Vascular Surgery
Every diagnosis, condition, procedure, complication, medication, classification, waveform, graft detail, wound care protocol, and management strategy in one place.
01 Vascular Anatomy — Arterial
Vascular surgery covers every blood vessel in the body except the coronary arteries (cardiothoracic/interventional cardiology) and the intracranial vessels (neurosurgery). The domain includes the entire aorta, all carotid, visceral, renal, iliac, extremity arteries, and all corresponding veins and lymphatic structures.
The Aorta
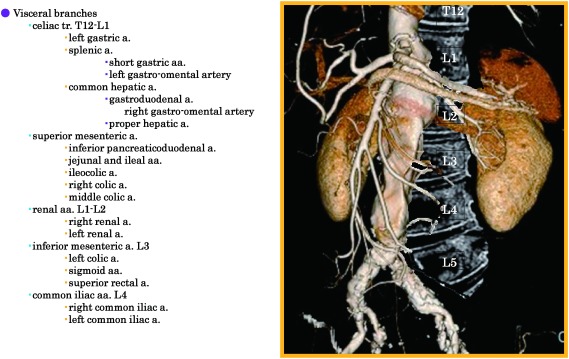
The aorta exits the heart as the ascending aorta, curves as the aortic arch (giving off the brachiocephalic trunk → right common carotid + right subclavian, then the left common carotid, then the left subclavian), descends through the thorax as the descending thoracic aorta (DTA), passes through the diaphragm at the aortic hiatus (T12), and becomes the abdominal aorta. Major abdominal branches: celiac trunk (T12), superior mesenteric artery (L1), renal arteries (L1–L2), inferior mesenteric artery (L3), then bifurcation into the common iliac arteries at L4 (approximately at the umbilicus). The infrarenal aorta (below the renal arteries) is the most common site of aortic aneurysmal disease.
Lower Extremity Arterial Tree
The sequence you must know cold: common iliac → external iliac (becomes the CFA after passing under the inguinal ligament) → common femoral artery (CFA) → bifurcates into the superficial femoral artery (SFA) and the profunda femoris (deep femoral artery, DFA). The SFA travels through the adductor canal (Hunter's canal) and emerges behind the knee as the popliteal artery. The popliteal trifurcates into the anterior tibial (AT), posterior tibial (PT), and peroneal (fibular) arteries. The AT becomes the dorsalis pedis (DP) on the dorsum of the foot. The PT gives off the medial and lateral plantar arteries on the sole. The profunda femoris is critical — it provides collateral circulation when the SFA is occluded and is almost never bypassed.
Upper Extremity Arterial Tree
Subclavian artery → axillary artery (at the lateral border of the first rib) → brachial artery → bifurcates in the antecubital fossa into the radial and ulnar arteries → form the superficial and deep palmar arches in the hand. The vertebral artery is the first branch of the subclavian and ascends through the transverse foramina of C6–C1 to supply the posterior circulation of the brain (relevant for subclavian steal syndrome).
Carotid System
The common carotid artery (CCA) bifurcates at approximately C3–C4 into the internal carotid artery (ICA) — supplying the brain (no extracranial branches) — and the external carotid artery (ECA) — supplying the face, scalp, meninges (branches: superior thyroid, ascending pharyngeal, lingual, facial, occipital, posterior auricular, superficial temporal, maxillary). The carotid bulb is the dilated area at the bifurcation and is the most common site of plaque accumulation due to flow turbulence. The carotid body (chemoreceptor) is located at the bifurcation — relevant for carotid body tumors.
Visceral (Mesenteric) Arteries
Three main arteries supply the GI tract: the celiac trunk (stomach, liver, spleen via left gastric, common hepatic, splenic arteries), the superior mesenteric artery (SMA) (small bowel, right/transverse colon), and the inferior mesenteric artery (IMA) (left/sigmoid colon, rectum). Key collateral pathways: the arc of Riolan and the marginal artery of Drummond connect the SMA and IMA territories. These collaterals are why chronic mesenteric ischemia usually requires stenosis/occlusion of at least 2 of 3 mesenteric vessels before symptoms develop.
02 Vascular Anatomy — Venous & Lymphatic
Deep Venous System (Lower Extremity)
Mirrors the arterial tree: posterior tibial veins, peroneal veins, anterior tibial veins → popliteal vein → femoral vein (FV) (formerly "superficial femoral vein" — this confusing name caused errors, now officially just "femoral vein") → common femoral vein (CFV) → external iliac vein → common iliac vein → inferior vena cava (IVC). The deep veins contain valves that ensure unidirectional flow toward the heart. Valve failure leads to deep venous reflux and contributes to CVI.
Superficial Venous System
The great saphenous vein (GSV) is the longest vein in the body. It originates from the dorsal venous arch of the foot, travels anterior to the medial malleolus, up the medial leg and thigh, and drains into the common femoral vein at the saphenofemoral junction (SFJ) in the groin. The small saphenous vein (SSV) runs posterior to the lateral malleolus, up the posterior calf, and drains into the popliteal vein at the saphenopopliteal junction (SPJ). Perforator veins connect the superficial to the deep system through the muscular fascia; they normally contain valves that allow flow from superficial → deep. When these valves fail, high pressure from the deep system transmits to the superficial veins, causing varicosities.
Lymphatic System
The lymphatic system is a network of thin-walled vessels that parallels the venous system, collecting interstitial fluid (lymph) and returning it to the central venous system via the thoracic duct (left side) and right lymphatic duct. Lymph nodes filter the fluid and are checkpoints for immune surveillance. Disruption of lymphatic drainage (by surgery, radiation, infection, or congenital malformation) causes lymphedema. The inguinal lymph nodes are particularly relevant in vascular surgery as they lie directly over the femoral vessels — surgical dissection in the groin carries risk of lymphatic injury, causing lymphoceles or lymphedema.
03 The Vascular Physical Exam
Pulse Examination
Pulses are assessed bilaterally and graded: 0 = absent, 1+ = diminished/thready, 2+ = normal, 3+ = bounding, D = detectable by Doppler only. Standard vascular pulse points: carotid, brachial, radial, ulnar, femoral, popliteal, dorsalis pedis (DP), posterior tibial (PT). For dialysis access patients, also assess the thrill (palpable vibration) and bruit (audible turbulence on auscultation) of the fistula or graft — a strong thrill/bruit indicates good flow, a weak or absent thrill suggests stenosis or thrombosis.
Radial: 2+/2+
Femoral: 2+/2+, no bruits
Popliteal: 2+/1+
DP: 2+/D
PT: 2+/D
Skin & Trophic Assessment
Signs of chronic arterial insufficiency: hair loss on the distal leg/dorsum of foot, shiny/atrophic skin, thickened dystrophic nails, muscle wasting, dependent rubor (foot turns red/purple when hanging down — engorged but poorly oxygenated capillary beds) and pallor on elevation (Buerger's test — elevate legs to 45° for 1–2 minutes; pallor indicates severe ischemia). Capillary refill > 3 seconds is abnormal. Skin temperature differences between limbs (palpate with the dorsum of your hand) indicate asymmetric perfusion.
Signs of venous disease: hemosiderin staining (brown discoloration of the gaiter area — lower medial leg — from red blood cell extravasation), stasis dermatitis (eczematous, itchy, inflamed skin), lipodermatosclerosis (indurated, hardened skin giving the classic "inverted champagne bottle" leg shape — narrow fibrotic lower calf, swollen upper calf), atrophie blanche (white, avascular, scar-like patches surrounded by dilated capillaries), varicosities, and pitting edema.
Edema Grading
| Grade | Depth | Rebound | Clinical |
|---|---|---|---|
| 1+ | 2 mm | Rapid (immediate) | Barely perceptible |
| 2+ | 4 mm | 15 seconds | Visible depression |
| 3+ | 6 mm | 30 seconds | Obvious swelling, limb distorted |
| 4+ | 8+ mm | > 30 seconds | Severe, tight skin, weeping |
Wound Assessment
Every wound is described with: location (be specific — plantar 1st MTP head, lateral 5th toe, medial malleolus, heel), dimensions (L × W × D in cm; use probe for depth/tunneling), wound bed (granulation tissue = healthy red/pink; slough = yellow/tan necrotic tissue; eschar = black/brown dry necrotic tissue; exposed bone/tendon), wound edges (well-defined vs undermined; epithelializing vs rolled), drainage/exudate (serous, serosanguinous, purulent, volume), periwound skin (erythema, warmth, induration, maceration, callus), and signs of infection (purulence, crepitus, malodor, fluctuance, lymphangitic streaking). Always note neurovascular status: sensation (Semmes-Weinstein monofilament for neuropathy screening), pulses, capillary refill.
Differentiating Arterial vs Venous Ulcers
| Feature | Arterial Ulcer | Venous Ulcer |
|---|---|---|
| Location | Toes, heel, lateral malleolus, pressure points | Medial malleolus ("gaiter area") |
| Pain | Severe, especially at night / with elevation | Mild-moderate, improved with elevation |
| Appearance | Punched-out, well-defined borders, pale/necrotic base | Irregular borders, shallow, pink/red granulating base |
| Surrounding Skin | Shiny, atrophic, hair loss, pallor | Hemosiderin staining, lipodermatosclerosis, eczema |
| Pulses | Diminished or absent | Usually normal |
| ABI | Low (< 0.9) | Normal (0.9–1.4) |
| Edema | Minimal | Significant |
| Treatment | Revascularization + wound care | Compression + wound care |
Neurological Exam (Carotid/Cerebrovascular)
For symptomatic carotid patients: assess cranial nerves (especially facial droop — CN VII, tongue deviation — CN XII), speech/language (aphasia or dysarthria), visual fields and acuity (amaurosis fugax), motor strength in all four extremities (scale 0–5), sensation, coordination, and gait. For post-CEA patients: specifically assess for new cranial nerve injuries — hypoglossal (tongue deviation), vagus/recurrent laryngeal (hoarseness), marginal mandibular branch of facial nerve (mouth asymmetry).
04 Peripheral Arterial Disease (PAD) Arterial
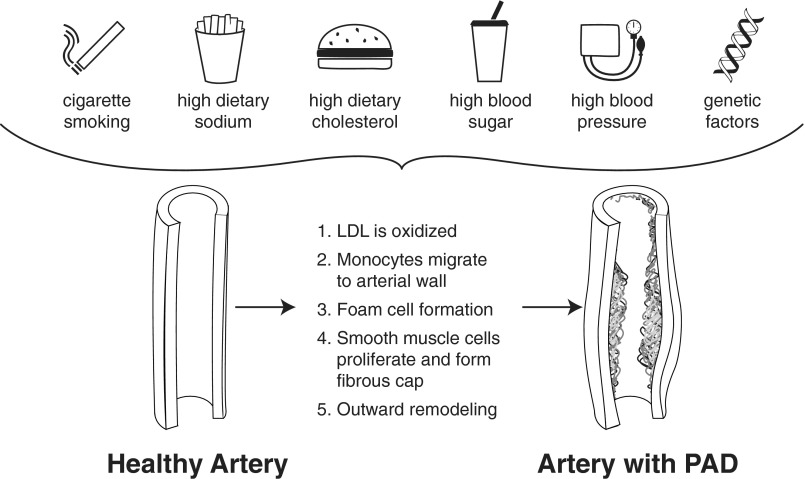
PAD is the single most common diagnosis in vascular surgery — atherosclerotic narrowing/occlusion of the arteries supplying the extremities, overwhelmingly the lower limbs. PAD is a systemic marker of atherosclerosis: patients have a 5–7× increased risk of MI, stroke, and cardiovascular death compared to age-matched controls without PAD.
Pathophysiology
Atherosclerotic plaque (lipids, calcium, fibrous tissue, inflammatory cells) gradually narrows the lumen. Most commonly affected segments: SFA at the adductor canal, aortoiliac segment, and tibial arteries (especially in diabetics). At rest, compensatory vasodilation and collateral development may maintain adequate flow. During exercise, the metabolic demand of working muscles outstrips supply, producing intermittent claudication.
Presentations by Level of Disease
| Level of Occlusion | Claudication Location | Other Features |
|---|---|---|
| Aortoiliac | Buttock, hip, thigh | Leriche syndrome (bilateral): buttock claudication + absent femoral pulses + erectile dysfunction |
| Femoropopliteal (SFA) | Calf (most common overall) | Diminished popliteal/pedal pulses, femoral pulse usually present |
| Tibial | Foot | Most common pattern in diabetics; pedal pulses absent, popliteal usually present |
| Multi-level | Thigh and calf, or buttock to foot | Severe disease, shorter walking distance, higher amputation risk |
ABI — The Critical Number
| ABI Value | Interpretation | Clinical Correlation |
|---|---|---|
| 1.00 – 1.40 | Normal | No significant PAD |
| 0.91 – 0.99 | Borderline | May need exercise ABI to unmask disease |
| 0.70 – 0.90 | Mild PAD | Asymptomatic or mild claudication |
| 0.50 – 0.69 | Moderate PAD | Moderate claudication, reduced walking distance |
| 0.40 – 0.49 | Severe PAD | Severe claudication, approaching CLTI territory |
| < 0.40 | Very severe / CLTI | Rest pain, tissue loss, limb threat |
| > 1.40 | Non-compressible (calcified) | Unreliable — use TBI (< 0.70 = PAD) or duplex |
Exercise ABI: If resting ABI is normal (0.91–1.40) but the patient has claudication symptoms, an exercise ABI is performed. The patient walks on a treadmill until symptoms occur, then ABIs are re-measured immediately. A drop of ≥ 20% in ankle pressure or ABI confirms PAD.
Fontaine & Rutherford — The Two Main Staging Systems
| Fontaine Stage | Rutherford Category | Clinical Description |
|---|---|---|
| I | 0 | Asymptomatic |
| IIa | 1 | Mild claudication (walking > 200m before pain) |
| IIb | 2 – 3 | Moderate to severe claudication (walking < 200m before pain) |
| III | 4 | Ischemic rest pain |
| IV | 5 | Minor tissue loss (non-healing ulcer, focal gangrene of toes) |
| IV | 6 | Major tissue loss (extending above TMA level, functional foot no longer salvageable) |
Management
First-line for claudication: Supervised exercise therapy (SET) — 3×/week for ≥12 weeks, walking to near-max pain, resting, repeating. SET doubles walking distance in most patients and is the most effective single intervention for claudication. Medical optimization: antiplatelet (ASA 81 mg or clopidogrel 75 mg daily), high-intensity statin (atorvastatin 40–80 mg), BP control (target <130/80), DM control (A1C <7%), and — most importantly — smoking cessation. Cilostazol (Pletal) 100 mg BID is the only FDA-approved drug for claudication symptoms (PDE3 inhibitor; vasodilator + antiplatelet); contraindicated in heart failure of any severity.
Revascularization — reserved for lifestyle-limiting claudication refractory to SET + meds, or for CLTI. Two approaches: endovascular (PTA ± stenting ± atherectomy — less invasive, shorter recovery, better for focal/short lesions) and open surgical bypass (more durable, better for long/complex lesions, requires adequate conduit and surgical fitness). Choice depends on anatomy (TASC classification, now being replaced by GLASS for CLTI), patient fitness, and expected durability needs.
05 Chronic Limb-Threatening Ischemia (CLTI) Arterial
CLTI is the advanced stage of PAD — ischemic rest pain, non-healing ulceration, or gangrene present for >2 weeks with objective evidence of arterial insufficiency. This is the modern term replacing "critical limb ischemia (CLI)." Without revascularization, 25–40% undergo major amputation within 6 months; 1-year mortality is 20–25%. CLTI patients are systemically sick — they almost universally have coexisting CAD, cerebrovascular disease, CKD, and diabetes.
WIfI Classification
The SVS Wound, Ischemia, foot Infection (WIfI) system grades each component 0–3 and provides an estimated risk of amputation and benefit from revascularization.
| Grade | W — Wound | I — Ischemia | fI — foot Infection |
|---|---|---|---|
| 0 | No ulcer/gangrene | ABI ≥ 0.80, AP ≥ 100, TP ≥ 60 | None |
| 1 | Small shallow ulcer, no gangrene | ABI 0.60–0.79, AP 70–100, TP 40–59 | Mild — skin/subQ only, ≤ 2 cm cellulitis |
| 2 | Deep ulcer ± exposed bone/tendon, gangrene limited to digits | ABI 0.40–0.59, AP 50–70, TP 30–39 | Moderate — deeper abscess, cellulitis > 2 cm, lymphangitis |
| 3 | Extensive ulcer/gangrene through forefoot or midfoot | ABI ≤ 0.39, AP < 50, TP < 30 | Severe — systemic sepsis, SIRS, wet gangrene |
AP = ankle pressure (mmHg), TP = toe pressure (mmHg)
Revascularization Planning
Uses the GLASS (Global Limb Anatomic Staging System) to grade the anatomic disease burden in the femoropopliteal and infrapopliteal segments separately, guiding endovascular-first vs surgery-first strategies. The SVS Global Vascular Guidelines (2019) use the PLAN framework: Patient risk (comorbidities, life expectancy), Limb staging (WIfI), ANatomic pattern (GLASS). The goal of revascularization in CLTI is to restore inline flow to the foot — at least one tibial or pedal artery with continuous flow to the foot (the "angiosome" concept suggests targeting the artery that specifically supplies the wound's territory).
06 Acute Limb Ischemia (ALI) Arterial
ALI is a surgical emergency — sudden decrease in limb perfusion threatening limb viability, typically within 14 days of onset. The 6 P's: Pain, Pallor, Pulselessness, Poikilothermia (cold), Paresthesia, Paralysis. Paresthesia and paralysis are the most ominous — indicating motor/sensory nerve ischemia that may become irreversible within 4–6 hours.
Etiology
| Cause | Features | % of ALI |
|---|---|---|
| Embolism | Sudden onset, no prior claudication, contralateral limb normal, cardiac source (AFib most common, mural thrombus post-MI, valvular disease, paradoxical embolism through PFO). Often lodges at bifurcation points (femoral bifurcation most common peripheral site). | ~30% |
| Thrombosis in situ | Background of PAD/claudication, gradual worsening then acute event, often on pre-existing stenosis. May have collaterals providing some perfusion. | ~40% |
| Graft/stent thrombosis | Prior bypass or stent suddenly stops working. Present with return of ischemic symptoms. | ~20% |
| Other (trauma, dissection, popliteal aneurysm thrombosis, ergotism, hypercoagulable) | Context-dependent | ~10% |
Rutherford Classification for ALI
| Category | Clinical | Sensory Loss | Motor Deficit | Arterial Doppler | Venous Doppler | Management |
|---|---|---|---|---|---|---|
| I — Viable | Not immediately threatened | None | None | Audible | Audible | Elective workup, anticoagulate |
| IIa — Marginally threatened | Salvageable if treated promptly | Minimal (toes only) | None | Often inaudible | Audible | Urgent revascularization (CDT or surgical) |
| IIb — Immediately threatened | Salvageable with immediate intervention | Beyond toes, rest pain | Mild–moderate | Inaudible | Audible | Emergent revascularization (surgical preferred) |
| III — Irreversible | Major tissue loss or permanent nerve damage inevitable | Profound, anesthetic | Profound (paralysis, rigor) | Inaudible | Inaudible | Primary amputation |
Treatment
Immediate IV heparin bolus (5,000–10,000 units) followed by drip (target aPTT 60–80) to prevent thrombus propagation. Then: surgical thromboembolectomy (cutdown, Fogarty balloon catheter — typically via CFA), catheter-directed thrombolysis (CDT) (tPA infused directly into clot over 12–48 hours via catheter — better for IIa; allows identification of underlying stenosis), percutaneous mechanical thrombectomy (PMT) (devices like AngioJet, Inari FlowTriever), or surgical bypass. Choice depends on severity (IIb → OR for embolectomy or bypass; IIa → CDT often reasonable), duration of ischemia, and underlying anatomy.
After revascularization of an acutely ischemic limb, reperfusion of damaged tissue releases toxic metabolites into the systemic circulation: hyperkalemia (can cause cardiac arrest — check K+ immediately, treat with calcium gluconate, insulin/dextrose, kayexalate), metabolic acidosis (lactic acid washout), myoglobinuria (rhabdomyolysis — dark urine, elevated CK/CPK, risk of acute tubular necrosis — aggressive IV fluids, target UOP 1–2 mL/kg/hr).
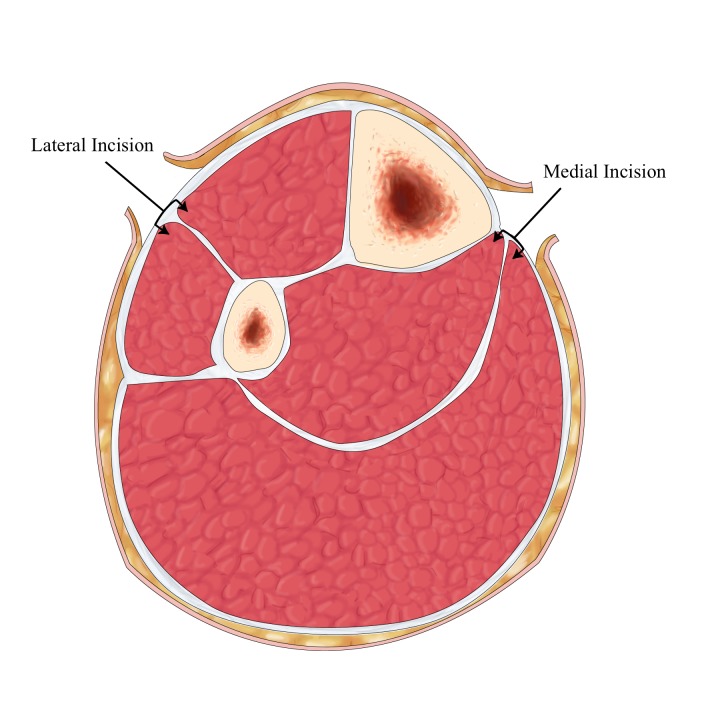
Compartment syndrome: reperfusion causes tissue edema within the tight fascial compartments of the lower leg, raising intracompartmental pressure above perfusion pressure, causing secondary ischemia. Signs: increasing pain (especially with passive stretch of muscles in the compartment), tenseness of compartments on palpation, pain out of proportion to exam. Treatment: emergent four-compartment fasciotomy (anterior, lateral, superficial posterior, deep posterior compartments). Fasciotomy should be performed prophylactically if ischemia time > 4–6 hours.
07 Carotid Artery Disease Arterial
Atherosclerotic stenosis at the carotid bifurcation/ICA. The symptomatic vs asymptomatic distinction is the single most important factor in treatment decisions.
Symptomatic: Ipsilateral stroke, TIA, or amaurosis fugax within the past 6 months. TIA symptoms: sudden onset contralateral motor weakness, sensory loss, speech difficulty (aphasia), or ipsilateral transient monocular blindness (amaurosis fugax — "curtain coming down over one eye"). Symptoms resolve completely, and current definitions focus on the absence of acute infarction rather than a strict 24-hour cutoff; stroke implies persistent deficit and/or infarction.
Asymptomatic: Discovered incidentally — bruit on exam, duplex for other reasons, or incidental finding on CT/MRI.
Stenosis Grading by Duplex Criteria
| Stenosis | PSV (cm/sec) | EDV (cm/sec) | ICA/CCA Ratio |
|---|---|---|---|
| < 50% | < 125 | < 40 | < 2.0 |
| 50–69% | 125–230 | 40–100 | 2.0–4.0 |
| ≥ 70% | > 230 | > 100 | > 4.0 |
| Near-occlusion | Variable (may be low due to reduced flow) | Variable | Variable |
| Total occlusion | No flow detected | — | — |
Treatment Thresholds
Symptomatic stenosis: CEA is generally recommended for 70–99% stenosis when perioperative risk is acceptable, and is considered on a case-by-case basis for 50–69% stenosis. When revascularization is chosen, treatment should ideally occur within 14 days of the neurological event. CAS or TCAR are alternatives in selected patients, especially when CEA risk is high or neck anatomy is unfavorable for open surgery.
Asymptomatic high-grade stenosis: Best medical therapy is mandatory. CEA, CAS, or TCAR may be considered only in carefully selected patients with high-grade stenosis, acceptable procedural risk, and reasonable life expectancy; this is not a blanket recommendation for every asymptomatic lesion above a single threshold.
All patients: Best medical therapy — antiplatelet therapy when indicated, high-intensity statin, BP control, diabetes management, and smoking cessation.
Procedures — CEA, CAS, TCAR
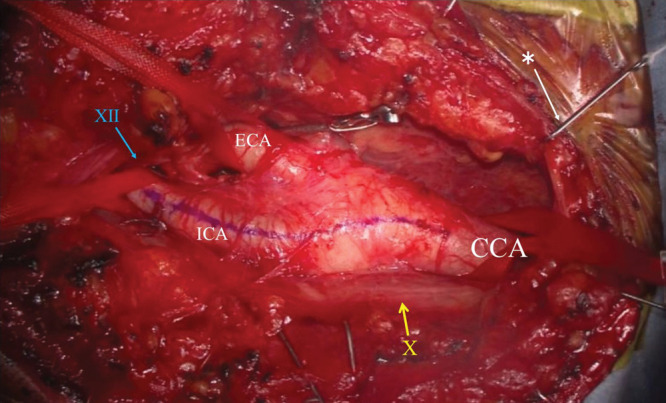
CEA (Carotid Endarterectomy): Gold standard. Incision along anterior border of sternocleidomastoid → expose CCA/ICA/ECA bifurcation → systemic heparin → clamp arteries → arteriotomy → plaque removal (endarterectomy plane between intima and media) → close arteriotomy with patch (bovine pericardium, Dacron, or ePTFE) to prevent restenosis → unclamp → restore flow. Intraoperative monitoring: EEG or stump pressure to assess cerebral perfusion during clamping; shunt placed if inadequate. Performed under general or regional (cervical block) anesthesia.
CAS (Carotid Artery Stenting): Transfemoral (or transradial) catheter approach. Embolic protection device (EPD) deployed (distal filter or proximal balloon occlusion with flow reversal), stent deployed across stenosis, post-dilation as needed. Reserved for patients who are high surgical risk for CEA: prior neck radiation, recurrent stenosis post-CEA, high cervical ICA lesion, contralateral carotid occlusion, severe cardiac/pulmonary disease.
TCAR (Transcarotid Artery Revascularization): Hybrid approach. Direct surgical cutdown to the CCA via small incision above the clavicle. A sheath is placed directly into the CCA, connected to a flow-reversal circuit with an external filter that returns filtered blood to the femoral vein. Stent is deployed under flow reversal (no emboli reach the brain). Combines the embolic safety of proximal flow reversal with the minimally invasive nature of stenting.
08 Subclavian Steal Syndrome Arterial
Caused by significant stenosis or occlusion of the proximal subclavian artery (before the vertebral artery origin). The pressure distal to the stenosis drops, and during arm exercise, blood "steals" from the brain by flowing retrograde down the vertebral artery to supply the arm — robbing the posterior cerebral circulation.
Clinical Features
Arm claudication (exercise-induced arm fatigue/pain), blood pressure discrepancy between arms (> 20 mmHg systolic difference is suggestive; > 40 mmHg is highly suggestive), subclavian bruit, and vertebrobasilar insufficiency symptoms (dizziness, vertigo, ataxia, diplopia, syncope, drop attacks) — especially provoked by arm exercise. Symptoms may be subtle or absent if collateral flow is adequate.
Diagnosis
Bilateral arm blood pressures (the critical screening test), carotid/vertebral duplex ultrasound (shows retrograde flow in the ipsilateral vertebral artery — pathognomonic), CTA or MRA for definitive anatomy.
Treatment
Symptomatic patients: subclavian artery stenting (endovascular — first line, high success rate) or carotid-subclavian bypass (open surgical — for lesions not amenable to stenting). Also important: subclavian steal can cause coronary steal in patients with a left internal mammary artery (LIMA) graft for CABG — the LIMA arises from the subclavian, so proximal subclavian stenosis can steal blood from the coronary bypass.
09 Upper Extremity Arterial Disease Arterial
Much less common than lower extremity PAD but still seen. Etiologies include atherosclerosis (subclavian/axillary arteries), emboli (from cardiac or proximal sources — causing acute hand ischemia with "blue finger" or "trash hand"), thoracic outlet syndrome (compression of subclavian artery), and repetitive trauma (hypothenar hammer syndrome — ulnar artery damage in workers who use the palm as a hammer).
Hypothenar hammer syndrome: Thrombosis of the ulnar artery at Guyon's canal from repetitive palmar trauma. Presents with ischemic fingers (especially ring and small fingers), cold sensitivity, and digital ulceration. Positive Allen's test (incomplete palmar arch filling). Treatment: smoking cessation, calcium channel blockers, and surgical revascularization or thrombolysis if severe.
10 Raynaud's Phenomenon Arterial
Episodic vasospasm of digital arteries causing the classic triphasic color change: white (pallor — vasospasm), blue (cyanosis — deoxygenation), red (rubor — reactive hyperemia on rewarming). Triggered by cold exposure or emotional stress.
Primary Raynaud's (Raynaud's disease): No underlying cause, typically young women, symmetric, benign course, no tissue loss. Treatment: cold avoidance, hand warmers, smoking cessation, calcium channel blockers (nifedipine or amlodipine — first-line pharmacotherapy).
Secondary Raynaud's (Raynaud's phenomenon): Associated with an underlying disease — most commonly connective tissue diseases (scleroderma, lupus, MCTD, rheumatoid arthritis), hypothenar hammer syndrome, thoracic outlet syndrome, or medications (beta-blockers, ergotamine). Can cause digital ulceration, gangrene, and auto-amputation. Workup includes ANA, anti-Scl-70, anti-centromere antibodies. Treatment: address underlying disease + vasodilators (CCBs, PDE5 inhibitors like sildenafil, topical nitroglycerin, iloprost infusion for severe cases).
11 Buerger's Disease (Thromboangiitis Obliterans) Arterial
Inflammatory, non-atherosclerotic, segmental thrombotic occlusion of small and medium arteries and veins of the distal extremities. Almost exclusively in young (20–45 years) male smokers (or users of any form of tobacco, including smokeless tobacco and marijuana). Affects hands and feet, sparing the proximal large vessels.
Presentation: distal extremity ischemia (rest pain, digital ulcers, gangrene) with normal proximal pulses, Raynaud's phenomenon, superficial thrombophlebitis (often migratory — "migratory thrombophlebitis"), and claudication of the foot arches or calves. Angiography shows segmental occlusions of distal arteries with "corkscrew" collaterals (a classic but not pathognomonic finding). No large-vessel atherosclerosis.
Diagnosis: clinical — young smoker with distal ischemia, no atherosclerotic risk factors (except tobacco), no embolic source, no autoimmune disease, no hypercoagulable state, and angiographic findings. Biopsy rarely needed but shows intraluminal thrombus with inflammatory infiltrate and intact vessel wall architecture (unlike vasculitis, which destroys the wall).
Treatment: Absolute tobacco cessation — this is the only treatment that alters disease course. Even a single cigarette can cause disease progression. If the patient quits completely, the disease usually stabilizes. Iloprost (prostacyclin analog) can be used for severe ischemia. Revascularization is rarely feasible because the disease affects small distal vessels. Amputation may be necessary for gangrene.
12 Popliteal Entrapment Syndrome Arterial
Anatomic compression of the popliteal artery by an aberrant medial head of the gastrocnemius muscle (or other musculotendinous structures) in the popliteal fossa. Classically presents in young, athletic males (20–30s) — often runners, cyclists, or military personnel — with calf claudication, exercise-induced leg pain, and paresthesias. Can lead to popliteal artery stenosis, thrombosis, post-stenotic aneurysm, and distal embolization if untreated.
Diagnosis: clinical suspicion in a young patient without atherosclerotic risk factors who has claudication. Provocative maneuver: active plantar flexion or passive dorsiflexion diminishes pedal pulses. Confirmed with MRI (shows anomalous muscle anatomy) or CTA/MRA with provocative positioning. Duplex with maneuvers can show flow reduction.
Treatment: surgical release of the anomalous muscular band/medial head of gastrocnemius ± popliteal artery reconstruction (bypass with saphenous vein graft) if the artery is damaged or aneurysmal.
13 Abdominal Aortic Aneurysm (AAA) Aortic
An aneurysm is dilation of an artery to ≥ 1.5× its normal diameter. For the infrarenal aorta (normal ~2.0 cm), AAA is defined as ≥ 3.0 cm. AAAs are the 13th leading cause of death in the US. Rupture mortality: > 80% (most die before reaching the OR); even with emergent repair, mortality is ~50%.
Risk Factors
Male sex, age > 65, smoking (strongest modifiable RF), family history (especially first-degree relatives), hypertension, and other atherosclerotic disease are major AAA risk factors. Some hereditary connective tissue disorders can also predispose to aneurysmal disease. Screening: USPSTF (2019) recommends one-time abdominal ultrasound for all men aged 65–75 who have ever smoked.
Surveillance & Repair Thresholds
| Diameter | Management | Surveillance |
|---|---|---|
| < 3.0 cm | Normal — no follow-up | — |
| 3.0–3.9 cm | Small AAA — surveillance | Every 3 years |
| 4.0–4.9 cm | Moderate — surveillance | Every 12 months |
| 5.0–5.4 cm | Approaching threshold | Every 6 months |
| ≥ 5.5 cm (men) / ≥ 5.0 cm (women) | Elective repair indicated | — |
| Growth > 0.5 cm / 6 months | Repair regardless of size | — |
| Symptomatic (any size) | Urgent/emergent repair | — |
Symptoms of Impending Rupture
Most AAAs are asymptomatic. Symptoms that suggest expansion or impending/contained rupture: sudden severe abdominal or back pain (often radiating to the flank or groin), pulsatile abdominal mass, hypotension, tachycardia. The classic triad of ruptured AAA is: abdominal/back pain + hypotension + pulsatile abdominal mass. However, this full triad is present in only ~50% of ruptured AAAs at presentation.
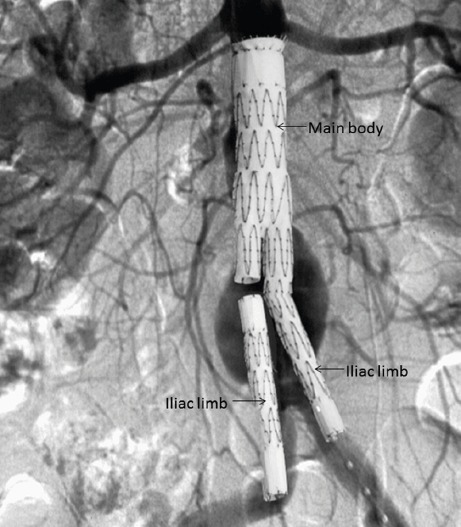
EVAR vs Open Repair
| Feature | EVAR | Open Repair |
|---|---|---|
| Approach | Bilateral femoral access, stent-graft deployed under fluoroscopy | Midline laparotomy or retroperitoneal approach |
| Perioperative mortality | 1–2% | 3–5% |
| Recovery | 1–3 days hospital, 2 weeks to baseline | 5–10 days hospital, 6–12 weeks to baseline |
| Anatomic requirements | Adequate proximal neck (≥ 10–15 mm infrarenal), suitable iliac arteries | No anatomic restrictions |
| Durability | Less durable — higher reintervention rate (5–20% at 5 years) | Very durable — low reintervention rate |
| Surveillance | Lifelong CT or ultrasound (endoleak monitoring) | Minimal follow-up needed |

Endoleak Classification
| Type | Description | Management |
|---|---|---|
| I | Leak at attachment site (Ia = proximal; Ib = distal) | Requires intervention — balloon molding, extension cuff, or conversion to open |
| II | Retrograde flow from branch arteries (lumbar, IMA) | Most common type. Observe unless sac expanding → then coil embolization |
| III | Graft defect — component separation or fabric tear | Requires intervention — relining or open conversion |
| IV | Graft porosity | Rare, self-limited with modern grafts |
| V (endotension) | Sac expansion without identifiable leak | Controversial — may need open conversion |
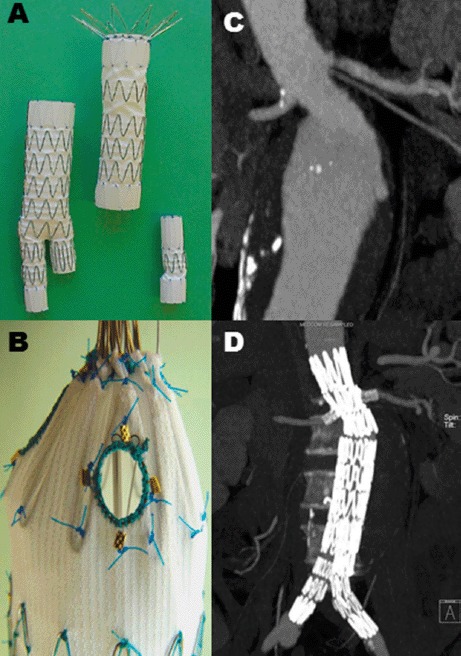
Complex Aortic Repair
When the aneurysm extends to or above the renal arteries (juxtarenal, suprarenal, or pararenal AAA), standard EVAR cannot be used because there is no adequate infrarenal landing zone. Options: fenestrated EVAR (FEVAR) — custom-made stent-graft with holes (fenestrations) or scallops to maintain flow to the renal and visceral arteries; branched EVAR — graft with side branches that connect to the visceral arteries; or open repair with suprarenal clamping and reimplantation of visceral vessels. These are among the most complex procedures in vascular surgery.
14 Thoracic & Thoracoabdominal Aneurysms Aortic
Thoracic aortic aneurysms (TAA) affect the ascending aorta, arch, or descending thoracic aorta (DTA). Vascular surgeons primarily manage DTA aneurysms; ascending aortic and arch aneurysms are usually managed by cardiothoracic surgery. Repair threshold: generally ≥ 5.5–6.0 cm for DTA; ≥ 4.5–5.0 cm for patients with connective tissue disorders (Marfan, Loeys-Dietz). TEVAR is now the standard approach for most DTA aneurysms that meet anatomic criteria (adequate proximal and distal landing zones ≥ 2 cm).
Thoracoabdominal aneurysms (TAAA) involve both the thoracic and abdominal aorta and are commonly described by the modified Crawford classification:
| Type | Extent |
|---|---|
| I | Most or all of DTA + upper abdominal aorta (to above renals) |
| II | Most or all of DTA + most or all of abdominal aorta (the most extensive) |
| III | Lower DTA + varying abdominal aorta |
| IV | Most or all of abdominal aorta including visceral segment |
| V | Lower DTA + upper abdominal aorta (above renals) |
TAAA repair carries risk of paraplegia (spinal cord ischemia from intercostal artery sacrifice — the artery of Adamkiewicz, typically T9–T12), renal failure, and visceral ischemia. Spinal cord protection strategies include CSF drainage (lumbar drain to reduce spinal cord pressure), hypothermia, and sequential clamping.
15 Aortic Dissection & Acute Aortic Syndromes Aortic
Tear in the aortic intima allowing blood to dissect between layers, creating a true lumen and false lumen. Stanford Type A = involves ascending aorta → emergent open repair (cardiac surgery). Stanford Type B = descending aorta only (distal to left subclavian) → managed by vascular surgery.
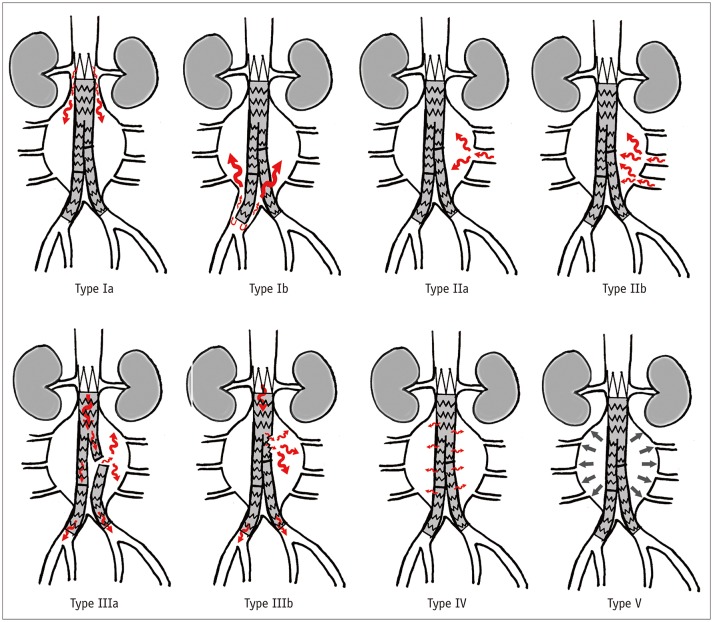
DeBakey: Type I = entire aorta; Type II = ascending only; Type IIIa = descending to diaphragm; Type IIIb = descending below diaphragm.
Type B Dissection Management
Uncomplicated: Medical — aggressive BP/HR control (target SBP < 120, HR < 60). IV esmolol or labetalol acutely → oral beta-blocker (amlodipine if beta-blocker intolerant). Pain control (often requires IV opioids initially). Serial imaging. Even uncomplicated Type B dissections may benefit from TEVAR in the subacute phase (INSTEAD-XL trial suggested improved aortic remodeling).
Complicated (any of: malperfusion of organs/limbs, rupture or hemodynamic instability, rapid false lumen expansion, refractory pain or hypertension): Urgent TEVAR — cover the primary intimal tear entry point with a thoracic stent-graft, restoring true lumen flow. May require additional fenestration/stenting of branch vessels if malperfusion persists.
Other Acute Aortic Syndromes
Intramural hematoma (IMH): Hemorrhage within the aortic wall (likely from ruptured vasa vasorum) without a visible intimal flap or false lumen on imaging. CT shows crescent-shaped high-density wall thickening. Considered a variant/precursor of dissection. Managed like dissection (Type A → surgery; Type B → medical ± TEVAR).
Penetrating atherosclerotic ulcer (PAU): Ulcerated atherosclerotic plaque eroding through the intima into the media. Can cause IMH, pseudoaneurysm, or rupture. More common in elderly patients with advanced atherosclerosis. CT shows focal contrast-filled outpouching through the aortic wall. Management: medical for stable PAU; TEVAR for symptomatic/expanding/ruptured.
16 Popliteal Artery Aneurysm Aortic
The most common peripheral aneurysm (70% of all peripheral aneurysms). Defined as popliteal artery diameter ≥ 2 cm (normal ~0.7–1.0 cm). Strong association with AAA — 40–50% of patients with popliteal aneurysms have a concomitant AAA, and 50–70% have bilateral popliteal aneurysms. Always screen for AAA and contralateral popliteal aneurysm.
The primary danger is not rupture (rare) but thrombosis and distal embolization. Thrombus forms within the aneurysm sac, and small emboli shower into the tibial arteries, gradually destroying the runoff vessels ("trash foot"). Acute popliteal aneurysm thrombosis presents as ALI and is a common cause of acute limb ischemia, especially in elderly men.
Indications for Repair
Diameter ≥ 2 cm, presence of mural thrombus (even if small), any prior embolic event, or if the patient is symptomatic. Some advocate for repair of any popliteal aneurysm with mural thrombus regardless of size, because embolization can occur from small aneurysms.
Repair Options
Open surgical repair: Medial approach, ligation of aneurysm above and below, interposition bypass with saphenous vein (preferred conduit) or prosthetic graft. Endovascular repair: Covered stent-graft across the aneurysm (concerns about long-term patency in this mobile joint segment). Posterior approach: For small aneurysms, direct aneurysmorrhaphy or interposition grafting.
17 Visceral Artery Aneurysms Aortic
Aneurysms of the celiac, splenic, hepatic, SMA, renal, or other visceral arteries. The most common visceral artery aneurysm is splenic artery aneurysm (60% of all visceral aneurysms), followed by hepatic artery aneurysm. Splenic artery aneurysms are more common in women and have a specific rupture risk during pregnancy (maternal mortality up to 70%). Repair threshold: generally ≥ 2 cm, or any size in women of childbearing age, or symptomatic. Treatment: coil embolization (endovascular — first line) or surgical excision/ligation.
SMA aneurysms are rare but have a high rupture rate (~40%) and require repair when diagnosed. Renal artery aneurysms are usually managed with observation unless ≥ 2 cm, symptomatic, in women of childbearing age, or enlarging.
18 Pseudoaneurysms Aortic
A pseudoaneurysm (false aneurysm) is a contained rupture — blood leaks through a breach in the arterial wall but is contained by surrounding tissue (adventitia and/or hematoma), creating a pulsatile sac that communicates with the arterial lumen through a "neck." Unlike true aneurysms, pseudoaneurysms do not contain all three layers of the arterial wall.
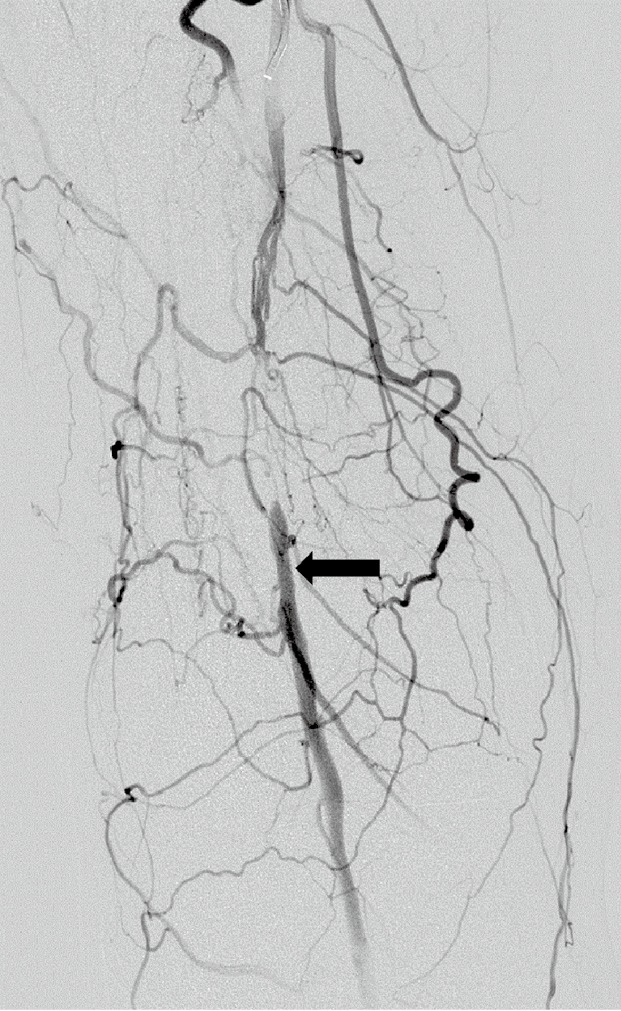
The most common cause you will see: femoral artery pseudoaneurysm after catheterization (post-cardiac catheterization, post-angiography, post-endovascular procedure). Presents as a pulsatile, expanding, painful groin mass with a bruit and thrill. Diagnosed by duplex ultrasound showing the classic "to-and-fro" flow pattern in the neck.
Treatment
Ultrasound-guided thrombin injection: First-line treatment for post-catheterization femoral pseudoaneurysms. Under ultrasound guidance, thrombin (a clotting factor) is injected directly into the pseudoaneurysm sac (avoiding the neck), causing immediate thrombosis. Success rate > 95%. Ultrasound-guided compression: Older technique — the ultrasound probe is used to compress the pseudoaneurysm neck for 10–30 minutes to promote thrombosis. Less comfortable for the patient, lower success rate. Surgical repair: For large, expanding, or infected pseudoaneurysms, or those not amenable to percutaneous treatment.
Other causes of pseudoaneurysms: anastomotic pseudoaneurysms (at bypass graft anastomoses, especially prosthetic-to-artery connections — can occur years after surgery), trauma, and infection (mycotic).
19 Mycotic / Infected Aneurysms & Graft Infections Aortic
Mycotic aneurysms are infected aneurysms (the name is misleading — they are usually bacterial, not fungal). Common organisms: Staphylococcus aureus (most common), Salmonella (especially in patients with pre-existing atherosclerosis), Streptococcus species, and Pseudomonas. Can arise from: hematogenous seeding of a pre-existing aneurysm, direct extension from adjacent infection, or infection of a traumatic/iatrogenic pseudoaneurysm.
Presentation: fever, elevated WBC, positive blood cultures, tender/expanding pulsatile mass, surrounding inflammation. CT shows irregular aneurysm wall, periaortic stranding/gas, and saccular morphology (mycotic aneurysms are typically saccular, unlike degenerative aneurysms which are fusiform).
Treatment: prolonged IV antibiotics (6+ weeks) + surgical excision of the infected arterial segment + extra-anatomic bypass (routing the bypass graft through clean, uninfected tissue planes to avoid reinfection) or in-situ reconstruction with antibiotic-impregnated graft or autologous tissue (femoral vein — the "neoaortoiliac system" or NAIS procedure).
Prosthetic Graft Infections
One of the most devastating complications in vascular surgery. Can be early (within 4 months — usually aggressive organisms like MRSA/MSSA, presenting with wound infection, sepsis) or late (months to years — often low-virulence organisms like Staphylococcus epidermidis, presenting with insidious graft thrombosis, sinus tract, pseudoaneurysm at anastomosis, or aortoenteric fistula). Aortic graft infections carry mortality rates of 25–75%.
Aortoenteric fistula (AEF): Erosion of an aortic graft (or native aortic aneurysm) into the duodenum (most commonly the 3rd or 4th portion). Presents as GI bleeding — the classic presentation is a "herald bleed" (small self-limited GI bleed) followed hours to days later by massive hemorrhage. Any patient with a history of aortic surgery who presents with GI bleeding has an AEF until proven otherwise. Diagnosed by CT (gas around graft, loss of fat plane between aorta and bowel, contrast extravasation) and/or endoscopy (seeing the graft eroding through the bowel wall). Treatment: emergent surgery — graft excision, bowel repair, and extra-anatomic bypass.
20 Deep Vein Thrombosis (DVT) Venous
Thrombus formation in a deep vein, most commonly in the lower extremity. Pathogenesis: Virchow's triad — stasis (immobilization, long travel, heart failure), endothelial injury (surgery, trauma, central lines), hypercoagulability (cancer, pregnancy, OCP/HRT, inherited thrombophilias: Factor V Leiden, Prothrombin G20210A mutation, antiphospholipid syndrome, Protein C/S deficiency, Antithrombin III deficiency).
Classification
Proximal DVT (popliteal vein and above): significant PE risk → requires anticoagulation. Distal DVT (calf veins — soleal, gastrocnemial, peroneal, tibial): lower PE risk; management varies (serial duplex surveillance vs anticoagulation based on risk factors). Iliofemoral DVT: most extensive, involving common femoral ± iliac veins; highest risk of PE and post-thrombotic syndrome; may benefit from pharmacomechanical thrombectomy (especially in young patients with low bleeding risk and symptom duration < 14 days).
Massive DVT Emergencies
Phlegmasia alba dolens ("painful white swelling"): extensive DVT causing massive leg swelling with pale/white appearance (arterial inflow still present but venous outflow obstructed). Phlegmasia cerulea dolens ("painful blue swelling"): near-total venous outflow obstruction → cyanotic, severely swollen, tense limb → can progress to venous gangrene and limb loss if not treated emergently. Treatment: elevation, systemic anticoagulation, and usually catheter-directed thrombolysis or surgical venous thrombectomy.
Anticoagulation
First-line: DOACs — rivaroxaban (Xarelto) or apixaban (Eliquis). Alternative: heparin bridge (UFH or enoxaparin) → warfarin (target INR 2.0–3.0). Duration: 3 months for provoked DVT (identifiable transient trigger), 3–6 months for first unprovoked DVT, indefinite for recurrent unprovoked DVT or strong thrombophilia. Consider thrombophilia workup for unprovoked DVT in young patients.
IVC Filter
Indications: DVT/PE with contraindication to anticoagulation (active bleeding, recent surgery, hemorrhagic stroke), or recurrent PE despite adequate anticoagulation. Retrievable filters should be removed when no longer needed (within weeks to months). Long-term IVC filters carry risks: filter thrombosis, IVC penetration, migration, and paradoxically increased DVT risk.
21 Pulmonary Embolism (PE) — Vascular Role Venous
PE is primarily managed by emergency medicine, pulmonology, and critical care, but vascular surgeons are involved when: catheter-directed thrombolysis is needed for massive/submassive PE, IVC filter placement is indicated, or surgical pulmonary embolectomy is required (rare, typically for massive PE with hemodynamic collapse and contraindication to systemic lysis).
PE severity classification: Massive PE — hemodynamic instability (SBP < 90, requiring vasopressors, cardiac arrest). Submassive PE — hemodynamically stable but with RV strain (elevated troponin, BNP, RV dilation on CT or echo). Low-risk PE — stable, no RV strain. Massive PE requires systemic thrombolysis (tPA), catheter-directed thrombolysis, or surgical embolectomy. Submassive PE is debated — systemic anticoagulation ± escalation to catheter-directed therapy if deteriorating.
22 Chronic Venous Insufficiency (CVI) Venous
Incompetent venous valves → sustained venous hypertension → swelling, skin changes, and ultimately ulceration. Most commonly results from prior DVT (post-thrombotic syndrome) or primary valvular incompetence. Duplex ultrasound with reflux testing confirms the diagnosis: reflux is defined as retrograde flow > 0.5 sec in superficial veins or > 1.0 sec in deep veins during Valsalva or calf compression-release maneuvers. (Classification updated in the 2020 CEAP revision.)
CEAP Classification
| C | Clinical Finding |
|---|---|
| C0 | No visible/palpable venous disease |
| C1 | Telangiectasias (spider veins) or reticular veins (< 3 mm) |
| C2 | Varicose veins (≥ 3 mm) |
| C2r | Recurrent varicose veins |
| C3 | Edema (without skin changes) |
| C4a | Pigmentation (hemosiderin) or eczema |
| C4b | Lipodermatosclerosis or atrophie blanche |
| C4c | Corona phlebectatica (fan-shaped intradermal veins at ankle) |
| C5 | Healed venous ulcer |
| C6 | Active venous ulcer |
| C6r | Recurrent active venous ulcer |
Additional CEAP descriptors: E (Etiologic: Ep = primary, Es = secondary/post-thrombotic, En = no cause, Ec = congenital), A (Anatomic: As = superficial, Ad = deep, Ap = perforator), P (Pathophysiologic: Pr = reflux, Po = obstruction, Pr,o = both).
Treatment: compression therapy (30–40 mmHg graduated stockings or multi-layer bandaging) is the cornerstone. Compression is contraindicated if ABI < 0.5 (severe arterial disease — always check before prescribing compression). Additional treatments: leg elevation, exercise, weight loss, treatment of superficial reflux (ablation), wound care for ulcers, and venous stenting for iliocaval obstruction.
23 Varicose Veins & Superficial Venous Disease Venous
Dilated, tortuous superficial veins ≥ 3 mm from incompetent valves in the GSV, SSV, or tributaries. Symptoms: heaviness, aching, throbbing, burning, itching, night cramps, restless legs. Complications: superficial thrombophlebitis, bleeding (can be dramatic from thin-walled varicosities), progression to CVI with skin changes and ulceration.
Treatment Modalities
| Procedure | Mechanism | Notes |
|---|---|---|
| Endovenous radiofrequency ablation (RFA / ClosureFast) | Thermal energy via catheter seals the vein | First-line for GSV/SSV reflux. Office-based under tumescent local anesthesia. |
| Endovenous laser ablation (EVLA/EVLT) | Laser energy via fiber seals the vein | Similar efficacy to RFA. Various wavelengths (810, 980, 1320, 1470 nm). |
| Mechanochemical ablation (MOCA / ClariVein) | Rotating wire + sclerosant injection | Non-thermal → no tumescent anesthesia needed. Less post-op pain. |
| Cyanoacrylate closure (VenaSeal) | Medical-grade glue injected into vein | Non-thermal, non-tumescent. No compression stockings needed post-procedure. |
| Sclerotherapy (liquid or foam) | Chemical destruction of vein wall (sodium tetradecyl sulfate, polidocanol) | For spider veins, reticular veins, small varicosities, residual tributaries after truncal ablation. |
| Ambulatory phlebectomy | Micro-incisions (2–3 mm) to remove varicose tributaries | Often done same session as truncal ablation. |
| High ligation & stripping | Open surgical ligation of SFJ + stripping of GSV | Largely replaced by endovenous techniques. Still used occasionally. |
24 Superficial Thrombophlebitis Venous
Thrombosis and inflammation of a superficial vein. Presents as a palpable, tender, indurated, erythematous cord along the course of a superficial vein. Most commonly affects the GSV or its tributaries. Risk factors: varicose veins, IV catheters, trauma, immobilization, hypercoagulable states, and malignancy (recurrent migratory superficial thrombophlebitis — Trousseau syndrome — is a paraneoplastic phenomenon associated with occult visceral malignancy, especially pancreatic cancer).
Superficial thrombophlebitis of the GSV within 3 cm of the saphenofemoral junction carries risk of extension into the deep venous system (DVT). Treatment: warm compresses, NSAIDs, compression. If extensive (> 5 cm), within 3 cm of SFJ, or in patients with DVT risk factors → anticoagulation (prophylactic or therapeutic dose depending on proximity to deep system). Duplex ultrasound should be performed to rule out concurrent DVT.
25 Post-Thrombotic Syndrome (PTS) Venous
The chronic sequela of DVT. After DVT, the thrombus either recanalizes (partially or completely) with residual valve damage, or persists as a chronic obstruction. The result is chronic deep venous reflux and/or obstruction → chronic venous hypertension → all the features of CVI (edema, pain, heaviness, skin changes, ulceration). PTS develops in 20–50% of patients after proximal DVT, even with adequate anticoagulation.
Severity is graded using the Villalta scale (assesses 5 symptoms and 6 signs on a 0–3 scale; score > 14 or presence of ulcer = severe PTS). Prevention: adequate anticoagulation duration after DVT, early ambulation, and possibly compression stockings (controversial after ATTRACT trial). Treatment: compression therapy (primary), exercise, elevation, treatment of superficial reflux, and for severe PTS with iliocaval obstruction → venous stenting (e.g., May-Thurner physiology post-DVT).
26 May-Thurner Syndrome Venous
Compression of the left common iliac vein by the overlying right common iliac artery against the lumbar spine. This anatomic compression causes intimal hyperplasia (spur formation) within the left iliac vein, predisposing to left-sided iliofemoral DVT. Classically seen in young to middle-aged women.
Should be suspected in any patient (especially young female) with unprovoked left-sided iliofemoral DVT. Diagnosis: IVUS (intravascular ultrasound — gold standard for identifying the compression and spur), CTV (CT venography), or MRV. Treatment: anticoagulation for the DVT, catheter-directed thrombolysis to clear the clot, then iliac vein stenting to relieve the compression and prevent recurrence.
27 Nutcracker Syndrome Venous
Compression of the left renal vein between the superior mesenteric artery (anteriorly) and the aorta (posteriorly). This causes left renal venous hypertension, leading to: hematuria (often microscopic but can be gross), left flank pain, left-sided varicocele (in men), pelvic congestion symptoms (in women), and proteinuria. A posterior variant exists where a retroaortic left renal vein is compressed between the aorta and the spine.
Diagnosis: CTA showing narrow aortomesenteric angle with left renal vein dilation, pressure gradient across the compressed segment (measured during venography — gradient > 3 mmHg is suggestive), and Doppler showing elevated velocities at the compression point. Treatment: conservative initially (young patients often improve with growth/weight gain); for severe/persistent symptoms → left renal vein transposition, SMA transposition, or renal vein stenting.
28 Pelvic Congestion Syndrome Venous
Chronic pelvic pain in women caused by dilated, incompetent pelvic (ovarian/internal iliac) veins — essentially "pelvic varicose veins." Presents with dull, aching pelvic pain worse with prolonged standing, worse premenstrually, associated with dyspareunia, and often with visible vulvar or thigh varicosities. Often misdiagnosed or underdiagnosed.
Diagnosis: transvaginal ultrasound showing dilated pelvic veins, MRV, or pelvic venography. Treatment: ovarian vein embolization (coils and/or sclerosant injected via catheter into the incompetent ovarian veins — first-line interventional treatment), possibly combined with treatment of associated iliac vein compression (May-Thurner) if present.
29 Mesenteric Ischemia Special
Acute Mesenteric Ischemia (AMI)
Surgical emergency with 60–80% mortality. Causes: SMA embolism (~50% — usually cardiac source), SMA thrombosis (~25% — on pre-existing atherosclerosis), mesenteric venous thrombosis (~10% — hypercoagulable states), non-occlusive mesenteric ischemia (NOMI ~10–15% — vasospasm in critically ill, low-flow states, vasopressors).
Classic presentation: "pain out of proportion to exam" — severe periumbilical/diffuse abdominal pain with benign abdominal exam (early). As bowel infarction develops → peritoneal signs, bloody diarrhea, hemodynamic instability, acidosis, elevated lactate. CTA is the study of choice (shows filling defects in mesenteric vessels, bowel wall thickening/pneumatosis, mesenteric stranding, portal venous gas).
Treatment: resuscitation, broad-spectrum antibiotics, systemic anticoagulation (heparin), and urgent revascularization — surgical embolectomy (SMA) or bypass for thrombosis, ± exploratory laparotomy for bowel viability assessment (second-look laparotomy often planned at 24–48 hours). Endovascular options (aspiration thrombectomy, catheter-directed thrombolysis, SMA stenting) are increasingly used for select cases. NOMI: treat underlying cause, stop vasopressors if possible, consider intra-arterial papaverine infusion.
Chronic Mesenteric Ischemia (CMI)
"Intestinal angina." Requires significant stenosis/occlusion of ≥ 2 of the 3 mesenteric arteries (celiac, SMA, IMA) due to extensive collateral networks. Classic triad: postprandial abdominal pain (within 15–30 minutes of eating, lasting 1–3 hours), sitophobia (food fear — patients stop eating to avoid pain), and weight loss (often dramatic — 20–40 lbs). CTA shows mesenteric vessel stenosis/occlusion. Treatment: mesenteric stenting (endovascular, first-line for most patients) or surgical bypass (antegrade or retrograde bypass from aorta or iliac to SMA/celiac). Open bypass may be more durable but carries higher perioperative risk.
30 Median Arcuate Ligament Syndrome (MALS) Special
The median arcuate ligament (MAL) is a fibrous arch connecting the diaphragmatic crura. In some patients, it crosses low, compressing the celiac trunk extrinsically. This can cause postprandial abdominal pain, nausea, and weight loss — mimicking CMI but in younger patients without atherosclerosis. The diagnosis is controversial because MAL compression is present in up to 13–50% of asymptomatic individuals, so the clinical significance of the compression must be carefully established.
Diagnosis: CTA with sagittal reconstructions showing focal celiac compression with characteristic hook-shaped narrowing that worsens with expiration (the ligament tightens during expiration). Duplex showing elevated celiac artery PSV (> 200 cm/sec during expiration). Treatment: laparoscopic or open release of the MAL (dividing the fibrous bands compressing the celiac axis), sometimes combined with celiac artery stenting if there is residual stenosis from post-stenotic changes or intimal damage.
31 Renal Artery Stenosis & FMD Special
Atherosclerotic RAS
Ostial/proximal renal artery narrowing due to aortic atherosclerotic plaque. Typically in older patients with other atherosclerotic disease. Causes renovascular hypertension (suspect when: HTN onset > 55 years, resistant HTN requiring 3+ meds, flash pulmonary edema, progressive CKD with no other cause, unilateral small kidney, bruit over the renal artery) and ischemic nephropathy. Screening: renal artery duplex (PSV > 200 cm/sec or renal-to-aortic ratio > 3.5 = ≥ 60% stenosis). Treatment: primarily medical (BP control, statin, antiplatelet). Renal artery stenting is reserved for failed medical therapy, flash pulmonary edema (Pickering syndrome), or rapidly declining renal function — the CORAL trial (2014) showed no benefit of stenting over medical therapy alone for most patients.
Fibromuscular Dysplasia (FMD)
Non-atherosclerotic, non-inflammatory vascular disease causing stenosis, aneurysm, and dissection of medium-sized arteries. Most commonly affects the mid-to-distal renal artery (60–75%) and carotid/vertebral arteries (25–30%). Classically in young to middle-aged women. The most common type is medial fibroplasia, which produces the pathognomonic "string of beads" appearance on angiography (alternating areas of stenosis and dilation). Treatment of renal FMD: balloon angioplasty (PTA) without stenting — excellent results with durable blood pressure improvement. Treatment of carotid FMD: antiplatelet therapy; PTA for symptomatic or high-grade stenosis.
32 Thoracic Outlet Syndrome (TOS) Special
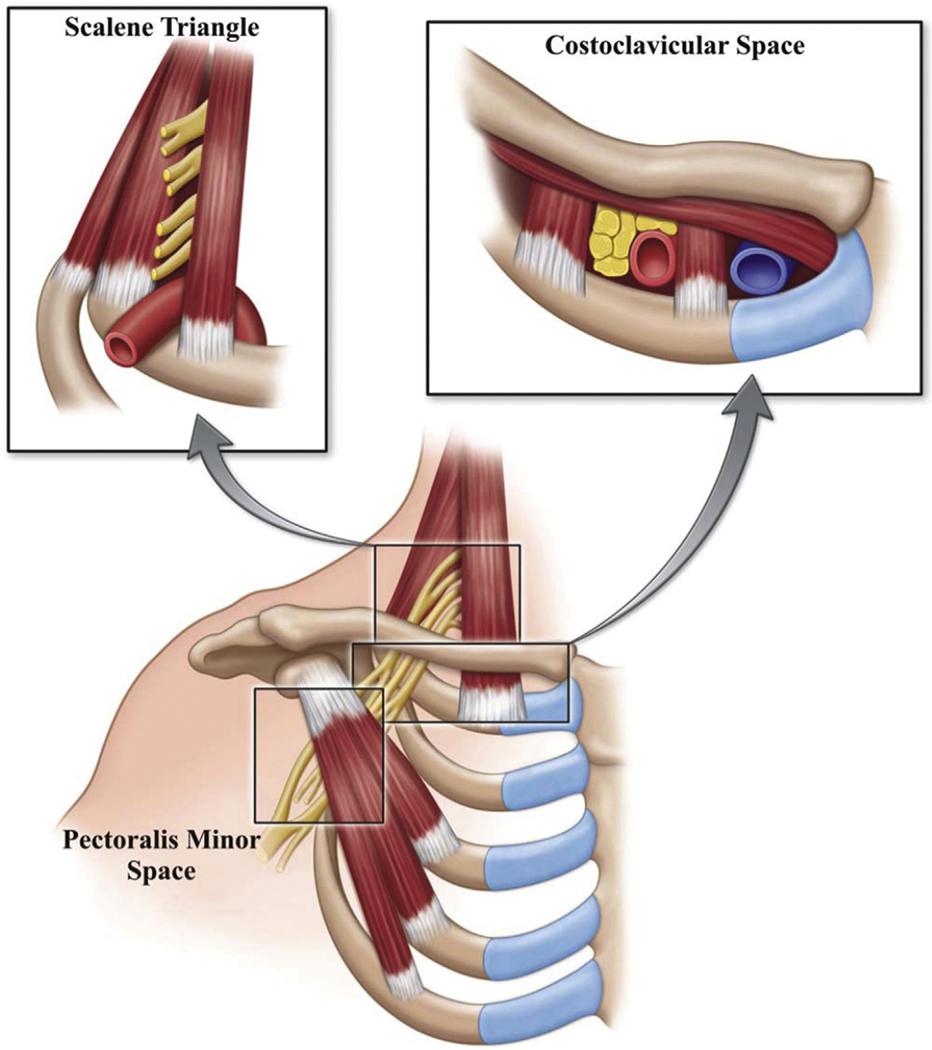
Compression of the neurovascular bundle (brachial plexus, subclavian artery, subclavian vein) in the thoracic outlet — between the clavicle, first rib, and scalene muscles. Three types:
Neurogenic TOS (nTOS, ~95%): Brachial plexus compression. Pain/numbness/tingling in the arm/hand, exacerbated by overhead activity. Provocative tests: Adson's (head turned toward affected side, deep inspiration — diminishes radial pulse), Wright's/hyperabduction (arm abducted and externally rotated), EAST/Roos test (arms at 90° abduction/external rotation, opening and closing fists for 3 minutes — reproduces symptoms, arm fatigues/drops). Treatment: physical therapy first (6–12 months); surgical first rib resection for refractory cases.
Venous TOS (vTOS / Paget-Schroetter, ~3–5%): Subclavian vein compression → DVT. "Effort thrombosis" — sudden arm swelling, pain, cyanosis after repetitive overhead activity (swimmers, baseball pitchers, weightlifters). Treatment: anticoagulation → CDT → first rib resection (to prevent recurrence).
Arterial TOS (aTOS, ~1%): Subclavian artery compression (often by cervical rib or anomalous first rib) → post-stenotic aneurysm → distal embolization (blue finger/hand). Treatment: first rib resection + arterial reconstruction ± embolectomy.
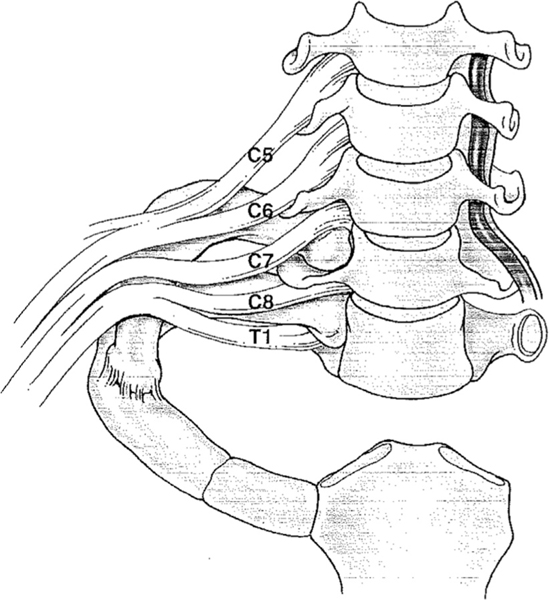
33 Dialysis Access (AVF, AVG, CVC) Special
Preferred order (Fistula First initiative): AVF > AVG > CVC.
AVF: Direct artery-to-vein anastomosis. Forearm: radiocephalic (Brescia-Cimino). Upper arm: brachiocephalic, brachiobasilic (basilic vein must be transposed superficially). Rule of 6's for maturity: ≥ 600 mL/min flow, ≥ 6 mm diameter, ≤ 6 mm depth, ≥ 6 cm length, wait ≥ 6 weeks. Best patency, lowest infection rate.
AVG: Prosthetic conduit (ePTFE) connecting artery to vein. Configurations: forearm loop (brachial artery to cephalic/basilic vein), upper arm straight (brachial to axillary), thigh loop (femoral). Can be cannulated in 2–3 weeks (some early-access grafts in 24–72 hours). Higher rates of thrombosis, stenosis (especially venous outflow), and infection than AVF.
CVC: Tunneled (Permacath — tip in right atrium, cuff in subcutaneous tunnel reduces infection) or non-tunneled (temporary). Right internal jugular is preferred site. Subclavian vein catheters should be avoided (risk of central venous stenosis that can render the ipsilateral arm unusable for future access). Highest infection rates.
Common Access Problems
| Problem | Presentation | Management |
|---|---|---|
| Stenosis (juxta-anastomotic or venous outflow) | Elevated venous pressures on dialysis, poor clearance, prolonged bleeding after needle removal, arm swelling (central venous stenosis) | Fistulogram → PTA ± stenting |
| Thrombosis | Loss of thrill/bruit, access not functioning | Surgical or pharmacomechanical thrombectomy, then treat underlying stenosis |
| Steal syndrome | Cold, painful, numb hand distal to access (arterial blood diverted through low-resistance access) | DRIL procedure (Distal Revascularization with Interval Ligation), banding, or access ligation if severe |
| Aneurysm/Pseudoaneurysm | Focal dilation at cannulation sites | Monitor if small; surgical revision if rapid expansion, skin compromise, or risk of rupture |
| Infection | Erythema, warmth, tenderness, purulence, fever | AVF infection rare but serious; AVG infection may require partial or complete graft excision; CVC infection → line removal + antibiotics |
| High-output cardiac failure | CHF symptoms from high-flow access (flow > 2 L/min) | Access banding to reduce flow, or ligation if CHF refractory |
34 Carotid Body Tumors (Paragangliomas) Special
Rare, typically benign, hypervascular tumors arising from the carotid body (a chemoreceptor at the carotid bifurcation). Present as a slowly enlarging, painless lateral neck mass at the angle of the mandible. The mass is pulsatile, has a transmitted bruit, and characteristically moves laterally but not vertically (Fontaine's sign). CTA shows a hypervascular mass at the carotid bifurcation that "splays" the ICA and ECA apart (pathognomonic "lyre sign"). Shamblin classification: Type I (small, easily dissected from vessels), Type II (partially surrounding vessels), Type III (encasing vessels — requires carotid reconstruction). Treatment: surgical excision ± preoperative embolization (to reduce intraoperative bleeding). Screen for bilateral tumors (~5% sporadic, up to 30% in familial cases) and pheochromocytoma (both are paragangliomas).
35 Vasculitis — Takayasu's & Giant Cell Arteritis Special
Takayasu's Arteritis
Large-vessel vasculitis affecting the aorta and its branches. Classically in young Asian women < 40 years. Causes stenosis, occlusion, and aneurysmal dilation of the aorta, arch branches (carotid, subclavian), renal, and mesenteric arteries. Presentation: constitutional symptoms (fever, malaise, weight loss), arm claudication, BP discrepancy between arms, absent pulses ("pulseless disease"), carotid/subclavian bruits, and renovascular hypertension. Diagnosis: CTA/MRA showing concentric wall thickening and stenosis in characteristic distribution + elevated ESR/CRP. Treatment: immunosuppression (high-dose corticosteroids ± methotrexate/tocilizumab), revascularization (bypass or stenting) reserved for limb/organ-threatening ischemia — ideally performed during disease quiescence.
Giant Cell Arteritis (GCA / Temporal Arteritis)
Large-vessel vasculitis affecting medium and large arteries, most classically the temporal artery. Typically in patients > 50 years, more common in women, northern European descent. Associated with polymyalgia rheumatica (PMR) in ~50% of cases. Presentation: new severe headache (especially temporal), scalp tenderness, jaw claudication, visual disturbances (amaurosis fugax, diplopia — ophthalmic artery involvement can cause permanent blindness), constitutional symptoms. Temporal artery may be tender, thickened, and pulseless. Markedly elevated ESR (> 50, often > 100) and CRP. Diagnosis: temporal artery biopsy (gold standard — shows granulomatous inflammation with giant cells; skip lesions can cause false negatives, so take a long specimen ≥ 2 cm). Treatment: immediate high-dose corticosteroids (prednisone 60–80 mg/day, do NOT wait for biopsy results if clinical suspicion is high — vision loss can be prevented but not reversed). Tocilizumab for steroid-sparing. Vascular surgeons may be involved in biopsy and in managing large-vessel complications (aortic aneurysm, subclavian/carotid stenosis).
36 Arteriovenous Malformations (AVM) Special
Congenital vascular anomalies with abnormal direct connections between arteries and veins without an intervening capillary bed. Can occur anywhere in the body (extremities, pelvis, viscera, brain/spinal cord — the latter managed by neurosurgery). Classified by the Schobinger staging system: Stage I (quiescence — warm, pink skin, AV shunting), Stage II (expansion — pulsation, thrill, bruit, enlarging), Stage III (destruction — pain, ulceration, bleeding, tissue necrosis), Stage IV (decompensation — high-output cardiac failure from massive AV shunting).
Diagnosis: clinical + duplex (shows high-velocity, low-resistance arterial flow feeding the nidus) + MRI (best for defining extent) + angiography (shows the nidus, feeding arteries, draining veins). Treatment: challenging — surgery alone has high recurrence if the nidus is not completely excised. Embolization (ethanol, Onyx, coils) to obliterate the nidus is first-line for many, often combined with surgical excision. Compression, sclerotherapy for superficial components.
37 Lymphedema Special
Protein-rich interstitial fluid accumulation from impaired lymphatic drainage. Primary: congenital lymphatic malformation (lymphedema praecox < 35 yrs; lymphedema tarda > 35 yrs). Secondary (far more common): surgery (axillary/inguinal lymph node dissection), radiation, infection (filariasis — #1 worldwide), malignancy, trauma.
Presentation: progressive, initially pitting then non-pitting edema that worsens through the day and improves overnight; asymmetric; does not resolve with elevation (unlike venous edema, which improves significantly). Positive Stemmer's sign (inability to pinch skin fold on dorsum of 2nd toe/finger). Over time: skin thickening, fibrosis, "peau d'orange" texture, hyperkeratosis, papillomatosis, recurrent cellulitis/lymphangitis.
Treatment: lifelong complete decongestive therapy (CDT) — manual lymphatic drainage massage, multi-layer compression bandaging (intensive phase), fitted compression garments (maintenance phase), exercise, skin care. Rarely surgical: vascularized lymph node transfer, lymphovenous anastomosis (supermicrosurgery), or debulking/liposuction for advanced fibrotic lymphedema.
38 Diabetic Foot Disease Special
The diabetic foot is a convergence of three pathologies: peripheral neuropathy (loss of protective sensation → unrecognized trauma → ulceration), PAD (diabetics preferentially develop tibial artery disease → impaired healing), and immunopathy (impaired WBC function → susceptibility to infection). Add to this altered foot biomechanics from motor neuropathy (intrinsic muscle atrophy → claw toes, prominent metatarsal heads → abnormal pressure points) and autonomic neuropathy (loss of sweating → dry, cracked skin → portal of entry for bacteria).
Assessment: Semmes-Weinstein monofilament test (10g monofilament pressed against plantar surface at multiple points — inability to feel = loss of protective sensation), 128 Hz tuning fork (vibratory perception), ABI/TBI (vascular status), X-ray or MRI of foot (evaluate for osteomyelitis, Charcot changes), and probe-to-bone test (if a wound can be probed down to bone, osteomyelitis is likely — positive predictive value ~89% in high-risk patients).
Charcot neuroarthropathy (Charcot foot): Destructive process of bones and joints in the neuropathic foot, leading to fractures, dislocations, and severe deformity ("rocker-bottom" foot). Acute Charcot presents with a hot, red, swollen foot that mimics cellulitis or DVT but is not infected. Treatment: total contact casting for offloading, strict non-weight-bearing, and close monitoring. Can take 3–12 months to resolve.
39 Wound Care — Dressings, VAC, HBO Special
Wound Healing Principles
Wound healing requires: adequate blood supply (revascularize if ischemic), absence of infection (debride and treat), moist wound environment (not too wet, not too dry), offloading of pressure (total contact cast, diabetic shoes, wheelchair for non-healing plantar ulcers), and nutritional optimization (albumin > 3.0, prealbumin > 15, adequate calories and protein).
Dressing Types
| Dressing Type | When to Use | Examples |
|---|---|---|
| Gauze (wet-to-dry or wet-to-moist) | Mechanical debridement; deep or tunneling wounds | Kerlix, Nu-Gauze |
| Foam | Moderate to heavy exudate; protection over granulating wounds | Mepilex, Allevyn |
| Alginate | Heavy exudate; hemostasis in bleeding wounds | Kaltostat, Aquacel |
| Hydrogel | Dry wounds; autolytic debridement of eschar/slough | IntraSite, DuoDERM Gel |
| Hydrocolloid | Shallow wounds with mild exudate; partial-thickness wounds | DuoDERM |
| Silver-containing | Infected or biofilm-colonized wounds | Aquacel Ag, Mepilex Ag |
| Collagen | Stalled wounds that need matrix support | Promogran, PuraPly |
| Skin substitutes / grafts | Clean, granulating wound ready for closure | Apligraf, Dermagraft, EpiFix, split-thickness skin graft (STSG) |
Negative Pressure Wound Therapy (NPWT / Wound VAC)
A sealed foam or gauze dressing connected to a vacuum pump that applies continuous or intermittent subatmospheric pressure (typically -125 mmHg). Benefits: removes excess exudate, reduces edema, promotes granulation tissue formation, draws wound edges together (macro-deformation), and provides a semi-closed environment. Commonly used for: large open wounds, post-debridement defects, open amputations, skin graft bolsters, infected wounds after debridement. Contraindications: untreated osteomyelitis, malignancy in wound, exposed blood vessels or organs, necrotic tissue with eschar (debride first), and fistulas to organs/body cavities.
Hyperbaric Oxygen Therapy (HBO)
Patient breathes 100% O₂ in a pressurized chamber (2.0–2.4 ATA) for 90–120 minutes per session, typically 20–40 sessions. Increases tissue O₂ levels → promotes angiogenesis, WBC function, fibroblast activity, and collagen synthesis. Indicated for: diabetic foot ulcers not responding to standard care (Medicare criteria: Wagner grade ≥ 3 with failed 30 days of standard treatment), compromised skin grafts/flaps, chronic refractory osteomyelitis, and radiation-induced tissue damage (osteoradionecrosis). TcPO₂ > 200 mmHg in the chamber predicts a good response.
Total Contact Casting (TCC)
Gold standard for offloading plantar diabetic foot ulcers. A well-molded total contact cast redistributes weight from the ulcer across the entire plantar surface. Changed weekly. Produces healing rates of ~90% for neuropathic plantar ulcers when used properly. Alternative: removable walking boots (but compliance is lower).
40 Amputation Levels & Limb Salvage Special
| Level | Description | Key Considerations |
|---|---|---|
| Toe amputation | Single or multiple toe removal | Preserve as much length as possible; "fillet" flap can close over bone |
| Ray amputation | Toe + corresponding metatarsal | Usually 1st or 5th ray; alters foot biomechanics less than central rays |
| TMA (transmetatarsal) | All toes + metatarsal heads | Preserves ankle function; high re-ulceration rate at stump if Achilles tight (may need Achilles lengthening) |
| Chopart | Midfoot disarticulation | Difficult to fit prosthesis; equinus deformity risk; rarely done |
| Syme | Ankle disarticulation with heel pad preserved | Good end-bearing stump; rarely performed now |
| BKA (below-knee) | Tibial transection, typically 12–15 cm below tibial tuberosity | Preserves knee → vastly better rehabilitation and prosthetic use than AKA; healing depends on adequate perfusion (TcPO₂ > 40 mmHg at calf predicts healing) |
| AKA (above-knee) | Femoral transection | When BKA will not heal (insufficient perfusion, flexion contracture, non-ambulatory patient); higher energy cost for ambulation with prosthesis |
| Hip disarticulation | Removal at hip joint | Severe cases (trauma, malignancy, extensive infection); very limited prosthetic options |
The goal is always to preserve the most distal, functional level possible. Pre-amputation assessment includes vascular status (ABI, TBI, TcPO₂, duplex — revascularize first if possible), nutritional status (albumin, prealbumin — malnourished patients have higher wound complication rates), and functional status (ambulatory potential, rehabilitation goals, patient preferences).
41 Endovascular Procedures Procedures
Balloon Angioplasty (PTA)
A catheter with a deflated balloon is advanced across the stenosis under fluoroscopic guidance. The balloon is inflated to compress the plaque against the arterial wall, widening the lumen. Used alone for short, focal lesions (especially iliac, tibial) or as an adjunct before/after stenting.
Stenting
Balloon-expandable stents: Mounted on a balloon, deployed by inflating the balloon. Precise placement, strong radial force. Used for ostial lesions (renal artery, iliac origin), calcified lesions, and carotid stenting. Less flexible — not ideal for mobile segments. Self-expanding stents: Compressed on a delivery system, expand to preset diameter when unsheathed. More flexible, conform to tortuous anatomy. Used for SFA/popliteal, iliac, and venous stenting. Covered stents (stent-grafts): Stent with fabric covering (ePTFE or Dacron). Used to treat aneurysms (EVAR/TEVAR), perforations, pseudoaneurysms, and AV fistulas.
Atherectomy
| Type | Device Examples | Mechanism | Best For |
|---|---|---|---|
| Directional | SilverHawk, TurboHawk, HawkOne | Rotating blade shaves plaque into a collection nosecone | Eccentric, non-calcified plaques; SFA/popliteal |
| Rotational | Jetstream (Boston Scientific) | Rotating blades + aspiration of debris | Calcified and thrombotic lesions; in-stent restenosis |
| Orbital | Diamondback 360 (CSI) | Diamond-coated crown orbits eccentrically, sanding plaque | Calcified lesions; creates smooth surface for DCB/stent |
| Laser | Turbo-Tandem (Spectranetics/Philips) | Excimer laser vaporizes plaque and thrombus | In-stent restenosis, thrombus-containing lesions |
42 Bypass Grafting Procedures
Conduit Choices
| Conduit | Advantages | Disadvantages | Typical Use |
|---|---|---|---|
| Autologous great saphenous vein (reversed or in-situ) | Best long-term patency (especially below knee), lowest infection rate | Requires adequate vein (≥ 3 mm), harvest site morbidity, not always available | Infrainguinal bypass (fem-pop below-knee, fem-tibial) |
| ePTFE (Gore-Tex) | Readily available, no harvest required | Inferior patency below-knee, higher infection rate, thrombogenic | Above-knee fem-pop, AV graft, hemodialysis access |
| Dacron (polyester) | Good for large-diameter, high-flow situations | Higher infection risk than vein, not suitable for small-caliber distal bypass | Aortobifemoral bypass, aortic repair |
| Arm veins (cephalic, basilic) | Alternative when GSV unavailable | Smaller caliber, more technically demanding | Infrainguinal bypass as second-line conduit |
| Cryopreserved vein allograft | Available "off the shelf" | Lower patency than autologous vein, immune-mediated degeneration | Infected field bypass, no autologous vein available |
Common Bypass Configurations
| Bypass | Indication | Notes |
|---|---|---|
| Aortobifemoral (ABF) | Aortoiliac occlusive disease (Leriche) | Y-shaped Dacron graft; excellent patency (85–90% at 5 yrs); requires laparotomy |
| Axillofemoral / Axillobifemoral | Aortoiliac disease in patients unfit for ABF | "Extra-anatomic" — graft tunneled subcutaneously from axillary to femoral artery; lower patency than ABF |
| Femoral-femoral crossover | Unilateral iliac occlusion with patent contralateral iliac | Graft tunneled suprapubically; simple, low morbidity |
| Fem-pop above-knee | SFA disease with patent above-knee popliteal | Vein preferred but ePTFE acceptable above-knee |
| Fem-pop below-knee | SFA/popliteal disease with patent below-knee popliteal | Vein strongly preferred — prosthetic has poor below-knee patency |
| Fem-tibial / fem-pedal | CLTI with tibial/pedal target | Vein mandatory — prosthetic to tibial arteries has unacceptable failure rate |
Vein Mapping
Pre-operative venous duplex ultrasound to assess the GSV (and arm veins if needed). Documents: vein diameter at multiple levels (groin, thigh, knee, calf), patency, compressibility, presence of sclerosis/thrombus, variations (duplicated segments, tributaries), and course. A vein ≥ 3.0 mm throughout is generally suitable. If GSV is absent or inadequate (prior harvest for CABG, prior ablation, varicose/thrombosed), alternate sources include the contralateral GSV, arm veins, or small saphenous vein.
43 Carotid Procedures (CEA, CAS, TCAR) Procedures
Covered in depth in Section 07. Key procedural details:
CEA: Key operative details: type of anesthesia (general vs cervical block), use of intraoperative shunt (yes/no), type of arteriotomy closure (primary vs patch — bovine pericardium, Dacron, ePTFE), intraoperative findings (extent of plaque, thrombus, ulceration), estimated blood loss, drain placement (yes/no — Jackson-Pratt drain sometimes placed), post-op neuro exam (intact cranial nerves, no focal deficits).
CAS/TCAR: Key operative details: access site (femoral, radial, or direct carotid), type of embolic protection device, stent type and size, pre/post-stent angiographic stenosis, any complications (dissection, spasm, bradycardia/hypotension from carotid body manipulation).
44 Aortic Repair (EVAR, TEVAR, Open) Procedures
Covered in depth in Sections 13–15. Key management and follow-up points:
EVAR: Key operative details: device manufacturer/model (Cook Zenith, Gore Excluder, Medtronic Endurant), main body and limb sizes, deployment accuracy, completion angiogram findings (endoleak type/presence), femoral access closure technique (Perclose ProGlide, surgical cutdown), and any intraoperative complications.
Open AAA repair: Key operative details: approach (midline transperitoneal vs retroperitoneal), clamp level (infrarenal, suprarenal), graft type and size (tube vs bifurcated, Dacron vs ePTFE), reimplantation of IMA if needed, EBL, urine output, and lower extremity pulses after unclamping.
TEVAR: Similar to EVAR plus: coverage of left subclavian artery (may require carotid-subclavian bypass or transposition), CSF drainage (lumbar drain for spinal cord protection), and post-op neuro checks for spinal cord ischemia (motor/sensory exam of lower extremities q1–2hr).
45 Venous Procedures Procedures
Endovenous ablation (RFA/EVLT): GSV or SSV accessed percutaneously under ultrasound guidance near the knee. Catheter advanced to the SFJ (positioned 2 cm below the junction to avoid extension into deep system). Tumescent anesthesia injected around the vein. Thermal energy delivered. Vein seals shut. Patient walks immediately.
Catheter-directed thrombolysis (CDT) for DVT: Catheter placed through popliteal or femoral vein access into the thrombus. tPA infused over 12–48 hours with serial monitoring (fibrinogen, aPTT, hemoglobin). Often combined with mechanical thrombectomy (pharmacomechanical thrombectomy — PMT). Post-thrombolysis venography performed to identify underlying stenosis (May-Thurner) → stenting if present.
Iliac vein stenting: For iliocaval obstruction (May-Thurner, post-thrombotic, extrinsic compression). Self-expanding stents (Wallstent, VENOVO) deployed under IVUS guidance. IVUS is critical — it detects intraluminal webs, spurs, and compression that may be missed on venography.
IVC filter placement/retrieval: Typically placed via jugular or femoral venous access under fluoroscopic guidance. Infrarenal IVC is the standard position. Retrievable filters should be retrieved when no longer indicated — retrieval rates historically low, increasing with dedicated filter retrieval programs.
46 Drug-Coated Devices (DCB & DES) Procedures
Drug-coated balloons (DCB) and drug-eluting stents (DES) deliver anti-proliferative drugs (primarily paclitaxel for peripheral, sirolimus for coronary) directly to the vessel wall during angioplasty or stenting to inhibit neointimal hyperplasia (the smooth muscle cell proliferation that causes restenosis). In the peripheral vasculature, paclitaxel-coated devices are used primarily in the femoropopliteal segment (SFA/popliteal artery), where restenosis rates after plain balloon angioplasty or bare metal stenting are high (up to 40–60% at 1 year).
The Paclitaxel Safety Controversy
In 2018, a meta-analysis by Katsanos et al. suggested increased all-cause mortality after 2 years in patients treated with paclitaxel-coated devices in the femoropopliteal arteries (1 excess death per 50 patients treated at 5 years). This triggered an FDA review, temporary reduction in use, and extensive re-analysis. Subsequent large-scale studies, registries, and FDA analysis found no definitive signal of increased mortality attributable to paclitaxel devices, and FDA ultimately concluded that the benefits continued to outweigh the risks. However, the controversy is still referenced frequently, and informed consent for DCB/DES procedures typically includes a discussion of this issue. Device-specific examples: IN.PACT Admiral (Medtronic DCB), Lutonix (BD DCB), Zilver PTX (Cook DES).
47 Post-Op Management by Procedure Procedures
| Procedure | Key Post-Op Monitoring | Common Complications |
|---|---|---|
| CEA | Neuro checks q1hr × 24hr (speech, motor, CN function), BP management (avoid hypo/hypertension — both dangerous), drain output, neck swelling (hematoma) | Stroke (1–3%), cranial nerve injury (hypoglossal, vagus), neck hematoma (can cause airway compromise — emergent bedside evacuation), hyperperfusion syndrome (headache, seizures, ICH from sudden restoration of flow to chronically hypoperfused brain) |
| EVAR | Femoral access site (hematoma, pseudoaneurysm), pedal pulses (limb occlusion), renal function (contrast), CT at 1 month (endoleak screening) | Endoleak (Type I/III require intervention), limb thrombosis, access site complications, contrast-induced nephropathy, post-implant syndrome (fever, elevated WBC — self-limited inflammatory response) |
| Open AAA repair | ICU admission, hemodynamics, urine output (renal perfusion), lower extremity pulses, abdominal exam (ileus expected), wound | MI (leading cause of post-op death), renal failure, colonic ischemia (monitor for bloody diarrhea — IMA ligation), graft infection, incisional hernia, sexual dysfunction (autonomic nerve damage) |
| Lower extremity bypass | Pulse checks q1hr × 24hr (pedal pulses or Doppler signals), ABI comparison, wound/incision sites, graft surveillance duplex before discharge | Graft thrombosis (early = technical; late = progression of disease or intimal hyperplasia), wound infection (especially groin), lymphocele, seroma |
| Endovenous ablation | Walk immediately, compression stockings × 2 weeks, duplex at 1 week (rule out EHIT — endovenous heat-induced thrombosis extending into deep system) | EHIT (extension of thrombus from ablated saphenous into common femoral vein — may require anticoagulation), skin burns (rare), paresthesia from nerve damage |
| Amputation | Stump wound, drain output, pain control (phantom limb pain prophylaxis), early PT/OT consult for rehabilitation, psychology/social work | Wound dehiscence/infection, stump revision, phantom limb pain (gabapentin, pregabalin), contracture (keep knee extended after BKA) |
48 Graft Complications Procedures
| Complication | Timing | Presentation | Management |
|---|---|---|---|
| Graft thrombosis | Early (< 30 days) or late | Return of ischemic symptoms, absent pulse/thrill | Surgical or endovascular thrombectomy → identify and treat cause (kink, stenosis, poor runoff) |
| Anastomotic stenosis (intimal hyperplasia) | 6 months – 2 years | Recurrent claudication, duplex shows elevated velocities at anastomosis | PTA ± patch angioplasty or jump graft |
| Anastomotic pseudoaneurysm | Months – years | Pulsatile mass at anastomosis site | Surgical repair — resection and re-anastomosis or interposition graft |
| Graft infection | Early or late | Wound drainage, exposed graft, sepsis, sinus tract, perigraft fluid/gas on CT | Excision of infected graft, extra-anatomic bypass, long-term antibiotics. MRSA/MSSA coverage (vancomycin + piperacillin-tazobactam empirically). |
| Aortoenteric fistula | Months – years after aortic graft | "Herald bleed" → massive GI hemorrhage. Any patient with aortic graft history + GI bleed = AEF until proven otherwise | Emergent surgery: graft excision, bowel repair, extra-anatomic bypass or in-situ reconstruction |
| Graft-enteric erosion | Similar to AEF but without full-thickness bowel perforation | Chronic low-grade infection, perigraft inflammation | Same principles as AEF |
| Seroma / lymphocele | Days – weeks | Fluid collection around graft (especially groin dissection) | Usually resolves spontaneously; aspiration if large/symptomatic; surgical drainage if persistent |
Graft Surveillance
All bypass grafts require long-term duplex surveillance to detect stenoses before they cause thrombosis. Typical schedule: 1 month, 3 months, 6 months, then every 6–12 months thereafter. A PSV > 300 cm/sec or PSV ratio > 3.5 at any point in the graft or at an anastomosis suggests ≥ 70% stenosis and warrants intervention (PTA, patch, or revision) to prevent graft failure.
49 Perioperative Cardiac Risk Procedures
Vascular surgery patients have extremely high rates of coexisting coronary artery disease (often asymptomatic due to limited exercise capacity). Major vascular surgery carries the highest cardiac risk of any surgical category. The Revised Cardiac Risk Index (RCRI / Lee Index) predicts risk of major cardiac events (MI, pulmonary edema, cardiac arrest, complete heart block) with 6 factors: high-risk surgery (intraperitoneal, intrathoracic, suprainguinal vascular), history of ischemic heart disease, history of CHF, history of cerebrovascular disease, preoperative insulin therapy for DM, and preoperative creatinine > 2.0 mg/dL.
Pre-operative cardiac workup: if RCRI ≥ 2 and functional capacity is poor (< 4 METs — cannot climb a flight of stairs or walk 2 blocks), consider pharmacologic stress testing (dobutamine stress echo or nuclear stress test). Coronary revascularization (PCI or CABG) before vascular surgery is only indicated if the cardiac condition itself independently warrants it — the CARP trial (NEJM, 2004) showed that prophylactic coronary revascularization before vascular surgery did not reduce mortality.
Perioperative beta-blocker management: patients already on beta-blockers should continue them (abrupt discontinuation increases cardiac risk). Starting beta-blockers de novo before surgery is controversial — may reduce MI but can increase stroke risk if titrated too aggressively (POISE trial, Lancet 2008).
50 Imaging & Diagnostics
| Modality | What It Shows | Clinical Interpretation Notes |
|---|---|---|
| ABI | Ratio of ankle:arm systolic BP | Record exact bilateral values; note exercise ABI if done |
| TBI | Toe:arm systolic BP ratio | Used when ABI > 1.4 (calcified); TBI < 0.7 = PAD |
| Arterial duplex | Anatomy + flow velocities (PSV, EDV) | Report PSV at each level, stenosis locations, and waveform type |
| Venous duplex | DVT (compressibility), reflux | Report which veins are involved, compressibility, and reflux duration |
| Carotid duplex | Carotid stenosis grading | PSV, EDV, ICA/CCA ratio — always report all three |
| CTA | 3D arterial anatomy | Check Cr/eGFR pre-contrast; routine IV contrast usually does not require holding metformin unless AKI, eGFR < 30, or local policy says otherwise |
| MRA | Arterial imaging without radiation | Gadolinium agent choice matters; group II agents are preferred in advanced CKD because NSF risk is very low |
| DSA (conventional angiography) | Gold standard real-time imaging | Invasive; mainly done during interventions |
| Segmental pressures | BP at thigh/calf/ankle levels | > 20 mmHg gradient = significant stenosis at that level |
| PVR (Pulse Volume Recording) | Plethysmographic waveforms | Qualitative: sharp upstroke = normal, blunted/flat = disease |
| TcPO₂ | Tissue oxygenation | > 40 mmHg = heals; < 30 mmHg = critical; used for amputation level selection |
| IVUS | Intravascular cross-sectional imaging | Used during venous stenting (iliac compression), aortic procedures |
51 Arterial Waveforms & Duplex Interpretation
You will hear the terms "triphasic," "biphasic," and "monophasic" on every single duplex ultrasound report. Understanding these waveforms is fundamental.
| Waveform | Components | What It Means |
|---|---|---|
| Triphasic (normal) | 1. Sharp systolic forward flow peak 2. Brief early diastolic flow reversal 3. Late diastolic forward flow | Normal peripheral arterial waveform. Indicates high-resistance vascular bed with no significant proximal stenosis. |
| Biphasic (mild disease) | 1. Systolic forward flow (peak may be blunted) 2. Diastolic reversal still present but reduced 3. No late diastolic forward flow | Early/mild arterial disease. Loss of the third component. Can be normal in older patients. |
| Monophasic (significant disease) | 1. Blunted systolic peak, no sharp upstroke 2. No diastolic reversal — continuous low-level forward flow throughout diastole | Significant proximal stenosis or occlusion. The continuous forward flow indicates distal vasodilation; when sampled distal to a proximal stenosis, this can produce a tardus-parvus pattern. |
Key Duplex Parameters
Peak Systolic Velocity (PSV): The highest velocity during systole. Elevated PSV at a specific point indicates stenosis at that location (the jet effect — blood accelerates through a narrow segment). A PSV ratio > 2.0 (velocity at stenosis ÷ velocity in normal upstream segment) indicates ≥ 50% stenosis. A PSV ratio > 4.0 indicates ≥ 75% stenosis.
End-Diastolic Velocity (EDV): Velocity at end-diastole. Elevated EDV indicates severe stenosis (high-grade narrowing maintains high velocity even in diastole). In the carotid, EDV > 100 cm/sec = ≥ 70% ICA stenosis.
Post-stenotic turbulence: Immediately downstream of a stenosis, the jet of blood creates turbulent, disorganized flow (spectral broadening on the Doppler tracing — loss of the clear "window" under the systolic peak). This is a qualitative sign of upstream stenosis.
52 Classification Systems — All in One Place
| System | Classifies | Key Features |
|---|---|---|
| Fontaine | Chronic PAD (symptoms) | I (asymptomatic), IIa (claudication >200m), IIb (<200m), III (rest pain), IV (tissue loss) |
| Rutherford (chronic) | Chronic PAD (symptoms + hemodynamics) | 0–6; adds objective ABI/PVR/treadmill data to clinical symptoms |
| Rutherford (acute) | ALI severity | I (viable), IIa (marginal), IIb (immediately threatened), III (irreversible) |
| WIfI | CLTI / limb threat | Wound (0–3), Ischemia (0–3), foot Infection (0–3); predicts amputation risk |
| CEAP | Chronic venous disease | C0–C6(r), E(p/s/n/c), A(s/d/p), P(r/o) |
| Villalta | Post-thrombotic syndrome severity | Score 0–33 based on 5 symptoms + 6 signs; ≥14 or ulcer = severe |
| Stanford | Aortic dissection | A (ascending involved) vs B (descending only) |
| DeBakey | Aortic dissection | I (whole aorta), II (ascending only), IIIa (descending to diaphragm), IIIb (below) |
| Crawford / modified Crawford | Thoracoabdominal aneurysm extent | Classically Types I–IV; many modern references include a modified Type V |
| TASC II | PAD lesion morphology | A–D based on lesion length/location; guides endo vs surgical approach |
| GLASS | Infrainguinal anatomy in CLTI | Grades FP and infrapopliteal disease separately; newer replacement for TASC in CLTI |
| Wagner | Diabetic foot ulcers | 0 (pre-ulcerative) to 5 (whole-foot gangrene) |
| Shamblin | Carotid body tumors | I (small, easily resected), II (partially surrounding vessels), III (encasing vessels) |
| Schobinger | AVM staging | I (quiescence) → IV (decompensation/heart failure) |
53 Medications Master Table Meds
Antiplatelet Agents
| Drug (Brand) | Mechanism | Dose | Vascular Use | Hold Before Surgery |
|---|---|---|---|---|
| Aspirin | Irreversible COX-1 inhibitor | 81 or 325 mg daily | All PAD, post-CEA, post-stent | 7 days (often continued perioperatively) |
| Clopidogrel (Plavix) | Irreversible P2Y12 blocker | 75 mg daily | PAD, DAPT post-stent | 5 days |
| Ticagrelor (Brilinta) | Reversible P2Y12 blocker | 90 mg BID | Post-ACS, some PAD | 5 days |
| Prasugrel (Effient) | Irreversible P2Y12 blocker | 10 mg daily | Post-PCI (coronary mainly) | 7 days |
| Cilostazol (Pletal) | PDE3 inhibitor | 100 mg BID | Claudication | Contraindicated in CHF |
| Dipyridamole+ASA (Aggrenox) | Nucleoside transport inhibitor + COX | 200/25 mg BID | Stroke prevention | — |
Anticoagulants
| Drug (Brand) | Mechanism | Monitoring | Reversal |
|---|---|---|---|
| Heparin (UFH) | Antithrombin III activator → anti-IIa, anti-Xa | aPTT (target 60–80 sec typically) | Protamine sulfate |
| Enoxaparin (Lovenox) | LMWH — primarily anti-Xa | Anti-Xa levels (if needed) | Protamine (partial reversal) |
| Warfarin (Coumadin) | Vitamin K antagonist (II, VII, IX, X) | INR (target 2.0–3.0 usually) | Vitamin K, FFP, PCC (Kcentra) |
| Rivaroxaban (Xarelto) | Direct Xa inhibitor | No routine monitoring | Andexanet alfa (Andexxa) |
| Apixaban (Eliquis) | Direct Xa inhibitor | No routine monitoring | Andexanet alfa |
| Dabigatran (Pradaxa) | Direct thrombin (IIa) inhibitor | No routine monitoring | Idarucizumab (Praxbind) |
Other Essential Medications
| Drug | Category | Relevance |
|---|---|---|
| Atorvastatin 40–80 mg (Lipitor) | High-intensity statin | All PAD patients — pleiotropic effects beyond lipid-lowering |
| Rosuvastatin 20–40 mg (Crestor) | High-intensity statin | Alternative to atorvastatin |
| Rivaroxaban 2.5 mg BID + ASA (COMPASS/VOYAGER dosing) | Low-dose DOAC + antiplatelet | Proven to reduce MACE + MALE in PAD patients (landmark trials) |
| Metoprolol / Atenolol | Beta-blocker | Perioperative cardiac protection, HR/BP control, dissection management |
| Amlodipine / Nifedipine | CCB | HTN, Raynaud's treatment |
| Lisinopril / Losartan | ACE-I / ARB | HTN, renal protection in DM |
| Metformin | Biguanide | Usually continued for routine IV iodinated contrast if there is no AKI and eGFR is at least 30; otherwise follow local contrast protocol |
| Pentoxifylline (Trental) | Methylxanthine | Improves RBC deformability; modest claudication benefit; less effective than cilostazol |
| Gabapentin / Pregabalin | Neuromodulator | Phantom limb pain, neuropathic pain |
54 Abbreviations Master List
Anatomy
Diagnoses
Procedures & Imaging
Medications & Labs
55 Risk Factors & Comorbidities
Smoking: Pack-years + quit date. Active smoking is the #1 modifiable risk factor for PAD, AAA, carotid disease, graft failure, and amputation. Smoking cessation counseling should occur at every visit.
Diabetes mellitus: Type 1 or 2, A1C, current regimen (insulin, metformin, sulfonylureas, GLP-1 agonists, SGLT2 inhibitors), neuropathy status, nephropathy (Cr, eGFR), retinopathy.
Hypertension: Current meds, BP control, target < 130/80.
Hyperlipidemia: LDL, statin type/dose. All PAD patients should be on high-intensity statin.
CKD/ESRD: Cr, eGFR, dialysis status, access type. Affects contrast use, medication dosing, and calcification patterns (falsely elevated ABI).
CAD: Prior MI, stents, CABG, EF%, recent stress test. Cardiac risk assessment before major vascular surgery.
AFib: Embolic risk source for carotid/mesenteric/ALI. Anticoagulation status.
CHF: EF%, NYHA class. Contraindicates cilostazol. Affects fluid management perioperatively.
Connective tissue disorders: Marfan, Ehlers-Danlos, Loeys-Dietz — earlier aneurysm repair thresholds, affect surgical approach and graft choice.
References & Figure Sources
Figures
- Figure 1 — Human Arterial System. Mariana Ruiz Villarreal (LadyofHats). Wikimedia Commons. Public domain.
- Figure 2 — Venous System of the Lower Extremity. BruceBlaus. Wikimedia Commons. Licensed under CC BY 3.0.
- Figure 3 — Carotid Artery Bifurcation. After Gray's Anatomy (1918). Wikimedia Commons. Public domain.
- Figure 4 — Aortic Dissection Classification (Stanford). Wikimedia Commons. Public domain.
- Figure 5 — Peripheral Artery Disease. Wikimedia Commons. Licensed under CC BY-SA 3.0.
- Figure 6 — Ankle-Brachial Index. Wikimedia Commons. Licensed under CC BY-SA 3.0.
- Figure 7 — Abdominal Aortic Aneurysm. NIH/NLM. Wikimedia Commons. Public domain.
- Figure 8 — Deep Vein Thrombosis. BruceBlaus. Wikimedia Commons. Licensed under CC BY 3.0.
- Figure 9 — Venous Valve Incompetence. BruceBlaus. Wikimedia Commons. Licensed under CC BY 3.0.
- Figure 10 — Thoracic Outlet Anatomy. Wikimedia Commons. Public domain.
- Figure 11 — Arteriovenous Fistula (AVF). BruceBlaus. Wikimedia Commons. Licensed under CC BY 3.0.
- Figure 12 — The Lymphatic System. BruceBlaus. Wikimedia Commons. Licensed under CC BY 3.0.
- Figure 13 — Lower Extremity Amputation Levels. Wikimedia Commons. Public domain.
- Figure 14 — Balloon Angioplasty and Stenting. NIH/NHLBI. Wikimedia Commons. Public domain.
- Figure 15 — Normal Arterial Doppler Waveform. Wikimedia Commons. Licensed under CC BY-SA 4.0.
Key Trials & Guidelines
- Conte MS, Bradbury AW, Kolh P, et al. Global Vascular Guidelines on the management of chronic limb-threatening ischemia. J Vasc Surg. 2019;69(6S):3S-125S.e40. PMID: 31159978
- Cooper CJ, Murphy TP, Cutlip DE, et al. Stenting and medical therapy for atherosclerotic renal-artery stenosis (CORAL trial). N Engl J Med. 2014;370(1):13-22. PMID: 24678939
- Eikelboom JW, Connolly SJ, Bosch J, et al. Rivaroxaban with or without aspirin in stable cardiovascular disease (COMPASS trial). N Engl J Med. 2017;377(14):1319-1330. PMID: 28498520
- Bonaca MP, Bauersachs RM, Anand SS, et al. Rivaroxaban in peripheral artery disease after revascularization (VOYAGER PAD trial). N Engl J Med. 2020;382(21):1994-2004. PMID: 32222135
- McFalls EO, Ward HB, Moritz TE, et al. Coronary-artery revascularization before elective major vascular surgery (CARP trial). N Engl J Med. 2004;351(27):2795-2804. PMID: 15625331
- Katsanos K, Spiliopoulos S, Kitrou P, et al. Risk of death following application of paclitaxel-coated balloons and stents in the femoropopliteal artery of the leg: a systematic review and meta-analysis. J Am Heart Assoc. 2018;7(24):e011245. PMID: 30561254
- Nienaber CA, Kische S, Rousseau H, et al. Endovascular repair of type B aortic dissection: long-term results of the randomized investigation of stent grafts in aortic dissection trial (INSTEAD-XL). Circ Cardiovasc Interv. 2013;6(4):407-416. PMID: 23918683
- POISE Study Group. Effects of extended-release metoprolol succinate in patients undergoing non-cardiac surgery (POISE trial). Lancet. 2008;371(9627):1839-1847. PMID: 18485482
- Vedantham S, Goldhaber SZ, Julian JA, et al. Pharmacomechanical catheter-directed thrombolysis for deep-vein thrombosis (ATTRACT trial). N Engl J Med. 2017;377(23):2240-2252. PMID: 29202613
- US Preventive Services Task Force. Screening for abdominal aortic aneurysm: US Preventive Services Task Force recommendation statement. JAMA. 2019;322(22):2211-2218. PMID: 31821437
- Lurie F, Passman M, Meisner M, et al. The 2020 update of the CEAP classification system and reporting standards. J Vasc Surg Venous Lymphat Disord. 2020;8(3):342-352. PMID: 32113854
Textbooks & Reference Works
- Rutherford's Vascular Surgery and Endovascular Therapy. 10th ed. Sidawy AN, Perler BA, eds. Elsevier; 2022.
- Haimovici's Vascular Surgery. 6th ed. Ascher E, ed. Wiley-Blackwell; 2012.
- Gray's Anatomy: The Anatomical Basis of Clinical Practice. 42nd ed. Standring S, ed. Elsevier; 2020.