Surgical Oncology
Every diagnosis, classification, procedure, technique, medication, complication, and management algorithm across the full scope of surgical oncology in one place.
01 Cancer Biology & Tumor Kinetics
Surgical oncology is built on an understanding of tumor biology. The decision to operate, the extent of resection, the role of neoadjuvant or adjuvant therapy, and the approach to surveillance all derive from fundamental principles of how cancers grow, invade, and metastasize.
Hallmarks of Cancer
Hanahan and Weinberg described the original six hallmarks (PMID: 10647931), later expanded to ten: sustaining proliferative signaling (e.g., EGFR, HER2 overexpression), evading growth suppressors (loss of Rb, p53), resisting cell death (Bcl-2 overexpression), enabling replicative immortality (telomerase activation), inducing angiogenesis (VEGF pathway), activating invasion & metastasis (E-cadherin loss, EMT), deregulating cellular energetics (Warburg effect — aerobic glycolysis), avoiding immune destruction (PD-L1 upregulation), genome instability & mutation (DNA repair defects — MSI, BRCA), and tumor-promoting inflammation.
Tumor Growth Kinetics
Tumor growth follows a Gompertzian curve — exponential early growth that progressively decelerates as the tumor enlarges. At small sizes the growth fraction (proportion of cells actively dividing) is highest, making the tumor most susceptible to cytotoxic chemotherapy. The doubling time varies enormously: aggressive lymphomas may double in days, while indolent thyroid cancers double over years. A 1-cm tumor contains approximately 109 (1 billion) cells; a clinically detectable tumor typically reaches 109–1010 cells. Death generally occurs at ~1012 cells (~1 kg tumor burden).
The Metastatic Cascade
Metastasis is an inefficient, multistep process: local invasion (loss of cell adhesion, basement membrane degradation by MMPs) → intravasation (tumor cells enter blood or lymphatic vessels) → survival in circulation (most circulating tumor cells die from shear stress or immune attack; platelet coating provides protection) → extravasation (arrest at distant capillary bed, migration through endothelium) → colonization (adaptation to the new microenvironment, angiogenesis, formation of a macrometastasis). The seed-and-soil hypothesis (Paget, 1889) explains non-random metastatic patterns: colorectal cancer preferentially metastasizes to the liver (portal venous drainage), sarcomas to the lungs (systemic venous drainage), and breast cancer to bone, liver, lung, and brain.
Tumor Angiogenesis
Tumors cannot grow beyond ~2 mm without developing their own blood supply. The angiogenic switch is triggered by hypoxia-induced upregulation of VEGF (vascular endothelial growth factor), which stimulates endothelial cell proliferation and new vessel formation. Anti-angiogenic therapy (bevacizumab, a monoclonal antibody against VEGF) exploits this dependence. Tumor neovasculature is disorganized and leaky, contributing to elevated interstitial pressure that paradoxically impairs drug delivery — "vessel normalization" by anti-VEGF agents may transiently improve chemotherapy delivery.
Tumor Immunology
The cancer-immunity cycle involves: tumor antigen release → antigen presentation by dendritic cells → T-cell priming → T-cell trafficking to the tumor → tumor infiltration → recognition of cancer cells → killing of cancer cells. Tumors evade this cycle through multiple mechanisms: downregulation of MHC class I, secretion of immunosuppressive cytokines (TGF-beta, IL-10), recruitment of regulatory T cells and myeloid-derived suppressor cells, and — crucially — upregulation of immune checkpoint ligands (PD-L1) that engage inhibitory receptors (PD-1) on T cells, inducing T-cell exhaustion. Checkpoint inhibitors (anti-PD-1: pembrolizumab, nivolumab; anti-CTLA-4: ipilimumab) block these inhibitory signals and unleash anti-tumor immunity.
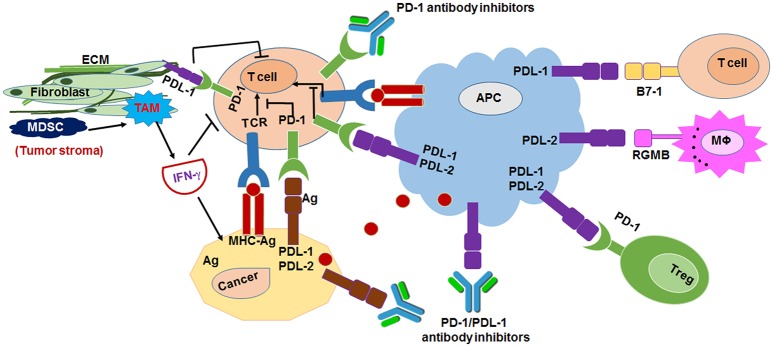
Tumor Microenvironment
The tumor microenvironment (TME) comprises tumor cells, immune cells (tumor-infiltrating lymphocytes [TILs], macrophages, myeloid-derived suppressor cells), fibroblasts (cancer-associated fibroblasts [CAFs]), extracellular matrix, and vasculature. The TME profoundly influences tumor behavior and therapeutic response. "Hot" tumors (high TIL infiltration, high mutational burden) respond better to immunotherapy. "Cold" tumors (low TIL, immunosuppressive TME) are resistant to checkpoint inhibitors. Strategies to convert cold tumors to hot include: combination immunotherapy (anti-PD-1 + anti-CTLA-4), radiation (releases tumor antigens, abscopal effect), oncolytic viruses, and intratumoral injections. The tumor mutational burden (TMB) — the number of somatic mutations per megabase of DNA — correlates with neoantigen load and immunotherapy responsiveness. TMB-high (≥10 mutations/Mb) is an FDA-approved agnostic biomarker for pembrolizumab.
Cancer Genetics — Oncogenes & Tumor Suppressors
| Gene | Type | Function | Associated Cancers |
|---|---|---|---|
| KRAS | Oncogene | GTPase signal transduction | Colorectal (40%), pancreatic (90%), lung |
| HER2 (ERBB2) | Oncogene | Receptor tyrosine kinase | Breast (20%), gastric |
| BRAF | Oncogene | Serine/threonine kinase (MAPK pathway) | Melanoma (50%), CRC (8%), thyroid (PTC) |
| KIT | Oncogene | Receptor tyrosine kinase | GIST (80%) |
| MYC | Oncogene | Transcription factor (cell proliferation) | Burkitt lymphoma, breast, liver |
| TP53 | Tumor suppressor | "Guardian of the genome" — cell cycle arrest, apoptosis | Most common mutation in human cancer (~50%); Li-Fraumeni syndrome |
| RB1 | Tumor suppressor | Cell cycle regulation (G1/S checkpoint) | Retinoblastoma, osteosarcoma, small cell lung |
| APC | Tumor suppressor | Wnt signaling pathway regulation | Colorectal (FAP, sporadic CRC) |
| BRCA1/2 | Tumor suppressor | DNA double-strand break repair (homologous recombination) | Breast, ovarian, pancreatic, prostate |
| VHL | Tumor suppressor | HIF degradation (oxygen sensing) | Renal cell carcinoma (clear cell), pheochromocytoma |
| Biomarker | Tumor Type | Clinical Significance |
|---|---|---|
| ER / PR | Breast | Positive → hormonal therapy (tamoxifen, AIs); better prognosis |
| HER2 | Breast, gastric | Overexpression → trastuzumab; more aggressive biology |
| KRAS / NRAS / BRAF | Colorectal | RAS mutation → no anti-EGFR therapy; BRAF V600E = poor prognosis |
| MSI / dMMR | Colorectal, endometrial | MSI-high → excellent response to checkpoint inhibitors; screen for Lynch syndrome |
| KIT / PDGFRA | GIST | KIT exon 11 → best imatinib response; PDGFRA D842V → imatinib-resistant, avapritinib |
| BRCA1 / BRCA2 | Breast, ovarian, pancreatic | Hereditary cancer risk; PARP inhibitor sensitivity; bilateral mastectomy consideration |
| PD-L1 (CPS / TPS) | Multiple | Predicts checkpoint inhibitor response in melanoma, NSCLC, gastric, others |
| RET, BRAF | Thyroid (MTC, PTC) | RET mutation → MEN2 screening; BRAF V600E in PTC → more aggressive behavior |
02 Surgical Oncology Principles
Resection Classification
The completeness of surgical resection is the single most important prognostic factor for most solid tumors.
| Classification | Definition | Clinical Implications |
|---|---|---|
| R0 | Microscopically negative margins — no tumor at the inked resection margin | Goal of curative surgery; associated with best overall survival in virtually all solid tumors |
| R1 | Microscopically positive margins — tumor cells at the inked margin | Associated with higher local recurrence; may require re-excision, adjuvant RT, or systemic therapy |
| R2 | Macroscopically positive — gross residual tumor left behind | Palliative intent; considered only for symptom control or when R0/R1 is technically impossible |
Margin Assessment Principles
Adequate surgical margins vary by tumor type: melanoma requires 1–2 cm margins based on Breslow depth; breast cancer requires "no ink on tumor" per SSO-ASTRO consensus; soft tissue sarcoma requires ≥1 cm or an intact fascial plane; pancreatic cancer margins are assessed at the SMA margin (retroperitoneal), where R1 rates are highest. Intraoperative frozen section is used for margin assessment — particularly in breast (lumpectomy cavity margins), pancreas (bile duct and pancreatic neck margins), and head/neck tumors. Touch-prep cytology is faster but less accurate than frozen section.
Sentinel Lymph Node Concept
The sentinel lymph node (SLN) is the first lymph node to receive lymphatic drainage from the primary tumor site. The concept, pioneered by Morton for melanoma and Krag/Giuliano for breast cancer, is based on the orderly progression of lymphatic metastases. If the SLN is negative, the remaining regional nodes are overwhelmingly likely to be negative — sparing the patient a complete lymph node dissection and its associated morbidity (especially lymphedema). Technique involves injection of blue dye (isosulfan blue / methylene blue), radiotracer (technetium-99m sulfur colloid), or indocyanine green (ICG) at the tumor site, followed by identification of the draining "hot" and/or "blue" node.
Multidisciplinary Tumor Board
The multidisciplinary tumor board (MDT) is the cornerstone of modern oncologic care. Studies consistently demonstrate improved staging accuracy, treatment plan adherence to guidelines, and overall survival when cases are discussed in a multidisciplinary setting. The MDT typically includes: surgical oncologist, medical oncologist, radiation oncologist, diagnostic radiologist, pathologist, and specialized support (genetics counselor, palliative care, nutrition, social work). All new cancer diagnoses and complex recurrent/metastatic cases should be presented.
Enhanced Recovery After Surgery (ERAS) in Oncologic Surgery
ERAS protocols have been adopted across oncologic surgery — particularly colorectal, hepatobiliary, and pancreatic resections. Key elements: preoperative carbohydrate loading, avoidance of prolonged fasting, multimodal analgesia (minimizing opioids), early mobilization, early enteral feeding, goal-directed fluid therapy, and avoidance of unnecessary drains/tubes. ERAS reduces length of stay by 2–3 days and reduces complication rates by 30–50% without increasing readmission rates (PMID: 20395846).
Surgical Oncology Volume-Outcome Relationship
High-volume centers performing complex oncologic procedures (pancreatectomy, esophagectomy, hepatectomy, CRS/HIPEC) have significantly lower perioperative mortality and better long-term outcomes. The "Leapfrog effect" demonstrates that centralizing high-risk cancer surgery at experienced centers saves lives. Minimum volume thresholds (Finks et al., PMID: 21631325): pancreatectomy ≥11/year, esophagectomy ≥13/year. Multidisciplinary care, dedicated ICU support, interventional radiology for complication management, and established clinical pathways all contribute to the volume-outcome relationship.
Hereditary Cancer Syndromes — Surgical Implications
| Syndrome | Gene | Associated Cancers | Surgical Considerations |
|---|---|---|---|
| BRCA1/2 | BRCA1, BRCA2 | Breast, ovarian, pancreatic, prostate | Bilateral mastectomy option; risk-reducing BSO; PARP inhibitor sensitivity |
| Lynch syndrome (HNPCC) | MLH1, MSH2, MSH6, PMS2, EPCAM | CRC, endometrial, ovarian, gastric, urinary | Extended colectomy (subtotal) over segmental for CRC; prophylactic hysterectomy/BSO; annual surveillance |
| FAP | APC | CRC (100% risk), desmoid tumors, thyroid, duodenal | Prophylactic total proctocolectomy + IPAA by age 25; duodenal surveillance; desmoid management |
| Li-Fraumeni | TP53 | STS, osteosarcoma, breast, brain, adrenocortical | Avoid radiation when possible (radiation-induced second malignancies); consider mastectomy over BCS |
| VHL | VHL | RCC (clear cell), pheochromocytoma, CNS hemangioblastomas, pancreatic NETs | Nephron-sparing surgery when possible; screen for pheo before any surgery; multifocal/bilateral tumors common |
| MEN1/2 | MEN1, RET | See Section 21 | Prophylactic thyroidectomy (MEN2); parathyroid surgery (MEN1); screen for pheo (MEN2) |
| Peutz-Jeghers | STK11 | GI (stomach, small bowel, colon), breast, pancreatic, ovarian | Small bowel surveillance; polypectomy; increased breast/pancreatic screening |
03 Oncologic Staging & Molecular Markers
TNM Staging System
The AJCC/UICC TNM system (currently 8th edition, 2017) is the universal framework for cancer staging. T = extent of the primary tumor (size, depth of invasion, or local extension), N = regional lymph node involvement (number and/or location), M = distant metastasis (M0 or M1). These three components are combined into a stage group (I–IV) that predicts prognosis and guides treatment. Clinical staging (cTNM) is based on physical examination and imaging before any treatment. Pathologic staging (pTNM) incorporates surgical and pathologic findings. Post-neoadjuvant staging uses the prefix "yp" (ypTNM).
Staging Modalities
| Modality | Best For | Limitations |
|---|---|---|
| CT chest/abdomen/pelvis | Most solid tumors — lung, colorectal, pancreatic, renal; staging of distant metastases | Limited soft tissue contrast compared to MRI; radiation exposure |
| MRI | Rectal cancer (T-staging), liver metastases (hepatocyte-specific agents), brain metastases, soft tissue sarcoma | Cost, availability, claustrophobia, contraindicated with certain implants |
| PET-CT (FDG) | Melanoma, esophageal, lung, lymphoma — detecting distant metastases; evaluating treatment response | False positives (inflammation, infection); low sensitivity for mucinous tumors, HCC, low-grade tumors |
| EUS (endoscopic ultrasound) | T-staging of esophageal, gastric, rectal, and pancreatic tumors; FNA of suspicious lymph nodes | Operator-dependent; cannot cross strictures |
| Diagnostic laparoscopy | Gastric cancer, pancreatic cancer, peritoneal disease — detecting occult carcinomatosis missed on imaging | Invasive; adds operative time |
Role of Molecular and Genomic Markers
Beyond anatomic staging, molecular profiling increasingly guides surgical decision-making. Oncotype DX (21-gene recurrence score) in ER-positive, node-negative breast cancer can spare patients from adjuvant chemotherapy if the recurrence score is low (TAILORx trial, PMID: 29860917). MSI testing / mismatch repair (MMR) status in colorectal cancer guides both chemotherapy selection (MSI-high tumors do not benefit from 5-FU alone but respond dramatically to checkpoint inhibitors) and Lynch syndrome screening. ctDNA (circulating tumor DNA) is emerging as a tool for post-operative surveillance — detectable ctDNA after curative resection predicts recurrence and may guide adjuvant therapy decisions (DYNAMIC trial).
- Tissue diagnosis: Core needle biopsy preferred (preserves architecture); avoid excisional biopsy of masses suspected to be sarcoma (disrupts tissue planes)
- Complete staging: CT CAP, PET-CT, MRI, or EUS as appropriate; diagnostic laparoscopy for gastric/pancreatic cancers
- Molecular profiling: ER/PR/HER2 (breast), RAS/BRAF/MSI (colorectal), KIT/PDGFRA (GIST), BRAF/RET (thyroid)
- Functional assessment: Performance status (ECOG/Karnofsky), cardiopulmonary reserve (PFTs for lung resection, cardiac stress testing)
- Nutrition: Albumin <3.0 g/dL or >10% weight loss → consider preoperative nutritional optimization (prehabilitation)
- Tumor board review: Multidisciplinary discussion before definitive surgery
- Genetic counseling: If hereditary syndrome suspected (young age, family history, bilateral disease, multiple primaries)
04 Breast Anatomy & Pathology
Breast Anatomy
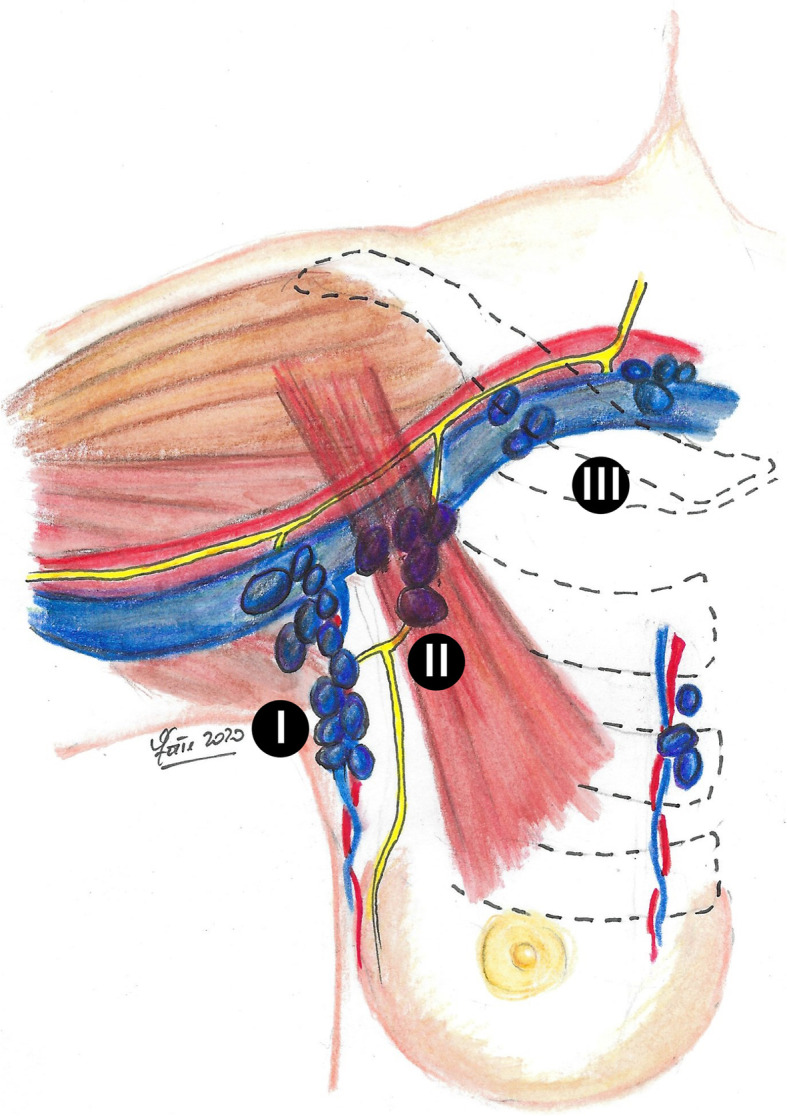
The breast extends from the 2nd to the 6th rib vertically and from the sternal edge to the midaxillary line horizontally. It lies on the pectoralis major fascia with a retromammary bursa (potential space) separating breast tissue from the pectoral muscles. The breast contains 15–20 lobes, each drained by a lactiferous duct converging at the nipple. The axillary tail of Spence extends toward the axilla and is a common location for palpable lumps mistaken for lymph nodes.
Blood supply: internal mammary artery (medial perforators supply ~60% of the breast, especially the medial and central portions), lateral thoracic artery, thoracoacromial artery, and intercostal perforators. Lymphatic drainage: ~75% drains to the axillary lymph nodes (Level I = lateral to pectoralis minor; Level II = behind pectoralis minor; Level III = medial to pectoralis minor / infraclavicular). Internal mammary nodes receive drainage from the medial breast (~25%). The Rotter nodes (interpectoral) lie between the pectoralis major and minor.
Screening & Diagnosis
Screening mammography reduces breast cancer mortality by approximately 20–30% in women aged 50–74 (USPSTF). The BI-RADS (Breast Imaging Reporting and Data System) classification standardizes mammographic reporting:
| BI-RADS | Assessment | Cancer Risk | Action |
|---|---|---|---|
| 0 | Incomplete — needs additional imaging | — | Diagnostic mammogram, US, or MRI |
| 1 | Negative | ~0% | Routine screening |
| 2 | Benign finding | ~0% | Routine screening |
| 3 | Probably benign | <2% | Short-interval follow-up (6 months) |
| 4 | Suspicious (4A: 2–10%, 4B: 10–50%, 4C: 50–95%) | 2–95% | Tissue sampling (core needle biopsy) |
| 5 | Highly suggestive of malignancy | >95% | Tissue sampling |
| 6 | Known biopsy-proven malignancy | 100% | Awaiting definitive treatment |
Core needle biopsy (CNB) is the standard diagnostic approach — 14-gauge spring-loaded needle under ultrasound or stereotactic guidance. CNB provides tissue architecture, receptor status (ER/PR/HER2), and grade. Fine-needle aspiration (FNA) provides cytology only and is generally reserved for cyst aspiration or axillary node sampling. Excisional biopsy should be avoided as the initial diagnostic step because it may compromise subsequent oncoplastic surgery and complicates margin assessment.
Molecular Subtypes
| Subtype | Receptor Profile | Frequency | Prognosis | Systemic Therapy |
|---|---|---|---|---|
| Luminal A | ER+/PR+, HER2−, low Ki-67 | ~40% | Best prognosis | Hormonal therapy; often no chemo (low Oncotype DX) |
| Luminal B | ER+/PR±, HER2±, high Ki-67 | ~20% | Intermediate | Hormonal therapy + chemotherapy; ± trastuzumab if HER2+ |
| HER2-enriched | ER−/PR−, HER2+ | ~15% | Aggressive; improved with targeted therapy | Trastuzumab + pertuzumab + chemotherapy |
| Triple-negative (basal-like) | ER−/PR−, HER2− | ~15% | Worst prognosis; high recurrence rate | Chemotherapy ± immunotherapy (pembrolizumab if PD-L1+) |
DCIS (Ductal Carcinoma In Situ)
DCIS is a non-invasive precursor — malignant cells confined within the ductal basement membrane. It accounts for ~20–25% of screen-detected breast cancers. Management: lumpectomy + radiation (standard; NSABP B-17 showed radiation reduces ipsilateral recurrence by ~50%) or mastectomy (for extensive/multicentric DCIS or patient preference). SLNB is recommended if mastectomy is planned (no opportunity for later SLNB), or for large/high-grade DCIS with planned lumpectomy (upgrade to invasive cancer found in ~20% on final pathology). The Van Nuys Prognostic Index (incorporates size, margin width, grade, age) helps stratify DCIS recurrence risk and guide radiation decisions.
05 Breast-Conserving Surgery & Mastectomy
Breast-Conserving Surgery (BCS / Lumpectomy)
The NSABP B-06 trial (Fisher, 1985; 20-year follow-up PMID: 12065600) established that lumpectomy + whole-breast radiation provides equivalent overall survival to mastectomy for stage I–II breast cancer. BCS is now the preferred approach for the majority of early-stage breast cancers.
Contraindications to BCS: multicentric disease (cancer in different quadrants), diffuse malignant-appearing microcalcifications, prior radiation to the chest wall, positive margins after reasonable re-excision attempts, large tumor-to-breast ratio with poor cosmetic outcome (consider neoadjuvant chemotherapy to downstage), inflammatory breast cancer, pregnancy (first/second trimester — radiation cannot be delivered), and connective tissue disorders with radiation intolerance (scleroderma, active SLE).
Margin adequacy (invasive cancer): The SSO-ASTRO consensus (2014) defines an adequate margin as "no ink on tumor" — wider margins do not further reduce local recurrence when whole-breast radiation is administered (PMID: 24501721). For DCIS, the SSO-ASTRO-ASCO guideline recommends a 2-mm margin.
Oncoplastic Techniques
Oncoplastic surgery combines oncologic resection with plastic surgery techniques to improve cosmetic outcomes while maintaining or improving margin clearance. Level I oncoplastic techniques involve undermining, advancement, and rotation flaps for small to moderate defects (<20% breast volume). Level II techniques involve therapeutic mammoplasty (reduction pattern lumpectomy — using a reduction mammoplasty design to excise larger tumors with good cosmesis) and volume displacement/replacement techniques. The contralateral breast may require symmetrization.
Mastectomy Types
| Type | What Is Removed | Indications |
|---|---|---|
| Simple (total) mastectomy | All breast tissue including nipple-areola complex (NAC) and skin envelope | Prophylactic mastectomy; DCIS not amenable to BCS; palliation |
| Skin-sparing mastectomy (SSM) | All breast tissue and NAC, preserving native skin envelope | Most common for immediate reconstruction; lower local recurrence than previously feared |
| Nipple-sparing mastectomy (NSM) | All breast tissue, preserving NAC and skin | Selected patients: tumor >2 cm from NAC, no nipple involvement on imaging, no Paget's disease; excellent cosmesis |
| Modified radical mastectomy (MRM) | Breast tissue + Level I/II axillary lymph nodes | Clinically node-positive disease; rarely performed since SLNB era |
| Radical mastectomy (Halsted) | Breast + pectoralis major/minor + Level I–III nodes | Historical — virtually never performed; only if pectoral muscle is directly invaded |
Reconstruction Considerations
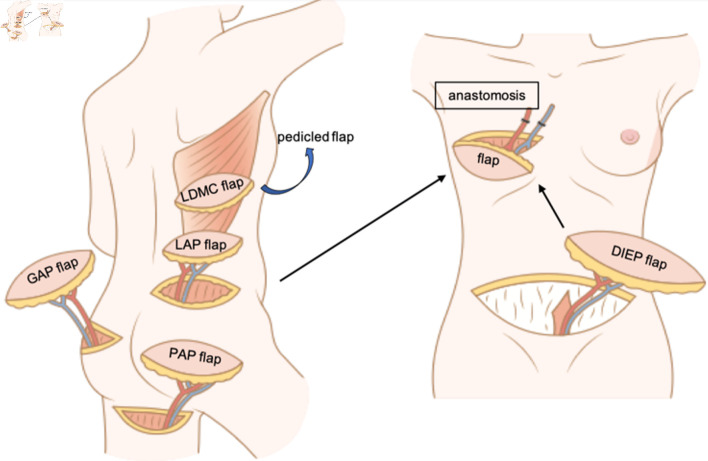
Immediate reconstruction (at the time of mastectomy) is preferred — avoids a second operation, better cosmesis, and psychosocial benefit. Delayed reconstruction is used when adjuvant radiation is anticipated (radiation damages implants and flaps) or for patient preference. Options: implant-based (tissue expander → exchange to permanent implant; or direct-to-implant with ADM/mesh), autologous tissue (DIEP flap — deep inferior epigastric perforator, gold standard for autologous reconstruction; TRAM flap; latissimus dorsi flap), or combined. Pre-pectoral implant placement (on top of the pectoralis major, covered by ADM) is increasingly used and avoids animation deformity.
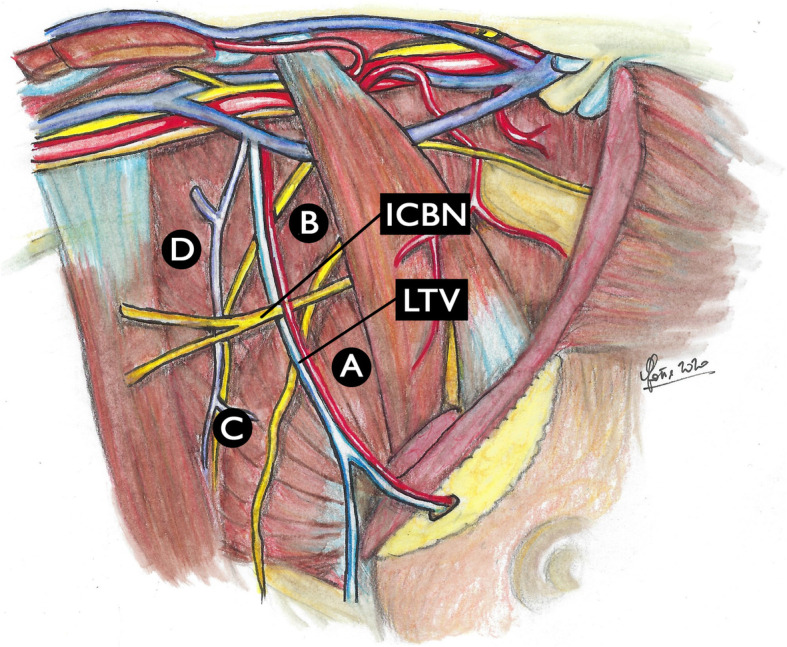
06 Axillary Management & SLNB
Sentinel Lymph Node Biopsy for Breast Cancer
SLNB has replaced routine axillary lymph node dissection (ALND) for clinically node-negative breast cancer. The dual-tracer technique (radiotracer + blue dye) achieves identification rates >97% and false-negative rates <5%. At least 2–3 SLNs should be removed. Intraoperative assessment options: touch-prep cytology, frozen section, or OSNA (one-step nucleic acid amplification).
ACOSOG Z0011 Trial
The landmark ACOSOG Z0011 trial (PMID: 21304082) demonstrated that patients with T1–T2 invasive breast cancer, 1–2 positive sentinel lymph nodes, undergoing BCS with whole-breast radiation do NOT require completion ALND. At 10-year follow-up, there was no difference in overall survival, disease-free survival, or regional recurrence between SLNB alone vs ALND. This trial fundamentally changed axillary management and spares thousands of patients annually from the morbidity of ALND (lymphedema, seroma, shoulder dysfunction).
- T1 or T2 invasive breast cancer
- 1–2 positive sentinel lymph nodes (micro- or macrometastases)
- Undergoing breast-conserving surgery (not mastectomy in the original trial)
- Whole-breast radiation planned
- No matted/fixed axillary nodes, no extranodal extension grossly
- No prior neoadjuvant chemotherapy (in original trial)
AMAROS Trial
The AMAROS trial (PMID: 25304656) showed that axillary radiation provides equivalent regional control to ALND for patients with positive SLN, with significantly less lymphedema (11% vs 23% at 5 years). This offers an alternative to ALND for patients who are Z0011-ineligible (e.g., those undergoing mastectomy with 1–2 positive SLN).
Axillary Management After Neoadjuvant Chemotherapy
For patients who were clinically node-positive at diagnosis and convert to clinically node-negative after neoadjuvant chemotherapy, targeted axillary dissection (TAD) is an emerging approach: the positive node is clipped at diagnosis, and at surgery, both the clipped node and SLN(s) are removed. If negative, ALND may be omitted. The ACOSOG Z1071, SENTINA, and SN FNAC trials showed that removing ≥3 SLNs or using dual tracers reduces the false-negative rate to an acceptable level (~7–10%) in the post-neoadjuvant setting.
07 Neoadjuvant Therapy & Special Situations
Neoadjuvant Chemotherapy (NAC) in Breast Cancer
NAC is given before surgery to: downstage locally advanced or large tumors to allow BCS instead of mastectomy, assess in vivo chemosensitivity (pathologic complete response [pCR] is a prognostic biomarker), and initiate systemic therapy early for aggressive subtypes. NAC is standard for: locally advanced breast cancer (stage IIIA–IIIC), inflammatory breast cancer (always NAC first), HER2-positive tumors (high pCR rates with trastuzumab + pertuzumab + taxane), and triple-negative breast cancer (KEYNOTE-522: addition of pembrolizumab to neoadjuvant chemotherapy improves pCR and event-free survival).
Pathologic complete response (pCR): Defined as no residual invasive cancer in the breast and axillary nodes (ypT0/is ypN0). pCR rates vary by subtype: HER2-positive ~60–80%, triple-negative ~40–60%, luminal A <10%. The CREATE-X trial demonstrated that patients with residual invasive disease after NAC who have HER2-negative tumors benefit from adjuvant capecitabine. The KATHERINE trial showed that HER2-positive patients with residual disease after NAC benefit from switching to T-DM1 (ado-trastuzumab emtansine).
Inflammatory Breast Cancer
Inflammatory breast cancer (IBC) presents with erythema, edema (peau d'orange), warmth, and rapid onset — caused by dermal lymphatic invasion. It is a clinical diagnosis (T4d). Always stage III at minimum. Treatment: trimodality therapy — neoadjuvant chemotherapy first → modified radical mastectomy (BCS is contraindicated) → post-mastectomy radiation. SLNB alone is not adequate; ALND is performed.
Hereditary Breast Cancer (BRCA)
BRCA1/BRCA2 carriers have a lifetime breast cancer risk of 60–80%. Surgical options include enhanced surveillance (MRI + mammography alternating every 6 months) or risk-reducing bilateral mastectomy (reduces risk by >95%). For carriers diagnosed with breast cancer: ipsilateral mastectomy ± contralateral prophylactic mastectomy (CPM) is discussed, though BCS + radiation remains oncologically acceptable. Risk-reducing bilateral salpingo-oophorectomy is recommended by age 35–40 for BRCA1 and 40–45 for BRCA2 carriers.
Post-Mastectomy Radiation (PMRT)
Indications for PMRT: positive margins after mastectomy, T3/T4 tumors, ≥4 positive axillary lymph nodes. PMRT is increasingly considered for 1–3 positive nodes based on the MA.20 and SUPREMO trials. PMRT reduces locoregional recurrence and improves OS in high-risk patients. When PMRT is anticipated, reconstruction planning is affected: tissue expander placement allows radiation during the expansion phase → delayed exchange to permanent implant. Autologous reconstruction is generally delayed until radiation is complete (radiation damages flaps — fat necrosis, fibrosis, contracture). The timing of radiation relative to reconstruction remains an area of active investigation (NCCN recommends discussing with the entire multidisciplinary team).
- Stage I–II, single tumor, adequate breast size: BCS (lumpectomy) + SLNB + whole-breast radiation (standard of care)
- Multicentric disease or large tumor/breast ratio: Mastectomy ± reconstruction; consider neoadjuvant chemo to downstage
- Clinically node-negative: SLNB (dual-tracer technique)
- 1–2 positive SLN + BCS + RT: Observation (Z0011 criteria) — no ALND needed
- Clinically node-positive before NAC, converts to cN0: Targeted axillary dissection (TAD)
- Inflammatory breast cancer: NAC first → MRM + ALND → PMRT (BCS contraindicated)
- BRCA carrier: BCS acceptable; bilateral mastectomy discussed for risk reduction
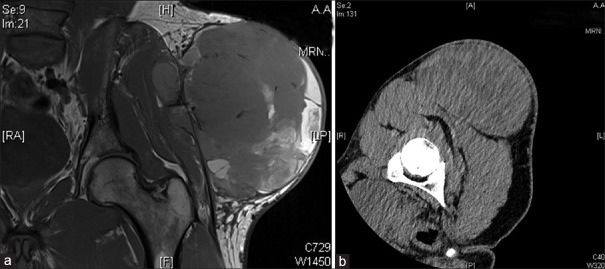
08 Esophageal Cancer
Epidemiology & Pathology
Two main histologic types: squamous cell carcinoma (SCC) — associated with smoking and alcohol, most common in the upper/middle esophagus and worldwide; adenocarcinoma — associated with Barrett's esophagus and GERD, most common in the distal esophagus/GEJ and rising in incidence in Western countries. Staging requires EUS (T/N staging) + PET-CT (distant staging).
Neoadjuvant Therapy — CROSS Trial
The CROSS trial (PMID: 22646630) established neoadjuvant chemoradiation (carboplatin + paclitaxel + 41.4 Gy radiation) followed by surgery as the standard of care for resectable esophageal cancer (T1N1 or T2–T3, any N). The trial demonstrated a median OS of 49 months with neoadjuvant CRT vs 24 months with surgery alone. pCR was achieved in 29% of patients (49% for SCC, 23% for adenocarcinoma). The CheckMate 577 trial subsequently showed that adjuvant nivolumab after neoadjuvant CRT + surgery improves DFS in patients without pCR.
Surgical Approaches
| Approach | Description | Indications |
|---|---|---|
| Ivor Lewis esophagectomy | Laparotomy (gastric mobilization) + right thoracotomy (esophageal resection, intrathoracic anastomosis) | Mid-to-distal esophageal and GEJ tumors; most common approach |
| McKeown (3-field) esophagectomy | Right thoracotomy + laparotomy + left cervical anastomosis | Upper/middle esophageal tumors; cervical leak is less catastrophic than intrathoracic leak |
| Transhiatal esophagectomy | Laparotomy + cervical incision — blunt mediastinal dissection without thoracotomy | Avoids thoracotomy; used for distal esophageal/GEJ tumors; less complete lymphadenectomy |
| MIE (minimally invasive) | Thoracoscopic + laparoscopic or robotic | Increasingly standard; TIME trial showed reduced pulmonary complications and shorter LOS |
Conduit Options for Esophageal Reconstruction
The gastric conduit (stomach tube) is the preferred conduit for esophageal replacement — excellent blood supply, adequate length to reach the neck, and single anastomosis required. The stomach is tubularized along the greater curvature using a linear stapler, preserving the right gastroepiploic arcade as the primary blood supply. Alternative conduits (when stomach unavailable): colon interposition (using the left, transverse, or right colon — technically demanding, higher complication rate) or jejunal interposition (short segment, limited reach — primarily for short-segment defects or revisional surgery). Route of conduit passage: posterior mediastinal (orthotopic — shortest route, preferred), substernal (used when posterior mediastinum is hostile — prior radiation, recurrent tumor), or subcutaneous (rare, for palliation).
Barrett's Esophagus & Dysplasia Management
Barrett's esophagus (intestinal metaplasia of the distal esophagus) is the primary risk factor for esophageal adenocarcinoma. Progression: no dysplasia → low-grade dysplasia (LGD) → high-grade dysplasia (HGD) → invasive adenocarcinoma. Management: no dysplasia — PPI therapy + surveillance endoscopy q3–5 years. LGD — endoscopic eradication therapy (radiofrequency ablation [RFA]) or surveillance q6–12 months. HGD — endoscopic eradication (RFA, endoscopic mucosal resection [EMR] for visible lesions) is first-line; esophagectomy reserved for endoscopic treatment failure or occult invasive cancer. T1a esophageal adenocarcinoma (mucosal, no lymphovascular invasion): EMR may be curative (<2% lymph node metastasis risk). T1b (submucosal invasion): esophagectomy recommended (lymph node metastasis risk ~20%).
09 Gastric Cancer
Staging & Preoperative Evaluation
EUS for T-staging, CT CAP for distant metastases, and diagnostic laparoscopy with peritoneal washings (positive cytology upstages to M1 in AJCC 8th edition) are standard. Lauren classification divides gastric cancer into intestinal (glandular, more differentiated, better prognosis) and diffuse (signet-ring cell, linitis plastica, worse prognosis, younger patients) types.
Extent of Gastrectomy
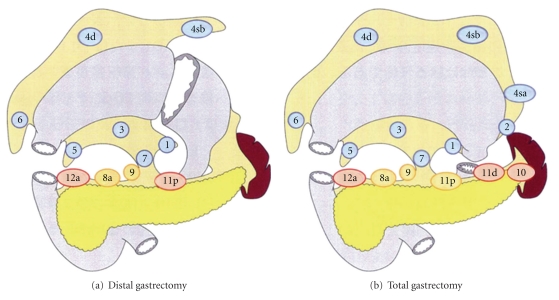
Distal (subtotal) gastrectomy is preferred for antral/distal tumors — equivalent survival to total gastrectomy with better quality of life and nutritional status (requires ≥5 cm proximal margin for intestinal type, ≥8 cm for diffuse type). Total gastrectomy is required for proximal tumors, linitis plastica, or when adequate proximal margins cannot be achieved. Reconstruction after distal gastrectomy: Billroth I (gastroduodenostomy), Billroth II (gastrojejunostomy), or Roux-en-Y gastrojejunostomy (preferred — lowest bile reflux). After total gastrectomy: Roux-en-Y esophagojejunostomy.
Lymphadenectomy — D1 vs D2
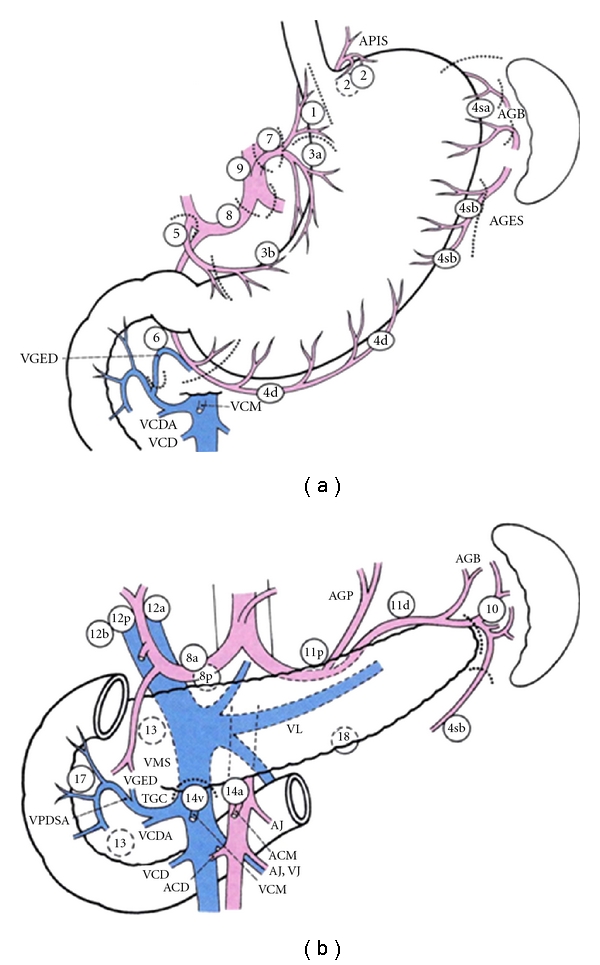
The extent of lymphadenectomy is a central debate. A D1 lymphadenectomy removes perigastric lymph nodes (stations 1–6). A D2 lymphadenectomy additionally removes nodes along the left gastric, common hepatic, celiac, and splenic arteries (stations 7–12). Japanese centers have long advocated D2 dissection, with superior survival outcomes. Western randomized trials (Dutch DGCT trial, UK MRC trial) initially showed no survival benefit and increased morbidity with D2, but long-term 15-year follow-up of the Dutch trial showed a cancer-specific survival benefit for D2 (PMID: 20880531). Current NCCN guidelines recommend D2 lymphadenectomy by experienced surgeons when safely feasible, with a minimum of 16 lymph nodes examined for adequate staging.
Perioperative Chemotherapy — MAGIC & FLOT
The MAGIC trial established perioperative chemotherapy (ECF: epirubicin, cisplatin, 5-FU) for resectable gastric cancer, demonstrating improved 5-year OS (36% vs 23%, PMID: 17615602). The FLOT4 trial subsequently showed that FLOT (5-FU, leucovorin, oxaliplatin, docetaxel) is superior to ECF/ECX, with improved median OS (50 vs 35 months, PMID: 30797662). FLOT is now the standard perioperative regimen for resectable gastric/GEJ adenocarcinoma (4 pre-op cycles + surgery + 4 post-op cycles).
HER2 Testing & Immunotherapy in Gastric Cancer
All advanced gastric/GEJ adenocarcinomas should be tested for HER2 overexpression (IHC 3+ or IHC 2+ with FISH amplification, ~15–20%). HER2-positive metastatic gastric cancer is treated with trastuzumab + chemotherapy (ToGA trial). PD-L1 testing (combined positive score, CPS) guides the use of checkpoint inhibitors: the CheckMate 649 trial showed nivolumab + chemotherapy improves OS in PD-L1 CPS ≥5 gastric/GEJ adenocarcinoma. MSI-high gastric cancers (~5%) have excellent responses to pembrolizumab.
10 Pancreatic Cancer
Resectability Assessment
Pancreatic ductal adenocarcinoma (PDAC) is resectable in only ~15–20% of patients at diagnosis. Classification:
| Category | Definition (Vessel Involvement) | Management |
|---|---|---|
| Resectable | No arterial contact (celiac, SMA, CHA); no venous involvement or ≤180° SMV/PV contact without irregularity | Upfront surgery → adjuvant therapy |
| Borderline resectable | Arterial: ≤180° SMA/celiac contact; Venous: >180° SMV/PV contact, or venous irregularity/thrombosis with suitable vessel for reconstruction | Neoadjuvant therapy → restaging → surgery if favorable response |
| Locally advanced | Arterial: >180° SMA/celiac contact; unreconstructible SMV/PV; aortic involvement | Systemic chemotherapy ± radiation; surgery only if significant downstaging |
| Metastatic | Distant metastases (liver, peritoneum, lung) | Palliative systemic therapy; no role for surgical resection of primary |
Surgical Procedures
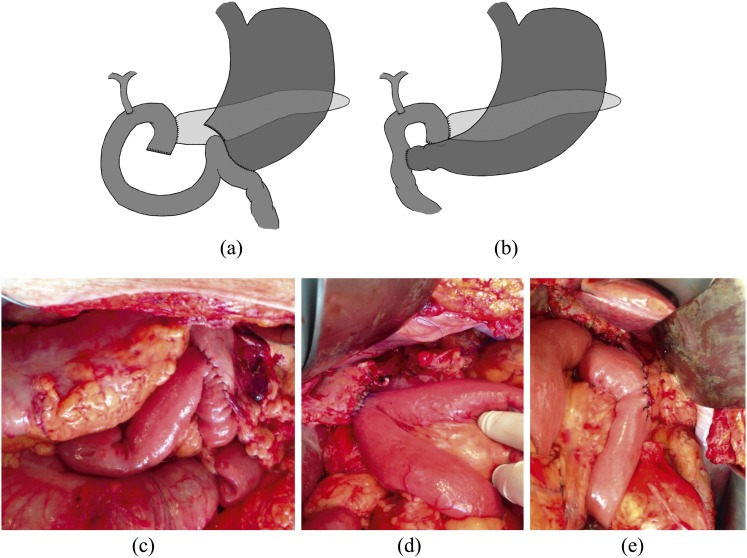
Pancreaticoduodenectomy (Whipple procedure) is the standard for pancreatic head tumors. Resected structures: head of pancreas, duodenum, distal common bile duct, gallbladder, and distal stomach (classic Whipple) or pylorus-preserving (PPPD — preserves the pylorus and proximal duodenum). Three anastomoses are created: pancreaticojejunostomy (or pancreaticogastrostomy), hepaticojejunostomy, and gastro/duodenojejunostomy. Distal pancreatectomy + splenectomy is performed for body/tail tumors. Total pancreatectomy is reserved for multicentric disease or positive pancreatic neck margin on frozen section.
Neoadjuvant Therapy
Neoadjuvant therapy is standard for borderline resectable disease and increasingly used for clearly resectable PDAC. FOLFIRINOX (5-FU, leucovorin, irinotecan, oxaliplatin) is the preferred regimen for fit patients (improved OS vs gemcitabine in metastatic setting, PMID: 21612468; extrapolated to neoadjuvant). Gemcitabine + nab-paclitaxel is an alternative for patients who cannot tolerate FOLFIRINOX. Adjuvant therapy after resection: modified FOLFIRINOX for 6 months (PRODIGE 24 trial showed superior DFS and OS vs gemcitabine alone, PMID: 30575490).
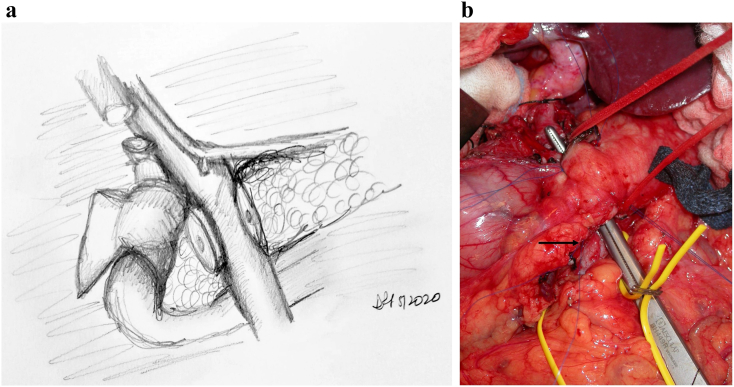
Vascular Resection in Pancreatic Surgery
Venous resection (SMV/PV) during pancreatectomy is commonly performed when the tumor involves the venous confluence. Types: tangential (lateral) resection with primary venorrhaphy, segmental resection with primary end-to-end anastomosis (if ≤2–3 cm segment), or interposition graft (internal jugular vein, synthetic PTFE) for longer defects. Venous resection to achieve R0 does not worsen outcomes compared to standard pancreatectomy without venous resection. Arterial resection (SMA, CHA) is more controversial — associated with higher morbidity and mortality — and should be performed only at high-volume centers in selected patients after neoadjuvant therapy, with careful patient selection and MDT discussion.
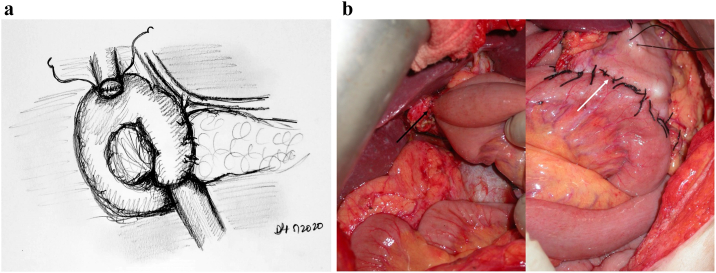
Palliative Surgery in Pancreatic Cancer
For unresectable pancreatic cancer with biliary obstruction: endoscopic metal stent (ERCP) is preferred for palliation (patency 6–12 months). For gastric outlet obstruction: endoscopic duodenal stent or surgical gastrojejunostomy (open or laparoscopic). If unresectability is found at the time of planned pancreatectomy, a prophylactic gastrojejunostomy should be considered (prevents future gastric outlet obstruction in ~20% of patients). Celiac plexus neurolysis (percutaneous or EUS-guided) provides pain relief in ~70–80% of patients with pancreatic cancer pain.
11 Colorectal Cancer Surgery
Surgical Principles
Curative colorectal cancer resection requires: adequate margins (5-cm proximal/distal margins for colon cancer; for rectal cancer, a 1–2 cm distal margin is acceptable with TME), adequate lymphadenectomy (minimum 12 lymph nodes examined for accurate staging — AJCC recommendation), and high ligation of the feeding vessel at its origin. The type of resection is dictated by the tumor location and its vascular supply.
| Tumor Location | Procedure | Vessels Ligated |
|---|---|---|
| Cecum, ascending colon | Right hemicolectomy | Ileocolic, right colic, right branch of middle colic |
| Hepatic flexure | Extended right hemicolectomy | Ileocolic, right colic, middle colic |
| Transverse colon | Transverse colectomy or extended right/left | Middle colic |
| Splenic flexure, descending colon | Left hemicolectomy | Left colic (IMA branches) |
| Sigmoid colon | Sigmoid colectomy | IMA (high ligation) or sigmoid branches |
| Upper rectum (above peritoneal reflection) | Low anterior resection (LAR) | IMA; TME |
| Mid/low rectum | LAR with TME + defunctioning ileostomy | IMA; total mesorectal excision |
| Very low rectum / sphincter involvement | Abdominoperineal resection (APR) | IMA; permanent colostomy |
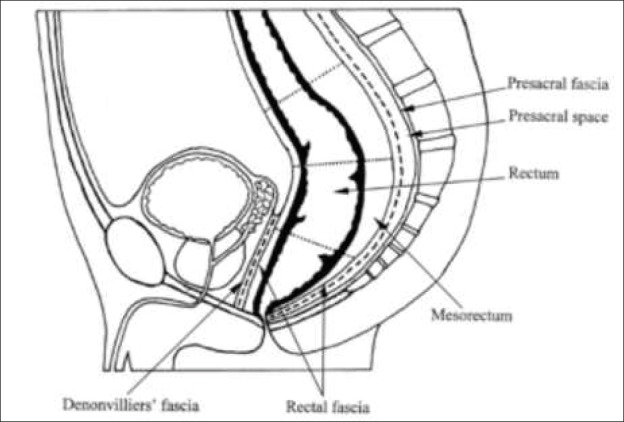
Total Mesorectal Excision (TME)
Described by Heald (1982), TME involves sharp dissection along the mesorectal fascia (the "holy plane"), excising the rectum with an intact mesorectal envelope. TME reduced local recurrence rates from 30–40% to <5–10%. The quality of the TME specimen is graded: complete (intact mesorectal fascia), nearly complete (minor irregularities), or incomplete (defects down to the muscularis propria — associated with higher recurrence). For upper rectal cancers, a tumor-specific mesorectal excision (partial TME, with at least 5 cm of mesorectum distal to the tumor) is acceptable.
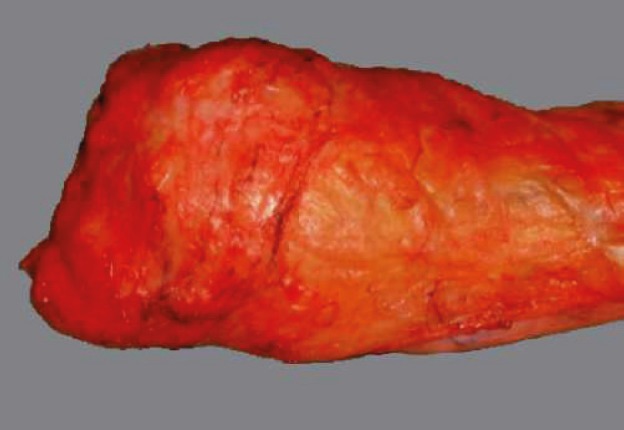
Complete Mesocolic Excision (CME)
Analogous to TME for rectal cancer, CME with central vascular ligation (CVL) for colon cancer involves dissection in the embryologic plane between the mesocolic fascia and Toldt's fascia, with ligation of the feeding artery at its origin. Proponents (Hohenberger) report improved oncologic outcomes with higher lymph node yields and reduced local recurrence compared to conventional colectomy.
Molecular Markers in CRC
All colorectal cancers should be tested for: MSI / MMR status (MSI-high tumors are right-sided, mucinous, poorly differentiated, with dense lymphocytic infiltrate — better prognosis stage for stage; do not benefit from 5-FU alone; screening for Lynch syndrome if MSI-high; dramatic response to checkpoint inhibitors in metastatic setting), RAS mutations (KRAS/NRAS — present in ~50% of CRC; precludes use of anti-EGFR therapy such as cetuximab and panitumumab), BRAF V600E (present in ~8–10%; associated with poor prognosis; if MSI-stable, very aggressive disease; BRAF-mutant metastatic CRC treated with encorafenib + cetuximab, BEACON trial).
| Marker | Frequency | Clinical Impact |
|---|---|---|
| MSI-high / dMMR | ~15% (higher in stage II) | Excellent prognosis; no benefit from 5-FU alone in stage II; checkpoint inhibitor response in stage IV; Lynch screening |
| KRAS exon 2–4 | ~40% | No anti-EGFR therapy (cetuximab/panitumumab); standard chemotherapy backbone |
| NRAS | ~5% | No anti-EGFR therapy |
| BRAF V600E | ~8–10% | Poor prognosis (MSS); encorafenib + cetuximab (BEACON); if MSI-H + BRAF: somatic, not Lynch |
| HER2 amplification | ~3–5% | Trastuzumab + pertuzumab (MyPathway, HERACLES); typically RAS/BRAF wild-type |
| NTRK fusion | <1% | Larotrectinib or entrectinib (tumor-agnostic approval) |
| ctDNA (post-op) | Variable | Positive ctDNA after curative resection predicts recurrence; guides adjuvant therapy decisions (DYNAMIC trial) |
Surgical Considerations for Left vs Right Colon Cancer
The distinction between left-sided and right-sided CRC has emerged as clinically significant. Right-sided (cecum to hepatic flexure): more commonly MSI-high, BRAF-mutated, mucinous histology; present with anemia/occult blood; better prognosis with immunotherapy; worse prognosis with conventional chemotherapy. Left-sided (splenic flexure to rectum): more commonly MSS, chromosomal instability pathway, more responsive to anti-EGFR therapy (cetuximab/panitumumab); present with obstruction, bleeding, change in stool caliber; generally better overall prognosis with chemotherapy. Surgical technique: right hemicolectomy includes ligation of the ileocolic and right colic arteries with an ileocolonic anastomosis. Left-sided resections require attention to splenic flexure mobilization for tension-free anastomosis and consideration of proximal diversion for low rectal anastomoses.
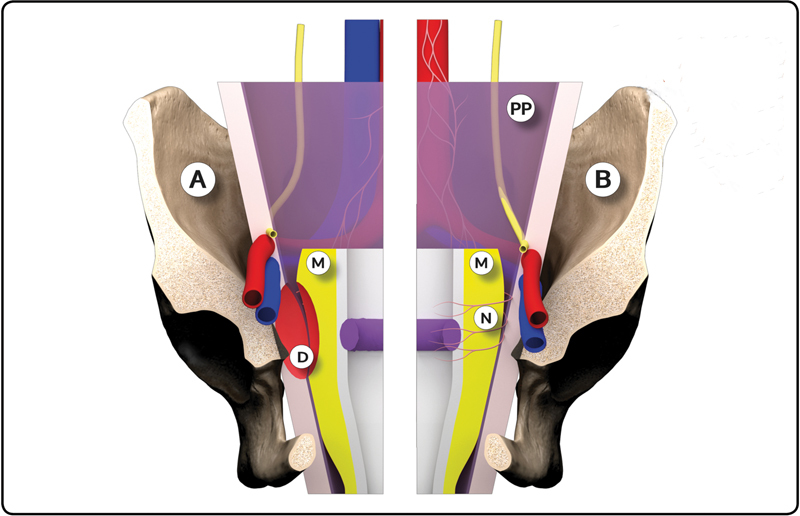
Rectal Cancer — Total Neoadjuvant Therapy (TNT)
Rectal cancer management has evolved from postoperative chemoradiation to total neoadjuvant therapy (TNT) — delivering all chemotherapy and radiation before surgery. The RAPIDO trial and PRODIGE 23 trial both demonstrated improved disease-free survival with TNT approaches. The OPRA trial investigated a watch-and-wait strategy for patients achieving clinical complete response (cCR) after TNT — organ preservation was achieved in ~50% of patients without compromising overall survival. Assessment of cCR requires: DRE, endoscopy with biopsy (if needed), and MRI showing no residual tumor. Surveillance for watch-and-wait: every 3–4 months for 2 years, then every 6 months (DRE, endoscopy, MRI, CEA).
Anal Cancer — Nigro Protocol
Squamous cell carcinoma of the anal canal is treated with definitive chemoradiation (Nigro protocol) — 5-FU + mitomycin C + concurrent radiation — NOT primary surgery. The Nigro protocol achieves complete response in ~85% of patients, with sphincter preservation. Surgery (APR) is reserved for treatment failure or recurrence after chemoradiation. Staging is clinical (tumor size and nodal status). HPV vaccination (Gardasil 9) may reduce anal cancer incidence.
12 GIST & Neuroendocrine Tumors
Gastrointestinal Stromal Tumors (GIST)
GISTs are the most common mesenchymal tumors of the GI tract, arising from the interstitial cells of Cajal. Most common location: stomach (60%) > small bowel (30%) > colorectal (5%) > esophagus (<5%). Driven by gain-of-function mutations in KIT (CD117, ~80%) or PDGFRA (~10%). Diagnosis: immunohistochemistry positive for CD117 (KIT) and DOG1.
GIST Risk Stratification — Miettinen/NIH Criteria
| Risk Category | Size | Mitotic Rate | Recurrence Risk |
|---|---|---|---|
| Very low | ≤2 cm | ≤5 / 50 HPF | <2% |
| Low | 2.1–5 cm | ≤5 / 50 HPF | 2–5% |
| Intermediate | ≤5 cm AND >5/50 HPF; OR 5.1–10 cm AND ≤5/50 HPF | — | 10–25% |
| High | >10 cm OR >5/50 HPF with size >5 cm OR any size >10/50 HPF | — | 40–90% |
Surgery: Complete (R0) resection with negative margins is curative. Lymph node dissection is NOT required (GISTs rarely metastasize to lymph nodes). Margins of 1–2 cm are adequate; the goal is an intact pseudocapsule. GISTs metastasize hematogenously to the liver and peritoneum; lung/bone metastases are rare. Laparoscopic resection is appropriate for small to moderate-sized tumors. Avoid tumor rupture (spillage seeds the peritoneum).
Imatinib (Gleevec): A tyrosine kinase inhibitor targeting KIT. Adjuvant imatinib (400 mg daily for 3 years) is standard for high-risk GISTs after R0 resection (SSG XVIII/AIO trial, PMID: 22089421). Neoadjuvant imatinib is used to downsize large/borderline resectable GISTs and enable less extensive surgery. KIT exon 11 mutations have the best response to imatinib. PDGFRA D842V mutations are imatinib-resistant — avapritinib is the specific agent. For imatinib-resistant GISTs: sunitinib (2nd line), regorafenib (3rd line), ripretinib (4th line).
Neuroendocrine Tumors (NETs)
NETs arise from the diffuse neuroendocrine cell system throughout the GI tract, pancreas, and lungs. Classified by WHO grading:
| Grade | Ki-67 | Mitotic Rate | Differentiation | Behavior |
|---|---|---|---|---|
| G1 (low) | <3% | <2 / 10 HPF | Well-differentiated | Indolent; often curable with surgery |
| G2 (intermediate) | 3–20% | 2–20 / 10 HPF | Well-differentiated | Intermediate prognosis |
| G3 (high) | >20% | >20 / 10 HPF | Well-differentiated NET or poorly differentiated NEC | Aggressive; NEC treated like small cell carcinoma (cisplatin/etoposide) |
Functional NETs produce hormones: carcinoid (serotonin — flushing, diarrhea, right-sided valvular heart disease; diagnose with 24-hr urine 5-HIAA and chromogranin A), insulinoma (hypoglycemia — Whipple triad; localize with endoscopic US ± intraoperative US; 90% benign — enucleation), gastrinoma (Zollinger-Ellison syndrome — recurrent peptic ulcers, diarrhea; 60% malignant; duodenal wall > pancreas; associated with MEN1 in 25%). Surgical resection is the primary treatment for localized NETs. Hepatic metastases from NETs may be resected or debulked if >90% of disease is resectable (cytoreduction improves symptoms and survival).
13 Hepatocellular Carcinoma
BCLC Staging & Treatment Algorithm
The Barcelona Clinic Liver Cancer (BCLC) staging system integrates tumor burden, liver function (Child-Pugh), and performance status to guide treatment:
| BCLC Stage | Tumor Status | Liver Function / PS | Treatment |
|---|---|---|---|
| 0 (Very early) | Single ≤2 cm | Child-Pugh A, PS 0 | Resection, ablation, or transplant |
| A (Early) | Single or up to 3 nodules ≤3 cm | Child-Pugh A–B, PS 0 | Resection (if single, preserved liver function), transplant (within Milan), or ablation |
| B (Intermediate) | Multinodular, beyond Milan | Child-Pugh A–B, PS 0 | TACE (transarterial chemoembolization) |
| C (Advanced) | Vascular invasion or extrahepatic spread | Child-Pugh A–B, PS 1–2 | Systemic therapy: atezolizumab + bevacizumab (IMbrave150, PMID: 32402160) |
| D (Terminal) | Any | Child-Pugh C, PS 3–4 | Best supportive care |
Surgical Resection
Resection is the treatment of choice for HCC in patients with well-preserved liver function (Child-Pugh A, no portal hypertension, adequate future liver remnant [FLR]). Indications for resection: single tumor, no vascular invasion, adequate FLR (≥20% in normal liver, ≥30% in steatotic liver, ≥40% in cirrhotic liver). Preoperative assessment includes: ICG-R15 (indocyanine green retention at 15 min — <10% is favorable for major hepatectomy), CT volumetry for FLR calculation, and portal hypertension assessment (hepatic venous pressure gradient ≥10 mmHg = significant portal hypertension → resection has high morbidity).
Liver Transplantation — Milan Criteria
The Milan criteria (Mazzaferro, 1996, PMID: 8596091) define the gold standard for liver transplant candidacy in HCC: single tumor ≤5 cm or up to 3 tumors each ≤3 cm, no vascular invasion, no extrahepatic disease. Within Milan criteria, 5-year post-transplant survival exceeds 70% with <10% recurrence. Extended criteria (UCSF criteria: single tumor ≤6.5 cm or ≤3 tumors each ≤4.5 cm with total tumor diameter ≤8 cm) modestly expand the eligible population. Bridging therapy (TACE, ablation, Y-90) is used to prevent tumor progression while awaiting transplant.
Ablation Techniques for HCC
Radiofrequency ablation (RFA) and microwave ablation (MWA) are curative options for BCLC 0/A tumors not suitable for resection. For tumors ≤2 cm, ablation has outcomes comparable to resection. RFA creates a thermal zone of coagulative necrosis using alternating current (target temperature 60–100°C). MWA generates electromagnetic waves that agitate water molecules — faster heating, larger ablation zones, less susceptible to heat-sink effect near vessels. Cryoablation uses argon gas to freeze tissue (risk of cryoshock syndrome with large ablation zones). Irreversible electroporation (IRE / NanoKnife) is non-thermal — preserves adjacent bile ducts and vessels — used for tumors near critical structures.
Locoregional Therapies for HCC
TACE (transarterial chemoembolization): Standard treatment for BCLC B (intermediate) HCC. Catheter-based delivery of chemotherapy (doxorubicin or cisplatin) mixed with embolic agents (Lipiodol + gelfoam, or drug-eluting beads) into the tumor-feeding hepatic artery branches. HCC derives >90% of its blood supply from the hepatic artery (normal liver parenchyma is predominantly portal vein supplied), enabling selective tumor targeting. Response assessed by mRECIST (modified RECIST — evaluates arterially enhancing viable tumor, not just size). TARE / Y-90 (transarterial radioembolization): Yttrium-90 labeled microspheres delivered into the hepatic artery; provides selective internal radiation. Used for intermediate/advanced HCC, particularly with portal vein invasion (contraindication to TACE). Requires pre-treatment MAA scan to assess lung shunt fraction (>20% = contraindication).
- Single tumor ≤2 cm, Child-Pugh A: Resection OR ablation (equivalent outcomes)
- Single tumor, no portal HTN, adequate FLR: Surgical resection
- Within Milan criteria, not resectable: Liver transplantation (bridge with TACE/ablation/Y-90)
- Multinodular, preserved liver function: TACE
- Vascular invasion or extrahepatic disease: Atezolizumab + bevacizumab (if Child-Pugh A/B7)
- Child-Pugh C / poor performance status: Best supportive care
14 Liver Metastases & Cholangiocarcinoma
Colorectal Liver Metastases (CRLM)
The liver is the most common site of colorectal cancer metastasis (~50% of patients). Surgical resection of CRLM offers 5-year survival of 40–58%, compared with <5% without treatment. Resectability criteria have evolved from tumor-based (number, size) to patient-based: the key question is whether an R0 resection can be achieved while leaving adequate FLR with intact vascular inflow, outflow, and biliary drainage.
Fong Clinical Risk Score (CRS)
| Risk Factor | Points |
|---|---|
| Node-positive primary | 1 |
| Disease-free interval <12 months | 1 |
| Number of metastases >1 | 1 |
| Largest metastasis >5 cm | 1 |
| CEA >200 ng/mL | 1 |
| Score 0: 5-yr OS ~60%; Score 5: 5-yr OS ~14% (PMID: 10493478) | |
Strategies to Increase Resectability
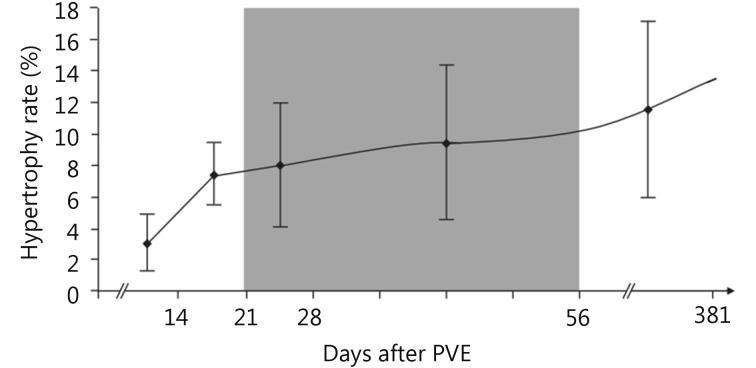
Portal vein embolization (PVE): Embolization of the portal vein branch supplying the lobe to be resected induces compensatory hypertrophy of the FLR over 4–6 weeks. Used when FLR is insufficient for major hepatectomy. Two-stage hepatectomy: Stage 1 — resect metastases from the FLR; perform PVE → wait for hypertrophy → Stage 2 — resect the remaining metastases with major hepatectomy. ALPPS (Associating Liver Partition and Portal vein ligation for Staged hepatectomy): Stage 1 — in situ liver splitting + portal vein ligation → rapid hypertrophy (1–2 weeks) → Stage 2 — completion hepatectomy. ALPPS achieves faster hypertrophy but carries higher morbidity (Clavien-Dindo ≥IIIa in ~30%) and should be reserved for experienced centers.
Hepatic Artery Infusion (HAI) Pump
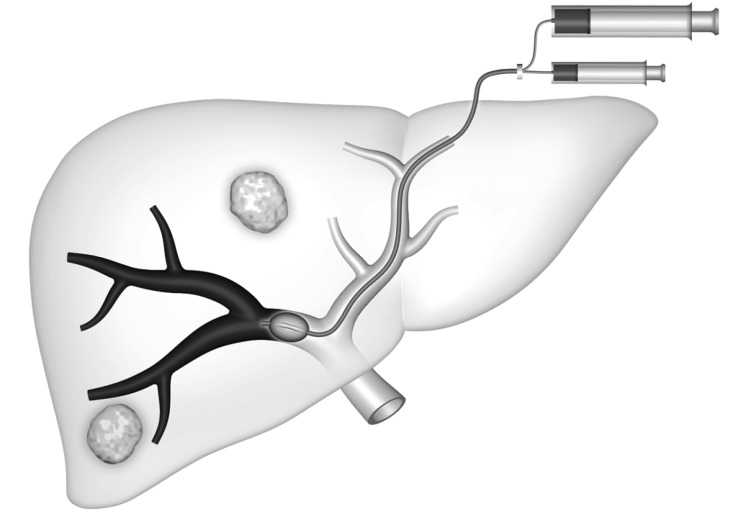
HAI delivers floxuridine (FUDR) directly into the hepatic artery, exploiting the hepatic arterial blood supply of liver metastases. Combined with systemic chemotherapy, HAI achieves response rates of 60–80% and can convert unresectable CRLM to resectable in ~50% of patients (MSKCC experience). HAI is technically demanding — requires pump implantation and meticulous attention to hepatic artery anatomy. Complications: biliary sclerosis (prevented by dexamethasone in the pump), gastric/duodenal ulceration (from inadvertent perfusion — requires cholecystectomy and ligation of extrahepatic branches at pump placement).
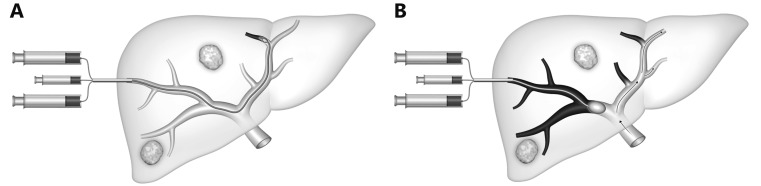
Cholangiocarcinoma
Classification by location: intrahepatic (iCCA, ~10%), perihilar / Klatskin tumor (pCCA, ~60%), and distal (dCCA, ~30%). The Bismuth-Corlette classification for perihilar cholangiocarcinoma guides surgical planning:
| Type | Description | Surgical Approach |
|---|---|---|
| I | Below the confluence of right and left hepatic ducts | Bile duct resection + regional lymphadenectomy |
| II | Reaches the confluence | Bile duct resection + regional lymphadenectomy |
| IIIa | Involves the right hepatic duct | Right hepatectomy + bile duct resection + caudate lobectomy |
| IIIb | Involves the left hepatic duct | Left hepatectomy + bile duct resection + caudate lobectomy |
| IV | Involves both right and left hepatic ducts | Generally unresectable; transplant protocols in select centers |
15 Gallbladder Cancer & Peritoneal Surface Malignancies
Gallbladder Cancer
Gallbladder cancer is frequently an incidental finding after cholecystectomy for presumed benign disease (~0.2–0.7% of cholecystectomies). Management depends on T-stage:
| T-Stage | Depth of Invasion | Management |
|---|---|---|
| Tis / T1a | Carcinoma in situ / invasion of lamina propria | Simple cholecystectomy is curative (ensure negative cystic duct margin) |
| T1b | Invasion of muscular layer | Radical cholecystectomy (re-resection of gallbladder bed — segments IVb/V hepatectomy + portal lymphadenectomy) |
| T2 | Perimuscular connective tissue, no serosal penetration | Radical cholecystectomy + excision of any laparoscopic port sites (risk of port-site metastasis after laparoscopic cholecystectomy) |
| T3–T4 | Serosal penetration / liver or adjacent organ invasion | Extended hepatectomy + portal lymphadenectomy if R0 resection achievable; many are unresectable |
Peritoneal Surface Malignancies — CRS/HIPEC
Peritoneal carcinomatosis was historically considered a terminal condition. Cytoreductive surgery (CRS) + hyperthermic intraperitoneal chemotherapy (HIPEC), pioneered by Sugarbaker, has transformed outcomes for selected patients. CRS involves peritonectomy procedures (parietal peritoneal stripping) and visceral resections (omentectomy, bowel resections, splenectomy, etc.) to achieve complete macroscopic cytoreduction (CC-0: no visible disease, or CC-1: residual nodules <2.5 mm).
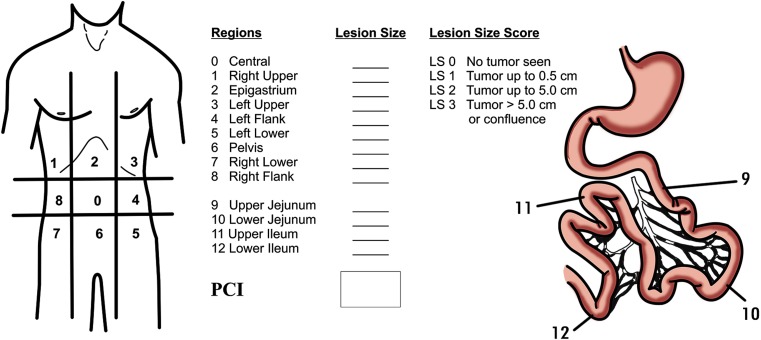
Peritoneal Cancer Index (PCI)
The PCI score (Sugarbaker) divides the abdomen into 13 regions; each is scored 0–3 based on the largest implant size. Total score range: 0–39. Lower PCI = better outcomes after CRS/HIPEC. Generally, PCI >20 for colorectal origin or >20–25 for appendiceal mucinous neoplasms carries a poor prognosis even with complete cytoreduction.
Indications for CRS/HIPEC: appendiceal mucinous neoplasms with pseudomyxoma peritonei (best outcomes — 10-year survival ~60–70%), colorectal peritoneal metastases (PRODIGE 7 trial questioned HIPEC benefit over CRS alone for CRC, PMID: 30192068), peritoneal mesothelioma, selected ovarian cancer (interval debulking + HIPEC — OVHIPEC trial), and selected gastric cancer with limited peritoneal disease. Common HIPEC agents: mitomycin C, oxaliplatin, cisplatin. Temperature: 41–43°C for 60–90 minutes.
Pseudomyxoma Peritonei (PMP)
PMP is a clinical syndrome of mucinous ascites and peritoneal implants, most commonly arising from ruptured appendiceal mucinous neoplasms. Classification: low-grade (DPAM — disseminated peritoneal adenomucinosis) — scant cellularity, pushing invasion, excellent prognosis with CRS/HIPEC (10-year survival 60–70%); high-grade (PMCA — peritoneal mucinous carcinomatosis) — higher cellularity, destructive invasion, worse prognosis (10-year survival 10–20%); intermediate (PMCA-I). Treatment: complete CRS + HIPEC (mitomycin C for 90 min). Completeness of cytoreduction is the single most important prognostic factor. Serial debulking procedures may be needed for recurrence. The "redistribute" phenomenon — mucinous tumor tracks to the greater omentum, right diaphragm, pelvis, and right paracolic gutter preferentially due to peritoneal fluid circulation — guides the surgical approach.
- PCI >20 for colorectal carcinomatosis (or >25 for appendiceal mucinous neoplasms)
- ECOG performance status ≥3
- Unresectable extra-peritoneal disease (liver metastases — exception: limited resectable liver disease may be included)
- Extensive small bowel involvement precluding safe resection (short gut risk)
- Bilaterally obstructing ureteral disease
- Massive mesenteric root disease
- Poor nutritional status (albumin <2.5 g/dL) without optimization
16 Melanoma — Staging, Excision, & SLNB
Melanoma Staging
Melanoma staging relies on Breslow depth (measured in mm from the granular layer of the epidermis to the deepest point of tumor invasion — the single most important prognostic factor for localized melanoma), Clark level (I–V based on anatomic depth of invasion — largely supplanted by Breslow depth but still reported), ulceration (upstages the T category), mitotic rate (removed from AJCC 8th edition T1 substaging but still prognostically relevant), and sentinel lymph node status (the most powerful predictor of recurrence for intermediate-thickness melanoma).
Wide Local Excision Margins
| Breslow Depth | Recommended Margin | Evidence |
|---|---|---|
| In situ (Tis) | 0.5–1 cm | NCCN guidelines; no RCT data |
| ≤1.0 mm (T1) | 1 cm | WHO Melanoma Program trial; Swedish trial |
| 1.01–2.0 mm (T2) | 1–2 cm | Intergroup Melanoma Surgical Trial; UK MSG trial |
| 2.01–4.0 mm (T3) | 2 cm | Intergroup trial; Swedish trial |
| >4.0 mm (T4) | 2 cm | No additional benefit from margins >2 cm (UK MSG trial) |
Sentinel Lymph Node Biopsy for Melanoma
The MSLT-I trial (PMID: 24295718) established SLNB as a staging procedure for intermediate-thickness melanoma (1–4 mm Breslow). SLNB provides critical prognostic information: SLN-positive patients have a 5-year survival of ~72% vs ~90% for SLN-negative patients. Indications: Breslow depth ≥0.8 mm, or any thickness with ulceration or high mitotic rate. SLNB is not routinely recommended for melanoma in situ or Breslow <0.8 mm without adverse features.
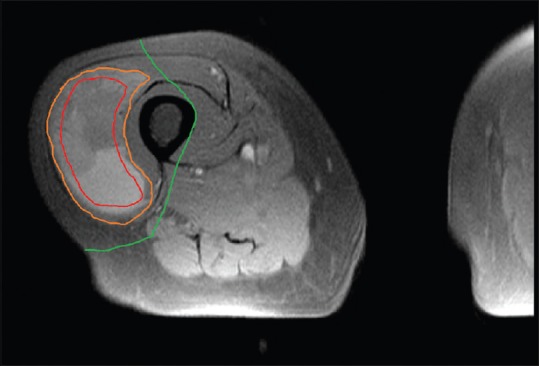
MSLT-II Trial — Completion Lymphadenectomy
The MSLT-II trial (PMID: 28614720) demonstrated that completion lymph node dissection (CLND) does NOT improve melanoma-specific survival in patients with SLN-positive melanoma compared to ultrasound surveillance of the nodal basin. CLND did improve regional disease control and provided prognostic information (non-SLN positivity). Current practice: CLND is no longer routinely recommended; observation with ultrasound surveillance of the nodal basin is the standard for most SLN-positive melanoma patients.
17 Advanced Melanoma & Non-Melanoma Skin Cancer
Melanoma AJCC 8th Edition — T-Stage Summary
| T Category | Breslow Depth | Ulceration |
|---|---|---|
| T1a | <0.8 mm | Without ulceration |
| T1b | <0.8 mm with ulceration, or 0.8–1.0 mm with or without ulceration | — |
| T2a | >1.0–2.0 mm | Without ulceration |
| T2b | >1.0–2.0 mm | With ulceration |
| T3a | >2.0–4.0 mm | Without ulceration |
| T3b | >2.0–4.0 mm | With ulceration |
| T4a | >4.0 mm | Without ulceration |
| T4b | >4.0 mm | With ulceration |
In-Transit Disease
In-transit metastases are cutaneous/subcutaneous deposits >2 cm from the primary but within the regional nodal basin. Treatment options: surgical excision (if limited), isolated limb infusion (ILI) or isolated limb perfusion (ILP) with melphalan ± TNF-alpha (higher temperatures in ILP → higher response rates ~60–80%), intralesional injection (talimogene laherparepvec / T-VEC — oncolytic herpes virus, FDA-approved for unresectable melanoma), and systemic immunotherapy.
Immunotherapy in Melanoma
Checkpoint inhibitors have revolutionized melanoma treatment. Adjuvant immunotherapy: nivolumab (CheckMate 238) or pembrolizumab (KEYNOTE-054) for resected stage III melanoma improves recurrence-free survival. Neoadjuvant immunotherapy is an active area of investigation — the SWOG S1801 trial showed improved event-free survival with neoadjuvant + adjuvant pembrolizumab vs adjuvant pembrolizumab alone for resectable stage III–IV melanoma. For BRAF-mutant melanoma (V600E/K, ~50% of cutaneous melanoma), targeted therapy with dabrafenib + trametinib is an alternative.
- Stage 0 (in situ): WLE with 0.5–1 cm margins; no SLNB needed
- Stage I (T1–T2a, N0): WLE with 1 cm margin (T1) or 1–2 cm (T2a); SLNB for T1b (≥0.8 mm) and all T2
- Stage II (T2b–T4b, N0): WLE with 1–2 cm margins; SLNB; consider adjuvant nivolumab (CheckMate 76K) or pembrolizumab for stage IIB–IIC (recent data)
- Stage III (any T, N+): WLE + SLNB; no routine CLND (MSLT-II); adjuvant immunotherapy (nivolumab or pembrolizumab x 1 year) or dabrafenib + trametinib if BRAF-mutant; consider neoadjuvant (S1801 paradigm)
- Stage IV (distant metastases): Combination immunotherapy (nivolumab + ipilimumab) or single-agent anti-PD-1; BRAF/MEK inhibitors if BRAF V600 mutant; surgical metastasectomy for limited resectable disease (oligometastatic)
Merkel Cell Carcinoma
Rare, aggressive neuroendocrine skin cancer associated with Merkel cell polyomavirus (MCPyV, ~80% of cases) and UV exposure / immunosuppression. Presents as a rapidly growing, painless, violaceous nodule — commonly misdiagnosed initially. Management: wide local excision (1–2 cm margins) + SLNB (high rate of nodal metastasis ~30%). Adjuvant radiation to the primary site and regional nodes improves local control. Checkpoint inhibitors (avelumab, pembrolizumab) are first-line for advanced/metastatic disease — response rates ~30–60%.
Non-Melanoma Skin Cancer Requiring Surgical Oncology
Most BCC and SCC are managed by dermatology/Mohs surgery. Surgical oncology is involved for: locally advanced BCC refractory to local therapy (hedgehog pathway inhibitors — vismodegib, sonidegib — for unresectable or metastatic BCC), SCC with perineural invasion (especially named-nerve involvement — adjuvant radiation recommended), cutaneous SCC with regional lymph node metastasis (therapeutic lymph node dissection + adjuvant radiation ± cemiplimab for advanced disease), and dermatofibrosarcoma protuberans (DFSP) — wide local excision with 2–3 cm margins or Mohs surgery; rarely metastasizes but has high local recurrence if inadequately excised; imatinib for unresectable/metastatic disease (COL1A1-PDGFB fusion gene).
18 Soft Tissue Sarcoma & Retroperitoneal Sarcoma
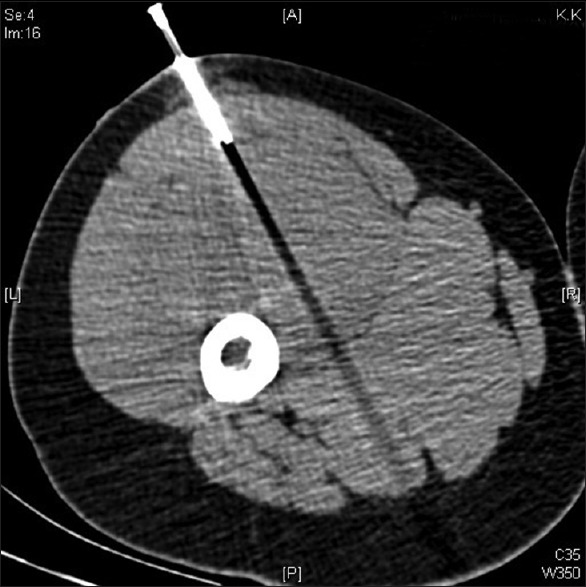
Diagnosis & Biopsy
Soft tissue sarcomas (STS) are rare mesenchymal tumors (~1% of adult malignancies) with >50 histologic subtypes. The most common subtypes are undifferentiated pleomorphic sarcoma (UPS), liposarcoma (well-differentiated, dedifferentiated, myxoid, pleomorphic), and leiomyosarcoma. Biopsy approach is critical: core needle biopsy is preferred (multiple passes for adequate tissue; avoids disrupting tissue planes). If incisional biopsy is necessary, the incision must be placed longitudinally along the extremity so it can be excised with the definitive resection specimen. Excisional biopsy of a suspected sarcoma (the "whoops" procedure) contaminates tissue planes and dramatically complicates definitive surgery.
FNCLCC Grading System
| Parameter | Score |
|---|---|
| Tumor differentiation: 1 = resembles normal tissue, 2 = certain histologic typing, 3 = undifferentiated/embryonal | 1–3 |
| Mitotic count: 1 = 0–9/10 HPF, 2 = 10–19/10 HPF, 3 = ≥20/10 HPF | 1–3 |
| Tumor necrosis: 0 = none, 1 = <50%, 2 = ≥50% | 0–2 |
| Total: Grade 1 (2–3), Grade 2 (4–5), Grade 3 (6–8). Grade is the most important prognostic factor for STS. | |
Surgical Management of Extremity STS
Limb-sparing surgery is the standard of care — equivalent survival to amputation when combined with radiation (landmark Rosenberg NCI trial, PMID: 6333478). The goal is R0 resection with a cuff of normal tissue (≥1 cm or an intact fascial plane). Lymph node dissection is not routinely performed (STS rarely metastasizes to lymph nodes — exceptions: synovial sarcoma, epithelioid sarcoma, rhabdomyosarcoma, clear cell sarcoma). STS metastasizes hematogenously to the lungs — pulmonary metastasectomy can provide long-term survival in select patients with isolated lung metastases.
Radiation Sequencing
Preoperative radiation (50 Gy in 25 fractions) vs postoperative radiation (66 Gy in 33 fractions) was compared in the Canadian SR2 trial (PMID: 12091249). Preoperative RT: smaller field, lower dose, better long-term function, but higher wound complication rate (35% vs 17%, especially for lower extremity). Postoperative RT: lower wound complications but higher late fibrosis. Current preference favors preoperative RT for most extremity STS.
Systemic Therapy for STS
Adjuvant chemotherapy for STS is not routinely recommended (unlike many solid tumors) — a pooled meta-analysis showed a modest 6% absolute survival benefit with doxorubicin-based chemotherapy, and it remains controversial. Neoadjuvant chemotherapy may be considered for large, high-grade, deep tumors where the goal is to shrink the tumor for limb salvage. Standard agents: doxorubicin ± ifosfamide (AIM regimen). For specific subtypes: myxoid liposarcoma is notably radiosensitive and chemosensitive (trabectedin effective); angiosarcoma responds to taxanes; synovial sarcoma is relatively chemosensitive (ifosfamide-based); solitary fibrous tumor may respond to temozolomide + bevacizumab.
Pulmonary Metastasectomy for Sarcoma
STS metastasizes predominantly to the lungs. Surgical resection of pulmonary metastases is the only potentially curative treatment for isolated lung disease. Selection criteria for metastasectomy: (1) controlled primary site, (2) no extrapulmonary metastases, (3) all pulmonary disease can be completely resected, (4) adequate pulmonary reserve. Wedge resection (preserving lung parenchyma) is preferred over anatomic lobectomy. Five-year survival after complete pulmonary metastasectomy ranges from 20–40%. Prognostic factors: disease-free interval, number of metastases, and ability to achieve complete resection.
Retroperitoneal Sarcoma
Retroperitoneal sarcomas (RPS) present as large masses (often >15 cm) and are predominantly liposarcoma or leiomyosarcoma. Complete surgical resection with negative margins is the goal, but true negative margins are often impossible due to critical adjacent structures (aorta, IVC, kidneys). Compartmental resection (en bloc removal of the tumor with contiguous organs — nephrectomy, colectomy, psoas excision) is advocated by some centers to reduce local recurrence. The STRASS trial (PMID: 31986257) evaluated preoperative RT for RPS and did not show an overall benefit in abdominal recurrence-free survival, though a pre-specified subgroup analysis suggested benefit for liposarcoma. Management should be centralized at high-volume sarcoma centers.
19 Desmoid Tumors & GIST (Sarcoma Context)
Desmoid Tumors (Aggressive Fibromatosis)
Desmoid tumors are locally aggressive, non-metastasizing fibroproliferative neoplasms arising from musculoaponeurotic tissues. They are associated with familial adenomatous polyposis (FAP) / Gardner syndrome (mesenteric desmoids), trauma, and pregnancy. Management has shifted dramatically toward active surveillance as the initial approach — many desmoids stabilize or regress spontaneously (~50% stabilize or regress without treatment over 3–5 years). Surgery carries a high recurrence rate (20–40%) because achieving wide margins is often impossible and positive margins paradoxically do not always predict recurrence.
Current treatment algorithm: observe → if progressive or symptomatic: systemic therapy (NSAIDs + tamoxifen/raloxifene for mild disease and slow progression; sorafenib for moderate progressive disease — PFS benefit in the DESMOFIB trial; doxorubicin-based chemotherapy or methotrexate + vinblastine for aggressive/life-threatening disease; nirogacestat [gamma-secretase inhibitor] — the DeFi trial showed a 41% objective response rate and 76% PFS at 2 years, now FDA-approved) → surgery reserved for life-threatening complications (bowel obstruction, ureteral obstruction, mesenteric vessel encasement) or isolated, easily resectable tumors.
GIST — Sarcoma Perspective
GIST is the most common sarcoma of the GI tract (covered in detail in Section 12). From a sarcoma perspective, key points: GISTs are classified separately from other STS because of their distinct biology (KIT/PDGFRA-driven) and response to targeted therapy (imatinib). Unlike other STS, GISTs do not benefit from conventional chemotherapy or radiation therapy. Mutational analysis is essential for treatment planning: KIT exon 11 (most common, best imatinib response), KIT exon 9 (higher imatinib dose — 800 mg, or sunitinib), PDGFRA D842V (avapritinib), and wild-type/SDH-deficient GIST (pediatric type, distinct biology).
20 Thyroid Cancer & Adrenal Tumors
Thyroid Nodule Evaluation — Bethesda System
Thyroid nodules are evaluated by ultrasound (ACR TI-RADS scoring) and FNA biopsy, reported using the Bethesda System for Reporting Thyroid Cytopathology:
| Bethesda Category | Description | Malignancy Risk | Management |
|---|---|---|---|
| I | Non-diagnostic | 5–10% | Repeat FNA in 4–6 weeks |
| II | Benign | 0–3% | Clinical follow-up |
| III | Atypia of undetermined significance (AUS/FLUS) | 10–30% | Repeat FNA or molecular testing (Afirma, ThyroSeq) |
| IV | Follicular neoplasm / suspicious for FN | 25–40% | Diagnostic lobectomy or molecular testing |
| V | Suspicious for malignancy | 50–75% | Lobectomy or total thyroidectomy |
| VI | Malignant | 97–99% | Surgery (lobectomy or total thyroidectomy) |
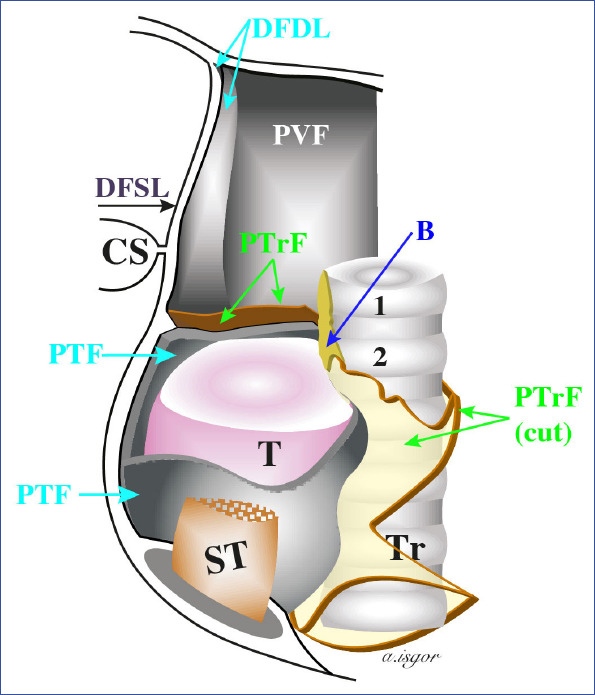
Thyroid Cancer — Surgical Management
Differentiated thyroid cancer (DTC) — papillary (PTC, ~80%) and follicular (FTC, ~10%): Lobectomy is sufficient for low-risk PTC (≤4 cm, no extrathyroidal extension, no nodal metastasis, no vascular invasion, no aggressive histologic variants). Total thyroidectomy is indicated for tumors >4 cm, bilateral disease, extrathyroidal extension, positive nodes, or when RAI therapy is planned. Central neck dissection (level VI) is performed for clinically evident nodal disease; prophylactic central dissection is controversial. Lateral neck dissection (levels II–V) for clinically/radiologically positive lateral nodes.
Medullary thyroid carcinoma (MTC) arises from parafollicular C cells; produces calcitonin and CEA. Surgery: total thyroidectomy + central neck dissection (always). Lateral neck dissection if lateral nodes involved. MTC does not take up radioiodine. ~25% are hereditary (MEN2A, MEN2B, familial MTC) — screen for RET mutations. Vandetanib and cabozantinib are TKIs approved for advanced/metastatic MTC. Selpercatinib and pralsetinib target RET-mutant MTC with high response rates. Anaplastic thyroid carcinoma (ATC) is the most aggressive thyroid cancer with median survival <6 months. Surgery is palliative (rarely R0); consider BRAF/MEK inhibitor combination (dabrafenib + trametinib) if BRAF V600E mutant — response rates ~60%, dramatically improving outcomes in a historically uniformly fatal disease.
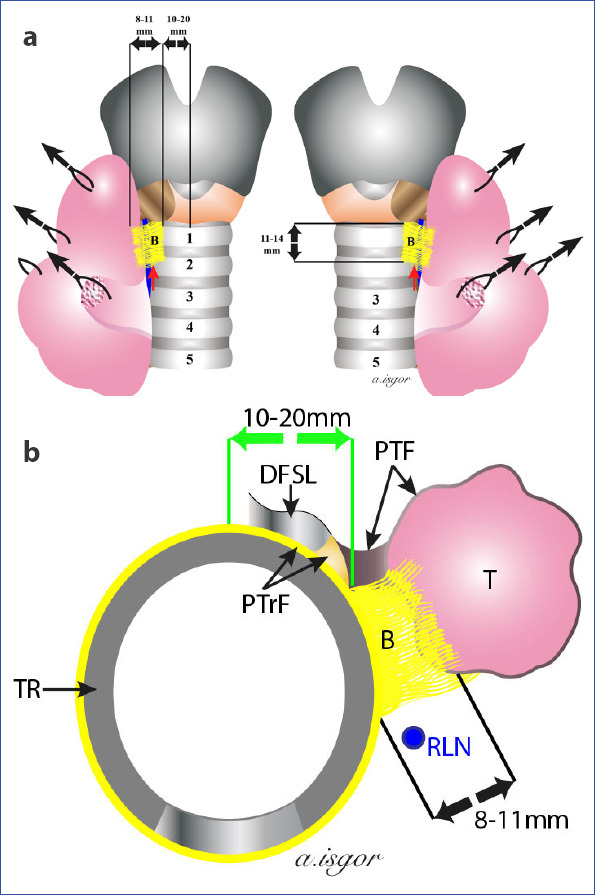
Molecular Testing in Thyroid Nodules
Molecular testing is transforming the management of indeterminate thyroid nodules (Bethesda III/IV). Afirma Genomic Sequencing Classifier (GSC): A "benign" result has a high negative predictive value (~96%) — avoids unnecessary diagnostic lobectomy. ThyroSeq v3: A multi-gene panel that identifies specific mutations (BRAF, RAS, RET/PTC, PAX8/PPARgamma) — both a rule-in and rule-out test. These molecular tests can shift ~50% of indeterminate nodules to "likely benign" or "likely malignant," reducing the need for diagnostic surgery. However, molecular testing does not replace surgical decision-making — results must be interpreted in the clinical and ultrasound context.
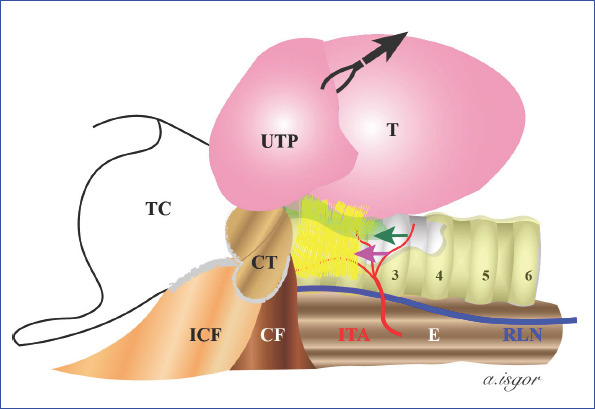
Adrenal Tumors
Adrenal incidentalomas (≥1 cm found incidentally on imaging) require functional workup (cortisol — 1 mg dexamethasone suppression test; catecholamines — plasma metanephrines/normetanephrines; aldosterone/renin ratio if hypertensive) and imaging characterization (Hounsfield units on non-contrast CT: ≤10 HU = likely lipid-rich adenoma; >10 HU = indeterminate → washout CT or MRI). Indications for adrenalectomy: functional tumor, suspected malignancy (size >4–6 cm, heterogeneous appearance, rapid growth), or pheochromocytoma.
Pheochromocytoma: Diagnose with plasma free metanephrines (sensitivity >97%). Preoperative alpha-blockade (phenoxybenzamine or doxazosin) for 10–14 days before surgery is mandatory to prevent hypertensive crisis during manipulation. Beta-blockers added ONLY after adequate alpha-blockade. Liberal salt and fluid intake to expand intravascular volume. Laparoscopic adrenalectomy is standard. Rule of 10s (approximate): 10% bilateral, 10% malignant, 10% extra-adrenal (paraganglioma), 10% pediatric, ~40% hereditary (MEN2, VHL, SDH mutations, NF1).
Adrenocortical carcinoma (ACC): Rare, aggressive tumor. Presents with Cushing syndrome (cortisol-producing, most common), virilization (androgen-producing), feminization, or as a non-functional mass. Typically large at diagnosis (>6 cm). Imaging: heterogeneous, irregular, HU >10 on non-contrast CT, calcifications, necrosis. Surgery: open adrenalectomy (laparoscopic approach is controversial due to risk of capsular violation and peritoneal seeding — generally not recommended for tumors >6 cm or with suspected malignancy). R0 resection is the most important prognostic factor. Adjuvant mitotane is standard for high-risk ACC (Ki-67 >10%, R1 margins, stage III). The FIRM-ACT trial showed cisplatin-based chemotherapy + mitotane for advanced disease. Weiss score ≥3 diagnostic criteria distinguish ACC from adenoma.
Radioactive Iodine (RAI) Therapy — Surgical Oncology Interface
After total thyroidectomy for differentiated thyroid cancer (PTC/FTC), radioactive iodine (I-131) therapy is used for remnant ablation (low-risk), adjuvant therapy (intermediate-risk), and treatment of known residual/metastatic disease (high-risk). RAI requires TSH stimulation — either thyroid hormone withdrawal (allow TSH to rise >30 mIU/L) or recombinant human TSH (Thyrogen) injection. Low-iodine diet for 1–2 weeks before RAI. Post-treatment whole-body scan at 5–7 days identifies iodine-avid metastases. Thyroglobulin (Tg) is the tumor marker for differentiated thyroid cancer post-thyroidectomy — a rising Tg (especially stimulated Tg >2 ng/mL with negative neck US) should prompt investigation for recurrence (CT neck/chest, PET-CT, or diagnostic RAI scan). RAI-refractory disease (progressive disease despite RAI, or loss of RAI avidity) is treated with multikinase inhibitors (lenvatinib — SELECT trial; sorafenib — DECISION trial).
21 Parathyroid Cancer & MEN Syndromes
Parathyroid Carcinoma
Rare (<1% of primary hyperparathyroidism). Suspect when: markedly elevated calcium (>14 mg/dL), very high PTH (>5–10 times normal), palpable neck mass, and renal/skeletal complications. Surgery: en bloc resection (parathyroidectomy with ipsilateral thyroid lobectomy + excision of any adherent tissue — avoid capsular violation/tumor spillage). R0 resection is the only curative treatment. No effective chemotherapy; radiation is of uncertain benefit. Monitor with serial PTH and calcium — rising PTH indicates recurrence.
MEN Syndromes — Surgical Implications
| Syndrome | Gene | Components | Key Surgical Considerations |
|---|---|---|---|
| MEN1 | MEN1 (menin) | Parathyroid hyperplasia (95%), pancreatic NETs (40% — gastrinoma most common), pituitary adenoma (30%) | Subtotal (3.5 gland) parathyroidectomy or total + autotransplant; pancreatic NETs: resect if >2 cm or functional |
| MEN2A | RET proto-oncogene | MTC (95%), pheochromocytoma (50%), primary hyperparathyroidism (20–30%) | Prophylactic thyroidectomy based on RET mutation risk (highest risk: codon 634 → thyroidectomy by age 5; moderate risk → by age 5–10); screen for pheo BEFORE thyroidectomy |
| MEN2B | RET (M918T, most common) | MTC (100%, earliest and most aggressive), pheochromocytoma (50%), mucosal neuromas, marfanoid habitus | Prophylactic thyroidectomy ideally in the first 6 months of life; MTC develops very early |
22 Sentinel Lymph Node Biopsy Technique
Tracers & Mapping Agents
| Agent | Mechanism | Timing | Advantages / Disadvantages |
|---|---|---|---|
| Technetium-99m sulfur colloid | Radiotracer detected by gamma probe | Injected 1–4 hours preoperatively (or day before for lymphoscintigraphy) | Gold standard; allows preoperative lymphoscintigraphy to map drainage basins; requires nuclear medicine facility |
| Isosulfan blue / methylene blue | Visual identification of blue-stained lymphatics and nodes | Injected 5–10 min before incision | Simple, inexpensive; rare anaphylaxis risk (isosulfan ~1%); blue discoloration of skin/urine; interferes with pulse oximetry |
| Indocyanine green (ICG) | Near-infrared fluorescence detected by camera | Injected at time of surgery | Real-time visualization; no radiation; no anaphylaxis; excellent in obese patients; requires NIR camera system |
| Superparamagnetic iron oxide (SPIO / Magtrace) | Magnetic detection with handheld probe | Injected 1–7 days preoperatively | No radiation; flexible timing; brown skin staining at injection site |
SLNB Technique — Step by Step
(1) Injection: Peritumoral or subareolar (breast) / intradermal at the primary site (melanoma). Radiotracer injected preoperatively; blue dye and/or ICG injected intraoperatively. (2) Lymphoscintigraphy (optional, primarily for melanoma): preoperative imaging to identify drainage basins — especially important for trunk melanoma with ambiguous drainage. (3) Incision: Over the axilla (breast) or mapped nodal basin (melanoma). (4) Identification: Use gamma probe to locate "hot" nodes (counts ≥10% of the hottest node ex vivo are considered sentinel); visual identification of blue/fluorescent nodes. (5) Excision: Careful dissection to remove SLN(s) with minimal disruption. (6) Intraoperative assessment: Touch-prep cytology, frozen section, or OSNA depending on institution. (7) Pathologic evaluation: Serial sectioning with H&E and immunohistochemistry (cytokeratin staining for breast; S-100, HMB-45, Melan-A for melanoma).
SLN by Tumor Type
Breast cancer: SLN identification rate >97%; false-negative rate ~5–7%. Inject peritumoral or subareolar (equivalent SLN identification for axillary staging). Melanoma: SLN identification rate ~97%; inject intradermally at the primary site (not the WLE scar). Merkel cell carcinoma: SLNB recommended for tumors >1 cm; SLN-positive in ~30%. Vulvar cancer: GOG 173 validated SLNB for squamous cell carcinoma of the vulva (≥2 cm tumors); negative SLN spares inguinofemoral lymphadenectomy. Penile cancer: Dynamic SLNB for cN0 patients with ≥T1b disease. Colon cancer: Experimental — not standard of care.
- Dual tracer technique: Using both radiotracer AND blue dye (or ICG) increases identification rate to >99% and reduces false-negative rate
- Remove all hot and/or blue nodes: The 10% rule — any node with ≥10% of the hottest node's radioactivity (ex vivo count) should be removed
- Palpate the nodal basin: Remove any clinically suspicious nodes even if not hot/blue
- Verify with back-table counts: Confirm radioactivity of excised nodes; ensure background counts are low before closing
- Avoid prior excision when possible: SLNB is most accurate when performed before or at the same time as wide local excision; prior excision disrupts lymphatic drainage
- Special situations: Prior axillary surgery reduces SLNB accuracy; drainage to internal mammary nodes (breast) or epitrochlear nodes (upper extremity melanoma) should prompt consideration of biopsy at these sites
23 Completion Lymphadenectomy & Lymphedema
When to Perform Completion Lymph Node Dissection (CLND)
| Tumor Type | CLND After Positive SLN? | Key Trial / Evidence |
|---|---|---|
| Breast cancer | Often omitted (Z0011 criteria); axillary RT as alternative (AMAROS) | ACOSOG Z0011, AMAROS |
| Melanoma | No — observation with US surveillance | MSLT-II, DeCOG-SLT |
| Merkel cell | Recommended if SLN positive (adjuvant RT often added) | NCCN guidelines |
| Vulvar cancer | Yes — inguinofemoral lymphadenectomy + adjuvant RT if >1 positive node | GOG 173 |
Lymphedema
Lymphedema is the most significant long-term morbidity of lymph node surgery. Incidence after ALND: 15–25%; after SLNB alone: 5–8%. Risk factors: extent of nodal surgery, adjuvant radiation to nodal basin, obesity, infection (cellulitis), and taxane-based chemotherapy. Staging (International Society of Lymphology): Stage 0 (subclinical — detectable by bioimpedance spectroscopy), Stage I (pitting edema, reversible with elevation), Stage II (non-pitting, fibrotic — does not resolve with elevation), Stage III (lymphostatic elephantiasis).
Prevention: SLNB instead of ALND when oncologically safe; axillary reverse mapping (ARM — identifies and preserves arm-draining lymphatics during ALND); prospective surveillance with bioimpedance spectroscopy or perometry to detect subclinical lymphedema early. Treatment: Complete decongestive therapy (CDT) — manual lymphatic drainage (MLD), compression bandaging, exercise, skin care. Compression garments for maintenance. Surgical options for refractory cases: lymphovenous anastomosis (LVA — supermicrosurgery connecting lymphatic vessels to venules), vascularized lymph node transfer (VLNT), and suction-assisted protein lipectomy (SAPL) for fibrofatty chronic lymphedema.
Lymph Node Sampling Strategies by Tumor Type
| Tumor Type | Nodal Assessment Strategy | Minimum Node Count / Targets |
|---|---|---|
| Breast | SLNB for cN0; ALND or axillary RT for cN+ (post-NAC: TAD) | ≥2–3 SLN examined; ALND includes Level I–II (typically 10–20 nodes) |
| Melanoma | SLNB for ≥0.8 mm Breslow; CLND no longer routine if SLN+ | At least 1 SLN; CLND yield varies by basin (inguinal ~10, axillary ~15–20, cervical ~15–30) |
| Colorectal | En bloc mesenteric lymphadenectomy with high vascular ligation | ≥12 nodes (AJCC minimum for adequate staging); CME may yield 30–40 nodes |
| Gastric | D2 lymphadenectomy (stations 1–12) | ≥16 nodes examined (AJCC); D2 typically yields 25–40 nodes |
| Esophageal | Two-field (mediastinal + abdominal) or three-field (+ cervical) | ≥15 nodes recommended (AJCC) |
| Pancreatic | Standard lymphadenectomy along peripancreatic vessels | ≥15 nodes examined (AJCC); extended lymphadenectomy not beneficial (Johns Hopkins RCT) |
| Thyroid (DTC) | Central dissection (level VI) for clinically evident nodes; lateral (levels II–V) if clinically positive | Variable; prophylactic central dissection debated for PTC |
24 Margin Assessment & Intraoperative Pathology
Frozen Section Analysis
Intraoperative frozen section provides rapid (~20 min) histologic assessment of surgical margins or tissue identification. The tissue is snap-frozen (cryostat at −20 to −30°C), sectioned (5–10 micrometers), and stained with H&E. Advantages: immediate margin assessment allows re-excision during the same operation. Limitations: freezing artifact degrades tissue quality; sampling error (only a small portion of the margin is examined); less accurate for lobular breast carcinoma, DCIS, and fatty tissue. Accuracy: ~95–98% for most tumors, lower for breast (especially DCIS). Final (permanent) sections always supersede frozen section results.
Touch-Prep / Imprint Cytology
The cut surface of excised tissue is gently pressed onto a glass slide, air-dried or fixed, and stained (Diff-Quik or H&E). Faster than frozen section (~5 min) and does not consume tissue. Used primarily for SLN assessment (particularly in breast cancer SLNB) and rapid intraoperative consultations. Sensitivity for detecting SLN metastasis: ~60–70% (better for macrometastases, poor for micrometastases and ITC).
Specimen Orientation & Handling
Proper specimen orientation is essential for accurate margin assessment. Lumpectomy specimens should be oriented with sutures (e.g., short suture = superior, long suture = lateral) or clips, and the surgeon should communicate the orientation to pathology. Specimens should NOT be bivalved by the surgeon — send intact to pathology for inking and sectioning. For mastectomy specimens, the pathologist should identify the closest margin and report the distance of tumor from each inked margin.
Cavity Shave Margins
The cavity shave margin (CSM) technique involves excising additional tissue from all six walls (anterior, posterior, superior, inferior, medial, lateral) of the lumpectomy cavity after the primary specimen is removed. CSMs are sent as separate, oriented specimens. Studies have shown that routine CSMs significantly reduce positive margin rates (from ~30% to ~10%) and re-excision rates after lumpectomy. This technique adds minimal operative time and tissue volume while substantially decreasing the need for second surgeries. CSMs are now widely adopted as standard practice during BCS.
Intraoperative Specimen Radiography
For non-palpable lesions localized by wire, radioactive seed (RSL), magnetic seed (Magseed), or radar reflector (SAVI SCOUT), intraoperative specimen radiography confirms that the target lesion (microcalcifications, clip, or localization device) is contained within the excised specimen. The specimen is sent for radiography immediately after excision — if the target is absent or at the specimen edge, additional tissue is excised. This step is essential for ensuring adequate excision of non-palpable breast lesions and reduces the need for re-operation.
Intraoperative Ultrasound (IOUS)
IOUS is invaluable in hepatobiliary oncologic surgery for: identifying additional liver metastases missed on preoperative imaging (changes operative plan in ~10–15% of cases), guiding parenchymal transection planes, identifying vascular structures (hepatic veins, portal pedicles), and guiding ablation probe placement. A high-frequency (7.5–10 MHz) linear or T-shaped probe provides excellent resolution for lesions within the hepatic parenchyma.
25 Ablation, HIPEC, & Minimally Invasive Oncologic Surgery
Ablation Techniques
| Technique | Mechanism | Best For | Key Considerations |
|---|---|---|---|
| RFA | Alternating current → ionic agitation → frictional heat (60–100°C) | HCC ≤3 cm, CRLM ≤3 cm, renal tumors | Heat-sink effect near vessels >3 mm reduces efficacy; multiple overlapping ablations needed for >3 cm tumors |
| MWA | Electromagnetic waves → water molecule rotation → heat | HCC, CRLM, lung tumors | Faster, higher temperatures, larger zones than RFA; less affected by heat-sink; preferred for tumors near vessels |
| Cryoablation | Argon gas → Joule-Thomson effect → ice ball (−40°C) | Renal tumors, lung, bone mets, breast (investigational) | Visible ice ball on imaging; risk of cryoshock (hemodynamic instability, coagulopathy) with large ablations |
| IRE (NanoKnife) | Short high-voltage DC pulses → nanopore formation → cell death | Pancreatic tumors near vessels, hilar CCA | Non-thermal → preserves adjacent bile ducts and vessels; requires general anesthesia with cardiac synchronization |
HIPEC Technique
After maximum cytoreductive surgery (CRS), the abdomen is filled with heated (41–43°C) chemotherapy solution and circulated for 60–90 minutes via inflow/outflow catheters. Two techniques: open (Coliseum) technique — abdomen held open with a retractor, chemotherapy solution circulated with the viscera exposed (better distribution, temperature monitoring) vs closed technique — abdomen closed temporarily with running suture (less heat loss, less environmental contamination). Common agents: mitomycin C (CRC, appendiceal), cisplatin (ovarian, mesothelioma), oxaliplatin (CRC — used in the PRODIGE 7 trial). Staff safety requires appropriate PPE (chemotherapy-rated gowns, double gloving, face shields).
Minimally Invasive Oncologic Surgery
Laparoscopic colectomy for colorectal cancer: COST, COLOR, and CLASICC trials confirmed equivalent oncologic outcomes (lymph node harvest, margins, survival) with benefits of reduced pain, shorter LOS, and faster recovery. Robotic surgery (da Vinci platform): increasingly used for rectal cancer (TME), pancreatic surgery, hepatectomy, and complex pelvic exenteration — advantages include 3D visualization, wristed instruments, tremor filtration. Laparoscopic/robotic gastrectomy: non-inferior to open for distal gastrectomy in early gastric cancer (KLASS-01, JCOG0912 trials). MIE (minimally invasive esophagectomy): TIME trial showed reduced pulmonary complications.
Port Placement & Venous Access
Implantable venous access ports (port-a-cath) are placed for long-term chemotherapy administration. Preferred veins: internal jugular (right-sided preferred — straighter path to the SVC), subclavian, or cephalic vein (cut-down technique — lower pneumothorax risk). Tip position: confirmed fluoroscopically at the cavoatrial junction. Complications: pneumothorax (subclavian approach ~1–3%), catheter malposition, port pocket infection, catheter thrombosis, pinch-off syndrome (catheter compression between the clavicle and first rib — use of the lateral subclavian approach reduces this risk), and catheter fracture/embolization.
Image-Guided Surgery & Emerging Technologies
Intraoperative fluorescence imaging (ICG): ICG binds to plasma proteins and fluoresces under near-infrared light. Applications: assessment of bowel/flap/anastomotic perfusion (reduced anastomotic leak rates), hepatobiliary anatomy visualization (biliary tree maps after IV injection), SLN mapping, and tumor identification (ICG accumulates in HCC and liver metastases). Intraoperative navigation: CT-based stereotactic navigation systems are used for hepatic tumor localization (image-guided liver surgery, IGLS), particularly for small, deep, non-palpable lesions. Intraoperative radiation therapy (IORT): Single-dose radiation delivered directly to the tumor bed during surgery — used in selected breast cancer (TARGIT-A trial), pancreatic, and rectal cancer. Intraoperative CT/MRI: Used primarily in neurosurgical oncology for real-time resection assessment.
Oncologic Emergency Procedures
Surgical oncologists manage several oncologic emergencies:
| Emergency | Tumor Type | Management |
|---|---|---|
| Malignant bowel obstruction | CRC, ovarian, peritoneal carcinomatosis | Resection if feasible; bypass or diverting ostomy; endoscopic stenting for palliation; octreotide + dexamethasone for inoperable |
| Tumor perforation | CRC, lymphoma, GIST | Emergent resection; Hartmann's procedure if unstable; primary anastomosis if stable and favorable conditions |
| Hemorrhage from tumor | GI tumors, hepatic tumors | Endoscopic hemostasis; angioembolization (IR); surgical resection if other measures fail |
| Obstructive jaundice (malignant) | Pancreatic head, cholangiocarcinoma, periampullary | ERCP with stent (metal for palliation, plastic if surgery planned); PTC if ERCP fails |
| Superior vena cava syndrome | Lung cancer, lymphoma, thymoma | Endovascular stenting for acute symptoms; definitive treatment of underlying malignancy; corticosteroids if lymphoma suspected |
| Tumor-related sepsis (post-biopsy, post-TACE) | HCC, liver mets | Broad-spectrum antibiotics; percutaneous drainage of infected necrotic tumor; supportive care |
26 Neoadjuvant & Adjuvant Chemotherapy
Neoadjuvant Regimens by Tumor Type
| Tumor | Regimen | Key Trial |
|---|---|---|
| Breast (HER2+) | TCHP (docetaxel, carboplatin, trastuzumab, pertuzumab) | NeoSphere, TRYPHAENA |
| Breast (TNBC) | AC-T + pembrolizumab (anthracycline, cyclophosphamide → paclitaxel + pembrolizumab) | KEYNOTE-522 |
| Esophageal / GEJ | Carboplatin + paclitaxel + 41.4 Gy RT | CROSS |
| Gastric / GEJ | FLOT (5-FU, leucovorin, oxaliplatin, docetaxel) x4 pre-op, x4 post-op | FLOT4-AIO |
| Rectal | Total neoadjuvant therapy (TNT): mFOLFOX6 → CRT (5-FU + 50.4 Gy) OR FOLFOX → short-course RT | RAPIDO, PRODIGE 23, OPRA |
| Pancreatic (borderline) | Modified FOLFIRINOX or gemcitabine + nab-paclitaxel | Alliance A021501 |
Adjuvant Chemotherapy Principles
Adjuvant therapy targets micrometastatic disease remaining after curative surgery. The benefit depends on the absolute recurrence risk (higher risk = greater absolute benefit from chemotherapy) and the chemosensitivity of the tumor. Key principles: (1) Timing — adjuvant chemotherapy should begin within 4–8 weeks of surgery (delays beyond 8–12 weeks are associated with worse outcomes). (2) Duration — varies by regimen (typically 3–6 months). (3) Assessment — adjuvant therapy is empiric (no measurable disease to assess response); the decision is based on pathologic risk factors (stage, grade, margins, molecular features).
Adjuvant Regimens by Tumor Type
Colon cancer (stage III): CAPOX (capecitabine + oxaliplatin) for 3 months (low-risk: T1–3, N1) or 6 months (high-risk: T4 or N2) — IDEA collaboration (PMID: 29527974). Pancreatic (resected): Modified FOLFIRINOX for 6 months (PRODIGE 24, PMID: 30575490). Breast (HR+/HER2−): Guided by Oncotype DX recurrence score; endocrine therapy for all; chemotherapy if high recurrence score. Gastric: Perioperative FLOT (pre-op + post-op). Melanoma (stage III): Nivolumab (CheckMate 238) or pembrolizumab (KEYNOTE-054) for 1 year.
Adjuvant Therapy for Special Situations
Stage II colon cancer: The benefit of adjuvant chemotherapy in stage II disease is modest (~3–5% absolute OS benefit). Use is guided by high-risk features: T4 tumor, poorly differentiated histology, lymphovascular invasion, perineural invasion, bowel obstruction/perforation at presentation, positive margins, or <12 lymph nodes examined. MSI-high stage II colon cancer has an excellent prognosis and does NOT benefit from 5-FU-based adjuvant chemotherapy — surveillance alone is recommended. Residual disease after neoadjuvant (breast): CREATE-X trial — HER2-negative patients with residual invasive disease after NAC receive adjuvant capecitabine (6–8 cycles). KATHERINE trial — HER2-positive patients with residual disease switch from trastuzumab to T-DM1 for 14 cycles. dMMR/MSI-high rectal cancer: The landmark MSKCC study (Cercek et al., 2022) showed 100% clinical complete response with dostarlimab (anti-PD-1) for dMMR locally advanced rectal cancer, potentially eliminating the need for surgery and radiation — paradigm-shifting, albeit in a small cohort requiring validation.
| Agent | Key Toxicity | Monitoring / Prevention |
|---|---|---|
| Doxorubicin | Cardiotoxicity (dilated cardiomyopathy) | LVEF (echo/MUGA) at baseline, q3 months; lifetime cumulative dose <450 mg/m2; dexrazoxane cardioprotection |
| Oxaliplatin | Peripheral neuropathy (cumulative, dose-limiting) | Grade 2+ neuropathy → dose reduce or discontinue; cold avoidance during infusion; "stop-and-go" strategies |
| 5-FU | DPD deficiency → fatal toxicity | DPD genotyping before treatment recommended in some guidelines; 5% of population heterozygous for DPYD variants |
| Irinotecan | Severe diarrhea (UGT1A1*28 homozygosity) | UGT1A1 genotyping before treatment; loperamide for late diarrhea; atropine for early cholinergic diarrhea |
| Trastuzumab | Cardiotoxicity (usually reversible) | LVEF monitoring q3 months; hold if LVEF drops >10% or below LLN; do not give concurrently with anthracyclines |
| Bevacizumab | GI perforation (1–2%), wound healing impairment | Hold 4–6 weeks pre-/post-surgery; monitor BP; urine protein |
27 Targeted Therapy, Immunotherapy, & Hormonal Therapy
Targeted Therapies
| Agent | Target | Indication | Key Side Effects |
|---|---|---|---|
| Imatinib (Gleevec) | KIT, PDGFRA, BCR-ABL | GIST (adjuvant, neoadjuvant, metastatic); CML; DFSP | Edema, diarrhea, nausea, muscle cramps; hepatotoxicity |
| Trastuzumab (Herceptin) | HER2 | HER2+ breast and gastric cancer | Cardiotoxicity (monitor LVEF q3 months); infusion reactions |
| Bevacizumab (Avastin) | VEGF | CRC, HCC (with atezo), ovarian, cervical, renal | Hypertension, proteinuria, bleeding, GI perforation, impaired wound healing — hold 4–6 weeks before/after surgery |
| Cetuximab / Panitumumab | EGFR | RAS wild-type metastatic CRC | Acneiform rash (correlates with response), hypomagnesemia, infusion reactions |
| Sorafenib / Lenvatinib | Multi-kinase (VEGFR, RAF) | HCC, DTC (RAI-refractory), desmoid tumors | Hand-foot syndrome, diarrhea, hypertension, hepatotoxicity |
| Sunitinib | Multi-kinase (VEGFR, KIT, PDGFR) | Imatinib-resistant GIST, RCC, pNET | Hand-foot syndrome, hypothyroidism, cardiotoxicity |
Immunotherapy (Checkpoint Inhibitors)
| Agent | Target | Key Indications in Surgical Oncology |
|---|---|---|
| Pembrolizumab (Keytruda) | PD-1 | Melanoma (adj/neoadj), MSI-high CRC, TNBC (neoadj), Merkel cell, gastric (PD-L1+), esophageal |
| Nivolumab (Opdivo) | PD-1 | Melanoma (adjuvant), esophageal (adj — CheckMate 577), HCC (+ ipilimumab), MSI-high CRC |
| Ipilimumab (Yervoy) | CTLA-4 | Melanoma (often combined with nivolumab); HCC (with nivolumab) |
| Atezolizumab (Tecentriq) | PD-L1 | HCC (+ bevacizumab — IMbrave150); TNBC (with nab-paclitaxel) |
| Avelumab (Bavencio) | PD-L1 | Merkel cell carcinoma, urothelial (maintenance) |
| Dostarlimab | PD-1 | dMMR rectal cancer — 100% clinical complete response in MSKCC study (investigational) |
Checkpoint inhibitors can cause autoimmune toxicity in any organ. Most common: colitis (diarrhea — hold therapy, steroids), hepatitis (elevated transaminases — hold therapy, steroids), pneumonitis (dyspnea, cough — may be life-threatening), endocrinopathies (thyroiditis, hypophysitis, adrenal insufficiency, type 1 diabetes), dermatitis (rash, vitiligo), and nephritis. Management: grade 1 — monitor; grade 2 — hold therapy, consider steroids; grade 3–4 — hold/discontinue therapy, high-dose steroids (methylprednisolone 1–2 mg/kg), consider infliximab for steroid-refractory colitis. Surgical teams must be aware of irAEs mimicking acute surgical conditions (e.g., immune-mediated colitis mimicking perforation).
Hormonal Therapy
Tamoxifen: Selective estrogen receptor modulator (SERM). Standard for premenopausal ER+ breast cancer (5–10 years). Side effects: hot flashes, increased risk of endometrial cancer (2–7x), DVT/PE. Aromatase inhibitors (AIs): Letrozole, anastrozole, exemestane. Block peripheral estrogen synthesis. Standard for postmenopausal ER+ breast cancer (or premenopausal with ovarian suppression — SOFT/TEXT trials). Side effects: musculoskeletal pain, osteoporosis (monitor DEXA), lipid changes. Ovarian suppression: GnRH agonists (goserelin/leuprolide) or bilateral salpingo-oophorectomy — combined with AI for high-risk premenopausal patients. Extended adjuvant endocrine therapy (10 years total) improves outcomes in node-positive disease (MA.17R trial).
28 Staging Systems Master Table
| System | Application | Key Components |
|---|---|---|
| AJCC TNM (8th ed.) | All solid tumors | T (primary tumor), N (nodes), M (metastasis) → Stage I–IV; site-specific criteria |
| BCLC | HCC | Stages 0, A, B, C, D; integrates tumor burden, liver function (Child-Pugh), performance status |
| Bismuth-Corlette | Perihilar cholangiocarcinoma | Types I, II, IIIa, IIIb, IV based on extent of biliary involvement |
| PCI (Sugarbaker) | Peritoneal surface malignancies | 13 abdominal regions scored 0–3; total 0–39; guides CRS/HIPEC candidacy |
| FNCLCC | Soft tissue sarcoma grading | Differentiation + mitotic count + necrosis → Grade 1–3 |
| Bethesda System | Thyroid FNA cytology | Categories I–VI with associated malignancy risk and management |
| BI-RADS | Breast imaging | Categories 0–6 with associated cancer risk and recommended action |
| Miettinen / NIH | GIST risk stratification | Size + mitotic rate + location → very low, low, intermediate, high risk |
| Fong CRS | Colorectal liver metastases | 5 clinical factors (0–5 points); predicts 5-year survival after hepatectomy |
| ISGPF | Pancreatic fistula | Grades A (biochemical leak), B (requires intervention), C (organ failure/death) |
| Breslow / Clark | Melanoma | Breslow: depth in mm; Clark: anatomic level I–V |
| WHO NET Grading | Neuroendocrine tumors | G1 (<3% Ki-67), G2 (3–20%), G3 (>20%); well-differentiated NET vs poorly differentiated NEC |
| Lauren | Gastric cancer | Intestinal (glandular, better prognosis) vs diffuse (signet ring, worse prognosis) |
| Clavien-Dindo | Surgical complications | Grade I–V based on intervention required (see Section 29) |
| ECOG Performance Status | All cancers | 0 = fully active; 1 = restricted strenuous; 2 = ambulatory but unable to work; 3 = limited self-care; 4 = bedbound; 5 = dead |
| Karnofsky Performance Scale | All cancers | 100% = normal; 80% = normal activity with effort; 50% = requires considerable assistance; 0% = dead |
Surveillance Schedules After Curative Resection
| Tumor Type | Surveillance Protocol (NCCN Summary) |
|---|---|
| Breast cancer | History/physical q3–6 months for 5 years, then annually; annual mammogram (ipsilateral if BCS); no routine labs/imaging for asymptomatic patients (except mammogram) |
| Colorectal cancer (stage II–III) | CEA q3–6 months x 5 years; CT CAP annually x 5 years; colonoscopy at 1 year, then q3–5 years |
| Melanoma (stage IIB–IV resected) | History/physical + skin exam q3–6 months x 3 years, then q6–12 months; CT/PET-CT q3–12 months based on stage; brain MRI if high risk |
| Pancreatic cancer (resected) | CA 19-9 + CT q3–6 months x 2 years, then q6–12 months; generally poor prognosis — median recurrence at 12 months |
| GIST (resected, high-risk) | CT abdomen/pelvis q3–6 months x 5 years on imatinib, then q6 months off imatinib; no role for PET in surveillance |
| Soft tissue sarcoma | Physical exam + chest imaging (CT or CXR) q3–6 months x 2–3 years, then q6–12 months; MRI of resection site for high-risk |
29 Complications & Management
Surgical Complications in Oncologic Surgery
| Complication | Incidence | Prevention / Management |
|---|---|---|
| Surgical site infection (SSI) | 5–15% (varies by wound class) | Prophylactic antibiotics within 60 min of incision; normothermia; glycemic control; wound opening and drainage if infected |
| Anastomotic leak | Colorectal 3–10%; esophageal 5–15%; pancreaticojejunostomy 10–20% | Tension-free, well-vascularized anastomosis; ICG angiography; defunctioning stoma for low rectal; NPO, drainage, antibiotics, and reoperation if needed |
| Postoperative pancreatic fistula | 10–30% after Whipple | Duct-to-mucosa pancreaticojejunostomy; drain amylase monitoring; octreotide (evidence equivocal); CT-guided drainage for collections |
| Bile leak | 5–10% after hepatectomy | Intraoperative leak test; drain monitoring (bilirubin in drain fluid); ERCP with stent for major leaks; percutaneous drainage of bilomas |
| Post-hepatectomy liver failure | 1–5% (major hepatectomy) | Adequate FLR (≥20–40%); PVE if needed; ICG clearance assessment; supportive care, MARS/transplant in severe cases |
| Lymphedema | 15–25% after ALND; 5–8% after SLNB | SLNB when possible; prospective surveillance; CDT; compression garments; LVA for refractory cases |
| Chyle leak | 1–3% after neck/thoracic dissection | NPO or medium-chain triglyceride diet; TPN if high output; thoracic duct ligation/embolization if persistent |
| Wound dehiscence | 1–3% midline laparotomy | 4:1 suture-to-wound-length ratio; small bites (STITCH trial); nutrition optimization; manage fascial evisceration emergently |
| VTE (DVT/PE) | 3–10% in oncologic surgery without prophylaxis | LMWH or UFH perioperatively; extended thromboprophylaxis 4 weeks post-op for major abdominal/pelvic cancer surgery (ENOXACAN II trial) |
Clavien-Dindo Classification
| Grade | Definition |
|---|---|
| I | Any deviation from normal course; no pharmacological, surgical, endoscopic, or radiologic intervention |
| II | Requires pharmacological treatment (including blood transfusion, TPN) |
| IIIa | Requires intervention NOT under general anesthesia |
| IIIb | Requires intervention UNDER general anesthesia |
| IVa | Single-organ dysfunction (including ICU management) |
| IVb | Multi-organ dysfunction |
| V | Death |
Oncologic Surgery-Specific Complications
Tumor lysis syndrome (TLS): Rare in solid tumors but can occur with bulky, rapidly responsive tumors (lymphoma, germ cell) or after hepatic artery embolization for large liver tumors. Features: hyperkalemia, hyperphosphatemia, hyperuricemia, hypocalcemia → acute renal failure. Prevention: aggressive hydration, allopurinol or rasburicase. Seroma: Very common after mastectomy + ALND (prevalence 30–50%). Managed with serial aspiration; surgical drains reduce but do not eliminate seromas. Phantom breast pain: Neuropathic pain perceived in the absent breast after mastectomy (30–50% of patients). Managed with gabapentin/pregabalin, duloxetine, or topical therapies. Dumping syndrome: Occurs after gastrectomy — early dumping (osmotic fluid shift into bowel lumen → vasomotor symptoms) and late dumping (reactive hypoglycemia). Management: dietary modification (small, frequent meals; avoid simple carbohydrates; separate liquids from solids); octreotide for refractory cases.
Oncologic Wound Healing Considerations
Cancer patients have impaired wound healing due to: malnutrition (cancer cachexia — affects 50–80% of advanced cancer patients), prior radiation (fibrosis, poor vascularity), immunosuppression (from chemotherapy or the disease itself), and corticosteroid use. Timing of surgery relative to chemotherapy: allow 3–4 weeks after last chemotherapy cycle before elective surgery (adequate bone marrow recovery — ANC >1500, platelets >100,000). After surgery, chemotherapy should be delayed 3–4 weeks to allow wound healing. Bevacizumab requires a 4–6 week washout period before and after surgery due to impaired angiogenesis.
Extended VTE Prophylaxis
Cancer patients undergoing major abdominal/pelvic surgery have a 2–3x higher risk of VTE compared to non-cancer patients. The ENOXACAN II trial demonstrated that extended thromboprophylaxis with LMWH for 4 weeks after major abdominal cancer surgery significantly reduces VTE compared to 1 week of prophylaxis (4.8% vs 12%). This 4-week prophylaxis is now standard of care for major oncologic abdominal surgery. Risk factors for VTE in cancer: active malignancy (Khorana score), major surgery, prolonged immobilization, central venous catheters, and chemotherapy (especially platinum-based and anti-angiogenic agents).
30 Key Landmark Trials
| Trial | Tumor | Question | Key Finding |
|---|---|---|---|
| NSABP B-06 | Breast | Lumpectomy + RT vs mastectomy | Equivalent OS at 20 years; BCS became standard of care |
| ACOSOG Z0011 | Breast | SLNB alone vs ALND for 1–2 positive SLN | No OS or DFS difference; ALND can be omitted (with BCS + RT) |
| AMAROS | Breast | Axillary RT vs ALND for positive SLN | Equivalent regional control; less lymphedema with RT |
| CROSS | Esophageal | Neoadjuvant CRT + surgery vs surgery alone | Median OS 49 vs 24 months; neoadjuvant CRT is standard |
| MAGIC | Gastric | Perioperative ECF chemo vs surgery alone | 5-yr OS 36% vs 23%; perioperative chemo became standard |
| FLOT4 | Gastric / GEJ | FLOT vs ECF/ECX perioperative | FLOT superior: median OS 50 vs 35 months |
| PRODIGE 24 | Pancreatic | Adjuvant mFOLFIRINOX vs gemcitabine | mFOLFIRINOX superior: median OS 54 vs 35 months |
| MSLT-I | Melanoma | SLNB as staging procedure | SLNB provides critical prognostic information; DFS benefit for SLN+ with early CLND |
| MSLT-II | Melanoma | CLND vs observation after positive SLN | No melanoma-specific survival benefit from CLND |
| SSG XVIII/AIO | GIST | 3 years vs 1 year adjuvant imatinib | 3 years superior: 5-yr OS 92% vs 82% for high-risk GIST |
| STRASS | RPS | Preoperative RT + surgery vs surgery alone | No overall benefit; possible benefit in liposarcoma subgroup |
| PRODIGE 7 | CRC peritoneal | CRS + HIPEC vs CRS alone | No OS benefit from HIPEC addition; CRS alone may suffice for CRC |
| Milan Criteria | HCC | Transplant criteria for HCC | Single ≤5 cm or ≤3 tumors ≤3 cm: 5-yr survival >70% |
| IMbrave150 | HCC | Atezo + bev vs sorafenib for advanced HCC | Superior OS and PFS; new first-line standard for advanced HCC |
| IDEA | Colon (stage III) | 3 vs 6 months adjuvant oxaliplatin | 3 months non-inferior for low-risk (T1–3N1) with CAPOX |
| KEYNOTE-522 | Breast (TNBC) | Neoadjuvant pembro + chemo vs chemo alone | Higher pCR and improved EFS with pembrolizumab |
31 Medications Master Table
| Drug | Class | Mechanism | Primary Indications | Key Toxicity |
|---|---|---|---|---|
| 5-Fluorouracil (5-FU) | Antimetabolite | Thymidylate synthase inhibitor | CRC, gastric, esophageal, anal, breast | Mucositis, diarrhea, hand-foot syndrome; DPD deficiency → fatal toxicity |
| Capecitabine (Xeloda) | Antimetabolite (oral 5-FU prodrug) | Converted to 5-FU in tumor tissue | CRC (CAPOX), breast (CREATE-X) | Hand-foot syndrome, diarrhea |
| Oxaliplatin | Platinum | DNA crosslinking | CRC (FOLFOX, CAPOX), gastric (FLOT) | Peripheral neuropathy (dose-limiting, cumulative), cold sensitivity |
| Irinotecan | Topoisomerase I inhibitor | Prevents DNA unwinding | CRC (FOLFIRI, FOLFIRINOX) | Diarrhea (early cholinergic; late — dose-limiting), myelosuppression; UGT1A1*28 polymorphism |
| Docetaxel | Taxane | Microtubule stabilization | Breast, gastric (FLOT), prostate | Neutropenia, fluid retention, neuropathy, nail changes |
| Paclitaxel | Taxane | Microtubule stabilization | Breast, ovarian, esophageal (CROSS) | Neuropathy, alopecia, hypersensitivity (cremophor formulation) |
| Gemcitabine | Antimetabolite | Nucleoside analog | Pancreatic, biliary | Myelosuppression, flu-like symptoms, pulmonary toxicity (rare) |
| Doxorubicin | Anthracycline | Topoisomerase II inhibitor, DNA intercalation | Breast (AC), STS, hepatocellular (TACE) | Cardiotoxicity (cumulative, max ~450 mg/m2), myelosuppression, alopecia |
| Mitomycin C | Alkylating agent | DNA crosslinking | Anal (Nigro protocol), HIPEC (CRC, appendiceal) | Myelosuppression (delayed), HUS (rare) |
| Tamoxifen | SERM | Competitive ER blockade | ER+ breast (premenopausal) | Hot flashes, endometrial cancer (2–7x risk), DVT/PE |
| Letrozole / Anastrozole | Aromatase inhibitor | Blocks estrogen synthesis | ER+ breast (postmenopausal) | Arthralgia, osteoporosis, lipid changes |
| Trastuzumab | Monoclonal antibody | Anti-HER2 | HER2+ breast, HER2+ gastric | Cardiotoxicity (monitor LVEF); generally well-tolerated |
| Pembrolizumab | Checkpoint inhibitor | Anti-PD-1 | Melanoma, MSI-high, TNBC, esophageal, gastric, Merkel | irAEs (see Section 27) |
| Nivolumab | Checkpoint inhibitor | Anti-PD-1 | Melanoma (adjuvant), esophageal (adjuvant — CM577), HCC | irAEs |
| Imatinib | TKI | KIT / PDGFRA / BCR-ABL inhibitor | GIST, CML, DFSP | Edema, nausea, diarrhea, muscle cramps |
| Bevacizumab | Monoclonal antibody | Anti-VEGF | CRC, HCC, ovarian | HTN, proteinuria, GI perforation, bleeding, wound healing impairment |
- Bevacizumab: Hold for 4–6 weeks before elective surgery; 4 weeks after major surgery before restarting
- Checkpoint inhibitors: No strict hold period required; coordinate with medical oncology; be aware of irAEs mimicking surgical emergencies
- Tamoxifen: Consider holding 2–4 weeks before major surgery (VTE risk) — weigh against cancer recurrence risk
- Imatinib: May be continued perioperatively for GIST; some surgeons hold 1–2 days pre-op
- Anticoagulation: See BRIDGE trial (routine bridging NOT recommended for AF); for cancer-associated VTE on LMWH, consider holding 24 hrs pre-op
32 Abbreviations & References
Abbreviations
| Abbreviation | Meaning |
|---|---|
| AJCC | American Joint Committee on Cancer |
| ALND | Axillary lymph node dissection |
| APR | Abdominoperineal resection |
| BCS | Breast-conserving surgery |
| BCLC | Barcelona Clinic Liver Cancer |
| BI-RADS | Breast Imaging Reporting and Data System |
| CAPOX | Capecitabine + oxaliplatin |
| CEA | Carcinoembryonic antigen |
| CLND | Completion lymph node dissection |
| CME | Complete mesocolic excision |
| CRC | Colorectal cancer |
| CRLM | Colorectal liver metastases |
| CRS | Cytoreductive surgery |
| CRT | Chemoradiation therapy |
| DCIS | Ductal carcinoma in situ |
| DFS | Disease-free survival |
| dMMR | Deficient mismatch repair |
| ERAS | Enhanced recovery after surgery |
| FLR | Future liver remnant |
| FLOT | 5-FU, leucovorin, oxaliplatin, docetaxel |
| FNCLCC | Federation Nationale des Centres de Lutte Contre le Cancer |
| GIST | Gastrointestinal stromal tumor |
| GEJ | Gastroesophageal junction |
| HCC | Hepatocellular carcinoma |
| HIPEC | Hyperthermic intraperitoneal chemotherapy |
| IBC | Inflammatory breast cancer |
| ICG | Indocyanine green |
| IOUS | Intraoperative ultrasound |
| irAE | Immune-related adverse event |
| IRE | Irreversible electroporation |
| LAR | Low anterior resection |
| MDT | Multidisciplinary team |
| MIE | Minimally invasive esophagectomy |
| MRM | Modified radical mastectomy |
| MSI | Microsatellite instability |
| MTC | Medullary thyroid carcinoma |
| MWA | Microwave ablation |
| NAC | Neoadjuvant chemotherapy |
| NET | Neuroendocrine tumor |
| NSM | Nipple-sparing mastectomy |
| OS | Overall survival |
| PCI | Peritoneal cancer index |
| pCR | Pathologic complete response |
| PDAC | Pancreatic ductal adenocarcinoma |
| POPF | Postoperative pancreatic fistula |
| PVE | Portal vein embolization |
| RAI | Radioactive iodine |
| RFA | Radiofrequency ablation |
| RPS | Retroperitoneal sarcoma |
| SLN / SLNB | Sentinel lymph node / biopsy |
| SSM | Skin-sparing mastectomy |
| STS | Soft tissue sarcoma |
| TACE | Transarterial chemoembolization |
| TME | Total mesorectal excision |
| TNBC | Triple-negative breast cancer |
| TNM | Tumor-Node-Metastasis |
| TNT | Total neoadjuvant therapy |
| VEGF | Vascular endothelial growth factor |
| VTE | Venous thromboembolism |
| WLE | Wide local excision |
Figures
- Figure 1 — Breast Anatomy. Wikimedia Commons. Public domain.
- Figure 2 — Pancreaticoduodenectomy (Whipple Procedure). Wikimedia Commons. Public domain.
- Figure 3 — Thyroid and Parathyroid Anatomy. Wikimedia Commons. Public domain.
Key Trials & Guidelines
- Hanahan D, Weinberg RA. The hallmarks of cancer. Cell. 2000;100(1):57-70. PMID: 10647931
- Fisher B, Anderson S, Bryant J, et al. Twenty-year follow-up of a randomized trial comparing total mastectomy, lumpectomy, and lumpectomy plus irradiation (NSABP B-06). N Engl J Med. 2002;347(16):1233-1241. PMID: 12065600
- Giuliano AE, Ballman KV, McCall L, et al. Effect of axillary dissection vs no axillary dissection on 10-year overall survival among women with invasive breast cancer and sentinel node metastasis (ACOSOG Z0011). JAMA. 2017;318(10):918-926. PMID: 21304082
- Donker M, van Tienhoven G, Straver ME, et al. Radiotherapy or surgery of the axilla after a positive sentinel node in breast cancer (EORTC 10981-22023 AMAROS). Lancet Oncol. 2014;15(12):1303-1310. PMID: 25304656
- Moran MS, Schnitt SJ, Giuliano AE, et al. SSO-ASTRO consensus guideline on margins for breast-conserving surgery with whole-breast irradiation in stages I and II invasive breast cancer. Ann Surg Oncol. 2014;21(3):704-716. PMID: 24501721
- van Hagen P, Hulshof MC, van Lanschot JJ, et al. Preoperative chemoradiotherapy for esophageal or junctional cancer (CROSS trial). N Engl J Med. 2012;366(22):2074-2084. PMID: 22646630
- Cunningham D, Allum WH, Stenning SP, et al. Perioperative chemotherapy versus surgery alone for resectable gastroesophageal cancer (MAGIC trial). N Engl J Med. 2006;355(1):11-20. PMID: 17615602
- Al-Batran SE, Homann N, Pauligk C, et al. Perioperative chemotherapy with FLOT versus ECF/ECX for resectable gastric or gastro-oesophageal junction adenocarcinoma (FLOT4-AIO). Lancet. 2019;393(10184):1948-1957. PMID: 30797662
- Conroy T, Hammel P, Hebbar M, et al. FOLFIRINOX or gemcitabine as adjuvant therapy for pancreatic cancer (PRODIGE 24). N Engl J Med. 2018;379(25):2395-2406. PMID: 30575490
- Conroy T, Desseigne F, Ychou M, et al. FOLFIRINOX versus gemcitabine for metastatic pancreatic cancer. N Engl J Med. 2011;364(19):1817-1825. PMID: 21612468
- Songun I, Putter H, Kranenbarg EMK, et al. Surgical treatment of gastric cancer: 15-year follow-up results of the Dutch D1D2 trial. Lancet Oncol. 2010;11(5):439-449. PMID: 20880531
- Morton DL, Thompson JF, Cochran AJ, et al. Final trial report of sentinel-node biopsy versus nodal observation in melanoma (MSLT-I). N Engl J Med. 2014;370(7):599-609. PMID: 24295718
- Faries MB, Thompson JF, Cochran AJ, et al. Completion dissection or observation for sentinel-node metastasis in melanoma (MSLT-II). N Engl J Med. 2017;376(23):2211-2222. PMID: 28614720
- Joensuu H, Eriksson M, Sundby Hall K, et al. One vs three years of adjuvant imatinib for operable gastrointestinal stromal tumor (SSG XVIII/AIO). JAMA. 2012;307(12):1265-1272. PMID: 22089421
- Bonvalot S, Gronchi A, Le Pechoux C, et al. Preoperative radiotherapy plus surgery versus surgery alone for patients with primary retroperitoneal sarcoma (STRASS). Lancet Oncol. 2020;21(10):1366-1377. PMID: 31986257
- Quenet F, Elias D, Roca L, et al. Cytoreductive surgery plus hyperthermic intraperitoneal chemotherapy versus cytoreductive surgery alone for colorectal peritoneal metastases (PRODIGE 7). Lancet Oncol. 2021;22(2):256-266. PMID: 30192068
- Mazzaferro V, Regalia E, Doci R, et al. Liver transplantation for the treatment of small hepatocellular carcinomas in patients with cirrhosis (Milan criteria). N Engl J Med. 1996;334(11):693-699. PMID: 8596091
- Finn RS, Qin S, Ikeda M, et al. Atezolizumab plus bevacizumab in unresectable hepatocellular carcinoma (IMbrave150). N Engl J Med. 2020;382(20):1894-1905. PMID: 32402160
- Fong Y, Fortner J, Sun RL, Brennan MF, Blumgart LH. Clinical score for predicting recurrence after hepatic resection for metastatic colorectal cancer. Ann Surg. 1999;230(3):309-321. PMID: 10493478
- Rosenberg SA, Tepper J, Glatstein E, et al. The treatment of soft-tissue sarcomas of the extremities: prospective randomized evaluations of limb-sparing surgery plus radiation therapy compared with amputation. Ann Surg. 1982;196(3):305-315. PMID: 6333478
- O'Sullivan B, Davis AM, Turcotte R, et al. Preoperative versus postoperative radiotherapy in soft-tissue sarcoma of the limbs (Canadian SR2 trial). Lancet. 2002;359(9325):2235-2241. PMID: 12091249
- Sparano JA, Gray RJ, Makower DF, et al. Adjuvant chemotherapy guided by a 21-gene expression assay in breast cancer (TAILORx). N Engl J Med. 2018;379(2):111-121. PMID: 29860917
- Grothey A, Sobrero AF, Shields AF, et al. Duration of adjuvant chemotherapy for stage III colon cancer (IDEA collaboration). N Engl J Med. 2018;378(13):1177-1188. PMID: 29527974
- Gustafson P. ERAS protocol adherence and outcomes after colorectal surgery. Br J Surg. 2010;97(8):1128-1136. PMID: 20395846
Textbooks & Reference Works
- Schwartz's Principles of Surgery. 11th ed. Brunicardi FC, ed. McGraw-Hill; 2019.
- Sabiston Textbook of Surgery: The Biological Basis of Modern Surgical Practice. 21st ed. Townsend CM, ed. Elsevier; 2021.
- Cancer: Principles and Practice of Oncology (DeVita, Hellman, and Rosenberg's). 12th ed. DeVita VT, Lawrence TS, Rosenberg SA, eds. Wolters Kluwer; 2023.
- The MD Anderson Surgical Oncology Handbook. 6th ed. Feig BW, Ching CD, eds. Wolters Kluwer; 2018.
- AJCC Cancer Staging Manual. 8th ed. Amin MB, Edge SB, Greene FL, et al., eds. Springer; 2017.
- NCCN Clinical Practice Guidelines in Oncology. National Comprehensive Cancer Network. nccn.org. Updated annually.
- Fischer's Mastery of Surgery. 7th ed. Fischer JE, ed. Wolters Kluwer; 2018.
- Blumgart's Surgery of the Liver, Biliary Tract and Pancreas. 6th ed. Jarnagin WR, ed. Elsevier; 2017.
- Harris JR, Lippman ME, Morrow M, Osborne CK. Diseases of the Breast. 5th ed. Wolters Kluwer; 2014.
- Balch CM, Gershenwald JE, Soong SJ, et al. Cutaneous Melanoma. In: AJCC Cancer Staging Manual. 8th ed. Springer; 2017.
- NCCN Guidelines: nccn.org — Gold standard treatment algorithms for all cancer types; updated multiple times yearly
- PubMed / MEDLINE: pubmed.ncbi.nlm.nih.gov — Literature search for latest evidence
- ClinicalTrials.gov: clinicaltrials.gov — Search for open trials for patients who may benefit from investigational therapies
- AJCC Cancer Staging: cancerstaging.org — Staging definitions, calculators, and resources
- Surgical Oncology societies: SSO (Society of Surgical Oncology), ESSO (European Society of Surgical Oncology), AHPBA (Americas Hepato-Pancreato-Biliary Association)