Oral & Maxillofacial Surgery
Every diagnosis, fracture pattern, surgical technique, pathology, implant procedure, classification, complication, medication, and management algorithm across the full scope of oral and maxillofacial surgery in one place.
01 Facial & Oral Anatomy
Facial Skeleton
The facial skeleton (viscerocranium) is composed of 14 bones that form the structural framework for the orbits, nasal cavity, and oral cavity. The key bones in OMS practice include:
Mandible: The only mobile bone of the facial skeleton. A U-shaped bone consisting of the body (bearing the alveolar process with teeth), angle, ramus, coronoid process (attachment of temporalis muscle), and condylar process (articulates with the temporal bone at the TMJ). The mandibular foramen on the medial ramus surface transmits the inferior alveolar nerve and vessels. The mental foramen exits on the buccal cortex, typically inferior to the second premolar apex, transmitting the mental nerve. The lingula is a bony projection medial to the mandibular foramen that serves as a landmark for the inferior alveolar nerve block. Mandibular bone is thicker at the inferior border and thinner at the alveolar crest; the weakest points are the angle (third molar region), condylar neck, and parasymphysis (mental foramen region).
Maxilla: Paired bones that form the central midface. Each maxilla has four processes: frontal (ascending), zygomatic, alveolar (bearing upper teeth), and palatine. The maxillary sinus (antrum of Highmore) is the largest paranasal sinus — its floor is in close proximity to the premolar and molar roots (average distance 1-5 mm). The infraorbital foramen is located approximately 7 mm below the infraorbital rim, transmitting the infraorbital nerve (V2). The paired maxillae are joined at the intermaxillary suture and contribute to the nasal septum, orbital floor, and hard palate.
Zygoma (Zygomatic Bone): Forms the malar prominence ("cheekbone") and contributes to the lateral orbital wall and floor. Articulates with the maxilla (zygomaticomaxillary buttress — a critical vertical buttress), frontal bone (zygomaticofrontal suture), temporal bone (zygomatic arch), and greater wing of sphenoid. The zygomaticomaxillary buttress is the most important vertical buttress of the midface and must be reconstructed in Le Fort fracture repair.
Nasal Bones: Paired rectangular bones forming the bridge of the nose. The most commonly fractured facial bone. Supported inferiorly by the upper lateral cartilages and the cartilaginous septum.
Orbital Bones: The orbit is composed of seven bones: frontal (roof), maxilla (floor medially), zygoma (floor laterally and lateral wall), lacrimal (medial wall), ethmoid/lamina papyracea (medial wall — thinnest orbital wall at 0.2-0.4 mm), sphenoid (greater wing — lateral wall; lesser wing — roof posteriorly), and palatine (small contribution to floor). The orbital floor is the most commonly fractured orbital wall. The infraorbital groove and canal transmit the infraorbital nerve across the floor.
Frontal Bone: Forms the forehead, supraorbital rim, and roof of the orbit. Contains the frontal sinuses — paired pneumatized spaces posterior to the glabella. The supraorbital nerve exits through the supraorbital notch/foramen.
Muscles of Mastication
All four muscles of mastication are innervated by the mandibular division of the trigeminal nerve (V3):
| Muscle | Origin | Insertion | Action | Clinical Note |
|---|---|---|---|---|
| Masseter | Zygomatic arch | Lateral ramus & angle of mandible | Elevation (jaw closing), strongest bite force | Hypertrophy causes facial widening; Botox injection for bruxism |
| Temporalis | Temporal fossa | Coronoid process & anterior ramus | Elevation & retrusion | Attaches to coronoid — coronoid fractures may displace superiorly |
| Medial Pterygoid | Medial surface of lateral pterygoid plate; tuberosity of maxilla | Medial surface of ramus & angle | Elevation, protrusion, lateral excursion | Forms a sling with masseter around the angle — unfavorable angle fractures displace due to this sling |
| Lateral Pterygoid | Superior head: greater wing of sphenoid; Inferior head: lateral pterygoid plate | Superior head: TMJ disc & capsule; Inferior head: condylar neck (pterygoid fovea) | Depression (mouth opening), protrusion, contralateral excursion | Pulls condyle anteromedially in condylar fractures; superior head stabilizes disc during closing |
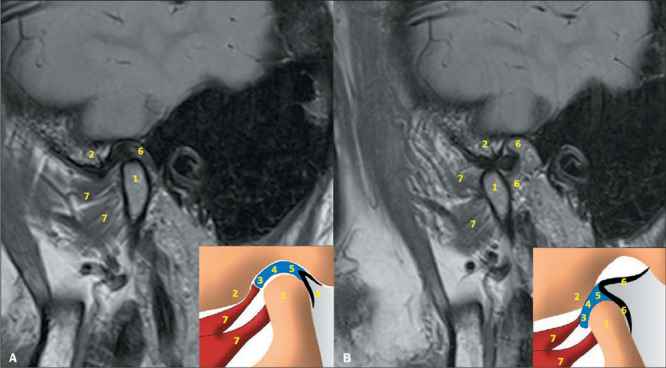
TMJ Anatomy
The temporomandibular joint (TMJ) is a ginglymoarthrodial (combined hinge and sliding) joint between the mandibular condyle and the glenoid fossa (mandibular fossa) of the temporal bone. Key components:
Articular Disc: A biconcave fibrocartilaginous structure dividing the joint into superior and inferior compartments. The disc has three zones: anterior band (thicker), intermediate zone (thinnest — where condyle articulates), and posterior band (thickest). The posterior attachment (bilaminar zone/retrodiscal tissue) is highly vascularized and innervated — a source of pain when the disc displaces anteriorly. The disc is attached laterally and medially to the condylar poles.
Condyle: Ovoid structure approximately 20 mm mediolaterally and 8-10 mm anteroposteriorly. The articular surface is covered by fibrocartilage (not hyaline cartilage — this allows remodeling capacity). The condyle contains active growth centers in children — condylar fractures in children can cause growth disturbance and facial asymmetry.
Articular Eminence: The anterior boundary of the glenoid fossa. The condyle-disc complex must translate past the eminence during full mouth opening. A steep eminence predisposes to disc displacement; a flat eminence predisposes to hypermobility/subluxation.
Dental Anatomy
Universal Numbering System (USA): Permanent teeth numbered 1-32 starting at the upper right third molar (#1), proceeding across to the upper left third molar (#16), then dropping to the lower left third molar (#17), proceeding across to the lower right third molar (#32). Primary teeth are lettered A-T.
FDI (Fédération Dentaire Internationale) System: Two-digit system — first digit indicates quadrant (1 = upper right, 2 = upper left, 3 = lower left, 4 = lower right; primary: 5-8), second digit indicates tooth position from midline (1 = central incisor to 8 = third molar). Example: tooth 36 = lower left first molar.
Tooth Morphology: Each tooth has a crown (covered by enamel — hardest substance in the body, 96% hydroxyapatite) and root (covered by cementum). Dentin forms the bulk of the tooth. The pulp contains neurovascular tissue. The periodontium consists of gingiva, periodontal ligament (PDL — 0.15-0.38 mm wide, provides proprioception and shock absorption), cementum, and alveolar bone. The PDL is essential for tooth reimplantation after avulsion — drying and damage to PDL cells dramatically reduces success.
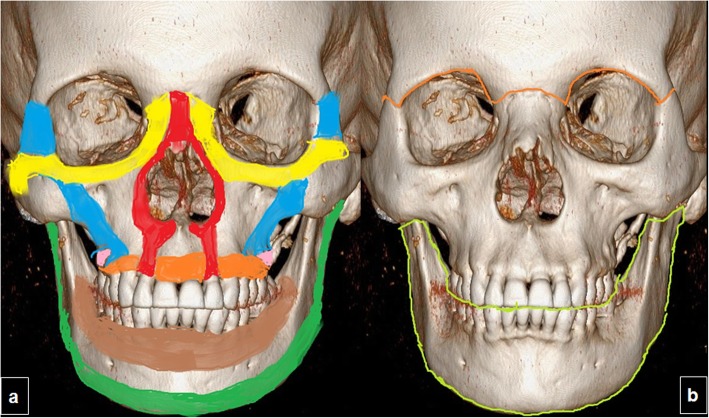
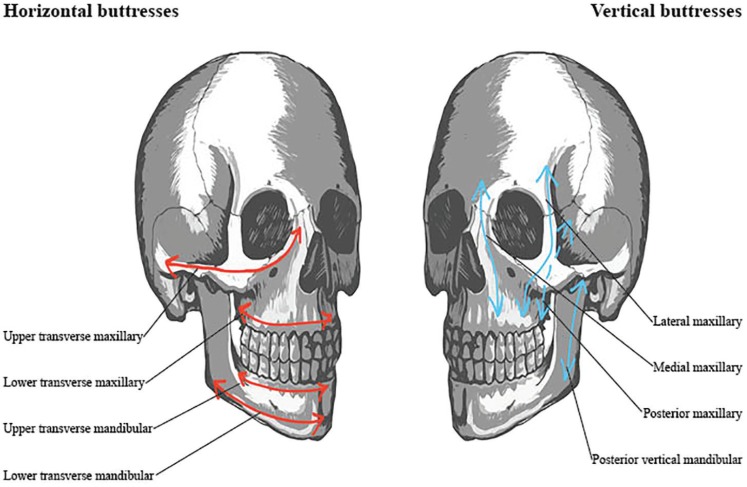
Facial Buttress System
The midface is organized around a system of vertical and horizontal buttresses that transmit masticatory forces to the cranial base. Understanding this buttress anatomy is critical for fracture repair — restoration of buttress continuity is the fundamental principle of midface reconstruction:
Vertical Buttresses (3 paired):
- Nasomaxillary (medial): Maxillary alveolus → piriform aperture → frontal process of maxilla → frontal bone. Transmits forces from the canine/premolar region.
- Zygomaticomaxillary (lateral): Maxillary alveolus → lateral maxillary wall → zygoma → frontal bone. The strongest vertical buttress — transmits forces from the molar region. Must be reconstructed in all Le Fort fractures.
- Pterygomaxillary (posterior): Maxillary tuberosity → pterygoid plates → sphenoid bone. Deepest buttress; fractured in all Le Fort fracture types.
Horizontal Buttresses: Frontal bar (supraorbital rim), infraorbital rim, hard palate, and maxillary alveolus. These resist telescoping (posterior displacement) of the midface. The palate is a critical horizontal buttress — palatal split in Le Fort I fractures requires fixation.
02 Neurovascular Anatomy
Trigeminal Nerve (CN V)
The trigeminal nerve is the dominant sensory nerve of the face and the motor nerve to the muscles of mastication. It has three divisions:
V1 — Ophthalmic Division: Purely sensory. Exits the skull via the superior orbital fissure. Branches: supraorbital nerve (forehead, scalp), supratrochlear nerve (medial forehead, upper eyelid), lacrimal nerve, nasociliary nerve (anterior and posterior ethmoidal nerves, infratrochlear nerve). V1 supplies sensation to the upper eyelid, forehead, scalp to the vertex, dorsum of the nose, and cornea (corneal reflex afferent).
V2 — Maxillary Division: Purely sensory. Exits via the foramen rotundum. Key branches: infraorbital nerve (exits infraorbital foramen — supplies midface, upper lip, lateral nose), posterior superior alveolar nerve (PSA — molar roots), middle superior alveolar nerve (MSA — premolar roots), anterior superior alveolar nerve (ASA — incisor and canine roots), zygomatic nerve, greater and lesser palatine nerves (hard and soft palate), nasopalatine nerve (anterior hard palate through incisive foramen).
V3 — Mandibular Division: Mixed (sensory + motor). Exits via the foramen ovale. Motor branches to all four muscles of mastication plus mylohyoid, anterior belly of digastric, tensor veli palatini, and tensor tympani. Key sensory branches:
- Inferior alveolar nerve (IAN): Enters the mandibular foramen, courses through the mandibular canal, supplies the mandibular teeth and lower lip/chin (as the mental nerve exiting the mental foramen). The IAN runs approximately 4.2 mm (average) below the apices of the lower third molar — this distance is critical for extraction risk assessment. The incisive branch continues anteriorly from the mental foramen to supply the premolars, canines, and incisors.
- Lingual nerve: Courses along the medial surface of the mandible, intimately related to the third molar region — lies an average of 2.06 mm inferior and 0.58 mm medial to the alveolar crest at the third molar. At highest risk during third molar surgery and submandibular gland excision. Carries taste (via chorda tympani from CN VII) and general sensation to the anterior two-thirds of the tongue.
- Buccal nerve (long buccal): Crosses the anterior border of the ramus, supplies the buccal mucosa and gingiva of the posterior mandible. Anesthetized separately from the IAN block.
- Auriculotemporal nerve: Passes posterior to the condylar neck, supplies sensation to the TMJ, external ear, and temporal region. Can be injured in condylar surgery — results in Frey syndrome (gustatory sweating).
Facial Nerve (CN VII)
The facial nerve exits the skull through the stylomastoid foramen, enters the parotid gland, and divides into five terminal branches (mnemonic: "Ten Zebras Bit My Cat"):
| Branch | Motor Supply | Clinical Significance |
|---|---|---|
| Temporal | Frontalis, orbicularis oculi (upper), corrugator supercilii | Crosses the zygomatic arch within the superficial temporal fascia (Pitanguy line: 0.5 cm below tragus to 1.5 cm above lateral brow). At risk in coronal approaches and zygomatic arch surgery. Injury causes inability to raise eyebrow. |
| Zygomatic | Orbicularis oculi (lower) | Cross-innervation with temporal branch often present. At risk in midface approaches. |
| Buccal | Buccinator, orbicularis oris (upper), zygomaticus major/minor, levator labii superioris | Multiple anastomoses — injury usually recovers spontaneously. Crosses over the buccal fat pad. |
| Marginal mandibular | Depressors of the lower lip (depressor anguli oris, depressor labii inferioris, mentalis) | Courses 1-2 cm below the mandibular border in 20% of patients (up to 3 cm below in some studies). At risk in submandibular approaches and mandibular angle surgery. Injury causes asymmetric smile and drooling. |
| Cervical | Platysma | Least clinically significant branch. Runs deep to the platysma in the neck. |
Blood Supply
The face has a rich arterial supply derived primarily from the external carotid artery:
Maxillary artery (internal maxillary artery — IMA): The terminal branch of the external carotid artery. It passes deep to the condylar neck (first part), courses through the infratemporal fossa (second part), and enters the pterygopalatine fossa (third part). Key branches: middle meningeal artery (enters foramen spinosum — epidural hematoma when torn), inferior alveolar artery (enters mandibular foramen with IAN), posterior superior alveolar artery, infraorbital artery, sphenopalatine artery (major source of posterior epistaxis), greater and lesser palatine arteries, buccal artery.
Facial artery: Crosses the mandibular body at the anterior border of the masseter (palpable pulse point). Ascends toward the medial canthus as the angular artery. Branches: submental artery (supplies the chin and floor of mouth), inferior and superior labial arteries.
Superficial temporal artery (STA): The other terminal branch of the external carotid. Courses anterior to the tragus over the zygomatic arch. Bifurcates into frontal and parietal branches. Used as a recipient vessel in microvascular reconstruction.
03 The OMS Examination
Cranial Nerve Examination
A focused cranial nerve exam is essential in facial trauma and pathology. Key elements: CN V — test light touch and pinprick in V1 (forehead), V2 (cheek), and V3 (chin/lower lip) distributions bilaterally; CN VII — assess frontalis (eyebrow raise), orbicularis oculi (forced eye closure), buccinator (puff cheeks), orbicularis oris (pucker lips), and depressor labii (show lower teeth); CN III/IV/VI — extraocular movements and pupillary response (critical in orbital fractures); CN XII — tongue protrusion (lingual nerve is CN V3, not XII — a common examination error).
Occlusion Assessment — Angle Classification
Occlusion is the single most important parameter in assessing and treating facial fractures and orthognathic surgery. The Angle Classification describes the mesiodistal (anteroposterior) relationship of the first molars:
| Class | Molar Relationship | Skeletal Pattern | Clinical Description |
|---|---|---|---|
| Class I (Neutrocclusion) | Mesiobuccal cusp of upper first molar aligns with buccal groove of lower first molar | Normal skeletal relationship | Normal AP relationship; may still have crowding, spacing, or rotations |
| Class II (Distocclusion) | Lower molar is positioned distally (posteriorly) relative to Class I | Retrognathic mandible and/or prognathic maxilla | Division 1: proclined upper incisors (increased overjet). Division 2: retroclined upper incisors (deep bite) |
| Class III (Mesiocclusion) | Lower molar is positioned mesially (anteriorly) relative to Class I | Prognathic mandible and/or retrognathic maxilla | Underbite; anterior crossbite; concave facial profile |
Dental Examination
Systematic dental assessment includes: tooth count (missing, mobile, or fractured teeth), caries assessment, periodontal probing (pocket depths > 4 mm indicate periodontal disease), tooth mobility grading (Grade I: < 1 mm horizontal; Grade II: > 1 mm horizontal; Grade III: vertical mobility), pulp vitality testing (electric pulp test, cold test with ethyl chloride or CO&sub2; snow), and assessment of existing restorations. In trauma patients, all teeth in the line of fracture must be evaluated for viability — a tooth in the fracture line is generally retained if it is not grossly mobile, does not prevent fracture reduction, and is not the source of infection (periapical pathology).
TMJ Examination
Range of motion: Maximum interincisal opening (MIO) — normal 40-55 mm; lateral excursion — normal 8-12 mm; protrusion — normal 8-10 mm. Measure with a ruler or Boley gauge. Auscultation/palpation: Clicking (early/intermediate/late — early click often indicates anterior disc displacement with reduction), popping (louder, suggests intermittent locking), crepitus (suggests osteoarthritis/degenerative joint disease). Deviation: Deviation toward the affected side with opening (suggests ipsilateral intracapsular restriction, e.g., disc displacement without reduction or ankylosis); deflection away from affected side (less common). Palpation: Lateral pole of the condyle (preauricular), posterior capsule (via external auditory canal), muscles of mastication (masseter, temporalis, lateral and medial pterygoid).
Facial Symmetry Assessment
Evaluate malar projection (flattening suggests ZMC fracture), nasal deviation, mandibular deviation (chin point), orbital height asymmetry (suggests orbital floor fracture with globe descent), step deformities along the orbital rim or zygomatic arch. Always compare to the contralateral side. Submental vertex view is valuable for assessing zygomatic arch flattening and midface width.
Eye Examination in Facial Trauma
Every patient with orbital or midface fractures must have a focused eye examination documented:
- Visual acuity: Snellen chart or finger counting. Decreased acuity may indicate globe injury, retrobulbar hematoma, or optic nerve injury. Document before any surgical intervention.
- Pupil examination: Direct and consensual light reflexes. A relative afferent pupillary defect (RAPD/Marcus Gunn pupil) indicates optic nerve or extensive retinal injury — urgent ophthalmology consultation required.
- Extraocular movements: Restriction in upgaze (orbital floor entrapment), medial gaze (medial wall entrapment). Pain with eye movement is significant even without frank restriction.
- Globe position: Enophthalmos (globe recessed — measured with Hertel exophthalmometer; > 2 mm difference is significant), hypoglobus (vertical descent), proptosis (may indicate retrobulbar hematoma).
- Infraorbital sensation: V2 hypoesthesia is present in 50-90% of orbital floor fractures.
Emergency Retrobulbar hematoma: Bleeding into the orbital space causes rapidly increasing intraorbital pressure, compressing the optic nerve and central retinal artery. Signs: proptosis, tense globe, decreased visual acuity, afferent pupillary defect, elevated IOP. This is an ophthalmologic emergency — permanent vision loss occurs within 90-120 minutes if untreated. Immediate treatment: lateral canthotomy and cantholysis (cut the lateral canthal tendon and release the inferior crus of the lateral canthal tendon to decompress the orbit). This bedside procedure can be performed by any OMS surgeon or emergency physician and should not wait for ophthalmology consultation. IV mannitol (1-2 g/kg) and acetazolamide (500 mg IV) to reduce IOP are adjunctive measures.
04 Anesthesia & Sedation in OMS
Local Anesthetic Techniques
OMS practitioners use a wide range of local anesthetic injections. The most common agents are 2% lidocaine with 1:100,000 epinephrine (maximum dose 7 mg/kg with epi, 4.4 mg/kg without) and 4% articaine with 1:100,000 epinephrine (maximum dose 7 mg/kg — superior bone penetration due to thiophene ring). 0.5% bupivacaine with 1:200,000 epinephrine is used for prolonged postoperative analgesia (duration 4-9 hours; maximum dose 1.3 mg/kg with epi).
| Technique | Target Nerve | Landmark/Approach | Area Anesthetized |
|---|---|---|---|
| IAN Block | Inferior alveolar nerve | Palpate coronoid notch, inject medial to ramus at the level of the lingula (approximately 1 cm above the occlusal plane of mandibular molars); needle contacts bone at pterygomandibular raphe | Ipsilateral mandibular teeth, lower lip, chin; lingual nerve usually blocked simultaneously (anterior 2/3 tongue, lingual gingiva) |
| PSA Block | Posterior superior alveolar nerve | Insert at the mucobuccal fold above the maxillary second molar, advance posteriorly, superiorly, and medially along the posterior maxilla (45° angle) | Maxillary molars (except mesiobuccal root of first molar in 28% — MSA territory), adjacent buccal gingiva, maxillary sinus |
| MSA Block | Middle superior alveolar nerve | Inject at the mucobuccal fold above the maxillary second premolar | Premolars, mesiobuccal root of first molar, adjacent buccal gingiva |
| ASA Block | Anterior superior alveolar nerve | Inject at the mucobuccal fold above the maxillary canine (infraorbital injection is an alternative) | Maxillary incisors and canines, anterior gingiva, upper lip, lateral nose |
| Mental/Incisive Block | Mental nerve (and incisive branch) | Inject at the mucobuccal fold at or just anterior to the mental foramen (below second premolar); apply pressure to direct solution into the foramen for incisive block | Mental: lower lip, chin, labial gingiva of premolars. Incisive: adds premolars, canine, incisors |
| Infiltration | Terminal nerve endings | Inject directly at the mucobuccal fold adjacent to the target tooth | Highly effective in maxilla (thin cortex); in mandible, 4% articaine has improved buccal infiltration success for posterior teeth |
IV Sedation & Office-Based General Anesthesia
OMS is one of very few specialties licensed for in-office deep sedation and general anesthesia. The OMS anesthesia team model requires the surgeon to serve as both operator and anesthetist. Standard agents include:
- Midazolam (0.05-0.1 mg/kg IV) — anxiolysis, amnesia, anticonvulsant
- Propofol (25-75 mcg/kg/min infusion) — rapid onset/offset, antiemetic properties
- Fentanyl (0.5-1 mcg/kg IV) — analgesia
- Ketamine (0.5-1 mg/kg IV) — dissociative anesthesia, maintains airway reflexes and respiratory drive; useful in pediatric patients
- Dexmedetomidine (0.5-1 mcg/kg loading, 0.2-0.7 mcg/kg/hr) — sedation without respiratory depression
Airway management: OMS practitioners must be proficient in nasal intubation (preferred for operations requiring assessment of occlusion), oral intubation, submental intubation (tube passed through a submental incision to avoid nasal route in midface/skull base fractures), and tracheostomy. Fiberoptic nasal intubation is the standard for predicted difficult airways and patients with limited mouth opening.
Local Anesthetic Toxicity
Signs of systemic toxicity (dose-dependent progression): perioral numbness, metallic taste, tinnitus, visual disturbances → muscle twitching, tremors → tonic-clonic seizures → respiratory depression → cardiovascular collapse (bradycardia, hypotension, asystole). Bupivacaine is the most cardiotoxic local anesthetic — cardiac arrest from bupivacaine toxicity is resistant to standard resuscitation. Treatment: Stop injection, airway management, benzodiazepine for seizures, 20% lipid emulsion (Intralipid) — 1.5 mL/kg IV bolus, then 0.25 mL/kg/min infusion. Lipid emulsion is the specific antidote and should be immediately available in every OMS office. Epinephrine dose should be limited (≤ 1 mcg/kg) as high-dose epinephrine may worsen bupivacaine cardiotoxicity.
Vasoconstrictor Considerations
Epinephrine in local anesthetics provides hemostasis, prolongs duration, slows systemic absorption, and reduces toxicity. Maximum recommended dose: 0.2 mg (11 cartridges of 1:100,000 concentration or 5.5 cartridges of 1:50,000). In patients with cardiovascular disease, limit to 0.04 mg (2 cartridges of 1:100,000). Absolute contraindications to epinephrine: pheochromocytoma, uncontrolled hyperthyroidism, severe uncontrolled hypertension, use of non-selective beta-blockers (propranolol — risk of hypertensive crisis). Epinephrine is NOT contraindicated in patients taking tricyclic antidepressants at normal local anesthetic doses, despite historical teaching.
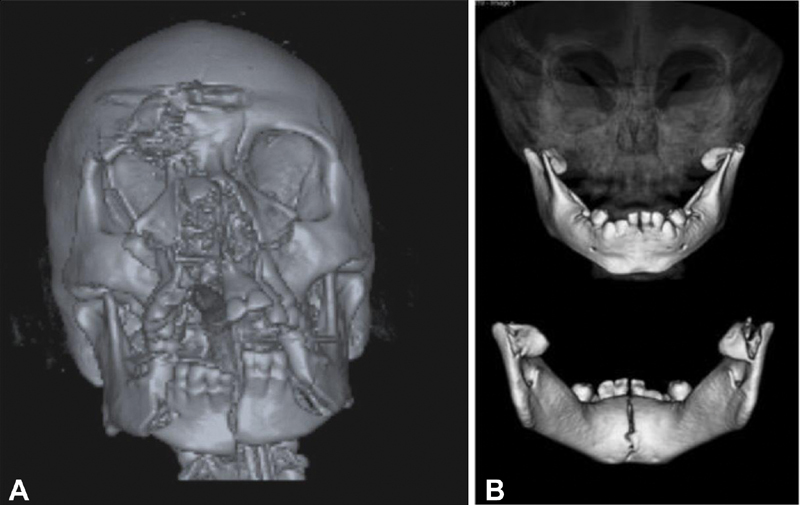
05 Mandibular Fractures
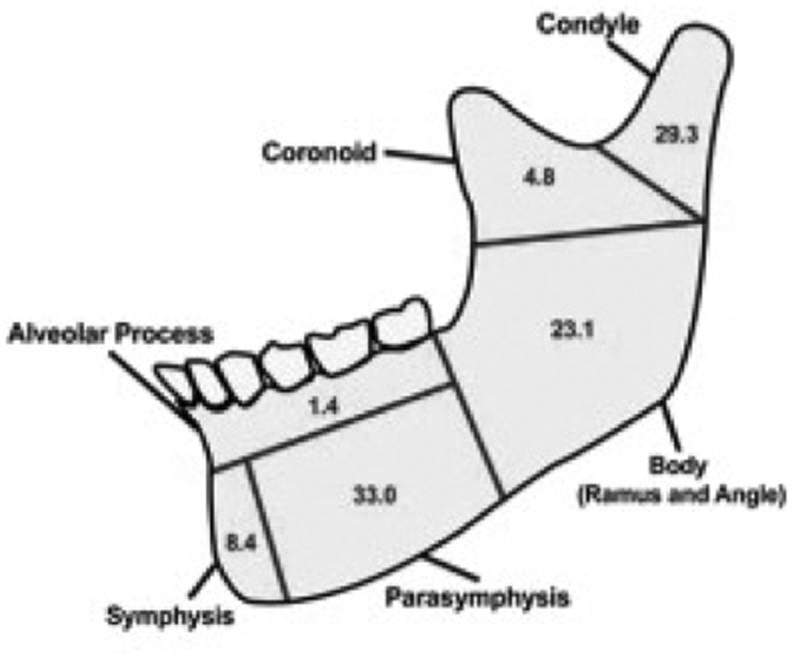
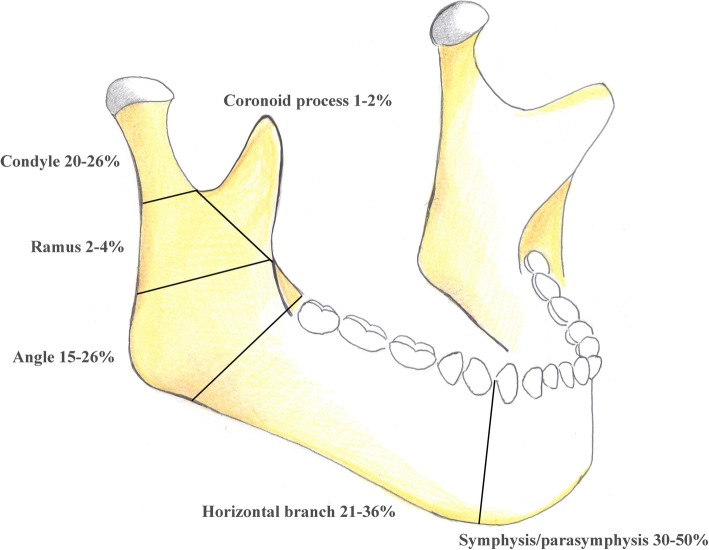
Classification by Location
Mandibular fractures are classified by anatomic site. The mandible acts as a ring structure — fractures commonly occur at two sites simultaneously (similar to a pretzel):
| Site | Frequency | Key Features |
|---|---|---|
| Condyle/Subcondylar | 25-35% | Most common site overall; classified as intracapsular (condylar head), subcondylar (neck), or low subcondylar (base); lateral pterygoid pulls condylar fragment anteromedially |
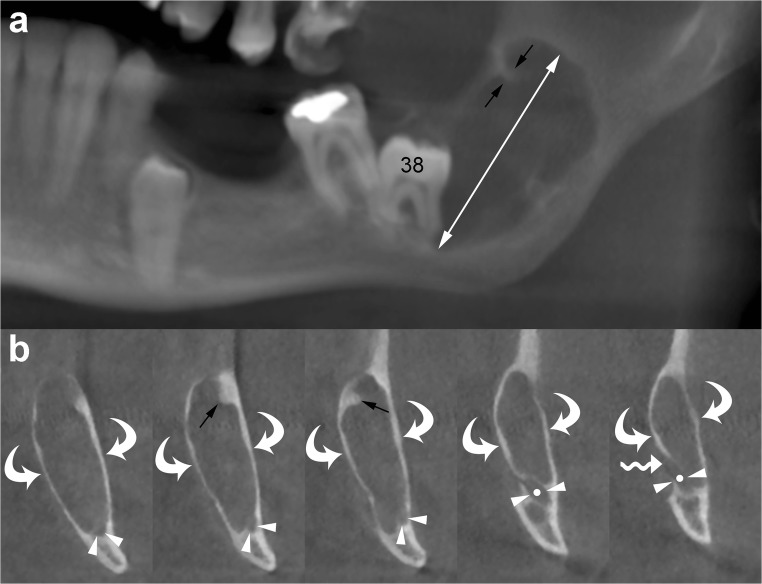
| Angle | 20-25% | Third molar region; influenced by masseter/pterygoid sling; impacted third molars increase angle fracture risk 3-fold |
| Body | 15-25% | Canine to angle; mental foramen at risk; IAN injury possible |
| Parasymphysis | 15-20% | Canine to canine region; mental nerve at risk; commonly paired with contralateral condyle fracture |
| Symphysis | 5-10% | Midline; often from direct anterior blow; bilateral condylar fractures may accompany |
| Ramus | 3-5% | Between angle and condylar neck; masseter splinting provides natural stability |
| Coronoid | 1-2% | Rare; temporalis pulls fragment superiorly; often managed conservatively |
| Alveolar | Variable | Isolated tooth-bearing segment; often accompanies dentoalveolar trauma |
Favorable vs. Unfavorable Fracture Lines
A favorable fracture is one where the muscle pull resists displacement of the fragments (muscles act to compress the fracture). An unfavorable fracture is one where the muscle pull displaces the fragments. This concept is assessed in two planes:
- Vertical (horizontal favorability): At the angle — a fracture angled from posterosuperior to anteroinferior is favorable (masseter/pterygoid sling compresses fragments); the reverse is unfavorable.
- Horizontal (vertical favorability): A fracture angled medially from the lateral cortex (buccal) to the medial cortex (lingual) is favorable — the medial pterygoid pulls the fragment medially into the other fragment. The reverse is unfavorable.
Treatment
Closed Reduction with MMF (Maxillomandibular Fixation): Appropriate for nondisplaced/minimally displaced fractures, favorable fracture patterns, condylar fractures in many cases, and patients who cannot undergo general anesthesia. Methods: arch bars (Erich arch bars — gold standard, 24-26 gauge wire to teeth), IMF screws (faster application, less soft tissue injury), and Ivy loops. Duration: typically 2-4 weeks for adults, 10-14 days for children.
ORIF — Champy Ideal Lines of Osteosynthesis: Michel Champy demonstrated that monocortical miniplates placed along the zones of tension provide adequate fixation for mandibular fractures while the inferior border acts as the zone of compression:
- Symphysis/parasymphysis: Two plates — one at the inferior border (2.0 mm locking plate) and one above (1.5-2.0 mm plate near the alveolar crest) to resist torsional forces
- Body: One plate along the tension band (external oblique ridge) or two plates for additional stability
- Angle: One plate along the external oblique ridge (Champy technique) — the single most validated plating location in the mandible; 2.0 mm monocortical miniplate with 6-8 mm screws; alternatively, one larger plate at the inferior border
Load-Bearing vs. Load-Sharing Fixation: Load-sharing plates (miniplates, 2.0 mm) share the functional load with the bone — require intact bone on both sides of the fracture for the bone to bear some load. Load-bearing plates (2.4-2.7 mm reconstruction plates, locked with bicortical screws) bear the entire functional load — used when bone is comminuted, atrophic, or there is a defect (bone loss). Reconstruction plates require at least three bicortical screws (6-8 mm shaft diameter) on each side of the fracture.
Condylar Fractures — Open vs. Closed Debate
Condylar fractures remain the most controversial area of mandible fracture management. Absolute indications for open treatment (ORIF): (1) displacement of condyle into the middle cranial fossa, (2) inability to obtain adequate occlusion with closed treatment, (3) lateral extracapsular displacement, (4) foreign body (e.g., gunshot). Relative indications: bilateral condylar fractures with comminuted midface, bilateral condylar fractures in edentulous patients. Approaches for open treatment: retromandibular (Risdon), preauricular, endoscopic transoral with trocar-assisted plating. Most intracapsular (condylar head) fractures are treated closed; subcondylar fractures increasingly treated with ORIF via endoscopic approaches. PMID: 23981784
Complications of Mandibular Fracture Treatment
Infection/hardware failure: Most common complication (5-15%). Risk factors: teeth in the fracture line with periapical pathology, substance abuse, noncompliance, delayed treatment > 72 hours, comminution. Malunion/malocclusion: Inadequate reduction or unstable fixation; may require osteotomy and re-plating if occlusion cannot be corrected orthodontically. Nonunion: Failure of bony healing after 6-8 weeks; risk factors: infection, inadequate fixation, poor blood supply (atrophic mandible), smoking, interposition of soft tissue. Treatment: debridement of fibrous tissue, revision fixation with load-bearing plate, consider bone graft. IAN injury: Paresthesia/hypoesthesia of the lower lip in 5-30% of mandibular fractures (particularly body and parasymphysis); most recover spontaneously within 6-12 months. TMJ ankylosis: Most feared long-term complication of condylar fractures (especially intracapsular fractures in children); results in progressive mouth opening limitation and facial growth restriction. Presents years after injury.
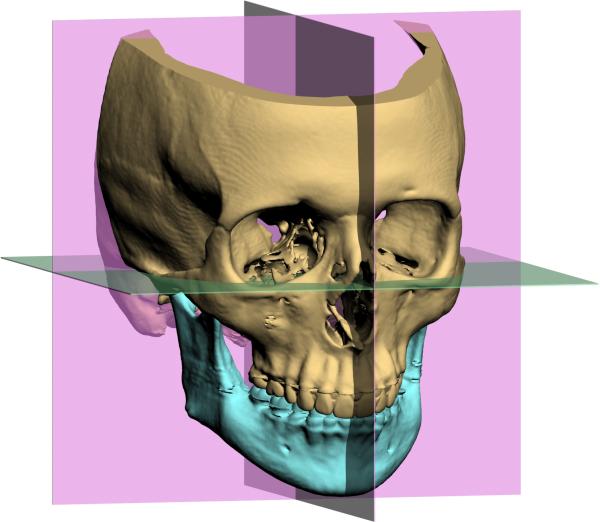
06 Midface Fractures
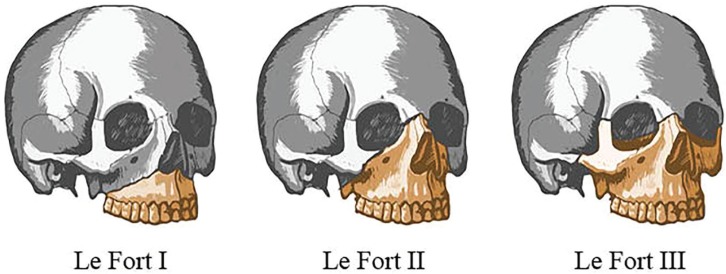
Le Fort Classification
René Le Fort (1901) described three patterns of midface fracture based on cadaver studies. Each Le Fort level separates a mobile maxillary segment from the stable cranial base:
| Type | Fracture Line | Key Features | Clinical Signs |
|---|---|---|---|
| Le Fort I (Guerin) | Horizontal fracture through the maxilla above the tooth apices: lateral nasal wall → lateral maxillary sinus wall → pterygoid plates (low) | Separates the palate and alveolar process from the upper midface; "floating palate" | Maxillary mobility at the palatal level; anterior open bite; malocclusion; perioral ecchymosis |
| Le Fort II (Pyramidal) | Pyramidal: nasion → medial orbit (through lacrimal bone) → infraorbital rim → lateral maxillary wall → pterygoid plates (mid) | Separates the central midface (maxilla + nose) from the zygoma and cranium; "floating maxilla" | Mobility of the nose and maxilla together; CSF rhinorrhea (cribriform plate involvement); periorbital ecchymosis (bilateral raccoon eyes); dish-face deformity; step deformity at infraorbital rim |
| Le Fort III (Craniofacial dysjunction) | Nasofrontal suture → medial and lateral orbital walls → zygomatic arch → pterygoid plates (high) | Complete separation of the facial skeleton from the cranium; "floating face" | Entire midface is mobile; massive facial edema; lengthening of face; CSF leak common; bilateral periorbital ecchymosis; dish-face deformity; often associated with skull base fractures |
Zygomatic Complex (ZMC/Tripod) Fractures
The zygoma articulates at four sutures: zygomaticofrontal (ZF), zygomaticomaxillary (ZM), zygomaticotemporal (ZT/zygomatic arch), and zygomaticosphenoid (within the orbit). A ZMC fracture involves disruption at all four articulations — hence the term "quadripod" is more accurate than "tripod."
Zingg Classification:
| Type | Description | Treatment |
|---|---|---|
| Type A | Incomplete: isolated zygomatic arch (A1), lateral orbital wall (A2), or infraorbital rim (A3) | A1: Gillies approach (temporal) or Keen approach (intraoral) for arch reduction; A2/A3: ORIF if displaced |
| Type B | Complete monofragment: intact tetrapod fracture with one piece (classic ZMC) | ORIF with 2-3 point fixation — key points: ZF suture (most accurate for reduction), infraorbital rim, zygomaticomaxillary buttress |
| Type C | Comminuted multifragment ZMC fracture | ORIF with wide exposure, multiple plates, and possible orbital floor reconstruction |
Naso-Orbito-Ethmoid (NOE) Fractures
NOE fractures involve the nasal bones, ethmoid labyrinth, lacrimal bones, and frontal process of the maxilla. The critical structure is the medial canthal tendon (MCT) — its integrity determines the classification and treatment.
Markowitz Classification:
| Type | Description | Treatment |
|---|---|---|
| Type I | Single large central fragment with MCT attached | Reduce and fixate the fragment — this repositions the MCT |
| Type II | Comminuted central fragment with MCT attached to a bone fragment | Identify the MCT-bearing fragment, reduce and plate it; reconstruct remaining fragments |
| Type III | Comminuted with MCT avulsion from bone | Transnasal canthopexy with wire/suture — pass the wire through the MCT, through the nasal bones, and anchor to the contralateral side. Most difficult repair; high incidence of telecanthus |
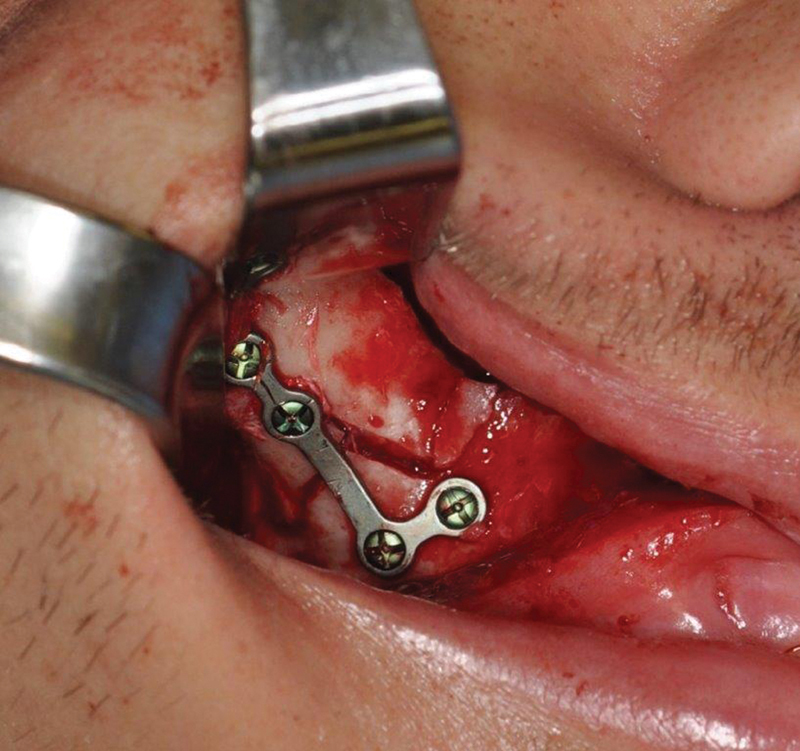
Midface Treatment Principles
The midface is organized around vertical and horizontal buttresses. The three paired vertical buttresses (nasomaxillary, zygomaticomaxillary, pterygomaxillary) transmit occlusal forces to the cranial base. Fixation strategy: restore vertical buttress height and alignment first (bottom-up approach — start with occlusion via MMF, then reconstruct buttresses from inferior to superior). Plate sizes: 1.5-2.0 mm miniplates for the midface; 1.3 mm microplates for orbital rim. Use at least two-point fixation per buttress.
07 Orbital Fractures
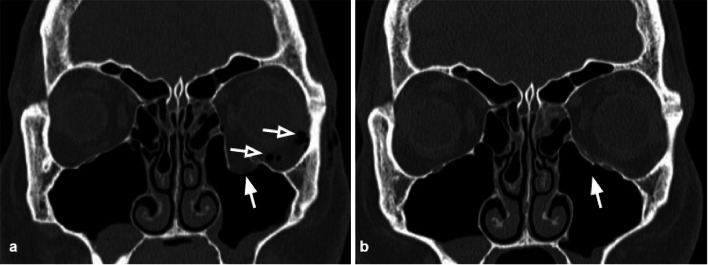
Types & Mechanisms
Orbital floor blowout: The most common isolated orbital fracture. Two mechanisms: (1) Buckling theory — force applied to the inferior orbital rim transmits through the orbital floor bone, causing it to buckle and fracture (most accepted for floor fractures); (2) Hydraulic theory — force applied directly to the globe increases intraorbital pressure, causing the thinnest wall (floor or medial wall) to fracture as a pressure relief valve.
Medial wall (lamina papyracea): The thinnest orbital wall (0.2-0.4 mm); commonly fractured along with the floor. Surgical access via transcaruncular or Lynch (frontoethmoidal) incision. Reconstruction with titanium mesh, porous polyethylene (Medpor), or resorbable plates.
Orbital roof: Uncommon; associated with frontal bone and skull base fractures; more common in children (due to undeveloped frontal sinus). May cause CSF leak, dural tear, or brain herniation into the orbit (pulsating exophthalmos). Neurosurgical consultation required.
Indications for Surgical Repair
- Persistent diplopia in primary or downgaze with positive forced duction test (indicating true entrapment vs. edema)
- Enophthalmos > 2 mm (or significant hypoglobus) — indicates large volume expansion of the orbit
- Large defect > 50% of the orbital floor (or > 2 cm²) — even without current enophthalmos, delayed enophthalmos will likely develop
- Entrapment with muscle ischemia — especially the trapdoor fracture / white-eyed blowout in children
Emergency White-eyed blowout fracture: Occurs predominantly in children and young adults. The orbital floor fractures in a greenstick/trapdoor pattern, herniating the inferior rectus muscle or periorbital fat into the maxillary sinus, then snapping back to trap the tissue. Signs: restricted upgaze with pain, bradycardia and nausea (oculocardiac reflex from muscle entrapment), minimal periorbital edema or ecchymosis (hence "white-eyed"). CT shows minimal floor displacement but soft tissue herniation. This is a surgical emergency — release must be performed within 24-48 hours to prevent permanent ischemic injury to the inferior rectus muscle and irreversible diplopia. PMID: 12140836
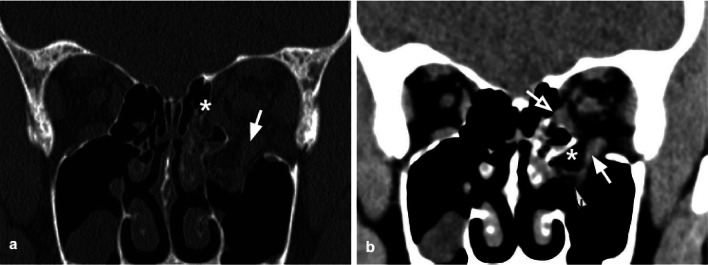
Implant Materials for Orbital Reconstruction
| Material | Type | Advantages | Disadvantages |
|---|---|---|---|
| Titanium mesh | Alloplastic, permanent | Strong, malleable, precontoured options, radiopaque (easy to assess on CT), low infection rate | Permanent implant, potential for globe adherence, may need removal in rare cases |
| Porous polyethylene (Medpor) | Alloplastic, permanent | Allows tissue ingrowth (200-μm pore size), stable fixation, easily trimmed, integrated well | Difficult removal once integrated, higher extrusion rate than titanium |
| Resorbable plates (PLLA, PGA, PDLLA) | Alloplastic, resorbable | No permanent implant, good for small-medium defects, preferred in pediatric patients | May not support large defects, foreign body reaction in 1-5% |
| Autogenous bone (calvarial, iliac crest) | Autogenous | No foreign body, biological integration | Donor site morbidity, unpredictable resorption, additional surgical site |
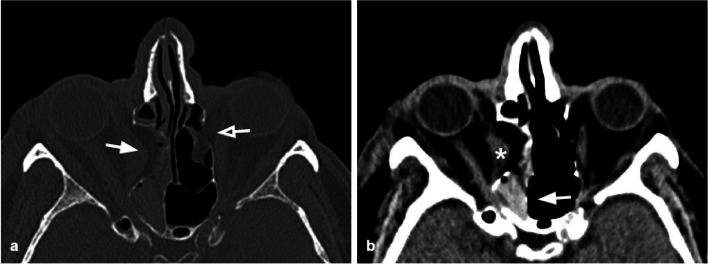
Surgical Approaches for Orbital Repair
Transconjunctival (subtarsal variant): The most commonly used approach for the orbital floor. Incision through the conjunctiva and lower eyelid retractors, dissecting in the preseptal plane to reach the orbital rim. Advantages: no visible scar, lower ectropion rate than subciliary. Can be combined with a lateral canthotomy (swinging eyelid approach) for wider exposure. Subciliary: Skin incision 2-3 mm below the lash line; dissected in skin-muscle flap or stepped fashion to the orbital rim. Higher risk of ectropion (5-15%), scleral show, and lower lid retraction. Subtarsal (mid-eyelid): Skin incision in a natural crease 5-7 mm below the lash line. Lower ectropion risk than subciliary. Transcaruncular: For medial orbital wall (lamina papyracea) fractures. Incision through the caruncle, dissecting posterior to the lacrimal sac into the medial orbit.
Intraoperative navigation: Computer-assisted navigation (stereotactic surgery) is increasingly used for complex orbital reconstruction — particularly for secondary orbital reconstruction, large medial wall defects, and cases requiring precise volume restoration. Preoperative CT is loaded into the navigation system, and the surgeon uses a tracked pointer or drill to verify implant position in real time. Reduces need for revision surgery in complex orbital fractures. PMID: 19837305
08 Nasal & Frontal Sinus Fractures
Nasal Fractures
The nasal bones are the most commonly fractured facial bones. Diagnosis is primarily clinical — palpation of crepitus, deviation, epistaxis, periorbital ecchymosis. CT is not required for isolated nasal fractures but should be obtained if more extensive injury is suspected.
Closed reduction: Performed within 2-3 hours of injury (before significant swelling) or at 5-10 days (after swelling resolves but before bony union at ~14 days). Technique: Walsham forceps for the septum, Asch forceps for the nasal bones, or Boies elevator. External splint applied for 7-10 days.
Emergency Septal hematoma: Collection of blood between the perichondrium and septal cartilage. Must be identified and drained immediately — the cartilage depends on the perichondrium for blood supply, and an undrained hematoma causes avascular necrosis of the cartilage within 48-72 hours, leading to septal perforation and saddle nose deformity. Treatment: incision and drainage, nasal packing or through-and-through quilting sutures to prevent reaccumulation, antibiotics (antistaphylococcal).
Frontal Sinus Fractures
The frontal sinus develops around age 6-8 and is fully pneumatized by age 12-18. It has a thick anterior table (average 4-8 mm) and a thin posterior table (average 0.4-1 mm). The nasofrontal outflow tract (NFOT, also called the frontal recess or nasofrontal duct) drains into the middle meatus. The management algorithm depends on which table is involved and whether the NFOT is obstructed:
| Fracture Pattern | NFOT Status | Management |
|---|---|---|
| Anterior table only, nondisplaced | Intact | Observation; serial imaging if concern for NFOT injury |
| Anterior table only, displaced | Intact | ORIF of anterior table (coronal approach) with 1.0-1.5 mm microplates |
| Anterior table, displaced | Obstructed | ORIF of anterior table + obliteration of sinus (strip mucosa, fill with fat, bone, or hydroxyapatite) |
| Posterior table, displaced > 1 table width | Any | Cranialization — remove posterior table, repair dura, allow brain to expand into the sinus space; obliterate NFOT |
| Through-and-through (both tables) | Obstructed | Cranialization + ORIF anterior table + obliterate NFOT + repair any dural tears |
09 Dentoalveolar Trauma
Tooth Avulsion — Reimplantation Protocol
Tooth avulsion (complete displacement of the tooth from the socket) is a true dental emergency. Successful reimplantation depends critically on extraoral dry time and PDL cell viability:
- Replant immediately if possible (< 5 min extraoral time = best prognosis)
- If the tooth is contaminated, rinse gently with saline — do NOT scrub or handle the root
- Storage media (in order of preference): (1) Hank's Balanced Salt Solution (HBSS) — Save-a-Tooth kit — maintains PDL viability for up to 24 hours, (2) milk — maintains viability for 1-3 hours, (3) saline, (4) saliva (buccal vestibule). Avoid water (hypotonic — lyses PDL cells).
- After replantation: flexible splint (composite + wire or titanium trauma splint) for 2 weeks; begin root canal therapy at 7-10 days (for teeth with closed apices); antibiotics (doxycycline or penicillin); tetanus booster if indicated
- Extraoral dry time > 60 minutes = poor prognosis, root resorption inevitable; reimplant for alveolar bone preservation but expect ankylosis and eventual replacement resorption
Ellis Classification of Tooth Fractures
| Class | Description | Treatment |
|---|---|---|
| Ellis I | Enamel only (white/chalky fracture surface) | Smooth sharp edges; composite bonding if esthetic concern; no urgency |
| Ellis II | Enamel + dentin (yellow/ivory fracture surface; tooth sensitivity to air/temperature) | Calcium hydroxide liner over exposed dentin + composite coverage to seal dentinal tubules. Prevent bacterial contamination of pulp. Refer to dentist within 24 hours |
| Ellis III | Enamel + dentin + pulp exposure (pink or red spot visible, bleeding from fracture site) | Emergency Dental emergency — direct pulp cap (MTA or calcium hydroxide) if exposure < 1 mm and < 24 hours; partial pulpotomy (Cvek) for larger exposures in teeth with open apices; root canal therapy for mature teeth with large exposures or delayed presentation |
Luxation Injuries
Concussion: Tender to percussion, no mobility, no displacement. Treatment: soft diet, monitoring. Subluxation: Increased mobility, no displacement, bleeding from gingival sulcus. Treatment: flexible splint for 2 weeks if very mobile. Lateral luxation: Tooth displaced labially/lingually/laterally, often locked in bone. Treatment: reposition with forceps under local anesthesia, flexible splint 4 weeks. Intrusion: Tooth driven into the socket — worst prognosis of all luxation injuries; high risk of ankylosis and root resorption. In permanent teeth with closed apices, orthodontic or surgical extrusion within 2-3 weeks. In immature teeth, allow spontaneous re-eruption. Extrusion: Tooth partially displaced axially out of socket. Reposition, flexible splint for 2 weeks.
Alveolar Fractures
Fracture of the alveolar bone segment, often with multiple teeth in the fragment. Diagnosis: mobility of a bone-tooth segment as a unit, occlusal derangement, mucosal laceration. Treatment: reduce under local anesthesia, stabilize with a rigid splint (arch bar) for 4 weeks, monitor pulp vitality of involved teeth.
Soft Tissue Injuries of the Face
Principles of facial laceration repair: The face has an excellent blood supply, allowing primary closure up to 24 hours after injury (longer than other body regions). Debridement should be conservative — remove clearly necrotic tissue only. Close in layers: muscle (4-0 or 5-0 resorbable), subcutaneous tissue (5-0 resorbable), skin (6-0 nylon or fast-absorbing gut). Key anatomic structures to identify and repair: facial nerve branches (repair with microsurgical technique if divided medial to a vertical line from the lateral canthus), parotid duct (Stensen's duct — repair over a stent if transected; located on a line from the tragus to the midpoint of the upper lip, crossing the masseter), lacrimal canaliculi (repair over silicone stent if medial eyelid injury), and the vermilion border (even 1 mm of malalignment is noticeable — align this first using a skin hook for precise apposition).
Lip lacerations: The vermilion border is the single most important landmark to align. For through-and-through lacerations: close in three layers — oral mucosa (4-0 chromic gut), orbicularis oris muscle (4-0 resorbable), and skin (6-0 nylon). Tissue loss < 25% of the lip length can usually be closed primarily; > 25% requires local flap (Abbe flap from the opposite lip for central defects, Karapandzic or Estlander flaps for commissure defects).
Ear avulsion/laceration: Preserve all tissue — even partially avulsed ear segments can survive due to random pattern blood supply. Close perichondrium-to-perichondrium to prevent cartilage exposure and secondary chondritis. Auricular hematoma must be drained (as with septal hematoma) and a bolster dressing applied to prevent cauliflower ear.
Dog bite facial lacerations: High-risk for infection (Pasteurella multocida, Staphylococcus, Streptococcus, anaerobes). Despite the general surgical principle that bite wounds should not be closed primarily, facial bite wounds ARE closed primarily due to the excellent facial blood supply and the unacceptable esthetic outcome of secondary healing. Management: copious irrigation (at least 250 mL), conservative debridement, layered primary closure, prophylactic antibiotics (amoxicillin-clavulanate 875/125 mg BID for 5-7 days), tetanus prophylaxis, and rabies assessment. Infection rate with this approach: 1-5% (much lower than bite wounds elsewhere on the body — 15-30%).
Gunshot wounds to the face: Classified by velocity — low velocity (handgun): limited tissue destruction, fracture patterns follow bullet trajectory, bullet fragments may be left in place if in non-critical locations. High velocity (rifle): massive tissue destruction, cavitation effect, extensive comminution and soft tissue loss requiring staged reconstruction. Management priorities: airway first (often requires emergent tracheostomy/cricothyrotomy), hemorrhage control, CT survey of facial skeleton and brain, debridement and primary soft tissue closure when possible, delay definitive skeletal reconstruction 48-72 hours for wound stabilization, definitive reconstruction with ORIF and free tissue transfer for tissue deficits.
10 Cephalometric Analysis
Key Landmarks
| Landmark | Abbreviation | Definition |
|---|---|---|
| Sella | S | Center of the sella turcica (pituitary fossa) |
| Nasion | N | Most anterior point of the frontonasal suture |
| A-point (Subspinale) | A | Deepest concavity on the anterior maxilla between ANS and alveolar crest — represents maxillary AP position |
| B-point (Supramentale) | B | Deepest concavity on the anterior mandible between Pogonion and alveolar crest — represents mandibular AP position |
| Pogonion | Pog | Most anterior point on the bony chin |
| Gnathion | Gn | Most anteroinferior point on the chin — intersection of facial plane and mandibular plane |
| Menton | Me | Most inferior point on the mandibular symphysis |
| Gonion | Go | Most posteroinferior point on the mandibular angle |
| Anterior nasal spine | ANS | Tip of the bony anterior nasal spine |
| Posterior nasal spine | PNS | Most posterior point on the hard palate |
Key Measurements
| Measurement | Normal Value | Clinical Significance |
|---|---|---|
| SNA | 82° ± 2° | Maxillary AP position relative to cranial base; > 84° = maxillary protrusion; < 80° = maxillary retrusion |
| SNB | 80° ± 2° | Mandibular AP position relative to cranial base; > 82° = mandibular prognathism; < 78° = mandibular retrognathism |
| ANB | 2° ± 2° | Skeletal relationship between maxilla and mandible; > 4° = skeletal Class II; < 0° = skeletal Class III |
| Mandibular plane angle (SN-GoMe) | 32° ± 5° | High angle (> 37°) = long face, hyperdivergent; Low angle (< 27°) = short face, hypodivergent |
| Occlusal plane to SN | 14° ± 3° | Steep occlusal plane associated with anterior open bite |
| Upper incisor to SN | 104° ± 6° | Excessive = proclined upper incisors (dental compensation for skeletal Class III) |
| Lower incisor to mandibular plane (IMPA) | 90° ± 5° | Excessive = proclined lower incisors (dental compensation for skeletal Class II) |
| Wits appraisal | M: 1 mm, F: 0 mm | Perpendicular projections of A and B points onto the occlusal plane; more reliable than ANB in patients with unusual cranial base angles |
Steiner Analysis: Uses SNA, SNB, ANB, upper incisor to NA (distance and angle), lower incisor to NB (distance and angle), and Holdaway ratio. McNamara Analysis: Uses nasion perpendicular to Frankfort horizontal as a reference — measures A-point to N-perpendicular (normal: 0-1 mm) and Pogonion to N-perpendicular (normal: -4 to 0 mm in adults). Effective lower face height and mandibular length are also measured.
Virtual Surgical Planning (VSP)
Modern orthognathic surgery increasingly uses 3D virtual surgical planning with CBCT-derived skeletal models and digital dental models (intraoral scan or scanned stone models). The surgical movements are planned digitally, and patient-specific cutting guides and intermediate/final splints are 3D-printed for intraoperative use. VSP improves accuracy of skeletal repositioning (particularly in asymmetry cases and three-piece Le Fort I osteotomies), reduces operative time, and improves surgeon-orthodontist communication. The workflow: (1) CBCT scan + dental impressions/intraoral scan, (2) virtual planning session with the surgeon and planning engineer, (3) fabrication of cutting guides, splints, and custom plates, (4) surgery using the guides. PMID: 24795279
11 Orthognathic Procedures
Le Fort I Osteotomy
The workhorse maxillary osteotomy. The osteotomy line follows the Le Fort I fracture pattern — horizontal cut above the tooth apices through the lateral maxillary wall, lateral nasal wall, and nasal septum. The maxilla is then downfractured and mobilized.
- Maxillary advancement: For maxillary retrusion (common in cleft patients). Average advancement 4-8 mm. Relapse rate is low with rigid fixation (four L-shaped plates at piriform rims and zygomaticomaxillary buttresses).
- Maxillary impaction: For vertical maxillary excess (gummy smile, long lower face). Superior repositioning of the maxilla. Requires removal of a bone strip from the lateral wall. Most stable movement in orthognathic surgery — relapse < 10%.
- Maxillary inferior repositioning (downgraft): For short face syndrome. Requires bone grafts to fill the gap. Least stable movement.
- Segmental Le Fort I: The maxilla is divided into segments (usually 2 or 3 pieces) to correct transverse discrepancies (expansion) or AP discrepancies (anterior setback/advancement of a segment).
Key surgical risk: Descending palatine artery — runs in the greater palatine canal approximately 3-5 mm posterior to the third molar. Must be protected or controlled during pterygomaxillary separation. Hemorrhage from the pterygoid venous plexus or internal maxillary artery can be severe.
Bilateral Sagittal Split Osteotomy (BSSO)
The most commonly performed mandibular orthognathic procedure. Originally described by Trauner and Obwegeser (1957), modified by Dal Pont (1961). The osteotomy splits the mandibular ramus sagittally into a proximal (condyle-bearing) segment and a distal (tooth-bearing) segment:
- Medial horizontal cut above the lingula (on the medial ramus surface)
- Sagittal cut through the buccal cortex of the ramus and body to the inferior border
- The split extends through the cancellous bone, carrying the IAN with the distal segment (the IAN lies in the medullary bone between the buccal and lingual cortical plates)
Mandibular advancement: Most common indication; advance the distal segment and fixate with three bicortical positional screws (Epker technique, 2.0 mm diameter, 60° angle) or a plate with monocortical screws on each side. Advancement > 10 mm has higher relapse rates and may require concurrent genioplasty. IAN injury (hypoesthesia/paresthesia of the lower lip) occurs in 5-30% of cases; most recover within 6-12 months. Mandibular setback: For mandibular prognathism. Remove a section of bone from the overlap. Generally very stable.
Genioplasty (Sliding Advancement/Reduction)
A horizontal osteotomy of the chin below the mental foramen (at least 5 mm below the mental foramen and tooth apices to avoid injury). The inferior chin segment is mobilized and repositioned: advancement (most common), setback, vertical reduction, lengthening (with bone graft), or lateral shifting. Fixation: single plate or step plate with monocortical screws. Can be performed as an isolated procedure or in conjunction with BSSO/Le Fort I. Genioplasty does not change occlusion.
Surgical-First Approach
Traditionally, orthognathic surgery follows 12-18 months of presurgical orthodontics to decompensate the dentition. The surgery-first approach performs orthognathic surgery before any orthodontic treatment, followed by postoperative orthodontics. Benefits: shorter total treatment time (reduced by 6-12 months), immediate facial improvement. Challenges: requires very accurate 3D surgical planning (virtual surgical planning/VSP), limited to straightforward single-jaw or double-jaw cases, postoperative orthodontic finishing may be more complex. PMID: 24295848
12 Distraction Osteogenesis
Principles
Distraction osteogenesis (DO) generates new bone by gradually separating two osteotomized bone segments. First described by Ilizarov (1950s, orthopedics), adapted for the craniofacial skeleton by McCarthy (1992). The process has three phases:
| Phase | Duration | Description |
|---|---|---|
| Latency | 5-7 days | Time between osteotomy and the start of distraction; allows initial callus formation |
| Activation (Distraction) | Variable (rate: 1 mm/day) | Active separation of bone segments; standard rate is 1 mm/day (0.5 mm twice daily). New bone, blood vessels, and soft tissue form in the distraction gap. |
| Consolidation | 6-12 weeks (2× the distraction period as a rule) | Maturation and mineralization of the regenerate bone; device remains in place |
Applications in OMS
Mandibular distraction: Used for (1) neonatal mandibular hypoplasia causing airway obstruction (Pierre Robin sequence — avoids tracheostomy), (2) hemifacial microsomia (Pruzansky-Kaban classification), (3) severe mandibular retrognathia requiring advancement > 10 mm (where BSSO alone has high relapse). Internal (buried) or external (pin-based) devices available.
Midface distraction: Le Fort III distraction for syndromic craniosynostosis (Apert, Crouzon, Pfeiffer syndromes) with severe midface hypoplasia. Performs a Le Fort III osteotomy and applies an internal or external (RED — rigid external distraction) device. Allows gradual advancement of the entire midface (15-25 mm possible), avoiding the relapse and soft tissue limitations of acute advancement.
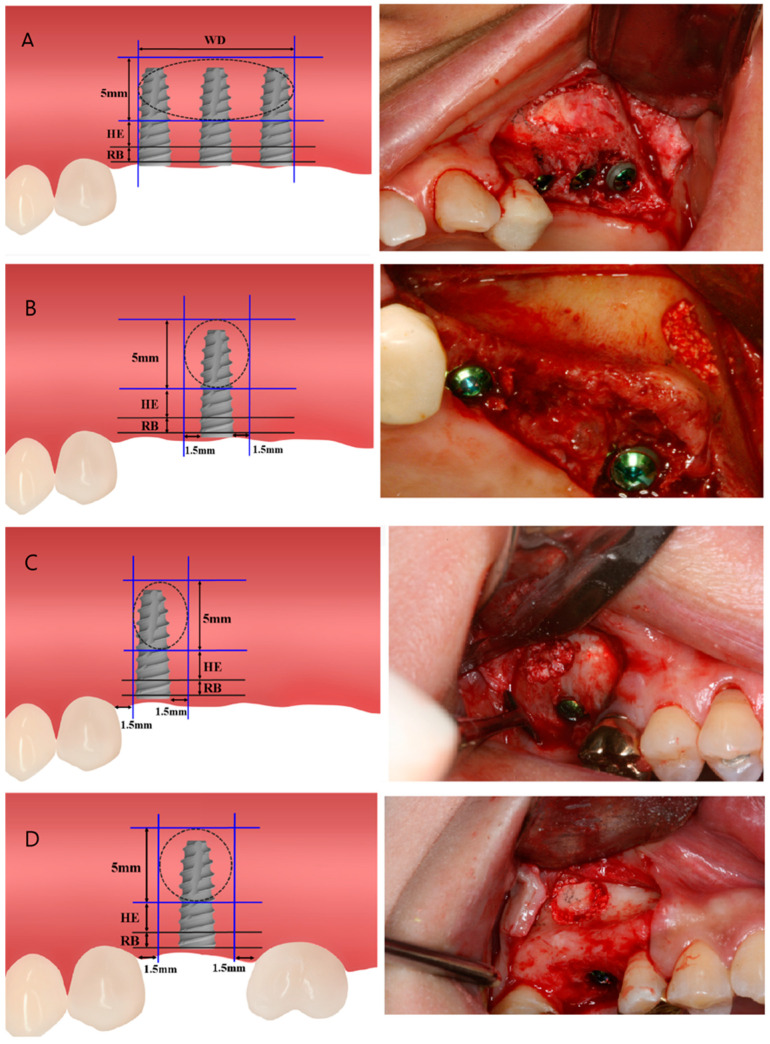
Alveolar distraction: Vertical augmentation of a deficient alveolar ridge prior to implant placement. Performs a horizontal osteotomy and gradually distracts the transport segment superiorly (or inferiorly in the maxilla). Advantages over block bone grafting: simultaneous bone and soft tissue generation, no donor site. Disadvantages: technique sensitivity, device complications, vector control difficulty.
Cleft Lip & Palate — OMS Role
OMS surgeons play a key role in the multidisciplinary cleft team, particularly in:
- Alveolar bone grafting: Performed at age 7-9 years (mixed dentition, before canine eruption). Iliac crest cancellous bone graft placed into the alveolar cleft defect to close the oronasal fistula, provide bone support for eruption of the canine, and stabilize the premaxilla. Timing guided by dental development (canine root 1/2 to 2/3 formed). Success rate: 85-95% when performed at optimal timing.
- Orthognathic surgery: 20-25% of cleft lip and palate patients develop maxillary hypoplasia requiring Le Fort I advancement at skeletal maturity (age 16-18 years). The scarred palate limits advancement and increases relapse — distraction osteogenesis (Le Fort I with internal or external distraction) is an alternative for large advancements (> 8 mm).
- Distraction osteogenesis: For severe maxillary hypoplasia where conventional Le Fort I advancement would relapse (advancement > 8-10 mm). Also for mandibular hypoplasia in associated conditions (Pierre Robin sequence).
13 Obstructive Sleep Apnea Surgery
MMA — Maxillomandibular Advancement
MMA is the most effective skeletal surgical treatment for moderate-to-severe obstructive sleep apnea (OSA). It combines a Le Fort I maxillary advancement with a BSSO mandibular advancement (typically 10-12 mm each), expanding the entire posterior airway space (PAS). PAS increases by approximately 50-75% after MMA. Success rate (defined as AHI < 20 and ≥ 50% reduction): 85-90%, making it the most predictably effective surgical treatment for OSA. PMID: 20456774
Patient selection: BMI < 35 preferred (though MMA can be effective at higher BMI). Failed or intolerant of CPAP. Cephalometric findings of mandibular retrognathia (SNB < 78°) and narrow PAS (< 11 mm at the base of the tongue). Skeletal Class II patients are ideal candidates.
Complications of MMA
IAN paresthesia (30-50% initial, most resolve within 6-12 months), velopharyngeal insufficiency (rare — 1-2%, more common with large advancements), relapse (5-10% with rigid fixation), unfavorable esthetic change (widened alar base — perform alar cinch suture; thinned upper lip — perform V-Y closure of the vestibular incision), condylar resorption (particularly in young females with preexisting condylar resorption — idiopathic condylar resorption/ICR), hemorrhage from pterygoid venous plexus or descending palatine artery, bad splits during BSSO (unfavorable fracture extending to the inferior border or condylar neck — salvage with additional plates or conversion to vertical ramus osteotomy).
Other OMS Procedures for OSA
Genioglossus advancement (GA): A rectangular osteotomy of the genial tubercle (origin of the genioglossus muscle) is advanced anteriorly and rotated 90° to prevent retraction. Modest improvement in AHI. Often combined with hyoid suspension. Hyoid suspension (myotomy-suspension): The hyoid bone is advanced and suspended anteriorly to the mandible or thyroid cartilage with sutures or tape. Prevents posterior tongue base collapse. UPPP (Uvulopalatopharyngoplasty): Removal or reshaping of the uvula, soft palate, and redundant pharyngeal tissue. Addresses retropalatal obstruction. Success rate alone: 40-50%. Often combined with other procedures in a multilevel approach.
14 Odontogenic Cysts
Classification & Key Features
| Cyst | Origin | Radiographic Appearance | Key Features | Treatment |
|---|---|---|---|---|
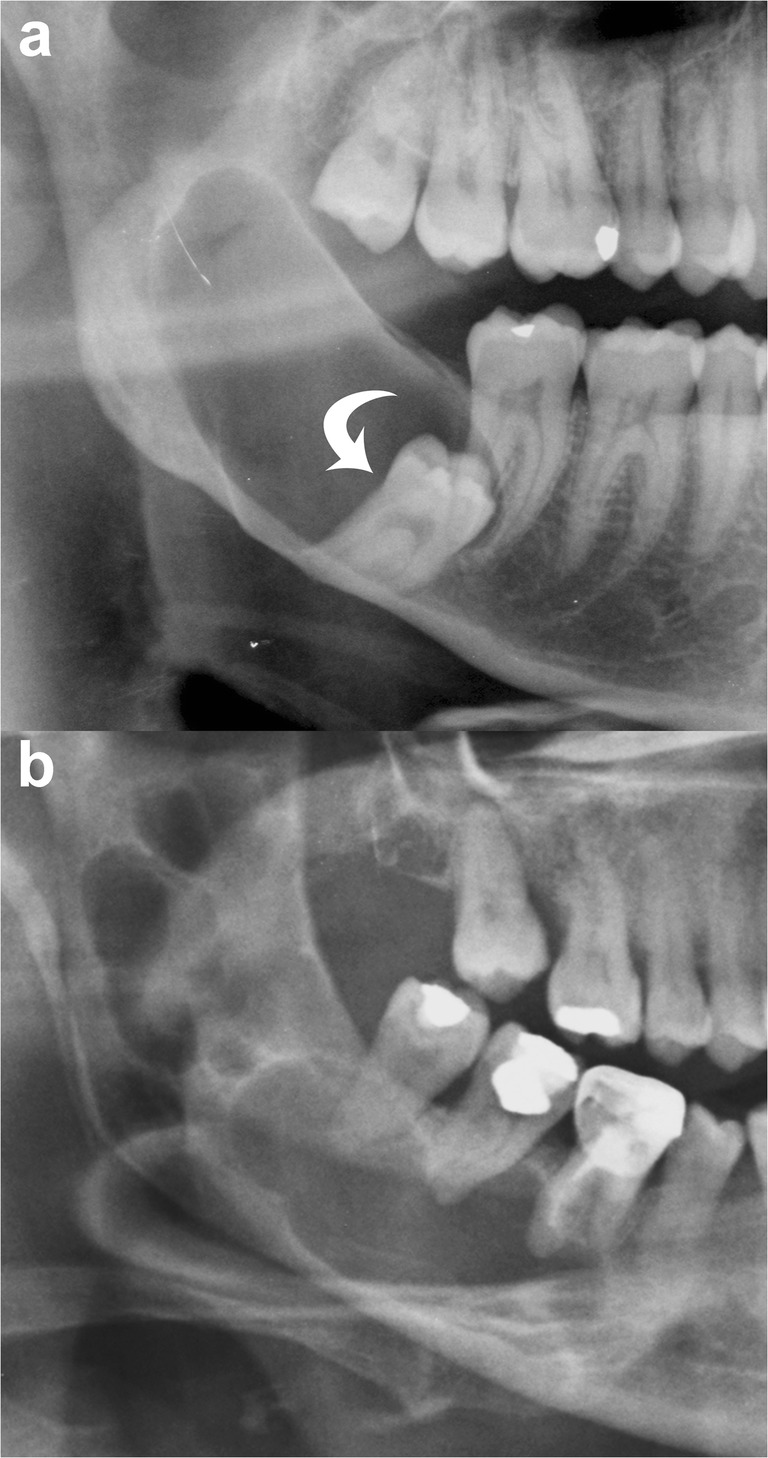
| Dentigerous (Follicular) Cyst | Reduced enamel epithelium (dental follicle) | Well-defined unilocular radiolucency attached to the CEJ of an unerupted tooth (most often mandibular third molar) | Second most common odontogenic cyst; may cause significant bone expansion; attached at the cementoenamel junction (CEJ); rarely undergoes malignant transformation (SCC or ameloblastoma) | Enucleation with extraction of the associated tooth; marsupialization in children to preserve permanent tooth eruption |
| Odontogenic Keratocyst (OKC) / Keratocystic Odontogenic Tumor | Dental lamina rests | Well-defined unilocular or multilocular radiolucency; posterior mandible most common; may extend along the ramus with minimal expansion | High recurrence rate (25-60% with simple curettage); lined by parakeratinized stratified squamous epithelium (6-8 cell layers, palisaded basal layer); grows AP along the medullary cavity; budding of daughter cysts in the wall. Associated with Gorlin syndrome (Basal Cell Nevus Syndrome) — multiple OKCs + basal cell carcinomas + skeletal anomalies (bifid ribs) + calcified falx cerebri; PTCH1 gene mutation (chromosome 9q) | Enucleation + peripheral ostectomy (Carnoy's solution or cryotherapy to the bony walls); curettage alone has unacceptable recurrence. Long-term follow-up (minimum 5 years with annual panoramic radiographs) is mandatory. Resection for multiply recurrent lesions. |
| Radicular (Periapical) Cyst | Rests of Malassez (epithelial cell rests of the periodontal ligament) | Well-defined radiolucency at the apex of a nonvital (carious/traumatized) tooth | Most common odontogenic cyst overall; results from chronic periapical inflammation stimulating epithelial rests; contains cholesterol clefts on histology; lined by nonkeratinized stratified squamous epithelium; residual cyst remains after extraction without curettage | Root canal therapy (most resolve); apicoectomy + enucleation if persistent; extraction + curettage |
| Lateral Periodontal Cyst | Dental lamina rests | Small (< 1 cm), well-defined radiolucency lateral to a vital tooth root (premolar region most common) | Uncommon; lined by thin nonkeratinized epithelium with focal plaques (clear cells); botryoid variant (multilocular, grape-like — higher recurrence) | Enucleation; teeth can usually be preserved |
Non-Odontogenic Cysts of the Jaws
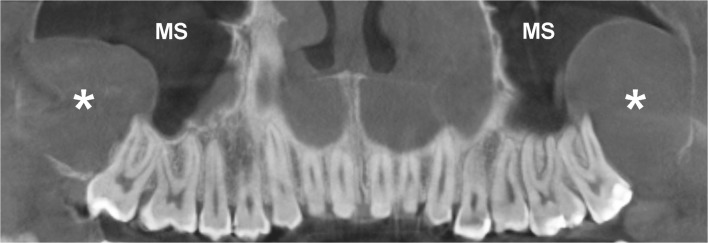
Nasopalatine duct cyst (incisive canal cyst): Most common non-odontogenic cyst of the jaws. Arises from epithelial remnants of the nasopalatine duct. Radiograph: well-defined, heart-shaped radiolucency between the roots of the maxillary central incisors. Teeth are vital (distinguishes from radicular cyst). Treatment: enucleation; low recurrence. Nasolabial cyst: Soft tissue cyst in the nasolabial fold area (extraosseous). Presents as a fluctuant swelling that obliterates the nasolabial fold and may elevate the alar base. Treatment: excision via sublabial approach. Stafne bone cavity (static bone cyst): Not a true cyst — a developmental concavity on the lingual surface of the mandible below the inferior alveolar canal, containing a lobe of the submandibular gland. Radiolucency below the canal, typically in the posterior mandible. No treatment needed — pathognomonic appearance on CT confirms diagnosis.
15 Odontogenic Tumors
Key Entities
| Tumor | Origin | Radiographic Appearance | Behavior | Treatment |
|---|---|---|---|---|
| Ameloblastoma (Conventional/Solid) | Odontogenic epithelium (enamel organ, dental lamina, epithelial lining of dentigerous cyst) | Multilocular radiolucency ("soap bubble" or "honeycomb" pattern); posterior mandible (80%); root resorption common | Locally aggressive; high recurrence with curettage alone (50-90%); does not metastasize (usually) but invades bone beyond radiographic margins by 1-2 cm; histologic subtypes: follicular, plexiform, acanthomatous, granular, desmoplastic | Resection with 1-1.5 cm bony margin; marginal mandibulectomy for small lesions, segmental mandibulectomy for large lesions; reconstruction with fibula free flap. Long-term follow-up (≥ 10 years) is essential — recurrence can appear 15-20 years later. |
| Unicystic Ameloblastoma | Odontogenic epithelium | Unilocular radiolucency, often associated with an impacted third molar (resembles dentigerous cyst) | Less aggressive than conventional; three subtypes: luminal (Type I), intraluminal (Type II — plunging into the lumen), mural (Type III — invading the cyst wall). Mural type behaves like conventional ameloblastoma. | Luminal & intraluminal: enucleation + Carnoy's solution (recurrence 10-20%). Mural: resection with margins (as for conventional). |
| Peripheral Ameloblastoma | Odontogenic epithelium (extraosseous) | No bony radiolucency (soft tissue lesion only); may cause superficial cortical bone saucerization | Least aggressive; arises in the gingiva/alveolar mucosa; no bony invasion | Local excision with adequate soft tissue margins; recurrence < 20% |
| Odontoma (Compound) | Odontogenic tissue (fully differentiated) | Cluster of small tooth-like structures (denticles) with a surrounding radiolucent capsule; anterior maxilla most common | Most common odontogenic tumor; hamartoma rather than true neoplasm; prevents eruption of associated permanent tooth | Conservative enucleation; does not recur |
| Odontoma (Complex) | Odontogenic tissue (disorganized) | Amorphous radiopaque mass with a surrounding radiolucent halo; posterior mandible most common | Disorganized dental tissue; hamartomatous | Conservative enucleation; does not recur |
| Odontogenic Myxoma | Dental papilla (ectomesenchyme) | Multilocular radiolucency with thin, angular septa ("tennis racket" or "sunburst" pattern) | Locally aggressive; gelatinous gross appearance; invades surrounding bone without a capsule | Curettage for small lesions; resection with margins for large lesions (high recurrence with curettage alone — 25%) |
| Ameloblastic Carcinoma | Odontogenic epithelium (malignant) | Destructive, irregular radiolucency with cortical perforation | Malignant counterpart of ameloblastoma; may metastasize to lungs and regional lymph nodes | Wide resection with margins + neck dissection if nodal disease + adjuvant radiation |
16 Oral Mucosal Pathology & Premalignancy
Potentially Malignant Disorders
Leukoplakia: A white plaque that cannot be characterized clinically or pathologically as any other disease (diagnosis of exclusion). Prevalence: 1-5% of adults. Malignant transformation rate: 1-9% overall (higher for nonhomogeneous/speckled or verrucous types, floor of mouth/ventral tongue location). Proliferative verrucous leukoplakia (PVL) is a distinct, aggressive subtype with very high malignant transformation (> 70%).
Erythroplakia: A red velvety patch that cannot be diagnosed as any other condition. Carries the highest malignant potential of any oral mucosal lesion — up to 50% are carcinoma in situ or invasive SCC at time of biopsy. Must be biopsied immediately. More common on the floor of mouth, lateral tongue, and soft palate.
Oral Lichen Planus (OLP): Chronic inflammatory mucosal disease. Six clinical forms: reticular (most common — Wickham striae), erosive/ulcerative (most symptomatic, highest malignant potential — 0.5-2%), atrophic, plaque, papular, bullous. Histology: band-like lymphocytic infiltrate at the epithelial-connective tissue interface with "saw-tooth" rete ridges, basal cell degeneration, and civatte bodies (apoptotic keratinocytes). Treatment: topical corticosteroids (fluocinonide, clobetasol), tacrolimus for refractory cases.
Oral Submucous Fibrosis (OSF): Progressive fibrosis of the oral submucosa caused by betel quid (areca nut) chewing. Progressive trismus (inability to open the mouth), blanching of oral mucosa, burning sensation, dysphagia. Malignant transformation rate: 7-13%. No definitive treatment — cessation of habit, intralesional corticosteroids, collagenase injections, surgical release of fibrotic bands for severe trismus.
Biopsy Techniques in OMS
Incisional biopsy: Preferred for lesions > 1 cm. Take a wedge-shaped specimen from the most representative area (the periphery of an ulcerated lesion, including both abnormal and adjacent normal tissue). For suspected malignancy, biopsy should include the transition zone between lesion and normal mucosa. Avoid necrotic centers. Depth should include submucosa. Excisional biopsy: Preferred for small lesions (< 1 cm), suspected benign lesions (fibromas, mucoceles, papillomas), and pigmented lesions (to evaluate the entire specimen for melanoma). Include 2-3 mm margin of normal tissue. Brush biopsy (OralCDx): Screening adjunct for suspicious mucosal lesions. A stiff brush samples all epithelial layers, which are analyzed by computer-assisted cytology. Positive or atypical results require scalpel biopsy for definitive diagnosis. Sensitivity 71-100%, specificity 32-94% (wide range limits reliability as standalone diagnostic). Fine needle aspiration (FNA): Used for salivary gland masses and cervical lymph nodes. 25-gauge needle with syringe aspiration. Sensitivity for salivary tumors: 73-98%. Cannot differentiate tumor grade or invasion — useful for preoperative planning but not definitive treatment planning.
17 Oral Squamous Cell Carcinoma
Risk Factors
Tobacco (smoking and smokeless — 5-10× increased risk), alcohol (synergistic with tobacco — combined use increases risk 15-30×), betel quid/areca nut chewing, HPV (types 16/18 — primarily oropharyngeal, less commonly oral cavity), chronic trauma (controversial), UV radiation (lip SCC), immunosuppression, and prior history of head and neck cancer (3-5% annual risk of second primary).
AJCC 8th Edition TNM Staging (Oral Cavity)
The AJCC 8th edition (2017) introduced depth of invasion (DOI) as a key modifier in T-staging, which is a significant change from previous editions:
| T Stage | Criteria |
|---|---|
| T1 | Tumor ≤ 2 cm, DOI ≤ 5 mm |
| T2 | Tumor ≤ 2 cm with DOI > 5 mm and ≤ 10 mm; OR tumor > 2 cm and ≤ 4 cm with DOI ≤ 10 mm |
| T3 | Tumor > 2 cm and ≤ 4 cm with DOI > 10 mm; OR tumor > 4 cm with DOI ≤ 10 mm |
| T4a | Moderately advanced: invasion of cortical bone (mandible or maxilla), maxillary sinus, or skin of face |
| T4b | Very advanced: invasion of masticator space, pterygoid plates, skull base, or encases internal carotid artery |
Surgical Management
Wide local excision: Primary tumor excised with at least 1 cm clinical margins (confirmed as ≥ 5 mm histologic margins). Mucosal margins checked with frozen sections intraoperatively.
Mandibulectomy:
- Marginal (rim) mandibulectomy: Removes a portion of the mandibular rim (usually the superior alveolar portion) while preserving continuity of the inferior border. Indicated when tumor is adherent to or minimally invading the periosteum/cortex. Requires ≥ 1 cm of residual mandibular height at the inferior border for structural integrity.
- Segmental mandibulectomy: Removes a full-thickness segment of the mandible. Indicated for frank cortical invasion, tumor encircling the IAN canal, or involvement of the medullary bone. Requires reconstruction to restore mandibular continuity (fibula free flap preferred).
Neck Dissection Levels:
| Level | Boundaries | Key Contents |
|---|---|---|
| I (IA/IB) | IA: submental triangle; IB: submandibular triangle | Submental and submandibular lymph nodes; floor of mouth tumors drain here first |
| II (IIA/IIB) | Upper jugular: skull base to hyoid; IIA anterior to CN XI, IIB posterior | Jugulodigastric nodes; primary drainage for oral tongue, FOM, oropharynx |
| III | Middle jugular: hyoid to cricoid | Middle jugular nodes |
| IV | Lower jugular: cricoid to clavicle | Lower jugular and supraclavicular nodes |
| V (VA/VB) | Posterior triangle: posterior border of SCM to anterior border of trapezius | Spinal accessory nodes (VA) and transverse cervical/supraclavicular (VB) |
Elective neck dissection (supraomohyoid: levels I-III) is recommended for clinically N0 oral cavity SCC when the occult metastasis risk exceeds 15-20% — generally for tumors with DOI > 4 mm or T2+ tumors. The SEND trial established the benefit of elective neck dissection for early oral SCC over therapeutic neck dissection at recurrence. PMID: 26286723
N-Staging (AJCC 8th Edition — Oral Cavity)
| N Stage | Criteria (Clinical) |
|---|---|
| N0 | No regional lymph node metastasis |
| N1 | Single ipsilateral node ≤ 3 cm, ENE-negative |
| N2a | Single ipsilateral node > 3 cm and ≤ 6 cm, ENE-negative |
| N2b | Multiple ipsilateral nodes ≤ 6 cm, ENE-negative |
| N2c | Bilateral or contralateral nodes ≤ 6 cm, ENE-negative |
| N3a | Any node > 6 cm, ENE-negative |
| N3b | Any node with clinical extranodal extension (ENE+) |
Adjuvant Therapy
Adjuvant radiation therapy (60-66 Gy in 30-33 fractions) is indicated for: advanced T-stage (T3-T4), positive or close margins (< 5 mm), perineural invasion, lymphovascular invasion, multiple positive lymph nodes, or any positive node in the setting of other adverse features. Adjuvant concurrent chemoradiation (cisplatin 100 mg/m² Q3 weeks × 3 cycles + radiation) is indicated for the two highest-risk features: positive surgical margins and extranodal extension. This was established by the EORTC 22931 and RTOG 9501 trials.
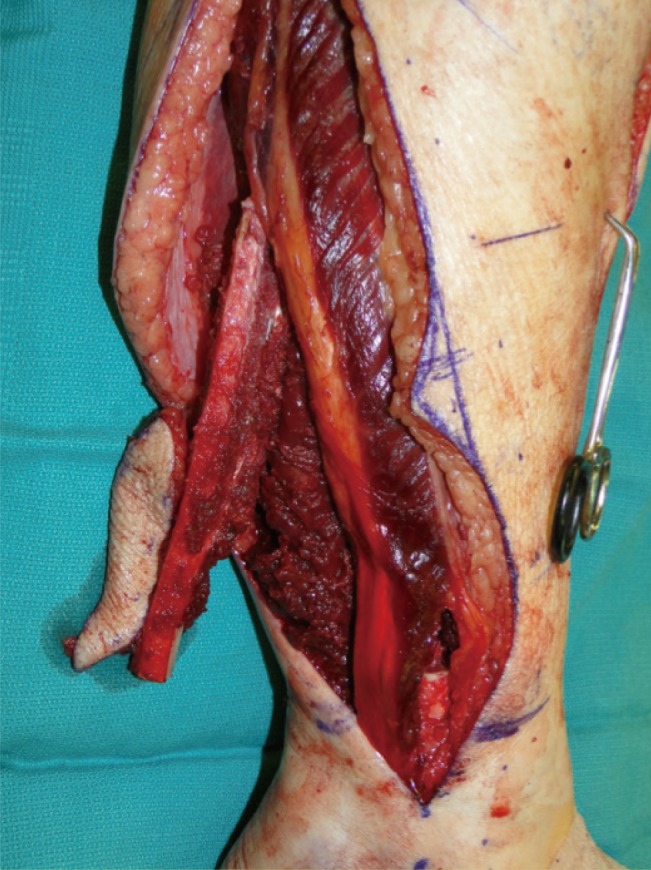
Reconstruction
| Flap | Type | Pedicle | Best Applications |
|---|---|---|---|
| Fibula free flap | Osseous/osteocutaneous | Peroneal artery & vein | Gold standard for mandibular reconstruction; up to 25 cm of bone; can accept dental implants; skin paddle for intraoral lining |
| Radial forearm free flap (RFFF) | Fasciocutaneous | Radial artery & vein | Thin, pliable — ideal for tongue, floor of mouth, and soft palate reconstruction. Positive Allen's test is a contraindication. |
| Scapula free flap | Osseous/osteocutaneous | Circumflex scapular artery or thoracodorsal | Combined soft tissue and bone; useful when fibula unavailable (PVD, absent peroneal artery) |
| ALT (anterolateral thigh) free flap | Fasciocutaneous/myocutaneous | Descending branch of lateral circumflex femoral artery | Versatile — large soft tissue defects, through-and-through defects, can be thinned; minimal donor site morbidity |
18 Salivary Gland Disease
Obstructive Conditions
Sialolithiasis: Calcified stones in the salivary duct or gland. 80-90% occur in the submandibular gland (Wharton's duct) — the submandibular gland produces mucous (thicker) saliva that must flow uphill against gravity, and the duct has a tortuous course with a narrow orifice. Stones are composed of calcium phosphate and hydroxyapatite. Presentation: recurrent painful swelling of the gland during eating (meal-time syndrome). Diagnosis: bimanual palpation, occlusal radiograph, CT, ultrasound, or sialography. Treatment: small anterior stones — transoral sialolithotomy; larger or proximal stones — sialendoscopy, extracorporeal shock wave lithotripsy (ESWL); recurrent or gland destruction — submandibular gland excision (protect the marginal mandibular and lingual nerves).
Sialadenitis: Inflammation of the salivary gland, usually from retrograde bacterial infection. Acute suppurative sialadenitis most commonly affects the parotid gland in dehydrated, postoperative, or debilitated elderly patients. Organism: Staphylococcus aureus most common. Treatment: hydration, sialogogues (lemon drops, pilocarpine), massage, warm compresses, antibiotics (amoxicillin-clavulanate or clindamycin). Incision and drainage if abscess forms.
Mucocele & Ranula: Mucocele is a mucus extravasation phenomenon (ruptured minor salivary gland duct — no true epithelial lining) most common on the lower lip. A ranula is a large mucocele in the floor of the mouth arising from the sublingual gland. Simple ranula: confined to the sublingual space. Plunging ranula: extends through the mylohyoid muscle into the submandibular or parapharyngeal space. Treatment: mucocele — excision with associated minor salivary gland; ranula — marsupialization or excision of the sublingual gland (most definitive, lowest recurrence).
Salivary Gland Tumors
| Tumor | Type | Key Features | Treatment |
|---|---|---|---|
| Pleomorphic Adenoma (Benign mixed tumor) | Benign | Most common salivary tumor overall (60-70%); parotid gland most common site (80%); well-circumscribed but has pseudopods that extend through the capsule (recurrence with enucleation: 20-50%); malignant transformation = carcinoma ex pleomorphic adenoma (5-15% lifetime risk) | Superficial parotidectomy with facial nerve preservation (for parotid tumors); complete excision of gland for submandibular tumors. Never enucleate — always excise with cuff of normal tissue. |
| Warthin Tumor (Papillary cystadenoma lymphomatosum) | Benign | Second most common benign salivary tumor; almost exclusively in parotid; bilateral in 10-15%; strong male and smoking association; papillary/cystic on FNA. "Hot" on Tc-99m pertechnetate scan (only salivary tumor that concentrates technetium). | Superficial parotidectomy or excision; observation may be appropriate in elderly/comorbid |
| Mucoepidermoid Carcinoma | Malignant | Most common malignant salivary tumor; parotid most common site; grading: low, intermediate, high. Low grade: cystic, mucous-producing, favorable prognosis (5-year survival > 90%). High grade: solid, aggressive, poor prognosis (5-year survival 40-50%); most common salivary malignancy in children. | Wide excision (total parotidectomy with nerve preservation if possible for parotid); neck dissection for high-grade or clinical nodes; adjuvant radiation for high-grade, close/positive margins, perineural invasion, nodal disease |
| Adenoid Cystic Carcinoma | Malignant | Second most common malignant salivary tumor; most common malignancy of the submandibular, sublingual, and minor salivary glands; hallmark: perineural invasion (found in 80% — tumor tracks along nerves for centimeters beyond the clinical/radiographic margin); three histologic patterns: cribriform ("Swiss cheese" — best prognosis), tubular, solid (worst prognosis). Indolent but relentless course — late distant metastases (lung) can appear 10-20 years after treatment. | Wide resection with attention to nerve margins (perineural invasion extends far beyond gross tumor); adjuvant radiation therapy (often neutron beam or proton therapy); neck dissection only if nodes clinically positive (nodal metastasis is rare — 5-10%) |
19 Medication-Related Osteonecrosis of the Jaw (MRONJ)
Definition & Risk Factors
MRONJ is defined by the AAOMS as exposed bone or bone that can be probed through an intraoral or extraoral fistula in the maxillofacial region, persisting for > 8 weeks, in a patient with current or previous exposure to antiresorptive or antiangiogenic therapy, and with no history of radiation to the jaws.
Causative medications:
- Bisphosphonates (nitrogen-containing): Zoledronic acid (IV — highest risk, cumulative dose-dependent), pamidronate (IV), alendronate (oral), risedronate (oral). IV bisphosphonates for cancer: risk 1-15%; oral bisphosphonates for osteoporosis: risk 0.01-0.1%.
- Denosumab (Prolia/Xgeva): RANKL inhibitor; MRONJ risk similar to or slightly higher than IV bisphosphonates for cancer patients (1-2%); unlike bisphosphonates, the effect is reversible (not incorporated into bone matrix).
- Antiangiogenic agents: Bevacizumab (Avastin), sunitinib — MRONJ risk 0.2-1%.
Risk factors for MRONJ: Dentoalveolar surgery (extraction is the most common precipitant — 50-60% of cases), dental implant placement, periodontal disease, ill-fitting dentures, duration and potency of drug exposure, concurrent corticosteroid use, concurrent chemotherapy.
AAOMS Staging & Treatment
| Stage | Description | Treatment |
|---|---|---|
| At Risk | No apparent necrotic bone; patient on antiresorptive/antiangiogenic therapy | Patient education; maintain good oral hygiene; complete necessary dental work before starting drug therapy |
| Stage 0 | No clinical evidence of necrotic bone, but nonspecific symptoms or radiographic findings (osteosclerosis, thickening/non-remodeling of PDL, widened PDL space) | Systemic management (pain control, antibiotics if infection); adjust antiresorptive therapy if possible (coordinate with prescribing physician) |
| Stage 1 | Exposed/necrotic bone or fistula probing to bone; asymptomatic; no evidence of infection | Antimicrobial rinses (0.12% chlorhexidine BID); clinical follow-up quarterly; patient education |
| Stage 2 | Exposed/necrotic bone with evidence of infection (pain, erythema, purulent drainage) | Antibiotics (penicillin, amoxicillin, or clindamycin; metronidazole added for anaerobic coverage); antimicrobial rinses; pain control; superficial debridement of necrotic bone without exposing uninvolved bone |
| Stage 3 | Exposed/necrotic bone with infection and one or more of: pathologic fracture, extraoral fistula, oroantral/oronasal communication, osteolysis extending to the inferior border or sinus floor | Antibiotics + surgical resection of necrotic bone (sequestrectomy/marginal or segmental resection); reconstruction as needed; may require long-term suppressive antibiotics |
20 Surgical Extractions & Impacted Third Molars
Classification of Impacted Third Molars
Pell-Gregory Classification (Depth & Ramus Relationship):
| Depth (Relative to Occlusal Plane of 2nd Molar) | Ramus Relationship (Space Available) | ||
|---|---|---|---|
| Position A | Highest point of tooth is at or above the occlusal plane of the 2nd molar | Class I | Sufficient space between the ramus and distal of the 2nd molar to accommodate the mesiodistal width of the 3rd molar |
| Position B | Highest point is between the occlusal plane and the cervical line of the 2nd molar | Class II | Space available is less than the mesiodistal width of the 3rd molar (partially impacted in the ramus) |
| Position C | Highest point is below the cervical line of the 2nd molar (deepest impaction) | Class III | All or most of the 3rd molar is within the ramus |
Winter Classification (Angulation): Describes the angle of the impacted third molar relative to the long axis of the adjacent second molar: Mesioangular (most common, ~43%), Horizontal, Vertical, Distoangular (most difficult to extract — unfavorable path of withdrawal toward the ramus), Buccoangular, Linguoangular, Inverted (rarest).
Indications for Third Molar Removal
Current evidence supports removal for: (1) pericoronitis (recurrent or severe), (2) caries on the third molar or distal surface of the second molar, (3) periodontal disease (probing > 5 mm distal to the second molar), (4) associated pathology (cyst, tumor), (5) orthodontic treatment planning, (6) teeth in the line of a fracture, (7) prior to orthognathic surgery (remove at least 6 months before BSSO), (8) prophylactic removal of asymptomatic impacted third molars is supported by AAOMS when the risk of pathology outweighs the risk of surgery — particularly for younger patients with deeply impacted teeth that are more likely to develop cysts or cause second molar damage with time. PMID: 27919600
Surgical Technique
Standard approach for impacted lower third molars: (1) Full-thickness mucoperiosteal flap (envelope or triangular with anterior releasing incision); (2) Bone removal with a surgical handpiece and round bur under copious saline irrigation (remove bone overlying the crown — buccal and distal bone); (3) Tooth sectioning (crown from roots, and/or mesial root from distal root) to reduce the amount of bone removal needed and create a path of delivery; (4) Tooth elevation and delivery; (5) Curettage of socket, irrigation; (6) Primary closure or partial closure with resorbable sutures.
Nerve Injury Risk
IAN injury: Risk 0.5-5%. High-risk radiographic signs: darkening of the root at the canal, deflection/narrowing of the canal, diversion of the canal, interruption of the white line of the canal. CBCT is recommended when the panoramic radiograph shows high-risk signs. If the root is intimately associated with the IAN canal, consider coronectomy (intentional partial odontectomy) — the crown is sectioned and removed, leaving the roots undisturbed in the socket. Contraindications to coronectomy: active infection at the root, horizontal impaction with roots above the canal, mobile tooth fragment.
Lingual nerve injury: Risk 0.2-2% (temporary), 0.1-0.5% (permanent). Occurs from lingual flap retraction, lingual plate perforation, or distal extension of the incision into the lingual tissues. Avoidance: stay buccal with bone removal, avoid lingual flap retraction with Howarth elevator (controversial), careful incision design.
21 Dental Implants & Osseointegration
Osseointegration Principles
Per-Ingvar Brånemark (1969) defined osseointegration as a direct structural and functional connection between ordered, living bone and the surface of a load-carrying implant. Modern dental implants are made of commercially pure titanium (cpTi, grade 4) or titanium alloy (Ti-6Al-4V). The implant surface is typically textured (SLA — sandblasted, large-grit, acid-etched; or TiUnite — anodized) to increase surface area and promote osteoblast adhesion.
Standard implant dimensions: Diameter: 3.5-5.0 mm (standard: 3.75-4.0 mm); Length: 6-16 mm (standard: 10-13 mm). A minimum of 1 mm of bone around the implant is recommended in all dimensions. Key distances: implant to adjacent tooth — at least 1.5 mm; implant to implant — at least 3 mm. These distances preserve the interimplant bone crest and papilla.
Surgical Protocol
Two-stage protocol (Brånemark): Stage 1: implant placed and submerged beneath the mucosa for 3-6 months (3 months mandible, 6 months maxilla — due to softer bone). Stage 2: re-entry to expose the implant and place a healing abutment. One-stage protocol: Implant placed and healing abutment connected at the same appointment — transgingival healing. Equivalent success rates to two-stage. Immediate loading: Prosthesis attached within 48 hours of implant placement. Requires primary stability (≥ 35 Ncm insertion torque, ISQ ≥ 65), good bone quality, and controlled occlusal loading. All-on-4 concept (tilted posterior implants for full-arch rehabilitation) uses immediate loading.
Immediate vs. delayed placement: Immediate placement (at time of extraction) reduces treatment time and number of surgeries but requires intact buccal bone and adequate apical bone beyond the socket for primary stability. Delayed placement (3-6 months after extraction) allows socket healing and is more predictable when bone defects are present. Early placement (4-8 weeks post-extraction) is a compromise — soft tissue healed but bone not fully remodeled.
Bone Augmentation Procedures
Sinus lift (maxillary sinus augmentation): The most common bone augmentation procedure in the posterior maxilla, where pneumatization of the maxillary sinus reduces available bone height.
- Lateral window approach (Caldwell-Luc): A bony window is created in the lateral maxillary wall; the Schneiderian membrane is elevated from the sinus floor; bone graft material is packed between the membrane and the sinus floor. Indicated when residual bone height is < 5 mm. Implant placement simultaneously (if ≥ 5 mm residual bone for primary stability) or delayed (4-9 months).
- Crestal approach (osteotome/transcrestal): Bone graft is pushed into the sinus through the osteotomy site using osteotomes. Less invasive but limited to 1-3 mm of elevation. Indicated when residual bone is ≥ 6-8 mm. Complications: Schneiderian membrane perforation (10-30%), BPPV (benign paroxysmal positional vertigo — from osteotome tapping).
Implant Complications
Early failure (before loading): failure of osseointegration due to overheating during osteotomy (drill speed > 1500 rpm without irrigation raises bone temperature above the critical threshold of 47°C for 1 minute), infection, premature loading, systemic factors (uncontrolled diabetes, smoking, radiation). Late failure (after loading): peri-implantitis (inflammatory bone loss around a functioning implant — incidence 10-20% at 5-10 years; treatment: debridement, decontamination, bone grafting for infrabony defects; implant removal for advanced cases), mechanical failure (screw loosening, abutment fracture, implant fracture — risk with narrow-diameter implants under high occlusal loads), and prosthetic complications. Contraindications: Active chemotherapy/radiation to the jaws within the past 12 months (relative), IV bisphosphonates for cancer (relative — higher MRONJ risk), uncontrolled diabetes (HbA1c > 8%), severe immune compromise, inadequate bone without augmentation, active periodontal disease (must be treated first), smoking (> 10 cigarettes/day doubles implant failure rate). PMID: 17138169
Guided bone regeneration (GBR): Barrier membrane (resorbable — collagen, or non-resorbable — ePTFE/titanium-reinforced) placed over a bone graft to exclude soft tissue cells and allow osteoblast proliferation. The membrane maintains space and prevents connective tissue ingrowth. Based on the PASS principle: Primary closure, Angiogenesis, Space maintenance, Stability of the graft. PMID: 22288944
22 Bone Grafting & Augmentation
Graft Types & Properties
| Graft Type | Source | Properties | Examples |
|---|---|---|---|
| Autogenous (autograft) | Patient's own bone | Gold standard — only graft with all three properties: osteogenesis (living cells), osteoinduction (BMPs), osteoconduction (scaffold). Disadvantage: donor site morbidity, limited quantity | Mandibular ramus, chin/symphysis (intraoral); iliac crest (largest volume), calvarium (membranous — least resorption), tibia (cancellous) |
| Allograft | Human cadaver bone | Osteoconductive + weakly osteoinductive; no living cells (osteogenic); processed to eliminate immunogenicity | FDBA (freeze-dried bone allograft), DFDBA (demineralized FDBA — exposes BMPs, more osteoinductive) |
| Xenograft | Animal bone (usually bovine) | Osteoconductive only; very slow resorption (maintains volume well); anorganic (deproteinized) — minimal immune response | Bio-Oss (deproteinized bovine bone mineral — most studied xenograft in dental literature) |
| Alloplast (synthetic) | Laboratory-made | Osteoconductive only; unlimited supply; no disease transmission risk | Hydroxyapatite (HA), beta-tricalcium phosphate (β-TCP), biphasic calcium phosphate (BCP), bioactive glass |
Growth Factors
BMP-2 (rhBMP-2, Infuse): Recombinant human bone morphogenetic protein-2. Potent osteoinductive agent. FDA-approved for sinus augmentation and localized alveolar ridge augmentation. Applied on an absorbable collagen sponge carrier. Risk: significant swelling (especially in anterior mandible), potential for exuberant bone formation, cost. Off-label use for mandibular continuity defects has been reported. PMID: 17513840
PRF (Platelet-Rich Fibrin): Autologous concentrate prepared from the patient's blood without anticoagulants. Blood is drawn and centrifuged — the fibrin clot contains platelets, leukocytes, and growth factors (PDGF, TGF-β, VEGF). Used as a membrane, mixed with graft material, or placed in extraction sockets to promote soft tissue healing and early bone formation. Simple, low-cost, no regulatory concerns.
PRP (Platelet-Rich Plasma): Similar concept to PRF but uses anticoagulants and a two-spin centrifugation protocol. The platelet concentrate is activated with calcium chloride and thrombin. Contains growth factors but lacks the fibrin scaffold of PRF. Less commonly used in OMS currently compared to PRF.
Principles of Bone Healing
Primary (direct) bone healing: Occurs when bone fragments are rigidly fixed with anatomic reduction and no gap (< 0.1 mm). Osteonal remodeling (cutting cones of osteoclasts followed by osteoblasts) crosses the fracture line directly — no callus formation. Requires rigid internal fixation (lag screws, compression plates). This is the goal of ORIF in mandibular fractures.
Secondary (indirect) bone healing: Occurs with some motion at the fracture site (semi-rigid fixation, closed reduction with MMF). Follows the sequence: hematoma formation → inflammatory phase (1-7 days) → soft callus (cartilage — weeks 1-3) → hard callus (woven bone — weeks 3-12) → remodeling (months to years). Micromotion (up to 150 μm) actually stimulates callus formation. Excessive motion leads to fibrous nonunion.
Wolff's Law: Bone adapts to the loads placed upon it. Stress-shielding from excessively rigid plates can cause local osteopenia. This is one rationale for using titanium miniplates (which are more flexible and allow functional loading) rather than rigid reconstruction plates for simple fractures.
Osteomyelitis of the Jaws
Acute osteomyelitis: Suppurative infection of bone, most commonly from odontogenic infection. Mandible far more common than maxilla (poorer blood supply — single inferior alveolar artery vs. rich maxillary collateral supply). Presentation: deep, boring pain; swelling; fever; purulent drainage; loose teeth; paresthesia of the IAN (Vincent's symptom); elevated WBC, ESR, and CRP. CT shows early cortical erosion and periosteal reaction. Treatment: IV antibiotics (ampicillin-sulbactam or clindamycin), surgical drainage, debridement of necrotic bone, extraction of involved teeth. Culture and sensitivity to guide antibiotic therapy.
Chronic osteomyelitis: Results from inadequately treated acute osteomyelitis or low-grade chronic infection. Presents with persistent pain, draining fistulae, sequestrum formation (necrotic bone separated from viable bone by a lucent zone), involucrum (new periosteal bone surrounding sequestrum). CT/CBCT shows mixed radiolucent-radiopaque pattern, sequestra, and involucrum. Treatment: sequestrectomy (remove necrotic bone), saucerization (decortication of the involved area), prolonged antibiotics (4-6 weeks IV, then PO), hyperbaric oxygen therapy (adjunctive — increases oxygen delivery to ischemic bone, stimulates angiogenesis and osteogenesis). Resection for refractory cases.
Osteoradionecrosis (ORN): Chronic non-healing exposed bone in an irradiated field (≥ 50 Gy to the jaws). Pathophysiology: radiation causes hypovascular, hypocellular, and hypoxic tissue (Marx theory of the "3 H's"). Unlike MRONJ, ORN has no drug association — it results from prior radiation therapy for head and neck cancer. Prevention: complete all dental extractions at least 14-21 days before radiation begins; maintain meticulous oral hygiene; fluoride trays for life (to prevent radiation caries). Treatment by severity: conservative (debridement, antibiotics, HBO) for early disease; resection with free flap reconstruction for advanced ORN (Marx Stage III — pathologic fracture, orocutaneous fistula). Hyperbaric oxygen (HBO): Marx protocol — 20 dives pre-extraction, 10 dives post-extraction at 2.4 atm for 90 min each for patients requiring extractions in previously irradiated fields. The HOPON trial raised questions about HBO efficacy, but it remains standard of care at many centers. PMID: 15592106
23 Preprosthetic Surgery & TMJ Surgery
Preprosthetic Surgery
Alveolar ridge augmentation: See bone grafting section. Onlay block grafts (from ramus or chin), GBR with particulate graft, or ridge split technique with expansion and graft for horizontally deficient ridges. Vertical augmentation is more challenging — options include distraction osteogenesis, tent-pole grafting, and titanium mesh with particulate graft.
Vestibuloplasty: Deepening of the vestibular sulcus to increase denture-bearing surface area. Techniques: Clark's technique (supraperiosteal dissection), Kazanjian's technique (transposition of the mucosa), or free mucosal/skin grafts. Less commonly performed since the advent of dental implants.
Torus removal: Torus palatinus (midline hard palate) and torus mandibularis (lingual mandible, bilateral in premolar region) are benign bony exostoses. Removal indicated when they interfere with denture fabrication/seating, cause recurrent ulceration, or require the bone for grafting. Technique: mucoperiosteal flap, bur or chisel to remove the torus, smooth edges, primary closure.
Frenectomy: Excision of an abnormally positioned frenum. Labial frenectomy — for maxillary midline diastema in children (after orthodontic closure), or for denture interference. Lingual frenectomy — for ankyloglossia (tongue-tie, Kotlow classification). Technique: diamond excision, Z-plasty, or laser.
Odontogenic Space Infections
Deep space infections of the head and neck are potentially life-threatening and require aggressive management. The fascial spaces commonly involved in odontogenic infections include:
Ludwig's Angina: Emergency Bilateral infection of the submandibular, sublingual, and submental spaces, typically arising from infected lower second or third molars (roots extend below the mylohyoid line). Hallmarks: bilateral submandibular swelling ("bull neck"), floor of mouth elevation, trismus, dysphagia, and airway compromise. The tongue is pushed posterosuperiorly, causing potential airway obstruction. Management: (1) Secure the airway immediately — fiberoptic nasal intubation or awake tracheostomy if intubation fails; (2) IV antibiotics: ampicillin-sulbactam 3 g Q6H or penicillin G + metronidazole; (3) CT with contrast to identify abscess vs cellulitis; (4) Surgical drainage of involved spaces via submandibular approach; (5) Source control — extract the offending tooth. Mortality was > 50% in the pre-antibiotic era; now < 5% with aggressive management, but airway loss remains the leading cause of death.
Parapharyngeal space infection: Danger — proximity to the carotid sheath (internal carotid artery, internal jugular vein, vagus nerve). Complications: carotid artery erosion, internal jugular vein thrombophlebitis (Lemierre syndrome — Fusobacterium necrophorum), cranial nerve deficits. Retropharyngeal space infection: Danger — can track into the mediastinum (danger space — extends from skull base to diaphragm). Descending necrotizing mediastinitis has a mortality rate of 25-40%. Signs: neck stiffness, dysphagia, stridor, widened retropharyngeal space on lateral neck radiograph (> 7 mm at C2, > 22 mm at C6).
TMJ Surgery
TMJ surgery is indicated when nonsurgical management (splint therapy, physical therapy, medications, behavioral modification) has failed for 3-6 months. The procedures form a stepwise escalation:
| Procedure | Indication | Technique | Success Rate |
|---|---|---|---|
| Arthrocentesis | Closed lock (disc displacement without reduction) < 6 months, adhesions, acute inflammatory conditions | Two needles inserted into the superior joint space (one for inflow, one for outflow); lavage with 200-300 mL lactated Ringer's under pressure; lyses adhesions, washes out inflammatory mediators; can inject HA or corticosteroid at the end | 70-80% improvement in pain and mouth opening |
| Arthroscopy | Disc displacement with or without reduction, adhesions, synovitis; failed arthrocentesis | Cannula with camera inserted into the superior joint space; allows visualization, lysis of adhesions, disc repositioning, synovectomy; lavage with direct visualization | 80-90% improvement; lower morbidity than open surgery |
| Open disc repositioning (Disc plication) | Disc displacement without reduction, failed arthroscopy, disc with potential for salvage | Preauricular or endaural approach; disc repositioned posteriorly and sutured to the posterior capsule/retrodiscal tissue; may use a Mitek anchor | 70-80%; redisplacement is possible |
| Discectomy | Severely degenerated, perforated, or nonsalvageable disc | Preauricular approach; removal of the disc; may place a temporalis fascia or dermal graft as interpositional material | 70-90% pain relief; concern for long-term degenerative changes (though studies show favorable outcomes at 20+ years) |
| Modified condylotomy | Disc displacement; closed lock; avoids direct TMJ surgery | Vertical subsigmoid osteotomy of the mandibular ramus — allows the condyle to sag inferiorly and anteriorly, indirectly recapturing the disc. Fixated with MMF for 2-4 weeks | 80-90% for disc displacement; alternative to arthrotomy |
| Total joint replacement (TJR) | End-stage TMJ disease: ankylosis, failed previous surgery, severe degenerative joint disease, avascular necrosis of condyle, tumor reconstruction | Stock (TMJ Concepts, Biomet/Zimmer) or custom (patient-specific) prosthesis; fossa component and condylar/ramus component. Requires coronoidectomy and aggressive heterotopic bone removal in ankylosis cases. | 85-95% improvement; long-term data improving; reoperation rate 5-15% |
Wilkes Classification of TMJ Internal Derangement
| Stage | Clinical | Imaging | Surgical Findings |
|---|---|---|---|
| I (Early) | Painless clicking; no restriction of motion | Slight anterior disc displacement with reduction; normal disc morphology | Normal disc, slight displacement |
| II (Early-Intermediate) | Occasional painful clicking; intermittent locking; headaches | Anterior disc displacement with reduction; early disc deformity (thickening of posterior band) | Anterior disc displacement; early disc deformity |
| III (Intermediate) | Frequent pain; joint tenderness; locking; restricted motion; altered function | Anterior disc displacement with or without reduction; disc deformity | Marked disc displacement and deformity; no hard tissue changes |
| IV (Intermediate-Late) | Chronic pain; restricted motion; crepitus | Disc displacement without reduction; degenerative bony changes (flattening, sclerosis, osteophytes on CT) | Adhesions; disc perforation may be present; early bony changes |
| V (Late) | Crepitus; variable pain; chronic restriction | Disc perforation or gross deformity; severe degenerative joint disease; subchondral cyst, osteophytes, flattened condyle | Disc perforation; gross bony degeneration; fibrillation of articular surfaces |
24 Classification Systems
Fracture Classifications
| Classification | Application | Categories |
|---|---|---|
| Le Fort I/II/III | Midface fractures | I: horizontal (floating palate); II: pyramidal (floating maxilla); III: craniofacial dysjunction (floating face). All involve pterygoid plates. |
| Zingg (ZMC) | Zygomatic complex fractures | A: incomplete (A1 arch, A2 lateral wall, A3 infraorbital rim); B: complete monofragment; C: comminuted multifragment |
| Markowitz (NOE) | Naso-orbito-ethmoid fractures | I: single fragment, MCT attached; II: comminuted, MCT on bone fragment; III: comminuted, MCT avulsed from bone (requires transnasal canthopexy) |
| Pell-Gregory | Third molar impaction depth & ramus relationship | Depth: A/B/C; Ramus: Class I/II/III |
| Winter | Third molar angulation | Mesioangular, horizontal, vertical, distoangular, buccoangular, linguoangular, inverted |
| Ellis (Tooth Fracture) | Crown fractures | I: enamel only; II: enamel + dentin; III: enamel + dentin + pulp exposure |
| Wilkes (TMJ) | TMJ internal derangement staging | I-V: early to late (disc displacement to degenerative disease) |
| AAOMS MRONJ | Medication-related osteonecrosis of the jaw | At Risk, Stage 0-3 (no exposed bone to pathologic fracture/fistula) |
| AJCC 8th Ed (Oral Cavity) | Oral SCC staging | T1-T4 incorporates depth of invasion (DOI); N staging includes ENE (extranodal extension) |
| Angle (Occlusion) | Molar relationship / Skeletal classification | Class I: normal AP; Class II: mandible posterior (Div 1/2); Class III: mandible anterior |
Panfacial Fractures — Sequencing of Repair
Panfacial fractures involve the upper face (frontal sinus/supraorbital), midface (Le Fort, ZMC, NOE, orbit), and lower face (mandible) simultaneously. Sequencing is critical because each repaired structure serves as a reference for the next. Two major philosophies:
Bottom-up, inside-out approach (most widely taught): (1) Mandible ORIF to restore the lower facial width and projection (start with condyles/ramus to establish posterior facial height, then body/symphysis); (2) Establish occlusion with MMF; (3) Reconstruct the palate and maxillary buttresses (Le Fort repair — use palatal splint if palatal split); (4) ZMC and zygomatic arches (establishes facial width and midface projection); (5) NOE complex; (6) Orbital floor/walls; (7) Frontal sinus and supraorbital rim (last — high-to-low). This approach uses known landmarks sequentially — the mandible establishes the foundation, occlusion guides midface position.
Top-down approach (alternative): Start with stable cranial structures and work inferiorly. Used when the frontal bone and cranial base are intact and reliable reference points. (1) Frontal bar/supraorbital rims; (2) ZMC and zygomatic arches; (3) NOE; (4) Le Fort-level fractures; (5) Mandible and occlusion last.
Other Important Classifications
| Classification | Application | Categories |
|---|---|---|
| Pruzansky-Kaban | Hemifacial microsomia (mandibular hypoplasia) | I: small but normal condyle/ramus; IIA: condyle/ramus present but abnormal; IIB: condyle absent, ramus rudimentary; III: complete absence of ramus, condyle, and glenoid fossa |
| House-Brackmann | Facial nerve function | I: normal; II: slight weakness; III: moderate dysfunction; IV: moderately severe; V: severe (barely perceptible motion); VI: total paralysis |
| Sunderland | Nerve injury severity | I: neuropraxia (conduction block); II: axonotmesis (axon disrupted, endoneurium intact); III: endoneurium disrupted; IV: perineurium disrupted; V: neurotmesis (complete transection) |
| Misch Bone Density | Bone quality for implant placement | D1: dense cortical (anterior mandible); D2: thick cortical + coarse trabecular (posterior mandible, anterior maxilla); D3: thin cortical + fine trabecular (posterior maxilla); D4: fine trabecular, minimal cortical (posterior maxilla — worst for implants) |
| Samuels & Chuong (Mandible Fracture) | Mandible fracture favorability | Fractures are favorable when muscle pull compresses (reduces) the fracture, unfavorable when muscle pull distracts (displaces) fragments. Assessed in both horizontal and vertical planes. |
| Kotlow (Ankyloglossia) | Severity of tongue-tie | Class I: mild (12-16 mm free tongue); Class II: moderate (8-11 mm); Class III: severe (3-7 mm); Class IV: complete (< 3 mm free tongue) |
Plate & Screw Specifications in OMS
| Application | Plate Size | Screw Type | Screw Length | Key Notes |
|---|---|---|---|---|
| Mandible — miniplate (angle, body) | 2.0 mm | Monocortical | 6-8 mm | Champy technique; 4 screws minimum (2 per side of fracture) |
| Mandible — reconstruction plate | 2.4-2.7 mm | Bicortical, locking | 12-18 mm | Load-bearing; minimum 3 screws per side; for comminuted fractures, defects, atrophic mandible |
| Mandible — lag screw (symphysis) | N/A (screw only) | Bicortical positional | 24-30 mm | Provides interfragmentary compression; glide hole in near cortex, thread hole in far cortex |
| Midface — miniplate | 1.5-2.0 mm | Monocortical | 4-6 mm | For Le Fort buttresses, ZMC, piriform rim |
| Orbital rim — microplate | 1.0-1.3 mm | Monocortical | 3-5 mm | Low profile; infraorbital rim, frontozygomatic suture |
| Orthognathic — Le Fort I | L-shaped or straight 2.0 mm | Monocortical | 6-8 mm | 4 plates (bilateral piriform + bilateral ZM buttress) |
| Orthognathic — BSSO | Plate or positional screws | Bicortical positional screws (2.0 mm × 3) or plate with monocortical screws | Screws: 16-18 mm | Positional screws placed at 60° angle through buccal cortex |
25 Medications Master Table
Antibiotics
| Drug | Dose (Adult) | Spectrum/Indication | Notes |
|---|---|---|---|
| Amoxicillin | 500 mg PO TID or 875 mg PO BID | First-line for odontogenic infections; covers oral streptococci and anaerobes | Most common OMS antibiotic; add clavulanate (Augmentin 875/125 BID) if resistant organisms suspected |
| Penicillin V | 500 mg PO QID | Odontogenic infections — narrow spectrum | Less commonly prescribed now due to QID dosing; still effective for simple infections |
| Clindamycin | 300-450 mg PO TID-QID | Penicillin-allergic patients; excellent bone penetration; anaerobic coverage | Risk of C. difficile colitis (0.5-2%); excellent for osteomyelitis |
| Metronidazole | 500 mg PO TID | Anaerobic coverage; often combined with penicillin for severe odontogenic space infections | Disulfiram-like reaction with alcohol; metallic taste |
| Ampicillin-Sulbactam (Unasyn) | 3 g IV Q6H | Inpatient treatment of severe odontogenic space infections (Ludwig's angina, parapharyngeal/retropharyngeal abscess) | Covers oral flora including β-lactamase producers |
| Ceftriaxone | 1-2 g IV Q24H | Hospital-acquired or resistant infections | Good bone penetration; broad gram-negative coverage |
Analgesics & Anti-Inflammatories
| Drug | Dose | Notes |
|---|---|---|
| Ibuprofen | 400-600 mg PO Q6H | First-line analgesic for dental/surgical pain; 400 mg ibuprofen = superior to 1000 mg acetaminophen for dental pain; max 3200 mg/day |
| Acetaminophen | 500-1000 mg PO Q6H | Combined with ibuprofen (alternating) provides analgesia comparable to opioids; max 4 g/day (2 g/day for hepatic disease) |
| Ketorolac | 15-30 mg IV/IM Q6H (max 5 days) | Potent NSAID; useful for acute postoperative pain; renal and GI precautions |
| Dexamethasone | 8-10 mg IV/IM preop or 4 mg PO BID × 3 days | Reduces postoperative edema significantly (40-60%); standard for orthognathic surgery and third molar extractions; antiemetic |
| Hydrocodone/Acetaminophen | 5/325 mg, 1-2 tabs PO Q4-6H PRN | Reserve for moderate-severe pain uncontrolled by NSAID/acetaminophen combination; limit quantity to 12-20 tablets for routine extractions |
Local Anesthetics
| Agent | Concentration | Vasoconstrictor | Max Dose (with epi) | Onset/Duration |
|---|---|---|---|---|
| Lidocaine | 2% | 1:100,000 epi | 7 mg/kg (500 mg absolute max) | Onset: 2-3 min; Duration: 60-90 min (pulpal), 3-5 hr (soft tissue) |
| Articaine | 4% | 1:100,000 or 1:200,000 epi | 7 mg/kg | Onset: 1-2 min; Duration: 60-75 min (pulpal); best buccal infiltration efficacy |
| Mepivacaine 3% | 3% | None | 6.6 mg/kg (400 mg absolute max) | Onset: 1.5-2 min; Duration: 20-40 min (pulpal); useful when vasoconstrictor is contraindicated |
| Bupivacaine | 0.5% | 1:200,000 epi | 1.3 mg/kg (90 mg absolute max) | Onset: 6-10 min; Duration: 4-9 hr (soft tissue); ideal for postoperative analgesia |
Bisphosphonates & Antiresorptives
| Drug | Route/Dose | Indication | MRONJ Risk |
|---|---|---|---|
| Alendronate (Fosamax) | 70 mg PO weekly | Osteoporosis | Low (0.01-0.1%); increases with duration > 4 years |
| Zoledronic acid (Zometa) | 4 mg IV Q3-4 weeks (cancer); 5 mg IV yearly (osteoporosis) | Bone metastases; osteoporosis | Highest among bisphosphonates (cancer dose: 1-15%) |
| Denosumab (Prolia/Xgeva) | 60 mg SC Q6 months (osteoporosis); 120 mg SC Q4 weeks (cancer) | Osteoporosis; bone metastases | Similar to IV bisphosphonates in cancer patients; effect reversible (6-month duration) |
Corticosteroids in OMS
| Drug | Dose | Indication | Protocol |
|---|---|---|---|
| Dexamethasone | 8-10 mg IV/IM intraop | Third molar surgery, orthognathic surgery, facial fracture ORIF | Single dose preop or intraop; may continue 4 mg PO BID for 2-3 days postop for major surgery |
| Methylprednisolone (Medrol Dosepak) | Taper over 6 days (24 mg → 4 mg) | TMJ inflammatory conditions, moderate postoperative edema | Start day of surgery; taper as directed |
Corticosteroid benefits in OMS: reduces postoperative edema by 40-60%, decreases trismus, decreases postoperative nausea (dexamethasone is an effective antiemetic), and may reduce pain. Risks with short-term use are minimal — adrenal suppression does not occur with courses < 7 days. Caution in diabetic patients (monitor blood glucose — transient hyperglycemia expected).
Anticoagulant Management for OMS Procedures
General principle: Most dental extractions and minor OMS procedures can be performed safely without discontinuing anticoagulants — the risk of thromboembolism from stopping anticoagulation outweighs the risk of postoperative bleeding, which can be controlled with local measures (sutures, oxidized cellulose, tranexamic acid rinse).
- Warfarin: Proceed with surgery if INR ≤ 3.5 for simple extractions; INR ≤ 3.0 for surgical extractions. Use local hemostatic agents. For major OMS procedures (orthognathic surgery, tumor resection), coordinate with the prescribing physician for bridging therapy.
- DOACs (apixaban, rivarelbaan, dabigatran): For minor procedures, hold the morning dose. For major procedures, hold for 24-48 hours (coordinate with cardiologist/hematologist). No reliable reversal for all agents (idarucizumab for dabigatran; andexanet alfa for anti-Xa agents).
- Antiplatelet agents (aspirin, clopidogrel): Generally do NOT discontinue for dental extractions. For major surgery, discontinue clopidogrel 5-7 days prior if cardiologist approves.
26 Imaging & Diagnostics
| Modality | Indications in OMS | Advantages | Limitations |
|---|---|---|---|
| Panoramic Radiograph (OPG) | Screening for impacted teeth, mandibular fractures, cysts/tumors, TMJ condylar morphology; preoperative planning for third molar surgery | Single image of entire mandible, maxilla, teeth, TMJs, and sinuses; low dose (~0.01 mSv); widely available | 2D projection — overlap and magnification artifacts; poor visualization of symphysis/parasymphysis (superimposition of spine); not reliable for orbital or midface fractures |
| CBCT (Cone Beam CT) | Implant planning, third molar-IAN relationship, TMJ osseous evaluation, small jaw lesions, orthodontic/orthognathic planning | 3D imaging with lower dose than conventional CT (0.05-0.5 mSv); excellent for hard tissue; high spatial resolution; small field of view options | Poor soft tissue contrast (cannot evaluate muscles, lymph nodes, or soft tissue tumors); motion artifact; not suitable for malignancy staging |
| CT Facial Bones | Facial trauma (midface, orbit, mandible), tumor staging, complex fracture planning, preoperative virtual surgical planning (VSP) | Excellent bony detail; multiplanar and 3D reconstruction; widely available; rapid acquisition; superior to CBCT for facial trauma evaluation | Higher dose than CBCT (0.3-1.5 mSv); cost; less available than panoramic; dental artifact from metallic restorations |
| MRI (TMJ Protocol) | TMJ disc position and morphology, TMJ effusion, soft tissue tumors, salivary gland tumors, perineural tumor spread | Gold standard for TMJ disc imaging; excellent soft tissue contrast; no ionizing radiation; can assess disc displacement with and without reduction (open/closed mouth views) | Cost; long acquisition time; not useful for bony fractures; contraindicated with certain implants; claustrophobia |
| Sialography | Salivary duct obstruction, strictures, Sjögren syndrome (sialectasia pattern) | Dynamic imaging of duct anatomy; can be combined with CT (CT-sialography) for enhanced visualization | Invasive (contrast injection into the duct); contraindicated in acute sialadenitis; largely replaced by ultrasound and MR-sialography |
| Ultrasound | Superficial masses (salivary gland tumors, lymph nodes), abscess vs cellulitis, sialolithiasis, guided FNA biopsy | No radiation; real-time; portable; inexpensive; excellent for superficial structures; can guide biopsy | Operator dependent; limited for deep structures; poor for bony evaluation |
| PET-CT (FDG) | Staging of oral SCC (advanced disease), detection of distant metastases, evaluation for recurrence, unknown primary with cervical metastasis | Detects metabolically active tumor; whole-body assessment for distant metastases; combined anatomic and functional information | False positives from inflammation/infection; poor for small primary tumors < 1 cm; cost; radiation exposure |
| CT Angiography (CTA) | Preoperative vascular mapping for free flap surgery (Allen test equivalent for fibula — CT angiography of lower extremities), evaluation of vascular injury in penetrating facial trauma | Defines vascular anatomy; identifies anomalous peroneal artery (absent in 5-8%, which contraindicates ipsilateral fibula harvest); rapid acquisition | Contrast allergy risk; renal considerations; radiation |
Diagnostic Algorithm for Jaw Lesions
When a radiolucent lesion is discovered in the jaws on imaging, the systematic approach includes:
- Step 1 — Location: Is it odontogenic (related to a tooth — at the apex = periapical/radicular cyst; at the crown of an unerupted tooth = dentigerous cyst or ameloblastoma; between roots = lateral periodontal cyst) or non-odontogenic (in bone not associated with teeth)?
- Step 2 — Radiographic features: Unilocular vs multilocular (multilocular suggests ameloblastoma, OKC, myxoma, central giant cell granuloma); well-defined vs ill-defined borders (ill-defined suggests malignancy or osteomyelitis); effect on adjacent teeth (root resorption, tooth displacement).
- Step 3 — Tooth vitality: Adjacent teeth vital = non-inflammatory (likely developmental or neoplastic); nonvital = likely inflammatory (radicular cyst, periapical granuloma).
- Step 4 — Patient demographics: Age, sex, and race affect differential diagnosis. Ameloblastoma peak: 30-40 years. OKC peak: 20-30 years. Central giant cell granuloma: young females < 30 years. Osteosarcoma: bimodal — 10-20 years and > 60 years.
- Step 5 — Biopsy: Aspiration first (straw-colored fluid = cyst; blood = vascular lesion or AVM — proceed with caution; nothing or white cheesy material = OKC). Then incisional or excisional biopsy as appropriate.
27 Abbreviations Master List
| Abbreviation | Full Term |
|---|---|
| OMS | Oral and Maxillofacial Surgery |
| TMJ | Temporomandibular Joint |
| TMD | Temporomandibular Disorder |
| IAN | Inferior Alveolar Nerve |
| IMA | Internal Maxillary Artery |
| ZMC | Zygomaticomaxillary Complex |
| NOE | Naso-Orbito-Ethmoid |
| MCT | Medial Canthal Tendon |
| ICD | Intercanthal Distance |
| MMF | Maxillomandibular Fixation |
| ORIF | Open Reduction Internal Fixation |
| BSSO | Bilateral Sagittal Split Osteotomy |
| MMA | Maxillomandibular Advancement |
| MIO | Maximum Interincisal Opening |
| NFOT | Nasofrontal Outflow Tract |
| CSF | Cerebrospinal Fluid |
| CBCT | Cone Beam Computed Tomography |
| OPG | Orthopantomogram (Panoramic Radiograph) |
| FDI | Fédération Dentaire Internationale |
| PDL | Periodontal Ligament |
| CEJ | Cementoenamel Junction |
| OKC | Odontogenic Keratocyst |
| SCC | Squamous Cell Carcinoma |
| DOI | Depth of Invasion |
| ENE | Extranodal Extension |
| MRONJ | Medication-Related Osteonecrosis of the Jaw |
| AAOMS | American Association of Oral and Maxillofacial Surgeons |
| GBR | Guided Bone Regeneration |
| BMP | Bone Morphogenetic Protein |
| PRF | Platelet-Rich Fibrin |
| PRP | Platelet-Rich Plasma |
| SLA | Sandblasted, Large-grit, Acid-etched |
| FDBA | Freeze-Dried Bone Allograft |
| DFDBA | Demineralized Freeze-Dried Bone Allograft |
| OSA | Obstructive Sleep Apnea |
| AHI | Apnea-Hypopnea Index |
| PAS | Posterior Airway Space |
| UPPP | Uvulopalatopharyngoplasty |
| SNA/SNB/ANB | Sella-Nasion-A point / Sella-Nasion-B point / A point-Nasion-B point angles |
| VSP | Virtual Surgical Planning |
| FNA | Fine Needle Aspiration |
| RFFF | Radial Forearm Free Flap |
| ALT | Anterolateral Thigh (Flap) |
| TJR | Total Joint Replacement |
| PSA/MSA/ASA | Posterior/Middle/Anterior Superior Alveolar (nerve/block) |
| HBSS | Hank's Balanced Salt Solution |
| ORN | Osteoradionecrosis |
| HBO | Hyperbaric Oxygen Therapy |
| OAC | Oroantral Communication |
| OPG | Orthopantomogram (Panoramic Radiograph) |
| GA | Genioglossus Advancement |
| DO | Distraction Osteogenesis |
| RED | Rigid External Distraction (device) |
| IOP | Intraocular Pressure |
| RAPD | Relative Afferent Pupillary Defect |
28 Key Guidelines & Landmark Studies
| Study/Guideline | Year | Key Finding/Recommendation | Reference |
|---|---|---|---|
| Champy et al. — Ideal Lines of Osteosynthesis | 1978 | Defined the zones of tension and compression in the mandible; established monocortical miniplate fixation along the external oblique ridge for angle fractures as biomechanically sufficient | PMID: 356722 |
| Brånemark — Osseointegration | 1969-1977 | Established the concept of direct bone-to-titanium integration; pioneered the two-stage dental implant protocol; demonstrated long-term success of endosseous implants | PMID: 331479 |
| AAOMS Position Paper on MRONJ | 2014 (updated 2022) | Defined MRONJ diagnostic criteria, staging system (0-3), and management recommendations including prevention protocols for patients initiating antiresorptive therapy | PMID: 25234529 |
| AAOMS Parameters for Third Molar Management | 2017 | Evidence-based guidelines for management of impacted third molars; supported prophylactic removal before pathology develops in appropriate patients; addressed coronectomy indications | PMID: 27919600 |
| McCarthy — Mandibular Distraction Osteogenesis | 1992 | First application of distraction osteogenesis to the human mandible; demonstrated feasibility of gradual bone lengthening in the craniofacial skeleton | PMID: 1597162 |
| Obwegeser-Dal Pont — Sagittal Split Osteotomy | 1955-1961 | Developed the sagittal split osteotomy of the mandibular ramus — became the foundation for modern mandibular orthognathic surgery (BSSO) | PMID: 13436795 |
| AJCC 8th Edition — Oral Cavity Staging | 2017 | Incorporated depth of invasion (DOI) into T-staging for oral cavity SCC and extranodal extension (ENE) into N-staging; improved prognostic accuracy over 7th edition | PMID: 28125762 |
| D'Eramo (OMS Anesthesia Safety) | 1999-2003 | Massachusetts Board of Registration data: OMS office anesthesia mortality rate of 1:365,000 — comparable to hospital-based anesthesia; established OMS anesthesia team model as safe | PMID: 12538055 |
| Holte & Bhatt — MMA for OSA Meta-Analysis | 2019 | MMA demonstrated 85.5% surgical success rate (AHI < 20 + ≥ 50% reduction) for moderate-to-severe OSA; mean AHI reduction from 49 to 9 | PMID: 20456774 |
| Le Fort — Cadaver Fracture Studies | 1901 | René Le Fort described three reproducible patterns of midface fractures (Le Fort I/II/III) based on cadaveric impact studies — remains the classification standard over a century later | Le Fort R. Etude expérimentale sur les fractures de la mâchoire supérieure. Rev Chir Paris. 1901. |
| SEND Trial — Elective Neck Dissection for Oral SCC | 2015 | Multicenter RCT showing that elective neck dissection for early (T1-T2 N0) oral SCC improved overall survival and disease-free survival compared to therapeutic neck dissection at recurrence (80% vs 67.5% OS at 3 years) | PMID: 26286723 |
| Ellis — Condylar Fracture Treatment | 2000-2015 | Series of studies demonstrating that ORIF of subcondylar fractures produces better functional outcomes (mouth opening, occlusion, protrusive movement) than closed treatment with comparable complication rates | PMID: 23981784 |
| Tatum — Maxillary Sinus Augmentation | 1977-1986 | Described the lateral window approach for maxillary sinus floor augmentation to enable dental implant placement in the atrophic posterior maxilla — remains the standard technique | PMID: 3514733 |
| Marx — Osteoradionecrosis & HBO Protocol | 1983 | Established the pathophysiology of ORN as a hypovascular-hypocellular-hypoxic tissue problem (not radiation osteomyelitis); proposed and validated the HBO protocol (20 dives pre-procedure, 10 post) for dental extractions in irradiated patients | PMID: 15592106 |
| EORTC 22931 / RTOG 9501 — Adjuvant Chemoradiation | 2004-2005 | Concurrent cisplatin + radiation improves locoregional control and disease-free survival over radiation alone for high-risk resected head and neck SCC (positive margins, extranodal extension); established the standard of care for adjuvant treatment | PMID: 15128893 |
Complications Master Reference
| Complication | Etiology/Context | Management |
|---|---|---|
| Oroantral communication (OAC) | Extraction of maxillary premolars/molars with roots close to the sinus floor | Small (< 2 mm): no treatment, heals spontaneously with precautions (avoid nose blowing, no straws, sneeze with mouth open). Medium (2-6 mm): primary closure with buccal advancement flap. Large (> 6 mm) or chronic fistula: buccal fat pad flap or palatal rotation flap. Antibiotics (amoxicillin + decongestant) for all. |
| Dry socket (alveolar osteitis) | Premature loss of the blood clot from the extraction socket (2-5% of extractions, 25-30% of mandibular third molars). Risk: smoking, oral contraceptives, difficult extraction, poor hygiene | Gentle irrigation to remove debris; medicated dressing (eugenol on iodoform gauze, or dry socket paste); change dressing every 2-3 days until symptoms resolve (5-7 days). NOT an infection — antibiotics unnecessary unless concurrent infection. |
| Postextraction hemorrhage | Inadequate hemostasis, anticoagulants, coagulopathy, traumatic extraction | Local measures: pressure with gauze, suturing, gelfoam/surgicel (oxidized cellulose) in socket, topical tranexamic acid (5% mouthwash), bone wax for bony bleeders. Rarely requires systemic intervention. |
| Displaced root into maxillary sinus | Excessive apical pressure during maxillary molar extraction | Do NOT attempt retrieval through the socket. Obtain CT to confirm location. Caldwell-Luc approach for retrieval. If the root is small and the sinus is healthy, some advocate observation — the root may be encapsulated or expelled through the ostium. |
| Mandibular fracture during extraction | Excessive force on an atrophic mandible (< 20 mm height), deeply impacted third molars, pathologic bone (cyst, tumor, osteonecrosis) | Recognized intraoperatively: ORIF with plate fixation. Recognized postoperatively: MMF and/or ORIF depending on displacement and patient factors. |
| Frey syndrome (gustatory sweating) | Aberrant regeneration of parasympathetic nerve fibers (auriculotemporal nerve) into the sympathetic innervation of sweat glands after parotid surgery or condylar fracture surgery | Usually mild; Botox injections to the affected skin area for symptomatic cases; interposition of fascia or acellular dermal matrix during surgery to prevent |