly increased 14-day mortality (21.1% vs 17.9%). Corticosteroids should NOT be used for the treatment of raised ICP in TBI (CRASH, PMID: 15474134).
Anticoagulation Reversal
Rapid reversal of anticoagulation is critical in patients with intracranial hemorrhage.
| Anticoagulant | Reversal Agent | Dosing | Target |
|---|---|---|---|
| Warfarin | 4-factor PCC (Kcentra) + Vitamin K | PCC: INR-based dosing (25–50 units/kg); Vitamin K: 10 mg IV (slow infusion) | INR ≤1.4 within 30 minutes |
| Unfractionated heparin | Protamine sulfate | 1 mg per 100 units of heparin (max 50 mg); dose reduced based on time since last heparin dose | Normalize aPTT |
| Dabigatran (Pradaxa) | Idarucizumab (Praxbind) | 5 g IV (two 2.5 g vials) | Immediate reversal of dabigatran |
| Rivaroxaban, apixaban, edoxaban (Xa inhibitors) | Andexanet alfa (Andexxa) or 4-factor PCC | Andexanet: high or low dose based on drug/timing; PCC: 50 units/kg if Andexanet unavailable | Normalize anti-Xa levels |
| Antiplatelet agents (aspirin, clopidogrel) | Platelet transfusion (controversial), DDAVP | DDAVP 0.3 mcg/kg IV; platelets if active surgical bleeding | Platelet function (clinical assessment) |
Chemotherapy — Temozolomide (TMZ)
The standard chemotherapy agent for gliomas (GBM and high-grade astrocytomas). Mechanism: alkylating agent that methylates DNA at O6-guanine, leading to mismatch repair-triggered apoptosis. Oral bioavailability ~100%; crosses the BBB effectively. Stupp protocol: concurrent phase — 75 mg/m2/day for 42 days during radiation; adjuvant phase — 150–200 mg/m2/day for 5 days per 28-day cycle, for 6–12 cycles. Key side effects: myelosuppression (lymphopenia — requires PJP prophylaxis with TMP-SMX during concurrent phase), nausea (antiemetic prophylaxis), fatigue, hepatotoxicity (monitor LFTs). MGMT promoter methylation is the strongest predictive biomarker for TMZ responsiveness — methylation silences the MGMT repair enzyme, rendering cells more susceptible to TMZ-induced DNA damage.
Other Agents
Bevacizumab (Avastin): anti-VEGF monoclonal antibody; FDA-approved for recurrent GBM. Reduces edema dramatically (steroid-sparing effect) and improves PFS, but does NOT improve overall survival. Risks: wound healing impairment (hold perioperatively), hemorrhage, thromboembolism, GI perforation. Lomustine (CCNU): nitrosourea alkylating agent used in combination with other agents (PCV regimen: procarbazine, CCNU, vincristine) for oligodendrogliomas and low-grade gliomas. Carmustine (BCNU) wafers (Gliadel): biodegradable polymer wafers impregnated with BCNU, placed in the resection cavity; modest survival benefit; risks include cerebral edema, seizures, and wound healing complications.
Perioperative Antibiotics
Standard prophylactic antibiotics for craniotomy: cefazolin 2 g IV within 60 minutes of incision (re-dose q4h for long cases or estimated blood loss >1500 mL). For patients with MRSA risk or beta-lactam allergy: vancomycin 15 mg/kg IV (begin infusion 120 minutes before incision to account for slow administration). For spinal surgery with instrumentation: cefazolin ± gentamicin powder applied locally (evidence for reduced SSI, though not universally adopted). Antibiotics are typically continued for 24 hours postoperatively (not extended further unless drains or EVD are in place — EVD-related infection prophylaxis protocols vary by institution).
Anticoagulation in Neurosurgical Patients
Preoperative: warfarin held 5 days before surgery (target INR ≤1.4); DOACs held 48–72 hours (longer for renal impairment); aspirin held 7 days (hold vs continue depends on indication — may continue for coronary stents if neurosurgical bleeding risk is acceptable); clopidogrel held 7 days; bridging with LMWH for high-risk patients (mechanical heart valve, recent VTE) — last dose ≥24 hours before surgery.
Postoperative: DVT prophylaxis with SCD/TED stockings from admission. Pharmacologic DVT prophylaxis (LMWH or UFH 5,000 units SQ BID–TID) initiated 24–48 hours after craniotomy if no hemorrhage on postoperative CT. For spine surgery: begin 24 hours postoperatively. Full therapeutic anticoagulation: generally safe to restart 7–14 days after craniotomy if no hemorrhagic complications (earlier if compelling indication such as mechanical heart valve or PE).
29 Classification Systems Master Table
| Classification | Application | Key Grades / Scores |
|---|---|---|
| WHO CNS Tumor Grading (2021) | Brain tumor malignancy | Grade 1 (low proliferative potential, possible cure) → Grade 4 (malignant, rapid growth, necrosis); integrated with molecular markers (IDH, 1p/19q, TERT, EGFR) |
| Hunt-Hess | SAH clinical severity | I (mild headache) → V (coma); correlates with surgical mortality |
| Modified Fisher | SAH CT appearance — vasospasm risk | 0 (no blood) → 4 (thick SAH + IVH); higher grade = higher vasospasm risk |
| WFNS | SAH severity (GCS-based) | I (GCS 15) → V (GCS 3–6); combines GCS with focal deficit |
| Spetzler-Martin | AVM surgical risk | Grade I (small, non-eloquent, superficial drainage) → Grade V (large, eloquent, deep drainage); Grade VI = inoperable |
| Simpson | Meningioma resection extent | I (complete + bone/dura) → V (biopsy only); lower grade = lower recurrence |
| GCS | Level of consciousness | 3 (unresponsive) → 15 (fully alert); E(1–4) + V(1–5) + M(1–6) |
| ASIA Impairment Scale | Spinal cord injury | A (complete) → E (normal); B–D = incomplete injuries of increasing function |
| TLICS | Thoracolumbar fracture management | ≤3 non-operative; 4 equivocal; ≥5 surgical |
| SLICS | Subaxial cervical injury management | ≤3 non-operative; 4 equivocal; ≥5 surgical |
| Anderson-D'Alonzo | Odontoid fracture | Type I (tip), Type II (base — highest nonunion), Type III (body) |
| Meyerding | Spondylolisthesis degree | I (0–25%) → V (spondyloptosis >100%) |
| House-Brackmann | Facial nerve function | I (normal) → VI (total paralysis) |
| Engel | Epilepsy surgery outcome | Class I (seizure-free) → Class IV (no improvement) |
| SINS | Spinal instability — neoplastic | 0–6 stable; 7–12 indeterminate; 13–18 unstable |
| Karnofsky Performance Status (KPS) | Functional status in neuro-oncology | 100 (normal) → 0 (dead); KPS ≥70 generally required for active treatment |
| ETVSS (ETV Success Score) | ETV outcome prediction | 0–90; higher score = higher success probability; based on age, etiology, prior shunt |
30 Complications & Management
Surgical Site Infection
Craniotomy wound infection rate ~1–3%. Superficial wound infection: erythema, swelling, drainage — antibiotics ± wound opening and irrigation. Bone flap infection / osteomyelitis: requires bone flap removal, prolonged IV antibiotics (6–8 weeks), and delayed cranioplasty (typically 3–6 months later). Empyema (subdural or epidural): surgical drainage + IV antibiotics. Most common organisms: S. aureus, coagulase-negative staphylococci, Propionibacterium acnes (indolent — may present months after surgery with bone flap loosening).
CSF Leak
Occurs after skull base surgery (especially transsphenoidal), posterior fossa surgery, and spine surgery (durotomy). Post-craniotomy CSF leak presents as clear drainage from the wound or CSF rhinorrhea/otorrhea. Management: conservative — bed rest, head elevation, lumbar drain (CSF diversion to allow dural healing), acetazolamide (reduce CSF production); surgical repair if conservative measures fail. CSF leak after transsphenoidal surgery (~2–5%) — managed with lumbar drain and possible re-exploration for nasoseptal flap repair. Test for beta-2 transferrin or beta-trace protein to confirm CSF.
Postoperative Hemorrhage
May occur in the epidural, subdural, or intracerebral compartments after any intracranial procedure. Risk factors: coagulopathy, hypertension, incomplete hemostasis, tumor bed bleeding. Presentation: neurologic deterioration, headache, new deficit, or failure to awaken from anesthesia as expected. Management: emergent CT, correct coagulopathy, and re-exploration if hemorrhage causes mass effect or neurologic decline. Intraoperative hemostasis with bipolar cautery, hemostatic agents (Surgicel, Gelfoam, thrombin), and meticulous technique are preventive.
Cerebral Edema
Vasogenic edema: BBB disruption (tumors, infection, radiation necrosis) — responds to dexamethasone. Cytotoxic edema: cellular swelling from ischemia — does NOT respond to steroids (requires osmotic therapy, decompressive surgery). Interstitial (transependymal) edema: periventricular CSF seepage in hydrocephalus — treatment is CSF diversion.
Venous Thromboembolism (DVT/PE)
Neurosurgical patients are at high risk (~20–30% DVT rate without prophylaxis) due to immobility, surgery duration, malignancy, and paresis. Prevention: mechanical (SCDs, graduated compression stockings) starting on admission; pharmacologic (LMWH or UFH) initiated 24–48 hours postoperatively once hemostasis is confirmed. In brain tumor surgery, the risk of VTE (~20–30%) outweighs the risk of postoperative hemorrhage from prophylactic anticoagulation (~1–2%). Treatment of established DVT/PE requires systemic anticoagulation — timing must balance the risk of intracranial hemorrhage (usually safe to start 1–2 weeks after craniotomy if no hemorrhagic complications). IVC filter considered if anticoagulation is contraindicated.
Seizures
Postoperative seizures occur in ~5–20% of supratentorial craniotomies (higher with cortical tumors, intraoperative cortical manipulation, and prior seizure history). New-onset seizures require imaging to rule out hemorrhage, edema, or ischemia. Management: IV levetiracetam or lorazepam for acute seizure; AED adjustment as needed. Status epilepticus protocol: lorazepam → levetiracetam or fosphenytoin → if refractory, intubation and IV anesthetics (propofol, midazolam, or pentobarbital) with continuous EEG monitoring.
Neurological Deficit
May result from direct surgical injury, retraction injury, vascular compromise, or postoperative hemorrhage/edema. SMA syndrome: supplementary motor area retraction/resection causes contralateral akinesia and mutism, which is typically transient (resolves within days to weeks in most cases). Posterior fossa/cerebellar mutism syndrome: occurs in ~25% of pediatric posterior fossa tumor resections, typically 1–2 days postoperatively; characterized by mutism, emotional lability, hypotonia, and ataxia; recovery may take weeks to months and may be incomplete.
Hyponatremia in Neurosurgical Patients
Common and potentially dangerous. Two major mechanisms must be distinguished:
| Feature | SIADH | Cerebral Salt Wasting (CSW) |
|---|---|---|
| Mechanism | Excessive ADH secretion → water retention | Excessive renal sodium loss (natriuretic peptide release) |
| Volume status | Euvolemic to mildly hypervolemic | Hypovolemic (dehydrated) |
| Urine output | Decreased (concentrated urine) | Increased (high urine output) |
| Urine sodium | Elevated (>40 mEq/L) | Elevated (>40 mEq/L) |
| Serum uric acid | Low | Low (but may normalize with volume repletion) |
| Treatment | Fluid restriction (1–1.5 L/day); salt tablets; severe cases: 3% saline or vasopressin receptor antagonists (tolvaptan — use cautiously, overcorrection risk) | Aggressive IV normal saline or 3% saline replacement; fludrocortisone 0.1–0.2 mg BID; do NOT fluid restrict (worsens hypovolemia and may precipitate vasospasm in SAH) |
Diabetes Insipidus (DI) After Pituitary Surgery
Occurs in ~10–20% of patients after transsphenoidal surgery (usually transient). Caused by injury to the posterior pituitary or pituitary stalk, resulting in decreased ADH secretion. Presents with polyuria (>250 mL/hr or >3 L/day of dilute urine), hypernatremia, and intense thirst. Diagnostic criteria: urine specific gravity <1.005, urine osmolality <200 mOsm/kg, serum sodium >145 mEq/L. Treatment: DDAVP (desmopressin) 1–2 mcg IV/SC or 10–20 mcg intranasal; titrate to maintain Na 135–145; monitor closely for overcorrection (SIADH phase may follow). The classic "triphasic response" after stalk injury: DI (days 1–5) → SIADH (days 5–10, release of stored ADH from degenerating posterior pituitary) → permanent DI (after day 10 if stalk injury is severe). Not all patients exhibit all three phases.
Pseudomeningocele
A collection of CSF in the soft tissues outside the dura, usually following spine surgery (incidental durotomy) or posterior fossa surgery. Presents as a fluctuant subcutaneous swelling at the surgical site, headache, and wound drainage. Most small pseudomeningoceles resolve with conservative management (pressure dressing, bed rest, wound care). Large or symptomatic pseudomeningoceles may require lumbar drain placement or surgical repair of the dural defect.
31 Medications Master Table
| Drug | Class | Indication | Dose | Key Monitoring / Notes |
|---|---|---|---|---|
| Levetiracetam | Antiepileptic | Seizure prophylaxis / treatment | 500–1500 mg IV/PO BID | Renal dosing; behavioral side effects; no drug interactions |
| Phenytoin / Fosphenytoin | Antiepileptic | Seizure prophylaxis / status epilepticus | Load 15–20 mg/kg; maint 100 mg TID | Level 10–20 mcg/mL; CYP inducer; hypotension with rapid IV; use fosphenytoin when possible |
| Dexamethasone | Corticosteroid | Vasogenic edema (tumors, radiation) | 10 mg load, 4 mg q6h; taper | Hyperglycemia, GI bleeding, immunosuppression; contraindicated in TBI for ICP |
| Mannitol 20% | Osmotic diuretic | Raised ICP, brain relaxation | 0.25–1 g/kg IV bolus | Serum osm <320; causes hypovolemia, AKI; Foley catheter required |
| Hypertonic saline 23.4% | Osmotic agent | Raised ICP, brain relaxation | 30 mL via central line | No diuresis; augments volume; risk of CPM if rapid correction in hyponatremic patient |
| Nimodipine | Ca2+ channel blocker | Vasospasm prevention (SAH) | 60 mg PO/NG q4h × 21 days | Only calcium channel blocker shown to improve outcomes in SAH; hypotension (hold if SBP <100) |
| Temozolomide | Alkylating agent | GBM, high-grade gliomas | 75 mg/m²/day (concurrent RT); 150–200 mg/m² × 5 d/28 d (adjuvant) | Lymphopenia — PJP prophylaxis; CBC q2 weeks during concurrent phase; MGMT predicts response |
| Bevacizumab | Anti-VEGF mAb | Recurrent GBM | 10 mg/kg IV q2 weeks | Wound healing impairment; hold 4 weeks before/after surgery; hemorrhage, HTN, proteinuria |
| Tranexamic acid (TXA) | Antifibrinolytic | Reduce surgical bleeding | 1 g IV before incision (some use infusion) | Seizure risk at high doses; used in spine surgery and craniotomy to reduce blood loss |
| Aminocaproic acid | Antifibrinolytic | SAH (before aneurysm secured) | 4 g IV load, then 1 g/hr | Short-term use only (increases thromboembolic risk); used if aneurysm securing is delayed |
32 Abbreviations Master List
| Abbreviation | Full Term |
|---|---|
| ACA | Anterior cerebral artery |
| ACDF | Anterior cervical discectomy and fusion |
| AICA | Anterior inferior cerebellar artery |
| ASIA | American Spinal Injury Association |
| AVM | Arteriovenous malformation |
| BBB | Blood-brain barrier |
| BTF | Brain Trauma Foundation |
| CBF | Cerebral blood flow |
| CBV | Cerebral blood volume |
| CPA | Cerebellopontine angle |
| CPP | Cerebral perfusion pressure |
| CSF | Cerebrospinal fluid |
| CTA | CT angiography |
| DAI | Diffuse axonal injury |
| DBS | Deep brain stimulation |
| DCI | Delayed cerebral ischemia |
| DSA | Digital subtraction angiography |
| DWI | Diffusion-weighted imaging |
| EDH | Epidural hematoma |
| EEG | Electroencephalography |
| EMG | Electromyography |
| ETV | Endoscopic third ventriculostomy |
| EVD | External ventricular drain |
| FLAIR | Fluid-attenuated inversion recovery |
| GBM | Glioblastoma |
| GCS | Glasgow Coma Scale |
| GPi | Globus pallidus internus |
| IAC | Internal auditory canal |
| ICA | Internal carotid artery |
| ICH | Intracerebral hemorrhage |
| ICP | Intracranial pressure |
| IDH | Isocitrate dehydrogenase |
| IONM | Intraoperative neuromonitoring |
| IVH | Intraventricular hemorrhage |
| LMN | Lower motor neuron |
| MAP | Mean arterial pressure |
| MCA | Middle cerebral artery |
| MEP | Motor evoked potential |
| MGMT | O6-methylguanine-DNA methyltransferase |
| MIS | Minimally invasive surgery |
| MSCC | Metastatic spinal cord compression |
| MVD | Microvascular decompression |
| NPH | Normal pressure hydrocephalus |
| OPLL | Ossification of the posterior longitudinal ligament |
| PCA | Posterior cerebral artery |
| PComm | Posterior communicating artery |
| PICA | Posterior inferior cerebellar artery |
| PLC | Posterior ligamentous complex |
| SAH | Subarachnoid hemorrhage |
| SCA | Superior cerebellar artery |
| SDH | Subdural hematoma |
| SEEG | Stereoelectroencephalography |
| SINS | Spinal Instability Neoplastic Score |
| SLICS | Subaxial Injury Classification and Severity |
| SMA | Supplementary motor area |
| SRS | Stereotactic radiosurgery |
| SSEP | Somatosensory evoked potential |
| SSS | Superior sagittal sinus |
| STN | Subthalamic nucleus |
| SWI | Susceptibility-weighted imaging |
| TBI | Traumatic brain injury |
| TCD | Transcranial Doppler |
| TLICS | Thoracolumbar Injury Classification and Severity |
| TMZ | Temozolomide |
| UMN | Upper motor neuron |
| VIM | Ventral intermediate nucleus (thalamus) |
| VNS | Vagus nerve stimulation |
| VP shunt | Ventriculoperitoneal shunt |
| WFNS | World Federation of Neurosurgical Societies |
| WHO | World Health Organization |
| XLIF | Extreme lateral interbody fusion |
| ACDF | Anterior cervical discectomy and fusion |
| ACCF | Anterior cervical corpectomy and fusion |
| ALIF | Anterior lumbar interbody fusion |
| PLIF | Posterior lumbar interbody fusion |
| TLIF | Transforaminal lumbar interbody fusion |
| OLIF | Oblique lateral interbody fusion |
| BMP | Bone morphogenetic protein |
| PEEK | Polyether ether ketone |
| PCV | Procarbazine, CCNU (lomustine), vincristine |
| EOR | Extent of resection |
| GTR | Gross total resection |
| STR | Subtotal resection |
| KPS | Karnofsky Performance Status |
| NF1 | Neurofibromatosis type 1 |
| NF2 | Neurofibromatosis type 2 |
| VHL | Von Hippel-Lindau syndrome |
| TIVA | Total intravenous anesthesia |
| SDR | Selective dorsal rhizotomy |
| SCS | Spinal cord stimulation |
| ITB | Intrathecal baclofen |
| DIPG | Diffuse intrinsic pontine glioma |
| PCNSL | Primary CNS lymphoma |
References
Figures
- Figure 1 — Circle of Willis. Wikimedia Commons. Public domain.
- Figure 2 — Ventricular System. Gray's Anatomy (1918). Wikimedia Commons. Public domain.
- Figure 3 — Spinal Cord Cross-Section. Wikimedia Commons. Public domain.
Key Trials & Guidelines
- Stupp R, et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med. 2005;352(10):987-996. PMID: 15758009
- Stummer W, et al. Fluorescence-guided surgery with 5-aminolevulinic acid for resection of malignant glioma. Lancet Oncol. 2006;7(5):392-401. PMID: 16456148
- Buckner JC, et al. Radiation plus procarbazine, CCNU, and vincristine in low-grade glioma (RTOG 9802). N Engl J Med. 2016;374(14):1344-1355. PMID: 27050206
- Molyneux AJ, et al. International Subarachnoid Aneurysm Trial (ISAT). Lancet. 2002;360(9342):1267-1274. PMID: 12414204
- Mohr JP, et al. Medical management with or without interventional therapy for unruptured brain arteriovenous malformations (ARUBA). Lancet. 2014;383(9917):614-621. PMID: 24444842
- Patchell RA, et al. A randomized trial of surgery in the treatment of single metastases to the brain. N Engl J Med. 1990;322(8):494-500. PMID: 2405271
- Brown PD, et al. Hippocampal avoidance during whole-brain radiotherapy plus memantine for patients with brain metastases (NRG CC001). J Clin Oncol. 2020;38(10):1019-1029. PMID: 31454575
- Cooper DJ, et al. Decompressive craniectomy in diffuse traumatic brain injury (DECRA). N Engl J Med. 2011;364(16):1493-1502. PMID: 21434843
- Hutchinson PJ, et al. Trial of decompressive craniectomy for traumatic intracranial hypertension (RESCUEicp). N Engl J Med. 2016;375(12):1119-1130. PMID: 27602507
- CRASH Trial Collaborators. Effect of intravenous corticosteroids on death within 14 days in 10,008 adults with clinically significant head injury. Lancet. 2004;364(9442):1321-1328. PMID: 15474134
- NASCET Collaborators. Beneficial effect of carotid endarterectomy in symptomatic patients with high-grade carotid stenosis. N Engl J Med. 1991;325(7):445-453. PMID: 1852179
- Vahedi K, et al. Early decompressive surgery in malignant infarction of the middle cerebral artery: pooled analysis. Lancet Neurol. 2007;6(3):215-222. PMID: 17290030
- Jüttler E, et al. Hemicraniectomy in older patients with extensive middle-cerebral-artery stroke (DESTINY II). N Engl J Med. 2014;370(12):1091-1100. PMID: 24694544
- Mendelow AD, et al. Early surgery versus initial conservative treatment in patients with spontaneous supratentorial intracerebral haematomas (STICH). Lancet. 2005;365(9457):387-397. PMID: 15639681
- Weinstein JN, et al. Surgical versus nonsurgical therapy for lumbar spinal stenosis (SPORT). N Engl J Med. 2008;358(8):794-810. PMID: 18299459
- Patchell RA, et al. Direct decompressive surgical resection in the treatment of spinal cord compression caused by metastatic cancer. Lancet. 2005;366(9486):643-648. PMID: 15689584
- Fehlings MG, et al. Early versus delayed decompression for traumatic cervical spinal cord injury (STASCIS). PLoS One. 2012;7(2):e32037. PMID: 22290395
- Fisher RS, et al. Electrical stimulation of the anterior nucleus of thalamus for treatment of refractory epilepsy (SANTE). Epilepsia. 2010;51(5):899-908. PMID: 20307616
- Adzick NS, et al. A randomized trial of prenatal versus postnatal repair of myelomeningocele (MOMS). N Engl J Med. 2011;364(11):993-1004. PMID: 21306277
- Temkin NR, et al. A randomized, double-blind study of phenytoin for the prevention of post-traumatic seizures. N Engl J Med. 1990;323(8):497-502. PMID: 2299096
Additional Key Trials
- Wiebe S, et al. A randomized, controlled trial of surgery for temporal-lobe epilepsy (ERSET). N Engl J Med. 2001;345(5):311-318. PMID: 22150006
- Berkhemer OA, et al. A randomized trial of intraarterial treatment for acute ischemic stroke (MR CLEAN). N Engl J Med. 2015;372(1):11-20. PMID: 25517348
- Nogueira RG, et al. Thrombectomy 6 to 24 hours after stroke with a mismatch between deficit and infarct (DAWN). N Engl J Med. 2018;378(1):11-21. PMID: 29129157
- Albers GW, et al. Thrombectomy for stroke at 6 to 16 hours with selection by perfusion imaging (DEFUSE 3). N Engl J Med. 2018;378(8):708-718. PMID: 29364767
- Hanley DF, et al. Efficacy and safety of minimally invasive surgery with thrombolysis in intracerebral haemorrhage evacuation (MISTIE III). Lancet. 2019;393(10175):1021-1032. PMID: 30739747
- Mendelow AD, et al. Early surgery versus initial conservative treatment in patients with spontaneous supratentorial lobar intracerebral haematomas (STICH II). Lancet. 2013;382(9890):397-408. PMID: 23726393
- Fiorella D, et al. Middle meningeal artery embolization for chronic subdural hematoma (EMBOLISE). N Engl J Med. 2024;391:568-578. PMID: 37856610
Textbooks
- Greenberg MS. Handbook of Neurosurgery. 9th ed. Thieme; 2020.
- Winn HR. Youmans and Winn Neurological Surgery. 8th ed. Elsevier; 2022.
- Rhoton AL. Rhoton's Cranial Anatomy and Surgical Approaches. Oxford University Press; 2019.
- Benzel EC. The Cervical Spine. 5th ed. Lippincott Williams & Wilkins; 2012.
- Vaccaro AR, et al. Spine Surgery: Techniques, Complication Avoidance, and Management. 4th ed. Elsevier; 2021.
- Louis DN, et al. WHO Classification of Tumours of the Central Nervous System. 5th ed. IARC; 2021.
- Brain Trauma Foundation. Guidelines for the Management of Severe Traumatic Brain Injury. 4th ed. 2016.