Transplant Surgery
Every organ system, donor evaluation, immunosuppression protocol, rejection classification, surgical technique, complication, and management algorithm across the full scope of transplant surgery in one place.
01 Transplant Immunology
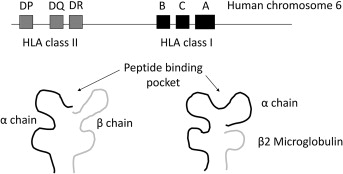
The MHC / HLA System
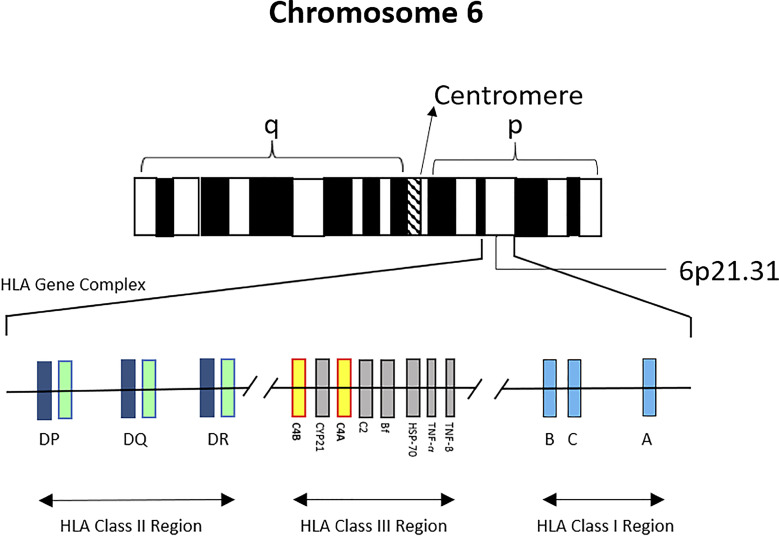
The major histocompatibility complex (MHC) is encoded on the short arm of chromosome 6 (6p21.3) and is the most polymorphic region in the human genome. In humans, MHC molecules are termed human leukocyte antigens (HLA). Two classes are critical for transplantation:
| Feature | MHC Class I | MHC Class II |
|---|---|---|
| Loci | HLA-A, HLA-B, HLA-C | HLA-DR, HLA-DQ, HLA-DP |
| Expression | All nucleated cells & platelets | APCs (dendritic cells, macrophages, B cells); activated T cells & endothelium |
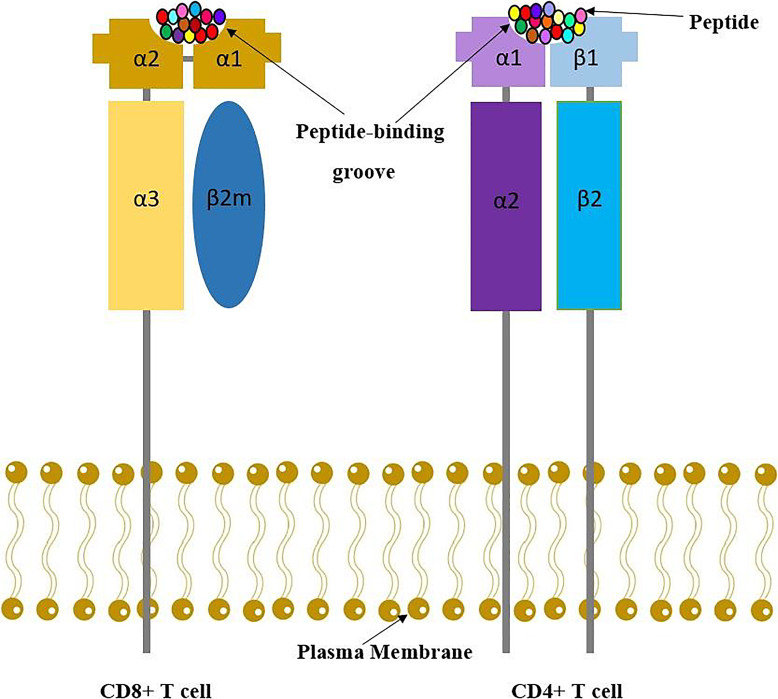
| Structure | α-chain + β2-microglobulin | α-chain + β-chain |
| Peptide binding | 8-10 amino acid peptides from cytosolic proteins | 13-25 amino acid peptides from extracellular proteins |
| Recognized by | CD8+ cytotoxic T cells | CD4+ helper T cells |
| Importance in transplant | HLA-A, -B matching moderately impacts graft survival | HLA-DR matching has greatest impact on kidney graft survival |
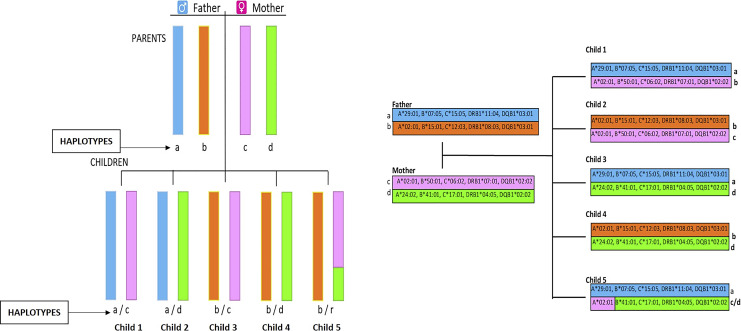
Each individual inherits one haplotype (a linked set of alleles on one chromosome 6) from each parent, giving two alleles at each locus. A zero-antigen-mismatch (0-ABDR-MM) kidney (0-0-0 at HLA-A, -B, -DR) confers the best graft survival — mandatorily shared nationally by UNOS policy regardless of geographic distance. HLA-identical sibling donors (2-haplotype match, 0-0-0-0-0-0 at A, B, C, DR, DQ, DP) provide the best possible immunologic match with half-lives > 25 years and the option for immunosuppression minimization.
The polymorphism of HLA is staggering: > 35,000 HLA alleles have been identified (IPD-IMGT/HLA Database), though many cluster into common allele groups. This diversity evolved to optimize pathogen peptide presentation but creates the immunologic barrier to transplantation.
Allorecognition Pathways
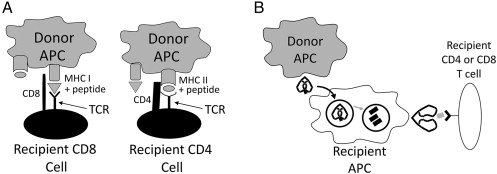
Recipient T cells recognize donor antigens via three distinct pathways:
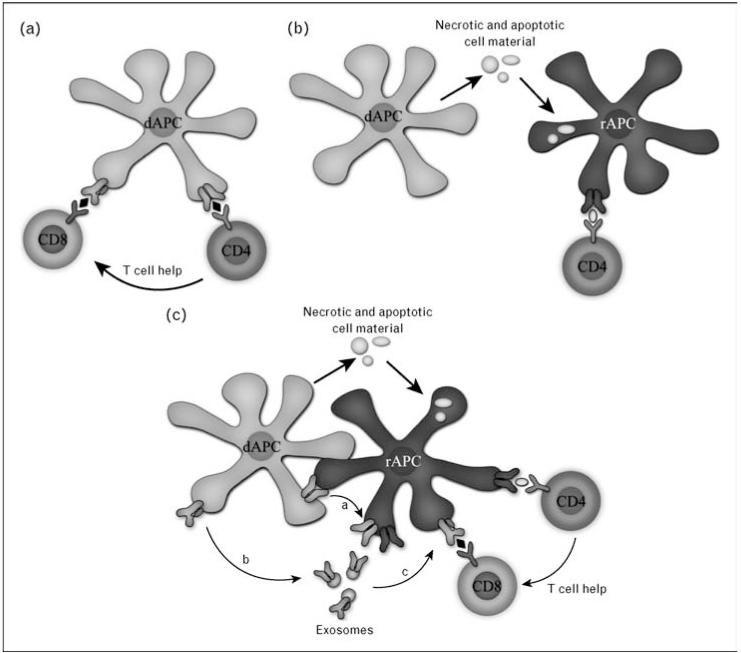
Direct allorecognition: Recipient T cells recognize intact donor MHC molecules on the surface of donor antigen-presenting cells (APCs) — especially donor dendritic cells that migrate from the graft to recipient lymphoid tissue ("passenger leukocytes"). This pathway dominates early after transplant and drives acute cellular rejection. Remarkably, 1-10% of the T-cell repertoire can directly recognize foreign MHC (compared to < 0.001% for conventional antigen recognition).
Indirect allorecognition: Recipient APCs process shed donor MHC molecules into peptide fragments and present them on recipient MHC class II to recipient CD4+ T cells — analogous to conventional antigen processing. This pathway predominates in chronic rejection and is responsible for generation of alloantibodies (donor-specific antibodies, DSA).
Semidirect allorecognition: Recipient APCs acquire intact donor MHC molecules via cell-to-cell contact or extracellular vesicles (trogocytosis/cross-dressing) and present them directly to T cells without processing. This pathway links direct and indirect pathways and may contribute to both acute and chronic rejection.
T-Cell Activation — The Three-Signal Model
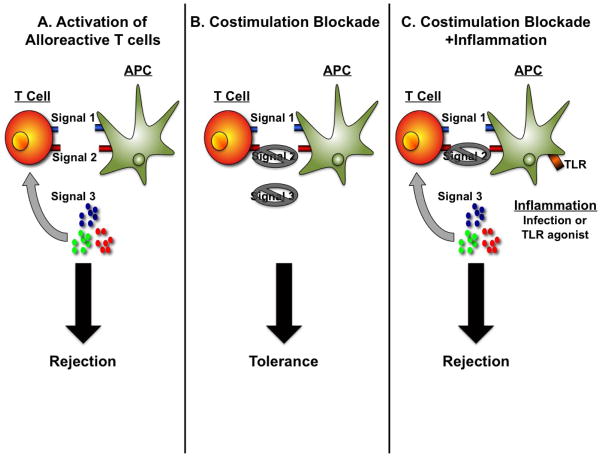
Full T-cell activation requires three distinct signals, each targeted by specific immunosuppressants:
| Signal | Mechanism | Drug Target |
|---|---|---|
| Signal 1 — Antigen recognition | TCR binds MHC-peptide complex → activates calcineurin → NFAT dephosphorylation → IL-2 gene transcription | Tacrolimus, cyclosporine (calcineurin inhibitors) |
| Signal 2 — Costimulation | CD28 on T cell binds B7 (CD80/CD86) on APC → amplifies Signal 1; CD40L on T cell binds CD40 on APC | Belatacept (CTLA4-Ig, blocks B7-CD28) |
| Signal 3 — Proliferation | IL-2 binds IL-2 receptor (CD25) → activates mTOR pathway → cell cycle progression G1→S | Basiliximab (anti-CD25); sirolimus, everolimus (mTOR inhibitors) |
Without Signal 2 (costimulation), T-cell receptor engagement leads to anergy (functional unresponsiveness) rather than activation — the basis for costimulation blockade strategies.
Complement & Antibody-Mediated Pathways
Preformed or de novo donor-specific antibodies (DSA) bind donor HLA on graft endothelium, activating the classical complement cascade: C1q binding → C4 cleavage → C4d deposition (a split product that covalently binds endothelium and serves as a "footprint" of antibody-mediated rejection) → C3 activation → membrane attack complex (C5b-9) formation → endothelial injury. C4d staining on biopsy is a hallmark of antibody-mediated rejection (AMR). Antibodies also cause injury via Fc receptor-mediated mechanisms (ADCC — antibody-dependent cell-mediated cytotoxicity by NK cells and macrophages) independent of complement.
02 Donor Evaluation & Organ Procurement
Brain Death Criteria
Brain death is defined as the irreversible cessation of all functions of the entire brain, including the brainstem. Determination requires: (1) known cause of coma; (2) exclusion of confounders (hypothermia < 36°C, drug intoxication, severe metabolic derangement); (3) clinical examination demonstrating absent brainstem reflexes — pupillary, corneal, oculocephalic, oculovestibular (cold calorics), gag, cough; (4) apnea test — disconnect ventilator, supply O2 via tracheal cannula, observe 8-10 minutes for respiratory effort, confirm PaCO2 ≥ 60 mmHg (or ≥ 20 mmHg rise from baseline) with no respiratory drive. Confirmatory tests (when clinical exam is unreliable): cerebral angiography (gold standard — no intracranial flow), EEG (electrocerebral silence), nuclear brain scan (no uptake — "hollow skull" sign), transcranial Doppler (reverberating flow or small systolic peaks).
Donation after Brain Death (DBD) vs. Donation after Circulatory Death (DCD)
| Feature | DBD | DCD |
|---|---|---|
| Definition | Donor declared dead by neurological criteria while on ventilator support | Donor declared dead after controlled withdrawal of life-sustaining treatment (Maastricht category III — most common) or uncontrolled cardiac arrest (category I, II) |
| Warm ischemia | Minimal — organs perfused until cross-clamp | Significant — period from withdrawal to death to cold perfusion (goal < 30 min) |
| Organ quality | Generally superior | Higher rates of DGF in kidneys (~50% vs ~25%); higher biliary complications in livers |
| % of deceased donors (US) | ~75% | ~25% (increasing) |
| Acceptable organs | All organs | Kidneys (most common), liver, lungs, pancreas; heart (emerging with NRP) |
Maastricht Classification — DCD Categories
| Category | Description | Control | Use |
|---|---|---|---|
| I | Dead on arrival (unwitnessed cardiac arrest) | Uncontrolled | Rarely used; organs often unsuitable |
| II | Unsuccessful resuscitation (witnessed cardiac arrest, failed CPR) | Uncontrolled | Used in some European centers with NRP; emerging in US |
| III | Awaiting cardiac arrest after withdrawal of life-sustaining treatment | Controlled | Most common DCD category in US (~95% of DCD); planned withdrawal in OR |
| IV | Cardiac arrest in a brain-dead donor (after brain death determination but before organ procurement) | Uncontrolled | Rare |
| V | Cardiac arrest in a hospitalized patient (expected cardiac arrest while on inpatient) | Controlled | Used in some countries |
In DCD category III: after withdrawal of life support, death is declared after 5 minutes of observed circulatory arrest (no-touch period). If death does not occur within 60-90 minutes of withdrawal, procurement is aborted and the patient returns to comfort care. The functional warm ischemia time (FWIT) is measured from the onset of significant hemodynamic compromise (systolic BP < 50-60 mmHg, SpO2 < 70%) to cold perfusion — FWIT > 30 minutes is associated with worse outcomes, particularly for liver grafts.
Normothermic Regional Perfusion (NRP) for DCD
NRP is an emerging technique where, after declaration of circulatory death, the DCD donor's abdominal (or thoracoabdominal) circulation is reestablished using ECMO to perfuse organs in-situ at normothermic temperature. This allows functional assessment of organs (liver: lactate clearance, bile production; kidney: urine output) before procurement. Benefits: improved graft quality, reduced biliary complications in DCD livers (NAS rate may decrease from ~15-30% to < 5%), potential to recover hearts from DCD donors. Ethical considerations: requires ligation of arch vessels to prevent brain reperfusion. NRP is established in Spain, UK, and Australia; pilot programs expanding in the US.
Donor Management — Hormonal Resuscitation
Brain death triggers a catecholamine storm followed by cardiovascular collapse and hormonal deficiency. Donor management goals: MAP ≥ 60 mmHg, CVP 4-10 mmHg, urine output 1-3 mL/kg/hr, PaO2/FiO2 > 300, Na+ < 155 mEq/L, pH 7.30-7.45, glucose < 180 mg/dL. Standard hormonal resuscitation protocol: T3/T4 (levothyroxine 20 μg IV bolus then 10 μg/hr), vasopressin (1 unit bolus then 0.5-4 units/hr — treats diabetes insipidus and supports MAP), methylprednisolone (15 mg/kg IV bolus — reduces inflammation), insulin drip (target glucose 120-180 mg/dL).
Organ Preservation
| Solution | Type | Key Features |
|---|---|---|
| UW (University of Wisconsin / Belzer) | Intracellular | Gold standard for liver, kidney, pancreas; contains lactobionate, raffinose, hydroxyethyl starch; high K+ (~125 mEq/L), low Na+ |
| HTK (Custodiol / Bretschneider) | Intracellular | Low viscosity — rapid flush; histidine buffer; lower K+ (~10 mEq/L); preferred for cardiac preservation; requires larger volumes (5-7 L vs 1-2 L for UW) |
| IGL-1 (Institut Georges Lopez) | Extracellular | PEG-based instead of HES; low K+, high Na+; comparable to UW with fewer electrolyte derangements on reperfusion |
| Celsior | Extracellular | Designed for heart; contains mannitol, glutamate, reduced glutathione; low viscosity |
Cold Ischemia Time (CIT) Limits
| Organ | Acceptable CIT | Maximum CIT | Notes |
|---|---|---|---|
| Kidney | < 24 hours | ~36 hours (with pump) | DGF rate increases significantly > 24 hr |
| Liver | < 10 hours | ~12 hours | Each hour > 8 increases PNF risk by ~10% |
| Pancreas | < 12 hours | ~18 hours | Optimal < 12 hr for whole organ |
| Heart | < 4 hours | ~6 hours | Most time-sensitive organ |
| Lung | < 6 hours | ~8 hours | Ex-vivo lung perfusion (EVLP) extends to 12+ hours |
| Intestine | < 8 hours | ~10 hours | Highly susceptible to ischemia-reperfusion injury |
Machine Perfusion — The Future of Organ Preservation
Hypothermic machine perfusion (HMP) for kidneys is well-established — pulsatile perfusion at 4°C reduces DGF compared to static cold storage (DGF rate ~20% vs ~27%; RR 0.77). PMID: 19246328. Parameters monitored: flow rate, resistance index (goal < 0.40), perfusate lactate. Normothermic machine perfusion (NMP) for liver — allows functional assessment of the graft ex-vivo (bile production, lactate clearance, pH) and extends preservation time; shown to reduce biliary complications in DCD livers. Ex-vivo lung perfusion (EVLP) — normothermic perfusion of donor lungs with Steen solution, allows reassessment of marginal lungs; INSPIRE trial showed non-inferior outcomes with EVLP-assessed lungs. PMID: 29211530
Multi-Organ Procurement — Surgical Sequence
The standard multi-organ procurement operation follows a precise sequence to minimize ischemia for each organ:
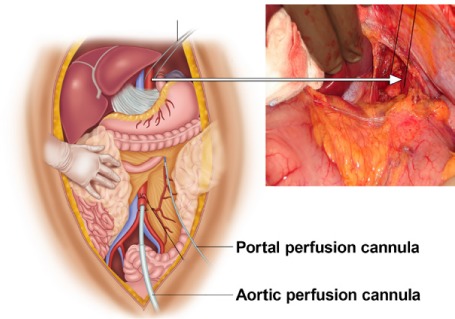
Incision & exposure: Midline incision from suprasternal notch to pubis; median sternotomy for cardiothoracic organs. Full abdominal exploration to assess organ quality (cirrhosis, masses, anatomic variants).
Dissection phase: Identify hepatic arterial anatomy (standard vs. replaced/accessory right hepatic artery from SMA — present in ~15-20%, or replaced left hepatic artery from left gastric — ~10%). Mobilize the liver, isolate the portal vein, identify the bile duct. Mobilize the kidneys bilaterally. Dissect the iliac vessels for conduit grafts (donor iliac artery Y-graft used for hepatic artery reconstruction or pancreas arterial graft).
Cannulation & flush: Aortic cannula placed in the distal aorta (infrarenal or at the iliac bifurcation); portal vein cannula for portal flush (some teams); supraceliac aortic cross-clamp applied after cardiac team places aortic root cardioplegia cannula. Cold preservation solution flushed antegrade through the aortic cannula (UW or HTK — typically 3-5 liters) and retrograde through portal vein if cannulated. Venting via IVC (in chest — right atrial incision).
Order of organ removal: (1) Heart (most time-sensitive — after cardioplegia); (2) Lungs (after bilateral pulmonary arterial flush with Perfadex); (3) Liver (with adequate aortic and portal flush); (4) Pancreas (en bloc with duodenal C-loop and spleen); (5) Kidneys (en bloc or separately, with ureters and aortic patch/Carrel patches for renal arteries). Iliac vessels, aortic conduits, and additional tissues (corneas, skin, bone, heart valves) harvested last.
03 Recipient Evaluation & Listing
UNOS Organ Allocation System
The United Network for Organ Sharing (UNOS), operating as the Organ Procurement and Transplantation Network (OPTN), manages organ allocation in the United States. Key principles: medical urgency, waiting time, geographic proximity (since 2021, allocation uses 250-nautical-mile concentric circles from the donor hospital rather than DSA-based boundaries for kidneys and livers), and immunologic compatibility. Each organ has a distinct allocation algorithm.
MELD / PELD Scores
The Model for End-Stage Liver Disease (MELD) score predicts 90-day mortality in liver disease and is used for liver allocation. Current MELD 3.0 formula (adopted January 2024):
MELD 3.0 = 1.33 × (female sex) + 4.56 × ln(bilirubin) + 0.82 × (137 − Na) − 0.24 × (137 − Na) × ln(creatinine) + 9.09 × ln(INR) + 11.14 × ln(creatinine) + 1.85 × (3.5 − albumin) − 1.83 × (3.5 − albumin) × ln(creatinine) + 6
Previously used MELD-Na: MELD-Na = MELD + 1.32 × (137 − Na) − [0.033 × MELD × (137 − Na)]; Na capped at 125-137 mEq/L. Range 6-40. MELD exception points are granted for conditions where mortality is not reflected by lab values (e.g., HCC, hepatopulmonary syndrome, portopulmonary hypertension).
PELD (Pediatric End-Stage Liver Disease) — for patients < 12 years: PELD = 4.80 × ln(bilirubin) + 18.57 × ln(INR) − 6.87 × ln(albumin) + 4.36 (< 1 year) + 6.67 (growth failure).
Waitlist Management & Organ Acceptance
Once listed, candidates are managed actively: regular reassessment of clinical status, update of laboratory values (MELD recertification every 7-90 days depending on score), optimization of comorbidities, and bridging therapies where applicable. Organ offers are generated by UNOS computer algorithm matching donor characteristics to candidate priority. Accepting a marginal organ (high KDPI kidney, DCD liver) vs. waiting for an optimal organ requires individualized risk-benefit analysis. Studies show that for most candidates, accepting a higher-risk organ yields better survival than remaining on the waitlist (survival benefit of transplant vs. dialysis is clear even with KDPI 85-100% kidneys for most recipients). PMID: 27428555
Kidney Allocation — KDPI / EPTS
The Kidney Donor Profile Index (KDPI) estimates the quality of a deceased donor kidney relative to all donors — based on 10 donor factors (age, height, weight, ethnicity, hypertension, diabetes, cause of death, creatinine, HCV, DCD status). KDPI 0-20% = highest quality; KDPI > 85% = expanded criteria (previously ECD). The Estimated Post-Transplant Survival (EPTS) score ranks candidates by expected survival — top 20% EPTS candidates are preferentially matched with top 20% KDPI kidneys ("longevity matching"). The Kidney Donor Risk Index (KDRI) predicts relative graft failure risk; KDPI is the percentile mapping of KDRI.
Crossmatch Testing
| Test | Method | Sensitivity | Clinical Significance |
|---|---|---|---|
| CDC crossmatch | Recipient serum + donor lymphocytes + complement; cell lysis = positive | Lowest (detects ~1 μg/mL DSA) | Positive T-cell CDC crossmatch = absolute contraindication to transplant (hyperacute rejection) |
| Flow cytometry crossmatch | Recipient serum + donor lymphocytes; fluorescent anti-IgG detection by FACS | 10-50x more sensitive than CDC | Positive T-cell flow = increased AMR risk; positive B-cell flow = controversial (may detect non-HLA antibodies) |
| Virtual crossmatch | Compare recipient antibody profile (Luminex SAB) to donor HLA typing | Highest (detects DSA at MFI > 1,000-3,000) | Allows prospective avoidance of incompatible donors; enables organ sharing over distance |
Panel Reactive Antibody (PRA) / Calculated PRA (cPRA)
cPRA estimates the percentage of deceased donors against whom a candidate has preformed HLA antibodies, based on the frequency of unacceptable antigens in the donor population. cPRA ≥ 98% designates a highly sensitized patient — receives priority points under UNOS policy. Causes of sensitization: prior transplant (most immunogenic), pregnancy, blood transfusions. Desensitization protocols for highly sensitized patients include: plasmapheresis, IVIG (2 g/kg), rituximab (375 mg/m2), and in some centers bortezomib (proteasome inhibitor targeting plasma cells).
Pregnancy Post-Transplant
Pregnancy is possible and increasingly common in transplant recipients (especially kidney). Key considerations: (1) Wait at least 1-2 years post-transplant with stable graft function, no recent rejection, and minimal immunosuppression; (2) Teratogenic medications must be discontinued before conception: MMF/MPA (category X — congenital anomalies in ~25% of exposed pregnancies — ear, facial, cardiac, digital malformations), mTOR inhibitors; replace with azathioprine (category D — safer in pregnancy, though not risk-free); (3) Maintain tacrolimus (levels may decrease in pregnancy due to increased volume of distribution and CYP3A4 activity — monitor more frequently); (4) Continue prednisone at lowest effective dose; (5) Complications: pre-eclampsia (~30-40%), preterm delivery (~40-50%), low birth weight, graft dysfunction (risk of rejection — especially if IS reduced). Outcomes: live birth rate ~70-80% (kidney Tx); graft loss within 2 years of pregnancy ~5-10%. Breastfeeding: tacrolimus excreted in breast milk at low levels — expert opinion generally permits breastfeeding with close monitoring.
Pediatric Transplantation
Pediatric transplant has unique considerations: (1) Growth: Corticosteroids impair linear growth — steroid-free/minimization protocols are critical. Catch-up growth occurs after steroid withdrawal. (2) Adherence: Non-adherence is the #1 cause of graft loss in adolescents — rates of 30-50% in teenage recipients. Transition programs from pediatric to adult care are critical. (3) EBV/PTLD: Higher risk due to EBV-seronegative status at transplant — primary EBV infection under immunosuppression carries highest PTLD risk. (4) Unique indications: Kidney — congenital anomalies (posterior urethral valves, renal dysplasia), FSGS, hemolytic uremic syndrome. Liver — biliary atresia (#1 indication for pediatric liver transplant, ~50% of cases), metabolic diseases. Heart — congenital heart disease, cardiomyopathy. (5) Surgical considerations: Size mismatch (adult donor to pediatric recipient), vascular anatomy challenges, split/reduced-size liver grafts. ABO-incompatible heart transplant is feasible in infants < 12-18 months (immature immune system, absence of isoagglutinins).
04 Histocompatibility & Crossmatching
ABO Compatibility
ABO-compatible transplantation follows standard blood group rules (identical or compatible). ABO-incompatible (ABOi) kidney transplantation is performed at experienced centers using desensitization: target anti-A or anti-B isoagglutinin titers ≤ 1:8-1:16 via plasmapheresis/immunoadsorption, plus rituximab and standard induction. One-year graft survival approaches ABO-compatible rates (~95-97%). ABOi liver transplant is performed in children < 2 years (immature isoagglutinins) with excellent results. ABOi heart transplant is similarly performed in infants < 12-18 months.
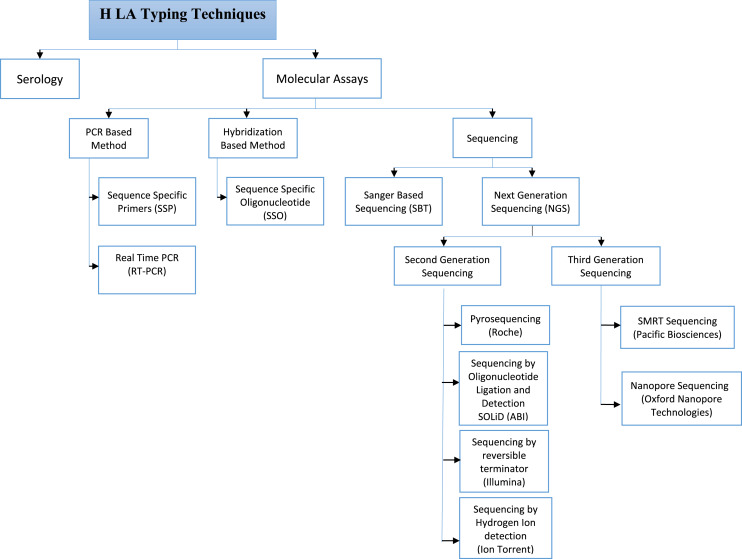
HLA Typing Methods
| Method | Resolution | Technique | Current Use |
|---|---|---|---|
| Serologic typing | Low (antigen-level) | Complement-dependent lymphocytotoxicity with known antisera | Largely replaced |
| SSP (sequence-specific primers) | Low-intermediate | PCR with allele-specific primers | Rapid typing; deceased donor |
| SSO (sequence-specific oligonucleotides) | Intermediate | PCR + hybridization to bead-bound probes (Luminex) | Standard for initial typing |
| SBT (sequence-based typing) | High (allele-level) | Sanger sequencing of HLA genes | Resolving ambiguities |
| NGS (next-generation sequencing) | Highest | Massively parallel sequencing | Increasingly adopted; unambiguous allele-level typing |
Antibody Identification — Luminex Single-Antigen Bead (SAB) Assay
The Luminex SAB assay is the standard for identifying HLA-specific antibodies. Beads coated with individual recombinant HLA antigens are incubated with patient serum; bound IgG is detected with PE-conjugated anti-human IgG. Results reported as mean fluorescence intensity (MFI). Typical thresholds: MFI > 1,000-3,000 considered positive (center-dependent); MFI > 5,000-10,000 associated with positive flow crossmatch. Limitations include prozone effect (false-negative at very high antibody titers — treat serum with EDTA or DTT to mitigate), shared epitope reactivity, and denatured antigens on beads.
Donor-Derived Disease Transmission
Rare but important consideration in transplantation. Types: (1) Infectious disease transmission: Donor screening includes HIV, HBV, HCV, CMV, EBV, RPR, HTLV, toxoplasma, strongyloides, Trypanosoma cruzi (in endemic areas), West Nile virus (seasonal). Despite screening, window period infections can occur. HCV-positive donor organs (viremic, NAT+) are now increasingly used for HCV-negative recipients with planned post-transplant DAA treatment — expanding the donor pool. HIV-positive donors can be used for HIV-positive recipients under the HOPE Act. (2) Malignancy transmission: Donor history of cancer requires careful risk assessment. Low risk: non-melanoma skin cancer, small renal cell carcinoma (treated), cervical CIS. Intermediate risk: breast cancer in remission > 5 years, colon cancer in remission. High risk / absolute contraindication: active malignancy (except non-metastatic primary brain tumor, non-melanoma skin cancer), metastatic cancer, melanoma. CNS tumors with VP shunts carry risk of peritoneal seeding. Donor-derived cancer incidence: ~0.01-0.05%.
Acceptable Mismatch Programs & Epitope Matching
For highly sensitized patients (cPRA ≥ 98%), acceptable mismatch programs identify HLA antigens to which the patient does NOT have antibodies — expanding the potential donor pool. HLAMatchmaker analyzes HLA compatibility at the eplet level (polymorphic amino acid configurations on the molecular surface that are targets of alloantibodies) rather than whole-antigen level, allowing more refined assessment of immunologic risk. Eplet mismatch load correlates with de novo DSA development and chronic rejection risk.
05 Kidney Transplant — Surgical Technique
Living Donor Nephrectomy
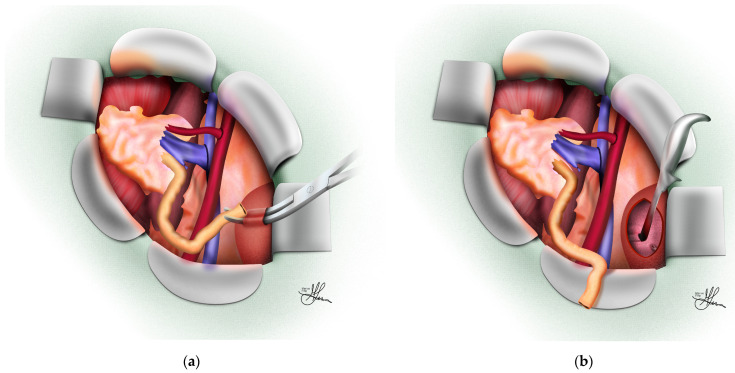
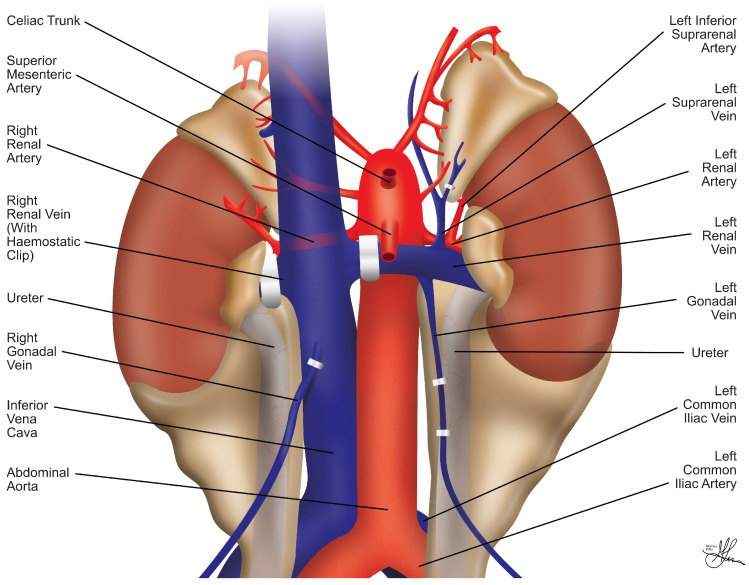
Laparoscopic donor nephrectomy is the standard approach (left kidney preferred due to longer renal vein). Technique: lateral decubitus position, 3-4 ports, transperitoneal approach; identify ureter (preserve periureteral blood supply — "golden rule"), gonadal vein, renal vein, renal artery; secure artery with Hem-o-lok clips or vascular stapler (no metal clips on artery — risk of dislodgment); divide ureter distally with adequate length; extract kidney through Pfannenstiel incision. Warm ischemia time goal: < 3-5 minutes. Donor mortality: ~0.03% (3 per 10,000). PMID: 20215610
Bench Preparation
The kidney is flushed on the back table with cold preservation solution. Excess perinephric fat is removed. Vascular anatomy is assessed — multiple arteries require reconstruction (side-to-side anastomosis creating a common ostium, or separate anastomoses). The renal vein is prepared; if right kidney (shorter vein), IVC cuff may be used or vein extension with donor gonadal or IVC vein. The ureter is trimmed to appropriate length, preserving periureteral tissue and the "golden triangle" (tissue between ureter, lower pole, and gonadal vein).
Recipient Operation
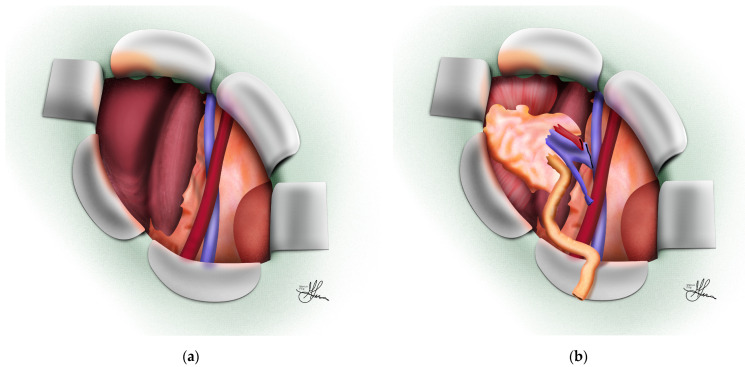
Incision: Curvilinear (Gibson/hockey-stick) incision in the right iliac fossa (preferred — external iliac vessels more superficial and horizontal); left iliac fossa if prior right transplant or if pancreas transplant planned for right side. Retroperitoneal approach — peritoneum reflected medially.
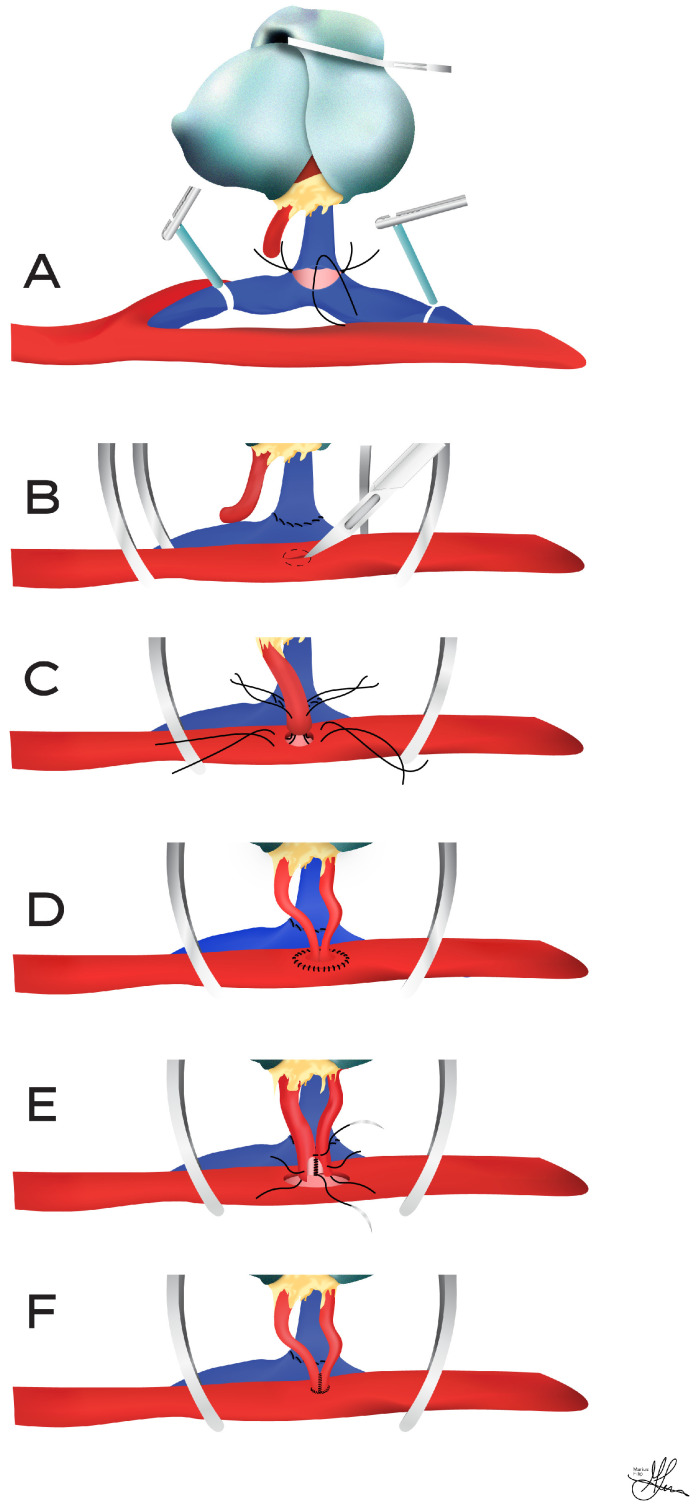
Vascular anastomoses: Renal vein to external iliac vein (end-to-side) — performed first to allow venous drainage; renal artery to external iliac artery (end-to-side) — or internal iliac artery (end-to-end, less common now). Running 5-0 or 6-0 polypropylene suture. Clamp time goal: < 30-45 minutes.
Ureteroneocystostomy (Lich-Gregoir): The extravesical technique — an anterior cystotomy is made in the bladder detrusor muscle, the ureter is anastomosed to the mucosa with 5-0 or 6-0 absorbable suture, and a submucosal tunnel is created by re-approximating the detrusor over the ureter (anti-reflux mechanism). A double-J ureteral stent is placed (removed at 4-6 weeks). Alternative: Leadbetter-Politano (intravesical technique — rarely used now).
Intraoperative Assessment
After unclamping, the kidney should pink up uniformly within minutes, become turgid, and begin producing urine. Signs of adequate perfusion: uniform color (no patchy areas suggesting accessory artery not reconstructed), turgor, visible peristalsis of the ureter, and urine output. If the kidney remains dusky: check for vascular kink, twist, or compression; confirm no residual thrombus; consider additional heparin flush. Intraoperative Doppler confirms arterial and venous flow. Furosemide (40-100 mg) and mannitol (12.5-25 g) are commonly given at reperfusion. Hemodynamic goals: maintain systolic BP > 120 mmHg, CVP 10-15 mmHg to maximize graft perfusion.
06 Kidney Transplant — Rejection
Hyperacute Rejection
Emergency Occurs within minutes to hours of reperfusion. Caused by preformed antibodies (anti-donor HLA or anti-ABO) that bind graft endothelium → complement activation → diffuse thrombotic microangiopathy → graft thrombosis. The kidney turns blue/mottled and fails to produce urine. Treatment: Immediate nephrectomy — no rescue therapy exists. Prevention: pretransplant crossmatch testing. Essentially eliminated in modern practice by crossmatching.
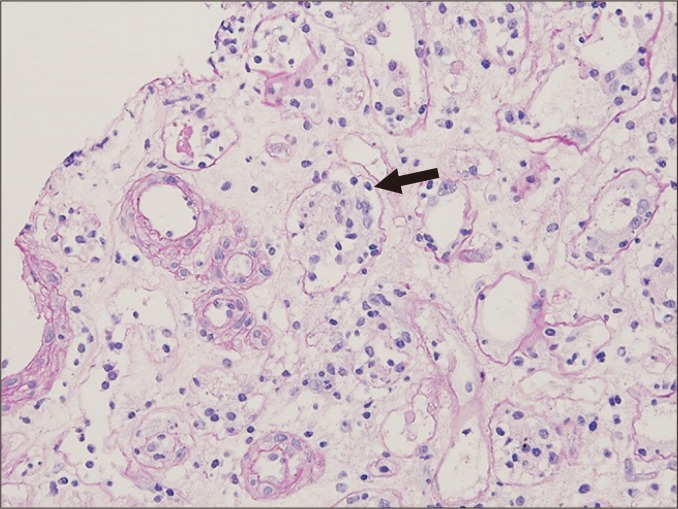
Acute Cellular Rejection (ACR) — Banff Classification
T-cell mediated rejection diagnosed on biopsy. The Banff classification (last updated 2022) grades tubulitis and interstitial inflammation:
| Grade | Histologic Criteria | Description |
|---|---|---|
| Borderline | t1-t2, i0-i1 | "Suspicious for rejection" — mild tubulitis with minimal interstitial inflammation; may treat or observe |
| IA | t2, i2-i3 | Moderate tubulitis (> 4 mononuclear cells per tubular cross-section) with significant interstitial inflammation (≥ 25% of cortex) |
| IB | t3, i2-i3 | Severe tubulitis (> 10 mononuclear cells per tubular cross-section) with significant interstitial inflammation |
| IIA | v1 | Intimal arteritis — mild to moderate (mononuclear cells under endothelium in < 25% of luminal area) |
| IIB | v2 | Severe intimal arteritis (≥ 25% luminal compromise) |
| III | v3 | Transmural arteritis and/or fibrinoid necrosis of arterial wall |
Banff scoring components: i (interstitial inflammation: i0-i3), t (tubulitis: t0-t3), v (intimal arteritis: v0-v3), g (glomerulitis: g0-g3), ptc (peritubular capillaritis: ptc0-ptc3), C4d (C4d staining: C4d0-C4d3), ci (interstitial fibrosis: ci0-ci3), ct (tubular atrophy: ct0-ct3), cv (vascular fibrous intimal thickening: cv0-cv3), cg (transplant glomerulopathy: cg0-cg3), mm (mesangial matrix expansion: mm0-mm3), ah (arteriolar hyalinosis: ah0-ah3).
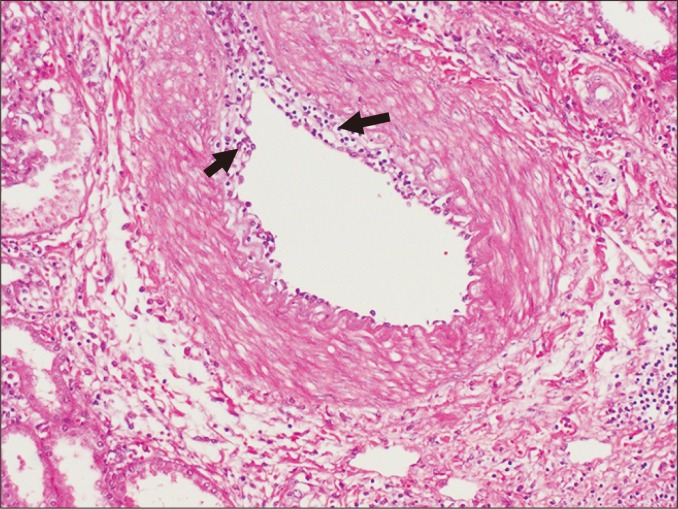
Acute Antibody-Mediated Rejection (AMR)
Requires all three criteria (Banff 2022): (1) histologic evidence of acute tissue injury — microvascular inflammation (g + ptc score ≥ 2), intimal or transmural arteritis, acute thrombotic microangiopathy, or acute tubular injury in absence of other causes; (2) evidence of antibody interaction with endothelium — C4d staining in peritubular capillaries (C4d2-3 by IF or IHC) OR microvascular inflammation (g + ptc ≥ 2) OR increased gene expression of validated transcripts (molecular AMR); (3) serologic evidence of DSA (HLA or non-HLA antibodies).
Acute Rejection — Clinical Presentation
Acute rejection in kidney transplant typically presents as: (1) asymptomatic rise in serum creatinine (most common — detected on routine labs); (2) decreased urine output; (3) graft tenderness/swelling (less common with modern immunosuppression); (4) fever (nonspecific). Importantly, modern immunosuppression has made "textbook" rejection presentation (fever, graft tenderness, oliguria) uncommon — most rejection episodes are now detected by surveillance bloodwork. This underscores the importance of frequent creatinine monitoring (twice weekly for 4 weeks, then weekly for 2 months, then biweekly, then monthly). Any unexplained creatinine rise of ≥ 20% warrants investigation: Doppler ultrasound (rule out obstruction, vascular complications, collections) and graft biopsy if no surgical cause identified.
Chronic Active Antibody-Mediated Rejection & IFTA
Chronic active AMR is characterized by transplant glomerulopathy (cg ≥ 1 — double contours on light microscopy or duplication of GBM on EM), peritubular capillary basement membrane multilayering (≥ 7 layers on EM), and arterial intimal fibrosis. This is the leading cause of late graft loss. Interstitial fibrosis and tubular atrophy (IF/TA) — previously termed "chronic allograft nephropathy" — is a nonspecific histologic pattern (ci + ct scores) representing the common endpoint of multiple injury mechanisms (chronic rejection, CNI toxicity, BK nephropathy, recurrent disease). Treatment of chronic AMR remains challenging — no proven effective therapy; options include optimizing maintenance immunosuppression, tocilizumab (anti-IL-6R), and investigational agents.
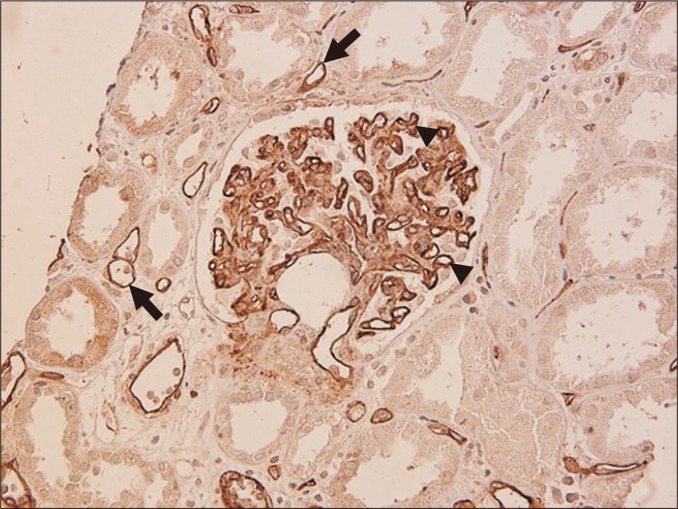
07 Delayed Graft Function
Definition & Epidemiology
Delayed graft function (DGF) is defined as the need for dialysis within the first 7 days post-transplant. Incidence: ~25% in DBD kidneys, ~50% in DCD kidneys, < 5% in living donor kidneys. DGF is associated with increased acute rejection risk (OR ~1.4), reduced long-term graft survival, and increased cost/hospitalization.
Risk Factors
| Donor Factors | Recipient Factors | Surgical Factors |
|---|---|---|
| Extended criteria donor (KDPI > 85%) | High PRA / sensitized | Prolonged CIT (> 24 hr) |
| DCD donation | Prolonged time on dialysis | Prolonged warm ischemia time |
| Advanced donor age (> 55 yr) | High BMI | Reoperation / vascular complications |
| Terminal creatinine > 1.5 mg/dL | Diabetes | Machine perfusion resistance > 0.40 |
| Donor hypertension/diabetes | Retransplant | Absence of machine perfusion |
Management
Continue dialysis as needed. Maintain adequate hydration (avoid hypovolemia — CVP 8-12 mmHg). Doppler ultrasound to confirm vascular patency (rule out renal artery/vein thrombosis). Avoid nephrotoxins (minimize CNI exposure — consider delayed CNI introduction with thymoglobulin induction). Protocol biopsy recommended at post-transplant day 7-10 if no improvement — to differentiate DGF from rejection (which occurs in ~10-30% of DGF cases). ATN on biopsy supports DGF diagnosis; tubulitis/interstitial inflammation suggests superimposed rejection requiring treatment.
Slow Graft Function vs. DGF vs. Primary Non-Function
| Term | Definition | Prognosis |
|---|---|---|
| Immediate graft function | Prompt diuresis, creatinine decline without need for dialysis | Best outcome |
| Slow graft function (SGF) | Serum creatinine fails to decrease by ≥ 10%/day in first 3 days OR creatinine > 3.0 at day 5, but NO dialysis needed | Intermediate; usually recovers; slightly worse long-term than IGF |
| Delayed graft function (DGF) | Dialysis required within 7 days post-transplant | Associated with increased rejection risk and reduced graft survival |
| Primary non-function (PNF) | Permanent failure to function from the time of transplant; requires nephrectomy and return to dialysis | Graft loss; retransplant needed |
Ischemia-Reperfusion Injury — Pathophysiology
During cold ischemia, ATP depletion leads to Na+/K+-ATPase failure → cellular swelling → calcium influx → mitochondrial injury. Upon reperfusion, reactive oxygen species (ROS) are generated by xanthine oxidase and the mitochondrial electron transport chain → endothelial activation → upregulation of adhesion molecules (P-selectin, ICAM-1) → neutrophil infiltration → further tissue injury. The innate immune response (TLR4 activation, complement, DAMPs) amplifies the adaptive immune response, increasing immunogenicity of the graft and rejection risk. This mechanistic link explains why DGF increases acute rejection risk and supports the strategy of more potent induction immunosuppression (rATG) in patients at high risk for DGF.
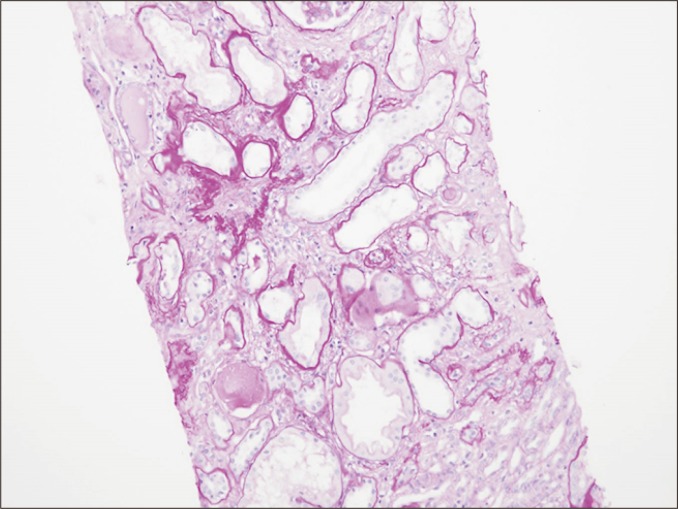
08 Post-Kidney Transplant Complications
Urologic Complications
Urine leak / urinoma: Usually at the ureteroneocystostomy (1-5% incidence); presents with pain, swelling, rising creatinine with stable serum BUN, perinephric fluid on imaging. Diagnosis: high creatinine in drain fluid (drain fluid Cr >> serum Cr), nuclear renal scan shows extravasation. Treatment: percutaneous drainage + ureteral stent placement; surgical revision if persistent.
Ureteral stenosis/stricture: 2-10% incidence; ischemic etiology most common (distal ureter has tenuous blood supply from the renal artery). Early (< 3 months): edema, kinking, blood clot. Late: fibrosis, BK virus, rejection. Presents as rising creatinine and hydronephrosis on ultrasound. Treatment: percutaneous nephrostomy + antegrade stent; balloon dilation; surgical reimplantation if refractory.
Lymphocele: 1-15% incidence; collection of lymphatic fluid from disrupted iliac lymphatics during recipient dissection. Usually asymptomatic; large lymphoceles may compress ureter (hydronephrosis) or iliac vein (leg edema/DVT). Diagnosis: ultrasound-guided aspiration — fluid creatinine = serum creatinine (distinguishes from urinoma), low triglycerides (distinguishes from chyle). Treatment: percutaneous drainage (high recurrence ~50%), laparoscopic marsupialization (fenestration into peritoneal cavity — definitive treatment, success > 90%).
Vascular Complications
Renal artery stenosis: 1-5% incidence; usually at the anastomosis; presents months post-transplant with refractory hypertension, graft dysfunction, flash pulmonary edema. Bruit over graft. Diagnosis: Doppler US (elevated PSV > 200-250 cm/s, tardus-parvus waveform distally), confirmed by CTA or MRA. Treatment: percutaneous transluminal angioplasty ± stenting (technical success ~80-90%); surgical revision if refractory.
Renal vein thrombosis: Emergency ~0.5-4% incidence; usually within first week. Presents with sudden graft swelling, pain, hematuria, absent/reversed diastolic flow on Doppler. Treatment: emergent surgical thrombectomy (if < 24-48 hr); graft nephrectomy if salvage fails. Risk factors: technical (vein kinking, compression by lymphocele/hematoma), hypercoagulability, DGF with low flow.
Renal artery thrombosis: Emergency < 1% incidence; usually technical (intimal flap, kinking). Absent arterial and venous flow on Doppler — complete graft infarction. Almost always leads to graft loss. Emergent surgical exploration may salvage very early cases.
Medical Complications
Post-transplant diabetes mellitus (PTDM): 10-30% incidence; risk factors: tacrolimus (greatest risk — impairs insulin secretion), corticosteroids (insulin resistance), obesity, age, HCV, family history. Management: lifestyle modification, insulin initially; transition to oral agents (metformin preferred if GFR allows; avoid sulfonylureas with CYP3A4 inhibitors). Consider conversion from tacrolimus to cyclosporine or belatacept in severe cases.
BK polyomavirus nephropathy: 1-10% incidence; BK virus reactivates under immunosuppression. Screening: BK viremia by PCR (quantitative BK viral load in plasma) at months 1, 2, 3, 6, 9, 12 post-transplant. BK viremia > 10,000 copies/mL ("presumptive BK nephropathy") triggers intervention. Biopsy confirms: viral cytopathic changes, positive SV40 immunostaining, tubulointerstitial nephritis. Treatment: reduce immunosuppression (decrease tacrolimus target to 4-6 ng/mL, reduce/stop mycophenolate). No proven antiviral therapy (cidofovir, leflunomide have limited evidence). IVIG and fluoroquinolones sometimes used.
Recurrent Disease in the Kidney Allograft
| Disease | Recurrence Rate | Timing | Notes |
|---|---|---|---|
| FSGS | 30-50% (up to 80% if prior graft lost to recurrence) | Hours to days (can recur within hours of reperfusion) | Circulating permeability factor (suPAR?); treat with plasmapheresis + rituximab. Living related donor may have higher recurrence |
| IgA nephropathy | 20-60% histologic; ~10% graft loss | Months to years | Histologic recurrence common but clinical impact often mild |
| Membranous nephropathy | 30-40% | Months to years | Anti-PLA2R antibody-positive patients have higher recurrence |
| MPGN / C3 glomerulopathy | 50-80% | Months | Complement-mediated; eculizumab under investigation |
| aHUS | 50-80% (without treatment) | Days to weeks | Eculizumab prophylaxis (begin pre-transplant) dramatically reduces recurrence |
| Lupus nephritis | 2-10% | Years | Low recurrence rate; transplant during disease quiescence |
| Diabetic nephropathy | ~100% histologic over years | Years | Histologic changes appear by 2 years; clinically significant by 5-10 years; SPK may reduce/prevent recurrence |
| Oxalosis (primary hyperoxaluria type 1) | Nearly 100% in kidney alone | Immediate | Combined liver-kidney transplant required (liver corrects the enzyme defect) |
09 Living Donor Kidney Transplantation
Living Donor Evaluation
Comprehensive assessment per OPTN policy: medical history & physical exam, psychosocial evaluation by independent living donor advocate (ILDA), ABO/HLA typing, crossmatch. Medical workup: GFR (must be > 80 mL/min by nuclear GFR or iothalamate clearance), 24-hr urine protein (< 300 mg/day), creatinine clearance, urinalysis, metabolic panel, CBC, coagulation studies, HbA1c (< 6.5%), lipid panel, fasting glucose, infectious screening (HIV, HBV, HCV, CMV, EBV, RPR, TB), age-appropriate cancer screening, CT angiogram of kidneys (vascular anatomy — number of arteries/veins, kidney size, ureteral anatomy, incidental findings). Exclusion criteria: uncontrolled HTN, DM, GFR < 80, proteinuria > 300 mg/day, BMI > 35 (relative), active malignancy, HIV (relative — living HIV-positive to HIV-positive donation under HOPE Act), substance abuse, psychiatric instability.
Outcomes
Living donor kidney transplant offers superior outcomes: 1-year graft survival ~98% vs ~93-95% (deceased donor); 5-year graft survival ~90% vs ~80%; half-life ~15-20 years (living) vs ~10-12 years (deceased). Lower DGF rate (< 5%). Donor risks: perioperative mortality ~0.03%, long-term ESRD risk slightly elevated but absolute risk remains very low (0.3-0.5% lifetime risk vs 0.15% general population). PMID: 24284516
Paired Kidney Exchange (KPD)
When a willing living donor is incompatible with their intended recipient (ABO-incompatible or positive crossmatch), kidney paired donation (KPD) allows swapping donors between incompatible pairs. Types: two-way exchange (simplest), three-way chain, domino chains (initiated by a non-directed/altruistic donor — chain can extend to 30+ transplants). UNOS operates a national KPD program. Non-simultaneous extended altruistic donor (NEAD) chains enable longer chains by allowing "bridge" donors to donate days-weeks after their paired recipient receives a kidney.
ABO-Incompatible Protocols
When paired exchange is not possible, ABOi transplantation is performed with desensitization: (1) determine baseline anti-A or anti-B isoagglutinin titers (IgG by column agglutination); (2) plasmapheresis sessions (typically 3-5) to reduce titer to ≤ 1:8-1:16; (3) rituximab 375 mg/m2 IV (given 2-4 weeks pre-transplant — depletes B cells producing isoagglutinins); (4) standard immunosuppression induction; (5) post-transplant monitoring of titers (daily initially). Accommodation (graft tolerance despite low-level circulating antibodies) develops in most cases. Graft survival: ~95% at 1 year, comparable to compatible transplants. PMID: 15084934
10 Liver Transplant — Indications & Listing
Indications
Decompensated cirrhosis (most common indication — ~60%): variceal hemorrhage, ascites, hepatic encephalopathy, hepatorenal syndrome, hepatopulmonary syndrome. Etiologies: alcohol-associated liver disease (now #1 cause in the US), NASH/MASLD (rising), hepatitis C (declining with DAA therapy), hepatitis B, autoimmune hepatitis, primary biliary cholangitis (PBC), primary sclerosing cholangitis (PSC).
Hepatocellular carcinoma (HCC): Within Milan criteria (see Section 14) — transplant is the best treatment as it addresses both the tumor and the underlying cirrhotic liver.
Acute liver failure (ALF): UNOS Status 1A — highest priority; etiologies: acetaminophen overdose (#1 in US/UK), viral hepatitis, drug-induced liver injury, Wilson disease, acute fatty liver of pregnancy, Budd-Chiari. King's College criteria guide listing.
Metabolic diseases: Alpha-1 antitrypsin deficiency, Wilson disease, hemochromatosis, familial amyloid polyneuropathy, primary hyperoxaluria, urea cycle defects, maple syrup urine disease. In some metabolic diseases, the liver is structurally normal but produces a toxic/absent protein — "domino transplantation" (the explanted liver can be transplanted into another recipient).
MELD-Na Score Details
MELD-Na range: 6 (least ill) to 40 (most ill). Determines liver allocation priority. Lab values capped: creatinine 1.0-4.0 mg/dL (if on dialysis ≥ 2x/week, Cr set to 4.0), bilirubin minimum 1.0, INR minimum 1.0, Na 125-137 mEq/L. MELD exception points are awarded for conditions that carry waitlist mortality disproportionate to MELD (e.g., HCC — standardized exception score starting at MELD equivalent with 10% 3-month mortality; hepatopulmonary syndrome — PaO2 ≤ 60 mmHg; cholangiocarcinoma — select protocols; portopulmonary hypertension — post-treatment mPAP < 35 mmHg).
Contraindications to Liver Transplant
Absolute: Active extrahepatic malignancy, uncontrolled sepsis, severe cardiopulmonary disease (irreversible), active alcohol or substance use (most programs require 6-month abstinence; "early transplant" protocols exist for select patients with severe alcoholic hepatitis PMID: 22070476), anatomic contraindications (complete portal vein thrombosis extending to SMV — relative), AIDS (relative — HIV+ patients can be transplanted with well-controlled disease).
Relative: Advanced age (> 70), severe portal hypertension, extensive prior abdominal surgery, morbid obesity, poor social support.
King's College Criteria — Acute Liver Failure
Criteria for listing for emergent liver transplant in acute liver failure. Two categories:
| Acetaminophen-Induced ALF | Non-Acetaminophen ALF |
|---|---|
| Arterial pH < 7.30 after resuscitation (regardless of grade of encephalopathy) | INR > 6.5 (PT > 100 sec) regardless of encephalopathy grade |
| OR | |
| All three of: INR > 6.5, creatinine > 3.4 mg/dL, and Grade III-IV encephalopathy | Any three of: (1) Age < 10 or > 40; (2) Etiology: non-A, non-B hepatitis, halothane, idiosyncratic drug reaction; (3) Jaundice > 7 days before encephalopathy; (4) INR > 3.5; (5) Bilirubin > 17.5 mg/dL |
Sensitivity ~70%, specificity ~90% for death without transplant. Additional prognostic markers: arterial lactate > 3.5 mmol/L after early fluid resuscitation (acetaminophen ALF), MELD score > 30, factor V level < 20% (Clichy criteria — used in France).
Hepatopulmonary Syndrome & Portopulmonary Hypertension
Hepatopulmonary syndrome (HPS): Triad of liver disease + intrapulmonary vascular dilatations + impaired oxygenation (PaO2 < 80 mmHg or A-a gradient ≥ 15 mmHg on room air). Diagnosed by contrast-enhanced echocardiography (agitated saline — microbubbles appearing in left atrium after 3-6 cardiac cycles indicates intrapulmonary shunt). Severity: mild (PaO2 ≥ 80), moderate (60-79), severe (50-59), very severe (< 50). Liver transplant is the only definitive treatment — MELD exception granted for PaO2 ≤ 60 mmHg. Post-transplant resolution: > 80% improve, though may take 6-12 months. Very severe HPS (PaO2 < 50) has higher post-transplant mortality.
Portopulmonary hypertension (POPH): Pulmonary arterial hypertension in the setting of portal hypertension. Defined: mPAP ≥ 25 mmHg, PAWP ≤ 15 mmHg, PVR ≥ 240 dynes·sec/cm5. Severity: mild (mPAP 25-34), moderate (35-44), severe (≥ 45). Liver transplant is contraindicated if mPAP ≥ 45 mmHg (high risk of intraoperative RV failure and death) or PVR ≥ 400. MELD exception granted if medical therapy reduces mPAP < 35 mmHg and PVR < 400. Treatment: pulmonary vasodilators (epoprostenol, treprostinil, ambrisentan, sildenafil).
11 Liver Transplant — Surgical Technique
Recipient Hepatectomy
The most challenging phase — extensive portal hypertension, coagulopathy, and adhesions (in redo cases). The operation has three phases: dissection, anhepatic, and neohepatic.
Dissection phase: Bilateral subcostal incision with midline extension (Mercedes/chevron). Mobilization of the liver by dividing falciform, triangular, and coronary ligaments. Hilum dissected: hepatic artery, portal vein, bile duct divided. Two techniques for hepatectomy:
Classical technique (caval replacement): The retrohepatic IVC is resected with the liver, requiring complete vascular isolation — suprahepatic and infrahepatic IVC clamping. Requires venovenous bypass (femoral vein + portal vein → centrifugal pump → axillary vein) to maintain venous return during the anhepatic phase. Used less frequently now.
Piggyback technique (caval preservation): The liver is dissected off the anterior surface of the IVC, preserving the recipient retrohepatic IVC. Hepatic veins are stapled or clamped at their confluence. Advantages: no need for venovenous bypass, hemodynamically better tolerated, shorter anhepatic phase. Now the standard technique at most centers.
Implantation (Anastomoses)
| Anastomosis | Classical | Piggyback |
|---|---|---|
| Suprahepatic IVC | Donor suprahepatic IVC to recipient suprahepatic IVC (end-to-end) | Donor suprahepatic IVC/hepatic veins to recipient hepatic vein confluence (end-to-side or piggyback "common channel") |
| Infrahepatic IVC | Donor infrahepatic IVC to recipient infrahepatic IVC (end-to-end) | Ligated (donor infrahepatic IVC oversewn) |
| Portal vein | End-to-end; growth factor technique if size mismatch; thrombectomy/venoplasty if PVT present | |
| Hepatic artery | End-to-end (most commonly donor celiac to recipient proper/common hepatic artery); interposition graft from donor iliac artery if needed | |
| Bile duct | Duct-to-duct (choledochocholedochostomy) preferred; Roux-en-Y hepaticojejunostomy if duct size mismatch, PSC, or prior biliary surgery | |
Order of anastomosis: Suprahepatic IVC → infrahepatic IVC (classical) or oversew (piggyback) → portal vein → reperfusion (portal then arterial, or simultaneous) → hepatic artery → bile duct. Reperfusion triggers "post-reperfusion syndrome" in ~30%: hypotension, bradycardia, hyperkalemia, metabolic acidosis — due to cold, acidotic, hyperkalemic effluent from the graft. Treatment: calcium chloride, epinephrine, sodium bicarbonate.
Intraoperative Challenges
Coagulopathy management: The anhepatic phase is associated with severe coagulopathy (loss of hepatic synthetic function, fibrinolysis, dilution). Thromboelastography (TEG) or rotational thromboelastometry (ROTEM) guide targeted blood product administration: FFP for prolonged R-time/CT, cryoprecipitate for low fibrinogen (< 150 mg/dL), platelets for low MA/MCF, aminocaproic acid or tranexamic acid for fibrinolysis. Massive transfusion may be required — average blood loss 5-10 units pRBC (less with piggyback technique). Cell salvage is used unless contraindicated (malignancy).
Hemodynamic instability: Portal vein clamping reduces cardiac preload by ~50%. Piggyback technique with partial IVC clamping is better tolerated than complete IVC clamping. Vasopressors (norepinephrine, vasopressin) are commonly required. Post-reperfusion syndrome: defined as ≥ 30% decrease in MAP for ≥ 1 minute within 5 minutes of reperfusion — caused by release of cold, acidotic, hyperkalemic preservation solution and vasoactive mediators from the graft. Prevention: flushing the graft with 500-1,000 mL albumin before portal reperfusion to clear preservation solution.
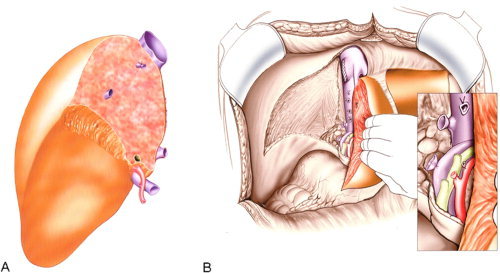
Living Donor Liver Transplant (LDLT)
Right lobe graft (segments V-VIII ± I) — used for adult recipients; requires graft-to-recipient weight ratio (GRWR) ≥ 0.8% (ideally ≥ 1.0%) to avoid small-for-size syndrome. Donor operation: right hepatectomy (preserving middle hepatic vein for donor safety; right hepatic vein and any significant accessory veins reconstructed on bench). Donor risk: mortality ~0.2-0.5%, morbidity ~30% (bile leak most common).
Left lateral segment graft (segments II-III) — used for pediatric recipients; technically simpler, safer for donor.
Split liver transplant: A deceased donor liver is divided: right trisegment (segments I, IV-VIII) for an adult + left lateral segment (segments II-III) for a child. Maximizes organ utilization. In-situ (preferred — lower complication rate) vs. ex-situ splitting.
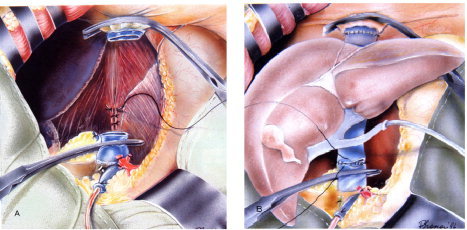
12 Post-Liver Transplant Complications
Primary Non-Function (PNF)
Emergency Defined as graft failure within 7 days requiring retransplantation or causing death. Incidence: 2-6%. Characterized by severe coagulopathy (rising INR), metabolic acidosis, hypoglycemia, encephalopathy, minimal bile production, markedly elevated transaminases (AST/ALT > 5,000). Risk factors: prolonged CIT, DCD donor, severe steatosis (> 30% macrovesicular), elderly donor, hypernatremic donor (Na > 155). Treatment: emergent relisting (UNOS Status 1A) and retransplantation.
Hepatic Artery Thrombosis (HAT)
Emergency The most feared vascular complication and the #1 cause of graft loss from surgical complications. Incidence: 2-5% in adults, up to 10% in children. The bile ducts receive their blood supply exclusively from the hepatic artery (the liver parenchyma has dual supply from portal vein + hepatic artery, but the bile ducts do not) — arterial thrombosis leads to ischemic cholangiopathy, biliary necrosis, liver abscess, graft failure.
Early HAT (< 30 days): Presents with acute graft failure — rising LFTs, coagulopathy, absent arterial flow on Doppler. Treatment: emergent surgical thrombectomy and revision of anastomosis (if < 24 hr); catheter-directed thrombolysis; retransplantation if graft unsalvageable.
Late HAT (> 30 days): May be asymptomatic (collateral formation) or present with biliary strictures, recurrent cholangitis, liver abscess. Treatment: biliary interventions (ERCP/PTC); retransplantation for severe cholangiopathy.
Biliary Complications
Overall incidence: 10-25% ("Achilles heel" of liver transplantation). Two categories:
Anastomotic strictures: Most common biliary complication (~5-15%); usually at the duct-to-duct anastomosis; typically presents 3-6 months post-transplant. Etiology: technical (tension, ischemia, small duct). Treatment: ERCP with balloon dilation + stenting (success ~70-80%); surgical revision (Roux-en-Y hepaticojejunostomy) if refractory.
Non-anastomotic strictures (NAS) / ischemic-type biliary lesions: Intrahepatic diffuse stricturing; etiology: HAT, prolonged CIT/WIT (especially DCD grafts), ABO incompatibility, chronic rejection. Much more difficult to treat — may require retransplantation. Incidence in DCD livers: up to 30% (vs 5-10% in DBD).
Bile leak: 2-10%; usually at cystic duct stump, T-tube site, or anastomosis. Presents with pain, bilious drain output, biloma on imaging. Treatment: ERCP with sphincterotomy + stent placement; percutaneous drainage of biloma.
Portal Vein Thrombosis / Stenosis
Incidence: 1-3%. Presents with graft dysfunction, ascites, variceal bleeding. Diagnosis: Doppler US (absent/reduced portal flow). Treatment: anticoagulation for partial thrombosis; surgical thrombectomy/revision for early complete thrombosis; TIPS for late stenosis; balloon angioplasty ± stenting.
Hepatic Vein Outflow Obstruction
More common with piggyback technique (0.5-3%): anastomotic stenosis or kinking of hepatic veins. Presents like Budd-Chiari: ascites, hepatomegaly, graft dysfunction. Diagnosis: Doppler US (monophasic hepatic vein waveforms, reduced/absent flow). Treatment: balloon angioplasty ± stenting via transjugular approach; surgical revision.
Early Allograft Dysfunction (EAD)
Defined as the presence of one or more of: (1) bilirubin ≥ 10 mg/dL on post-operative day 7; (2) INR ≥ 1.6 on POD 7; (3) peak AST or ALT > 2,000 within the first 7 days. EAD is distinct from PNF — the graft functions but poorly. Incidence: 20-30%. Risk factors: donor age, steatosis, prolonged CIT, DCD status. EAD is associated with increased mortality, graft loss, and longer ICU/hospital stay. The L-GrAFT score (Liver Graft Assessment Following Transplantation) uses daily AST, ALT, bilirubin, INR, and platelet count during the first 10 post-operative days to predict 3-month and 12-month graft failure risk — allowing early identification of grafts in trouble.
Metabolic Complications After Liver Transplant
Metabolic syndrome: Extremely common post-liver transplant — obesity (BMI increase of 2-5 kg/m2 in first year), diabetes (PTDM ~15-30%), hypertension (~60-70%), dyslipidemia (~45-70%). Contributed to by immunosuppressive agents (steroids, CNIs). Management: lifestyle modification, statin therapy (pravastatin preferred — least CYP3A4 interaction; avoid simvastatin/lovastatin with CNIs), ACE inhibitors/ARBs for hypertension (additional benefit of reducing proteinuria and PTDM-associated kidney disease).
Chronic kidney disease (CKD): GFR < 60 mL/min develops in ~20% of liver transplant recipients by 5 years, primarily due to CNI nephrotoxicity. Prevention: minimize CNI levels over time, use of mTOR inhibitors as CNI-sparing agents (SPARE-the-Nephron trial: everolimus + reduced tacrolimus improved GFR at 12 months vs. standard tacrolimus). CKD post-liver transplant is associated with significantly increased mortality — combined liver-kidney transplant is performed when GFR is < 30 mL/min for > 90 days or on dialysis ≥ 6 weeks.
Bone disease: Pre-existing hepatic osteodystrophy (especially in cholestatic liver diseases) is worsened by corticosteroids. Bone density nadir occurs at 3-6 months post-transplant. Prevention: calcium, vitamin D, bisphosphonates (or denosumab). Avascular necrosis (AVN) of the femoral head occurs in ~5-10%, related to corticosteroid use.
13 Liver Transplant — Rejection & Recurrent Disease
Acute Cellular Rejection (ACR) — Banff RAI Score
Incidence: 15-30% in the first year. Diagnosed on biopsy using the Rejection Activity Index (RAI), which scores three histologic components:
| Component | Score 0 | Score 1 | Score 2 | Score 3 |
|---|---|---|---|---|
| Portal inflammation | None | Mild — mostly lymphocytic, confined to some portal tracts | Moderate — mixed infiltrate in most portal tracts | Severe — dense infiltrate expanding most portal tracts, spillover into periportal parenchyma |
| Bile duct damage | None | Minority of ducts — degenerative changes (nuclear pleomorphism, cytoplasmic vacuolization) | Most/all ducts — degenerative changes | Most/all ducts — degenerative changes with duct disruption or luminal sloughing |
| Venous endotheliitis | None | Mild — subendothelial lymphocytic infiltrate involving some portal or hepatic venules | Moderate — subendothelial infiltrate involving most portal/hepatic venules | Severe — moderate to severe perivenular hepatocyte necrosis involving most hepatic venules |
Interpretation: RAI 0-2 = no rejection; RAI 3 = borderline/indeterminate; RAI 4-5 = mild ACR; RAI 6-7 = moderate ACR; RAI 8-9 = severe ACR. Treatment: mild-moderate ACR responds to pulse methylprednisolone (1 g IV daily × 3 days) in > 80% of cases. Steroid-resistant ACR: thymoglobulin.
Chronic Ductopenic Rejection
Characterized by progressive loss of interlobular bile ducts (ductopenia — loss of bile ducts in ≥ 50% of portal tracts on biopsy). Leads to chronic cholestasis and graft failure. Incidence: < 5% (decreased with modern immunosuppression). Often the result of untreated or undertreated acute rejection. Treatment: optimize immunosuppression; conversion to tacrolimus (if on cyclosporine); sirolimus addition; retransplantation for advanced ductopenic rejection.
Antibody-Mediated Rejection in Liver
Historically considered rare due to the liver's resistance to AMR (Kupffer cells clear circulating antibodies, large sinusoidal surface area). However, increasingly recognized — criteria (Banff 2016): (1) histopathology (portal/periportal edema, ductular reaction, portal and/or central microvascular inflammation, C4d staining of portal microvessels); (2) DSA positive; (3) exclusion of other causes. More common in ABO-incompatible transplants, highly sensitized recipients, and crossmatch-positive transplants.
Recurrent Disease
Hepatitis C: Historically, universal recurrence (100%) post-transplant with accelerated fibrosis; now effectively cured with direct-acting antivirals (DAAs — sofosbuvir/velpatasvir, glecaprevir/pibrentasvir) with > 95% SVR rate. Treat pre- or post-transplant.
Hepatitis B: Prevented with combination of hepatitis B immune globulin (HBIG) + nucleos(t)ide analogue (entecavir or tenofovir) — recurrence < 5%. Some centers now use HBIG-free protocols with potent antivirals. HBV recurrence is defined as reappearance of HBsAg and/or detectable HBV DNA in serum. De novo hepatitis B can occur in HBV-naive recipients receiving grafts from anti-HBc positive donors (occult infection in the graft) — these recipients require lifelong antiviral prophylaxis.
Autoimmune hepatitis: Recurrence 20-40%; de novo AIH also occurs.
PSC: Recurrence 20-25%; diagnosis of exclusion (cholangiographic findings of intrahepatic biliary strictures without HAT or other cause). No proven prophylaxis.
NASH/MASLD: Recurrence common (60-100% steatosis, 20-40% steatohepatitis by 5 years), driven by metabolic risk factors + immunosuppressive agents.
Alcohol-associated liver disease: Relapse to heavy drinking ~15-20%; graft loss from recurrent alcoholic liver disease ~5-10% at 5 years.
De Novo Malignancy After Liver Transplant
In addition to PTLD (see Section 24), liver transplant recipients are at increased risk for de novo solid organ malignancies. Colorectal cancer screening is particularly important in PSC patients (often have concurrent ulcerative colitis with high CRC risk — annual colonoscopy recommended). Skin cancer risk is elevated as in all transplant recipients. Oropharyngeal and esophageal cancers are increased, especially with alcohol and tobacco history. HCC can develop de novo in the new graft, particularly if underlying risk factors persist (ongoing HBV/HCV, NASH). Overall cancer incidence: ~10-15% at 10 years post-liver transplant.
Operational Tolerance in Liver Transplantation
The liver is considered an "immunoprivileged" organ — it has lower rejection rates than kidney or heart, and ~20% of select liver transplant recipients can achieve operational tolerance (complete withdrawal of immunosuppression with stable graft function). Characteristics favoring tolerance: long time since transplant (> 5-10 years), older recipient age, single HLA mismatch, absence of autoimmune etiology. Prospective trials (WISP-R, iWITH, LIFT) are systematically attempting immunosuppression withdrawal in stable recipients. Biomarkers predicting tolerance: specific gene expression signatures in peripheral blood (iron metabolism genes, NK cell genes), regulatory T cell profiles, and liver biopsy findings (absence of inflammation/fibrosis). Operational tolerance remains experimental — immunosuppression withdrawal should only be attempted in formal clinical trials or expert centers with close monitoring.
14 HCC & Liver Transplantation
Milan Criteria
The gold standard for HCC transplant eligibility, proposed by Mazzaferro et al. (1996) PMID: 8632270:
| Parameter | Milan Criteria |
|---|---|
| Single tumor | ≤ 5 cm diameter |
| Multiple tumors | ≤ 3 nodules, each ≤ 3 cm |
| Macrovascular invasion | Absent |
| Extrahepatic spread | Absent |
| 5-year survival | ~70-75% |
| Recurrence rate | < 15% |
UCSF Criteria (Expanded)
Proposed by Yao et al.: single tumor ≤ 6.5 cm, OR ≤ 3 nodules with largest ≤ 4.5 cm and total tumor diameter ≤ 8 cm, with no macrovascular invasion. 5-year survival comparable to Milan (~70%). Not used for UNOS allocation but supported by some centers and validates expanded criteria.
Downstaging Protocols
Patients beyond Milan criteria may undergo locoregional therapy to reduce tumor burden within Milan, then be listed for transplant. UNOS downstaging criteria (since 2017): single tumor 5.1-8 cm, OR 2-3 tumors each ≤ 5 cm with total ≤ 8 cm, OR 4-5 tumors each ≤ 3 cm with total ≤ 8 cm. Must achieve within Milan for ≥ 6 months to receive MELD exception. Successful downstaging yields outcomes comparable to patients initially within Milan.
Bridging / Locoregional Therapy
Used to treat HCC while awaiting transplant (prevent progression and dropout). Options: transarterial chemoembolization (TACE) — most commonly used; transarterial radioembolization (TARE/Y-90); radiofrequency ablation (RFA) or microwave ablation (MWA) — best for tumors < 3 cm; stereotactic body radiation (SBRT). Bridging recommended when expected wait time > 6 months.
RETREAT Score — Post-Transplant Recurrence
The RETREAT (Risk Estimation of Tumor Recurrence After Transplant) score predicts HCC recurrence post-transplant based on explant pathology:
| Variable | Points |
|---|---|
| Microvascular invasion | +1 |
| Largest viable tumor diameter (per cm beyond 1 cm) | +1 per cm |
| AFP at transplant > 20 ng/mL | +2 |
| AFP at transplant > 200 ng/mL | +3 |
| AFP at transplant > 1,000 ng/mL | +4 |
Score 0 = < 3% recurrence at 5 years; score ≥ 5 = > 75% recurrence at 5 years. PMID: 28187827
AFP Thresholds in HCC & Transplant Listing
Alpha-fetoprotein (AFP) is the most commonly used biomarker for HCC. In transplant candidacy, AFP levels influence eligibility and prognosis:
| AFP Level | Clinical Significance |
|---|---|
| < 20 ng/mL | Low risk; favorable post-transplant prognosis |
| 20-200 ng/mL | Moderate risk; acceptable for transplant listing within Milan |
| 200-1,000 ng/mL | Higher risk; consider biological behavior (response to locoregional therapy, AFP trend) |
| > 1,000 ng/mL | High risk for microvascular invasion and recurrence; UNOS policy (since 2023): AFP > 1,000 disqualifies for MELD exception UNLESS AFP decreases to ≤ 500 with treatment |
AFP response to locoregional therapy is a powerful predictor of biology — tumors that respond (AFP decline > 50%) have better post-transplant outcomes than non-responders. Total tumor volume (TTV) combined with AFP has been proposed as a more accurate selection tool than Milan criteria alone.
15 Pancreas Transplantation
Categories
| Type | Definition | % of Pancreas Tx | 1-Year Graft Survival |
|---|---|---|---|
| SPK (Simultaneous Pancreas-Kidney) | Pancreas + kidney from same donor, performed simultaneously | ~75% | ~87-90% |
| PAK (Pancreas After Kidney) | Pancreas transplant in patient with prior functioning kidney transplant | ~15% | ~80-85% |
| PTA (Pancreas Transplant Alone) | Pancreas transplant in patient with preserved native kidney function | ~10% | ~75-80% |
Indications: Type 1 diabetes mellitus (primary); select type 2 DM with low C-peptide; hypoglycemia unawareness (brittle diabetes). SPK is the most common and most successful. Goal: insulin independence and prevention of secondary diabetic complications.
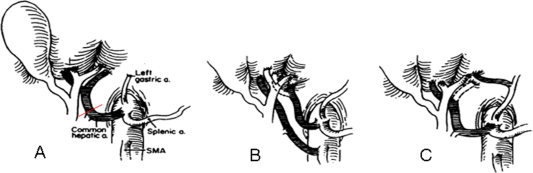
Surgical Technique
Donor procurement: En bloc pancreas with duodenal segment (C-loop — 2nd portion of duodenum), spleen, and vascular pedicles (SMA, splenic artery). Bench preparation: Y-graft reconstruction — donor iliac artery bifurcation graft anastomosed to SMA and splenic artery, creating a single arterial inflow.
Exocrine drainage:
Enteric drainage (preferred, ~90% of cases): Donor duodenal segment anastomosed to recipient jejunum (side-to-side). More physiologic; lower metabolic complications.
Bladder drainage (historic, ~10%): Donor duodenal segment anastomosed to recipient bladder. Allows urinary amylase monitoring for rejection. Complications: hematuria, metabolic acidosis (bicarbonate loss), UTIs, chemical cystitis, urethritis. Often converted to enteric drainage.
Venous drainage:
Systemic venous drainage: Donor portal vein to recipient iliac vein or IVC. Results in peripheral hyperinsulinemia (bypasses hepatic first-pass).
Portal venous drainage: Donor portal vein to recipient SMV. More physiologic insulin delivery but technically more challenging. No clear long-term survival benefit demonstrated.
Rejection Monitoring
Pancreas rejection is difficult to detect — no equivalent of serum creatinine. For SPK: kidney rejection (rising creatinine) is a surrogate marker (both organs rejected simultaneously). For PAK/PTA: serum lipase (not very sensitive), serum amylase (if bladder-drained — urine amylase decline), glucose elevation (late sign — > 80% islet loss by the time hyperglycemia occurs). Protocol biopsies (percutaneous or endoscopic of duodenal segment) provide definitive diagnosis. Rejection rate: ~15-20% for SPK, higher for PAK/PTA.
Outcomes & Survival
SPK has the best pancreas graft survival of all three categories. IPTR data: SPK 1-year pancreas graft survival ~87-90%, 5-year ~73-80%; PAK 1-year ~80-85%, 5-year ~60-65%; PTA 1-year ~75-80%, 5-year ~55-60%. Patient survival is excellent (> 95% at 1 year for all categories). SPK provides superior patient survival compared to remaining on dialysis with continued insulin therapy. Successful pancreas transplantation normalizes HbA1c (typically < 6%), prevents recurrence of diabetic nephropathy in the kidney graft, stabilizes or improves diabetic neuropathy and retinopathy (though benefit takes years), and significantly improves quality of life.
Complications Specific to Pancreas Transplantation
| Complication | Incidence | Management |
|---|---|---|
| Graft thrombosis | 5-10% (most common cause of early graft loss) | Anticoagulation prophylaxis (heparin drip, then aspirin + heparin); surgical thrombectomy if early; graft pancreatectomy if nonviable |
| Pancreatitis (allograft) | 10-20% | Usually self-limited; NPO, IV fluids; may mimic rejection |
| Enteric leak | 5-10% (enteric-drained) | Surgical exploration and repair; may require graft pancreatectomy |
| Bladder complications (bladder-drained) | 20-30% | Chemical cystitis, UTI, hematuria, metabolic acidosis (HCO3 loss); enteric conversion in ~15-25% |
| Peripancreatic abscess/infection | 5-10% | CT-guided drainage; antibiotics; surgical debridement if needed |
16 Islet Cell Transplantation
Islet Isolation
The pancreas is digested with collagenase (Liberase or collagenase blend) via intraductal infusion, followed by mechanical dissociation and density gradient purification (COBE 2991 cell processor). Goal: obtain ≥ 5,000 islet equivalents (IEQ) per kg recipient body weight (typically requires 2-3 donor pancreases). Islet purity: ideally > 50%. Viability testing: fluorescein diacetate/propidium iodide staining.
Edmonton Protocol
Landmark protocol (Shapiro et al., 2000) PMID: 10911004 that achieved insulin independence in 7/7 patients. Key innovations:
(1) Steroid-free immunosuppression: daclizumab induction (anti-CD25) + sirolimus + low-dose tacrolimus — avoided diabetogenic corticosteroids.
(2) Adequate islet mass: ≥ 10,000 IEQ/kg from 2+ donors (fresh, immediate transplantation).
(3) Intraportal infusion: Islets infused into the portal vein via percutaneous transhepatic approach under fluoroscopic guidance; islets lodge in hepatic sinusoids and revascularize.
Outcomes
Short-term: insulin independence achieved in 50-70% at 1 year. Long-term: only 10-20% remain insulin independent at 5 years (progressive islet loss). However, C-peptide positivity (graft function) persists in 80%+ at 5 years — providing glycemic stability, reduced hypoglycemia, and improved HbA1c. Complications: portal vein thrombosis (~5%), bleeding (~10% — usually self-limited), hepatic steatosis at infusion sites.
Emerging Approaches
Stem cell-derived beta cells: Pluripotent stem cells (iPSC or ESC) differentiated into insulin-producing beta-like cells. Phase I/II trials show promise — VX-880 (Vertex Pharmaceuticals) demonstrated insulin independence in early patients with type 1 diabetes (requires immunosuppression). VX-264 uses an encapsulation device to protect cells from immune attack, potentially eliminating the need for immunosuppression.
Encapsulation strategies: Macro- or micro-encapsulation of islets in biocompatible materials (alginate, PEG) to create an immune barrier while allowing glucose/insulin diffusion. Challenges: foreign body response, fibrosis around capsule, oxygen diffusion limitation. The ViaCyte PEC-Encap and Beta-O2 devices are in clinical trials.
Alternative sites: Research explores non-hepatic sites for islet delivery to avoid the instant blood-mediated inflammatory reaction (IBMIR) that destroys ~50% of infused islets in the portal system. Sites under investigation: subcutaneous (with prevascularization), omentum (pouch technique — phase I/II trials showing safety), bone marrow, muscle.
17 Heart Transplantation
Indications & Evaluation
Primary indication: advanced/end-stage heart failure (NYHA Class III-IV) refractory to maximal medical therapy. Most common etiologies: dilated cardiomyopathy (~50%), ischemic cardiomyopathy (~40%), other (restrictive, congenital, valvular, retransplant). VO2 max < 12 mL/kg/min (on beta-blocker) or < 14 mL/kg/min (without beta-blocker) predicts poor 1-year survival without transplant. Heart failure survival score (HFSS) and SHFM (Seattle Heart Failure Model) aid prognostication.
Key evaluation elements: Right heart catheterization (must assess pulmonary vascular resistance — PVR > 5 Wood units or transpulmonary gradient > 15 mmHg = risk of acute RV failure post-transplant; if elevated, test reversibility with nitroprusside/milrinone/iNO); cardiopulmonary exercise testing; panel reactive antibody (highly sensitized patients — virtual crossmatch); psychosocial evaluation.
UNOS Heart Allocation (revised 2018)
Six-tier system (Status 1-6) replacing the prior three-tier system. Accounts for mechanical circulatory support device type, temporary support, and clinical acuity. Status 1-3 require evidence of hemodynamic compromise or device complications. Geographic allocation: 250-nautical-mile zones. Median wait time: ~4-6 months.
Bridging with Ventricular Assist Devices (VAD)
Approximately 40-50% of heart transplant recipients are bridged with a VAD. LVAD (left ventricular assist device) — continuous-flow devices (HeartMate 3 — centrifugal flow, fully magnetically levitated impeller; now the dominant device). Bridge-to-transplant (BTT) outcomes: > 80% survival to transplant. Post-transplant outcomes for VAD-bridged patients are comparable to non-bridged patients.
Key Surgical Considerations
Orthotopic transplant — bicaval technique (standard) superior to biatrial (less tricuspid regurgitation, fewer arrhythmias, better hemodynamics). Cold ischemia time limit: < 4 hours. Denervated heart: resting HR ~90-110 (no vagal tone), does not respond to atropine or carotid sinus massage, delayed chronotropic response to exercise (reliance on circulating catecholamines). Outcomes: median survival ~12-14 years; 1-year survival ~90%, 5-year ~80%. Leading cause of death at 1 year: rejection/infection; at 5+ years: cardiac allograft vasculopathy (CAV) — a diffuse, concentric intimal proliferative process affecting the entire coronary tree (not amenable to conventional PCI/CABG). PMID: 29187358
Cardiac Allograft Vasculopathy (CAV)
CAV is the leading cause of late graft loss and death after the first year. Unlike conventional atherosclerosis (focal, eccentric, proximal), CAV is diffuse, concentric, and affects the entire coronary tree including intramyocardial branches — making it largely not amenable to PCI or CABG. Incidence: ~30% at 5 years, ~50% at 10 years by angiography. ISHLT CAV grading: CAV 0 (none), CAV 1 (mild — left main < 50%, primary vessel < 70%, no graft dysfunction), CAV 2 (moderate — left main ≥ 50%, or single primary ≥ 70%, or branch stenosis ≥ 70% in 2 systems), CAV 3 (severe — meets CAV 2 criteria plus graft dysfunction). Pathogenesis: immune-mediated (both cellular and humoral — chronic rejection of the endothelium) with contribution from traditional cardiovascular risk factors. Prevention: statins (started universally post-transplant — pravastatin and simvastatin showed reduction in CAV in landmark trials), mTOR inhibitors (everolimus reduces intimal thickening in randomized trials). Monitoring: annual coronary angiography or intravascular ultrasound (IVUS — more sensitive, detects early intimal thickening). Treatment: limited options — retransplantation for severe CAV with graft dysfunction.
Heart Transplant Rejection — Endomyocardial Biopsy
Protocol endomyocardial biopsies remain the gold standard for rejection surveillance: typically performed weekly for 6 weeks, biweekly for 3 months, monthly to 6 months, then every 3-6 months in year 2, and annually thereafter. Accessed via right internal jugular vein; bioptome directed to the RV septum under fluoroscopic guidance. Minimum 3-4 pieces of myocardium required. ISHLT grading: 0R (no rejection), 1R (mild — focal/diffuse infiltrate without myocyte damage), 2R (moderate — multifocal infiltrate with myocyte damage), 3R (severe — diffuse infiltrate with myocyte necrosis). Treatment: 1R — no treatment usually; 2R — pulse steroids ± adjustment of maintenance IS; 3R — pulse steroids + rATG; hemodynamic compromise — mechanical support.
18 Lung Transplantation
Indications
| Disease | % of Lung Tx | Type of Tx | Key Notes |
|---|---|---|---|
| COPD/emphysema | ~25% | Single or bilateral | Bilateral preferred (better survival); native hyperinflation with single lung (contralateral mediastinal shift) |
| Idiopathic pulmonary fibrosis (IPF) | ~25% | Single or bilateral | Bilateral increasingly preferred; single lung acceptable with LAS benefit |
| Cystic fibrosis (CF) | ~15% | Bilateral only | Bilateral mandatory (infected native lungs would contaminate a single allograft); Burkholderia cenocepacia = relative contraindication |
| Pulmonary arterial hypertension (PAH) | ~5% | Bilateral | Bilateral preferred; heart-lung for Eisenmenger with irreversible cardiac defect |
| Alpha-1 antitrypsin deficiency | ~5% | Single or bilateral | Bilateral preferred |
| Other (ILD, sarcoidosis, LAM, bronchiectasis) | ~25% | Variable | Disease-specific considerations |
Lung Allocation Score (LAS)
The LAS (range 0-100) determines allocation priority, balancing waitlist urgency (estimated days of life without transplant) and post-transplant survival (estimated days of life with transplant). Calculated from: diagnosis, FVC, 6-minute walk distance, O2 requirement, pulmonary artery pressures, PCO2, BMI, diabetes, functional status, age, creatinine. Higher LAS = higher priority. Replaced time-based allocation in 2005 — reduced waitlist mortality and wait times. PMID: 16818847
Primary Graft Dysfunction (PGD)
Lung-specific form of ischemia-reperfusion injury; the leading cause of early mortality. Graded by ISHLT consensus (PaO2/FiO2 ratio at T0, T24, T48, T72 hours):
| PGD Grade | PaO2/FiO2 | Radiographic Infiltrates |
|---|---|---|
| Grade 0 | > 300 | Absent |
| Grade 1 | > 300 | Present |
| Grade 2 | 200-300 | Present |
| Grade 3 | < 200 | Present |
PGD grade 3 at 72 hours: mortality ~40-50%; risk factors include donor smoking, recipient PAH, high BMI, CPB use. Treatment: supportive (protective ventilation, iNO, ECMO for refractory cases). PGD grade 3 at 72 hours is also a risk factor for chronic rejection (CLAD).
Chronic Lung Allograft Dysfunction (CLAD)
Affects ~50% of lung transplant recipients by 5 years — the major limitation to long-term survival. Two phenotypes:
Bronchiolitis obliterans syndrome (BOS): Most common form (~70% of CLAD). Obstructive physiology — progressive decline in FEV1 to < 80% of baseline (BOS 1), < 65% (BOS 2), < 50% (BOS 3). Pathology: obliterative bronchiolitis (fibrotic obliteration of small airways). Treatment: azithromycin (anti-inflammatory — ~30% respond), montelukast, photopheresis, total lymphoid irradiation, re-transplantation. Median survival after BOS onset: ~3-5 years.
Restrictive allograft syndrome (RAS): ~30% of CLAD. Restrictive physiology — decline in TLC to < 90% of baseline with upper lobe-predominant infiltrates/fibrosis on CT. More aggressive course than BOS; median survival ~1.5 years after onset. No effective treatment. Pathology: pleuroparenchymal fibroelastosis.
Lung Transplant — Surgical Technique
Single lung: Posterolateral thoracotomy (4th-5th intercostal space). Pneumonectomy of the native lung; anastomoses: bronchial (end-to-end), pulmonary artery (end-to-end), pulmonary venous cuff (left atrial cuff of donor to recipient left atrium). Bronchial anastomosis is the most vulnerable — blood supply depends on bronchial artery collaterals (not reanastomosed); telescope technique may improve healing.
Bilateral sequential: Bilateral anterolateral thoracotomies (clamshell incision — transverse sternotomy connecting both thoracotomies) or bilateral anterior thoracotomies without sternal transection. Each lung transplanted sequentially (worse lung first). CPB/ECMO standby required — used in ~30-40% (for PAH, hemodynamic instability, or severe V/Q mismatch during single-lung ventilation).
19 Intestinal & Multivisceral Transplantation
Indications
Primary indication: irreversible intestinal failure with life-threatening complications of total parenteral nutrition (TPN). Specific criteria for transplant listing: (1) loss of ≥ 2 central venous access sites; (2) recurrent line sepsis (≥ 2 episodes/year of sepsis or single episode of fungemia); (3) TPN-associated liver disease (progressive fibrosis/cirrhosis); (4) severe dehydration despite IV supplementation. Underlying conditions: short bowel syndrome (most common — post-surgical from mesenteric ischemia, NEC, volvulus, Crohn's, trauma), motility disorders (chronic intestinal pseudo-obstruction), mucosal diseases (microvillus inclusion disease).
Types of Intestinal Transplantation
| Type | Organs Included | Indication |
|---|---|---|
| Isolated intestinal | Small bowel only | Intestinal failure with preserved liver function |
| Combined liver-intestine | Liver + small bowel (en bloc or separate) | Intestinal failure with TPN-associated liver disease |
| Multivisceral | Stomach + duodenum + pancreas + small bowel ± liver ± colon | Diffuse splanchnic disease (mesenteric thrombosis, hollow visceral myopathy, desmoid tumors) |
| Modified multivisceral | Multivisceral without liver | Multivisceral indications with preserved liver function |
Outcomes & Challenges
Intestinal transplantation has the highest rejection rate of any solid organ transplant (~30-50% in the first year) due to the massive lymphoid tissue in the gut (GALT — gut-associated lymphoid tissue, Peyer's patches). One-year graft survival: ~70-80%; 5-year: ~50-60%. Monitoring: protocol ileoscopy with zoom endoscopy and biopsies (villous architecture, apoptotic bodies, inflammatory infiltrate). Acute rejection histology: villous blunting, crypt cell apoptosis (≥ 6 apoptotic bodies/10 crypts = moderate rejection), mucosal denudation (severe). Other complications: GVHD (donor lymphocytes attack recipient — 5-10%), infection, PTLD (highest risk of all solid organ transplants — up to 20%), graft-versus-host disease. Citrulline levels correlate with functioning enterocyte mass and are used as a biomarker for rejection (citrulline < 10 μmol/L suggests significant mucosal injury).
Intestinal Rejection — Grading
| Grade | Histologic Findings | Management |
|---|---|---|
| Indeterminate | Mild increase in crypt apoptosis; minimal inflammation | Observation, repeat biopsy in 3-5 days |
| Mild | Increased crypt apoptosis (6-10 per 10 crypts); mild villous blunting; mild inflammation | Increase maintenance IS; pulse steroids |
| Moderate | Crypt injury with focal dropout; moderate villous blunting and inflammation; > 10 apoptotic bodies per 10 crypts | Pulse steroids; consider rATG |
| Severe | Extensive crypt damage/loss; mucosal ulceration; severe villous blunting to denudation | rATG; consider graft enterectomy if unresponsive |
Surgical Technique — Intestinal Transplant
Isolated intestinal transplant: Donor SMA anastomosed to recipient infrarenal aorta (Carrel patch or direct); donor portal vein to recipient SMV (or systemic drainage to IVC). Intestinal anastomosis: proximal — donor jejunum to recipient jejunum/duodenum; distal — Bishop-Koop or end ileostomy (allows endoscopic surveillance). The ileostomy is essential for monitoring — allows direct visualization and biopsy for rejection surveillance.
Multivisceral transplant: En bloc graft including stomach, duodenum, pancreas, and small bowel (with or without liver and colon). Arterial supply: aortic conduit carrying the celiac trunk and SMA. Venous drainage: donor portal vein to recipient IVC (if liver included — caval replacement or piggyback). Gastrointestinal continuity restored with gastrogastrostomy proximally and ileostomy or ileocolostomy distally.
Post-Intestinal Transplant Nutrition & Adaptation
After intestinal transplant, enteral nutrition is initiated early (within days) via feeding tube or oral intake, starting with elemental formula and advancing gradually. Full enteral autonomy (complete TPN independence) is achieved in ~70-80% of patients by 6-12 months. Monitoring: stool output (> 2 L/day may indicate rejection), weight, nutritional labs (prealbumin, zinc, magnesium, fat-soluble vitamins). The transplanted intestine undergoes adaptive responses — villous hypertrophy and functional upregulation of absorptive capacity — over the first 1-2 years. Inclusion of the ileocecal valve and colon (when feasible) improves fluid and electrolyte absorption.
Retransplantation
Retransplantation is considered when graft failure is irreversible. Organ-specific considerations: (1) Kidney retransplantation is common (~15% of kidney transplants are retransplants); sensitization from prior graft increases immunologic risk. Failed allograft nephrectomy is performed if symptomatic (pain, hematuria, chronic rejection syndrome) or if discontinuation of immunosuppression is planned (prevent sensitization flare). (2) Liver retransplantation: ~5-10% of liver transplants; indications: PNF, HAT, chronic rejection, recurrent disease. Technically more challenging (adhesions, portal hypertension, coagulopathy). Outcomes: inferior to primary transplant (~60-70% 1-year survival). (3) Heart retransplantation: ~2-4% of heart transplants; main indication: CAV with graft failure; outcomes depend on timing (> 1 year from primary: better; early retransplant for PGD: worse). (4) Lung retransplantation: controversial; main indication: CLAD; limited by organ scarcity and ethical considerations regarding resource allocation.
20 Induction Therapy
Overview
Induction therapy provides intense immunosuppression at the time of transplant — the period of highest immunologic risk. Used in > 80% of kidney transplants, less commonly in liver transplants (lower immunologic risk). Two categories: depleting and non-depleting agents.
| Agent | Type | Mechanism | Primary Use | Duration of Effect |
|---|---|---|---|---|
| Basiliximab | Non-depleting | Anti-CD25 (IL-2Rα) | Low-risk kidney Tx | ~4-6 weeks |
| rATG (Thymoglobulin) | Depleting (T cell) | Polyclonal anti-thymocyte | High-risk kidney Tx, DGF anticipated | Weeks-months |
| Alemtuzumab | Depleting (pan-lymphocyte) | Anti-CD52 | Off-label; minimization protocols | Months (CD4 recovery 6-12 mo) |
Choice of induction agent is risk-stratified: depleting agents (rATG) for sensitized patients, retransplants, AA race recipients, and when DGF is anticipated (allows delayed CNI introduction — reduces additional nephrotoxic insult to the ischemic graft). Non-depleting agent (basiliximab) for low-risk first transplants with living donors.
Non-Depleting Agents
Basiliximab (Simulect): Chimeric murine-human monoclonal antibody targeting the IL-2 receptor α-chain (CD25) on activated T cells. Blocks IL-2-driven T-cell proliferation (Signal 3). Dose: 20 mg IV on day 0 and day 4. Well-tolerated, minimal side effects. Saturates IL-2 receptors for ~4-6 weeks. Used preferentially in low-immunologic-risk kidney transplants. Does NOT cause cytokine release syndrome. PMID: 11794211
Depleting Agents
Rabbit anti-thymocyte globulin (rATG / Thymoglobulin): Polyclonal antibody derived from rabbits immunized with human thymocytes. Causes profound T-cell depletion (CD3+ count < 50 cells/μL). Dose: 1.5 mg/kg/day IV for 3-7 days (total dose 4.5-10.5 mg/kg). Preferred for high-immunologic-risk patients (sensitized, retransplant, AA race, DGF anticipated — allows delayed CNI introduction). Side effects: cytokine release syndrome (fever, rigors, hypotension — premedicate with methylprednisolone, diphenhydramine, acetaminophen), leukopenia, thrombocytopenia, serum sickness, increased infection/malignancy risk. Monitor: CD3+ count, CBC (hold dose if WBC < 2,000 or platelets < 50,000). PMID: 10071074
Alemtuzumab (Campath-1H): Humanized monoclonal antibody targeting CD52 (expressed on T cells, B cells, NK cells, monocytes). Causes profound and prolonged lymphocyte depletion. Dose: 30 mg IV single dose (or 20 mg × 2). Recovery of CD4+ T cells takes 6-12 months; B cells recover faster. Used off-label in transplantation — enables minimization protocols (early steroid withdrawal, tacrolimus monotherapy). Risk: increased opportunistic infections, PTLD, autoimmune cytopenias.
21 Maintenance Immunosuppression
Triple Therapy — Standard Regimen
The cornerstone of transplant immunosuppression is triple therapy: calcineurin inhibitor (CNI) + antimetabolite + corticosteroids.
| Drug | Mechanism | Target Trough Level | Key Side Effects |
|---|---|---|---|
| Tacrolimus (FK506) | Binds FKBP-12 → inhibits calcineurin → blocks NFAT → suppresses IL-2 transcription | Months 0-3: 8-12 ng/mL Months 3-12: 6-10 ng/mL > 12 months: 5-8 ng/mL | Nephrotoxicity (#1), PTDM, tremor, alopecia, hyperkalemia, hypomagnesemia, neurotoxicity (PRES), diarrhea |
| Mycophenolate mofetil (MMF/CellCept) | Prodrug of MPA; inhibits IMPDH → blocks de novo purine synthesis (lymphocytes lack salvage pathway) | MPA AUC 30-60 μg·h/mL (if monitored); dose 1 g BID | GI (diarrhea, nausea — #1 reason for dose reduction), leukopenia, anemia, teratogenicity (pregnancy category X) |
| Prednisone | Blocks NF-κB and AP-1 → suppresses cytokine transcription; blocks prostaglandins | Dose-based (not level-based): 200 mg post-op taper to 5-10 mg/day by month 1-3; some protocols withdraw by 1 week | PTDM, HTN, hyperlipidemia, osteoporosis, cataracts, weight gain, adrenal insufficiency, infection, AVN, growth retardation (pediatric) |
Calcineurin Inhibitor Toxicity
Acute CNI nephrotoxicity: Afferent arteriolar vasoconstriction (reversible); isometric vacuolization of proximal tubular cells on biopsy. Chronic CNI nephrotoxicity: "Striped" interstitial fibrosis (alternating areas of fibrosis and normal tissue), arteriolar hyalinosis (nodular — pathognomonic), global glomerulosclerosis, tubular atrophy. A major contributor to chronic allograft nephropathy. Management: dose reduction, conversion to belatacept or mTOR inhibitor.
Belatacept (Nulojix) — Costimulation Blockade
Belatacept is a fusion protein (CTLA4-Ig) that blocks the B7-CD28 costimulatory pathway (Signal 2). Given IV monthly (5 mg/kg initially, then 5 mg/kg every 4 weeks). Advantages: no nephrotoxicity, improved long-term GFR, better cardiovascular risk profile, no drug level monitoring. BENEFIT trial: 7-year data showed superior GFR with belatacept vs. cyclosporine (+16 mL/min) with comparable graft/patient survival. PMID: 26816011. Disadvantages: higher rate of acute rejection (especially Banff ≥ IIA), requires IV infusion, contraindicated in EBV-seronegative recipients (10x higher PTLD risk — black box warning). Only approved for kidney transplant.
mTOR Inhibitors
Sirolimus (rapamycin) and everolimus: Bind FKBP-12 (same binding protein as tacrolimus, but different downstream target) → inhibit mTOR → block cell cycle progression G1→S phase. Not nephrotoxic (but may potentiate CNI nephrotoxicity if co-administered — typically used as CNI-sparing or CNI-free). Target trough: sirolimus 5-15 ng/mL; everolimus 3-8 ng/mL. Side effects: impaired wound healing (avoid early post-transplant or with BMI > 30), hyperlipidemia, proteinuria, oral ulcers, pneumonitis, cytopenias, lymphocele. Antitumor properties — used preferentially in patients with history of or high risk for malignancy (skin cancer, Kaposi sarcoma).
Other Maintenance Agents
Azathioprine (Imuran): Purine analogue; prodrug converted to 6-mercaptopurine → inhibits DNA/RNA synthesis. Largely replaced by MMF (superior efficacy in the Mycophenolate Mofetil Renal Study Group trial — lower rejection rates). Still used when MMF is not tolerated (GI intolerance) or in pregnancy (category D — less teratogenic than MMF which is category X). Critical interaction: allopurinol inhibits xanthine oxidase, which metabolizes azathioprine → 4-fold increase in active metabolite levels → potentially fatal pancytopenia. If allopurinol is necessary, reduce azathioprine dose to 25% of standard.
Steroid avoidance / early withdrawal: Multiple trials have explored steroid-free or early steroid withdrawal protocols (within 7 days). Generally safe in low-risk patients with potent induction (thymoglobulin or alemtuzumab) and tacrolimus + MMF maintenance. Benefits: reduced PTDM, HTN, hyperlipidemia, osteoporosis, weight gain, growth retardation (pediatric). Risks: slightly higher rejection rate in some studies (2-5% absolute increase). Not recommended for high-risk patients (sensitized, AA race, retransplant). PMID: 18256394
Organ-Specific Immunosuppression Differences
| Organ | Typical Induction | Maintenance Intensity | Special Considerations |
|---|---|---|---|
| Kidney | rATG (high risk) or basiliximab (low risk) | Standard triple therapy | Most studied; most protocols available |
| Liver | Often none or basiliximab | Lower intensity (liver is "immunoprivileged") | ~20% achieve operational tolerance (off all IS); steroid withdrawal common by 3-6 months |
| Heart | rATG or basiliximab | Higher intensity | No steroid withdrawal; mTOR inhibitors for CAV prevention and renal sparing |
| Lung | rATG or basiliximab or alemtuzumab | Highest intensity | Highest rejection rate among thoracic organs; no steroid withdrawal; azithromycin for BOS |
| Pancreas | rATG (most common) | Standard to high | Rejection monitoring difficult; rely on kidney as sentinel (SPK) |
| Intestine | rATG or alemtuzumab | Highest intensity (often + steroids) | Highest rejection and PTLD rates; bone marrow augmentation protocols in some centers |
22 Rejection Treatment Protocols
Acute Cellular Rejection (ACR) Treatment
| Severity | First-Line | Second-Line (Steroid-Resistant) |
|---|---|---|
| Borderline / Banff IA | Pulse methylprednisolone 250-500 mg IV daily × 3 days; optimize maintenance IS | rATG 1.5 mg/kg/day × 5-7 days |
| Banff IB-IIA | Pulse methylprednisolone 500-1,000 mg IV daily × 3 days | rATG 1.5 mg/kg/day × 7-10 days |
| Banff IIB-III (vascular rejection) | rATG 1.5 mg/kg/day × 7-14 days (consider as first-line for grade III) | Higher-dose rATG; OKT3 (historic); plasmapheresis if mixed AMR component |
After treatment: optimize maintenance immunosuppression (ensure therapeutic tacrolimus levels, adequate MMF dose, consider adding/increasing prednisone if on steroid-free protocol). Repeat biopsy in 1-2 weeks to confirm resolution.
Antibody-Mediated Rejection (AMR) Treatment
AMR is more difficult to treat than ACR and carries worse prognosis. Standard approach (no universal consensus):
Acute AMR: (1) Plasmapheresis (5-7 sessions on alternate days — removes circulating DSA); (2) IVIG (100-400 mg/kg after each plasma exchange, or 2 g/kg total — provides immunomodulation and anti-idiotypic antibodies); (3) Rituximab 375 mg/m2 IV (anti-CD20 — depletes B cells producing DSA); (4) Optimize CNI levels; (5) Pulse methylprednisolone. Additional agents for refractory cases: bortezomib (proteasome inhibitor — targets antibody-producing plasma cells), eculizumab (anti-C5 — blocks terminal complement), tocilizumab (anti-IL-6R), carfilzomib, imlifidase (IdeS — IgG-cleaving enzyme).
Chronic active AMR: Most difficult to treat — no proven effective therapy. Options: optimize tacrolimus (trough 8-10), rituximab, IVIG, tocilizumab (showing promise in small studies PMID: 28556838), costimulation blockade. Many patients progress to graft failure despite treatment.
Desensitization Protocols for Highly Sensitized Patients
Patients with cPRA ≥ 98% or known DSA against an available donor may undergo desensitization to enable transplantation. Two major approaches:
IVIG-based protocol (Cedars-Sinai): IVIG 2 g/kg (max 140 g) monthly while awaiting transplant → reduces PRA and desensitizes. Post-transplant: IVIG + plasmapheresis if flow crossmatch positive at time of transplant. Results: ~80% transplanted within 5 years; comparable graft survival to non-sensitized patients at 5 years.
Plasmapheresis + IVIG + rituximab protocol (Johns Hopkins): For positive crossmatch living donor transplantation: pre-transplant plasmapheresis every other day until crossmatch converts negative → rituximab 375 mg/m2 → IVIG 100 mg/kg after each session. Results: successful transplantation in > 90% of desensitized patients; slightly higher AMR rate (~30% in first year) but acceptable long-term outcomes.
Novel agents: Imlifidase (IdeS) — an IgG-degrading enzyme from Streptococcus pyogenes; cleaves all IgG in circulation within hours, converting positive crossmatch to negative. Approved in EU for highly sensitized patients. Phase 3 trials in the US (IDEALIST). Limitation: IgG rebounds within days; requires concomitant rituximab, IVIG, and potentially bortezomib to prevent rebound DSA. PMID: 34469215
Complement Inhibition in Transplantation
Eculizumab (anti-C5): Terminal complement inhibitor that blocks C5a generation and MAC formation. Uses in transplantation: (1) Prevention of AMR in positive crossmatch transplants — mixed results; (2) Treatment of acute AMR (rescue therapy); (3) Prevention of recurrent aHUS in kidney transplants (well-established indication — reduces recurrence from ~80% to < 10%); (4) Prevention of AMR in ABOi transplants. Risks: Neisseria meningitidis infection (vaccination mandatory pre-treatment), cost ($500,000+/year). PMID: 24599934
23 Post-Transplant Infections
Timeline of Post-Transplant Infections
| Period | Type | Common Pathogens |
|---|---|---|
| 0-1 month | Nosocomial / surgical site / donor-derived | Wound infections (S. aureus, enterococcus), UTI (E. coli, Klebsiella), pneumonia (hospital flora), C. difficile, line infections, donor-derived (HSV, Candida) |
| 1-6 months | Opportunistic infections (peak immunosuppression) | CMV (most important — see below), BK polyomavirus, PJP (Pneumocystis jirovecii), Aspergillus, Nocardia, Listeria, Cryptococcus, EBV (driving PTLD), toxoplasmosis (heart Tx), HCV/HBV reactivation |
| > 6 months | Community-acquired; late opportunistic in over-immunosuppressed | Community-acquired pneumonia, UTI, influenza, COVID-19; late CMV in those with inadequate prophylaxis; JC virus (PML), cryptococcal meningitis, TB reactivation |
CMV — The Most Important Post-Transplant Infection
CMV serostatus mismatch (D+/R−) = highest risk (~60% viremia without prophylaxis). Risk stratification: D+/R− (highest), D+/R+ or D−/R+ (intermediate), D−/R− (lowest). Two prevention strategies: (1) Universal prophylaxis: valganciclovir 900 mg daily for 3-6 months (kidney), 3-6 months (liver), 6-12 months (lung, heart) — reduces CMV disease but late-onset CMV after discontinuation in D+/R−. (2) Preemptive therapy: Weekly CMV PCR monitoring and treat only when viremia detected (> threshold, typically > 1,000 copies/mL). Treatment of active CMV disease: IV ganciclovir 5 mg/kg q12h × 14-21 days, then oral valganciclovir 900 mg BID until viremia clears + 2 negative PCRs. Reduce immunosuppression. Ganciclovir-resistant CMV (UL97 mutation): foscarnet (nephrotoxic) or cidofovir; maribavir (UL97-independent, approved 2021) for refractory/resistant CMV. PMID: 34724827
Standard Prophylaxis Protocols
| Infection | Prophylaxis | Duration |
|---|---|---|
| PJP (Pneumocystis) | TMP-SMX 400/80 mg daily (or 800/160 3x/week) | 6-12 months (lifelong in lung Tx); restart during rejection treatment |
| CMV | Valganciclovir 900 mg daily (renally adjusted) | 3-6 months (kidney/liver); 6-12 months (heart/lung) for D+/R− and R+ |
| Candida (liver Tx) | Fluconazole 400 mg daily or micafungin 100 mg daily | 2-4 weeks post-liver transplant |
| HSV/VZV | Covered by valganciclovir; if not on CMV prophylaxis: acyclovir 400 mg BID | 3-6 months minimum |
| Hepatitis B | Entecavir or tenofovir ± HBIG | Lifelong |
| TB (if LTBI) | Isoniazid 300 mg daily + pyridoxine (avoid rifampin — CYP3A4 inducer, drastically reduces CNI levels) | 9 months |
BK Virus — Detailed Management
BK polyomavirus nephropathy is unique to kidney transplantation. Screening protocol: quantitative BK PCR in plasma at months 1, 2, 3, 6, 9, 12 post-transplant (per AST/IDCOP guidelines). Interpretation:
| BK Viremia Level | Interpretation | Action |
|---|---|---|
| < 1,000 copies/mL | Low-level viremia | Monitor; no intervention |
| 1,000-10,000 copies/mL | Moderate viremia — rising trend concerning | Consider IS reduction; recheck in 2-4 weeks |
| > 10,000 copies/mL | "Presumptive BK nephropathy" | Reduce IS (tacrolimus trough to 4-6 ng/mL, reduce/stop MMF); consider biopsy |
| > 100,000 copies/mL | High-level viremia — high risk of nephropathy | Aggressive IS reduction; biopsy recommended |
Biopsy findings: viral cytopathic changes (enlarged nuclei with ground-glass appearance), positive SV40 immunostain (cross-reacts with BK virus large T antigen), tubulointerstitial nephritis. BK nephropathy can mimic acute rejection on light microscopy — SV40 stain is essential to differentiate. Treating rejection in the setting of BK nephropathy (further immunosuppression) worsens outcomes; treating BK nephropathy (reduction of IS) in the setting of rejection risks graft loss from rejection. This clinical dilemma requires careful biopsy-guided management.
24 Post-Transplant Malignancy
Overall Risk
Transplant recipients have a 3-5 fold increased overall cancer risk compared to the general population; for certain cancers, the increase is dramatic: non-melanoma skin cancer (~65-250x), PTLD/lymphoma (~8-12x), Kaposi sarcoma (~200-500x), lip cancer (~20x), renal cell carcinoma (~15x), cervical/vulvar cancer (~3-5x). Cancer is the leading cause of death after cardiovascular disease in long-term transplant recipients. Cumulative incidence of any cancer: ~20-30% at 20 years post-transplant.
Post-Transplant Lymphoproliferative Disorder (PTLD)
PTLD is a spectrum of lymphoid proliferations occurring in the setting of immunosuppression, primarily driven by Epstein-Barr virus (EBV). Incidence: 1-2% in kidney/liver, 2-5% in heart, 5-10% in lung, up to 20% in intestinal transplant. Highest risk: EBV D+/R− (seronegative recipient). Median onset: 6-12 months (early, EBV-driven) or years (late, often EBV-negative).
PTLD Classification (WHO)
| Category | Description | EBV Association |
|---|---|---|
| Non-destructive (early lesions) | Plasmacytic hyperplasia, infectious mononucleosis-like, florid follicular hyperplasia | Usually EBV+ |
| Polymorphic | Polymorphic B-cell proliferations that destroy tissue architecture but do not fulfill criteria for lymphoma | Usually EBV+ |
| Monomorphic | Fulfill criteria for a recognized lymphoma subtype: DLBCL (most common, ~60-80%), Burkitt, plasmacytoma, T-cell lymphoma, Hodgkin | Variable (DLBCL ~60-70% EBV+) |
| Classic Hodgkin lymphoma-type | Meets criteria for classic Hodgkin lymphoma | Usually EBV+ |
PTLD Treatment
(1) Reduction of immunosuppression (RI): First-line for all PTLD — reduce CNI by 50%, discontinue antimetabolite (MMF/AZA). Complete response in ~30-60% of early lesions and polymorphic PTLD with RI alone. (2) Rituximab (anti-CD20): For CD20+ PTLD not responding to RI — 375 mg/m2 weekly × 4; response rate ~50-60%. (3) R-CHOP chemotherapy: For CD20+ monomorphic PTLD not responding to RI + rituximab; or for aggressive/high-stage disease. (4) Radiation, surgery for localized disease. (5) EBV-specific cytotoxic T lymphocytes (adoptive immunotherapy) — investigational, promising. Monitor EBV viral load by PCR — rising EBV may precede PTLD; trigger for preemptive RI.
Skin Cancer
Most common malignancy post-transplant. Squamous cell carcinoma (SCC) exceeds basal cell carcinoma (BCC) by 2-5:1 ratio (reversed from general population). SCC in transplant recipients: more aggressive, higher metastatic rate (5-8% vs < 1% in immunocompetent). Risk factors: cumulative UV exposure, fair skin, HPV co-infection, degree of immunosuppression. Prevention: sun protection, regular dermatologic screening (every 6-12 months), conversion to mTOR inhibitor (sirolimus/everolimus — 50% reduction in new skin cancers). PMID: 22458902
Kaposi Sarcoma
HHV-8-driven; ~200-500x increased risk. Higher incidence in certain geographic regions (Mediterranean, Middle East, Africa). Treatment: reduction of immunosuppression (conversion to mTOR inhibitor — sirolimus has anti-KS properties); local therapy (radiation) for limited disease; chemotherapy (liposomal doxorubicin, paclitaxel) for disseminated disease.
Screening Recommendations Post-Transplant
| Cancer | Screening | Frequency |
|---|---|---|
| Skin cancer | Full-body dermatologic exam | Every 6-12 months (annually at minimum) |
| Cervical cancer | Pap smear + HPV testing | Annually (more frequent than general population) |
| Colorectal cancer | Colonoscopy (especially if PSC/UC) | Per general population guidelines; annually for PSC/UC |
| Breast cancer | Mammography | Per general population guidelines |
| Renal cell carcinoma | Ultrasound of native kidneys | Annual (for patients with ACKD on chronic dialysis) |
| PTLD | EBV viral load monitoring (if high risk) | Monthly for 6-12 months, then every 3-6 months in first 2 years |
| Hepatocellular carcinoma | AFP + liver ultrasound (if cirrhosis/HBV in native liver) | Every 6 months |
25 Classification Systems
Banff Classification — Kidney Allograft Rejection (2022)
| Category | Criteria |
|---|---|
| 1. Normal | No significant histologic changes |
| 2. Antibody-mediated changes | Active AMR: microvascular inflammation (g + ptc ≥ 2) OR v > 0 OR TMA; + C4d or molecular AMR markers; + DSA. Chronic active AMR: cg ≥ 1 OR PTCBMML ≥ 7 layers; + antibody criteria; + DSA |
| 3. Borderline | t1-t2 with i0-i1, or t1 with i2-i3 — suspicious for acute T-cell mediated rejection |
| 4. T-cell mediated rejection | IA (i2-3, t2), IB (i2-3, t3), IIA (v1), IIB (v2), III (v3 — transmural arteritis/fibrinoid necrosis) |
| 5. Interstitial fibrosis / tubular atrophy (IF/TA) | Grade I: ci1-ct1 (mild, ≤ 25%); Grade II: ci2-ct2 (moderate, 26-50%); Grade III: ci3-ct3 (severe, > 50%) |
| 6. Other | Changes not attributable to rejection: CNI toxicity, BK nephropathy, recurrent disease, PTLD, etc. |
Banff RAI — Liver Allograft Rejection
| Component | 0 (None) | 1 (Mild) | 2 (Moderate) | 3 (Severe) |
|---|---|---|---|---|
| Portal inflammation | None | Mild, some portal tracts | Most portal tracts, mixed | Dense, expanding, spillover |
| Bile duct damage | None | Minority of ducts | Most/all ducts | Disruption/sloughing |
| Venous endotheliitis | None | Some venules | Most venules | Perivenular necrosis |
RAI 0-2: no rejection; 3: indeterminate; 4-5: mild; 6-7: moderate; 8-9: severe.
Milan Criteria (HCC)
Single tumor ≤ 5 cm OR up to 3 tumors each ≤ 3 cm; no macrovascular invasion; no extrahepatic disease.
KDPI / KDRI — Kidney Donor Quality
KDRI incorporates 10 donor variables: age, height, weight, ethnicity, hypertension, diabetes, cause of death, serum creatinine, HCV status, DCD status. KDPI = percentile mapping of KDRI against prior year's donors. KDPI 0-20%: highest quality; 21-85%: standard; > 85%: marginal (formerly ECD).
Lung Allocation Score (LAS)
Range 0-100. Based on: waitlist urgency (predicted survival without transplant) and post-transplant survival. Inputs: diagnosis group (A: obstructive, B: PAH, C: CF/bronchiectasis, D: restrictive), FVC, O2 at rest, 6MWD, PA systolic pressure, PCO2, continuous mechanical ventilation, age, BMI, diabetes, functional status, creatinine. LAS replaced time-based lung allocation in 2005, reducing waitlist mortality by ~40%. The composite allocation score (CAS) — implemented 2023 — replaces LAS with a broader scoring system incorporating medical urgency, post-transplant outcomes, candidate biology, patient access equity, and placement efficiency.
CLAD Phenotype Classification
| Feature | BOS (Bronchiolitis Obliterans Syndrome) | RAS (Restrictive Allograft Syndrome) |
|---|---|---|
| Frequency | ~70% of CLAD | ~30% of CLAD |
| Physiology | Obstructive — FEV1 decline | Restrictive — TLC decline (< 90% baseline) |
| CT findings | Air trapping, mosaic attenuation | Upper lobe fibrosis, ground glass, consolidation |
| Pathology | Obliterative bronchiolitis | Pleuroparenchymal fibroelastosis |
| Prognosis | Median survival ~3-5 years after onset | Median survival ~1.5 years after onset |
| Response to azithromycin | ~30% respond | Generally non-responsive |
Transplant Outcomes Summary — Survival by Organ
| Organ | 1-Year Graft Survival | 5-Year Graft Survival | Median Graft Half-Life | Leading Cause of Late Graft Loss |
|---|---|---|---|---|
| Kidney (living donor) | ~97-98% | ~90-92% | ~15-20 years | Chronic active AMR / IF/TA |
| Kidney (deceased donor) | ~93-95% | ~80-83% | ~10-12 years | Chronic active AMR / IF/TA |
| Liver | ~88-92% | ~73-78% | ~12-15 years | Recurrent disease, biliary complications |
| Heart | ~88-90% | ~77-80% | ~12-14 years | Cardiac allograft vasculopathy |
| Lung | ~83-87% | ~55-60% | ~6-7 years | CLAD (BOS/RAS) |
| Pancreas (SPK) | ~87-90% | ~73-80% | ~10-12 years | Thrombosis, rejection |
| Intestine | ~70-80% | ~50-60% | ~5-7 years | Rejection, infection, PTLD |
ISHLT Heart Rejection Grading
| Grade (2004 Revised) | Description |
|---|---|
| 0R | No rejection |
| 1R (mild) | Interstitial/perivascular infiltrate with up to one focus of myocyte damage |
| 2R (moderate) | Two or more foci of infiltrate with myocyte damage |
| 3R (severe) | Diffuse infiltrate with multifocal myocyte damage ± edema, hemorrhage, vasculitis |
ISHLT Primary Graft Dysfunction — Lung Grading
| Grade | PaO2/FiO2 | Chest Radiograph |
|---|---|---|
| 0 | > 300 | Clear |
| 1 | > 300 | Infiltrates present |
| 2 | 200-300 | Infiltrates present |
| 3 | < 200 | Infiltrates present |
Cold Ischemia Time Limits by Organ (Summary)
| Organ | Ideal CIT | Max CIT |
|---|---|---|
| Heart | < 4 hr | ~6 hr |
| Lung | < 6 hr | ~8 hr (12+ with EVLP) |
| Liver | < 10 hr | ~12 hr |
| Pancreas | < 12 hr | ~18 hr |
| Kidney | < 24 hr | ~36 hr (with HMP) |
| Intestine | < 8 hr | ~10 hr |
26 Medications Master Table
| Drug | Class | Mechanism | Dose / Target Level | Key Side Effects | Drug Interactions |
|---|---|---|---|---|---|
| Tacrolimus | CNI | FKBP-12 → calcineurin inhibition → blocks IL-2 | 0.05-0.1 mg/kg BID; trough 5-12 ng/mL (time-dependent) | Nephrotoxicity, PTDM, tremor, hyperkalemia, hypomagnesemia, alopecia, PRES | CYP3A4 substrate: ↑ by azole antifungals, diltiazem, erythromycin; ↓ by rifampin, phenytoin, carbamazepine |
| Cyclosporine | CNI | Cyclophilin → calcineurin inhibition → blocks IL-2 | Trough 100-300 ng/mL (C0) or C2 800-1,200 ng/mL | Nephrotoxicity, HTN, hyperlipidemia, gingival hyperplasia, hirsutism, tremor | Same CYP3A4 interactions as tacrolimus |
| Mycophenolate (MMF) | Antimetabolite | IMPDH inhibitor → blocks de novo purine synthesis in lymphocytes | 1 g BID (kidney); 1-1.5 g BID (liver) | GI (diarrhea, nausea), leukopenia, anemia, teratogenic (Category X) | Levels ↓ by cholestyramine, antacids; avoid with azathioprine |
| Azathioprine | Antimetabolite | Purine analogue → inhibits DNA/RNA synthesis | 1-2 mg/kg/day | Leukopenia, hepatotoxicity, pancreatitis | Allopurinol inhibits xanthine oxidase → 4x increase in AZA levels → fatal pancytopenia; reduce AZA to 25% dose |
| Sirolimus | mTOR inhibitor | FKBP-12 → mTOR inhibition → blocks G1→S transition | Trough 5-15 ng/mL | Impaired wound healing, hyperlipidemia, proteinuria, oral ulcers, pneumonitis, cytopenias | CYP3A4 substrate; same interactions as CNIs |
| Everolimus | mTOR inhibitor | Same as sirolimus | Trough 3-8 ng/mL | Same as sirolimus | CYP3A4 substrate |
| Belatacept | Costimulation blocker | CTLA4-Ig → blocks B7-CD28 (Signal 2) | 5-10 mg/kg IV on schedule | Higher ACR rate, PTLD (EBV− recipients), PML | Minimal drug interactions |
| Basiliximab | Anti-CD25 mAb | Blocks IL-2 receptor on activated T cells | 20 mg IV day 0 and day 4 | Well tolerated; rare hypersensitivity | None significant |
| rATG (Thymoglobulin) | Polyclonal depleting Ab | T-cell depletion via multiple mechanisms | 1.5 mg/kg/day × 3-7 days | Cytokine release syndrome, leukopenia, thrombocytopenia, serum sickness, infection, PTLD | None significant |
| Alemtuzumab | Anti-CD52 mAb | Depletes T cells, B cells, NK cells, monocytes | 30 mg IV × 1 (or 20 mg × 2) | Profound lymphopenia, infections, autoimmune cytopenias, PTLD | None significant |
| Rituximab | Anti-CD20 mAb | B-cell depletion | 375 mg/m2 IV | Infusion reactions, hepatitis B reactivation, PML (rare), hypogammaglobulinemia | None significant |
| Prednisone | Corticosteroid | NF-κB / AP-1 inhibition → broad anti-inflammatory | Taper from 200 mg to 5 mg/day over weeks | PTDM, HTN, osteoporosis, cataracts, AVN, weight gain, adrenal suppression | CYP3A4 substrate; ↓ by rifampin |
| Valganciclovir | Antiviral | Guanosine analogue → inhibits CMV DNA polymerase | 900 mg daily (prophylaxis); 900 mg BID (treatment); renal dosing required | Leukopenia, neutropenia, anemia, thrombocytopenia | Additive myelosuppression with MMF |
| TMP-SMX | Antibiotic | Folate synthesis inhibition | SS (400/80 mg) daily or DS 3x/week | Hyperkalemia, rash, cytopenias, interstitial nephritis | May increase creatinine (tubular secretion block — not true nephrotoxicity); potentiates methotrexate |
27 Imaging & Diagnostics
Doppler Ultrasound — Graft Assessment
Renal allograft ultrasound: First-line imaging for transplant kidney dysfunction. Assess: (1) graft size (normal transplant kidney slightly larger than native); (2) hydronephrosis (ureteral obstruction); (3) perigraft collections (lymphocele, hematoma, urinoma, abscess); (4) Doppler parameters: main renal artery PSV (normal 80-150 cm/s; > 200-250 = stenosis), resistive index (RI = [PSV − EDV] / PSV; normal 0.5-0.8; > 0.8 suggests rejection, ATN, or renal vein thrombosis), acceleration time (> 0.1 sec in segmental arteries = proximal stenosis), absent/reversed diastolic flow (renal vein thrombosis, severe rejection).
Hepatic allograft ultrasound: Assess hepatic artery (normal RI 0.5-0.8; absent or very low signal = HAT; tardus-parvus = hepatic artery stenosis), portal vein (normal flow velocity 20-40 cm/s; absent flow = portal vein thrombosis), hepatic veins (normal triphasic waveform; monophasic = outflow obstruction), bile ducts (dilatation suggests stricture or obstruction).
Protocol Biopsy Schedules
| Organ | Indication Biopsy | Protocol (Surveillance) Biopsy |
|---|---|---|
| Kidney | Rising creatinine without clear cause | Some centers: 3, 6, 12 months; increasingly replaced by donor-derived cell-free DNA (dd-cfDNA) monitoring |
| Liver | Rising LFTs, cholestasis | Less commonly performed; some centers: 1 year |
| Heart | Clinical suspicion of rejection | Standard: weekly × 6 weeks, then biweekly × 3 months, then monthly × 6 months, then every 3-6 months × year 2, annually thereafter; increasingly supplemented by dd-cfDNA (AlloMap/AlloSure) |
| Lung | Declining FEV1, infiltrates | Transbronchial biopsy: 1, 3, 6, 9, 12 months, then annually |
| Intestine | Diarrhea, stomal output changes | Ileoscopy with biopsy: weekly × 4, then biweekly, then monthly |
MRCP & CT Angiography
MRCP (magnetic resonance cholangiopancreatography): Non-invasive evaluation of biliary anatomy in liver transplant recipients. Indications: suspected biliary stricture (anastomotic or non-anastomotic), biliary leak, choledocholithiasis. Advantages: no radiation, no contrast needed for biliary imaging. Limitations: motion artifact, T-tube artifact, spatial resolution inferior to ERCP. MRCP findings guide decision between ERCP (therapeutic — stenting, dilation) and PTC (if Roux-en-Y anatomy precludes ERCP).
CT angiography: Used for vascular evaluation in all transplant types. Indications: suspected renal artery stenosis (if Doppler equivocal), hepatic artery stenosis/thrombosis, portal vein thrombosis. Living donor evaluation: CT angiogram of kidneys (arterial, venous, and excretory phases) is the standard for assessing donor renal vascular anatomy, kidney volume, and collecting system anatomy. Living donor liver evaluation: CT volumetry calculates future liver remnant and graft volume.
Nuclear Medicine Studies
MAG3 renal scan (Tc-99m mercaptoacetyltriglycine): Assesses graft perfusion and function. Findings: (1) Normal: prompt radiotracer uptake and excretion; (2) ATN/DGF: preserved perfusion with delayed/absent excretion; (3) Rejection: decreased perfusion AND delayed excretion; (4) Obstruction: normal perfusion with progressively increasing activity (obstructed renogram curve); (5) Urine leak: radiotracer extravasation outside the collecting system.
Hepatobiliary iminodiacetic acid (HIDA) scan: Assesses hepatic uptake and biliary excretion in liver transplants. Used for suspected bile leak (radiotracer seen outside biliary tree) or biliary obstruction (non-visualization of bowel activity at 60 minutes). Hepatocyte function correlates with hepatic uptake rate.
Non-Invasive Rejection Monitoring
Donor-derived cell-free DNA (dd-cfDNA): DNA fragments released from dying graft cells into recipient blood. Measured as a fraction of total cfDNA. Thresholds: kidney — dd-cfDNA > 1% suggests active rejection (sensitivity ~85% for AMR, ~60% for ACR); heart — dd-cfDNA > 0.2% (AlloSure) correlates with rejection/injury. Advantages: non-invasive, can detect subclinical rejection, high negative predictive value (> 95%). Commercially available: CareDx AlloSure (kidney, heart), Natera Prospera (kidney). PMID: 28428346
Gene expression profiling: AlloMap (heart) — 20-gene panel on peripheral blood mononuclear cells; score < 34 indicates low probability of moderate-severe ACR (high NPV). Used to reduce frequency of endomyocardial biopsies. CARGO II study validated utility. PMID: 20413602
Torque Teno Virus (TTV) — Emerging Biomarker
TTV is a ubiquitous, non-pathogenic DNA virus that infects > 90% of the population. TTV viral load in blood correlates with the degree of immunosuppression — serving as a potential "immune function" biomarker. High TTV loads (> 107-108 copies/mL) suggest over-immunosuppression (higher infection risk); low TTV loads (< 103-104 copies/mL) suggest under-immunosuppression (higher rejection risk). The TTV-GUIDE-IT randomized trial is prospectively evaluating TTV-guided immunosuppression adjustment in kidney transplant recipients. If validated, TTV monitoring could provide a personalized approach to immunosuppression dosing — a significant advance over current empiric protocols.
Percutaneous Graft Biopsy Technique
Kidney allograft biopsy: Ultrasound-guided, 16-18 gauge automated spring-loaded needle; target the lower pole cortex (avoiding medulla and hilum); minimum 2 cores with ≥ 7 glomeruli and 1 artery required for adequate sampling (Banff criteria). Complications: bleeding (~1% clinically significant), AV fistula (~1-5% by Doppler — most resolve spontaneously), perinephric hematoma, graft loss from biopsy (< 0.1%).
Liver allograft biopsy: Percutaneous, ultrasound-guided, intercostal approach; 16-18 gauge needle. Adequate sample: ≥ 11 portal tracts, ≥ 20 mm length. Complications: bleeding, bile leak, pneumothorax, hemobilia. Transjugular approach used if coagulopathy (INR > 1.5) or ascites.
Heart — endomyocardial biopsy: Right internal jugular vein approach; flexible bioptome advanced to RV septum under fluoroscopic guidance. 3-5 pieces obtained from different sites. Complications: tricuspid regurgitation, RV perforation/tamponade (rare — < 0.5%), pneumothorax, arrhythmia.
28 Abbreviations Master List
| Abbreviation | Full Term |
|---|---|
| ACR | Acute cellular rejection |
| AMR | Antibody-mediated rejection |
| APC | Antigen-presenting cell |
| ATG / rATG | Anti-thymocyte globulin / rabbit ATG |
| BOS | Bronchiolitis obliterans syndrome |
| CAV | Cardiac allograft vasculopathy |
| CDC | Complement-dependent cytotoxicity |
| CIT | Cold ischemia time |
| CLAD | Chronic lung allograft dysfunction |
| CMV | Cytomegalovirus |
| CNI | Calcineurin inhibitor |
| cPRA | Calculated panel reactive antibody |
| DAA | Direct-acting antiviral |
| DBD | Donation after brain death |
| DCD | Donation after circulatory death |
| dd-cfDNA | Donor-derived cell-free DNA |
| DGF | Delayed graft function |
| DSA | Donor-specific antibody |
| EBV | Epstein-Barr virus |
| EPTS | Estimated post-transplant survival |
| EVLP | Ex-vivo lung perfusion |
| GALT | Gut-associated lymphoid tissue |
| HAT | Hepatic artery thrombosis |
| HLA | Human leukocyte antigen |
| HMP | Hypothermic machine perfusion |
| HTK | Histidine-tryptophan-ketoglutarate (Custodiol) |
| IF/TA | Interstitial fibrosis and tubular atrophy |
| IMPDH | Inosine monophosphate dehydrogenase |
| IS | Immunosuppression |
| KDPI | Kidney Donor Profile Index |
| KDRI | Kidney Donor Risk Index |
| KPD | Kidney paired donation |
| LAS | Lung Allocation Score |
| LDLT | Living donor liver transplant |
| LVAD | Left ventricular assist device |
| MELD | Model for End-Stage Liver Disease |
| MFI | Mean fluorescence intensity |
| MHC | Major histocompatibility complex |
| MMF | Mycophenolate mofetil |
| MPA | Mycophenolic acid |
| mTOR | Mammalian target of rapamycin |
| NAS | Non-anastomotic stricture |
| NFAT | Nuclear factor of activated T cells |
| NMP | Normothermic machine perfusion |
| NRP | Normothermic regional perfusion |
| OPTN | Organ Procurement and Transplantation Network |
| PAK | Pancreas after kidney |
| PELD | Pediatric End-Stage Liver Disease (score) |
| PGD | Primary graft dysfunction |
| PNF | Primary non-function |
| PRA | Panel reactive antibody |
| PTA | Pancreas transplant alone |
| PTDM | Post-transplant diabetes mellitus |
| PTLD | Post-transplant lymphoproliferative disorder |
| RAI | Rejection Activity Index (liver) |
| RAS | Restrictive allograft syndrome |
| SAB | Single-antigen bead (Luminex) |
| SPK | Simultaneous pancreas-kidney |
| TCR | T-cell receptor |
| UNOS | United Network for Organ Sharing |
| UW | University of Wisconsin (preservation solution) |
| WIT | Warm ischemia time |
| ABOi | ABO-incompatible |
| ADCC | Antibody-dependent cell-mediated cytotoxicity |
| ACKD | Acquired cystic kidney disease |
| ALF | Acute liver failure |
| CAS | Composite allocation score |
| DAMPs | Damage-associated molecular patterns |
| EAD | Early allograft dysfunction |
| FWIT | Functional warm ischemia time |
| GVHD | Graft-versus-host disease |
| HBIG | Hepatitis B immune globulin |
| HPS | Hepatopulmonary syndrome |
| IBMIR | Instant blood-mediated inflammatory reaction |
| IEQ | Islet equivalents |
| ILDA | Independent living donor advocate |
| MAC | Membrane attack complex (C5b-9) |
| MASLD | Metabolic dysfunction-associated steatotic liver disease |
| NASH | Non-alcoholic steatohepatitis |
| POPH | Portopulmonary hypertension |
| PTCBMML | Peritubular capillary basement membrane multilayering |
| SVR | Sustained virologic response |
| TTV | Torque teno virus |
29 Landmark Trials
| Trial | Year | Key Finding | PMID |
|---|---|---|---|
| BENEFIT | 2010 (7-yr: 2016) | Belatacept vs. cyclosporine in kidney Tx: belatacept achieved superior 7-year GFR (+16 mL/min), comparable graft/patient survival despite higher early rejection rate | PMID: 26816011 |
| ELITE-Symphony | 2007 | Low-dose tacrolimus + MMF + corticosteroids + daclizumab induction → lowest acute rejection rate (12.3%), best renal function, best graft survival at 1 year compared to standard cyclosporine, low-dose cyclosporine, or low-dose sirolimus arms | PMID: 18094378 |
| CONVERT | 2009 | Conversion from CNI to sirolimus in kidney Tx: safe in patients with GFR ≥ 40 and proteinuria < 500 mg/day; improved GFR. Not beneficial (and potentially harmful) with GFR < 40 or proteinuria > 500 mg/day | PMID: 19155978 |
| Milan Criteria (Mazzaferro) | 1996 | Defined tumor criteria (single ≤ 5 cm or ≤ 3 tumors each ≤ 3 cm) for liver Tx in HCC → 75% 4-year survival, 8% recurrence; became the worldwide standard | PMID: 8632270 |
| Edmonton Protocol | 2000 | Steroid-free islet transplantation with adequate islet mass (from 2+ donors) achieved insulin independence in 7/7 T1DM patients — proof of concept for islet transplantation | PMID: 10911004 |
| SPLIT (Studies of Pediatric Liver Tx) | 2001+ | Multicenter registry establishing outcomes benchmarks for pediatric liver transplantation; identified risk factors for graft failure | PMID: 11689788 |
| Meier-Kriesche (Half-life data) | 2004 | Living donor kidneys: half-life ~15-20 years; deceased donor: ~10-12 years; preemptive transplantation (before dialysis) yields best outcomes | PMID: 15084934 |
| USRDS/SRTR Annual Data Report | Annual | Comprehensive US transplant outcomes data: 1-year kidney graft survival ~95% (living), ~93% (deceased); 1-year liver ~90%; 1-year heart ~90%; 1-year lung ~85% | — |
| Lefaucheur (DSA and AMR) | 2010 | Demonstrated that de novo DSA is a strong independent risk factor for AMR and graft loss; established the importance of post-transplant DSA monitoring | PMID: 20581746 |
| INSPIRE (EVLP) | 2017 | Portable EVLP (Organ Care System Lung) for standard-criteria donor lungs: non-inferior composite outcome vs. standard cold storage; validated EVLP technology | PMID: 29211530 |
| Mateo (Alcoholic Hepatitis Tx) | 2011 | Early liver transplantation (without 6-month sobriety) for severe alcoholic hepatitis non-responsive to medical therapy: 77% 6-month survival (transplanted) vs. 23% (not transplanted) | PMID: 22070476 |
| STCS (Swiss Transplant Cohort Study) — PTLD | 2012 | EBV D+/R− mismatch increases PTLD risk 10-fold; incidence highest in first year; intestinal Tx highest overall risk | PMID: 22458902 |
| VOCAL (Valganciclovir) | 2010 | Valganciclovir 900 mg BID × 21 days then 900 mg daily × 49 days for CMV disease: viremia clearance ~45% at day 21; established treatment duration standard | PMID: 20215610 |
| SOLARIS (Maribavir) | 2022 | Maribavir superior to investigator-assigned therapy for refractory/resistant CMV: 55.7% vs. 23.9% viremia clearance at week 8 | PMID: 34724827 |
| TRANSFORM (Everolimus) | 2019 | Everolimus + reduced tacrolimus vs. standard tacrolimus + MMF in kidney Tx: non-inferior efficacy with better eGFR at 12 months; supports CNI-sparing strategy | PMID: 30573365 |
| THINKER (HCV+ to HCV−) | 2017 | HCV-viremic kidneys transplanted into HCV-negative recipients with immediate DAA treatment: 100% SVR12; excellent graft function; expanded donor pool paradigm | PMID: 28614720 |
| A2ALL (Adult-to-Adult Living Donor Liver) | 2009+ | Multicenter US study of LDLT: donor mortality 0.2-0.5%; recipient outcomes comparable to DDLT when performed at experienced centers; learning curve matters | PMID: 19409011 |
| ISHLT Registry (Annual) | Annual | Comprehensive global heart and lung transplant outcomes data; median heart Tx survival ~12-14 years; median lung Tx survival ~6-7 years; CAV incidence ~50% at 10 years | PMID: 29187358 |
| Kaplan (Deceased Donor Score) | 2010 | Developed KDPI/KDRI scoring system to quantify deceased donor kidney quality; enabled risk-stratified allocation | PMID: 20659094 |
| Moers (Machine Perfusion) | 2009 | Hypothermic machine perfusion vs. static cold storage for deceased donor kidneys: reduced DGF (20.8% vs. 26.5%), improved 1-year graft survival | PMID: 19246328 |
| Bloom (dd-cfDNA) | 2017 | Donor-derived cell-free DNA > 1% in plasma discriminates active rejection from stable grafts in kidney transplant; established dd-cfDNA as a non-invasive biomarker | PMID: 28428346 |
| ELIXA (Imlifidase) | 2021 | IgG-degrading enzyme enables desensitization and successful transplantation in highly sensitized patients with positive crossmatch; rapid conversion of positive to negative crossmatch within hours | PMID: 34469215 |
Key Drug Interactions in Transplantation
| Drug | Effect on CNI Levels | Mechanism | Clinical Action |
|---|---|---|---|
| Azole antifungals (fluconazole, voriconazole, posaconazole) | ↑↑ (2-5x increase) | CYP3A4 inhibition | Reduce tacrolimus dose by 50-75%; monitor levels daily |
| Diltiazem, verapamil | ↑ (1.5-2x) | CYP3A4 inhibition | Some centers use intentionally to reduce CNI dose/cost |
| Erythromycin, clarithromycin | ↑ (1.5-3x) | CYP3A4 inhibition | Use azithromycin instead (no CYP3A4 effect) |
| Grapefruit juice | ↑ (variable) | Intestinal CYP3A4 inhibition | Avoid |
| Rifampin | ↓↓↓ (80-90% decrease) | Potent CYP3A4/P-gp induction | Absolutely contraindicated; use rifabutin if TB treatment needed |
| Phenytoin, carbamazepine | ↓↓ (50-70% decrease) | CYP3A4 induction | Use levetiracetam instead (no CYP interaction) |
| St. John's wort | ↓↓ | CYP3A4 induction | Absolutely contraindicated |
| Metoclopramide | ↑ (1.5x) | Increased GI absorption | Monitor levels |
Medication Non-Adherence
Non-adherence to immunosuppressive medications is a major cause of late graft loss — estimated to contribute to 20-36% of kidney graft failures. Risk factors: adolescents/young adults (highest risk group — ~30-50% non-adherence), psychiatric illness, substance abuse, poor social support, complex regimens, medication side effects, high cost/lack of insurance. Tacrolimus variability (high coefficient of variation in trough levels > 30-40%) is a surrogate marker for non-adherence and independently predicts graft loss and de novo DSA development. Interventions: simplification of regimens (once-daily extended-release tacrolimus), electronic reminders, nurse coordinator follow-up, mental health support, medication assistance programs. The 2020 US legislation extending Medicare coverage for immunosuppression beyond 36 months post-transplant (Immunosuppressive Drug Coverage Act) addresses a major barrier to adherence.
Tolerance Induction — The Holy Grail
The ultimate goal of transplant immunology is to achieve immune tolerance — specific unresponsiveness to the graft while maintaining competent immunity to infections and malignancy, without chronic immunosuppression. Approaches under investigation:
Mixed chimerism: Combined kidney and bone marrow transplant from the same donor. Recipient undergoes non-myeloablative conditioning (cyclophosphamide, rituximab, thymic irradiation, ATG) followed by kidney transplant + donor bone marrow infusion. Goal: establishment of mixed hematopoietic chimerism (coexistence of donor and recipient immune cells) → central and peripheral tolerance. Stanford/MGH protocols have achieved immunosuppression-free graft survival in select patients for > 10 years. PMID: 22003520
Regulatory T cells (Tregs): Ex-vivo expanded recipient Tregs (CD4+CD25+FoxP3+) infused post-transplant to suppress alloreactive effector T cells. Phase I/II trials (ONE Study, TRACT) demonstrate safety; efficacy studies ongoing. Donor-specific Tregs may be more effective than polyclonal Tregs.
Costimulation blockade-based tolerance: Belatacept (CTLA4-Ig) blocks CD28-B7 costimulation, theoretically inducing T-cell anergy. Combined with donor bone marrow and short-course immunosuppression in preclinical models. Clinical translation in progress.
Mesenchymal stromal cells (MSCs): Multipotent cells with immunomodulatory properties (suppress T-cell proliferation, promote Treg generation, inhibit DC maturation). Phase I/II trials show safety in kidney transplantation; potential to reduce rejection and allow immunosuppression minimization. Autologous vs. donor-derived MSCs both under investigation. Timing of infusion (pre- vs. peri- vs. post-transplant) remains to be optimized.
CAR-Treg therapy: Chimeric antigen receptor-engineered regulatory T cells that specifically recognize donor HLA antigens — combining the specificity of targeted cellular therapy with the suppressive function of Tregs. Preclinical data show potent antigen-specific tolerance. Phase I trials in kidney transplant are underway. This represents the cutting edge of personalized tolerance induction.
Xenotransplantation — Emerging Frontier
Genetically modified pig organs represent a potential solution to the critical organ shortage — over 100,000 patients are waiting for organ transplants in the US alone, with ~17 patients dying daily while waiting. Key genetic modifications to pig donors: (1) knockout of α-Gal (galactose-α1,3-galactose) — the major xenoantigen that triggers hyperacute rejection; (2) knockout of CMAH (Neu5Gc) and β4GalNT2 — additional carbohydrate antigens; (3) insertion of human complement regulatory proteins (CD46, CD55, CD59) — prevent complement-mediated injury; (4) insertion of human thrombomodulin — prevent thrombotic microangiopathy; (5) insertion of human CD47 — "don't eat me" signal to prevent macrophage phagocytosis. Ten-gene-edited pigs are now available. In 2022-2024, pig-to-human heart xenotransplants were performed in compassionate use cases — the first patient (David Bennett, University of Maryland) survived 60 days; subsequent cases have extended survival. Pig kidney xenotransplants in brain-dead recipients have demonstrated function for weeks, with creatinine normalization and sustained urine output. The first living patient to receive a pig kidney (Tong, Massachusetts General Hospital, 2024) survived over two months. Phase I clinical trials are anticipated. Challenges remain: porcine CMV (porcine cytomegalovirus — a herpesvirus distinct from human CMV) contamination was identified as a contributor to early graft failure in some cases; porcine endogenous retrovirus (PERV) integration risk, long-term immune response patterns, and ethical/regulatory frameworks. The FDA is developing specific regulatory pathways for xenotransplantation clinical trials. If successful, xenotransplantation has the potential to eliminate the organ shortage entirely.