Orthopedic Surgery
Every fracture, joint pathology, classification system, implant, surgical approach, physical exam maneuver, medication, and management strategy in one place.
01 Musculoskeletal Anatomy
Orthopedic surgery covers the entire musculoskeletal system: bones, joints, ligaments, tendons, muscles, cartilage, and the peripheral nerves that innervate them. The specialty's scope extends from skull base (craniocervical junction) to the tips of the fingers and toes, and includes the spine. Understanding skeletal anatomy, joint mechanics, and neurovascular relationships is the foundation of every orthopedic decision.
Bone Classification
The 206 bones of the adult skeleton are classified by shape, and each type has distinct fracture patterns and healing characteristics.
| Bone Type | Examples | Structure | Clinical Relevance |
|---|---|---|---|
| Long bones | Femur, tibia, humerus, radius, ulna, fibula, metacarpals, phalanges | Diaphysis (shaft) of cortical bone surrounding a medullary canal; metaphysis (flared transition zone); epiphysis (articular end) with subchondral bone | Fracture location described as proximal, mid-shaft, or distal; amenable to intramedullary nailing |
| Short bones | Carpals (scaphoid, lunate, triquetrum, pisiform, trapezium, trapezoid, capitate, hamate), tarsals (talus, calcaneus, navicular, cuboid, cuneiforms) | Mostly cancellous (trabecular) bone with thin cortical shell | Vulnerable to avascular necrosis due to retrograde blood supply (scaphoid, talus) |
| Flat bones | Scapula, ilium, sternum, skull | Two layers of cortical bone (tables) with cancellous diploe between | Fractures often from high-energy mechanisms; scapula fracture implies massive force |
| Irregular bones | Vertebrae, sacrum, facial bones | Variable cortical/cancellous ratio | Complex 3D anatomy makes fracture classification and fixation challenging |
| Sesamoid bones | Patella, hallux sesamoids | Embedded within tendons | Patella is the largest sesamoid; fractures disrupt the extensor mechanism |
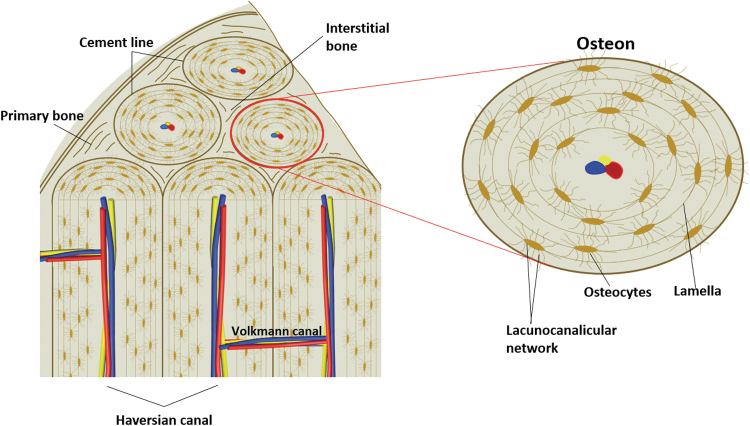
Bone Microstructure
Cortical (compact) bone constitutes ~80% of skeletal mass. It is organized into osteons (Haversian systems) — concentric lamellae of mineralized collagen around a central Haversian canal containing blood vessels and nerves. Osteons are connected by Volkmann's canals running perpendicular to the long axis. Cortical bone provides mechanical strength, particularly in resisting bending and torsional forces. Cancellous (trabecular/spongy) bone fills the metaphyses and epiphyses. Its trabecular architecture aligns along lines of mechanical stress (Wolff's law) and provides compressive strength with less weight. Cancellous bone has a much higher surface area and turnover rate, making it more metabolically active and more susceptible to osteoporosis.
Joint Classification
| Joint Type | Movement | Examples |
|---|---|---|
| Synarthrosis (fibrous) | No movement | Skull sutures, distal tibiofibular syndesmosis |
| Amphiarthrosis (cartilaginous) | Limited movement | Intervertebral discs (symphysis), pubic symphysis, growth plates (synchondrosis) |
| Diarthrosis (synovial) | Free movement | Ball-and-socket (hip, shoulder), hinge (elbow, interphalangeal), pivot (atlantoaxial, proximal radioulnar), saddle (1st CMC), condyloid (MCP, wrist), plane/gliding (acromioclavicular, intercarpal) |
Synovial joints share a common architecture: articular (hyaline) cartilage covering the bony surfaces, a fibrous joint capsule lined by synovium, synovial fluid for lubrication and nutrition, and stabilizing ligaments. The labrum (hip, shoulder) is a fibrocartilaginous ring that deepens the socket and enhances stability.
Major Muscle Groups & Innervation
| Region | Key Muscles | Nerve | Root | Action Tested |
|---|---|---|---|---|
| Shoulder | Deltoid | Axillary | C5–C6 | Arm abduction (15–90°) |
| Shoulder | Supraspinatus | Suprascapular | C5–C6 | Arm abduction (0–15°, initiation) |
| Shoulder | Infraspinatus / teres minor | Suprascapular / axillary | C5–C6 | External rotation |
| Shoulder | Subscapularis | Upper & lower subscapular | C5–C7 | Internal rotation |
| Arm | Biceps brachii | Musculocutaneous | C5–C6 | Elbow flexion, forearm supination |
| Arm | Triceps | Radial | C6–C8 | Elbow extension |
| Forearm | Wrist extensors (ECRL, ECRB, ECU) | Radial / posterior interosseous | C6–C7 | Wrist extension |
| Hand | Intrinsics (lumbricals, interossei) | Median (lateral 2 lumbricals), ulnar (rest) | C8–T1 | MCP flexion, IP extension; finger abduction/adduction |
| Hip | Iliopsoas | Femoral nerve, direct branches L1–L3 | L1–L3 | Hip flexion |
| Hip | Gluteus medius/minimus | Superior gluteal | L4–S1 | Hip abduction (Trendelenburg test) |
| Thigh | Quadriceps | Femoral | L2–L4 | Knee extension |
| Thigh | Hamstrings | Sciatic (tibial division) | L5–S2 | Knee flexion |
| Leg | Tibialis anterior | Deep peroneal (fibular) | L4–L5 | Ankle dorsiflexion |
| Leg | Gastrocnemius/soleus | Tibial | S1–S2 | Ankle plantarflexion |
| Leg | Peroneus longus/brevis | Superficial peroneal | L5–S1 | Ankle eversion |
| Foot | Extensor hallucis longus | Deep peroneal | L5 | Great toe dorsiflexion (key L5 test) |
Blood Supply to Bone
Long bones receive blood from three sources: the nutrient artery (enters the diaphysis through the nutrient foramen, supplies the inner 2/3 of the cortex and medullary canal), periosteal arteries (supply the outer 1/3 of the cortex — critically important when the nutrient artery is disrupted by fracture or reaming), and metaphyseal/epiphyseal arteries (enter at the bone ends). Certain bones have tenuous blood supply making them vulnerable to avascular necrosis (AVN) after fracture: femoral head (medial femoral circumflex artery via retinacular vessels), scaphoid (dorsal branch of radial artery enters distally, flows retrograde), talus (artery of the tarsal canal, deltoid branches), and the proximal pole of the lunate.
02 The Orthopedic Physical Exam
The orthopedic exam follows a systematic approach: Look (alignment, deformity, swelling, ecchymosis, skin integrity, muscle wasting), Feel (point tenderness, crepitus, effusion, temperature, pulses), Move (active and passive range of motion, stability testing), and Neurovascular status (motor, sensory, vascular). Every injured extremity requires documentation of neurovascular status before and after any intervention.
Range of Motion — Normal Values
| Joint | Motion | Normal ROM |
|---|---|---|
| Shoulder | Forward flexion / extension | 180° / 60° |
| Shoulder | Abduction / adduction | 180° / 45° |
| Shoulder | External rotation / internal rotation | 90° / 70° |
| Elbow | Flexion / extension | 150° / 0° (hyperextension up to 10° in some) |
| Forearm | Pronation / supination | 80° / 80° |
| Wrist | Flexion / extension | 80° / 70° |
| Wrist | Radial deviation / ulnar deviation | 20° / 30° |
| Hip | Flexion / extension | 120° / 30° |
| Hip | Abduction / adduction | 45° / 30° |
| Hip | Internal rotation / external rotation | 35° / 45° |
| Knee | Flexion / extension | 135° / 0° |
| Ankle | Dorsiflexion / plantarflexion | 20° / 50° |
| Ankle | Inversion / eversion | 35° / 15° |
Special Tests by Joint — Shoulder
| Test | Technique | Positive Finding | Pathology Assessed |
|---|---|---|---|
| Neer impingement | Stabilize scapula, passively forward flex shoulder with arm pronated | Pain at ~90° flexion | Subacromial impingement / rotator cuff tendinopathy |
| Hawkins-Kennedy | Forward flex shoulder to 90°, forcibly internally rotate | Pain | Subacromial impingement |
| Empty can (Jobe's) | Arms abducted 90°, forward flexed 30°, thumbs pointing down; resist downward force | Weakness or pain | Supraspinatus tear |
| External rotation lag | Passively externally rotate the arm; ask patient to hold position | Arm falls into internal rotation | Infraspinatus/teres minor tear |
| Belly press / lift-off (Gerber) | Hand on abdomen, push against belly / hand behind back, lift off | Cannot maintain pressure / cannot lift hand off back | Subscapularis tear |
| Speed's test | Resist forward flexion with elbow extended, forearm supinated | Bicipital groove pain | Biceps tendinopathy / SLAP lesion |
| Apprehension / relocation | Abduct 90°, externally rotate; then apply posterior force to humeral head | Apprehension resolves with relocation | Anterior glenohumeral instability |
| Sulcus sign | Pull arm inferiorly with elbow at side | Visible sulcus below acromion >2 cm | Multidirectional / inferior instability |
| O'Brien's test | Arm flexed 90°, adducted 10°, internally rotated; resist downward force; repeat in supination | Pain with pronation relieved by supination | SLAP lesion / AC joint pathology |
| Cross-body adduction | Forward flex arm 90°, adduct across body | Pain at AC joint | AC joint pathology |
Special Tests — Knee
| Test | Technique | Positive Finding | Pathology |
|---|---|---|---|
| Lachman | Knee at 20° flexion, stabilize femur, pull tibia anteriorly | Increased anterior translation, soft/absent endpoint | ACL tear (most sensitive test) |
| Anterior drawer | Knee at 90° flexion, pull tibia forward | Anterior translation >6 mm | ACL tear |
| Pivot shift | Internal rotation + valgus stress during flexion from extension | Clunk as tibia reduces from subluxed position | ACL tear (most specific; best under anesthesia) |
| Posterior drawer | Knee at 90°, push tibia posteriorly | Posterior translation | PCL tear |
| Posterior sag sign | Both knees flexed 90°, compare tibial plateau prominence | Tibial plateau sags posteriorly on affected side | PCL tear |
| Valgus stress test | Apply valgus force at 0° and 30° of flexion | Medial joint opening >5 mm at 30°; instability at 0° indicates combined injury | MCL tear (Grade I–III) |
| Varus stress test | Apply varus force at 0° and 30° | Lateral joint opening | LCL/posterolateral corner injury |
| McMurray's | Flex knee fully, apply valgus + external rotation, then extend; repeat with varus + internal rotation | Painful click or catching | Meniscal tear |
| Thessaly test | Patient stands on affected leg, knee flexed 20°, rotates body | Locking, catching, or pain at joint line | Meniscal tear |
| Patellar apprehension | Push patella laterally with knee in slight flexion | Patient grabs examiner's hand, resists | Patellar instability / history of dislocation |
Special Tests — Hip
| Test | Technique | Positive Finding | Pathology |
|---|---|---|---|
| FABER (Patrick's) | Flex, Abduct, Externally Rotate hip; lower knee toward table | Groin pain = hip pathology; sacral pain = SI joint | Intra-articular hip / SI joint |
| FADIR | Flex, Adduct, Internally Rotate hip | Groin pain / catching | Femoroacetabular impingement (FAI), labral tear |
| Trendelenburg | Stand on one leg | Contralateral pelvis drops | Gluteus medius weakness / superior gluteal nerve injury |
| Thomas test | Flex opposite hip fully; observe tested leg | Tested hip cannot remain flat on table | Hip flexion contracture |
| Ober's test | Side-lying, abduct and extend hip, allow to adduct | Leg stays abducted, cannot adduct past midline | IT band contracture |
| Log roll | Gently internally/externally rotate leg in extension | Pain with minimal rotation | Hip fracture / effusion / synovitis |
Neurovascular Examination
Every injured extremity must be assessed for neurovascular integrity. Motor testing: grade strength 0–5 (0 = no contraction, 1 = flicker, 2 = movement with gravity eliminated, 3 = against gravity, 4 = against resistance, 5 = normal). Sensory testing: light touch and two-point discrimination in the distribution of each peripheral nerve (median = thenar eminence and index fingertip; ulnar = small finger; radial = first dorsal web space; peroneal = first web space of foot; tibial = sole of foot). Vascular: palpate pulses (radial, ulnar, DP, PT), check capillary refill (<2 seconds is normal), and assess for compartment syndrome signs (pain with passive stretch, tense compartments, pain out of proportion).
03 Fracture Fundamentals
Fracture Description System
Every fracture is described using a standardized system. The complete description includes: which bone, location (proximal, mid-shaft, distal; metaphyseal vs diaphyseal vs epiphyseal), pattern (transverse, oblique, spiral, comminuted, segmental, butterfly fragment), displacement (amount in mm or % and direction), angulation (degrees and direction — described by the apex of the angle: apex volar, apex lateral, etc.), shortening (in mm), rotation, articular involvement (intra-articular vs extra-articular), and open vs closed.
| Fracture Pattern | Mechanism | Stability | Clinical Implication |
|---|---|---|---|
| Transverse | Direct blow / bending force | Stable after reduction (resists shortening) | Good bone-to-bone contact; amenable to plating |
| Oblique | Combined bending and compression | Unstable (tendency to shorten) | Requires lag screw or plate fixation to prevent shortening |
| Spiral | Torsional / rotational force | Unstable (shortening and rotation) | Longer fracture line = more surface area for healing but difficult to hold reduced |
| Comminuted | High-energy; >2 fragments | Unstable | Cannot rely on cortical contact for stability; may need bridge plating or nail |
| Segmental | High-energy; isolated bone segment between two fracture lines | Very unstable | Middle segment at risk for AVN due to stripped periosteum; usually requires nailing |
| Greenstick | Bending in pediatric bone; one cortex breaks, other bows | Partially stable | Must complete the fracture or accept angulation; risk of re-fracture |
| Torus (buckle) | Axial compression in pediatric bone | Stable | Metaphyseal compression; treated with removable splint, heals in 3–4 weeks |
| Avulsion | Tendon/ligament pulls off bone fragment | Displaced by muscle pull | May need fixation if fragment large or involves joint surface |
| Pathologic | Fracture through weakened bone (tumor, osteoporosis, Paget's) | Variable | Must evaluate for underlying etiology; may need biopsy before fixation |
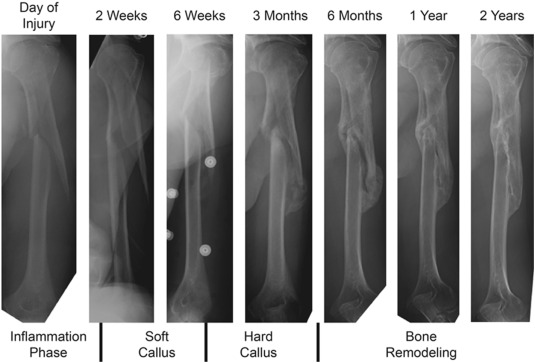
Fracture Healing Biology
Fracture healing proceeds through a predictable sequence. Inflammatory phase (days 0–7): fracture hematoma forms, providing a scaffold rich in platelets, macrophages, and growth factors (PDGF, TGF-beta, BMP). Osteocytes in damaged bone undergo apoptosis. Mesenchymal stem cells are recruited from periosteum, endosteum, and bone marrow. Soft callus phase (weeks 1–3): fibroblasts and chondroblasts produce a cartilaginous callus bridging the fracture gap (endochondral ossification). This is visible radiographically as hazy periosteal new bone. Hard callus phase (weeks 3–12): the soft callus is progressively mineralized and replaced by woven bone through osteoblast activity. Radiographically, the callus becomes denser and more organized. Remodeling phase (months to years): osteoclast-mediated resorption of excess callus and osteoblast-mediated deposition of lamellar bone along stress lines (Wolff's law) restores near-normal bone architecture.
Factors Affecting Healing
| Factor | Promotes Healing | Impairs Healing |
|---|---|---|
| Blood supply | Intact periosteum, good soft tissue envelope | Open fracture, periosteal stripping, smoking (vasoconstriction) |
| Stability | Appropriate fixation, good reduction | Inadequate fixation, excessive motion at fracture site |
| Fracture gap | Minimal gap, bone-to-bone contact | Bone loss, excessive distraction, interposed soft tissue |
| Nutrition | Adequate protein, calcium, vitamin D | Malnutrition (albumin <3.0), vitamin D deficiency |
| Systemic factors | Young age, healthy metabolism | Diabetes, hypothyroidism, renal failure, immunosuppression |
| Medications | BMP (adjunct), PTH analogs (teriparatide) | NSAIDs (controversial but avoid in high-risk nonunion), corticosteroids, some chemotherapeutics |
| Infection | Clean wound, prophylactic antibiotics | Osteomyelitis, contaminated open fractures |
| Smoking | Cessation | Active smoking doubles nonunion risk; nicotine impairs osteoblast function and neovascularization (Castillo et al., 2005) |
Nonunion & Malunion
Delayed union: healing has not occurred within the expected time frame but is still progressing (callus visible, fracture line still present). Nonunion: fracture has failed to heal and no further progress is expected — typically defined as no radiographic progression of healing for 3 consecutive months or failure to heal by 9 months. Two types: hypertrophic nonunion (abundant callus, "elephant foot" or "horse hoof" appearance — biology is intact but stability is insufficient; treatment = improve stability with revision fixation) and atrophic nonunion (no callus, sclerotic bone ends — biology is deficient; treatment = debridement of sclerotic bone, restoration of canal, bone grafting + stable fixation). Malunion: fracture has healed in a non-anatomic position (angulation, rotation, shortening) — may require corrective osteotomy if symptomatic.
Open Fracture Classification — Gustilo-Anderson
Open fractures involve a communication between the fracture site and the external environment. The Gustilo-Anderson classification is the universal system, though it has significant inter-observer variability. Definitive grading occurs in the operating room after wound exploration and debridement.
| Type | Wound Size | Soft Tissue Injury | Contamination | Bone Injury | Infection Rate |
|---|---|---|---|---|---|
| I | <1 cm | Minimal | Clean | Simple fracture pattern | 0–2% |
| II | 1–10 cm | Moderate, no flaps or avulsions | Moderate | Moderate comminution | 2–10% |
| IIIA | >10 cm | Extensive but adequate soft tissue coverage of bone possible | High | Severe comminution, segmental | 10–25% |
| IIIB | >10 cm | Extensive soft tissue loss requiring flap coverage (local or free) | High | Severe with periosteal stripping | 25–50% |
| IIIC | Any | Any open fracture with arterial injury requiring repair | Variable | Variable | 25–50% |
Antibiotics: Administer within 1 hour of presentation (Lack et al., 2015). Type I–II: cefazolin 2 g IV (or clindamycin 900 mg if penicillin allergy). Type III: add gentamicin 5 mg/kg IV. Farm/water contamination: add penicillin 4 million units IV for Clostridium coverage. Tetanus prophylaxis per immunization history. Irrigation and debridement (I&D): Perform in the OR within 24 hours (evidence no longer supports the rigid "6-hour rule," but earlier is preferred for type IIIB/C — Schenker et al., 2012). Use low-pressure lavage with normal saline (high-pressure shown to damage tissue and impair healing). Debride all nonviable tissue. Repeat I&D at 48–72 hours as needed. Stabilization: Temporary external fixation for type III injuries; definitive fixation (typically intramedullary nail for tibial/femoral shaft fractures) when soft tissues allow. Wound management: Type I/II can often undergo primary closure. Type IIIB requires soft tissue coverage — goal within 72 hours to 7 days (the "fix and flap" paradigm).
04 Shoulder Pathology Upper Extremity
Rotator Cuff Disease
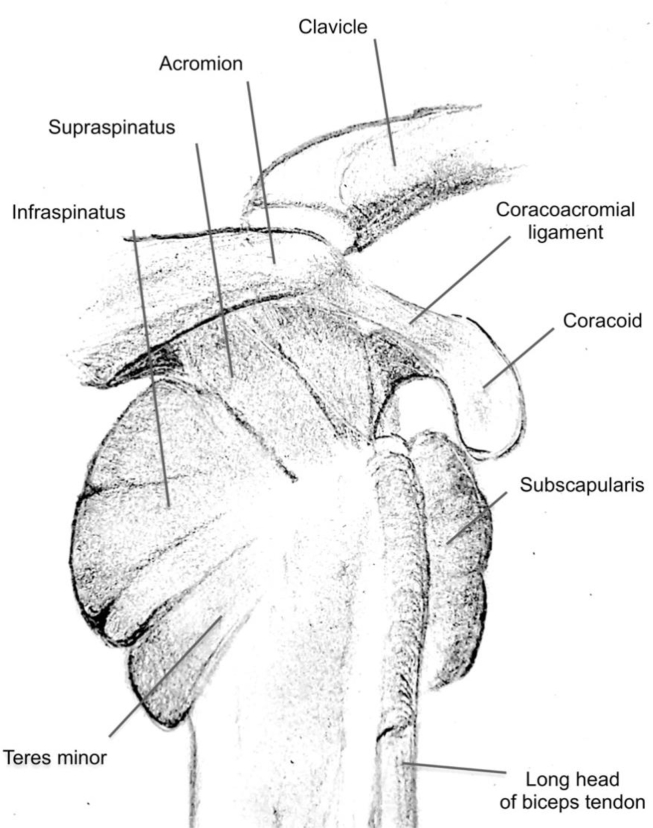
The rotator cuff consists of four muscles — supraspinatus (abduction initiation), infraspinatus (external rotation), teres minor (external rotation), and subscapularis (internal rotation) — forming a musculotendinous cuff that dynamically stabilizes the humeral head within the glenoid. The supraspinatus tendon is the most commonly torn, particularly in its "critical zone" (an area of relative hypovascularity ~1 cm proximal to its insertion on the greater tuberosity).
Impingement syndrome is the clinical spectrum from tendinopathy to partial-thickness tears to full-thickness tears. Neer's stages: Stage I = edema/hemorrhage (age <25, reversible), Stage II = fibrosis/tendinitis (age 25–40, chronic), Stage III = tendon degeneration/tear (age >40, usually requires surgery if full-thickness). Subacromial impingement occurs when the rotator cuff tendons are compressed between the humeral head and the coracoacromial arch (acromion, coracoacromial ligament, AC joint). A type III (hooked) acromion on supraspinatus outlet view increases impingement risk (Bigliani classification: Type I = flat, Type II = curved, Type III = hooked).
Treatment: Partial tears and tendinopathy: activity modification, physical therapy (eccentric strengthening, scapular stabilization), NSAIDs, subacromial corticosteroid injection (limit to 3 per year — repeated injections weaken tendon). Full-thickness tears: surgical repair (arthroscopic preferred) indicated for acute traumatic tears in active patients, chronic tears with significant functional limitation despite conservative management, or progressive tear enlargement. Repair techniques include single-row, double-row, and transosseous-equivalent (suture bridge) fixation to the greater tuberosity using suture anchors. Massive irreparable tears may be managed with superior capsular reconstruction (SCR), balloon spacer (InSpace), or reverse total shoulder arthroplasty.
Glenohumeral Instability
The shoulder trades bony constraint for mobility — the glenoid covers only ~25% of the humeral head surface. Stability depends on static restraints (labrum, capsule, glenohumeral ligaments) and dynamic restraints (rotator cuff). Anterior dislocation accounts for ~95% of traumatic dislocations — mechanism is typically forced abduction + external rotation. The humeral head displaces anteroinferiorly. Associated injuries: Bankart lesion (avulsion of the anteroinferior labrum from the glenoid — the "essential lesion" of recurrent anterior instability), Hill-Sachs lesion (compression fracture of the posterosuperior humeral head from impaction against the glenoid rim), and axillary nerve injury (test deltoid function and "regimental badge area" sensation). Recurrence risk is strongly age-dependent: >90% in patients under 20, ~25% in patients over 40.
Posterior dislocation accounts for ~2–5% and is classically associated with seizures, electrocution, and electroconvulsive therapy (bilateral posterior dislocations are virtually pathognomonic for seizure). The AP radiograph may appear deceptively normal ("lightbulb sign" — internally rotated humeral head appears rounded). A reverse Hill-Sachs lesion (McLaughlin lesion — compression fracture of the anteromedial humeral head) may be present. Always obtain axillary lateral or scapular-Y views to rule out posterior dislocation.
Treatment of recurrent anterior instability: Arthroscopic Bankart repair (reattachment of the torn labrum with suture anchors) is the standard for younger patients with soft tissue Bankart lesions. In the presence of significant glenoid bone loss (>20–25% — "inverted pear" glenoid on arthroscopy, or critical bone loss on CT), or a large engaging Hill-Sachs lesion, a bony procedure is required: Latarjet procedure (transfer of the coracoid process with the conjoined tendon to the anterior glenoid — provides bone augmentation + dynamic sling effect) or iliac crest bone graft (Eden-Hybinette procedure).
Acromioclavicular (AC) Joint Injuries
The Rockwood classification grades AC joint separations I through VI based on the degree of displacement of the clavicle relative to the acromion and the integrity of the AC and coracoclavicular (CC) ligaments.
| Type | AC Ligament | CC Ligament | Displacement | Treatment |
|---|---|---|---|---|
| I | Sprain | Intact | None | Sling, ice, early ROM — nonoperative |
| II | Torn | Sprain | Slight vertical (clavicle elevated <100% CC distance) | Nonoperative (sling 1–2 weeks, PT) |
| III | Torn | Torn | Clavicle elevated 100% of CC distance | Controversial; nonoperative initially for most; surgery for young athletes/laborers |
| IV | Torn | Torn | Clavicle displaced posteriorly into trapezius | Surgical (CC reconstruction) |
| V | Torn | Torn | Clavicle elevated 200–300% of CC distance | Surgical |
| VI | Torn | Torn | Clavicle displaced inferiorly (subcoracoid or subacromial) | Surgical (very rare, high-energy) |
05 Proximal Humerus & Clavicle Fractures Upper Extremity
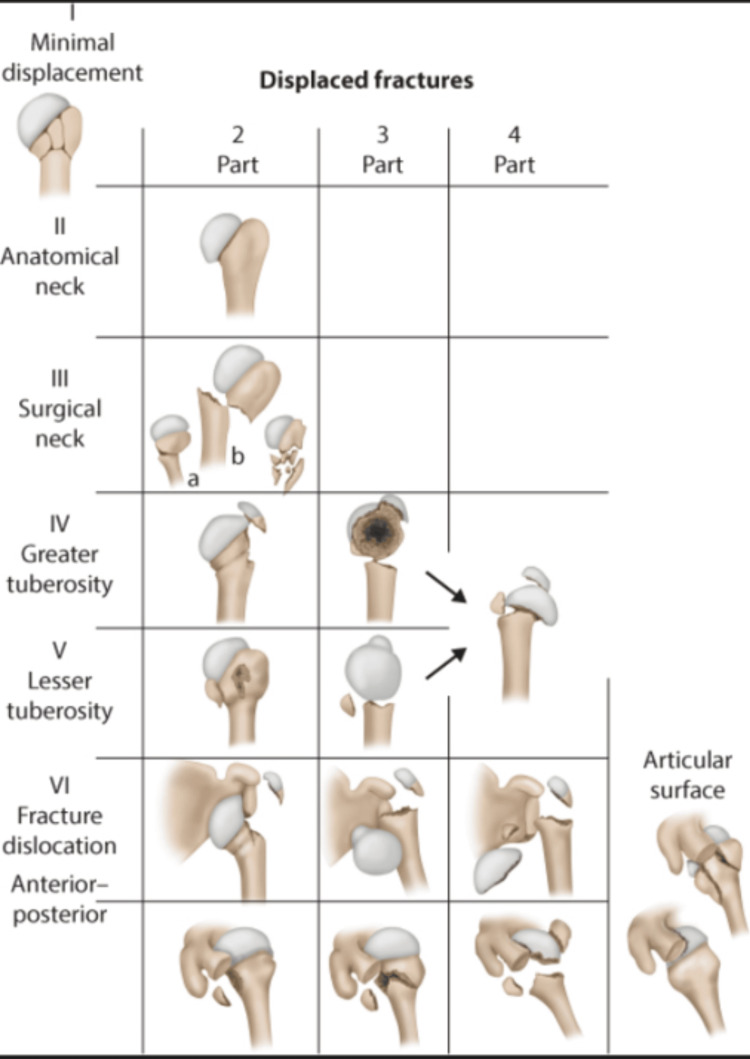
Proximal Humerus Fractures — Neer Classification
The proximal humerus has four "parts" (Neer's parts): greater tuberosity, lesser tuberosity, articular segment (humeral head), and shaft. A part is considered displaced when there is >1 cm of displacement or >45° of angulation. The Neer classification counts the number of displaced parts.
| Neer Classification | Description | Treatment |
|---|---|---|
| 1-part (minimally displaced) | No part meets displacement criteria; ~80% of proximal humerus fractures | Sling, early pendulum exercises, progress ROM at 2–3 weeks |
| 2-part | One part displaced: surgical neck (most common 2-part), greater tuberosity, lesser tuberosity, or anatomic neck | Surgical neck: ORIF with plate (Philos plate) if displaced >1 cm or angulated. Greater tuberosity: ORIF if displaced >5 mm (some say >3 mm in overhead athletes) |
| 3-part | Two parts displaced; humeral head typically malrotated | ORIF in younger patients; hemiarthroplasty or reverse TSA in elderly |
| 4-part | All four parts displaced; head segment detached from blood supply | High AVN risk; reverse total shoulder arthroplasty (rTSA) preferred in elderly; ORIF attempted in young patients |
| Head-splitting | Fracture line through articular surface | Arthroplasty (head not reconstructable) |
| Fracture-dislocation | Any pattern with associated glenohumeral dislocation | Urgent reduction; surgical management based on fracture pattern |
Clavicle Fractures
The clavicle is the most commonly fractured bone in the body (~2.6% of all fractures). Middle third fractures account for ~80% — the clavicle is thinnest here and lacks muscular/ligamentous protection. The lateral third (~15%) and medial third (~5%) are less common.
Treatment of middle third fractures: Non-displaced or minimally displaced: sling for comfort, early ROM as tolerated, heals in 6–12 weeks. Operative indications include: shortening >2 cm, comminution with displacement, open fracture, skin tenting, associated neurovascular injury, floating shoulder (ipsilateral clavicle + scapular neck fracture). Fixation options: superior plate (Acumed, DePuy precontoured anatomic plates) or anterior-inferior plate (theoretical lower prominence), or intramedullary fixation (Rockwood pin, TEN nail — less soft tissue disruption but less rotational control). A landmark RCT (the Canadian Orthopaedic Trauma Society, 2007) showed operative fixation of displaced midshaft fractures reduces nonunion rate from ~15% to ~3% and improves functional outcomes.
Lateral third fractures: Neer classification: Type I = lateral to CC ligaments (stable, nonoperative); Type II = medial to CC ligaments (unstable, high nonunion rate ~30%, often requires fixation — hook plate, CC screw, suture button like TightRope); Type III = intra-articular (uncommon).
06 Elbow Pathology & Fractures Upper Extremity
Distal Humerus Fractures
Distal humerus fractures in adults are complex intra-articular injuries, often from falls or high-energy trauma. The distal humerus forms two columns (medial and lateral) supporting the trochlea and capitellum. The AO/OTA classification divides them into extra-articular (type A), partial articular (type B — affecting one column), and complete articular (type C — both columns, often with intercondylar split). Treatment of displaced intra-articular fractures is ORIF via a posterior approach (typically olecranon osteotomy or triceps-reflecting [Bryan-Morrey] approach) using dual-column plating (perpendicular or parallel 90-90 plating technique). In elderly patients with severely comminuted fractures and poor bone quality, total elbow arthroplasty (TEA) may be preferred over ORIF (the McKee et al., 2003 trial showed TEA had better outcomes than ORIF in elderly patients with comminuted distal humerus fractures).
Olecranon Fractures
The olecranon is subcutaneous and vulnerable to direct blows and falls on the flexed elbow. The triceps inserts on the olecranon, so any displaced fracture disrupts the extensor mechanism. If the patient cannot actively extend the elbow against gravity, the fracture is functionally displaced and requires surgery. Treatment: Non-displaced, stable fractures with intact extensor mechanism: long arm splint at 90°, early ROM. Displaced: tension band wiring (TBW) — two K-wires + a figure-of-8 wire that converts the triceps' tensile force into compression at the articular surface during elbow flexion. This is the classic technique for simple transverse fractures. Comminuted fractures: plate fixation (precontoured olecranon plates — Synthes, Acumed) is superior to TBW for comminuted and oblique patterns. Excision of the fragment with triceps reattachment is an option for elderly/low-demand patients with comminuted fractures involving <50% of the articular surface.
Radial Head Fractures — Mason Classification
| Mason Type | Description | Treatment |
|---|---|---|
| I | Non-displaced or minimally displaced (<2 mm) | Sling, early ROM within 48 hours. Aspirate hemarthrosis + inject lidocaine for pain relief and to assess for mechanical block |
| II | Displaced >2 mm, partial articular (involving >30% of head, >2 mm step-off, or mechanical block) | ORIF with headless compression screws or mini plate |
| III | Comminuted, entire radial head | Radial head arthroplasty (metal prosthesis — Mason, Evolve); excision only if no associated ligamentous injury (contraindicated with Essex-Lopresti or MCL tear) |
| IV (Johnston modification) | Any Mason type with associated elbow dislocation | Reduce dislocation; treat radial head as above + address associated ligament injuries |
Lateral Epicondylitis (Tennis Elbow)
Lateral epicondylitis is a degenerative tendinopathy (not a true inflammatory process) of the extensor carpi radialis brevis (ECRB) origin at the lateral epicondyle. Peak incidence ages 35–55. Pathology shows angiofibroblastic hyperplasia (Nirschl), not acute inflammation — hence "tendinosis" is more accurate than "tendinitis." Treatment: 90% resolve with nonoperative management: activity modification (avoid repetitive gripping/wrist extension), counterforce bracing (forearm strap), eccentric wrist extensor stretching/strengthening, and topical NSAIDs. PRP injections have shown benefit in some studies (Gosens et al., 2011). Corticosteroid injection provides short-term relief but worsens long-term outcomes compared to wait-and-see. Surgical debridement (open or arthroscopic Nirschl debridement) is reserved for refractory cases after 6–12 months.
Medial Epicondylitis (Golfer's Elbow)
Tendinopathy of the flexor-pronator mass (primarily flexor carpi radialis and pronator teres) at the medial epicondyle. Less common than lateral epicondylitis (~10:1 ratio). Must evaluate for concomitant ulnar neuropathy at the cubital tunnel (present in 20–60% of medial epicondylitis cases) — Tinel's at the cubital tunnel, assess intrinsic hand strength and small/ring finger numbness. Treatment parallels lateral epicondylitis; surgery includes debridement with or without ulnar nerve transposition.
07 Wrist & Hand Upper Extremity
Distal Radius Fractures
The most common fracture in the upper extremity and the most common fracture in the ED. Bimodal distribution: young adults (high-energy) and elderly women with osteoporosis (low-energy FOOSH — fall on outstretched hand). Key named patterns: Colles' fracture (dorsally displaced and angulated — the classic "dinner fork" deformity), Smith's fracture (volarly displaced — "reverse Colles'"), Barton's fracture (intra-articular fracture-subluxation — dorsal or volar), Chauffeur's fracture (radial styloid fracture — intra-articular).
Acceptable reduction parameters (Lafontaine criteria for instability when not met): radial inclination >15° (normal 22°), radial height >7 mm (normal 11 mm), volar tilt 0–15° (normal 11° volar), articular step-off <2 mm, ulnar variance <3 mm. Treatment: Stable, extra-articular, acceptable alignment: closed reduction + sugar-tong splint, then short arm cast at 1–2 weeks for a total 6 weeks. Operative indications: intra-articular fractures with >2 mm step-off, loss of reduction, unstable fracture patterns (dorsal comminution, initial shortening >5 mm, volar tilt >20°), associated ulnar styloid base fracture with DRUJ instability. Fixation: volar locking plate (Synthes VA-LCP, Acumed Acu-Loc) is the most common technique — allows early ROM. Fragment-specific fixation for complex articular patterns. External fixation (spanning or non-spanning) for severely comminuted fractures.
Scaphoid Fractures
The scaphoid is the most commonly fractured carpal bone and the most clinically important because of its tenuous blood supply. The dorsal branch of the radial artery enters the scaphoid distally and supplies the proximal pole via retrograde flow — fractures through the waist or proximal pole disrupt this supply, leading to avascular necrosis (AVN) of the proximal fragment in 20–40% of displaced proximal pole fractures. Diagnosis: Tenderness in the anatomical snuffbox (sensitivity ~90%, specificity ~40%) and tenderness with axial compression of the thumb (scaphoid compression test). Initial radiographs may be negative in up to 20% of cases. If clinical suspicion is high with negative radiographs, immobilize in a thumb spica splint and obtain MRI (gold standard, 99% sensitivity) or repeat radiographs at 10–14 days.
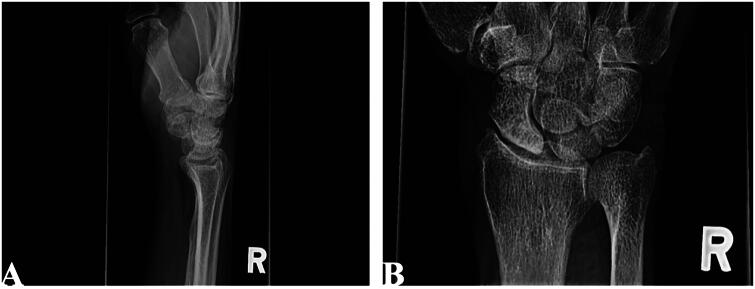
Treatment: Non-displaced waist fractures: thumb spica cast for 8–12 weeks (short arm thumb spica is generally adequate; long arm thumb spica adds no benefit per recent evidence). Non-displaced proximal pole fractures: higher nonunion risk, lower threshold for surgical fixation. Displaced (>1 mm step-off on CT) or proximal pole fractures: percutaneous or open headless compression screw fixation (Acutrak, Herbert screw). Established nonunion: open reduction, bone grafting (vascularized or non-vascularized — the 1,2-intercompartmental supraretinacular artery [1,2 ICSRA] vascularized bone graft for proximal pole AVN), and screw fixation. End-stage: scaphoid excision and four-corner fusion (SLAC/SNAC wrist reconstruction).
Carpal Tunnel Syndrome
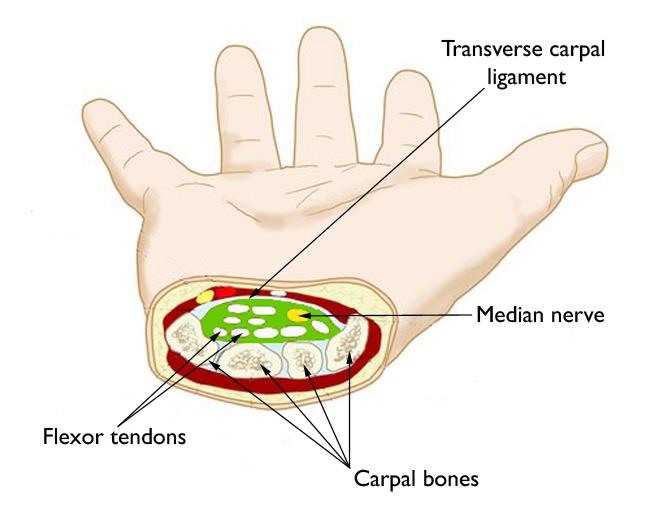
The most common peripheral nerve entrapment. The median nerve is compressed within the carpal tunnel — a fibro-osseous tunnel bounded by the carpal bones dorsally and the transverse carpal ligament (flexor retinaculum) volarly. Nine flexor tendons (4 FDS, 4 FDP, FPL) accompany the median nerve through the tunnel. Symptoms: Numbness and paresthesias in the median nerve distribution (thumb, index, middle, radial half of ring finger) — classically worse at night and with sustained wrist flexion (Phalen's test positive in 30–60 seconds). Physical exam: Tinel's sign (percussion over the carpal tunnel produces paresthesias), Phalen's test (wrist flexion for 60 seconds), Durkan's (direct compression). Late findings: thenar atrophy (abductor pollicis brevis), decreased two-point discrimination. Diagnosis: Nerve conduction studies (NCS) / electromyography (EMG) confirm median nerve entrapment — distal motor latency >4.2 ms, distal sensory latency >3.5 ms. Treatment: Mild/intermittent: night splinting in neutral wrist position, activity modification, corticosteroid injection (temporary relief, diagnostic utility). Moderate-severe or refractory: carpal tunnel release (CTR) — division of the transverse carpal ligament. Open (2–3 cm incision in line with the ring finger ray) or endoscopic (Agee single-portal or Chow two-portal technique). Success rates exceed 90%.
Trigger Finger (Stenosing Tenosynovitis)
Thickening of the A1 pulley at the level of the MCP joint causes catching or locking of the flexor tendon during finger motion. The ring finger and thumb are most commonly affected. Quinnell grading: Grade I = pain/tenderness at A1 pulley without triggering, Grade II = triggering but patient can actively extend, Grade III = triggering requiring passive extension, Grade IV = fixed locked position. Treatment: Splinting (MCP in extension), corticosteroid injection into the tendon sheath (60–90% success rate for first injection; diabetic patients have lower success rates). Refractory: A1 pulley release (open or percutaneous) — simple and definitive.
De Quervain's Tenosynovitis
Stenosing tenosynovitis of the first dorsal compartment (abductor pollicis longus [APL] and extensor pollicis brevis [EPB]). Finkelstein's test (ulnar deviation of the wrist with the thumb grasped in the fist — pain over the radial styloid) is the classic provocative maneuver. Treatment: thumb spica splint, corticosteroid injection into the first dorsal compartment (85% resolution rate — ensure injection enters the EPB subsheath, which exists as a separate compartment in 30% of patients), and surgical release for refractory cases.
08 Hip Pathology & Fractures Lower Extremity
Hip Fracture Overview
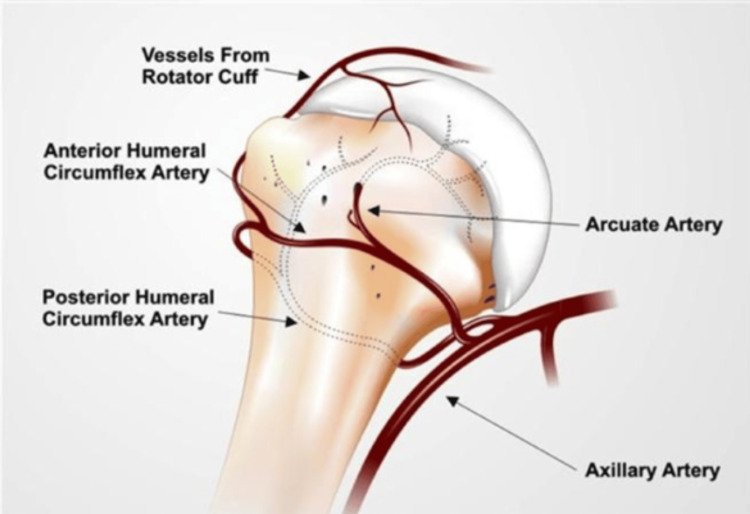
Hip fractures are one of the most consequential injuries in orthopedics — 1-year mortality ranges from 15–30% in the elderly. The primary distinction is intracapsular (femoral neck) vs extracapsular (intertrochanteric, subtrochanteric). This distinction dictates management because the femoral neck is intracapsular, and displaced fractures disrupt the retinacular blood supply to the femoral head, creating high AVN and nonunion risk.
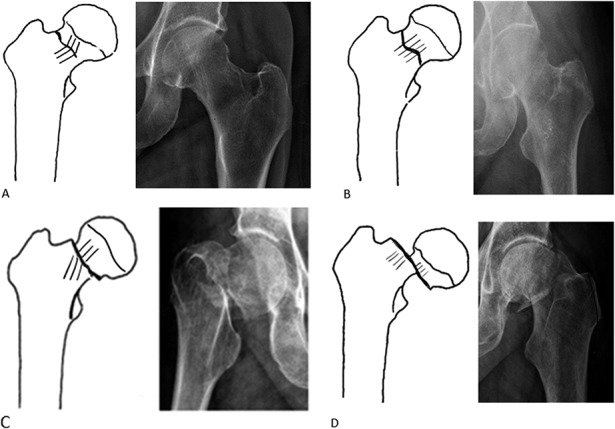
Femoral Neck Fractures — Garden Classification
| Garden Type | Description | Radiographic Finding | Treatment |
|---|---|---|---|
| I | Incomplete / valgus impacted | Inferior cortex intact, femoral head tilted into valgus | Percutaneous cannulated screws (3 parallel screws in inverted triangle) or sliding hip screw (SHS) |
| II | Complete, non-displaced | Complete fracture line, no displacement, trabeculae align normally | Cannulated screws or SHS; urgent fixation (<24 hours to reduce AVN risk) |
| III | Complete, partially displaced (varus) | Femoral head tilted into varus, trabeculae of head and acetabulum do not align | Age-dependent (see below) |
| IV | Complete, fully displaced | Femoral head returns to neutral in acetabulum, completely dissociated from neck | Age-dependent (see below) |
Young patients (<60–65 years): Attempt to save the native femoral head — emergent closed/open reduction and internal fixation (ORIF) with cannulated screws or sliding hip screw. Time to reduction is critical: goal <6–12 hours to minimize AVN risk (though evidence is mixed on the exact threshold — Upadhyay et al., 2004). Capsulotomy to decompress intracapsular hematoma may improve femoral head perfusion.
Elderly patients (≥65 years), active: Total hip arthroplasty (THA) — better functional outcomes and lower reoperation rates than hemiarthroplasty in active elderly patients. The HEALTH trial (Bhandari et al., NEJM 2019) showed THA had fewer secondary procedures but similar mortality and hip function scores compared to hemiarthroplasty at 24 months.
Elderly patients, low-demand / limited life expectancy / cognitive impairment: Hemiarthroplasty (unipolar [Austin-Moore, Thompson — cemented] or bipolar [has inner and outer bearings]) — shorter operative time, lower dislocation risk than THA.
Intertrochanteric Fractures
Extracapsular fractures between the greater and lesser trochanters. The blood supply to the femoral head is not at risk, so AVN is not a concern. These are treated surgically in virtually all cases (nonoperative management only for non-ambulatory patients with unacceptable surgical risk). Classification — AO/OTA 31-A: A1 = simple (2-part, stable after reduction), A2 = multifragmentary (comminuted, including loss of posteromedial buttress — unstable), A3 = reverse obliquity or transverse (subtrochanteric extension — very unstable).
Fixation: Stable patterns (A1): sliding hip screw (SHS) — lag screw into the femoral head with a side plate. Allows controlled collapse along the screw axis. Tip-apex distance (TAD) <25 mm predicts against cut-out (the most common mode of failure — Baumgaertner et al., 1995). Unstable patterns (A2, A3): cephalomedullary nail (Gamma nail, Synthes TFN/TFNA, Smith+Nephew InterTAN, Stryker Gamma3) — intramedullary device with a lag screw or helical blade into the femoral head. The intramedullary position provides a shorter moment arm and better load sharing than an SHS in unstable fractures. Reverse obliquity (A3) patterns must not be treated with SHS (the fracture will displace along the screw axis).
Avascular Necrosis (AVN) of the Femoral Head
AVN (osteonecrosis) results from disruption of the blood supply to the femoral head, leading to bone cell death and eventual subchondral collapse and secondary osteoarthritis. Etiologies: femoral neck fracture (post-traumatic, most common), corticosteroids (dose-dependent — risk increases significantly above 20 mg/day prednisone for >3 months), alcohol abuse, sickle cell disease, SLE, HIV/antiretroviral therapy, diving (caisson disease), Gaucher's disease, radiation, idiopathic. Ficat & Arlet classification: Stage 0 = preclinical (only biopsy); Stage I = pre-radiographic (normal X-ray, positive MRI/bone scan); Stage II = abnormal X-ray (sclerosis, cysts) but no collapse; Stage III = subchondral collapse (crescent sign on X-ray); Stage IV = secondary OA with acetabular involvement.
Treatment: Pre-collapse (Ficat I–II): core decompression (drilling a channel into the necrotic area to relieve intraosseous pressure and stimulate revascularization — can add bone graft/BMP/stem cells). Non-vascularized or vascularized fibular bone grafting (free vascularized fibula graft — Urbaniak technique). Bisphosphonates may delay collapse. Post-collapse (Ficat III–IV): total hip arthroplasty is the definitive treatment. Young patients with limited head involvement and preserved joint congruency may be candidates for rotational osteotomy (Sugioka).
Hip Dislocation
Posterior dislocation (~90%) — classically from a dashboard injury (knee strikes dashboard, axial force drives the femur posteriorly out of the acetabulum). The leg is held in a characteristic position: flexed, adducted, and internally rotated. Associated injuries: sciatic nerve palsy (10–20% — usually the peroneal division, leading to foot drop), posterior wall acetabular fracture (up to 70%), femoral head fracture (Pipkin classification). Anterior dislocation (~10%) — forced abduction and external rotation. The leg is held in extension, abduction, and external rotation. Associated with femoral head impaction fractures. Both types require emergent closed reduction within 6 hours to minimize AVN risk (each hour of delay increases AVN rate). Post-reduction CT scan is mandatory to assess for acetabular fractures, loose bodies, and concentric reduction.
09 Knee Pathology & Fractures Lower Extremity
ACL Tear
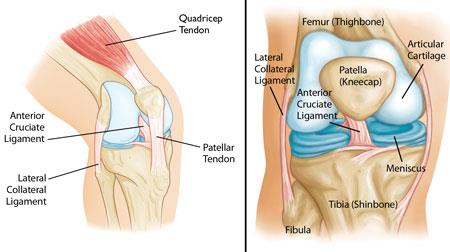
The anterior cruciate ligament (ACL) is the primary restraint to anterior tibial translation and a secondary stabilizer to rotation. It originates from the posteromedial aspect of the lateral femoral condyle and inserts on the anterior tibial spine. Mechanism of injury: non-contact pivoting/deceleration (70%), valgus + rotation, or hyperextension. Physical exam: positive Lachman (most sensitive), positive anterior drawer, pivot shift. MRI confirms the diagnosis and evaluates for associated injuries: meniscal tears (up to 50%), MCL injury ("unhappy triad" = ACL + MCL + medial meniscus), bone bruises of the lateral femoral condyle and posterior lateral tibial plateau (pathognomonic pattern).
Treatment: Non-operative: acceptable for low-demand, older patients without functional instability; involves PT focusing on hamstring strengthening and proprioception. ACL reconstruction is indicated for young/active patients, those with functional instability (giving way episodes), and associated repairable meniscal tears. Graft options: bone-patellar tendon-bone (BTB) autograft (gold standard for return to cutting/pivoting sports — rigid bone-to-bone healing, higher anterior knee pain), hamstring tendon autograft (gracilis + semitendinosus, quadrupled — less donor site morbidity, soft tissue-to-bone healing slower), quadriceps tendon autograft (gaining popularity — thick, strong graft with less anterior knee pain than BTB), and allograft (no donor site morbidity, but higher re-tear rate in young athletes <25 years — Kaeding et al., 2011). Tunnel placement is the most critical factor for success — the femoral tunnel should be in the center of the anatomic ACL footprint.
PCL, MCL, LCL & Multi-Ligament Knee Injuries
PCL tear: Most common mechanism is a dashboard injury (posterior force on the proximal tibia with the knee flexed) or hyperflexion. Isolated PCL tears are often managed non-operatively with quadriceps-intensive rehabilitation. Surgical reconstruction is considered for grade III injuries with combined instability, persistent functional limitation, or multi-ligament injury. MCL tear: Valgus stress mechanism. Graded I–III by medial joint opening (I = 0–5 mm, II = 5–10 mm, III = >10 mm). Isolated MCL tears heal well with protected weight-bearing and hinged knee brace; surgery is rarely needed for isolated injuries. LCL / posterolateral corner (PLC) injury: Varus or hyperextension force. PLC structures include the LCL, popliteus tendon, and popliteofibular ligament. PLC injuries almost always require surgical repair or reconstruction, especially when combined with cruciate tears. Must check peroneal nerve function.
Knee dislocation (multi-ligament injury involving ≥2 cruciate and/or collateral ligaments) is an emergency requiring immediate assessment for popliteal artery injury — occurs in 5–40% of knee dislocations. Mandatory vascular exam: ABI, followed by CTA if ABI <0.9 or any clinical concern. Spontaneous reduction may have occurred before presentation — a multi-ligament injury without a documented dislocation should still prompt vascular assessment.
Meniscal Tears
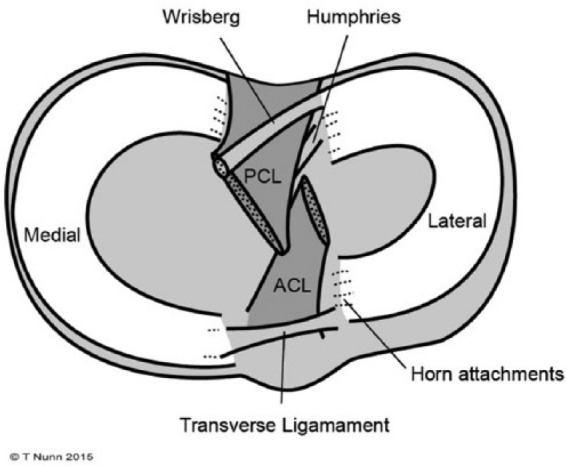
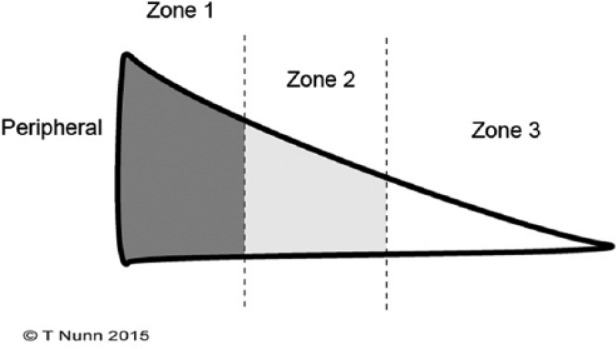
The menisci (medial and lateral) are C-shaped fibrocartilaginous structures that deepen the tibial plateau, distribute load (transmit 50–70% of the load in extension, up to 85% in flexion), absorb shock, and aid in joint lubrication and proprioception. The vascular zones are critical for treatment decisions: red-red zone (peripheral 1/3 — vascularized, good healing potential, suitable for repair), red-white zone (middle 1/3 — intermediate vascularity, repair may succeed), white-white zone (inner 1/3 — avascular, no healing capacity, typically partial meniscectomy). Tear patterns: vertical longitudinal (bucket-handle tears cause locked knee), radial, horizontal cleavage, flap/oblique, complex/degenerative.
Treatment: Arthroscopic partial meniscectomy for irreparable tears in the white-white zone — remove only the unstable fragment, preserve as much meniscus as possible (total meniscectomy accelerates OA). Meniscal repair for tears in the red-red or red-white zone, especially in young patients and when performed concurrently with ACL reconstruction (the ACL drilling creates a healing response that improves meniscal repair success). Repair techniques: inside-out (gold standard), outside-in, all-inside (meniscal repair devices — FasT-Fix, RapidLoc). Meniscal transplantation (allograft) is considered for young patients with a complete or near-complete meniscectomy who develop pain from the meniscus-deficient compartment, before advanced chondral damage occurs.
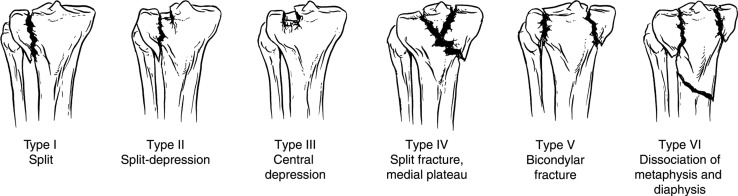
Tibial Plateau Fractures — Schatzker Classification
| Schatzker Type | Description | Mechanism / Population | Treatment |
|---|---|---|---|
| I | Lateral split (wedge fragment) | Valgus force in younger patients with strong subchondral bone | ORIF if displaced >2 mm or condylar widening >5 mm; percutaneous screws often sufficient |
| II | Lateral split-depression | Most common type; valgus force in older/osteoporotic bone | ORIF: elevate depressed articular fragment, bone graft void, buttress plate (lateral periarticular plate) |
| III | Lateral pure depression | Low-energy in osteoporotic bone | Percutaneous elevation + bone graft/cement + screws; or ORIF if depression >5 mm |
| IV | Medial plateau fracture (split or depression) | Higher energy, varus force | ORIF mandatory — medial plateau bears 60% of load; associated with peroneal nerve injury, popliteal artery injury |
| V | Bicondylar (medial + lateral split) | High energy (axial load) | Dual-column fixation (medial + lateral plates); staged approach if severe soft tissue injury |
| VI | Plateau fracture with metadiaphyseal dissociation | Highest energy | Spanning external fixation initially; definitive ORIF or hybrid fixation after soft tissue recovery |
Knee Osteoarthritis
OA is the most common joint disease, affecting >30 million Americans. The knee is the most commonly affected large joint. Pathology: progressive loss of articular cartilage, subchondral bone sclerosis, osteophyte formation, synovial inflammation. Weight-bearing radiographs show: joint space narrowing, subchondral sclerosis, osteophytes, subchondral cysts. Kellgren-Lawrence grading: 0 = normal, 1 = doubtful (minute osteophytes), 2 = mild (definite osteophytes, possible joint space narrowing), 3 = moderate (moderate joint space narrowing, some sclerosis), 4 = severe (bone-on-bone, large osteophytes, sclerosis, deformity). Medial compartment OA is most common, leading to varus (bow-legged) deformity.
Non-operative management: Weight loss (each 1 lb of weight loss = 4 lbs less force across the knee), physical therapy (quadriceps strengthening), activity modification, acetaminophen, topical NSAIDs, oral NSAIDs (ibuprofen, naproxen, meloxicam — shortest duration at lowest effective dose), intra-articular corticosteroid injection (3–4 months relief), hyaluronic acid injection (viscosupplementation — Synvisc, Euflexxa; modest benefit, insurance coverage varies), unloader brace for unicompartmental disease. Surgical: Arthroscopy for OA has no benefit over sham surgery (Kirkley et al., 2008). High tibial osteotomy (HTO) for young patients (<60) with isolated medial compartment OA and varus alignment. Total knee arthroplasty is the definitive treatment for end-stage disease.
10 Ankle & Foot Lower Extremity
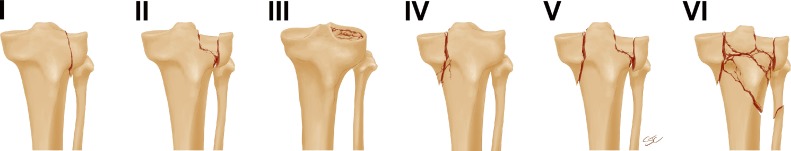
Ankle Fractures — Weber Classification
The Weber classification is based on the level of the fibular fracture relative to the syndesmosis (the ligamentous complex connecting the distal tibia and fibula — anterior inferior tibiofibular ligament [AITFL], posterior inferior tibiofibular ligament [PITFL], transverse ligament, and interosseous membrane).
| Weber Type | Fibula Fracture Level | Syndesmosis | Stability | Treatment |
|---|---|---|---|---|
| A | Below the syndesmosis (lateral malleolus tip, avulsion) | Intact | Stable | Usually nonoperative: short leg walking boot or cast |
| B | At the level of the syndesmosis (spiral fracture beginning at the joint line) | May be intact or disrupted | Potentially unstable — stress test needed | Stable (negative stress test, no medial tenderness): nonoperative. Unstable: ORIF |
| C | Above the syndesmosis (proximal fibula — Maisonneuve pattern at extreme) | Disrupted | Unstable | ORIF of fibula + syndesmotic fixation (screws or suture button [TightRope]) |
Lauge-Hansen Classification
The Lauge-Hansen system describes the mechanism of ankle fractures based on foot position (first word: supination or pronation) and direction of force (second word: adduction, external rotation, or abduction). It predicts the pattern of ligamentous and bony injury in a sequential manner.
| Type | Sequence of Injury | Frequency |
|---|---|---|
| Supination–External Rotation (SER) | Stage I: AITFL tear → Stage II: spiral oblique fibula fracture at syndesmosis → Stage III: PITFL tear or posterior malleolus fracture → Stage IV: deltoid ligament tear or medial malleolus fracture | ~60% (most common) |
| Supination–Adduction (SAD) | Stage I: lateral ligament tear or transverse lateral malleolus avulsion → Stage II: vertical medial malleolus fracture | ~20% |
| Pronation–External Rotation (PER) | Stage I: medial malleolus transverse fracture or deltoid tear → Stage II: AITFL tear → Stage III: high spiral fibula fracture (above syndesmosis) → Stage IV: PITFL tear or posterior malleolus fracture | ~10% |
| Pronation–Abduction (PAB) | Stage I: medial malleolus fracture or deltoid tear → Stage II: AITFL tear → Stage III: comminuted/butterfly fibula fracture at or above syndesmosis | ~10% |
Achilles Tendon Rupture
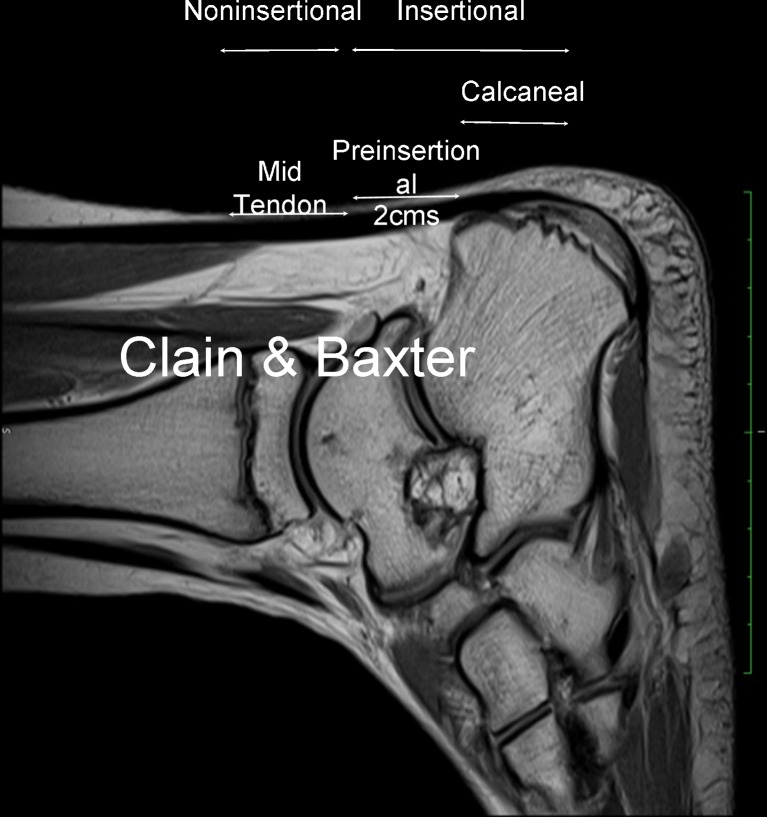
Typically occurs in 30–50-year-old men during sports ("weekend warrior" demographic). The tendon ruptures 2–6 cm above its calcaneal insertion — the watershed zone of poorest vascularity. Patients report a "pop" or sensation of being kicked. Physical exam: Palpable gap in the tendon, positive Thompson test (squeezing the calf does not produce plantar flexion of the foot), loss of resting tension (foot hangs in dorsiflexion when prone). Treatment: Operative repair (Krackow suture technique, with or without augmentation) vs functional non-operative treatment (equinus casting followed by progressive dorsiflexion, early weight-bearing with a boot and heel wedges). Landmark studies (STAR trial — Costa et al., BMJ 2020) suggest similar outcomes with accelerated functional rehabilitation vs surgery, though re-rupture rates are slightly higher non-operatively (4–6% vs 1–2%). Operative repair is generally preferred for young, active patients and competitive athletes.
Lisfranc Injury

Injuries to the tarsometatarsal (TMT) joint complex, named for the Lisfranc ligament — a strong ligament connecting the medial cuneiform to the base of the 2nd metatarsal. This is the "keystone" of the midfoot. Mechanism: axial load on a plantarflexed foot (classically a horse stirrup injury, now more often motor vehicle or athletic). Frequently missed — subtle injuries present with midfoot swelling, plantar ecchymosis (pathognomonic), and inability to bear weight. Diagnosis: Weight-bearing AP foot X-ray: look for diastasis between the 1st and 2nd metatarsal bases (>2 mm) and loss of alignment between the medial border of the 2nd metatarsal and the medial border of the middle cuneiform. CT scan for subtle or equivocal cases. Treatment: Purely ligamentous injuries: primary arthrodesis (fusion) of the TMT joints (better outcomes than ORIF for ligamentous injuries — Ly & Coetzee, 2006). Fracture-dislocations: anatomic ORIF with screws and/or plates.
Calcaneal Fractures
The most commonly fractured tarsal bone, usually from a fall from height (axial loading). 10% bilateral, 10% associated with lumbar spine compression fracture (always check the spine). The Bohler's angle (normally 20–40°) measures the relationship between the posterior facet and the tuberosity — a decreased or negative Bohler's angle indicates subtalar joint depression and predicts worse outcomes. The Sanders classification (based on coronal CT through the widest part of the posterior facet) guides treatment: Type I = non-displaced → nonoperative; Type II = 2 fragments → ORIF; Type III = 3 fragments → ORIF; Type IV = >3 fragments (comminuted) → primary subtalar arthrodesis. The extensile lateral approach (L-shaped incision) provides excellent visualization but has wound complication rates of 10–25%; sinus tarsi approach (minimally invasive) has lower wound complications and is increasingly favored for Type II fractures.
11 Cervical Spine Spine
Cervical Spine Fractures
Atlas (C1) fractures — Jefferson fracture: Axial load (diving, fall onto head) fractures the C1 ring at 2–4 points. Lateral mass overhang >6.9 mm (combined bilateral) on open-mouth odontoid view indicates transverse ligament rupture (unstable — Rule of Spence). Stable Jefferson: rigid cervical collar. Unstable: halo vest or surgical fusion (C1–C2).
Axis (C2) fractures: Three main types: Odontoid (dens) fractures — Anderson & D'Alonzo classification: Type I = tip avulsion (rare, generally stable, may be unstable if associated with occipitocervical dissociation); Type II = base of the dens (most common and most problematic — high nonunion rate of 20–40% in elderly with displacement >5 mm, age >50; treatment: rigid collar for non-displaced, halo or anterior odontoid screw for displaced, posterior C1–C2 fusion [Harms technique: C1 lateral mass screws + C2 pedicle or pars screws] for elderly/nonunion); Type III = extends into the C2 body (good cancellous healing potential, usually treated in cervical collar). Hangman's fracture (bilateral C2 pars interarticularis fracture) — Levine classification: Type I = <3 mm displacement, no angulation → rigid collar; Type II = >3 mm or >11° angulation → halo vs surgical fusion; Type IIA = minimal displacement but severe angulation (flexion-distraction) → do NOT apply traction → halo or surgery; Type III = bilateral facet dislocation → surgery.
Subaxial cervical fractures (C3–C7): Classified by the Subaxial Cervical Spine Injury Classification (SLIC) system, which scores morphology (compression = 1, burst = 2, distraction = 3, translation/rotation = 4), disco-ligamentous complex integrity (intact = 0, indeterminate = 1, disrupted = 2), and neurological status (intact = 0, root injury = 1, complete cord = 2, incomplete cord = 3, ongoing compression with neuro deficit = +1). Score ≤3: nonoperative; Score = 4: surgeon discretion; Score ≥5: surgical stabilization.
Facet injuries: Unilateral facet dislocation = ~25% anterior subluxation on lateral X-ray + radiculopathy; bilateral facet dislocation = ~50% anterior subluxation + high risk of spinal cord injury. Treatment: closed reduction with traction (after MRI to rule out disc herniation in alert patients), then anterior or posterior stabilization.
Cervical Myelopathy
Cervical spondylotic myelopathy (CSM) is the most common cause of spinal cord dysfunction in adults >55. Caused by spinal cord compression from disc osteophyte complexes, ligamentum flavum hypertrophy, and OPLL (ossification of the posterior longitudinal ligament). Symptoms: progressive gait difficulty (broad-based, spastic), hand clumsiness (loss of fine motor function — difficulty with buttons, dropping objects), upper extremity numbness/weakness, urinary urgency/retention (late). Signs: hyperreflexia, clonus, Hoffmann's sign (flicking the middle fingertip produces thumb/index flexion), Babinski sign, inverted radial reflex (brachioradialis tap produces finger flexion instead of elbow flexion), Lhermitte's sign (neck flexion causes electric shock sensation down the spine). Treatment: Surgery for moderate-severe myelopathy — CSM is progressive and rarely improves spontaneously. Anterior cervical discectomy and fusion (ACDF) for 1–2 level anterior compression. Anterior cervical corpectomy and fusion (ACCF) for retrovertebral compression. Laminoplasty (expansive open-door technique) or laminectomy with posterior fusion for multi-level compression (≥3 levels) with maintained cervical lordosis.
Cervical Radiculopathy
Nerve root compression from disc herniation (younger patients) or foraminal stenosis from osteophytes (older patients). The most commonly affected levels are C5–C6 (C6 root) and C6–C7 (C7 root). Key dermatome/myotome relationships:
| Root | Disc Level | Motor Deficit | Reflex | Sensory Distribution |
|---|---|---|---|---|
| C5 | C4–C5 | Deltoid, biceps weakness | Biceps ↓ | Lateral arm (regimental badge area) |
| C6 | C5–C6 | Wrist extension, biceps weakness | Brachioradialis ↓ | Lateral forearm, thumb, index finger |
| C7 | C6–C7 | Triceps, wrist flexion, finger extension | Triceps ↓ | Middle finger |
| C8 | C7–T1 | Finger flexion (FDP), intrinsics | None reliable | Ring and small fingers, medial forearm |
| T1 | T1–T2 | Intrinsic hand muscles (interossei) | None | Medial arm |
Treatment: 80–90% resolve with conservative management: activity modification, NSAIDs, oral corticosteroid taper (methylprednisolone dose pack), cervical epidural steroid injection (fluoroscopic-guided transforaminal or interlaminar). PT with cervical traction. Surgery (ACDF, posterior cervical foraminotomy) for failure of 6–12 weeks conservative treatment, progressive neurological deficit, or intractable pain.
12 Thoracolumbar Spine Spine
Thoracolumbar Fractures
The thoracolumbar junction (T11–L2) is the most common site of spinal fractures — it is the transition zone between the rigid thoracic spine (stabilized by the rib cage) and the mobile lumbar spine. The Denis three-column model divides the spine into: anterior column (anterior 2/3 of vertebral body and disc, anterior longitudinal ligament), middle column (posterior 1/3 of vertebral body and disc, posterior longitudinal ligament), and posterior column (pedicles, facets, laminae, spinous process, posterior ligamentous complex [PLC — supraspinous ligament, interspinous ligament, ligamentum flavum, facet joint capsules]). Instability requires involvement of ≥2 columns.
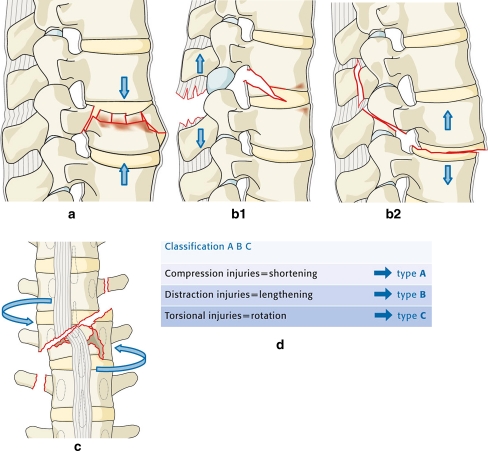
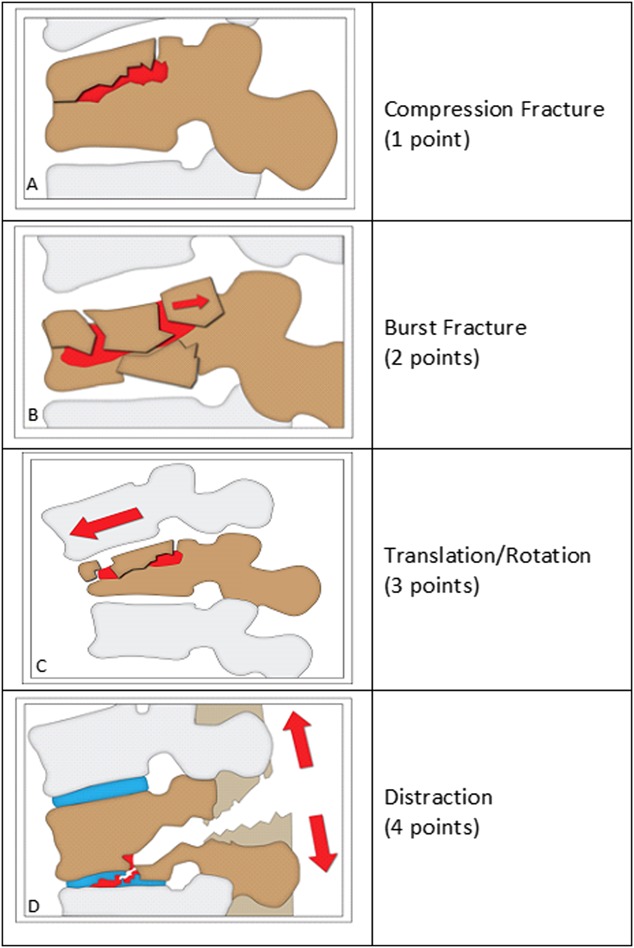
Thoracolumbar Injury Classification and Severity Score (TLICS)
| Parameter | Finding | Points |
|---|---|---|
| Morphology | Compression | 1 |
| Burst | 2 | |
| Translation/rotation | 3 | |
| Distraction | 4 | |
| PLC Integrity | Intact | 0 |
| Indeterminate / suspected | 2 | |
| Disrupted | 3 | |
| Neurological Status | Intact | 0 |
| Nerve root injury | 2 | |
| Complete cord / conus | 2 | |
| Incomplete cord / conus | 3 |
Score ≤3: nonoperative (TLSO brace). Score = 4: surgeon discretion. Score ≥5: operative stabilization. Neurological deficit from canal compromise generally warrants surgical decompression.
Fracture Types
Compression fracture: Flexion mechanism, anterior column failure only. Loss of anterior vertebral body height with intact posterior wall and middle column. Kyphotic angulation >30° or >50% height loss suggests PLC injury — obtain MRI. Treatment: TLSO brace for 8–12 weeks if stable; vertebral augmentation (kyphoplasty or vertebroplasty) for intractable pain in osteoporotic fractures. Burst fracture: Axial compression mechanism, anterior and middle column failure. Retropulsed bone fragments may compromise the spinal canal. Treatment depends on neurological status, PLC integrity, and degree of canal compromise — may be braced if neurologically intact with intact PLC, or may require surgical stabilization. Flexion-distraction (Chance fracture): Distraction mechanism with failure of the posterior and middle columns in tension — "seatbelt fracture." May be purely bony (extends through the vertebral body — better prognosis, can heal in extension brace) or ligamentous/combined (PLC disrupted — requires surgical stabilization). Associated with abdominal injuries (small bowel, mesentery, pancreas) in up to 50% — CT abdomen/pelvis required. Fracture-dislocation: All three columns fail, with translation or rotation — most unstable pattern. High rate of neurological injury. Requires surgical stabilization (posterior pedicle screw fixation ± anterior column support).
Lumbar Spinal Stenosis
Central canal narrowing causing neurogenic claudication — bilateral leg pain, heaviness, and weakness with walking or standing that is relieved by sitting or leaning forward (flexion opens the central canal). Distinguished from vascular claudication by the "shopping cart sign" (patients lean forward over shopping carts for relief). Diagnosis: MRI shows central canal stenosis, often at multiple levels (L3–L4, L4–L5 most common). The SPORT trial (Weinstein et al., NEJM 2008) demonstrated that surgical decompression provided significantly better outcomes than conservative treatment for moderate-severe stenosis at 4-year follow-up, though crossover rates were high. Treatment: Conservative: PT, epidural steroid injections, activity modification. Surgical: laminectomy (decompression of the central canal) — the standard procedure. If associated instability or spondylolisthesis: laminectomy with posterolateral fusion ± pedicle screw instrumentation. Interspinous process spacers (Superion, X-STOP) are a minimally invasive option for mild-moderate stenosis.
13 Degenerative Disc Disease & Spondylolisthesis Spine
Degenerative Disc Disease (DDD)
The intervertebral disc consists of the nucleus pulposus (gelatinous center, high water content, provides compressive resistance) and the annulus fibrosus (concentric collagen lamellae providing tensile strength). Disc degeneration begins in the 2nd decade — progressive desiccation, loss of disc height, annular fissuring, and altered biomechanics. The disc is largely avascular (nutrition by diffusion from the vertebral endplates), so healing capacity is minimal. Disc herniation refers to displacement of disc material beyond the normal disc space margin: protrusion (base wider than dome), extrusion (dome wider than base), sequestration (free fragment, separated from parent disc).
Lumbar disc herniation: Most commonly posterolateral (the posterior longitudinal ligament is weakest laterally). L4–L5 and L5–S1 account for 95% of lumbar herniations. A posterolateral herniation at L4–L5 compresses the traversing L5 nerve root (not the exiting L4 root). A far lateral (foraminal) herniation at L4–L5 compresses the exiting L4 root. Key lumbar root findings:
| Root | Disc Level | Motor | Reflex | Sensory |
|---|---|---|---|---|
| L4 | L3–L4 | Quadriceps (knee extension), tibialis anterior | Patellar ↓ | Medial leg |
| L5 | L4–L5 | EHL (great toe dorsiflexion), tibialis anterior, gluteus medius (hip abduction) | None reliable (medial hamstring in some) | Lateral leg, dorsum of foot, first web space |
| S1 | L5–S1 | Gastrocnemius/soleus (plantarflexion), peroneus longus/brevis (eversion) | Achilles ↓ | Lateral foot, sole |
Treatment: 90% of lumbar disc herniations resolve with conservative management: NSAIDs, activity modification (avoid prolonged sitting), PT (McKenzie extension exercises, core stabilization), and epidural steroid injections. Surgical indication: microdiscectomy (standard of care) for progressive neurological deficit, cauda equina syndrome (surgical emergency — saddle anesthesia, urinary retention, bilateral leg weakness; requires emergent decompression within 24–48 hours), or failure of 6–12 weeks of conservative treatment with persistent radiculopathy. The SPORT trial showed microdiscectomy provided faster recovery, though long-term (8-year) outcomes were similar between operative and nonoperative groups.
Spondylolisthesis
Anterior displacement of one vertebra relative to the one below. Wiltse classification by etiology: Type I = dysplastic (congenital facet abnormality), Type II = isthmic (pars interarticularis defect — spondylolysis; most common at L5–S1 in young athletes, especially gymnasts and football linemen), Type III = degenerative (facet arthropathy and disc degeneration; most common at L4–L5 in elderly women), Type IV = traumatic, Type V = pathologic (tumor, infection), Type VI = iatrogenic (post-surgical). Meyerding grading by percentage of slip: Grade I = 0–25%, Grade II = 25–50%, Grade III = 50–75%, Grade IV = 75–100%, Grade V = spondyloptosis (>100%).
Treatment of isthmic spondylolisthesis: Low-grade (I–II), asymptomatic or mild symptoms: activity modification, PT (core and hamstring flexibility), bracing for symptomatic pars defect in adolescents (TLSO brace for 3–6 months may allow pars healing). Surgery for refractory symptoms, neurological deficit, or high-grade slip: posterolateral or interbody fusion (PLIF, TLIF) with pedicle screw instrumentation. Reduction of slip is controversial for high-grade — incomplete reduction is often accepted to avoid neurological injury (L5 root stretch). Treatment of degenerative spondylolisthesis: The SPORT trial showed surgery (decompressive laminectomy + fusion) was superior to conservative treatment for symptomatic degenerative spondylolisthesis with stenosis at 4 years.
14 Pelvic & Acetabular Fractures Trauma
Pelvic Ring Fractures — Young-Burgess Classification
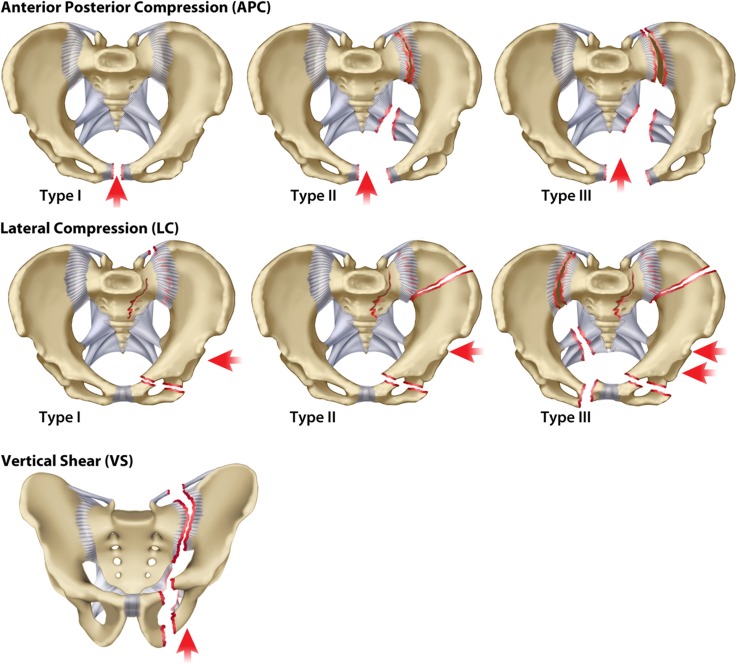
The pelvis is a ring structure: disruption at one point implies disruption (bony or ligamentous) at a second point. The posterior ring (sacroiliac joints, sacrum, posterior ligamentous complex) contributes ~60% of pelvic stability. Pelvic fractures are classified by the mechanism of injury.
| Type | Mechanism | Pattern | Stability | Hemorrhage Risk |
|---|---|---|---|---|
| Lateral Compression (LC) | Side impact (MVC T-bone, fall onto side) | LC-I: sacral compression fracture + ipsilateral pubic rami fractures. LC-II: crescent (iliac wing) fracture. LC-III: LC-I or LC-II + contralateral open-book (windswept pelvis) | LC-I: stable. LC-II: rotationally unstable. LC-III: rotationally + vertically unstable | LC-I lowest. LC-III very high |
| Anteroposterior Compression (APC) | Head-on collision, crush, straddle | APC-I: symphysis diastasis <2.5 cm, posterior ligaments intact. APC-II: diastasis >2.5 cm, anterior SI ligaments torn, posterior SI intact ("open book"). APC-III: complete SI disruption | APC-I: stable. APC-II: rotationally unstable. APC-III: rotationally + vertically unstable | APC-II/III: very high (posterior venous plexus disruption) |
| Vertical Shear (VS) | Fall from height, axial load through one limb | Vertical displacement of hemipelvis through posterior ring (SI joint or sacral fracture) + anterior ring (rami or symphysis) | Rotationally + vertically unstable | Very high |
| Combined Mechanism (CM) | High-energy, mixed | Features of multiple patterns | Unstable | High |
Pelvic fractures can cause massive hemorrhage (up to 3–5 L of blood loss into the retroperitoneum). Sources: posterior pelvic venous plexus (~85%), arterial bleeders (~15%), and cancellous bone surfaces. Initial steps: (1) Pelvic binder (T-POD, SAM Pelvic Sling) or sheet wrap at the level of the greater trochanters — reduces pelvic volume and tamponades venous bleeding. (2) Massive transfusion protocol (1:1:1 PRBC:FFP:platelets). (3) Preperitoneal pelvic packing (PPP) — increasingly performed in the resuscitation bay for ongoing instability; direct pressure on the presacral space. (4) Angiography and embolization for arterial bleeders (CT blush or persistent instability despite binder and packing). (5) REBOA (Resuscitative Endovascular Balloon Occlusion of the Aorta) — Zone III deployment as a bridge. (6) Definitive fixation after hemodynamic stabilization — external fixation (anterior frame) acutely, then delayed posterior fixation (iliosacral screws) when stable.
Acetabular Fractures — Letournel Classification
The Letournel system describes acetabular fractures based on which columns and walls are involved. The acetabulum is formed by two columns: the anterior column (iliopectineal line on X-ray — pubis to anterior AIIS) and the posterior column (ilioischial line — ischium to greater sciatic notch). The anterior wall and posterior wall are the articular portions of their respective columns.
| Category | Type | Description |
|---|---|---|
| Elementary (5) | Posterior wall | Most common acetabular fracture (~25%); associated with posterior hip dislocation |
| Posterior column | Fracture extends from greater sciatic notch to ischial tuberosity | |
| Anterior wall | Rare; fracture of the anterior articular surface | |
| Anterior column | Fracture extends from iliac crest to pubic rami | |
| Transverse | Divides the acetabulum into superior (iliac) and inferior (ischiopubic) segments | |
| Associated (5) | Posterior column + posterior wall | Combination of posterior column and wall fractures |
| Transverse + posterior wall | Transverse fracture with posterior wall component — second most common | |
| T-type | Transverse fracture with inferior vertical extension (separates anterior and posterior columns inferiorly) | |
| Anterior column + posterior hemitransverse | Anterior column fracture with transverse component through the posterior column | |
| Both columns | All articular segments are dissociated from the intact ilium; pathognomonic "spur sign" on obturator oblique view |
Treatment principles: Anatomic reduction of the articular surface is essential for long-term outcomes. Operative indications: >2 mm articular step-off, hip instability, incarcerated fragments, posterior wall fractures involving >40% of the wall (hip unstable), associated fracture patterns. Surgical approaches: Kocher-Langenbeck (posterior — for posterior wall/column, transverse, T-type), Ilioinguinal (anterior — for anterior column/wall, both column, anterior column + posterior hemitransverse), modified Stoppa (intrapelvic — alternative anterior approach with better visualization of the quadrilateral plate). Timing: ideally within 5–10 days (after resuscitation, before callus formation complicates reduction). In elderly patients with severe comminution and osteoporosis, acute THA with acetabular reconstruction may be preferable to ORIF.
15 Femoral & Tibial Shaft Fractures Trauma
Femoral Shaft Fractures
High-energy injuries in young adults (MVC, fall from height); low-energy in elderly with osteoporosis. The femur is the longest and strongest bone in the body. Fractures cause significant hemorrhage (each femur fracture can lose 1–1.5 L of blood into the thigh). Winquist-Hansen classification by comminution: Type 0 = no comminution, Type I = small butterfly fragment (<25% cortex), Type II = butterfly 25–50% cortex, Type III = >50% cortex comminution (no cortical contact after nailing), Type IV = segmental bone loss (circumferential comminution).
Treatment: Antegrade intramedullary nailing (IMN) is the gold standard for virtually all femoral shaft fractures. Entry point: piriformis fossa (standard) or greater trochanter tip (trochanteric entry nail — Synthes Expert, Smith+Nephew T2). Reaming the canal before nail insertion improves cortical blood flow long-term and allows a larger nail diameter. Static locking (interlocking screws at both ends) controls length and rotation. Dynamization (removal of one set of interlocking screws at 3–4 months) may be performed for delayed union to allow axial compression. Retrograde nailing (entry through the intercondylar notch via the knee) is an alternative for ipsilateral femoral neck/shaft combination, distal fractures, bilateral fractures (allows nailing in supine position), obesity, or pregnant patients. Plate fixation (submuscular/bridge plating) is reserved for narrow canal, adolescents with open physis, severe deformity, or periprosthetic fractures around implants. External fixation: damage control for polytrauma (see below).
Tibial Shaft Fractures
The tibia is the most commonly fractured long bone. The anteromedial surface is subcutaneous (no muscle coverage), which explains the high rate of open fractures (20–30% of tibial shaft fractures are open). Treatment: Non-displaced, stable, closed fractures: long leg cast (bent at knee to prevent rotation) for 4–6 weeks, then patellar tendon-bearing (PTB) short leg cast/functional brace for another 6–12 weeks. Operative indications: displaced, unstable, open fracture, associated compartment syndrome, floating knee (ipsilateral femur + tibia fractures), polytrauma. Intramedullary nailing is the gold standard for displaced fractures — entry at the proximal tibia through a suprapatellar or infrapatellar approach. Reamed nailing provides better union rates than unreamed nailing for closed fractures. For open fractures, either reamed or unreamed nailing is acceptable (the SPRINT trial — Study to Prospectively Evaluate Reamed Intramedullary Nails in Tibial Fractures, 2008 — showed reamed nailing reduced the need for secondary procedures). External fixation for severe open fractures (temporary or definitive in Gustilo IIIB/C). Plate fixation (MIPO — minimally invasive plate osteosynthesis) for proximal or distal metaphyseal fractures where nailing is difficult.
16 Open Fractures Trauma
Open fractures involve a breach in the skin and soft tissues communicating with the fracture site. The management principles are detailed in Section 03 (Gustilo-Anderson classification and acute management protocol). This section covers additional operative principles and special considerations.
Surgical Debridement Principles
The cornerstone of open fracture management is thorough, systematic debridement. Skin: Extend the wound to visualize the entire zone of injury; excise non-viable skin edges (minimal). Subcutaneous tissue and fascia: Excise all non-viable tissue. Muscle: Apply the "4 C's" test — Color (viable muscle is beefy red, not pale or dark), Consistency (not mushy), Contractility (twitches when stimulated with forceps or cautery), and Capacity to bleed (viable muscle bleeds when cut). Bone: Remove small, completely devitalized cortical fragments that are not attached to soft tissue. Preserve larger fragments and those with periosteal attachment. Neurovascular structures: Preserve and tag injured nerves and vessels. Repeat debridement at 48–72 hours is standard for type III injuries to reassess tissue viability.
Soft Tissue Coverage
The goal is definitive soft tissue coverage within 72 hours to 7 days of injury. Options by defect location (tibial shaft — the most common site requiring flap coverage): Proximal third: gastrocnemius rotational flap (medial head most commonly used). Middle third: soleus rotational flap. Distal third: free tissue transfer (free latissimus dorsi, free rectus abdominis, free anterolateral thigh [ALT] flap) — requires microsurgical anastomosis. Negative pressure wound therapy (VAC — vacuum-assisted closure) is a temporizing measure between debridements but should not delay definitive coverage.
Antibiotic Prophylaxis Duration
Current evidence supports 24 hours of prophylactic antibiotics after definitive wound management for open fractures (Lack et al., 2015). Extended courses do not reduce infection rates and increase antibiotic resistance. For contaminated wounds, additional targeted antibiotics may be appropriate based on the contamination source.
17 Pathologic Fractures & Polytrauma Trauma
Pathologic Fractures
A pathologic fracture occurs through bone weakened by an underlying process — most commonly metastatic disease (breast, prostate, lung, kidney, thyroid — "BLT with a Kosher Pickle"), multiple myeloma, osteoporosis, Paget's disease, or primary bone tumors. Impending fracture: The Mirels' scoring system predicts pathologic fracture risk (site, pain, type of lesion, size — score ≥9 out of 12 warrants prophylactic fixation). Workup: Obtain staging studies before fixation (unless the patient is in immediate need of stabilization): chest CT, bone scan or PET-CT, labs (CBC, CMP, alkaline phosphatase, PSA in males, SPEP/UPEP for myeloma), and biopsy if the primary is unknown (biopsy track must be in line with the planned surgical approach). Treatment: Stabilize the fracture to restore function and reduce pain. Options: intramedullary nail (protects the entire bone from additional metastatic fractures), plate fixation (with PMMA cement augmentation for lytic defects), or endoprosthetic reconstruction (proximal femoral replacement for extensive proximal femur disease). Post-operative radiation therapy to treated sites prevents disease progression and improves fixation longevity.
Polytrauma Priorities — Damage Control Orthopedics (DCO)
In the multiply injured patient, the "lethal triad" — hypothermia, acidosis, and coagulopathy — drives mortality. Damage control orthopedics prioritizes physiological resuscitation over definitive fracture fixation in the unstable polytrauma patient. Principles: (1) Hemorrhage control and resuscitation take priority. (2) Temporary stabilization of long bone fractures with external fixation ("ex-fix and resuscitate") rather than prolonged definitive surgery. (3) Pelvic binder or external fixation for unstable pelvic fractures. (4) Fasciotomy if compartment syndrome is suspected. (5) Definitive fixation (conversion to intramedullary nail or ORIF) performed during the "window of opportunity" — typically days 5–10, after resuscitation and before the immunologic "second hit."
The decision between Early Total Care (ETC) — definitive fixation within 24 hours — and DCO depends on patient physiology. ETC is preferred for hemodynamically stable patients and is standard for isolated femoral shaft fractures (early nailing within 24 hours reduces ARDS, fat embolism, and ICU length of stay). DCO is indicated for: ISS >40, hypothermia (<35°C), coagulopathy (PT >1.5x normal), acidosis (pH <7.24, base deficit >-8), massive transfusion (>10 units PRBC), bilateral femur fractures in the hemodynamically borderline patient, and associated severe thoracic injury (bilateral pulmonary contusions, flail chest).
18 Total Hip Arthroplasty (THA) Joint Replacement
Indications
End-stage osteoarthritis (most common), inflammatory arthritis (RA), AVN, post-traumatic arthritis, displaced femoral neck fracture in the elderly, failed prior fixation, dysplasia, and tumor reconstruction. Non-operative management should be exhausted before considering THA (activity modification, weight management, PT, NSAIDs, injections).
Surgical Approaches
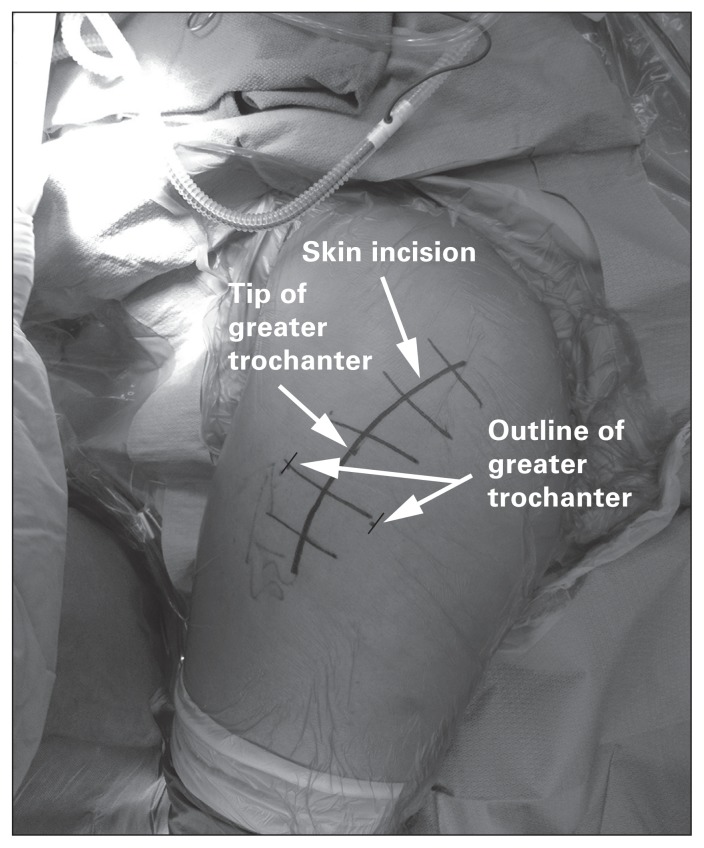
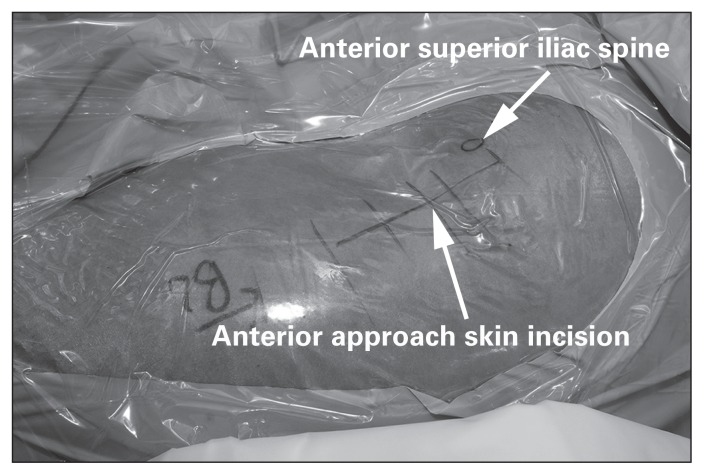
| Approach | Interval | Structures at Risk | Advantages | Disadvantages |
|---|---|---|---|---|
| Posterior (Moore/Southern) | Gluteus maximus splitting, short external rotators detached | Sciatic nerve, short external rotators | Excellent exposure, versatile, familiar; preserves abductors | Higher dislocation rate (mitigated by posterior capsular repair), sciatic nerve risk |
| Direct Anterior (DAA, Smith-Petersen) | Tensor fasciae latae (TFL) and sartorius — true internervous plane (superior gluteal vs femoral nerve) | Lateral femoral cutaneous nerve (LFCN), femoral nerve/artery | Muscle-sparing, lower dislocation rate, faster early recovery, supine fluoroscopy possible | Learning curve, femoral canal exposure challenging in obese, proximal femur fracture risk |
| Anterolateral (Watson-Jones) | Between TFL and gluteus medius | Superior gluteal nerve (limits proximal dissection to 5 cm above greater trochanter tip) | Moderate exposure, lower dislocation than posterior | Abductor damage risk, Trendelenburg limp |
| Direct Lateral (Hardinge) | Anterior third of gluteus medius/minimus detached from greater trochanter | Superior gluteal nerve | Good exposure, low dislocation rate | Abductor detachment → Trendelenburg limp (5–20%) |
Implant Components
A THA has four components: (1) Acetabular cup — most commonly an uncemented titanium hemispherical shell with porous surface (press-fit, ± supplemental screws) + a polyethylene, ceramic, or metal liner. (2) Femoral stem — uncemented (press-fit with porous coating for bone ongrowth/ingrowth) or cemented (PMMA cement fixation — preferred in elderly with Dorr type C femurs and narrow, osteoporotic canals). Common stems: Corail (DePuy), Taperloc (Biomet), Accolade II (Stryker), Exeter (cemented). (3) Femoral head — metal (CoCr) or ceramic (alumina, BIOLOX delta). Sizes 28 mm, 32 mm, 36 mm, 40 mm — larger heads reduce dislocation risk but increase volumetric wear and risk of trunionosis. (4) Bearing surface (liner) — determines wear characteristics.
Bearing Surfaces
| Bearing | Advantages | Disadvantages | Modern Use |
|---|---|---|---|
| Metal-on-Polyethylene (MoP) | Long track record, low cost, forgiving of malpositioning | Polyethylene wear → osteolysis; conventional PE worse than XLPE | XLPE (highly cross-linked polyethylene) with Vitamin E stabilization is the most widely used modern bearing |
| Ceramic-on-Polyethylene (CoP) | Lower wear than MoP, excellent wettability | Ceramic fracture (rare with modern alumina matrix composites, ~0.004%) | Increasingly popular, especially with XLPE |
| Ceramic-on-Ceramic (CoC) | Lowest volumetric wear, bioinert debris | Squeaking (1–10%), stripe wear, ceramic fracture, unforgiving of malpositioning | Used in young, active patients |
| Metal-on-Metal (MoM) | Low volumetric wear, large head sizes possible | Adverse local tissue reactions (ALTR), metallosis, pseudotumors, elevated serum cobalt/chromium, carcinogenesis concerns | Largely abandoned (ASR recall, DePuy 2010). Only used in select hip resurfacing |
THA Complications
Dislocation: 2–5% overall; highest with posterior approach (mitigated by posterior capsular repair and larger heads). Most dislocations are posterior — hip is flexed, adducted, and internally rotated. Prevention: precautions (avoid hip flexion >90°, adduction past midline, internal rotation), larger femoral heads (36 mm+), dual-mobility cups (articulation within an articulation — increasingly used for high-risk patients). Periprosthetic joint infection (PJI): See Section 20. Leg length discrepancy (LLD): Ideally <1 cm; intraoperative assessment with templating, shuck test, and landmarks. Periprosthetic fracture: See Section 20. DVT/PE: Chemoprophylaxis is mandatory — aspirin 81 mg BID (low-risk) or enoxaparin 40 mg daily or rivaroxaban 10 mg daily (higher-risk) for 2–6 weeks post-op. Heterotopic ossification (HO): Ectopic bone formation in soft tissues; prophylaxis with indomethacin 75 mg daily for 6 weeks or single-dose radiation (700 cGy) within 72 hours post-op.
19 Total Knee Arthroplasty (TKA) Joint Replacement
Indications & Planning
End-stage knee OA (most common), inflammatory arthritis, post-traumatic arthritis, and severe valgus/varus deformity with pain. Pre-operative planning includes weight-bearing full-length standing radiographs (hip-to-ankle mechanical axis film) to assess alignment, templating, and evaluation of bone stock and deformity.
Alignment Philosophy
The historic goal has been mechanical alignment — restoring the mechanical axis (a straight line from the center of the femoral head to the center of the ankle) to neutral (0 ± 3°). Femoral component: cut at 5–7° of valgus relative to the femoral shaft axis to produce a cut perpendicular to the mechanical axis. Tibial component: cut perpendicular to the tibial mechanical axis, with 3–7° of posterior slope. Kinematic alignment is an alternative philosophy that aims to restore the patient's constitutional (pre-disease) joint line obliquity rather than forcing neutral mechanical alignment. Proponents argue it produces more natural kinematics and better patient satisfaction; critics cite concerns about long-term survivorship of residual malalignment. Robotic-assisted TKA (Mako/Stryker, ROSA/Zimmer-Biomet, CORI/Smith+Nephew) improves precision of bone cuts and component positioning, though long-term outcome advantages over conventional TKA have not been conclusively demonstrated.
Implant Types
| Type | Constraint | PCL | Indication |
|---|---|---|---|
| Cruciate-retaining (CR) | Minimal | Preserved | Intact, functional PCL; preserves more natural femoral rollback |
| Posterior-stabilized (PS) | Moderate (cam-post mechanism) | Sacrificed | Deficient PCL, inflammatory arthritis, significant deformity; more predictable kinematics |
| Varus-valgus constrained (VVC) | High | Sacrificed | Collateral ligament deficiency, significant bone loss, revision |
| Rotating hinge | Highest | Sacrificed | Global ligament deficiency, massive bone loss, complex revision, tumor reconstruction |
| Unicompartmental (UKA) | Minimal | Preserved | Isolated medial or lateral compartment OA with intact ACL and MCL, correctable deformity, BMI <35; faster recovery than TKA |
The most common bearing in modern TKA is cobalt-chromium femoral component on highly cross-linked polyethylene (XLPE) tibial insert. Tibial component: titanium or CoCr baseplate with modular PE insert. Patellar component: all-polyethylene dome resurfacing (most common); debate persists on whether to routinely resurface the patella, but most high-volume centers resurface. Fixation: cemented TKA is the gold standard (hybrid or fully cemented); cementless TKA (porous-coated titanium for biologic fixation) is gaining acceptance but lacks long-term data comparable to cemented.
TKA Complications
Stiffness: Flexion <90° at 6–12 weeks warrants manipulation under anesthesia (MUA). Instability: Flexion vs extension gap mismatch, ligament incompetence — may require revision to higher-constraint implant. Periprosthetic fracture: Supracondylar femur fractures are most common — treatment depends on implant stability and fracture location (retrograde nail, plate, or revision). Extensor mechanism disruption: Patellar tendon rupture, quadriceps tendon rupture, patellar fracture — devastating complications, often require allograft reconstruction. Aseptic loosening: Most common cause of late TKA failure — polyethylene wear debris triggers osteolysis. Component malalignment: Varus tibial component or internally rotated femoral component leads to poor outcomes and early failure.
20 Revision Arthroplasty, Periprosthetic Fractures & PJI Joint Replacement
Periprosthetic Joint Infection (PJI)
PJI is the most devastating complication of joint replacement, occurring in 1–2% of primary THA/TKA. The Musculoskeletal Infection Society (MSIS) 2018 criteria define PJI with major and minor criteria. Major criteria (either one is diagnostic): Two positive cultures of the same organism from periprosthetic tissue, or a sinus tract communicating with the joint. Minor criteria (combined score ≥6 is diagnostic): elevated serum CRP (>10 mg/L, 2 points), elevated D-dimer (>860 ng/mL, 2 points), elevated synovial WBC (>3,000 for knee, >3,000 for hip, 3 points), elevated synovial PMN% (>80%, 2 points), positive alpha-defensin (3 points), elevated synovial CRP (>6.9 mg/L, 1 point), positive purulence (3 points), one positive culture (2 points).
Treatment: Acute PJI (<4 weeks from surgery or <3 weeks of symptoms with a well-fixed implant): DAIR (debridement, antibiotics, irrigation, and retention of implant) — exchange the modular components (polyethylene liner, femoral head), aggressive debridement, followed by 6 weeks of IV antibiotics + prolonged oral suppression. Success rate ~50–70%. Chronic PJI: Two-stage exchange arthroplasty is the gold standard — Stage 1: remove all implants and cement, thorough debridement, place an antibiotic-impregnated cement spacer (static or articulating), 6–8 weeks of IV antibiotics. Stage 2: reimplantation of new prosthesis after infection markers normalize (ESR, CRP trending down, joint aspiration negative). Success rate: 85–95%. One-stage exchange (removal and reimplantation in a single procedure) is gaining acceptance for select patients with a known organism, adequate soft tissue, and good bone stock — common in Europe, growing in North America.
Periprosthetic Fractures — Vancouver Classification (Hip)
| Type | Location | Stem Stability | Bone Quality | Treatment |
|---|---|---|---|---|
| AG | Greater trochanter | Stable | N/A | Usually nonoperative (unless displaced >2 cm with abductor insufficiency) |
| AL | Lesser trochanter | Stable | N/A | Usually nonoperative |
| B1 | Around or just distal to the stem | Stable (well-fixed) | Good | ORIF (plate + cerclage wires/cables); leave the stem in place |
| B2 | Around or just distal to the stem | Loose | Good | Revision to a longer, distally-fixed stem (extensively porous-coated or tapered fluted — Wagner SL, Stryker Restoration) |
| B3 | Around or just distal to the stem | Loose | Poor (severe bone loss) | Revision with structural allograft (strut grafts) + long revision stem, or proximal femoral replacement (megaprosthesis) |
| C | Well distal to the stem tip | Stable | Variable | Treat as a standard femur fracture (plate or retrograde nail, independent of the prosthesis) |
Aseptic Loosening
The most common cause of late arthroplasty failure. Mechanism: particulate wear debris (polyethylene, metal, ceramic) is phagocytosed by macrophages, triggering a cytokine cascade (TNF-alpha, IL-1, IL-6, RANKL) that activates osteoclasts → periprosthetic osteolysis → progressive loosening. Radiographic signs: progressive radiolucent lines at the bone-implant interface (≥2 mm or progressive), component migration, subsidence, or cement fracture. Treatment: revision arthroplasty — address bone loss with grafting, augments, or revision implants with increased constraint.
21 Bone Tumors Oncology
Approach to Bone Lesions
The initial evaluation of a bone lesion uses radiographic features to narrow the differential. Key descriptors: location in bone (epiphyseal, metaphyseal, diaphyseal), pattern of bone destruction (geographic [well-defined, slow-growing], moth-eaten [intermediate aggression], permeative [aggressive, malignant]), margin (sclerotic rim = slow-growing vs ill-defined = aggressive), periosteal reaction (solid/uninterrupted = benign; interrupted/sunburst/onion-skin/Codman triangle = aggressive/malignant), matrix (osteoid = cloud-like density; chondroid = rings-and-arcs calcification), and soft tissue mass.
Common Benign Bone Tumors
| Tumor | Age / Location | Characteristics | Treatment |
|---|---|---|---|
| Osteochondroma | Most common benign bone tumor; 10–30 years; metaphysis of long bones (distal femur, proximal tibia) | Pedunculated or sessile cartilage-capped bony exostosis; cortex and medullary canal are continuous with the parent bone; growth stops at skeletal maturity | Observation. Excision if symptomatic, growing after maturity, or cartilage cap >2 cm (risk of secondary chondrosarcoma, 1% solitary, 10% in hereditary multiple exostoses) |
| Enchondroma | 20–50 years; small bones of hands (phalanges, metacarpals) most common | Central, lytic, well-defined; chondroid matrix (rings-and-arcs). Ollier's disease = multiple enchondromas; Maffucci syndrome = multiple enchondromas + soft tissue hemangiomas (both carry significant malignant transformation risk) | Observation if asymptomatic. Curettage + bone grafting if pathologic fracture or in long bones where differentiation from low-grade chondrosarcoma is difficult |
| Giant Cell Tumor (GCT) | 20–40 years; epiphysis of long bones (distal femur, proximal tibia most common); must have closed physis | Eccentric, lytic, epiphyseal, well-defined without sclerotic margin; "soap bubble" appearance. Locally aggressive, rarely metastasizes to lungs (~3%) | Extended curettage + local adjuvant (phenol, liquid nitrogen, or argon beam) + bone graft/cement. Denosumab (RANKL inhibitor) for unresectable/recurrent. En bloc resection for recurrence or expendable bones |
| Osteoid Osteoma | 5–25 years; cortex of femur or tibia | <1.5 cm nidus with surrounding reactive sclerosis; intense night pain relieved by NSAIDs (prostaglandin-mediated). CT shows characteristic nidus with central dot of mineralization | NSAIDs (indomethacin) for 2–3 years (some resolve). CT-guided radiofrequency ablation (RFA) — 90% success rate. Surgical excision (en bloc or curettage) if RFA fails |
| Non-ossifying Fibroma (NOF) | Most common incidental finding in children; metaphysis of distal femur, proximal/distal tibia | Eccentric, cortical-based, lytic with sclerotic scalloped border; "bubbly" appearance. Histology: spindle cell fibroblastic tissue with storiform pattern | Observation — most resolve with skeletal maturity. Curettage + bone graft only if large (>50% of cortical diameter) with pathologic fracture risk |
Primary Malignant Bone Tumors
| Tumor | Age / Location | Characteristics | Treatment |
|---|---|---|---|
| Osteosarcoma | Most common primary malignant bone tumor; bimodal: 10–20 years (conventional) and >60 years (secondary to Paget's). Metaphysis of distal femur, proximal tibia, proximal humerus | Aggressive: permeative destruction, sunburst periosteal reaction, Codman triangle, osteoid matrix, soft tissue mass. Labs: elevated ALP. Lungs = #1 metastatic site | Neoadjuvant chemotherapy (MAP: methotrexate, doxorubicin [Adriamycin], cisplatin) → wide resection (limb salvage with endoprosthetic reconstruction vs amputation) → adjuvant chemo. Evaluate percent tumor necrosis on resected specimen (>90% necrosis = good response) |
| Ewing Sarcoma | 5–15 years; diaphysis of long bones (femur), flat bones (pelvis). More common in Caucasians | "Onion-skin" periosteal reaction, permeative destruction, large soft tissue mass. t(11;22) translocation → EWS-FLI1 fusion. Histology: small round blue cell tumor, CD99 positive, PAS positive (glycogen) | Neoadjuvant chemo (VDC/IE: vincristine, doxorubicin, cyclophosphamide alternating with ifosfamide, etoposide) → wide resection or radiation (Ewing is radiosensitive, unlike osteosarcoma) → adjuvant chemo |
| Chondrosarcoma | 40–70 years; pelvis, proximal femur, proximal humerus | Central, lytic, chondroid matrix (rings-and-arcs), endosteal scalloping (>2/3 cortical thickness = concerning). Low-grade (I) vs high-grade (II–III) | Wide surgical resection — chondrosarcoma is chemo- and radio-resistant. Intralesional procedures have unacceptable recurrence rates for conventional chondrosarcoma. Grade I central lesions of the extremity may be treated with extended curettage in select cases |
22 Metabolic Bone Disease Special
Osteoporosis
Systemic skeletal disease characterized by low bone mass and microarchitectural deterioration, leading to increased fracture risk. Affects ~10 million Americans; causes ~2 million fractures annually (hip, vertebral compression, distal radius). Diagnosis: DEXA scan (dual-energy X-ray absorptiometry) — T-score compares BMD to a young adult reference: T-score ≥ -1.0 = normal, -1.0 to -2.5 = osteopenia, ≤ -2.5 = osteoporosis, ≤ -2.5 with fragility fracture = severe osteoporosis. Screening recommended for all women ≥65 and men ≥70, or younger patients with risk factors.
Pharmacologic treatment (indicated for T-score ≤ -2.5, or T-score -1.0 to -2.5 with fragility fracture or FRAX 10-year hip fracture risk ≥3% or major osteoporotic fracture risk ≥20%):
| Class | Drug (Brand) | Mechanism | Dose | Key Pearl |
|---|---|---|---|---|
| Bisphosphonate | Alendronate (Fosamax) | Osteoclast inhibition (binds hydroxyapatite, induces osteoclast apoptosis) | 70 mg PO weekly | Take on empty stomach with full glass of water, remain upright 30 min (esophagitis risk). Drug holiday after 5 years (oral) or 3 years (IV) |
| Bisphosphonate | Zoledronic acid (Reclast) | Same | 5 mg IV annually | Most potent bisphosphonate; flu-like symptoms common after first infusion. Renal function must be adequate (CrCl >35) |
| RANKL inhibitor | Denosumab (Prolia) | Monoclonal antibody against RANKL → inhibits osteoclast formation | 60 mg SQ every 6 months | No drug holiday — rebound vertebral fractures occur after discontinuation. Must transition to bisphosphonate if stopping |
| PTH analog | Teriparatide (Forteo) | Anabolic — intermittent PTH stimulates osteoblasts | 20 mcg SQ daily × 2 years | Only anabolic option for severe osteoporosis; must follow with antiresorptive. Black box: osteosarcoma in rats at high doses (avoid in Paget's, unexplained ALP elevation, prior radiation) |
| Sclerostin inhibitor | Romosozumab (Evenity) | Anti-sclerostin antibody → dual action (increases formation, decreases resorption) | 210 mg SQ monthly × 12 months | Most rapid BMD increase of any agent. Black box: cardiovascular risk — contraindicated in patients with recent MI or stroke within past year |
Paget's Disease of Bone
Chronic disorder of bone remodeling with excessive osteoclastic resorption followed by disorganized osteoblastic new bone formation, producing enlarged, structurally weak bone. Affects 1–3% of adults >55; often asymptomatic and discovered incidentally. Common sites: pelvis, femur, tibia, skull, spine. Radiographic features: cortical thickening, coarsened trabecular pattern, "cotton wool" skull, "picture frame" vertebrae, bowing deformities of long bones. Labs: markedly elevated alkaline phosphatase (ALP) with normal calcium and phosphorus. Complications: pathologic fracture (transverse "chalk-stick" pattern), secondary OA (from deformed bone/joints — TKA and THA are technically challenging in Paget's), sarcomatous degeneration (osteosarcoma — rare, ~1%, but prognosis is dismal), hearing loss (skull base involvement), high-output cardiac failure (increased bone vascularity). Treatment: Zoledronic acid 5 mg IV (single infusion normalizes ALP in >90%, effect lasts years) or risedronate 30 mg daily × 2 months.
23 Pediatric Orthopedics Special
Growth Plate (Physis) Injuries — Salter-Harris Classification
The physis (growth plate) is the weakest link in the pediatric musculoskeletal system — it is 2–5x weaker than surrounding ligaments and bone. Physeal injuries account for 15–30% of childhood fractures. The Salter-Harris classification (mnemonic: SALTR) is universal:
| Type | Fracture Pattern | Mnemonic | Prognosis | Treatment |
|---|---|---|---|---|
| I | Through the physis only (separation at the growth plate) | Straight across (Slip) | Excellent; growth disturbance rare | Immobilization; closed reduction if displaced |
| II | Through the physis + metaphyseal fragment (Thurston-Holland fragment) | Above | Excellent; most common SH type (~75%) | Closed reduction + immobilization; rarely ORIF |
| III | Through the physis + epiphyseal fragment (intra-articular) | Lower (beLow) | Good if anatomically reduced; risk of growth arrest and OA | Anatomic reduction required; ORIF if displaced >2 mm (screws parallel to physis, not crossing it) |
| IV | Through metaphysis, physis, AND epiphysis | Through all | Higher risk of growth arrest; physeal bar formation | ORIF with anatomic reduction of articular surface and physis |
| V | Crush injury to the physis (compression) | Ruined (cRush) | Worst prognosis — often not recognized initially; growth arrest likely | Diagnosis often retrospective; treat growth disturbance as it develops |
Supracondylar Humerus Fracture
The most common elbow fracture in children (ages 5–7). >95% are extension-type (fall on outstretched hand with elbow hyperextended). Gartland classification: Type I = non-displaced (posterior fat pad sign may be the only finding; treat with long arm cast at 90°). Type II = displaced with intact posterior cortex (angulated but hinged; closed reduction + long arm cast or percutaneous pinning). Type III = completely displaced (no cortical contact; closed reduction + percutaneous pin fixation [2 lateral pins or crossed medial/lateral pins — medial pin risks ulnar nerve injury]). Neurovascular complications: anterior interosseous nerve (AIN) injury is most common (loss of FPL and FDP to index finger — unable to make "OK" sign); brachial artery injury (check radial pulse, hand perfusion — "pink and pulseless" hand may be observed vs explored). Volkmann's ischemic contracture — compartment syndrome of the forearm from brachial artery injury or post-reduction swelling — is the most feared complication; leads to irreversible flexor muscle fibrosis and claw-hand deformity.
Developmental Dysplasia of the Hip (DDH)
Spectrum from mild acetabular dysplasia to complete hip dislocation. Risk factors: female sex (4:1), firstborn, breech presentation, family history, oligohydramnios. Screening: Barlow test (adduct and push posteriorly — does the hip dislocate?) and Ortolani test (abduct with anterior lift — does a dislocated hip reduce with a palpable "clunk"?). Asymmetric skin folds, limb length discrepancy, and limited abduction are additional signs. Ultrasound screening at 6 weeks for high-risk infants (or those with abnormal exam). Treatment by age: 0–6 months: Pavlik harness (flexion/abduction positioning — 95% success for reducible hips; contraindicated in irreducible dislocations and children >6 months). 6–18 months: closed reduction under anesthesia + spica cast ± adductor tenotomy; arthrogram to confirm concentric reduction. 18 months–5 years: open reduction (medial or anterior approach) + pelvic osteotomy (Pemberton, Dega, or Salter). >5 years: treatment becomes increasingly complex with poorer outcomes.
Slipped Capital Femoral Epiphysis (SCFE)
The femoral epiphysis slips posteriorly and inferiorly relative to the metaphysis through the physis. Affects obese adolescents (10–16 years) during the growth spurt. Bilateral in 20–40%. Presents with hip or referred knee pain (always examine the hip when a child presents with knee pain), antalgic gait, and obligate external rotation with hip flexion. Radiographs: AP pelvis + frog-lateral — the frog-lateral is more sensitive; look for the Klein's line (a line along the superior femoral neck should intersect the lateral epiphysis — in SCFE it does not) and Southwick angle (measures slip severity: <30° mild, 30–60° moderate, >60° severe). Classification: Stable (able to bear weight, even with crutches) vs Unstable (unable to bear weight). This distinction is the most important prognostic factor — unstable SCFE has a 20–50% AVN rate vs <5% for stable. Treatment: In situ percutaneous screw fixation (single cannulated screw across the physis, centered in the epiphysis) — for all stable SCFE, even severe slips. Do NOT attempt to reduce a stable slip (reduction maneuvers increase AVN risk). Unstable SCFE: urgent surgical stabilization; gentle reduction (capsulotomy to decompress hematoma may be beneficial — Ziebarth et al., 2009) + screw fixation. Prophylactic contralateral pinning is controversial but often performed in high-risk patients (age <12, endocrinopathy, Modified Oxford Score).
Legg-Calve-Perthes Disease
Idiopathic AVN of the femoral head in children, typically ages 4–8. Four phases: (1) necrosis/initial, (2) fragmentation, (3) re-ossification, (4) remodeling. Younger age at onset and greater femoral head involvement correlate with better outcomes (the acetabulum can remodel around a deformed head). The Herring lateral pillar classification predicts prognosis: Group A (lateral pillar maintained at full height — good prognosis), Group B (>50% lateral pillar height maintained — intermediate), Group C (<50% lateral pillar height — poor prognosis in older children). Treatment: containment of the femoral head within the acetabulum — abduction bracing or surgical containment (femoral varus osteotomy or Salter innominate osteotomy) for older children (>8 years) with lateral pillar B or C disease.
24 Compartment Syndrome & CRPS Special
Acute Compartment Syndrome
A limb-threatening emergency where increased pressure within a closed fascial compartment compromises tissue perfusion. Most common in the leg (four compartments: anterior, lateral, superficial posterior, deep posterior) and forearm (three compartments: volar, dorsal, mobile wad). Etiologies: fractures (tibial shaft #1), crush injuries, vascular injury, burns, post-ischemic reperfusion, tight casts/dressings, bleeding in anticoagulated patients.
Diagnosis is clinical: the "5 P's" — Pain (out of proportion to injury, especially with passive stretch of the muscles in the affected compartment — the earliest and most reliable sign), Pressure (tense/firm compartment on palpation), Paresthesias (nerve ischemia), Paralysis (late — indicates irreversible damage), Pulselessness (very late — compartment syndrome occurs at pressures far below arterial occlusion pressure). Intracompartmental pressure measurement: Stryker needle or arterial line manometer. Absolute pressure >30 mmHg or delta pressure (ΔP) <30 mmHg (ΔP = diastolic blood pressure − compartment pressure) warrants fasciotomy. In a hypotensive patient, compartment syndrome occurs at lower absolute pressures — always use delta pressure.
Treatment: Emergent fasciotomy — release ALL compartments at risk. Leg: two-incision technique (anterolateral incision releases anterior and lateral compartments; posteromedial incision releases superficial and deep posterior compartments). Forearm: volar Henry approach (curvilinear incision from proximal to carpal tunnel) releases all three volar compartments; dorsal incision if dorsal compartment involved. Wounds are left open with a vessel-loop shoelace or negative-pressure dressing; delayed primary closure or skin grafting at 48–72 hours when swelling resolves. Delayed fasciotomy (>8 hours) is associated with significantly higher rates of amputation, infection, and permanent nerve damage.
Complex Regional Pain Syndrome (CRPS)
A chronic pain condition characterized by pain disproportionate to the inciting event, accompanied by autonomic, sensory, motor, and trophic changes. CRPS Type I (formerly reflex sympathetic dystrophy [RSD]): occurs without a definable nerve lesion — commonly after fractures, surgery, or immobilization. CRPS Type II (formerly causalgia): occurs after a defined peripheral nerve injury. Diagnosis uses the Budapest criteria: (1) continuing pain disproportionate to the inciting event, (2) at least one symptom in 3 of 4 categories (sensory [hyperesthesia/allodynia], vasomotor [temperature/color asymmetry], sudomotor/edema [swelling, sweating changes], motor/trophic [weakness, tremor, dystonia, nail/hair/skin changes]), (3) at least one sign at evaluation in 2 of 4 categories, (4) no other diagnosis better explains the findings.
Treatment: Multimodal and multidisciplinary. Physical/occupational therapy (graded motor imagery, mirror therapy, progressive loading) is the cornerstone. Medications: gabapentin/pregabalin (neuropathic pain), low-dose naltrexone, NSAIDs, topical lidocaine, short courses of oral corticosteroids in acute inflammatory CRPS. Interventional: sympathetic nerve blocks (stellate ganglion block for upper extremity, lumbar sympathetic block for lower extremity), spinal cord stimulation for refractory cases. Bisphosphonates (particularly IV alendronate or pamidronate) have shown benefit in randomized trials. Vitamin C 500 mg daily after distal radius fracture reduces CRPS incidence (Zollinger et al.).
25 Fracture Fixation Hardware Procedures
Plates & Screws
Cortical screws: Fully threaded, engage both cortices. Thread pitch designed for dense cortical bone. Used for interfragmentary compression (lag technique — gliding hole in near cortex, threads engage only far cortex → compression). Cancellous screws: Partially threaded (threads only at the tip), larger thread pitch for purchase in softer cancellous bone. Used in metaphyseal and epiphyseal regions (e.g., cannulated screws for femoral neck fractures). Locking screws: Thread into the plate as well as the bone, creating a fixed-angle construct. Locking plates function as internal external fixators — they do not require bone-plate friction for stability, making them ideal for osteoporotic bone, periarticular fractures, and bridge plating where anatomic reduction is not possible.
Plate types: Dynamic compression plate (DCP) — uses the oval hole design to generate interfragmentary compression as screws are tightened. Limited-contact DCP (LC-DCP) — reduced plate-bone contact to preserve periosteal blood supply. Locking compression plate (LCP) — combination holes accept both locking and conventional screws. Reconstruction plate — can be contoured in three dimensions (used for pelvis, clavicle). Periarticular anatomic plates — precontoured for specific anatomic locations (proximal humerus [Philos], distal radius [VA-LCP], proximal/distal tibia, distal femur). Bridge plating — plate spans comminuted zone without compressing individual fragments, relying on secondary bone healing (relative stability). One-third tubular plate — low-profile plate for lateral malleolus and other subcutaneous bones.
Intramedullary Nails
Nails are load-sharing devices placed within the medullary canal that provide alignment, length, and rotational control for long bone fractures. Advantages: biological fixation (preserves periosteal blood supply — no need to expose the fracture site), load-sharing (rather than load-bearing as with plates), early weight-bearing. Reaming the canal before insertion increases cortical contact and allows a larger-diameter nail, but strips the endosteal blood supply (temporarily — recovers within weeks). Static interlocking (screws through holes in both ends of the nail) controls length and rotation; dynamic interlocking (screws at one end only) allows axial compression. Common nails: tibial nail (suprapatellar or infrapatellar entry), femoral nail (piriformis or trochanteric entry, antegrade; or intercondylar, retrograde), cephalomedullary nail (trochanteric entry with lag screw into femoral head — for intertrochanteric/subtrochanteric fractures — Gamma3, TFN, InterTAN), humeral nail (antegrade through rotator cuff or retrograde through olecranon fossa).
External Fixation
Pins placed into bone proximal and distal to the fracture, connected by external bars or rings. Indications: temporary stabilization in damage control (polytrauma, open fractures), definitive treatment when internal fixation is contraindicated (severe soft tissue compromise, infection), periarticular fractures (spanning ex-fix bridges the joint), distraction osteogenesis (Ilizarov method for bone transport and limb lengthening), correction of complex deformities (Taylor Spatial Frame — hexapod frame that can correct six axes of deformity simultaneously using a computer-generated correction plan). Complications: pin site infection (the most common complication, 2–30%), pin loosening, fracture through pin sites, joint stiffness.
Other Fixation Devices
Kirschner wires (K-wires): Smooth pins of varying diameter used for temporary fixation, small bone fractures (phalanges, metacarpals), and as guide wires. Tension band: Converts tensile forces to compression (olecranon, patella, greater tuberosity). Cerclage wires/cables: Circumferential wires around bone — used to hold fracture fragments together (periprosthetic fractures, trochanteric fractures) or to reduce spiral/oblique fractures before plating. Suture anchors: Devices placed into bone that provide a suture attachment point for soft tissue repair (rotator cuff, labrum, ligaments). Interference screws: Used to fix soft tissue grafts within bone tunnels (ACL reconstruction).
26 Arthroscopy Procedures
General Principles
Arthroscopy is the insertion of a small camera (arthroscope, typically 4.0 mm, 30° angled lens) and instruments through small portals into a joint for diagnostic and therapeutic purposes. Benefits over open surgery: smaller incisions, less soft tissue damage, faster recovery, ability to visualize the entire joint.
Shoulder Arthroscopy
Indications: rotator cuff repair, labral repair (Bankart, SLAP), subacromial decompression (acromioplasty), distal clavicle excision (Mumford procedure for AC joint arthritis), capsular release for adhesive capsulitis, biceps tenotomy/tenodesis, removal of loose bodies. Standard portals: posterior (primary viewing portal, placed 2 cm inferior and 1 cm medial to the posterolateral corner of the acromion), anterior (working portal, in the rotator interval), lateral (subacromial space — for subacromial decompression and rotator cuff repair). The patient is positioned in beach chair or lateral decubitus.
Knee Arthroscopy
The most commonly performed orthopedic arthroscopic procedure. Standard portals: anterolateral and anteromedial (flanking the patellar tendon). Indications: meniscal surgery (partial meniscectomy, meniscal repair), ACL/PCL reconstruction, loose body removal, synovectomy, cartilage procedures (microfracture, OATS, ACI/MACI), lateral release for patellar maltracking. The systematic diagnostic arthroscopy evaluates: suprapatellar pouch, patellofemoral joint, medial gutter, medial compartment (medial meniscus, MFC, medial tibial plateau), intercondylar notch (ACL, PCL), lateral compartment, and lateral gutter.
Hip Arthroscopy
Rapidly growing field. Indications: femoroacetabular impingement (FAI) — cam (femoral-sided bump) and/or pincer (acetabular over-coverage) resection, labral repair/reconstruction, loose body removal, iliopsoas release, ligamentum teres debridement, capsular plication for instability. Performed in the supine or lateral position on a fracture table with traction to distract the joint. Portals: anterolateral, mid-anterior, and distal anterolateral accessory. Traction neuropraxia of the pudendal or lateral femoral cutaneous nerve is a recognized complication — limit traction time and force.
Cartilage Restoration Procedures
Microfracture: Arthroscopic awl creates perforations in the subchondral bone plate to allow marrow elements (MSCs, growth factors) to fill a chondral defect with fibrocartilage. Best for lesions <2 cm². Fibrocartilage is biomechanically inferior to hyaline cartilage but provides functional improvement. OATS (Osteochondral Autograft Transfer System): Cylindrical plug(s) of osteochondral tissue harvested from a non-weight-bearing area (lateral trochlear ridge) and transplanted into the defect. Best for lesions 1–4 cm². Osteochondral allograft (OCA): Fresh cadaveric osteochondral grafts for large defects (>2–4 cm²). ACI/MACI (Autologous Chondrocyte Implantation / Matrix-assisted ACI): Two-stage procedure — first stage harvests cartilage cells, which are expanded in culture; second stage implants them into the defect under a periosteal patch (ACI) or on a collagen scaffold (MACI — now the standard). Best for large lesions (>2 cm²) in young patients.
27 Arthroplasty Implants & Bearing Surfaces Procedures
Implant details for THA and TKA are covered in Sections 18 and 19. This section covers additional arthroplasty concepts.
Cemented vs Uncemented Fixation
Cemented fixation uses polymethylmethacrylate (PMMA) bone cement to fill the gap between the implant and bone, providing immediate mechanical fixation. Modern cementing technique (3rd generation): pulsatile lavage of the canal, retrograde cement insertion with a cement gun, canal plug (restrictor) to pressurize cement, centralization of the stem. Best for: elderly patients, osteoporotic bone (Dorr type C), cemented all-poly tibial components, revision with bone loss. Uncemented fixation relies on press-fit initial stability followed by biological fixation (bone ongrowth/ingrowth into porous surface coatings — hydroxyapatite, titanium plasma spray, trabecular metal [tantalum]). Best for: young, active patients with good bone quality. The trend in primary THA is toward uncemented acetabular cups (near-universal) and uncemented stems (majority in North America); cemented stems remain the standard in the UK/Scandinavia (excellent long-term registry data). In primary TKA, cemented fixation remains the gold standard globally.
Special Implant Concepts
Dual-mobility cups: An articulation within an articulation — the inner bearing (small head within a mobile polyethylene liner) and the outer bearing (the liner within the metal shell). Significantly reduces dislocation risk (<1% in primary THA vs 2–5% with fixed bearings). Increasingly used for fracture THA, revision, and patients at high dislocation risk. Constrained liners: Mechanically prevent dislocation by capturing the head within a locking ring — reserved for recurrent dislocation after failed closed reduction and soft tissue procedures, or abductor deficiency. Megaprostheses (endoprosthetic reconstruction): Large modular implants that replace segments of bone (proximal femur, distal femur, proximal tibia, proximal humerus). Used after tumor resection or massive periprosthetic bone loss. Systems: Stryker GMRS, Biomet OSS, Zimmer Segmental.
28 Spine Surgery Techniques Procedures
Anterior Cervical Discectomy and Fusion (ACDF)
The most commonly performed cervical spine surgery. Approach: Smith-Robinson anterior approach — transverse skin incision on the left side of the neck (protects the recurrent laryngeal nerve, which has a more consistent course on the left), dissect between the carotid sheath laterally and the trachea/esophagus medially. Retract the longus colli muscles laterally to expose the anterior cervical spine. Use fluoroscopy to confirm the level. Perform complete discectomy, decompress the neural foramen, place an interbody spacer (PEEK cage, titanium cage, allograft, or polyetheretherketone) filled with bone graft, and apply an anterior cervical plate with screws. Complications: dysphagia (most common, 2–50% depending on definition; usually transient), recurrent laryngeal nerve injury (hoarseness), esophageal perforation (rare, 0.1–0.3%), vertebral artery injury (rare), graft/hardware failure, adjacent segment disease (the most important long-term concern — accelerated degeneration at levels adjacent to the fusion, occurring in 2.9% per year). Cervical disc replacement (CDR) is an alternative that preserves motion and may reduce adjacent segment disease — FDA-approved devices include Prestige (Medtronic), ProDisc-C (Synthes), Mobi-C (Zimmer-Biomet).
Posterior Cervical Surgery
Laminectomy: Removal of the lamina to decompress the spinal canal — typically combined with posterior instrumented fusion (lateral mass screws at C3–C6, pedicle screws at C2 and C7) if instability is present or created. Laminoplasty: The lamina is hinged open on one side (open-door technique — Hirabayashi) or both sides (French-door technique) and held in the expanded position with spacers or plates. Preserves motion, avoids fusion, maintains posterior tension band. Best for multilevel stenosis with maintained lordosis. Posterior cervical foraminotomy: Keyhole decompression of a single nerve root without fusion — ideal for lateral disc herniations causing radiculopathy, allowing motion preservation.
Lumbar Fusion Techniques
| Technique | Description | Advantages | Disadvantages |
|---|---|---|---|
| Posterolateral fusion (PLF) | Bone graft placed along the transverse processes ± pedicle screw instrumentation | Simple, avoids the disc space, familiar | Lower fusion rate without instrumentation; does not restore disc height or lordosis |
| PLIF (Posterior Lumbar Interbody Fusion) | Posterior approach, bilateral laminotomy, disc removal, interbody cage(s) + posterior instrumentation | Direct canal decompression, anterior column support | Significant neural retraction, higher dural tear and nerve injury risk |
| TLIF (Transforaminal Lumbar Interbody Fusion) | Unilateral posterior approach through the facet joint, oblique cage insertion + posterior instrumentation | Less neural retraction than PLIF, can decompress contralateral side | Unilateral access may limit bilateral decompression in some cases |
| MIS-TLIF | TLIF through tubular retractors and percutaneous pedicle screws | Less muscle damage, less blood loss, faster recovery | Steep learning curve, limited visualization, radiation exposure |
| ALIF (Anterior Lumbar Interbody Fusion) | Anterior retroperitoneal approach (often with a vascular surgeon for L5–S1); large interbody cage | Best disc height/lordosis restoration, no posterior muscle dissection | Risk of vascular injury (iliac vessels), retrograde ejaculation (superior hypogastric plexus injury at L5–S1), requires supplemental posterior fixation at some levels |
| XLIF/DLIF (Lateral Interbody Fusion) | Direct lateral transpsoas approach (patient in lateral decubitus) | Large cage, indirect decompression, minimal posterior dissection | Psoas muscle injury, lumbar plexus injury (thigh numbness/weakness), not usable at L5–S1 (iliac crest obstruction) |
| OLIF (Oblique Lateral Interbody Fusion) | Anterior to psoas approach | Avoids the psoas muscle and lumbar plexus (lower neuro risk than XLIF) | Risk of vascular injury, limited to specific levels |
Pedicle Screw Fixation
The foundation of modern spine instrumentation. Screws are placed through the pedicle into the vertebral body, connected by rods to create a rigid construct. Provides three-column fixation (most stable posterior construct). Screw diameter, length, and trajectory are determined by pedicle anatomy (CT-guided planning, navigation, or robotic assistance improves accuracy). Complications: screw misplacement (medial breach → spinal cord/nerve root injury; lateral breach → vascular/visceral injury), screw loosening (especially in osteoporotic bone — augment with PMMA cement, use larger screws, extend the construct, or use cortical bone trajectory screws), rod fracture (at the level of pseudarthrosis), adjacent segment disease, proximal junctional kyphosis (PJK) after long constructs.
29 Imaging & Diagnostics
Imaging Modalities in Orthopedics
| Modality | Best For | Key Points |
|---|---|---|
| Plain Radiography (X-ray) | First-line for all fractures, arthritis, alignment, hardware | Always obtain ≥2 orthogonal views. Weight-bearing views for arthritis (joint space narrowing not apparent on non-weight-bearing). Include joints above and below for long bone injuries |
| CT Scan | Complex fractures (acetabulum, tibial plateau, calcaneus, spine), pre-operative planning, occult fractures | Superior bony detail. 3D reconstructions for surgical planning. Thin-cut (1–2 mm) axial images with sagittal and coronal reconstructions |
| MRI | Soft tissue pathology (ligaments, menisci, rotator cuff, labrum, disc herniation, AVN, infection, tumor) | Gold standard for soft tissue evaluation. T1: anatomy (fat is bright). T2/STIR: pathology (fluid/edema is bright). Gadolinium contrast for tumors and infection. MRI detects occult fractures (bone marrow edema) when X-rays are negative |
| Bone Scan (Scintigraphy) | Stress fractures, metastatic survey, infection, loosening around implants | Technetium-99m MDP: high sensitivity, low specificity. Triple-phase bone scan (angiographic, blood pool, delayed) helps differentiate infection from other causes. Positive within 24–72 hours of injury |
| Ultrasound | Rotator cuff tears, effusions, soft tissue masses, pediatric hip (DDH screening), guided injections | Dynamic evaluation (can assess impingement in real-time). Operator-dependent. No radiation |
| DEXA | Bone mineral density assessment (osteoporosis screening) | T-score and Z-score. Measured at hip and lumbar spine. False elevations from vertebral osteophytes, aortic calcification, prior fracture |
Key Radiographic Lines & Angles
| Measurement | Normal Value | Clinical Use |
|---|---|---|
| Bohler's angle (calcaneus) | 20–40° | Decreased angle indicates posterior facet depression; predicts need for surgical reduction |
| Critical angle of Gissane (calcaneus) | 130–145° | Increased angle indicates subtalar joint disruption |
| Radial inclination (wrist) | 22° | Measured on AP view; loss indicates distal radius fracture displacement |
| Volar tilt (wrist) | 11° volar | Measured on lateral; dorsal tilt indicates displacement (Colles' pattern) |
| Radial height (wrist) | 11 mm | Shortening indicates fracture impaction or malunion |
| Tip-apex distance (hip) | <25 mm | Predictor of lag screw cut-out in hip fracture fixation |
| Klein's line (hip) | Intersects lateral epiphysis on AP pelvis | Failure to intersect indicates SCFE |
| Southwick angle (hip) | <30° mild, 30–60° moderate, >60° severe | Quantifies SCFE severity on frog-lateral view |
| Mechanical axis (knee) | 0 ± 3° (neutral) | Full-length standing film; target for TKA alignment |
| Insall-Salvati ratio (knee) | 0.8–1.2 | Patellar tendon length / patellar length; >1.2 = patella alta (dislocation risk) |
30 Classification Systems (All)
This section consolidates all major classification systems in orthopedic surgery for quick reference. Detailed descriptions are found in the relevant clinical sections.
Fracture Classifications
| Classification | Structure | Categories | Section |
|---|---|---|---|
| AO/OTA | Universal fracture classification | Bone number (1–9) + segment (1–3) + type (A, B, C) + group/subgroup. Example: 31-A1 = femur (3), proximal (1), extra-articular simple (A1) = intertrochanteric simple | All fractures |
| Gustilo-Anderson | Open fracture severity | I, II, IIIA, IIIB, IIIC — based on wound size, soft tissue injury, contamination, and vascular injury | §03, §16 |
| Neer (proximal humerus) | Number of displaced parts | 1-part (non-displaced), 2-part, 3-part, 4-part, head-splitting, fracture-dislocation | §05 |
| Garden (femoral neck) | Displacement | I (valgus impacted), II (complete non-displaced), III (partially displaced/varus), IV (fully displaced) | §08 |
| Weber (ankle) | Fibula fracture level relative to syndesmosis | A (below), B (at), C (above) | §10 |
| Lauge-Hansen (ankle) | Mechanism-based | SER, SAD, PER, PAB — with sequential stages of injury | §10 |
| Schatzker (tibial plateau) | Fracture pattern | I (lateral split), II (lateral split-depression), III (pure depression), IV (medial), V (bicondylar), VI (with metadiaphyseal dissociation) | §09 |
| Mason (radial head) | Displacement and comminution | I (non-displaced), II (displaced partial), III (comminuted), IV (with dislocation) | §06 |
| Young-Burgess (pelvis) | Mechanism-based | LC-I/II/III, APC-I/II/III, VS, CM | §14 |
| Letournel (acetabulum) | Anatomic pattern | 5 elementary (posterior wall, posterior column, anterior wall, anterior column, transverse) + 5 associated | §14 |
| Sanders (calcaneus) | CT-based posterior facet | I (non-displaced), II (2-part), III (3-part), IV (comminuted) | §10 |
| Salter-Harris (physeal) | Growth plate involvement | I through V (SALTR mnemonic) | §23 |
| Gartland (supracondylar humerus) | Displacement | I (non-displaced), II (angulated/hinged), III (completely displaced) | §23 |
| Vancouver (periprosthetic hip) | Location and stem stability | AG, AL, B1, B2, B3, C | §20 |
| Anderson & D'Alonzo (odontoid) | Fracture location | Type I (tip), Type II (base), Type III (body) | §11 |
Grading & Staging Systems
| System | Entity | Summary |
|---|---|---|
| Kellgren-Lawrence | Osteoarthritis severity | 0–4 based on joint space narrowing, osteophytes, sclerosis, and deformity |
| Ficat & Arlet | Femoral head AVN | 0–IV: pre-clinical → subchondral collapse → secondary OA |
| Rockwood | AC joint separation | I–VI based on clavicle displacement and ligament integrity |
| Bigliani | Acromion morphology | Type I (flat), II (curved), III (hooked) — hooked increases impingement risk |
| TLICS | Thoracolumbar injury | Score morphology + PLC integrity + neuro status: ≤3 nonop, ≥5 operative |
| SLIC | Subaxial cervical injury | Score morphology + DLC integrity + neuro status: ≤3 nonop, ≥5 operative |
| Meyerding | Spondylolisthesis | I–V based on % slip of vertebral body: I (0–25%) to V (spondyloptosis >100%) |
| Herring lateral pillar | Legg-Calve-Perthes | A (full height), B (>50%), C (<50%) — prognosis worsens A → C |
| Mirels' | Pathologic fracture risk | Score 4 variables (site, pain, lesion type, size): ≥9 = prophylactic fixation |
| MSIS criteria | Periprosthetic joint infection | Major criteria (diagnostic alone) + minor criteria (combined score ≥6) |
31 Medications Master Table
| Class | Drug (Brand) | Mechanism | Dose | Indication | Critical Pearl |
|---|---|---|---|---|---|
| NSAID | Ketorolac (Toradol) | Non-selective COX inhibitor | 15–30 mg IV/IM q6h (max 5 days) | Acute post-op pain, fracture pain | Limit to 5 days; avoid in renal impairment. Theoretical concern for fracture healing impairment — controversial but avoid in high-risk nonunion situations |
| NSAID | Meloxicam (Mobic) | Preferential COX-2 inhibitor | 7.5–15 mg PO daily | OA, inflammatory arthritis | Once-daily dosing improves compliance; lower GI risk than non-selective NSAIDs |
| NSAID | Indomethacin (Indocin) | Non-selective COX inhibitor | 25–50 mg PO TID or 75 mg SR BID | Heterotopic ossification prophylaxis, gout, osteoid osteoma | GI side effects limit use; 6-week course for HO prophylaxis post-THA |
| NSAID | Celecoxib (Celebrex) | Selective COX-2 inhibitor | 100–200 mg PO BID | OA, RA, post-operative pain | Lower GI risk; cardiovascular risk with long-term use. Avoid in sulfa allergy |
| Opioid | Oxycodone (OxyContin, Roxicodone) | Mu-opioid receptor agonist | 5–10 mg PO q4–6h PRN | Moderate-severe acute pain (fractures, post-op) | Short-course only; multimodal analgesia reduces opioid requirements |
| Gabapentinoid | Gabapentin (Neurontin) | Binds alpha-2-delta subunit of voltage-gated Ca2+ channels | 100–300 mg PO TID, titrate up | Neuropathic pain, CRPS, radiculopathy, peri-operative multimodal analgesia | Sedation, dizziness. Renal dosing required. Pre-op gabapentin reduces post-op opioid use |
| Gabapentinoid | Pregabalin (Lyrica) | Same as gabapentin (more potent) | 75–150 mg PO BID | Neuropathic pain, fibromyalgia, CRPS | More linear pharmacokinetics than gabapentin; scheduled drug (Class V) |
| Muscle relaxant | Cyclobenzaprine (Flexeril) | Central acting; structurally related to TCAs | 5–10 mg PO TID | Muscle spasm (acute back pain, post-fracture) | Sedation is primary side effect; avoid in elderly. Short course (<2–3 weeks) |
| Corticosteroid (injection) | Triamcinolone (Kenalog) | Anti-inflammatory, inhibits phospholipase A2 | 40 mg (large joint) / 10–20 mg (small joint) | OA, tendinopathy, bursitis | Limit to 3–4 injections/year/joint. Hold THA/TKA for 3 months after intra-articular injection (PJI risk) |
| Corticosteroid (injection) | Methylprednisolone acetate (Depo-Medrol) | Same | 40–80 mg (large joint) | OA, epidural steroid injection | Longer-acting than triamcinolone; avoid intravascular injection during epidurals (particulate steroid risks embolism in cervical ESI) |
| Bisphosphonate | Alendronate (Fosamax) | Osteoclast inhibitor | 70 mg PO weekly | Osteoporosis | Drug holiday after 5 years; atypical femoral fracture risk. Take upright, empty stomach |
| Bisphosphonate | Zoledronic acid (Reclast) | Osteoclast inhibitor (most potent bisphosphonate) | 5 mg IV annually | Osteoporosis, Paget's disease | Single infusion for Paget's. Renal function requirement (CrCl >35 mL/min) |
| RANKL inhibitor | Denosumab (Prolia / Xgeva) | Anti-RANKL monoclonal antibody → inhibits osteoclast formation | Prolia: 60 mg SQ q6 months. Xgeva: 120 mg SQ monthly | Prolia: osteoporosis. Xgeva: bone metastases, GCT | Rebound vertebral fractures after discontinuation — must transition to bisphosphonate. No renal dose adjustment |
| PTH analog | Teriparatide (Forteo) | Anabolic — intermittent PTH stimulates osteoblasts | 20 mcg SQ daily × 24 months | Severe osteoporosis, failure of antiresorptive | Black box: osteosarcoma in rats. Follow with antiresorptive agent. Off-label for fracture nonunion |
| Sclerostin inhibitor | Romosozumab (Evenity) | Anti-sclerostin → dual anabolic + antiresorptive | 210 mg SQ monthly × 12 months | Osteoporosis (high fracture risk) | CV risk: avoid if recent MI/stroke. Most rapid BMD gains of any agent |
| Antibiotic | Cefazolin (Ancef) | 1st-gen cephalosporin (cell wall synthesis inhibition) | 2 g IV (3 g if >120 kg), within 60 min pre-incision; redose at 4 hours | Surgical prophylaxis (all clean orthopedic procedures) | Standard of care for surgical prophylaxis in orthopedics. Add vancomycin if MRSA risk |
| Antibiotic | Vancomycin | Cell wall synthesis inhibition (glycopeptide) | 15 mg/kg IV, infuse over 1–2 hours (start 2 hours pre-incision) | MRSA prophylaxis, PJI treatment | Requires slow infusion (red man syndrome). Vancomycin powder in wounds is an emerging adjunct to reduce SSI |
| Anticoagulant | Enoxaparin (Lovenox) | LMWH — inhibits factor Xa | 40 mg SQ daily or 30 mg SQ BID | VTE prophylaxis post-THA/TKA, pelvic/acetabular fractures | Hold 12 hours before neuraxial anesthesia. Renal dosing (30 mg daily if CrCl <30) |
| Anticoagulant | Rivaroxaban (Xarelto) | Direct factor Xa inhibitor | 10 mg PO daily × 12 days (TKA) or 35 days (THA) | VTE prophylaxis post-arthroplasty | FDA-approved for arthroplasty VTE prophylaxis. No routine monitoring. Avoid in severe renal/hepatic impairment |
| Anticoagulant | Aspirin (ASA) | Irreversible COX-1 inhibitor → antiplatelet | 81 mg PO BID × 4 weeks | VTE prophylaxis post-arthroplasty (low-risk patients) | AAOS/ACS support ASA for VTE prophylaxis in standard-risk arthroplasty patients. Lower bleeding risk than LMWH/DOACs |
| Vitamin | Vitamin C | Antioxidant, collagen synthesis cofactor | 500 mg PO daily | CRPS prophylaxis after distal radius fracture/surgery | Started day of injury/surgery, continued 50 days. Low cost, minimal risk |
| Local anesthetic | Liposomal bupivacaine (Exparel) | Extended-release local anesthetic (bupivacaine in multivesicular liposomes) | 266 mg (20 mL) infiltrated locally | Periarticular injection during TKA/THA for post-op pain | Provides up to 72 hours of local analgesia. Do not mix with other local anesthetics (disrupts liposomes). Part of multimodal pain protocols |
32 Abbreviations Master List
| Abbreviation | Meaning |
|---|---|
| ACL | Anterior cruciate ligament |
| ACDF | Anterior cervical discectomy and fusion |
| ACI | Autologous chondrocyte implantation |
| AIN | Anterior interosseous nerve |
| ALIF | Anterior lumbar interbody fusion |
| ALP | Alkaline phosphatase |
| AO/OTA | Arbeitsgemeinschaft fur Osteosynthesefragen / Orthopaedic Trauma Association |
| AVN | Avascular necrosis |
| BMD | Bone mineral density |
| BMP | Bone morphogenetic protein |
| BTB | Bone-patellar tendon-bone (autograft) |
| CoCr | Cobalt-chromium alloy |
| CRPS | Complex regional pain syndrome |
| CR | Cruciate-retaining (TKA) |
| CTR | Carpal tunnel release |
| DAA | Direct anterior approach (THA) |
| DCO | Damage control orthopedics |
| DDH | Developmental dysplasia of the hip |
| DDD | Degenerative disc disease |
| DEXA | Dual-energy X-ray absorptiometry |
| DRUJ | Distal radioulnar joint |
| DVT | Deep vein thrombosis |
| ECRB | Extensor carpi radialis brevis |
| EHL | Extensor hallucis longus |
| EMG | Electromyography |
| ESI | Epidural steroid injection |
| ETC | Early total care |
| Ex-fix | External fixation / fixator |
| FAI | Femoroacetabular impingement |
| FDP | Flexor digitorum profundus |
| FDS | Flexor digitorum superficialis |
| FPL | Flexor pollicis longus |
| GCT | Giant cell tumor |
| HO | Heterotopic ossification |
| HTO | High tibial osteotomy |
| I&D | Irrigation and debridement |
| IMN | Intramedullary nail |
| IP | Interphalangeal |
| K-wire | Kirschner wire |
| LCL | Lateral collateral ligament |
| LCP | Locking compression plate |
| LLD | Leg length discrepancy |
| MACI | Matrix-assisted autologous chondrocyte implantation |
| MCL | Medial collateral ligament |
| MCP | Metacarpophalangeal |
| MIPO | Minimally invasive plate osteosynthesis |
| MUA | Manipulation under anesthesia |
| NCS | Nerve conduction study |
| NOF | Non-ossifying fibroma |
| NVI | Neurovascularly intact |
| OA | Osteoarthritis |
| OATS | Osteochondral autograft transfer system |
| ORIF | Open reduction and internal fixation |
| PCL | Posterior cruciate ligament |
| PE | Pulmonary embolism |
| PEEK | Polyetheretherketone (interbody cage material) |
| PJI | Periprosthetic joint infection |
| PLC | Posterior ligamentous complex (spine) / posterolateral corner (knee) |
| PLIF | Posterior lumbar interbody fusion |
| PMMA | Polymethylmethacrylate (bone cement) |
| PS | Posterior-stabilized (TKA) |
| PT | Physical therapy / posterior tibial (context-dependent) |
| RA | Rheumatoid arthritis |
| ROM | Range of motion |
| rTSA | Reverse total shoulder arthroplasty |
| SCFE | Slipped capital femoral epiphysis |
| SHS | Sliding hip screw |
| SLAP | Superior labrum anterior-posterior (tear) |
| TAD | Tip-apex distance |
| TBW | Tension band wiring |
| TEA | Total elbow arthroplasty |
| THA | Total hip arthroplasty |
| TKA | Total knee arthroplasty |
| TLIF | Transforaminal lumbar interbody fusion |
| TLICS | Thoracolumbar Injury Classification and Severity Score |
| TLSO | Thoracolumbosacral orthosis (brace) |
| TMT | Tarsometatarsal |
| TSA | Total shoulder arthroplasty |
| UKA | Unicompartmental knee arthroplasty |
| VTE | Venous thromboembolism |
| XLPE | Highly cross-linked polyethylene |
33 Risk Factors & Comorbidities
Fracture Risk Factors
| Risk Factor | Mechanism / Relevance |
|---|---|
| Osteoporosis | Reduced bone mineral density → fragility fractures (vertebral compression, hip, distal radius) with minimal trauma. T-score ≤ -2.5 on DEXA |
| Advanced age | Progressive bone loss, sarcopenia (fall risk), impaired balance, polypharmacy (sedatives, antihypertensives → falls) |
| Smoking | Impairs osteoblast function, neovascularization, and oxygen delivery. Doubles nonunion risk. Increases surgical site infection rate by 2–4x |
| Diabetes mellitus | Microvascular disease impairs healing. Perioperative hyperglycemia (glucose >200 mg/dL) increases infection risk. A1C >8% associated with higher complication rates in arthroplasty |
| Chronic kidney disease | Renal osteodystrophy (secondary hyperparathyroidism → increased bone turnover and fragility). Impaired vitamin D activation |
| Malnutrition | Albumin <3.5 g/dL and lymphocyte count <1,500 predict poor wound healing and increased infection risk. Pre-optimize before elective surgery |
| Obesity (BMI >40) | Increased wound complications, higher implant loads (accelerated wear), technical surgical difficulty, higher DVT risk. Some centers use BMI >40 as relative contraindication for elective arthroplasty |
| Vitamin D deficiency | 25-OH vitamin D <20 ng/mL → impaired calcium absorption and bone mineralization. Supplementation improves fracture healing and implant fixation |
| Corticosteroid use | Dose-dependent osteoporosis, AVN (femoral head, humeral head). >5 mg prednisone daily for >3 months warrants osteoporosis screening and prophylaxis |
| Alcohol abuse | Direct toxic effect on osteoblasts, AVN, falls, nutritional deficiency, liver disease (coagulopathy) |
| Metastatic disease | Lytic lesions weaken bone → pathologic fractures. Most common primaries: breast, prostate, lung, kidney, thyroid |
Surgical Complication Risk Factors
| Complication | Key Risk Factors |
|---|---|
| Surgical site infection (SSI) | Diabetes (A1C >7–8%), malnutrition, obesity, smoking, immunosuppression, prolonged operative time, prior surgery at same site, active skin infection, Staph aureus nasal carriage (decolonize with mupirocin + chlorhexidine bathing) |
| VTE (DVT/PE) | Prior VTE (strongest risk factor), active malignancy, prolonged immobilization, obesity, age >60, lower extremity fracture, pelvic surgery, general anesthesia (vs neuraxial), thrombophilia |
| Dislocation (THA) | Posterior approach (without capsular repair), female sex, neuromuscular disease, cognitive impairment, alcohol abuse, prior hip surgery, component malpositioning, small femoral head size (<32 mm) |
| Nonunion | Smoking (#1 modifiable factor), open fracture, bone loss, infection, inadequate fixation, NSAID use (controversial), poor nutrition, comminution, distraction at fracture site |
| Hardware failure | Non-compliance with weight-bearing restrictions, obesity, osteoporosis (poor screw purchase), inadequate fixation construct, infection |
34 Key References
The following landmark studies and guidelines are referenced throughout this specialty page and form the evidence base for modern orthopedic practice.
| Reference | Topic | Key Finding |
|---|---|---|
| Canadian Orthopaedic Trauma Society, JBJS 2007 | Displaced midshaft clavicle fractures | Operative fixation reduced nonunion from ~15% to ~3% and improved functional outcomes compared to nonoperative treatment |
| Baumgaertner et al., JBJS 1995 | Hip fracture fixation — tip-apex distance | TAD <25 mm is the strongest predictor against lag screw cut-out in intertrochanteric fracture fixation |
| Bhandari et al., NEJM 2019 (HEALTH trial) | Displaced femoral neck fractures in elderly | THA had fewer secondary procedures but similar mortality and functional outcomes compared to hemiarthroplasty at 24 months |
| Kirkley et al., NEJM 2008 | Arthroscopic surgery for knee OA | Arthroscopic debridement provided no benefit over sham surgery or conservative treatment for knee OA |
| Weinstein et al., JBJS 2008 (SPORT) | Lumbar spinal stenosis | Surgical decompression provided significantly better outcomes than conservative treatment at 4-year follow-up |
| SPRINT investigators, JBJS 2008 | Reamed vs unreamed tibial nailing | Reamed nailing reduced the need for secondary procedures in closed tibial shaft fractures |
| Costa et al., BMJ 2020 (STAR trial) | Achilles tendon rupture | Functional rehabilitation produced similar outcomes to surgical repair; operative group had slightly lower re-rupture rate |
| Ly & Coetzee, JBJS 2006 | Lisfranc injuries | Primary arthrodesis produced superior outcomes to ORIF for purely ligamentous Lisfranc injuries |
| Lack et al., JBJS 2015 | Open fracture antibiotic timing | Antibiotics administered within 66 minutes of presentation significantly reduced infection rate; 24-hour course is sufficient |
| Schenker et al., JBJS 2012 | Open fracture debridement timing | No evidence that the traditional "6-hour rule" for debridement affects infection rate; early antibiotics are more important |
| McKee et al., JBJS 2003 | Distal humerus fractures in elderly | TEA produced better outcomes than ORIF for comminuted distal humerus fractures in patients over 65 |
| Kaeding et al., AJSM 2011 | ACL graft selection | Allograft ACL reconstruction in young athletes (<25) had significantly higher re-tear rates than autograft |
| Ziebarth et al., JBJS 2009 | Unstable SCFE | Modified Dunn procedure with surgical hip dislocation and capsulotomy reduced AVN rate in unstable SCFE |