Plastic & Reconstructive Surgery
Every diagnosis, procedure, flap, graft, classification, complication, medication, and management algorithm across the full scope of plastic and reconstructive surgery in one place.
01 Skin & Soft-Tissue Anatomy
Skin Layers
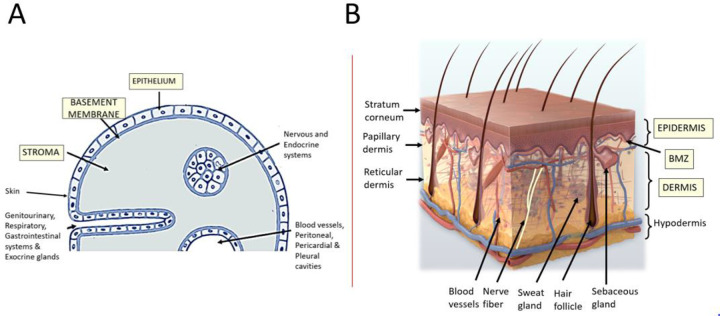
The skin is the largest organ of the body, comprising approximately 15% of total body weight. It consists of two primary layers: the epidermis (stratified squamous epithelium, 0.04–1.5 mm thick) and the dermis (connective tissue, 0.5–5 mm thick). Beneath lies the hypodermis (subcutaneous fat), which is not technically part of the skin but is critical in plastic surgery.
The subcutaneous tissue (hypodermis) consists of fat lobules separated by fibrous septa (Cooper ligaments in the breast). The superficial fatty layer (Camper fascia) and membranous layer (Scarpa fascia) are distinct in the lower abdomen. Scarpa fascia is important in abdominoplasty — preserving it and suturing it to the Scarpa fascia of the inferior flap during closure reduces dead space and seroma formation. Fat thickness varies enormously by body site, sex, and BMI, and determines the suitability of donor sites for fat grafting and flap harvest.
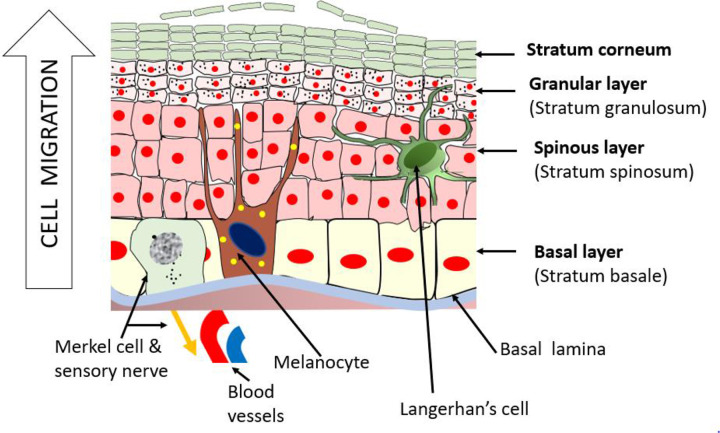
Epidermis: Five layers from deep to superficial — stratum basale (germinativum), stratum spinosum, stratum granulosum, stratum lucidum (only in thick skin of palms/soles), and stratum corneum. The stratum basale contains melanocytes, Merkel cells, and keratinocyte stem cells. Epidermal turnover time is approximately 28 days. Melanocytes are present in a 1:10 ratio with keratinocytes across all races; skin colour differences result from melanin production and distribution, not melanocyte number.
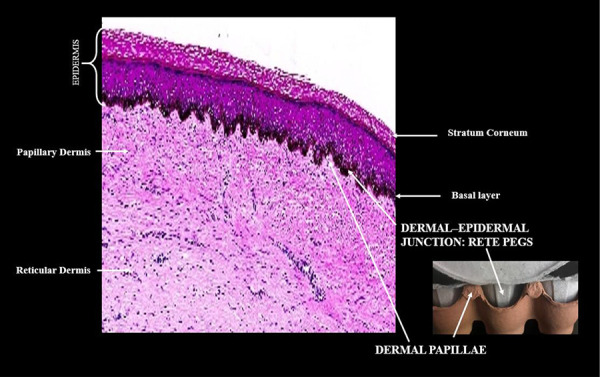
Dermis: Divided into the thin papillary dermis (loose connective tissue with type III collagen, Meissner corpuscles) and the thick reticular dermis (dense irregular connective tissue with type I collagen, Pacinian corpuscles). Contains hair follicles, sebaceous glands, eccrine sweat glands, blood vessels, nerves, and lymphatics. The dermis is ~80% type I collagen and ~15% type III collagen in mature skin.
Blood Supply & Angiosomes
Cutaneous blood supply follows a predictable layered pattern. The musculocutaneous perforators arise from source arteries, pass through muscle, and reach the skin via the subdermal plexus. The septocutaneous (fasciocutaneous) perforators travel through fascial septa between muscles. The concept of angiosomes (Taylor & Palmer, 1987) divides the body into 40 three-dimensional vascular territories, each supplied by a named source artery. Adjacent angiosomes are linked by choke vessels that can dilate over time (the delay phenomenon).
Fascia & the Fasciocutaneous System
The deep fascia invests muscles and provides a scaffold for perforating vessels. The fasciocutaneous system is the anatomical basis for fasciocutaneous flap design (Cormack & Lamberty classification). Scarpa fascia in the abdomen is a distinct layer of the superficial fascia that can be preserved during abdominoplasty to reduce seroma rates.
Dermatomes & Sensory Innervation
Knowledge of dermatomes is essential for sensory flap design and nerve block placement. Key landmarks: C5 — lateral arm (deltoid); T4 — nipple line; T10 — umbilicus; L1 — inguinal ligament; S2-S4 — perineum. Cutaneous nerve territories often overlap between adjacent dermatomes.
Mathes-Nahai Muscle Flap Classification
The Mathes-Nahai system classifies muscles by their vascular anatomy, which determines their reliability as flaps.
| Type | Vascular Pattern | Example Muscles |
|---|---|---|
| I | One vascular pedicle | Tensor fasciae latae, gastrocnemius (medial/lateral heads), abductor digiti minimi |
| II | Dominant pedicle + minor pedicle(s) | Gracilis, soleus, sternocleidomastoid, trapezius, vastus lateralis |
| III | Two dominant pedicles | Rectus abdominis, gluteus maximus, serratus anterior |
| IV | Segmental vascular pedicles | Sartorius, tibialis anterior, external oblique |
| V | One dominant pedicle + secondary segmental pedicles | Latissimus dorsi, pectoralis major |
02 Wound Healing Physiology
Phases of Wound Healing
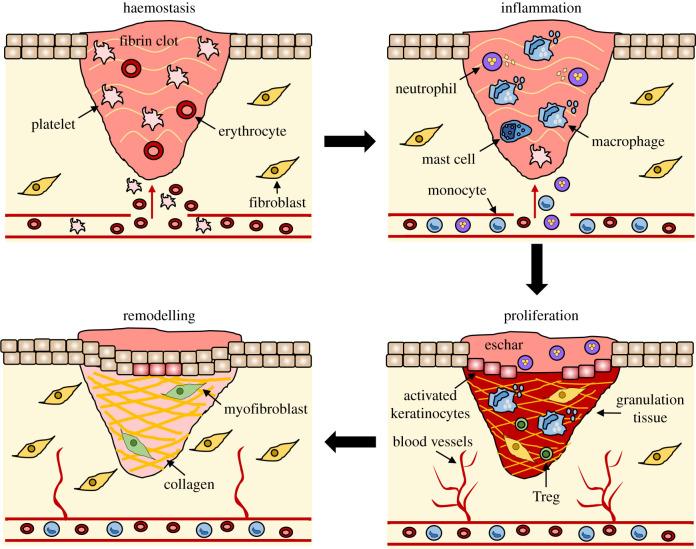
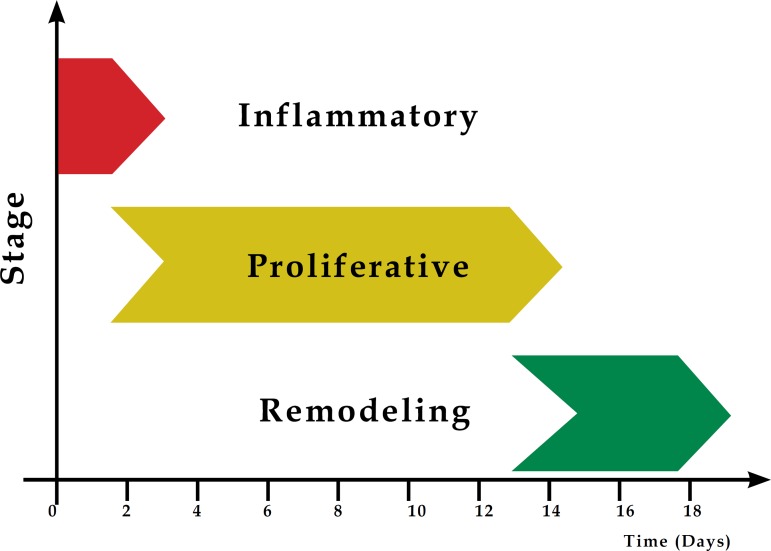
Wound healing proceeds through four overlapping phases: hemostasis (minutes), inflammation (hours to days), proliferation (days to weeks), and remodelling (weeks to years).
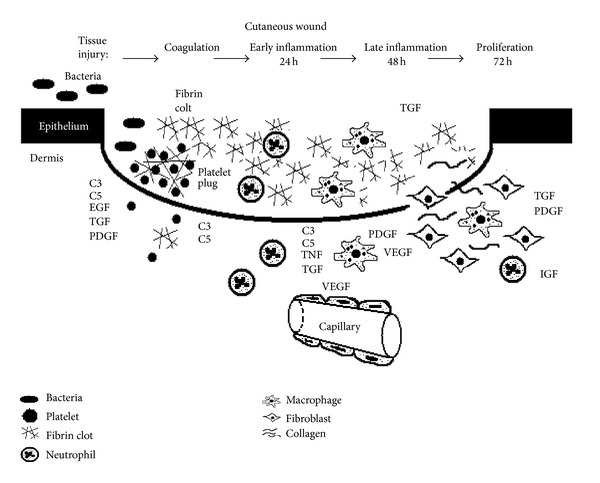
Hemostasis (0–minutes): Vasoconstriction, platelet adhesion via exposed collagen & von Willebrand factor, platelet aggregation, fibrin clot formation. Platelets degranulate releasing PDGF (chemotactic for fibroblasts/macrophages), TGF-β (stimulates collagen synthesis), and VEGF (angiogenesis).
Inflammation (1–4 days): Neutrophils arrive first (within 24 hours), followed by macrophages (48–72 hours) — the most critical cell in wound healing. Macrophages phagocytose debris, release cytokines (IL-1, TNF-α), and transition the wound from inflammation to proliferation. A wound can heal without neutrophils but cannot heal without macrophages.
Proliferation (4–21 days): Fibroblast migration and collagen deposition (initially type III collagen), angiogenesis (new blood vessel formation), granulation tissue formation, and epithelialisation. Fibroblasts are the dominant cell type. Myofibroblasts (containing α-smooth muscle actin) drive wound contraction — the most important mechanism of wound closure in open wounds.
Remodelling (21 days–2 years): Type III collagen is replaced by type I collagen (ratio shifts from 3:1 type III:I to the normal 1:3). Collagen is cross-linked by lysyl oxidase. Maximum wound tensile strength is reached at ~8 weeks and only reaches 80% of original tensile strength — a wound never regains 100% strength.
Key Growth Factors
| Growth Factor | Source | Function |
|---|---|---|
| PDGF | Platelets, macrophages | Chemotaxis for fibroblasts & macrophages; mitogen for fibroblasts & smooth muscle cells |
| TGF-β | Platelets, macrophages, lymphocytes | Stimulates collagen synthesis & deposition; inhibits proteases; promotes fibrosis; immunosuppressive |
| FGF | Macrophages, endothelial cells | Angiogenesis; fibroblast proliferation |
| VEGF | Keratinocytes, macrophages | Potent angiogenesis factor |
| EGF | Platelets, macrophages, salivary glands | Epithelial cell migration & proliferation |
| IGF-1 | Liver, fibroblasts | Stimulates collagen synthesis; cell growth |
Abnormal Wound Healing — Keloid vs. Hypertrophic Scar
| Feature | Hypertrophic Scar | Keloid |
|---|---|---|
| Extension beyond wound | No — remains within wound borders | Yes — extends beyond original wound |
| Regression | Often regresses spontaneously over 1–2 years | Rarely regresses; tends to recur after excision |
| Collagen pattern | Type III collagen; nodular arrangement parallel to skin surface | Thick, disorganised type I & III collagen bundles |
| Prevalence | Common; any race | Higher in African, Asian, Hispanic populations (15× higher in dark-skinned individuals) |
| Common sites | Flexor surfaces, across joints | Earlobes, sternum, shoulders, upper back |
| Genetics | Not hereditary | Familial tendency; autosomal dominant with variable penetrance |
| Treatment | Pressure, silicone sheeting, steroid injection | Triamcinolone injection, excision + adjuvant (radiation, 5-FU), pressure earrings |
Collagen Synthesis
Collagen synthesis begins intracellularly in the fibroblast ribosome. The key steps: (1) transcription and translation of procollagen α-chains; (2) hydroxylation of proline and lysine residues by prolyl hydroxylase and lysyl hydroxylase (requires vitamin C, Fe2+, α-ketoglutarate, and O2); (3) glycosylation; (4) triple helix formation (procollagen); (5) secretion into extracellular space; (6) cleavage of propeptides to form tropocollagen; (7) cross-linking by lysyl oxidase (requires copper) to form mature collagen fibrils. The wound initially deposits type III collagen, which is gradually replaced by type I collagen during remodelling (ratio shifts from 30% type III to the normal ~10% type III by 1–2 years).
Factors impairing wound healing: Infection (>105 organisms/g tissue), diabetes (impaired neutrophil function, microangiopathy), malnutrition (vitamin C deficiency impairs collagen hydroxylation; zinc deficiency impairs cell division), smoking (vasoconstriction, CO reduces O2 delivery), steroids (inhibit inflammatory phase & collagen synthesis — effect can be reversed by vitamin A), radiation (obliterative endarteritis, fibrosis), chemotherapy, and foreign bodies.
03 The Reconstructive Exam & Tissue Assessment
History & Physical Examination
The plastic surgery consultation begins with a thorough assessment of the wound or defect, patient comorbidities, and reconstructive goals. Key history points: mechanism of injury, time since injury, prior radiation or surgery, smoking status, diabetes control (HbA1c), nutritional status (albumin, prealbumin), immunosuppression, and anticoagulation.
Physical examination: Defect dimensions (length × width × depth), tissue type exposed (skin, subcutaneous tissue, muscle, bone, tendon, hardware, joint), wound bed quality (granulating vs. necrotic vs. irradiated), surrounding tissue quality and laxity, vascular status of the limb/region, and sensory/motor nerve function.
Vascular Assessment
The Allen test assesses dual blood supply to the hand (radial and ulnar arteries) via the palmar arches. Essential before radial forearm free flap harvest or radial artery catheterisation. A positive Allen test (incomplete refill with one artery occluded) is a contraindication to harvesting the radial forearm flap from that extremity.
Handheld Doppler assessment is used to identify perforator locations preoperatively, confirm pedicle patency intraoperatively, and monitor free flap perfusion postoperatively. An audible arterial Doppler signal is triphasic in normal vessels. Ankle-brachial index (ABI) ≥ 0.9 indicates adequate perfusion for wound healing; ABI < 0.5 suggests limb-threatening ischaemia and likely inability to heal a wound without revascularisation.
Tissue Perfusion Assessment — Advanced Modalities
Transcutaneous oxygen tension (TcPO2): Measures skin oxygen diffusion; values > 40 mmHg predict adequate healing; values < 20 mmHg indicate hypoxia and impaired healing potential. Particularly useful in determining amputation level in peripheral arterial disease. Laser Doppler flowmetry: Measures microvascular blood flow in real time; used for burn depth assessment (differentiate deep partial-thickness from full-thickness) and flap monitoring. Fluorescein angiography: IV fluorescein (10–15 mg/kg) with Wood’s lamp examination; areas that fluoresce are perfused; non-fluorescing areas are ischaemic. Useful for intraoperative assessment of flap perfusion and mastectomy flap viability. Being supplanted by ICG fluorescence angiography (SPY system), which is more accurate and does not cause skin staining.
Wound Bed Preparation
The TIME framework guides wound bed preparation: T — Tissue (debridement of nonviable tissue); I — Infection/Inflammation (control bacterial burden, manage biofilm); M — Moisture balance (optimal moist environment); E — Edge (assess epithelial advancement, non-healing edges may indicate malignancy — biopsy Marjolin ulcer).
Negative pressure wound therapy (NPWT) applies subatmospheric pressure (typically –125 mmHg continuous or intermittent) to promote granulation tissue formation, reduce oedema, increase blood flow, and remove exudate. Indications include open wounds, skin graft bolstering, dehisced surgical wounds, and bridge therapy before definitive closure. Contraindications: malignancy in the wound, unexplored fistulae, exposed vessels without coverage, and necrotic tissue with eschar.
Skin Cancer Reconstruction
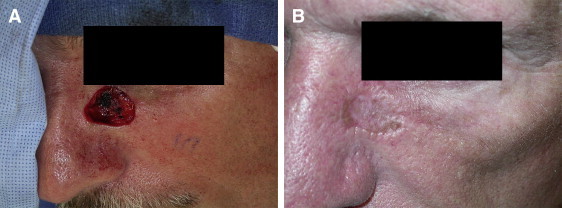
Plastic surgeons frequently reconstruct defects following Mohs micrographic surgery or wide local excision of cutaneous malignancies. Basal cell carcinoma (BCC) is the most common skin cancer (80% of non-melanoma skin cancers); locally destructive but very rarely metastasises (< 0.1%). Squamous cell carcinoma (SCC) accounts for ~20% of non-melanoma skin cancers; metastatic rate ~2–5% overall (higher for immunosuppressed patients, lip SCC, and recurrent tumours). Melanoma excision margins: in situ — 0.5–1 cm; ≤ 1 mm thickness — 1 cm; 1.01–2 mm — 1–2 cm; > 2 mm — 2 cm. Sentinel lymph node biopsy is recommended for melanomas ≥ 0.8 mm thickness or with ulceration.
Reconstruction follows the subunit principle on the face: if > 50% of a facial aesthetic subunit is lost, excise the remainder and reconstruct the entire subunit for the best cosmetic result. The nose is divided into subunits: dorsum, tip, columella, soft triangle, paired alar lobules, and paired sidewalls. The eyelids are divided into anterior lamella (skin + orbicularis) and posterior lamella (tarsus + conjunctiva) — at least one lamella must have its own blood supply in reconstruction.

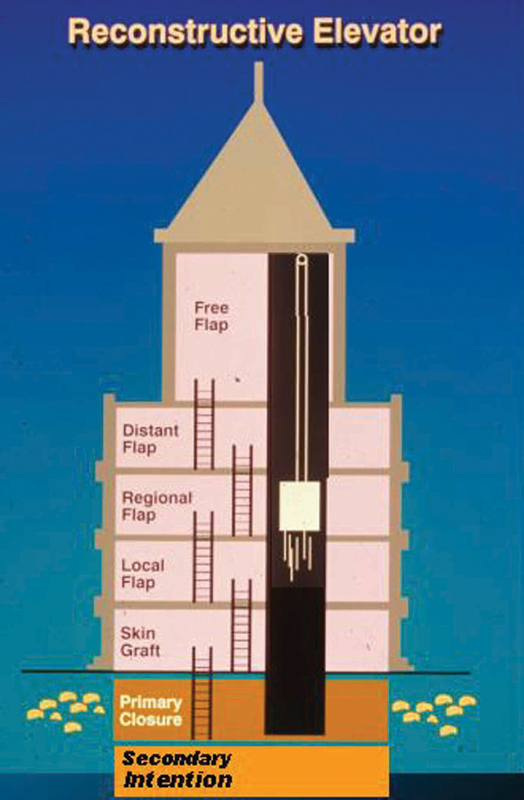
04 The Reconstructive Ladder & Elevator
The Reconstructive Ladder is a conceptual framework for wound closure, progressing from simple to complex. The Reconstructive Elevator (Gottlieb & Krieger) modernises this concept by allowing the surgeon to skip rungs and proceed directly to the optimal reconstruction rather than exhausting simpler options first.
| Rung | Method | Indications |
|---|---|---|
| 1 | Secondary intention | Small, superficial, clean wounds; concave surfaces (medial canthus, temple, ear conchal bowl) |
| 2 | Primary closure | Clean wounds with adequate tissue laxity; tension-free approximation possible |
| 3 | Skin graft (STSG/FTSG) | Wounds with well-vascularised bed; no exposed bone/tendon/hardware without periosteum/paratenon |
| 4 | Local flap | Small-to-moderate defects with adequate adjacent tissue; need for tissue match (face) |
| 5 | Regional/pedicled flap | Larger defects requiring bulk; recipient site within arc of rotation |
| 6 | Free tissue transfer | Large/complex defects; inadequate local tissue; need for specific tissue (bone, muscle, skin) |
| 7 | Composite tissue allotransplantation | Devastating tissue loss (face transplant, hand transplant); requires lifelong immunosuppression |

05 Skin Grafts
Split-Thickness vs. Full-Thickness Skin Grafts
| Feature | STSG (Split-Thickness) | FTSG (Full-Thickness) |
|---|---|---|
| Components | Epidermis + partial dermis (0.008–0.018 in) | Epidermis + entire dermis |
| Donor site healing | Re-epithelialises from adnexal structures (7–14 days) | Requires primary closure or grafting |
| Contraction | More secondary contraction (up to 50%) | Less secondary contraction (~10–20%) |
| Primary contraction | Less (less elastic dermis) | More (full dermis with elastic fibres) |
| Graft take reliability | Higher (thinner = less metabolic demand) | Lower (thicker = greater metabolic demand) |
| Colour/texture match | Poor (shiny, hyperpigmented) | Better (more dermis = better match) |
| Sensation | Limited recovery | Better sensory recovery |
| Donor sites | Thigh (most common), buttock, scalp | Groin, postauricular, supraclavicular, inner arm, abdomen |
| Best indications | Large wounds, burns, temporary coverage | Face, hands, over joints |
Graft Take Physiology
Skin graft survival depends on three sequential phases:
1. Plasmatic imbibition (0–48 hours): The graft absorbs nutrients via diffusion from wound bed plasma. The graft swells and may gain 40% in weight. This is the most critical phase — anything that prevents graft-bed contact (hematoma, seroma, shearing) prevents graft take.
2. Inosculation (48–72 hours): Direct anastomosis of graft vessels with recipient bed vessels. Capillary alignment occurs.
3. Revascularisation (Day 4–7): Neovascularisation — ingrowth of new vessels from the wound bed into the graft. By day 4–5, the graft has an established blood supply.
Dermal Substitutes
Integra Dermal Regeneration Template: Bilayer construct consisting of a bovine collagen/glycosaminoglycan dermal matrix with a silicone epidermal analogue. Applied to wounds lacking dermal coverage (exposed bone/tendon). The dermal matrix vascularises over 2–3 weeks, then the silicone layer is removed and a thin STSG is applied. Useful for burns, trauma, and reconstruction where dermal thickness is needed but FTSG donor sites are limited. AlloDerm: Acellular human dermis that can be used as a dermal substitute or to augment soft tissue. MatriDerm: Bovine collagen/elastin matrix applied in a single stage beneath a skin graft. These substitutes provide a vascularised dermal scaffold that improves graft quality, reduces contraction, and provides better aesthetic and functional outcomes than STSG alone.
Meshing & Bolster Dressings
STSGs may be meshed at ratios of 1:1, 1:1.5, 1:3, or 1:6 to allow expansion over larger wound areas, permit fluid drainage, and conform to irregular surfaces. Higher mesh ratios result in more prominent mesh pattern scarring. A bolster (tie-over) dressing immobilises the graft against the wound bed to prevent shearing — typically left in place for 5–7 days. NPWT (VAC) is increasingly used as an alternative bolster, particularly for irregular surfaces.
Composite Grafts
A composite graft contains two or more tissue types (e.g., skin + cartilage). Most commonly used for nasal alar rim reconstruction, where an auricular composite graft (skin-cartilage-skin from the helix or antihelix) provides structural support and lining. Maximum graft size: ~1.5 cm (limited by diffusion distance for nutrients). Survival depends on peripheral revascularisation; these grafts characteristically appear white, then cyanotic (“take cycle”), then pink over 5–7 days. Cool the graft site for the first 5 days to reduce metabolic demand. Used also for fingertip reconstruction in children and eyelid margin repair.
06 Local Flaps
Flap Design Principles
Local flaps transfer tissue adjacent to the defect with its own blood supply. Key principles: plan in reverse (design the flap first, then create the defect), maintain adequate length-to-width ratios for random-pattern flaps (generally ≤ 3:1 on the body, up to 5:1 on the face due to rich blood supply), orient final scars along relaxed skin tension lines (RSTL), and avoid distortion of free margins (eyelids, lips, nasal ala).
Advancement Flaps
Single advancement flap: Tissue slides directly forward into the defect. The Burow triangle is excised at the base to prevent standing cone deformity. Bilateral advancement (H-plasty): Two opposing advancement flaps. V-Y advancement: A V-shaped incision where the island of tissue is advanced and the donor closed in a Y configuration — useful for fingertip reconstruction and release of contracted scars.
Rotation Flaps
A rotation flap pivots around a point into the defect. The arc of rotation should be 4–8 times the diameter of the defect. A back-cut toward the pivot point increases mobility but reduces the pedicle base. A Burow triangle may be excised at the base of the arc to allow closure.
Transposition Flaps
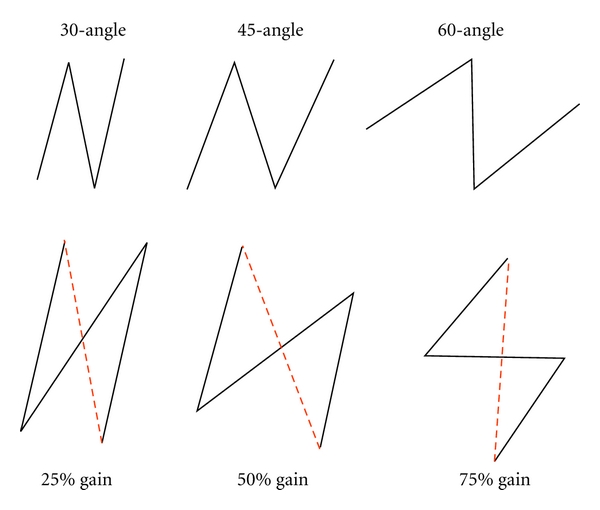
Z-plasty: Two triangular transposition flaps that interchange position. The central limb is placed along the scar or contracture. Standard Z-plasty uses 60° angles, which produces a theoretical 75% gain in length along the central limb. Angles of 30° gain ~25% length; 45° gain ~50%; 60° gain ~75%. Z-plasty also reorients the scar direction by 90°.
Rhomboid (Limberg) flap: A parallelogram-shaped flap designed adjacent to a rhomboid (60° and 120° angles) defect. The short diagonal of the rhomboid is extended, and the flap is raised adjacent to this extension. There are four possible Limberg flaps for any given rhomboid defect — the surgeon chooses the one that places closure tension along RSTL and borrows from the area of greatest tissue laxity.
Bilobed flap (Zitelli modification): A double transposition flap most commonly used for nasal defects of the distal third (1–1.5 cm). The first lobe is the same size as the defect and is transposed 45°; the second lobe is half the size and transposed an additional 45° (total arc 90°). The secondary defect is closed primarily.
W-Plasty & Geometric Broken Line Closure
W-plasty: Excision of a scar in a zigzag pattern (series of small triangles) to irregularise the scar line. Unlike Z-plasty, W-plasty does not lengthen the scar but breaks up the linear shadow that makes scars conspicuous. Best for scars that cross relaxed skin tension lines. Geometric broken line closure (GBLC): Random geometric shapes (triangles, semicircles, rectangles) are designed along the scar line, providing even greater scar irregularity than W-plasty. Both techniques are used for scar revision on the face.
Interpolation Flaps
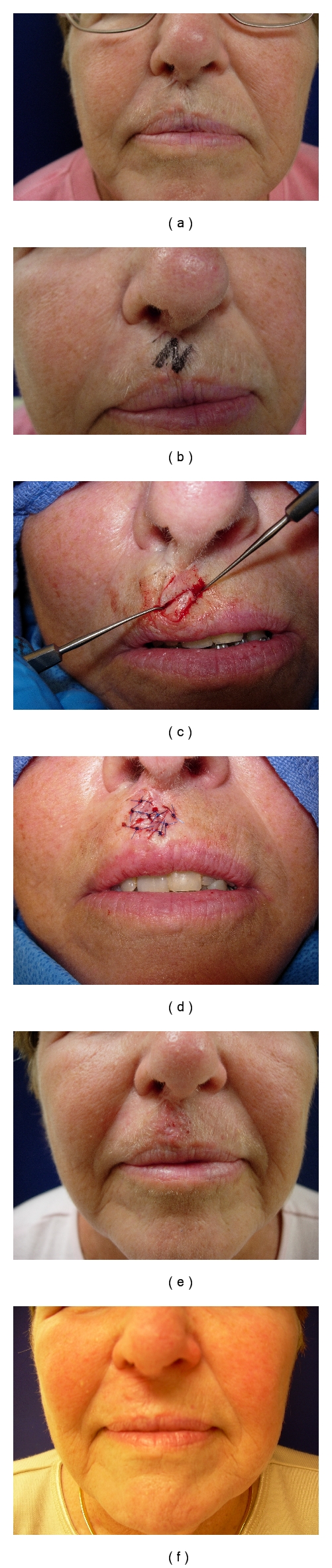
Paramedian forehead flap: The workhorse flap for nasal reconstruction. Based on the supratrochlear artery. Provides well-vascularised tissue with excellent colour and texture match for the nose. Requires a two-stage procedure: flap inset (Stage 1) and pedicle division at 3 weeks (Stage 2). The forehead donor site can be closed primarily up to ~3 cm width.
Melolabial (nasolabial) flap: An interpolation or transposition flap based on the angular artery or random blood supply from the rich facial vasculature. Used for alar and lateral nasal defects. Can be superiorly or inferiorly based.
07 Regional & Pedicled Flaps
Common Pedicled Muscle & Musculocutaneous Flaps
| Flap | Type (M-N) | Pedicle | Arc of Rotation | Common Uses |
|---|---|---|---|---|
| Pectoralis major | V | Thoracoacromial artery (dominant); internal mammary perforators (segmental) | Head & neck, sternum, chest wall | Head & neck reconstruction, sternal wound coverage |
| Latissimus dorsi | V | Thoracodorsal artery (dominant); segmental posterior intercostals & lumbar perforators | Chest, axilla, head & neck, upper extremity | Breast reconstruction, chest wall, scalp |
| Rectus abdominis (VRAM/TRAM) | III | Superior epigastric (dominant proximally); deep inferior epigastric (dominant distally) | Chest, groin, perineum | Breast reconstruction (pedicled TRAM), perineal reconstruction |
| Gracilis | II | Medial circumflex femoral artery (dominant) | Perineum, groin, vagina | Perineal reconstruction, vaginal reconstruction, facial reanimation (free) |
| Gastrocnemius (medial head) | I | Medial sural artery | Proximal tibia, knee | Proximal third tibial defects, exposed knee prosthesis |
| Soleus | II | Popliteal artery branches (proximal dominant); posterior tibial artery branches (distal) | Middle third tibia | Middle third tibial defects |
| Gluteus maximus | III | Superior gluteal artery; inferior gluteal artery | Sacrum, ischium, trochanter | Sacral & ischial pressure injuries |
Fasciocutaneous Flaps
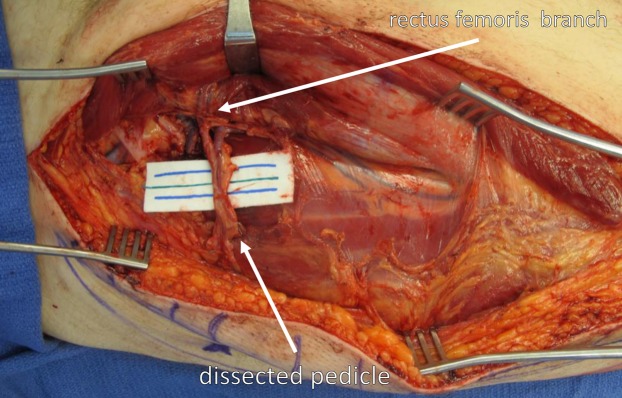
The anterolateral thigh (ALT) flap is the workhorse fasciocutaneous flap, based on septocutaneous or musculocutaneous perforators from the descending branch of the lateral circumflex femoral artery. Pedicle length averages 8–12 cm, vessel diameter 2–2.5 mm. Can be harvested as fasciocutaneous, adipofascial, or with vastus lateralis muscle. Used pedicled for groin/abdominal defects or free for head & neck, extremity, and trunk reconstruction. The perforator is located within a 3 cm radius of the midpoint of a line from the ASIS to the superolateral patella in approximately 90% of cases.
Propeller Flaps
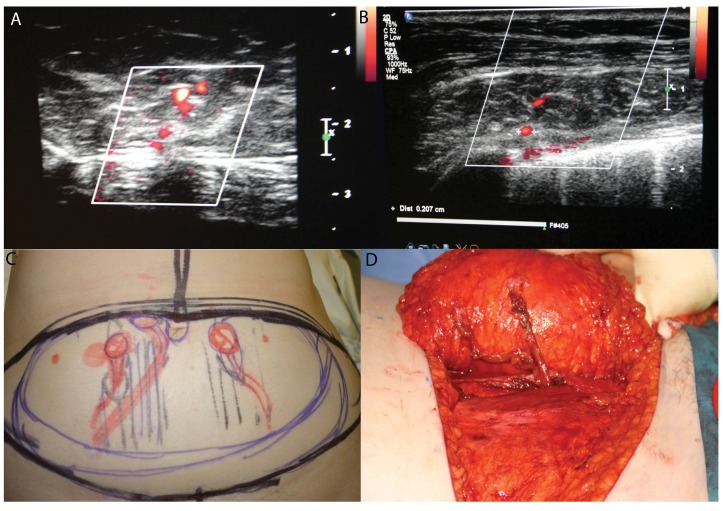
A propeller flap is an island flap that rotates up to 180° on a single perforator vessel (the “propeller blade” design). The perforator is identified with a handheld Doppler preoperatively. The flap is raised, skeletonised on its perforator, and rotated into the defect. Propeller flaps provide local tissue with similar colour and texture match while avoiding the need for microsurgery. Commonly used in lower extremity reconstruction, especially for small to moderate defects where free tissue transfer would be excessive. Success rates exceed 90% in experienced hands, though venous congestion is the most common complication (10–15%). PMID: 20140696
08 Free Tissue Transfer (Microsurgery)
Principles of Microsurgery
Free tissue transfer involves detaching a flap from its donor site, transferring it to a distant recipient site, and re-establishing blood flow via microvascular anastomosis. Requires an operating microscope (6–40× magnification), microsurgical instruments, and 8-0 to 10-0 nylon or Prolene suture. Vessel diameter must be ≥ 0.5 mm (practical minimum ~1.0 mm). Success rates at experienced centres exceed 95–98%.
Anastomosis technique: End-to-end or end-to-side arterial and venous anastomoses. A coupling device (Synovis/GEM coupler) can be used for venous anastomosis (90-second coupler technique) with patency rates equivalent to hand-sewn. At least one artery and one vein (preferably two veins) are anastomosed. Antithrombotic protocols vary by institution — commonly include aspirin 325 mg, heparin 5,000 units IV intraoperatively, and dextran 40 or heparin drip postoperatively.
Common Free Flaps
| Flap | Pedicle | Tissue Components | Pedicle Length/Diameter | Primary Indications |
|---|---|---|---|---|
| DIEP | Deep inferior epigastric artery & vein | Skin, fat | 10–14 cm / 2–3 mm | Breast reconstruction (gold standard autologous) |
| ALT | Descending branch of LCFA | Skin, fat ± muscle (VL) | 8–12 cm / 2–2.5 mm | Head & neck, extremity, trunk |
| Radial forearm | Radial artery & venae comitantes | Thin, pliable skin & fat | 15–20 cm / 2.5–3 mm | Oral cavity, pharynx, hand |
| Fibula | Peroneal artery & vein | Bone (up to 25 cm) ± skin paddle | 6–8 cm / 2–3 mm | Mandible, maxilla, long bone reconstruction |
| Scapula/parascapular | Circumflex scapular artery | Skin, fat ± bone (lateral scapular border) | 5–7 cm / 2.5–3 mm | Head & neck, upper extremity |
| Gracilis | Medial circumflex femoral artery | Muscle ± skin | 6–8 cm / 1.5–2 mm | Facial reanimation (functional), perineal, breast (TUG) |
| Latissimus dorsi | Thoracodorsal artery & vein | Muscle ± skin | 8–12 cm / 2–3 mm | Large soft-tissue defects, breast, scalp |
Perforator Flap Concept
A perforator flap is a flap based on a single or few perforating vessels that pass through or between deep tissues (usually muscle) to supply the skin and subcutaneous fat. The key advantage over traditional musculocutaneous flaps is preservation of the underlying muscle, reducing donor-site morbidity. Perforator flap nomenclature follows the Gent consensus: named by the source artery with the suffix “-AP” (artery perforator), e.g., DIEP = deep inferior epigastric artery perforator. Dissection of the perforator through the muscle is the most technically demanding step and may take 1–2 hours.
Recipient Vessel Selection
Recipient vessel choice depends on the location of the defect, vessel calibre match (ideally ≤ 2:1 size discrepancy), proximity to the defect, and vessel quality (avoid irradiated, atherosclerotic, or previously operated vessels). Common recipient vessels by site: head & neck — facial artery, superior thyroid artery, transverse cervical artery; breast — internal mammary artery (3rd or 4th intercostal space), thoracodorsal artery; lower extremity — anterior tibial, posterior tibial, dorsalis pedis; upper extremity — radial, ulnar arteries. End-to-side anastomosis preserves flow to the distal limb and is preferred in traumatised or single-vessel extremities.
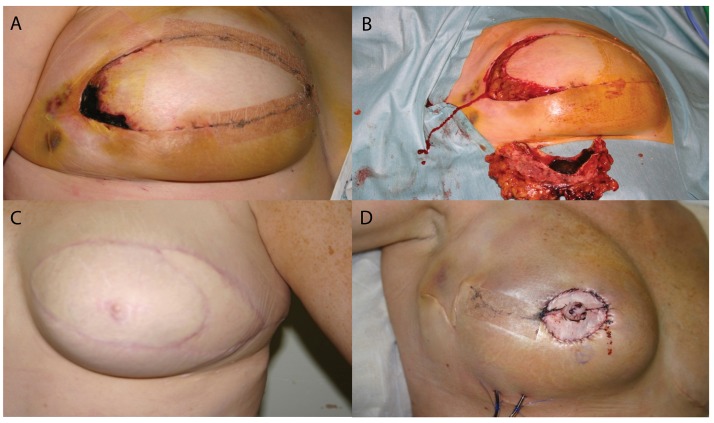
09 Breast Reconstruction
Timing
Immediate reconstruction is performed at the time of mastectomy. Advantages: preserved breast skin envelope, superior aesthetic outcome, single anaesthesia. Delayed reconstruction is performed weeks to months later — chosen when postmastectomy radiation is planned (radiation to an implant increases capsular contracture from ~8% to ~40%; radiation to an autologous flap increases fat necrosis, fibrosis, and volume loss). Delayed-immediate reconstruction uses a tissue expander at mastectomy, radiation through the expander, then definitive reconstruction after radiation is complete.
Implant-Based Reconstruction
Two-stage (tissue expander → implant): Most common approach. A tissue expander is placed subpectoral (partial muscle coverage ± acellular dermal matrix [ADM] for inferolateral coverage) or prepectoral (above the muscle, with ADM wrap). Serial expansion over 6–12 weeks, then exchange to permanent implant (silicone or saline) at a second operation. Direct-to-implant (DTI): One-stage placement of a permanent implant at mastectomy — requires adequate skin flap perfusion and an ADM or mesh sling. Success requires well-perfused mastectomy flaps (intraoperative assessment with SPY fluorescence angiography or ICG).
Acellular dermal matrix (ADM): Products include AlloDerm (human cadaveric dermis), FlexHD, DermACELL, and SurgiMend (bovine). ADM provides inferolateral pole support, reduces capsular contracture rates, and allows prepectoral placement. Complications: seroma (10–15%), infection (5–10%), red breast syndrome.
Autologous Reconstruction
| Flap | Pedicle | Pros | Cons |
|---|---|---|---|
| DIEP | Deep inferior epigastric | Gold standard; preserves muscle; natural ptosis; abdominoplasty donor site | Long operative time (6–8 hr); requires microsurgery; perforator dissection technically demanding |
| Pedicled TRAM | Superior epigastric | No microsurgery required; reliable | Sacrifices rectus muscle; abdominal wall morbidity (bulge ~10%, hernia ~5%); less reliable blood supply than DIEP |
| Free TRAM | Deep inferior epigastric | Better blood supply than pedicled TRAM | Microsurgery required; muscle sacrifice |
| Latissimus dorsi + implant | Thoracodorsal | Reliable; good for partial breast/thin patients | Usually requires implant; back scar; seroma (~30%) |
| SGAP/IGAP | Superior/inferior gluteal artery perforator | Alternative when abdomen unavailable | Short pedicle; difficult patient positioning; gluteal contour deformity |
| TUG (gracilis) | Medial circumflex femoral | Small to moderate breast; hidden scar | Small flap volume; short pedicle |
| PAP | Profunda artery perforator | Posterior thigh tissue; hidden scar; moderate volume | Requires microsurgery; limited volume for large breasts |
Radiation & Breast Reconstruction
Postmastectomy radiation therapy (PMRT) significantly impacts reconstruction outcomes. For implant-based reconstruction: radiation increases capsular contracture rates from ~8% to ~40%, implant loss from ~2% to ~15%, and need for revisional surgery. Prepectoral implant placement in the irradiated field has higher complication rates than submuscular. For autologous reconstruction: radiation to the flap increases fat necrosis (15–25%), fibrosis, volume loss, and flap contracture, but overall flap loss rates remain low (< 3%). The delayed-immediate approach (expander at mastectomy, radiation, then autologous reconstruction) avoids direct radiation to the permanent reconstruction and is preferred at many centres. The timing debate continues, but meta-analyses consistently show superior aesthetic outcomes with autologous over implant-based reconstruction in the irradiated breast. PMID: 24572840
Oncoplastic Breast Surgery
Oncoplastic surgery combines oncological resection with plastic surgery techniques to allow wider excision margins while maintaining breast shape. Techniques include volume displacement (reshaping the remaining breast tissue using local tissue rearrangement, reduction mammaplasty, or mastopexy patterns) and volume replacement (filling the lumpectomy defect with local or distant flaps). Allows resection of up to 20–50% of breast volume while avoiding mastectomy. Contralateral symmetry surgery (reduction, augmentation, or mastopexy) is often performed simultaneously.
Nipple-Areolar Complex Reconstruction
Typically performed 3–6 months after breast mound reconstruction. The C-V flap and skate flap are common techniques for nipple projection. The areola is recreated with a full-thickness skin graft (inner thigh, contralateral areola) or medical tattooing (3D tattoo techniques have become the preferred method for many patients). Nipple-sparing mastectomy avoids the need for NAC reconstruction.
10 Head & Neck Reconstruction
Principles
Head and neck reconstruction aims to restore form and function after ablative surgery for cancer, trauma, or congenital deformity. Critical functional goals: oral competence, speech, swallowing, airway patency, and eye protection. The “like with like” principle guides tissue selection — thin pliable flaps for intraoral lining, bone-containing flaps for mandibular continuity, skin flaps matched for colour and texture.
Defect-Based Approach
| Defect Site | First-Line Reconstruction | Alternatives |
|---|---|---|
| Oral cavity (tongue, floor of mouth) | Radial forearm free flap (thin, pliable) | ALT (if thin), ulnar forearm |
| Mandible | Fibula free flap (gold standard — up to 25 cm bone, accepts osseointegrated implants) | Scapula, iliac crest, reconstruction plate |
| Maxilla | Fibula, scapula, or ALT/radial forearm + obturator | Iliac crest |
| Scalp | Local rotation/advancement flaps; free latissimus dorsi + STSG for large defects | Tissue expansion (delayed) |
| External nose | Paramedian forehead flap (gold standard) | Melolabial flap, free flap for total nasal |
| Pharyngoesophageal (circumferential) | Free jejunal flap or tubed ALT/radial forearm | Gastric pull-up |
| Midface/cheek | ALT, radial forearm, or cervicofacial advancement | Submental island flap |
The Fibula Free Flap for Mandible Reconstruction
The fibula free flap (Taylor, Hidalgo) is the workhorse for mandibular reconstruction. The fibula provides up to 25 cm of bicortical bone supplied by the peroneal artery (endosteal blood supply) and can be osteotomised at multiple points (each segment must retain a periosteal cuff for blood supply). A skin paddle can be included based on septocutaneous or musculocutaneous perforators. Virtual surgical planning (VSP) with 3D-printed cutting guides improves accuracy of osseous reconstruction. Osseointegrated dental implants can be placed primarily or secondarily.
11 Lower Extremity Reconstruction
Gustilo-Anderson Classification of Open Fractures
| Type | Wound Size | Soft-Tissue Injury | Contamination | Treatment Approach |
|---|---|---|---|---|
| I | < 1 cm | Minimal | Clean | I&D, primary closure |
| II | 1–10 cm | Moderate, no flap needed | Moderate | I&D, wound care ± skin graft |
| IIIA | > 10 cm | Severe, but adequate soft-tissue coverage possible | High | Serial debridement, local/regional flap |
| IIIB | > 10 cm | Severe, with periosteal stripping and bone exposure requiring flap | High | Flap coverage required (local or free) |
| IIIC | Any | Vascular injury requiring repair | Variable | Emergency Vascular repair + flap coverage; consider amputation |
Zone-Based Reconstruction of the Tibia
The classic teaching (Godina principle, adapted) assigns flap coverage based on the zone of tibial exposure:
| Zone | Location | Recommended Flap | Rationale |
|---|---|---|---|
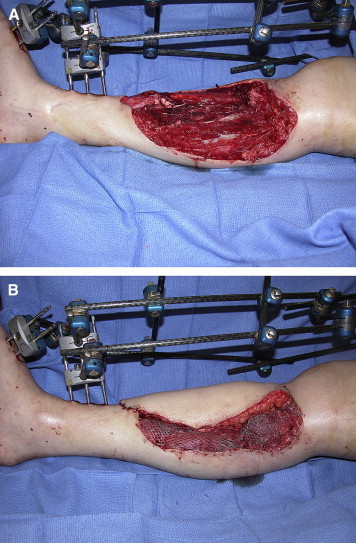
| Proximal 1/3 | Tibial plateau to mid-shaft | Gastrocnemius muscle flap (medial head preferred) | Type I muscle; reliable arc of rotation to proximal tibia |
| Middle 1/3 | Mid-shaft | Soleus muscle flap (hemisoleus or full) | Type II muscle; covers mid-shaft; can be distally or proximally based |
| Distal 1/3 | Distal tibia, ankle | Free flap (ALT, gracilis, latissimus dorsi, radial forearm) | No local muscle flaps reach distal third; free tissue transfer required |
Timing of Coverage
Godina (1986) demonstrated that early free flap coverage (within 72 hours of injury) was associated with a flap failure rate of 0.75%, compared with 12% for flaps performed between 72 hours and 3 months, and 9.5% for flaps after 3 months. Infection rates were also lowest with early coverage (1.5% vs. 17.5%). Modern practice targets definitive soft tissue coverage within 7 days of injury when patient condition allows. The British Orthopaedic Association/British Association of Plastic Surgeons (BOA/BAPS) standard recommends soft tissue coverage within 72 hours for Gustilo IIIB fractures. The “fix and flap” approach advocates combined orthopaedic fixation and soft tissue reconstruction in a single operative session when possible. PMID: 3514036
Diabetic Foot Reconstruction
Diabetic foot ulcers affect 15–25% of diabetics and are the leading cause of non-traumatic amputation. The Wagner classification grades ulcer severity: 0 — intact skin at risk; 1 — superficial ulcer; 2 — deep ulcer to tendon/bone; 3 — deep ulcer with abscess/osteomyelitis; 4 — localised gangrene; 5 — extensive gangrene. Reconstruction options include local muscle flaps (abductor hallucis, abductor digiti minimi, flexor digitorum brevis), medial plantar artery flap (sensate coverage for weight-bearing heel), and free tissue transfer for larger defects. Vascular assessment (ABI, CTA or duplex) and infection control are prerequisites. Offloading (total contact casting) is essential for healing and preventing recurrence.
Limb Salvage vs. Amputation
The MESS (Mangled Extremity Severity Score) was developed to guide limb salvage vs. amputation decisions. Scores ≥ 7 have been associated with amputation, though the score is more useful for predicting which limbs will not require amputation (high negative predictive value). The LEAP trial (Lower Extremity Assessment Project) showed no significant difference in functional outcomes at 2 years between reconstruction and amputation for severe lower extremity trauma, emphasising shared decision-making. PMID: 12473698
12 Trunk & Abdominal Wall Reconstruction
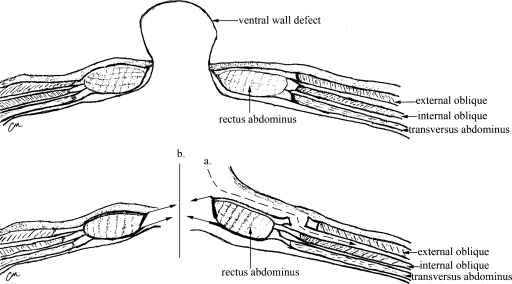
Component Separation Technique
Anterior component separation (Ramirez, 1990): Release of the external oblique aponeurosis 1–2 cm lateral to the rectus sheath from costal margin to inguinal ligament. This allows medial advancement of the rectus complex: up to 10 cm at the waist, 5 cm at the epigastrium, and 3 cm at the suprapubic region. Bilateral release achieves up to 20 cm of midline closure.
Posterior component separation (transversus abdominis release, TAR): Division of the transversus abdominis muscle in the retromuscular plane, posterior to the rectus abdominis. Provides equivalent or greater medial advancement than anterior CS with less disruption of the anterior abdominal wall blood supply (perforators from the deep inferior epigastric artery are preserved). The TAR technique has become the preferred approach for complex ventral hernia repair at many centres. PMID: 22067400
Mesh & Biologic Options
Synthetic mesh (polypropylene, polyester) provides durable repair but should not be placed in contaminated fields due to infection risk. Biologic mesh (AlloDerm, Strattice, Surgisis) derived from decellularised human or porcine dermis/intestinal submucosa can be used in contaminated fields; revascularises and remodels over time but has higher recurrence rates (up to 30% at 5 years). Biosynthetic mesh (GORE BIO-A, Phasix) offers intermediate properties — absorbable scaffold with synthetic strength.
Free Flap Options for Abdominal Wall
For massive abdominal wall defects where component separation is insufficient, free tissue transfer may be required. The tensor fasciae latae (TFL) flap provides vascularised fascia lata for structural support. The ALT flap can provide large skin/soft tissue coverage. Composite reconstruction combining mesh reinforcement with flap coverage may be necessary for full-thickness abdominal wall loss.
Chest Wall Reconstruction
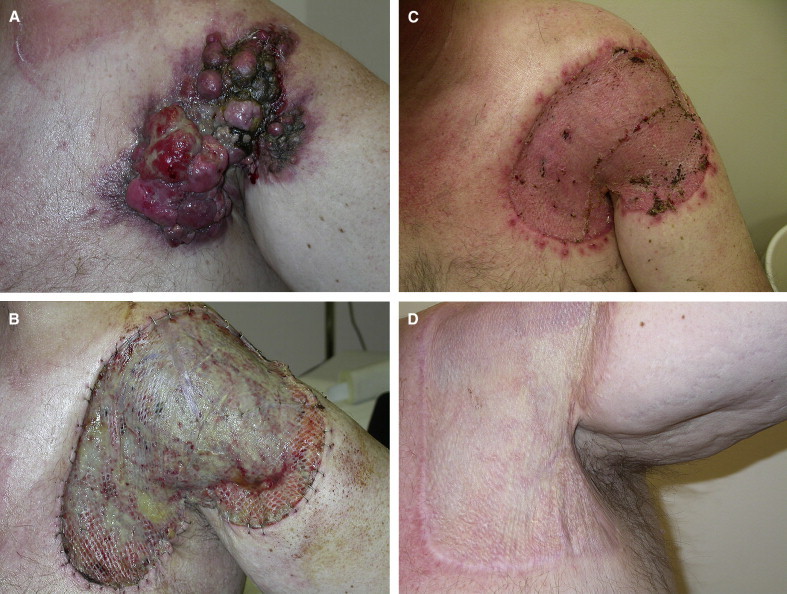
Full-thickness chest wall defects (following tumour resection, radiation necrosis, or infection) require skeletal stabilisation and soft tissue coverage. Skeletal reconstruction: rigid fixation with titanium plates, mesh (polypropylene or PTFE), or methylmethacrylate sandwich (mesh-cement-mesh). Defects < 5 cm or posterior defects protected by the scapula may not require rigid reconstruction. Soft tissue coverage: pectoralis major, latissimus dorsi, rectus abdominis, or omental flaps depending on defect location. Sternal wound infections after cardiac surgery (incidence 1–5%, mortality 10–40%) are treated with debridement and muscle flap closure (bilateral pectoralis major advancement flaps are the workhorse for sternal wounds; rectus abdominis flap for inferior sternal defects; omental flap for deep mediastinal dead space).
13 Pressure Injuries (Decubitus Ulcers)
NPUAP Staging
| Stage | Description | Tissue Involved |
|---|---|---|
| I | Non-blanchable erythema of intact skin | Epidermis intact |
| II | Partial-thickness skin loss (blister or shallow ulcer) | Epidermis & partial dermis |
| III | Full-thickness skin loss; fat visible; no bone/tendon/muscle exposure | Through dermis into subcutaneous fat |
| IV | Full-thickness tissue loss with exposed bone, tendon, or muscle | Through all layers to deep structures |
| Unstageable | Full-thickness loss, base obscured by slough or eschar | Cannot determine depth until debrided |
| Deep tissue injury (DTI) | Purple/maroon discoloured intact skin or blood-filled blister | Damage to underlying soft tissue from pressure/shear |
Flap Options by Location
| Location | First-Line Flap | Alternative Flaps |
|---|---|---|
| Sacral | Superior gluteal artery perforator (SGAP) flap; gluteus maximus V-Y advancement | Bilateral V-Y advancement flaps, posterior thigh rotation flap |
| Ischial | Posterior thigh flap (inferior gluteal artery); hamstring V-Y advancement | Gracilis flap, gluteus maximus rotation flap |
| Trochanteric | TFL (tensor fasciae latae) flap; vastus lateralis flap | Rectus femoris flap, ALT flap |
Management Principles
Successful treatment of pressure injuries requires addressing the underlying cause: pressure offloading (specialty mattresses, turning schedules q2h), nutrition optimisation (albumin > 3.0, prealbumin > 15), management of incontinence, and spasticity control. Surgical reconstruction is indicated for Stage III/IV ulcers that fail conservative management. The recurrence rate for pressure injury flaps is 19–61%, underscoring the importance of modifiable risk factor management. Biopsy non-healing wounds to rule out Marjolin ulcer (SCC).
14 Burns
Burn Depth Classification
| Depth | Old Term | Clinical Findings | Healing |
|---|---|---|---|
| Superficial | 1st degree | Erythema, pain, dry; no blisters (sunburn) | 3–5 days; no scarring |
| Superficial partial-thickness | 2nd degree (superficial) | Blisters, weeping, painful, brisk capillary refill, blanches | 7–14 days from adnexal structures; minimal scarring |
| Deep partial-thickness | 2nd degree (deep) | Pale/mottled, decreased sensation, sluggish capillary refill, may not blanch | 3–6 weeks; significant scarring; may need excision & grafting |
| Full-thickness | 3rd degree | Leathery, waxy, painless (nerve destruction), thrombosed vessels, no blanching | Will not heal without surgery (no adnexal structures remain); requires excision & grafting |
| Subdermal (4th degree) | 4th degree | Extension into muscle, tendon, bone | Requires flap coverage or amputation |
TBSA Calculation
Rule of 9s (Wallace): Adults — head 9%, each upper extremity 9%, anterior trunk 18%, posterior trunk 18%, each lower extremity 18%, perineum 1%. Note: palmar surface (hand including fingers) of the patient = ~1% TBSA (useful for estimating scattered burns). Lund-Browder chart is more accurate, especially in children, as it adjusts for age-related body proportion changes (e.g., a child’s head is proportionally larger).
Fluid Resuscitation — Parkland Formula
Parkland formula: 4 mL × body weight (kg) × %TBSA burned (second- and third-degree only). Give half in the first 8 hours from the time of burn (not from arrival), and the remaining half over the next 16 hours. Use lactated Ringer’s solution. Titrate to urine output: adults 0.5–1.0 mL/kg/hr, children 1.0–1.5 mL/kg/hr.
Emergency Escharotomy is performed for circumferential full-thickness burns causing compartment syndrome of the extremity or respiratory compromise from trunk burns. Incisions are made through the eschar (insensate full-thickness burn) down to subcutaneous fat along the mid-lateral and mid-medial lines. No anaesthesia is needed for full-thickness burns. Chest escharotomy uses bilateral anterior axillary line incisions connected by a transverse incision at the costal margins.
Burn Wound Management
Silver sulfadiazine (Silvadene): Broad-spectrum topical antimicrobial; may cause transient leukopenia (5–15%); do not use on face (causes discolouration) or in sulfa allergy. Mafenide acetate (Sulfamylon): Penetrates eschar; painful on application; carbonic anhydrase inhibitor (can cause metabolic acidosis); excellent for ear cartilage burns. Silver nitrate (0.5%): Broad-spectrum; stains everything black; can cause electrolyte abnormalities (hyponatremia, hypocalcaemia).
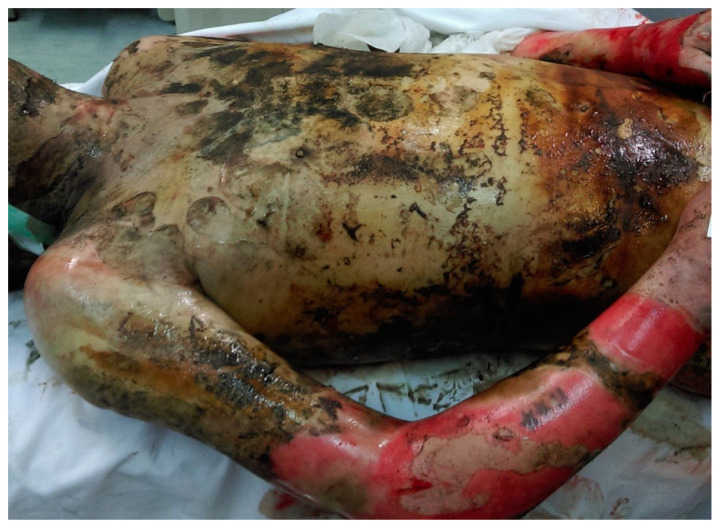
Early excision and grafting (within 24–72 hours) for deep partial-thickness and full-thickness burns has reduced mortality and hospital stay compared to conservative management. Tangential excision removes sequential thin layers of burned tissue until punctate bleeding is seen (viable dermis). Fascial excision is reserved for massive burns.
Inhalation Injury
Inhalation injury is the leading cause of death in house fires and significantly increases burn mortality (from ~5% to ~30% when combined with cutaneous burns). Diagnosis: history of closed-space exposure, singed nasal hairs, carbonaceous sputum, hoarseness, stridor. Confirmed by fibreoptic bronchoscopy (erythema, oedema, soot deposition, mucosal sloughing). Emergency Early endotracheal intubation is mandatory if inhalation injury is suspected, as airway oedema progresses rapidly and may become impossible to intubate within hours. Carbon monoxide poisoning: carboxyhaemoglobin (COHb) levels > 20% are significant; treat with 100% FiO2 (half-life of COHb: 4–5 hours on room air, reduced to 60–90 minutes on 100% O2). Cyanide poisoning from combustion of synthetic materials: treat with hydroxocobalamin (Cyanokit) 5 g IV.
Electrical Burns
Electrical burns cause tissue injury far beyond the visible surface wound (“iceberg” phenomenon). Current follows the path of least resistance (nerves, blood vessels > muscle > skin > bone). Complications: cardiac arrhythmias (ECG monitoring mandatory for high-voltage > 1,000 V injuries), rhabdomyolysis (monitor CK, maintain UOP > 1 mL/kg/hr, alkalinise urine), compartment syndrome (early fasciotomy), delayed vascular thrombosis, and progressive tissue necrosis requiring serial debridements. Maintain urine output at 1–2 mL/kg/hr (higher than thermal burns due to myoglobinuria risk).
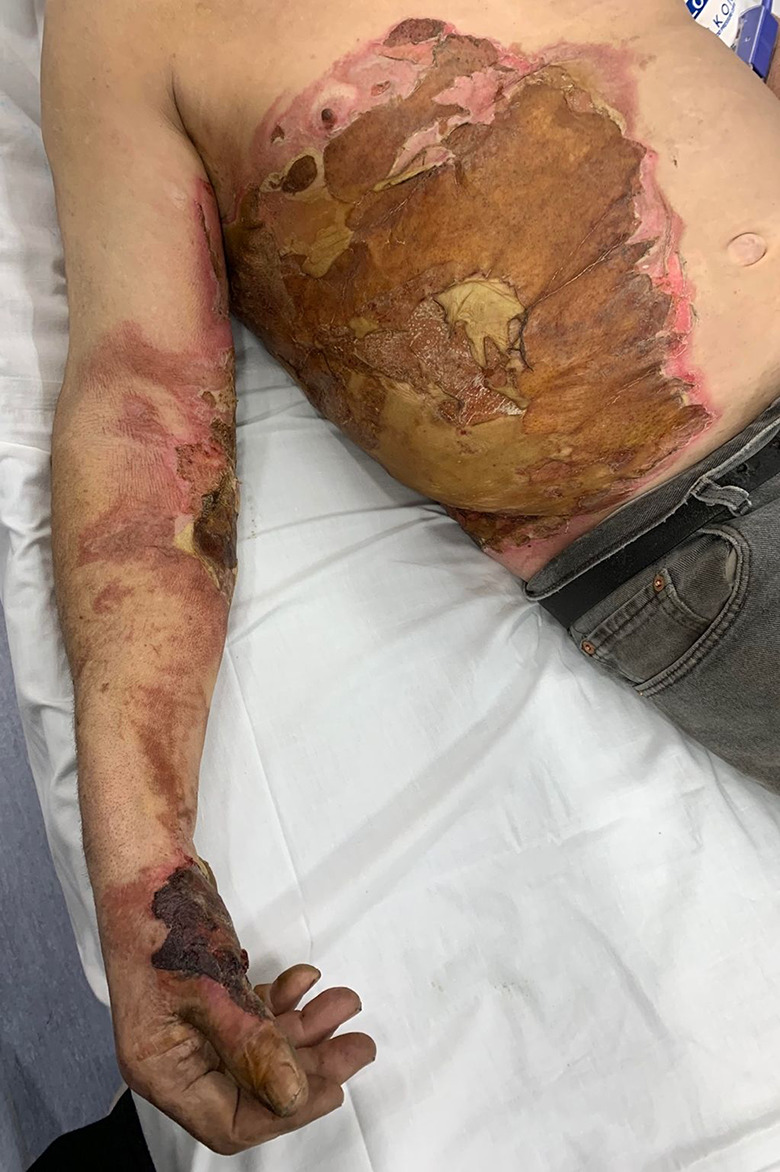
Burn Reconstruction
Scar management begins early: pressure garments (23 hours/day for 12–18 months), silicone sheeting, splinting to prevent contractures. Surgical reconstruction for burn contractures includes Z-plasty, skin grafting, local flaps, tissue expansion, and free tissue transfer. Burn scar contractures across joints may severely limit function and require release with FTSG or flap coverage. Tissue expansion is particularly valuable in burn reconstruction, allowing expansion of adjacent unburned skin to provide excellent colour and texture match. Expanders are inflated serially over 6–12 weeks, then the expanded flap is advanced into the scar-excised defect.
15 Cleft Lip & Palate
Embryology & Epidemiology
Cleft lip results from failure of fusion of the medial nasal prominence with the maxillary prominence during weeks 4–7 of gestation. Cleft palate results from failure of fusion of the palatal shelves during weeks 8–12. Cleft lip ± palate (CL/P) occurs in ~1:700 live births; isolated cleft palate (CP) in ~1:2,000. CL/P is more common in males and in Asian populations; isolated CP is more common in females. Approximately 70% of cleft cases are non-syndromic; 30% are associated with syndromes (Pierre Robin sequence, Van der Woude, Stickler, velocardiofacial/22q11.2 deletion).
Classification Systems
Veau Classification:
| Class | Description |
|---|---|
| I | Soft palate only |
| II | Soft and hard palate (to incisive foramen) |
| III | Complete unilateral cleft (lip, alveolus, and palate) |
| IV | Complete bilateral cleft (lip, alveolus, and palate) |
Kernahan striped-Y classification provides a visual diagram of the cleft, with each segment of the Y representing a portion of the lip, alveolus, and palate bilaterally. Used for documentation and communication.
Timing of Repair — Rule of 10s
Lip repair: ~3 months (10 weeks old, 10 lbs, Hgb 10 g/dL). Palate repair: ~10–12 months (before speech development). Alveolar bone graft: 6–9 years (mixed dentition, before canine eruption). Rhinoplasty/orthognathic surgery: After skeletal maturity (16–18 years).
Surgical Techniques
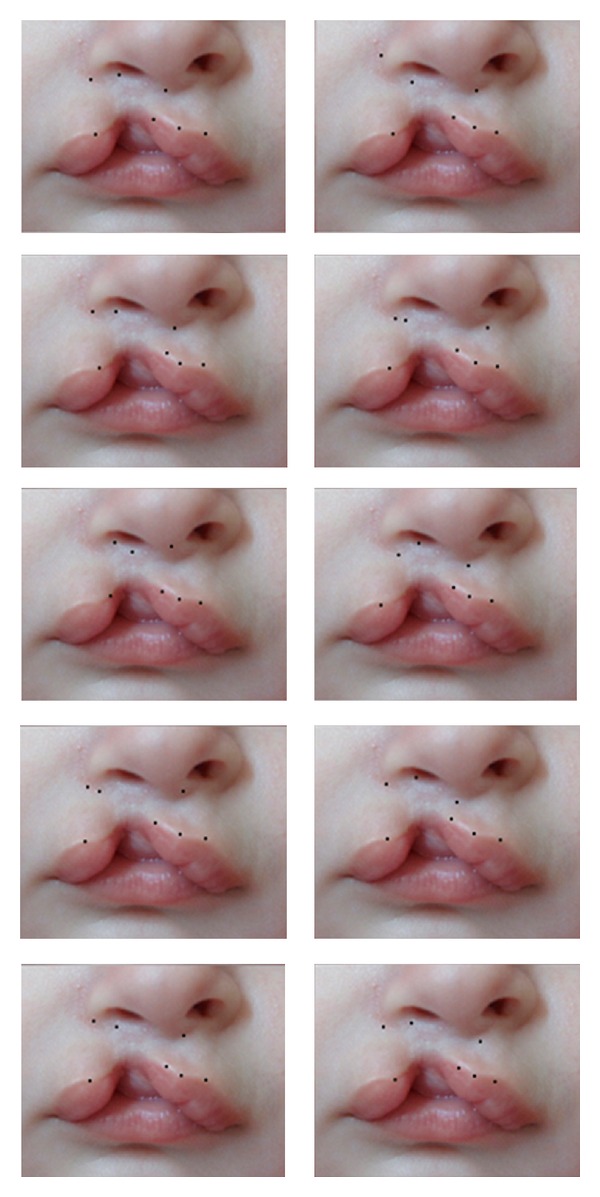
Cleft lip repair: The Millard rotation-advancement technique is the most widely used method for unilateral cleft lip repair. The medial lip element is rotated downward (rotation flap) and the lateral element is advanced into the gap (advancement flap). Preserves the philtral column and Cupid’s bow. The Tennison-Randall (triangular flap) technique is an alternative that produces a predictable lip length but leaves a scar across the philtrum.
Cleft palate repair: The Furlow double-opposing Z-plasty lengthens the palate by re-orienting the levator veli palatini muscles in the soft palate, creating a functional muscular sling. The Bardach two-flap palatoplasty raises bilateral mucoperiosteal flaps that are mobilised medially for midline closure. The intravelar veloplasty (IVVP) detaches the levator muscles from their abnormal insertion on the hard palate and repositions them transversely.
Velopharyngeal Insufficiency (VPI)
VPI occurs in 10–30% of patients after palate repair, causing hypernasal speech. Diagnosed with nasendoscopy and videofluoroscopy. Treatment: pharyngeal flap (superiorly based midline pharyngeal flap sutured to the soft palate — best for sagittal closure pattern), sphincter pharyngoplasty (bilateral palatopharyngeus muscle flaps inset into the posterior pharyngeal wall — best for circular closure pattern), or Furlow palatoplasty (if initial repair was a straight-line technique).
16 Craniofacial Surgery
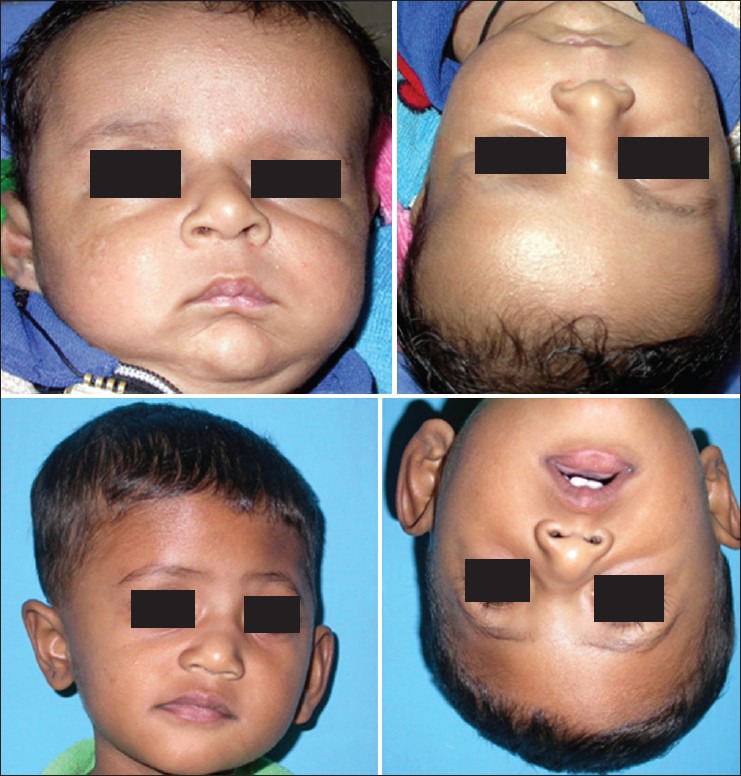
Craniosynostosis
Craniosynostosis is premature fusion of one or more cranial sutures, resulting in restricted growth perpendicular to the fused suture and compensatory growth parallel to it (Virchow’s law). Incidence: ~1:2,000–2,500 live births. 80–90% are non-syndromic (single suture); 10–20% are syndromic.
| Fused Suture | Head Shape | Frequency |
|---|---|---|
| Sagittal | Scaphocephaly/dolichocephaly (long, narrow) | Most common (~40–55%) |
| Coronal (unilateral) | Anterior plagiocephaly (flattened forehead on affected side, “harlequin” orbit) | ~20–25% |
| Metopic | Trigonocephaly (triangular forehead, hypotelorism) | ~10–15% |
| Lambdoid (unilateral) | Posterior plagiocephaly (flattened occiput on affected side, ipsilateral ear displaced posteroinferiorly) | ~1–3% |
| Coronal (bilateral) | Brachycephaly (short, wide) | ~5–10% |
Syndromic craniosynostosis: Crouzon syndrome (FGFR2 mutation; bicoronal synostosis, midface hypoplasia, proptosis, no limb anomalies); Apert syndrome (FGFR2; bicoronal synostosis, severe midface hypoplasia, bilateral symmetric syndactyly of hands and feet — “mitten hands”); Pfeiffer syndrome (FGFR1/FGFR2; broad thumbs/great toes, variable craniosynostosis); Muenke syndrome (FGFR3 P250R; unicoronal or bicoronal, hearing loss).
Le Fort Fractures
| Type | Fracture Pattern | Clinical Findings |
|---|---|---|
| Le Fort I | Transverse fracture through the maxilla above the teeth (horizontal maxillary fracture) | Mobile upper dental arch; midface swelling |
| Le Fort II | Pyramidal fracture through nasofrontal suture, medial orbital wall, infraorbital rim, and pterygoid plates | Mobile midface (nose and maxilla move together); bilateral periorbital ecchymosis (“raccoon eyes”); CSF rhinorrhoea |
| Le Fort III | Craniofacial dysjunction — fracture through nasofrontal suture, orbital walls, and zygomatic arches separating the face from the skull | Entire face mobile relative to cranium (“dish face”); significant oedema; CSF leak; associated intracranial injuries |
Orbital Fractures
Orbital floor (blowout) fractures: Caused by increased intraorbital pressure (direct blow to globe). Findings: enophthalmos, hypoglobus, infraorbital nerve hypoaesthesia (V2), diplopia (entrapment of inferior rectus or periorbital fat herniation). CT findings: “teardrop sign” (herniated contents in maxillary sinus). Surgical indications: diplopia with positive forced duction test, enophthalmos > 2 mm, large floor defect (> 50% of floor or > 2 cm2). Emergency Trapdoor fracture in children: “white-eyed blowout” with incarcerated inferior rectus causing oculocardiac reflex (bradycardia, nausea) — requires urgent repair within 24–48 hours to prevent permanent muscle ischaemia and fibrosis.
Zygomatic & Nasal Fractures
Zygomaticomaxillary complex (ZMC) fractures involve disruption at the zygomaticofrontal suture, zygomaticomaxillary buttress, zygomatic arch, and orbital floor/lateral wall. Assessment: malar flattening, trismus (coronoid impingement), infraorbital nerve hypoaesthesia (V2), step deformity at the infraorbital rim. CT with 3D reconstruction is the imaging of choice. Treatment: ORIF via multiple approaches (lateral brow, subciliary/transconjunctival, upper buccal sulcus incisions) with mini-plate fixation at 2–3 points. An isolated zygomatic arch fracture (depressed arch causing trismus) may be reduced via a Gillies approach (temporal incision, elevator placed deep to the temporal fascia).
Nasal fractures are the most common facial fracture. Diagnosis is clinical (swelling, tenderness, deviation, crepitus, epistaxis). CT is not routinely needed unless other fractures are suspected. Septal haematoma must be ruled out (bilateral boggy, purple swelling of the septum) — Emergency requires immediate drainage to prevent cartilage necrosis and subsequent saddle nose deformity. Closed reduction of nasal fractures should be performed within 2 weeks of injury (ideally 5–10 days, after swelling subsides but before bony union).
Distraction Osteogenesis
Distraction osteogenesis (DO) applies the principle of gradual mechanical tension to generate new bone (Ilizarov). Used in craniofacial surgery for mandibular lengthening (Pierre Robin sequence, hemifacial microsomia), midface advancement (syndromic craniosynostosis), and cranial vault expansion. Protocol: osteotomy → latency (5–7 days) → distraction (1 mm/day in 2 increments) → consolidation (6–8 weeks for bone mineralisation). PMID: 1597224
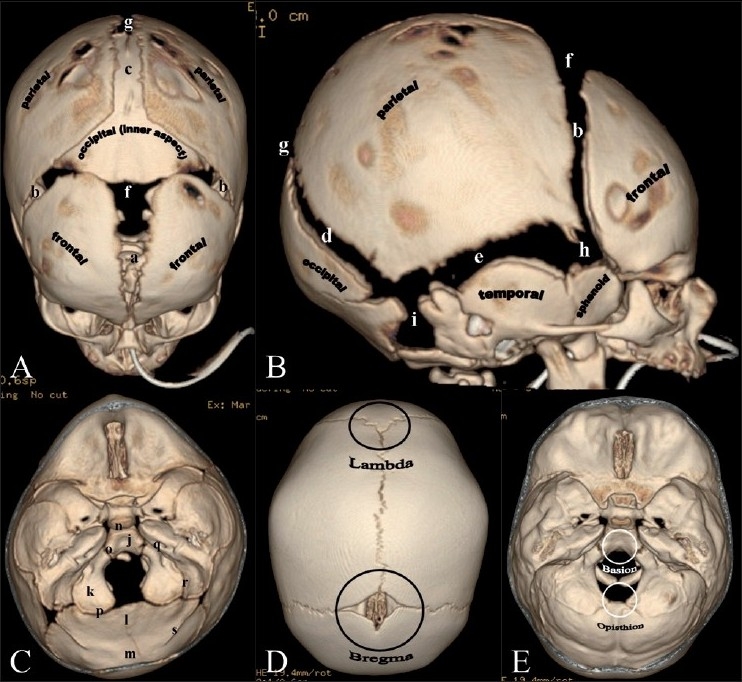
17 Congenital Hand Anomalies
Swanson Classification (Modified by IFSSH)
| Category | Description | Examples |
|---|---|---|
| I | Failure of formation (arrest of development) | Radial club hand (radial longitudinal deficiency), phocomelia, transverse deficiency (congenital amputation) |
| II | Failure of differentiation (separation) | Syndactyly, symphalangism, camptodactyly, clinodactyly, radiohumeral synostosis |
| III | Duplication | Polydactyly (preaxial = thumb/radial side; postaxial = small finger/ulnar side; central) |
| IV | Overgrowth | Macrodactyly |
| V | Undergrowth | Thumb hypoplasia (Blauth classification), brachydactyly |
| VI | Constriction band syndrome | Amniotic band syndrome |
| VII | Generalised skeletal anomalies | Achondroplasia, Marfan syndrome |
Trigger Thumb & Hand Surgery Basics
Trigger thumb (paediatric): Palpable nodule (Notta’s node) at the A1 pulley. Present in ~3% of children by age 1. Observation for first 12 months (spontaneous resolution in 30–60%); if persistent, A1 pulley release. Tendon repair in the hand: The flexor sheath is divided into zones (I–V). Zone II (“no man’s land” from the A1 pulley to the FDS insertion) was historically considered irreparable due to adhesion formation; modern 4-strand core suture + epitendinous repair followed by early active motion protocols has yielded good results. Key pulleys to preserve: A2 (proximal phalanx) and A4 (middle phalanx) to prevent bowstringing.
Syndactyly
Fusion of adjacent digits; incidence ~1:2,000 live births. Simple (skin only) vs. complex (bony fusion). Complete (extends to fingertip) vs. incomplete (does not reach fingertip). Most common: 3rd web space (long-ring fingers). Repair at 12–18 months; earlier (6 months) for border digits (thumb-index, ring-small) where growth discrepancy causes angular deformity. Technique: interdigitating triangular flaps with FTSG to cover defects on the lateral surfaces of the separated digits.
Polydactyly
Preaxial (thumb) polydactyly: Wassel classification (Types I–VII based on level of duplication). Most common type: Wassel IV (duplicated proximal phalanx). Treatment: ablation of the less functional digit with reconstruction of the retained thumb (collateral ligament, tendon, and nail fold reconstruction). Postaxial (ulnar) polydactyly: Type A (well-formed digit) requires formal surgical excision; Type B (pedunculated “nubbin”) can be treated with suture ligation in the neonatal period (though surgical excision is preferred by some to avoid residual bumps).
Thumb Hypoplasia — Blauth Classification
| Type | Features | Treatment |
|---|---|---|
| I | Minor hypoplasia; all structures present | No surgery needed |
| II | Hypoplastic thumb with tight first web space, UCL laxity, thenar muscle deficiency | First web deepening, UCL reconstruction, opponensplasty (FDS ring finger transfer) |
| IIIA | Type II features + stable CMC joint, extrinsic tendon abnormalities | Reconstruction as Type II with tendon transfers |
| IIIB | Type II features + unstable or aplastic CMC joint | Pollicisation (index finger transfer to thumb position) |
| IV | Floating thumb (pouce flottant) — attached only by skin pedicle | Pollicisation |
| V | Absent thumb | Pollicisation |
Radial Longitudinal Deficiency (Radial Club Hand)
Spectrum from hypoplastic thumb to complete absence of the radius. Associated with VACTERL association (Vertebral, Anal atresia, Cardiac, TE fistula, Renal, Limb anomalies), Holt-Oram syndrome (cardiac defects), TAR syndrome (Thrombocytopenia-Absent Radius), and Fanconi anaemia (bone marrow failure). Workup: echocardiography, renal ultrasound, spinal radiographs, CBC, and chromosomal breakage analysis (Fanconi). Treatment: serial splinting/stretching, then centralisation or radialisation of the carpus on the ulna at 6–12 months, followed by pollicisation (index finger transfer to thumb position) if the thumb is absent or severely hypoplastic (Blauth III–V).
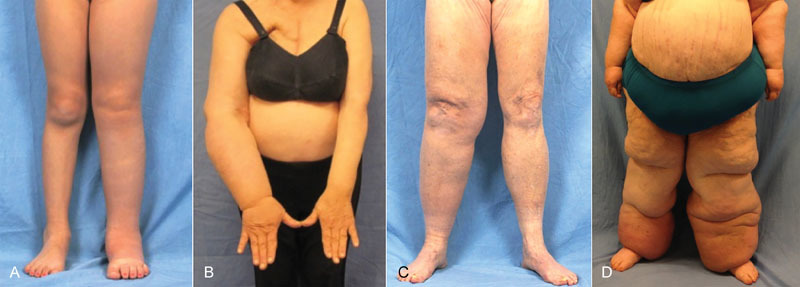
18 Lymphedema
Classification
Primary lymphedema: Congenital absence or dysfunction of lymphatics. Milroy disease (congenital, at birth; VEGFR3 mutation); lymphedema praecox (onset at puberty, most common form of primary lymphedema, predominantly female); lymphedema tarda (onset > 35 years).
Secondary lymphedema: Acquired obstruction or destruction of lymphatics. Most common cause worldwide: filariasis (Wuchereria bancrofti). Most common cause in developed countries: cancer treatment (axillary lymph node dissection for breast cancer — incidence 20–30%; sentinel lymph node biopsy alone — incidence 5–7%). Also caused by radiation, trauma, infection, obesity.
ISL Staging
| Stage | Description |
|---|---|
| 0 (Subclinical) | Impaired lymphatic drainage but no visible swelling; detectable by lymphoscintigraphy or bioimpedance |
| I (Reversible) | Pitting oedema that resolves with elevation; minimal fibrosis |
| II (Spontaneously irreversible) | Non-pitting oedema that does not resolve with elevation; progressive fibrosis and fat deposition |
| III (Lymphostatic elephantiasis) | Severe swelling with skin changes (papillomatosis, hyperkeratosis), recurrent infections (cellulitis), fat hypertrophy |
Treatment
Conservative: Complete decongestive therapy (CDT) — manual lymphatic drainage, compression bandaging/garments, exercise, skin care. Remains the first-line treatment for all stages.
Microsurgical: Lymphovenous anastomosis (LVA) connects lymphatic channels to subdermal venules (supermicrosurgery, vessels < 0.8 mm). Best results in early-stage (ISL I–II) lymphedema. Vascularised lymph node transfer (VLNT) transfers a lymph node flap (groin, submental, supraclavicular, or lateral thoracic nodes) to the affected extremity. The transferred nodes promote lymphangiogenesis. Best for moderate-to-severe lymphedema or when LVA is not feasible. PMID: 22286048
Debulking: Suction-assisted protein lipectomy (SAPL / liposuction) removes adipose hypertrophy in chronic lymphedema with non-pitting component (ISL Stage II–III). Requires lifelong compression garment use post-procedure. The Charles procedure (radical excision of skin, subcutaneous tissue, and fascia with STSG) is reserved for severe elephantiasis refractory to other treatments.
19 Gender-Affirming Surgery
Overview
Gender-affirming surgical procedures are performed as part of a multidisciplinary approach following WPATH (World Professional Association for Transgender Health) Standards of Care. Referral requirements typically include persistent, well-documented gender dysphoria, informed consent, and appropriate duration of hormone therapy (12 months for genital surgery).
Masculinising Procedures
Chest masculinisation (top surgery): Double-incision mastectomy with free nipple grafts (most common for larger breasts); periareolar/keyhole technique for smaller breasts. Phalloplasty: Radial forearm free flap (most common), ALT flap, or fibula flap. Requires microsurgical expertise. Urethroplasty, glansplasty, and testicular prostheses may be staged. Complication rates are significant: urethral fistula (15–50%), stricture (25–40%). Metoidioplasty: Release and advancement of the hormonally enlarged clitoris; smaller phallus but fewer complications and preserved erogenous sensation.
Feminising Procedures
Vaginoplasty: Penile inversion vaginoplasty (most common technique) uses penile and scrotal skin to create the vaginal canal. Peritoneal pull-through vaginoplasty and sigmoid colon vaginoplasty are alternatives when skin is insufficient. Requires lifelong vaginal dilation postoperatively to maintain canal patency. Breast augmentation: After minimum 12 months of oestrogen therapy; techniques similar to cisgender breast augmentation. Facial feminisation surgery (FFS): Brow bone reduction (Type III osteoplasty), rhinoplasty, jawline contouring, tracheal shave (chondrolaryngoplasty).
Vascular Anomalies
Vascular anomalies are classified by the ISSVA (International Society for the Study of Vascular Anomalies) into vascular tumours (proliferative, e.g., infantile haemangioma) and vascular malformations (structural, present at birth, grow with the child). Infantile haemangiomas are the most common tumours of infancy (4–5% of infants, 3:1 female:male). They follow a characteristic course: rapid proliferation (0–12 months), then slow involution (1–10 years; 50% resolved by age 5, 90% by age 9). Most require observation only. Treatment (when indicated for functional impairment, ulceration, or disfigurement): propranolol 2–3 mg/kg/day PO (first-line since the HEMANGEOL trial; monitor heart rate, blood pressure, and blood glucose). PMID: 25643702
Vascular malformations: Classified by flow rate and vessel type. Low-flow: capillary (port-wine stain — treat with pulsed dye laser), venous (compressible, blue, enlarge with Valsalva; treat with sclerotherapy), lymphatic (microcystic or macrocystic; macrocystic amenable to sclerotherapy). High-flow: arteriovenous malformations (AVM) — Schobinger staging (I: quiescence; II: expansion; III: destruction; IV: decompensation/heart failure). AVMs do not involute and may worsen with incomplete treatment. Definitive treatment: complete surgical excision after preoperative embolisation.
20 Rhinoplasty
Approaches
Open (external) rhinoplasty: Transcolumellar incision connected to bilateral marginal incisions. Provides direct visualisation of the nasal framework. Preferred for complex revision rhinoplasty, tip work, and cases requiring precise graft placement. Disadvantage: columellar scar (usually inconspicuous). Closed (endonasal) rhinoplasty: All incisions inside the nose (intercartilaginous, transfixion, marginal). Advantages: no external scar, less tip oedema, shorter operating time. Disadvantage: limited exposure.
Septoplasty & Turbinate Reduction
Often combined with rhinoplasty for functional nasal obstruction. Septoplasty corrects a deviated nasal septum through a hemitransfixion or Killian incision. A minimum of 1.5 cm of dorsal and caudal strut (L-strut) must be preserved to maintain nasal tip support and dorsal profile. Inferior turbinate hypertrophy contributes to nasal obstruction and is addressed with submucosal reduction (radiofrequency, microdebrider, or outfracture).
Nasal Anatomy & Analysis
The nose is divided into thirds: upper (nasal bones), middle (upper lateral cartilages, septum), and lower (lower lateral/alar cartilages). The internal nasal valve (angle between the upper lateral cartilage and septum, normally 10–15°) is the area of greatest airflow resistance. Collapse here causes nasal obstruction. A spreader graft (autologous cartilage placed between the upper lateral cartilage and septum) widens the internal valve angle and is the standard treatment.
Complications of Rhinoplasty
Early: bleeding/epistaxis (1–4%), infection (< 1%), septal haematoma. Late: asymmetry, over- or under-resection, persistent dorsal hump, pollybeak deformity (supratip fullness from inadequate cartilage reduction or excessive scar tissue), inverted-V deformity (visible transition between nasal bones and upper lateral cartilages after hump removal — prevented by spreader grafts), saddle nose deformity (excessive dorsal resection or septal perforation), tip ptosis, nasal obstruction (internal valve collapse). Revision rhinoplasty rate: 5–15%. Rib cartilage grafting is often necessary in revision cases when septal and auricular cartilage have been previously harvested.
Structural Grafting
Columellar strut: Cartilage graft between the medial crura to provide tip support. Shield graft: Placed at the tip to define the tip-defining points. Alar batten graft: Placed lateral to the existing lower lateral cartilage to prevent external valve collapse. Spreader grafts: Dorsal onlay grafts between the upper lateral cartilage and septum. Graft sources: septal cartilage (preferred), auricular cartilage, costal cartilage (for revision or when septal/auricular cartilage is insufficient).
Tip Dynamics
The tripod concept (Anderson) describes the nasal tip as a tripod: two lateral crura and a conjoined medial crural unit. Shortening or lengthening any leg changes tip position, projection, and rotation. Tip projection is assessed by the Goode ratio (nasal projection/nasal length = 0.55–0.60). Tip rotation is measured by the nasolabial angle (90–95° in males, 95–110° in females).
21 Blepharoplasty & Facelift
Upper Blepharoplasty
Removes excess skin (dermatochalasis) and protruding orbital fat from the upper eyelids. Functional indication: visual field obstruction (> 30% superior field loss on formal visual field testing). The incision is placed in the supratarsal crease (8–10 mm from the lid margin). At least 20 mm of skin must remain between the brow and upper lid margin after excision to allow complete eyelid closure (pinch test). The medial fat pad (white, denser, medial to the orbital septum) is distinct from the central (preaponeurotic) fat pad (yellow, softer).
Lower Blepharoplasty
Transcutaneous approach: Subciliary incision 2 mm below the lash line; allows skin/muscle excision and fat repositioning. Risk of lower lid retraction/ectropion. Transconjunctival approach: Incision through the conjunctiva (no external scar); preferred for fat removal/repositioning without skin excess. Lower lid laxity (positive snap-back test, distraction > 6 mm) requires concurrent lid-tightening (canthopexy or canthoplasty) to prevent postoperative ectropion.
Rhytidectomy (Facelift)
The SMAS (superficial musculoaponeurotic system) is a fibromuscular layer continuous with the platysma inferiorly and the temporoparietal fascia superiorly. Modern facelift techniques address the SMAS to achieve long-lasting, natural-appearing results:
SMAS plication: SMAS is folded and sutured without dissection deep to it. Simplest technique; least risk to facial nerve. SMASectomy: Excision of a strip of SMAS followed by closure. SMAS flap (deep plane): The SMAS is elevated as a composite flap with the overlying skin (deep plane facelift — extended sub-SMAS dissection). Provides more powerful lifting of the midface and nasolabial fold. Higher risk to the facial nerve, especially the marginal mandibular branch (most commonly injured) and the frontal (temporal) branch.
Danger zones for facial nerve injury: The frontal branch crosses the zygomatic arch within the temporoparietal fascia (superficial temporal fascia) — dissection should stay superficial to the deep temporal fascia in the temporal region. The marginal mandibular nerve courses superficial to the facial artery and vein at the angle of the mandible — vulnerable 1–2 cm below the mandibular border.
Brow Lift
Endoscopic brow lift: Three to five small incisions behind the hairline; subperiosteal dissection with release of the periosteum at the orbital rim and the conjoined tendon (zone of adhesion between the orbital ligament and the temporal fusion line). The brow is resuspended with cortical tunnels, bone anchors, or Endotine fixation devices. Advantages over coronal brow lift: shorter scar, less sensory loss, less alopecia. Contraindicated in patients with high hairline (use direct, mid-forehead, or pretrichial approach instead). The temporal branch of the facial nerve courses within the temporoparietal fascia (superficial to the deep temporal fascia) — dissection in the temple must remain in the subperiosteal plane (deep to both layers) or superficial to the temporoparietal fascia to avoid injury.
22 Abdominoplasty & Body Contouring
Abdominoplasty
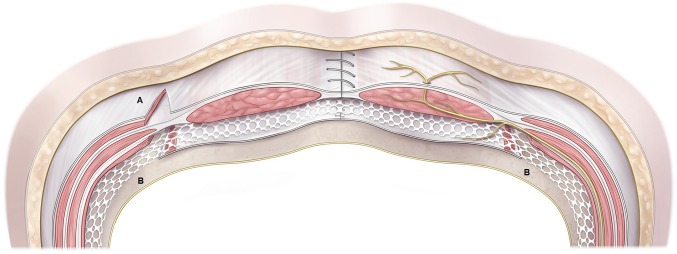
Full abdominoplasty: Low transverse incision (hip to hip), umbilical transposition, undermining of abdominal skin flap to the costal margins, rectus diastasis repair (plication of the anterior rectus sheath in the midline), and excision of redundant skin and fat. Mini-abdominoplasty: Shorter incision, no umbilical transposition, limited undermining for infraumbilical laxity only.
Perfusion zones: Blood supply to the abdominal flap after undermining is primarily from the intercostal, subcostal, and lumbar perforators laterally. The deep inferior epigastric perforators are divided during flap elevation. Smoking is the greatest risk factor for flap necrosis and must be ceased ≥ 4 weeks preoperatively. Combining abdominoplasty with liposuction was historically controversial; the current evidence supports limited liposuction of the flanks at the time of abdominoplasty if lateral undermining is avoided.
Massive Weight Loss Body Contouring
Following bariatric surgery or significant weight loss (> 50 lbs), patients develop significant tissue laxity affecting multiple body regions. Common procedures:
| Procedure | Target Area | Key Technique Points |
|---|---|---|
| Circumferential body lift (belt lipectomy) | Abdomen, flanks, mons pubis, buttock, lateral thigh | Circumferential excision of excess tissue; includes abdominoplasty + buttock/thigh lift |
| Brachioplasty | Upper arm | Medial arm incision from axilla to medial epicondyle; visible scar is main drawback |
| Medial thighplasty | Inner thigh | Vertical or horizontal (groin crease) excision pattern; risk of scar migration, labial distortion |
| Upper body lift | Back, bra line | Horizontal excision of back rolls; often combined with breast procedures |
Patients should be at a stable weight for at least 6 months before body contouring surgery. Nutritional deficiencies (protein, iron, B12, vitamin D) from malabsorptive bariatric procedures must be corrected preoperatively. Liposuction (suction-assisted lipectomy) removes localised fat deposits through small cannulas via tumescent technique (infiltration of dilute lidocaine + epinephrine in saline). Safe removal limit: ≤ 5 L of aspirate for outpatient procedures. Alternatives: ultrasound-assisted liposuction (VASER), power-assisted liposuction (PAL), and laser-assisted liposuction. Complications: contour irregularity (most common), seroma, skin necrosis, fat embolism, lidocaine toxicity (maximum safe tumescent dose: 35–55 mg/kg due to slow absorption from adipose tissue).
23 Breast Augmentation & Mastopexy
Implant Types
Silicone gel implants: Cohesive silicone gel (“gummy bear” = form-stable, highly cohesive). More natural feel than saline. If ruptured, gel may remain within the capsule (“silent rupture”) — FDA recommends MRI screening at 5–6 years post-implantation, then every 2–3 years. Saline implants: Filled intraoperatively (adjustable volume). If ruptured, deflation is immediate and obvious; saline is absorbed harmlessly. Less natural feel; more visible rippling, especially in thin patients.
Implant Pocket Planes
| Plane | Description | Pros | Cons |
|---|---|---|---|
| Subglandular | Above pectoralis major muscle | Easier dissection; less animation deformity; better for ptotic breasts | Higher capsular contracture rate; more visible/palpable implant; mammography interference |
| Submuscular (total) | Completely beneath pectoralis major | More tissue coverage; lower capsular contracture rate | Animation deformity; more painful; “bottoming out” |
| Dual plane | Upper pole subpectoral, lower pole subglandular (muscle released from parenchyma inferiorly) | Balanced coverage and aesthetics; most versatile | More complex dissection; requires precise pocket creation |
Baker Classification of Capsular Contracture
| Grade | Description |
|---|---|
| I | Breast looks and feels natural (normal) |
| II | Minimal firmness; breast looks normal but feels slightly firm |
| III | Moderate firmness; breast feels firm and appears distorted |
| IV | Severe contracture; breast is hard, painful, and distorted |
Breast Implant Illness
Breast implant illness (BII) is a term used by patients to describe systemic symptoms attributed to breast implants, including fatigue, cognitive dysfunction (“brain fog”), arthralgias, myalgias, and autoimmune-like symptoms. While a definitive causal link has not been established, the FDA mandated a boxed warning on breast implants in 2020. Many patients report symptom improvement after explantation with en bloc capsulectomy. Shared decision-making is essential.
Treatment for symptomatic capsular contracture (Baker III/IV): capsulectomy (complete removal of capsule) with implant exchange, change of pocket plane (e.g., subglandular to submuscular), or conversion to autologous reconstruction. Capsulotomy (scoring of the capsule without removal) has a high recurrence rate.
Breast Implant-Associated Anaplastic Large Cell Lymphoma (BIA-ALCL)
A rare T-cell lymphoma associated primarily with textured surface implants. Estimated incidence: 1:2,207 to 1:86,029 depending on implant type. Presents as a late seroma (> 1 year post-implantation), capsular mass, or lymphadenopathy. Diagnosis: cytology and flow cytometry of periprosthetic fluid (CD30+, ALK–). Treatment: total capsulectomy with implant removal (most cases are localised and cured by surgery alone). PMID: 28296502
Mastopexy
Breast lift surgery corrects ptosis (Regnault classification: Grade I — nipple at the inframammary fold; Grade II — nipple below the fold but above the lowest breast contour; Grade III — nipple at the lowest breast contour; Pseudoptosis — nipple above the fold but inferior pole descends). Techniques: periareolar (Benelli, for mild ptosis), vertical (lollipop scar, for moderate ptosis), and Wise-pattern (inverted T/anchor scar, for severe ptosis).
24 Injectables & Non-Surgical Aesthetics
Neurotoxins (Botulinum Toxin)
Botulinum toxin type A (Botox/onabotulinumtoxinA, Dysport/abobotulinumtoxinA, Xeomin/incobotulinumtoxinA) inhibits acetylcholine release at the neuromuscular junction, causing temporary muscle paralysis. Onset: 2–5 days; peak effect: 1–2 weeks; duration: 3–4 months. Common aesthetic uses: glabellar lines (corrugator supercilii, procerus), forehead lines (frontalis), crow’s feet (orbicularis oculi), platysmal bands, masseter reduction (facial slimming). Dosing is not interchangeable between products (Botox:Dysport ratio approximately 1:2.5–3).
Dermal Fillers
| Filler Type | Material | Duration | Reversibility | Common Uses |
|---|---|---|---|---|
| Hyaluronic acid (HA) | Cross-linked HA (Juvederm, Restylane families) | 6–18 months | Yes (hyaluronidase) | Lips, nasolabial folds, marionette lines, tear troughs, cheeks |
| Calcium hydroxylapatite (CaHA) | Radiesse | 12–18 months | Not directly reversible | Cheeks, jawline, hands; biostimulator |
| Poly-L-lactic acid (PLLA) | Sculptra | ~2 years (gradual collagen stimulation) | No | Volume restoration (temples, midface, cheeks); HIV lipoatrophy |
| Polymethylmethacrylate (PMMA) | Bellafill | Permanent | No (requires excision) | Nasolabial folds, acne scars |
Danger Zones for Filler Injection
Inadvertent intravascular injection can cause tissue necrosis or blindness. Critical danger zones:
Glabella / dorsal nose: The supratrochlear and dorsal nasal arteries communicate with the ophthalmic artery via the angular artery. Retrograde embolisation can cause retinal artery occlusion and blindness (reported with all filler types, most commonly in the glabella, nose, and nasolabial fold). Occurs within seconds of injection.
Management of suspected vascular occlusion: Emergency Stop injection immediately; aspirate if possible; inject hyaluronidase (at least 200–500 units, flood the area) if HA filler was used; apply warm compresses (vasodilatation); prescribe aspirin 325 mg; apply nitroglycerin paste 2%; consider hyperbaric oxygen; urgent ophthalmology referral for visual symptoms. Inject hyaluronidase even if unsure of filler type, as the benefit outweighs the risk.
Thread Lifts & Energy-Based Devices
Thread lifts use absorbable barbed sutures (PDO, PLLA, or PCL) placed subcutaneously to lift sagging tissue and stimulate collagen production. Results are modest and temporary (12–18 months). Complications include asymmetry, dimpling, thread palpability, migration, and infection. Energy-based devices include radiofrequency (RF) microneedling (Morpheus8, Vivace) for skin tightening and textural improvement, high-intensity focused ultrasound (HIFU) (Ultherapy) for non-surgical lifting of the brow and submentum, fractional CO2 laser for scar resurfacing and rejuvenation, and intense pulsed light (IPL) for pigmentation and vascular lesions. Chemical peels (glycolic, TCA, phenol) resurface the skin at varying depths: superficial (epidermis only), medium (to papillary dermis), and deep (to reticular dermis — phenol/croton oil, single application).
Autologous Fat Grafting
Harvested via lipoaspiration, processed (centrifugation, decantation, or washing), and injected in small aliquots (Coleman technique: inject in multiple passes, 0.1 mL per pass, multiple tissue planes). Graft survival: 40–60% at 1 year; higher survival with atraumatic harvest, minimal processing, and small-volume injection. Uses: facial rejuvenation, breast augmentation (fat grafting alone or as supplement to implant), gluteal augmentation (Brazilian butt lift [BBL]). Emergency BBL mortality: Fat embolism to the gluteal veins is the leading cause of death in aesthetic surgery. Mortality rate historically 1:3,000; reduced to < 1:15,000 with subcutaneous-only injection (avoid intramuscular injection, which is now strongly recommended against). PMID: 29370056
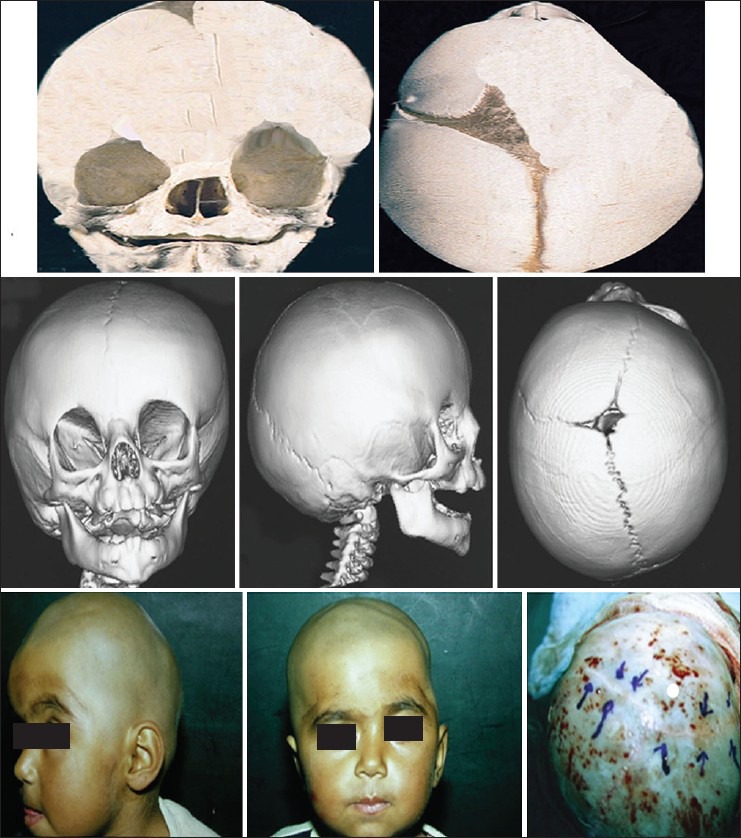
25 Microsurgical Complications
Flap Failure & Salvage
Overall free flap success rate at experienced centres: 95–98%. Flap compromise is most common in the first 24–48 hours. The most common cause of free flap failure is venous thrombosis (~80% of thrombotic events), followed by arterial thrombosis (~20%). Return to the operating room (take-back) rate: 5–10%. Successful salvage rates when thrombosis is detected early: 50–75%.
Venous Congestion vs. Arterial Insufficiency
| Feature | Venous Congestion | Arterial Insufficiency |
|---|---|---|
| Colour | Blue/purple/dusky | Pale/white |
| Turgor | Tense, swollen, engorged | Soft, flaccid, wrinkled |
| Capillary refill | Brisk (rapid — < 1 second) | Sluggish or absent (> 3 seconds) |
| Pin-prick bleeding | Rapid, dark blood | Absent or minimal bleeding |
| Doppler signal | Arterial signal may be present; venous signal diminished | No arterial signal |
| Temperature | Warm initially, then cool | Cool |
| Cause | Venous thrombosis, external compression, pedicle kinking | Arterial thrombosis, vasospasm, kinking |
| Emergent treatment | Release sutures/dressings; leech therapy (Hirudo medicinalis); operative re-exploration | Emergency Immediate operative re-exploration; thrombectomy; reanastomosis ± vein graft |
Risk Factors for Flap Failure
Patient factors: Smoking (relative risk 2–3×), peripheral vascular disease, diabetes, obesity, hypercoagulable states, prior radiation to the recipient site, and vasopressor use. Surgical factors: Vessel size mismatch > 2:1, tension on the pedicle, pedicle kinking or compression, inadequate vessel preparation, intimal damage during dissection, and prolonged ischaemia time (> 4 hours warm ischaemia is associated with increased risk, though muscle flaps are more sensitive than fasciocutaneous flaps). Postoperative factors: Hypotension, hypothermia, external compression from dressings or positioning, and haematoma at the anastomosis site.
Salvage Protocols
Medicinal leeches (Hirudo medicinalis): Applied for venous congestion when operative revision is not possible or as a bridge therapy. The leech removes 5–15 mL of blood during feeding and secretes hirudin (direct thrombin inhibitor) that allows continued oozing for 6–48 hours. Complications: blood loss requiring transfusion, Aeromonas hydrophila infection (prophylactic antibiotics — fluoroquinolone or TMP-SMX — are mandatory during leech therapy). PMID: 16327836
26 Wound Complications
Hematoma
Incidence varies by procedure: rhytidectomy 1–4% (most common complication of facelift; risk higher in males, hypertension), breast surgery 1–3%, abdominoplasty 1–2%. Emergency Expanding hematomas after neck/face surgery can cause airway compromise and require immediate evacuation. Risk factors: uncontrolled hypertension, anticoagulants/antiplatelets, NSAIDs, postoperative straining/vomiting.
Seroma
Collection of serous fluid in a dead space. Common after abdominoplasty (10–15%), latissimus dorsi flap harvest (~30%), and breast procedures. Prevention: drain placement, quilting sutures (progressive tension sutures in abdominoplasty reduce seroma from ~15% to < 5%), and compression garments. Treatment: serial aspiration; sclerotherapy (doxycycline, talc, fibrin glue) for recurrent seromas.
Surgical Site Infection (SSI)
Clean surgical cases (class I) have baseline SSI rates of 1–3%. Clean-contaminated (class II, e.g., intraoral flaps): 5–10%. Factors increasing SSI risk: diabetes, smoking, obesity, immunosuppression, implanted materials. Prophylactic antibiotics: cefazolin 2 g IV within 60 minutes of incision for clean cases involving implants or prolonged procedures (> 3 hours). Gram-negative coverage (cefazolin + metronidazole or ampicillin-sulbactam) for head & neck cases involving intraoral incisions.
Dehiscence
Wound separation risk factors: excessive tension, infection, malnutrition, steroid use, radiation, and smoking. Management depends on the setting: clean dehiscence of a skin closure may be managed with secondary intention or delayed primary closure; fascial dehiscence requires operative repair; flap dehiscence may require revision or additional tissue coverage.
Fat Necrosis
Common after autologous breast reconstruction (DIEP flap: 10–15%), fat grafting, and trauma. Presents as a firm, palpable mass that may mimic malignancy on clinical examination and imaging. Diagnosis: ultrasound (oil cyst), mammography (calcification patterns), or MRI (fat signal with rim enhancement). Management: observation for small asymptomatic areas; core needle biopsy to rule out malignancy when imaging is equivocal; excision for symptomatic or enlarging masses. Prevention: ensure adequate flap perfusion (avoid including poorly perfused tissue in the flap design; use ICG angiography).
Nerve Injury
Nerve injuries in plastic surgery include the Seddon classification (neurapraxia — focal demyelination, full recovery in weeks; axonotmesis — axon damage with intact endoneurium, recovery possible at 1 mm/day; neurotmesis — complete nerve division, requires surgical repair) and the Sunderland classification (5 degrees of increasing severity). Nerve repair principles: tension-free epineural or group fascicular repair within 72 hours ideally; nerve grafts (sural nerve donor, up to 30–40 cm available) or conduits for gaps > 3 cm; processed nerve allografts (Avance) for gaps ≤ 3 cm. Nerve transfers are increasingly used when the distal nerve stump is unavailable or denervation time exceeds 12–18 months.
27 Post-operative Monitoring
Free Flap Monitoring
Free flaps require intensive monitoring in the immediate postoperative period to detect vascular compromise early and allow timely re-exploration. Standard protocol:
| Parameter | Method | Frequency |
|---|---|---|
| Clinical assessment | Colour, turgor, capillary refill, temperature, pin-prick test | Every 1 hour × 48 hours, then every 2 hours × 24 hours, then every 4 hours |
| Handheld Doppler | Audible arterial/venous signals over the pedicle or skin paddle | Every 1–2 hours × 48 hours |
| Implantable Doppler (Cook-Swartz) | Continuous audible venous signal; alarm for loss of signal | Continuous × 5–7 days |
| Tissue oximetry (ViOptix/NIRS) | Near-infrared spectroscopy measuring tissue oxygen saturation | Continuous monitoring with alarm thresholds |
The implantable Doppler (Cook-Swartz probe) is placed around the venous pedicle at the time of surgery and provides continuous monitoring with a high sensitivity for detecting venous thrombosis. Studies show earlier detection and higher salvage rates compared to clinical monitoring alone.
Implant Surveillance
Breast implant monitoring: FDA recommends MRI screening beginning at 5–6 years after silicone implant placement, then every 2–3 years, to detect silent rupture. Ultrasound is an alternative screening modality (lower sensitivity, higher specificity). Patients should be counselled that implants are not lifetime devices — the average implant life is 10–20 years, and revision surgery may be needed.
Replantation & Digital Replantation
Replantation (reattachment of a completely amputated part) requires microsurgical repair of arteries, veins, nerves, tendons, and bone. Indications for replantation (attempt strongly recommended): thumb amputation, multiple digit amputation, hand/wrist amputation, paediatric amputation (any level), and individual digits distal to the FDS insertion (sharp mechanism). Relative contraindications: severe crush/avulsion mechanism, multilevel injury, prolonged warm ischaemia (> 12 hours for digits, > 6 hours for major limb), single digit proximal to the FDS insertion in an adult, significant comorbidities. Cold ischaemia time tolerance: digits (no muscle) tolerate up to 24 hours of cold ischaemia; muscle-containing parts (hand, arm) should be replanted within 6–12 hours. Preservation: wrap the amputated part in saline-moistened gauze, place in a sealed plastic bag, and place the bag on ice (do NOT place the part directly on ice). Replantation success rates at centres of excellence: 80–90% for clean sharp amputations.
Microsurgical Anticoagulation Protocols
No universally standardised protocol exists. Common regimens: Aspirin 325 mg PO daily (started preoperatively or day of surgery) for 2–4 weeks. Heparin 5,000 units IV intraoperatively after anastomosis completion. Dextran 40 (low molecular weight) at 20–25 mL/hr for 48–72 hours postoperatively (decreasing use due to anaphylaxis risk and lack of clear benefit). Enoxaparin 40 mg SC daily for DVT prophylaxis. Avoid hypotension and dehydration, which predispose to pedicle thrombosis.
Tissue Expansion
Tissue expansion exploits the skin’s ability to generate new tissue in response to mechanical stretch (biological creep — new cell division and collagen deposition). Silicone expanders with an integrated or remote port are placed in a subcutaneous or submusculofascial pocket adjacent to the defect. Serial inflation occurs every 1–2 weeks over 6–12 weeks until adequate tissue is generated. The expanded flap provides tissue of matching colour, texture, and sensation. Common applications: breast reconstruction (tissue expander/implant), scalp reconstruction, burn scar revision, and congenital nevi excision. Complication rate: 5–15% (infection, expander exposure, hematoma, pain). Intraoperative tissue expansion (acute expansion) takes advantage of mechanical creep (stress relaxation of existing collagen fibres) to gain 1–2 cm of additional tissue for immediate closure.
Tissue Engineering & Regenerative Medicine
Emerging technologies include: acellular dermal matrices (ADM) for soft-tissue augmentation; bioprinted skin and composite tissues; adipose-derived stem cells (ASCs) for fat grafting enrichment (cell-assisted lipotransfer) and wound healing; platelet-rich plasma (PRP) for wound healing augmentation and hair restoration; and nerve conduits and nerve allografts for bridging peripheral nerve gaps. Vascularised composite allotransplantation (VCA) — face and hand transplantation — represents the frontier of reconstructive surgery, with > 40 face transplants performed worldwide since 2005, though lifelong immunosuppression remains a significant limitation.
28 Classification Systems
Mathes-Nahai Muscle Flap Classification
| Type | Pattern | Examples |
|---|---|---|
| I | One vascular pedicle | Tensor fasciae latae, gastrocnemius, abductor digiti minimi |
| II | Dominant + minor pedicle(s) | Gracilis, soleus, sternocleidomastoid, vastus lateralis, platysma |
| III | Two dominant pedicles | Rectus abdominis, gluteus maximus, serratus anterior, temporalis |
| IV | Segmental pedicles | Sartorius, tibialis anterior, external oblique |
| V | One dominant + secondary segmental | Latissimus dorsi, pectoralis major, internal oblique |
Cormack-Lamberty Fasciocutaneous Flap Classification
| Type | Blood Supply Pattern | Description |
|---|---|---|
| A | Multiple unnamed fasciocutaneous perforators at the base | Random-pattern skin flap; blood enters at base |
| B | Single fasciocutaneous perforator (moderate, consistent) | Can be islanded on the perforator |
| C | Multiple small perforators along a fascial septum | Axial flap based on a septocutaneous vessel (e.g., radial forearm flap) |
| D | Osteomusculocutaneous | Composite flap including bone (e.g., fibula osteocutaneous flap) |
Gustilo-Anderson Open Fracture Classification
| Type | Wound | Soft-Tissue Injury | Bone Injury |
|---|---|---|---|
| I | < 1 cm, clean | Minimal | Simple fracture, minimal comminution |
| II | 1–10 cm | Moderate, no flap required | Moderate comminution |
| IIIA | > 10 cm | Extensive but can cover bone | Usually segmental or severe comminution |
| IIIB | > 10 cm | Periosteal stripping, flap coverage needed | Segmental, severe comminution |
| IIIC | Any size | Vascular injury requiring repair | Variable |
NPUAP Pressure Injury Staging
| Stage | Description |
|---|---|
| I | Non-blanchable erythema of intact skin |
| II | Partial-thickness loss with exposed dermis; blister or shallow ulcer |
| III | Full-thickness loss; subcutaneous fat visible; no deep structure exposure |
| IV | Full-thickness loss with exposed bone, tendon, or muscle |
| Unstageable | Obscured by slough or eschar |
| DTI | Deep tissue injury — intact skin with purple/maroon discolouration |
Baker Capsular Contracture
| Grade | Description |
|---|---|
| I | Normal — looks and feels natural |
| II | Minimal firmness; looks normal, feels slightly firm |
| III | Moderate firmness; feels firm, appears distorted |
| IV | Severe; hard, painful, distorted |
Le Fort Fracture Classification
| Type | Fracture Line | Segment Mobilised |
|---|---|---|
| I | Horizontal maxillary (above teeth, below piriform aperture) | Hard palate and alveolus |
| II | Pyramidal (nasofrontal junction, medial orbit, infraorbital rim, pterygoid plates) | Maxilla and nose as a unit |
| III | Craniofacial dysjunction (nasofrontal, orbital walls, zygomatic arches) | Entire midface from cranial base |
Burn Depth Classification
| Depth | Layers Involved | Healing |
|---|---|---|
| Superficial | Epidermis only | 3–5 days, no scar |
| Superficial partial-thickness | Epidermis + superficial dermis | 7–14 days, minimal scar |
| Deep partial-thickness | Epidermis + deep dermis | 3–6 weeks, significant scar; often needs excision & grafting |
| Full-thickness | Entire dermis destroyed | Will not heal; requires excision & grafting |
| Subdermal (4th degree) | Muscle, tendon, bone | Requires flap or amputation |
Wassel Classification — Thumb Polydactyly
| Type | Level of Duplication | Frequency |
|---|---|---|
| I | Bifid distal phalanx | 2% |
| II | Duplicated distal phalanx | 15% |
| III | Bifid proximal phalanx | 6% |
| IV | Duplicated proximal phalanx | 43% (most common) |
| V | Bifid metacarpal | 10% |
| VI | Duplicated metacarpal | 4% |
| VII | Triphalangism | 20% |
29 Medications Master Table
| Category | Drug | Dose/Route | Key Notes |
|---|---|---|---|
| Antibiotics (Perioperative) | Cefazolin | 2 g IV (3 g if > 120 kg) | First-line prophylaxis for clean cases with implants; re-dose every 3–4 hours intraoperatively |
| Ampicillin-sulbactam | 3 g IV | Head & neck cases with intraoral incisions (covers oral flora) | |
| Clindamycin | 900 mg IV | Alternative for penicillin-allergic patients | |
| Vancomycin | 15 mg/kg IV | MRSA coverage; infuse over 1 hour; for revision implant cases or known MRSA colonisation | |
| Topical Antimicrobials (Burns) | Silver sulfadiazine (Silvadene) | 1% cream topically BID | Broad-spectrum; transient leukopenia; avoid face; contraindicated in sulfa allergy |
| Mafenide acetate (Sulfamylon) | 5% cream or 5% solution | Penetrates eschar; painful; carbonic anhydrase inhibitor → metabolic acidosis; best for ear burns | |
| Silver nitrate | 0.5% solution | Broad-spectrum; stains black; electrolyte abnormalities (hyponatremia, hypochloremia) | |
| Anticoagulants (Microsurgery) | Aspirin | 325 mg PO daily | Platelet inhibitor; start day of surgery; continue 2–4 weeks |
| Heparin (unfractionated) | 5,000 units IV bolus | Given intraoperatively after anastomosis; some centres use continuous infusion post-op | |
| Enoxaparin | 40 mg SC daily | DVT prophylaxis; start 12–24 hours post-op | |
| Scar Management | Triamcinolone acetonide | 10–40 mg/mL intralesional injection | First-line for keloids & hypertrophic scars; inject into scar every 4–6 weeks; risk of skin atrophy, hypopigmentation |
| 5-Fluorouracil (5-FU) | 50 mg/mL intralesional | Adjunct to triamcinolone for keloids; inhibits fibroblast proliferation; often mixed with steroid | |
| Silicone sheeting/gel | Topical, continuous | First-line prevention and treatment of hypertrophic scars; mechanism: hydration and occlusion; use ≥ 12 hours/day for ≥ 3 months | |
| Vasospasm Management | Papaverine | 30 mg/mL topical irrigation | Smooth muscle relaxant; applied topically to vessels during microsurgery for vasospasm |
| Verapamil | 2.5 mg/mL topical or intralesional | Calcium channel blocker; topical vasodilator for microsurgery; also used intralesionally for keloids | |
| Reversal Agents | Hyaluronidase (Hylenex) | 200–500+ units intralesional | Reverses hyaluronic acid fillers; Emergency use for vascular occlusion from HA filler |
| Phentolamine | 5 mg in 10 mL saline, local injection | Alpha-blocker; reversal of vasoconstriction from epinephrine (digital injection) |
30 Imaging & Diagnostics
CT Angiography (CTA)
Preoperative CTA for perforator mapping is the gold standard for planning DIEP and other perforator flaps. Identifies the location, calibre, and intramuscular course of perforators from the deep inferior epigastric artery. Reduces operative time, improves flap design, and reduces the risk of perforator injury. Also used for fibula free flap planning (identifies peroneal artery and confirms three-vessel runoff to the foot) and ALT flap planning.
MRI
Breast MRI: Screening for silicone implant rupture (intracapsular: “linguine sign”; extracapsular: silicone outside the capsule). Extremity MRI: Evaluation of soft-tissue tumours, assessment of flap donor sites, and evaluation of lymphedema (fluid-sensitive sequences show oedema patterns). MRA: Alternative to CTA for vascular mapping when contrast allergy or renal insufficiency precludes CT contrast.
Lymphoscintigraphy & ICG Lymphography
Lymphoscintigraphy: Technetium-99m-labelled colloid injected intradermally; gamma camera imaging of lymphatic drainage. Used for lymphedema staging and preoperative planning. ICG lymphography: Indocyanine green (ICG) injected intradermally; near-infrared fluorescence imaging provides real-time, high-resolution mapping of superficial lymphatic channels. Superior spatial resolution compared to lymphoscintigraphy. Essential for planning lymphovenous anastomosis (identifies functional lymphatic channels and their relationship to subdermal venules). MD Anderson classification of ICG patterns: linear (normal/early disease) → splash → stardust → diffuse (severe disease).
Other Imaging Modalities
SPY fluorescence angiography (ICG): Intraoperative assessment of tissue perfusion using IV ICG (0.1–0.3 mg/kg IV) and near-infrared camera. Used to assess mastectomy flap perfusion before implant placement (areas with absent perfusion should be excised to prevent necrosis, reducing mastectomy flap necrosis from ~30% to ~5%), evaluate free flap perfusion after anastomosis, and assess burn wound depth. Duplex ultrasound: Non-invasive evaluation of vessel patency, perforator localisation, and vascular malformations. Duplex can map perforators for DIEP flap planning with accuracy comparable to CTA in experienced hands. Laser Doppler: Measures microvascular perfusion in flaps and wound beds.
3D Photography & Virtual Surgical Planning
3D photography (Vectra, Canfield) provides volumetric analysis for breast surgery planning, facial analysis for rhinoplasty, and outcome documentation. Virtual surgical planning (VSP) uses CT data to create 3D models and patient-specific cutting guides for bony reconstruction (mandible with fibula flap, midface reconstruction, cranial vault remodelling). VSP improves accuracy of osteotomy placement, reduces operative time, and enables precise pre-bending of reconstruction plates. Custom 3D-printed titanium implants are emerging for cranial, orbital, and midface reconstruction.
31 Abbreviations Master List
| Abbreviation | Full Term |
|---|---|
| ADM | Acellular dermal matrix |
| ALT | Anterolateral thigh (flap) |
| BIA-ALCL | Breast implant-associated anaplastic large cell lymphoma |
| CaHA | Calcium hydroxylapatite |
| CDT | Complete decongestive therapy |
| CL/P | Cleft lip and/or palate |
| CP | Cleft palate |
| CS | Component separation |
| CTA | Computed tomography angiography |
| DIEP | Deep inferior epigastric perforator (flap) |
| DO | Distraction osteogenesis |
| DTI | Direct-to-implant; deep tissue injury (context-dependent) |
| FFS | Facial feminisation surgery |
| FTSG | Full-thickness skin graft |
| HA | Hyaluronic acid |
| ICG | Indocyanine green |
| IGAP | Inferior gluteal artery perforator (flap) |
| ISL | International Society of Lymphology |
| IVVP | Intravelar veloplasty |
| LCFA | Lateral circumflex femoral artery |
| LEAP | Lower Extremity Assessment Project |
| LVA | Lymphovenous anastomosis |
| MESS | Mangled Extremity Severity Score |
| M-N | Mathes-Nahai (classification) |
| NAC | Nipple-areolar complex |
| NIRS | Near-infrared spectroscopy |
| NPUAP | National Pressure Ulcer Advisory Panel |
| NPWT | Negative pressure wound therapy |
| PAP | Profunda artery perforator (flap) |
| PLLA | Poly-L-lactic acid |
| PMMA | Polymethylmethacrylate |
| RSTL | Relaxed skin tension lines |
| SAPL | Suction-assisted protein lipectomy |
| SGAP | Superior gluteal artery perforator (flap) |
| SMAS | Superficial musculoaponeurotic system |
| SSI | Surgical site infection |
| STSG | Split-thickness skin graft |
| TAR | Transversus abdominis release |
| TBSA | Total body surface area |
| TFL | Tensor fasciae latae |
| TRAM | Transverse rectus abdominis myocutaneous (flap) |
| TUG | Transverse upper gracilis (flap) |
| VACTERL | Vertebral, Anal, Cardiac, TE fistula, Renal, Limb anomalies |
| VLNT | Vascularised lymph node transfer |
| VPI | Velopharyngeal insufficiency |
| VRAM | Vertical rectus abdominis myocutaneous (flap) |
| VSP | Virtual surgical planning |
| VTE | Venous thromboembolism |
| WALANT | Wide Awake Local Anaesthesia No Tourniquet |
| WPATH | World Professional Association for Transgender Health |
32 Landmark Studies & Guidelines
| Study/Guideline | Year | Key Finding | Reference |
|---|---|---|---|
| Godina — Early Microsurgical Reconstruction of Complex Trauma | 1986 | Free flap coverage within 72 hours of injury has lowest failure rate (0.75%), lowest infection rate (1.5%), and shortest hospitalisation | PMID: 3514036 |
| LEAP Trial (Lower Extremity Assessment Project) | 2002 | No significant difference in functional outcomes (SIP scores) at 2 years between reconstruction and amputation for severe lower extremity trauma (Gustilo IIIB/IIIC) | PMID: 12473698 |
| Taylor & Palmer — Angiosome Concept | 1987 | Defined 40 angiosomes of the body; established the vascular basis for flap design and the concept of choke vessels between adjacent angiosomes | PMID: 3625564 |
| Mathes & Nahai — Muscle Flap Classification | 1981 | Classified muscles into five types based on their vascular anatomy, providing the foundation for reliable muscle flap surgery | PMID: 7301062 |
| Ramirez — Component Separation | 1990 | Described the anterior component separation technique for complex abdominal wall reconstruction, achieving up to 10 cm of midline advancement per side | PMID: 2138306 |
| Hidalgo — Fibula Free Flap for Mandible | 1989 | Described the fibula free flap as the ideal vascularised bone graft for mandibular reconstruction due to its length, shape, and reliable blood supply | PMID: 2681572 |
| Allen & Treece — DIEP Flap | 1994 | Described the deep inferior epigastric perforator flap for breast reconstruction, preserving the rectus abdominis muscle entirely | PMID: 8171237 |
| Millard — Rotation-Advancement Cleft Lip Repair | 1957 | Described the rotation-advancement technique for unilateral cleft lip repair, which became the most widely used method worldwide | PMID: 13484405 |
| Gustilo & Anderson — Open Fracture Classification | 1976 | Classified open fractures into Types I–III based on wound size, soft-tissue injury, and contamination; later refined with IIIA/B/C subtypes (1984) | PMID: 1085588 |
| ASPS Evidence-Based Guidelines — Breast Reconstruction | 2022 | Comprehensive evidence-based clinical practice guidelines for breast reconstruction following mastectomy covering implant, autologous, and combined approaches | PMID: 35675701 |
| Furlow — Double-Opposing Z-Plasty Palatoplasty | 1986 | Described double-opposing Z-plasty technique for cleft palate repair that re-orients the levator veli palatini muscles, improving velopharyngeal closure and lengthening the palate | PMID: 3514056 |
| Ilizarov — Distraction Osteogenesis | 1989 | Described the tension-stress effect: gradual mechanical distraction of living tissue generates new bone, cartilage, and soft tissue — the foundation for mandibular and cranial distraction osteogenesis | PMID: 2912611 |
| Multi-Society Aesthetic Surgery Task Force — BBL Safety | 2017 | Established that gluteal fat grafting mortality is primarily due to fat embolism from intramuscular injection; recommended subcutaneous-only injection technique | PMID: 29370056 |
| Novak et al. — BIA-ALCL | 2017 | Systematic review establishing the association between textured breast implants and anaplastic large cell lymphoma; recommended capsulectomy as curative treatment | PMID: 28296502 |