Trauma Surgery
Every diagnosis, classification, procedure, technique, medication, complication, and management algorithm across the full scope of trauma surgery and acute care in one place.
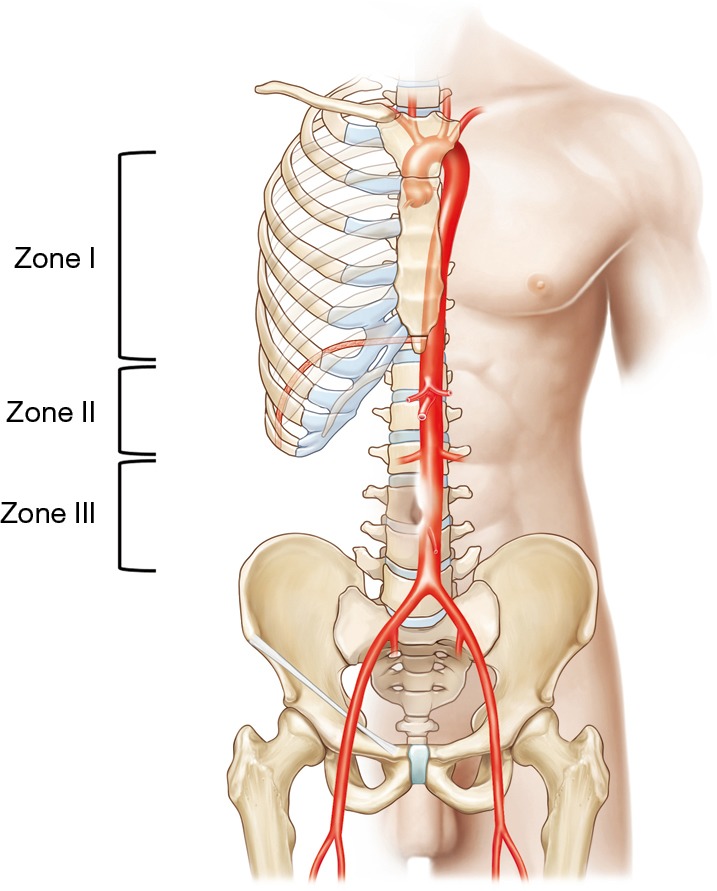
01 Trauma Anatomy — Zones, Regions, & Vascular Landmarks
Trauma surgery demands rapid anatomical reasoning under pressure. Knowing regional anatomy — the zones of the neck, thoracic landmarks, abdominal quadrants and retroperitoneal zones, and vascular territories — determines the operative approach, the urgency of exploration, and the structures at risk for every mechanism of injury.
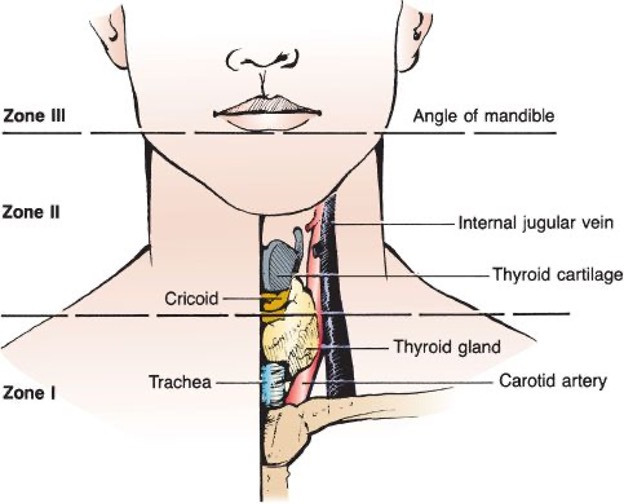
Zones of the Neck
The neck is divided into three zones based on external landmarks, each with distinct vascular, aerodigestive, and neurologic structures at risk:
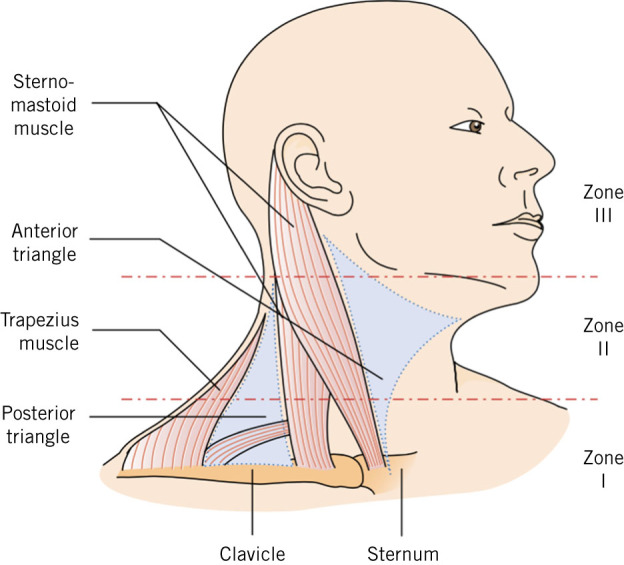
| Zone | Boundaries | Key Structures | Management Considerations |
|---|---|---|---|
| Zone I | Clavicles / sternal notch to cricoid cartilage | Subclavian vessels, common carotid origin, vertebral artery origin, innominate vessels, trachea, esophagus, thoracic duct, lung apex | Most difficult surgical exposure; may require sternotomy or thoracotomy; CTA recommended first |
| Zone II | Cricoid cartilage to angle of mandible | Carotid artery (bifurcation), internal jugular vein, pharynx, larynx, esophagus, vagus/recurrent laryngeal nerve | Most accessible — historically explored for all penetrating injuries with platysma violation; now selective management with CTA is standard |
| Zone III | Angle of mandible to skull base | Distal internal carotid, vertebral artery (V3 segment), pharynx, cranial nerves IX–XII | Difficult surgical access; angiography with embolization preferred for vascular injuries |
Thoracic Anatomy for Trauma
The thorax is divided by the mediastinum into left and right pleural cavities, each capable of holding >2 L of blood. Key landmarks: the cardiac box (bordered by the clavicles superiorly, nipple lines laterally, and costal margin inferiorly) defines the area where penetrating wounds mandate evaluation for cardiac injury. The thoracic inlet (suprasternal notch to T1) is a zone of transition where neck vessels become mediastinal structures. The left subclavian artery arises directly from the aortic arch and courses over the lung apex. The thoracic aorta is tethered at the ligamentum arteriosum just distal to the left subclavian artery origin — this is the most common site of traumatic aortic injury from deceleration.
The intercostal neurovascular bundle runs along the inferior border of each rib in the order Vein-Artery-Nerve (VAN, from superior to inferior). This is why chest tubes and thoracentesis needles are inserted over the SUPERIOR border of the rib, and why rib fractures can cause significant intercostal bleeding. The internal mammary artery (internal thoracic artery) runs 1 cm lateral to the sternum — laceration from sternal fractures or parasternal stab wounds can cause significant hemorrhage requiring ligation.
Abdominal Regions & Organ Vulnerability
The abdomen is divided into the peritoneal cavity (intraperitoneal organs: spleen, liver, stomach, small bowel, transverse colon, sigmoid colon), the retroperitoneum (kidneys, ureters, aorta, IVC, duodenum [2nd–4th parts], pancreas, ascending and descending colon), and the pelvis (bladder, rectum, iliac vessels, uterus). The thoracoabdominal region (from the 4th intercostal space anteriorly / tip of scapula posteriorly to the costal margin) overlaps thorax and abdomen — penetrating wounds here can injure the diaphragm, liver, or spleen.
Retroperitoneal Zones
| Zone | Location | Contents | Exploration Policy |
|---|---|---|---|
| Zone 1 (central/midline) | Aorta to IVC, diaphragm to sacral promontory | Aorta, IVC, proximal renal vessels, pancreas, duodenum | ALWAYS explore (blunt and penetrating) |
| Zone 2 (lateral/perinephric) | Flanks, from renal hilum laterally | Kidneys, renal vessels, ureters, adrenals, ascending/descending colon | Explore if penetrating; blunt — observe if contained and hemodynamically stable (CT-staged) |
| Zone 3 (pelvic) | Below the sacral promontory | Iliac vessels, rectum, bladder | Explore if penetrating; blunt — do NOT open (pelvic fracture hematoma will worsen; use angioembolization/preperitoneal packing) |
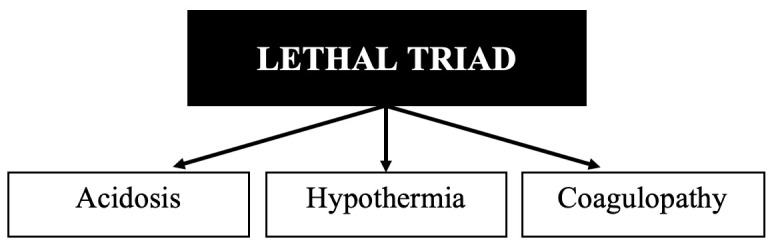
02 Trauma Physiology — Hemorrhagic Shock & the Lethal Triad
Classes of Hemorrhagic Shock (ATLS)
| Parameter | Class I | Class II | Class III | Class IV |
|---|---|---|---|---|
| Blood loss (mL) | <750 | 750–1500 | 1500–2000 | >2000 |
| Blood loss (% BV) | <15% | 15–30% | 30–40% | >40% |
| Heart rate | <100 | 100–120 | 120–140 | >140 |
| Blood pressure | Normal | Normal | Decreased | Decreased |
| Pulse pressure | Normal | Narrowed | Narrowed | Narrowed |
| Respiratory rate | 14–20 | 20–30 | 30–40 | >35 |
| Urine output (mL/hr) | >30 | 20–30 | 5–15 | Negligible |
| Mental status | Slightly anxious | Mildly anxious | Anxious, confused | Confused, lethargic |
| Initial fluid | Crystalloid | Crystalloid | Crystalloid + blood | MTP activation |
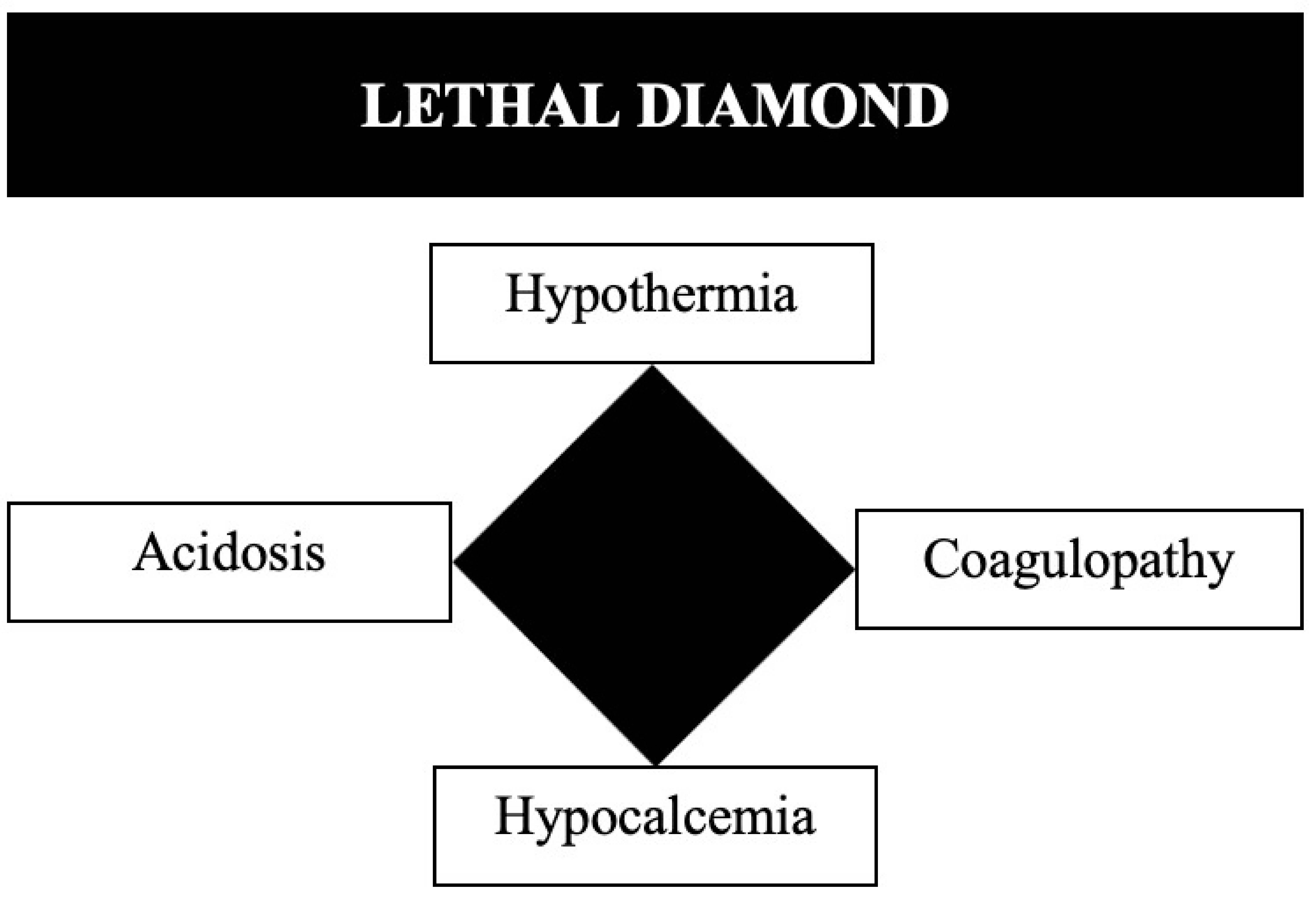
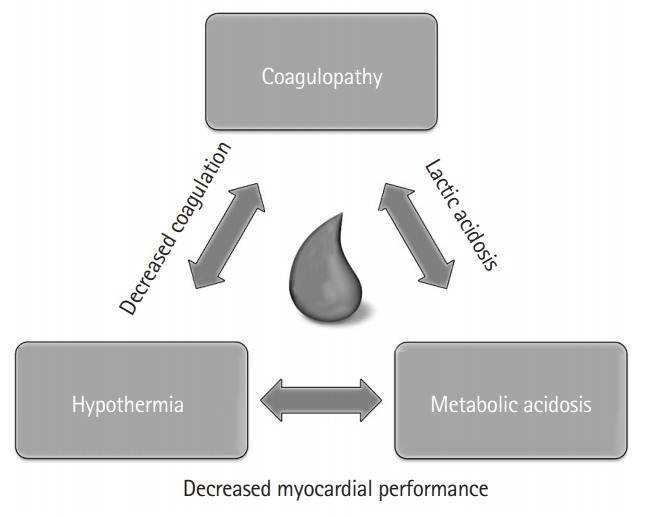
The Lethal Triad
The three mutually reinforcing pathologic states that drive mortality in trauma:
Hypothermia (core temp <35 C): impairs platelet function, slows coagulation enzyme kinetics (10% decrease in clotting factor activity per 1 C drop), increases cardiac irritability, and worsens acidosis. Every trauma patient must have active rewarming measures initiated immediately — warm IV fluids, forced-air warming blankets (Bair Hugger), warm environment, warm lavage fluids.
Acidosis (pH <7.2, base deficit > −6): result of tissue hypoperfusion and anaerobic metabolism producing lactic acid. Impairs myocardial contractility, reduces catecholamine responsiveness, and worsens coagulopathy. Treatment is restoration of perfusion (hemorrhage control + volume resuscitation), NOT routine bicarbonate administration.
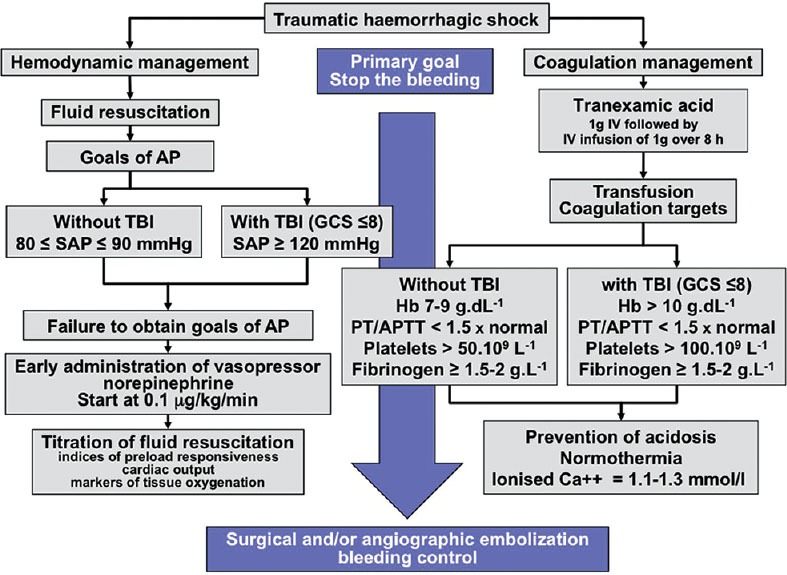
Coagulopathy: multifactorial — dilutional (crystalloid resuscitation), consumptive (DIC from tissue injury), hypothermia-induced, and acidosis-induced. Acute traumatic coagulopathy (ATC) begins within minutes of injury from tissue hypoperfusion and activated protein C pathways. Treated with balanced blood product resuscitation (1:1:1), TXA, correction of hypothermia/acidosis, and calcium replacement.
Resuscitation Physiology
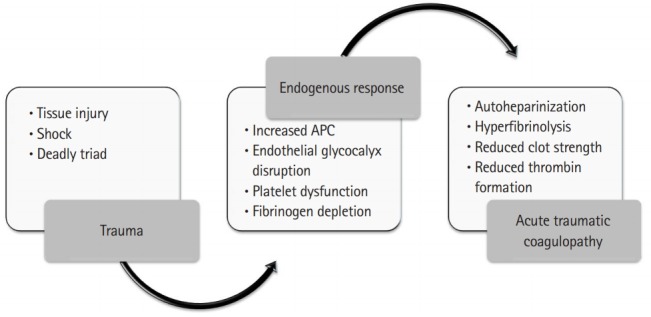
Acute Traumatic Coagulopathy (ATC)
ATC is a distinct entity from the coagulopathy caused by dilution, hypothermia, and acidosis. It develops within minutes of injury through two main pathways: (1) tissue hypoperfusion activates thrombomodulin on endothelium → activated protein C → consumption of factors V and VIII + inhibition of PAI-1 → hyperfibrinolysis; (2) tissue factor release from injured tissue triggers the extrinsic pathway, leading to consumptive coagulopathy. ATC is present in ~25% of severely injured patients on arrival and independently predicts mortality (4x higher mortality). Detection: prolonged PT/INR on arrival, or more precisely via TEG/ROTEM showing prolonged clotting time and hyperfibrinolysis. Treatment: early MTP activation with 1:1:1 resuscitation, TXA, and minimizing crystalloid dilution.
Permissive hypotension: in penetrating torso trauma, targeting a systolic BP of 80–90 mmHg (MAP ~50 mmHg) until surgical hemorrhage control is achieved reduces re-bleeding from disrupted clots, limits dilutional coagulopathy, and improves survival. This strategy does NOT apply to TBI patients (who require MAP >80 to maintain CPP) or to blunt trauma with likely TBI. Damage control resuscitation (DCR): the overarching strategy combining permissive hypotension, limited crystalloid use, early balanced blood product transfusion (1:1:1 ratio of pRBC:FFP:platelets), early TXA, and rapid surgical hemorrhage control. The PROPPR trial (PMID: 25647203) demonstrated that 1:1:1 ratios achieved hemostasis more effectively and reduced death from exsanguination compared to 1:1:2 ratios.
Shock Index
The Shock Index (SI) = heart rate / systolic blood pressure. Normal SI = 0.5–0.7. An SI >0.9 suggests significant hemorrhage and need for transfusion. SI >1.0 is associated with high mortality and should prompt MTP activation. Modified Shock Index (MSI) = HR / MAP is more specific. The Assessment of Blood Consumption (ABC) score uses four binary variables (penetrating mechanism, SBP ≤90, HR ≥120, positive FAST) to predict MTP need — score ≥2 has good sensitivity for massive transfusion requirement.
Non-Hemorrhagic Causes of Shock in Trauma
Not all shock in trauma is hemorrhagic. The trauma surgeon must simultaneously consider and differentiate:
| Type | Mechanism | Key Distinguishing Features | Treatment |
|---|---|---|---|
| Hemorrhagic | Blood loss → decreased preload | Tachycardia, flat neck veins, cool extremities, narrow pulse pressure | Hemorrhage control + blood products |
| Cardiogenic (tamponade) | Pericardial blood → impaired diastolic filling | Beck's triad, distended neck veins, pulsus paradoxus, PEA | Pericardiocentesis → thoracotomy |
| Obstructive (tension PTX) | Mediastinal shift → kinked great veins | Distended neck veins, absent breath sounds, tracheal deviation, hyperresonance | Needle decompression → tube thoracostomy |
| Neurogenic | Spinal cord injury above T6 → loss of sympathetic tone | Hypotension + BRADYCARDIA (unlike hemorrhagic shock), warm/vasodilated extremities, loss of motor/sensory function | Vasopressors (phenylephrine), atropine for bradycardia, cautious fluids |
03 Trauma Assessment Principles & Injury Mechanisms
Mechanism of Injury
Blunt trauma accounts for ~80% of trauma in civilian settings (motor vehicle collisions, falls, assaults, pedestrian strikes). Key kinematics: frontal impact MVC — down-and-under (femur fracture, posterior hip dislocation, patellar fracture) or up-and-over (steering wheel injuries: sternal fracture, cardiac contusion, aortic transection). Lateral impact — contralateral cervical spine fracture, ipsilateral thoracic/abdominal organ injury. Ejection from vehicle has the highest mortality. Falls from height: calcaneal fractures (lover's fracture), lumbar burst fractures, bilateral wrist fractures (Colles').
Penetrating trauma: low-velocity (stab wounds) — injury limited to the wound tract; one-third rule for anterior abdominal stab wounds — approximately 1/3 do not penetrate the peritoneum, 1/3 penetrate but do not cause significant injury, 1/3 cause injury requiring repair. High-velocity (gunshot wounds) — cavitation creates a cone of tissue destruction around the permanent wound tract. The temporary cavity from a high-velocity round can be 10–25x the bullet diameter, causing distant organ injury and microvascular thrombosis. Shotgun wounds at close range (<3 meters) behave like high-velocity injuries. Military rifle rounds (5.56 mm, 7.62 mm) produce devastating temporary cavities, and fragment/tumble inside tissue creating unpredictable wound tracks.
Blast injury has four mechanisms: primary (pressure wave — tympanic membrane rupture [most common], blast lung, bowel perforation); secondary (shrapnel/debris — penetrating injuries); tertiary (body displaced by blast wave — blunt injuries, crush); quaternary (burns, inhalation, radiation, chemical exposure). Blast lung is the most common fatal primary blast injury — presents with dyspnea, hemoptysis, and "butterfly" bilateral infiltrates on CXR. Treatment is supportive; avoid positive pressure ventilation if possible (risk of air embolism through alveolar-capillary disruption).
Transfer Criteria to Trauma Center
Field triage guidelines (CDC, ACS) identify patients who benefit from Level I/II trauma center care. Step 1 (physiologic criteria): GCS <14, SBP <90, RR <10 or >29. Step 2 (anatomic criteria): penetrating injuries to head/neck/torso/extremities proximal to elbow/knee, chest wall instability or deformity (flail chest), ≥2 proximal long bone fractures, crushed/degloved/mangled extremity, amputation proximal to wrist/ankle, pelvic fracture, open/depressed skull fracture, paralysis. Step 3 (mechanism criteria): fall >20 feet (adults) or >10 feet (children), high-risk auto crash (intrusion >12 inches occupant side or >18 inches any site, ejection, death in same passenger compartment, vehicle telemetry data consistent with high risk), auto vs pedestrian/bicyclist thrown/run over or with significant impact, motorcycle crash >20 mph. Step 4 (special considerations): age >55, anticoagulants/bleeding disorders, burns, pregnancy >20 weeks, EMS provider judgment.
Predictive Indices & Triage
| Score | Components | Use |
|---|---|---|
| Revised Trauma Score (RTS) | GCS (weighted 0.9368) + SBP (weighted 0.7326) + RR (weighted 0.2908) | Field triage; RTS <4 = Level I trauma center |
| Injury Severity Score (ISS) | Sum of squares of the 3 highest AIS scores from 6 body regions | ISS >15 = major trauma; ISS >25 = high mortality |
| TRISS | Combines RTS, ISS, age, and mechanism (blunt vs penetrating) | Probability of survival; benchmark for trauma center quality assessment |
Immediate deaths (seconds to minutes): massive CNS injury, aortic disruption, cardiac injury — preventable only by injury prevention. Early deaths (minutes to hours): the "Golden Hour" — hemorrhage, tension pneumothorax, cardiac tamponade, airway obstruction — preventable by rapid prehospital transport and skilled trauma team resuscitation. Late deaths (days to weeks): sepsis, multiorgan failure — preventable by optimal ICU care, early definitive surgery, and infection control. Modern trauma systems have blurred this trimodal distribution, but it remains a foundational teaching concept.
04 ATLS Primary Survey (ABCDE)
The ATLS primary survey is a systematic, priority-based approach to identifying and treating immediately life-threatening injuries. Each step must be completed before moving to the next, and the survey is repeated if the patient deteriorates.
Assess: Can the patient speak clearly? Look for stridor, hoarseness, gurgling, drooling, facial/neck burns, expanding neck hematoma, tracheal deviation. Intervene: chin lift / jaw thrust (NOT head tilt in trauma), suction, oropharyngeal or nasopharyngeal airway (NPA contraindicated in suspected basilar skull fracture), endotracheal intubation with in-line stabilization (maintain C-spine precautions — one person holds manual inline stabilization while another intubates). If unable to intubate → surgical cricothyroidotomy (needle cricothyroidotomy in children <12). C-spine: maintain immobilization with rigid cervical collar until cleared clinically or radiographically.
Assess: Expose the chest. Look for asymmetric chest rise, tracheal deviation, respiratory rate, chest wall instability (flail segment), open wounds, subcutaneous emphysema. Auscultate both lung fields. Percuss (hyperresonance = pneumothorax; dullness = hemothorax). Immediately life-threatening: tension pneumothorax (needle decompression 2nd ICS MCL or 4th/5th ICS MAL → tube thoracostomy), open pneumothorax (3-sided occlusive dressing → tube thoracostomy), massive hemothorax (>1500 mL initial output or >200 mL/hr → thoracotomy), flail chest with pulmonary contusion (intubation if respiratory failure).
Assess: Pulse quality, rate, regularity, skin color/temperature, capillary refill, mental status. Intervene: Direct pressure on external hemorrhage. Two large-bore (≥16-gauge, ideally 14-gauge) peripheral IVs in the antecubital fossae. If peripheral access fails → intraosseous (IO) access (proximal tibia, proximal humerus) or central venous access (femoral preferred in trauma). Begin warm crystalloid (1–2 L bolus) then transition to blood products. Assess for sources of hemorrhage — external (scalp, extremities); chest (hemothorax on CXR/eFAST); abdomen/pelvis (FAST exam); retroperitoneum (CT or pelvic XR); long bones (femur fracture = 1–2 L blood loss per side). Activate MTP if indicated. Apply pelvic binder if pelvic fracture suspected.
Assess: GCS score (eye opening + verbal + motor responses, scored 3–15), pupil size and reactivity (unilateral fixed dilated pupil = ipsilateral uncal herniation until proven otherwise), lateralizing signs (hemiparesis), and spinal cord level. Intervene: GCS ≤8 → intubate for airway protection. Identify and treat reversible causes of altered consciousness: hypoglycemia (check glucose), hypoxia, hypotension (shock), and drug/alcohol intoxication. Avoid secondary brain injury — maintain SBP >100 mmHg, PaO2 >60 mmHg, PaCO2 35–45 mmHg.
Completely undress the patient to identify all injuries. Log-roll to examine the back, spine, and perineum (blood at the urethral meatus, perineal ecchymosis, high-riding prostate suggest urethral injury — do NOT insert a Foley before retrograde urethrogram). Maintain normothermia with warm blankets, forced-air warming, warm IV fluids, and warm environment. Hypothermia is a preventable contributor to the lethal triad.
05 Secondary Survey & Adjuncts
The secondary survey is a head-to-toe physical examination performed only after the primary survey is complete and the patient is hemodynamically responding to resuscitation. It includes a complete history (AMPLE: Allergies, Medications, Past medical history, Last meal, Events of the injury), detailed physical exam of every body region, and review of all imaging.
Adjuncts to the Primary Survey
ECG: all blunt chest trauma (look for arrhythmias from cardiac contusion, PEA from tamponade). Chest X-ray (portable AP): widened mediastinum (>8 cm) suggests aortic injury; hemothorax; pneumothorax; rib fractures (1st/2nd rib fractures = high-energy mechanism, evaluate for aortic/great vessel injury). Pelvic X-ray: AP pelvis in hemodynamically unstable patients with suspected pelvic fracture. Urinary catheter: monitor urine output as marker of perfusion (goal >0.5 mL/kg/hr in adults, >1 mL/kg/hr in children); contraindicated until urethral injury excluded if blood at meatus. Gastric tube: decompress the stomach, reduce aspiration risk; use orogastric (not nasogastric) if suspected basilar skull fracture.
Adjuncts to the Secondary Survey
CT scan: the definitive imaging modality in hemodynamically stable trauma patients. Pan-scan (CT head/C-spine/chest/abdomen/pelvis with IV contrast) is standard for significant mechanisms. CT angiography for suspected vascular injury. Laboratory studies: CBC, type and crossmatch (or type and screen), BMP, coagulation studies (PT/INR, PTT, fibrinogen), lactate, base deficit, ethanol level, urine drug screen, urinalysis, pregnancy test (all women of childbearing age), TEG/ROTEM if available.
06 FAST & eFAST Examination
The Focused Assessment with Sonography in Trauma (FAST) is a bedside ultrasound performed during the primary survey to detect free intraperitoneal fluid (a surrogate for hemoperitoneum). The extended FAST (eFAST) adds bilateral thoracic views to evaluate for pneumothorax and hemothorax.
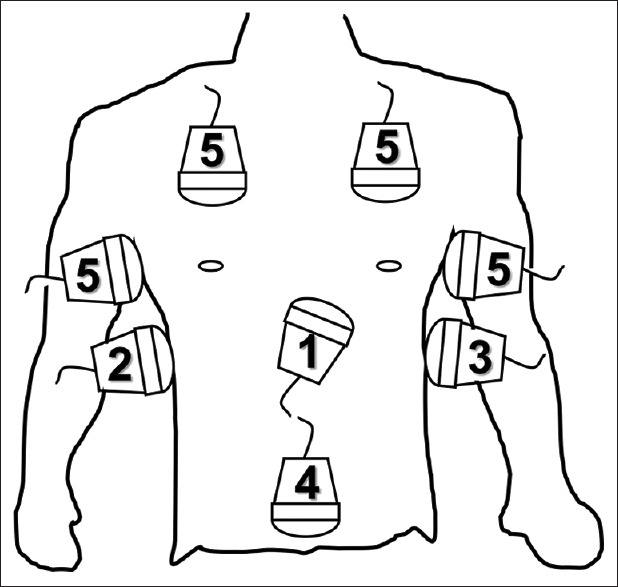
FAST Windows
| View | Probe Position | Structures Evaluated | Findings |
|---|---|---|---|
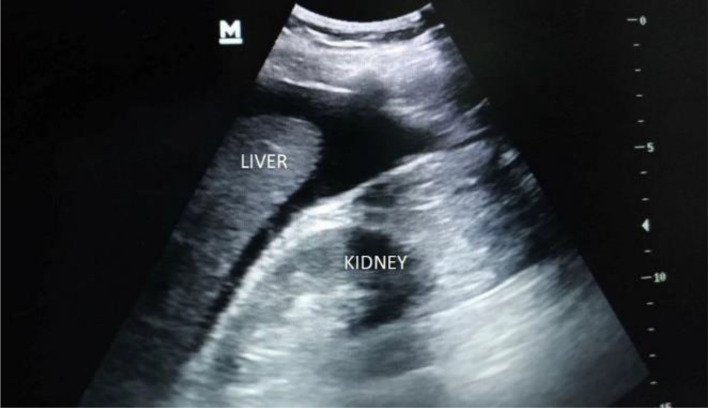
| RUQ (Morrison's pouch) | Right mid-axillary line, 8th–11th intercostal space | Hepatorenal space, right pleural space, subdiaphragmatic space | Anechoic stripe between liver and kidney; most sensitive view (~60% of positive FASTs detected here first) |
| LUQ (splenorenal) | Left posterior axillary line, 6th–9th intercostal space | Splenorenal space, left pleural space, subdiaphragmatic space | Anechoic stripe between spleen and kidney; often most technically difficult view |
| Subxiphoid (pericardial) | Subxiphoid, angled toward left shoulder | Pericardial space | Anechoic stripe around the heart; pericardial effusion/tamponade |
| Suprapubic (pelvic) | Suprapubic, sagittal and transverse | Pouch of Douglas (women) / rectovesical pouch (men) | Free fluid surrounding the bladder |
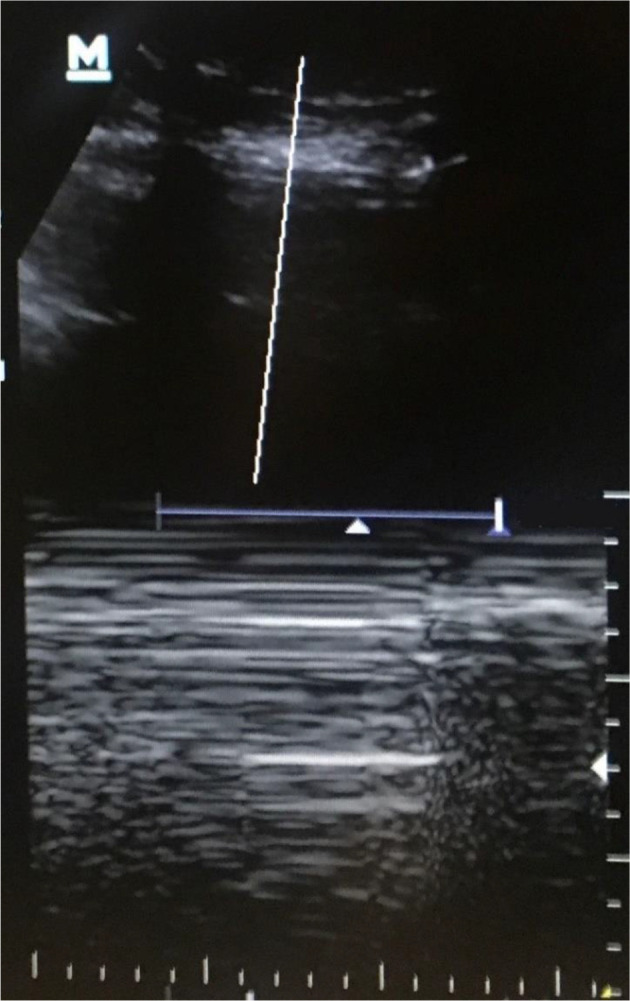
| Bilateral thoracic (eFAST) | Anterior chest, 2nd–3rd ICS MCL | Pleural sliding, B-lines | Absent lung sliding + absent comet-tail artifacts = pneumothorax; "barcode sign" on M-mode confirms |
Hemodynamically unstable + positive FAST → immediate exploratory laparotomy (do not delay for CT). Hemodynamically unstable + negative FAST → consider other sources of hemorrhage (chest, pelvis, retroperitoneum, external); repeat FAST; consider DPL if equivocal. Hemodynamically stable + positive FAST → CT scan for characterization of injury and grading. Hemodynamically stable + negative FAST → FAST sensitivity for free fluid is only ~85%; a negative FAST does NOT exclude injury — proceed with CT if mechanism or clinical exam warrants. FAST cannot detect retroperitoneal hemorrhage, isolated hollow viscus injury, or diaphragmatic injury.
07 Massive Transfusion & Damage Control Resuscitation
Massive Transfusion Protocol (MTP)
Definition: transfusion of ≥10 units of pRBCs within 24 hours, or ≥4 units in 1 hour with ongoing hemorrhage. Modern MTP delivers blood products in a 1:1:1 ratio of pRBC:FFP:platelets (the PROPPR trial standard). Coolers are pre-packaged and delivered in rounds (e.g., 6 units pRBC, 6 units FFP, 1 apheresis platelet unit per round).
Tranexamic Acid (TXA) — CRASH-2 Trial
The landmark CRASH-2 trial (PMID: 20554319) demonstrated that TXA given within 3 hours of injury reduces all-cause mortality in bleeding trauma patients. Dose: 1 g IV over 10 minutes, then 1 g IV over 8 hours. Key finding: TXA given >3 hours after injury INCREASED mortality — timing is critical. TXA is an antifibrinolytic (lysine analog that blocks plasminogen binding to fibrin).
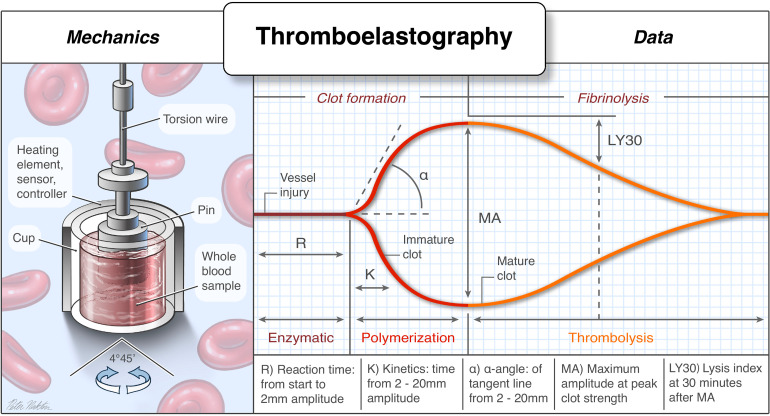
TEG/ROTEM-Guided Resuscitation
Thromboelastography (TEG) and rotational thromboelastometry (ROTEM) provide real-time, viscoelastic assessment of the entire coagulation cascade — from clot formation to fibrinolysis. They guide component therapy more precisely than standard coagulation tests (PT/INR/PTT take 30–60 min; TEG/ROTEM results available in 5–10 min).
| TEG Parameter | ROTEM Equivalent | What It Measures | Abnormal → Treatment |
|---|---|---|---|
| R time (reaction time) | CT (clotting time) | Time to initial fibrin formation (clotting factor activity) | Prolonged → FFP (10–15 mL/kg) |
| K time / alpha angle | CFT / alpha angle | Clot kinetics (fibrinogen contribution) | Prolonged K / low alpha → cryoprecipitate (goal fibrinogen >150 mg/dL) |
| MA (maximum amplitude) | MCF (max clot firmness) | Clot strength (platelet function + fibrinogen) | Low MA → platelets (goal platelet count >50,000) |
| LY30 (lysis at 30 min) | ML (maximum lysis) | Fibrinolysis | LY30 >3% → TXA or aminocaproic acid |
Citrate in stored blood products chelates ionized calcium. Hypocalcemia (iCa <1.0 mmol/L) impairs myocardial contractility, causes coagulopathy, and potentiates vasopressor resistance. Monitor ionized calcium with every blood gas. Replace with calcium chloride 1 g IV (preferred over calcium gluconate in resuscitation — 3x more bioavailable ionized calcium, but must be given through central or large-bore peripheral line due to tissue necrosis risk with extravasation) or calcium gluconate 3 g IV peripherally. Rule of thumb: give 1 g CaCl2 for every 4 units of blood products transfused.
08 Traumatic Brain Injury
Glasgow Coma Scale (GCS)
| Component | Response | Score |
|---|---|---|
| Eye Opening (E) | Spontaneous | 4 |
| To voice | 3 | |
| To pain | 2 | |
| None | 1 | |
| Verbal (V) | Oriented | 5 |
| Confused | 4 | |
| Inappropriate words | 3 | |
| Incomprehensible sounds | 2 | |
| None | 1 | |
| Motor (M) | Obeys commands | 6 |
| Localizes pain | 5 | |
| Withdraws from pain | 4 | |
| Abnormal flexion (decorticate) | 3 | |
| Extension (decerebrate) | 2 | |
| None | 1 |
Mild TBI: GCS 13–15. Moderate TBI: GCS 9–12. Severe TBI: GCS 3–8 (requires intubation).
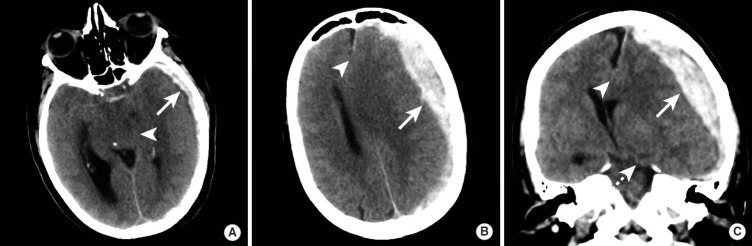
Types of Intracranial Hemorrhage
| Type | Source | CT Appearance | Clinical Features | Management |
|---|---|---|---|---|
| Epidural hematoma (EDH) | Middle meningeal artery (temporal bone fracture); occasionally dural venous sinus | Biconvex (lens-shaped), hyperdense; does NOT cross suture lines | "Lucid interval" — brief LOC → transient improvement → rapid deterioration with ipsilateral pupil dilation (uncal herniation) | Emergent craniotomy if >30 mL, >15 mm thick, or >5 mm midline shift; surgical emergency |
| Subdural hematoma (SDH) | Bridging veins (between cortex and dural venous sinuses) | Crescent-shaped, hyperdense (acute) or mixed/hypodense (chronic); crosses suture lines, does NOT cross midline | More common in elderly/anticoagulated patients; higher mortality than EDH due to underlying brain injury | Craniotomy if >10 mm thick or >5 mm midline shift; chronic SDH may be drained via burr holes |
| Subarachnoid hemorrhage (tSAH) | Cortical vessels, often from contusion | Hyperdensity in sulci and cisterns | Headache, meningismus; traumatic SAH has better prognosis than aneurysmal SAH | Observation, serial imaging, seizure prophylaxis (7 days), consider nimodipine if vasospasm |
| Diffuse axonal injury (DAI) | Shearing of white matter axons from rotational acceleration/deceleration | CT often normal initially; MRI shows punctate hemorrhages at grey-white junction, corpus callosum, brainstem | Coma disproportionate to CT findings; mechanism = high-speed MVC or shaking | Supportive; ICP monitoring; prognosis depends on extent and location (brainstem worst) |
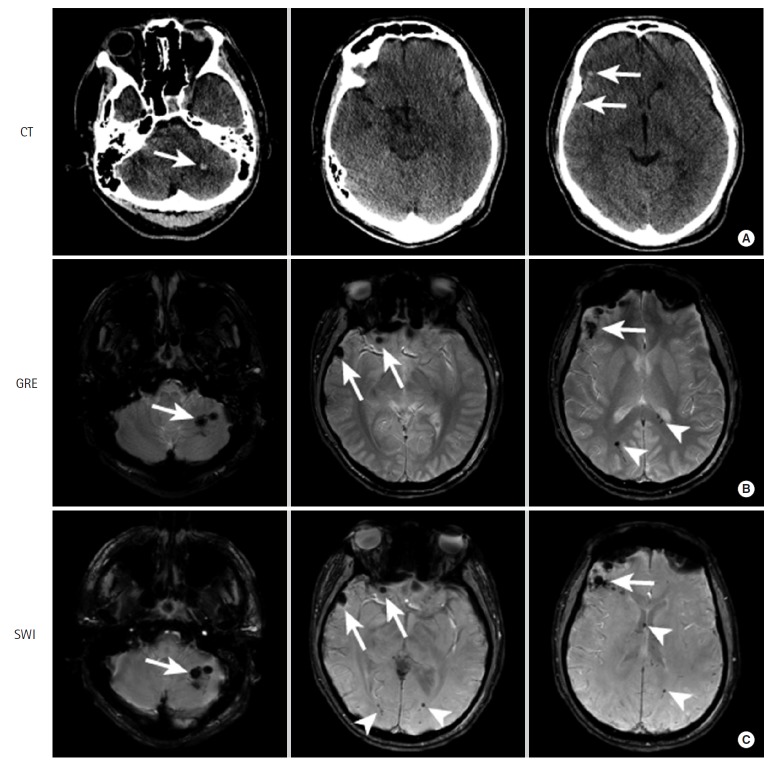
ICP Management (Brain Trauma Foundation Guidelines)
Normal ICP: 5–15 mmHg. Treatment threshold: ICP >22 mmHg (BTF 4th edition). Cerebral perfusion pressure (CPP) = MAP − ICP; target CPP 60–70 mmHg.
Tier 1: Head of bed elevation 30 degrees, midline head position (optimize venous drainage), sedation and analgesia (propofol or midazolam + fentanyl), normothermia, normocarbia (PaCO2 35–45), treat seizures, EVD drainage of CSF if available.
Tier 2: Hyperosmolar therapy — mannitol 0.25–1 g/kg IV (check serum osmolality, hold if >320 mOsm/L or osmolar gap >10) or hypertonic saline 23.4% 30 mL bolus via central line (or 3% NaCl infusion to target Na 145–155). Neuromuscular blockade (cisatracurium).
Tier 3: Decompressive craniectomy (DECRA trial for diffuse injury — PMID: 21434843; RESCUEicp trial for refractory ICP — PMID: 28899894). Barbiturate coma (pentobarbital) — last resort, significant hemodynamic depression. Mild hypothermia (35–36 C) is no longer routinely recommended (Eurotherm3235 trial showed harm).
09 Cervical Spine Injury & Clearance
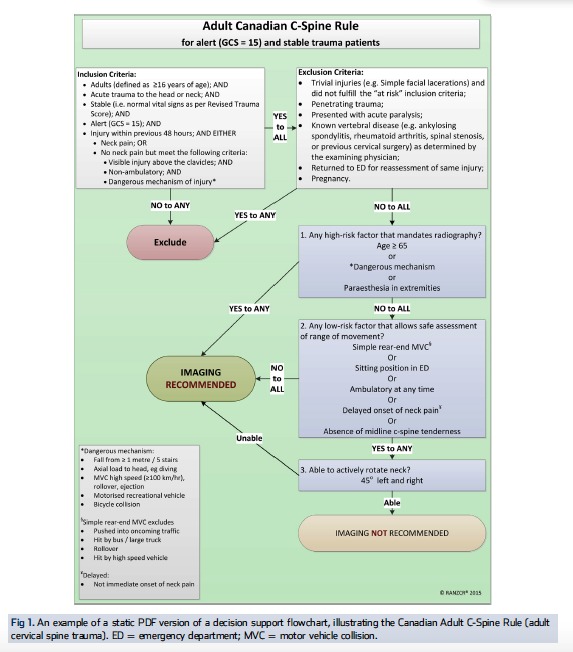
Clinical Clearance Rules
| Criteria | NEXUS | Canadian C-Spine Rule (CCR) |
|---|---|---|
| Applicability | All blunt trauma patients | Alert (GCS 15), stable patients only |
| Key criteria to clear | All 5 must be met: no midline tenderness, no focal neurologic deficit, normal alertness, no intoxication, no distracting injury | Step 1: Any high-risk factor? (age ≥65, dangerous mechanism, paresthesias) → imaging. Step 2: Any low-risk factor allowing ROM testing? (simple rear-end MVC, seated in ED, ambulatory, delayed onset pain, no midline tenderness) → Step 3. Step 3: Can actively rotate neck 45 degrees L and R? → Clear. |
| Sensitivity | 99.6% | 99.4% (more specific, fewer unnecessary images) |
Common C-Spine Injury Patterns
C1 (atlas) fracture — Jefferson fracture: burst fracture from axial loading (diving into shallow water); bilateral lateral mass displacement. Stable if transverse ligament intact (ADI <3 mm adults). C2 (axis) fracture — Odontoid fractures: Type I (tip avulsion — stable), Type II (base of dens — most common, highest nonunion rate, often requires surgical fixation), Type III (extends into C2 body — usually heals with halo). Hangman's fracture: bilateral C2 pars interarticularis fracture — usually stable due to canal widening; treated with collar or halo unless significantly displaced/angulated. Subaxial injuries: facet dislocations (unilateral = rotational instability; bilateral = complete ligamentous disruption, high risk of cord injury — requires closed reduction if awake, open reduction if obtunded).
ASIA Impairment Scale (AIS)
| Grade | Description |
|---|---|
| A — Complete | No motor or sensory function preserved in sacral segments S4–S5 |
| B — Sensory incomplete | Sensory but NOT motor function preserved below the level, including S4–S5 |
| C — Motor incomplete | Motor function preserved below the level; more than half of key muscles have grade <3 |
| D — Motor incomplete | Motor function preserved below the level; at least half of key muscles have grade ≥3 |
| E — Normal | Motor and sensory function are normal |
Sacral sparing (any sensory or motor function in S4–S5 segments — perineal sensation, voluntary anal contraction, great toe flexion) distinguishes incomplete (AIS B–D) from complete (AIS A) injuries and carries a significantly better prognosis for recovery.
Spinal Cord Injury Syndromes
| Syndrome | Mechanism | Deficits | Prognosis |
|---|---|---|---|
| Central cord | Hyperextension in elderly with stenosis | Upper extremity weakness >> lower; variable sensory loss; bladder dysfunction | Best prognosis; arms recover last |
| Anterior cord | Flexion injury or anterior spinal artery occlusion | Loss of motor function, pain, and temperature BELOW the level; preserved proprioception and vibration (posterior columns spared) | Worst prognosis of incomplete injuries |
| Brown-Sequard | Hemisection of cord (penetrating injury) | Ipsilateral motor loss + proprioception loss; contralateral pain/temperature loss | Best overall prognosis of all incomplete injuries |
| Posterior cord | Rare; hyperextension | Loss of proprioception and vibration; motor/pain preserved | Good |
| Cauda equina | Below L1-L2 (below conus medullaris) | Saddle anesthesia, urinary retention, lower extremity weakness (LMN pattern) | Variable; surgical emergency for decompression |
10 Penetrating Neck Trauma
Hard vs Soft Signs
| Hard Signs (mandate exploration) | Soft Signs (require workup) |
|---|---|
| Active arterial hemorrhage or expanding/pulsatile hematoma | History of significant hemorrhage at scene |
| Airway compromise (stridor, hoarseness, air bubbling from wound) | Non-expanding, stable hematoma |
| Hematemesis or massive hemoptysis | Dysphagia or odynophagia |
| Stroke / focal neurologic deficit | Subcutaneous emphysema |
| Air bubbling through wound | Proximity of wound to major structures |
Hard signs present → immediate operative exploration (approach depends on zone: Zone II = anterior SCM incision; Zone I = may need sternotomy/thoracotomy; Zone III = may need mandibular subluxation or angiographic intervention). No hard signs, wound penetrates platysma → CT angiography (CTA) of the neck. CTA has sensitivity >95% and specificity >97% for clinically significant vascular injury. If CTA negative → observation. If CTA positive → operative repair, endovascular stenting, or embolization depending on injury. Wound does NOT penetrate platysma → wound care and discharge.
Evaluate the aerodigestive tract: esophagoscopy + esophagography (combined sensitivity ~95% for esophageal injury), laryngoscopy/bronchoscopy for airway injury. Missed esophageal injury has high mortality — delayed diagnosis leads to mediastinitis and sepsis.
11 Maxillofacial Trauma
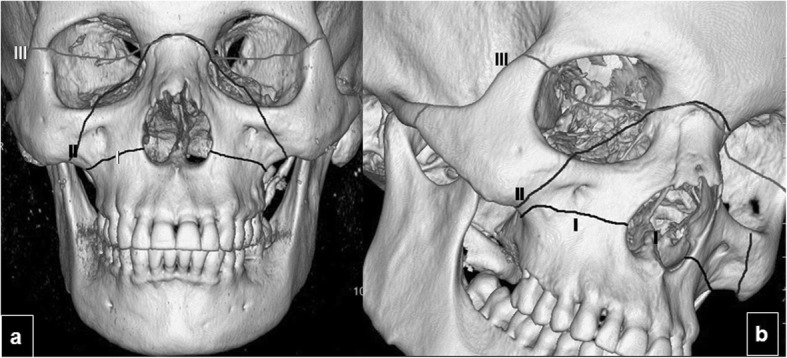
Le Fort Fracture Classification
| Type | Fracture Line | Clinical Finding | Key Association |
|---|---|---|---|
| Le Fort I | Horizontal fracture through maxilla above the alveolar ridge, separating the hard palate from the upper face | Mobile hard palate ("floating palate") | Dental malocclusion |
| Le Fort II | Pyramidal fracture through nasion, medial orbit, and infraorbital rim | Mobile midface (nose + maxilla move as a unit); periorbital ecchymosis ("raccoon eyes") | CSF rhinorrhea (cribriform plate involvement); infraorbital nerve numbness |
| Le Fort III | Craniofacial disjunction through nasofrontal suture, orbits, and zygomatic arches | Entire face mobile relative to cranium ("dish face") | High risk of CSF leak, airway compromise; often requires tracheostomy |
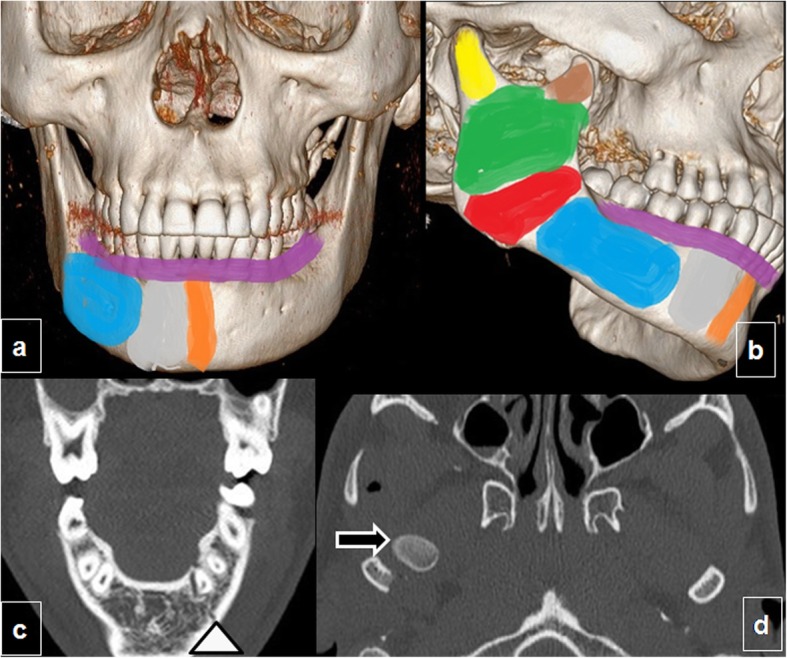
Mandible fractures are the second most common facial fracture. The mandible is a ring structure — fractures often occur in two places (analogous to pelvic ring injuries). Most common sites: condyle (36%), body (21%), angle (20%). Tongue blade bite test: patient bites down on a tongue depressor — inability to fracture it with molar teeth suggests mandibular fracture (negative predictive value ~95%). Management: closed reduction with intermaxillary fixation (wires/arch bars) for non-displaced fractures; ORIF for displaced fractures.
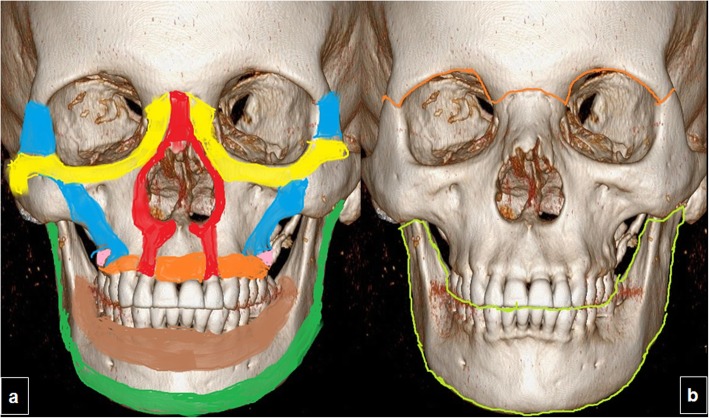
Orbital Fractures
Orbital blowout fracture: isolated fracture of the orbital floor (most common) or medial wall from blunt force to the globe. Clinical findings: periorbital ecchymosis, enophthalmos, infraorbital nerve hypesthesia (numbness of the cheek and upper lip), diplopia on upgaze (inferior rectus entrapment in the fracture), restricted extraocular movement. CT (coronal cuts) is diagnostic — shows fracture with herniation of orbital fat/muscle into the maxillary sinus. Indications for surgical repair (orbital floor plating or mesh): persistent diplopia after 2 weeks, enophthalmos >2 mm, or large floor defect (>50% of floor). Urgency: "white-eyed" blowout fracture in children (trapdoor fracture trapping inferior rectus — bradycardia from oculocardiac reflex) requires urgent repair within 24–48 hours.
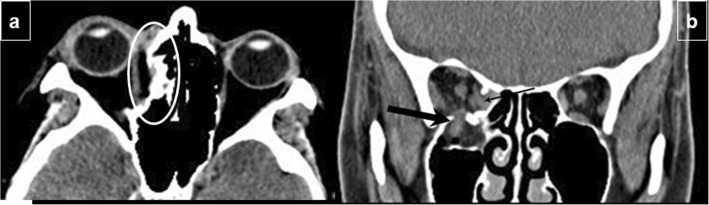
Zygomaticomaxillary Complex (ZMC) Fractures
The tripod fracture involves three suture lines: zygomaticofrontal, zygomaticomaxillary, and zygomaticotemporal. Clinical features: cheek flattening, periorbital ecchymosis, trismus (impingement of zygomatic arch on coronoid process of mandible), step-off at the infraorbital rim, and infraorbital nerve numbness. Non-displaced fractures may be observed; displaced fractures require ORIF at 2–3 of the fracture points.
12 Pneumothorax & Hemothorax
Pneumothorax Types
| Type | Pathophysiology | Clinical Features | Immediate Management |
|---|---|---|---|
| Simple pneumothorax | Air in pleural space without mediastinal shift | Decreased breath sounds, hyperresonance on ipsilateral side; may be asymptomatic if small | Small (<20%, asymptomatic) → observation with serial CXR; large or symptomatic → tube thoracostomy (28–32 Fr, 4th/5th ICS anterior to mid-axillary line) |
| Tension pneumothorax | One-way valve effect → progressive air accumulation → mediastinal shift → kinking of great veins → decreased venous return → cardiovascular collapse | Hypotension, tachycardia, distended neck veins, tracheal deviation AWAY from affected side, absent breath sounds; this is a CLINICAL diagnosis | Do NOT wait for CXR. Needle decompression (14-gauge needle, 2nd ICS MCL or 4th/5th ICS MAL — the SALT-2 study found the 4th/5th ICS MAL has higher success rate) → immediate tube thoracostomy |
| Open pneumothorax | Chest wall defect >2/3 tracheal diameter → air preferentially enters through wound ("sucking chest wound") | Audible sucking sound with respiration, respiratory distress | Three-sided occlusive dressing (tape 3 sides, leave 1 open as flutter valve) → tube thoracostomy placed REMOTE from the wound → subsequent wound debridement and closure |
Hemothorax
Blood in the pleural space — most commonly from intercostal or internal mammary vessel laceration; less commonly from lung parenchyma (usually self-limited) or great vessel/cardiac injury.
Massive hemothorax is defined as >1500 mL of blood immediately evacuated on chest tube placement, OR ongoing output >200 mL/hr for 2–4 hours. Indications for urgent thoracotomy include:
- Initial output >1500 mL
- Ongoing output >200 mL/hr for 2–4 consecutive hours
- Increasing hemothorax despite tube drainage (retained clot)
- Hemodynamic instability not responding to resuscitation
- Need for continued blood transfusion to maintain hemodynamics
Most thoracic hemorrhage (85%) can be managed with tube thoracostomy alone. Autotransfusion of chest tube blood should be utilized when available (blood is collected, filtered, and reinfused).
Rib Fractures
The most common thoracic injury. First and second rib fractures indicate high-energy mechanism — evaluate for aortic/great vessel injury and brachial plexus injury. Fractures of ribs 9–12 should prompt evaluation for hepatic (right) or splenic (left) injury. Mortality increases with number of ribs fractured, especially in elderly patients (mortality doubles with ≥3 rib fractures in patients >65). Pain management is critical to prevent splinting, atelectasis, and pneumonia: multimodal analgesia with acetaminophen + NSAIDs (if no contraindication) + regional anesthesia (thoracic epidural is the gold standard for multiple rib fractures; alternatives include paravertebral block, serratus anterior plane block, and erector spinae block). Incentive spirometry and early mobilization reduce pulmonary complications.
13 Cardiac Injury & Tamponade
Cardiac Tamponade
Accumulation of blood in the pericardial sac (as little as 75–100 mL acutely) compresses the heart, impairs diastolic filling, and causes obstructive shock.
Beck's triad (present in only ~30% of cases): hypotension, distended neck veins (JVD), muffled heart sounds. Additional findings: pulsus paradoxus (>10 mmHg drop in SBP during inspiration), Kussmaul's sign (JVD worsening with inspiration), electrical alternans on ECG (beat-to-beat variation in QRS amplitude), PEA on cardiac monitor. Diagnosis: FAST exam (subxiphoid view) shows pericardial fluid. In the trauma setting, any pericardial fluid in a hemodynamically unstable patient should be treated as tamponade until proven otherwise.
Management: Pericardiocentesis (subxiphoid approach, needle directed toward left shoulder under ultrasound guidance) is a temporizing measure — removing even 15–20 mL can dramatically improve hemodynamics. Definitive treatment is operative exploration — sternotomy (preferred for anterior wounds) or left anterolateral thoracotomy (preferred in ED/resuscitative thoracotomy). Cardiac injuries are repaired with pledgeted horizontal mattress sutures (avoid coronary arteries), and lacerations near coronaries may require passage of sutures beneath the artery.
Blunt Cardiac Injury (Cardiac Contusion)
Mechanism: steering wheel impact, high-speed deceleration. The right ventricle is most commonly injured (anterior position against the sternum). Diagnosis: ECG abnormalities (new RBBB, ST changes, frequent PVCs, atrial fibrillation) + troponin elevation. Management: continuous cardiac monitoring for 24–48 hours. Complications: arrhythmias, ventricular wall rupture (rare, usually fatal), valvular injury (tricuspid most common), cardiac herniation through pericardial tear.
Penetrating Cardiac Injury — Operative Approach
The right ventricle is most commonly injured (40%) due to its anterior position, followed by the left ventricle (35%), right atrium (15%), and left atrium (5%). Most penetrating cardiac injuries present as tamponade rather than exsanguination (the pericardium contains the hemorrhage). Stab wounds have higher survival than gunshot wounds (60–80% vs 10–30%). Repair principles: anterior right ventricular lacerations are repaired with pledgeted horizontal mattress sutures of 3-0 or 4-0 Prolene. Injuries near coronary arteries require sutures placed BENEATH the artery to avoid ligation (coronary ligation can cause infarction). Atrial injuries can be controlled with a side-biting Satinsky clamp and repaired with running Prolene. Posterior cardiac injuries are accessed by elevating the apex ("cradling the heart") — this can cause profound hypotension and arrhythmias.
14 Aortic & Great Vessel Injury
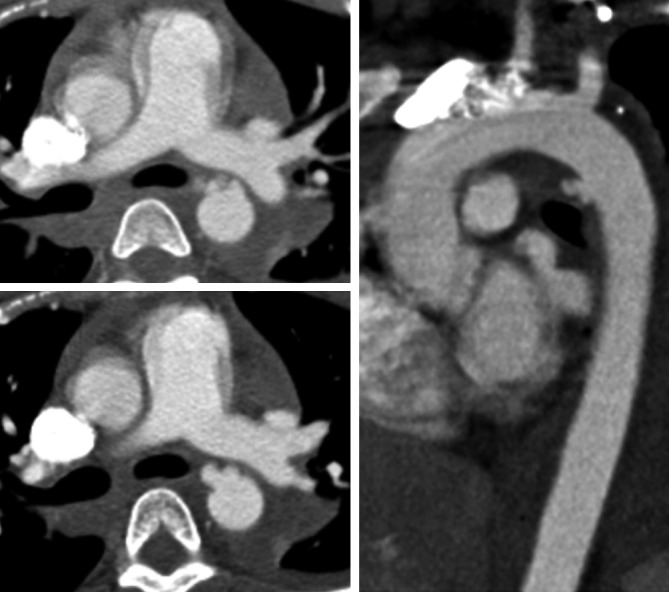
Traumatic Aortic Injury (TAI)
The second leading cause of death in blunt trauma (after TBI). Mechanism: rapid deceleration (high-speed MVC, fall from height) → shearing force at points of anatomic fixation. The aortic isthmus (just distal to the left subclavian artery, at the ligamentum arteriosum) is the most common site (~90% of tears). Other sites: aortic root (usually fatal), mid-descending aorta, diaphragmatic hiatus. Most patients with complete transection die at the scene (80–90% prehospital mortality); those who survive to the hospital have a contained rupture (adventitia and mediastinal pleura tamponade the leak — "pseudoaneurysm"). Of those who arrive alive, 50% will die within 24 hours without intervention.
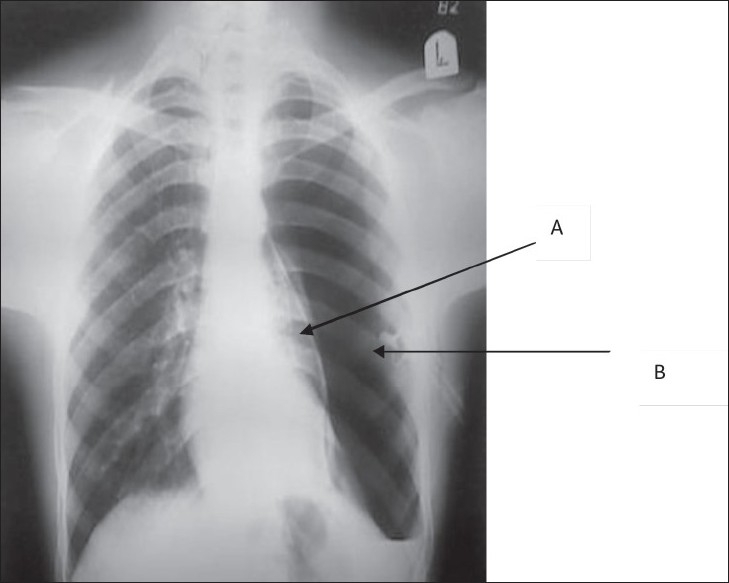
CXR Findings Suggestive of Aortic Injury
Widened mediastinum >8 cm (most sensitive, ~90%); loss of aortic knob contour; deviation of trachea or NG tube to the right; depression of left mainstem bronchus; left apical cap (blood tracking over lung apex); left hemothorax; 1st/2nd rib fractures. None of these are specific — CXR is a screening tool only.
CT angiography of the chest is the diagnostic standard (sensitivity and specificity >98%). Aortic injury is graded: Grade I = intimal tear; Grade II = intramural hematoma; Grade III = pseudoaneurysm; Grade IV = free rupture (rarely survives to imaging).
TEVAR (thoracic endovascular aortic repair) has replaced open repair as the treatment of choice for most traumatic aortic injuries. The AAST prospective multicenter study (PMID: 21307738) showed TEVAR has lower mortality than open repair (7.2% vs 23.5%). For Grade I–II injuries, medical management with anti-impulse therapy (beta-blockers to target HR <80, SBP <120) and serial imaging may be appropriate. Grade III–IV injuries require urgent repair.
Anti-impulse therapy: esmolol infusion (titrate to HR <80 and SBP <120) or labetalol IV boluses. Avoid nitroprusside (causes reflex tachycardia, increases dP/dt). If beta-blockers insufficient, add nicardipine infusion.
15 Pulmonary & Airway Injury
Pulmonary Contusion
The most common potentially lethal chest injury. Hemorrhage and edema within the lung parenchyma from blunt force — appears on CXR/CT as a non-anatomic (non-lobar) opacity in the lung underlying the site of impact, often with rib fractures. Worsens over 24–48 hours. Management: supplemental O2, judicious fluid resuscitation (avoid fluid overload), aggressive pulmonary toilet, intubation/mechanical ventilation if PaO2/FiO2 <200 or progressive respiratory failure. Avoid over-resuscitation — third-spacing of fluid into contused lung worsens gas exchange.
Flail Chest
Defined as ≥3 consecutive ribs fractured in ≥2 places, creating a free-floating segment that moves paradoxically with respiration. The clinical significance is NOT the paradoxical motion but the underlying pulmonary contusion. Management: adequate analgesia (epidural preferred, or paravertebral/serratus anterior nerve block), aggressive pulmonary toilet, non-invasive ventilation (BiPAP), intubation if needed. Surgical rib fixation (rib plating) is increasingly used for severe flail segments — evidence suggests reduced pneumonia, shorter ventilator days, and reduced ICU stay (PMID: 23694879).
Tracheobronchial Injury
Rare but life-threatening. Suspect when: large, persistent air leak through chest tube; pneumomediastinum; subcutaneous emphysema; failure of the lung to fully re-expand after tube thoracostomy. Most injuries occur within 2.5 cm of the carina. Diagnosis: bronchoscopy. Management: small tears (<1/3 circumference) without respiratory compromise may heal with conservative management (selective intubation past the injury); large tears require operative repair via thoracotomy.
Diaphragmatic Injury
Left-sided injury more common (3:1) — the liver protects the right hemidiaphragm. Blunt mechanism causes large radial tears; penetrating causes small perforations that may enlarge over time. CXR findings: elevated hemidiaphragm, NG tube coiled in the chest, bowel gas pattern above the diaphragm. CT sensitivity is ~60–80% (can miss small injuries). Diagnosis may require diagnostic laparoscopy or thoracoscopy. All diaphragmatic injuries require operative repair — primary closure with interrupted non-absorbable sutures (polypropylene or nylon) in blunt trauma; laparoscopic repair may be appropriate for small, penetrating injuries found incidentally.
16 Resuscitative Thoracotomy
Indications
Indicated (highest survival): penetrating cardiac injury with witnessed cardiac arrest or signs of life within the past 15 minutes — survival up to 35%. Selectively indicated: penetrating thoracic injury with loss of vital signs <15 min (survival ~15%); blunt trauma with loss of vital signs <10 min (survival <2%). Contraindicated / futile: blunt trauma with no signs of life for >10 min; penetrating trauma with no signs of life for >15 min; traumatic arrest with asystole and no pericardial fluid.
"Signs of life" = pupillary reactivity, spontaneous movement, organized cardiac electrical activity, or measurable blood pressure at any point during prehospital or ED care. Narrow complex PEA is more likely to have a treatable cause than wide complex PEA. Overall survival for ED thoracotomy across all indications is approximately 7.4% (higher for penetrating cardiac injury, lowest for blunt trauma).
Technique
The standard approach is a left anterolateral thoracotomy in the 4th or 5th intercostal space, from the sternum to the posterior axillary line. Steps:
- Incision: Left anterolateral thoracotomy through the 4th/5th intercostal space. Extend across the sternum to the right chest ("clamshell") if needed for bilateral access.
- Open pericardium: longitudinal incision ANTERIOR to the phrenic nerve → evacuate blood/clot → repair cardiac laceration with pledgeted 3-0 Prolene horizontal mattress sutures (or digital occlusion / Foley catheter balloon tamponade as temporizing measures).
- Cross-clamp the descending thoracic aorta: retract the left lung anteriorly and superiorly → identify the descending aorta against the spine → apply a large vascular (Satinsky) clamp. This redistributes blood flow to the heart and brain and controls subdiaphragmatic hemorrhage.
- Internal cardiac massage: bimanual (two-hand) technique, compressing from apex to base toward the sternum.
- Control pulmonary hilum: twist and clamp the hilum (hilar twist) to control massive pulmonary hemorrhage or air embolism.
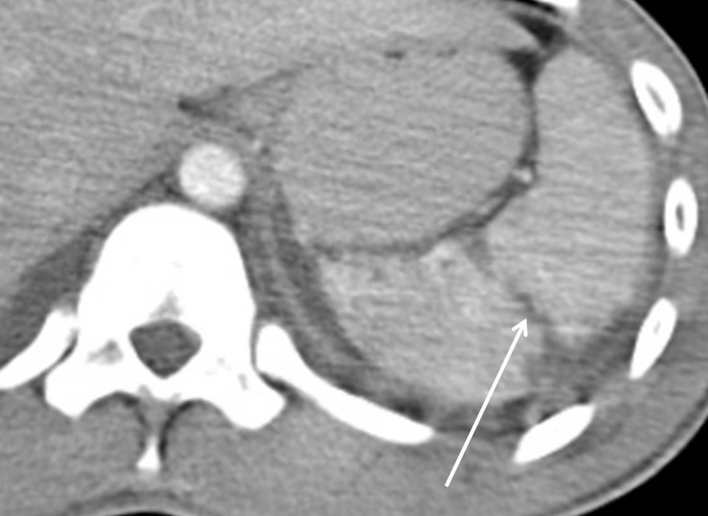
17 Splenic Injury
AAST Spleen Injury Scale
| Grade | Injury Description | Management |
|---|---|---|
| I | Subcapsular hematoma <10% surface area; capsular laceration <1 cm depth | NOM |
| II | Subcapsular hematoma 10–50% surface area; intraparenchymal hematoma <5 cm; laceration 1–3 cm depth | NOM |
| III | Subcapsular hematoma >50% or expanding; ruptured subcapsular/intraparenchymal hematoma ≥5 cm; laceration >3 cm depth | NOM if hemodynamically stable; consider angioembolization for contrast blush on CT |
| IV | Laceration involving segmental or hilar vessels with major devascularization (>25%) | Angioembolization if stable; splenectomy if unstable |
| V | Shattered spleen; hilar vascular injury with complete splenic devascularization | Splenectomy |
Nonoperative Management (NOM)
Prerequisites: hemodynamic stability (or rapid response to resuscitation), no peritonitis, no need for other laparotomy, ability for serial exams and monitoring (ICU or step-down), blood products available. Protocol: bed rest, serial abdominal exams (q4–6h), serial hemoglobin (q6h for 24h, then q12h), NPO initially. NOM success rate is ~90% for grades I–III. Angioembolization has extended NOM to higher-grade injuries — contrast blush (active extravasation) on CT is an indication for embolization even in stable patients. Failure of NOM (hemodynamic instability, falling hemoglobin despite transfusion, peritonitis) → splenectomy.
Post-Splenectomy Considerations
Splenectomy creates lifelong risk of overwhelming post-splenectomy infection (OPSI) — fulminant sepsis from encapsulated organisms (Streptococcus pneumoniae, Haemophilus influenzae, Neisseria meningitidis). Mortality of OPSI is 50–70%. Vaccinate ≥14 days post-splenectomy (or pre-discharge if the patient may not follow up): pneumococcal (PCV20 or PCV15 + PPSV23), meningococcal (MenACWY + MenB), Hib. Patients should carry a medical alert card and receive early antibiotics for any febrile illness.
18 Liver Injury
AAST Liver Injury Scale
| Grade | Injury Description | Management |
|---|---|---|
| I | Subcapsular hematoma <10%; laceration <1 cm depth | NOM |
| II | Subcapsular hematoma 10–50%; intraparenchymal hematoma <10 cm; laceration 1–3 cm depth, <10 cm length | NOM |
| III | Subcapsular hematoma >50% or expanding/ruptured; intraparenchymal hematoma >10 cm; laceration >3 cm depth | NOM if stable; angioembolization for active blush |
| IV | Parenchymal disruption involving 25–75% of lobe; 1–3 Couinaud segments within a single lobe | NOM with angioembolization if stable; operative if unstable |
| V | Parenchymal disruption >75% of lobe; juxtahepatic venous injury (hepatic vein/IVC) | Operative — damage control packing; consider liver transplant for severe injury |
| VI | Hepatic avulsion | Usually fatal; damage control if survivable |
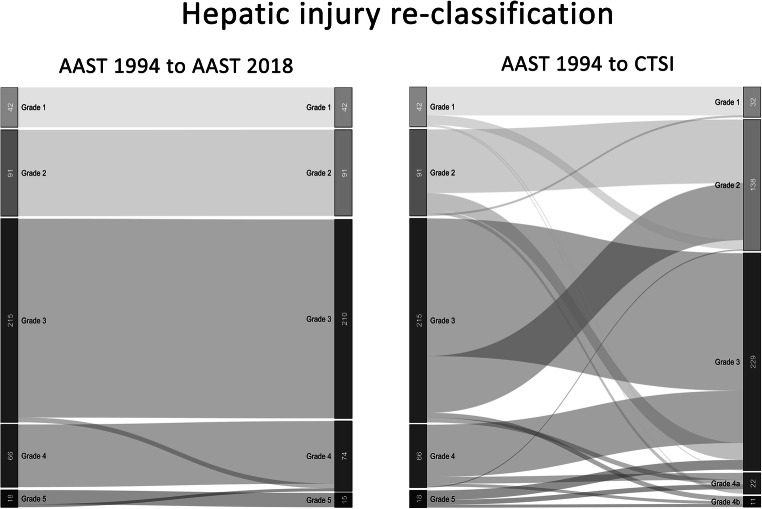
Operative Techniques for Hepatic Hemorrhage
Pringle maneuver: manual or vascular clamp compression of the hepatoduodenal ligament (portal triad — hepatic artery + portal vein + common bile duct) to temporarily control hepatic inflow. Safe for intermittent clamping (15–20 min on, 5 min off). If bleeding stops with Pringle → source is hepatic artery or portal vein (inflow). If bleeding continues → source is hepatic vein or retrohepatic IVC (outflow).
Perihepatic packing: the cornerstone of damage control for liver hemorrhage. Laparotomy pads placed above and below the liver, compressing the liver between the diaphragm and packs. Effective for controlling venous hemorrhage and parenchymal oozing. Leave packs in place, perform temporary abdominal closure, resuscitate in ICU, and return to OR in 24–48 hours for pack removal and definitive repair.
Finger fracture technique: for deep lacerations, the surgeon can digitally fracture hepatic parenchyma along the line of injury to expose bleeding vessels and bile ducts for individual ligation — the hepatic parenchyma is friable but vessels/ducts are more resistant and remain intact for identification and ligation.
Additional techniques: direct suture hepatorrhaphy (horizontal mattress with large chromic/Vicryl suture for simple lacerations), topical hemostatic agents (fibrin sealant, Surgicel, thrombin-soaked Gelfoam), hepatic artery ligation (right or left — the liver tolerates hepatic artery ligation due to dual blood supply, but carries risk of biliary necrosis), resectional debridement (non-anatomic resection of devitalized tissue), and in extreme cases, total hepatic vascular isolation (Pringle + suprahepatic and infrahepatic IVC clamping — extremely poorly tolerated hemodynamically).
19 Hollow Viscus & Mesenteric Injury
Small Bowel Injury
The most commonly injured hollow viscus in penetrating abdominal trauma. In blunt trauma, small bowel injuries are uncommon (~5% of laparotomies for blunt trauma) but easily missed — the CT finding of free intraperitoneal fluid WITHOUT solid organ injury should raise high suspicion. Mechanism in blunt trauma: compression against the spine (especially the fixed duodenojejunal junction at the ligament of Treitz and the distal ileum at the ileocecal valve), or mesenteric tearing with bowel devascularization. CT signs: bowel wall thickening, mesenteric stranding/hematoma, extraluminal air, free fluid without solid organ injury. Management: simple lacerations → primary repair (single or double layer); extensive injury or devascularized segments → segmental resection with anastomosis. In damage control situations, bowel is stapled off (not anastomosed) and left in discontinuity for second-look.
Colon Injury
Primary repair is appropriate for most colon injuries, including in the setting of penetrating trauma — the older practice of mandatory diversion (colostomy) for all colon injuries has been largely abandoned based on multiple prospective studies. Indications for diversion (colostomy): delayed diagnosis (>12 hours), massive fecal contamination, damage control setting, destructive rectal injuries, hypotension requiring vasopressors, or large colon tissue loss requiring tenuous anastomosis. Extraperitoneal rectal injuries: presacral drainage and proximal diversion remain standard.
Gastric Injury
The stomach is relatively protected by the rib cage but can be injured by penetrating trauma (GSW, stab wound) or severe blunt force (full stomach rupture on impact). Most gastric injuries are found during laparotomy for other indications. Management: debridement of devitalized tissue → primary two-layer repair (inner running absorbable, outer interrupted seromuscular Lembert sutures). Always inspect BOTH the anterior and posterior walls — a through-and-through injury requires opening the lesser sac (dividing the gastrocolic ligament) to inspect the posterior stomach wall. Missed posterior gastric wall injuries are a classic pitfall.
Mesenteric Injury
Mesenteric hematomas may compromise bowel blood supply even without direct bowel wall injury. Operative management: inspect the mesenteric border for any compromised bowel (dusky, non-peristaltic, non-bleeding from cut edges → resect). Control mesenteric hemorrhage with suture ligation. Avoid ligating the SMA or large mesenteric branches — loss of significant mesenteric vasculature may require resection of the affected bowel segment. The "seatbelt sign" (ecchymosis across the abdominal wall from a lap belt) is associated with a 5–10x higher incidence of bowel and mesenteric injury — maintain a high index of suspicion in these patients and have a low threshold for CT or even diagnostic laparoscopy.
20 Pancreatic & Duodenal Injury
Pancreatic Injury
Uncommon (~2% of abdominal trauma) but high morbidity. Mechanism: blunt epigastric force compressing the pancreas against the spine (classically, bicycle handlebar injury in children, steering wheel in adults, or assault). The key determinant of management is ductal integrity.
| AAST Grade | Description | Management |
|---|---|---|
| I | Minor contusion/laceration, no duct injury | Drainage (closed suction drain adjacent to pancreas) |
| II | Major contusion/laceration, no duct injury or tissue loss | Drainage |
| III | Distal transection or parenchymal injury WITH duct injury | Distal pancreatectomy ± splenectomy |
| IV | Proximal transection or parenchymal injury involving the ampulla | Drainage + distal pancreatectomy if distal duct injured; complex reconstruction if proximal |
| V | Massive disruption of pancreatic head | Damage control with drainage; delayed Whipple rarely performed (high mortality in trauma) |
Ductal assessment: MRCP (non-invasive, preferred in stable patients), ERCP (diagnostic and therapeutic — stenting for small ductal disruptions), or intraoperative assessment (transection visible, or intraoperative secretin injection with ductal cannulation). CT findings suggestive of pancreatic injury: pancreatic laceration/transection (often subtle), peripancreatic fluid, fluid between the splenic vein and the pancreas (separating the pancreas from its retroperitoneal bed), and thickening of the left anterior renal fascia. CT sensitivity for pancreatic injury is only ~60–80%, especially in the first 12 hours — maintain a high index of suspicion with handlebar or seatbelt mechanism injuries.
Duodenal Injury
Most duodenal injuries result from penetrating trauma. Blunt injuries are uncommon but easily missed. Management depends on location and severity:
Simple lacerations (<50% circumference, no devascularization): primary repair in two layers (inner absorbable, outer seromuscular). Complex injuries (>50% circumference, devascularized, or delayed diagnosis): Roux-en-Y duodenojejunostomy (serosal patch for small defects), pyloric exclusion (close pylorus from inside with absorbable suture + gastrojejunostomy — pylorus reopens spontaneously in 2–3 weeks), or rarely pancreaticoduodenectomy (Whipple) for combined severe pancreatic head and duodenal injuries. Duodenal hematoma (intramural, typically from blunt trauma in children): usually resolves with nasogastric decompression and TPN over 1–3 weeks; operates only if obstruction persists beyond 2–3 weeks or if exploration is required for other reasons.
21 Renal & Urologic Trauma
AAST Kidney Injury Scale
| Grade | Description | Management |
|---|---|---|
| I | Contusion or non-expanding subcapsular hematoma; no laceration | NOM — observation |
| II | Non-expanding perirenal hematoma; cortical laceration <1 cm without collecting system involvement | NOM |
| III | Cortical laceration >1 cm without collecting system rupture or urinary extravasation | NOM if stable |
| IV | Laceration into collecting system with urinary extravasation; renal artery or vein injury with contained hemorrhage; segmental artery/vein thrombosis | NOM if stable; ureteral stenting for collecting system injury; angioembolization for vascular blush |
| V | Shattered kidney; avulsion of renal hilum (devascularized kidney); main renal artery/vein laceration or thrombosis | Exploration — attempt renorrhaphy if possible; nephrectomy if damage control needed or unreconstructable |
Nonoperative management is successful in >90% of blunt renal injuries (grades I–IV). CT with delayed images (10-min delay) assesses collecting system integrity and urinary extravasation. Renal artery thrombosis from blunt deceleration has a very narrow window for revascularization (<4–6 hours of warm ischemia); beyond this, nephrectomy or observation (if contralateral kidney is normal) is preferred.
Ureteral Injury
Rare in trauma (most are iatrogenic from surgery). If identified operatively: simple laceration → primary repair over a stent; complete transection → debridement of devitalized tissue + spatulated, tension-free, stented reanastomosis. Distal ureteral injury → ureteroneocystostomy (reimplantation into the bladder with a psoas hitch or Boari flap for length). Mid-ureteral loss → transureteroureterostomy or ileal interposition graft.
Bladder Injury
Extraperitoneal rupture (80%): associated with pelvic fractures; managed with Foley catheter drainage alone for 10–14 days; heals spontaneously in >95%. Intraperitoneal rupture (20%): results from blunt force to a full bladder (seatbelt injury); requires operative repair — two-layer closure with absorbable suture + Foley drainage. If bladder injury is found during laparotomy for other indications, repair both types operatively. Diagnosis: retrograde cystogram (instill at least 350 mL contrast via Foley, obtain filled and post-drainage films) or CT cystogram. Gross hematuria with pelvic fracture is an absolute indication for cystography.
Urethral Injury
Suspect in males with pelvic fractures. Classic triad: blood at the urethral meatus, perineal/scrotal ecchymosis ("butterfly hematoma"), and high-riding or non-palpable prostate on DRE. Do NOT insert a Foley catheter until urethral injury is excluded by retrograde urethrogram (RUG). Posterior urethral injury (membranous urethra, above the urogenital diaphragm): associated with pelvic fractures; initial management = suprapubic catheter placement; definitive repair (urethroplasty) delayed 3–6 months. Anterior urethral injury (bulbar/penile urethra, below the urogenital diaphragm): straddle injuries; may be managed with primary alignment over a catheter or delayed repair. A single gentle attempt at Foley placement is acceptable if blood at the meatus is absent and clinical suspicion is low, but any resistance mandates RUG.
22 Retroperitoneal Hematoma
Retroperitoneal hematomas (RPH) found during trauma laparotomy require a zone-based management strategy (see Section 01 for zone anatomy).
Zone 1 (central/midline) — ALWAYS explore regardless of mechanism. Central hematomas involve the aorta, IVC, or their major branches. Supramesocolic Zone 1: left medial visceral rotation (Mattox maneuver — mobilize the left colon, kidney, spleen, and pancreatic tail to expose the entire abdominal aorta from the hiatus to bifurcation). Inframesocolic Zone 1: right medial visceral rotation (Cattell-Braasch maneuver — mobilize cecum, ascending colon, and small bowel mesentery rightward to expose the IVC, right iliac vessels, and infrarenal aorta).
Zone 2 (lateral/perinephric) — explore if penetrating; observe if blunt (provided the hematoma is stable/non-expanding and CT shows a manageable injury). If exploration is required, mobilize the colon on the affected side, open Gerota's fascia, and obtain proximal vascular control of the renal hilum before entering the hematoma.
Zone 3 (pelvic) — explore if penetrating; do NOT explore if blunt. Blunt pelvic hematomas from fractures involve disrupted pelvic venous plexus and fractured cancellous bone — opening the hematoma removes tamponade and causes uncontrollable hemorrhage. Manage with pelvic binder, preperitoneal packing, and angioembolization.
23 Peripheral Vascular Injury
Hard vs Soft Signs of Vascular Injury
| Hard Signs (mandate exploration) | Soft Signs (require CTA/ABI) |
|---|---|
| Pulsatile hemorrhage | History of hemorrhage at scene (now controlled) |
| Expanding or pulsatile hematoma | Stable, non-expanding hematoma |
| Absent distal pulses | Diminished (but present) distal pulses |
| Signs of limb ischemia (6 P's: pain, pallor, pulselessness, paresthesias, paralysis, poikilothermia) | Proximity of wound to major vessel |
| Audible bruit / palpable thrill | Nerve deficit adjacent to a named vessel |
Ankle-Brachial Index (ABI): ratio of ankle SBP to brachial SBP measured by Doppler. ABI <0.9 = abnormal, requires CTA or operative exploration. ABI >0.9 in an asymptomatic patient with soft signs has a negative predictive value >99% for clinically significant vascular injury.
Vascular Repair Principles
Obtain proximal and distal vascular control before entering a hematoma. Options for repair: lateral arteriorrhaphy (for small lacerations — ensure no stenosis >50%); patch angioplasty (for larger lacerations that would stenose with primary repair); end-to-end anastomosis (for transections with <2 cm gap after debridement); interposition graft (autologous reversed saphenous vein preferred; PTFE if vein unavailable or for large-caliber vessels); temporary vascular shunt (Argyle, Javid, or Pruitt-Inahara shunt — used in damage control to restore distal flow while other life-threatening injuries are addressed or the patient is transferred). Shunts are secured with silk ties and can remain patent for 24–48 hours.
Mangled Extremity Severity Score (MESS)
| Component | Criteria | Points |
|---|---|---|
| Skeletal/soft tissue injury | Low energy (stab, simple fracture) | 1 |
| Medium energy (open/multiple fracture, dislocation) | 2 | |
| High energy (close-range shotgun, military, crush) | 3 | |
| Very high energy (above + gross contamination, soft tissue avulsion) | 4 | |
| Limb ischemia | Reduced pulse but normal perfusion | 1* |
| Pulseless, paresthesias, reduced capillary refill | 2* | |
| Cool, paralyzed, insensate, numb | 3* | |
| Shock | SBP transiently <90 | 1 |
| Persistent hypotension | 2 | |
| Age | <30 | 0 |
| 30–50 | 1 |
*Score doubled if ischemia time >6 hours. MESS ≥7 has historically predicted the need for amputation with high specificity, though it should not be used as the sole determinant — modern microsurgery and free tissue transfer have salvaged limbs with high MESS scores. The LEAP trial (PMID: 12473698) showed similar long-term functional outcomes between reconstruction and amputation for severe lower extremity injuries.
24 Compartment Syndrome & Fasciotomy
Pathophysiology
Elevated pressure within a closed fascial compartment compromises tissue perfusion, leading to ischemia and necrosis of muscle and nerve. Ischemic time >6 hours causes irreversible damage. Common causes: fractures (tibial shaft most common), crush injuries, vascular injury with ischemia-reperfusion, tight circumferential dressings/casts, burns, massive fluid resuscitation.
Diagnosis
The 6 P's (pain, pressure, paresthesias, paralysis, pallor, pulselessness) — but pain out of proportion to injury and pain with passive stretch of the compartment muscles are the earliest and most sensitive clinical findings. Loss of pulses is a LATE finding — do not wait for this. In obtunded patients, measure compartment pressures with a Stryker pressure monitor: absolute pressure >30 mmHg or delta pressure (diastolic BP minus compartment pressure) <30 mmHg is the threshold for fasciotomy (delta pressure is more reliable than absolute pressure).
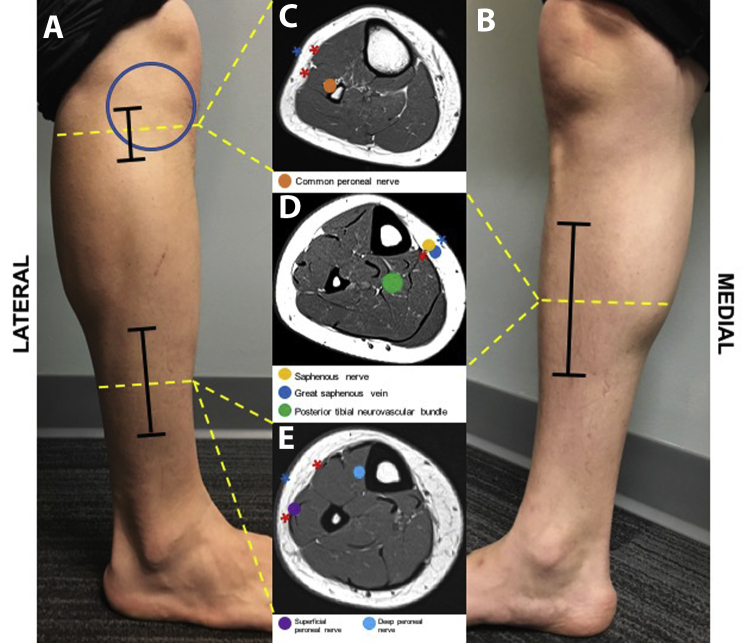
Four-Compartment Lower Leg Fasciotomy
Lateral incision: placed 2 cm anterior to the fibular shaft, from below the fibular head to 4 cm above the lateral malleolus. Through this incision, release the anterior compartment (tibialis anterior, EHL, EDL, deep peroneal nerve, anterior tibial artery) and the lateral compartment (peroneus longus/brevis, superficial peroneal nerve) — identify the intermuscular septum between them and open both compartments longitudinally.
Medial incision: placed 2 cm posterior to the medial tibial border. Through this incision, release the superficial posterior compartment (gastrocnemius, soleus, plantaris) and the deep posterior compartment (tibialis posterior, FHL, FDL, posterior tibial artery, tibial nerve) — the soleus bridge (attachment of soleus to the posteromedial tibial border) must be completely released to decompress the deep posterior compartment.
All fasciotomies: leave wounds open. Apply vacuum-assisted closure (VAC) or wet dressings. Return to OR in 48–72 hours for delayed primary closure, skin grafting, or further debridement.
25 Pelvic Fractures & Hemorrhage Control
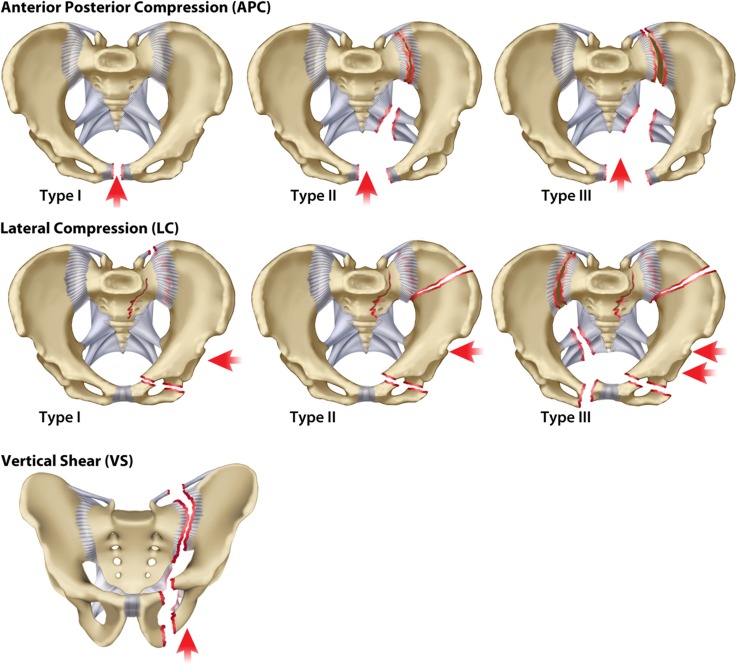
Classification (Young-Burgess)
| Type | Mechanism | Injury Pattern | Hemorrhage Risk |
|---|---|---|---|
| Lateral Compression (LC) | Lateral force (side-impact MVC, pedestrian struck) | LC-I: sacral buckle fracture + ipsilateral pubic ramus fractures. LC-II: + iliac wing fracture (crescent fracture). LC-III: + contralateral external rotation (windswept pelvis) | LC-I/II: low-moderate. LC-III: high |
| Anteroposterior Compression (APC) | Frontal force (motorcycle, pedestrian) | APC-I: symphysis diastasis <2.5 cm. APC-II: >2.5 cm diastasis + anterior SI ligament disruption ("open book"). APC-III: complete SI disruption + posterior ligaments torn | APC-II/III: very high (disrupted pelvic venous plexus) |
| Vertical Shear (VS) | Axial load (fall from height) | Complete hemipelvis displacement (Malgaigne fracture) — vertical displacement of hemipelvis through sacrum/SI joint + pubic rami | Very high (iliac vessel injury, bone surfaces, venous plexus) |
| Combined Mechanism (CM) | Mixed | Combination of above patterns | High |
Hemorrhage Control Algorithm
- Pelvic binder (T-POD, SAM sling, or bedsheet): apply at the level of the greater trochanters to close the pelvic ring and reduce volume (effect on venous and bone hemorrhage). Apply in the field or ED. Binders should NOT be left in place >24–48 hours (skin necrosis).
- Massive transfusion protocol activation if hemorrhagic shock.
- REBOA (Resuscitative Endovascular Balloon Occlusion of the Aorta) in Zone III (infrarenal aorta) — provides temporary hemorrhage control while definitive measures are prepared. Balloon inflation time should be limited (<60–90 min).
- Preperitoneal packing (PPP): midline infraumbilical incision → enter preperitoneal space (do NOT open the peritoneum) → pack 3 laparotomy pads into each side of the pelvic basin → close. Controls venous/bone hemorrhage. Can be performed in the ED or OR in <20 minutes.
- Angioembolization: for arterial hemorrhage (contrast blush on CT or persistent bleeding after packing/binder). Targets the internal iliac artery branches. Performed in angiography suite or hybrid OR.
- External fixation: temporary pelvic stability (anterior frame) if binder insufficient or for definitive management. C-clamp for posterior ring injuries.
26 Orthopedic Trauma Principles
Gustilo-Anderson Classification of Open Fractures
| Type | Wound Size | Soft Tissue Injury | Contamination | Infection Rate |
|---|---|---|---|---|
| I | <1 cm | Minimal, clean wound, inside-out mechanism | Minimal | 0–2% |
| II | 1–10 cm | Moderate soft tissue damage, no flap or avulsion | Moderate | 2–10% |
| IIIA | >10 cm | Extensive soft tissue damage but adequate coverage of bone | High | 10–25% |
| IIIB | >10 cm | Extensive soft tissue loss requiring flap coverage (periosteal stripping, bone exposed) | High | 25–50% |
| IIIC | Any | Any open fracture with arterial injury requiring repair | Variable | 25–50%; amputation rate 25–50% |
Antibiotic Prophylaxis for Open Fractures
Based on the EAST guidelines: Type I–II: first-generation cephalosporin (cefazolin 2 g IV q8h) for 24 hours. Type III: add an aminoglycoside (gentamicin 5 mg/kg IV q24h) for 72 hours or until soft tissue coverage. Farm/soil contamination: add penicillin G (for Clostridium). All patients: verify tetanus prophylaxis status.
Damage Control Orthopedics (DCO)
In polytrauma patients who cannot tolerate definitive fixation (hemodynamically unstable, coagulopathic, hypothermic, head injury), temporary external fixation of long bone fractures is performed to achieve skeletal stability without the "second hit" of prolonged definitive surgery. External fixators are converted to intramedullary nails or plates when the patient stabilizes (typically 5–10 days). Early Total Care (ETC) — definitive fixation within 24 hours — is appropriate for isolated long bone fractures in hemodynamically stable patients, particularly femoral shaft fractures (early fixation reduces pulmonary complications and ICU stay).
Traumatic Amputations
Management in the field: tourniquet application proximal to the amputation (leave in place until operative control), pressure dressing on the stump, wrap the amputated part in saline-moistened gauze → place in plastic bag → place on ice (NOT directly on ice — avoid freezing). Replantation criteria (consider for): thumb amputation, multiple digit amputations, mid-hand or more proximal amputations in adults, almost any level in children. Sharp amputations (clean-cut) have better replantation success than crush/avulsion amputations. Warm ischemia time limit: ~6 hours for digits, ~4 hours for major limbs. Cold ischemia tolerance: ~12 hours for digits, ~6 hours for major limbs.
Traumatic Joint Dislocations
Hip dislocation: 90% posterior (dashboard injury — flexed, adducted, internally rotated hip). Reduce within 6 hours to minimize risk of avascular necrosis (AVN) of the femoral head. Sciatic nerve injury in 10%. Knee dislocation: emergency — popliteal artery injury in 30–40% of cases. After reduction, mandatory ABI measurement: ABI <0.9 → CTA or surgical exploration. Even with normal ABI, serial vascular exams for 24–48 hours are required (intimal injuries may thrombose with delayed occlusion). Multi-ligament knee injury (ACL + PCL ± collaterals) requires vascular assessment regardless of dislocation status.
27 Damage Control Laparotomy
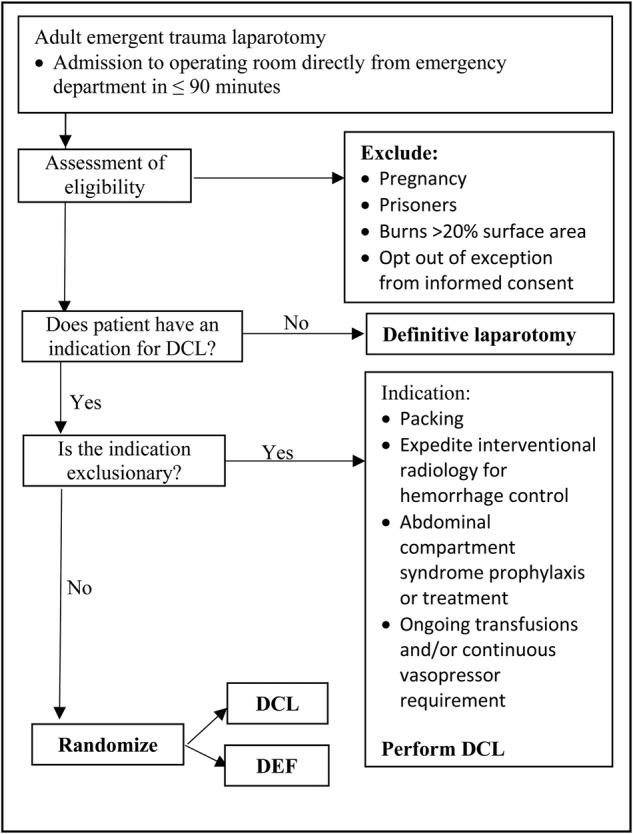
Indications for Damage Control
Damage control is triggered by the patient's physiology, NOT the anatomic injury severity:
- Hypothermia: core temperature <35 C (especially <34 C)
- Acidosis: pH <7.2 or base deficit worse than −8
- Coagulopathy: clinical non-surgical bleeding (diffuse oozing from all surfaces), INR >1.5, fibrinogen <100, transfusion of >10 units pRBC
- Massive blood loss with hemodynamic instability despite resuscitation
- Anticipated procedure time >90 minutes in an unstable patient
- Need for reassessment of bowel viability or injury severity
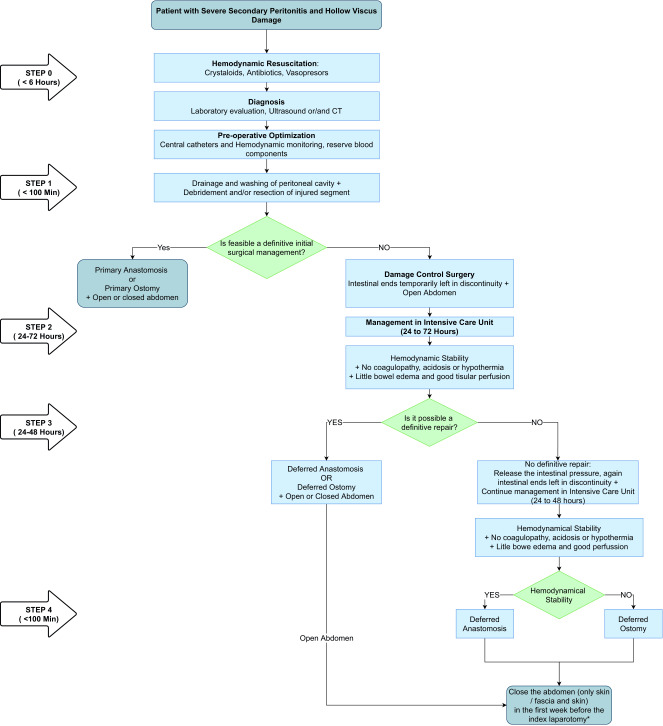
Abbreviated Laparotomy Technique
The damage control laparotomy follows a strict protocol of rapid hemorrhage control and contamination control, without definitive repair:
- Midline laparotomy: full xiphoid-to-pubis incision.
- Four-quadrant packing: pack all four quadrants with laparotomy pads → temporary hemorrhage control → systematic inspection.
- Hemorrhage control: perihepatic packing for liver injuries; splenectomy for severe splenic injury; vascular clamping or shunting for major vessel injuries; ligation of smaller vessels.
- Contamination control: staple across bowel injuries (GIA stapler) — do NOT perform anastomosis; temporarily close bowel ends with suture or stapler. Drain bile/urine if bile duct or bladder injury identified.
- Temporary abdominal closure: do NOT attempt fascial closure (risk of abdominal compartment syndrome). Apply negative pressure wound therapy (Barker vacuum pack or commercial VAC system) or use a Bogota bag (sterile IV bag sewn to skin edges).
- Transfer to ICU for damage control resuscitation.
Total OR time for abbreviated laparotomy should be <60–90 minutes. "You can stop anytime" — if the patient deteriorates, the surgeon should progress to closure immediately.
Damage control for vascular injuries: temporary intravascular shunts (Argyle, Javid, or Pruitt-Inahara) maintain distal flow while definitive repair is delayed. Shunts are particularly useful for extremity vascular injuries when the patient requires truncal surgery first, or when prolonged definitive repair would extend OR time unacceptably. Venous injuries can be ligated in the damage control setting (most single-vein ligations, including the IVC below the renals, are tolerated — expect significant lower extremity edema with IVC ligation). Damage control for urologic injuries: bladder injuries — drain with Foley and/or suprapubic catheter; ureteral injuries — tie off with clips, place ureteral stent, or create a nephrostomy tube for delayed repair.
28 ICU Resuscitation & Planned Reoperation
ICU Phase Goals
The ICU phase of damage control focuses on correcting the lethal triad before returning to the OR:
| Parameter | Goal | Interventions |
|---|---|---|
| Temperature | >36 C | Forced-air warming, warm IV fluids, warm ventilator circuit, bladder/peritoneal warm lavage if severe |
| Acidosis | pH >7.25, base deficit <−6, lactate trending down | Continued resuscitation, restore perfusion; avoid bicarbonate unless pH <7.1 |
| Coagulation | INR <1.5, fibrinogen >150, platelets >50K | Continue MTP products as needed; cryoprecipitate for low fibrinogen; TEG/ROTEM-guided therapy |
| Calcium | iCa >1.0 mmol/L | Calcium chloride or gluconate supplementation |
| Hemodynamics | MAP >65 (or >80 if TBI) | Volume resuscitation → vasopressors (vasopressin 0.04 U/min + norepinephrine) |
Planned Reoperation
Return to OR typically within 24–48 hours once the lethal triad is corrected. At reoperation: remove packing (soak with warm saline before removal to minimize re-bleeding), reassess bowel viability (resect any non-viable segments), perform definitive bowel anastomosis or stoma creation, perform definitive vascular repair (remove shunts, complete grafting), irrigate thoroughly, and attempt fascial closure if possible.
29 Open Abdomen Management
Temporary Abdominal Closure Techniques
| Technique | Description | Advantages | Disadvantages |
|---|---|---|---|
| Negative pressure wound therapy (NPWT / VAC) | Polyurethane sponge placed over viscera (with plastic layer separating sponge from bowel), covered with adhesive drape, connected to suction at −125 mmHg | Best fascial closure rates; most widely used; removes fluid; reduces edema | Requires commercial device; risk of fistula if sponge directly contacts bowel |
| Barker vacuum pack | Fenestrated polyethylene sheet over bowel, moist towels, suction drains, adhesive drape | Inexpensive, widely available, effective | Less controlled than commercial VAC |
| Bogota bag | Sterile IV bag sewn to fascial edges or skin | Simple, fast, inexpensive | Does not remove fluid; no tension on fascia; lower closure rates |
| Wittmann Patch | Velcro-like mesh sewn to fascial edges; progressively tightened at each OR visit | Excellent for staged closure; maintains fascial domain | Requires multiple OR visits; expensive |
Sequential Fascial Closure
The goal is primary fascial closure at the earliest safe opportunity. With each return to OR, the fascia is serially advanced (tightening the NPWT system or Wittmann patch). Fascial closure rates with NPWT are 60–80% when closure is achieved within 7–10 days. After 10–14 days, progressive loss of fascial domain makes primary closure increasingly difficult — the fascial edges retract laterally and the abdominal wall musculature shortens. If primary closure cannot be achieved, the abdomen is managed as a planned ventral hernia: NPWT continued until granulation tissue covers the viscera → split-thickness skin graft over granulation tissue → delayed ventral hernia repair (6–12 months after skin grafting, once the patient has fully recovered).
Techniques to facilitate delayed closure: component separation (external oblique release), progressive pneumoperitoneum (daily insufflation of air into the peritoneal cavity to stretch the abdominal wall and recover fascial domain), and biologic mesh bridging (porcine or human acellular dermal matrix placed as a bridge between fascial edges — vascularizes and incorporates, providing a scaffold for tissue ingrowth, though long-term hernia rates remain high).
30 Burn Assessment & Fluid Resuscitation
Burn Depth Classification
| Depth | Old Terminology | Appearance | Sensation | Healing |
|---|---|---|---|---|
| Superficial | 1st degree | Erythema, dry, no blisters (sunburn) | Painful | 3–5 days; no scarring |
| Superficial partial-thickness | 2nd degree (superficial) | Blisters, pink/red moist base, blanches with pressure | Very painful | 7–14 days; minimal scarring |
| Deep partial-thickness | 2nd degree (deep) | White or mottled pink, less moisture, sluggish capillary refill | Decreased sensation | 3–8 weeks; significant scarring; may require excision/grafting |
| Full-thickness | 3rd degree | White, waxy, leathery, or charred; thrombosed vessels visible; dry, inelastic | Insensate (nerve destruction) | Will not heal without surgery; excision + skin grafting required |
| 4th degree | — | Extends into muscle, bone, or tendon | Insensate | Requires extensive debridement, flaps, or amputation |
Total Body Surface Area (TBSA) Estimation
Rule of Nines (adults): head/neck = 9%; each upper extremity = 9%; anterior trunk = 18%; posterior trunk = 18%; each lower extremity = 18%; perineum = 1%. Lund-Browder chart: more accurate, especially in children (adjusts for proportional differences — e.g., an infant's head is 18% TBSA vs 9% in adults). Palmar method: the patient's palm (including fingers) ≈ 1% TBSA — useful for small or scattered burns. Only include partial-thickness and full-thickness burns in TBSA calculation for fluid resuscitation (superficial burns are excluded).
Fluid Resuscitation — Parkland Formula
4 mL x body weight (kg) x %TBSA burned = total volume of lactated Ringer's in the first 24 hours. Give half in the first 8 hours (from the time of burn, not arrival) and half over the next 16 hours. Titrate to urine output: 0.5–1.0 mL/kg/hr in adults; 1.0–1.5 mL/kg/hr in children. Avoid "fluid creep" (excessive resuscitation) — monitor for abdominal compartment syndrome, extremity compartment syndrome, and pulmonary edema. Consider colloid (albumin 5%) after 12–24 hours if resuscitation requirements are exceeding predicted volumes.

Escharotomy
Circumferential full-thickness burns create a non-elastic eschar that, as underlying tissue swells, can cause compartment syndrome of the extremity or restrict chest wall excursion (respiratory compromise). Indications: circumferential extremity burns with diminished pulses, poor capillary refill, or elevated compartment pressures; circumferential chest burns with impaired ventilation. Technique: full-thickness incision through eschar only (not into subcutaneous fat) along the medial and lateral mid-axial lines of extremities or along the anterior axillary lines of the chest. No anesthesia required (the eschar is insensate). Bleeding is usually minimal (full-thickness burns destroy dermal vasculature).
Burn Center Transfer Criteria (ABA)
Partial-thickness burns >10% TBSA; burns involving face, hands, feet, genitalia, perineum, or major joints; full-thickness burns of any size; electrical/chemical burns; inhalation injury; burns in patients with significant comorbidities; burns with concomitant trauma (where burn is the greatest risk); children in facilities without qualified pediatric burn capabilities.
Electrical Burns
Low voltage (<1000 V): primarily causes cutaneous thermal burns at entry/exit sites. High voltage (≥1000 V): current travels through tissues of least resistance (nerves, blood vessels, muscle) causing deep tissue destruction far exceeding the visible surface burn. Complications: rhabdomyolysis (aggressive IV fluids to maintain UOP >1–2 mL/kg/hr; alkalinize urine if myoglobinuria), cardiac arrhythmias (ECG monitoring for 24 hours — high voltage, loss of consciousness, or abnormal initial ECG), compartment syndrome (fasciotomy often required), and delayed neurologic deficits (transverse myelitis, peripheral neuropathy). Lightning injury: direct strike, side flash, ground current; cardiac arrest from asystole (often reversible with CPR, unlike VFib in other settings — "reverse triage" — treat the apparently dead first).
Chemical Burns
Treatment principle: copious water irrigation for at least 20–30 minutes — begin immediately. Remove all contaminated clothing. Do NOT attempt neutralization (exothermic reaction worsens injury). Specific agents: hydrofluoric acid (HF) — uniquely dangerous; fluoride ion penetrates tissue and chelates calcium, causing severe hypocalcemia, cardiac arrhythmias (QT prolongation), and progressive deep tissue necrosis. Treatment: topical calcium gluconate gel (2.5%) to the affected area; for significant exposures, intradermal or intra-arterial calcium gluconate injection; monitor serum calcium and ECG. Alkali burns (cement, lye, oven cleaners) cause liquefactive necrosis — deeper penetration than acid burns (coagulative necrosis with acid creates a protective eschar). Irrigate extensively.
31 Inhalation Injury, Hypothermia, & Frostbite
Inhalation Injury
Three components: supraglottic thermal injury (heat is efficiently dissipated by the oropharynx — direct thermal injury is rare below the glottis unless steam exposure); lower airway chemical injury (combustion products — acrolein, aldehydes, HCl — cause mucosal edema, bronchospasm, mucosal sloughing); systemic toxicity (carbon monoxide [CO] and cyanide [HCN]). Suspect inhalation injury with: facial burns, singed nasal hairs/eyebrows, soot in oropharynx/sputum, hoarseness, stridor, history of enclosed-space fire. Diagnosis confirmed by fiberoptic bronchoscopy (mucosal edema, erythema, carbonaceous deposits, ulceration). Management: early intubation (airway edema progresses rapidly — intubate prophylactically if signs of impending obstruction), aggressive pulmonary toilet, bronchodilators.
Carbon monoxide poisoning: CO has 200–250x greater affinity for hemoglobin than oxygen → carboxyhemoglobin (COHb) shifts the oxyhemoglobin dissociation curve left → tissue hypoxia. Pulse oximetry is falsely normal (reads COHb as O2Hb). Diagnosis: co-oximetry on ABG. Treatment: 100% FiO2 via NRB or mechanical ventilation (half-life of COHb: room air = 5 hours; 100% O2 = 60–90 min; hyperbaric O2 = 20–30 min). Hyperbaric O2 is indicated for COHb >25%, LOC, pregnancy, or end-organ dysfunction. Cyanide poisoning: suspect in enclosed-space fire victims with persistent lactic acidosis despite normal PaO2. Treatment: hydroxocobalamin 5 g IV (Cyanokit) — first-line; binds cyanide directly. Alternative: sodium thiosulfate.
Hypothermia
| Stage | Core Temp | Clinical Features | Management |
|---|---|---|---|
| Mild | 32–35 C | Shivering, tachycardia, vasoconstriction, impaired judgment | Passive external rewarming (warm blankets, warm environment); warm IV fluids |
| Moderate | 28–32 C | Shivering ceases, bradycardia, atrial fibrillation, decreased LOC | Active external rewarming (forced-air warming); warm IV fluids; cardiac monitoring (risk of Vfib) |
| Severe | <28 C | Loss of reflexes, Vfib/asystole, pulmonary edema, appears dead | Active internal rewarming (warm lavage — peritoneal, thoracic; warm humidified O2; ECMO/cardiopulmonary bypass for arrest); handle gently (Vfib triggered by rough handling) |
Frostbite
Classified by depth: 1st degree (superficial — erythema, edema, no blisters → full recovery); 2nd degree (partial-thickness — clear blisters, erythema → usually heals); 3rd degree (full-thickness — hemorrhagic blisters, skin necrosis); 4th degree (extends to muscle/bone — mummification, likely amputation). Treatment: rapid rewarming in 37–39 C water bath for 15–30 minutes (do NOT use dry heat, do NOT rub). TPA within 24 hours may improve limb salvage in severe frostbite with absent flow on angiography. Do NOT debride early — "frostbite in January, amputate in July" (allow complete demarcation before definitive surgery).
32 Chest Tube, Cricothyroidotomy, & DPL
Chest Tube Thoracostomy
Indication: pneumothorax (symptomatic or tension), hemothorax, post-thoracotomy. Position: 4th or 5th intercostal space, anterior to mid-axillary line, in the "safe triangle" (anterior border of latissimus dorsi, lateral border of pectoralis major, horizontal line at nipple level, apex of axilla). Size: 28–32 Fr for hemothorax; 24–28 Fr for pneumothorax. Technique: 2–3 cm skin incision over the rib below the target intercostal space → blunt dissection with Kelly clamp over the TOP of the rib (avoids the intercostal neurovascular bundle on the inferior rib margin) → puncture the parietal pleura → finger sweep to confirm entry into pleural space and sweep for adhesions → direct tube posteriorly and superiorly (for hemothorax drainage) → connect to water seal (−20 cm H2O) → secure with suture → CXR to confirm position. Document initial output volume.
Surgical Cricothyroidotomy
Indication: "can't intubate, can't oxygenate" scenario. Contraindicated in children <12 (use needle cricothyroidotomy). Technique: palpate the cricothyroid membrane (between thyroid cartilage and cricoid cartilage — the most accessible and avascular area of the airway). Stabilize the larynx. Horizontal stab incision through skin and membrane → insert tracheal hook to maintain airway → dilate with hemostats or Trousseau dilator → insert cuffed tracheostomy tube (6.0) or cuffed ETT (6.0). Surgical cricothyroidotomy should be converted to formal tracheostomy within 24–72 hours if prolonged airway is needed.
Diagnostic Peritoneal Lavage (DPL)
Largely replaced by FAST and CT, but remains useful when ultrasound is unavailable or equivocal in unstable patients. Technique: decompress the stomach (NG tube) and bladder (Foley). Open (infraumbilical or supraumbilical if pelvic fracture) or Seldinger technique. Aspirate: grossly positive if >10 mL of gross blood (→ laparotomy). If not grossly positive, infuse 1 L warm LR (10 mL/kg in children) → drain by gravity → send for lab analysis. Positive lavage criteria: RBC >100,000/mm3 (blunt) or >5,000/mm3 (penetrating); WBC >500/mm3; amylase >175 IU/dL; bacteria or bile on Gram stain; food particles. DPL is very sensitive (~98%) but not specific — it detects ANY intra-abdominal blood and does not grade injuries or guide nonoperative management.
33 REBOA
Resuscitative Endovascular Balloon Occlusion of the Aorta
REBOA is an endovascular adjunct for temporary aortic occlusion to control non-compressible torso hemorrhage (NCTH), serving as a less-invasive alternative to resuscitative thoracotomy with aortic cross-clamp.
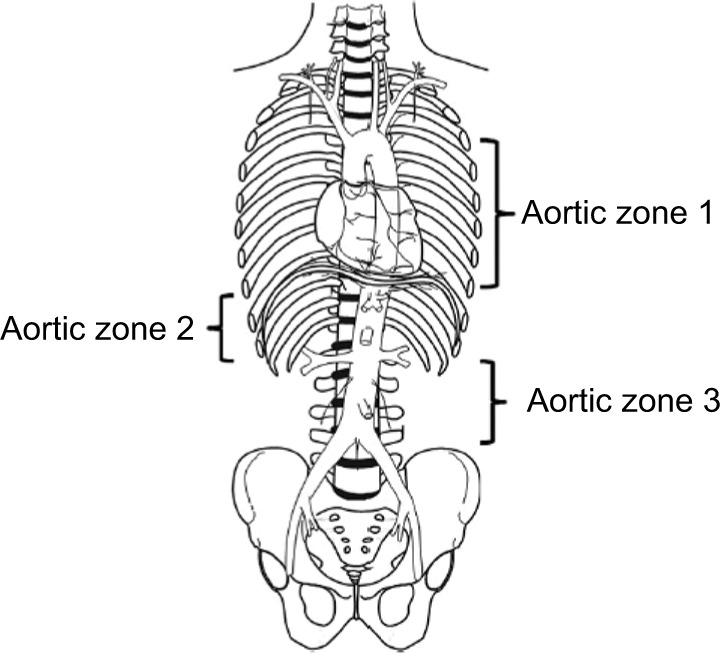
Zone I (descending thoracic aorta, from left subclavian to celiac trunk): occlusion for abdominal/junctional hemorrhage (analogous to aortic cross-clamp during resuscitative thoracotomy). Insertion depth ~46 cm from femoral artery.
Zone II (paravisceral aorta, celiac trunk to renal arteries): NO balloon inflation — risk of mesenteric and renal ischemia.
Zone III (infrarenal aorta, below renal arteries to bifurcation): occlusion for pelvic and junctional hemorrhage. Insertion depth ~28 cm from femoral artery.
Indications: hemorrhagic shock (SBP <90) from suspected non-compressible torso hemorrhage (abdominal or pelvic) refractory to resuscitation, as a bridge to definitive hemorrhage control (OR or angiography). Contraindications: suspected thoracic aortic injury, proximal femoral/iliac injury precluding access.
Technique: Common femoral artery access (ultrasound-guided, 7 Fr sheath). Advance balloon catheter (ER-REBOA catheter) to the appropriate zone under fluoroscopic guidance (or using external landmarks). Inflate balloon until loss of contralateral femoral pulse (Zone I) or until blood pressure response. Partial REBOA (partial inflation) may reduce ischemia-reperfusion injury. Complications: aortic injury, iliac/femoral injury, distal ischemia, balloon migration, ischemia-reperfusion injury on deflation (hyperkalemia, lactic acidosis, cardiac arrest). Time limit: Zone I inflation should be <60 minutes; Zone III <90 minutes. Transition to definitive care as rapidly as possible.
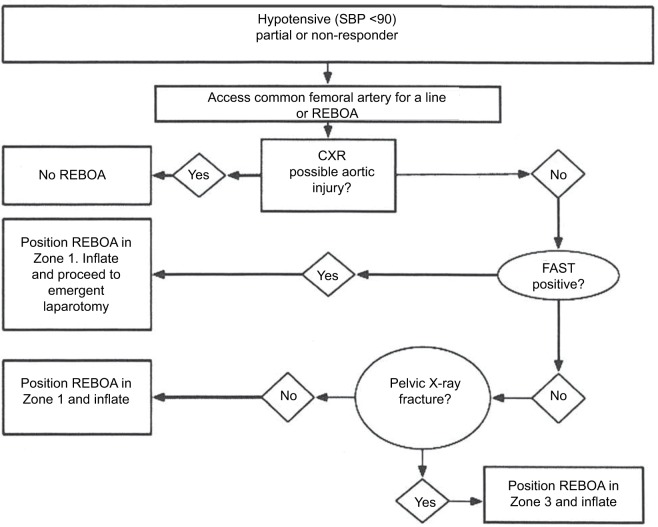
REBOA vs Resuscitative Thoracotomy
| Feature | REBOA | Resuscitative Thoracotomy |
|---|---|---|
| Access | Percutaneous femoral artery (7 Fr sheath) | Open left anterolateral thoracotomy |
| Skill set | Endovascular / procedural | Open surgical |
| Setup time | ~5–10 minutes | ~2–3 minutes |
| Best application | Subdiaphragmatic hemorrhage in patient NOT yet in arrest | Penetrating cardiac/thoracic injury, cardiac arrest |
| Aortic occlusion | Infrarenal (Zone III) or supraceliac (Zone I) | Descending thoracic aorta (cross-clamp) |
| Advantage | Less invasive, fewer blood products, can be performed in ED or angio suite | Allows pericardiotomy, cardiac repair, internal cardiac massage, hilar clamping |
| Limitation | Cannot address thoracic pathology; requires vascular access (may fail in severe shock) | Highly invasive; high complication rate in survivors |
34 Fasciotomy Techniques
Fasciotomy technique for the four-compartment lower leg release is detailed in Section 24. Additional fasciotomy sites:
Forearm Fasciotomy
Volar (flexor) compartment: curvilinear incision from proximal to the medial epicondyle, crossing the antecubital fossa, extending distally along the ulnar aspect of the forearm to the wrist, then crossing into the palm (carpal tunnel release). Release the lacertus fibrosus, flexor-pronator mass, and carpal tunnel. Dorsal (extensor) compartment: straight longitudinal incision over the mobile wad (lateral forearm). The mobile wad (brachioradialis, ECRL, ECRB) lies between the volar and dorsal compartments.
Thigh Fasciotomy
Three compartments: anterior (quadriceps), medial (adductors), posterior (hamstrings). Lateral incision from the greater trochanter to the lateral femoral condyle releases the anterior and posterior compartments through the lateral intermuscular septum. A separate medial incision is used for the medial compartment.
Foot Fasciotomy
Nine compartments in the foot. Two dorsal incisions (medial: over the 2nd metatarsal; lateral: over the 4th metatarsal) access interosseous and adductor compartments. A medial incision posterior to the medial malleolus accesses the calcaneal compartment. Most commonly performed for crush injuries or calcaneal fractures.
Prophylactic Fasciotomy Indications
Fasciotomy should be performed prophylactically (before clinical compartment syndrome develops) in the following high-risk situations: combined arterial and venous injury with >4–6 hours of ischemia before revascularization; crush injury with prolonged entrapment (>4 hours); massive fluid resuscitation (≥6 L crystalloid); high-velocity injury with significant soft tissue destruction; and after repair of any popliteal artery injury (prophylactic 4-compartment fasciotomy is recommended by many surgeons given the high rate of subsequent compartment syndrome). Delayed fasciotomy (>8 hours after onset of compartment syndrome) carries a high risk of infection and should be balanced against the likelihood of non-viable muscle requiring debridement.
35 Blood Products & Tranexamic Acid
Blood Product Overview
| Product | Contents | Volume | Effect | Key Notes |
|---|---|---|---|---|
| Packed RBCs (pRBC) | RBCs in additive solution (CPDA or AS-1), Hct ~55–80% | ~300 mL | 1 unit raises Hgb by ~1 g/dL | Universal donor: O-negative (females of childbearing age) or O-positive (males and post-menopausal females); stored at 1–6 C; shelf life 42 days |
| Fresh Frozen Plasma (FFP) | All coagulation factors, fibrinogen (~2 g/L) | ~250 mL | 1 unit raises factor levels by ~3–5% | Must be ABO-compatible; universal donor: AB; thaw time 20–30 min (keep thawed plasma available) |
| Platelets | Pooled (4–6 donor) or apheresis (single donor) | ~50 mL (single) or ~300 mL (apheresis) | 1 apheresis unit raises count by ~30–50K | Stored at room temperature with agitation; shelf life only 5 days; ABO matching preferred but not required |
| Cryoprecipitate | Fibrinogen (150–250 mg/unit), Factor VIII, vWF, Factor XIII | ~15 mL per unit | 10-unit pool raises fibrinogen by ~70 mg/dL | Primary indication: fibrinogen <150 mg/dL; also used for Factor XIII deficiency, hemophilia A (if concentrates unavailable) |
| Whole blood | RBCs + plasma + platelets + WBCs in physiologic ratios | ~500 mL | Balanced resuscitation in a single product | Increasingly used in military and civilian trauma; cold-stored (1–6 C) or warm fresh whole blood; limited civilian availability |
Tranexamic Acid (TXA) — Detailed
Mechanism: synthetic lysine analog that competitively inhibits plasminogen activation → prevents fibrinolysis → stabilizes clot. Dose: 1 g IV bolus over 10 min (within 3 hours of injury) → 1 g IV infusion over 8 hours. Key evidence: CRASH-2 (PMID: 20554319) — 1.5% absolute mortality reduction when given within 3 hours; TXA >3 hours INCREASED mortality. CRASH-3 (TBI) — modest benefit in mild-to-moderate TBI when given early; no benefit in severe TBI with GCS 3–8. MATTERs study (military) — TXA + MTP associated with 6.5% absolute mortality reduction. Contraindications: time >3 hours from injury, active thromboembolic disease, DIC with predominant thrombosis.
36 Reversal Agents, Vasopressors, & Adjuncts
Anticoagulant Reversal Agents
| Anticoagulant | Reversal Agent | Dose | Notes |
|---|---|---|---|
| Warfarin | 4-Factor PCC (Kcentra) + Vitamin K | PCC: INR 2–4 → 25 U/kg; INR 4–6 → 35 U/kg; INR >6 → 50 U/kg. Vitamin K 10 mg IV slow push | PCC works in minutes; Vitamin K takes 6–12 hours but provides sustained reversal. FFP is second-line (large volume, slower). |
| Dabigatran (Pradaxa) | Idarucizumab (Praxbind) | 5 g IV (two 2.5 g vials) | Monoclonal antibody fragment; reverses within minutes; RE-VERSE AD trial |
| Rivaroxaban, apixaban (Factor Xa inhibitors) | Andexanet alfa (Andexxa) | Low dose: 400 mg bolus + 480 mg infusion. High dose: 800 mg bolus + 960 mg infusion | Recombinant modified Factor Xa decoy; very expensive; 4-Factor PCC 50 U/kg is an alternative if andexanet unavailable |
| Heparin (UFH) | Protamine sulfate | 1 mg per 100 units of heparin given in prior 2–3 hours (max 50 mg) | Partially reverses LMWH (~60%); risk of anaphylaxis (fish allergy, prior protamine exposure) |
Vasopressors in Trauma
Volume resuscitation is the first-line treatment for hemorrhagic shock. Vasopressors are used as a bridge while hemorrhage control and blood product resuscitation are ongoing — they are NOT a substitute for volume.
| Agent | Dose | Mechanism | Role in Trauma |
|---|---|---|---|
| Vasopressin | 0.01–0.04 U/min (fixed dose, no titration) | V1 receptor agonist → vascular smooth muscle contraction | First-line in vasodilatory shock after hemorrhage; replenishes endogenous vasopressin (depleted in prolonged shock). AVERT trial showed benefit. |
| Norepinephrine | 0.01–0.5 mcg/kg/min | Alpha-1 > beta-1 agonist | Standard vasopressor for persistent hypotension after volume resuscitation |
| Phenylephrine | 0.1–0.5 mcg/kg/min | Pure alpha-1 agonist | Neurogenic shock (spinal cord injury with loss of sympathetic tone — hypotension + bradycardia) |
Additional Adjuncts
Tetanus prophylaxis: all trauma patients with wounds — give Td/Tdap if last dose >5 years ago; add TIG (tetanus immune globulin 250 U IM) if unknown vaccination history or <3 prior doses. Antibiotics for open fractures: see Section 26. VTE prophylaxis: all trauma patients are at high risk for DVT/PE. Start LMWH (enoxaparin 30 mg SC q12h) as early as safe — within 24–48 hours for most injuries (hold for active hemorrhage, intracranial hemorrhage, incomplete spinal cord injury, or high-grade solid organ injury with active blush). Mechanical prophylaxis (SCDs) from admission. IVC filter placement only if VTE prophylaxis is contraindicated for >72 hours AND high-risk features present. Dose adjustments: consider enoxaparin 40 mg SC q12h in obese patients (>120 kg) — measure anti-Xa levels (target 0.2–0.4 IU/mL for prophylaxis). Unfractionated heparin 5000 U SC q8h is an alternative in patients with CrCl <30 mL/min.
Stress ulcer prophylaxis: indicated in mechanically ventilated patients, coagulopathic patients, and those with TBI, burns >35% TBSA, or spinal cord injury. Proton pump inhibitors (pantoprazole 40 mg IV daily) or H2 blockers (famotidine 20 mg IV q12h). Transition to enteral PPI when tolerating feeds. Discontinue when risk factors resolve.
Nutritional support: early enteral nutrition (within 24–48 hours) is strongly recommended in trauma patients. Trophic feeds (10–20 mL/hr) via nasogastric or post-pyloric tube should be started as soon as resuscitation is complete. Goals: 25–30 kcal/kg/day, 1.5–2 g protein/kg/day (trauma patients are hypermetabolic/hypercatabolic). Parenteral nutrition is reserved for patients who cannot tolerate enteral feeds for >7 days. Open abdomen patients present a unique challenge — enteral feeding is still recommended (jejunal access preferred) and is safe even with exposed bowel, provided there is intestinal continuity.
37 Classification Systems (All)
AAST Organ Injury Scales — Summary
AAST grading for spleen (Section 17), liver (Section 18), kidney (Section 21), and pancreas (Section 20) are detailed in their respective sections. The grading system (I–V or I–VI) is universally used for operative decision-making and reporting.
Glasgow Coma Scale
Detailed in Section 08. Range: 3–15. Severe TBI ≤8, moderate 9–12, mild 13–15.
Hemorrhagic Shock Classes (ATLS)
Detailed in Section 02. Classes I–IV based on estimated blood loss and physiologic parameters.
Gustilo-Anderson Classification
Detailed in Section 26. Types I, II, IIIA, IIIB, IIIC for open fractures.
MESS (Mangled Extremity Severity Score)
Detailed in Section 23. Score ≥7 historically predictive of amputation.
AAST Organ Injury Scale — Diaphragm
| Grade | Description |
|---|---|
| I | Contusion |
| II | Laceration ≤2 cm |
| III | Laceration 2–10 cm |
| IV | Laceration >10 cm with tissue loss ≤25 cm2 |
| V | Laceration with tissue loss >25 cm2 |
Additional Classification Systems
| System | Use | Key Points |
|---|---|---|
| TLICS (Thoracolumbar Injury Classification and Severity) | Thoracolumbar spine fractures | Scores morphology (compression 1, burst 2, translational/rotational 3, distraction 4) + posterior ligamentous complex (intact 0, suspected 2, disrupted 3) + neurologic status (intact 0, nerve root 2, cord incomplete 3, cord complete 2, cauda equina 3). Score ≤3 → nonoperative; 4 → equivocal; ≥5 → surgical stabilization |
| SLICS (Subaxial Cervical Spine Injury Classification) | Subaxial (C3–C7) cervical spine injuries | Morphology + disco-ligamentous complex + neurologic status. Score ≤3 → nonoperative; 4 → equivocal; ≥5 → surgical |
| Young-Burgess Pelvic Fracture Classification | Pelvic ring injuries | LC, APC, VS, CM — detailed in Section 25 |
| Denver Screening Criteria | Blunt cerebrovascular injury (BCVI) | Risk factors for carotid/vertebral injury: Le Fort II/III, basilar skull fracture through carotid canal, cervical spine fracture (subluxation, transverse process of C1–C3), DAI with GCS <6, near-hanging, soft tissue neck injury (seatbelt sign). CTA screening → treat with antiplatelet or anticoagulation if positive |
| Clavien-Dindo Classification | Postoperative complications | Grade I–V. Grade I = deviation from normal course; II = pharmacologic treatment; IIIa = intervention without GA; IIIb = intervention with GA; IVa = single organ failure + ICU; IVb = multiorgan failure; V = death |
38 Complications — Missed Injury, ACS, ARDS, & DVT/PE
Missed Injury
Missed injury rate in major trauma: 2–10%. Most commonly missed: thoracolumbar spine fractures, small pneumothoraces, extremity fractures (especially in obtunded/intubated patients), hollow viscus injuries (CT sensitivity for bowel injury is only ~90%), diaphragmatic tears (especially small penetrating injuries to the left diaphragm), and peripheral nerve injuries.
Timing: within 24 hours of admission AND when the patient first becomes evaluable (awake, sober, cooperative). Components: complete re-examination head-to-toe, review of ALL imaging (not just the initial reads — re-review with radiology for missed findings), review of laboratory results (delayed rise in amylase, WBC, lactate may suggest evolving injury), and documentation of any new findings. Impact: tertiary surveys identify clinically significant missed injuries in 2–10% of trauma admissions and reduce delayed diagnosis rates by >50%. Late presentations of missed injuries include: peritonitis (hollow viscus perforation), respiratory distress (diaphragmatic hernia with bowel strangulation), sepsis (missed pancreatic or retroperitoneal injury), compartment syndrome (delayed swelling in extremity injuries), and delayed splenic rupture.
Abdominal Compartment Syndrome (ACS)
Sustained intra-abdominal pressure (IAP) >20 mmHg with new organ dysfunction. Normal IAP: 5–7 mmHg. Measured via bladder pressure transduction (instill 25 mL saline into Foley → measure pressure at end-expiration). Causes in trauma: massive fluid resuscitation, retroperitoneal hemorrhage, visceral edema, tight abdominal closure.
Grade I (IAP 12–15 mmHg): monitor. Grade II (16–20): diuresis, neuromuscular blockade, evacuate space-occupying lesions (blood, ascites). Grade III (21–25): likely requires decompressive laparotomy if organ dysfunction present. Grade IV (>25): decompressive laparotomy. Surgical decompression: re-open midline laparotomy (or create one) → leave fascia open → temporary abdominal closure (NPWT). Immediately improves ventilatory compliance, renal function, and cardiac output. Watch for reperfusion syndrome on decompression (hyperkalemia, hypotension, cardiac arrest — pretreat with calcium, bicarbonate, and prepare for aggressive resuscitation).
ARDS (Acute Respiratory Distress Syndrome)
Berlin Definition: acute onset (within 1 week of insult), bilateral opacities on CXR (not fully explained by effusions/atelectasis), respiratory failure not fully explained by cardiac failure, PaO2/FiO2 ratio: mild 200–300, moderate 100–200, severe <100 (on PEEP ≥5). Common causes in trauma: pulmonary contusion, aspiration, massive transfusion (TRALI), sepsis, fat embolism. Management: lung-protective ventilation (tidal volume 6 mL/kg ideal body weight, plateau pressure <30 cm H2O — ARDSNet protocol), PEEP titration, prone positioning for moderate-severe ARDS (PROSEVA trial — PMID: 23688302), conservative fluid strategy (FACTT trial), and neuromuscular blockade in severe cases.
DVT/PE
Trauma patients are at very high risk for VTE due to Virchow's triad: stasis (immobilization, pelvic/lower extremity fractures), endothelial injury (vascular trauma, surgery), and hypercoagulability (acute phase response, blood product transfusion). Prophylaxis: LMWH (enoxaparin 30 mg SC q12h) started as early as safely possible + mechanical compression devices. Screening duplex ultrasound is debated — some centers screen all high-risk patients before mobilization. Treatment of confirmed DVT/PE: therapeutic anticoagulation (heparin drip → transition to LMWH or DOAC). Massive PE with hemodynamic instability → thrombolysis (alteplase), catheter-directed therapy, or surgical thrombectomy.
Fat Embolism Syndrome
Classic triad: respiratory distress, neurologic changes (confusion, agitation), and petechial rash (upper chest, axillae, conjunctivae). Occurs 24–72 hours after long bone fractures (especially femoral shaft). Diagnosis is clinical (Gurd's criteria). Treatment: supportive — O2, mechanical ventilation if needed. Prevention: early fracture fixation reduces incidence.
Venous Thromboembolism — Specific Trauma Considerations
The highest-risk trauma populations for VTE include: spinal cord injury (DVT rate 60–80% without prophylaxis), pelvic fractures, lower extremity fractures requiring immobilization, and prolonged ICU stays. Risk stratification tools (e.g., Greenfield Risk Assessment Profile for trauma) can guide prophylaxis intensity. Duplex screening ultrasound before mobilization is used at some trauma centers for high-risk patients. IVC filters are reserved for patients who cannot receive anticoagulation for >72 hours AND have a high risk profile — retrievable filters should be removed as soon as anticoagulation can be safely initiated. Complications of IVC filters include DVT below the filter, IVC thrombosis, filter migration, IVC perforation, and failure to retrieve.
Infection & Sepsis
Post-traumatic infections include surgical site infection (SSI), pneumonia (particularly ventilator-associated pneumonia — VAP), urinary tract infection (catheter-associated), bloodstream infections (central line-associated — CLABSI), empyema, intra-abdominal abscess, and wound/soft tissue infections (necrotizing fasciitis in contaminated wounds). Empiric antibiotics for post-traumatic intra-abdominal sepsis: piperacillin-tazobactam or carbapenem. Duration guidelines: hollow viscus perforation repaired within 24 hours requires only 24 hours of antibiotics (not prolonged courses). Damage control patients with open abdomens are at particular risk for fungal infections (Candida) — consider empiric antifungal therapy if multi-drug resistant bacteria or clinical deterioration despite antibiotics.
39 Abbreviations Master List
| Abbreviation | Full Term |
|---|---|
| AAST | American Association for the Surgery of Trauma |
| ABI | Ankle-Brachial Index |
| ACS | Abdominal Compartment Syndrome |
| APC | Anteroposterior Compression (pelvic fracture) |
| ARDS | Acute Respiratory Distress Syndrome |
| ATC | Acute Traumatic Coagulopathy |
| ATLS | Advanced Trauma Life Support |
| BTF | Brain Trauma Foundation |
| CCR | Canadian C-Spine Rule |
| COHb | Carboxyhemoglobin |
| CPP | Cerebral Perfusion Pressure |
| CTA | Computed Tomography Angiography |
| DAI | Diffuse Axonal Injury |
| DCO | Damage Control Orthopedics |
| DCR | Damage Control Resuscitation |
| DCS | Damage Control Surgery |
| DPL | Diagnostic Peritoneal Lavage |
| DVT | Deep Vein Thrombosis |
| EAF | Enteroatmospheric Fistula |
| EDH | Epidural Hematoma |
| eFAST | Extended Focused Assessment with Sonography in Trauma |
| ETC | Early Total Care |
| EVD | External Ventricular Drain |
| FAST | Focused Assessment with Sonography in Trauma |
| FFP | Fresh Frozen Plasma |
| GCS | Glasgow Coma Scale |
| IAP | Intra-Abdominal Pressure |
| ICP | Intracranial Pressure |
| IO | Intraosseous |
| ISS | Injury Severity Score |
| IVC | Inferior Vena Cava |
| LC | Lateral Compression (pelvic fracture) |
| LMWH | Low Molecular Weight Heparin |
| MAP | Mean Arterial Pressure |
| MESS | Mangled Extremity Severity Score |
| MTP | Massive Transfusion Protocol |
| NCTH | Non-Compressible Torso Hemorrhage |
| NEXUS | National Emergency X-Radiography Utilization Study |
| NOM | Nonoperative Management |
| NPWT | Negative Pressure Wound Therapy |
| OPSI | Overwhelming Post-Splenectomy Infection |
| PCC | Prothrombin Complex Concentrate |
| PE | Pulmonary Embolism |
| PPP | Preperitoneal Packing |
| pRBC | Packed Red Blood Cells |
| REBOA | Resuscitative Endovascular Balloon Occlusion of the Aorta |
| ROTEM | Rotational Thromboelastometry |
| RPH | Retroperitoneal Hematoma |
| RTS | Revised Trauma Score |
| SAH | Subarachnoid Hemorrhage |
| SCI | Spinal Cord Injury |
| SDH | Subdural Hematoma |
| SI | Shock Index |
| SLICS | Subaxial Cervical Spine Injury Classification |
| TAI | Traumatic Aortic Injury |
| TBSA | Total Body Surface Area |
| TEG | Thromboelastography |
| TEVAR | Thoracic Endovascular Aortic Repair |
| TLICS | Thoracolumbar Injury Classification and Severity |
| TPA | Tissue Plasminogen Activator |
| TRALI | Transfusion-Related Acute Lung Injury |
| TRISS | Trauma and Injury Severity Score |
| TXA | Tranexamic Acid |
| VAC | Vacuum-Assisted Closure |
| VS | Vertical Shear (pelvic fracture) |
| VTE | Venous Thromboembolism |
References & Figure Sources
Figures
- Figure 1 — Neck Anatomy. Key anatomical structures of the neck relevant to penetrating trauma. After Gray's Anatomy (1918). Wikimedia Commons. Public domain.
Key Trials & Guidelines
- CRASH-2 Trial Collaborators. Effects of tranexamic acid on death, vascular occlusive events, and blood transfusion in trauma patients with significant haemorrhage (CRASH-2). Lancet. 2010;376(9734):23-32. PMID: 20554319
- Holcomb JB, Tilley BC, Baraniuk S, et al. Transfusion of plasma, platelets, and red blood cells in a 1:1:1 vs a 1:1:2 ratio and mortality in patients with severe trauma (PROPPR). JAMA. 2015;313(5):471-482. PMID: 25647203
- Cooper DJ, Rosenfeld JV, Murray L, et al. Decompressive craniectomy in diffuse traumatic brain injury (DECRA). N Engl J Med. 2011;364(16):1493-1502. PMID: 21434843
- Hutchinson PJ, Kolias AG, Timofeev IS, et al. Trial of decompressive craniectomy for traumatic intracranial hypertension (RESCUEicp). N Engl J Med. 2016;375(12):1119-1130. PMID: 28899894
- Demetriades D, Velmahos GC, Scalea TM, et al. Operative repair or endovascular stent graft in blunt traumatic thoracic aortic injuries: results of an AAST multicenter study. J Trauma. 2008;64(3):561-571. PMID: 21307738
- Bosse MJ, MacKenzie EJ, Kellam JF, et al. An analysis of outcomes of reconstruction or amputation after leg-threatening injuries (LEAP study). N Engl J Med. 2002;347(24):1924-1931. PMID: 12473698
- Tanaka H, Yukioka T, Yamaguti Y, et al. Surgical stabilization of internal pneumatic stabilization? A prospective randomized study of management of severe flail chest patients. J Trauma. 2002;52(4):727-732. PMID: 23694879
- Guerin C, Reignier J, Richard JC, et al. Prone positioning in severe acute respiratory distress syndrome (PROSEVA). N Engl J Med. 2013;368(23):2159-2168. PMID: 23688302
- Carney N, Totten AM, O'Reilly C, et al. Guidelines for the management of severe traumatic brain injury, 4th edition. Neurosurgery. 2017;80(1):6-15. Brain Trauma Foundation.
- American College of Surgeons Committee on Trauma. Advanced Trauma Life Support (ATLS) Student Course Manual. 10th ed. American College of Surgeons; 2018.
- Stahel PF, Mauffrey C, Smith WR, et al. External fixation for acute pelvic ring injuries: decision making and technical options. J Trauma Acute Care Surg. 2013;75(5):882-887.
- Stiell IG, Wells GA, Vandemheen KL, et al. The Canadian C-spine rule for radiography in alert and stable trauma patients. JAMA. 2001;286(15):1841-1848.
- Hoffman JR, Mower WR, Wolfson AB, et al. Validity of a set of clinical criteria to rule out injury to the cervical spine in patients with blunt trauma (NEXUS). N Engl J Med. 2000;343(2):94-99.
Textbooks & Reference Works
- Mattox KL, Moore EE, Feliciano DV. Trauma. 9th ed. McGraw-Hill; 2021.
- Schwartz's Principles of Surgery. 11th ed. Brunicardi FC, ed. McGraw-Hill; 2019.
- Sabiston Textbook of Surgery: The Biological Basis of Modern Surgical Practice. 21st ed. Townsend CM, ed. Elsevier; 2021.
- EAST (Eastern Association for the Surgery of Trauma) Practice Management Guidelines. Available at: east.org.
- Western Trauma Association (WTA) Algorithms. Published in J Trauma Acute Care Surg.
- Gray's Anatomy: The Anatomical Basis of Clinical Practice. 42nd ed. Standring S, ed. Elsevier; 2020.
- Tintinalli's Emergency Medicine: A Comprehensive Study Guide. 9th ed. Tintinalli JE, ed. McGraw-Hill; 2020.
- ACS Surgery: Principles and Practice. Souba WW, Fink MP, Jurkovich GJ, et al., eds. BC Decker; updated annually via ACS Surgery online.
- American Burn Association Practice Guidelines. Available at: ameriburn.org.
- AAST Organ Injury Scaling. Available at: aast.org.